Abstract
Introduction:
The Safety Pharmacology Society (SPS) conducted a membership survey to examine industry practices related mainly to cardiovascular (CV) safety pharmacology (SP).
Methods:
Questions addressed nonclinical study design, data analysis methods, drug-induced effects, and conventional and novel CV assays.
Results:
The most frequent therapeutic area targeted by drugs developed by the companies/institutions that employ survey responders was oncology. The most frequently observed drug-mediated effects included an increased heart rate, increased arterial blood pressure, hERG (IKr) block, decreased arterial blood pressure, decreased heart rate, QTc prolongation, and changes in body temperature. Broadly implemented study practices included Latin square crossover study design with n = 4 for nonrodent CV studies, statistical analysis of data (eg, analysis of variance), use of arrhythmia detection software, and the inclusion of data from all study animals when integrating SP studies into toxicology studies. Most responders frequently used individual animal housing conditions. Responders commonly evaluated drug effects on multiple ion channels, but in silico modeling methods were used much less frequently. Most responders rarely measured the J-Tpeak interval in CV studies. Uncertainties relative to Standard for Exchange of Nonclinical Data applications for data derived from CV SP studies were common. Although available, the use of human induced pluripotent stem cell cardiomyocytes remains rare. The respiratory SP study was rarely involved with identifying drug-induced functional issues. Responders indicated that the study-derived no observed effect level was more frequently determined than the no observed adverse effect level in CV SP studies; however, a large proportion of survey responders used neither.
Introduction
The assessment of novel drugs on the cardiovascular (CV) system has garnered much attention within safety pharmacology (SP) owing to the potentially insidious clinical consequences that can occur if a drug perturbs this vital organ system. 1 Since there is a relatively high prevalence of drug-induced effects on the CV system, this has resulted in allocation of considerable resources over the last decade by both drug safety groups in the pharmaceutical industry and regulatory authorities toward identification of testing methods to ascertain drug effects on QT/QTc interval prolongation and potential for proarrhythmia. As a consequence of a series of marketed drug withdrawals that occurred in the late 1990s, related to drug-mediated development of torsade de pointes (TdP) arrhythmia, the SP community rallied and chose to implement novel strategies utilizing innovative nonclinical methods and models to identify and limit development of drugs associated with this potentially lethal liability. 2,3 This focus resulted in a “hERG-centric” era within the SP community, with development and implementation of the ICH S7B guideline. 4 This testing paradigm, which focused on drug-mediated delay in cardiac repolarization due to blockade of only the IKr (or hERG) channel, may be replaced by a more holistic mechanistic evaluation of drug-associated proarrhythmic potential involving the evaluation of drug effects on multiple ion channels. 5 To complement the multiple ion channel evaluation, drug safety scientists could consider conduct of an in silico analysis. 5,6 The paradigm shift from inquisition of drug-induced effects only on cardiac repolarization to a more global approach involving the assessment of the proarrhythmic potential of the novel drug could lessen concerns of rejecting favorable therapeutics due to the isolated interpretation of IKr current blockade of the cardiac action potential and QT prolongation. Clinical results supporting a more complex mechanistic basis to drug-induced proarrhythmia 7 -9 culminated with development of the comprehensive in vitro proarrhythmia assay (CiPA). 9 -11 Briefly, CiPA is an integrated risk assessment strategy involving evaluation of data derived from clinical, in vitro, and in silico methods 12 to evaluate the proarrhythmic potential of a novel compound. Comprehensive in vitro proarrhythmia assay continues to fuel a plethora of research initiatives primarily within the SP community to delineate better predictive from erroneous nonclinical assays. Thus, although CiPA has become a dominant theme in SP, the landscape of CV safety concerns far exceeds this initiative and TdP risk alone. For example, inhibition of ion channels other than hERG can yield an increased proarrhythmic risk (ie, sodium channel inhibition producing altered cardiac conduction properties and increased risk for development of ventricular tachycardia or fibrillation in patients with structural heart disease), and altered blood pressure, heart rate, and cardiac contractility issues can elevate risk of adverse CV outcomes clinically. Compiling responses from global SP community members, the current industry survey attempts to depict a reasonably detailed portrait of current SP practices regarding the CV system as related to model selection, study design, and data analysis and interpretation.
Methods
This survey explored and assessed industry practices and strategies as well as common study outcomes as they relate to CV SP. Survey preparation involved an initial selection of survey question categories. Subsequently, a range of established and emerging issues considered relevant to the Safety Pharmacology Society (SPS) membership was interrogated by developing a series of specific questions within each category. The survey was distributed to individuals by the SPS for which the specific demographics are detailed in the Results section. Survey topics included nonclinical study design, data analysis methods, drug-induced CV effects/issues, and conventional and novel CV assays. The responses to the survey questions are reported in the Results section.
Results
All survey results are presented as the percentage of the total number of responses received per question.
Study Survey Demographics
Scientists (n = 1,349) from various fields of expertise and location were invited to participate in the survey and 137 (10%) responded. Of the 137 respondents, a preponderance were from North America (47%), followed by Europe (40%) and Asia (13%; Figure 1A). Respondents originated from diverse public private institution sizes (Figure 1B), with 52% employed at companies with >1,000 employees. Thus, it is anticipated that multiple respondents from the same organization may have contributed to this survey (an issue that was not quantified in the responses). Pharmaceutical companies developing small molecules (new chemical entities [NCEs]) employed most respondents (29%); however, almost as many respondents were involved in developing both NCE and large molecules/biologics (24%) or were from contract research organizations (CRO; 24%; Figure 1C). Interestingly, consultants accounted for 10% of responders, while government and regulators represented 7% of responders. When asked to describe their role in the drug development process, responders were primarily nonclinical safety pharmacologists (71%) followed by toxicologists (32%). Note, however, that more than one response was used to describe their role (Figure 1D). When asked, 66% of responders indicated that they provide general oversight to the overall design, review, analysis, and interpretation of data derived from the CV safety studies (Figure 1E). The survey identified an interesting fact regarding the primary therapeutic areas that were targeted by the companies employing survey respondents. Oncology (52%) followed by immunology/inflammation (47%), neurology (45%), and cardiometabolic disease (45%) were the most frequently targeted therapeutic areas reported for drug development (Figure 1F).
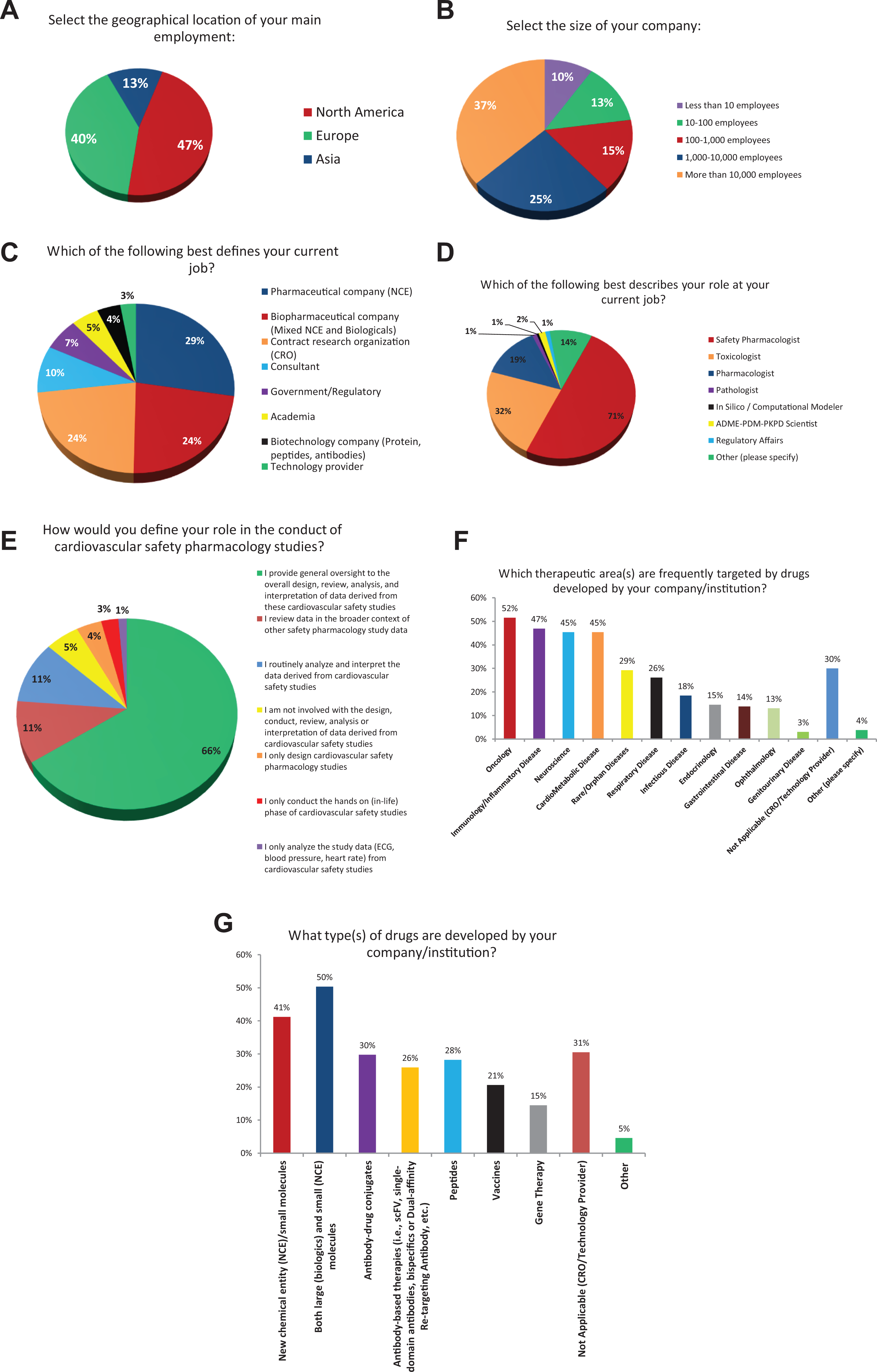
Cardiac study survey demographics. Panel A, Panel B, Panel C, Panel D, Panel E, Panel F, and Panel G. Panels A-F describe the characteristics of survey responders in terms of geographical location (A), size of the organization (B), current job (C), role in current job (D), role in the conduct of safety pharmacology studies (E), indication most frequently targeted by drugs developed (130 respondents; F), and type of drugs developed (G).
Study Design
When assuming the drug indication was not sex-specific, primarily male (56%) but sometimes both male and female (42%), nonclinical animal species were used in the conduct of Good Laboratory Practice (GLP) CV SP testing. In stand-alone CV SP studies, a group size of n = 4 was used most frequently by respondents; however, group sizes of n = 6 (23%) or n ≥ 6 (24%) were also frequently reported to be used (Figure 2). However, when the study end points from a CV SP study were incorporated into a standard toxicology study, most respondents (61%) reported that all animals (as opposed to only a subset of animals) were typically used in the safety assessment (Figure 3). In a stand-alone CV SP drug evaluation, most respondents reported using single dose administration of the drug with a Latin square cross-over study design. While these studies included a pharmacokinetic (PK) blood collection, it was often conducted in a subsequent session after the completion of the telemetry monitoring phase of the study (Table 1). For the PK evaluation, respondents reported that a very limited blood collection was conducted during the CV SP study. The purpose of the limited drug level quantitation was to confirm dosing and that an appropriate drug exposure was achieved in the SP study. This PK value is extrapolated to a complete PK profile for the drug conducted in a separate study in the same animal species using the same route of administration. The PK blood sample was collected after the time to maximal drug concentration (Tmax) occurred during telemetry monitoring. When testing large molecules/biologics, respondents reported the frequent use of a parallel study design; however, in some instances, the stand-alone CV SP study design with single dose administration (Table 2) was used. The survey identified the fact that during the conduct of repeat dose toxicology studies, both jacketed telemetry recording methods and PK blood collection after Tmax were implemented by a number of respondents.
