Abstract
Vedolizumab, a humanized monoclonal antibody approved for the treatment of adults with moderately to severely active ulcerative colitis or Crohn disease, targets α4β7 integrin and selectively blocks gut-specific lymphocyte trafficking. The potential effects of vedolizumab on development were assessed by standard preclinical toxicity studies in rabbits and cynomolgus monkeys. A single infusion of vedolizumab (0, 10, 30, or 100 mg/kg) was administered intravenously to pregnant rabbits on gestational day 7; rabbits were monitored to gestational day 29. Vedolizumab (0, 10, or 100 mg/kg) was administered intravenously every 2 weeks to pregnant cynomolgus monkeys beginning on gestational day 20 with the last dose on gestational day 132 (9 doses total). In rabbits, vedolizumab did not affect maternal net body weight or net gains, gravid uterine weights, or mean maternal food consumption, nor did it affect intrauterine growth or fetal survival. There were also no vedolizumab effects on embryo–fetal development compared to controls. In cynomolgus monkeys, there was no increase in prenatal loss/death or stillbirth and no maternal toxicity associated with vedolizumab. On day 28 postpartum, low levels of vedolizumab were detected in the breast milk of 3 of 11 monkeys in the 100 mg/kg group. No vedolizumab-related effects on the number of infants born, infant development, or animal hematology or clinical chemistry were noted. Administration of vedolizumab to pregnant rabbits and cynomolgus monkeys did not show any potential for maternal or developmental effects.
Introduction
Inflammatory bowel disease (IBD) is a chronic disabling condition with a prevalence of up to 0.5% of the general population in the Western world. 1 It is estimated that approximately 25% of female patients with IBD conceive after the diagnosis is made. 2 Although the course of the disease during pregnancy generally mirrors that of the nonpregnant IBD population, 3 active IBD in pregnant women has been associated with an increased risk of adverse pregnancy outcomes including increased rates of spontaneous abortion, preterm delivery, low birth weight, and severe preeclampsia, as well as increased likelihood of delivery by cesarean. 4 -7 Women with active disease at conception also have a higher risk of ongoing disease activity during pregnancy. 8 Maintaining adequate disease control during pregnancy is, therefore, essential to ensuring favorable maternal and neonatal outcomes. 2,9
Poor treatment compliance among pregnant women with IBD can stem from a lack of awareness of the harmful effects of IBD exacerbation during pregnancy and an overestimation of the harmful effects of medication. 10 In reality, active IBD is high risk to the mother and developing fetus and the benefit of disease treatment may outweigh the risks. 2,11 Biologics such as anti–tumor necrosis factor α (anti-TNFα) and anti-integrin agents are important treatment options for patients with IBD. Although many of these agents are known to cross the placenta at the end of the second trimester and after delivery, 12 -14 use of biologics for the management of IBD during pregnancy is considered low risk, 15,16 with no evidence of an increased risk of unfavorable pregnancy outcomes. 17,18 The ongoing prospective, multicenter Pregnancy in IBD and Neonatal Outcomes (PIANO) registry, with an enrollment of approximately 1,500 women in the United States, is providing insight into the safety of biologic medicines in pregnant women with IBD. 19 Analyses from PIANO have reported that the use of vedolizumab, a biologic, was not associated with an increase in adverse pregnancy outcomes, such as congenital abnormalities or abnormal infant growth and development, or infant infection rates. 20,21 Furthermore, in utero exposure to biologics did not have an impact on infant developmental milestones 22 or affect infant response to vaccines. 19 However, the long-term implications of in utero exposure to biologics, especially with respect to immune system development and function, are currently unknown. 15
Immunoglobulin G (IgG) is actively transported from mother to fetus across the placenta by the neonatal Fc receptor FcRn. This process begins at week 16 of gestation, but the majority of IgG is acquired by the fetus during the last 4 weeks of pregnancy. 23 Placental transfer of the IgG1 anti-TNFα agents adalimumab and infliximab is well-documented 12,14 ; however, certolizumab pegol, an IgG1 anti-TNFα agent which lacks an Fc region, is not actively transported across the placenta. 24 Placental transfer of natalizumab, a humanized IgG4 monoclonal antibody against the α4-integrin cell adhesion molecule, has also been reported. 13,15 Labels for adalimumab, infliximab, and certolizumab pegol state that women should inform their health-care provider if they are pregnant or plan on becoming pregnant because the agent will harm the unborn baby. 25 -27 The natalizumab label categorizes pregnancy as a specific population, indicating that use may cause fetal harm based on animal data. 28
Vedolizumab, a humanized IgG1 monoclonal antibody approved for the treatment of adults with moderately to severely active ulcerative colitis or Crohn disease, targets α4β7 integrin and selectively blocks gut-specific lymphocyte trafficking. 29,30 According to the drug prescribing information, vedolizumab should be used during pregnancy only if the benefits outweigh any potential risks to both the mother and the fetus. 29 The timing and mechanisms of placental transfer of vedolizumab are expected to be similar to that of other IgG1 antibodies, 31 and in a small number of vedolizumab-treated patients included in PIANO registry, placental transfer of vedolizumab has been confirmed. 32 Data for vedolizumab during pregnancy are currently limited. 31,33,34 The largest report comes from Mahadevan et al, which described pregnancy outcomes among 24 pregnancies in females with IBD and 15 pregnancies in partners of males who were exposed to vedolizumab in clinical trials, as well as 81 pregnancies from postmarketing safety data. 31 Although a single congenital abnormality was documented in a clinical trial, this was not deemed to be vedolizumab treatment related, and the data do not indicate safety concerns related to pregnancy outcomes with vedolizumab. 31
To further characterize the preclinical safety profile of vedolizumab, embryo–fetal and postnatal developmental studies were conducted to evaluate the potential of vedolizumab to induce maternal or developmental effects after gestational exposure in preclinical animal models. Pregnant rabbits were administered vedolizumab to assess maternal toxicity and fetal development. Cynomolgus monkeys were treated from gestational day (GD) 20 to GD 140 to evaluate the effects of vedolizumab on pregnancy, parturition, and lactation in mothers, as well on the survival, growth, and postnatal development of the offspring.
Materials and Methods
Animal protocols for both studies were reviewed and approved by an institutional review panel (Institutional Animal Care and Use Committee).
Rabbit Study
This study was conducted in accordance with the International Conference on Harmonisation Tripartite Guideline on Detection of Toxicity to Reproduction for Medicinal Products, Federal Register (September 22, 1994; Section 4.1.3) and the appropriate provisions of the United States Food and Drug Administration (FDA) Good Laboratory Practices (GLP) regulations (21 CFR Part 58).
Timed-mated, female, young adult to adult (5-9 months of age at study initiation), New Zealand white rabbits (n = 25 per group) were obtained from Covance Research Products, Inc (Kalamazoo, Michigan) and housed in WIL Research Laboratories (Ashland, Ohio), a facility that is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. Vedolizumab was administered as a single intravenous (IV) infusion at doses of 0 mg/kg (saline control), 10 mg/kg (human equivalent dose [HED]: 3.2 mg/kg), 30 mg/kg (HED: 9.7 mg/kg), or 100 mg/kg (HED: 32.3 mg/kg; Supplemental Table 1) via the marginal ear vein on GD 7; previous preclinical data demonstrated that a single infusion at the proposed doses resulted in clinically relevant vedolizumab exposure for up to 14 days. An infusion time of 60 minutes was chosen to minimize stress from prolonged restraint while optimizing the flow rate needed for infusion into the marginal ear vein. Additional rabbits were used for the toxicokinetic (TK) phase (n = 3 per group). Blood samples for quantification of analytes of interest were collected prior to dosing (0 hour), at the end of the infusion, and at 48, 168, and 337 hours after the end of the infusion.
