Abstract
Acrylamide (AA) is a food contaminant present in a wide range of frequently consumed foods, which makes human exposure to this toxicant unfortunately unavoidable. However, efforts to reduce the formation of AA in food have resulted in some success. This review aims to summarize the occurrence of AA and the potential mitigation strategies of its formation in foods. Formation of AA in foods is mainly linked to Maillard reaction, which is the first feasible route that can be manipulated to reduce AA formation. Furthermore, manipulating processing conditions such as time and temperature of the heating process, and including certain preheating treatments such as soaking and blanching, can further reduce AA formation. Due to the high exposure to AA, recognition of its toxic effect is necessary, especially in developing countries where awareness about AA health risks is still very low. Therefore, this review also focuses on the different toxic effects of AA exposure, including neurotoxicity, genotoxicity, carcinogenicity, reproductive toxicity, hepatotoxicity, and immunotoxicity.
Introduction
For thousands of years, people have used heat to cook their food. Thermal processing has a great importance in terms of microbiological safety, nutritional quality, and the desired sensory properties, such as color, texture, and flavor; however, undesirable chemicals have arisen as problems associated with food processing techniques. 1,2 Heating of food induces chemical reactions that lead to the formation of heat-induced toxic substances, the so-called thermal process contaminants. 3 One such compound that has received much scientific interest over the recent years is acrylamide (AA). 4 Before its discovery in food, AA was known as an industrial chemical compound used primarily as a building block in many industrial processes, such as in the production of plastics, glues, paper, component of cigarette smoke, and in the treatment of drinking water and wastewater, including sewage. 1 Acrylamide was also found in consumer products, such as caulking, food packaging, and some adhesives. 5
Although AA has probably been around as long as people have been baking, roasting, toasting, or frying foods, it was only in April 2002 when the Swedish National Food Administration (SNFA) announced that prolonged heat treatments of some foods could create significant amounts of AA. 6 This discovery by Swedish researchers resulted in the detection of AA in a wide range of foods, particularly starchy foods such as potato and grain products when processed at high temperatures. 7 Acrylamide does not appear in raw foods themselves; however, it is formed during the heating process when the temperature reaches 120°C or higher. 8 Maximum limits for AA in food have not been established, although the World Health Organization (WHO) guideline for AA in drinking water is 0.5 μg/L. 9 The SNFA and Stockholm University researchers reported moderate levels (5-50 μg/kg) and high levels (150-4,000 μg/kg) of AA in heated protein and carbohydrate-rich foods, respectively. 10 Foods that lack AA are those that are boiled or nonthermally treated. 11