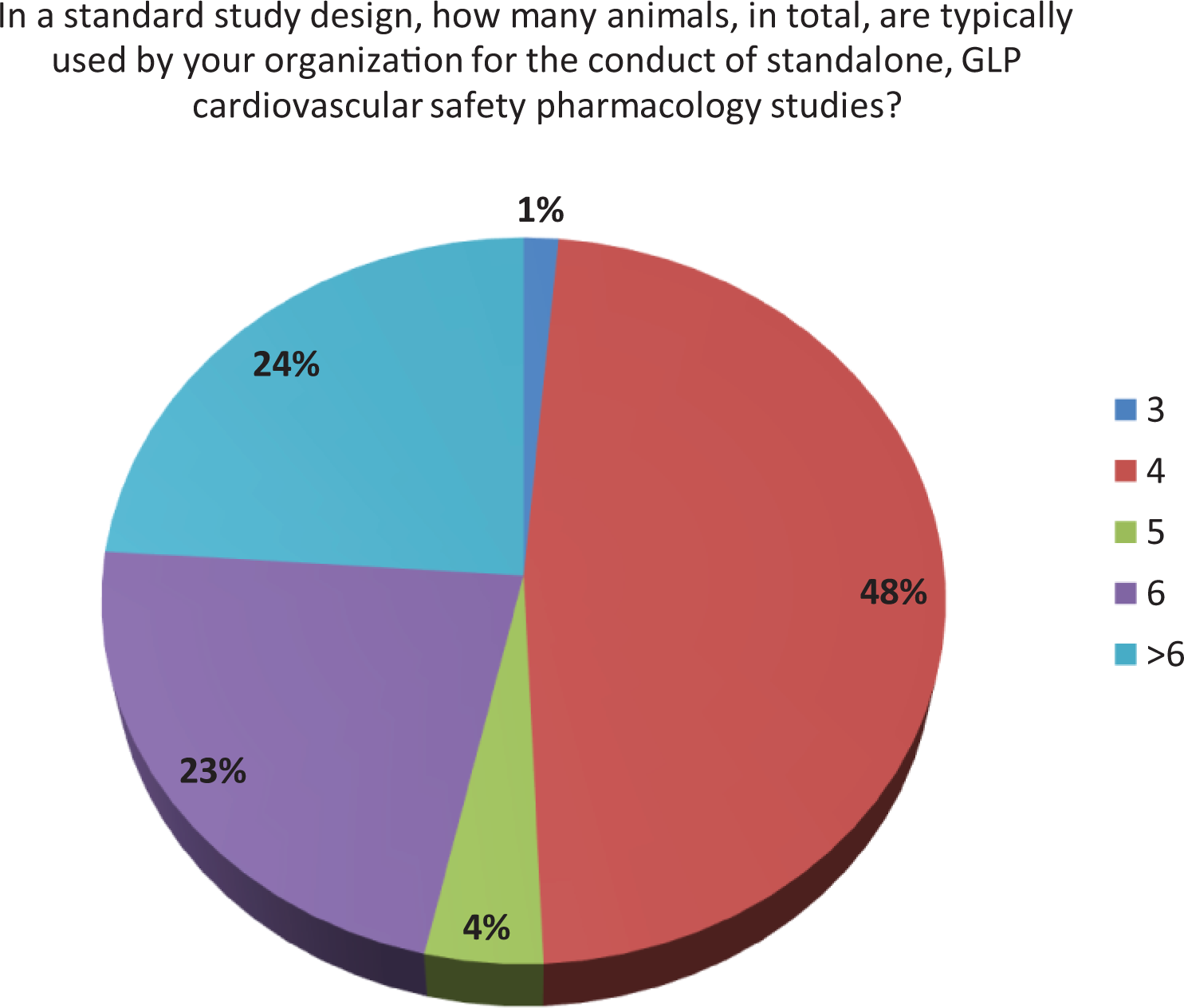
Group size in stand-alone cardiovascular safety pharmacology studies.
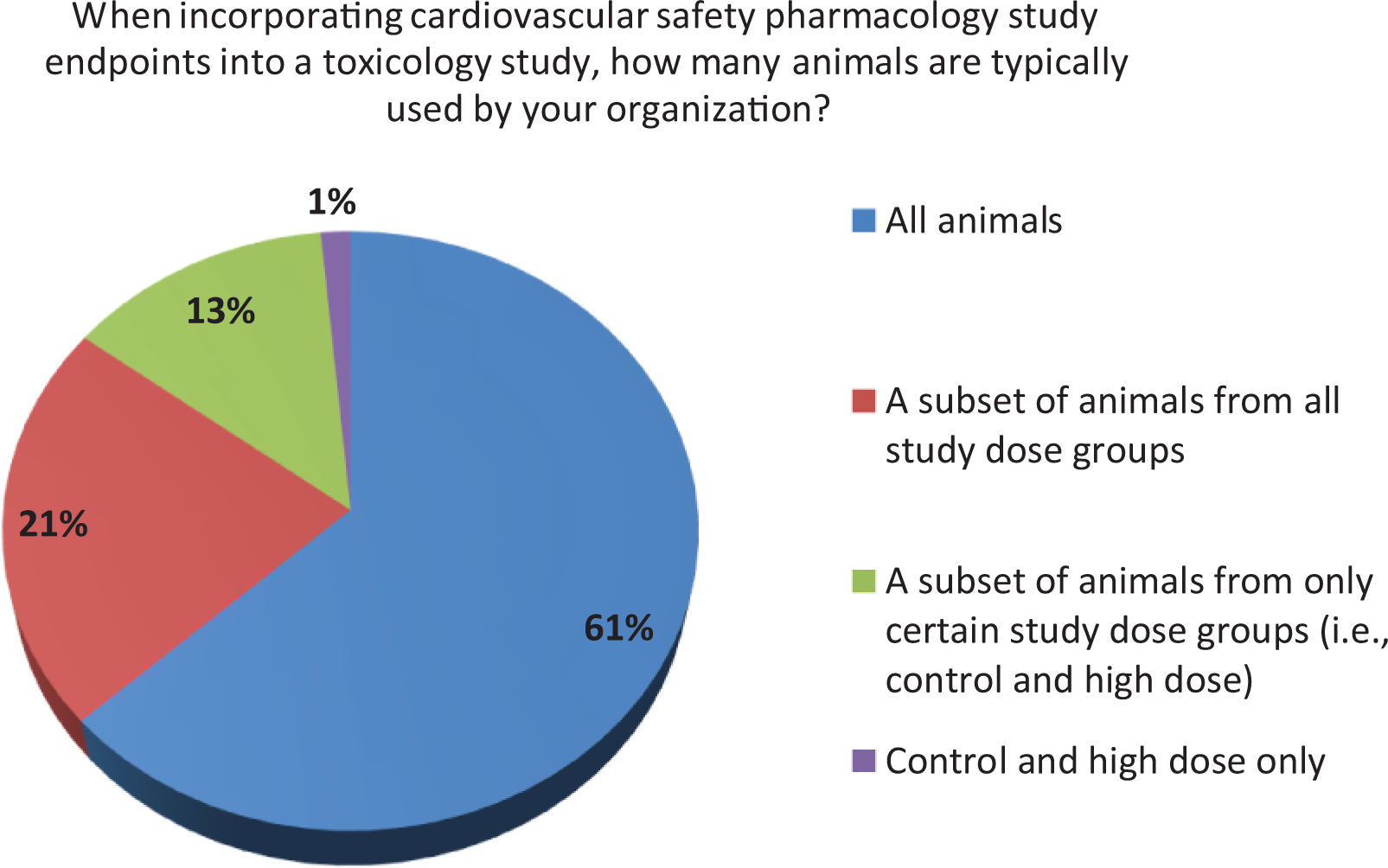
Group size for cardiovascular safety pharmacology into toxicology.
Experimental Design Features for NCE/Small-Molecule Cardiovascular Safety Pharmacology Studies.
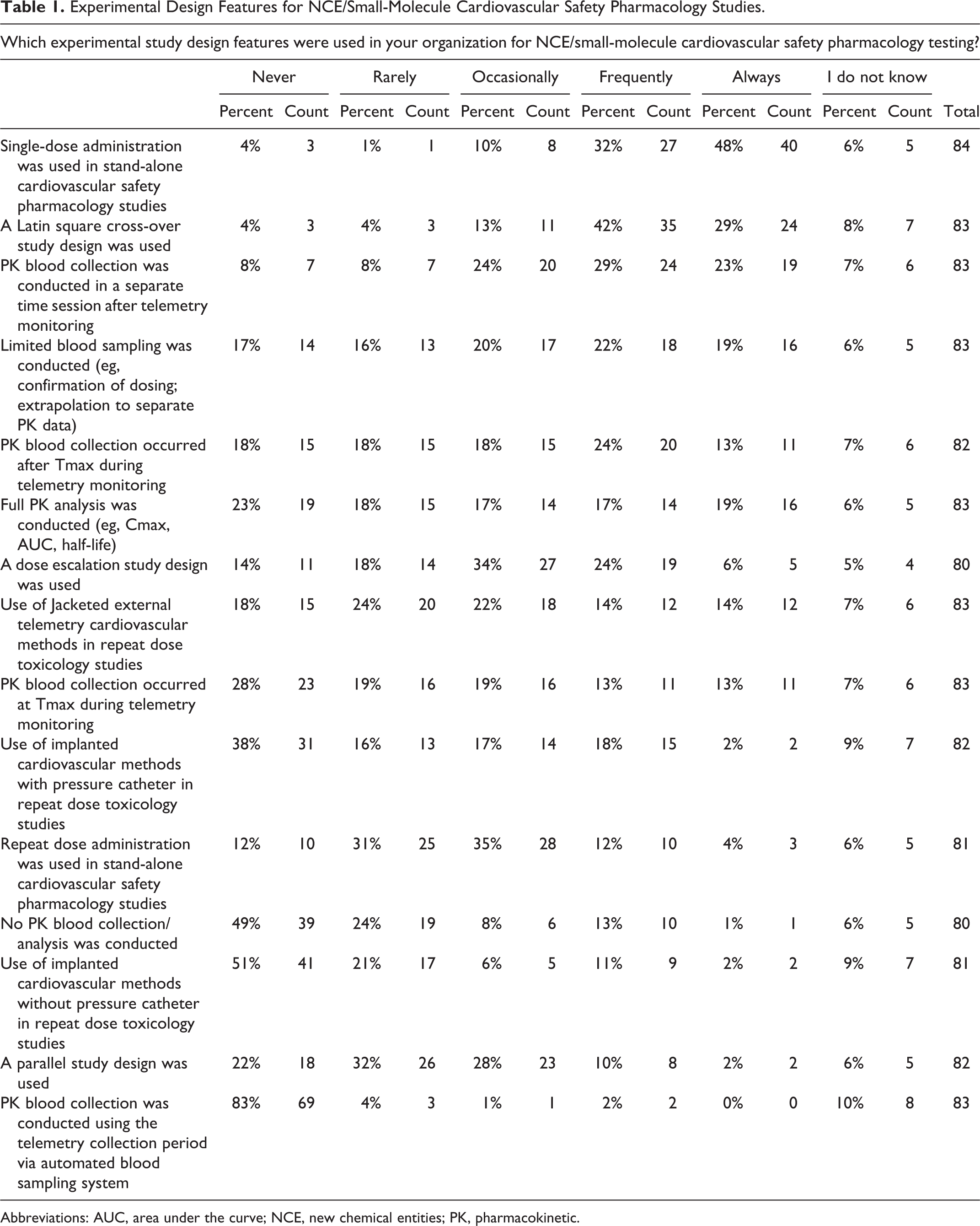
Abbreviations: AUC, area under the curve; NCE, new chemical entities; PK, pharmacokinetic.
Experimental Design Features for Cardiovascular Safety Pharmacology Studies With Large Molecules/Biologics.
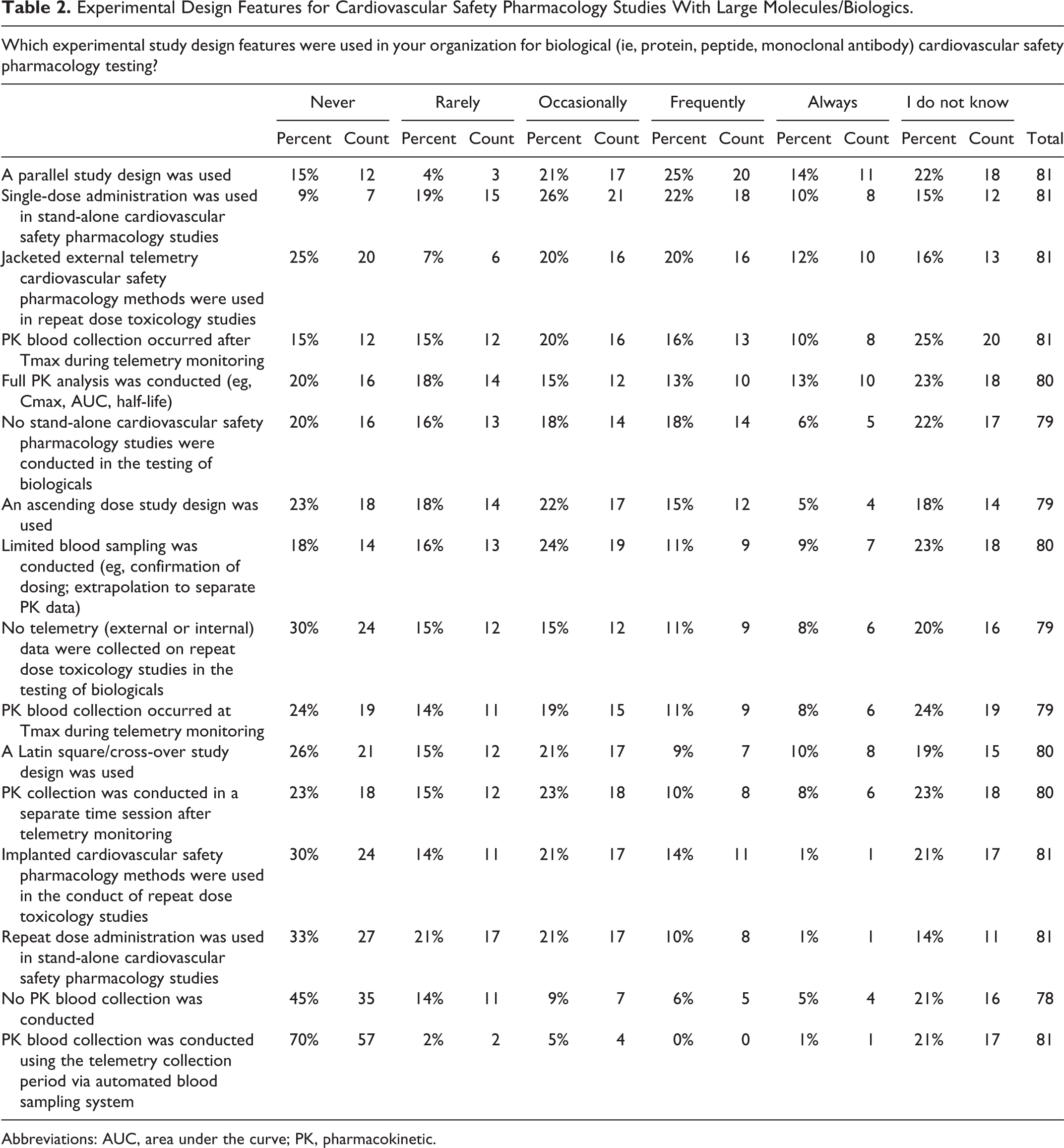
Abbreviations: AUC, area under the curve; PK, pharmacokinetic.
