Abstract
The use of organophosphates phosphate flame retardants, particularly isopropylated triphenyl phosphate (IPTPP), has increased in recent years as replacements for polybrominated diphenyl ethers. This is despite limited understanding of the hazards of IPTPP. To examine the general and endocrine toxicity of IPTPP, adult Wistar rats were fed for 90 days on diets containing IPTPP estimated to deliver daily doses of 5 to 140 mg/kg/d. Exposure to IPTPP caused a dose-related increase in liver and adrenal gland weight in both sexes. Cells in the zona fasciculate (ZF) of the adrenal cortex were observed to be filled with droplets that stained with Nile red, suggesting they contained neutral lipid. Despite marked structural changes, there was no change in basal or stress-induced serum levels of their major secreted ZF product corticosterone (B), suggesting cell function was not altered. There were no effects on responses to glucose or insulin challenge, but serum levels of fructosamine were elevated by IPTPP exposure, suggesting a slight tendency of exposed animals to be hyperglycemic. Serum levels of total cholesterol and high-density lipoprotein cholesterol were significantly elevated in both sexes at the 2 highest doses. This study demonstrates that IPTPP exposure causes hypertrophy and neutral lipid accumulation in adrenal cortex ZF cells but does not result in impaired B production.
Introduction
Isopropylated triphenyl phosphate (IPTPP) is used as an antiwear additive in hydraulic fluids and a flame-resistant plasticizer in polyvinyl chloride plastics. 1 It is also a flame retardant for flexible polyurethane foams and is a major component of a product (Firemaster 550) 2 which has seen increased use as a direct replacement for the banned pentabromodiphenyl ether mixtures. 3 As it is not covalently bound with PU polymers, IPTPP migrates out of household and workplace items and contaminates indoor environments. Technical IPTPP is a mixture of aryl phosphate ester congeners that vary in the number of phenyl moieties containing the isopropyl group. 4 Isopropylphenyl, diphenyl phosphate is the major congener in technical IPTPP, and its presence has been reported in indoor air from an electronics recycling plant in Sweden 5 and house dust in North Carolina. 6 In addition, several studies have identified an urinary metabolite of this congener in the majority of individuals in the United States. 6 –14 Given the recent increase and widespread presence and levels of other FM550 components (ie, tetrabromo diethylhexylphthalate and 2-ethylhexyl-2,3,4,5-tetrabromobenzoate), 3 including from homes in many cities across Canada, 15 it is reasonable to assume that human exposure to IPTPP is as at least as widespread as exposures to these throughout North America.
The consequences of IPTPP exposure are currently unclear. Recent studies report a relationship between maternal, but not paternal, IPTPP exposure and reduced fertilization rate, implantation, and live birth in women attending an infertility clinic. 9 –10 Currently, there are no published studies of mammalian toxicity of IPTPP. Limited mammalian toxicity information is available as summaries of regulatory toxicology studies, 1,16–17 suggesting liver and adrenal toxicity. There is no known dose that is without effect, and the health consequences of reported observations are not known. Data available in the public domain include one small study of the effects of early life exposure of rats to the full FM550 mixture. 18 Treatment of female rats through pregnancy and lactation of FM550, which contains approximately 55% IPTPP, 4 caused multiple effects on growth, thyroid hormone levels, behavior, and adverse metabolic outcomes including elevated adiposity and reduced glucose tolerance. 18 Given the high proportion of IPTPP in FM550, it is possible that metabolic and endocrine effects reported may be at least partially attributable to IPTPP. The aim of the current study is to examine the toxicity of IPTPP focusing on endocrine and metabolic effects. The current work describes functional and structural effects on the adrenal glands as well as effects on glucose metabolism.
Materials and Methods
Test Chemical and Diets
Isopropylated triphenyl phosphate was generously donated by Dr Mamta Behl (National Institute of Environmental Health Sciences, Durham, NC). Diets were prepared at Health Canada by mixing neat IPTPP with increasing amounts of powdered isoflavone-free rodent diet (TekLad Global 2014; Envigo, Laval, Quebec, Canada) to ensure very thorough mixing. Diets were formulated to deliver estimated daily doses of 140, 70, 20, and 5 mg/kg/d (assuming an average daily consumption of 70 g diet per kg body weight based on average food consumption from lower doses in NTP study of tricresyl phosphate 19 ). Final concentrations of IPTPP in diet were 2,000, 1,000, 300, and 75 mg/kg diet, respectively. Mixed powdered diets were wetted, pelleted, and dried. Pelleted diets were made fresh once per month and stored at −20°C until added to food hoppers.
Animal Exposures
Sexually mature Wistar rats (∼60 days of age) were purchased from Charles River (St Constance, Quebec, Canada). Rats were housed in clear plastic cages either singly (females) or in pairs (males). Cages were individually supplied with High Efficiency Particulate Air-filtered room air. All animals were housed under controlled light (12-hour light/12-hour dark), humidity (40%-70%), and temperature (20°C-24°C). All animal handling adhered to the guidelines of the Canadian Council on Animal Care, and experimental procedures were approved by the Health Canada Animal Care Committee (protocol # 2014-010) prior to start of the study. Animals were provided with control diet and water ad libitum upon arrival and were randomized to 5 treatment groups (n = 10 per sex per treatment). On the first day of dosing, animals were weighed and provided with diets containing appropriate amount of test substance. Animals were examined daily while body weight and food consumption were determined weekly.
As adrenal function, insulin, and glucose tolerance are strongly influenced by ovarian steroids, all endocrine measures in females were conducted during diestrus to minimize variability. Consequently, ovarian cycles were monitored in female animals beginning on treatment day 64 and continued daily until the end of the study. Vaginal cytology was examined by collecting vaginal cells using a small amount of water (∼200 µL) in a pliable plastic pipette. Cells were examined immediately by a trained technician who assigned stage of cycle (proestrus, estrus, or diestrus).
Animals were euthanized either on the 91st morning of the experiment (treated diets introduced on 1st morning) for males or on the anticipated first day of diestrus (based on daily examination of vaginal cytology) after 90 days of exposure for females. Animals were fasted overnight and euthanized by exsanguination and pneumothorax transection under isoflurane anesthesia. Blood was retained in serum separator tubes (Becton Dickinson, Thermo Fisher, Nepean, Ontario, Canada) for the preparation of serum per the manufacturer’s directions. All organs were removed and grossly examined by a pathologist. Liver, heart, brain, spleen, adrenal glands, kidneys, testes, epididymis, and periovarian (female) or epididymal (male) fat pads were weighed and processed (left for all paired organs) for histopathology or a sample snap-frozen in liquid nitrogen (right for paired organs) for molecular analyses. Histopathology was initially assessed in sections from 5 controls and 5 high-dose samples that were coded to blind treatment and examined by a pathologist. If a notable difference between control and treated was observed, the process was repeated for sections of this tissue for all animals in the study including reanalysis of all control and high-dose samples.
To assess the relative content of neutral lipid, adrenal glands were snap-frozen by immersion in liquid nitrogen and stored at −80°C until sectioning. Whole frozen tissue was mounted on the cryostat block in Cryomatrix embedding resin (Thermo Fisher) on a bed of dry ice. Sections (10 μm) were fixed in 4% paraformaldehyde and stained with 1 μg/mL each of Nile red bioreagent (Sigma Aldrich, St Louis, Missouri) and 4′,6-diamidino-2-phenylindole dihydrochloride dissolved in phosphate-buffered saline containing 0.2% wt/vol Triton. Sections were preserved with Prolong AntiFade (Thermo Fisher Scientific, Nepean, ON) and digital images collected using a Zeiss confocal microscope.
Insulin and Glucose Challenge
Male animals exposed to IPTPP for at least 70 days were subjected to an insulin tolerance test (ITT), and after 84 days of treatment, the same animals were subjected to an oral glucose tolerance test (OGTT). Both tests were initiated at 9
Drug-Metabolizing Enzymes
Hepatic activities of some phase I biotransforming enzymes were analyzed in liver homogenates using previously described methods. 20 Briefly, a snap-frozen piece of the liver, from a consistent lobe from each animal, was homogenized in 2.5 volumes (vol/wt) of ice-cold 0.05 M Tris–1.15% KCl buffer (pH 7.4). The 10,000g supernatant (S-9 fraction) was retained and assayed for the activities of CYP1A1/1A2, 2B, 1A2/3A, and 2B/3A using ethoxyresorufin-O-deethylase (EROD), pentoxyresorufin-O-dealkylase (PROD), and benzyloxyresorufin-O-dealkylase (BROD) activities, respectively. Reactions were carried out at 37°C with triplicate samples in black 96-well plates. The fluorescence of resorufin was measured with a SpectraMax M2 (Molecular Devices, Sunnyvale, California), with excitation and emission wavelengths of 530 and 585 nm.
