Abstract
Fulranumab, an anti-human nerve growth factor antibody, was evaluated in a series of nonclinical toxicology studies. No treatment effects were observed in adolescent cynomolgus monkeys in standard design, repeat-dose toxicology studies of up to 6 months. Adverse effects on the developing nervous system were observed in offspring of pregnant cynomolgus monkeys treated with fulranumab. Subsequent studies including detailed morphologic investigations of the nervous system did reveal fulranumab-related changes in adult cynomolgus monkeys; this article is focused on those findings. A single dose of ≥1 mg/kg fulranumab administered subcutaneously (SC) caused a decrease in neuron and sympathetic ganglion size (superior cervical ganglion), observed morphologically and stereologically, with a resulting appearance of increased glial cell density. Similar results were observed in repeat-dose (15 to 52 weeks) toxicity studies at ≤50 mg/kg/wk fulranumab SC. These effects recovered after a 3-month treatment-free period. Fulranumab did not cause any neuronal death, necrosis, apoptosis, or any apparent decrease in function of sympathetic neurons/ganglia at any time point examined. A no observed effect level (NOEL) was established at 0.25 mg/kg fulranumab SC every 4 weeks for 28 weeks.
Introduction
Nerve growth factor (NGF) is associated with pain and increased nociception in bone diseases such as osteoarthritis leading to NGF antagonists being investigated as analgesics. To support clinical development of fulranumab, a fully human anti-NGF immunoglobulin G2 monoclonal antibody, a series of standard general toxicology studies in adolescent cynomolgus monkeys were performed. Studies included a single-dose cardiovascular safety pharmacology study and repeat-dose toxicity studies of ≤6 months in duration. The autonomic nervous system was not specifically evaluated as part of these standard general toxicology studies. The no observed effect levels (NOELs) were the highest doses tested: a single dose of 300 mg/kg intravenously (IV) and 150 mg/kg/wk IV or subcutaneously (SC).
Treating pregnant cynomolgus monkeys in a combined embryo/fetal development and peri-/postnatal development study with 10 or 50 mg/kg/wk fulranumab SC, beginning on gestation day 20 through Caesarean delivery or parturition, had no adverse effects on maternal animals but resulted in adverse effects on offspring. Effects in infants were increased infant mortality/morbidity, skin lesions, and reduced body weight gain. In the postmortem evaluations of fetuses and infants, there was a decrease in the overall size of the sural nerve and a decrease in the number of neurons in sympathetic ganglia, including the cranial mesenteric ganglia, cervicothoracic ganglia, and the sympathetic trunk (with a corresponding decrease in overall size in the latter 2 structures) in both the 10 and 50 mg/kg/wk groups. Developmental effects were due to neuronal aplasia, hypoplasia, and/or early atrophy; a no adverse effect level (NOAEL) was not determined. These results were not unexpected based on the role of NGF in neuron development and effects observed in rodents. 1 -5 The results were similar but not identical to those reported for tanezumab (another anti-NGF mAb). 6
While it has been known for decades that NGF and its receptors, trkA and p75, play a role in nervous system development, the role of NGF in adults is still being elucidated (reviewed by Denk et al 7 ). Subsequent studies of fulranumab in adult monkeys included detailed functional and neuropathologic evaluations of the central and peripheral (including the autonomic) portions of the nervous system; this article is focused on those studies.
Methods
The toxicology studies were performed according to Good Laboratory Practices at SNBL USA (Everett, Washington), an Association for Assessment and Accreditation of Laboratory Animal Care International accredited test facility, according to an Institutional Animal Care and Use Committee approved protocol. Animal care and use were in accordance with the Animal Welfare Act and recommendations set forth in the Guide for the Care and Use of Laboratory Animals. 8
Fifteen-Week Study
Sexually mature cynomolgus monkeys (7/sex/group) were given 0 (saline), 10, or 50 mg/kg/wk fulranumab SC for 15 weeks (mean control body weight at the end of dosing: male = 6.2 kg; female = 3.5 kg) followed by a 12-week treatment-free recovery period (3/sex/group; mean control body weight at the end of recovery: male = 6.2 kg; female = 3.8 kg). Assessments included clinical observations, food consumption, body weight, menses checks, hormone levels, semen analysis, testicular volume, hematology, coagulation, serum chemistry, urinalysis, toxicokinetics (TK), anti-drug antibody (ADA), gross necropsy findings, organ weights, microscopic evaluations of standard tissues, and specialized histopathological evaluations (described below) focused on the central and peripheral nervous systems. Specific parameters assessed are shown in Table 1.
Specific Parameters Assessed in Each Study.a

aX = assessment was performed; – = assessment not performed.
Acute Study
Eight groups of sexually mature cynomolgus monkeys (3/sex/group) were given 0 (saline), 1, 10, or 50 mg/kg fulranumab SC as either a single dose or weekly (QW) for 5 weeks. Animals were necropsied 3 days after the last dose (on day 4 for single-dose [mean control body weight: male = 8.5 kg; female = 4.7 kg] or day 32 for 5-dose animals [mean control body weight: male = 6.7 kg; female = 3.2 kg]). Assessments included clinical observations, body weight, TK, gross necropsy, and specialized histopathological evaluations (described below) focused on the central and peripheral nervous systems. Sympathetic nervous system (SNS) function was assessed by ocular neurological observations, including pupil reactivity (using a pen light), eye movement (conjugate eye movement, strabismus, nystagmus) and dilation abnormalities (miosis and mydriasis). Specific parameters assessed are shown in Table 1.