Maternal body weight (BW) was recorded on GD 0, 4, 6 to 21 daily, 24, and 29; gravid uterine weight, net BW, and net BW change data were collected on GD 29; and food consumption was recorded on GD 4 to 29. Vedolizumab in serum was quantified via enzyme-linked immunosorbent assay (ELISA; mean peak serum vedolizumab concentration [Cmax], time to maximum concentration [Tmax], and area under the concentration–time curve from 0 to 337 hours after infusion [AUC0-337h]). On GD 7, at predose and 312 hours postdose, approximately 0.75 mL of whole blood was collected from the marginal ear vein for the measurement of rabbit antihuman (anti-vedolizumab) antibodies (RAHA). The blood samples for serum concentrations of vedolizumab and RAHA were processed into serum at room temperature and held on dry ice until stored at approximately −70°C. Serum samples were shipped on dry ice from WIL Research Laboratories, LLC, to Millennium Pharmaceuticals, Inc (Cambridge, Massachusetts). Blood samples were shipped directly to Millennium at room temperature for flow cytometry analysis to measure free α4β7 sites and bound vedolizumab on peripheral blood B and T cells. Qualitative determination of CD4+ T cells staining positive for free α4β7 sites and bound vedolizumab after infusion was evaluated by flow cytometry.
All rabbits were euthanized on GD 29. Postmortem end points included the number of fetuses, early and late resorptions, and pre- and postimplantation loss. Examinations of viable fetuses included BW and the appearance of the external body, the viscera, and a mid-coronal slice of the head. All external, visceral, and skeletal findings were recorded as developmental variations (alterations in anatomic structure considered to have no significant biological effect on animal health or body conformity or to occur at high incidence, representing slight deviations from normal) or malformations (structural abnormalities that alter general body conformity, disrupt or interfere with normal body function, or may be incompatible with life).
Monkey Study
This study was conducted at Covance Laboratories (Münster, Germany) in compliance with the GLP Regulations as outlined in German Chemical Law, annex 1 to §19a Chemikalien Gesetz, June 2002, the OECD Principles of GLP (revised 1997, issued January 1998) ENV/MC/CHEM (98),17, and the Consensus Document “The Application of the OECD Principles of GLP to the Organisation and Management of Multi-Site Studies” ENV/JM/MONO (2002)9. The study was conducted under compliance of these regulations and standards, fully met GLP regulations, and was acceptable to US FDA regulatory authorities.
Sexually mature female monkeys at least 3 years of age were purpose bred. Female monkeys were housed individually in a climate-controlled room with a minimum of 10 air changes/hour and artificial lighting controlled on a 12-hour light–dark cycle; enrichment was provided in each cage. Pregnancy was confirmed by ultrasound at postcoital days 18 to 20. Continuation of pregnancies was confirmed by ultrasound on GD 30, 44, 58, 72, 86, 100, 128, 142, and 156 (± 1 day); BWs were monitored on GD 19, 26, 33, 40, 47, 54, 61, 68, 75, 82, 89, 96, 103, 110, 117, 124, 131, 138, 145, 152, and 159 (± 1 day).
Vedolizumab was administered IV every 2 weeks (Q2W) to 36 nonfasted pregnant cynomolgus monkeys starting at GD 20 (total of 9 infusions given over 20-30 minutes on GD 20, 34, 48, 62, 76, 90, 104, 118, and 132). Mothers were randomly assigned to groups receiving doses of 0 mg/kg (saline control), 10 mg/kg (expected maximum human dose), or 100 mg/kg vedolizumab (10-fold the expected maximum human dose; Supplemental Table 1). Pregnant females delivered their offspring who were then monitored for 6 months. Maternal BWs in the postpartum period were recorded on postpartum days (PPD) 1, 7, 14, 21, and 28 and then at monthly intervals for up to 6 months. In addition, food consumption, excretions, and pelage were monitored regularly during the lactation period. Mothers and infants were euthanized on PPD 181 ± 1.
Mothers
Toxicokinetic assessments were conducted on serum from blood collected on GD 20, GD 132, postcoital day 277, and the day of planned necropsy of the infant (PPD 181 ± 1). In addition, breast milk was collected on PPD 28 ± 1 to determine vedolizumab concentration by direct ELISA. Mothers were monitored for clinical signs, morbidity and mortality, pregnancy status, and BW (Supplementary Table 2). Blood sample collection for primate anti-human antibody (PAHA) analysis was scheduled at the same time as that for TK assessments and evaluated by direct ELISA. Pharmacodynamic (PD) assessments (vedolizumab bound to α4β7 integrin in peripheral blood T cells by flow cytometry assays) were conducted on serum samples from blood collected as per the TK assessments. Hematology and clinical assessments included blood counts and standard blood chemistries.
Infants
Toxicokinetic assessments were conducted on blood samples collected on PPD 28 ± 1, PPD 120, and on necropsy (PPD 181 ± 1) to determine vedolizumab concentration by ELISA. Infants were sexed on day 1 and then examined for external abnormalities on PPD 1, 7, 14, 21, and 28 and then at monthly intervals for up to 6 months. Morphologic examinations were performed on PPD 1, 21, 50, 88, and 180 (±1 day). A neurobehavioral test battery was performed on PPD 1 and 7, and grip strength evaluation was conducted on PPD 28 (Supplementary Table 3). Blood samples were also collected on PPD 28 and 120 and at necropsy (PPD 181 ± 1); these were evaluated for PAHA concentration by ELISA and utilized for PD assessments (vedolizumab bound to α4β7 integrin in peripheral blood CD4+/CD45AR− T cells by flow cytometry assays). Full external and internal examinations were made of the infant monkeys following euthanasia.
Statistical Analyses
For the rabbit study, maternal BWs, BW changes, food consumption, gravid uterine weights, numbers of corpora lutea, implantation sites, viable fetuses, and fetal BWs were analyzed by 1-way parametric analysis of variance (ANOVA) to determine intragroup differences with a threshold of P < 0.05 to conclude significance. This analysis was followed by performing Dunnett tests to compare BWs between the vedolizumab treatment groups and the vehicle control group. No statistical analyses were performed on the serum concentration data or derived PK, PD, or RAHA parameters.
For the monkey study, maternal BWs, net maternal weight gain, infant BWs, infant organ weights, hematology data, clinical chemistry data, and organ weights were analyzed by ANOVA, with a threshold of P < 0.05 required to demonstrate statistical significance.
Results
Assessment of Maternal and Developmental Toxicity in Rabbits
Exposure to vedolizumab was dose proportional in pregnant rabbits (Table 1). The mean observed peak serum concentrations (Cmax) of vedolizumab were 244 µg/mL at 10 mg/kg, 818 µg/mL at 30 mg/kg, and 3,020 µg/mL at 100 mg/kg. The areas under the serum concentrations versus time curve (AUC0-337 hr) were 28,300 h·µg/mL at 10 mg/kg, 102,000 h·µg/mL at 30 mg/kg, and 36,600 h·µg/mL at 100 mg/kg.
Vedolizumab Pharmacokinetics in Pregnant Rabbits.
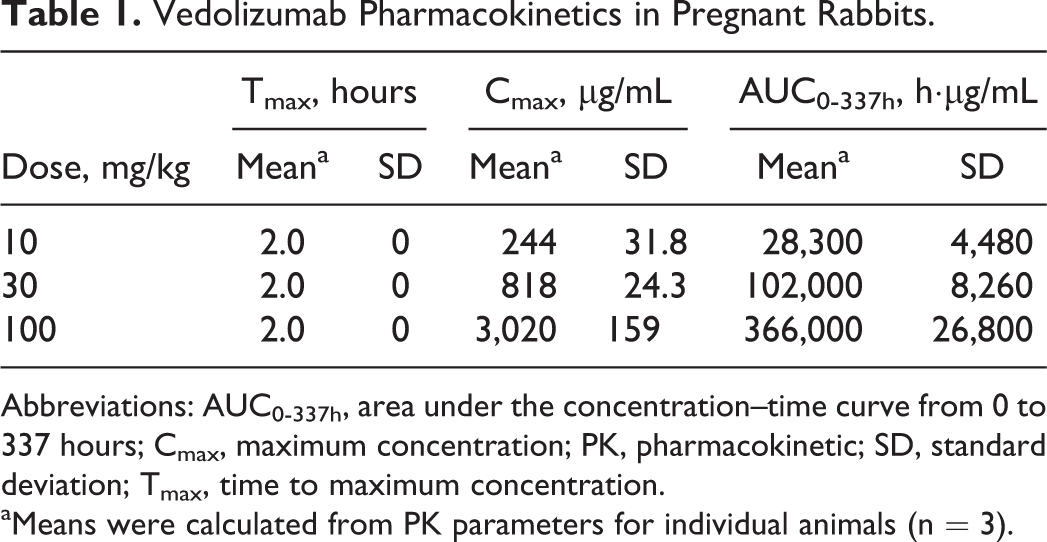
Abbreviations: AUC0-337h, area under the concentration–time curve from 0 to 337 hours; Cmax, maximum concentration; PK, pharmacokinetic; SD, standard deviation; Tmax, time to maximum concentration.
aMeans were calculated from PK parameters for individual animals (n = 3).
