Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) assessed the safety of 20 sorbitan esters; this report included sorbitan esters that were reviewed in 1985 and 2002, as well as 3 previously unreviewed sorbitan esters (sorbitan undecylenate, sorbitan sesquicaprylate, and sorbitan palmate). Most of the sorbitan esters are reported to function in cosmetics as surfactant-emulsifying agents. The Panel reviewed the data from previous sorbitan ester reports, as well as additional data included in this report, to determine the safety of these ingredients. The Panel concluded that the sorbitan esters included in this safety assessment are safe in cosmetics in the present practices of use and concentration.
Keywords
Introduction
In 1985, the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) published a safety assessment of 7 sorbitan esters; based on the data presented in that assessment, the panel concluded that sorbitan stearate, sorbitan laurate, sorbitan sesquioleate, sorbitan oleate, sorbitan tristearate, sorbitan palmitate, and sorbitan trioleate were safe as cosmetic ingredients under [then] present conditions of concentration and use. 1 In 2002, the Panel considered the safety of an additional 10 sorbitan fatty acid esters and, based on new data as well as data from the 1985 review, concluded that the sorbitan fatty acid esters were safe for use in cosmetic ingredients under the [then] present practices of use. 2
The Panel also determined that the data from those safety assessments, together with the new data presented on the sorbitan esters, support the safety of 3 additional esters that had not yet been reviewed (indicated below by bolded text), and the Panel reopened the safety assessment to add these esters. The ingredients included in this rereview are:
Fatty sorbitan monoesters
Sorbitan caprylate (2002)
Sorbitan laurate (1985) Sorbitan palmitate (1985) Sorbitan isostearate (2002) Sorbitan oleate (1985) Sorbitan stearate (1985)
Fatty sorbitan sesquiesters
Sorbitan sesquiisostearate (2002) Sorbitan sesquioleate (1985) Sorbitan sesquistearate (2002)
Fatty sorbitan diesters
Sorbitan diisostearate (2002) Sorbitan dioleate (2002) Sorbitan distearate (2002)
Fatty sorbitan triesters
Sorbitan triisostearate (2002) Sorbitan trioleate (1985) Sorbitan tristearate (1985)
Mixed-chain sorbitan esters
Sorbitan cocoate (2002) Sorbitan olivate (2002)
All 20 of these ingredients share an identical sorbitan structural core and only vary by the fatty acid substituents. Most of the sorbitan esters are reported to function as a surfactant-emulsifying agent in cosmetic ingredients 3 (Table 1).
Definition, Structure, and Function.
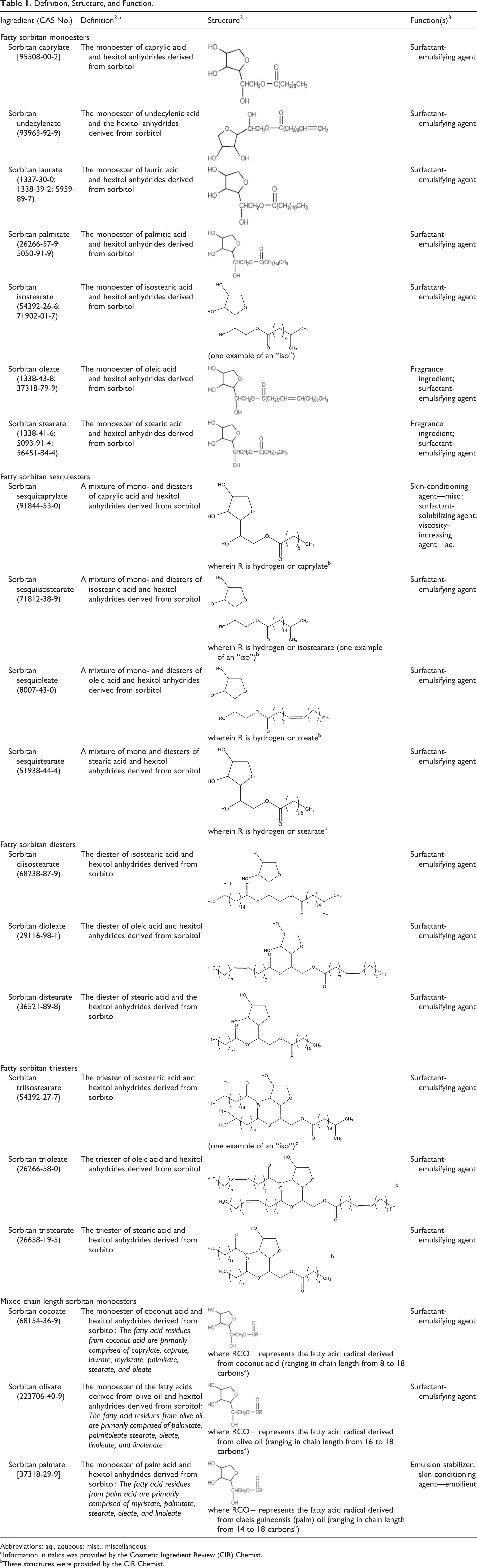
Abbreviations: aq., aqueous; misc., miscellaneous.
a Information in italics was provided by the Cosmetic Ingredient Review (CIR) Chemist.
b These structures were provided by the CIR Chemist.
Much of the new data included in this safety assessment were found on the European Chemicals Agency (ECHA) web site. 4 The ECHA web site provides summaries of information generated by industry, and it is those summary data that are reported in this safety assessment when ECHA is cited.
Chemistry
Definition and Structure
The sorbitan esters are mono-, di-, and triesters, and mixtures thereof, of fatty acids and 1,4-sorbitan. Generally, these ingredients can be depicted using a 5-membered ring shown as the tetrahydrofuran form (Figure 1). However, some of the 6-membered ring, tetrahydropyran, may also be present.
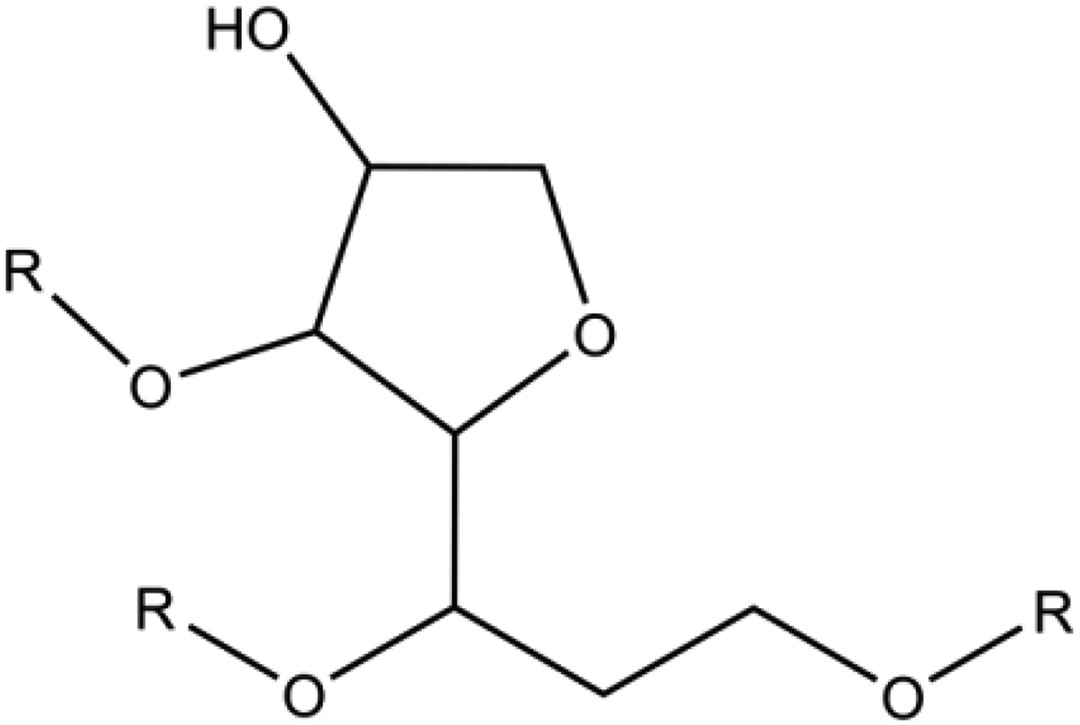
Sorbitan esters, wherein R represents either a fatty acid residue or hydrogen.