Chronic (28- to 52-Week) Study
Sexually mature cynomolgus monkeys were given saline or fulranumab SC every 4 weeks (Q4W) for 28 weeks or once QW for 28 or 52 weeks. Recovery was evaluated either 13 or 24 weeks following 28 weeks of dosing and 26 weeks following 52 weeks of dosing, as shown in Table 2. Assessments included clinical observations, food consumption, body weight, physical examination, hematology, coagulation, serum chemistry, electrocardiography (ECG), blood pressure, rectal temperature, respiratory rate, serum and semen TK, serum ADA, gross necropsy findings, organ weights, and microscopic evaluations of standard tissues (Table 1). Sympathetic nervous system function was assessed by ocular neurological observations and specialized histopathological evaluations were performed (described below) focused on the central and peripheral nervous systems. Mean control body weights prior to necropsy were as follows: 6 months, male = 5.8 kg, female = 3.3 kg; 9 months, male = 5.7 kg, female = 3.8 kg; 12 months, male = 6.3 kg, female = 4.0 kg; 18 months, male = 7.1 kg, female = 3.8 kg.
Chronic Study Design.

Abbreviation: Q4W, every 4 weeks; –, not applicable.
Neurohistopathology
Tissue preparation and neurohistopathology were performed as described previously. 6,9,10 Briefly, following euthanasia, whole-body perfusion with 4% paraformaldehyde was performed and the brain, spinal cord, peripheral nerves, dorsal root ganglia, sympathetic ganglia, and parasympathetic ganglia were dissected and preserved at SNBL or Tox Path Specialists, LLC (Frederick, Maryland). Tox Path Specialists performed the neurohistopathic evaluation. Morphologic evaluation of representative areas from the brain, spinal cord, peripheral nerves, cervicothoracic ganglia, sympathetic trunk ganglia, superior mesenteric ganglia, superior cervical ganglia, and thoracic dorsal root ganglia was performed utilizing standard (paraffin/H&E) and/or special staining/embedding techniques (Table 3). Morphometric evaluations of the myelinated nerve fibers in the sural nerve and unmyelinated nerve fibers in the epidermis (footpad) were conducted. Stereologic investigations of ganglion size, total neuron number, and average neuron size were performed on the superior cervical ganglion and thoracic dorsal root ganglion. Each ganglion (left and right separately) was cryoprotected, embedded in gelatin, frozen using dry ice on a freezing sliding microtome, then sectioned completely through. The section thickness was approximately 50 µm. Ten to 20 sections for stereology were mounted to glass slides and stained with Cresyl violet (Nissl stain). The volume of the ganglion was determined as the sum of the volumes of the individual sections examined (Cavalieri method), multiplied by the inverse of the section sampling fraction. Neuron counts were calculated using the optical fractionator method. The average cross-sectional neuron area was calculated based on the radius of each neuron in each section examined using the nucleator method (radius [r] of individual cells measured; area of individual neurons = π·r2).
Neural Tissues Evaluated in Cynomolgus Monkey Studies.

Abbreviations: ChAT, anti-choline acetyltransferase (stain for cholinergic neurons); CTG, cervicothoracic ganglion; CV, cresyl violet (Nissl stain to demonstrate neurons); FJB, Fluoro-Jade B (stain for necrotic neurons); GFAP, glial fibrillary acidic protein (stain for astrocyte reactions); H&E, hematoxylin and eosin (morphology stain); N, number of animals; Neuro Apop, stain for apoptosis; PGP 9.5, frozen section/protein gene product 9.5 (labels neuron cytoplasm including axons/dendrites); Q4W, every 4 weeks; QW, weekly; SCG, superior cervical ganglion; SMG, superior mesenteric ganglion; STG, sympathetic trunk ganglia; TB, osmium post-fixation/resin embedding/toluidine blue (stain for myelin); TDRG, thoracic dorsal root ganglion.
Results
Fifteen-Week Toxicity
A 15-week repeat-dose toxicity study in sexually mature cynomolgus monkeys was performed primarily to evaluate the effect of fulranumab on reproductive tissues and functions. This was the first fulranumab study to include detailed neuropathology evaluations in adult monkeys.
Monkeys received 0 (saline), 10, or 50 mg/kg/wk fulranumab SC for 15 weeks followed by a 12-week treatment-free recovery period. All fulranumab-treated animals were exposed to the test article continuously during the treatment period. Serum fulranumab concentrations were slightly less than dose proportional between the 10 and 50 mg/kg/wk dose groups. The highest mean serum fulranumab concentration following the 13th dose was ∼1,292 µg/mL. At the end of the 12-week recovery period, mean serum fulranumab concentrations were still quantifiable: ∼7 and 24 µg/mL on average for the 10 and 50 mg/kg dose groups, respectively. No differences were observed in the TK parameters between male and female animals. All animals were negative for ADA at the end of the treatment period. One recovery animal tested positive for ADA during the recovery phase.
Administration of ≤50 mg/kg/wk fulranumab SC for 15 weeks had no effect on clinical observations, body weight, food consumption, menstruation, semen parameters, testicular volume, hormone levels, hematology, serum chemistry, coagulation, urinalysis, gross pathology, organ weights, microscopic evaluations of standard tissues, the central nervous system, density of intraepidermal nerve fibers in footpads, total myelinated nerve fibers (and fiber size distribution) in the sural nerve, ganglion volume, neuron number, and average neuron size within the thoracic dorsal root ganglia.