Of the 100 timed-pregnant rabbits that were randomized to the 4 study groups, 92 were gravid. There were no significant differences in pregnant female mean net BW, net BW gains, and gravid uterine weights between the groups tested. There was also no statistically significant difference in mean maternal food consumption between the vedolizumab and control groups (Table 2). Of the gravid females, rates of fetal resorption and pre- or postimplantation loss were similar between the 4 study groups. Vedolizumab had no effect on the mean number of fetuses per litter (Table 2).
Clinical Outcomes of Pregnant Rabbits Exposed to Vedolizumab.
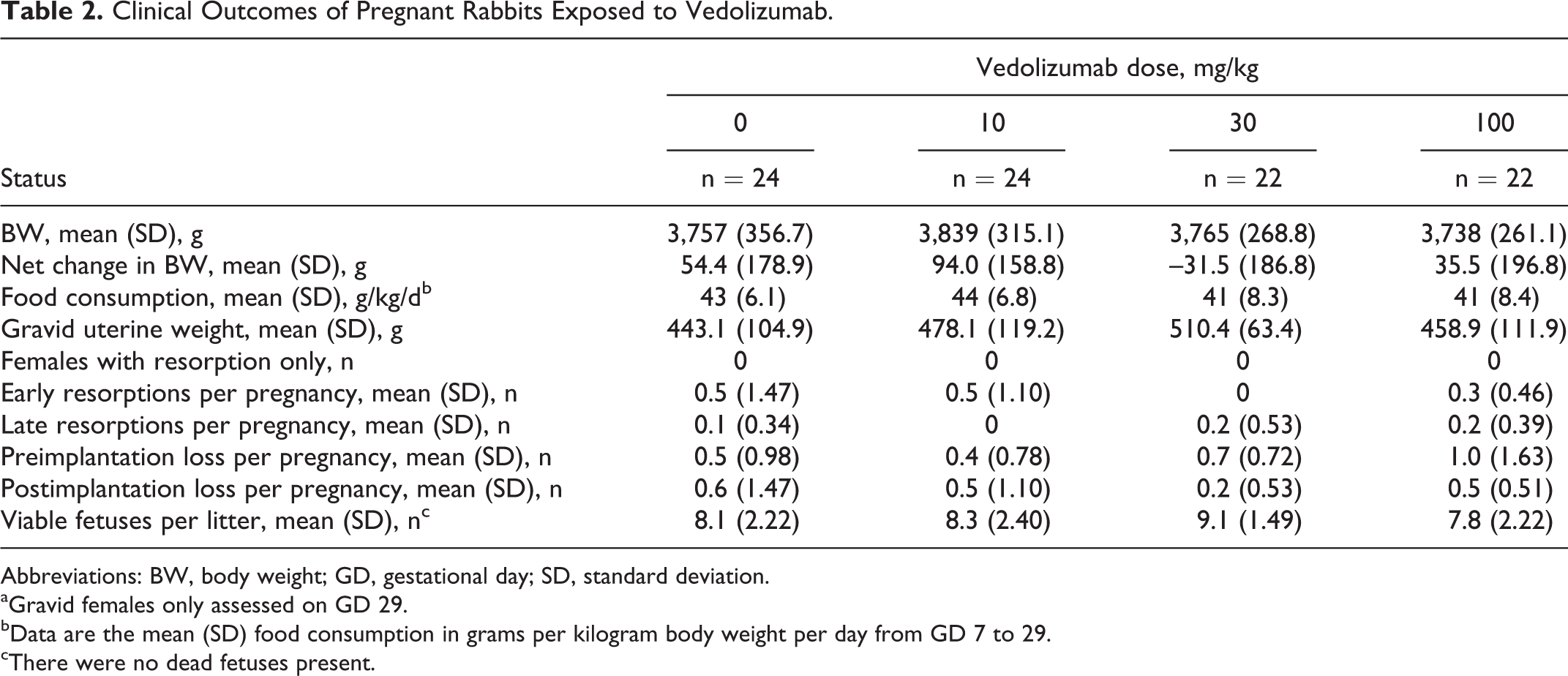
Abbreviations: BW, body weight; GD, gestational day; SD, standard deviation.
aGravid females only assessed on GD 29.
bData are the mean (SD) food consumption in grams per kilogram body weight per day from GD 7 to 29.
cThere were no dead fetuses present.
When fetuses were examined at GD 29, no vedolizumab-related internal findings were observed at any dose level. Fetal weights were comparable across the study groups. Overall, malformations were observed in 4 of 194, 3 of 200, 1 of 201, and 2 of 171 fetuses in the control and vedolizumab 10, 30, and 100 mg/kg dose groups, respectively; all were considered spontaneous in origin (Table 3).
Outcomes of Fetal Rabbits Exposed to Vedolizumab During Gestation.
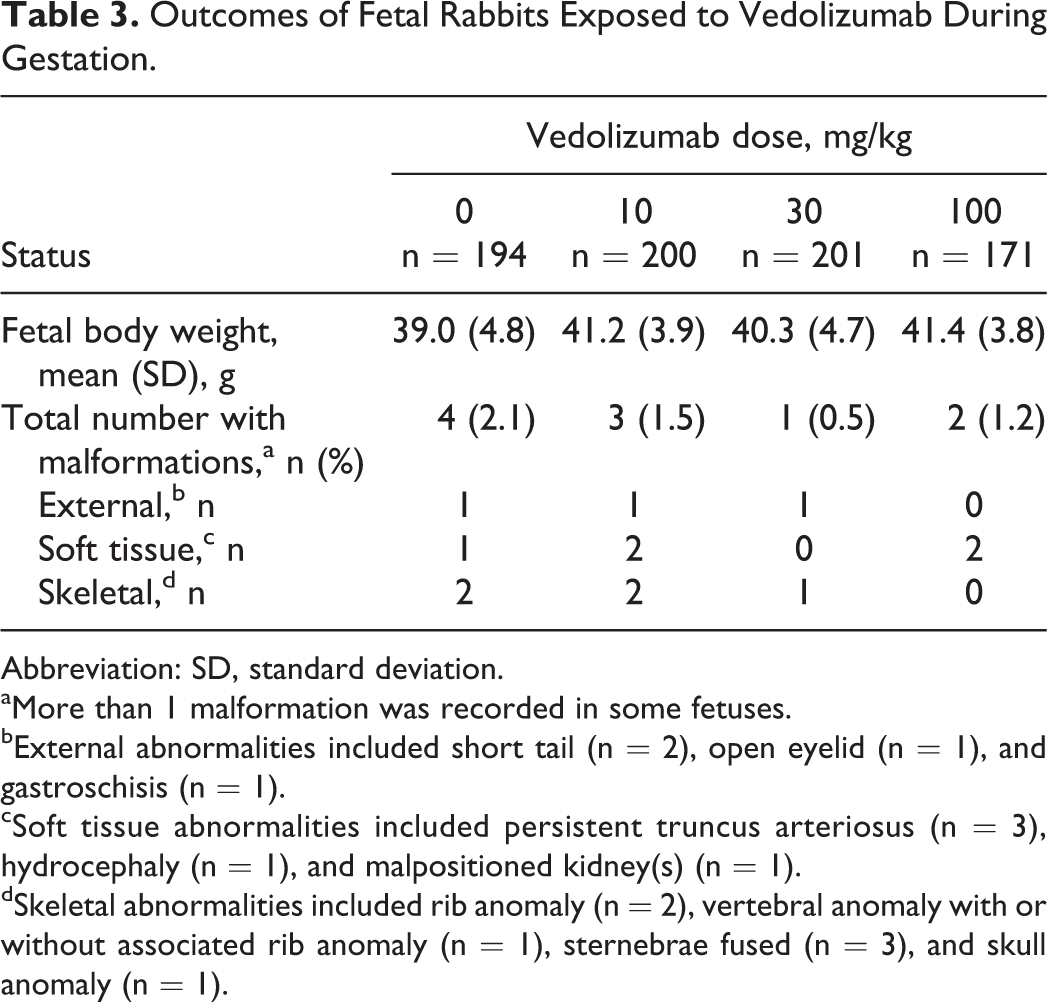
Abbreviation: SD, standard deviation.
aMore than 1 malformation was recorded in some fetuses.
bExternal abnormalities included short tail (n = 2), open eyelid (n = 1), and gastroschisis (n = 1).
cSoft tissue abnormalities included persistent truncus arteriosus (n = 3), hydrocephaly (n = 1), and malpositioned kidney(s) (n = 1).
dSkeletal abnormalities included rib anomaly (n = 2), vertebral anomaly with or without associated rib anomaly (n = 1), sternebrae fused (n = 3), and skull anomaly (n = 1).
