Abstract
Potential applications of gold nanoparticles in biomedicine have increasingly been reported on account of the ease of synthesis, bioinert characteristics, optical properties, chemical stability, high biocompatibility, and specificity. The safety of these particles remains a great concern, as there are differences among toxicity study protocols used. This article focuses on integrating results of research on the toxicological behavior of gold nanoparticles. This can be influenced by the physicochemical properties, including size, shape, surface charge, and other factors, such as methods used in the synthesis of gold nanoparticles, models used, dose, in vivo route of administration, and interference of gold nanoparticles with in vitro toxicity assay systems. Several researchers have reported toxicological studies with regard to gold nanoparticles, using various in vitro, in vivo, and in ovo models. The conflicting results concerning the toxicity of gold nanoparticles should thus be addressed to justify the safe use of gold nanoparticles in biomedicine.
Introduction
Recently, researchers have become more interested in nanomedicine due to more target-specific drug delivery. There is, however, still a paucity to apply new findings because of the lack of clarity with regard to short- and long-term clinical effects. This article tries to integrate the available literature to give a better understanding of factors that may contribute to the toxicity or safety of gold nanoparticles (AuNPs). Toxicological studies from 2009 to 2018 are used in this review because many earlier toxicity studies focusing on AuNPs have previously been reviewed. 1 –4
Promising applications of AuNPs in nanomedicine include plasmon-based labeling and imaging, optical and electrochemical sensing, and diagnostics, which facilitate early detection and treatment of cancers, 5 –8 hepatitis, 9 and tuberculosis. 10 –12 Gold nanoparticles may also be used in angiogenesis, as antibacterial agents, in photothermal and radiofrequency-mediated thermal therapies, 13,14 as well as delivery vehicles for genetic materials, 15 imaging agents, 5,16 and drugs. 17,18 These are applicable for various diseases, including cancers, 19 Alzheimer disease, 20 diabetes, 21 heart disease, 22 hepatitis, 23 tuberculosis, 24 rheumatoid arthritis, 25 and obesity. 26 In fact, gold nanoshells (AuroLase for lung cancer [NCT01679470], head and neck cancer [NCT00848042]), as well as tumor necrosis factor (TNF)-conjugated AuNPs (NCT00356980) have been approved by Food and Drug Administration for cancer trials. 27
These applications are based on the stability and unique properties of AuNPs 28,29 as well as the ability to easily translocate target cells, thereby enhancing sustained release of drugs and increasing therapeutic efficacy. 30 There is however a great concern since during synthesis and development for a wide range of applications, AuNPs of different sizes, shapes, and surface charges are generated and may be of health risk.
Nanoparticles
Particles smaller than 100 nm in diameter are classified as nanoparticles. Metallic nanoparticles are useful in the diagnosis and in facilitating early detection of cancers. 31,32 They are also used as therapeutic agents because these can easily translocate to target cells and bear high drug load. 33
Despite the versatility of nanoparticles, metal nanoparticles face challenges such as toxicity, inefficiency in translocation into cells, and clearance from tissues or organs. 34 To overcome some of these barriers, AuNPs are used in nanomedicine because of their diverse properties, including biocompatibility, conjugation to biomolecules, and tunable optical properties resultant of their shape, size, and surface area. 33,35,36
Gold Nanoparticles
Gold in its bulk form is unreactive and is therefore considered a noble metal. In its ionic state, however, gold is reactive. Due to the unreactive nature of bulk gold, it is often considered a major raw material in various biomedical applications, including dental prostheses, drug delivery microchips, endovascular stents, reconstructive surgery, and as a food additive. 37 Bulk gold can thus be considered nontoxic. Gold also exists in ionic or molecular forms (as gold salts), which allow the synthesis of gold as nanomaterials. In nanoscale form, gold possesses several properties that differentiate them from the bulk or molecular state, thereby making them a considerable powerful tool in nanomedical applications. 38 Such characteristics include facile synthesis, higher particle reactivity, greater surface area to volume ratio, ability to undergo surface modifications, and strong optical properties. 38,39
These unique physicochemical properties enable them to interact with biological fluids and intracellular structures or biomolecules, resulting in cellular and tissue toxicity. 37,40 These properties also affect the rate of excretion of nanoparticles, which can contribute to the toxicity.
Most nanotoxicity assessment studies are conducted using in vitro cell culture models, but results from these studies cannot fully approximate the complexities that occur in whole organisms in terms of absorption, distribution, biotransformation, and elimination. 2 Both in vivo and in ovo toxicity need to be considered. In this review, the focus will be on the possible factors contributing toward the toxicity of AuNPs as well as recent advances in the in vitro, in vivo and in ovo toxicities.
Toxicity of AuNPs
A number of researchers have reported the safety of naked or conjugated AuNPs, 26,41 –48 as well as the toxicity, 49 –57 using various in vitro, in vivo, and in ovo models. There are, however, few toxicological reports of AuNPs in the animal model, which is the preferred system for the toxicological evaluation of a novel agent. 58 The toxicologic behavior of AuNPs are guided by their small size, shape, and surface charges. 3
Factors Influencing the Toxicity of AuNPs
Several factors (Figure 1) are important in predicting the toxicity of AuNPs in vitro, in ovo, and most especially in vivo. Numerous studies have indicated that toxicity is highly dependent on the physicochemical properties of nanoparticles, although many of these unique properties make them appealing to nanomedicine and biological applications. 28
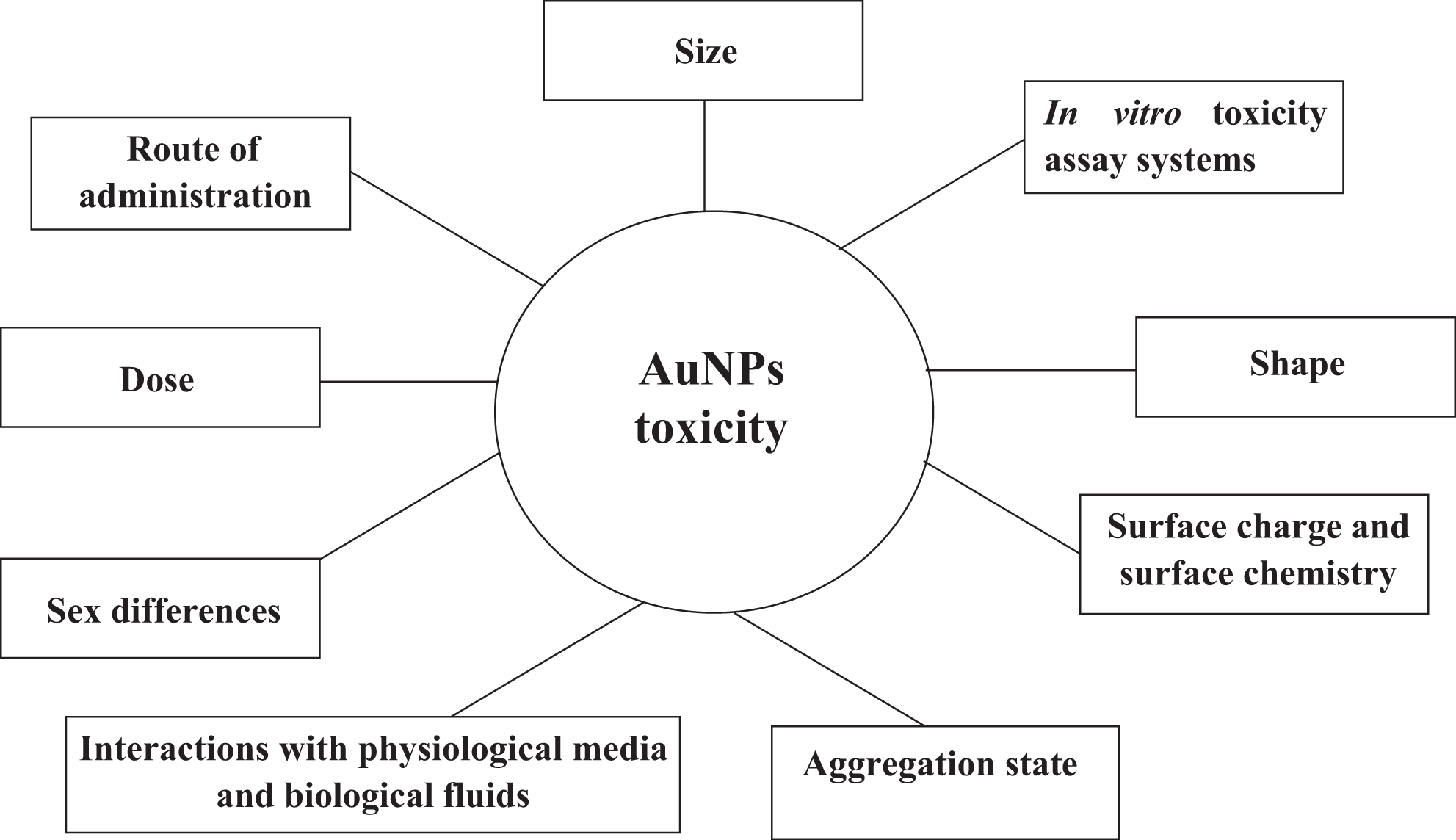
Factors influencing the toxicity of gold nanoparticles.
Interference of AuNPs With In Vitro Toxicity Assay Systems
The cytotoxicity of AuNPs has been assessed via various in vitro toxicity testing methods. Most often, these include (1) the 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT) assay, (2) the lactate dehydrogenase (LDH) assay, (3) neutral red, (4) the intracellular adenosine triphosphate level assay, 59 and (5) the colony-forming efficiency (CFE) assay, a label-free test. 60
It has been difficult to draw meaningful conclusions from the literature with regard to the toxicity of AuNPs based on the use of these assay methods because of the differences in size of the AuNPs used and the cell types implemented, 59 as well as variations in shape, model used, dose, route of administration, sex, surface charge, and surface chemistry. The contradictory cytotoxicity results obtained with the different assay methods were reportedly caused by possible interference of the tested AuNPs with these assay systems. 59,61 This could be linked to the several unique physicochemical properties displayed by nanoparticles. These include high surface area, which leads to increased adsorption capacity; optical properties, which allow interference with fluorescence or visible light absorption detection systems (as they absorb light in the visible region, ∼520 nm); and magnetic properties, which lead to interference with methods based on redox reactions. 61,62 This potential interference was reportedly eliminated using cell impedance technology. 59 As a result of this, the methods adopted for traditional toxicological studies cannot directly be applied to nanoparticle toxicological studies.
Dhawan and Sharma, 62 therefore, suggested the provision of a standardized nanoparticle reference material for toxicological studies, through which comparison of data can be made across different studies. The protocol of the CFE assay has been reported by Ponti et al 60 to be well-defined and has several advantages when compared to other in vitro toxicity-screening assays. These include lack of interference (because it is a label-free test) and higher sensitivity. The performance of this method was in at least 3 independent runs tested by 12 laboratories from countries, including France, Italy, Japan, Poland, Republic of Korea, South Africa, and Switzerland, which are in the frame of the Organisation for Economic Co-Operation and Development (OECD) working party of manufactured nanomaterials. This test has already been used in different in vitro systems to assess cytotoxicity of a wide range of nanomaterials, such as AuNPs, 63 silica nanoparticles, 64 and multi-wall carbon nanotubes. 65 The CFE assay was therefore suggested to serve as a first-choice method in defining dose–effect relationships for other in vitro assays in the early screening of toxicity of nanoparticles. 60
Size
The size of nanoparticles is thought to influence the biological activity of the cell.