An apparent increase in the density of glial cells (presumably predominantly satellite glial cells, but Schwann cells could not be excluded as contributing to the cell density) in one or more ganglia of the SNS (cervicothoracic, sympathetic trunk, superior mesenteric, superior cervical, and thoracic dorsal root) was observed histologically at both dosages in both sexes at the terminal and recovery necropsies (Figure 1). Stereology evaluations were performed on the superior cervical ganglion; decreases (as compared to concurrent controls) were observed in ganglion volume, neuron number, and neuron size (Figure 2). The decreases in total calculated neuron number and neuron size were not associated with any indications of neuronal cell necrosis or SNS dysfunction. The increase in density of satellite glial cells was attributed to the reduced ganglion volume (unaltered absolute number of glial cells within a reduced volume) and the reduced size of the neurons (bringing existing satellite glial cells closer together), not to any actual increase in the glial cell populations. Complete/partial recovery of these effects was demonstrated following the 12-week recovery interval. Overall, these changes were not considered adverse because they were not associated with any signs of SNS dysfunction.
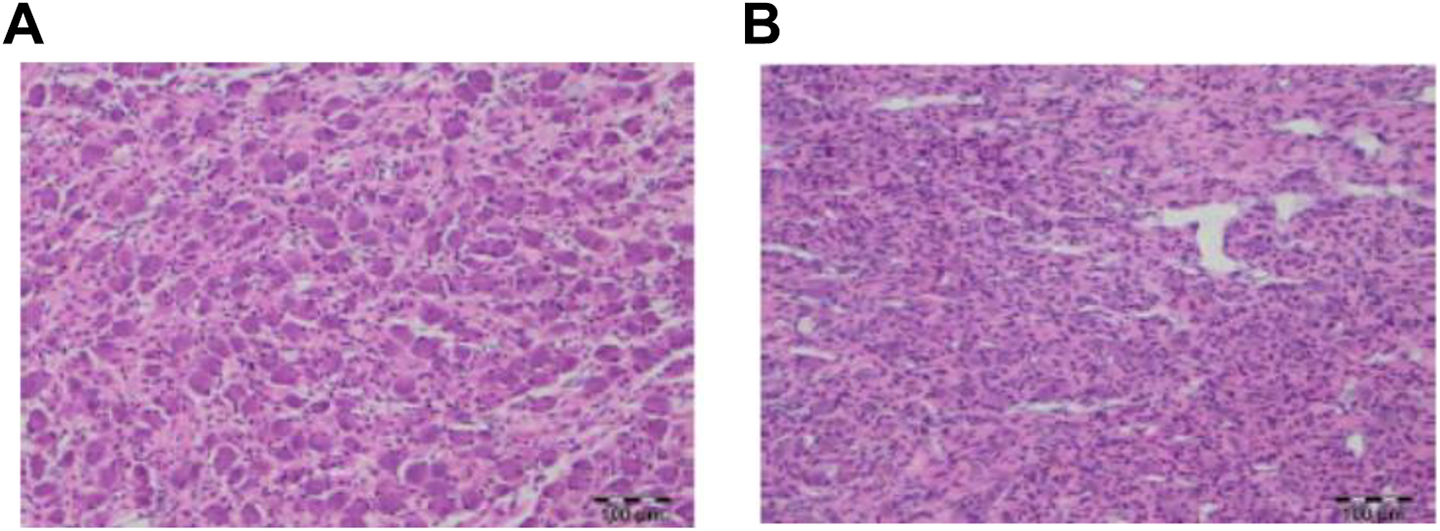
Representative images of superior cervical ganglia. A, Image from a control male with normal glial cell (small dark cells) density and neuron size (large cells). B, Image from a 50 mg/kg/wk fulranumab-treated male illustrating the diffuse increase in satellite glial cell density and decrease in neuron size (×10 magnification of H&E-stained paraffin-embedded sections): (A) control and (B) 50 mg/kg fulranumab.