Serum Analyses
A broad spectrum of steroid hormones were measured in serum collected at necropsy by solid-phase extraction of serum using C-18 columns followed by high-performance liquid chromatography–mass spectrometry/mass spectrometry by OpAns LLC (Durham, North Carolina). As serum levels of corticosterone (B) were outside the bounds of standards in these analyses, B was analyzed in a fresh aliquot of terminal serum and ITT serum samples using a commercial ELISA (Cat # K014-H1, Detect-X; Corticosterone EIA, Arbor Assay, Ann Arbor, Michigan). Serum nonesterified fatty acids were quantified using Serum Fatty acid detection kit by ZenBio (Cat SFA-5; ZenBio, Research Triangle Park, North Carolina) per the manufacturer’s specifications. An ABX Pentra 400 serum analyzer (Horiba Medical, Irvine, California) was used to analyze serum levels of albumin, calcium, total cholesterol, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, glucose, magnesium, total protein, total triacylglycerides, and urea nitrogen using reagents from Horiba and fructosamine using RaiChem fructosamine assay reagents (Clinica, San Marco, California). Analyses of all serum measures used samples stored at −80°C that had not been thawed until immediately prior to testing.
Statistical Analysis
Data for variables with observations in both sexes were analyzed by 2-way analysis of variance (ANOVA; IPTPP dose × sex) followed by post hoc Holm-Sidak test to compare means to control. Data for weekly body weight were analyzed by repeated-measures ANOVA separately for males and females. Significant treatment effects were further analyzed using post hoc (Holm-Sidak) test to compare means to control within each week. Where tests for assumptions of homogeneity of variance and normality failed (Shapiro-Wilk and Levene mean tests, respectively), data were log-transformed and retested. When homoscedasticity and normality were still not satisfied after this transformation, data were retested using Kruskal-Wallis (K-W) ANOVA on ranks for each sex separately. Significant K-W ANOVA was further evaluated using Dunn test. All statistical analyses were performed using Sigmaplot (12.5; Systat Software, San Jose, California).
Results
Dosing, and Body and Organ Weights
Exposure to IPTPP tended to cause a reduction in body weight gain in females, although only the highest dose was significantly different from control (P < 0.05) at weeks 8, 9, 11, 12, 13, and 14 (Figure 1A), while body weight in males was unaffected (Figure 1B). Food consumed per gram body weight declined markedly for all dose groups in both males and females (Supplementary Figure 1A and B). The weekly rate of food intake was remarkably similar between doses, indicating that any reduction in weight gain in the high-dose IPTPP-treated females cannot be explained by reduced food consumption. However, the decline in feed consumption per unit body weight does mean that the daily exposure to IPTPP also declined over the course of the study. Daily dose rates, based on food consumption, during the final week of the study were equivalent to 45% and 62.6% of that experienced in the first week for male and female animals, respectively. Based on these data, the estimated average nominal dose rate calculated for each group across the study in 75, 300, 1,000 and 2,000 mg/kg diet groups for females was 6.0 ± 0.3, 25.4 ± 1.6, 83.7 ± 4.0, and 166 ± 12.1 mg/kg/d, respectively, compared to 5.2 ± 0.2, 21.0 ± 1.0, 70.0 ± 1.1, 143 ± 5.4 mg/kg/d, respectively, for males.
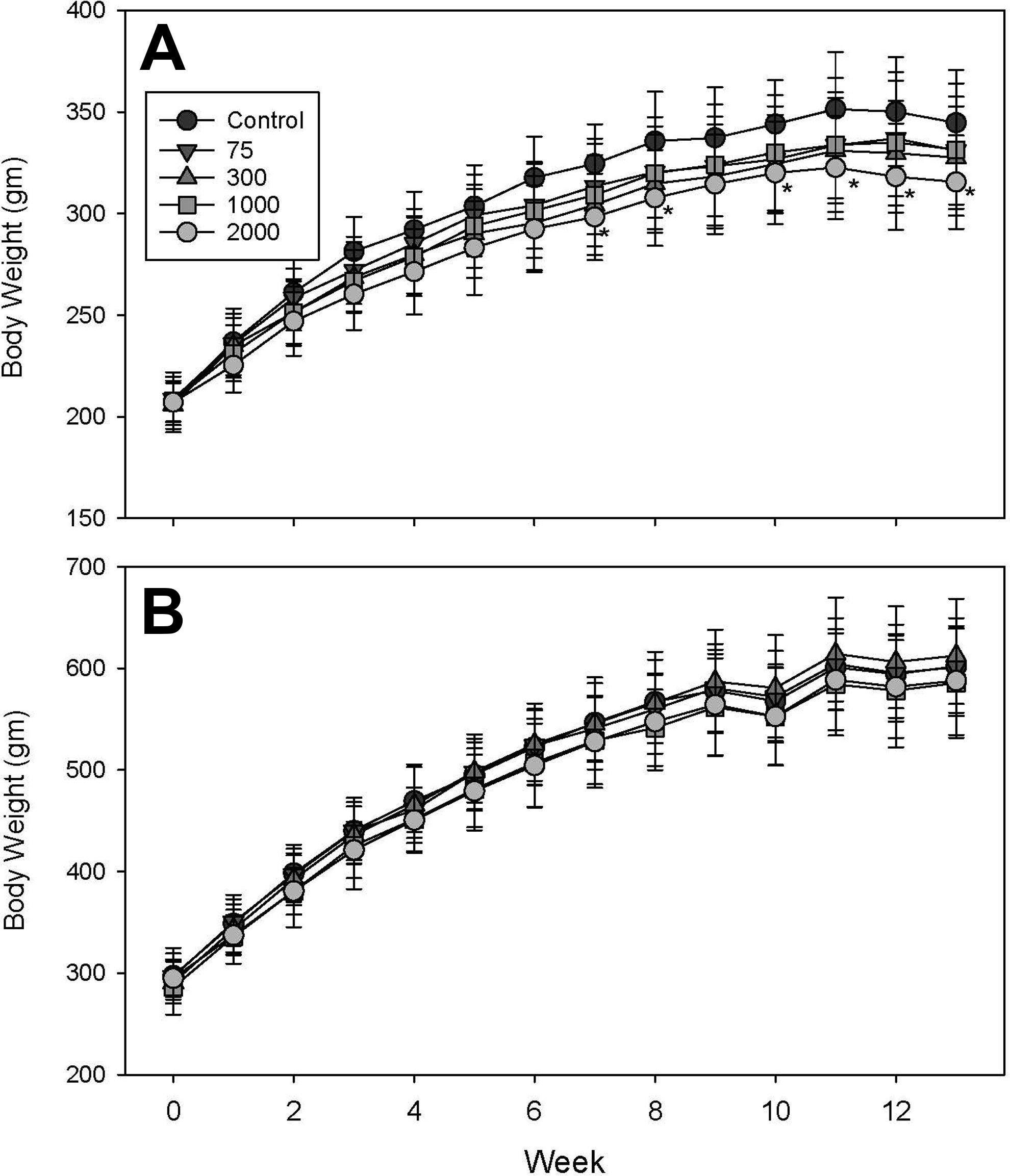
Weekly mean body weights of female (A) and male (B) rats exposed to isopropylated triphenyl phosphate (IPTPP) through diet for 13 weeks. Doses indicated in the legend reflect the nominal concentration of IPTPP in mg/kg diet. Data points are mean ± standard deviation of n = 10 animals per treatment except for control male data, where n = 11 for weeks 1 to 8 and n = 10 for weeks 9 to 13. *Significant difference (P < 0.05) from mean of control animals at the same time interval.
