Abstract
This is a safety assessment of Avena sativa (oat)–derived ingredients. The reported functions of these ingredients in cosmetics include abrasives, antioxidant, skin conditioning agents, absorbents, and bulking agents. The Panel reviewed relevant animal and human data related to these ingredients. Because final product formulations may contain multiple botanicals, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may lead to sensitization or other toxic effects. The Panel stated that industry should continue to use good manufacturing practices to limit impurities and concluded that all but one of the Avena sativa (oat)–derived ingredients are safe as cosmetic ingredients in the practices of use and concentration described in this safety assessment when formulated to be nonsensitizing; data are insufficient to come to a conclusion of safety for Avena Sativa (Oat) Meristem Cell Extract.
Introduction
This is a review of the available scientific literature and unpublished data provided by industry relevant for assessing the safety of Avena sativa (oat)–derived ingredients as used in cosmetics. The functions of these ingredients in cosmetics include: abrasives, antioxidant, skin conditioning agents, absorbents, and bulking agents (Table 1). The 21 ingredients included in this report are:

Definition and Function of Avena sativa–Derived Ingredients. 1
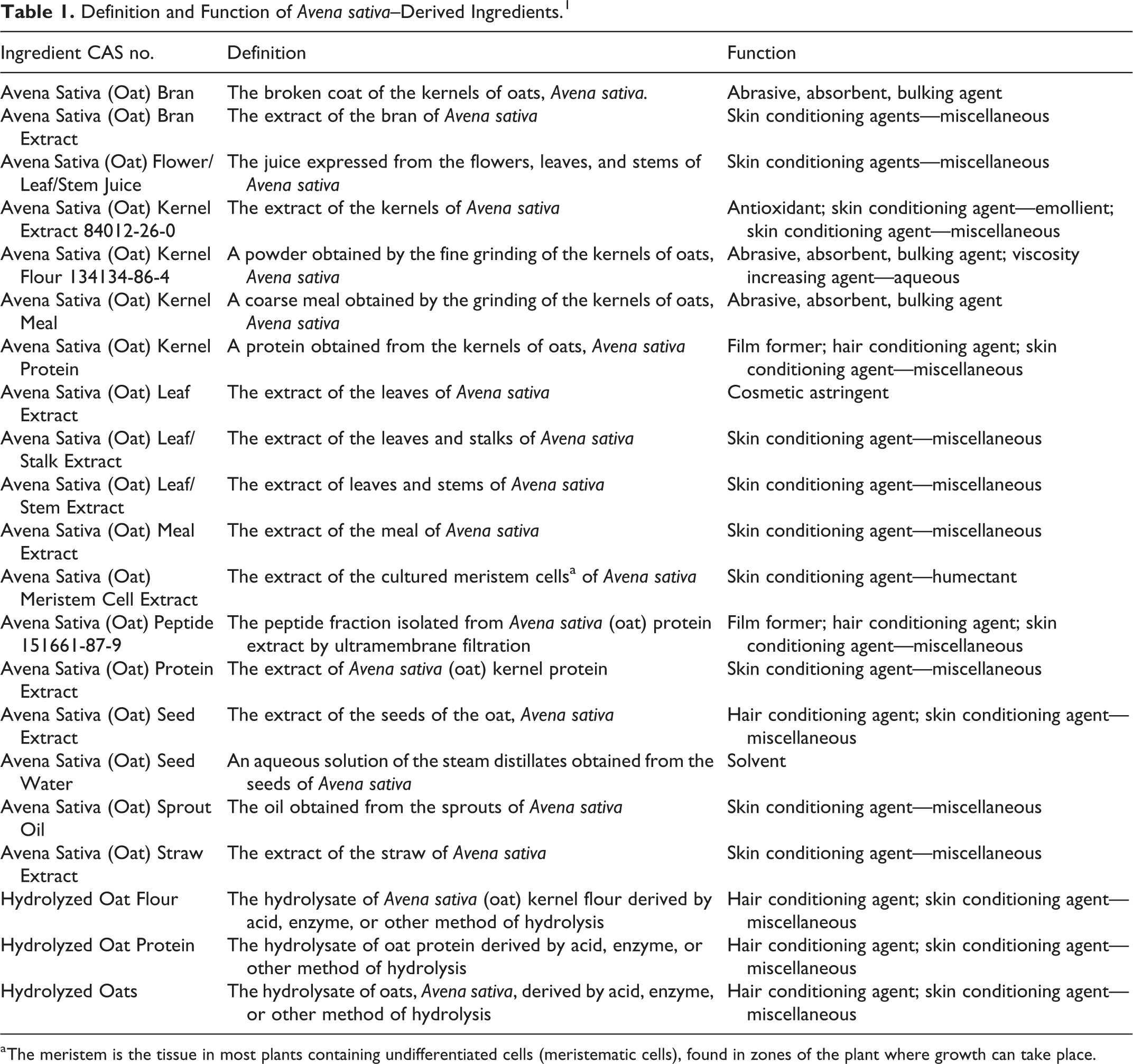
a The meristem is the tissue in most plants containing undifferentiated cells (meristematic cells), found in zones of the plant where growth can take place.
The International Cosmetic Dictionary and Handbook (Dictionary) defines colloidal oatmeal as finely ground oatmeal; the definition does not specify the species of oat from which it is derived. 1 Therefore, any oat species (ie, Avena abyssinica, Avena byzantina, Avena nuda, and Avena strigosa) may be used to manufacture this cosmetic ingredient. However, some information on colloidal oatmeal does specify the source species. Therefore, when the colloidal oatmeal is derived from A sativa, the data are included in this report for read-across.
The US Pharmacopeia (USP) Convention defines colloidal oatmeal as derived from only A sativa or A byzantina; the USP definition does not include A nuda or A strigosa. The USP indicates that oats used to make colloidal oatmeal must meet US standards for No. 1 or 2 grade oats (ie, 97% or 94% undamaged oats, respectively) and may contain, singly or in combination, not more than 25% wild oats and other grains for which standards have been established under the US Grain Standards Act. 2 [7CFR810.1001]
Even though “A sativa” is not included in the names of the Hydrolyzed Oat Flour or Hydrolyzed Oats, the Dictionary 1 does specify that these ingredients are derived from the A sativa plant, and therefore, these ingredients are appropriate for inclusion in this report.
Avena sativa (Oat) Kernel Oil was reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) in 2011 and the Panel concluded that it was safe as used in cosmetics. 3 Because some of these ingredients are hydrolyzed proteins, the Panel noted previously reviewed safety assessments of α-amino acids, animal- and plant-derived amino acids, hydrolyzed collagen, hydrolyzed corn protein, and triticum vulgare (wheat) gluten, and wherein the conclusions were that these ingredients are safe as used in cosmetic products. 4 -10 Avena sativa (oat) starch was concurrently reviewed as part of a safety assessment of polysaccharides. 11
Oats are included in the list of food grains and feed grains established under the United States Grain Standards Act [7CFR810.101]. Avena sativa grains are used extensively in both animal feed and human food and the other plant parts are used in animal feed, resulting in much larger oral exposures than would result from cosmetic uses. Therefore, the systemic toxicity potential of these cosmetic ingredients is mitigated.
Chemistry
Definition and Description
The definitions and functions of A sativa (oat)–derived ingredients are provided in Table 1. Avena sativa is a member of the Gramineae (grass) family. 12 The plant is an annual grass that grows up to 1.5 m tall. The stems are smooth and may be tufted or solitary and erect or bent at the base. The leaves are nonauriculate and green, with the sheaths rounded on the back. The cluster of flowers is a diffuse panicle with 2 to 3 florets, which can be either all bisexual or mostly bisexual, with the distal 1 or 2 flowers reduced in size and either male or sterile. The grain is tightly enclosed in the hard lemma and palea. The seed size varies with cultivar (plant strain) and commonly yields approximately 30,000 seeds per kilogram of harvested plants.
Physical and Chemical Properties
The solid components of an alcohol extract of ground and macerated A sativa seeds were reported to have a relative molecular mass of 1,000 to 10,000 Da, as characterized by ultrafiltration. 13 The average molecular weight of small peptides for a batch of Hydrolyzed Oats was reported to be 1,365 Da. 14 The average molecular weight for Hydrolyzed Oats was reported to be approximately 1,000 Da. 14,15
The high concentration of starch and β-glucan in colloidal oatmeal has a water-holding function; phenols (constituents of these botanical ingredients) reportedly have antioxidant and anti-inflammatory activity and act as ultraviolet absorbers. 16 The cleansing activity of oat is from the saponins.
Some of the flavonoid constituents with phenolic structures strongly absorb long-wavelength ultraviolet radiation (UVA) in the 320 to 370 nm range. 17
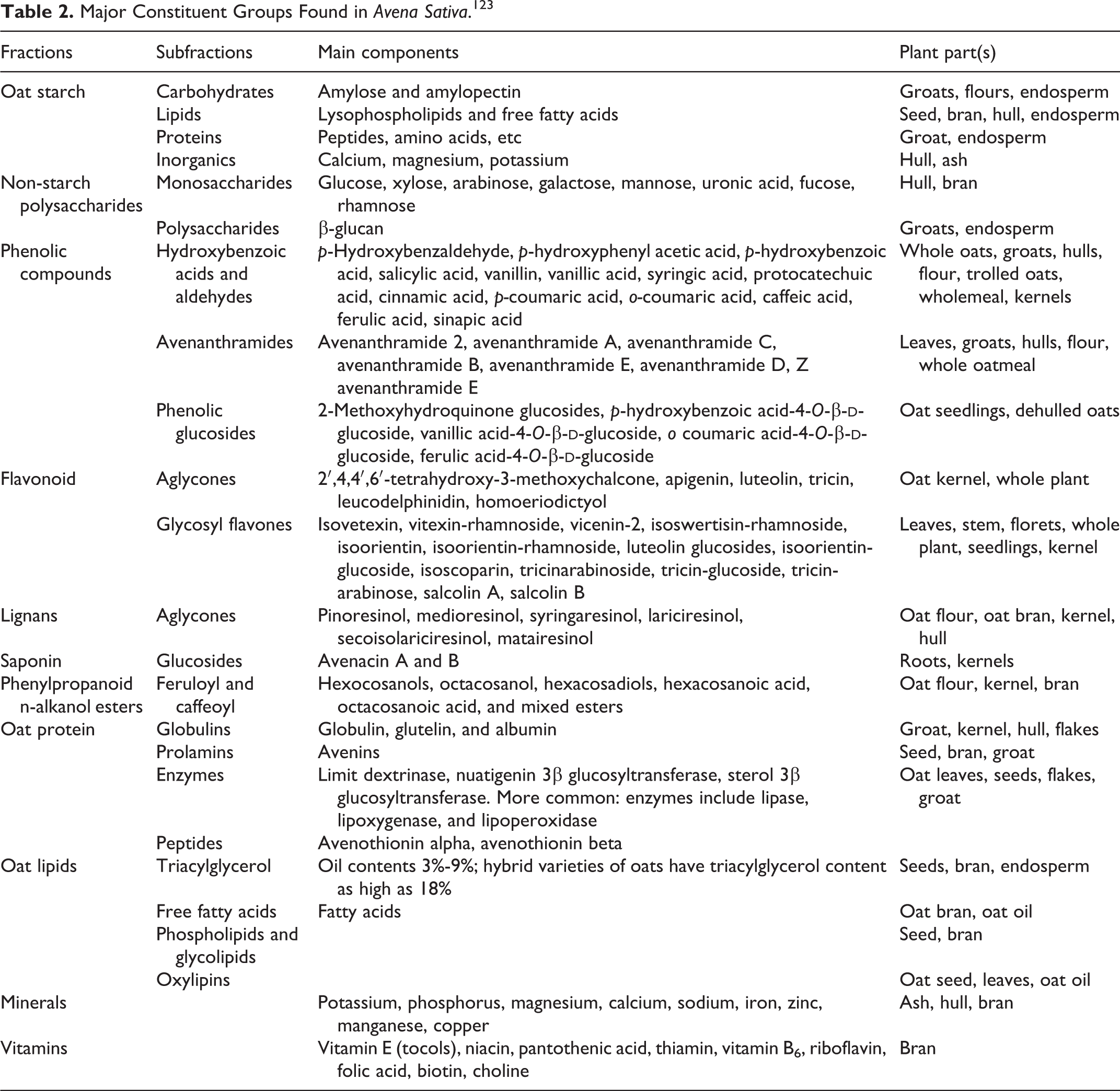
Constituents of A sativa
As in all plants, there are a number of constituents that make up A sativa grains and other plant parts. Table 2 presents an overview of the constituent groups and subgroups. The constituent groups include:
Amino acids: Oats are rich in the amino acid lysine, approximately 4%.