66
A number of researchers have reported small-sized AuNPs (
Evaluation of AuNPs in Different Cell Lines.
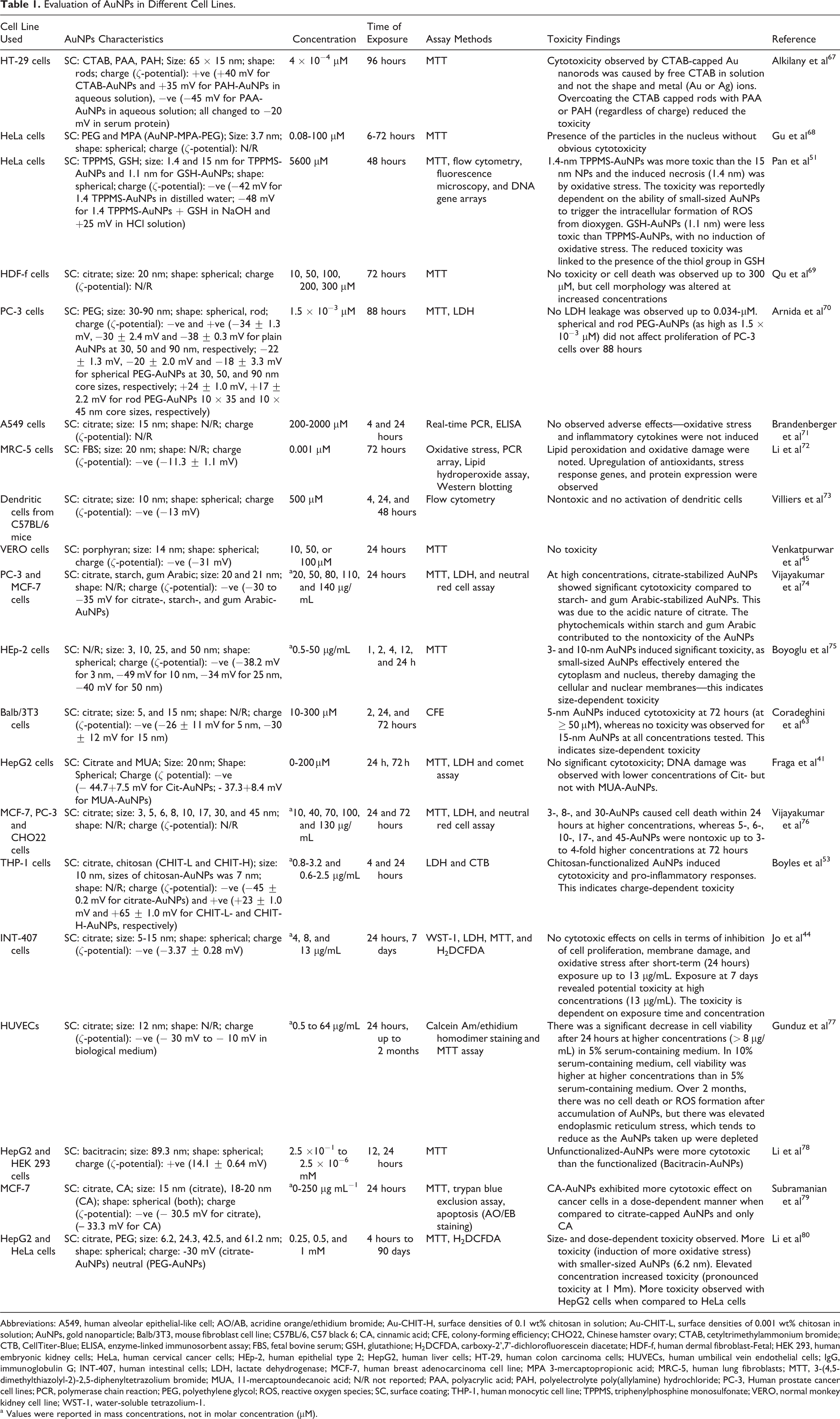
Abbreviations: A549, human alveolar epithelial-like cell; AO/AB, acridine orange/ethidium bromide; Au-CHIT-H, surface densities of 0.1 wt% chitosan in solution; Au-CHIT-L, surface densities of 0.001 wt% chitosan in solution; AuNPs, gold nanoparticle; Balb/3T3, mouse fibroblast cell line; C57BL/6, C57 black 6; CA, cinnamic acid; CFE, colony-forming efficiency; CHO22, Chinese hamster ovary; CTAB, cetyltrimethylammonium bromide; CTB, CellTiter-Blue; ELISA, enzyme-linked immunosorbent assay; FBS, fetal bovine serum; GSH, glutathione; H2DCFDA, carboxy-2’,7’-dichlorofluorescein diacetate; HDF-f, human dermal fibroblast-Fetal; HEK 293, human embryonic kidney cells; HeLa, human cervical cancer cells; HEp-2, human epithelial type 2; HepG2, human liver cells; HT-29, human colon carcinoma cells; HUVECs, human umbilical vein endothelial cells; IgG, immunoglobulin G; INT-407, human intestinal cells; LDH, lactate dehydrogenase; MCF-7, human breast adenocarcinoma cell line; MPA 3-mercaptopropionic acid; MRC-5, human lung fibroblasts; MTT, 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide; MUA, 11-mercaptoundecanoic acid; N/R not reported; PAA, polyacrylic acid; PAH, polyelectrolyte poly(allylamine) hydrochloride; PC-3, Human prostate cancer cell lines; PCR, polymerase chain reaction; PEG, polyethylene glycol; ROS, reactive oxygen species; SC, surface coating; THP-1, human monocytic cell line; TPPMS, triphenylphosphine monosulfonate; VERO, normal monkey kidney cell line; WST-1, water-soluble tetrazolium-1.
a Values were reported in mass concentrations, not in molar concentration (µM).
Effect of AuNPs on Zebrafish.
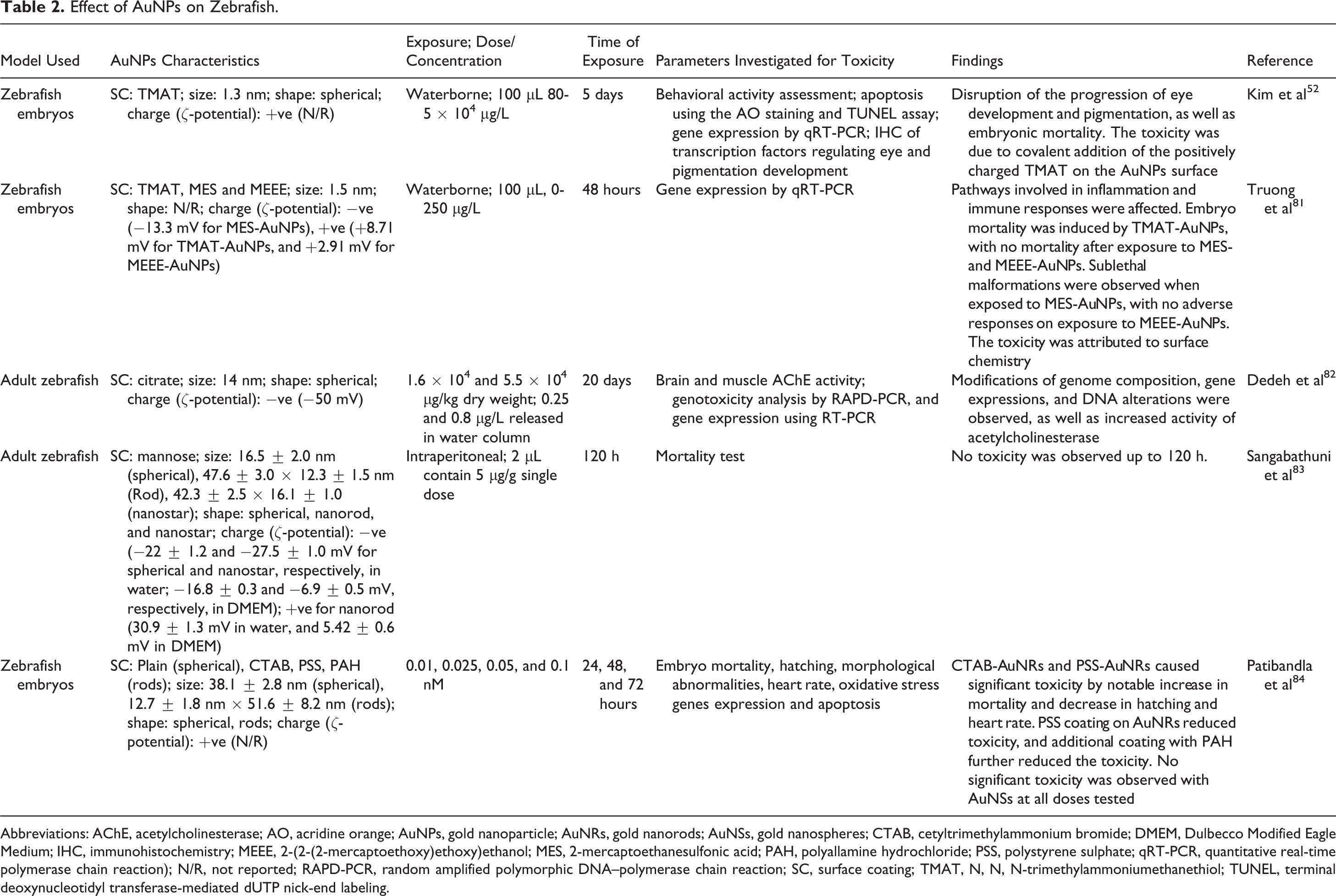
Abbreviations: AChE, acetylcholinesterase; AO, acridine orange; AuNPs, gold nanoparticle; AuNRs, gold nanorods; AuNSs, gold nanospheres; CTAB, cetyltrimethylammonium bromide; DMEM, Dulbecco Modified Eagle Medium; IHC, immunohistochemistry; MEEE, 2-(2-(2-mercaptoethoxy)ethoxy)ethanol; MES, 2-mercaptoethanesulfonic acid; PAH, polyallamine hydrochloride; PSS, polystyrene sulphate; qRT-PCR, quantitative real-time polymerase chain reaction); N/R, not reported; RAPD-PCR, random amplified polymorphic DNA–polymerase chain reaction; SC, surface coating; TMAT, N, N, N-trimethylammoniumethanethiol; TUNEL, terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling.
Effect of AuNPs in Mice.