At necropsy, there were no IPTPP-related effects on terminal body weight in either sex (Table 1). However, IPTPP exposure caused a dose-dependent increase in the relative weight of liver which reached significance in males of the 2,000 mg/kg diet group and females in both the 1,000 and 2,000 mg/kg diet groups (Table 1). Adrenal glands were the most sensitive of organs in either sex, with significant increases in relative weight observed at the 75 mg/kg dose in males and 300 mg/kg dose in females (Table 1). Histopathological analyses of liver revealed hypertrophy of hepatocytes throughout the sections of 1,000 and 2,000 mg/kg females and the 2,000 mg/kg males. In addition, there was an increased frequency of granulomas observed in all of these groups compared to control animals. No treatment-related histological changes were observed in heart, kidney, pancreas, or thyroid gland. Treatment-related changes observed in reproductive organs will be reported elsewhere (Manuscript in development).
Organ Weights Relative to Body Weight in Rats Exposed to Dietary IPTPP for 90 Days.a
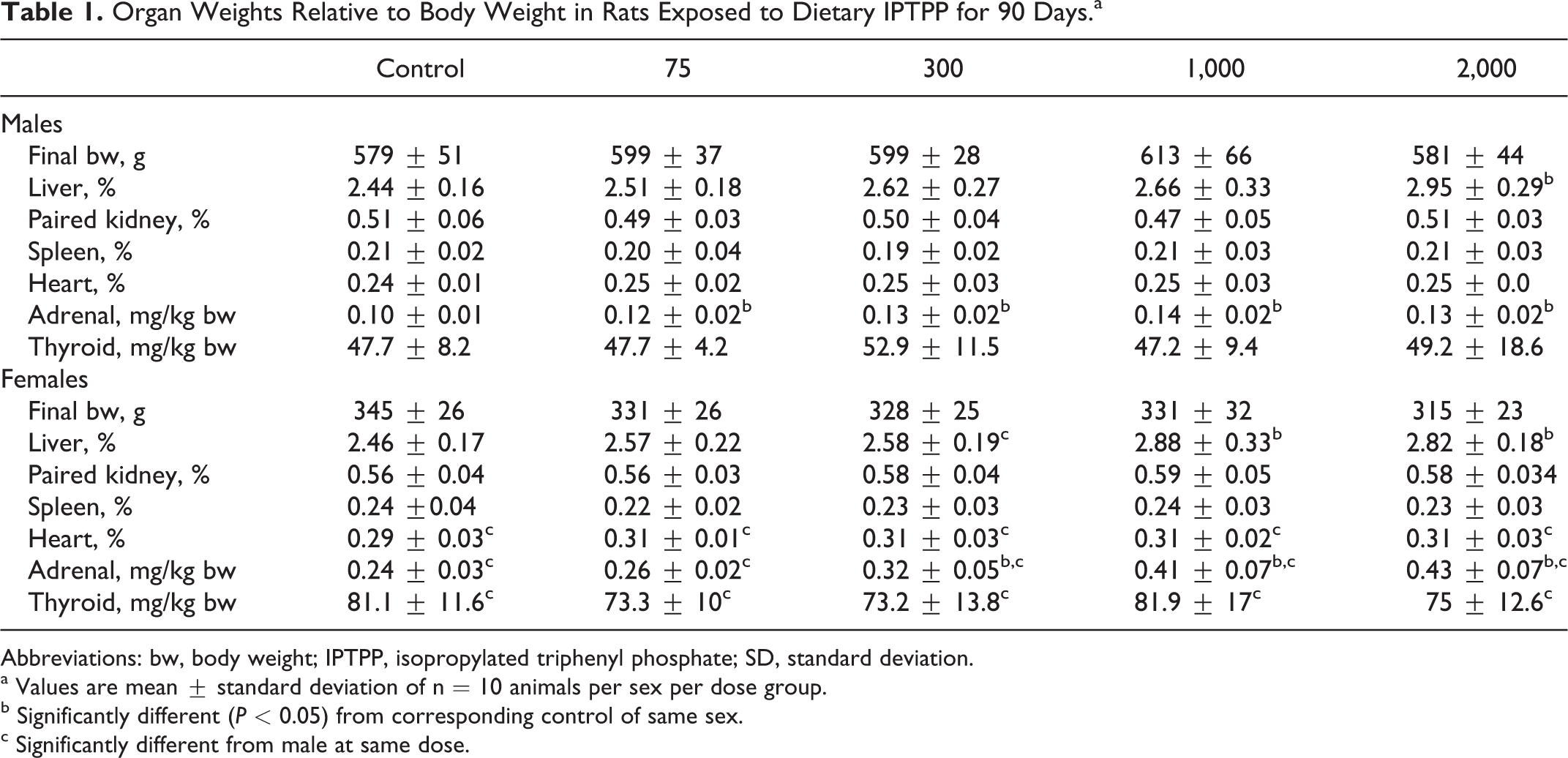
Abbreviations: bw, body weight; IPTPP, isopropylated triphenyl phosphate; SD, standard deviation.
a Values are mean ± standard deviation of n = 10 animals per sex per dose group.
b Significantly different (P < 0.05) from corresponding control of same sex.
c Significantly different from male at same dose.
Adrenal Effects
Histological examination of adrenal glands revealed a dose-related increase in the relative size of the adrenal cortex in treated females (Figure 2C and D) and males (Figure 2G and H) relative to controls (Figure 2A, B, E, and F, respectively). This was primarily due to a marked increase in the size of cells in the adrenal cortical zona fasciculate (ZF), which corresponded to an increase in the vacuolization of these cells. In females, this effect seemed to be associated with the cytoplasm being filled with small vacuoles in cells throughout the ZF, although cells located toward the interior part of the gland tended to have very large vacuoles (Figure 2C and D). In comparison, ZF cell hypertrophy in exposed males was associated with an increased level of very large vacuoles only (Figure 2 G and H). To provide a semi-quantitative assessment of the severity, each sample was assigned a score for 1 to 5 based on the apparent intensity of cell hypertrophy/vacuolization and the extent of its distribution throughout the ZF. Median score increased with increasing dose in both sexes, with the score for animals in groups consuming control, 75, 300, 1,000, and 2,000 mg IPTPP/kg diet being 1, 2, 3, 5, and 5 for females, respectively, and 1, 1, 2, 3.5 and 4 for males, respectively. In addition, the number of cell nuclei per area of ZF was counted in at least 3 high-power images of adrenal sections from each animal (Supplemental Figure 2). The density of nuclei declined in ZF in sections from females at 300 mg/kg and higher diets, while nuclei density was significantly reduced in males on 1,000 mg/kg or higher diets (Supplemental Figure 2). The vacuoles in the cytoplasm of the adrenal cortex from either control or high-dose animals stained intensely with Nile red indicating that their content was neutral lipid (Figure 3). The stained structures were far larger in the ZF of high-dose animals compared to controls, suggesting that the increased adrenal is largely a consequence of accumulation of neutral lipid primarily in ZF (Figure 3).