The ingredients in this safety assessment are the reaction products of fatty acids, ranging from 8 carbons in length (ie, sorbitan caprylate) to 18 (eg, sorbitan stearate), with hexitol anhydrides derived from sorbitol (eg, sorbitan tristearate; Figure 2). “Sorbitan” is a generic name for anhydrides (ie, cyclic ether tetrahydric alcohols) derived from sorbitol by the removal of 1 molecule of water with concomitant cyclization.
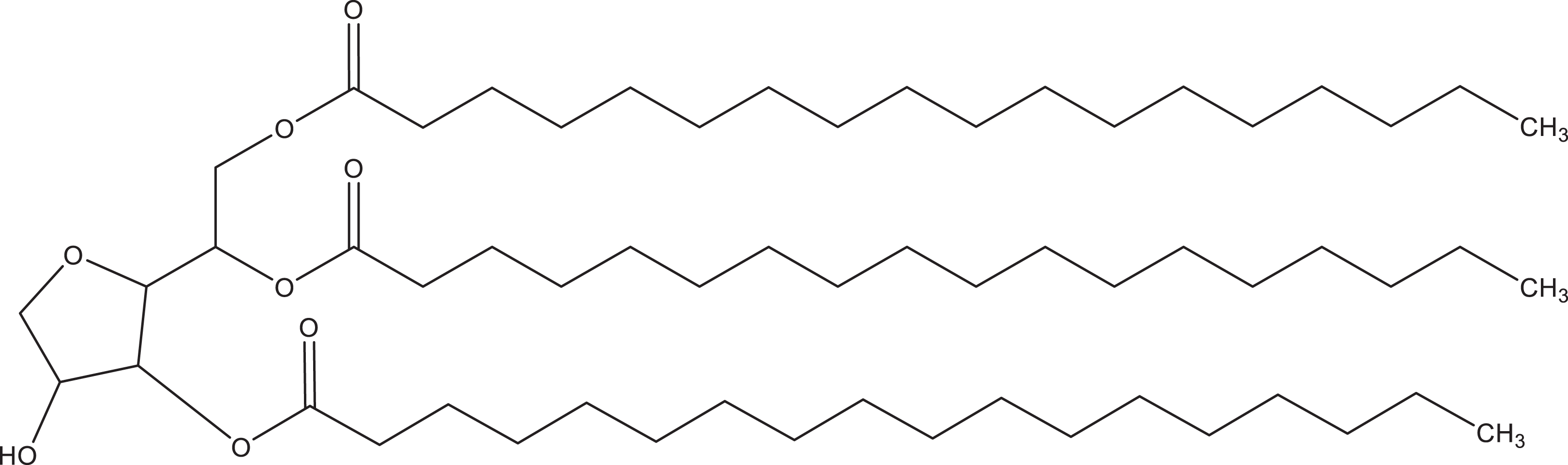
Sorbitan tristearate.
Chemical and Physical Properties
Sorbitan esters are waxy solids or viscous liquids soluble in inorganic solvents (Table 2). 1 Sorbitan esters are stable at pH ranging from 2 to 12. Hydrolysis of sorbitan fatty acid esters can occur in the presence of water at excessively high or low pH. Sorbitol, the compound from which the hexitol anhydrides of the sorbitan esters are derived, is a crystalline, hexahydric alcohol. 5
Methods of Manufacture
Sorbitan esters are manufactured by combining sorbitol with the appropriate fatty acid at elevated temperatures. 1
Composition and Impurities
Impurities such as free acid or alcohol, arsenic (3 ppm), lead (10 ppm), and water may be found in sorbitan fatty acid esters. 1 Pyranyl isomers of these sorbitan esters may be present.
Use
Cosmetic
Most of the sorbitan esters are reported to function in cosmetics as surfactant-emulsifying agents (Table 1). 3 In aqueous (aq.) formulations, the emulsifying agent function is a result of the classic surfactant structural combination of a polar head group (the sorbitan core) and apolar fatty chain(s).
The US Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetic formulations as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2014 6 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) 7 indicate that 13 of the 20 sorbitan esters named in this safety assessment are currently used in cosmetic formulations. Sorbitan stearate has the most reported uses, 968, followed by sorbitan isostearate, 401 reported uses, and several of the sorbitan esters have a few hundred uses (Table 3). The results of the concentration of use survey indicate that the sorbitan esters are used at less than 10% in cosmetic formulations; sorbitan triisostearate has the highest reported use concentration, 9.1% in rouges. Use concentration data were reported for sorbitan caprylate, but no uses were received in the VCRP; it should be presumed that there is at least one use in every category for which a concentration is reported. The ingredients not in use according to the VCRP and industry survey are listed in Table 4.
Physical and Chemical Properties.

Current and Historical Frequency and Concentration of Use According to Duration and Exposure.
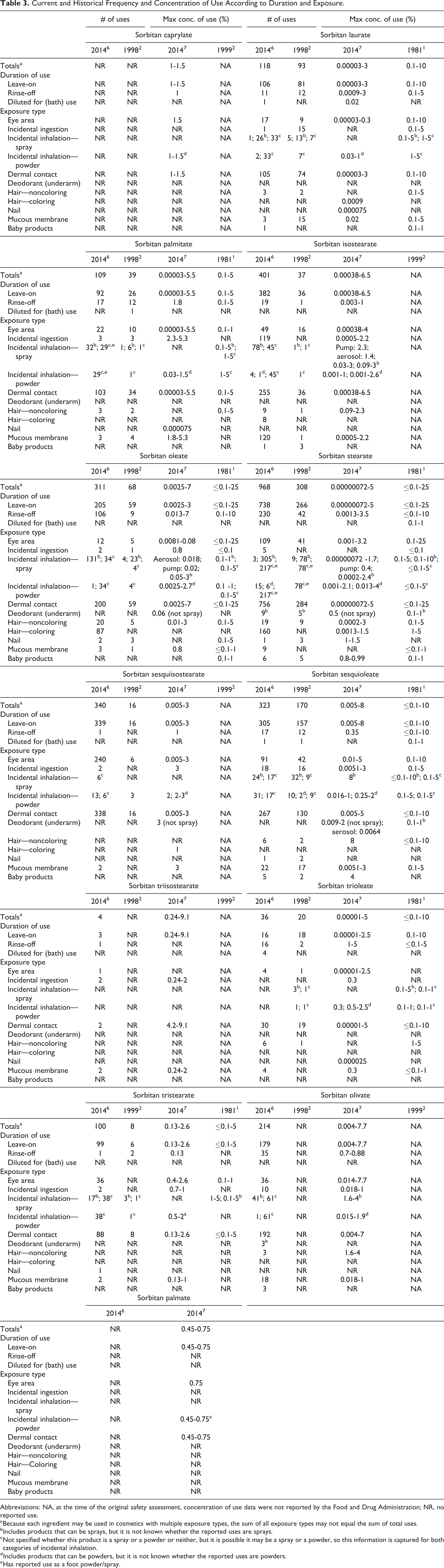
Abbreviations: NA, at the time of the original safety assessment, concentration of use data were not reported by the Food and Drug Administration; NR, no reported use.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b Includes products that can be sprays, but it is not known whether the reported uses are sprays.
c Not specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
d Includes products that can be powders, but it is not known whether the reported uses are powders.
e Has reported use as a foot powder/spray.
Ingredients Not Reported to Be Used.

Both historical and current use data are provided in Table 3. Concentration of use data were provided in the 1985 report, but these data were not available for the 2002 assessment; however, the 2002 assessment stated that the expected concentration of use was up to 20% in cosmetics. Additionally, it should be noted that frequency of use data for the 7 sorbitan esters included in the 1985 report were updated in the 2002 report; the updated information is included in Table 3. Although the frequency of use of these ingredients has increased, the concentration of use has not.
The sorbitan esters have many uses in the eye area; the highest concentration of use reported for eye products is 7.7% sorbitan olivate in mascara. 7 Some sorbitan esters are reported to be used in baby products (eg, 0.99% sorbitan stearate in baby lotions, oils, and creams), in products applied to the mucous membranes, or in products that could possibly be ingested (eg, 5.3% sorbitan palmitate in lipsticks).
