Abstract
Nicotine is a major component of tobacco plants and is responsible for the development of reproductive problems in smokers. Nicotine has been recognized to result in oxidative stress by inducing the generation of reactive oxygen species (ROS) in some parts of female reproductive system, but the effect of nicotine on endometrium that plays an important role in reproductive biology stays unexplored. The aim of this work was to clarify the direct effects of nicotine administration on the antioxidant defense system and lipid peroxidation in human endometrial cells. Human endometrial stromal primary cells were treated with nicotine (0, 10−11, 10−8, and 10−6 M) for 24 hours. On nicotine administration, the endometrial cells were associated with a decrease in antioxidant defense markers such as Glutathione (GSH) level, glutathione peroxidase (GPx), glutathione reductase (GR), and catalase (CAT) enzymes activity and higher levels of malondialdehyde (MDA) in a dose-dependent manner when compared to the control. We concluded that nicotine as a pro-oxidant affects the oxidative state of the endometrial cells.
Introduction
The tobacco epidemic is one of the main public health threats the world has ever faced and accounts for nearly 7 million deaths per year. World Health Organization estimated around 86% of those deaths is the result of direct tobacco use, while the remaining is the result of nonsmokers being exposed to second-hand smoke. 1
Nicotine is one of the main components present in cigarette smoke. 2 This naturally occurring alkaloid enters the body when inhaled during smoking and gets rapidly absorbed into the circulatory system and metabolized in the liver, 3 leading to serious systemic side effects on many organs including the heart, reproductive system, lung, kidney, and so on. In addition, it is highly addictive. 4 -7 Female reproductive system and its different parts is one of the most important organs that can be widely affected. Endometrium is the innermost glandular layer of the uterus and plays an important role in reproductive biology, embryo implantation, and fetal nourishment. Thus, normal endometrial function is an important factor that can lead to successful pregnancy outcome and reproductive health. 8 Although several studies have demonstrated that nicotine exposure can lead to chronic anovulation, irregular menstrual cycles, irregular bleeding, impairment of the oocyte production, shape, and maturation, 9,10 the direct effects of nicotine on the endometrial cells stay unexplored.
However, among different cellular mechanisms through which nicotine can affect different organs, the exact mechanism has not been completely elucidated to date; several studies have shown higher oxidative damage and reduced antioxidant status in smokers versus nonsmokers. Moreover, the usefulness and protection of different antioxidants against nicotine exposure 11 -13 suggest the involvement of oxidative stress as an important mechanism in nicotine-induced side effects.
Oxidative stress is caused as a result of an imbalance between pro-oxidants such as reactive oxygen species (ROS) and the body’s scavenging ability (antioxidants). 14,15 Although cellular ROS as the key signal molecules play an important role in physiology of the female reproductive system, in some animal and in vitro studies, the oxidative stress has been proposed as a contributing factor in several reproductive diseases including unexplained infertility. 14,16 Catalase together with glutathione and its related enzymes, glutathione peroxidase (GPx) and glutathione reducatase (GR), comprise a system that scavenges the free radicals and acts as a primary protection against excessive generation of damaging ROS. 17 In addition to the studies that show a decrease in the cellular antioxidant defense system can lead to apoptosis and some other cellular malfunctions, 18 -20 we recently reported the impact of oxidative stress on apoptosis, adhesion, and spread of the endometrial cells. 21
Our study extends upon this previous evidence that some changes in the endometrial stromal cells after nicotine exposure were independent of steroid hormones, and more extensive research is needed to explore the possible mechanism. 1 Therefore, based on the results of our previous study, as well as the previous consistent results, evidence indicates the involvement of ROS production in nicotine exposure 11,12,22,23 ; in this study, we aimed to clarify the direct effects of nicotine administration on the antioxidant defense system and lipid peroxidation in human endometrial cells.
Materials and Methods
Sample Preparation
Endometrial biopsies were obtained from reproductive age-fertile women (aged 20-40 years) with normal menstrual cycles and undergoing sterilization procedures. All experimental and surgical procedures were approved by the committee of investigations involving human participants of Shiraz University of Medical Sciences. Informed consent of the participants was obtained before the collection of any tissue samples for this study. No participant had used hormonal contraception within the 3 months preceding the operation. The part of the endometrium removed was fixed for conventional histology to confirm that the tissue was normal. Human endometrial stromal cultures were identified as described previously with some modifications. 24 Briefly, 5 healthy biopsies were collected and all tissues cut into 2- to 3-mm pieces, and all 5 biopsies were pooled to make a single cell suspension which was then expanded to get enough cells for the experiment. Biopsies incubated with 1 mg/mL of collagenase type 1 in DMEM/10% fetal calf serum (FCS) with stirring for 2 hours at 37°C. The suspension was then filtered through a 40-µm nylon sieve that allowed the stromal cells to pass through, while the intact glands were retained. At the end of the isolation procedure, the cells were counted in a hemocytometer, and viability was determined by the trypan blue exclusion test. Representative cells were stained for vimentin to ensure that the monolayers comprised of 98% or more stromal cells.