Mechanism of AA Formation in Food
A few months following the 2002 announcement by the SNFA, much attention became focused on the Maillard reaction after it was reported that AA formation in starchy foods during heating involves 2 natural components, namely, reducing sugars and the amino acid asparagine. 12 High temperatures and low moisture content in food favor the formation of AA. 13 Maillard reaction is not a single reaction but a complex series of reactions that occur during the thermal processing of food. During this nonenzymatic reaction, reducing sugars (glucose and fructose) condense with amino acids, mainly asparagine, to produce N-glycoside that usually rearranges to the Amadori rearrangement product, which in turn undergoes different steps to produce melanoidin, where a further decarboxylation of the Schiff base leads to AA formation (Figure 1). 14 -16 This reaction is primarily responsible for the brown color, crust, and characteristic tasty flavor of baked, fried, and toasted foods. 17 Research has shown that the reducing sugars are the limiting factors in potatoes, while asparagine is the limiting factor in cereal products. 18 Recent studies have indicated that one other compound called 3-aminopropionamide can also be formed during the Maillard reaction and can be converted to AA under aqueous conditions. 19 This compound has been identified in cocoa beans, coffee, and cereal products. 20 Maillard reaction is primarily a surface reaction, so AA in bread is located mainly in the crust with very low amounts in the crumb. 21 In potato crisps, one possible reason for the high AA content is that the crisp is essentially 2 thin surfaces with very little matter between them. 21 In addition, the darker in color the food product is (burnt toast, darker chips), the higher the AA content. 21
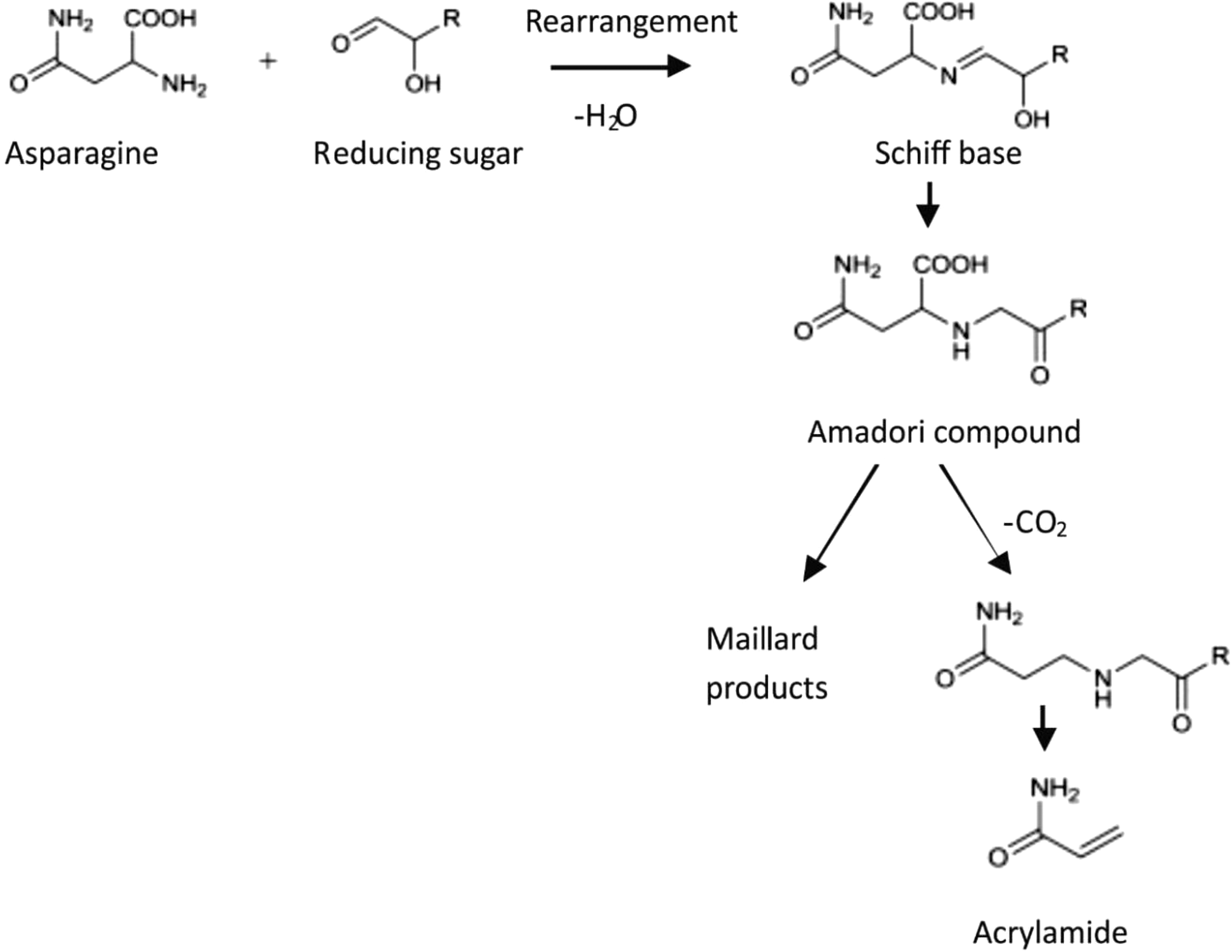
Main pathway of acrylamide formation in food.
In conclusion, free asparagine, free reducing sugar, high temperature (>120°C), and low moisture conditions at the surface of the food are key requirements for AA formation in heat-processed foods. 17
Acrylamide Exposure
Humans can be exposed to AA through oral, dermal, and inhalational routes. 22 Acrylamide is also present in nondietary sources such as tobacco smoke, which is, therefore, another source of exposure for both smokers and nonsmokers (through passive smoking). 23 For smokers, it was found that tobacco smoking is a more prominent source of AA exposure than food. 24 In addition, due to its wide variety of other nonfood industrial uses, many people can be exposed to AA in the workplace through dermal absorption or inhalation. 25 Therefore, AA exposure is a combination of exposures from different sources such as diet, smoking, drinking water, and occupational sources.
