Abstract
Background:
Acute pancreatitis (AP) is associated with risks of morbidity and mortality. The incidence of AP recently increased compared to that traditionally reported in the literature.
Objective:
The purpose of this study was to evaluate the possible association between AP and drugs using the Japanese Adverse Drug Event Report (JADER) database, which is a spontaneous reporting database of adverse drug events.
Methods:
Adverse event reports submitted to the JADER database between 2004 and 2017 were analyzed. Disproportionality analysis was performed by calculating the reporting odds ratio (ROR) with 95% confidence intervals for signal detection.
Results:
A total of 3,443 reports (0.17% of all adverse events) were identified as drug-induced AP, in which 431 different drugs were involved. Acute pancreatitis was frequently reported in men (58.5%) in their 60s (19.1%); 40.6% developed AP within 4 weeks after the treatment. Among the most frequently reported drugs, signals were detected for prednisolone, ribavirin, sitagliptin, mesalazine, tacrolimus, and
Conclusion:
Most of the identified drugs were already known to induce AP, but the likelihood of the reporting of AP varied among the drugs. Our results should raise physicians’ awareness of drugs associated with AP, but further investigation of these medications is warranted.
Keywords
Introduction
Acute pancreatitis (AP) is characterized by acute inflammation of the pancreas and classified into interstitial edematous pancreatitis and necrotizing pancreatitis. 1 Common clinical symptoms are upper abdominal pain, nausea, vomiting, fever, and elevated serum pancreatic enzymes. Although AP is mild to moderate in severity in the majority of patients, some develop persistent organ failure. 2 The most frequent causes of AP are gallstones and excessive alcohol consumption. 3 Drugs are responsible for 5% of AP incidents 4 ; however, the incidence recently increased compared to that traditionally reported in the literature, 1%. 5 As a result, drug-induced AP has gained more attention, promoting health-care providers to become more concerned about drug-induced AP.
The vast majority of reported drug-induced AP cases may have an idiosyncratic character 6 ; the onset of drug-induced AP is thought to be immune mediated. 7,8 Early diagnosis and discontinuation of the offending drug can reduce complications. 9 Classically, there have been several studies on drugs inducing AP 5 but little information about drugs that are most frequently responsible for AP in a real-world setting.
Pharmacovigilance is a growing field, and it may be a useful tool to screen for potential associations among adverse drug events. Identifying the drugs most frequently implicated in the occurrence of AP could improve prescribing practice. However, few studies have performed surveillance of drugs inducing AP using a spontaneous reporting database. 10 In the present study, we conducted a comprehensive nationwide overview of the risk of drug-induced AP and identification of drugs most frequently implicated in the occurrence of AP using the Japanese Adverse Drug Event Report (JADER) database, a spontaneous reporting database of the Pharmaceuticals and Medical Devices Agency (PMDA).
Materials and Methods
We used the JADER database of the PMDA as a spontaneous reporting database, which was established in April 2004. 11 -15 The JADER database is freely obtainable from the web site of the PMDA, and we accessed the data set to which adverse event reports were submitted between April 2004 and January 2017. The data structure of the database consists of 4 data sets: patient demographics, drug information, adverse reactions, and medical history. In the adverse reaction table, the Medical Dictionary for Regulatory Activities (MedDRA) is used to codify the adverse reactions, which are indicated as “preferred term (PT).”
After we removed duplicated data, each table was then linked using the ID number. In each case, the contribution of the medication to adverse reactions was classified into 3 categories: “suspected medicine,” “interaction,” and “concomitant medicine.” A “suspected medicine” is defined as a pharmaceutical product with which an adverse reaction is suspected to be associated. When the reporter suspects an interaction, he or she reports it as an “interaction.” A “concomitant medicine” is defined as another pharmaceutical product used at the time of the adverse reaction.