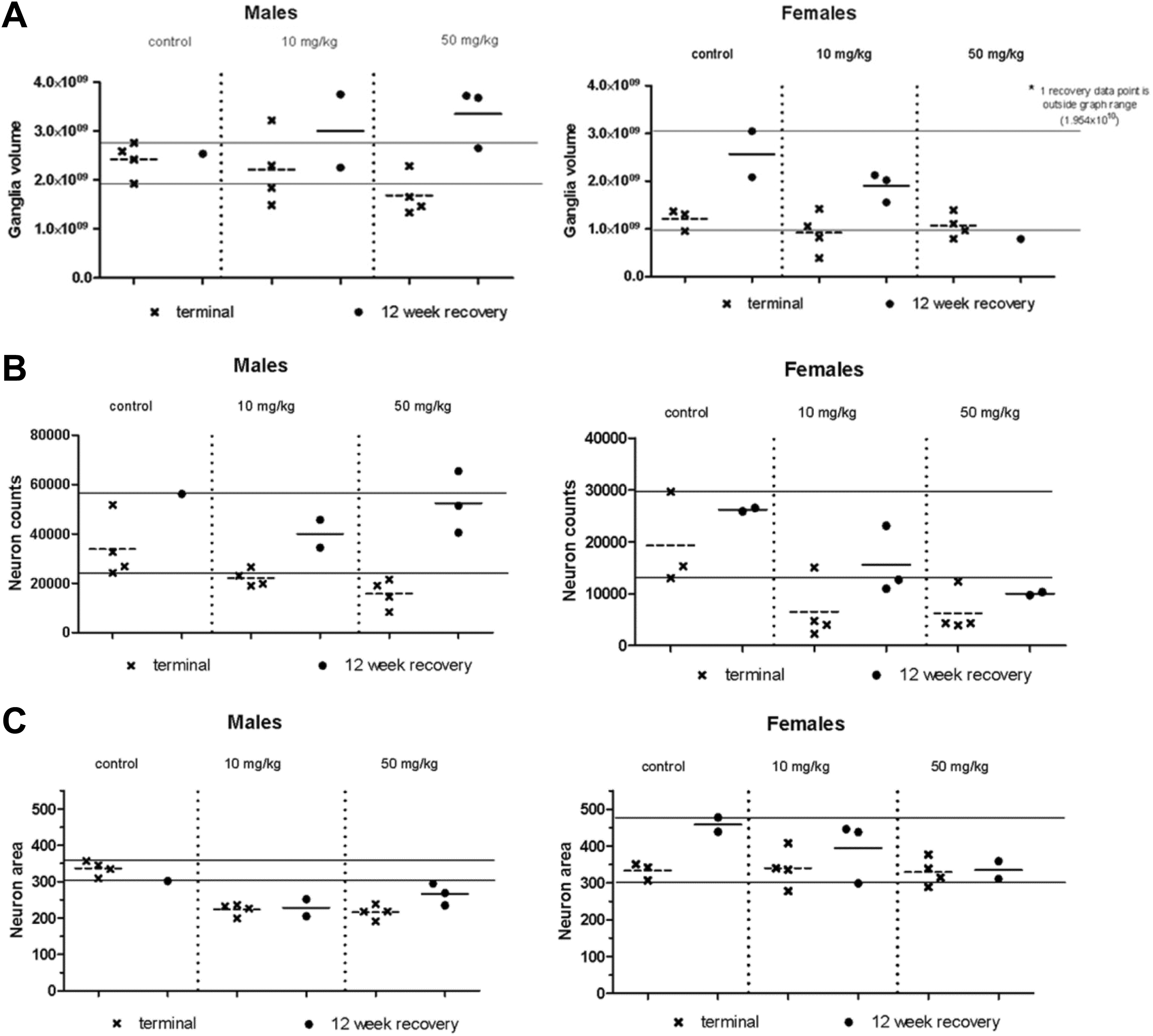
Fifteen-week study superior cervical ganglion stereology results. A, Ganglion volume (μm3). B, Neuron number. C, Neuron area (average neuron size, μm2). Data are plotted as individual animal values with the cross-bar representing the mean value. Data points are from the terminal necropsy (
Acute Toxicity
With many neurotoxicants, neuronal necrosis and/or apoptosis of susceptible neurons may occur within a small window of time following the initial exposure, and repeated administration does not always result in affecting additional neurons. 11 Therefore, an acute/subacute study was performed in sexually mature cynomolgus monkeys to detect any early neuronal necrosis that might occur 48 to 72 hours after a single dose or 5 QW doses. In addition, SNS function was assessed by ocular neurological observations (pupil reactivity, eye movement, and dilation).
Monkeys were given 0 (saline), 1, 10, or 50 mg/kg fulranumab SC as either a single dose or QW for 5 weeks. Monkeys were exposed to fulranumab throughout the dosing period. Serum fulranumab concentrations were dose proportional between the 1 and 10 mg/kg and less than dose proportional between the 10 and 50 mg/kg dose groups. No differences in the TK parameters between male and female animals were observed. Anti-drug antibody was not tested in this study.
Administration of 1, 10, or 50 mg/kg fulranumab SC as either a single dose or QW for 5 weeks had no effect on clinical observations (including no evidence of autonomic nervous system dysfunction), body weight, ocular neurological observations (Table 4), or gross necropsy.
Acute Study Ocular Neurological Observations.a
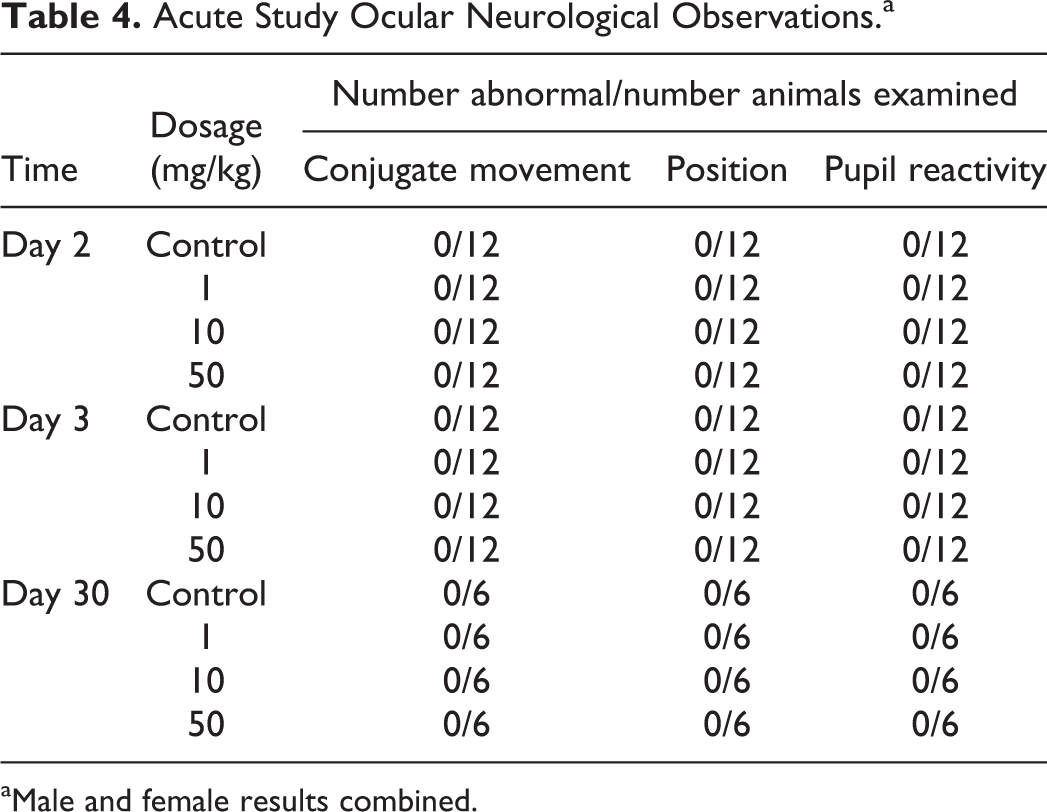
aMale and female results combined.