Novel CV Assays and End Points
Safety pharmacologists are well versed at developing, validating, and implementing novel nonclinical assays. An important aspect to this survey were questions related to the application of novel assay methods or the use of biomarkers in the conduct of CV SP studies. Interestingly, from a contemporary perspective (ie, within the last 5 years), both biomarkers (such as troponins and NT-proBNP) and echocardiography methods show limited use in standard CV SP studies. While 23% of responders used human induced pluripotent stem cell cardiac myocytes (hiPSC-CM), only 3% used human induced pluripotent stem cell–based neurons as a screen to investigate for drug-induced neurotoxicity (Figure 4).
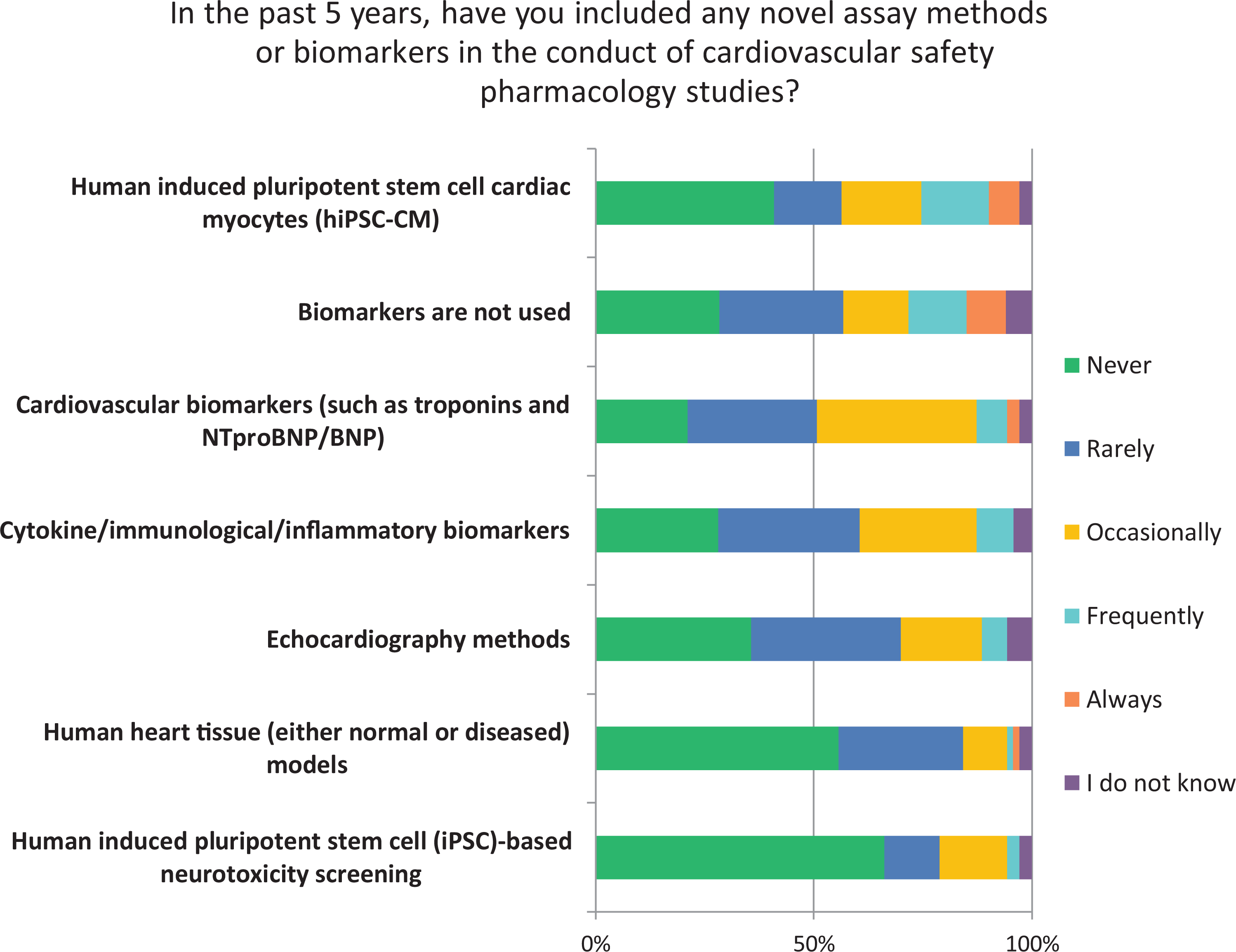
Novel assays or biomarkers in cardiovascular safety pharmacology.
Cardiovascular Experimental Study Variables
Most survey responders (72%) reported that body temperature was recorded during CV SP testing (Table 3). Similarly, most responders (73%) confirmed that a hERG IC50 value was frequently or always determined during in vitro ion channel testing. Most responders (62%) also reported the use of a single electroencephalography (ECG) lead and monitored physical activity levels (57%) in animals via telemetry recording methods. The use of an intravascular ECG lead was less common. Interestingly, only 42% of responders reported always, or only frequently, recording the left ventricular pressure; Table 3). In contrast, other useful study parameters including measurement of pulmonary arterial pressure, fractional shortening or speckle tracking/strain by echocardiography, central venous pressure, local arterial blood flow, cardiac output, and cardiac contractility (corrected for changes in heart rate or use of multiple ECG leads) were reported to never or rarely be used by most survey respondents.
Experimental Study Variables in Cardiovascular Safety Pharmacology Studies.
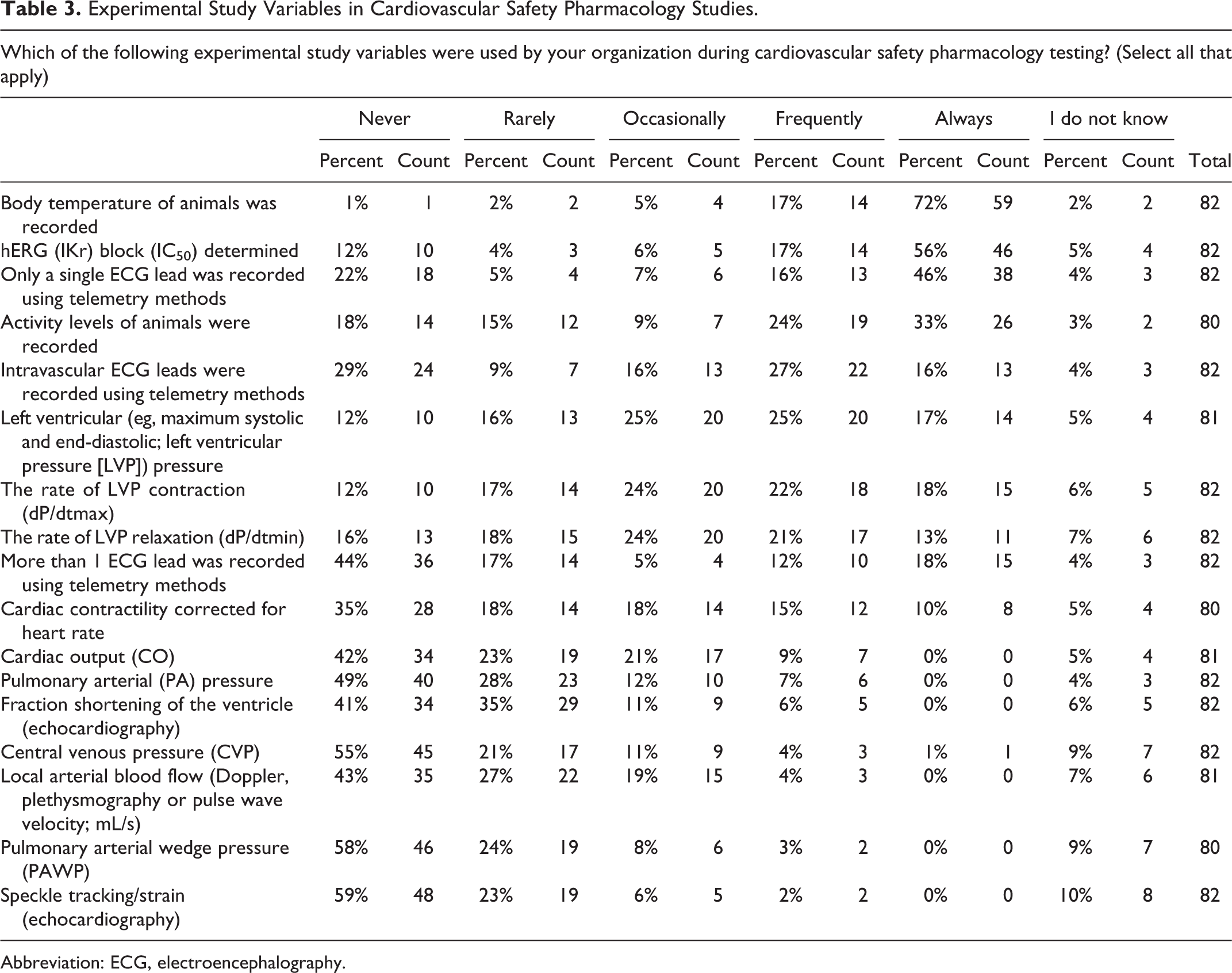
Abbreviation: ECG, electroencephalography.
Data Analysis in CV SP Studies
For GLP or non-GLP CV SP studies, most responders reported the frequent use of advanced statistical analysis methods that included analysis of variance (ANOVA) or analysis of variance covariate analysis (ANCOVA) and 2D graphical data representation (Tables 4 and 5). A power analysis calculation (used to determine the sample size that is required to detect an effect of a given magnitude with a given degree of confidence) for the standard CV SP study type was used by only 43% of responders. Even less frequently used by responders (33%) was the minimal detectable difference to determine on-study statistical sensitivity. In terms of drug-induced ECG effects, both individual animal QTc interval correction methods and standard QT (eg, Bazett, Fridericia or Van de Water) correction formulae were used (Tables 4 and 5). The use of larger data summary periods or superintervals (defined as average values of cardiac cycle–derived data usually greater than 1 hour in duration), simple statistical analysis (eg, Student t test), correction of the QT interval for body temperature, P-QRS-T amplitude measurements, PKPD modeling techniques, the use of J-Tpeak and Tp-e intervals, and 3D (ie, x, y, and z axis) graphical representation of data were rarely conducted by responders.
Analysis Methods in Non-GLP Cardiovascular Safety Pharmacology Studies.