18
Other amino acids, including
Avenacins and avenacosides: These are saponins. Avenacosides are biologically inactive until they are converted to antifungal monodesmosidic saponins (26-desglucoavenacosides A and B) in response to tissue damage.
19
The stem and leaves contain bidesmosidic steroidal saponins (eg, avenacosides A and B); triterpenoid saponins and avenacin have been also reported in the root.
19
-23
Enzymes: There are multiple enzymes found in A sativa (eg, superoxide dismutase).
24,25
Carbohydrates: Mucilage (β-glucan), 3% to 4% sugar (glucose, fructose), β-glucan, pentosans, saccharose, kestose, neokestose, bifurcose, neobifurcose, and acid galactoarabinoxylan have been reported.
21
Starch is the most abundant component of the oat grain, which is approximately 25% to 30% amylose.
21,26
Polysaccharide carbohydrates include starches and β-glucan.
27,28
Carbohydrates mostly consist of araban and xylan gums.
29
Flavonoids: The following flavonoids have been isolated from Avena Sativa Bran: kaempferol 3-O-(2”,3”-di-E-p-coumaroyl)-α-
Lipids: Avena sativa contains higher levels of lipids, particularly those containing a high content of unsaturated fatty acids, than other cereal-type grains. The most abundant lipids are unsaturated triglycerides.
33,34
The lipid content depends on genetic and environmental factors. The methods of extraction and analysis result in differences in the lipid content of the extracts. Various lipids, like stearyl esters, partial glycerides, free fatty acids, glycolipids, and phospholipids, were identified in oats.
21,35
Avena sativa starches contain lipids ranging from 1% to 3%, present in the starch possibly as amylose–lipid complexes.
34
Phenolic compounds: At various growth stages, A sativa has been found to contain a large number of phenolic compounds, including all major classes, in addition to avenanthramides: benzoic and cinnamic acids, quinones, flavones, flavonols, chalcones, flavanones, anthocyanidines, and aminophenolics.
17
Avena sativa oat flour contains the glyceryl esters of hydroxycinnamic, ferulic, p-coumaric, and caffeic acids.
36
Antioxidant activity is attributed to the presence of phenolic esters.
17,37
Avena sativa also contains various compounds with antioxidant activity, which serves to help protect the lipids from oxidation.
17
Avenanthramides are soluble, phenolic compounds that are minor components of A sativa (0.03% by weight).
38
-40
They have powerful antioxidative activity. They also have anti-inflammatory properties.
41
The stem and leaves contain phenolic compounds.
32,42,43
Major Constituent Groups Found in Avena Sativa. 123
The total phenol content of the n-hexane extract of an A sativa whole plant extract was 26.10 ± 2.31 mg/g, 75.79 ± 4.02 mg/g in an ethyl acetate extract, 39.34 ± 0.78 mg/g in an ethanol extract, and 46.02 ± 0.07 mg/g in a water extract.
31
Proteins: Avena sativa has a high level of total protein compared to other grasses.
44,45
The primary storage protein is globulin.
44
The proteins in the stem and leaves include membrane proteins and soluble proteins of chloroplasts.
19
Sterols: Sterols, sterylglycosides, acylated sterylglycosides, and steroidal saponins are present in oat leaves. The sterol moieties consisted mainly of sitosterol, stigmasterol, cholesterol, cholestenol, Δ5-avenasterol, Δ7-avenasterol, campesterol, campeslenol, lophenol, stigmastenol, Δ7-stigmastenol, and Δ7-cholestenol.
21,46
Vitamins and minerals: Avena sativa contains a variety of minerals and vitamins.
45
These include vitamin E, mostly as α-tocopherol, which is a major antioxidant component in crude oat lipids. β- and γ-tocopherol are present in minor amounts.
25
Constituents of concern
Quercetin: Quercetin has been reported to be in the hay of A sativa at 310 ppm. 47 This constituent was positive for genotoxicity in an Ames assay. 48 It was also consistently positive in in vitro tests of genotoxicity, and in some in vivo studies via intraperitoneal (IP) injections in mice and rats, but was consistently negative in oral exposure genotoxicity tests using mice and rats. 49
Characterization of Avena sativa–derived ingredients
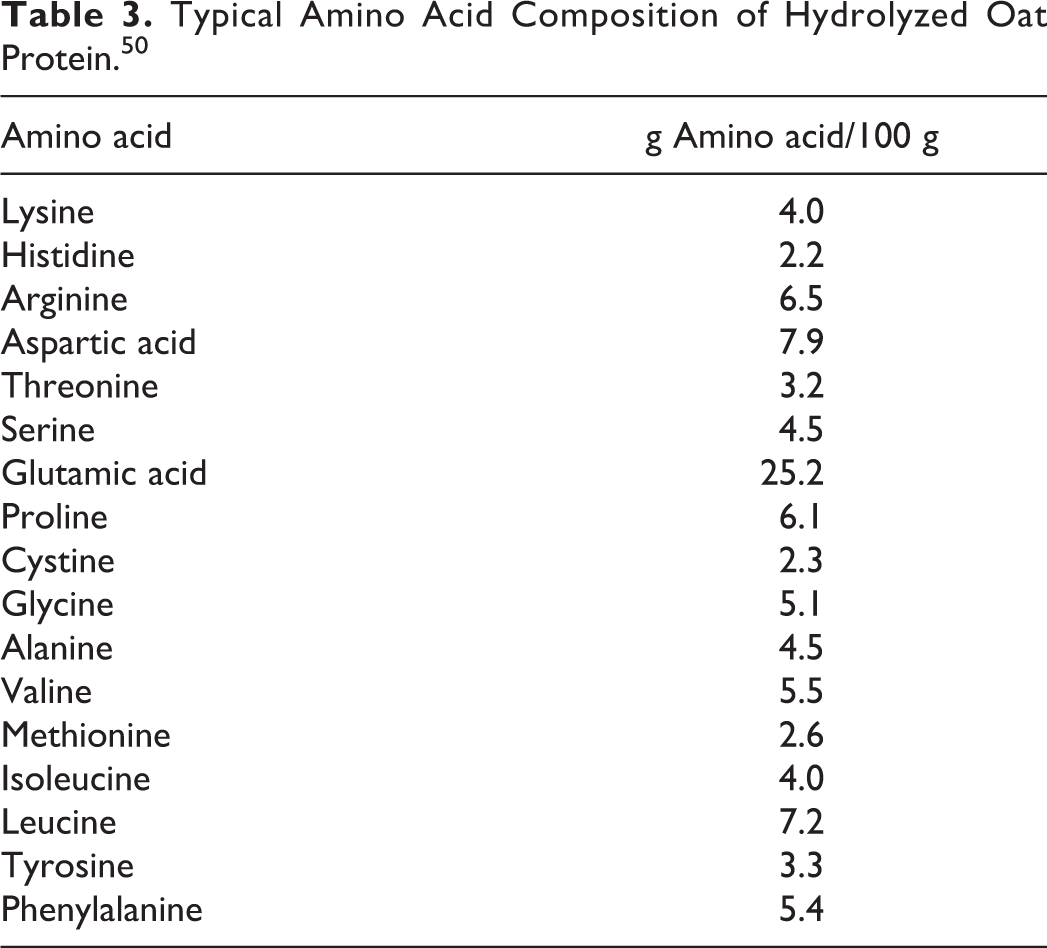
Avena sativa: The molecular weight (MW) of the peptides in Hydrolyzed Oat protein were reported to be 2,000 to 4,000. 50 Hydrolyzed oat protein contained 25.2% glutamic acid in a characterization by a supplier (Table 3).
Typical Amino Acid Composition of Hydrolyzed Oat Protein. 50
The composition of an Avena Sativa (Oat) Leaf/Stem Extract was reported to be sugars, minimum 60%; flavonoids, 7% to 10%; saponins, 1%. 51
The composition of Avena Sativa (Oat) Sprout Oil (100%) was reported to contain glycerides of fatty acid residues consisting of 43% linoleic acid, 37% oleic acid, and 14% palmitic acid. 51 This is similar to Avena Sativa (Oat) Kernel Oil (linoleic acid, 22.8%-43.1%; oleic acid, 31.4%-51.26%; palmitic acid, 13.9%-18.82%). 52
Method of Manufacture
Many solvents are used singly, serially, or in combination to make Avena Sativa (Oat) Kernel Extract, including ethanol, water, and glycerin. Avena Sativa (Oat) Kernel Extract can be manufactured by extracting the milled oat kernels with ethyl alcohol and water. 53 The ethyl alcohol is distilled off and the remaining extract is formulated in glycerin and water with potassium sorbate.
A supplier reported that the manufacturing process of Avena Sativa (Oat) Kernel Extract entails the maceration of oat kernels, glycerin, and water for several days followed by draining and pressing. 54 The product is sterilized and packaged. Samples are sent for final analysis before being released for use.
Another supplier reports that the manufacturing process of Avena Sativa (Oat) Kernel Extract entails the solubilizing of powdered Avena sativa kernels in water followed by enzymatic hydrolysis. 55 The product is heated, then filtered. Proteins are extracted by adsorption on an adjuvant. The soluble phase is concentrated, filtered, and sterilized.
The manufacturing process of Avena Sativa (Oat) Kernel Flour from dehulled, cleaned high-quality oats is completed under sanitary conditions. 56 Good manufacturing practices according to 21 CFR 110 and current USP monographs are followed. There are no other ingredients used in the process.
To extract proteins from oat kernels for potential use in cosmetics, a first extract was prepared from dried grains (200 g) by extracting the grains twice with aqueous sodium hydroxide at pH 8 (1 L) for 1 hour at room temperature. 57 After centrifugation, the supernatant was precipitated with aqueous hydrochloric acid (at pH 5.4) and centrifuged. The precipitate was suspended in water, dialyzed overnight at 4°C using a 6,000 to 8,000 Da molecular weight cutoff dialysis bags, and then lyophilized. A second extract was obtained from dried grains (40 g) by extracting with 200 mL of 70% ethanol (aqueous) for 1 hour at boiling temperature. This extract was then centrifuged and the precipitate dried. The second extract (2 g) was combined with the first one (1 g) to obtain the grain protein extract.
According to one supplier, to produce Avena Sativa (Oat) Leaf/Stem Extract, plantlets (young or small plants) are extracted with 80% acetone and water. 51 The resulting medium is filtered and concentrated to 0.9 L per 1 kg of engaged plant. After filtration of the aqueous concentrate, the extract is concentrated, filtered, and sterilized by filtration. It is further concentrated up to 40% to 50% of dried extract. The medium is then stabilized and dried with maltodextrin. The resulting composition of the extract is Avena Sativa (oat) leaf/stem extract, 75%; maltodextrin, 25%.