Dietary Exposure
The presence of AA in heat-processed foods is a worldwide health concern, since this substance has been classified as a probable human carcinogen by the International Agency for Research on Cancer (IARC). 26 Acrylamide is primarily formed in food products derived from raw materials that are rich in carbohydrates and low in proteins. 10,27 Fried, deep-fried, or baked food items, such as cake, bread, French fries, and chips are believed to contain the highest levels of AA as shown in Table 1. 13 Despite the fact that AA concentration in coffee is relatively low, it is a major contributor to AA exposure in adults because of the high amounts of coffee consumed. 29
Acrylamide Levels in Selected Food Groups.a
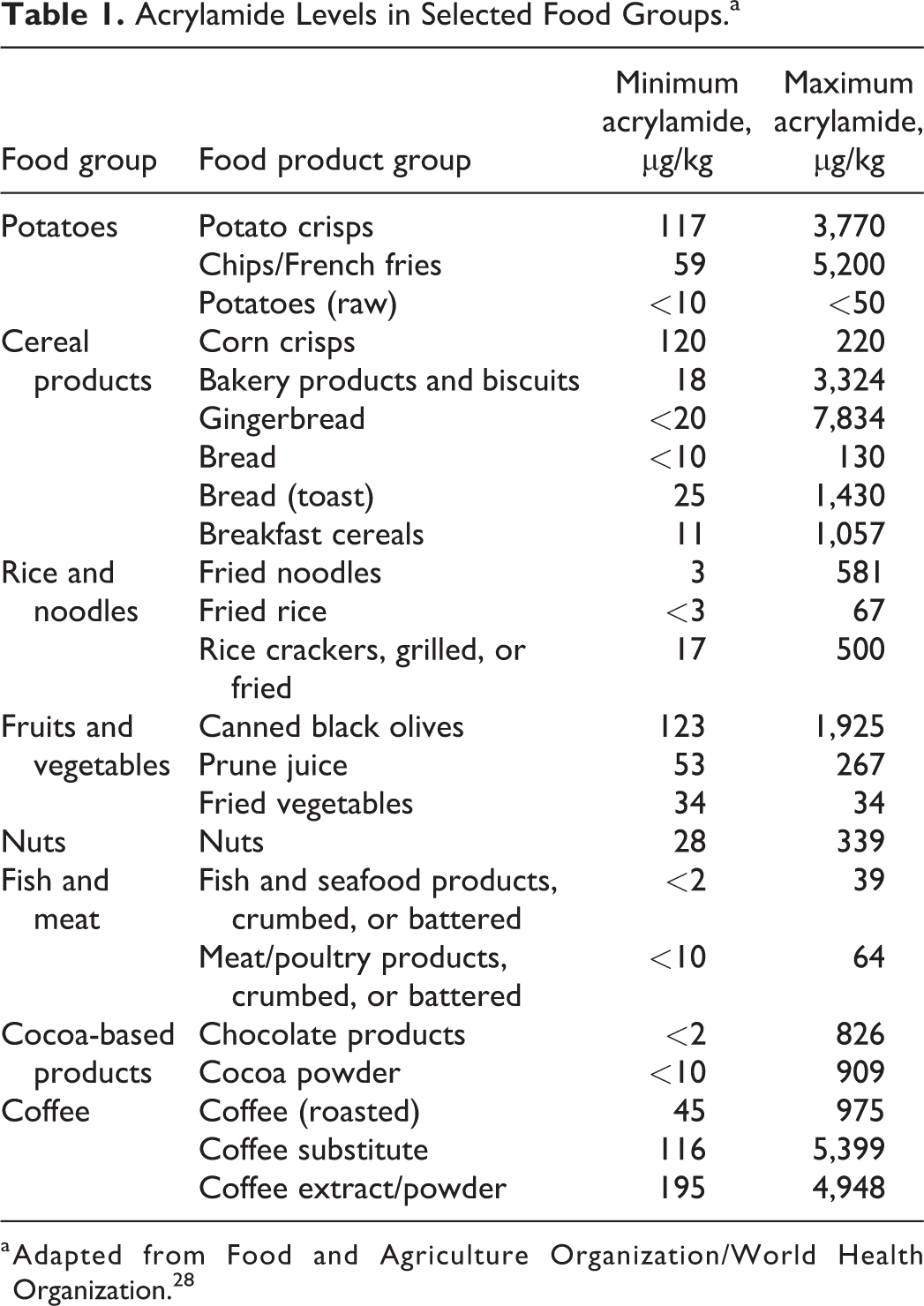
a Adapted from Food and Agriculture Organization/World Health Organization. 28
Estimates of the average intake of AA by consumers may differ between countries and according to dietary habits, but an average mean intake can be considered to be about 0.4 μg/kg body weight per day (bw/d), and the average intake for a high-level consumer to be about 1.0 μg/kg bw/d. 30 Other researchers estimated the acceptable daily intake to be 1 µg/AA/d, which is an amount exceeded in many regular food products. 31 The WHO states that AA has no reliably identifiable threshold of effects, meaning that exposure to low doses might be followed by a symptom silent period in which the detrimental effects of the chemical may not be clinically apparent, but nevertheless morphological and/or biochemical alterations may be present. 32 Tolerable daily intake for neurotoxicity from AA was estimated to be 40 mg/kg/d while that for cancer was estimated to be 2.6 and 16 mg/kg/d based on AA or glycidamide, respectively. 33
In a study done in Lebanon, the daily consumption of AA from potato and corn chips was found to be 7- to 40-fold higher than the risk intake set by WHO but was below the neurotoxic risk threshold. The cancer risk for the Lebanese population from AA exposure estimations appears to be significant, highlighting the need to conduct further epidemiological studies and to ensure monitoring of AA levels in food products. 34 Another Lebanese study on the amount of AA in caffeinated beverages showed that caffeinated beverages contributed an average of 29,176 μg/kg of AA, which was higher than the risk intake for carcinogenicity and neurotoxicity set by the WHO. 35 This study shows alarming results that call for the need to regulate the caffeinated product industry in Lebanon by setting legislations and standard protocols for product preparation in order to limit the AA content and protect the consumers.