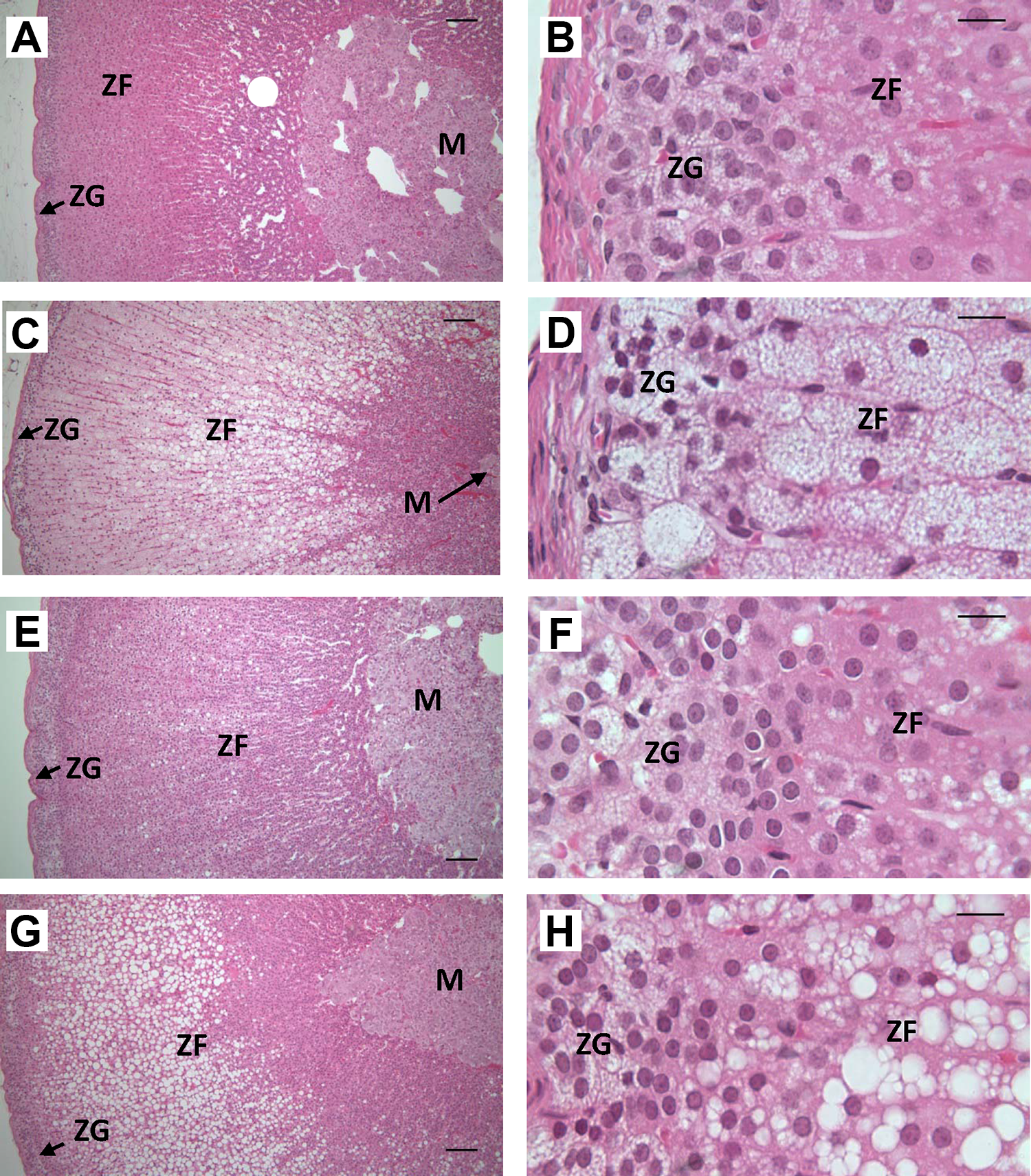
Effects of dietary isopropylated triphenyl phosphate (IPTPP) on adrenal gland morphology. Representative examples of adrenal histology from females on the control (A and B) or 2,000 mg/kg diet (C and D) dose groups and from males on control (E and F) or 2,000 mg/kg diet (G and H). Low-power (panels A, C, E, G; ×10 objective; bar = 200 µm) images show multiple cell layers within the cortex. while high-power images (B, D, F, H; ×40 objective; bar = 50 µm) show detail of zona glomerulosa and zona fasciculata cells. M indicates medulla; ZG, Zona glomerulosa; ZF, Zona Fasciculata.
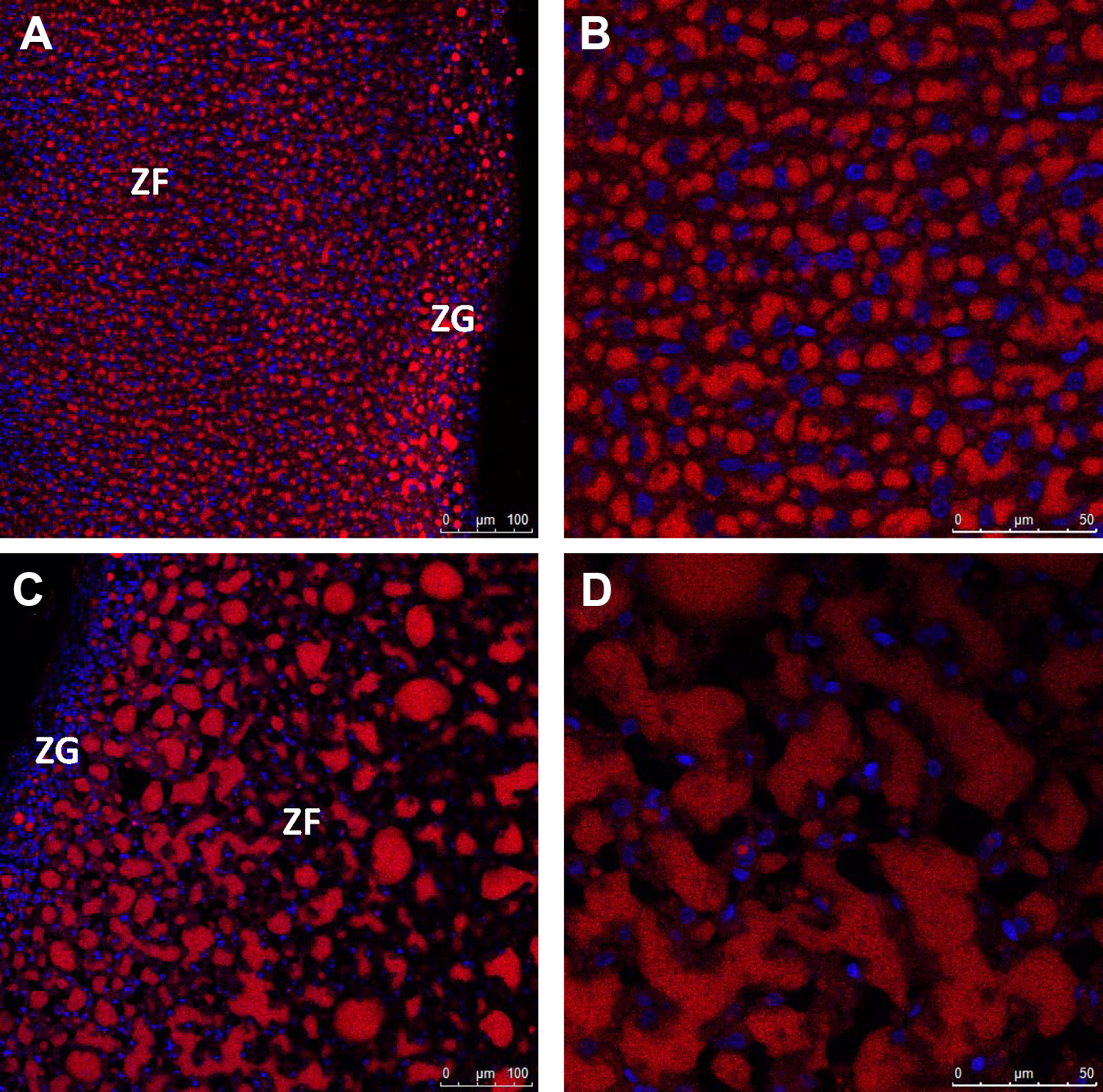
Representative images of Nile red and DAPI staining of frozen sections of adrenal glands from control (A and B) or 2,000 mg/kg diet-exposed (C and D) female rats. Similar results were observed in adrenal glands of male rats from these doses (data not shown). Images were collected under ×20 (A and C) or ×63 (B and D) objectives. Cells in high-power fields (B and D) are all within the ZF. ZF indicates zona fasciculata; ZG, zona glomerulosa.