Additionally, some of the sorbitan esters are used in cosmetic sprays and could possibly be inhaled; for example, sorbitan isostearate is reported to be used at 2.3% in pump hair sprays. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared to pump sprays. 8,9 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 10,11 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 10 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. All of the sorbitan esters named in this report are listed in the European Union inventory of cosmetic ingredients. 12
Noncosmetic
As described in the 1985 safety assessment, sorbitan esters are direct and indirect food additives. 1 Additionally, several sorbitan esters are inactive ingredients in drugs. 13 These ingredients are used in drugs that are administered via most possible routes, including topical, transdermal, inhalation oral, and/or across mucous membranes (Table 5). The greatest maximum potency concentration used in topically administered drugs is 8% sorbitan stearate and in orally administered drugs is 15% sorbitan oleate. Additionally, sorbitan oleate, sorbitan stearate, and sorbitan tristearate are diluents in color additive mixtures for drug use exempt from certification (ingested drugs) (21CFR73.1001).
Inactive Ingredient in Drugs.
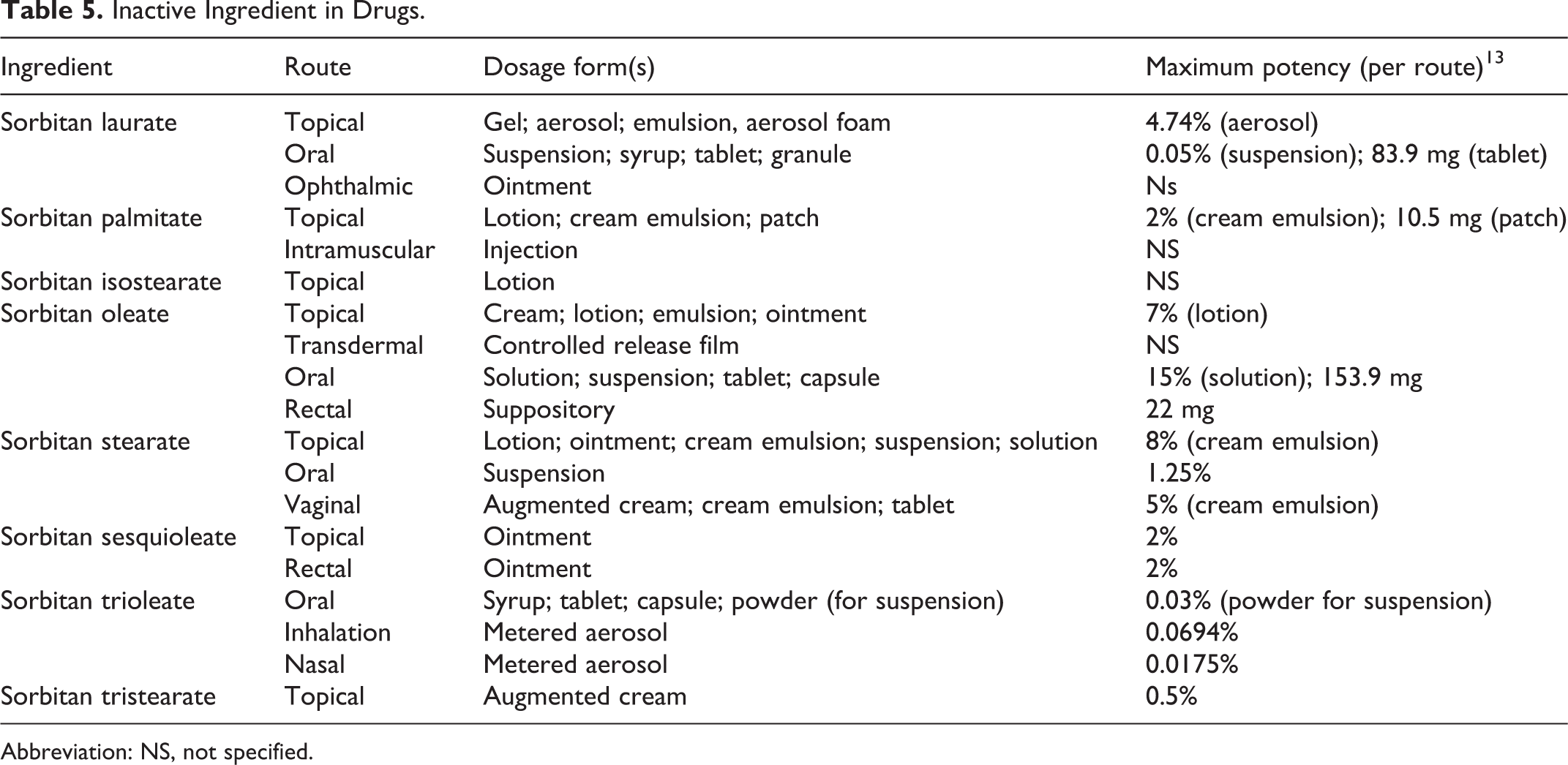
Abbreviation: NS, not specified.
Sorbitan sesquioleate is used as an emulsifier in fragrance mix I, a mixture used in patch testing. 14 Sorbitan laurate and sorbitan palmitate are used as emulsifiers in niosomes (nonionic surfactant-based vesicles). 15,16 Niosomes are microscopic vesicles composed of nonionic surface active agent bilayers, and the intended use of these vesicles is as a drug delivery system.
Toxicokinetics
Sorbitan stearate is hydrolyzed to stearic acid and anhydrides of sorbitol when ingested. 1 Approximately 90% of the sorbitan stearate is absorbed and hydrolyzed when fed to rats in oil solution, and 50% is absorbed and hydrolyzed when given as a water emulsion. Sorbitan stearate does not accumulate (<0.5%) to any appreciable amount in the fat stores of the rat body. Prolonged feeding (8 weeks) of sorbitan stearate to rats did not affect growth, and other studies indicated that sorbitan stearate had nutritive value for rats and dogs.
Toxicological Studies
The results of oral toxicity studies of sorbitan fatty acid esters indicated that these sorbitans in low concentration were relatively nontoxic via ingestion. 1 The lowest rat lethal dose, 50% (LD50) in the 20 sorbitan ester studies reported was 31 g/kg for sorbitan stearate. In subchronic feeding experiments of sorbitan laurate in a variety of species (chickens, rats, monkeys, and hamsters), no toxic effects were noticed when the ester concentration in the feed was less than 10%. When the feed concentration of sorbitan laurate was ≥10%, growth depression, decreased organ weights, diarrhea, unkempt appearance, hepatic and renal abnormalities, and gastrointestinal tract irritation were generally observed. Subchronic feeding of sorbitan oleate to rats produced no abnormalities until the ester was at least 10% of the feed. At this concentration, the same types of abnormalities occurred as those observed in the sorbitan laurate–fed animals.
Chronic feeding studies have been conducted with 3 sorbitans. At a 5% dietary concentration, sorbitan laurate and sorbitan oleate had no adverse effect on rats over a 2-year period. Dogs fed 5% sorbitan stearate for 20 months had no compound-related changes. A feed concentration of ≥10% sorbitan stearate was required to produce depressed growth and hepatic and renal abnormalities. Mice appeared more sensitive to toxic effects of sorbitan stearate than rats. A 0.5% dietary concentration produced growth depression in male rats, and a 4% dietary concentration produced renal abnormalities as well.
Subchronic dermal studies of 2% sorbitan palmitate and 4% sorbitan palmitate were negative for any systemic toxicity. No systemic toxicity was observed with dermal application of 5% sorbitan trioleate for 93 days.
Three clinical assessments have evaluated the oral toxicity of sorbitan stearate. One acute dose of 20 g was administered to 5 subjects, 2 of whom had increased gastric motility. One subject had an increase in free gastric acidity, and all subjects had normal gastric juices. Nine patients were given 3 g sorbitan stearate twice daily for 28 days. Seven patients had normal gas patterns (determined radiographically), one had more, and one had less at the end of the observation period. Seven patients had no change in gall bladder function, the eighth had increased emptying time, and the ninth patient had fainter visualization. Normal radiographic intestinal patterns were observed for all 9 patients. In an additional study, 42 subjects ingested 6 g sorbitan stearate daily for 28 days. Eleven subjects had albumin in their urine at the end of the study, and 4 had glycosuria; however, 1 of the 4 patients with glycosuria was diabetic, and another had an abnormal glucose tolerance test. No significant changes were found in hemoglobin content, hematocrit, red cell count, or red cell fragility; other blood chemistry values were normal except in 1 patient who had slightly elevated total serum bilirubin.