There was no evidence of neuron death, necrosis, or apoptosis in the SNS as determined through morphologic evaluations of the sympathetic trunk (with ganglia), the superior cervical, cervicothoracic, and superior mesenteric ganglion. A diffuse decrease in sympathetic neuron size and increased glial cell density in sympathetic ganglia were observed morphologically and a decrease in neuron number, neuron size, and ganglion volume was observed in stereological evaluations of the superior cervical ganglion in all fulranumab-treated groups after either a single dose or 5 QW doses compared to controls (Table 5, data were also reviewed in comparison to control ranges as shown in Figure 2; presented here in tabular form for brevity). Microscopic images were similar to those shown in Figure 1.
Acute Study Superior Cervical Ganglia Stereology Results.a
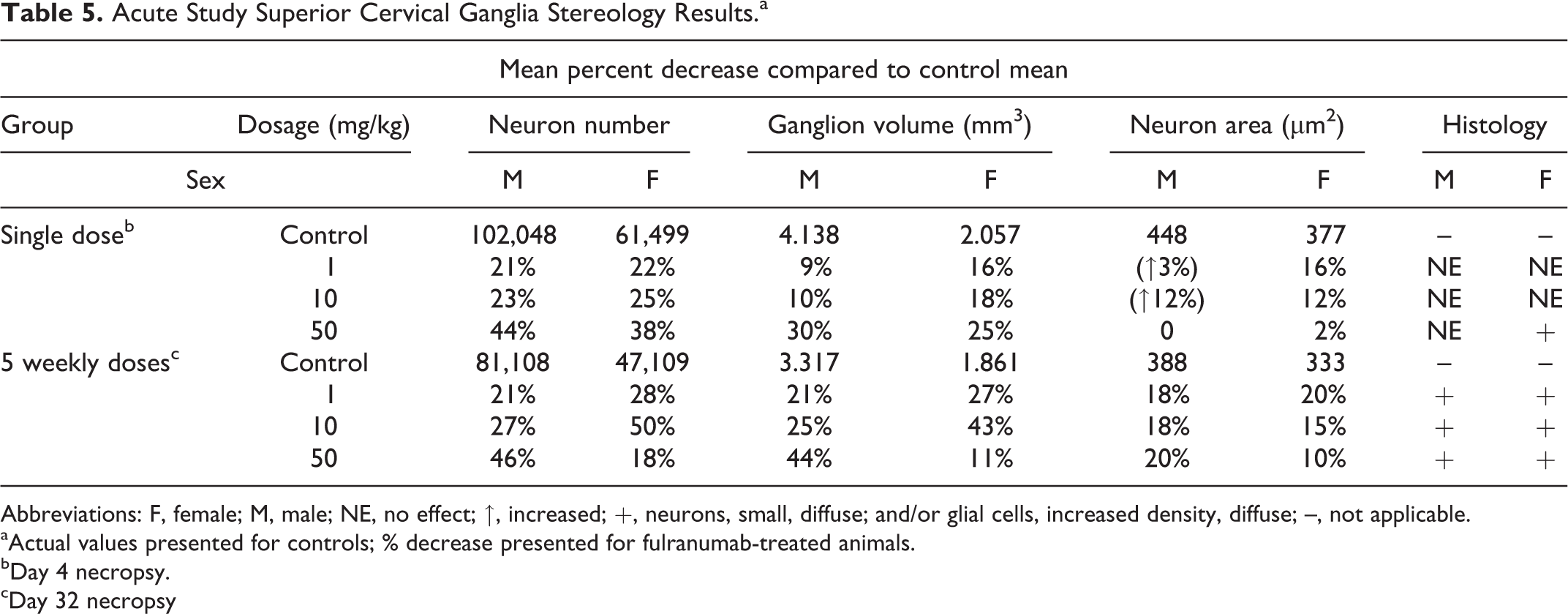
Abbreviations: F, female; M, male; NE, no effect; ↑, increased; +, neurons, small, diffuse; and/or glial cells, increased density, diffuse; –, not applicable.
aActual values presented for controls; % decrease presented for fulranumab-treated animals.
bDay 4 necropsy.
cDay 32 necropsy
The decrease in calculated neuron counts was attributed to under recognition of smaller (atrophied) neurons during the stereology investigation. This interpretation was supported by 2 main factors: the lack of morphologic observation of neuronal necrosis/apoptosis at any of the morphologic investigations using any of the staining or embedding techniques and the recovery/partial recovery of the calculated neuron counts during the recovery period. An actual loss of neurons would not be expected to show any recovery. As with the 15-week study described above, the increased glial cell density was attributed to the reduced ganglion volume and/or the reduced size of surrounding neurons, not to an absolute increase in glial cells. This interpretation was supported by the morphologic appearance of the ganglia, including a general return to a normal cellular density during the 12-week treatment-free period in the 15-week study.
Chronic (28- to 52-Week) Toxicity
A chronic toxicity study was performed to evaluate the toxicity of long-term treatment with fulranumab, to determine if the effects were reversible during a treatment-free recovery period, and to identify a NOAEL, if possible. The dosages in this study were chosen to yield at least a 5-fold higher exposure in monkeys (area under the curve [AUC] at steady state) than the exposure at the 3 mg Q4W dosage in clinical studies.