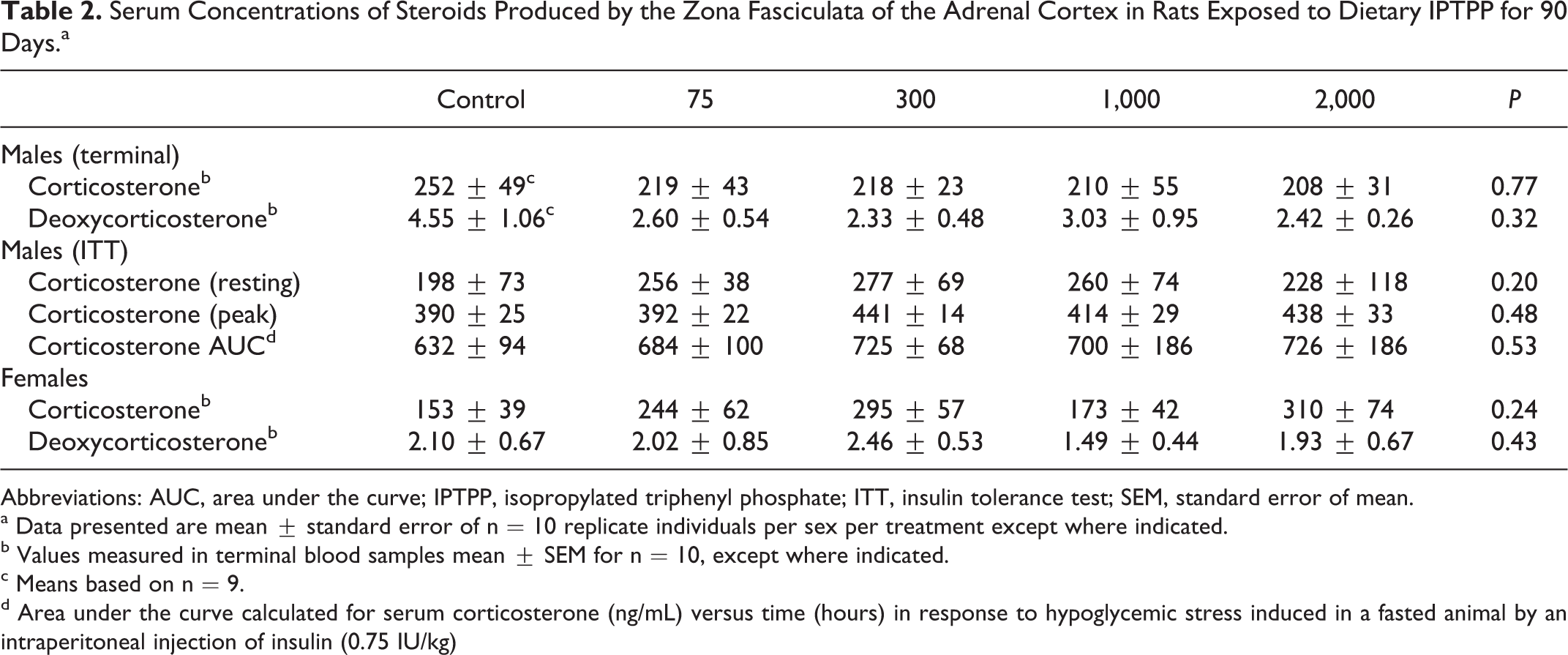
Rat adrenal ZF cells synthesize corticosterone (B), which is the active rodent glucocorticoid mediating the physiological response to stress. Corticosterone synthesis involves sequential enzymatic modifications from cholesterol. To determine whether IPTPP-induced hypertrophy was associated with a change in the ability of the adrenal gland to synthesize glucocorticoids, terminal serum samples were analyzed for the levels of B, and 11-deoxycorticosterone (DOC), the immediate upstream intermediates that are secreted by the ZF in rats. These analyses revealed that one of the control male animals returned data for both steroids that were notably higher than all of animals and Grubs test indicated that values of DOC for this animal were significant outliers (Grubb test P < 0.01). When data for this animal were removed from the analysis, there was no effect of IPTPP exposure on the serum levels of any ZF hormone product (Table 2; P > 0.05).
Serum Concentrations of Steroids Produced by the Zona Fasciculata of the Adrenal Cortex in Rats Exposed to Dietary IPTPP for 90 Days.a
Abbreviations: AUC, area under the curve; IPTPP, isopropylated triphenyl phosphate; ITT, insulin tolerance test; SEM, standard error of mean.
a Data presented are mean ± standard error of n = 10 replicate individuals per sex per treatment except where indicated.
b Values measured in terminal blood samples mean ± SEM for n = 10, except where indicated.
c Means based on n = 9.
d Area under the curve calculated for serum corticosterone (ng/mL) versus time (hours) in response to hypoglycemic stress induced in a fasted animal by an intraperitoneal injection of insulin (0.75 IU/kg)
To evaluate the dynamic response of the ZF to physiological stimulation, hypoglycemic stress was induced in the male animals and the serum B levels before and 30, 60, and 120 minutes after insulin injections were analyzed. There was no difference between IPTPP-treated groups in basal or stress-induced peak concentrations of B in serum, while the area under the curve of B versus time (an integrated measure of the dynamic response) also showed no differences between IPTPP treatment groups (Table 2). These data show that the morphological changes in ZF cells induced by IPTPP treatment did not impair the capacity of these cells to produce B.
Liver Effects
In addition to increasing liver weight, IPTPP treatment caused increased incidence of cellular hypertrophy in periportal hepatocytes in the 2 highest doses, particularly in females where every animal showed moderate effects (data not shown). Liver S-9 were analyzed for several CYP-related activities, because IPTPP congeners were reported to impair zebra fish embryo development via arylhydrocarbon (AhR) receptor activation. 2 Activity of EROD, a marker of AhR-induced CYP1a1 activity, was slightly but significantly increased in males, but not females, in a dose-related fashion with significant induction observed in the 2 highest doses only (Figure 4A). Treatment with IPTPP caused a more marked dose-related increase in the activities of PROD and BROD in both males and female, indicating that IPTPP induces activity of Cyp2B and Cyp3A, respectively, suggestive of the activation of the constitutive androstane receptor and/or the pregnane X receptor (Figure 4B and C).
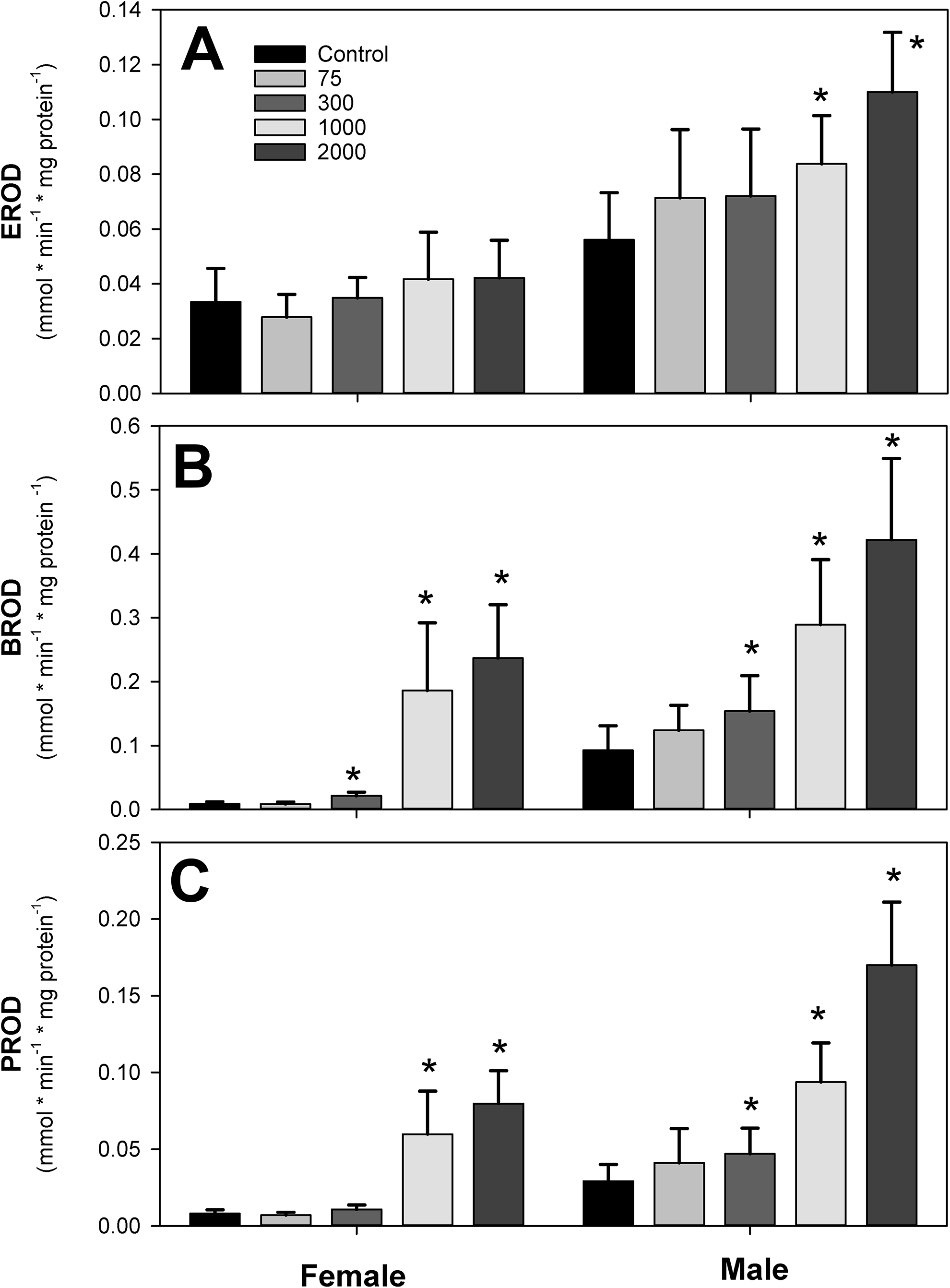
Effects of dietary isopropylated triphenyl phosphate (IPTPP) on the activity of (A) ethoxy-(EROD), (B) benzyloxy-(BROD), (C) pentoxy-(PROD), and (D) resorufin-O-de-ethylation measured in clarified homogenates (S-9 fraction) prepared from frozen liver samples as described in the text. Activity measured by changing substrate fluorescence was corrected by homogenate protein content and expressed as mean ± standard deviation of 10 replicate samples. *Significantly different (P < 0.05) from control within sex.