The no-effect dose of sorbitan stearate was 7.5 g/kg/d using rats fed the ingredient for 2 years. 2 The acute oral LD50 of sorbitan sesquiisostearate was 25 mL/kg in a study using female ddY mice.
Single-Dose (Acute) Toxicity
The original safety assessments of the sorbitan esters included data that indicated that this group of ingredients was relatively nontoxic upon ingestion of a single dose. Additional oral studies in mice and rats on sorbitan laurate, sorbitan palmitate, sorbitan isostearate, sorbitan stearate, sorbitan sesquioleate, and sorbitan tristearate support those findings (Table 6). 17 -22 In 4-hour inhalation exposure studies, sorbitan laurate and sorbitan trioleate had an lethal concentration 50% (LC50) in rats that was ≥5 mg/L. 17,23
Acute Toxicity Studies.
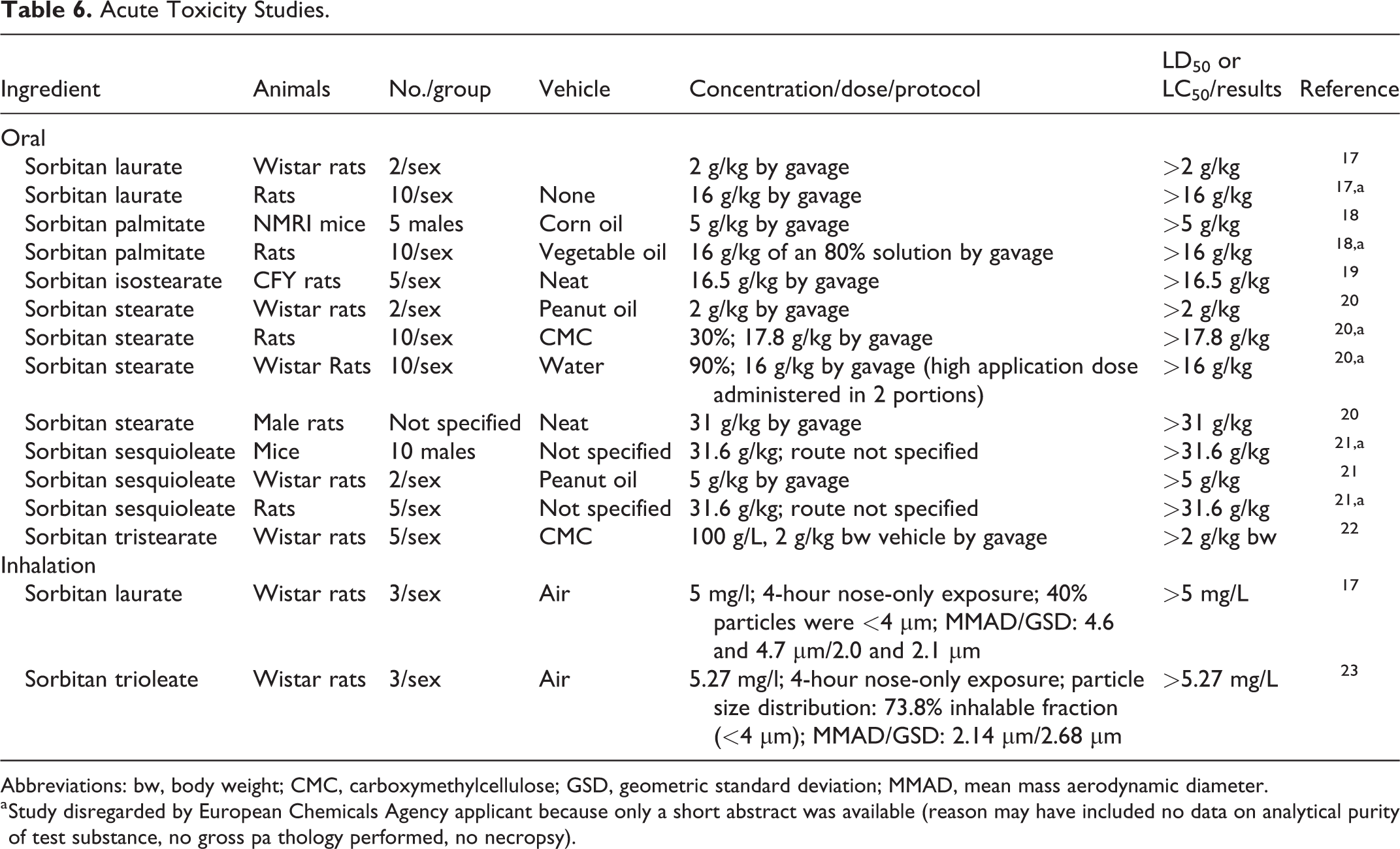
Abbreviations: bw, body weight; CMC, carboxymethylcellulose; GSD, geometric standard deviation; MMAD, mean mass aerodynamic diameter.
a Study disregarded by European Chemicals Agency applicant because only a short abstract was available (reason may have included no data on analytical purity of test substance, no gross pa thology performed, no necropsy).
Repeated Dose Toxicity
Oral
Male rats (number not specified) were fed a diet containing 5% sorbitan palmitate for 2 years. 18 The no-observable adverse effect level (NOAEL) was 5%. Thirty male rats were fed a diet with 5% sorbitan stearate, equivalent to 5,000 mg/kg body weight (bw)/d, for 2 years. 20 No effects on clinical signs, mortality, bw, feed consumption, hematology, clinical chemistry, gross or microscopic lesions, or pathology were reported. Sorbitan tristearate, 5% (equivalent to 5,000 mg/kg bw/day), was also administered in the diet to a group of 30 male rats for 2 years. 22 No effects on clinical signs or mortality, bw or weight gains, or gross or microscopic lesions were reported.
Reproductive and Developmental Toxicity
Fatty acids are normal components of diet for which (at the time of this report) no data were available concerning reproductive or developmental toxicity. 2 Sorbitol (2.5% to 10%) had no adverse effects on the reproduction of CD rats during a multigenerational feeding study. Hydrogenated starch hydrolysates (∼7% sorbitol) were not reproductive toxins at doses of 3,000 to 7,000 mg/kg/d for 2 years.
Gravid female Wistar rats, 20 per group, were dosed once daily by gavage with 0, 500, or 1,000 mg/kg bw/d sorbitan stearate on days 0 to 20 of gestation, and the animals were killed. 20 The NOAEL for maternal toxicity and for teratogenicity was 1,000 mg/kg bw/d. No test article–related embryotoxic results were reported.
Groups of 12 male and 12 female Sprague Dawley rats were dosed by gavage once daily with 0, 40, 200, or 1,000 mg/kg bw/d sorbitan stearate in water. 20 The females were dosed 2 weeks prior to mating until day 4 of lactation; the males were dosed for 42 days. No signs of toxicity; no effects on mortality, bw, or bw gains; and no gross or microscopic lesions were observed. Effects on feed consumption, hematology, and clinical chemistry were not examined.
Genotoxicity
Sorbitan stearate was not mutagenic in bacteria with or without metabolic activation systems. 1 Sorbitan stearate did not transform primary Syrian golden hamster embryo cells in vitro. Sorbitan oleate at a concentration of 0.01% inhibited in vitro DNA repair.
An unspecified sorbitan fatty acid ester had equivocal results in an Ames test and chromosome aberration assay using Chinese hamster fibroblasts. 2 In a feeding study using rats, the ester altered pharmacokinetic activity in the liver.
Sorbitan laurate, sorbitan palmitate, sorbitan stearate, sorbitan sesquicaprylate, sorbitan sesquioleate, and sorbitan trioleate were not mutagenic in the Ames test, with or without metabolic activation (Table 7). 17,18,20,21,23,24 Sorbitan laurate was not genotoxic to peripheral human lymphocytes in a chromosomal aberration assay, 17 but the results of a chromosomal aberration assay in Chinese hamster cells with sorbitan stearate were ambiguous. 20
Genotoxicity Studies.