We only extracted cases that were classified as “suspected medicine” and excluded patients with a history of chronic or autoimmune pancreatitis. We analyzed the reports of suspected drugs and adverse events, which we selected as “pancreatitis acute” in the PT coded in MedDRA (version 20). We compiled a cross-tabulation table based on 2 classifications: the presence or absence of AP and the presence or absence of the suspected medicine. Then, we calculated the reporting odds ratio (ROR). The ROR is rate of reporting a specific adverse reaction caused by a particular drug divided by the rate of the same adverse reactions caused by all other drugs present in the database. In addition, the ROR was frequently used with the spontaneous reporting database as an index of the relative risk of drug-associated adverse events. A signal 16 was considered to be present when the lower limit of the 95% confidence interval (CI) of the ROR was >1.
In this database, age, height, and weight information is indicated in the form of age in decades, height in centimeter-denominated ranges, and weight in kilogram-denominated ranges. Because these data are not continuous variables, we could not conduct multiple analyses using them. All analyses were performed with JMP Pro 12 (SAS Institute Inc, Cary, North Carolina).
Results
The total number of reported cases of AP associated with drugs was 3,443 (0.17% of all adverse events). The annual numbers of reports in 2004 to 2016 are shown in Table 1. Rates of death and nonrecovery from drug-induced AP varied widely from year to year and reached peaks of 12.6% and 18.4%, respectively. Table 2 shows characteristics of patients. Acute pancreatitis was frequently reported in men (58.5%) in their 60s (19.1%). As for the treatment period until AP, 40.6% of patients experienced AP within 4 weeks and 36.5% of reports did not specify an onset time. The outcomes of AP were distributed as follows: 5.7% (196 reports) of the patients died, 77.0% (2,650 reports) had recovered or were recovering, 5.2% (180 reports) had not recovered, 2.9% (100 reports) had after effects, and the outcome was unknown in the remaining 9.2% (317 reports).
Drug-Induced AP From 2004 to 2016.
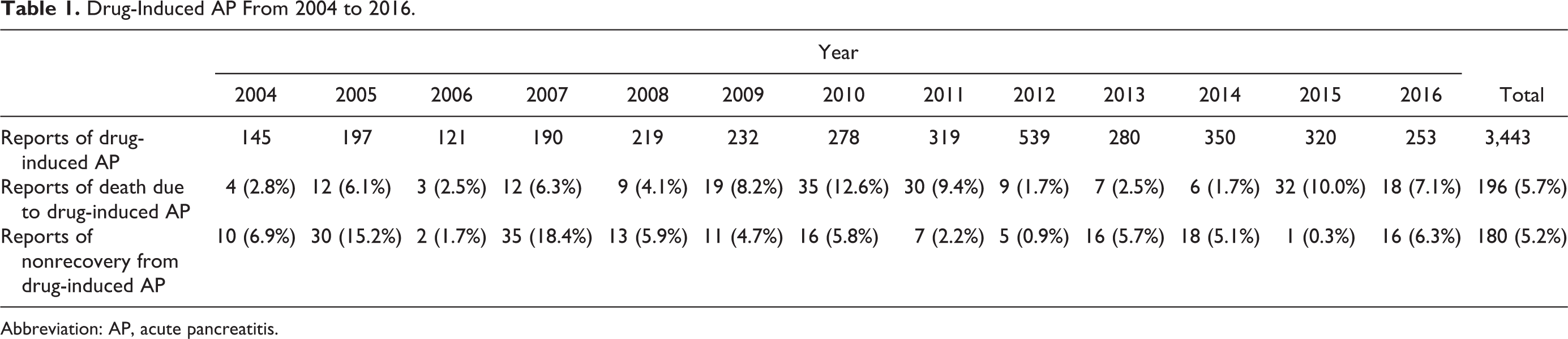
Abbreviation: AP, acute pancreatitis.
Characteristics of the Patients With Drug-Induced AP.

Abbreviations: AP, acute pancreatitis; N, number of reports.
In our analysis, 431 different drugs were “suspected” in cases of AP. Of those, we examined the 70 most frequently reported drugs because Nitsche et al
17
reported 62 drugs with a probable or definite association with AP. The most frequently reported drugs were prednisolone (180 reports), ribavirin (159 reports), sitagliptin (127 reports), mesalazine (127 reports), and tacrolimus (105 reports). Focusing on RORs, 39 drugs generated a signal, that is, the lower confidence limit of the ROR was >1. Of note, the association with AP was more noteworthy for
Most Frequently Reported Drugs Associated With AP.