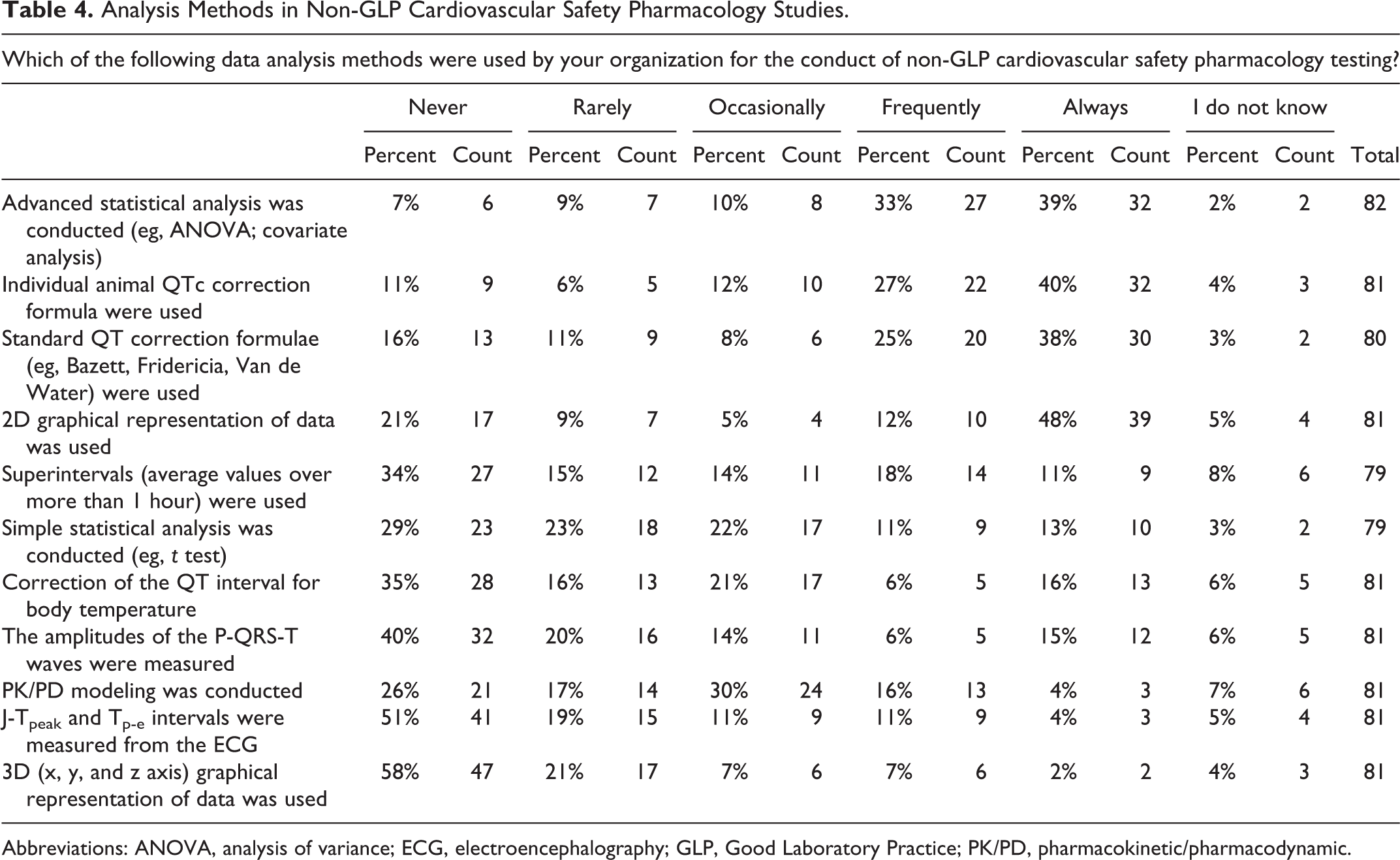
Abbreviations: ANOVA, analysis of variance; ECG, electroencephalography; GLP, Good Laboratory Practice; PK/PD, pharmacokinetic/pharmacodynamic.
Analysis Methods in GLP Cardiovascular Safety Pharmacology Studies.
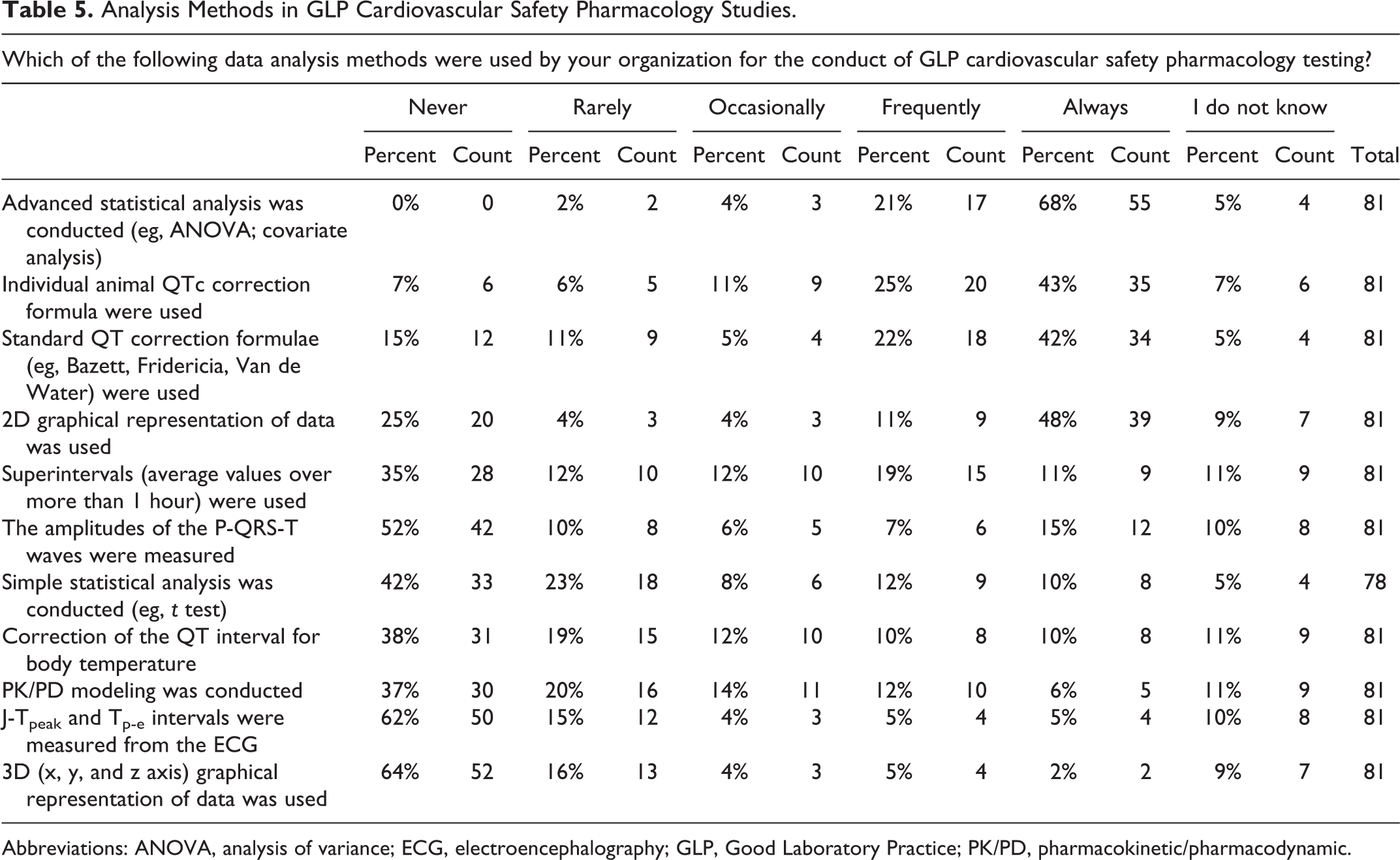
Abbreviations: ANOVA, analysis of variance; ECG, electroencephalography; GLP, Good Laboratory Practice; PK/PD, pharmacokinetic/pharmacodynamic.
A series of questions intended to determine the nature of the statistical analysis methods and study design aspects of CV SP studies were asked in the survey. A total of 87% of responders reported that studies were frequently, or always, designed with equal group sizes for each dose level tested, and 61% responded that studies are conducted with randomized dose groups (Table 6). The use of the term, no observed adverse effect level (NOAEL), a common toxicological term, has not been used extensively as a component of the SP lexicon defining the overall assessment of nonclinical safety. A review of the ICH S7A guideline provides no criteria for classifying an outcome as “adverse.” Thus, a large proportion of responders (46%) indicated that an NOAEL value was never, or rarely, determined from CV SP studies. Only 32% of responders used the term no observed effect level (NOEL), a similar toxicological term. The survey identified some interesting essentials regarding statistics. While responders did not conduct a statistical analysis with dose groups containing less than 4 animals per group, the use of blinded study groups was a rare study design practice.
Statistical Analysis Methods in Cardiovascular Safety Pharmacology Studies.
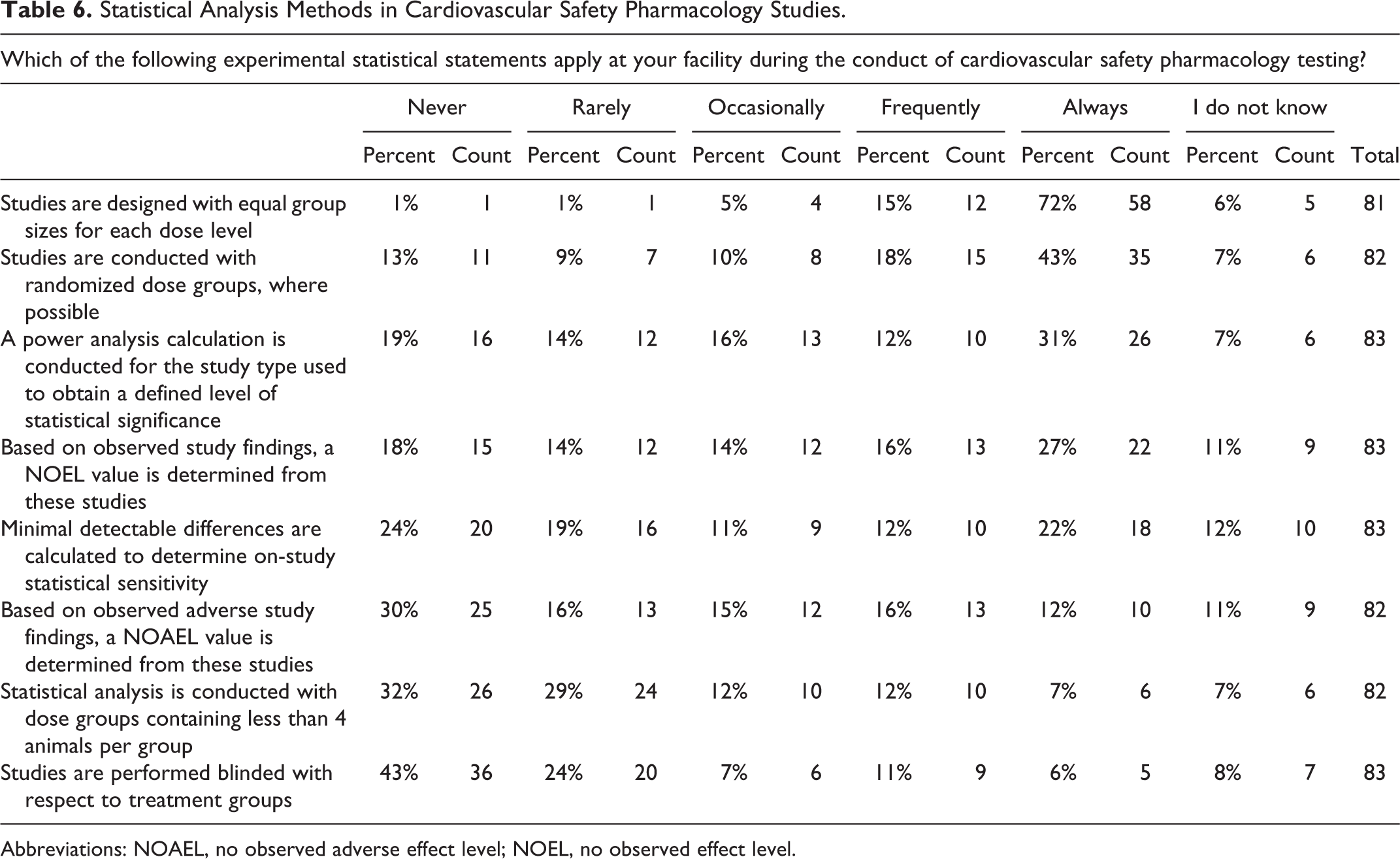
Abbreviations: NOAEL, no observed adverse effect level; NOEL, no observed effect level.
Cardiovascular and Other System Effects in Nonclinical and Clinical Studies
A majority of responders indicated that drug-induced arrhythmias observed in nonrodent studies were frequently, or always, confirmed by review (57%) and that an arrhythmia evaluation was included in the conduct of GLP repeat dose general toxicology studies in nonrodents (55%; Figure 5). However, responders indicated that a manual arrhythmia evaluation was not usually conducted at predetermined time points without automated (software based) detection.