Serum Biochemistry
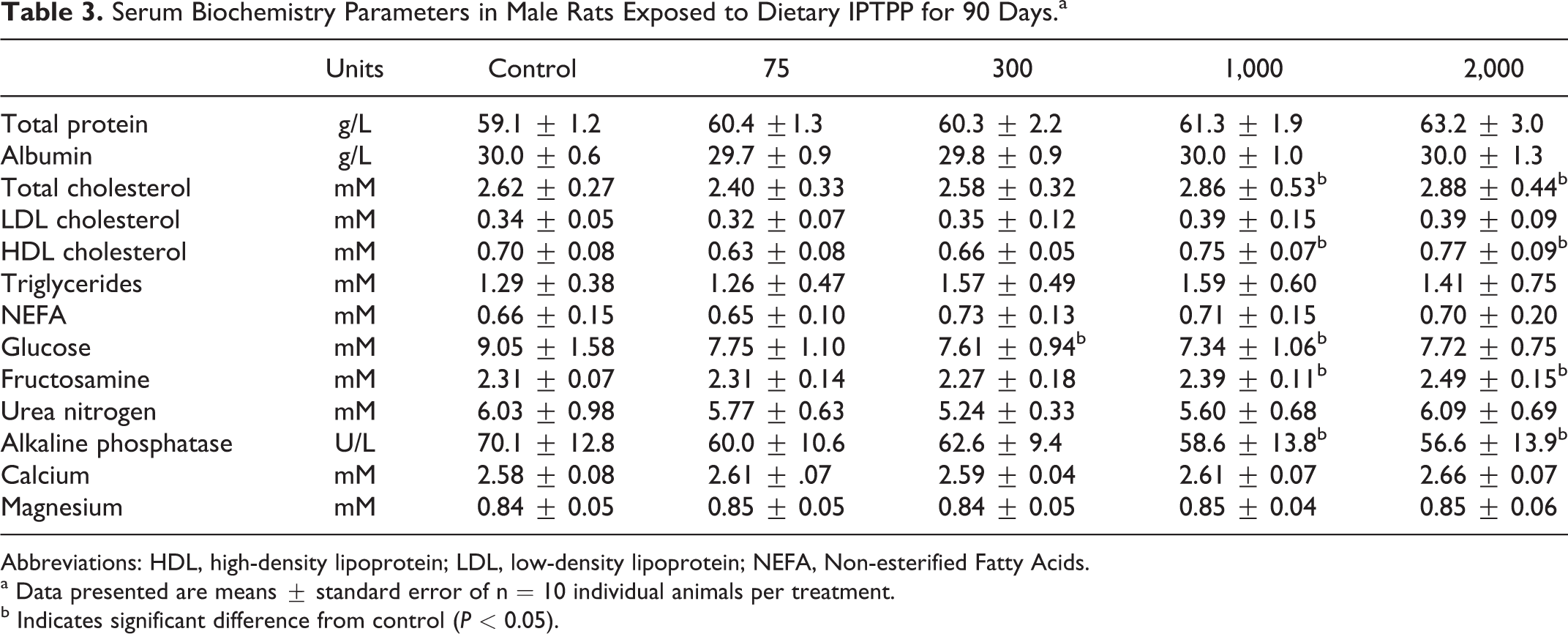
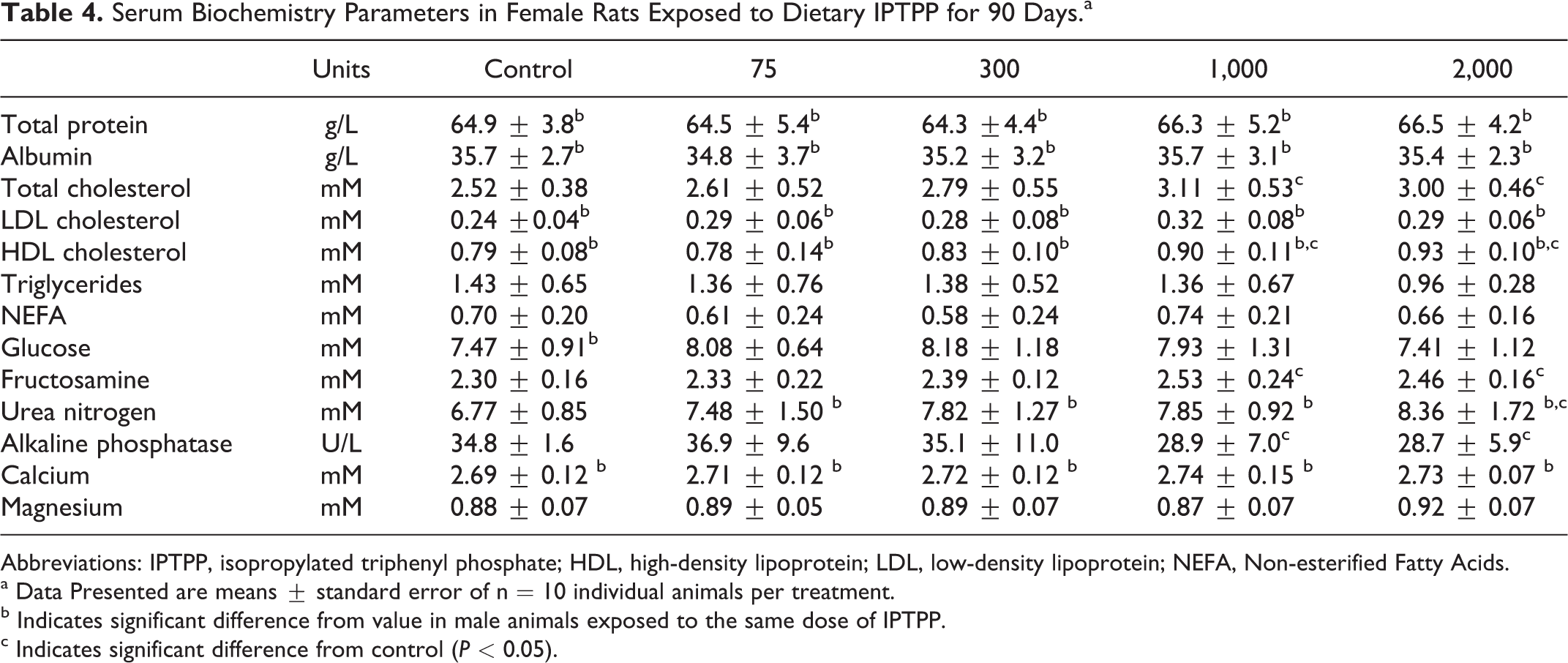
A variety of serum markers of general organ function or metabolic status were analyzed. The highest 2 doses of IPTPP showed altered cholesterol metabolism with increased total cholesterol and HDL-, but not LDL-, associated cholesterol (Tables 3 and 4), with a greater relative increase in females compared to males. Serum total cholesterol in high-dose animals was increased by 19% and 10% over control in females and males, respectively, while serum HDL cholesterol increases by 17% and 10% for females and males, respectively. Other serum lipid measures evaluated in serum, total triglycerides, and nonesterified fatty acids were not changed by IPTPP treatment. Serum activity of alkaline phosphatase was reduced in both sexes in a dose-related fashion, although only values from the 2 highest dose groups achieved significance (P < 0.05).