Cell Culture Procedures and Treatments
The cells extracted from biopsies were suspended in Dulbecco modified Eagle medium (DMEM) supplemented with FCS (10% v/v), 100 U/mL penicillin, and 100 µg/mL streptomycin. Aliquots of the stock cell suspension were added to 25-cm2 flasks containing DMEM with 10% FBS and then incubated in a humidified atmosphere of 5% CO2 and 95% air at 37°C. The medium was changed every 3 to 4 days. All experiments were done using cells in the passage numbers 2 to5 at 70% confluency. Treatment media were prepared by adding 0 (control), 10−11, 10−8, and 10−6 M nicotine. 1,25 All treatments were performed in triplicate within each experiment (for each experimental repeat, each condition was tested with three flasks of cells. Thus, the stock of healthy cells was divided into 12 flasks and every three flasks had the same treatment and conditions). Cells were incubated at 37°C with 5% CO2. Medium pH was not affected by the addition of nicotine. Cell lysate was prepared after 24-hour culture period and stored at −20°C.
Determination of Total Intracellular Glutathione
The assay of GSH with 5,5′-Dithiobis-(2-nitrobenzoic acid) (DTNB) was performed followed by a standard Ellman’s method described previously. 26,27 The standard curve was generated using a 1 mM solution of GSH. The clear supernatant from the cell lysate was analyzed for GSH levels. Moreover, 2.3 mL of potassium phosphate buffer (0.2 M, pH 7.6) was added to 0.2 mL of the cell lysate supernatant, followed by the addition of 0.5 mL DTNB (0.001 M) in a buffer. The absorbance of the reaction products was observed after 5 minutes at 412 nm.
Determination of GPx Activity
The procedures of Fecondo and Augusteyn 28 that monitor the continuous regeneration of reduced glutathione from oxidized glutathione (G–S–S–G), in the presence of GR and disodium (Na2) salt of reduced nicotinamide adenine dinucleotide phosphate (NADPH; Fluka Chemical Company, Switzerland) and t-BuOOH, were used for the determination of GPx activity with minor modifications. 29 The enzyme activity in the clear supernatant of the endometrial cell lysates was expressed as micromoles of NADPH oxidized per milligram of cell protein, using a molar extinction coefficient of 6.22 × 106 M−1 cm−1for NADPH. One unit of GPx is defined as mU/mg cell protein.
Determination of GR Activity
The activity of GR was assayed using the method described by Racker with minor modifications. 30 The GR assay was performed in a cuvette in a total volume of 1 mL that included 60 μM buffer, 5 mM EDTA (pH 8.0), 0.033 M GSSG, 2 mM NADPH, and the sample. The decrease in absorbance, which reflects the oxidation of NADPH during reduction of GSSG by GR present in the sample, was monitored spectrophotometrically at 340 nm for 3 minutes. Results were based on a molar extinction coefficient for NADPH of 6.22 × 106 M−1 cm−1. One unit of GR is defined as mU/mg cell protein.
Determination of Catalase Activity
Catalase was assayed spectrophotometrically by monitoring the decomposition of H2O2, using the procedure of Aebi. 31 To 100 μL of 30 mmol/L H2O2 solution in 350 μL phosphate buffer (pH 7.0), 50 μL of endometrial cell lysate was added, and the consumption of H2O2 was followed spectrophotometrically at 240 nm for 3 minutes at 25°C.
Catalase activity was expressed as mmol H2O2 consumed/min per mg endometrial cell lysate protein, using a molar extinction coefficient of 43.6 L/mol per cm for H2O2. An International Unit of catalase (CAT) (IU) is defined as the amount of enzyme causing the decomposition of 1 μmol H2O2 per minute at 25°C and pH 7.0.
Determination of Malondialdehyde
Malondialdehyde (MDA), as a marker of lipid peroxidation, was estimated according to a colorimetric method. 32 Briefly, 0.5 mL of the supernatant was added to 2 mL TBA reagent containing 0.375% thiobarbituric acid (TBA), 15% trichloroacetic acid, and 0.25 mol/L HCl. The mixture was boiled in a water bath at 95°C for 30 minutes and after fast cooling it was centrifuged at 8,000 g for 15 minutes at 4°C for4 minutes and after fast cooling it was centrifuged at 8,000 nd 0 nm. The mixture was boiled in a water bath at 95°C for 30 minutes and after fast cooling it was centrifuged at 8000g for 15 min at 4°C. The absorbance of the pink color supernatant was measured at 532 nm. Malondialdehyde concentration was calculated using 1,1,3,3-tetraethoxypropane (TEP) as standard and expressed as nmol /mg protein.
Protein Concentration
The protein concentration (mg/mL) was determined with the method of Bradford using bovine serum albumin as the standard. 33
Results
Effect of Nicotine on Cell Morphology
As shown in Figure 1, the cells were grown under confluent culture conditions at the same time. Treatment with different doses of nicotine did not induce a significant effect on the cell growth and morphology compared to the control cells.