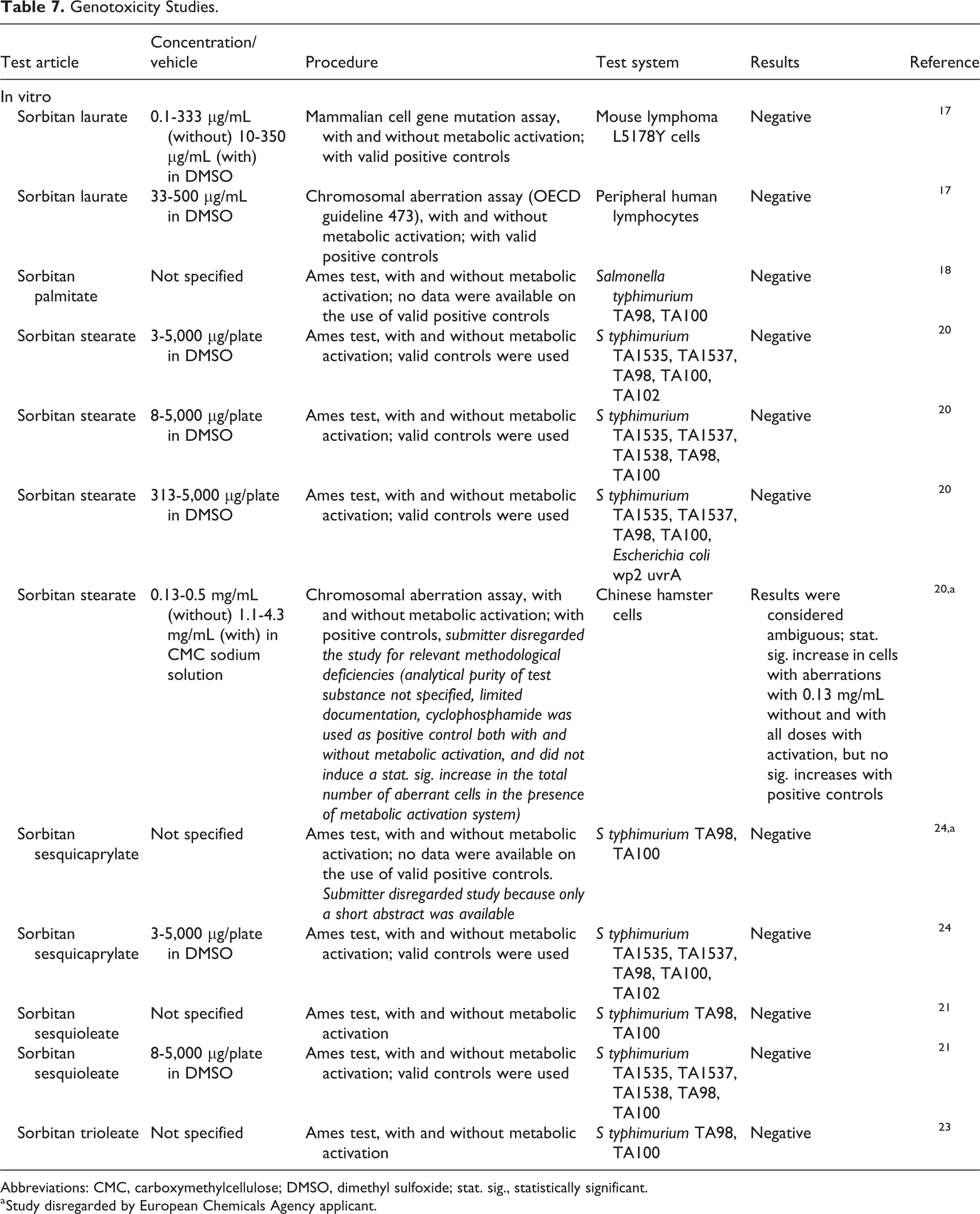
Abbreviations: CMC, carboxymethylcellulose; DMSO, dimethyl sulfoxide; stat. sig., statistically significant.
a Study disregarded by European Chemicals Agency applicant.
Carcinogenicity
Mice fed ≤4% sorbitan stearate for 80 weeks had no difference in tumor type and incidence when compared to control animals. 1 Sorbitan laurate was inactive as a carcinogen or tumor promoter when painted on mice skin for 70 weeks. However, in another study, sorbitan laurate was a tumor promoter when applied twice daily to mice skin after initiation by 7,12 dimethylbenz[a]anthracene (DMBA). In the same study, sorbitan oleate and sorbitan trioleate were inactive as tumor promoters. In undiluted form, sorbitan laurate and sorbitan trioleate were active as cocarcinogens on mouse skin when applied with DMBA (0.003%).
The sorbitan fatty acid esters had no antitumor activity against Ehrlich ascites tumors in mice. Sorbitan stearate was neither a mouse skin carcinogen nor tumor promoter. Sorbitan laurate and sorbitan trioleate were co-carcinogens in one mouse skin study, but the latter ester and sorbitan oleate were not tumor promoters in another.
Irritation and Sensitization
Dermal
Nonhuman
Numerous skin irritation studies in animals indicate that the sorbitans are minimal to mild irritants. 1 Skin irritation tests in which up to 60% sorbitan stearate in petrolatum and up to 100% sorbitan laurate, sorbitan oleate, and sorbitan trioleate were applied for 30 days produced no visible changes at 3 days but erythema and edema after 10 days. A formulation containing 4% sorbitan stearate applied for 7 days resulted in mild irritation. The dermal irritation in rabbits of a formulation containing 3% sorbitan sesquioleate was minimal. Undiluted sorbitan oleate was minimally irritating to rabbit skin when applied for 24 hours. Sorbitan palmitate, 50%, was not irritating to rabbit skin when applied for 24 hours. Sorbitan tristearate, 30%, was nonirritating when applied to the skin of rabbits for 24 hours.
Sorbitan isostearate, however, was a moderate irritant in one study using rabbits; intradermal injections of the ingredient caused mild to severe irritation in a study using guinea pigs. 2 Concentrations up to 100% sorbitan isostearate had low sensitization potential in guinea pigs. Sorbitan isostearate and sorbitan sesquiisostearate (10%) were non- to weak irritants to the intact and abraded skin of rabbits. The same concentrations caused weak cumulative irritation in a study using guinea pigs. In other studies, the ingredient did not produce significant irritation, sensitization, or comedone formation.
In studies published since the original assessments, several of the sorbitan esters were considered not irritating to rabbit skin (Table 8). Sorbitan palmitate, sorbitan isostearate, and sorbitan stearate were applied at concentrations of up to 100% using 24-hour occlusive patches. 18 -20 Sorbitan sesquioleate was applied for 24 hours using a semiocclusive patch, 21 40% aq. sorbitan tristearate was applied using a 24-hour occlusive patch, 22 and undiluted sorbitan sesquicaprylate and sorbitan trioleate were applied with a 4-hour semiocclusive patch. 23,24 Sorbitan sesquicaprylate was not a sensitizer in a guinea pig maximization test (GPMT); intradermal induction was conducted at a concentration of 5%, and topical induction and challenge were performed at 100%. 24 In a GPMT of sorbitan trioleate, intradermal induction (2%), topical induction (100%), and challenge (50% and 100%) resulted in slight to moderate erythema and slight edema were observed in test and control animals 23 ; at re-challenge (25 and 50%), slight erythema was observed in 2 test animals.
Dermal Irritation and Sensitization.