According to another supplier, to produce protein-free extracts of Avena sativa, young plants were air-dried and ground. 57 A 200 g sample of the dried, ground plant was extracted with 2 L acetone/water 80:20 (vol/vol) under constant agitation and refluxed for 1 hour. After filtration, the extract was concentrated to eliminate the acetone and precipitate lipophilic constituents. Filtration and drying produced a beige powder (yield 11.3%). An aliquot of the extract (2 g) was subjected to chromatography. Four fractions of eluent were collected by successive elution with 10 mL of 25% methanol (fraction 1), 10 mL 50% methanol (fraction 2), and 20 mL 100% methanol (fraction 3). The same operation was repeated 3 times and the corresponding fractions were pooled to obtain 4 g of fraction 1, 0.58 g of fraction 2, and 0.27 g of fraction 3.
For the preparation of A sativa plantlet protein extract, one supplier reported that fresh oat plantlets were homogenized in a buffered extraction medium containing Tris acetate, 100 mM (pH 7.5) lithium chloride, 50 mM dithiothreitol, 20 mM sodium dodecyl sulfate, 40 g/L of 3 M urea, and 1 M thiourea, followed by a 1-hour maceration at room temperature. After filtration, the extracted fraction was purified by precipitation from acetone.
In the kernel protein and plant protein extracts above, protein concentrations were determined as 20% (wt/wt) and 40% (wt/wt), respectively. Analysis of the protein-free plant extract by silver nitrate protein staining showed no protein (limit of detection of 0.3 ppm). 57
In another procedure to produce extracts (information was unclear on the exact plant parts and the solvents used) without detectable proteins, young A sativa plants are dried and crushed. 58 An extraction is performed with stirring for 1 hour. The extract is filtered and the residue is rinsed. The filtrate is then concentrated, delipidated, and dried, yielding an extract in powder form containing 2% to 15% flavonoids and 0.2% to 2% avenacosides A and B.
To manufacture Avena Sativa (Oat) Sprout Oil, one supplier reported the oil is extracted from oat sprouts with acetone and the extract is filtered. 51 The oil is then concentrated, followed by a final filtration. The oat flake raw material used in the manufacture of Hydrolyzed Oats is food grade; the resulting Hydrolyzed Oats are not used in human food. 14
Hydrolyzed Oats is manufactured by mixing the oat flake with water, then hydrolysis by enzymes. 59 The mixture is then filtered and evaporated. The liquid is spray-dried to create a powder form. The products are analyzed and packed. Another manufacturer reports that the process entails enzyme hydrolysis of oats, followed by purification steps that include enzyme denaturation, filtration, evaporation, and preservation. 15 The sodium hydroxide, enzymes, oats, potassium sorbate, and disodium EDTA are food grade. It is not known whether the hydrochloric acid and sodium benzoate are also food grade.
Impurities
Analysis of an Avena Sativa (Oat) Leaf/Stem extract and an Avena Sativa (Oat) Sprout Oil (100%) showed that allergens listed in European Union (EU) regulation 1223/2009 60 were below detection level as measured by gas chromatography–mass spectrometry; heavy metals (As, Cd, Cu, Fe, Hg, Ni, Pb, Zn, Ag, Ba, Se, Sb, Cr, and Co) totaled <20 ppm and that pesticide concentrations were compliant with EU Pharmacopeia. 51,61
There were no detectable proteins (limit of detection of enzyme-linked immunosorbent assay less than 0.5 ppm protein) in an extract of young A sativa plants (solvent(s) not specified). 58
Fusarium avenaceum, Pseudodiscosia avenae, and Sclerospora macrospora are among the species of fungi known to infect oat plants, including A sativa. 18 Two of 5 oat-based cereals tested positive for the mycotoxin deoxynivalenol (DON) at a concentration of 2.6 and 1.3 µg/g cereal. 62 Three of these products tested positive for zearalenone (ZEA) at an average concentration of 16 ng/g cereal. Aflatoxin B1 was not detected in these samples. The mycotoxins DON, 3-acetyl DON (3AcDON), nivalenol, neosolaniol, T-2 triol, T-2 toxin, and HT-2 toxin were detected in samples of recently harvested oats (species/varieties not provided). 63 Samples were obtained from both conventional and organic farms. In Avena Sativa Bran samples (n = 30), collected from grocery stores and health food stores in Spain, ZEA was detected in 17% of the samples, DON in 17%, and ochratoxin A in 20%. 64
Cadmium content in fresh A sativa grown in Finland ranged from 0.008 to 0.120 mg/kg dry weight. 65 There was no difference in cadmium content between conventionally and organically grown crops. Nitrogen fertilization increased cadmium content. Cadmium content may vary by strain and may exceed the safe level for human consumption set by the European Commission (0.1 mg/kg fresh mass). 66
Use
Cosmetic
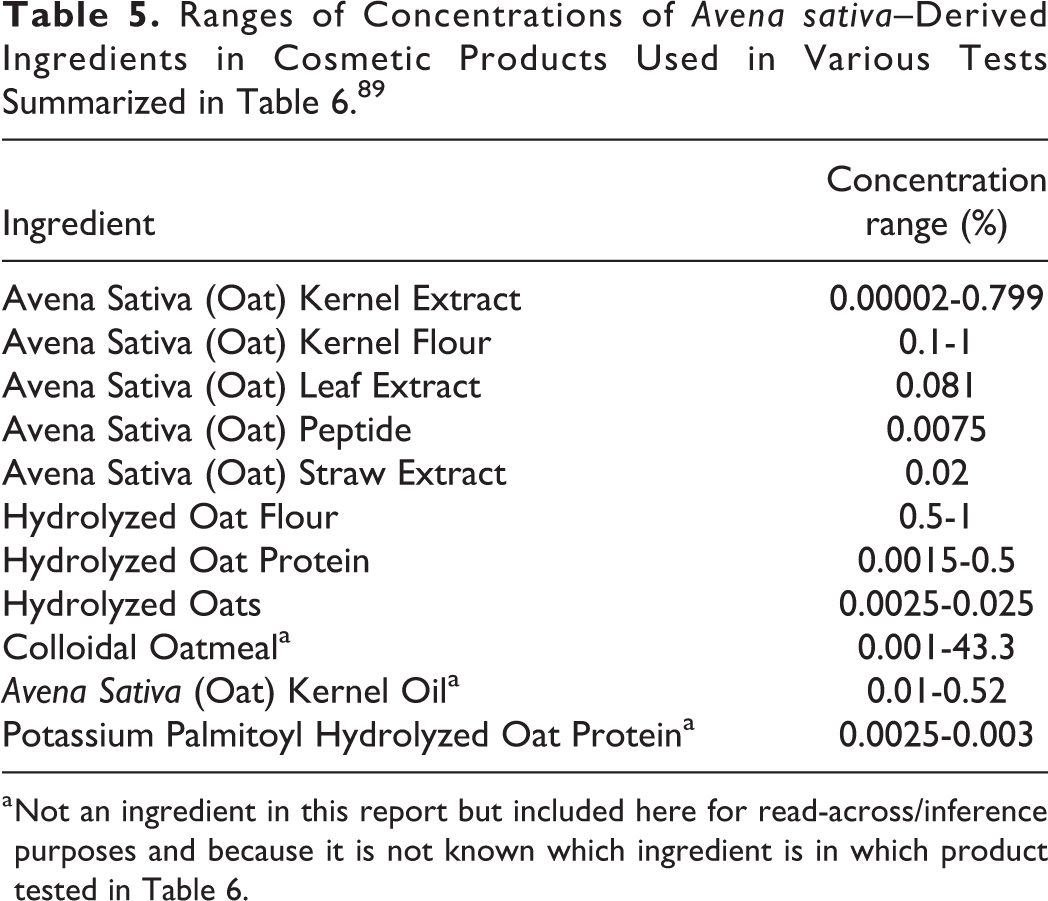
The A sativa (oat)–derived ingredients were reported to function in cosmetics as abrasives, antioxidants, skin conditioning agents, absorbents, and bulking agents. 1 Data on ingredient usage are provided to the Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (Table 4). 67 A survey was conducted by the Personal Care Products Council (Council) of the maximum use concentrations for these ingredients reported by industry. 68,69
Abbreviations: NR, not reported; Totals = rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
c Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
d It is possible these products may be powders, but it is not specified whether the reported uses are powders.
e Baby products are not powders.
f Pump spray.
Avena Sativa (Oat) Kernel Extract has the most reported uses, with 499 in cosmetic products. Avena Sativa (Oat) Kernel Flour has the highest reported use concentration of 84.4% in skin cleansing products; Avena sativa (Oat) Kernel Extract has the highest reported leave-on use concentration of 25% in face and neck products. 67 -69
There were no reported uses for: Avena Sativa (Oat) Flower/Leaf/Stem Juice Avena Sativa (Oat) Leaf/Stalk Extract Avena Sativa (Oat) Leaf/Stem Extract Avena Sativa (Oat) Meristem Cell Extract Avena Sativa (Oat) Seed Extract Avena Sativa (Oat) Seed Water Avena Sativa (Oat) Sprout Oil
Avena sativa (Oat) Kernel Extract was reported to be used in face and neck spray products in concentrations up to 0.0025% and Avena Sativa (Oat) Kernel Protein in pump hair sprays in concentrations up to 0.001%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 70 -73 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 70,72
Noncosmetic
Avena sativa–containing products are used medically as dermal moisturizers and to treat itchy skin due to dryness, chicken pox, poison ivy/oak/sumac, and insect bites. 74 They are also used to treat acne.
Colloidal oatmeal, including that derived from A sativa, is used in dermatological practice as an adjunctive therapy to treat many pruritic skin conditions such as cercarial dermatitis (swimmer’s itch), chicken pox, poison ivy, oak and sumac, insect bites, winter itch, atopic dermatitis, dry skin, allergic or irritant contact dermatitis, and ichthyosis. 39,40,75 -80 Other indications for colloidal oatmeal products include prickly heat, hives, sunburn, and rashes. It is regulated for these uses by the FDA as an over-the-counter drug and can be included in tub baths at a minimum concentration of 0.007% if alone or at a minimum concentration of 0.003% when combined with mineral oil [21 CFR347.10(f), 21 CFR347.10(o)]. Colloidal oatmeal is to be used in footbaths at a minimum concentration of 0.25% [21CFR347.20].
For agricultural purposes, the FDA specifies that oat grain consists of 50% or more of oats (A sativa L. and A byzantina C Koch) and may contain, singly or in combination, not more than 25% of wild oats and other grains for which standards have been established under the United States Grain Standards Act [7 CFR 810.1001].
The FDA defines the following foods derived from oats:
Oat bran: Oat bran is produced by grinding clean oat groats (hulled kernels) or rolled oats and separating the resulting oat flour into fractions such that the oat bran fraction is not more than 50% of the original starting material and provides at least 5.5% (dry weight basis [dwb]) β-glucan soluble fiber and a total dietary fiber content of 16% (dwb) and such that at least one-third of the total dietary fiber is soluble fiber [21 CFR 101.81].
Rolled oats: Rolled oats, also known as oatmeal, produced from 100% dehulled, clean oat groats by steaming, cutting, rolling, and flaking, provide at least 4% (dwb) β-glucan soluble fiber and total dietary fiber content of at least 10%.
Whole oat flour: Whole oat flour is produced from 100% dehulled, clean oat groats by steaming and grinding, such that there is no significant loss of oat bran in the final product, and provides at least 4% (dwb) β-glucan soluble fiber and total dietary fiber content of at least 10% (dwb).