Serum Biochemistry Parameters in Male Rats Exposed to Dietary IPTPP for 90 Days.a
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; NEFA, Non-esterified Fatty Acids.
a Data presented are means ± standard error of n = 10 individual animals per treatment.
b Indicates significant difference from control (P < 0.05).
Serum Biochemistry Parameters in Female Rats Exposed to Dietary IPTPP for 90 Days.a
Abbreviations: IPTPP, isopropylated triphenyl phosphate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; NEFA, Non-esterified Fatty Acids.
a Data Presented are means ± standard error of n = 10 individual animals per treatment.
b Indicates significant difference from value in male animals exposed to the same dose of IPTPP.
c Indicates significant difference from control (P < 0.05).
Glucose Tolerance and Metabolic Effects
Exposure to IPTPP did not significantly affect insulin tolerance in male animals at any dose (Supplemental Figure 3). In addition, there was no significant effect of IPTPP treatment on glucose tolerance in either sex (Figure 5). In contrast, fructosamine levels in serum from fasted animals collected at termination showed a slight but significant elevation at the highest 2 doses of IPTPP treatment (Tables 3 and 4).
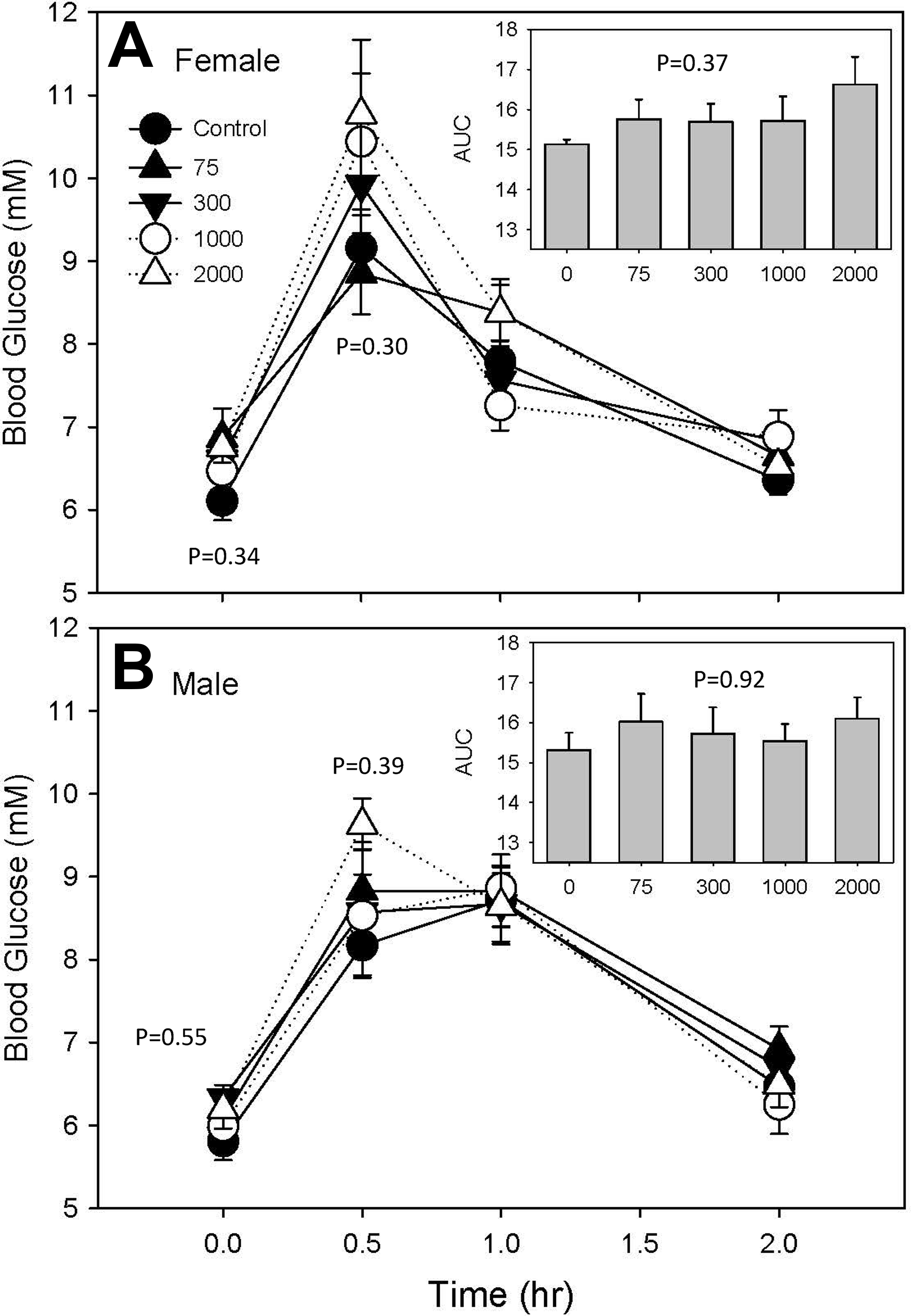
Blood glucose levels in female (A) and male (B) rats after gavage of 2 g/kg glucose. Total area under the curve (AUC) calculated for serum glucose levels integrated over time for each animal is depicted in the inset. Data are expressed as mean ± standard error of mean (SEM) of n = 10 replicate animals. P values indicated at each time point are derived from analysis of variance (ANOVA) comparisons of means at that time point only.
Discussion
Our work demonstrates that dietary exposure of adult rats to IPTPP induces hypertrophy of the adrenal glands and increased lipid accumulation in the adrenal cortex in both sexes at nominal doses below those previously studied. Despite very marked changes in the structure of glucocorticoid-secreting cells in the affected region, there was no evidence that the hormone-producing function of these cells was compromised by this treatment. Effects were also observed in the liver including histological changes and activity of xenobiotic-metabolizing enzymes. Other effects include increased serum cholesterol and hyperglycemic marker fructosamine, but these all occurred at higher levels of exposure than adrenal effects. These results are the first data on mammalian toxicity of IPTPP in the public domain.
The adrenal gland appears to be the most sensitive target of IPTPP toxicity, as adrenal weight is significantly increased in females consuming diets containing at least 300 mg/kg and in males consuming diets with at least 75 mg/kg IPTPP content. Histological examination revealed the accumulation of neutral lipid in the ZF occurred in both male and female animals consuming diets containing 300 mg IPTPP/kg diet or higher. No ZF lipid accumulation or other histological changes were observed in adrenal glands from male consuming the 75 mg/kg diet, suggesting that the increased adrenal weight observed in these animals may be consequent to additional, undetermined tissue effects. Despite the marked changes in ZF cell structure, no obvious changes in the physiological function of these cells was observed, as there was no effect of IPTPP exposure on terminal B or DOC levels or the increase in B induced by hypoglycemic stress. Hypoglycemia induced in a controlled fashion by insulin injection of a fasted animal is well characterized as physical (ie, nonpsychogenic) induction of the hypothalamic–pituitary adrenal stress response 21 and results in a rapid increase in B release due to increased release and action of adrenocorticotropin (ACTH). 22 However, we cannot definitively conclude a lack of effect on steroid hormone–producing capacity, as we were unable to measure circulating ACTH and cannot rule out the possibility that pituitary ACTH secretion was increased to compensate for IPTPP-induced effects.
Adrenal cortical ZF cells are normally richly endowed with lipid droplets that store steroid hormone precursor cholesterol esters (CEs). 23 Cholesterol esters must be converted to unesterified cholesterol by a neutral cholesterol esterase before their translocation into mitochondria where the first step in hormone synthesis can occur. 24 The enzyme that mediates this conversion is most likely hormone-sensitive lipase (Hsl), 25 although neutral cholesterol ester hydrolase 1 (Nceh1) may also play a role. 26
The accumulation of lipid droplets in ZF as seen in IPTPP-treated rats has also been observed in rats administered excess ACTH, 27 treated with chemical inhibitors of adrenal steroid synthesis (aminoglutethamide, 28 cyanoketone, 29 ketoconazole 30 ), or in transgenic mice lacking adrenal expression of some proteins involved in steroid hormone synthesis (eg, CYP11A1, 31 StAR, 32 TSPO 33 ) or lacking Hsl. 34 Lipid droplet accumulation in cells of the adrenal cortex is also observed in aging rats with high circulating ACTH but reduced B levels. 35 Mice fed a diet rich in sucrose also had enlarged adrenal glands with increased cortical lipid droplets, which exhibited elevated serum B levels in unstressed animals but a blunted increase in B in response to ACTH treatment. 36 These observations suggest that impaired synthesis of B or increased ACTH stimulation can lead to increased ZF lipid droplet accumulation.