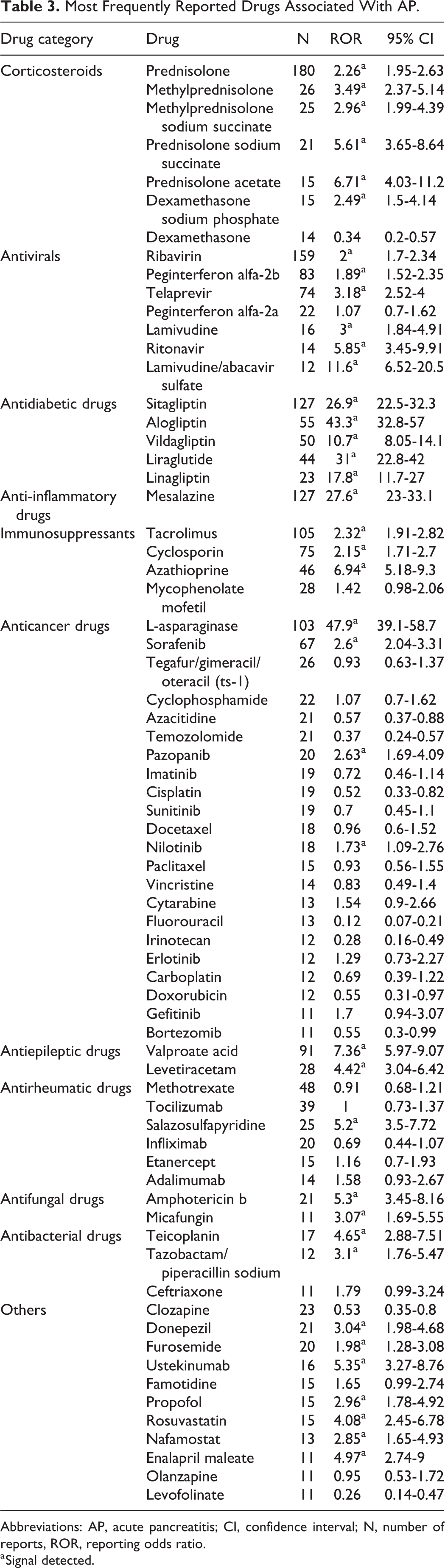
Abbreviations: AP, acute pancreatitis; CI, confidence interval; N, number of reports, ROR, reporting odds ratio.
a Signal detected.
Discussion
This is the first study to provide an apparent association between AP and several drugs and characteristics of patients with drug-induced AP, based on information from the JADER database. Death or nonrecovery from drug-induced AP was 10.9% of the total AP reports, being consistent with previous reports. 18 Regarding the characteristics of patients, AP tended to occur most frequently within 1 month of exposure and show a peak age of onset in patients in their 60s. The highest incidence was noted in patients in their 60s, which is consistent with a study that there is an association between increased risk of AP and polypharmacy. 19 Of note, among drugs with a signal, telaprevir, donepezil, and ustekinumab have not been reported as AP-inducing drugs.
In the study by Nango et al,
10
drug-induced AP was assessed using the US Food and Drug Administration’s adverse event reporting system, in which target drugs were limited to cortisol, cortisone, prednisolone, methylprednisolone, triamcinolone, dexamethasone, and betamethasone. In the present study, we assessed drugs that most frequently induced AP and identified various drugs. In addition, we found that the number of cases of drug-induced AP varied from a low of 145 in 2004 to a high of 539 in 2012. This is partly because several drugs were launched onto the market a few years before 2012: sitagliptin, clozapine, and nilotinib in 2009; alogliptin, vildagliptin, liraglutide, and levetiracetam in 2010; linagliptin, azacitidine, ustekinumab, and ritonavir in 2011; and pazopanib in 2012. Among drugs with a signal, telaprevir, donepezil, and ustekinumab have not been reported as AP-inducing drugs, and so the results might offer new and valuable insights. Focusing on RORs, the drugs with a high signal score included
The mechanisms of drug-induced AP include pancreatic duct constriction, cytotoxic and metabolic effects, the accumulation of a toxic metabolite or intermediary, and hypersensitivity reactions. 9 Mainly, intrinsic drug toxicity and idiosyncratic reactions treated with drugs are involved in the pathogenesis of drug-induced AP. The mechanism of intrinsic drug toxicity is dose dependent, develops in the short term, and is consistent from person to person regarding the frequency of development. Alternatively, idiosyncratic reactions are not dose dependent, are unpredictable, and the period of development depends on the person. Idiosyncratic reactions can be categorized into those secondary to hypersensitivity reactions and those caused by the accumulation of a toxic metabolite or an injurious intermediary substance. Hypersensitivity reactions tend to occur with a latency of within 1 month after exposure. When a patient is reexposed to a drug that caused hypersensitivity, the drug reaction is reproduced in a shorter time period than before. 7 In our study, the most frequent treatment period until AP was within 1 month. This is possibly due to hypersensitivity reactions or reexposure. Considering that hypersensitivity reactions are related to the development of drug-induced AP, we need to observe the condition.