Oatrim: The soluble fraction of α-amylase-hydrolyzed oat bran or whole oat flour. Oatrim is produced from either oat bran, as defined in paragraph (c)(2)(ii)(A)(1) of 21 CFR 101.81, or whole oat flour, as defined in paragraph (c)(2)(ii)(A)(3), by solubilizing the starch in the starting material using an α-amylase hydrolysis process, followed by centrifugation to remove the insoluble components consisting of a high portion of protein, lipid, insoluble dietary fiber, and the majority of the flavor and color components of the starting material. The FDA regulation specifies that oatrim shall have a β-glucan soluble fiber content up to 10% (dwb) and not less than that of the starting material (dwb) [21 CFR 101.81].
Toxicokinetics
Since these ingredients are complex botanical mixtures, obtaining useful and informative data on the toxicokinetics of A sativa–derived ingredients would not, in practicality, be possible. However, since these ingredients are safely consumed as food and by livestock in their feed and systemic exposure to the components of these ingredients via their presence in cosmetics is expected to be much lower than dietary exposure, concern over absorption is mitigated.
Toxicological Studies
Avena sativa oats and other plant parts are used extensively in human food, as well as in animal feed, resulting in much larger systemic exposures than would be possible from cosmetic uses. Thus, the concern for potential for systemic effects is mitigated. Accordingly, while all available, relevant systemic toxicity data have been included herein, the primary focus of this report is on the potential for local effects, such as irritation and sensitization.
Dermal Effects
Overview of Dermal Effects
The dermal effects of colloidal oatmeal derived from A sativa have been attributed to the anti-inflammatory and antipruritic properties of the avenanthramides. These constituents have been shown to reduce oxazolone-induced contact hypersensitivity, resiniferatoxin-induced neurogenic inflammation, and induced histamine-mediated itch. 81 In vitro, avenanthramides reduced histamine release from mast cells stimulated by substance P. 81 The buffering property of colloidal oatmeal (the pH of the skin surface is important for preservation of skin barrier function) was demonstrated when treatment with colloidal oatmeal reduced the elevated pH of diseased skin (eg, eczematous or pruritic) and alkali-treated normal skin to within the normal range. Other reported skin-barrier-related effects include the formation of a protective moisturizing barrier by the proteins and polysaccharides in colloidal oatmeal, which reduced transepidermal water loss. Colloidal oatmeal has also been shown to act as an emollient, humectant, and occlusive on the skin. 82 The application of A sativa extracts to sodium lauryl sulfate–treated skin has been reported to reduce irritation, demonstrating the anti-inflammatory effects of oats and suggesting potential benefits for the skin barrier. 83
In Vitro
Avena sativa extracts reportedly inhibited the phospholipase A2 PLA2-dependent mobilization of arachidonic acid from phospholipids in cultured human keratinocytes. 84 This extract also inhibited the formation of eicosanoids, expression of cytosolic phospholipase PLA2, and formation of metabolites of prostacyclin in keratinocytes, all of which are implicated in the regulation of inflammation. An A sativa extract oligomer reduced vasodilation induced by vasoactive intestinal peptide in human skin samples. 85 Treatment with the oligomer reduced edema and mean surface of dilated vessels. It has also been reported that colloidal A sativa extracts (both ethanol and phosphate buffer; with and without boiling) inhibited the activity of prostaglandin synthase of bull seminal vesicles. 13
When fibroblasts from cosmetic surgery patients were incubated with A sativa whole-young-plant extract (0.05%; solvent not provided), there was an increase in the proliferation of the cells and extension of a neoepithelium compared to untreated cells. 86 There were no differences in the number of basal layers up to day 20 postexposure and then there were more layers observed in the treated cells on day 22. The dermal equivalent was created in a petri dish by combining the dermal fibroblasts with collagen type I. A punch biopsy from skin left over from surgery was used as the source of epidermal cells, which were then placed on the dermal equivalent, where a multilayered epidermis developed.
Nonhuman
Avena sativa whole plant extract
In a wound healing experiment using the n-hexane, ethyl acetate, ethanol, and water extracts of whole A sativa plants, there were no adverse effects to Sprague Dawley rats (n = 6+) and Swiss albino mice (n = 6+) when the extracts (1%, 0.5 g in an ointment base) were administered to wounds daily for 9 days. 31 The ethanol extract increased wound healing activity, the other extracts did not. The rats and mice were anesthetized and either 2 incisions along either side of the backbone or biopsy punches were performed. The extracts were administered to the wounds once per day for 9 days. The rats and mice were killed and the wounds excised. The healing of the incisions was measured by tensile strength across the wound and the healing of the punches was measured by area of healing.
Human
Colloidal oatmeal
In a blind study of acute burn patients (n = 35), a shower/bath oil containing colloidal oatmeal (5% in liquid paraffin) resulted in no adverse effects. 87 The group using colloidal oatmeal had reduced itchiness compared to the group using paraffin oil alone. The subjects showered or bathed with the test material or the same product without the colloidal oatmeal for 30 days. Patients who had been admitted to intensive care were excluded from this study.
Complete or marked itch relief was reported by over 71% of the subjects (n = 139; aged 21-91) suffering from pruritic dermatoses when colloidal oatmeal was used as a bath and regular cleanser for 3 months with no adverse effects. 78
Pediatric subjects (n = 152) presenting with atopic dermatitis, contact dermatitis, fungus infections, or seborrheic dermatitis who were administered baths with colloidal oatmeal in an oil exhibited no adverse effects. 76
Reproductive and Developmental Toxicity
Data on the reproductive and developmental toxicity of A sativa (oat)–derived ingredients were not found in the published literature, nor were unpublished data provided.
Antiestrogenic Activity
When 23- to 24-day-old female rats (n = 5-10) were subcutaneously injected with any of 3 A sativa hay extracts (0.15 mL in olive oil) and 0.05 µg estradiol, uterine weights were less than in the rats injected with estradiol alone. 88 This result was consistent when the extraction solvent was ether, the chloroform-extract fraction of the ether extract, or the fraction obtained from the ether extract passed over an alumina column and eluted with chloroform. The extracts were processed by first extracting ground A sativa hay with HCl followed by precipitation with ethanol. The solids were filtered out and discarded. The ethanol was evaporated and the remaining aqueous phase was extracted with ether in a separating funnel. The residue was then extracted with chloroform.
Genotoxicity
Avena Sativa (Oat) Leaf/Stem Extract
In the Ames test performed following the Organization for Economic Cooperation and Development (OECD) test guideline (TG) 471, using Salmonella typhimurium (strains TA98, TA100, TA102, TA1535, and TA 1537), Avena Sativa (Oat) Leaf/Stem Extract (concentration not specified) was not mutagenic, with or without metabolic activation. 51
Avena Sativa (Oat) Leaf/Stem Extract (concentration not specified) was not mutagenic in a micronucleus test on mouse lymphoma cells (L5178Y/TK+/−) following OECD TG 487. 51 The test material did not exhibit an in vitro intrinsic genotoxic potential in conditions of this study with or without metabolic activation.
Avena Sativa (Oat) Sprout Oil
In 2 in vitro assays, Avena Sativa (Oat) Sprout Oil (concentration not specified) was not mutagenic. 51 In a fluctuation Ames test, the test material was not mutagenic with or without metabolic activation system. In a micronucleus test, performed in accordance with OECD TG 487, on Chinese hamster ovary cells, the test substance did not demonstrate intrinsic genotoxic potential up to 1,500 ppm, without metabolic activation, and up to 150 ppm with metabolic activation.
Carcinogenicity
Data on the carcinogenicity of A sativa (oat)–derived ingredients were not found in the published literature, nor were unpublished data provided.
Irritation and Sensitization
Dermal Irritation
Human
In a series of cumulative irritation tests (total N = 1,717), it was concluded that multiple products, each containing an A sativa (oat)–derived ingredient (Table 5), were not irritants (Table 6). 89 The maximum irritation score was 0.326% (nonirritant score = 2.9%-5.0%). Each of the products were administered neat under semi-occlusion 3 times per week for 2 weeks. Patches were left in place for 48 or 72 hours. Times of observations were not provided. The concentrations of A sativa–derived ingredients ranged from 0.00002% to 1% except for colloidal oatmeal, which ranged up to 43.3%. This information was presented in aggregate and the individual studies on the individual ingredient-containing products were not provided.
a Not an ingredient in this report but included here for read-across/inference purposes and because it is not known which ingredient is in which product tested in Table 6.
Summary Information of Irritation and Sensitization Tests of Various Cosmetic Products Containing Avena sativa–Derived Ingredients.a
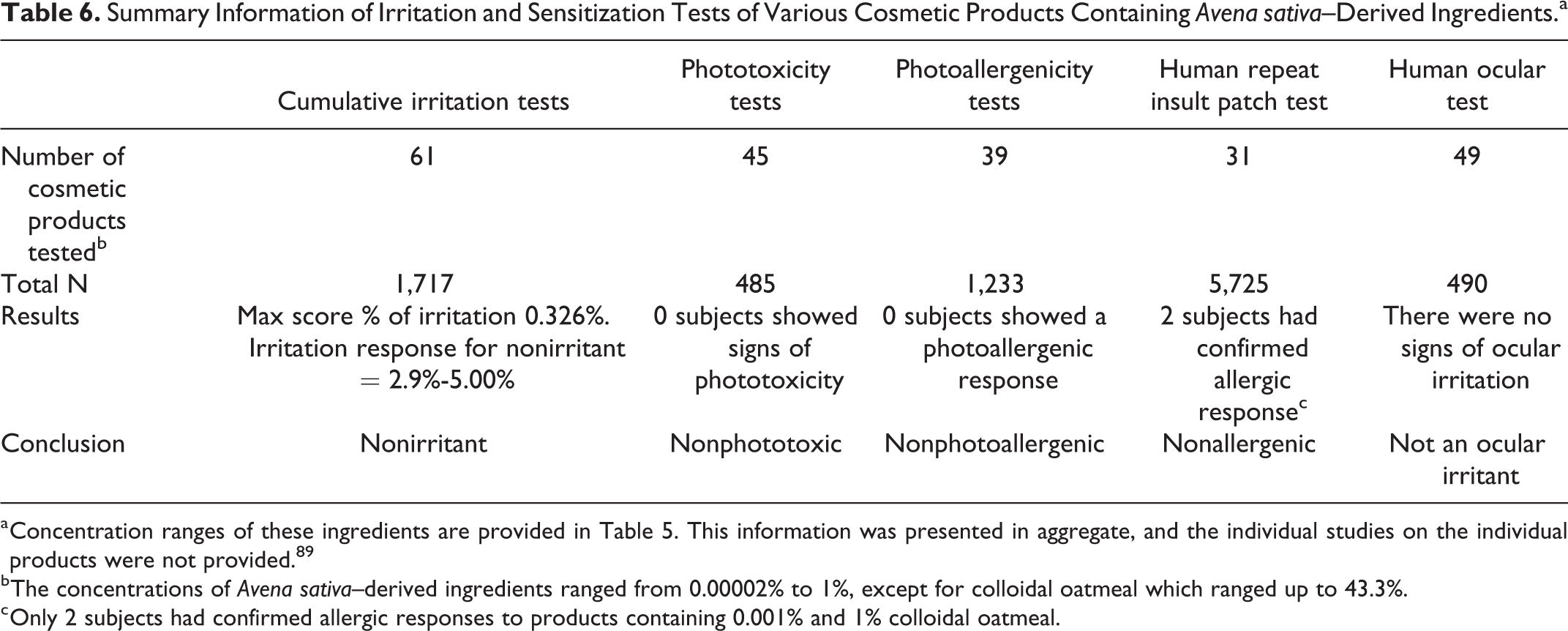
a Concentration ranges of these ingredients are provided in Table 5. This information was presented in aggregate, and the individual studies on the individual products were not provided. 89
b The concentrations of Avena sativa–derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which ranged up to 43.3%.
c Only 2 subjects had confirmed allergic responses to products containing 0.001% and 1% colloidal oatmeal.