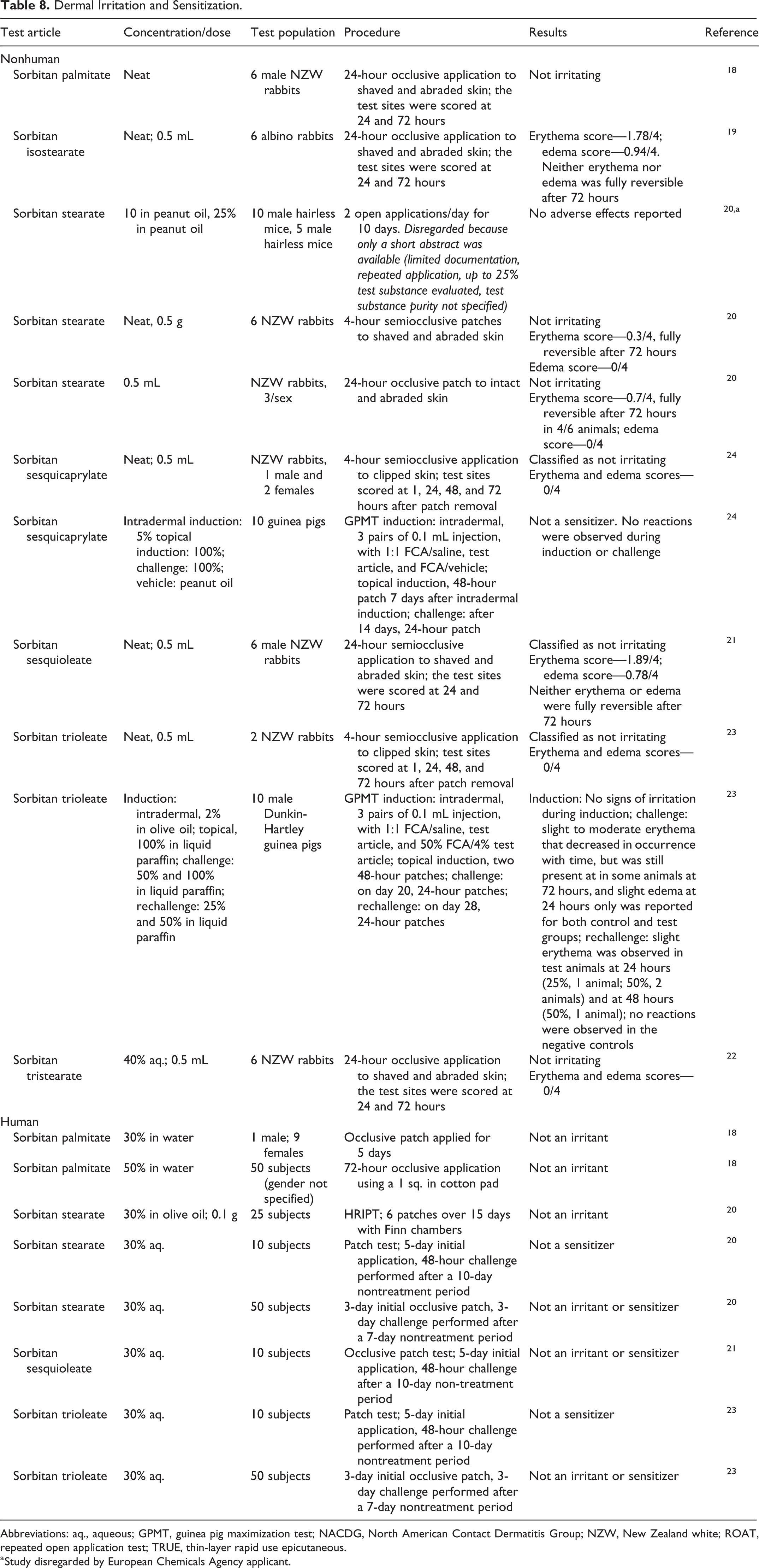
Abbreviations: aq., aqueous; GPMT, guinea pig maximization test; NACDG, North American Contact Dermatitis Group; NZW, New Zealand white; ROAT, repeated open application test; TRUE, thin-layer rapid use epicutaneous.
a Study disregarded by European Chemicals Agency applicant.
Human
The sorbitans are minimal to mild skin irritants in humans. 1 Results from 3 human repeated insult patch tests (HRIPTs; involving a total of 420 patients) indicated that sorbitan stearate at up to 4% is not a sensitizer. Products containing 2% to 4% of sorbitan stearate were mild irritants in 21-day cumulative irritation studies. A Schwartz prophetic patch test with 30% sorbitan laurate produced no irritation. Human skin tests for sensitivity to sorbitan sesquioleate were negative; 2 Schwartz prophetic patch tests with undiluted sorbitan sesquioleate produced no reactions; and in 5 HRIPTs, products containing 1% to 3% sorbitan sesquioleate were not sensitizers, but some patients did experience mild irritation. Several products containing 1.75% to 2.0% sorbitan oleate have been tested on humans. In four, 21-day cumulative irritation studies, the products tested were mildly irritating; in these tests using entire product formulations, the specific ingredient(s) causing irritation was not determined. Formulations containing up to 2% sorbitan oleate were nonsensitizers in several HRIPTs. No irritation was observed in a maximization test with a formulation containing 1.75% sorbitan oleate, but in a product usage test, a cream containing 1.75% sorbitan oleate produced mild irritation in 2 of 53 individuals. A Schwartz prophetic patch test with undiluted sorbitan tristearate produced no irritation in 201 patients. Formulations containing 4% sorbitan palmitate were found to be slightly irritating in humans in 21-day cumulative irritation tests (34 patients total). In a Shelanski/Jordan RIPT (206 patients), a skin care product containing 4% sorbitan palmitate was nonirritating and nonsensitizing. Several products containing 5% sorbitan trioleate were tested on humans; these products were slightly irritating in 21-day cumulative irritation tests, Shelanski/Jordan HRIPT, modified Schwartz-Peck predictive patch tests, and in a 4-week usage test.
Photosensitization assessments on products containing 2% sorbitan stearate or 2% sorbitan oleate classified both products as nonphototoxic and nonphotoallergenic. Sorbitan laurate, sorbitan sesquioleate, sorbitan palmitate, and sorbitan trioleate did not absorb radiation in the UV-A and UV-B range in UV spectral analysis.
In clinical studies, the sorbitan fatty acid esters were generally minimal to mild skin irritants in humans, and 20% sorbitan sesquioleate increased the incidence of irritation or sensitization reactions produced in 709 patients with suspected contact dermatitis. 2 Cross-sensitization was reported after 1,206 patients with eczema were treated with 5% to 20% sorbitans stearate, oleate, and sesquioleate, as well as 2 polysorbates. Sorbitan isostearate and sorbitan sesquiisostearate (10%) were nonirritating in a 24-hour occlusive patch test using 56 patients.
In clinical testing published since the original assessments, 30% sorbitan palmitate, 30% sorbitan stearate, 30% sorbitan sesquioleate, and 30% sorbitan trioleate were not irritants or sensitizers when applied for up to 5 days, followed by a challenge application after 7 to 10 days (Table 8). 18,20,21,23 Sorbitan palmitate, 50%, also was not an irritant after a 72-hour occlusive application. 18
Sorbitan sesquioleate is an emulsifier at a concentration of 5% in fragrance mix I, a mixture used as part of a patch test series. 14 Some researchers have stated that evidence has suggested that sensitization to sorbitan sesquioleate has been increasing, and the researchers have hypothesized that the increase may be due to an increased use of sorbitan sesquioleate as an emulsifier in corticosteroids. 14,25 Therefore, several researchers have postulated that a positive allergic reaction to a test mixture actually could be due to the emulsifier and reiterated the importance of patch testing the individual components of the mixture, in addition to the test mixture itself, to properly identify the contact allergen. 14,25 -29
Case reports
Several case studies of patients with diseased skin have been described (Table 9). Positive reactions to sorbitan laurate (0.4% incidence), 30 sorbitan oleate (0.6% incidence with 5%; up to 2.7% incidence with 20%), 14,30 -33 sorbitan stearate (2.3% incidence with 5%), 32 and sorbitan sesquioleate (up to 9.8% incidence with 20%) were observed in provocative tests in patients with contact allergy and contact dermatitis. 14,31 -34
Case Studies of Patients With Diseased Skin.