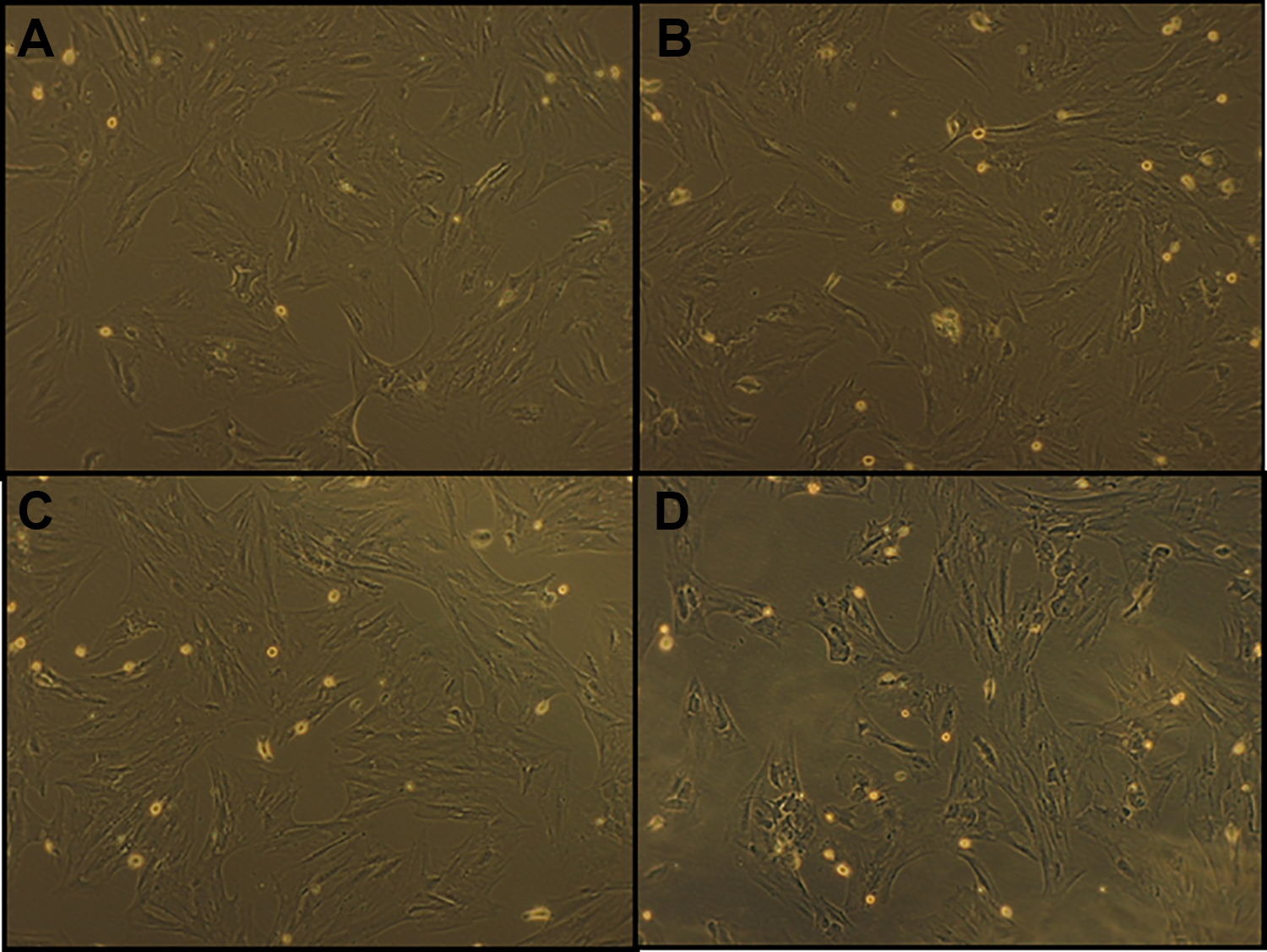
Phase-contrast micrograph of adherent human endometrial cells. Cells were grown under confluent culture conditions at the same time. (A) Control human endometrial cells, (B) human endometrial cells treated with 10−11 M of nicotine, (C) human endometrial cells treated with 10−8 M of nicotine, and (D) human endometrial cells treated with 10−6 M of nicotine (×100).