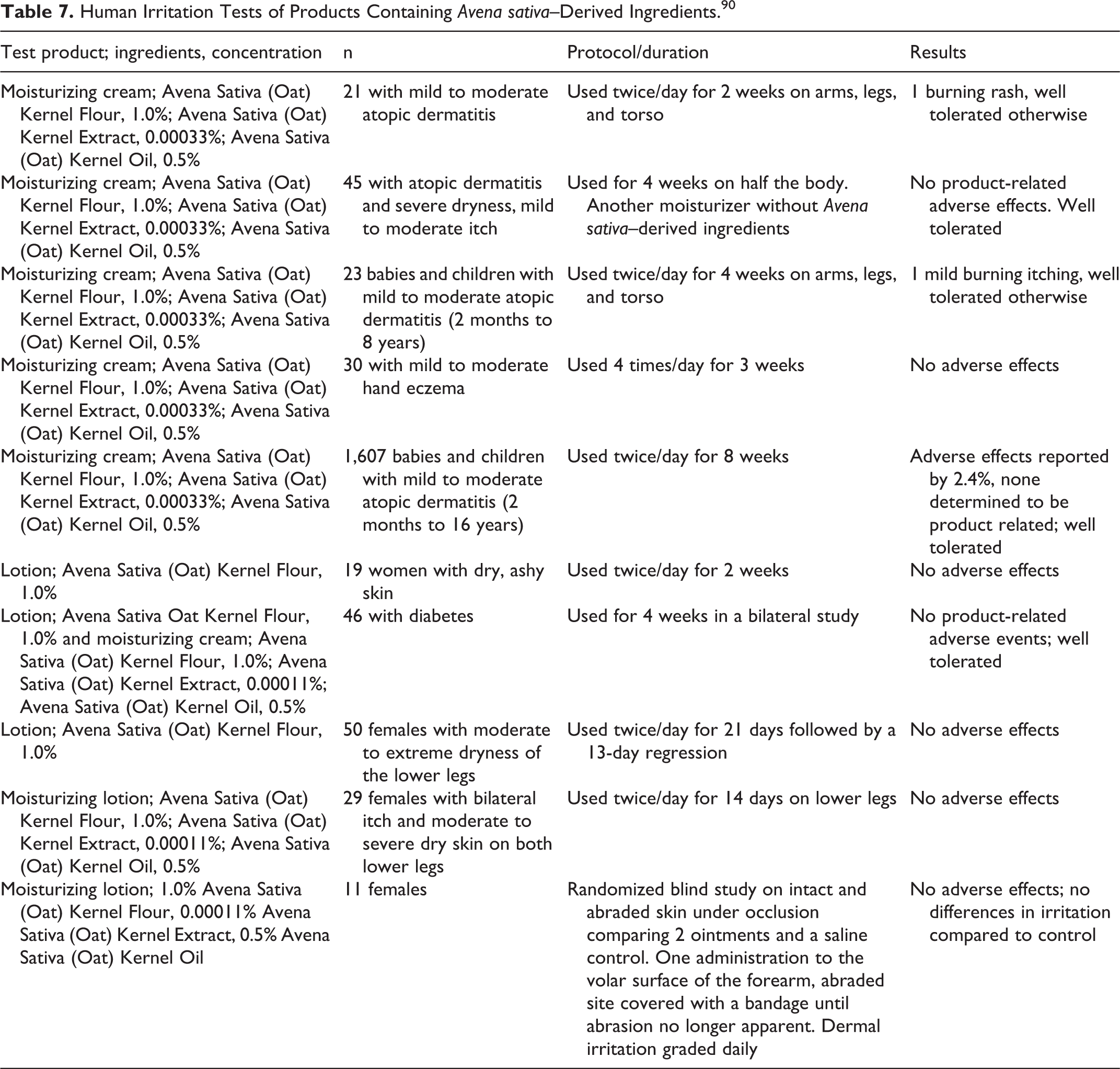
In another series of dermal studies of 10 moisturizing products that contain A sativa (oat)–derived ingredients (up to 1%) on subjects with various dermal issues, there were few adverse events and it was concluded in all tests that the test substance was well tolerated (Table 7). 90 Most of these products contained multiple A sativa–derived ingredients. Adverse events included burning rash and burning itching. There were no adverse events in subjects with diabetes or in babies and children.
Human Irritation Tests of Products Containing Avena sativa–Derived Ingredients. 90
When a cream containing an extract of young A sativa plants (information not clear on the type of extract, eg, Avena Sativa (Oat) Leaf/Stalk Extract and/or Avena Sativa (Oat) Leaf/Stem Extract; concentration, amount applied, and extract solvent not provided) was administered to female subjects (n = 16) with dry skin, there were no signs of irritation. 58 Sixty-three percent of the subjects used for this study had sensitive skin and 81% had sensitive eyes. The cream was administered to one or the other elbow fold twice daily for 4 days, then once more on day 5. The cream was also applied to one side of the face once daily.
In another study of the same product, no irritation was observed when the cream was administered to the tape-stripped skin of subjects (n = 19). Both elbow folds were stripped 6 times and the test material administered 72 hours later to one of the stripped sites. The test material was administered twice per day for 4 days, and once on day 5. The sites were examined for erythema, pruritus, heat, tingling, and burning on days 4, 5, 6, and 7. All subjects exhibited moderate to intense erythema after tape stripping prior to administration of the test material. No erythema was observed in 14 subjects by day 4 or in any subject by day 8. No subjects exhibited any symptoms of a reaction. 58
When an emollient containing an extract of young A sativa plants (concentration not specified), in addition to separately administered topical corticosteroids of both high or moderate potency, was administered to infants (<12 months old; n = 78, control = 70) with moderate to severe atopic dermatitis, the tolerance evaluation was good to very good in 89% of the subjects at day 21 and 94% at day 42 for A sativa emollient. 91 Three adverse events that were possibly treatment related were reported as mild and 3 as moderate. Two were severe and treatment was discontinued. All of the adverse events resolved spontaneously. Further details about the adverse events were not provided. The quantity of high-potency corticosteroids used by the parents on the subjects that were also administered the emollient reduced over time while the quantity of moderate-potency corticosteroids did not.
The information was not clear on the type of extract (eg, Avena sativa (Oat) Leaf/Stalk Extract and/or Avena Sativa (Oat) Leaf/Stem Extract) that was in the emollient. The control group was only administered the corticosteroids and the test group was administered the corticosteroids and the emollient containing the A sativa extract. The test substances were administered twice daily; the parents of the emollient group were instructed to administer the test substance “in sufficient amount on the dry, noninflammatory areas of the skin, over the whole body” for 21 days. The parents were supplied with 2 bottles of the emollient (400 mL each). The corticosteroids (high or moderate potency) were administered by the parents to the subjects as needed to treat the atopic dermatitis. The unused portions of the corticosteroids were returned for weighing. The subjects were evaluated on days 1, 21, and 42. 91
Colloidal oatmeal
In 12 use safety studies of various personal care products containing Avena Sativa Colloidal Oatmeal, there was a low percentage of subjects (0%-10.9%) who exhibited irritation; it was concluded that these products had a low potential for irritation (Table 8). 92 The concentrations of colloidal oatmeal were not provided. The products tested were a shower and bath oil, cream, moisturizing oil, shower gel, night cream, conditioning shampoo, body lotion, liquid hand wash, face and eye cleansing lotion (2 products), facial exfoliating cleanser, intimate wash, and baby milk. Assessments, conducted by a dermatologist, included visual examination of skin dryness and appearance of the skin, as well as tactile evaluation of skin roughness. A 10-cm visual analog scale was used, where 0 represented “none” and 10 was “severe”. The subjects self-assessed using a questionnaire with a 5-point scale. Measurements were made on the treated body areas (leg and inner forearm), as well as on an untreated area on the mid-thigh, which served as a control site. Clinical assessments were performed only on the treated leg and on the control area.
Use Safety Tests of Personal Care Products Containing Colloidal Oatmeal Derived From Avena sativa.a
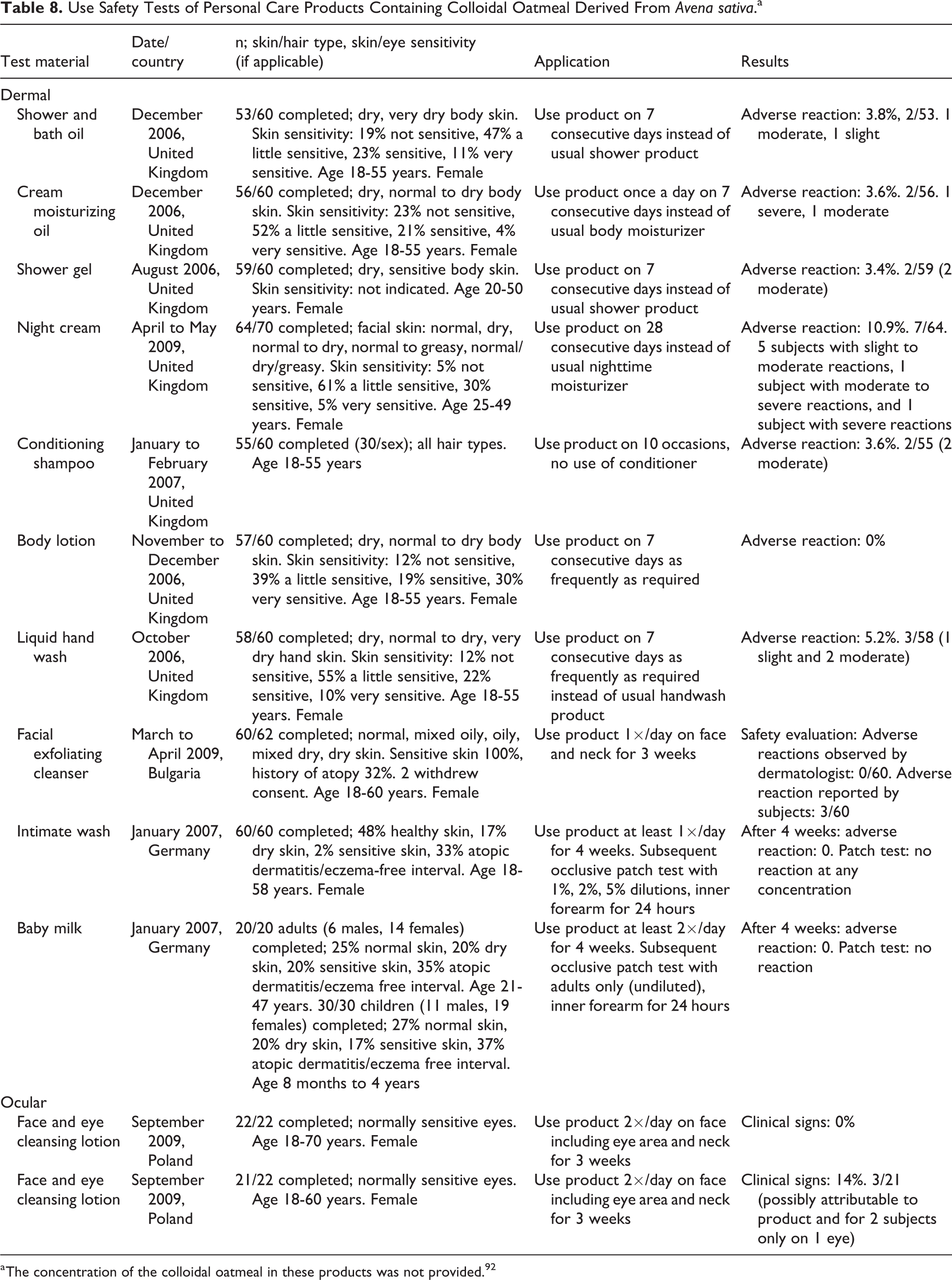
a The concentration of the colloidal oatmeal in these products was not provided. 92
There were no adverse effects reported for children (aged <14 years) with mild atopic dermatitis who used 5 different baby products (n = 55, 29, 75, 37, and 67) containing colloidal oatmeal (concentrations not specified) for 12 weeks. 93 Evaluation of their skin conditions were improved in 201 of 263 cases after 3 months of treatment (in 153/263 after 2 weeks), remained unchanged in 60 of 263 (in 108/263 after 2 weeks), and deteriorated in 2 of 263.