The Joint Expert Committee on Food Additives had reported that the major foods contributing to the total AA intake for most countries are potato crisps (6%-46%), potato chips (16%-30%), coffee (13%-39%), pastry and sweet biscuits (10%-20%), and bread (10%-30%). 36 Furthermore, food packages that contain polyacrylamide may lead to indirect exposure to AA monomer residual. 37 Although nonfood exposures may exist, the diet is assumed to be the major source of AA exposure for the general nonsmoking population, where around 38% of caloric uptake is provided by food sources that are known to contain AA. 38
Acrylamide Metabolism
After consumption, it is demonstrated that AA is rapidly and completely absorbed by the gastrointestinal tract in rats via the circulation and is distributed to the peripheral tissues. 39 The fate of AA in humans seems to be qualitatively similar to that in rodents. 39 One exploratory study in healthy volunteers have confirmed that AA can cross the blood–placenta barrier in a human placenta in vitro model as well as the blood–breast milk barrier in vivo in lactating mothers. 40 These studies may suggest that AA is able to reach any human tissue. Once absorbed, AA is metabolized via at least 2 main pathways. It may be conjugated to N-acetyl-S-(3-amino-3-oxopropyl) cysteine by glutathione-S-transferase (GST), or it may be converted to glycidamide in a reaction catalyzed by the cytochrome P450 enzyme complex (CYP450), where this metabolite is known to be more reactive toward DNA and proteins than the parent AA compound (Figure 2). 41,42
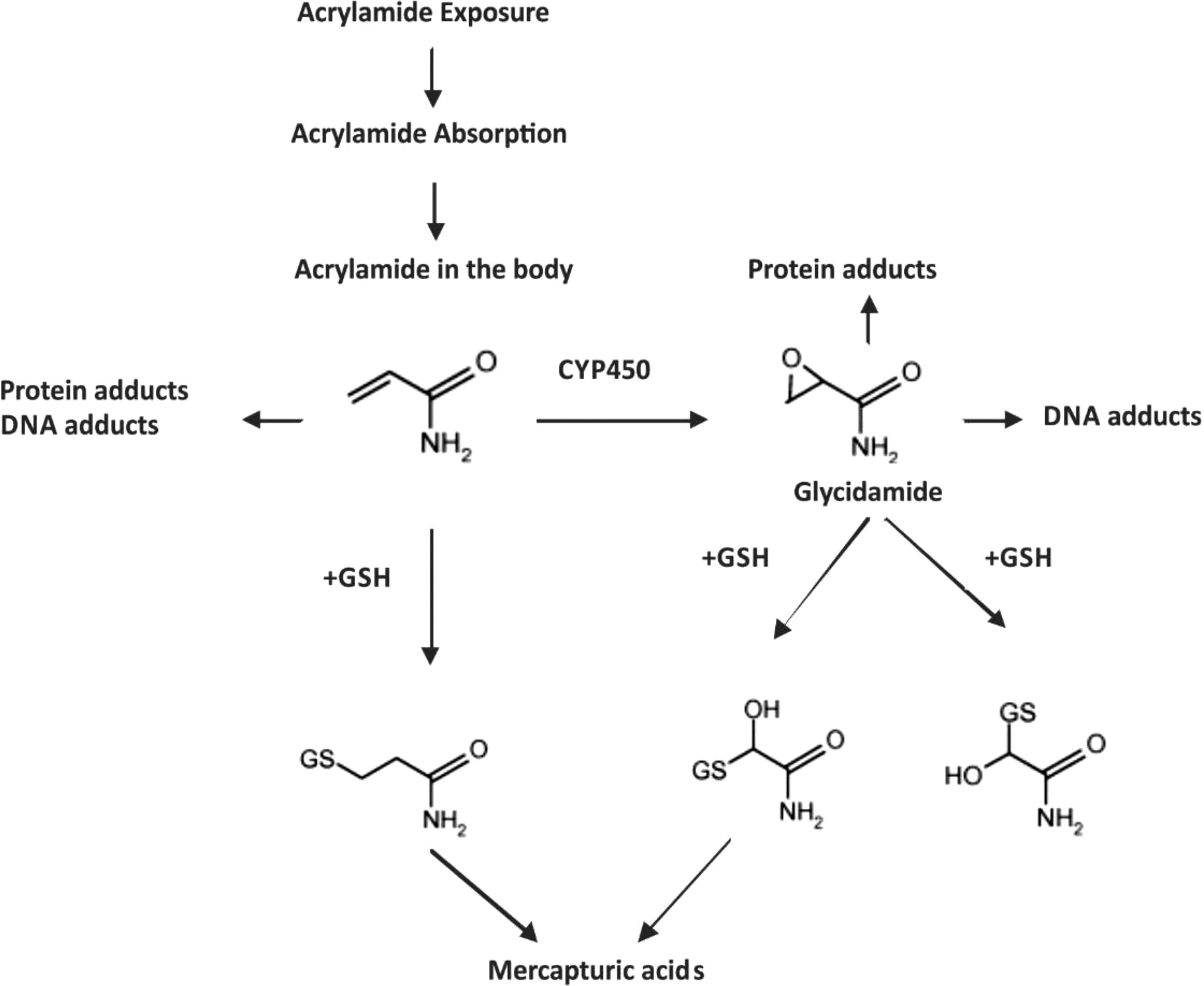
Proposed metabolic scheme of acrylamide.
Detoxification of both AA and glycidamide can proceed through conjugation with glutathione (GSH), mediated by GST and the GSH adducts thereafter are excreted in urine as by-products of mercapturic acids. 43 The mercapturic acids of AA and glycidamide represent the major metabolites, and their urinary excretion levels are proposed to be biomarkers of AA exposure. 43 Additionally, AA and glycidamide can also form adducts with DNA and amino acids in hemoglobin, and therefore, these adducts represent important biomarkers of AA exposure. 44 The importance of AA as a food contaminant was shown in 2002 when it was observed that feeding rats with fried feed showed a large increase in the level of a hemoglobin adduct. 1 Moreover, AA was found to be able to cross the placental barrier where studies showed the presence of AA-hemoglobin adducts in the neonatal blood. 45