Assessment of Maternal and Developmental Toxicity in Monkeys
A substantial and dose–proportional increase in vedolizumab serum levels was achieved rapidly in pregnant mothers after administration (Tmax at ∼0.9-3.4 hours; Table 4 and Supplementary Figure 1). At the 10 mg/kg dose, the mean Cmax was 333 µg/mL for GD 20 and 364 µg/mL for GD 132; mean AUC0-336 was 38,200 h·µg/mL for GD 20 and 62,200 h·µg/mL for GD 132. At the 100 mg/kg dose, the mean Cmax was 2,920 µg/mL for GD 20 and 4,140 µg/mL for GD 132; mean AUC0-336 was 440,000 h·µg/mL for GD 20 and 692,000 h·µg/mL for GD 132. Vedolizumab was detected at low levels (0.163-0.267 μg/mL) on PPD 28 in the breast milk of 3 of 11 monkeys in the 100 mg/kg group but was not detected in the breast milk of mothers who received the 10 mg/kg dose. Vedolizumab was present at low levels on PPD 28 in 4 of 7 infant monkeys in the 10 mg/kg group (concentration range, <0.125-6.9 μg/mL) and in 11 of 11 infants in the 100 mg/kg group (concentration range, 11.5-68.5 μg/mL). One infant in the 100 mg/kg group had a concentration of 0.166 μg/mL on PPD 120.
Vedolizumab Pharmacokinetics in Pregnant Cynomolgus Monkeys.
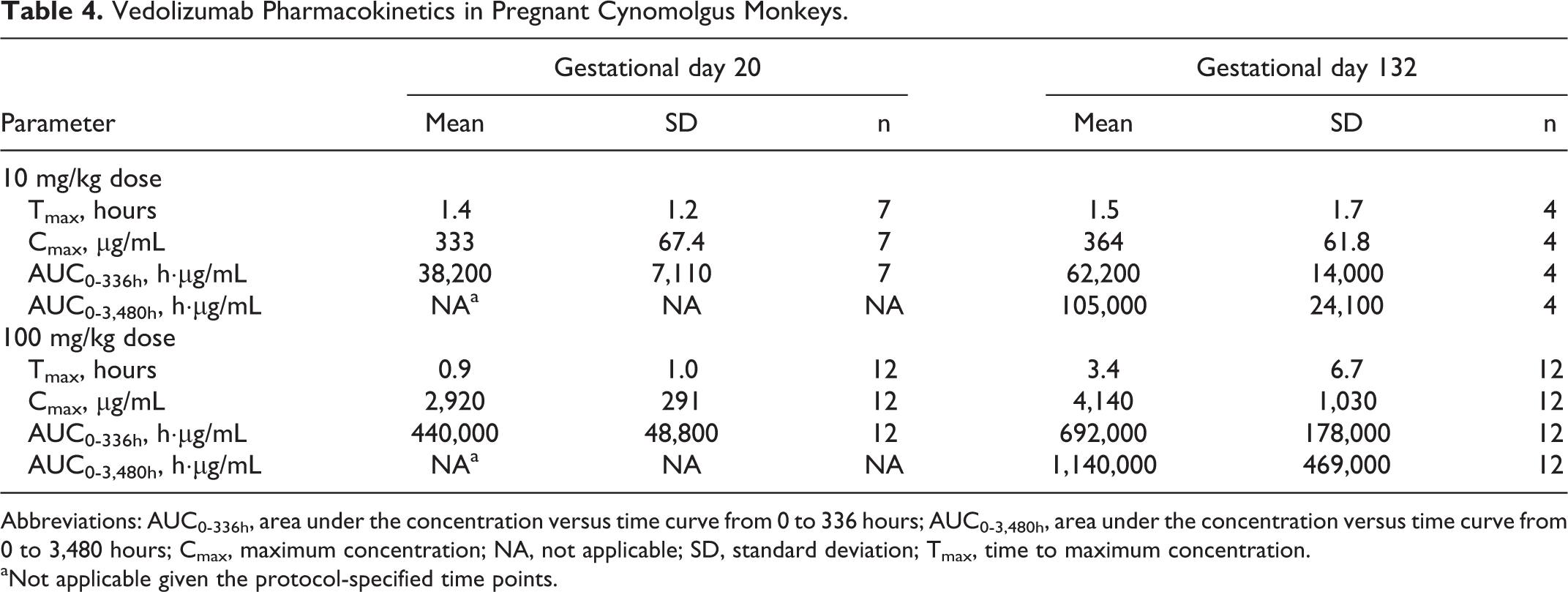
Abbreviations: AUC0-336h, area under the concentration versus time curve from 0 to 336 hours; AUC0-3,480h, area under the concentration versus time curve from 0 to 3,480 hours; Cmax, maximum concentration; NA, not applicable; SD, standard deviation; Tmax, time to maximum concentration.
aNot applicable given the protocol-specified time points.
There were no vedolizumab-related maternal deaths and no increase in the incidence of prenatal loss/death, stillbirth, cesarean delivery, or mean gestational length (Table 5). Two maternal deaths (euthanized in moribund condition) were reported, both of which were a consequence of bacterial infection (crypt microabscesses throughout the large intestine). This condition is a common finding in postpartum cynomolgus monkeys and was not considered related to vedolizumab treatment. Stress during gestation and a general weak condition after birth may have increased the susceptibility for infections. Both mothers delivered healthy infants. There were no vedolizumab-related changes in clinical signs including BW during pregnancy or in the postpartum period (Figure 1) and no gross evidence of vedolizumab-related maternal organ toxicity.
Gestational Outcomes of Cynomolgus Monkeys Exposed to Vedolizumab During Pregnancy.
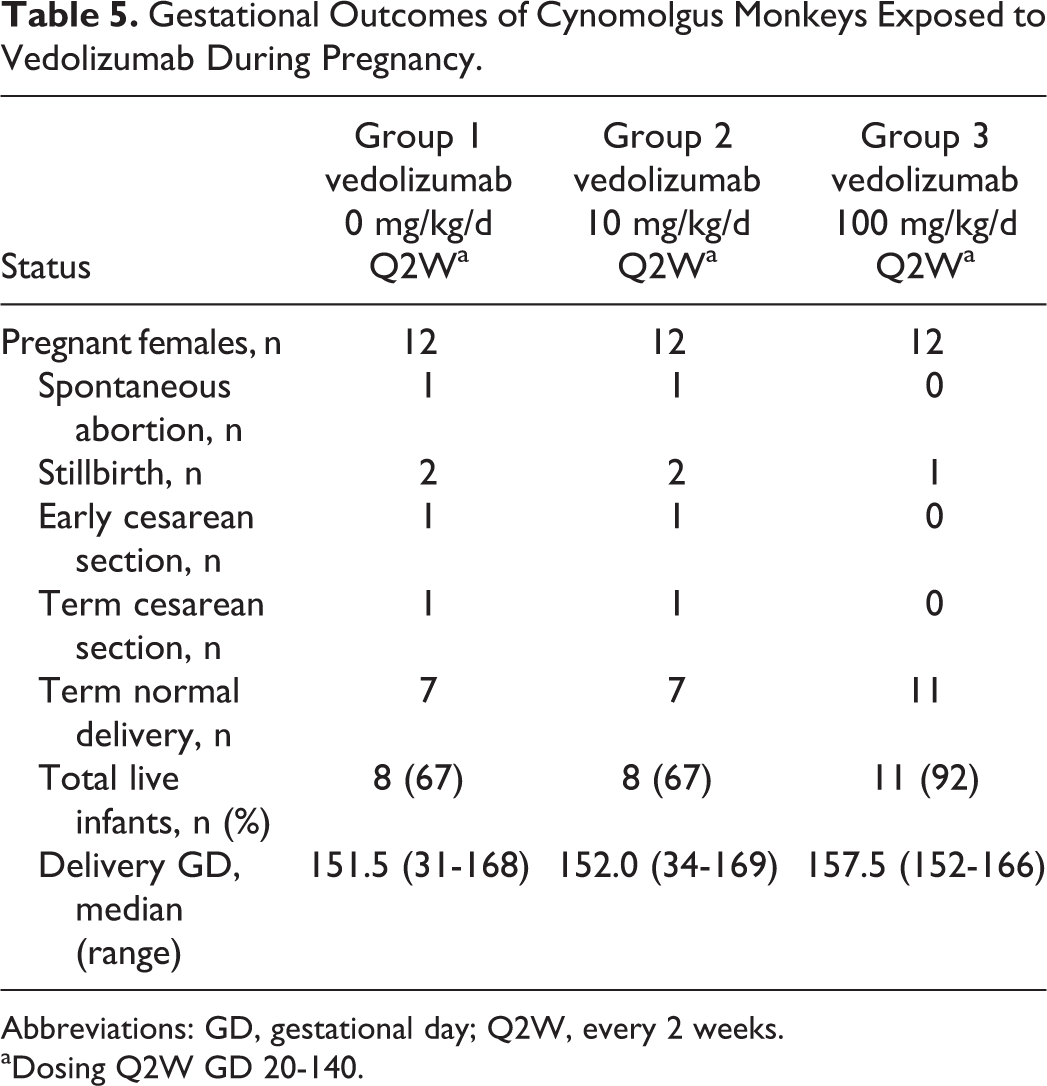
Abbreviations: GD, gestational day; Q2W, every 2 weeks.