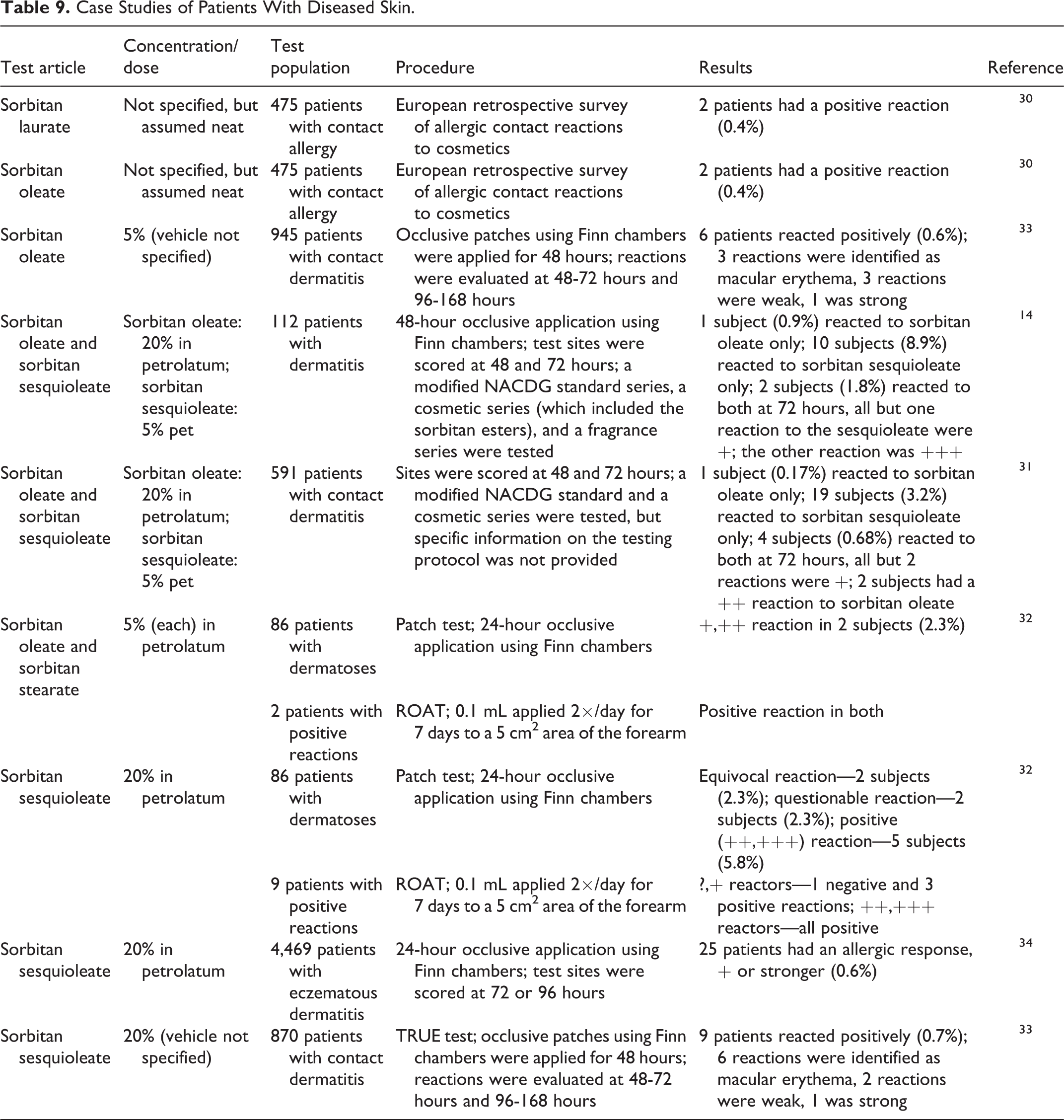
Case reports of contact dermatitis to sorbitan laurate, sorbitan oleate, and sorbitan sesquioleate have been described. For example, reactions to 2% 35 or 5% aq. sorbitan laurate 36 used as an emulsifier in a hydrocortisone cream have been reported; 3 of 23 patients with chronic leg ulcers had an allergic reaction to 20% sorbitan oleate in petrolatum 37 ; 3 patients with recalcitrant wounds from sensitization to a nonadhering dressing had positive reactions to sorbitan sesquioleate, a component of the dressing 38 ; and 6 pediatric patients with recalcitrant dermatitis had positive reactions to 20% sorbitan sesquioleate in petrolatum. 39
Effect on nonimmunologic immediate contact reactions
A 10 × 20 cm2 area of skin on the backs of 6 male and 6 female subjects was treated with 0.5 mL of sorbitan sesquioleate in petrolatum (20:80) 3 times per day for 2 days; a contralateral site was treated in a similar manner with petrolatum only. 40 On day 3, 10 µL of 31, 62, 125, 250, or 500 mM benzoic acid in petrolatum only or petrolatum containing 20% sorbitan sesquioleate was applied without occlusion to the pretreated areas on the back of each subject. Each site was scored visually for irritation 40 minutes after application of the benzoic acid. Additionally, cutaneous blood flow to the test sites was measured using laser Doppler flowmetry (LDF). When assessed with LDF, reactions to 125 or 250 mM benzoic acid in petrolatum only were stronger on the sites that were pretreated with sorbitan sesquioleate when compared to the areas pretreated with petrolatum only; these differences were not apparent using visual observation. No differences were observed with the lower concentrations of benzoic acid. However, a weaker reaction was observed, visually and with LDF, when 31 or 62 mM benzoic acid in petrolatum containing sorbitan sesquioleate was applied to skin pretreated with sorbitan sesquioleate and petrolatum alone, when compared to application of benzoic acid in petrolatum alone.
Ocular Irritation
Draize and modified Draize ocular irritation studies using rabbits were performed with all of the sorbitans in the original report. 1 One study of 30% sorbitan stearate was negative for ocular irritation, and a cream product containing 4% sodium stearate caused slight conjunctival irritation. Undiluted sorbitan sesquioleate produced no ocular irritation. A study with 30% sorbitan laurate and studies with up to undiluted sorbitan oleate, 40% sorbitan tristearate, and 30% sorbitan palmitate were negative for ocular irritation in the rabbit.
The sorbitan fatty acid esters were generally not ocular irritants. 2 In one study, sorbitan isostearate (10%) was nonirritating to the eyes of rabbits, whereas the same concentration of sorbitan sesquiisostearate was minimally irritating. In studies published since the original assessments, undiluted sorbitan stearate, sorbitan sesquicaprylate, sorbitan sesquioleate, and sorbitan trioleate were classified as not irritating to rabbit eyes following a single instillation without rinsing (Table 10). 20,21,23,24
Ocular Irritation Studies.