Toxicity of AA
Neurotoxicity
Neurotoxicity is a major consequence of AA exposure, and considerable attention has been drawn to this area of investigation. This compound is considered to be a cumulative neurotoxicant in rodents as well as in humans. 46 In rodent toxicity studies, exposure to repeated doses of 10 to 50 mg/kg bw/d AA had been reported to cause neuropathy in most laboratory animal species, while exposure to single doses of 100 to 200 mg/kg was fatal in most animals. 47 In vitro, AA was shown to induce apoptosis in rat primary astrocytes and cause mitochondrial dysfunction and apoptosis in BV-2 microglial cells. 48 Moreover, Chen and Chou showed that AA disrupted the nervous system by inhibiting human neuroblastoma and glioblastoma cellular differentiation. 49
Acrylamide neurotoxicity in occupationally exposed populations has been ascertained by various epidemiological studies. 50 General symptoms of neurotoxicity in humans are a characteristic ataxia, skeletal muscle weakness, weight loss, distal swelling, and degeneration of axons in the central and peripheral nervous systems. 51,52 A case report from Sweden described peripheral neuropathy in tunnel workers exposed to short-term but intensive doses of a grouting agent containing AA and N-methylolacrylamide. 52 There was a significant dose–response association between peripheral nervous symptoms and hemoglobin adducts that were used as biomarkers. 52
Genotoxicity and Carcinogenicity
The genotoxicity of AA and its major metabolite glycidamide had been investigated in several studies. A study by Alzahrani in mice showed that single doses of AA at 10, 20, and 30 mg/kg and repeated doses of 10 mg/kg for 1 and 2 weeks significantly induced DNA damage compared to the control group as shown by elevation in micronuclei and chromosome aberrations in mice bone marrow cells. 53
Moreover, prolonged exposure of animals to high concentrations of AA in the drinking water leads to tumor development at multiple sites in both male and female genders. 54 Although there is sufficient evidence for the carcinogenicity of AA in experimental animals, the few epidemiologic studies conducted to date on occupational and dietary exposure to AA have found no consistent evidence of its carcinogenic effects in humans. 55,56 Based on the current research, AA is currently classified as a “probable human carcinogen” by the IARC and as “reasonably anticipated to be a human carcinogen” by the US National Toxicology Program.