Monkeys were given 0.25 or 0.7 mg/kg fulranumab SC Q4W for 28 weeks and 0.2 or 1 mg/kg once QW for 28 or 52 weeks. Serum fulranumab concentrations were approximately dose proportional following the Q4W or QW dosing schedule (Table 6). Steady-state serum fulranumab concentrations were reached by day 85. Most recovery animals had serum fulranumab concentrations below the lowest quantifiable concentration (<0.04 µg/mL) at the end of the recovery period. The half-life of fulranumab ranged from 10 to 12 days (determined in the QW groups). No differences in the TK parameters between male and female animals were observed. On average, ∼25% of fulranumab-treated animals tested positive for ADA.
Chronic Study Mean (SD) Serum Fulranumab Exposures.
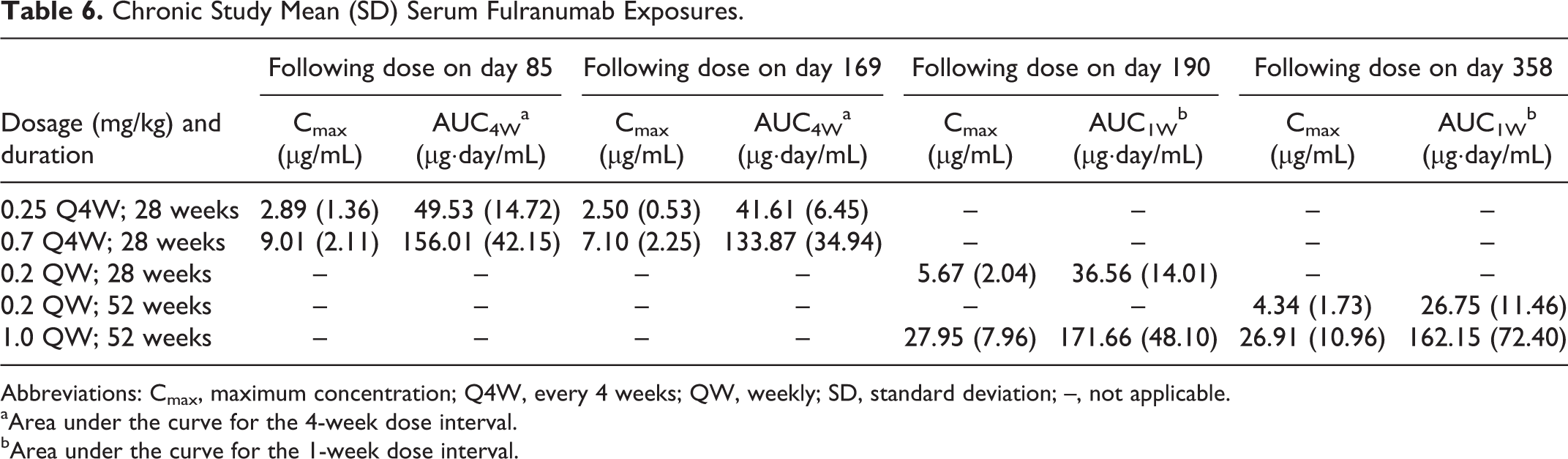
Abbreviations: Cmax, maximum concentration; Q4W, every 4 weeks; QW, weekly; SD, standard deviation; –, not applicable.
aArea under the curve for the 4-week dose interval.
bArea under the curve for the 1-week dose interval.
Administration of 0.25 or 0.7 mg/kg fulranumab SC Q4W for 28 weeks and 0.2 or 1 mg/kg once QW for 28 or 52 weeks had no effect on clinical observations, food consumption, body weight, physical examination, hematology, coagulation, serum chemistry, ocular neurological examinations (Table 7), ECG, blood pressure, rectal temperature, respiratory rate, gross necropsy findings, organ weights, or microscopic evaluations of standard tissues.