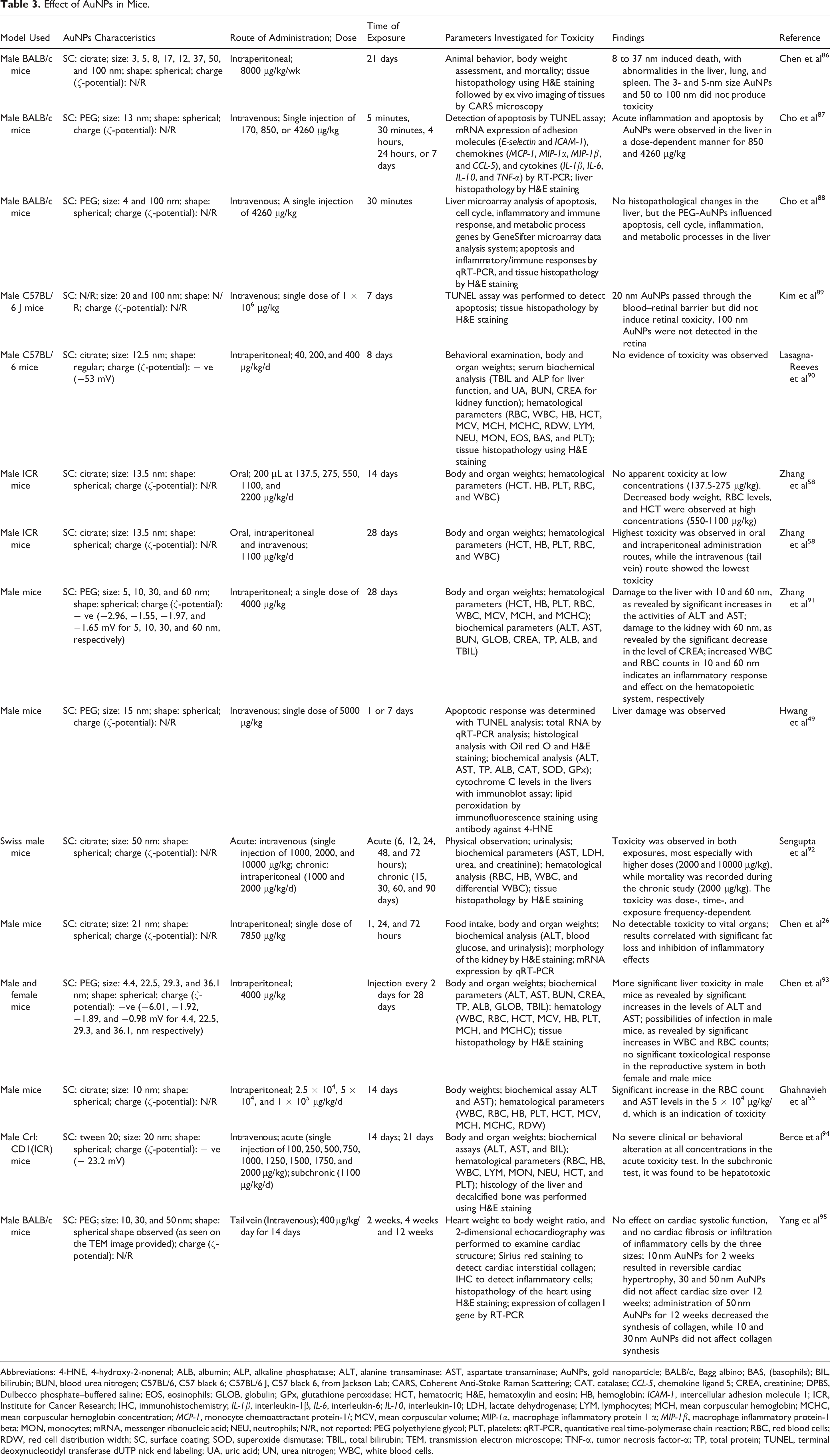
Abbreviations: 4-HNE, 4-hydroxy-2-nonenal; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine transaminase; AST, aspartate transaminase; AuNPs, gold nanoparticle; BALB/c, Bagg albino; BAS, (basophils); BIL, bilirubin; BUN, blood urea nitrogen; C57BL/6, C57 black 6; C57BL/6 J, C57 black 6, from Jackson Lab; CARS, Coherent Anti-Stoke Raman Scattering; CAT, catalase; CCL-5, chemokine ligand 5; CREA, creatinine; DPBS, Dulbecco phosphate–buffered saline; EOS, eosinophils; GLOB, globulin; GPx, glutathione peroxidase; HCT, hematocrit; H&E, hematoxylin and eosin; HB, hemoglobin; ICAM-1, intercellular adhesion molecule 1; ICR, Institute for Cancer Research; IHC, immunohistochemistry; IL-1β, interleukin-1β, IL-6, interleukin-6; IL-10, interleukin-10; LDH, lactate dehydrogenase; LYM, lymphocytes; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCP-1, monocyte chemoattractant protein-1/; MCV, mean corpuscular volume; MIP-1α, macrophage inflammatory protein 1 α; MIP-1β, macrophage inflammatory protein-1 beta; MON, monocytes; mRNA, messenger ribonucleic acid; NEU, neutrophils; N/R, not reported; PEG polyethylene glycol; PLT, platelets; qRT-PCR, quantitative real time-polymerase chain reaction; RBC, red blood cells; RDW, red cell distribution width; SC, surface coating; SOD, superoxide dismutase; TBIL, total bilirubin; TEM, transmission electron microscope; TNF-α, tumor necrosis factor-α; TP, total protein; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; UA, uric acid; UN, urea nitrogen; WBC, white blood cells.
Effect of AuNPs in Rabbits.
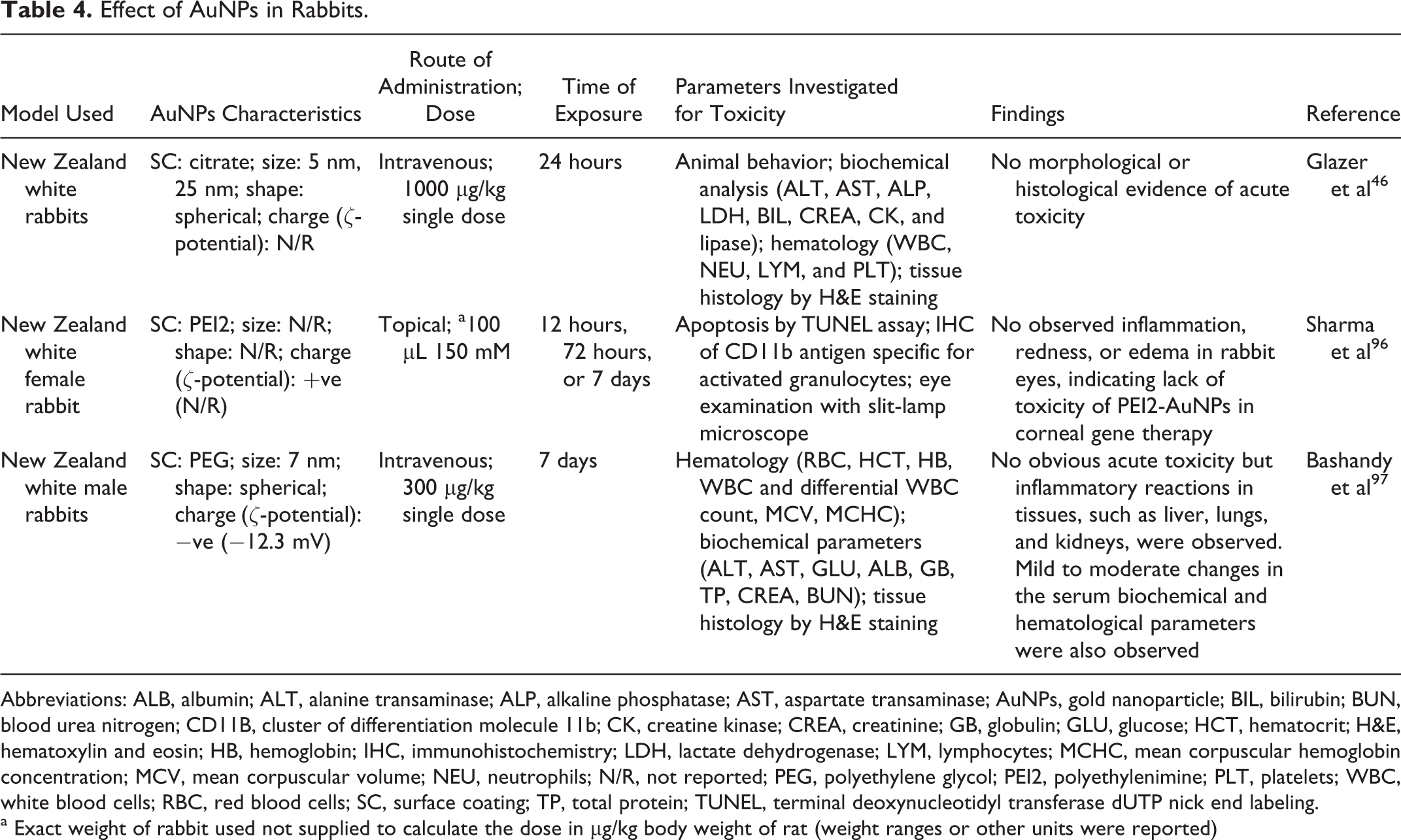
Abbreviations: ALB, albumin; ALT, alanine transaminase; ALP, alkaline phosphatase; AST, aspartate transaminase; AuNPs, gold nanoparticle; BIL, bilirubin; BUN, blood urea nitrogen; CD11B, cluster of differentiation molecule 11b; CK, creatine kinase; CREA, creatinine; GB, globulin; GLU, glucose; HCT, hematocrit; H&E, hematoxylin and eosin; HB, hemoglobin; IHC, immunohistochemistry; LDH, lactate dehydrogenase; LYM, lymphocytes; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; NEU, neutrophils; N/R, not reported; PEG, polyethylene glycol; PEI2, polyethylenimine; PLT, platelets; WBC, white blood cells; RBC, red blood cells; SC, surface coating; TP, total protein; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling.
a Exact weight of rabbit used not supplied to calculate the dose in μg/kg body weight of rat (weight ranges or other units were reported)
Several conflicting results have also been reported with regard to large-sized AuNPs (5 to 100 nm). Chen et al 86 reported that AuNPs ranging from 8 to 37 nm were toxic in mice (Table 3). Similar results were in agreement as shown in Tables 2, 57 3, 49,87 5, 109 while many other results contradict these reports as presented in Tables 3 26,52,90,95 and 5. 43 –45,48 It is therefore still difficult to draw a conclusion on the size of AuNPs that are toxic in vivo.
Effect of AuNPs in Rats.