No adverse effects were observed or reported by the subjects (n = 54) with various dry skin conditions in an efficacy study of moisturizing lotion containing colloidal oatmeal (concentration not specified). 21,94 Improvement in cutaneous lesions including erythema, scaling, scratching lesions, lichenification, and pruritus was reported in 52 out of 54 subjects. The lotion was used as the only treatment once a day for 3 weeks. Patients were allowed to use neutral cleansing daily.
In Vitro
Avena Sativa (Oat) Leaf/Stem Extract
Avena Sativa (Oat) Leaf/Stem Extract (100%) was rated as nonirritant in a Reconstructed Human Epidermis Model test (RHE SkinEthic). 51
Hydrolyzed oats
In an in vitro toxicity test using the MATREX system, Hydrolyzed Oats (100%) was not predicted to be a dermal irritant. 95 At 1%, 10%, and 100%, the viability after 1 hour was 97%, 121%, and 120%, respectively, compared to controls. Propylene glycol and morpholine served as the positive and negative controls, respectively. The test used a 3-dimensional construct of living cells on a collagen matrix that was to mimic human skin. Viability of the cells was measured photometrically after administration of tetrazolium salt (MTT).
In an in vitro toxicity test using the EpiDerm Skin Model, Hydrolyzed Oats (100%) was not predicted to be a dermal irritant. 96 At 1, 4.5, and 20 hours, the viability was 104%, 79%, and 99%, respectively, compared to controls. Triton X 100 served as the control. The test used human keratinocytes. Viability of the cells was measure by photometrically after administration of MTT.
Dermal Sensitization
Nonhuman
Avena Sativa (Oat) Leaf/Stem Extract
In a local lymph node assay (LLNA), using nongravid female mice (n = 5), of dermally administered Avena Sativa (Oat) Leaf/Stem Extract (1%, 10%, 25%, 50%, and 70% in diluted propylene glycol/water, 50/50), the stimulation indices (SI) were 0.7, 0.6, 0.9, 1.8, and 4.4, respectively. 51 The test substance was not a sensitizer, except at 70% (SI ≥ 3). The EC3 (estimated concentration needed to produce a stimulation index of 3) was 59%.
Avena Sativa (Oat) Sprout Oil
In an LLNA, Avena Sativa (Oat) Sprout Oil (2%, 10%, 30%, and 100%) did not induce delayed contact hypersensitivity when dermally administered to female CBA mice (n = 4) for 3 consecutive days. 51 The protocol followed those in OECD TG 429.
Human
In a series of human repeated insult patch test (HRIPT; total N = 5,725), it was concluded that multiple products, each containing an A sativa–derived ingredient (Table 5), were not sensitizing (Table 6). 89 Only 2 subjects had confirmed allergic responses to products containing 0.001% and 1% colloidal oatmeal. The follow-up data for these subjects were lost. The test substance (100%) was administered under occlusion 3 times per week for 3 weeks (for a total of 9 applications). Patches were left in place for 24 to 72 hours. After a 2-week rest period, a new patch was administered for 24 hours (times of observation were not provided). The concentrations of A sativa–derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which was applied at up to 43.3%. This information was presented in aggregate, and the individual studies on the individual ingredient-containing products were not provided.
Avena Sativa (Oat) Kernel Extract
A paste mask product containing Avena Sativa (Oat) Kernel Extract (25%) was not sensitizing in a double-blind HRIPT (n = 111). 97 No responses were observed at any phase of the study. The test material (150 µL) was administered, under semi-occlusion, 3 d/wk for 3 weeks, and removed after 24 hours. The challenge was administered on the fourth week of the study.
Avena Sativa (Oat) Kernel Flour
A face powder containing Avena Sativa (Oat) Kernel Flour (1%) was not sensitizing in an HRIPT (n = 51). 98 In the induction phase, the test material was administered to the backs of the subjects and the patches left in place for 24 hours. This was repeated 9 times consecutively. The test sites were observed immediately upon removal of the patch or on the Monday following the removal of the patch on a Saturday. After a 2-week rest, the test material was administered to a naive site and was left in place for 24 hours. The challenge site was observed at removal and at 48 and 72 hours. In an HRIPT (n = 56) following the same procedure, a blush containing Avena Sativa (Oat) Kernel Flour (1%) was not sensitizing. 99
A body lotion that contained Avena Sativa (Oat) Kernel Flour (0.1%) was not sensitizing in an HRIPT (n = 93). 100 One subject exhibited transient, low-level (±1) reactions accompanied by dryness, and another subject exhibited dryness. In the induction phase, 0.2 g of the test material was administered to the skin in the scapular region under occlusion. Induction exposure was repeated 9 times for 24 hours each. The challenge was 0.2 g of the test material administered to a naive site for 24 hours. The test site was observed at 24, 48, 72, and 96 hours after the challenge patch was removed.
Hydrolyzed oats
Hydrolyzed oats (100%; 0.2 mL) was not sensitizing in an HRIPT (n = 52). 101 There were no signs of irritation or sensitization during the test. The test substance was administered to the scapular region under occlusion on Monday, Wednesday, and Friday for 10 applications. All patches were removed after 24 hours. After approximately 14 days of rest, the challenge patch was administered to a naive site on the volar forearm.
Other A sativa–derived ingredients
In a use study of a cream and soap containing an extract of young A sativa plants, subjects (n = 8 females, 4 males) with a history of cereal-sensitized atopic dermatitis did not develop immediate or delayed-type hypersensitivity in response to the products after using them for 21 days. 102 The cream contained 12% and the soap contained 3% of the extract. Prior to and after the 21-day use study, none of the subjects displayed positive reactions in patch tests and skin prick tests of 5 fractions of the extract used in the products or the study cream. Total serum A sativa IgE levels analyzed before and after the use study did not change.
In the first 10 days of the use study of the cream and soap, open application tests, prick tests, and IgE tests of the A sativa extracts (colloidal 5%, phenolic 5%, acetonic 5%, enzyme-hydrolyzed phenolic 5%, and acetonic 5%) and the cream were conducted on all subjects. During these 10 days, the subjects used their own cream and soap (ingredients unknown). On day 11, the test cream was administered to one half of each body. The vehicle cream, without the A sativa extract, was administered to the other half of each body. The subjects showered 4 hours later using the test soap. The subjects then used the cream containing the extract twice per day and showered with the soap once per day for a total of 21 days. The patch test and a skin prick tests were repeated after the use part of the experiment, and total IgE and A sativa–specific IgE were measured. 102
There were no signs of irritation or sensitization in a HRIPT (n = 104) of a cream containing A sativa (concentration not provided; 50 µL). 58 The test material was administered in a Finn chamber on days 1, 3, 5, 8, 10, 12, 15, 17, and 19, for 48 or 72 hours. Two weeks later, the challenge patch was left on a naive site for 48 hours.
In a group of children (under 15 years of age) referred for allergy testing (n = 150 females, 152 males), 14.6% had positive results in a patch test of the A sativa young plant extract described above (1%, 3%, and 5%). 103 Sixteen of 44 subjects tested positive at 5%, 6 each for 3% and 5%, and 22 subjects reacted to all 3 concentrations. Of those sensitized, 15.6% (5/32) and 28% (7/25) tested positive in an oral food challenge and a repeated open application test.
In a skin prick test of the subjects in the previous study, 19.2% had positive reactions to oat pollen. Sensitization was observed in a total of 32.5% of the subjects demonstrated by either the patch or skin prick test; only 4 subjects tested positive in both tests. Sensitization decreased with the age of the subjects.
In a history survey of 67 of the subjects, no connection was found between sensitization and clinical signs (asthma, hay fever, atopic dermatitis severity); home location; proximity of cereal production; consumption of oats; skin prick test results to grass, cereal pollen, or wheat pollen; or oat- or wheat-specific IgE. In the patch test, 100% of the subjects that had not used products containing A sativa tested negative; only 66.7% of those that had used product containing A sativa had negative results (P = 0.0068). 103
In a commentary of the above history study, 103,104 it was proposed that the conclusion that children (who have immature epidermal barrier that could be more reactive) should avoid exposure to products containing A sativa–derived ingredients to avoid developing atopic dermatitis is not supported by the experiment. The authors of the commentary stated that this study is not enough evidence to come to this conclusion and that it does not experimentally connect the use of products containing A sativa–derived ingredients with sensitization. It was pointed out that the prick tests were carried out with oat pollen, not derivatives of the A sativa kernels or the plant, which are the source materials of A sativa (oat)–derived ingredients. It was also pointed out that there have been multiple other studies of products containing these ingredients, or these ingredients solely, with few or no reactions. 105 -107 It was also noted that there are millions of oat-containing products on the market and very few cases of allergic contact dermatitis to oats reported. 108,109 These authors noted their own experiment in which oat colloidal extract was unable to trigger any immunization reaction in mice with atopic dermatitis. 107 They proposed that a study on a large population of atopic children with repeated long-term use of emollients with and without A sativa-derived ingredients would be needed before coming to the conclusion proposed by the authors of the history survey.
Colloidal oatmeal
Children (n = 65; 6 months to 2 years of age) who were atopic or nonatopic, with and without previous exposure to Avena Sativa Colloidal Oatmeal, did not show signs of immediate or urticarial allergic reactions to either of 2 bath products containing Avena Sativa Colloidal Oatmeal at the expected use concentration (0.007% in water) or at an elevated concentration (0.7% in water). 106 These subjects were also nonreactive to Avena Sativa Colloidal Oat Flour (0.7% and 0.007% in water). The subjects were exposed to the bath products for 15 minutes. There were no reactions. Then a patch test using a pair of Finn chambers (50 µL) for each test substance and concentration was conducted. One of each pair of chambers was removed and the test sites observed after 24 hours, and the second set was removed after 48 hours. The skin under both sets of chambers was examined at 72 and 96 hours after removal.
In 12 HRIPTs (total N = 2,291) performed using 12 skin care products containing Avena Sativa Colloidal Oatmeal, the products did not produce signs of sensitization (Table 9). 92 The test substances comprised 3 lotions, 2 face creams, 1 serum product, 2 cleansing lotions, 1 exfoliating cleanser, 2 baby products (1 cream and 1 cleanser), and 1 hand cream. The concentrations of colloidal oatmeal in the products were not specified. Overall, 23 subjects experienced a reaction. A total of 34 transient low-level grade ± reactions (ie, faint, minimal erythema) were observed, including 1 subject with 8 consecutive faint erythema readings, 6 transient low-level grade 1 reactions in 6 subjects, and mild erythema in 1 subject. In the challenge period, 17 subjects had the following reactions: 18 transient low-level grade ± reactions in 14 subjects, 9 transient low-level grade 1 reactions in 7 subjects, and 5 grade 1 reactions with edema in 3 subjects. Edematous reactions were not confirmed in subsequent patch tests on 2 of the subjects. The other subjects’ reactions were confirmed for the complete product.