Pharmacovigilance can be a useful tool to screen potential associations in drug-induced AP. Indeed, the safety of drugs was primarily assessed in nonclinical and clinical studies in the development stage; however, rare but serious adverse events often go undetected during the development phase because the selection criteria are limited in clinical trials. Therefore, unexpected serious adverse drug events are sometimes identified postmarketing. An analysis using the JADER database as a self-reporting database has advantages in that it can detect unknown or severe side effects.
This study had some limitations. First, as a consequence of the self-reporting database, the data occasionally contain typing errors. In addition, we cannot know whether the reports covered all the incidences, and it is difficult to completely remove the reporting bias. Second, this database lacks detailed clinical information such as on unknown etiology of underlying disease and the possible role of disease in the reported adverse event and on alcohol consumption. 27 Third, the ROR may incidentally decrease if another drug-specific reaction is reported more consistently; this dilutes the association because of the stronger association with other events. For example, methotrexate is known to cause AP but did not show a significant ROR in the present data set analysis. This might be because the adverse drug event most frequently reported to the pharmacovigilance network for this drug is interstitial lung disease, resulting in a very small proportion of AP events.
Conclusion
In conclusion, analysis of real-world data suggests that the peak age at onset was in the 60s, the most frequent treatment period until AP was within 1 month, and the most implicated drug categories were corticosteroids and antivirals. We identified that telaprevir, donepezil, and ustekinumab were associated with AP. Most of the identified drugs were already known to induce AP, but the likelihood of reporting AP varied among them. Our results should raise physicians’ awareness of drugs associated with AP, but further studies are needed to confirm the associations identified in this study.
Supplemental Material
Supplemental Material, DS1_IJT_10.1177_1091581819870717 - Pharmacovigilance Assessment of Drug-Induced Acute Pancreatitis Using a Spontaneous Reporting Database
Supplemental Material, DS1_IJT_10.1177_1091581819870717 for Pharmacovigilance Assessment of Drug-Induced Acute Pancreatitis Using a Spontaneous Reporting Database by Iku Niinomi, Keiko Hosohata, Saki Oyama, Ayaka Inada, Tomohito Wakabayashi and Kazunori Iwanaga in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS2_IJT_10.1177_1091581819870717 - Pharmacovigilance Assessment of Drug-Induced Acute Pancreatitis Using a Spontaneous Reporting Database
Supplemental Material, DS2_IJT_10.1177_1091581819870717 for Pharmacovigilance Assessment of Drug-Induced Acute Pancreatitis Using a Spontaneous Reporting Database by Iku Niinomi, Keiko Hosohata, Saki Oyama, Ayaka Inada, Tomohito Wakabayashi and Kazunori Iwanaga in International Journal of Toxicology
Footnotes
Author Contributions
Iku Niinomi contributed to analysis and drafted the manuscript. Saki Oyama contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. Keiko Hosohata contributed to conception and design, contributed to acquisition and interpretation, drafted the manuscript, and critically revised the manuscript. Ayaka Inada contributed to conception and design, contributed to interpretation, and drafted the manuscript. Tomohito Wakabayashi contributed to analysis and drafted the manuscript. Kazunori Iwanaga contributed to acquisition, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: K. Hosohata received research support from the Science Research Promotion Fund.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