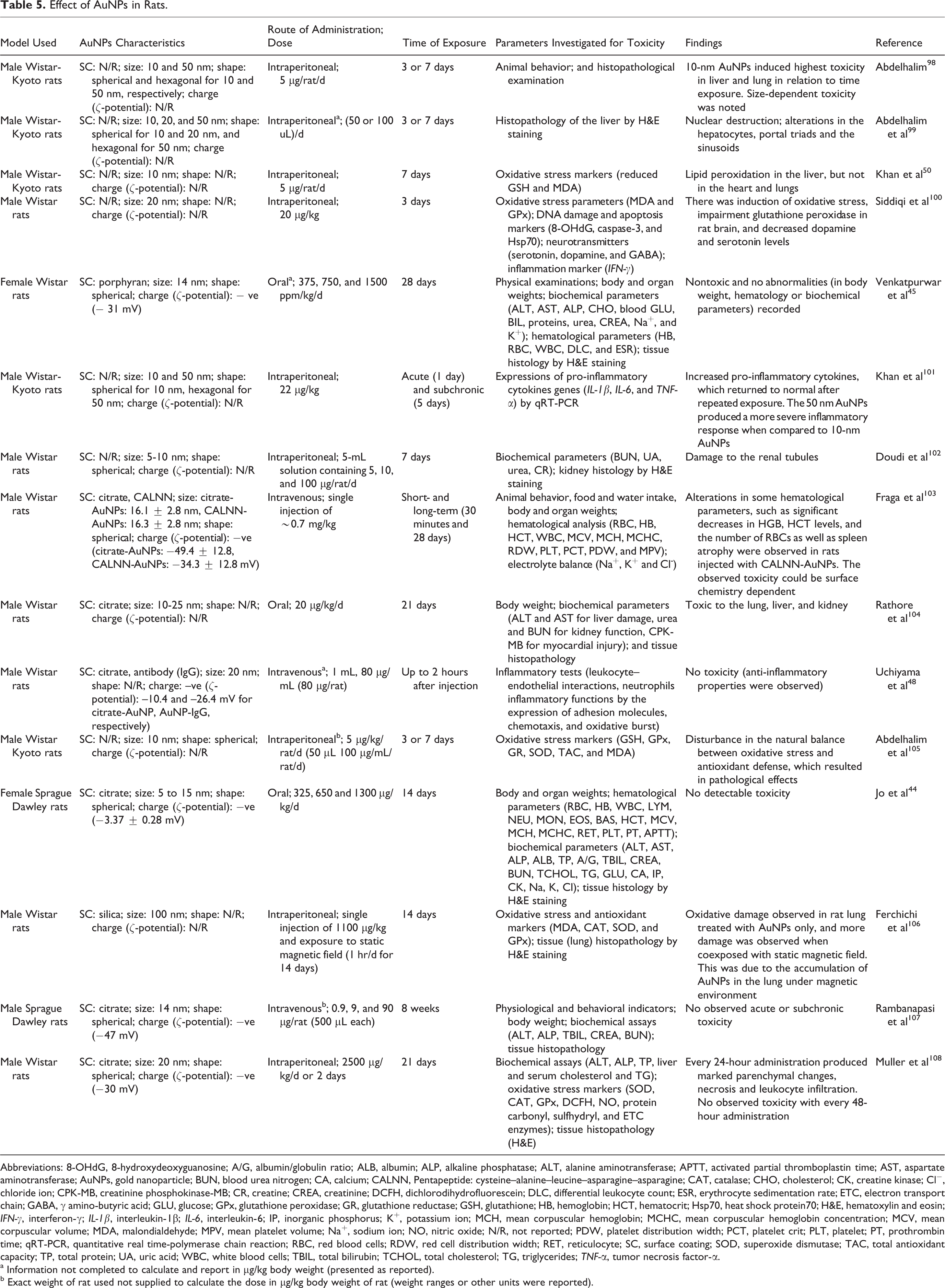
Abbreviations: 8-OHdG, 8-hydroxydeoxyguanosine; A/G, albumin/globulin ratio; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; APTT, activated partial thromboplastin time; AST, aspartate aminotransferase; AuNPs, gold nanoparticle; BUN, blood urea nitrogen; CA, calcium; CALNN, Pentapeptide: cysteine–alanine–leucine–asparagine–asparagine; CAT, catalase; CHO, cholesterol; CK, creatine kinase; Cl−, chloride ion; CPK-MB, creatinine phosphokinase-MB; CR, creatine; CREA, creatinine; DCFH, dichlorodihydrofluorescein; DLC, differential leukocyte count; ESR, erythrocyte sedimentation rate; ETC, electron transport chain; GABA, γ amino-butyric acid; GLU, glucose; GPx, glutathione peroxidase; GR, glutathione reductase; GSH, glutathione; HB, hemoglobin; HCT, hematocrit; Hsp70, heat shock protein70; H&E, hematoxylin and eosin; IFN-γ, interferon-γ; IL-1β, interleukin-1β; IL-6, interleukin-6; IP, inorganic phosphorus; K+, potassium ion; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MDA, malondialdehyde; MPV, mean platelet volume; Na+, sodium ion; NO, nitric oxide; N/R, not reported; PDW, platelet distribution width; PCT, platelet crit; PLT, platelet; PT, prothrombin time; qRT-PCR, quantitative real time-polymerase chain reaction; RBC, red blood cells; RDW, red cell distribution width; RET, reticulocyte; SC, surface coating; SOD, superoxide dismutase; TAC, total antioxidant capacity; TP, total protein; UA, uric acid; WBC, white blood cells; TBIL, total bilirubin; TCHOL, total cholesterol; TG, triglycerides; TNF-α, tumor necrosis factor-α.
a Information not completed to calculate and report in μg/kg body weight (presented as reported).
b Exact weight of rat used not supplied to calculate the dose in μg/kg body weight of rat (weight ranges or other units were reported).
In a study by Zhang et al, 110 it was reported that AuNPs less than 20 nm in diameter are useful for in vivo applications, as these were more readily excreted via the urinary and hepatobiliary systems of the body. There is, however, no guidance what the size of the AuNPs should be after conjugating with different sizes of conjugants. Reported research on size may be complicated by the stability of the conjugating agent or the possible toxicity of the capping agent. More studies are therefore suggested.
Shape
The shape of AuNPs is thought to be an important factor in establishing the cellular or biological responses to AuNPs. 111,112 Gold nanoparticles are presented in shapes, such as nanospheres, nanotriangles, nanoprisms, and nanorods. Other shapes include tetrahedral, sub-octahedral, octahedral, decahedral, icosahedral, multiple twined, and irregular shapes. 113 Spherical AuNPs and nanorods have mostly been studied and compared for toxicity. This may be connected to (1) the popularity of spherical AuNPs with regard to their small size (<100 nm), ease of fabrication, as well as the absorption wavelength in the visible spectral region (500-600 nm), and (2) the superior optical properties of gold nanorods in terms of surface plasmon resonances (longitudinal and transverse), as compared to other gold nanostructures. 114
It was reported that cellular uptake of rod-shaped AuNPs is lower than spherical AuNPs. 115 Moreover, gold nanorods were more toxic to cells in culture compared to spherical AuNPs, 1 as shown in Table 1. 67 This toxicity was reportedly caused by cetyltrimethylammonium bromide (CTAB), which is essential for the synthesis of Au nanorods, using the seed-growth Au nanorod synthesis method. 67,116 The toxicity of Au nanorods can therefore be reduced if coated with other polymer molecules, such as polyethylene glycol (PEG), phosphatidylcholine and poly(acrylic acid). 1,67,117,118
Different capping molecules are used to synthesize AuNPs, and therefore, the majority of articles in the literature comparing toxicity due to the shape may be complicated by different surface chemistry. Schaeublin et al 111 reported the role of the shape of nanoparticles in mediating cellular responses and found that substantial adverse effects were induced by PEG–gold nanorods on cells, whereas exposure of cells to 3–mercaptopropanesulfonate (MPS)–Au nanospheres did not show toxicity, although the surface coatings were different. It was further clarified that the surface coatings (PEG and MPS) were biocompatible. It was also reported that gold nanorods conjugated with PEG or citric acid ligands were less toxic, when compared to spherical AuNPs in human keratinocyte cells and human breast cancer cells (MCF–7), 119 which contradicts the report by Schaeublin et al. 111 In a report by Wang et al, 120 it was shown that CTAB–coated spherical, rods and polyhedral AuNPs resulted in developmental toxicity on zebrafish embryo, in a shape–dependent manner, with spherical AuNPs exhibiting more toxicity when compared to nanopolyhedrons and nanorods. It needs to be noted that CTAB contributes to cell toxicity. Bhamidipati and Fabris 121 compared the toxicity of spherical AuNPs, nanorods, and nanostars, each functionalized with CTAB, PEG, or human serum albumin (HSA). Cetyltrimethylammonium bromide–coated spherical nanoparticles had most cytotoxicity on the cell lines, while PEG– and HSA–coated nanoparticles did not display any shape–related effects. Inherent to differences in shape is a difference in size and or surface area, which can also affect toxicity. More toxicological studies on AuNPs, using several shapes, are thus encouraged.
Surface Chemistry
Gold nanoparticles are coated, stabilized, functionalized, or conjugated with different organic moieties to improve their stability or specificity, thereby forming a protective layer on the surface of the particles and preventing aggregation in biological fluids. 122 Most of these molecules have shown low cytotoxicity, with excellent biodistribution abilities. 36 A number of these agents, including nucleic acids, drugs, antibodies, PEG, 123 small-interfering RNA, 123,124 peptides, 125,126 and methoxypoly-ethylene glycol-graft-poly(L-lysine) copolymer, 122 have been used to facilitate the potential use of AuNPs in nanomedicine. These functionalized AuNPs are used for gene transfection and silencing, targeted drug or gene delivery, 125 intracellular detection, bioimaging, 127 cancer studies, 128,129 and as biosensors. 130 A specific protein, for example, upon adsorption to the nanoparticle surface, can facilitate the uptake of the nanoparticles through receptor-mediated endocytosis. 131
Polyethylene glycol, as a modifying agent, has gained popularity due to its amphiphilic and solubility characteristics, thereby protecting the AuNPs, and ensuring a high degree of biocompatibility and affinity of the nanocarrier for cell membranes. 15,132 Almeida 133 aimed at developing a gene delivery vector, based on the ability of AuNPs to transfect colorectal cancer (hematocrit-116) cells, with enhanced green fluorescent protein expression vector. In this study, no toxicity was recorded with 14 nm AuNPs-based formulations (PEG-AuNPs-functionalized with quaternary ammonium and pEGFP vector [AuNP-PEG-R4N+-pEGFP]), when compared to a commercial transfection reagent, Lipofectamine 2000 (a cationic liposome), 48 hours post-exposure to cells. A non-cytotoxic advantage of antibodies, peptides, and proteins over liposomes was therefore reported. 133
Zhang et al 110 reported that AuNPs coated with high molecular weight (5000 Da) PEG were more stable than the AuNPs coated with low molecular weight (2000 Da) PEG. High molecular weight PEG-stabilized nanoparticles were reported to be the less toxic, 134 and bifunctional PEG derivatives were also reported to be mostly employed for the PEGylation of nanoparticles. 135,136
In a study by Sosibo et al, 132 stabilization of AuNPs with 2 PEG molecules (1% PEG-biotin and 99% PEG-OH (2-{2-[2-(2-{2-[2-(1-mercaptoundec-11-yloxy)-ethoxy]-ethoxy}-ethoxy)-ethoxy]-ethoxy}-ethanol) PEG-biotin (N-(2-{2-[2-(2-{2-[2-(1-mercaptoundec-11-yloxy)-ethoxy]-ethoxy}-ethoxy)-ethoxy]-ethoxy}-ethyl) biotinamide) promote stability and prevent the aggregation of AuNPs. The PEG-OH was used because of its versatility with regard to solubility and biological compatibility, while the PEG-biotin allowed the immobilization of streptavidin—a linker between PEGylated biotin on the surface of AuNPs and biotinylated trans-activator of the transcription peptide. This approach reduced the risk of introducing toxic reagents during the coupling reaction and provides a suitable design of multilayered nanoconjugates for various applications in nanomedicine. Although PEG serves as a promising coating agent for AuNPs, more toxicological studies are suggested.