Human Repeat Insult Patch Tests of Personal Care Products That Contain Colloidal Oatmeal Derived From Avena sativa. 92
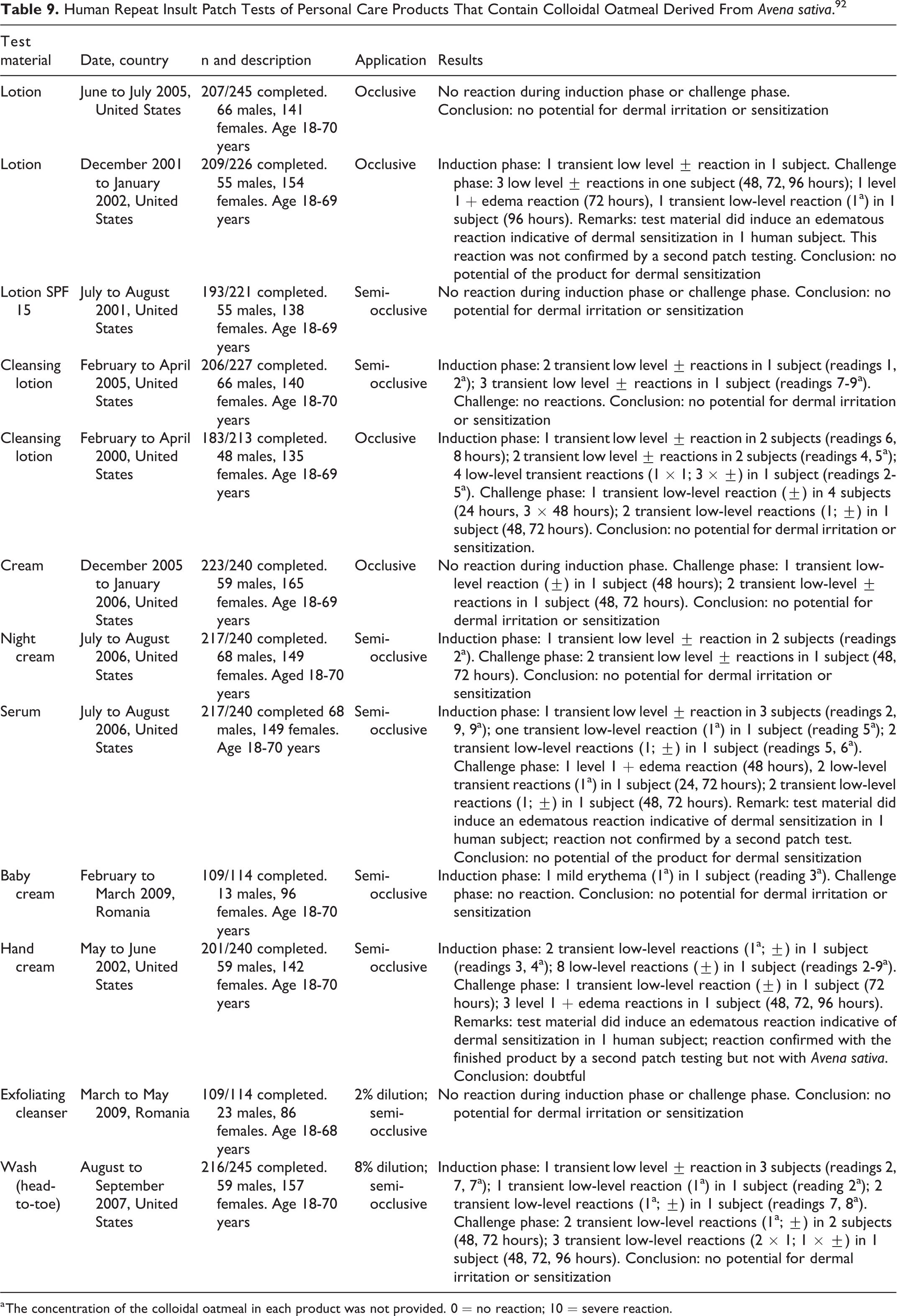
a The concentration of the colloidal oatmeal in each product was not provided. 0 = no reaction; 10 = severe reaction.
Photoirritation and Phototoxicity
In Vivo—Nonhuman
Avena sativa has been reported to cause photosensitization when consumed by cattle, goats, pigs, and sheep. 110 No further information was provided.
Avena Sativa (Oat) Leaf/Stem Extract
In a guinea pig maximization assay, Avena Sativa (Oat) Leaf/Stem Extract was not a photoirritant up to 70%, but was a slight photosensitizer (class II). 51 No further details were provided.
In Vivo—Human
In a series of phototoxicity tests (total N = 485) and photoallergy tests (total N = 1,233), it was concluded that multiple products, each containing an A sativa–derived ingredients (Table 5), were not phototoxic or photoallergenic (Table 6). 89 The maximum irritation score was 0.326% (nonirritant score = 2.9%-5.0%). The concentrations of A sativa (oat)–derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which ranged up to 43.3%. This information was presented in aggregate and the individual studies on the individual ingredient-containing products were not provided.
In the phototoxicity tests, the finished products were administered (100%) under occlusion on 2 sites on the subjects’ back for 24 hours. The patches were removed and one of the test sites exposed to long-wavelength ultraviolet light (UVA). Exact wavelengths and times of observation were not provided.
In the photoallergy tests, the finished products (100%) were administered on 2 sites on the subjects’ upper back for 24 hours. Following removal of the patch, one site was exposed to UVA and mid-wavelength UV (UVB). Exact wavelengths and times of observation were not provided. After a 2-week rest, 2 more patches were administered for 24 hours followed by the irradiation of one site with UVA. The subjects’ skin was classified as having Fitzpatrick skin types I, II, or III. 89
In Vitro—Human
Avena Sativa (Oat) Sprout Oil
Avena Sativa (Oat) Sprout Oil (100%) was not phototoxic in a Human Epidermis Model test (RHE SkinEthic). 51 In an in vitro 3T3 phototoxicity assay, the test substance was also not phototoxic. The test was performed according to OECD TG 432; no further details were provided.
Ocular Irritation
Human
In a series of human ocular tests (total N = 490), it was concluded that multiple products, each containing an A sativa–derived ingredient (Table 5), were not ocular irritants (Table 6). 89 The concentrations of A sativa–derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which ranged up to 43.3%. In vitro testing was conducted before these finished products were administered to humans. Irritation was determined by the measurement of lacrimation, stinging, and bulbar and palpebral redness. This information was presented in aggregate and the individual studies on the individual ingredient-containing products were not provided.
Colloidal oatmeal
In 2 use studies of a face and eye cleansing lotion containing Avena Sativa Colloidal Oatmeal (concentration not provided), the products caused little or no ocular irritation (Table 8). 92
In Vitro
Avena Sativa (Oat) Leaf/Stem Extract
In a human corneal epithelium (HCE) test, Avena Sativa (Oat) Leaf/Stem Extract was not predicted to be an irritant at 10% and 100%. 51 Negligible cytotoxicity was observed in a neutral red uptake assay. The extract (100%) was predicted to be slightly irritating in a Hen’s egg test–chorioallantoic membrane (HET-CAM) test.
Avena Sativa (Oat) Sprout Oil
In an HCE test, Avena Sativa (Oat) Sprout Oil was not predicted to be an irritant at 10% and 100%. 51 Negligible cytotoxicity was observed in a neutral red uptake assay. The extract (100%) was predicted to be slightly irritating in a HET-CAM test.
Type I Hypersensitivity
The binding of IgE in the sera of 40 adult atopic dermatitis patients (35 with severe, chronic atopic dermatitis, 4 with urticaria, and 1 with rhinitis) to proteins from oats (species and source not specified) and other grains in immunoblotting experiments was evaluated. 111 The sera of 35 of the 40 patients tested positive for IgE binding to oat proteins in the radioallergosorbent test (RAST). Four nonatopic subjects served as controls.
The authors prepared an acidic extract and a neutral extract from milled oats (“oat flour” or, essentially, colloidal oatmeal) and other milled grains, then, for each grain, mixed equal amounts of the acidic extract and the neutral extract for immunoblotting. They separated the components of the mixed extract of each grain by sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred the resultant protein bands to nitrocellulose sheets. The sera of 33 of the 40 patients bound to one or more of 10 protein bands of the oat extract mixture, including a 66 kDa protein, designated by the authors as the major allergen, and a 23 kDa and a 42 kDa protein, designated as “intermediate allergens.” The remaining 7 proteins were designated minor allergens. The sera of the 5 patients with negative RAST results tested positive in the immunoblotting experiment, and the sera of the 7 patients with negative immunoblotting results were positive in the RAST. The oat allergens appeared to cross-react only weakly with the wheat, rye, and barley allergens in this experiment. The authors stated that their results reveal the potential for proteins from oats and other grains to induce IgE-mediated type 1 immediate hypersensitivity reactions in adult atopic dermatitis patients. However, establishing a relationship between exposures to these substances and clinical allergic responses would require controlled elimination diet and challenge studies and characterization of the stability of the potential allergens after heating and in the gastrointestinal tract. 111
The same authors examined the potential for IgA and IgG from the same 40 adult atopic dermatitis patients to bind to the components of the protein extracts of the same grains, including oats. 112 They found that the immunoblotting binding patterns of IgA and IgG in the sera of the patients were indistinguishable from the binding patterns of these antibodies in the sera of the nonatopic controls, in contrast to the binding patterns of IgE, which were clearly different for the atopic patients compared to the nonatopic controls.
In a review of oat and wheat contact allergens, the authors note that different results among the studies of sensitization and contact dermatitis may be due to several factors such as study population, type of allergy tests, and type and concentration of allergens. 113 Although prick tests and serologic tests for antigen-specific IgE to oat are useful in detecting immediate reactions such as contact urticaria, patch testing may detect delayed reactions manifesting as contact dermatitis or flares of atopic dermatitis. Patch testing with oat proteins and extracts should be performed more frequently, especially in atopic children. It may help identify cutaneous sensitization and contact dermatitis, which may be the cause of flares in patients with atopic dermatitis.
Studies in the CIR report on hydrolyzed wheat protein showed that hydrolysates with weight average MW of approximately 3,000 or less exhibit no potential to elicit hypersensitivity reactions in sensitized individuals, in contrast to hydrolysates with weight average MW >10,000. 114 -116 Substantial experimental results support the theory that a polypeptide must be at least 30 amino acids long (ie, MW approximately 3,570, assuming an average of 119 Da/amino acid) to have the 2 IgE-binding epitopes needed to elicit type 1 hypersensitivity reactions.
The manufacturing process of personal care products may function like cooking in that it denatures the protein secondary structure to the point that the allergen loses the capacity to bind IgE and cause a type I response. However, T cells can react to short peptide sequences and may still elicit a type IV response even to finished products. 117 This means that type IV sensitivity may not be recognized when screening patients selected for antigen-specific IgE with skin prick tests or serologic tests.
It is possible for there to be proteins in the oils. It has been demonstrated that there are allergenic proteins in crude and refined peanut oil. 118 These proteins are the same size as 2 allergens previously described in peanut protein extracts.
Case Studies
A 4-month old infant with atopic dermatitis and allergy to cow’s milk tested positive in patch tests (++) for sensitization to oats (species not specified) and exhibited a sensitization to wheat, which the child had never ingested. 119 The authors suggested that, although sensitization to wheat in utero could not be eliminated, most likely the infant developed a cross-sensitization to wheat during exposure to a cream containing oats. At 1 year old, the child had results for the patch test to wheat identical to the results at 4 months of age and remained on an eviction diet.