Chronic Study Ocular Neurological Observations.a
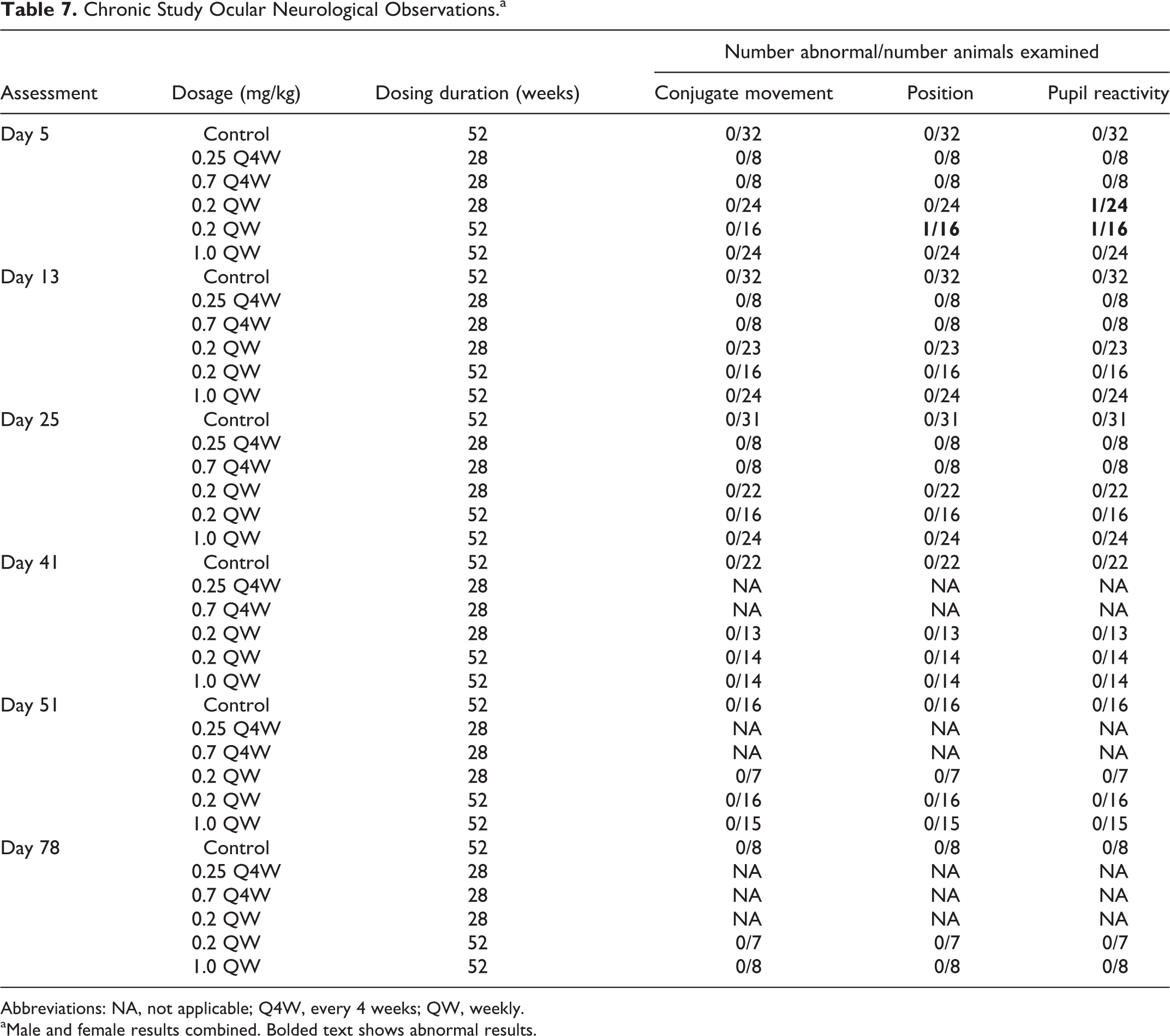
Abbreviations: NA, not applicable; Q4W, every 4 weeks; QW, weekly.
aMale and female results combined. Bolded text shows abnormal results.
No effects of treatment were observed in monkeys given 0.25 mg/kg fulranumab SC Q4W for 28 weeks (NOEL). Of the 8 animals in the group, 1 male was ADA positive.
Effects at higher doses were similar to those observed in previous studies diffuse small neurons and increased glial cell density in sympathetic ganglia and a decrease in total calculated neuron number, estimated neuron size, and ganglion volume of the superior cervical ganglion at the 6-month and 12-month necropsies. There was a complete recovery of the effects in the 0.2 mg/kg/wk group during the 13-week treatment-free period following 28 weeks of treatment and in the 0.2 and 1 mg/kg/wk groups during the 24-week treatment-free period following 52 weeks of treatment (Table 8).
Chronic Study Superior Cervical Ganglia Stereology Results.a
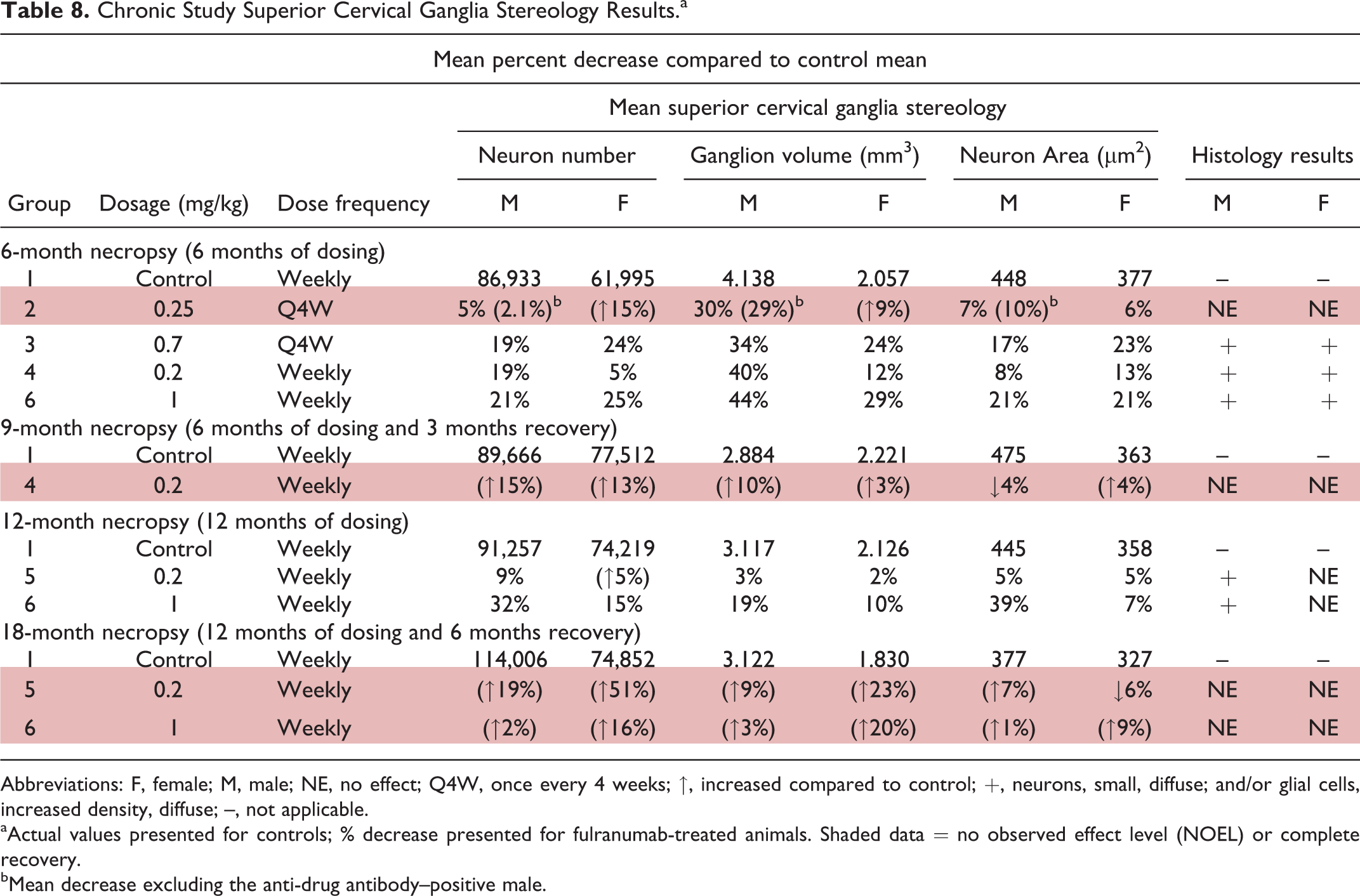
Abbreviations: F, female; M, male; NE, no effect; Q4W, once every 4 weeks; ↑, increased compared to control; +, neurons, small, diffuse; and/or glial cells, increased density, diffuse; –, not applicable.