Polyethylene glycol, especially in large quantities on the nanoparticle, can minimize binding interactions between surfaces and protein targets, which is a key factor to be considered for its potential use in the application of AuNPs as therapeutic agents. The use of glutathione (GSH) as an alternative to PEG in the design of AuNP therapeutics was therefore suggested. Glutathione proved to have a low immunogenicity and is biocompatible. 137 It was earlier reported by Pan et al 51 that 1.1 nm GSH-AuNPs were less toxic than 1.4-nm triphenylphosphine monosulfonate–AuNPs, which upregulate stress-related genes. The GSH-AuNPs, smaller than 3 nm, proved to be highly resistant to bind to serum protein and showed efficient renal clearance. 138
Different degrees of toxicities have been reported regarding the use of other capping, stabilizing or conjugating agents for AuNPs which include citrate, 74 CTAB, 67 sodium borohydride, polyelectrolyte poly(allylamine) hydrochloride, 139 and hydrazinium hydroxide. Researchers have recently focused on the synthesis of nanoparticles via biological methods (green chemistry) and physical methods (laser ablation in liquids).
The biological methods involve the use of biological systems such as plant and plant materials, 42,74,140 bacteria, 141,142 algae, 143 and fungi 144 in the synthesis of AuNPs. Benefits associated with these approaches include simplicity, cost-effectivity and efficiency, eco-friendliness, and low toxicity. 140,142,143
In a study by Vijayakumar et al, 74 the comparative cytotoxic effects of 3 different stabilizing agents (citrate, starch and gum Arabic [GA]) were tested on PC-3 and MCF-7 cell lines using MTT, neutral red cell assay, and LDH assays (Table 1). It was found that when compared to starch- and GA-AuNPs, citrate-AuNPs were cytotoxic at higher concentrations and argued that the toxicity may result from the acidic nature of citrate.
There are limited toxicity studies reported on these biosynthesized AuNPs. Tripathi et al 140 reported the nontoxic effect of spherical, herbal-AuNPs up to 100 µg/mL (average size 70 nm), using leaf extract of Achyranthes aspera, on cultured splenocyte cells. It is therefore suggested that more detailed toxicity studies, with improved production or synthesis of biosynthesized AuNPs, must be done so as to address some of the earlier reported disadvantages of this approach, which include particle aggregation, nonuniform shapes with biological materials, and poor monodispersity. 119
The physical methods (such as laser ablation in liquid), on the other hand, provide a free-ligand synthesized nanoparticles, due to possible interference of ligands with toxicological assays and particle properties. 145 This method has several advantages, such as simple procedural steps, high purity-synthesized colloidal nanoparticles free of chemical reagents (free-ligand), and excellent colloidal stability of nanoparticles. 146,147 The stability of ligand-free AuNPs (without stabilizing agents or ligands) in a variety of media may result from the partial oxidation of the gold surface during the process of ablation, thereby resulting in positively charged AuNPs. 147
Taylor et al 148 reported that cultured bovine immortalized cells (GM7373) co-incubated with 15 nm ligand-free AuNP (50 μM Au) for 2, 24, and 48 hours produced no toxic effects up to a gold concentration of 25 μM.
In a study by Tiedemann et al, 147 AuNPs were synthesized by pulsed laser ablation of a gold wire in liquid flow. After the synthesis, the AuNPs were coated with bovine serum albumin (BSA) to prevent the formation of protein corona upon exposure to biological proteins. No adverse effects were recorded after 2-hour incubation of porcine spermatozoa with BSA-coated AuNPs (20 nm, 10 µg/mL). Also, no sign of toxicity was observed when porcine cumulus–oocyte complexes was exposed to 6 nm and 20 nm AuNPs-BSA (10 µg/mL) and 6 nm BSA/citrate-coated AuNPs (10 and 30 µg/mL).
In another study, spherical AuNPs were synthesized by femtosecond laser ablation in 3 different liquid media (AuNPs prepared in pure deionized water, PEG, and dextran solutions). The toxic effects of these nanoparticles (0.1 μg/L to 10,000 μg/L) were investigated in human glioblastoma cell line (U87-MG) and neuroblastoma cell line (SK-N-SH) after 72 hours. It was reported that the AuNPs were biocompatible and did not induce any obvious cytotoxicity. 47
It should thus be noted that although the ligand-free AuNPs resulted in the absence of possible interference of ligands with toxicological assays and particle properties, the following points are also to be considered: (1) The ligand-free AuNPs need to be stabilized with agents, such as BSA, before exposing them to the biological environment. This is to prevent the adsorption of several proteins to the surface of the nanoparticles, which could form protein corona. 145,147 This corona could influence the biological nature of the particles. (2) In designing AuNPs as a drug carrier or delivery for therapeutic or diagnostic purposes, which requires conjugation with capping or stabilizing or coating or conjugating agents, it is important to investigate the biocompatibility and toxic effect of these agents, which are important in the synthesis of the bioconjugating nanomaterials; and (3) the capping agents introduced to the free-ligand AuNPs seem to influence the size of the AuNPs. 47,149 Nevertheless, there is no method that can be regarded as the best in the synthesis of AuNPs since each of these methods have several disadvantages.
Surface charge
The effect of surface charge was reported by Goodman et al, 150 where 2-nm cationic gold particles were moderately toxic, whereas the anionic counterparts were non-toxic in cells at the same concentration. The difference was related to the interactions of the cationic nanoparticles with the negatively charged cell membrane, thereby leading to membrane disruption. In another study, Kim et al 52 reported that the surface functionalization of 1.3-nm AuNPs with a cationic ligand monolayer N, N, N-trimethylammonium ethanethiol (TMAT-AuNPs) played a significant role in the toxicity results observed in the zebrafish embryos. It is, however, difficult to blame only the surface charge for the toxicity since these molecules were smaller than 1.3 nm, and molecules smaller than 5 nm seems to be more toxic (see discussion earlier under size section).
The effect of charge was also observed in zebrafish embryo, when exposed to 1.5 nm AuNP, but with different surface charges. It was found that neutrally charged 2-(2-(2-mercaptoethoxy)ethoxy)ethanol (MEEE)-AuNP did not cause behavioral defects, but negatively charged 2-mercaptoethanesulfonic acid (MES) resulted in larval behavioral abnormalities, which persisted into adulthood, while the positively charged TMAT-AuNP caused 100% death rate at the same concentration. 151
Boyles et al 53 showed that positively charged nanoparticles (7 nm) are taken up into the cell (via endocytosis) much faster than the negatively charged nanoparticles (10 nm). It is postulated that the charge of nanoparticles plays a role in the intracellular trafficking and effect on nucleus; however, the same effect is not consistently observed by all positively charged nanoparticles. Positively charged nanoparticles also seem to have a higher incidence of exocytosis which can initiate inflammation within an organism. 53 It remains very difficult to specify the effect of charge because it is impossible to have the same size of nanoparticles with the same capping agent having 2 different charges.
Aggregation State
Aggregation of nanoparticles influences cellular toxicity. 152 Aggregation of AuNPs could be influenced by a number of factors, such as charge, size and surface chemistry. 153 It was reported that cationic and oligocationic species resulted in particle aggregation, 154 while negatively charged AuNPs repel each other and inhibit aggregation. 155 A number of coating agents, including lipids 152 and polymers, 156 and the presence of BSA and proteins in cellular medium, prevent the aggregation of nanoparticles. 152,153 More studies investigating the toxicity of nanomaterials in relation to aggregation is encouraged.
Interactions With Physiological Media and Biological Fluids
The physicochemical properties of nanoparticles can be influenced by the immediate environment. 157 Nanoparticles interact with the components of a typical cell growth medium, consisting mainly of serum proteins, essential amino acids, vitamins, ionic salts, and other chemicals, including trace metals and antibiotics. These constituents influence the physiochemical properties, such as surface chemistry, surface charge, aggregation state, 2 and the hydrodynamic behavior of nanoparticles. 153 Alkilany and Murphy 2 reported that growth media proteins adsorb onto the surface of AuNPs and alters the effective surface charge, thereby increasing the hydrodynamic radius of these particles. Further, it was reported that the presence of a high concentration of electrolytes in media results in the destabilization of nanoparticles, 158 which could have an influence on the ability of AuNPs to interact with cells. 2
On exposure to biological fluids, AuNPs interact with various biological molecules, such as proteins, nucleic acids, lipids, and polysaccharides. It has been known that the formation of protein corona on exposure of AuNPs to biological fluid or media is due to the presence of proteins. In a review, Wang et al 159 pointed out that proteins, such as ubiquitin, TNF, serum albumin, polypeptides, fibrinogen, or cytochrome C, can easily be adsorbed onto the surface of AuNPs to form a protein corona. On interaction, the physicochemical properties of both AuNPs and proteins change and can induce a number of physiological and pathological changes, which include blood coagulation, aggregation of proteins, complement activation and configuration of bound proteins. 159 A number of effects were associated with the formation of a protein corona, which include (1) changes in the structures of adsorbed proteins 160 ; (2) loss of original targeting capabilities of proteins 161 ; and (3) induction of several cellular responses, such as activation of caspase-related pathways, apoptosis, increased lysosomal permeability, or inflammatory responses. 159,162
Formation of protein corona has significant effect on targeting ability, cellular uptake, accumulation and toxicity of nanoparticles in living systems, 77 since the formation of protein corona has the tendency to change the biological effects of the nanoparticles. 163 The structure and composition of protein corona is largely controlled by the surface coating. 39 It was reported that protein corona formation is minimized by functionalizing agents, such as coating of AuNPs with GSH and PEG, as these reportedly shield the surface of nanoparticles from serum protein binding (opsonization). 164,165 The direct role of protein corona on the toxicity of AuNPs, therefore, cannot be explained without reference to surface chemistry as well as many other factors that influence the formation of the complex (nanoparticle-protein corona). These include surface charge, size and shape of the AuNPs. 39,165,166
Different physiological results can be obtained from nanoparticles of different chemical properties. In a study by Yang et al, 39 a comparative toxicity profile of 11.4 to 13.3 mg/kg silver nanoparticles (AgNPs) and AuNPs (with identical surface coating) was investigated after intravenous injection in mice. Silver nanoparticles showed greater changes in gene expression (apoptosis, ion transport, and oxidative stress) when compared to AuNPs group, although no definite signs of toxicity were reported with physiological, biochemical, and histological evaluations between the nanoparticles. Diverse protein corona was formed on the surface of these nanoparticles, which could be linked to intrinsic properties of the nanoparticles. 39 It can therefore be supported that toxicity of nanoparticles is greatly influenced by chemical composition of nanoparticles.
Sex Differences
In in vivo models, the toxicity of AuNPs may be influenced by sex differences. Further, there are some factors to be considered when choosing sex of an in vivo model. These include (1) larger percentage of fat in relation to the total body weight of females than males; (2) the anatomical differences in the reproduction system and associated hormone cycle differences between males and females; and (3) faster total clearance of most substances in males than females. 167
In a study by Chen et al, 93 no significant toxicological responses were noted in the reproductive system in both male and female mice upon intraperitoneal injection of 200-μL PEG-AuNPs (Table 3). They reported that AuNPs caused more serious liver toxicity and infection in male mice than female mice. It was thus suggested that sex differences may be one of the important elements of in vivo toxicity of AuNPs. 93
Dose
The total concentration of AuNPs administered to an organism at a specific duration of exposure could influence the toxicity of the particles. Various conflicting results have been reported on the dosage of AuNPs. In fact, there has been no reported safe dose of these particles without emphasis on other factors, such as size, shape, and route of administration. Since the doses used in animal studies were generally higher than the actual doses humans are exposed to, and since nanoparticle pharmacokinetics is dose-dependent, a low-dose model, as shown in Table 3, 58 is therefore important. 168,169 The dose in a rat model for low and medium doses are ∼0.01 and ∼1.0 mg/kg (10 and 1000 µg/kg), respectively, and was therefore extrapolated to simulate low-dose kinetics of AuNP, 109 since pharmacokinetics is dose-dependent. Based on this, the results of Lin et al 170 provided a scientific basis for researchers to select the most appropriate animal model and dosing paradigms for conducting future nanomaterial studies so as to increase the research relevance to humans. In choosing dose for toxicity studies, the purpose of application of AuNPs should be considered.