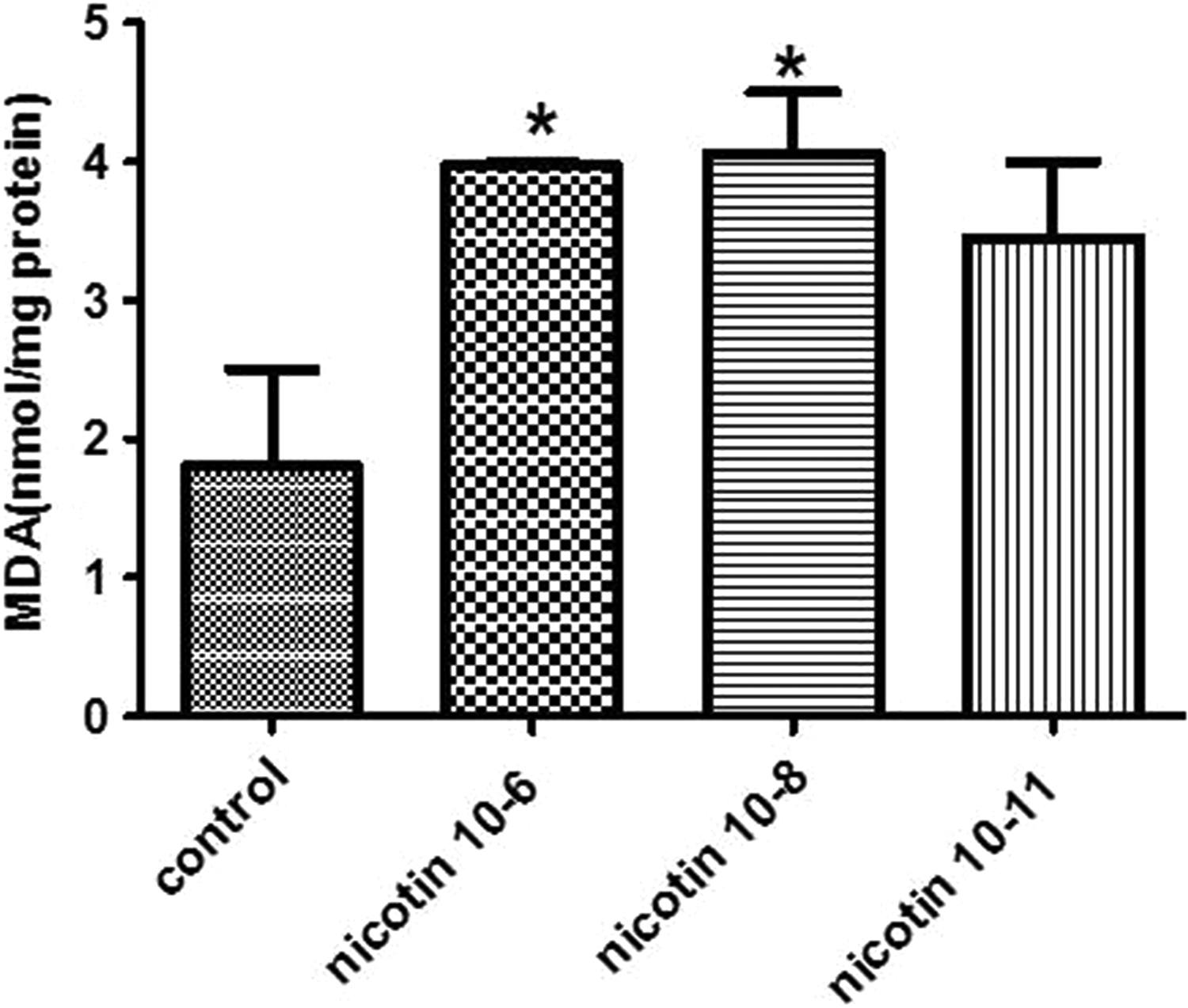
Effect of Nicotine on MDA Levels
Figure 2 shows MDA level alterations in the endometrial stromal cells following nicotine exposure. The results reveal that MDA level in the cells treated with 10−6 and 10−8 M concentrations of nicotine was significantly increased compared to untreated control cells (P < 0.05).
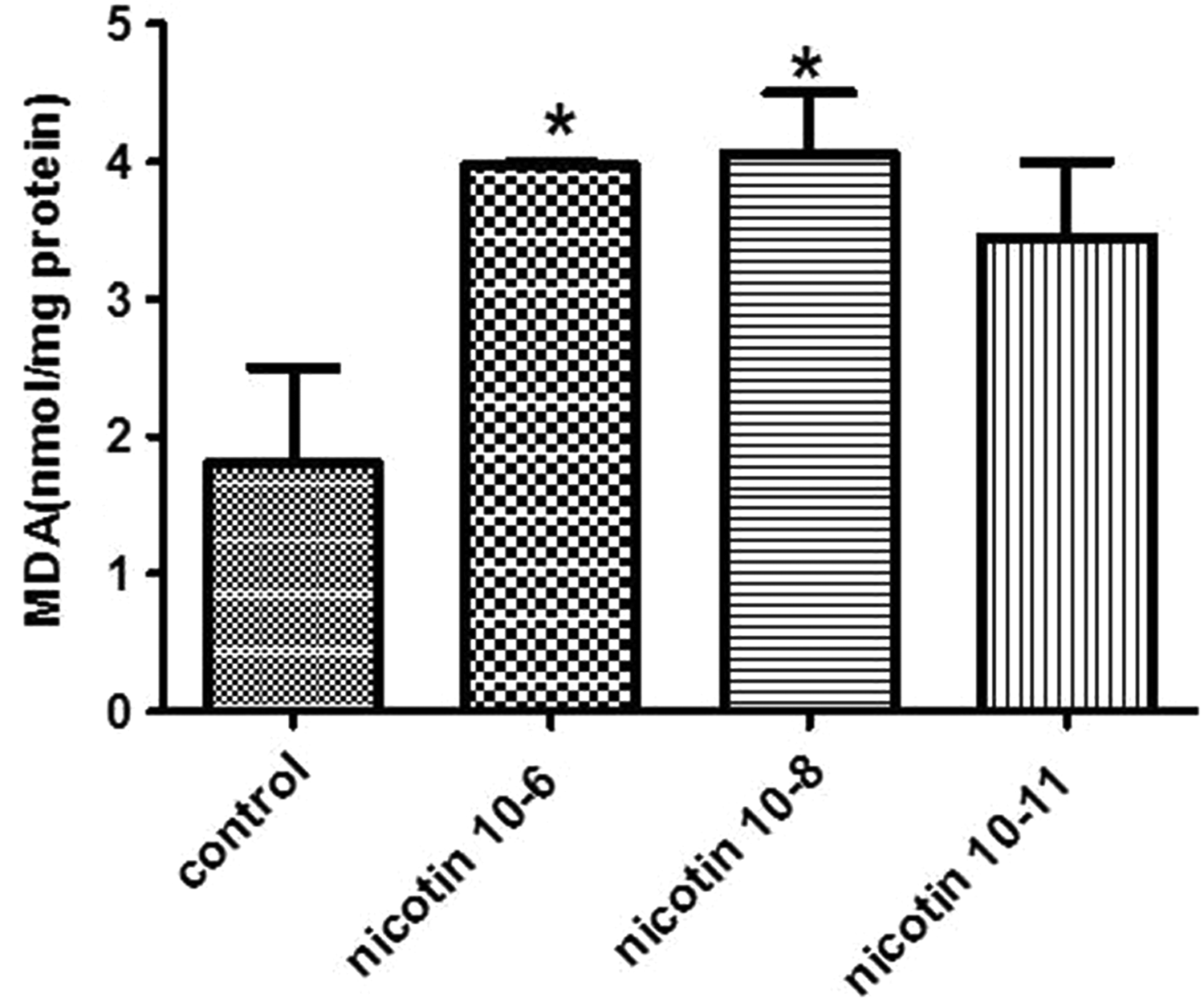
Effect of 10−6, 10−8, and 10−11 M of nicotine on the malondialdehyde (MDA) level in human endometrial cells. Data are presented as mean ± standard error of mean (SEM). *P < 0.05 for significant change compared with the control group.