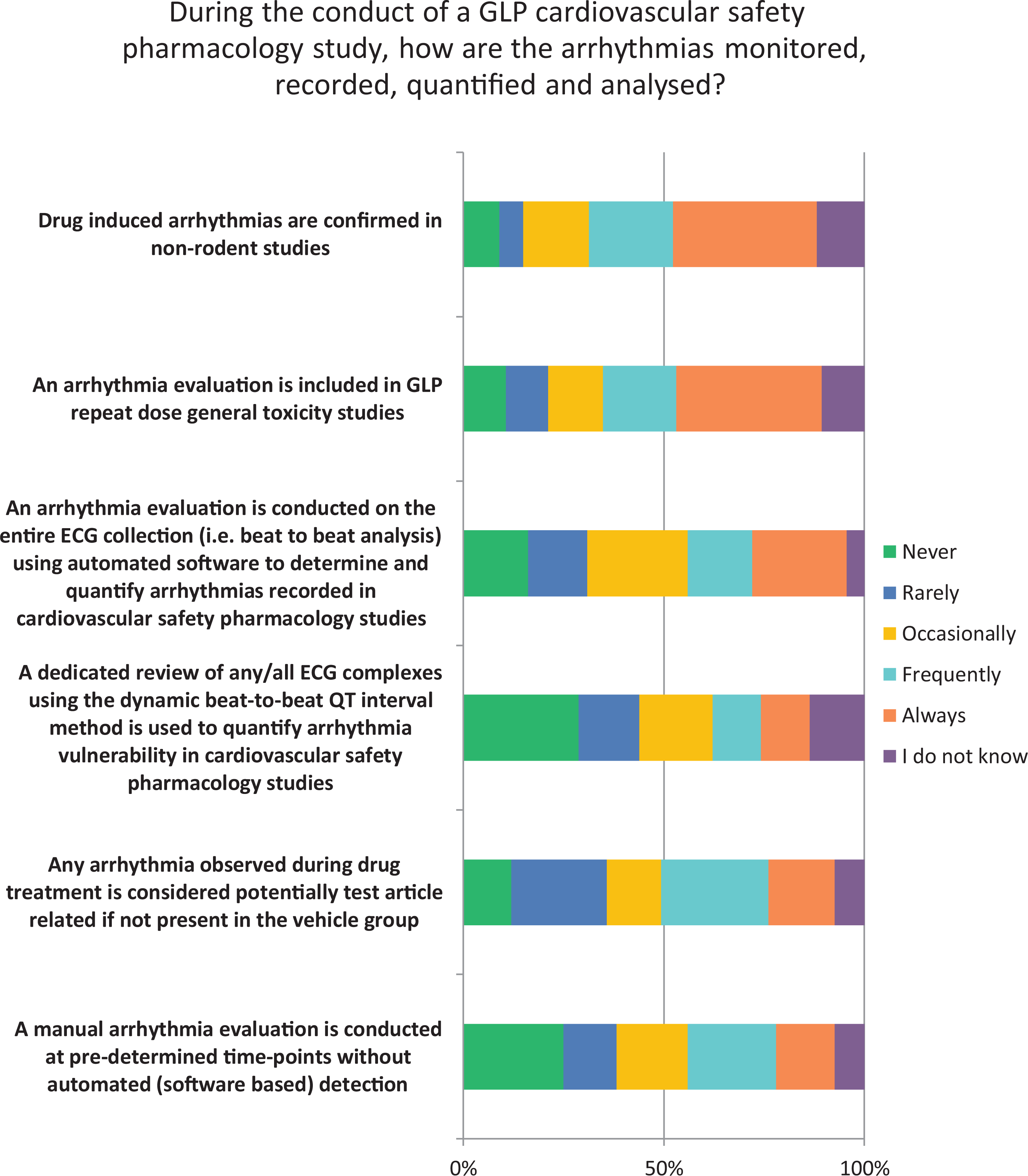
Arrhythmia analysis in cardiovascular safety pharmacology.
Support by responders for the use of study components, as proposed by the ongoing CiPA initiative, for inclusion in the nonclinical risk assessment of a drug candidate (and for regulatory submission) differed between the CiPA components interrogated (Figure 6). The in vitro assessment of drug effects on multiple cardiac ionic currents received high support from responders; however, conduct of the in silico computer modeling component that is used to predict proarrhythmic risk received limited support.
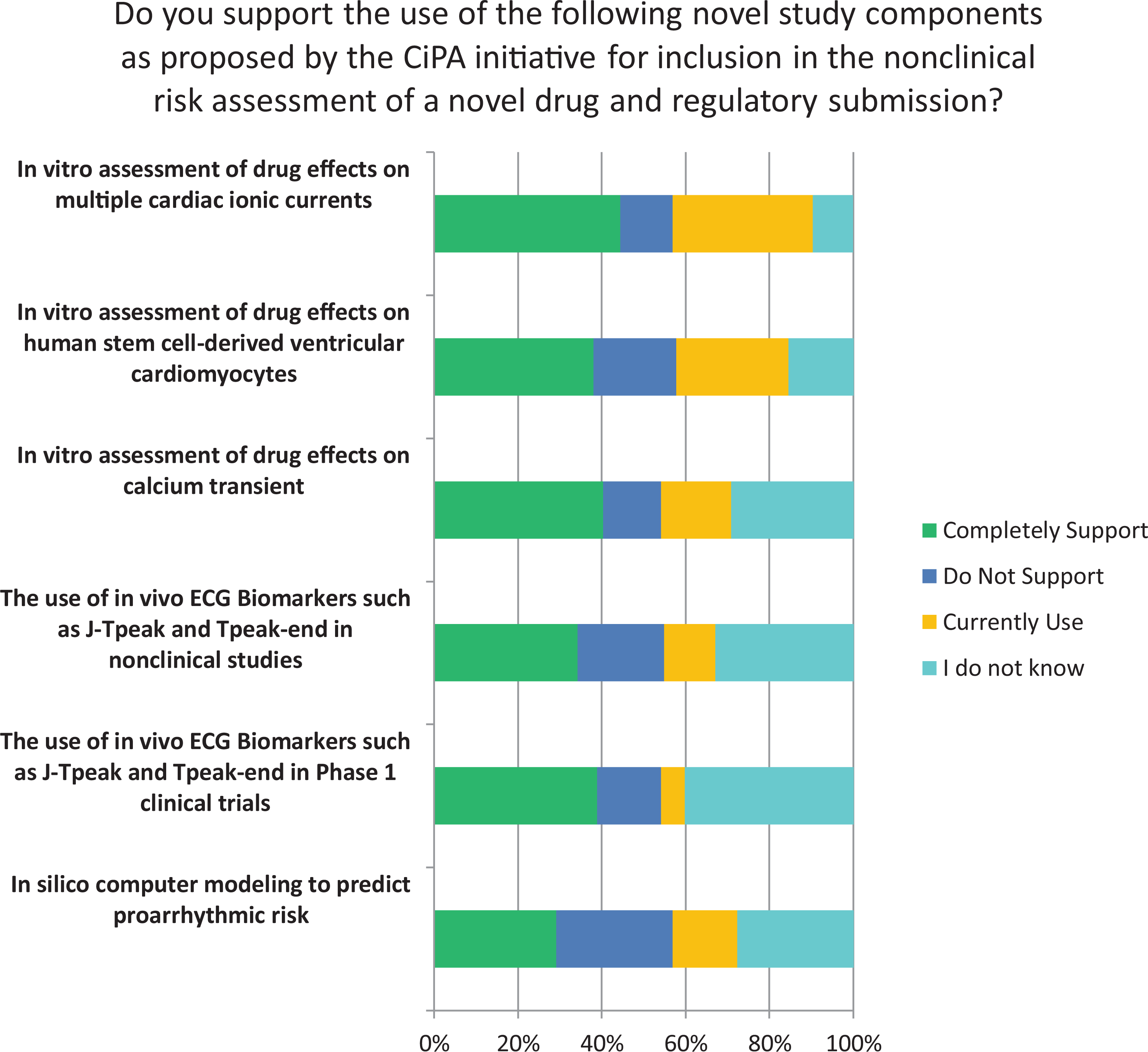
Support to Novel Study Components Proposed by CiPA. CiPA indicates comprehensive in vitro proarrhythmia assay.
When asked to provide details regarding those organ systems involved in drug-induced functional issues in CV SP testing, the largest number of survey responders observed chronotropic changes in the heart followed by changes to the ECG and arterial blood pressure (Figure 7). Since cardiac chronotropism was frequently observed by most survey responders (Table 7), it would be interesting to identify whether a threshold could be identified for the increase in heart rate using current telemetry methods. The next most frequently reported CV effect observed by responders included an increase in arterial blood pressure, hERG (IKr) block, decreased arterial blood pressure, decreased heart rate, QT/QTc prolongation, and body temperature changes. Prolongation of both the PR interval and QRS width of the ECG was observed by 61% and 46% of responders, respectively, which typically correlates with a negative chronotropic effect on the heart. This combination of effects was observed by 66% of responders, while a decrease in ventricular contractility was reported by 54% of responders. Effects that were less frequently observed in CV SP studies included delayed QT/QTc prolongation (suggestive of a hERG-trafficking effect), cardiac sodium channel block, drug-induced atrioventricular block, and QTc interval shortening. Most responders also reported that several organs systems were rarely involved with drug-induced functional concerns in nonclinical SP testing. These include the auditory, ocular, peripheral nervous, musculoskeletal, renal, and the respiratory system (Figure 7).
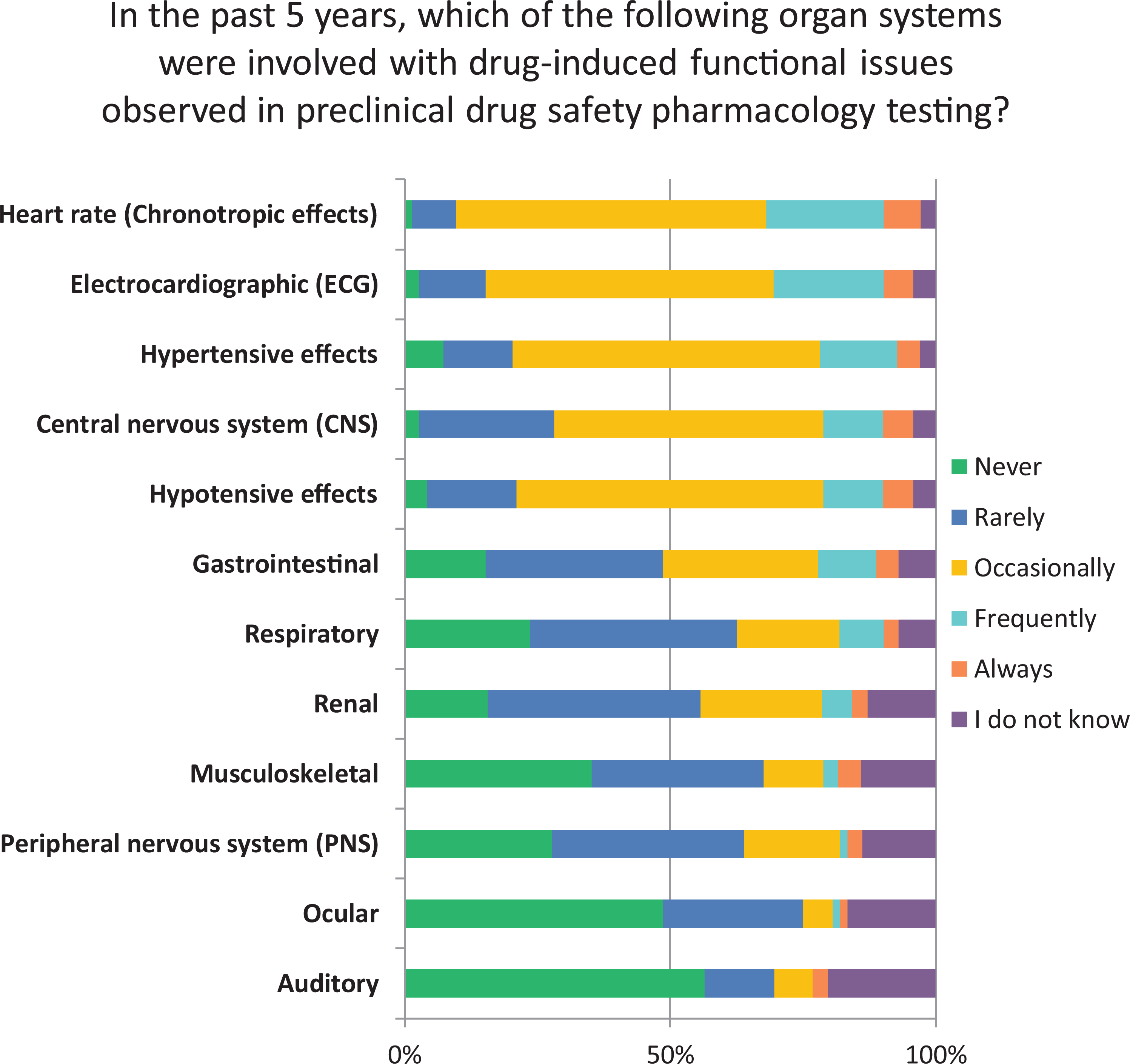
Organ systems involved with drug-induced functional issues in safety pharmacology testing.