Two different cohort studies were done on factory workers being exposed to high levels of AA for many years, but no statistically significant cancer mortality was reported. 57,58 However, other researchers have found some association between AA-hemoglobin adduct levels and incidence of estrogen receptor-positive breast cancer as well as between AA intake and endometrial and ovarian tumors in postmenopausal women. 59,60 Herein, it is important to note the need for further research on this topic to provide information about AA exposure and cancer risk in humans.
Reproductive Toxicity
Reproductive toxicity of AA in humans has not been demonstrated; however, in rats, the No-observed adverse effect level for reproductive toxicity was assessed to be 2 to 5 µg/kg/d. 54 The administration of 0.5 to 10 mg/kg of AA caused growth retardation in rats and reduction in epididymal sperm reserves compared to the control group. 54 In addition, repeated injections of AA (20 mg/kg) to male rats for 20 days caused decrease in testosterone and prolactin concentrations in a dose-dependent manner. 61 In another study, reproductive toxicity was also revealed in AA-treated female mice, where a decline in the viability of mouse granulosa cells, the number of corpora lutea, and progesterone production was observed. 62
Hepatotoxicity
Although AA is metabolized in the liver, reports of its hepatotoxicity in humans are still scarce. However, numerous studies in animals have reported the harmful effects of dietary AA in the liver due to oxidative stress. A high dose of 25 mg/kg AA administered for 21 days resulted in significant decrease in liver GSH level and total antioxidant status in experimental adult rats. Administration of AA also led to increase in serum level of liver enzymes (AST, ALT, and ALK) and decrease in superoxide dismutase and catalase activities, while total oxidant status and malondialdehyde levels increased. 63
Immunotoxicity
Studies regarding the adverse effects of AA on the immune system are limited compared to other end points. 54 Nevertheless, immunotoxicity of AA was found in female BALB/c mice, where AA decreased final bw, spleen, and thymus weights, and lymphocyte counts in addition to causing pathological changes in lymph glands, thymus, and spleen. 64 Acrylamide was also shown to cross the placenta and reach the fetus, but no significant associations were found between prenatal dietary exposure to AA and the investigated immune-related health outcomes or blood parameters at any age. 65
Mechanism of AA Detoxification
Once ingested, AA can be detoxified in the body if it is processed through CYP450 and converted into glycidamide or if it is bound to the sulfur-containing antioxidant glutathione as shown in Figure 2. 66 However, despite the metabolic pathways assisting in AA detoxification, it is still possible to overload the detoxification capability of these pathways and create health risks from excess exposure. 67 One way to possibly help lower the risk of toxicities from AA is to increase glutathione levels by consuming sulfur-containing foods such as onions, garlic, and cruciferous vegetables such as broccoli and brussels sprouts or foods that contain significant amounts of cysteine, which is an essential substrate for the synthesis of glutathione, such as onions, garlic, cruciferous vegetables, and red peppers. 67,68 Foods such as poultry, yogurt, and eggs also contain significant amounts of the amino acid cysteine. 67
Mitigation Strategies for AA
Since 2002, scientists from all over the world in collaboration with agencies such as FAO and WHO have been working on different ways to reduce the levels of AA in foods commonly consumed with the highest levels of AA such as potato and cereal products. 69,70 Although coffee is also a substantial contributor to the total dietary intake of AA, which is formed during coffee roasting, currently there are no feasible strategies to decrease AA levels in coffee. According to the Codex Code of Practice to reduce AA in foods, it is quoted “no commercial measures for reducing acrylamide in coffee are currently available.” 38 Nevertheless, work is presently underway to identify new strategies for AA mitigation without significantly impacting the significant organoleptic properties and acceptability of coffee. 71,72 Thus, this review discusses strategies that have been conducted for reduction of AA in foods such as potato and cereal products that were categorized into 4 different groups based on the effect of raw materials, additives, processing conditions as well as the effect of pH, water activity, and fermentation.