Effect of Nicotine on GPx Activity
Figure 3 demonstrates the effect of nicotine exposure on GPx activity. Based on the results, however, all the 3 nicotine concentrations decreased the GPx enzyme activity in the endometrial stromal cells; only the effect of highest concentration (10−6 M) was significant compared to the control group (P < 0.05).
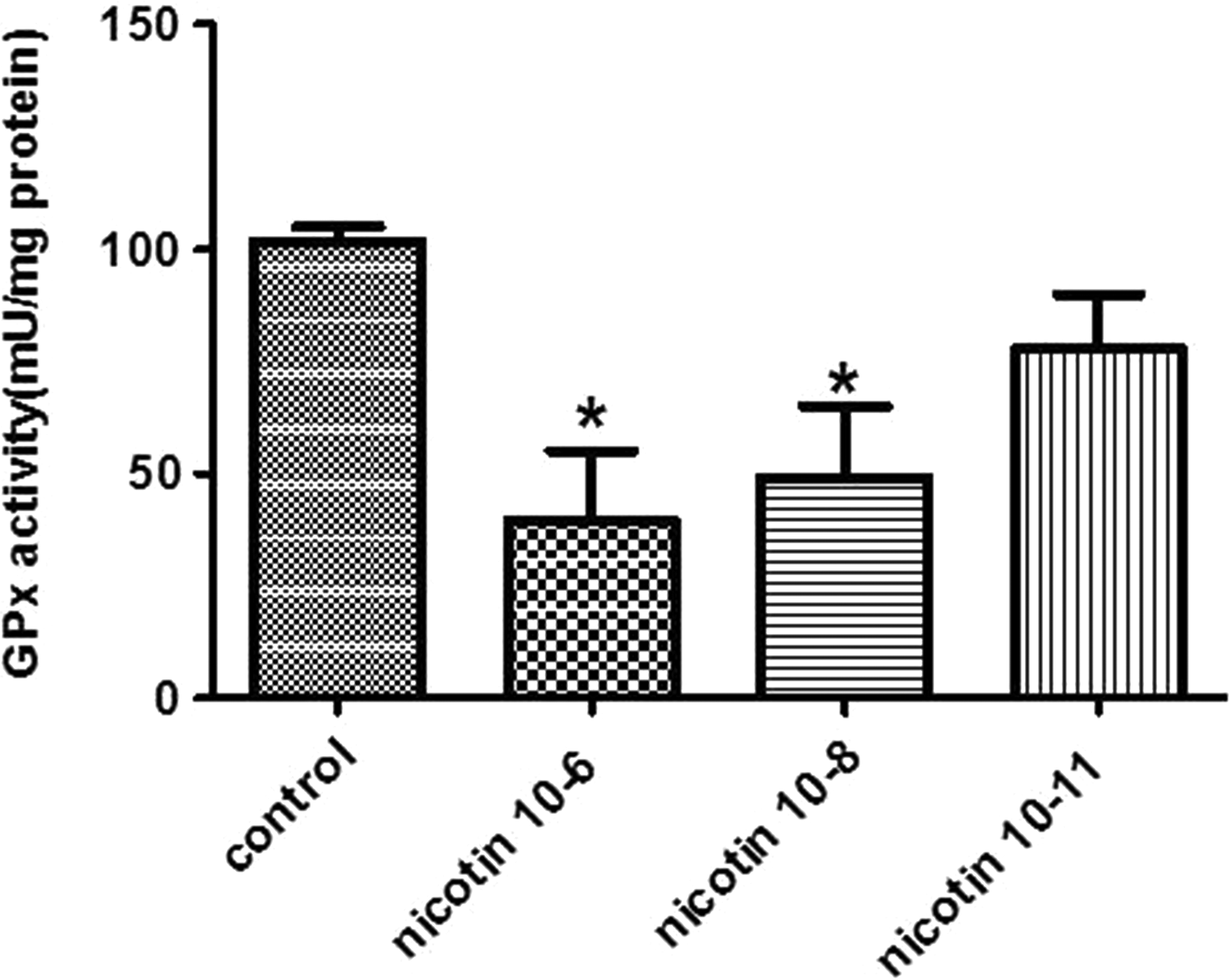
Effect of 10−6, 10−8, and 10−11 M of nicotine on the activity of glutathione peroxidase (GPx) in human endometrial cells. Data are presented as mean ± standard error of mean (SEM). *P < 0.05 for significant change compared to the control group.