aDosing Q2W GD 20-140.
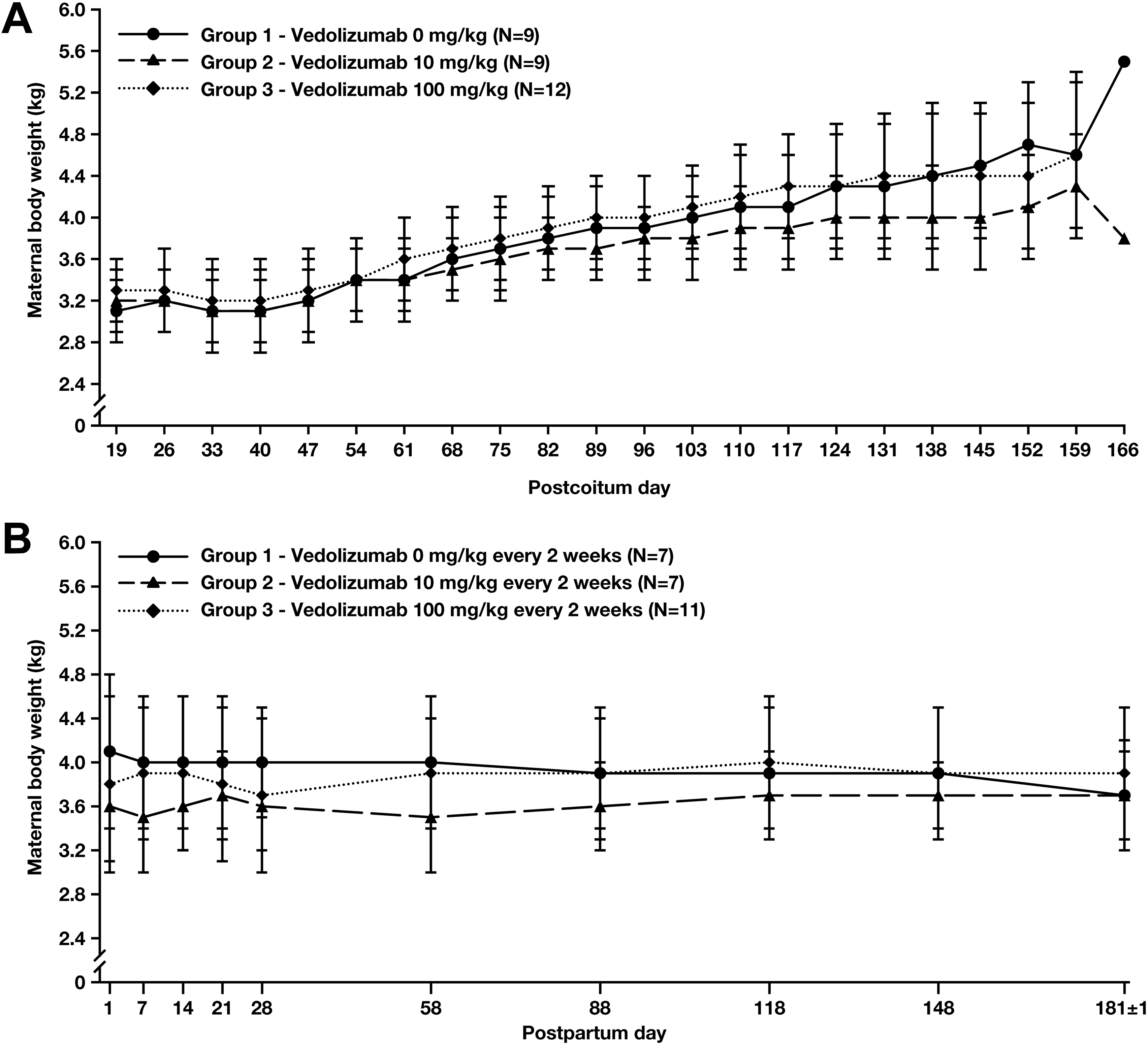
Mean maternal body weight during gestation (A) and lactation (B) in cynomolgus monkeys treated with 0 mg/kg (group 1), 10 mg/kg (group 2), or 100 mg/kg (group 3) vedolizumab once every 2 weeks from GD 20 to GD 140.
No vedolizumab-related effects on the development of infants were observed including postnatal BW gain (Figure 2) or grip strength (Table 6), nor were there vedolizumab-related effects on neurobehavioral or morphologic parameters (Supplemental Tables 4 and 5, respectively) or vedolizumab-related changes in hematology or clinical chemistry (Supplemental Table 6). The infant of one of the moribund mothers in the 100 mg/kg group was sacrificed at the same time as the mother due to the infant’s poor condition, which included mucosal atrophy in parts of the intestine, moderate atrophy in the bone marrow of the sternum, spleen, and thymus, and atrophy of the lymphatic system, as well as insufficient nutrition. This condition was considered related to insufficient nutrition secondary to the mother’s condition and not to vedolizumab treatment.