Effect of Raw Materials
The formation of AA in foods such as potato and cereal products has been widely studied. The amount of AA formed varies greatly in the same food items due to variable food composition (ie, nutrient contents) which is affected by several factors such as the climate and storage conditions, fertilization, and manufacturing. 73 Reducing sugars and asparagine content of potato and cereal products before thermal processing play significant roles in the formation of AA during later processing stages. 74 For example, since the amount of reducing sugars in potatoes is much higher than that of asparagine, controlling their level in the initial raw material can decrease AA formation in the finished fried potato products. Therefore, selecting potato varieties with low content of reducing sugars may help reduce AA, while maintaining the desirable product qualities. For instance, potatoes with less than 1 g/kg fresh weight of reducing sugars should be used for frying or roasting. 75 Additionally, lower levels of AA were detected in French fries made from the genetically modified potatoes (innate potatoes) than in fries made from conventional potatoes. 76 Innate potatoes were produced by silencing the asparagine synthetase-1 gene (Asn1) in the tuber resulting in lower levels of asparagine, which in turn decreases the formation of AA by 52% to 78% when fried or baked at high temperatures. 77
The reducing sugar content in potato tubers tend to decrease over the course of the growing period to reach a minimum level toward the end of the growing season which is a good indicator to harvest at the right time to reduce the potential for high AA formation during processing. 78 Acrylamide content is also affected by climatic conditions, where warm weather conditions (above 25°C-30°C) and cold climates (below 8°C-12°C) tend to increase sugar content of potato tubers and subsequently increase AA formation upon frying. 79 Therefore, it is proposed that the optimum temperature for tuber growth ranges between 15°C and 20°C. 79
Formation of AA is also related to the storage temperature of potato tubers. Storing potato tubers at 8°C or lower will lead to phenomenon called “low-temperature sweetening” which causes an increase in reducing sugar content and enhancement of the brown pigment during frying and hence higher amounts of AA. 80 For heat processing, potato tubers should be stored at 8°C to 12°C to avoid this increase in reducing sugar content. 81
Effect of Additives
Plant antioxidants
The effect of antioxidants and their extracts on the level of AA in foods has not been investigated satisfactorily. Both positive and negative results had been obtained when food researchers tried to use natural antioxidative extracts to inhibit AA formation in food. Some antioxidants were claimed to reduce AA formation, while others did not show any effect or showed an enhancing effect. 82,83
Bioactive products extracted from plants have been shown to reverse AA toxicity. For example, when rosemary extract, oil, and dried leaves were added to wheat dough, AA was reduced by 62%, 67%, and 57%, respectively, compared to wheat buns without addition of rosemary. 82 On the other hand, grape seed extract added to baked products did not have any effect on AA formation. 83 Similarly, other studies found that the addition of sesamol, vitamin E, and antioxidants such as 2,6-bis (1.1-dimethylethyl)-4-methylphenol to meat before heating led to increased AA formation. 84 In a model system based on wheat flour and water, which resembled crackers, the use of ascorbic acid and ascorbate showed a slight reduction in AA content. 85 Furthermore, the addition of bamboo leaves and green tea extracts significantly reduced AA formation in an asparagine–glucose model system. 86
In another study, chrysin, which is a natural biologically active flavonoid compound found in many plants, reduced AA-induced neurotoxicity in Wistar rats due to its high antioxidant power. 87 Additionally, quercetin, which is a polyphenolic flavonoid compound, contains a spectrum of antioxidants that are able to protect against AA-induced neurotoxicity. 88 Berry juices (bilberry, black mulberry, and raspberry) significantly restored the growth of AA-exposed yeast cells, Saccharomyces cerevisiae, and decreased the level of reactive oxygen species. 89
Enzymes
The use of asparaginase enzyme is an effective strategy at reducing AA formation in fried potatoes, as it catalyzes the hydrolysis of asparagine to aspartic acid and ammonia without affecting the final product aspects. 90 This approach was found to be more significant when the raw material was blanched prior to enzyme application. 91 A study has demonstrated that soaking of blanched potato strips in an asparaginase solution at 40°C for 20 minutes reduced AA by 60% when compared with blanched strips without the enzyme treatment. 91
Amino acids