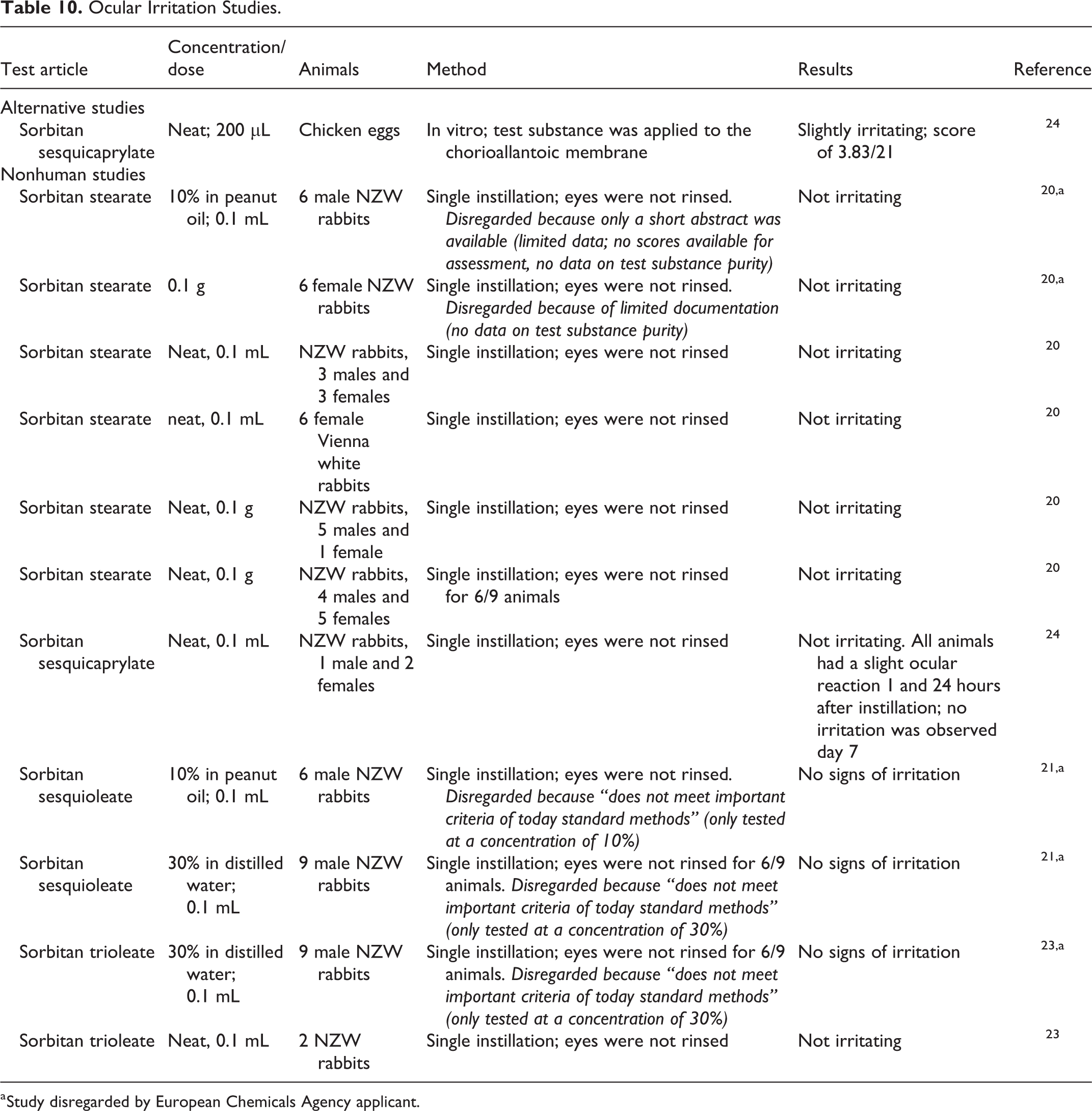
a Study disregarded by European Chemicals Agency applicant.
Summary
In 1985, the Panel reviewed the safety of the cosmetic use of 7 sorbitan esters, and in 2002, the Panel reviewed the safety of another 10 sorbitan esters; in both instances, the Panel concluded that the sorbitan fatty acid esters were safe as used in cosmetic ingredients. An additional 3 sorbitan esters have been identified and are reviewed in this assessment; all 20 of these esters share an identical sorbitan structural core and only vary by fatty acid substituents. Most of the sorbitan esters are reported to function as a surfactant-emulsifying agent in cosmetic ingredient
The VCRP data obtained from the FDA, and data received in response to surveys of the maximum reported use concentration by category that were conducted by the Council, indicate that 13 of the 20 sorbitan esters included in this safety assessment are used in cosmetic formulations. Sorbitan stearate has the most reported uses, 968, followed by sorbitan isostearate, 401 reported uses; several of the sorbitan esters have a few hundred uses. The sorbitan esters are used at less than 10% in cosmetic formulations; sorbitan triisostearate has the highest reported use concentration, 9.1% in rouges. The frequency of use of these ingredients has increased since the original safety assessments, but the concentration of use has not.
Sorbitan laurate, sorbitan palmitate, sorbitan isostearate, sorbitan stearate, sorbitan sesquioleate, and sorbitan tristearate were relatively nontoxic in mice and rats following a single oral exposure. In single-exposure inhalation studies, the LC50 in rats was ≥5 mg/L for sorbitan laurate and sorbitan trioleate. Administration of a diet containing 5% sorbitan palmitate, 5% sorbitan stearate, or 5% sorbitan tristearate to rats for 2 years did not result in any adverse effects. In oral reproductive toxicity studies in rats, the NOAEL for sorbitan stearate was 1,000 mg/kg bw/d, the highest dose administered.
Sorbitan laurate, sorbitan palmitate, sorbitan stearate, sorbitan sesquicaprylate, sorbitan sesquioleate, and sorbitan trioleate were not mutagenic in the Ames test, with or without metabolic activation. Sorbitan laurate was not genotoxic to peripheral human lymphocytes in a chromosomal aberration assay; the results of a chromosomal aberration assay in Chinese hamster cells with sorbitan stearate were ambiguous.
Several of the sorbitan esters were considered not irritating to rabbit skin; sorbitan palmitate, sorbitan isostearate, and sorbitan stearate were applied at concentrations of up to 100% using 24-hour occlusive patches, sorbitan sesquioleate was applied for 24 hours using a semiocclusive patch, 40% aq. sorbitan tristearate was applied under a 24-hour occlusive patch, and undiluted sorbitan sesquicaprylate and sorbitan trioleate were applied with a 4-hour semiocclusive patch. Sorbitan sesquicaprylate was not a sensitizer in a GPMT; intradermal induction was conducted at a concentration of 5%, and topical induction and challenge were performed at 100%. In a GPMT of sorbitan trioleate, concentration of 2% were used for intradermal induction, 100% for topical induction, 50% and 100% at challenge, and 25% and 50% at rechallenge. Slight to moderate erythema and slight edema were observed in test and control animals; at rechallenge, slight erythema was observed in only 2 test animals.
In clinical testing, sorbitan palmitate, sorbitan stearate, sorbitan sesquioleate, and sorbitan trioleate, all tested at 30%, were not irritants or sensitizers when applied for up to 5 days, followed by a challenge application after 7 to 10 days. Sorbitan palmitate, 50%, was also not an irritant after a 72-hour occlusive application. Case studies were performed in patients with diseased skin; reactions were observed in patients with contact allergy and contact dermatitis with sorbitan laurate (0.4% incidence), sorbitan oleate (0.6% incidence with 5%; up to 2.7% incidence with 20%), sorbitan stearate (2.3% incidence with 5%), and sorbitan sesquioleate (up to 9.8% incidence with 20%). Undiluted sorbitan stearate, sorbitan sesquicaprylate, sorbitan sesquioleate, and sorbitan trioleate were classified as not irritating to rabbit eyes following a single instillation without rinsing.
Discussion
In 1985, the Panel determined that 7 sorbitan esters were safe as used in cosmetic ingredients. In 2002, the Panel reviewed the safety of 10 additional sorbitan esters and issued an addendum to the 1985 report, concluding that the sorbitan fatty acid esters were safe as used in cosmetic ingredients. The Panel reaffirmed the safe as used conclusions of the 1985 and 2002 safety assessments. The Panel also determined that the data from those safety assessments, together with the new data presented on the sorbitan esters, support the safety of 3 additional esters that had not yet been reviewed, and the Panel reopened the safety assessment to add these esters.
Some of the components of the sorbitan esters are botanical ingredients. The Panel expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
The Panel stated that some of the sorbitan esters are used in products that could be incidentally inhaled, for example, sorbitan isostearate is reported to be used at 2.3% in pump hair sprays. Because single-dose, 4-hour inhalation studies of sorbitan laurate and sorbitan trioleate reported LC50 values > 5 mg/L, and because sorbitan trioleate has been reported to be used as an inactive ingredient in drugs at a maximum concentration of 0.0694% in a nasal aerosol product, the Panel was not concerned with the use of these ingredients in formulations that might be inhaled. The Panel also noted that in aerosol products, 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following 20 sorbitan esters are safe in cosmetics in the present practices of use and concentration described in this safety assessment. Sorbitan caprylate Sorbitan cocoate* Sorbitan diisostearate* Sorbitan dioleate* Sorbitan distearate* Sorbitan isostearate Sorbitan laurate Sorbitan oleate Sorbitan olivate Sorbitan palmate Sorbitan palmitate Sorbitan sesquicaprylate* Sorbitan sesquiisostearate Sorbitan sesquioleate Sorbitan sesquistearate* Sorbitan stearate Sorbitan triisostearate Sorbitan trioleate Sorbitan tristearate Sorbitan undecylenate*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
M. Fiume contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. W. Bergfeld contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. D. Belsito contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. R. Hill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. C. Klaassen contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. D. Liebler contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. J. Marks contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. R. Shank contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. T. Slaga contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. L. Gill contributed to analysis and interpretation and critically revised the manuscript. B. Heldreth contributed to analysis and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