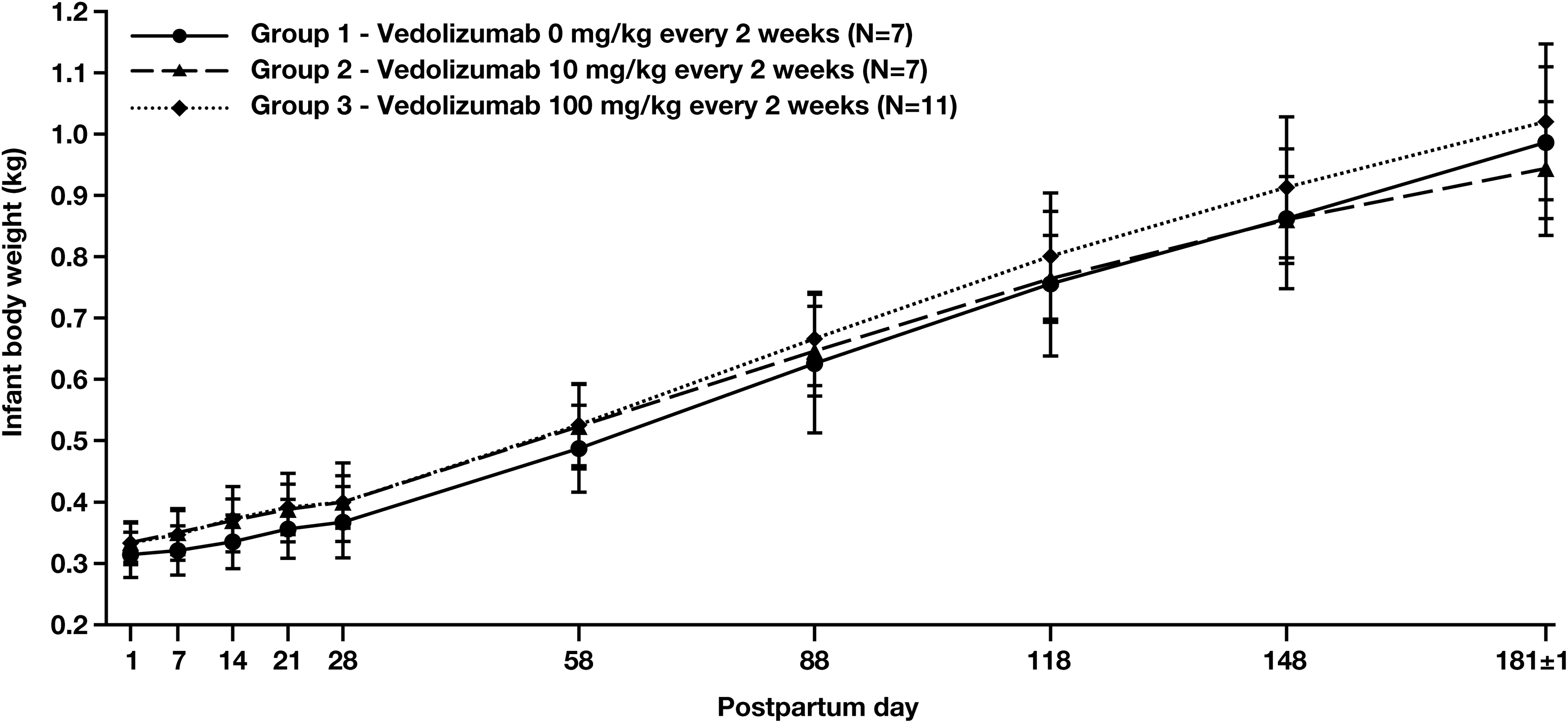
Mean infant body weight through PPD 181 in cynomolgus monkeys born to mothers treated with 0 mg/kg (group 1), 10 mg/kg (group 2), or 100 mg/kg (group 3) vedolizumab once every 2 weeks from GD 20 to GD 140.
Grip Strength of Infant Monkeys at Postpartum Day 28 Exposed to Vedolizumab In Utero.
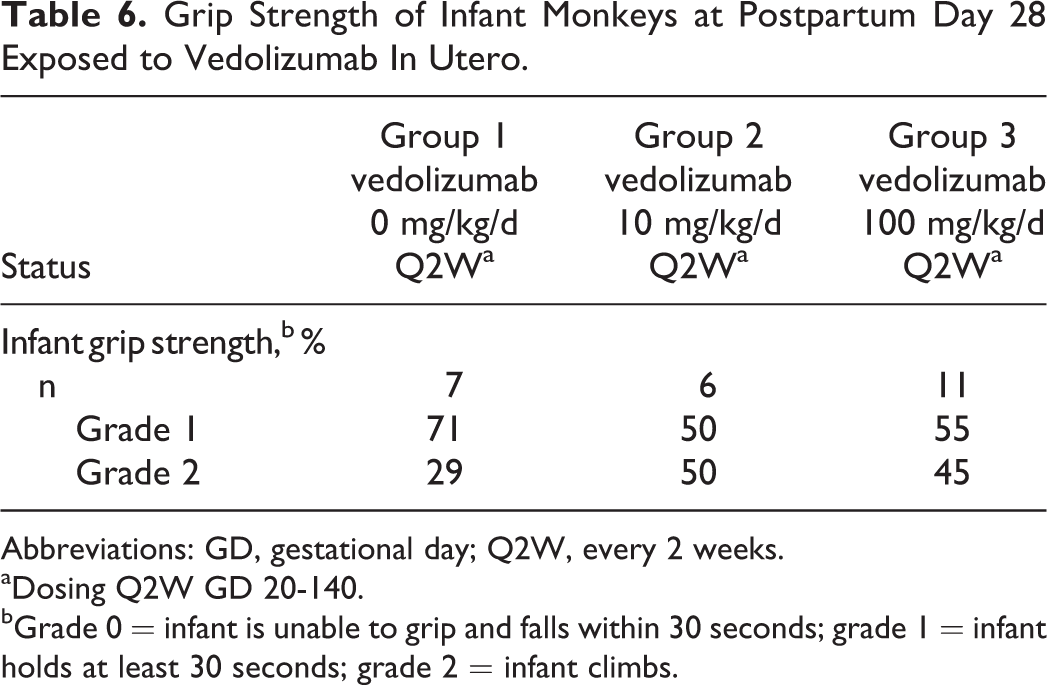
Abbreviations: GD, gestational day; Q2W, every 2 weeks.
aDosing Q2W GD 20-140.
bGrade 0 = infant is unable to grip and falls within 30 seconds; grade 1 = infant holds at least 30 seconds; grade 2 = infant climbs.
Assessment of PD
In rabbits, saturation of the α4β7 binding sites was achieved with the 30 and 100 mg/kg doses and persisted for the duration of study, supporting the sustained pharmacologic activity of vedolizumab at this dose level (Figure 3).
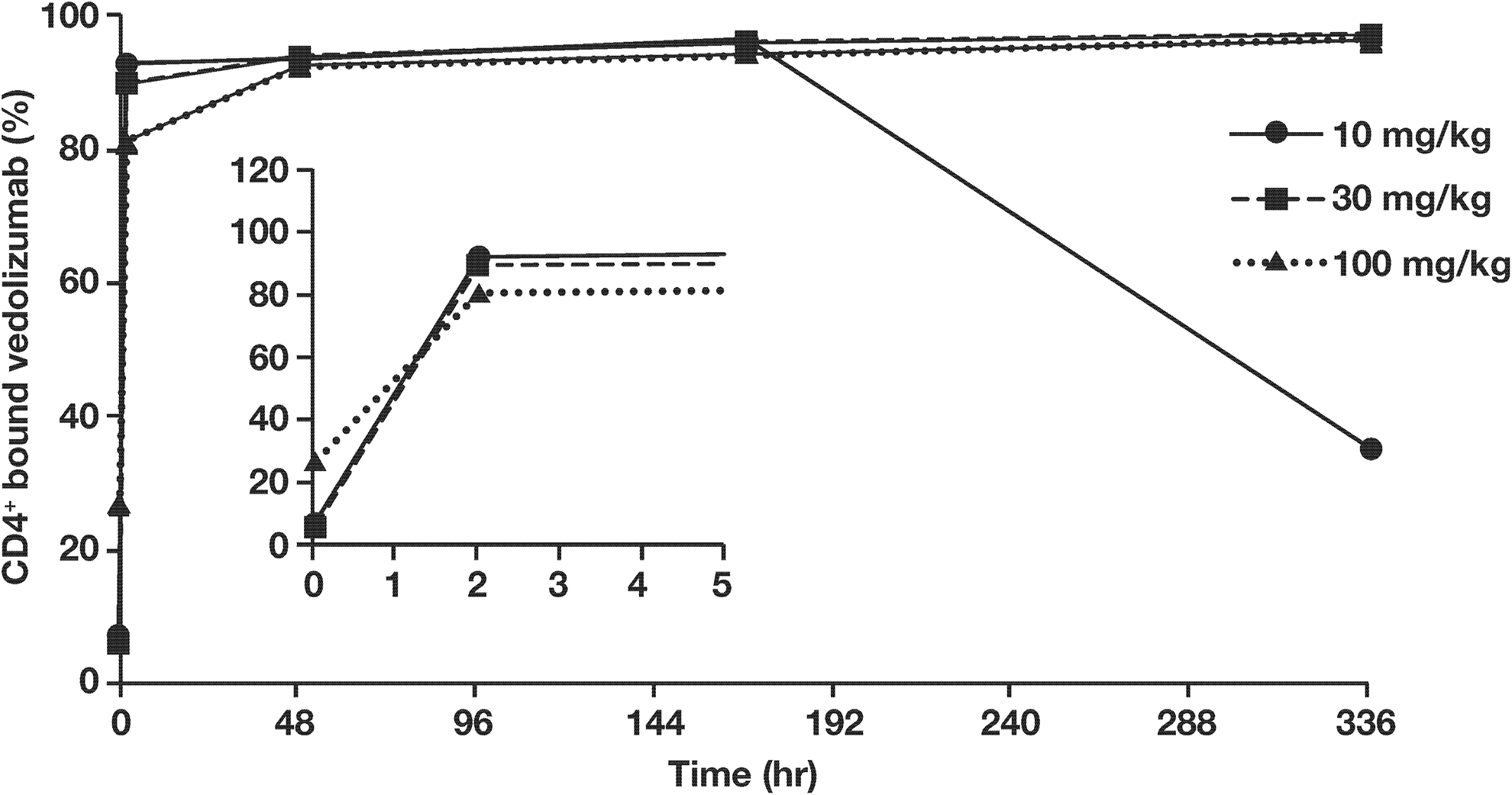
Proportion of CD4+ cells staining positive for bound vedolizumab versus time in pregnant rabbits (n = 3 per group). Time points are calculated from the initiation of vedolizumab infusion. The inset graph illustrates hours 0 to 5 from initiation of infusion.