Increased vacuolization of the ZF was also reported from adult male or female rats exposed to 2 other aryl phosphates with considerable structural similarity to IPTPP: tricresyl phosphate (TCrP) and tri-isobutylphenyl phosphate (TiBPP). 37 Animals exposed to a high dose of either TCrP (400 mg/kg) or TiBPP (1,700 mg/kg) showed hypertrophy in the adrenal cortex, which was associated with a marked increase in CE measurable in the gland (∼4-fold increase over control for TCrP and 1.74-fold for TiBPP). 38 Further, the CE activity—activity that could be attributed to either HSL or Nceh1—in the adrenal glands of TCrP- and TiBPP-treated animals was reduced to 4% and 44% of control levels, respectively. Levels of circulating B in animals treated with either TCrP or TiBPP did not differ from levels observed in untreated animals of the same sex, 39 which is consistent with these aryl phosphates having a similar mode of action to IPTPP. These observations and the very high degree of structural similarity between these substances and IPTPP make it tempting to speculate that inhibition of CE is the mechanism underlying the adrenal hypertrophy that is the common phenotype in exposed rats.
To our knowledge, ours is the first published study to report mammalian toxicity of technical IPTPP. One other mammalian study has reported effects of commercial flame-retardant mixture FM550 containing roughly 55% technical IPTPP. 18 These authors reported increased adiposity and reduced glucose tolerance in adult pups that were exposed early in life to FM550. Although our study design differed from that reported by Patisaul and colleagues, 18 we observed little effect of IPTPP treatment on markers relevant to metabolic outcomes such as fat pad weights, serum triglycerides, or free fatty acids nor did we observe marked effects on glucose or insulin tolerance beyond a significant increase in serum fructosamine. Also, these authors report effects on circulating thyroid hormones with a significant increase in circulating total thyroxine in the FM550-treated dams, while data from pups suggested a reduction but this did not reach statistical significance. We observed no effects on serum thyroxine (data not shown) or any changes in thyroid gland structure that might suggest IPTPP effects on the hypothalamic–pituitary–thyroid axis. Others, however, have reported on the effects of exposure to IPTPP or congeners found in technical IPTPP on the development 2,40 and behavior 41 of zebra fish. These studies reported that exposure of zebra fish embryos to technical IPTPP or to isopropylphenyl, diphenylphosphate during critical windows of development caused abnormalities of heart structure and function and activated 2 of the 3 isoforms of AhR possessed by zebra fish. 2 We observed a very weak induction of hepatic EROD activity at only the 2 highest doses in males only. Conversion of EROD to a fluorescent marker is catalyzed by AhR-induced enzymes of the CYP1 family, suggesting that neither technical IPTPP nor its constituent di-PDPP is robust activator of the mammalian AhR. This is consistent with the lack of induction of EROD activity by FM550 treatment in rats. 18 Activity of BROD and PROD was increased to a greater extent, and the increase was observed at the mid-dose indicating increased catalytic activity of CYP3A 42 and/or CYP2B. 43 Subchronic exposure of adult rats to higher doses of similar triaryl phosphates TCrP and tris (t-butylphenyl) phosphate also increased liver weights and hepatic levels of total cytochrome P450 levels to 3× control levels. 39
The relevance of the observed IPTPP effects in the current study for predicting human consequences of real-world exposure to IPTPP is unclear, as the doses used in the current study probably exceed the potential maximum human exposure. However, the paucity of information available for estimating human exposures, as has been recognized as a priority information gap by the United States Environmental Protection Agency, 44 make it difficult to determine the extent of this gap. Recently published measures of a major metabolite of IPTPP in urine samples from several study populations from the United States 6 –14 may provide a means to roughly estimate background levels of exposure from all sources. The levels of isopropyl phenyl, phenyl phosphate measured in urine (corrected for specific gravity) range from below the minimum detection limit 13 to 2.0 ng/mL 8 for median levels and 2.3 8 to 76.1 ng/mL. 14 Applying a nominal estimate of urine production (median normal adult daily urine volume of 1.4 45 ) and body weight (80 kg 46 ), this maximum urinary level of 76.1 µ/L yields a crude estimate of maximum daily excretion of a maximum daily excretion estimate of 1.3 µg/kg. There are no data for IPTPP toxicokinetics from which we can extrapolate total IPTPP exposure. However, if one assumes that humans metabolize IPTPP with the same kinetics as rats metabolize TCrP, one can estimate that this urinary level represents about 41% of the total original dose 47 and that this excretion estimate translates to a crude estimate of maximum daily exposure of 3.2 µg/kg. This value is at least 3 orders of magnitude below the lowest dose tested in the current study (5.5 mg/kg in males). Given that the study populations in which these urinary levels were observed did not attempt to identify and sample individuals at higher risk of exposure from occupational sources, it is possible that rates of human exposure higher than these may occur. However, this estimated exposure is based on many unstated assumptions and data limitations, and it should be stressed that the total human exposure to IPTPP and health consequences of this remain poorly characterized.
In conclusion, subchronic exposure to common phosphate flame-retardant technical IPTPP causes hypertrophy of the adrenal cortical ZF due to increased content of lipid-containing droplets. The similarity of this phenotype to rats treated with structurally similar organophosphates shown to inhibit adrenal CE and with mice lacking functional HSL suggest that IPTPP may be impairing the mobilization of free cholesterol from the pool of CE in these cells. The IPTPP-related increase in HDL cholesterol—the physiological source of CE for ZF cells—may be secondarily related to the inability for ZF cells to mobilize CE for steroid hormone production. As the use of this material in home furnishings is widespread and human exposure likely ubiquitous, these results suggest that investigation of the effects of human exposures on adrenal function and serum cholesterol is warranted. Moreover, the bioactivity of this material in adult animals raises concerns about the impact of fetal and early neonatal exposure to this material on the function of the hypothalamic–pituitary–adrenal axis, given the heightened sensitivity of early-life stages and potential for lifelong impacts.
Supplemental Material
Supplemental Material, DS1_IJT_10.1177_1091581819851502 - Toxicity of Flame Retardant Isopropylated Triphenyl Phosphate: Liver, Adrenal, and Metabolic Effects
Supplemental Material, DS1_IJT_10.1177_1091581819851502 for Toxicity of Flame Retardant Isopropylated Triphenyl Phosphate: Liver, Adrenal, and Metabolic Effects by Michael G. Wade, Alice Kawata, Marc Rigden, Don Caldwell and Alison C. Holloway in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS2_IJT_10.1177_1091581819851502 - Toxicity of Flame Retardant Isopropylated Triphenyl Phosphate: Liver, Adrenal, and Metabolic Effects
Supplemental Material, DS2_IJT_10.1177_1091581819851502 for Toxicity of Flame Retardant Isopropylated Triphenyl Phosphate: Liver, Adrenal, and Metabolic Effects by Michael G. Wade, Alice Kawata, Marc Rigden, Don Caldwell and Alison C. Holloway in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS3_IJT_10.1177_1091581819851502 - Toxicity of Flame Retardant Isopropylated Triphenyl Phosphate: Liver, Adrenal, and Metabolic Effects
Supplemental Material, DS3_IJT_10.1177_1091581819851502 for Toxicity of Flame Retardant Isopropylated Triphenyl Phosphate: Liver, Adrenal, and Metabolic Effects by Michael G. Wade, Alice Kawata, Marc Rigden, Don Caldwell and Alison C. Holloway in International Journal of Toxicology
Footnotes
Acknowledgments
The authors thank Karine Chamberland, Michelle Lalond, Julie Todd, and Scott Smith for their assistance with animal handling and Kevin Kittle for mixing and pelleting dosed feed. In addition, the authors thank Drs Errol Thomson and Ella Atlas for their expert comments on this manuscript.
Author Contributions
M. G. Wade contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. A. Kawata contributed to design, contributed to acquisition and analysis, and critically revised the manuscript. M. Rigden contributed to design, contributed to acquisition and analysis, and critically revised the manuscript. D. Caldwell contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. A. C Holloway contributed to conception and design, contributed to interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chemicals Management Plan Research Initiative of Health Canada.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