Routes of Administration
The administration route of AuNPs could influence the toxicity of the particles. Injection via the tail vein (intravenous) is the most promising route for AuNPs because it is a superficial and easily accessible procedure for animals and showed the lowest toxicity for AuNPs, as reported by Zhang et al 58 (Table 3).
Oral delivery of nanoparticles may have an effect on the mucosal cells of the gastrointestinal tract as a result of morphologic and physiologic absorption barriers. 171 Zhang et al 58 reported that there was no significant damage to the stomach, which indicates good absorption of AuNPs. A slight damage to the intestine was also reported, which may be an indication of the toxicity of the AuNPs. 58 No research reports could be found on the effect of the digestive enzymes on the surface coatings of the nanoparticles; however, one expects that nanoparticles will be subjected to relevant digestive activities in the gut until they are taken up.
Intraperitoneal injection was reported to be less toxic than oral administration at the dose of 1100 µg/kg. 58 It is generally accepted that due to the dense blood vessels and lymph in the murine peritoneum, drug absorption by intraperitoneal injection is rapid. More research is therefore suggested to test the toxic effects of AuNPs via intraperitoneal administration.
Uptake, Biodistribution, and Accumulation of AuNPs
Nanoparticles are generally taken up in a similar manner to biomolecules, depending on the size, shape, surface charge, surface functionality, dose, species, and chemical composition. 38,172 –174 In the liver, these particles are recognized by the Kupffer cells and internalized by endocytotic pathways, such as macropinocytosis, receptor-mediated (clathrin-mediated), and caveolin-mediated endocytosis. 173
It was earlier reported that the distribution of AuNPs in vivo is closely related to toxicity. 175 These nanoparticles are distributed to various organs and tissues before elimination and is largely dependent on the physicochemical properties including size, shape, surface charge, surface modification, and route of administration. 38,176 –178
Different sized nanoparticles (10, 50, 100, and 250 nm) accumulate in the liver after 24 hours, which could be linked to the high perfusion of blood through it. The 10-nm sized AuNPs distribute to the blood, spleen, liver, kidney, lung, testes, heart, thymus, and brain in rats, while the larger particles (50, 100, and 250 nm) were limited to the blood, liver, and spleen. 176 Intravenous administration of 13 nm-sized PEG-AuNPs to mice were distributed primarily to the liver and spleen 5 minutes to 7 days post injection. This presented with acute inflammation and apoptosis in the liver in a dose-dependent manner. 87
The liver and spleen presented with the highest levels of accumulation post intravenous injection (24 hours up to 2 months), 38,109,107 suggesting that the clearance mechanism of AuNPs could be linked to the hepatobiliary system. 107 This can thus be attributed to the fact that nanoparticles are taken up by reticuloendothelial organs (liver and spleen), 179 with the spleen having a higher filtering efficacy, and both organs having a high number of phagocytic cells and capillary beds. 127,177
It should be noted that many reported toxicological studies have not directly linked the effect or toxicity of AuNPs to tissue distribution and accumulation, but rather to the several physicochemical properties exhibited by the AuNPs. 38,103 This suggests that uptake, biodistribution, and bioaccumulation studies on AuNPs provide more information on the applications of AuNPs in diagnosis and therapy rather than toxicity, as AuNPs are distributed and accumulate in various tissues and body fluids for short or extended periods without inducing toxicity. 34,103,107,175,180 Due to the extended exposure of the liver and spleen to the nanoparticles, inflammation, connective tissue formation or the presence of a large number of phagosomes should be investigated in toxicity studies of chronic exposure to nanoparticles.
Clearance and Excretion
Nanoparticles with optimum clearance conditions tend to reduce the risks of toxicity, as a result of the reduction in the duration of exposure to the body. Elimination routes include (1) renal excretion (urine), which involves the filtration of small particles (<6-nm spherical particles) within hours to days post administration, through the glomerular capillary walls of filtration-size threshold ranging from 6 to 8 nm; (2) hepatobiliary excretion (bile to feces), which allows excretion of nanoparticles (few hours to weeks after administration) greater than the glomerular filtration threshold (>6 nm); (3) the reticuloendothelial system, which involves the trapping of non-degradable nanoparticles in the body for a prolonged period (>6 months) due to their stability and inability to be digested by enzymes located in the lysosomes of macrophages. 165,173
Choi et al 181 reported that nanoparticles (quantum dots) with diameters <5.5 nm are easily excreted via the kidney. This is because the filtration-size threshold of glomerular capillary walls is typically 6 to 8 nm, and particles with hydrodynamic diameter smaller than 6 nm can undergo rapid urinary excretion. 165,173,181 It was found that 1.9-nm AuNPs were cleared via the kidney and excreted 5 hours after intravenous injection in mice, 182 indicating that these particles are small enough to cross the glomerular filter.
Larger nanoparticles (>6 nm) in the systemic circulation, which were not excreted in urine, can be eliminated from the blood by the reticuloendothelial system. These are accumulated in the liver and spleen, or entrapped by the mononuclear phagocyte system, which could be cleared via hepatobiliary system or be retained in the body for a long time. 2,173,183 Sadauskas et al 184 reported an overtime elimination of 40 nm AuNPs primarily by the Kupffer cells of the liver and found the nanoparticles inside the lysosome/endosome-like vesicles in a clustered manner. This is a normal physiological way the body can neutralize unfamiliar/unwanted particles without any side effects, as long as there is not an overload. Based on their extrapolated data, it was therefore concluded that most of the AuNPs remain in the liver of the mouse, even for the normal full span, since the clearance of these particles may be very slow. Based on their extrapolated data from their mouse studies, Sadauskas et al 184 concluded that most of the AuNPs remain in the liver even for the normal full lifetime since the clearance of these particles may be very slow.
Surface chemistry is another factor that can influence the excretion of AuNPs. It was found that 20 nm PEG-AuNPs were cleared from the blood without further accumulation in the liver and spleen. 185 In a study by Zhang et al, 186 it was reported that 36% Au in GSH (2.1 nm)- and 1% Au in BSA (8.2 nm)-protected Au nanoclusters were excreted by urine after 24 hours. Also, 94% and <5% Au in GSH- and BSA-Au nanoclusters, respectively, were metabolized by renal clearance after 28 days. This resulted in a significant reduction in the toxicity of GSH-Au nanoclusters, whereas the BSA-Au nanoclusters accumulated in liver and spleen leading to irreparable toxicity response. It can therefore be concluded that the size of nanoparticles and surface chemistry contributes to its elimination from the body.
Models Previously Used to Screen the Toxicity of AuNPs
Looking at various in vitro, in vivo, and in ovo models (Figure 2) used to screen for toxicity of AuNPs, several researchers have reported the safety of naked or conjugated AuNPs, 26,41 –48 as well as the toxicity. 49 –57
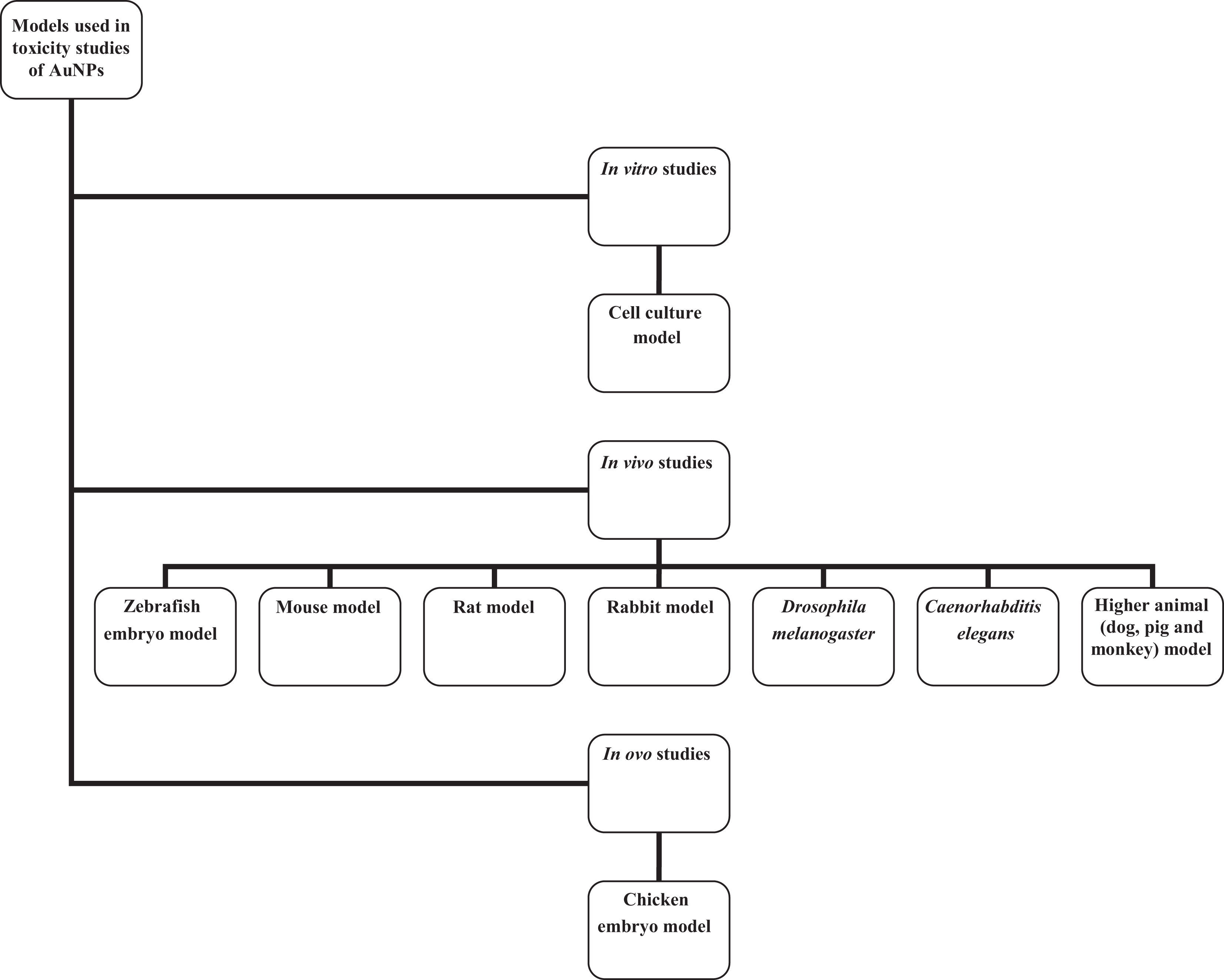
Models used in screening the toxicity of gold nanoparticles (AuNPs).