Effect of Nicotine on GR Activity
Figure 4 indicates changes in GR activity in the nicotine-exposed endometrial stromal cells. We found a significant decrease in GR enzyme activity after nicotine exposure by 10−6 and 10−8 M concentrations compared to the control group (P < 0.05).
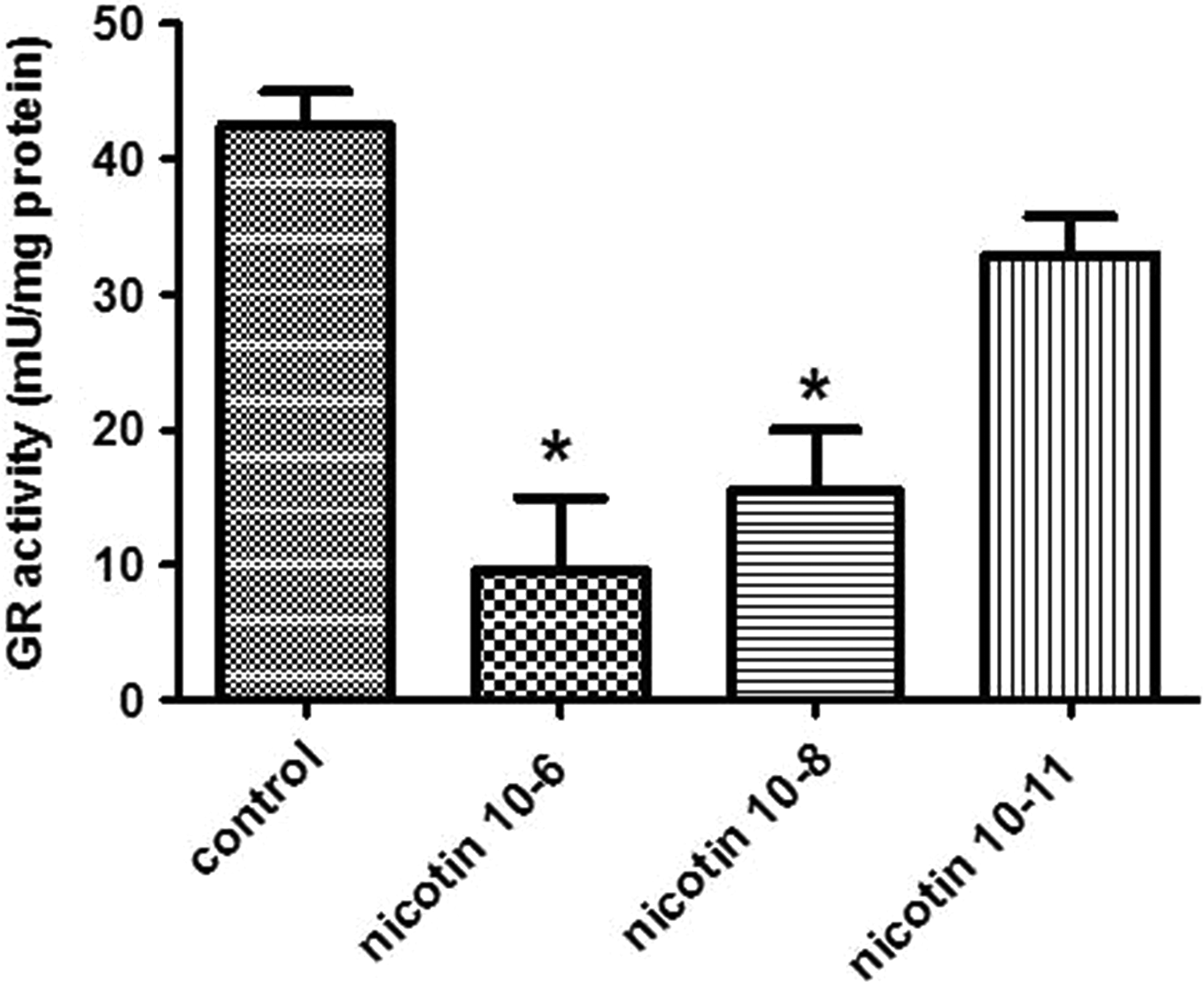
Effect of 10−6, 10−8, and 10−11 M of nicotine on the activity of glutathione reductase (GR) in human endometrial cells. Data are presented as mean ± standard error of mean (SEM). *P < 0.05 for significant change compared with the control group.