In the pregnant monkey study, there was no marked difference in PD response in mothers for peripheral CD4+/CD45AR− T cells at either the 10 mg/kg or the 100 mg/kg dose (Figure 4A and B). The PD response in infant monkeys whose mothers were treated with vedolizumab trended similarly to the PD response in the mothers (Figure 5A and B). All infants (11/11) in the 100 mg/kg group showed target saturation on PPD 28 and 3 of these also had target saturation on PPD 120. There was no target saturation in any infant by PPD 181. In the 10 mg/kg dose group, only 3 of the 7 infants were found to have vedolizumab present in serum on PPD 28; vedolizumab levels were low (<10 µg/mL) in all cases.
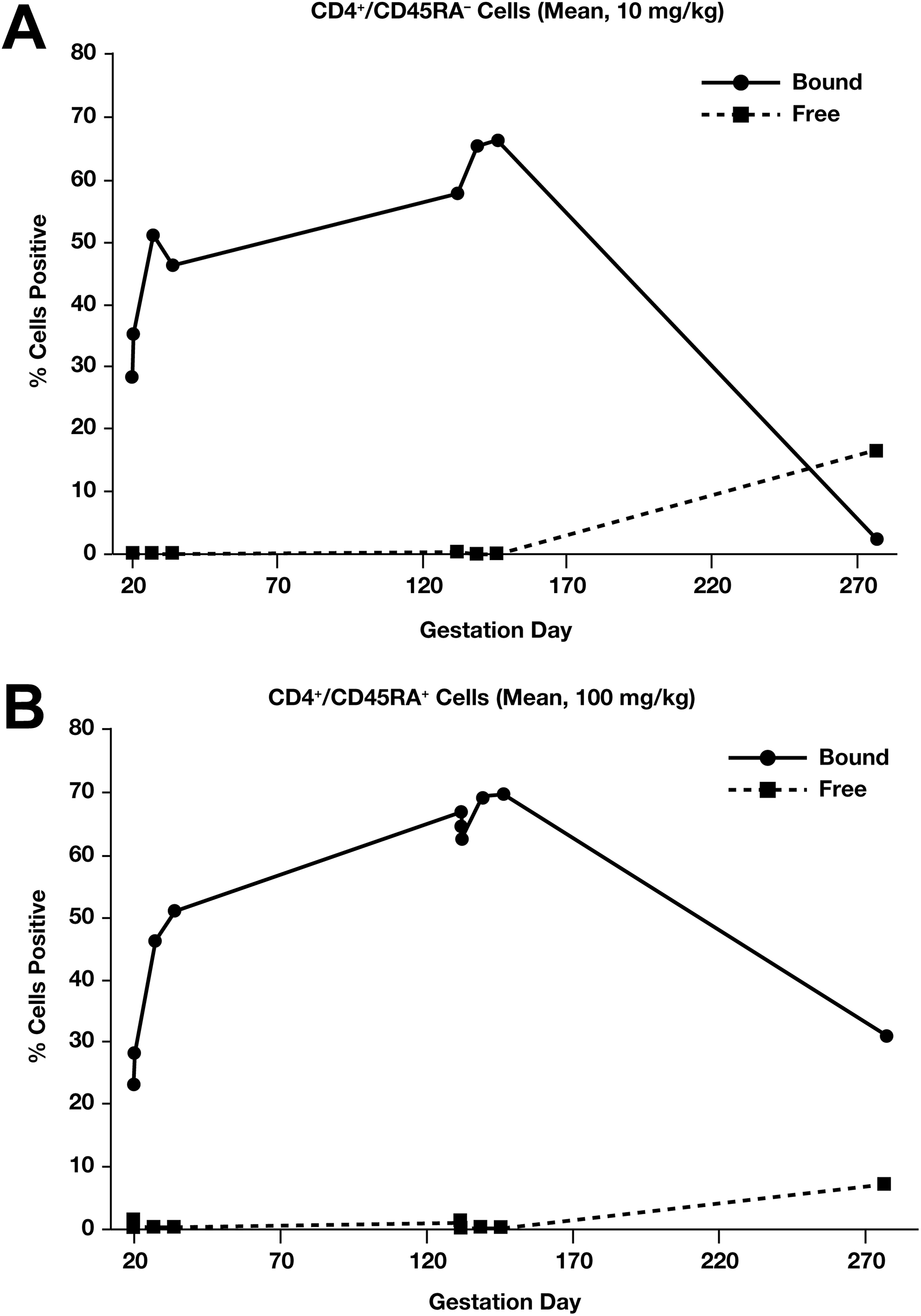
Maternal (mean %) CD4+/CD45RA− cells staining positive for vedolizumab-bound α4β7 sites and free α4β7 sites for pregnant cynomolgus monkeys in the 10 mg/kg vedolizumab group (A) and 100 mg/kg group (B).
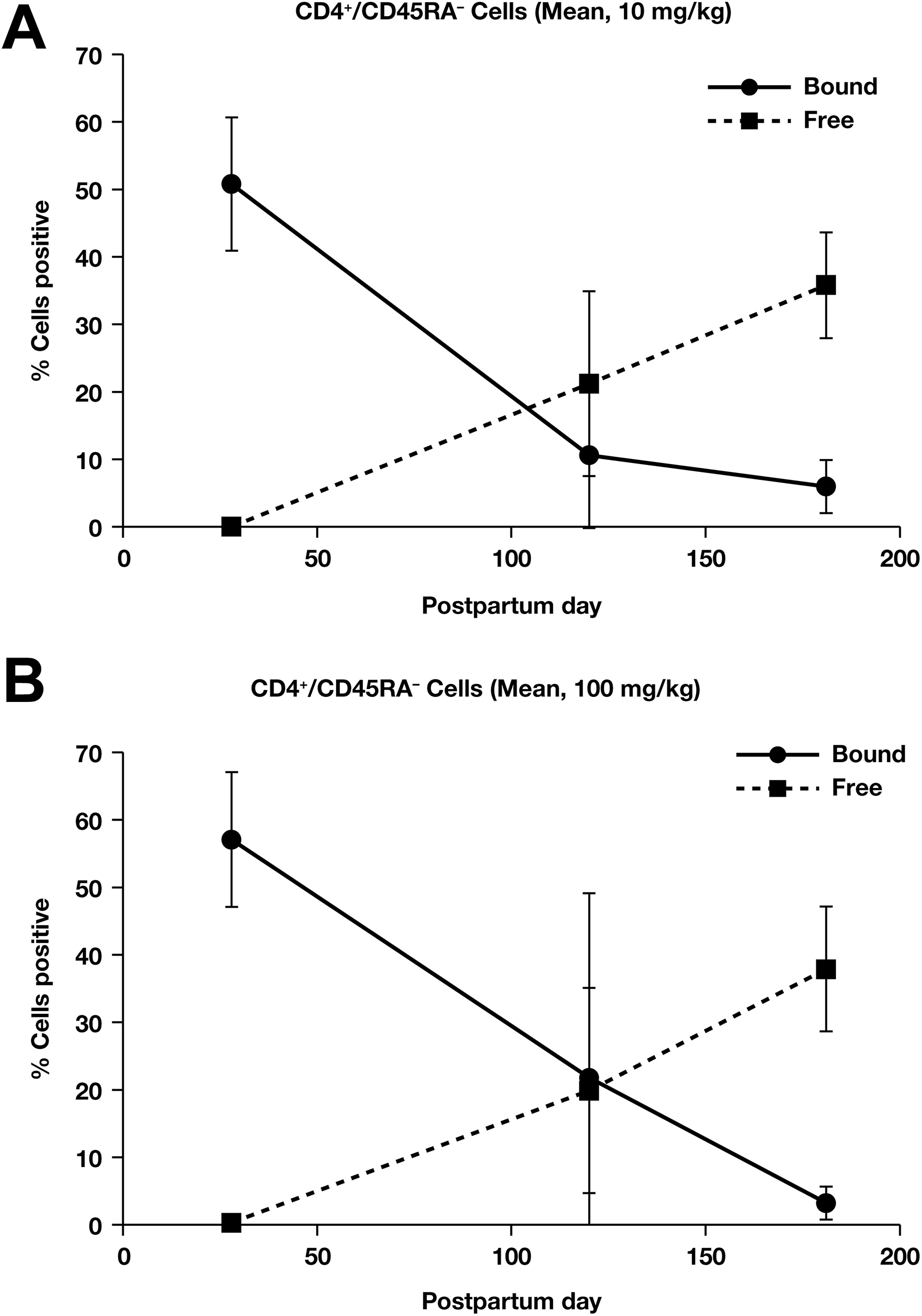
Mean % (±standard deviation) CD4+/CD45RA− cells staining positive for vedolizumab-bound α4β7 sites and free α4β7 sites in infant monkeys whose mothers were dosed with 10 mg/kg vedolizumab (A) and 100 mg/kg (B).