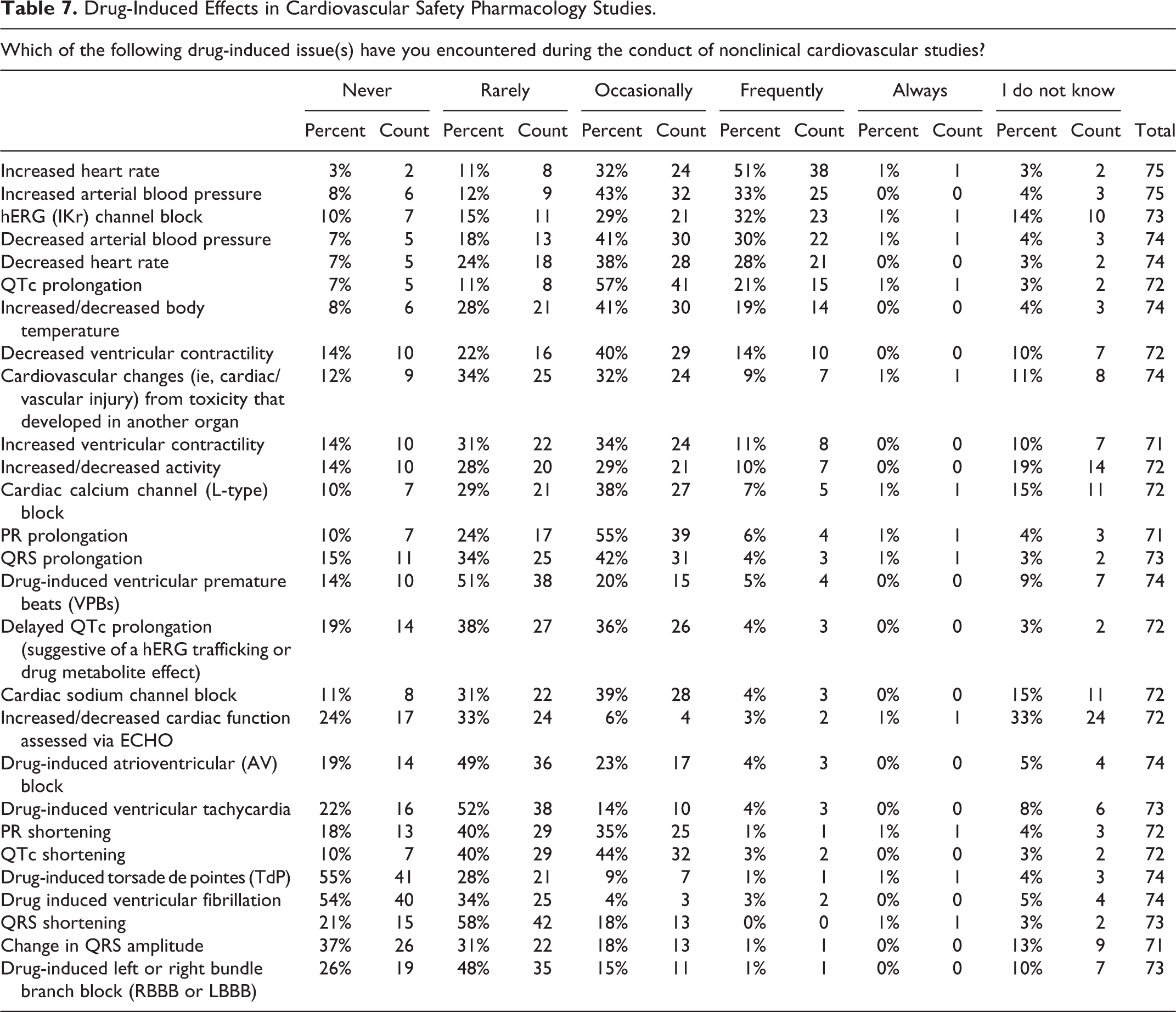
Drug-Induced Effects in Cardiovascular Safety Pharmacology Studies.
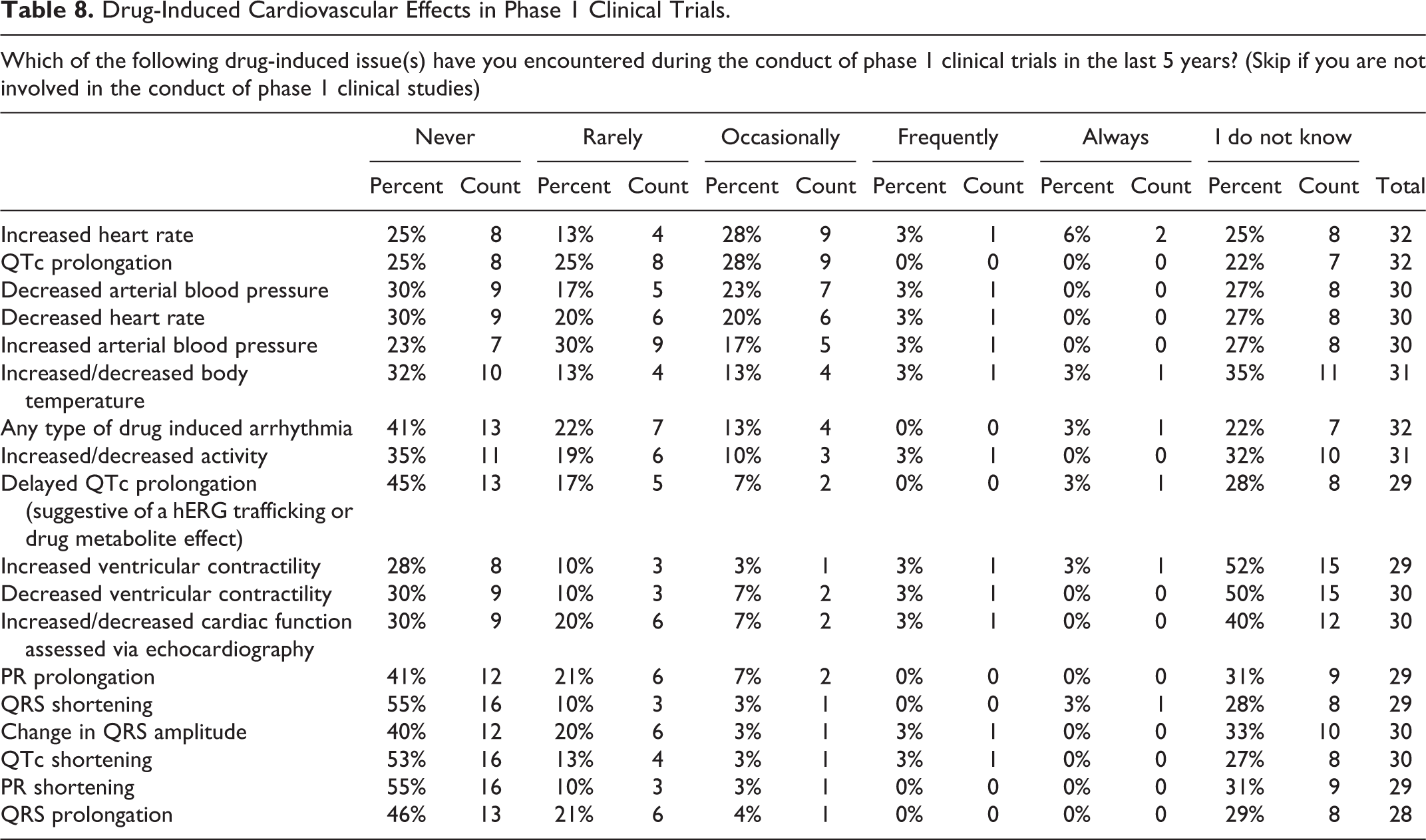
It is of particular interest that most responders declined to answer the survey question regarding the CV effects observed in phase 1 clinical studies. This suggests that those who conduct SP studies are unaware of clinical events for a given compound that advances to an early phase clinical development team. For those who did respond (n = 29-32), chronotropic effects, QTc interval prolongation, and changes in arterial blood pressure were most frequently observed (Table 8).
Drug-Induced Cardiovascular Effects in Phase 1 Clinical Trials.
Non-GLP and GLP CV SP Testing
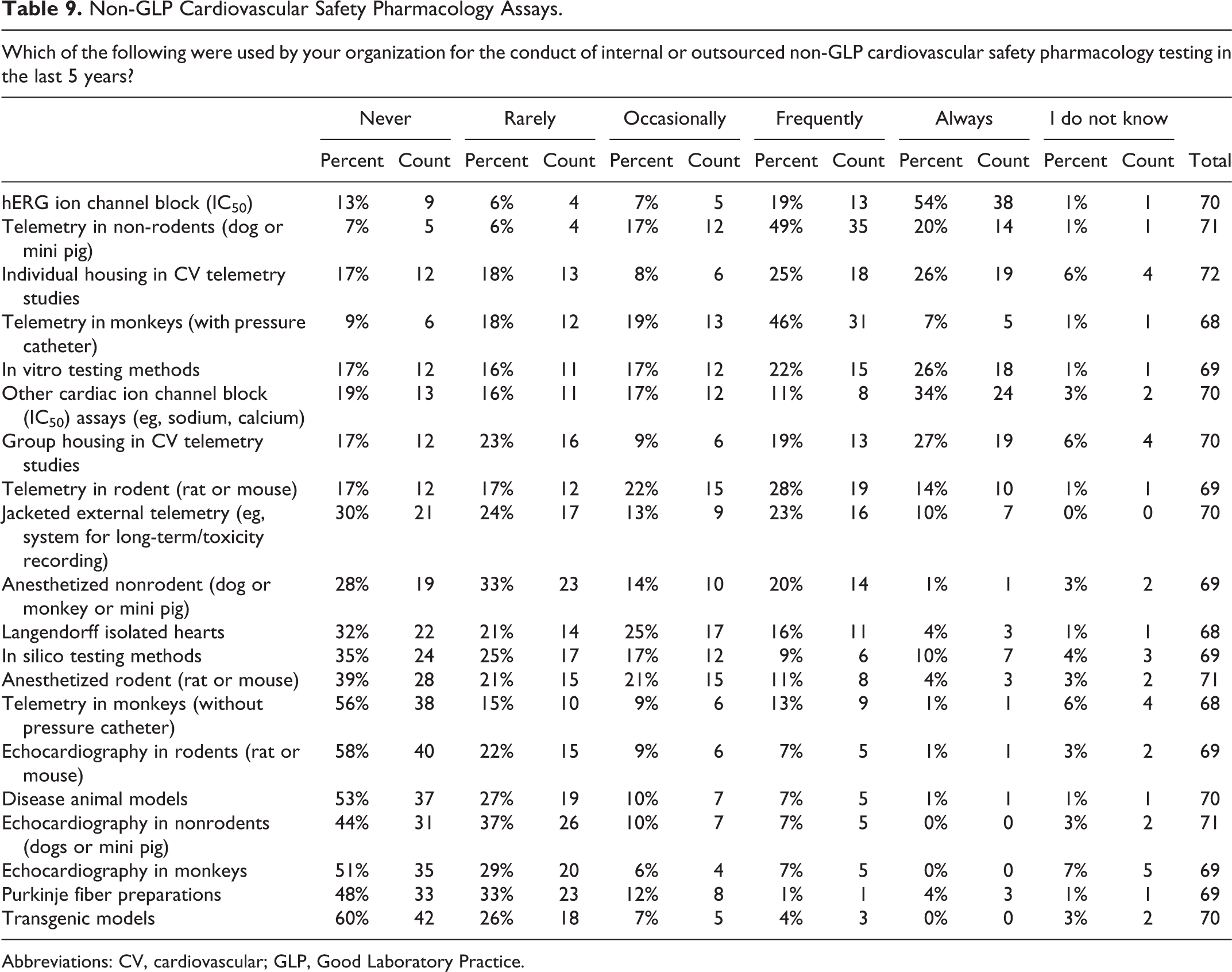
As expected, most responders determine the IC50 value for IKr (hERG) ion channel block and conduct an in vivo evaluation using telemetry methods in nonrodent species (ie, dogs, mini pigs, or nonhuman primates) for both non-GLP and GLP CV SP testing (Tables 9 and 10). Most responders (72%) primarily use non-GLP in vitro testing assays and rarely/never use other assays including Langendorff isolated hearts, anesthetized nonrodent models, echocardiography (all species), disease models, Purkinje fiber preparations, or transgenic models in their non-GLP or GLP CV SP testing paradigm. Most responders less frequently conducted GLP compliant in vitro testing of other cardiac ion channel (IC50) assays (eg, for sodium or calcium channels). Lastly and somewhat surprisingly, individual animal housing (rather than group housing) remains a frequent practice in the conduct of both GLP and non-GLP CV studies by most responders.
Non-GLP Cardiovascular Safety Pharmacology Assays.
Abbreviations: CV, cardiovascular; GLP, Good Laboratory Practice.
Stand-Alone GLP Cardiovascular Safety Pharmacology Assays.