Three children (14 months, 2 years, and 14 years of age) with atopic dermatitis had positive patch tests for oatmeal extract (species not specified). 109 The children all had histories of bathing with a product that contained an oatmeal extract. The eczema worsened after such baths. None of the subjects had a history of consuming oats.
A 3-year-old girl presented with an atopic dermatitis event on her arm and hands after using a moisturizer cream containing the young A sativa plant extract. 120 Serum IgE levels were elevated and a standard prick test was positive for Dermatophagoides farina and Dermatophagoides pteronyssinus. The subject had a family and personal history of other atopic maladies such as hay fever and rhinitis. Standard patch testing was positive for the cream at days 2 and 3 (++, ++). She was patch tested further with the ingredients of the cream (provided by the manufacturer) and was positive for the plant extract at days 2 and 3 (++, ++), but not for the zinc oxide and petrolatum. The atopic dermatitis did not reoccur when she no longer used the product.
A 7-year-old girl presented with swollen lesions where an oat cream had been applied after bathing. 108 The lesions appeared 15 minutes after application. She had a history of IgE-mediated allergic rhinoconjunctivitis, allergic asthma, and atopic dermatitis syndrome from the age of 3. The lesions were only on the application sites and resolved in less than 1 hour without treatment. Skin tests were positive for grass, rice, and oat pollens and were negative for the other pneumoallergens and foods. An open patch test was positive, and swollen lesions were apparent on the right forearm 10 minutes after the cream was administered, which resolved 30 minutes after administration of oral cetirizine. The oat-specific IgE assay was positive (0.76 kU/L) and negative for the other cereals. The girl ate foods containing oats with no adverse effects.
A 33-year-old female presented with a persistent rash that had linear streaks of eczema, mostly on the forearm, the sides of her face and neck, and less so on her waist and ankles. 121 The rash started 3 weeks after beginning a job weighing bird feeds that included oats. Patch test of the seeds had a ++ reaction to crushed oats at 48 hours and + reaction at 96 hours. She also had a ++ reaction to bran at 96 hours. The rash resolved when the subject avoided working with oats and bran. The rash reoccurred when she measured out oats and bran on 2 subsequent occasions.
A 33-year-old woman presented with atopic eczema and allergic rhinoconjunctivitis. 122 She had a history of type 1 hypersensitivity reactions to dust mites, cats, dogs, malassezia, nuts, shrimp, lobster, and asparagus. She had used a moisturizer that contained A sativa extract for 1 year. The reactions began to appear approximately 6 months after she began using the moisturizer. The reactions faded a few hours after application. The subject noted that she experienced itching and swelling of the lips and pruritic, erythematous papules, and patchy lesions on her trunk after eating breads containing oatmeal.
The patch test of the moisturizer was negative, but the prick test was positive. Her total serum IgE was slightly elevated. Further analysis of her serum revealed immunoreactivity to a “casual” A sativa extract, but not another A sativa extract with the proteins removed. The sera of 3 other cereal-sensitized subjects were tested with 5 different A sativa extracts, 1 without proteins. Two subjects reacted to all of the extracts; the third did not react to any. 122
Summary
This is a safety assessment of 21 A sativa-derived cosmetic ingredients. These ingredients are reported to function as abrasives, antioxidants, skin conditioning agents, absorbents, and bulking agents. This safety assessment does not include colloidal oatmeal, as the definition does not restrict the species of oats used to A sativa. However, data from colloidal oatmeal that were confirmed to be derived from this species were included for read-across/inference purposes.
Multiple fungi and their toxins have been reported in the plant, seed, dried hay, and/or in processed oat cereals. Avena Sativa (Oat) Kernel Extract has the most reported uses, with 499 in cosmetic products. Avena Sativa (Oat) Kernel Flour has the highest reported use concentration of 84.4% in skin cleansing products; Avena Sativa (Oat) Kernel Extract has the highest reported leave-on use concentration of 25% in face and neck products.
Dermal, anti-inflammatory, and buffering effects have been attributed to A sativa. Increased proliferation was observed in dermal cells incubated in extract of the whole plant of A sativa. Dermal administration of a whole plant ethanol extract of A sativa increased wound healing activity in rats and mice. There were no adverse effects when products containing colloidal oatmeal were used on subjects with damaged skin.
Female rats subcutaneously injected with any of 3 A sativa hay extracts (0.15 mL) and estradiol had reduced uterine weights compared to rats injected with estradiol alone. Avena Sativa (Oat) Leaf/Stem Extract was not mutagenic with or without metabolic activation in an Ames test and a micronucleus test. Avena Sativa (Oat) Sprout Oil was not mutagenic with or without metabolic activation in a fluctuation Ames test and a micronucleus test.
In a series of cumulative irritation tests (total N = 1,717), it was concluded that multiple products containing various A sativa-derived ingredients were not irritants. The concentrations of A sativa-derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which ranged up to 43.3%.
Avena Sativa (Oat) Leaf/Stem Extract and Avena Sativa (Oat) Sprout Oil at 100% was rated as nonirritant in an RHE test. Creams containing an extract of the entire young A sativa plant were not irritating when administered to the intact and tape-stripped skin of human subjects for up to 5 days. In 12 use safety studies of various personal care products containing colloidal oatmeal (concentrations not specified), there were a low percentage of subjects (0%-10.9%) who had positive reactions, and it was concluded that these products had a low potential to cause irritation. An emollient containing an extract of young A sativa plants, in addition to topical corticosteroids, administered to 78 infant subjects with moderate to severe atopic dermatitis was mostly well tolerated with 3 mild, 3 moderate, and 2 severe adverse events.
In a series of human ocular tests, it was concluded that multiple products containing various A sativa–derived ingredients were not ocular irritants. In 2 use studies of a face and eye cleansing lotion containing colloidal oatmeal, there was little or no ocular irritation. There were no adverse effects reported in children with mild atopic dermatitis who used several baby products containing colloidal oatmeal for 12 weeks.
Avena Sativa (Oat) Leaf/Stem Extract and Avena Sativa (Oat) Sprout Extract were not predicted to be ocular irritants at 10% and 100%. Negligible cytotoxicity was observed in a neutral red uptake assay. The extracts at 100% were predicted to be slightly irritating in a HET-CAM test.
In an LLNA, the EC3 of Avena Sativa (Oat) Leaf/Stem Extract was 59%. Avena Sativa (Oat) Sprout Oil up to 100% did not induce delayed contact hypersensitivity when dermally administered to mice on 3 consecutive days.
A paste mask product containing 25% Avena sativa (Oat) Kernel Extract was not sensitizing in a double blind HRIPT. A face powder containing 1% Avena Sativa (Oat) Kernel Flour, a blush containing 1% Avena Sativa (Oat) Kernel Flour, and a body lotion containing 0.1% Avena Sativa (Oat) Kernel Flour were not sensitizing in HRIPTs.
The use of a cream and soap containing the extract of young A sativa plants (12%, and 3%, respectively) for 21 days did not result in hypersensitivity. In a patch test of children referred for allergy testing, 14.6% tested positive for a young plant extract of A sativa at 1%, 3%, or 5%. In a skin prick test of the same subjects, 19.2% had positive reactions to A sativa pollen. An HRIPT of a cream containing an extract of the entire A sativa plant (concentration not provided) was negative in 104 subjects. In HRIPTs performed on skin care products containing Avena Sativa Colloidal Oatmeal (concentration not provided), the products did not yield signs of sensitization. In a series of HRIPTs (total N = 5,725), it was concluded that multiple products containing various A sativa-derived ingredients were not sensitizing; the concentrations of A sativa-derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which ranged up to 43.3%.
The sera of 33 of the 40 patients tested positive for IgE binding to oat proteins in an RAST. The immunoblotting binding patterns of IgA and IgG in the sera of the patients were indistinguishable from the binding patterns of these antibodies in the sera of the nonatopic controls, in contrast to the binding patterns of IgE.
In a series of phototoxicity and photoallergy tests, it was concluded that multiple products containing various A sativa–derived ingredients were not phototoxic or photoallergenic; the concentrations of A sativa–derived ingredients ranged from 0.00002% to 1%, except for colloidal oatmeal which ranged up to 43.3%. In a guinea pig maximization assay, Avena Sativa (Oat) Leaf/Stem Extract was not a photoirritant at up to 70% but was a slight photosensitizer. Avena Sativa (Oat) Sprout Oil at 100% was not phototoxic in an RHE test. There are several reported cases of atopic dermatitis as a result of using products containing Avena Sativa ingredients.
Discussion
The Panel acknowledged that A sativa grains are safely used in both animal feed and human food, resulting in much larger systemic exposures than would be possible from cosmetic uses. Therefore, the Panel was not concerned about the systemic toxicity potential of most of these cosmetic ingredients.
The Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities. The Panel noted that aflatoxins have been detected in A sativa plants, seeds, dried hay, and/or in processed oat cereals. They recognized the US Department of Agriculture designation of ≤15 ppb as corresponding to “negative” aflatoxin content and concluded that aflatoxins will not be present at levels of toxicological concern in A sativa–derived ingredients.
Because final product formulations may contain multiple botanicals, each possibly containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be lead to sensitization or other toxic effects. For A sativa–derived ingredients, the Panel was concerned about the presence of quercetin in cosmetics, which has tested positive for genotoxicity in an Ames assay, consistently tested positive in in vitro tests of genotoxicity, and positive in some in vivo studies via IP injections in mice and rats. Quercetin, however, has also had negative results in oral genotoxicity studies using rats and mice. Therefore, when formulating products, manufacturers should avoid reaching levels of this plant constituent and any other constituent that may cause sensitization or other adverse health effects.
The Panel discussed the issue of incidental inhalation exposure from face and neck spray products containing up to 0.0025% Avena Sativa (Oat) Kernel Extract and pump hair sprays containing up to 0.001% Avena Sativa (Oat) Kernel Protein. There were no inhalation toxicity data available. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, these ingredients are not likely to cause any direct toxic effects in the upper respiratory tract, based on data that show that these ingredients are not irritants. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel considered other data available to characterize the potential for A sativa–derived ingredients to cause irritation, sensitization, and genotoxicity. They noted the lack of systemic toxicity due to the use of these ingredients as food for humans and feed for animals. They also noted little or no dermal irritation, sensitization, or ocular irritation and the absence of genotoxicity in Ames tests and micronucleus tests.
The Panel discussed the potential for these ingredients to cause type 1 reactions in individuals. In the previous CIR report of hydrolyzed wheat protein, the Panel limited the size of proteins to 3,500 or less. The data provided for this assessment indicate that the ingredients in this report do not have the properties required to induce type 1 hypersensitivity; thus, the Panel concluded that these products had a low potential to cause sensitivity. Additionally, the Panel was not as concerned about the potential for protein in A sativa–derived ingredients to cause type I reactions, because, compared to wheat, soy, eggs, and nuts, oats are not a major food allergen.
There were no available data on the composition or concentration of use for Avena Sativa (Oat) Meristem Cell Extract. Because potential differences may exist between these meristem cells and the other ingredients for which data were provided, the Panel stated that composition and concentration of use data for Avena Sativa (Oat) Meristem Cell Extract were needed to support a conclusion of safety.
Conclusion
The CIR Expert Panel concluded that the following ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be nonsensitizing:
*Not in use. Were the ingredient in this group not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
However, the CIR Expert Panel concluded that the available data are insufficient to support to a conclusion of safety for Avena Sativa (Oat) Meristem Cell Extract.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
L. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, P. Snyder, and L. Gill contributed to conception and design, contributed to analysis and interpretation, and critically the revised manuscript. B. Heldreth contributed to analysis and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.