In Vitro Assessment of the Toxicity of AuNPs
Most of the toxicological studies are conducted in vitro in immortalized cell lines, using standard toxicological assays, such as the MTT assay and the LDH assay, which measure the mitochondrial enzymatic activity and disruption of the cell membrane, respectively, 56 CFE, which measures the ability of a single cell to form a colony, 60 as well as other tests, including carboxy-2’,7’-dichlorofluorescein diacetate assay and water-soluble tetrazolium-1 assay, which measures reactive oxygen species and cell proliferation, respectively. 44
The high proliferation rate of cell lines, simple procedures (which can be automated), low costs, short experimental period, and precise applied concentration of testing materials make cell-based screening tests popular. 187
Cell-based toxicity-screening tests
Cell culture assays are suitable for the study of basic mechanisms that include endocytosis, death pathways, and the interactions between AuNPs and subcellular components. A number of cell lines have been used to assess the effects of AuNPs 44,45,188 (Table 1).
The various toxicities observed are dependent on the size, charge, surface coating, assay type, and time of exposure (Table 1). For instance, large-sized citrate-AuNPs (>5 nm) were not toxic to cells—the negative charge on the citrate causes the particles to repel the negatively charged cell membranes. 53 Small-sized AuNPs are obtained from a higher concentration of sodium citrate used in the synthesis, 69,189 leading to increased acidity of the citrate-AuNPs and hence toxicity. It is suggested that the amount of sodium citrate be reduced to the level at which the stability and safety of AuNPs are guaranteed, depending on the desired size of the AuNPs under investigation. Surface modifications of AuNPs with agents, such as biomolecules, ligands, and polymers, also contribute largely to the reduction in the toxicity of AuNPs, irrespective of the size. This could result from the exchange of citrate on the surface of AuNPs with these ligands or biomolecules. 51,190
The results of cell-based toxicity could also be influenced by the type of assay employed. 76 The majority of toxicity assessments were done with MTT assays. Gold nanoparticles absorb light in the visible region (14 nm at 520 nm in milli-Q water and at 557 nm in RPMI), which means that the AuNP may interfere with colorimetric, fluorometric, and luminometric tests. 59
Results obtained from the LDH studies suggest that the U87 cell line might be more resistant to AuNP-induced toxicity when compared to fibroblasts and that, most likely, fibroblasts undergo nanoparticle-induced necrosis, as opposed to what was observed in U87 cells. 121 This means that the outcome of toxicity tests may be different for different cell lines.
Generally, the disadvantages of using cell-based assays include (1) the genome and the proliferation pattern of commonly used tumor cell lines (which are the mostly studied) are not comparable with normal, healthy cells and (2) the metabolism, routes of exposure, and organ-specific toxicity of intact animals cannot be mimicked in cell culture. 187 This approach does not represent the complexity and defense system of the whole organism, as it only reveals the toxicity at a cellular level, 191 which may not correspond to animal-based results. 38 In view of this and other reasons, animal-based models should be more emphasized in the final analysis of AuNPs toxicity, 38 although the cell culture model provides initial basic toxicity information.
Human embryonic stem cells
Apart from several reports on common cell lines used in the study of the toxicological behavior of AuNPs, a study using human embryonic stem cells (hESCs) has also been reported (Table 2). Senut et al 56 reported the advantages of using hESCs with reference to other (eg. animal-derived) systems, some of which are (1) the unlimited self-renewal capabilities and continuous growth in culture; (2) the ability to differentiate into various specialized cell types over an extended period of time; and (3) the ability to prevent interspecies variability, which makes this a better model in predicting human responses to toxins.
Senut et al 56 reported the effect of different conjugating agents with AuNPs of varying sizes (1.5 nm thiolate-, 4 nm mercaptosuccinic acid (MSA)- and 14 nm citrate-AuNPs) on hESCs. It was found that the cells exposed to 1.5 nm naked AuNPs (0.1 μg/mL) disintegrated into single cells within 48 hours, suggesting cell death; 1.5 thiolate-AuNPs (0.1 μg/mL) was toxic to hESC; 4-nm MSA-AuNPs showed a decrease in DNA methylation and a corresponding increase in DNA hydroxymethylation, while the 14-nm AuNPs showed no significant toxic effects on the hESCs at concentrations as high as 10 μg/mL. This can be related to size- and surface chemistry–dependent toxicity, as small-sized particles presented adverse effects on hESCs, with no significant toxicity with citrate-capped AuNPs.
This is the first report on the use of hESCs to evaluate the toxicity of AuNPs. It is therefore suggested that more studies be performed on hESCs using different concentrations and surface groups.
Determination of the In Vivo Toxicity of AuNPs
The pharmacokinetics and safety of AuNPs should be evaluated in laboratory animals prior to application in humans. It is thus crucial to understand interspecies differences in the pharmacokinetics of nanoparticles before generated data in laboratory animals can be used to establish comparisons in humans. 170
Studies using the zebrafish model
The zebrafish (Danio rerio) is a widely used vertebrate model for toxicity studies. Various characteristics, such as small size, easy maintenance, transparency of embryos, and the requirement of low amounts of testing compounds, make zebrafish suitable for the use as a toxicity-screening model. Several zebrafish embryos can easily be acquired, where critical organs may be fully developed within 7 days. The larvae are transparent, thereby enabling online visualization of the internal organs and tissues. 192
Studies have indicated some similarities between zebrafish and humans, including: (1) diploid genome structure; 193,194 (2) heart rates, action potential shape and duration, as well as electrocardiogram morphology; 195 and (3) drug metabolism—similar phase I and phase II metabolism, which may be attributed to the highly conserved genetic expression profiles in the liver. 196,197
The zebrafish model, although more complex than cultured cell models, is invasive and less expensive than large-scale biocompatibility studies in rats or mice. 198 Few studies have, however, been reported using this model (Table 2).
In Table 2, mortality of the zebrafish embryos was reported when AuNPs were conjugated to TMAT, but showed no adverse effects with MES- and MEEE-AuNPs. The toxicity could be a result of TMAT itself or the positively charged nanoparticles to the zebrafish embryos. On the other hand, the positively charged nanorods showed no mortality on zebrafish adults; however, the zebrafish embryos and zebrafish adults represent different models. Larger-sized citrate-AuNPs (>5 nm) can be considered nontoxic as observed with adult zebrafish. The study by Sangabathuni et al 83 also showed no toxicity whether AuNPs were spherical, rods or nanostars, even with positively charged nanorods. The zebrafish studies have the advantage of a longer exposure period (20 days) to be studied.
The zebrafish model is, however, not a representative replacement for other in vivo models, such as mice and rats, but has been shown to be highly suitable as an intermediate screening tool between preliminary toxicity assessment in cells and more conclusive and translationally relevant follow-up toxicity assessments in higher organisms. 198
Studies using mice as an in vivo model
Mice, another in vivo model used in toxicity screening of AuNPs, have several advantages. These include closer similarities to human in all mammalian organs and physiological features, possibilities of performing pharmacokinetic and toxicokinetic studies, as well as easy access and possibilities of withdrawing excess blood for analyses. 199 Conflicting results have been reported on the toxicity of AuNPs, which could be due to differences in size, surface chemistry, surface charge, dose, and route of administration, using the in vivo mouse model (Table 3).
Table 3 shows the difficulty to make conclusive remarks on toxicity of nanoparticles because some criteria that contributes to toxicity are not always stated, like the charge of the AuNPs. Chen et al 86 have also reported that citrate-AuNPs larger than 5 nm, but smaller than 50 nm, had fatal effects on the mice, but nanoparticles of 3, 5, 50, and 100 nm did not show toxicity. The authors gave no possible explanations, neither was the charge of the molecules mentioned. It is known that citrate concentration (as well as temperature) during synthesis determines the size of molecules but also the charge affects toxicity (as explained earlier). According to Table 3, nanoparticle concentrations up to 400 μg/kg/d are safe even at daily injections for 12 weeks. Considering the route of administration, oral, intraperitoneal, and intravenous administrations seem to be acceptable if dosages are not higher than 400 μg/kg/d, although with an unsafe dosage, intravenous injections were the least toxic. 58 This contradicts the report of Cho et al, 88 where toxicity was observed at the genetic level, 30 minutes after intravenous injection of PEG-AuNPs, however, in this study, the dose was 4260 μg/kg.
Studies using rabbits as an in vivo model
The toxicological evaluation of AuNPs using the rabbit model (Table 4) has been reported. Sharma et al 96 suggested the safe use of polyethylenimine-conjugated AuNPs for the cornea and for corneal gene therapy because of observed normalcy in rabbit eyes.
Studies using rats as in vivo model
Rats and pigs are more appropriate representative models of humans than mice in animal-to-human extrapolation of AuNPs pharmacokinetics, although the dose, size, age, and/or species/strain/breed are factors as well. 170 The spleen capillaries in rats and humans are both sinusoidal, where a large volume of blood flows through open-circulation routes with NP filtration at interendothelial cell slits, unlike the mouse spleen capillaries that are non-sinusoidal. 200 Further, the liver capillaries of mice, rats, pigs, and humans are all sinusoidal with open fenestrae, but the average number of fenestrae per square micrometer in mice is lower than that of rats and humans. 201
In addition, there are other parameters, such as surface coating, corona formation, as well as physiological parameters, including organ weight fraction of body weight and plasma volume fraction of body weight, which greatly affect the pharmacokinetics of NPs. 202 In most physiological parameters, rats are closer to humans than mice. 170 Several researchers have reported the toxicity of AuNPs using the rat model (Table 5).
It can be deduced that citrate-capped AuNPs between 5 and 20 nm were non-toxic to rats via intravenous administration at low doses (Table 5).
A study investigating species differences in the toxicity of spherically shaped 15-nm PEG-AuNPs (1000 mg/kg body weight), with ζ-potential −34.43 ± 2.71 mV, between mice (female BALB/c) and rats (female F344) 28 days post intravenous injection was reported by Bahamonde et al. 203 It was observed that there were no differences between the serum biochemical parameters, such as ALT, AST creatinine, urea nitrogen, and glucose in both species, which suggested no metabolic dysfunction or liver and kidney injury. There was, however, a notable macrophage response (increased serum levels of interleukin-18) 7 days post injection and granulomas in the liver of mice injected with the AuNPs. Mortality of some rats (3 of 19) 24 hours post injection was recorded, with no reported cause of death with necropsy. These indicate species-specific differences in the toxicity profile of AuNPs. 203
Studies using Drosophila melanogaster as an in vivo model of toxicity
The fruit fly, Drosophila melanogaster, is a well-established insect model for human disease. 204 –206 Pompa et al 206 studied the effects of 15-nm citrate-capped AuNPs using 6 concentrations (1.9, 3.8, 19, 38, 190, and 380 pmol/L) in the D melanogaster upon ingestion. It was observed that AuNPs caused DNA fragmentation, significant overexpression of the stress proteins, as well as significant reduction of their life span and fertility. This is the only report in this model to explore the toxicity of AuNPs.
Studies using Caenorhabditis elegans as in vivo model
In a study by Hu et al, 207 Caenorhabditis elegans was exposed to different composition of AuNPs (unfunctionalized AuNPs, as well as 0.5, 1, and 3 MUA-AuNPs of ratios 1:2, 1:1, and 3:1 MUA: Au, respectively) with sizes 6.45 ± 1.58, 1.83 ± 1.21, 1.26 ± 0.25, and 0.80 ± 0.12 nm, respectively. Greater reduction in worm body size, brood size, mobility, and worm population were observed within 72 hours with the MUA-AuNPs when compared to the unfunctionalized AuNPs. Viability (MTT assay) of cultured primary neurons of C elegans when exposed to MUA-AuNPs resulted in reduced neuronal survival with increasing MUA-Au ratios. This indicates the role of surface modification in the toxicity of AuNPs.