Inhibition of Maillard reaction, thus reducing AA formation in foods, could occur by using competitive compounds that are able to compete with asparagine for carbonyl groups. In fact, the addition of amino acids before heat processing of foods have been proposed as a possible strategy to reduce AA formation by competing with asparagine in the Maillard reaction or reacting with AA after its formation. 92 In one study, the formation of AA was reduced by more than 80% in potato slices soaked in 3% solution of either lysine or glycine prior to frying. 93
Vitamins
The formation of AA was reduced by more than 60% after the addition of approximately 1% of vitamin C and vitamin B1, whereas only 20% to 30% reduction was found after the addition of 1% vitamin B2 and vitamin B5 in an amino acid/sugar chemical model system. 94 Up to 75% reduction in AA formation in french fries was found by dipping potato cuts in 2% citric acid solutions for 1 hour before frying; however, there might be concerns about sour flavors in the resulting products. 95
Salt solution
Some mono- and divalent cations (eg, Na+ or Ca2+) were reported to effectively mitigate AA formation in food. 96 For example, potatoes dipped in CaCl2 solution showed 95% reduction in the amount of AA formed in fried potatoes without negatively affecting the sensory characteristics of the strips. 96 These ions interact with asparagine so that the formation of the Schiff base is inhibited, and thus AA generation during heating is reduced. 96
Effect of Processing Conditions
Frying Time and Temperature
Generally, frying temperature and time have been shown to significantly affect the amount of AA formed and are considered to be the most critical factors affecting its content in fried potato products. 97 Higher temperatures and longer duration of thermal processing are associated with higher AA content. 97
Soaking
A simple measure of presoaking potatoes before frying can reduce the formation of AA. 98 Washing raw french fries and soaking them in water reduced the formation of AA if they were fried to a lighter color. 98 This is because presoaking causes the glucose content in the potato strips to be reduced with increased soaking time. 98 Therefore, water soaking results in lower AA content due to the leaching of one important AA precursor such as glucose.
Blanching
Blanching is also performed in water to reduce the level of reducing sugars in raw potatoes, which could result in higher AA content. 99 Blanching of potato strips with sunflower oil for 43 seconds at 150°C had a greater reduction effect on the level of AA precursors (asparagine and reducing sugars) and thus on the final AA level, relative to soaking in water. 100 Moreover, blanching in hot or warm water was reported to reduce the amount of AA in french fries. 101 As the blanching temperature and duration increased, more glucose and asparagine are being leached out leading to french fries with lower AA levels. 101
Effect of pH, Water Activity, and Fermentation
Acids
It is widely established that pH levels influence the formation of AA. Lowering the pH of the soaking solution has been shown to stop the formation of the Schiff base (the nucleophilic amine group (NH2) is converted to the non-nucleophilic protonated-NH3+) that leads to AA formation. 102
Water Content
The total amount of water present in foods greatly influences AA content in food. It has been found that AA is formed in foods with water activity (aw) between 0.4 and 0.8. When the water activity is <0.4, the formation of AA is decreased. Water activity and moisture content are 2parameters that are linked together, so foods with moisture content <5% are more likely to follow the Maillard reaction and form AA. 97
Fermentation
Formation of AA in food was also found to be affected by fermentation. 103 For instance, lactic acid fermentation was reported to be suitable in reducing AA formation in potato products, especially when combined with blanching. 104
Conclusion
The report made by the SNFA in April 2002 about the presence of high levels of AA in carbohydrate-rich foods processed at high temperature (>120°C) evoked a worldwide health alarm. This announcement sparked intensive investigations into AA, its occurrence in foods, and its adverse health effects. At present, AA is considered a food-borne toxicant by many international organizations such as the Food and Drug Administration and WHO. Many studies described the potential health risks of AA; however, there is paucity of information on strategies to reduce the levels of AA in processed food. This review did not only present the toxicity of AA including neurotoxicity, mutagenicity, and carcinogenicity but also focused on various ways used in mitigating AA levels in food such as soaking, blanching, fermentation, enzymes, or antioxidant addition.
Footnotes
Author Contributions
Rifai, L. and Saleh, F. contributed to conception and design, drafted manuscript, critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