Vedolizumab Immunogenicity
In rabbits, a high RAHA titer (≥18,750) on GD 20 (14 days postinfusion) developed in 2 of 3 pregnant rabbits in the 10 mg/kg group. The RAHA response in these animals appeared to be neutralizing because their exposure to vedolizumab and PD responses were reduced compared to the animals in the group with a titer of 3,750. The immunogenicity of vedolizumab at 30 mg/kg and 100 mg/kg was minimal (titers ≤3,750).
In pregnant monkeys, PAHA had an effect on the TK and PD of vedolizumab in the 10 mg/kg group, resulting in lowered vedolizumab serum concentrations and reduced binding of vedolizumab to peripheral blood B cells and T cells. Positive PAHA titers (≥500) were detected in 1 of 12 mothers in the control group, 9 of 12 mothers in the 10 mg/kg group, and 9 of 12 mothers in the 100 mg/kg group. No relationship between dose and titer or dose and day of highest titer was apparent.
Vedolizumab was mildly immunogenic in infant monkeys whose mothers were treated with vedolizumab. The PAHAs were detected in 0 of 7 infants in the control group, 2 of 7 infants in the 10 mg/kg group, and 1 of 9 infants in the 100 mg/kg group.
Discussion
Inflammatory bowel disease can affect women in their reproductive years—consequently, the safety of therapeutic agents for IBD during pregnancy is an important consideration. Pregnant patients are commonly excluded from clinical studies, and contraceptive measures are typically required for female patients at risk of pregnancy, as was the case in the vedolizumab clinical program. As a result, there is currently limited experience with vedolizumab in pregnancy from clinical studies. 31 Although real-world evidence is being collated through postmarketing experience and registries such as PIANO and the Organization of Teratology Information Services observational pregnancy registry, 35 supporting data from animal studies can provide valuable information regarding placental transfer and potential effects on pregnant females and their offspring.
Rabbits and cynomolgus monkeys are among those species frequently used in preclinical assessment of monoclonal antibodies, and both are considered good models for developmental toxicity studies, although the maternal–fetal transfer of IgG antibodies with cynomolgus monkeys is closer to that of humans. 36,37 In the present study, placental transfer of vedolizumab was confirmed in cynomolgus monkeys. Exposure of vedolizumab showed an increasing trend in a greater than dose proportional manner. At 100 mg/kg (the highest dose examined), saturation of binding to α4β7 sites was achieved throughout the study period. Administration of high doses of vedolizumab did not show any maternal or developmental effects in rabbits or monkeys, with vedolizumab shown to be pharmacologically active in both animal models as assessed by target binding saturation. Vedolizumab 100 mg/kg, the highest dose tested in rabbits, resulted in Cmax and AUC0-312h values of 3,020 ± 159 µg/mL and 366,000 ± 26,800 µg·h/mL, respectively, and values in monkeys of 4,140 ± 1,030 µg/mL and 692,000 ± 178,000 µg·h/mL, respectively. Based on the lack of maternal and embryo–fetal effects, 100 mg/kg (the highest dose tested) was determined to be the no observed adverse effect level (NOAEL) in both rabbits and monkeys; this dose is approximately 20 times the recommended human dosage.
Anti-human antibodies were detected in most animals to which vedolizumab was administered directly, whereas vedolizumab was only mildly immunogenic in offspring. Immunogenicity to monoclonal antibodies in nonhuman primate models is known to influence PK and PD to varying degrees. 38 As observed with other monoclonal antibodies, the immunogenicity of vedolizumab in adult animals was greater at the lowest dose (10 mg/kg) than at higher doses of 30 mg/kg and 100 mg/kg, due to a tolerizing effect. The RAHA/PAHA responses in the lowest dosing group appeared to be neutralizing, as it affected both serum concentrations of vedolizumab and PD parameters, whereas this effect was less pronounced in higher dosing groups. A similar effect was observed with the natalizumab administration in pregnant cynomolgus monkeys, probably due to increased natalizumab clearance. 39 Despite eliciting an immunogenic response, pharmacologically active drug exposure was observed in both mothers and infants, with continuous saturation of the α4β7 sites on peripheral lymphocytes seen at the NOAEL of 100 mg/kg/d.
Clinical data in pregnancy are currently limited for all biologic agents used for the treatment of IBD. 15 The majority of the pregnancy safety information from the anti-TNFα agents adalimumab and infliximab suggests low risk. Less is known about the safety of the anti-integrins in pregnancy and the exact role of α4β7 integrin in fetal development. 15 As with other biologics, however, the current evidence suggests that in utero exposure to vedolizumab does not increase the risk of congenital abnormalities, which is supported by the animal data presented here. This correlates well with the current understanding that placental transfer of IgG begins after the crucial period of organogenesis in the first trimester. 15,31 Due to similarities in placental transfer of IgG between animal models and humans, preclinical data can supplement our understanding of the effects of in utero exposure to biologic therapies. 37 Cynomolgus monkeys may be of particular relevance in identifying safety concerns, since they are considered to be similar to humans in many aspects of reproductive physiology, including transfer via the chorioallantoic placenta. There are, however, limitations to consider when extrapolating these findings, such as the fact that these are small cohorts of non-disease-specific models. Another limitation is species differences in placental anatomy: Cynomolgus monkeys have bidiscoidal placentas, whereas humans have discoidal placentas, which could translate to differences in degree and timing of exposure between the 2 species. 36,37
Animal studies similar to the present study have been conducted with other biologics for IBD, with studies in cynomolgus monkeys (adalimumab and natalizumab), guinea pigs (natalizumab), and mice (infliximab) largely supporting their safety in pregnancy. 25,26,39 -42 Preclinical studies with natalizumab in cynomolgus monkeys did, however, observe an increased number of spontaneous abortions in 1 study cohort as well as hematologic effects (decreased lymphocyte and platelet counts) in offspring. 40 Nevertheless, as with vedolizumab, 31,33,34 data in humans for all of these agents indicate no increased risk of congenital abnormalities or adverse pregnancy outcomes. 15
In conclusion, vedolizumab did not show any potential for maternal or developmental toxicity in rabbits or monkeys. The data presented here support the growing body of clinical evidence that vedolizumab exposure represents a low risk to the mother or to the developing fetus in humans.
Supplemental Material
Supplemental Material, DS1_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS1_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS2_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS2_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS3_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS3_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS4_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS4_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS5_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS5_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS6_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS6_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS7_IJT_10.1177_1091581819864105 - Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate
Supplemental Material, DS7_IJT_10.1177_1091581819864105 for Evaluation of the Developmental Toxicity of Vedolizumab, an α4β7 Receptor Antagonist, in Rabbit and Nonhuman Primate by David Crawford and Mitchell Friedman in International Journal of Toxicology
Footnotes
Acknowledgments
Medical writing support was provided by Christopher Barnes, PhD, of ProEd Communications, Inc, and was sponsored by Takeda Pharmaceutical Company Ltd.
Author Contributions
D. Crawford and M. Friedman contributed to interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David F. Crawford and Mitchell Friedman are employees of Takeda and receive company stocks.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Takeda Pharmaceutical Company Ltd.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