Studies using higher animals as in vivo models
Other in vivo models, such as pig (swine), monkey, and dog (canine) models, have been used in studies involving potential application of AuNPs in nanomedicine. 208 Inadequate toxicological studies on AuNPs have been reported using these animals. Some studies, however, have reported evidence of toxicological behavior of AuNPs in these animals during investigation for use in biomedical application. For example, no short-term toxicity was observed in dogs with prostate cancer following a single intralesional injection of radioactive AuNPs coated with GA-AuNPs, 104,209 Gum arabic–coated AuNPs (GA-AuNPs, 12-15 nm), used as X-ray contrast agent for tumor imaging, was intratumorally injected to mice with breast cancer and to a dog presented with thyroid carcinoma and osteosarcoma and induced no toxicity. 210 There was also no noted toxicity in pig intravenously injected with GA-AuNPs (15-20 nm). 211 The non-toxic effect of GA-AuNPs (both radioactive and non-radioactive analogue) reported in these studies is linked to the presence of GA, a naturally occurring glycoprotein, on the nanoparticles. 209,211
The inadequate toxicological profile of AuNPs in higher animals may be due to high cost when compared to mice, rats, zebrafish and rabbits. Lower animals, such as mice, have, however, been used to extrapolate the non-toxic dosage for primates. For example, monkey dose can be obtained from mouse dose by dividing the no-observed-adverse-effect-level of mice by 4. 212
In Ovo Assessment of the Toxicity of AuNPs
Studies using chicken embryo as a model of toxicity
Chicken embryo has a rapid growth rate and is independent of the mother. 213 The embryo is highly sensitive to harmful factors 214 and has therefore been used as a model for cancer therapy research, 215 and the development of new drug delivery systems. 216 Only a few studies have recently been reported using the in ovo model (Table 6). The model is easily implemented and cost-effective, as it allows limited use of animals, but impossible to perform pharmacokinetic studies and no possibility of long-term studies when compared to in vivo model. 219
Effect of AuNPs on Chicken Embryos.
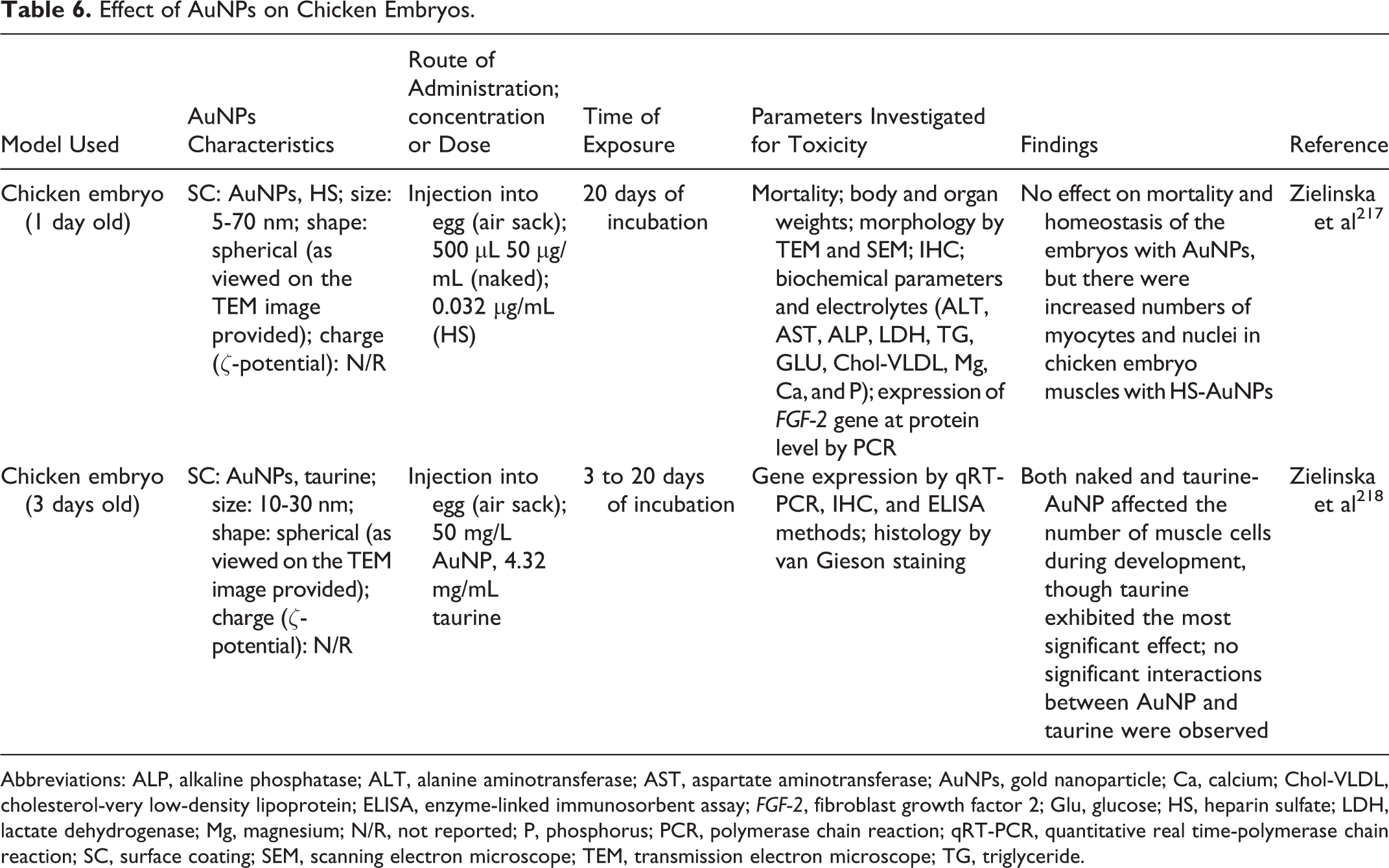
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; AuNPs, gold nanoparticle; Ca, calcium; Chol-VLDL, cholesterol-very low-density lipoprotein; ELISA, enzyme-linked immunosorbent assay; FGF-2, fibroblast growth factor 2; Glu, glucose; HS, heparin sulfate; LDH, lactate dehydrogenase; Mg, magnesium; N/R, not reported; P, phosphorus; PCR, polymerase chain reaction; qRT-PCR, quantitative real time-polymerase chain reaction; SC, surface coating; SEM, scanning electron microscope; TEM, transmission electron microscope; TG, triglyceride.
Further research was suggested to elucidate the mechanism of muscle fiber enlargement, 217 as this is a model that serves as an intermediate between in vitro and in vivo studies (due to reduction in number of animals used in in ovo) 219 to investigate the toxicity of AuNPs.
Conclusion
Gold nanoparticles have shown promising applications in the field of nanomedicine. This review aimed to give perspective on the current safety knowledge on the safety of AuNPs. Many conflicting results have posed serious concerns regarding their safety. This may be connected to the lack of standard protocols addressing the safety investigations. Cell lines are most often used, but inconsistency in experimental design and reporting of certain important factors such as charge, shape, or coating surface of nanoparticles make it impossible to draw a final conclusion in which molecules are most likely to be toxic. Although cell lines provide important information as an early screening tool, it needs to be kept in mind that in the body, cells are not exposed to nanoparticles for only 24 to 72 hours. According to animal studies, AuNPs are removed from the circulation and accumulate mainly in the liver and spleen.
To improve AuNP toxicity studies, the following are suggestions based on the research results: (1) CFE assay, a non-colorimetric and non-fluorescent assay method, is suggested as the first-line in vitro toxicity-screening test of AuNPs, since it has higher sensitivity when compared to other in vitro assays. Most in vitro studies centered on diseased (tumor) cells, which provide insight into the therapeutic potential of AuNPs; studies involving normal, healthy cells are encouraged for toxicological studies. (2) Since the aim is to use AuNPs in medicine, it can be suggested that more in vivo studies be conducted to address the complexity and interaction of different organs and to extrapolate to humans. Of the different kinds of in vivo models previously used, the rat model could be the best option since rats are more closely related to humans in several pharmacokinetics parameters. (3) The sex difference in toxicity still needs to be established in the rat model. The liver of male mice seems to be more vulnerable to AuNPs toxicity and infection than female mice, but the average number of fenestrae per square micrometer in mice is lower than that of rats and humans, and therefore, studies are needed to establish the preferred sex for toxicity studies in rats. (4) A dose model for rats ranging between ∼0.01 and ∼1.0 mg/kg is suggested, although other parameters, such as size, surface chemistry, and biological media of suspension of AuNPs, should be considered. It is recommended that authors should report uniform dose in relation to the weight of the animal used (in vivo) and provide full information about the nanoparticles administered, in form of the surface area, mass or molar concentration, as well as the volume. (5) Intravenous administration of AuNPs appears to be the safest route of administration. (6) The AuNPs between 5 and 100 nm appear to be the least toxic. (7) Negatively charged AuNPs appear to be non-toxic compared to positively charged AuNPs. (8) It is most likely that PEG-AuNPs-conjugated diagnostic or therapeutic agents, such as peptides, can prevent the toxic effects observed with PEG. Toxicity studies on 1 or 2 PEG molecule(s) 132 are suggested to ascertain its potential use as a stabilizing agent in designing multilayered nanoconjugates for nanomedicine applications. High-molecular-weight PEG has however been suggested as a stabilizing agent for molecules, including drugs, therapeutic, and targeting peptides, and in AuNPs-based formulations for biomedical applications, since PEG increases stability and biocompatibility of AuNPs to these agents. Toxicological studies of more conjugating agents, such as proteins/peptides, GSH, and antibodies, are suggested to provide insight on the safe use of AuNPs in biomedical applications. (9) Further investigations into the clearance and excretion pathways of AuNPs from the body is suggested. (10) Biological methods in the synthesis of AuNPs with clinically tested safe reducing agents, such as plant products, is suggested. Toxicity studies of the biosynthesized AuNPs should, however, be fully explored. (11) More studies on the stability and toxicity of free-ligand AuNPs, using laser ablation in liquid (physical method) synthesis, is suggested.
Generally, more studies on the toxicity of AuNPs are needed to shed more light on the available conflicting reports. Limited research reports were found to describe the excretion of AuNP over time. It is therefore recommended that the possible long-term effects of AuNPs be considered, since these nanoparticles remain in cells for extended periods. Inadequate information on model or assay protocols used is one of the factors limiting the discovery of the best AuNPs for biomedical applications. It is therefore recommended that researchers report fully the characteristics, such as size, shape, coating agents of AuNPs, dose, and other factors employed. Preliminary studies on AuNPs might present little or no toxicity; this does not justify the safety, as there may be cellular damage associated with the exposure. Further studies on tissue distribution, general behavioral changes, weight loss, mortality, inflammatory and oxidative stress responses, hematological analyses, tissue toxicological studies, and histopathological changes should be performed to justify the safe use of AuNPs in medicine. These should be investigated to provide adequate information on both short- and long-term effects of AuNPs. A standardized nanoparticle reference material for toxicological studies, through which comparison of data can be made across different studies, is therefore recommended.
Footnotes
Authors’ Note
This review was included in the PhD thesis of Olusola B. Adewale from Nelson Mandela University, South Africa.
Author Contribution
Adewale, O. contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Davids, H. and Roux, S. contributed to conception and design analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Cairncross, L. contributed to conception and acquisition; drafted the manuscript; and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation, South Africa [grant number 101132].