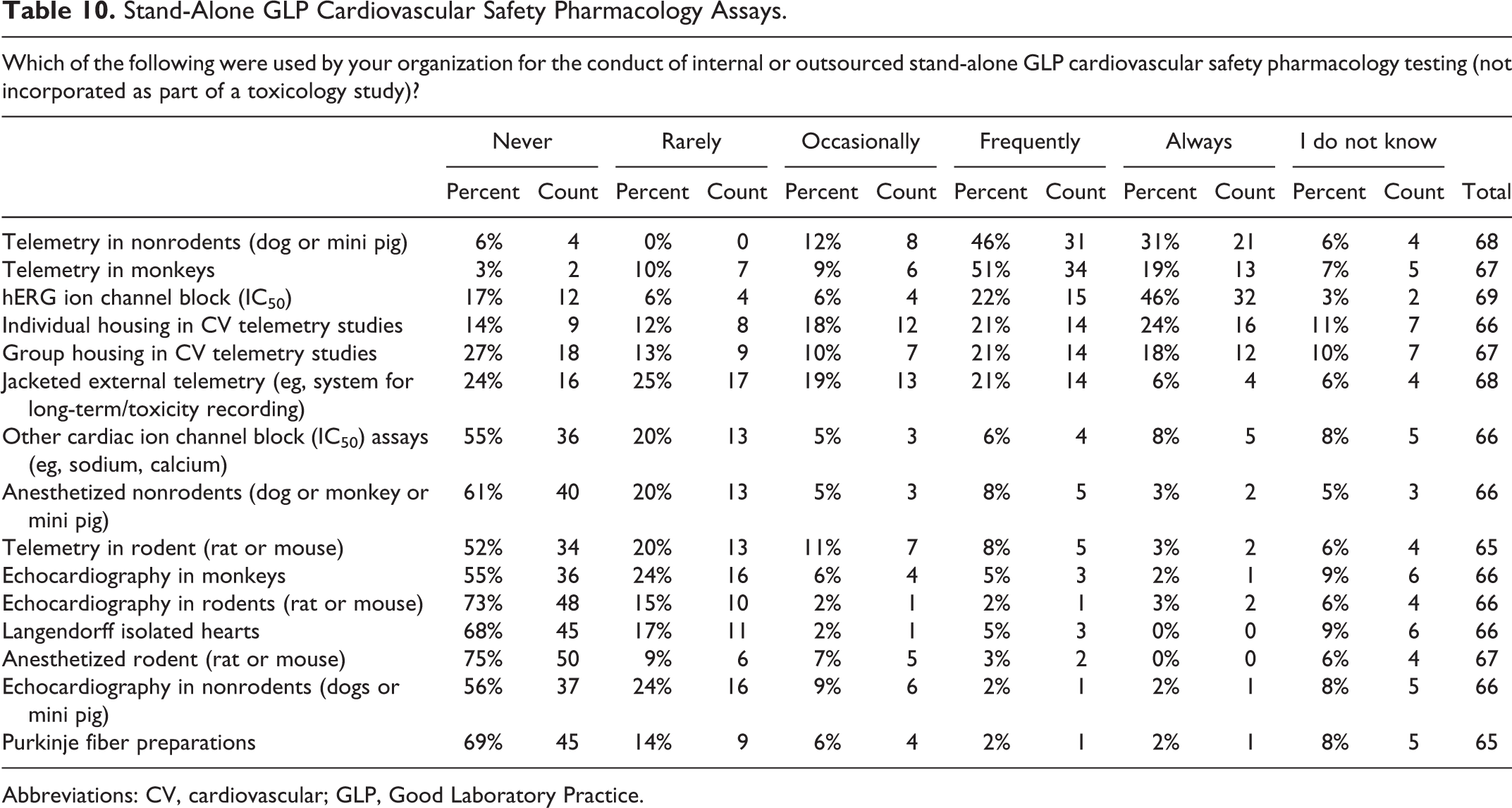
Abbreviations: CV, cardiovascular; GLP, Good Laboratory Practice.
Considerations in the Conduct of GLP CV SP Assays and SEND Data Sets
Most survey responders reported that doses that are chosen for evaluation in single-dose CV SP studies were not based on clinical doses and exposures (as these are not known at the time of study conduct) but rather doses were frequently selected based on findings from maximum tolerated dose (MTD)/dose-range finding toxicology studies (Table 11). The SP dose selection process considers the anticipated therapeutic index as doses should include, and exceed, the primary pharmacodynamic (PD) or therapeutic range; however, this was not incorporated into the survey questionnaire. Most responders reported that candidate drugs have only rarely/occasionally been eliminated from further development based on CV SP findings.
Considerations in the Conduct of Cardiovascular Safety Pharmacology Assays.
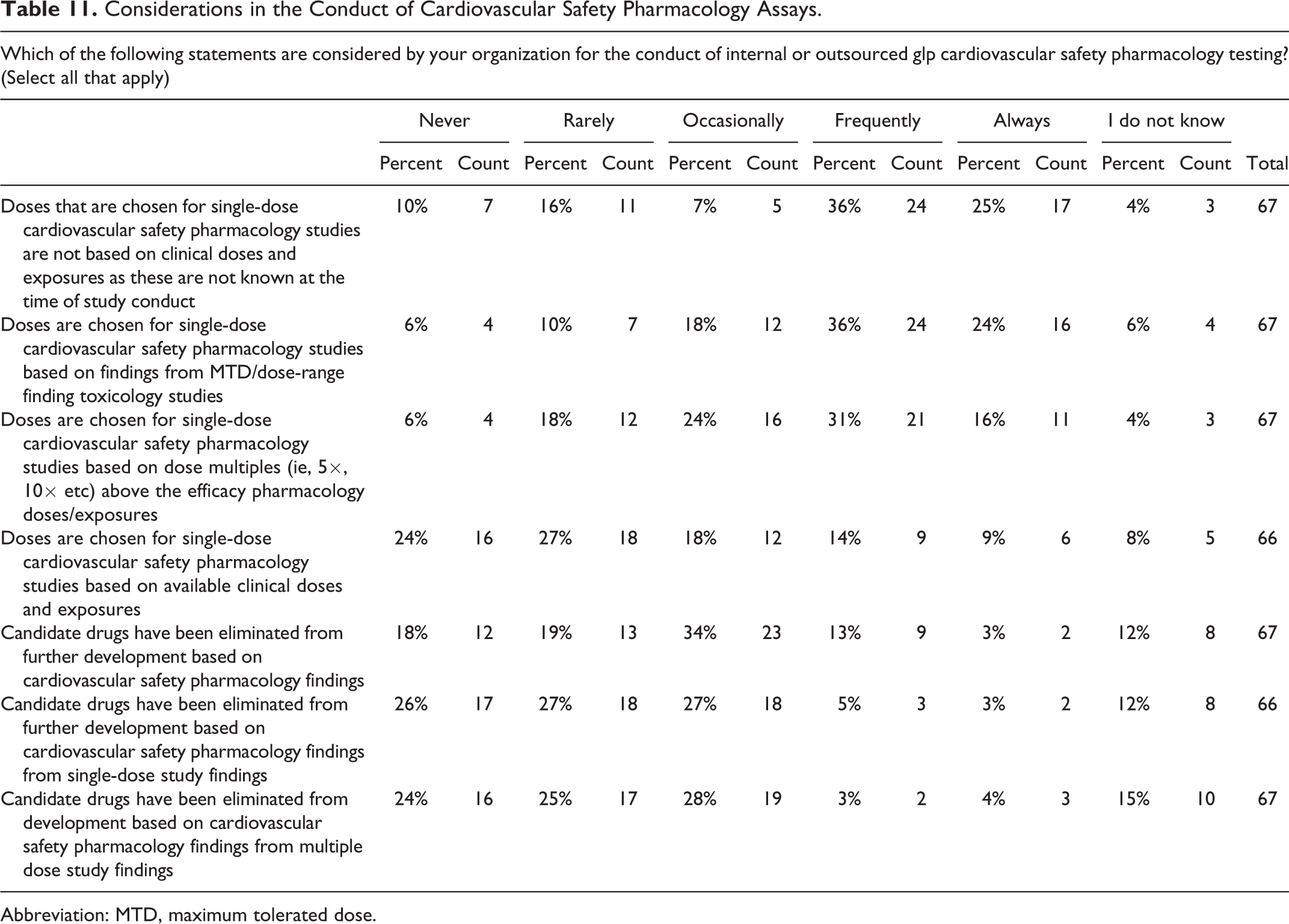
Abbreviation: MTD, maximum tolerated dose.
Standard for Exchange of Nonclinical Data (SEND) is the primary component of a national electronic system that has recently been developed for nonclinical studies and will be used by the US Food and Drug Administration (FDA) to track drug safety upon completion of marketing authorization. Thus, upon issuance of the FDA guidance, a SEND initiative was developed in order to provide an organization as well as structure and format for the standardization of nonclinical tabulation data sets for exchange of study data. The current SEND version 3.1 standard applies to the core battery of SP CV and respiratory studies but will expand to include additional SP topics including central nervous system (CNS) studies and also the hERG assay. Safety pharmacology groups that sponsor and/or perform these standard core battery studies must have these practices incorporated or work with their CROs to understand the guidelines, manage the data, and prepare for use. Several questions regarding the applicability of SEND in the conduct of CV SP studies were asked in the survey. The most frequent response for all questions related to SEND was “I do not know” (Table 12). The survey identified a number of fascinating facts including the fact that most responders did not know if the laboratory recording system used at their institution was able to utilize the assembled Controlled Terminology list for SEND studies. Of relevance to CV SP were a number of aspects including the fact that responders did not know if the system used was capable of describing the preclinical arrhythmias observed during the conduct of CV SP studies. Responders (52%) did not know whether the veterinary cardiologist was recording the results from their analysis in a SEND compatible electronic data set or if SEND study data sets were generated for all CV SP studies prior to study finalization (Table 12).
Standard for Exchange of Nonclinical Data in the Conduct of Cardiovascular Safety Pharmacology Assays.
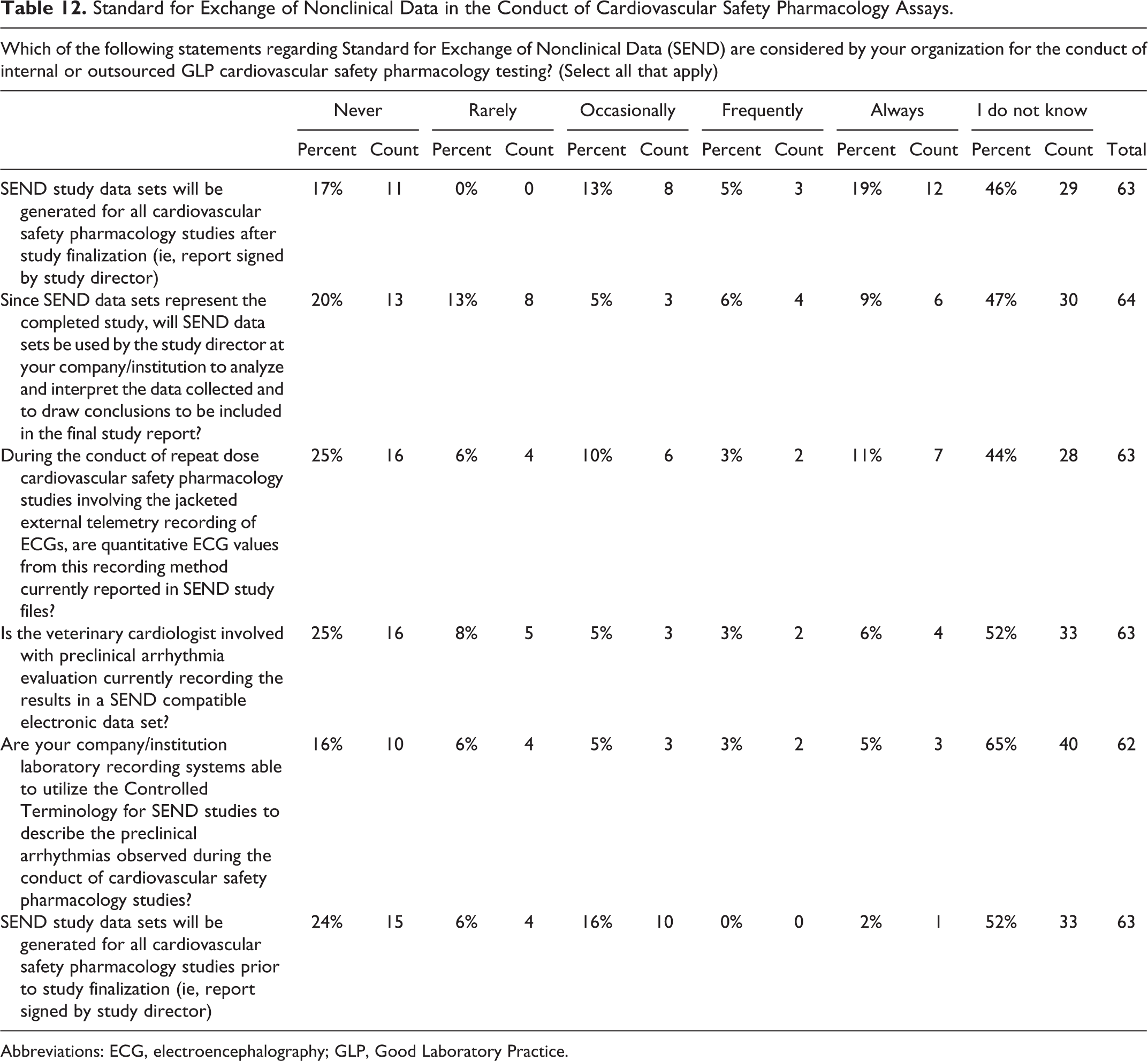
Abbreviations: ECG, electroencephalography; GLP, Good Laboratory Practice.