Effect of Nicotine on GSH Concentration
Figure 5 depicts the effects of nicotine treatment on the endometrial stromal cells. We observed that all the 3 nicotine concentrations significantly decreased the GSH level in the endometrial stromal cells compared to the control group (P < 0.05).
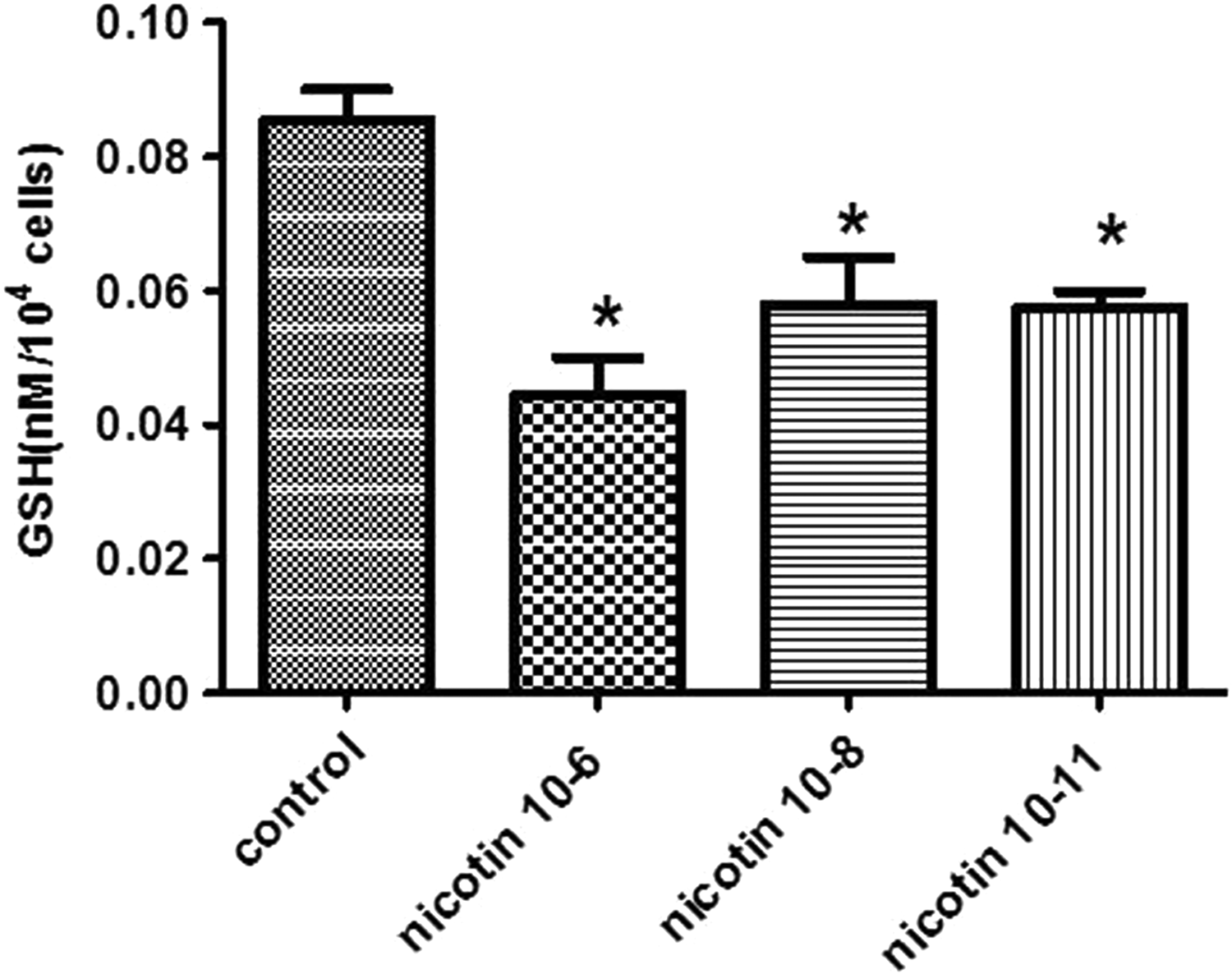
Effect of 10−6, 10−8, and 10−11 M of nicotine on the GSH level in human endometrial cells. Data are presented as mean ± standard error of mean (SEM). *P < 0.05 for significant change compared with the control group.
Effect of Nicotine on CAT Activity
The effects of nicotine exposure on CAT activity of the endometrial stromal cells are shown in Figure 6. Based on the results, only 10−6 and 10−8 M concentrations of nicotine could significantly decrease the CAT activity compared to the control group (P < 0.05).
Effect of 10−6, 10−8, and 10−11 M of nicotine on the activity of catalase (CAT) in human endometrial cells. Data are presented as mean ± standard error of mean (SEM). *P < 0.05 for significant change compared with the control group.