Discussion
Consistent with the SPS tradition of publishing industry surveys on SP-related topics to inform on trends and practices in the field, 13 -17 this survey explored important additional facets of CV SP. The present survey aimed to further examine current practices and strategies that are used principally by those drug safety scientists working in the pharmaceutical industry relative to CV SP model selection, study design, and data interpretation. The survey scope encompasses a range of areas including fundamental aspects of in vitro and in vivo assays. Additionally, the survey queries responders for their understanding regarding SP data use in early clinical trials and facets of the SEND initiative.
Survey demographics reflected the SPS membership with a balanced participation from both North America and Europe, however, with a trend toward higher participation from Asia compared to previous SPS surveys. 14 The distribution according to company size was very comparable to previous SPS surveys with 52% of responders from companies with >1,000 employees. The potential participation of several employees from the same organization and the lack of clinical expert representation were identified as a limitation to the survey as these aspects were not quantified. There was a noticeable increase in the number responders working for companies that develop both NCEs and biologics compared to previous surveys and a corresponding decline in the number of responders working for companies that only develop NCEs. However, such a change in dynamics is in keeping with the shifting focus of drug development portfolios within most pharmaceutical companies. The survey identified the primary therapeutic areas that were targeted by the companies employing survey respondents. Oncology remained the most frequently targeted therapeutic area (52%) but with an apparently lower proportion of responders than previous SPS surveys. Immunology/inflammation, neurology, and cardiometabolic disease remained among the most frequently targeted therapeutic areas at response rates (∼45%) that were similar to previous SPS surveys. However, unlike those surveys, the current survey provides increased granularity into certain topics such as the types of large molecules/biologics that are currently in development. These biologics now include antibody–drug conjugates, antibody-based therapies, and peptides currently being evaluated by about one-third of responders (Figure 1).
In stand-alone CV SP studies sex selection, group size and overall general study design is consistent when compared to a previous SPS survey conducted over a decade ago. 14 In contrast, the inclusion of a TK blood collection time point during the conduct of study telemetry monitoring represents an emerging practice for SP scientists. However, concerns persist within the scientific community regarding the potential impact on data quality due to sampling artifacts. Although the practice of including blood collection during telemetry monitoring was not a question included in previous SPS surveys, it was a topic of discussion at several SPS webinars. Some resolution is required in the near future. Likewise, in a previous SPS survey, 17 the potential limitations to the use of hiPSC-CM in drug safety evaluations appeared numerous and included aspects such as reliability of the cells to represent adult cardiomyocyte phenotypes, stability of cardiac ion channel expression, and the financial impact to establish the assay. 17 -20 However, responses in the current survey suggest that the implementation and use of hiPSC-CM has become less formidable, with 23% of responders now reporting frequent use of this in vitro assay. While this is likely attributed to both technological advances achieved with advanced engineering of hiPSC-CM as well as a plethora of published data using a multitude of drugs in recent years, there remains some concerns relative to the adult phenotype of the cells used in these emerging in vitro models. 5,21,22
It is important to compare and contrast findings derived from responses in this survey to previously conducted SPS survey as this informs whether change has occurred with regard to study design and practice over time during the conduct of SP studies. In a recent survey that investigated the developing CiPA initiative, 16 it was revealed that a majority (59%) of responders reported that their company had abandoned at least one pharmacophore (or potential “toxicophore”) based on data obtained from assays characterizing drug-mediated blockade of IKr. The negative correlation between body temperature and the QTc interval is well-documented in both anesthetized 23 and conscious dogs. 24 Herein, we report that body temperature is recorded by most responders during the conduct of CV SP studies reflecting the importance of knowing changes in body temperature during telemetry recording. Lastly, determination of the hERG IC50 for a novel drug candidate, the use of an ECG signal derived from a single lead, and monitoring of the physical activity of the animals remain consistently recorded by responders. These established CV practices indicate stability in the nature of the CV SP study paradigm.
Biostatistics remains an essential aspect to the conduct of contemporary CV SP investigations, 25 however, while the implementation of appropriate biostatistical methods remains a complex endeavor for many biological sciences, SP scientists have defined appropriate statistics for the types of studies conducted. The current survey results suggest a modest increase in the use of the power analysis, with at least 43% of responders frequently conducting a power analysis calculation for the study type used to obtain a defined level of statistical significance using formal group size estimations. When this responder rate is compared to the previous SPS survey by Lindgren et al, 14 this is an increase of 12%. While the use of advanced statistical analysis methods such as the ANOVA remains constant at 72% between survey responders, the use of ANCOVA has increased among responders reporting frequent use of such analyses in both non-GLP and GLP studies. In the 2008 SPS survey, 71% of responders indicated using both parametric and nonparametric data analysis methods. 14 Use of these statistical methods may be less robust for certain CV parameters that may require multifactorial analysis due to the interdependence of several functional parameters. The use of 3D graphs is considered to aid, characterize, and visualize these complex correlations. 26 The current survey highlights the frequent use of 2D graphical representations of data; however, despite the advantages attained using 3D graphs, this method has yet to become common practice.
No observed adverse effect level is a toxicological dose descriptor that denotes the level of exposure an organism receives at which there is no biologically (or statistically) significant increase in the frequency or severity of any adverse or toxic effect. 27 As it is a known toxicological term that is derived regularly in repeat dose toxicology studies, it has not been used extensively as a component of the SP lexicon to define the overall nonclinical safety assessment. In alignment with the recent publication by Baird et al 28 and Andersen et al, 29 responders of the survey confirm that the use of the term NOAEL is relatively rare in CV SP studies but that use of the descriptor NOEL is common.
Pharmacokinetic/PD modeling has become an integral component in the drug development process. It is a mathematical technique that predicts the efficacy and effect of drug dosing over a period. Despite well-established methodologies 30 and broad clinical use, 31 responders indicate that PKPD modeling remains only marginally used as a methodology.
The characterization of both arrhythmia incidence and severity in animal models 32 -34 and humans 35 is central to the interpretation of the potential for a drug to produce arrhythmias. 36 This survey attempted to examine the methodologies most often employed during the conduct of arrhythmia analysis in CV SP studies. With the implementation of improved computerized arrhythmia detection systems, 37,38 there is an expectation that methodologies will shift toward higher throughput approaches. However, this survey confirms that a minority of responders still use a manual arrhythmia evaluation at predetermined study time points without automated (software based) detection methods.
In a previous SPS survey, 92% of responders supported the CiPA initiative, 16 an expected response rate given the robust nonclinical and clinical pharmacological characterization that was ongoing at the time. 39 However, the current survey highlights development of a more complex position. While there is general acceptance for the use of multi-ion channel testing aspects of the paradigm, there is diminished support for the use of the in silico modeling. The testing of drug effects using hiPSC-CMs is currently used or endorsed by a high proportion (65%) of responders.
Recently, clinical data have emerged suggesting that use of the J to T-peak (JTp) interval of the ECG may differentiate between selective IKr blockers associated with a high proarrhythmic risk and drugs with a balanced multi-ion channel inhibition profile. 9 Similarly, a recent retrospective analysis of data from nonclinical studies in dogs and nonhuman primates suggests a similar discrimination for JTp nonclinically. 40 Despite this apparent concordance, only a small proportion of responders (34%) support the use of JTp as a novel proarrhythmia biomarker in CV SP studies. It should be noted that a comparable proportion (33%) were undecided regarding support for the use of JTp in SP studies.
The diverse range of nonclinical methods and models (in silico → in vitro → in vivo) that have developed since the inception of SP 20 years ago underscores the complex nature of the integrated proarrhythmic risk assessment. The key to its success as a measure of CV drug safety is the ability to translate signals derived from these models and ensure sufficient margins to limit the risk of drug-induced arrhythmia (ie, TdP) in humans. Thus, the concordance of preclinical and clinical data remains an everlasting priority in order to maintain the value of SP models. 41,42 Positive chronotropic and pressor effects on blood pressure were the most frequently observed adverse events in CV SP studies. The survey did not attempt to elucidate the basis for these changes, but it could be hypothesized that these effects were secondary to other drug-mediated physiological changes (eg, CNS-mediated CV effects through altered autonomic tone). Despite a thorough understanding of the hERG/IKr channel structure, resulting effects on ventricular repolarization, 6 and evolving medicinal chemistry methods to limit drug-association with the IKr channel protein, IKr block remained one of the most frequent drug-associated issues reported by responders. A drug-induced decrease in ventricular contractility was frequently observed by most responders (54%) in SP studies. However, since oncology remains the most frequently targeted indication in the current survey and several drugs in this therapeutic area are well characterized for their negative inotropic effects, 43 this finding likely contributes to this observation.
Without surprise, the 2 historical pillars of CV SP, hERG ion channel block (IC50) and nonrodent telemetry methods, were most frequently used in both non-GLP and GLP studies by responders. Cardiac repolarization investigations bring considerable value to support the discovery and development of safer medicines. 44 Interestingly, only minor differences in the frequency of use were noted between dogs/mini pigs and nonhuman primates. This is likely due to the relatively high proportion of companies developing large molecules/biologics in the current survey. Biologics often require the use of nonhuman primates as the most relevant nonclinical testing species. Surprisingly, the use of GLP compliant in vitro testing of other cardiac ion (IC50) channels (eg, sodium, calcium) was relatively rare among responders but is likely due to the acceptance of non-GLP data obtained from high-throughput screening platforms. Similarly, the Langendorff isolated heart model, which has an extensive history of use in SP, 45 is also now reported to be rarely used by responders. This may perhaps be due to the availability of low-cost/high-throughput screening in vitro assays and the generalized use of standardized in vivo models, the latter of which ultimately can be used to bridge the any data not obtained with the Langendorff isolated heart model.
The data also highlight the slow implementation of social housing in non-GLP and GLP SP telemetry studies. The relatively high frequency of CV monitoring with the single housing of animals may be due to the slow but progressive adoption of digital telemetry technologies enabling social housing during the CV monitoring phase of studies and the requirement for validation before use in the conduct of GLP studies. Expect that the transition will accelerate as validation programs tailored to the digital telemetry platforms reach maturity.
Dose selection for use in CV SP studies is only based on limited available information prior to the conduct of SP studies, and predicting human efficacious exposures remains a challenge prior to first in human studies. This survey highlights the importance of data derived from MTD/dose-range finding toxicology studies used in the establishment of dose levels for CV SP studies. Interestingly, the inclusion of CV monitoring in the MTD//dose-range finding toxicology studies may address potential CV risks early in the development spectrum. Similar to previous SPS survey results, most responders report that drug candidates are rarely eliminated from further development based on adverse CV findings in nonclinical studies. Only 10% of responders from the 2017 SPS survey reported that their company abandoned development of a drug due to an inability to eliminate hERG channel blockade, 16 recognized as the most prevalent drug-induced adverse effects in the heart.
This survey suggests that practices related to the application of SEND to CV SP data remain vastly uncharted. The highest levels of uncertainty by responders appeared related to the use of a SEND compatible acquisition platform by the veterinary cardiologist reviewing study ECGs and for recording arrhythmias. However, it is expected that this will evolve rapidly with the implementation of SEND across all areas of GLP nonclinical drug safety testing including SP.
Conclusion
The current SPS survey revealed interesting trends in the evolution of CV SP practices and strategies. A majority of the discipline remained remarkably stable over more than 2 decades using hERG testing and in vivo core battery study design methods and parameters. Oncology remained the most frequently targeted therapeutic indication with signs of an increased number of companies developing biologics. Respiratory SP methods were rarely involved with drug-induced functional issues in preclinical SP testing within the last 5 years for most survey responders. Areas with noticeable advancement included arrhythmia detection and statistical analyses, while advanced CV assessment methods remained less commonly used. Survey participant support for CiPA was strong, but the level differed considerably across assay types recommended for use in the paradigm. Future SPS surveys must establish the direction, progress level, and trends within this pivotal new chapter of SP.
Footnotes
Authors’ Note
This publication reflects the views of the authors and does not represent views or policies of any organization, including the FDA. The views of the authors do not represent the FDA’s views or policies.
Acknowledgments
The authors want to thank the Safety Pharmacology Society (SPS) for support in the conduct of this survey.
Author Contribution
Simon Authier contributed to conception, contributed to analysis, and drafted the manuscript. Matthew M. Abernathy contributed to conception and critically revised the manuscript. Krystle Correll contributed to conception and critically revised the manuscript. Ray W. Chui contributed to conception and critically revised the manuscript. Jill Dalton contributed to conception and critically revised the manuscript. C. Michael Foley contributed to acquisition and critically revised the manuscript. Gregory S. Friedrichs critically revised the manuscript. John E. Koerner contributed to conception and critically revised the manuscript. Mary-Jeanne Kallman critically revised the manuscript. Malar Pannirselvam critically revised the manuscript. William S. Redfern critically revised the manuscript. Vijay Urmaliya contributed to conception and critically revised the manuscript. Jean-Pierre Valentin contributed to conception and critically revised the manuscript. Todd Wisialowski to conception, contributed to analysis and critically revised the manuscript. Tanja S. Zabka critically revised the manuscript. Michael K. Pugsley contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