Discussion
To study the effects of nicotine on the endometrium, as an in vitro experimental model system, we used the culture of primary human endometrial stromal cells from endometrial biopsies obtained from reproductive-age fertile women (aged 20-40 years). In this study, we explored the direct effects of nicotine administration on the antioxidant defense system and lipid peroxidation. Nicotine-treated endometrial stromal cells were associated with a decrease in antioxidant defense markers and higher levels of MDA in a dose-dependent manner when compared ti the controls. A variety of no neuronal tissues have been demonstrated recently to express nicotinic acetylcholine receptors (nAChRs). As Krasnyi et al detected and studied for the first time that α7-Nicotinic acetylcholine receptor (α7nAChR) is the nicotine receptor in human endometrium; it seems that the effect of nicotine on endometrium is related to this receptor. 34
Reactive oxygen species are generated in aerobic processes in cellular interior and in limited quantities it has a unique role in female reproductive physiology. Thus, tightly controlled pro-oxidant–antioxidant ratio is crucial to secure normal reproductive function. Therefore, oxidative stress induction could be a potent, underlying mechanism for various reproductive abnormalities. 35 -37 Oxidative stress, inflammation, and ER stress are some proposed cellular mechanisms by which nicotine exposure can adversely influence the reproductive health outcomes. 38
Although harmful effects of nicotine on different parts of female reproductive system have been reported previously, 10,39,40 the relationship between nicotine and oxidative stress in the endometrial cells has not been sufficiently studied.
Malondialdehyde is one of the most used biomarkers of lipid peroxidation, indicating tissue damage caused by free radicals in biological fluids. The results obtained from our study indicated that in nicotine-exposed endometrial cells, the level of MDA was increased which is consistent with previous reports that already reported the same results in the plasma and liver tissues of nicotine-treated rats. 41,42 Increased levels of MDA in human hepatic HepG2 cell line following nicotine treatment were also observed in the findings of Yarahmadi et al. Moreover, in the studies conducted by Kamceva et al, MDA levels in the plasma were significantly increased in smokers compared to nonsmokers. 43 The enhanced lipid peroxidation after nicotine exposure is indicative of cellular membrane oxidative injury. After noticing the significant increase in MDA production, the level of GSH, known as the predominant scavenger which reacts directly with ROS and free radicals and maintains cellular stability, was evaluated.
The GSH levels were significantly decreased in all nicotine concentrations coinciding with the increase in MDA. This finding demonstrates that in comparison to other antioxidant enzymes, GSH is more prone to oxidative damage caused by enhanced ROS/free radicals, and nicotine showed a significant negative correlation between reduced glutathione and MDA. Reduced levels of GSH in the liver and testes of rats following chronic administration of nicotine were also observed in the findings of Husain et al. 44 Based on the findings of our experiment, we can hypothesize that oxidative stress may be a contributing factor in the development of reproductive abnormalities in female smokers. In order to explain this theory further, we decided to measure the levels of antioxidant enzymes known to be related in cellular regulation of ROS, that is, GPx, GR, and CAT.
Glutathione peroxidase has been named as the principal enzyme for the detoxification of hydrogen peroxide 45 and, therefore, plays an important part in the protection against ROS. Glutathione reductase is another important enzyme of the antioxidant defense system, and it is involved in reducing glutathione from its inactive form (GSSG) to GSH. 46 In this study, we observed that following endometrial cell treatment with different concentrations of nicotine, GPx and GR activities decreased.
Although activity of both enzymes was significantly decreased, GR activity reduction was more prominent. When GSH production by GR enzyme decreased more than its consumption by Gpx enzyme, the reduction observed in the level of GSH can be explained. This finding suggests the role of glutathione homeostasis alteration in oxidative stress and nicotine cytotoxicity.
Although some observations indicated that nicotine treatment caused a decrease in GR and CAT activity, while the glutathione concentration and Gpx activity were enhanced, 12,47 the findings of Jain et al are compatible with our results. They reported a significant decrease in the blood, liver, and brain activities of GPx and GR as a consequence of nicotine exposure in male Wistar rats. 11 A possible explanation in the observed differences could be the fact that the cell types between the studies were different. It is also worth mentioning that chronic nicotine exposure can lead to this discrepancy.
Conclusion
In conclusion, our findings suggest that nicotine has a harmful effect on response to oxidative stress through decreasing the levels of antioxidant enzymes and antioxidant processes that lead to uncompensated ROS production. However, the deleterious effects of nicotine in reproductive organs may be mediated through different mechanisms other than oxidative stress, but since the nicotine-induced oxidative stress may be an important and potentially modifiable pathway, investigating different aspects of this mechanism may lead to an inexpensive and noninvasive therapy for smoking-induced fertility problems and might be beneficial in the clinical recovery of the subject exposed to nicotine.
Footnotes
Acknowledgment
The authors would like to thank Shiraz University of Medical Sciences, Shiraz, Iran and also Center for Development of Clinical Research of Nemazee Hospital and Dr. Nasrin Shokrpour for editorial assistance.
Author Contributions
Zal, F. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Khademi , F. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Totonchi, H. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Mohammadi, N. contributed to conception, contributed to acquisition, drafted manuscript, and critically revised manuscript; Zare, R. contributed to conception, contributed to acquisition, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This article was supported by Grant Number 12605 from Vice-chancellor for Research Affairs of Shiraz University of Medical Sciences.