aActual values presented for controls; % decrease presented for fulranumab-treated animals. Shaded data = no observed effect level (NOEL) or complete recovery.
bMean decrease excluding the anti-drug antibody–positive male.
Discussion
The anatomical and functional effects of NGF inhibition were evaluated in a series of studies in adult cynomolgus monkeys. Fulranumab treatment had no effect on in-life evaluations or standard histopathology as is typically performed for general toxicity studies. Comprehensive evaluations of the central and peripheral (including the autonomic) portions of the nervous systems were performed. These evaluations included detailed morphologic evaluations of the brain, spinal cord, peripheral nerves, cervicothoracic ganglia, sympathetic trunk ganglia, superior mesenteric ganglia, superior cervical ganglia, and thoracic dorsal root ganglia utilizing standard (paraffin/H&E) and special staining/embedding staining techniques. Morphologic evaluations were augmented by stereology of the superior cervical ganglia and dorsal root ganglia. These evaluations revealed that fulranumab treatment caused decreases in neuron size and increases in glial cell density as observed by morphologic examinations of sympathetic ganglia and dose-related decreased neuron size, neuron count, and ganglion volume in the superior cervical ganglia by stereology, but did not cause neuronal death, necrosis, or apoptosis. Observed decreases in estimated neuron counts and the appearance of increased density of glial cells were interpreted to be due to atrophy of the neurons, which resulted in the glial cells surrounding the neuronal cell body (satellite glial cells) and the glial cells surrounding neuronal processes (Schwann cells) being closer together in the ganglion. Morphologic evidence supporting this interpretation included lack of evidence of glial cell proliferation (ie, altered mitotic rate on H&E stained sections), return of neuron counts to control levels during the recovery period (indicating no prior actual loss of neurons), return of neuron size (resolution of the atrophy) to control levels during the recovery period, and a return of glial cell density to control levels associated with the return to normal neuron size.
The functional effect of fulranumab treatment on SNS function was assessed in a prior cardiovascular safety pharmacology study in telemetered cynomolgus monkeys and by evaluations of clinical observations, physical examinations, ECG, blood pressure, rectal temperature, and respiration rate in repeat-dose monkey studies. Fulranumab treatment had no effect on these endpoints. In addition, ocular neurological observations, including pupil reactivity (using a pen light), eye movement (conjugate eye movement, strabismus, nystagmus), and dilation abnormalities (miosis and mydriasis), were performed in the acute and chronic toxicity studies. In humans, cervicothoracic SNS block results in collection of signs and symptoms including ipsilateral pupil constriction, ptosis, and enophthalmos known as Horner's syndrome. Sympathetic nervous system block can occur for a variety of reasons including injection of anesthesia into the cervical plexus for pain relief, from carotid artery surgery, neck tumors, and accidental or congenital severing of the cervical sympathetic trunk. 12 -14 Differential diagnosis includes lack of pupillary reactivity to changes in light and lack of pupil dilation with cocaine eye drops. Thus, ocular neurologic examinations are a sensitive and clinically translatable end point for evaluation on SNS effects. Treatment with fulranumab had no effect on ocular neurological parameters.
The anatomical methods and results presented here for fulranumab are similar to those reported for tanezumab. 9 For both NGF inhibitors, the main finding in adult cynomolgus monkeys was a reversible diffuse decrease in neuron size in the sympathetic ganglia accompanied by the appearance of increased glial cell density. Whether these anatomical changes correlated with functional effects was also evaluated. Compensatory responses to postural changes and pupillary responses are both mediated by cervical sympathetic ganglion. An assessment of orthostatic hypotension using a tilt chair was performed for tanezumab 9 and ocular neurological observations were performed for fulranumab. No functional effects were observed for either NGF inhibitor.
Conclusion
There were no effects, including morphological effects, on the central and peripheral nervous systems of fulranumab treatment (0.25 mg/kg SC Q4W for 28 weeks; NOEL) in adult cynomolgus monkeys at a 5-fold higher exposure (AUC at steady state) than the exposure at the 3 mg Q4W dosage in clinical studies. At other doses, up to the highest doses tested (50 mg/kg/wk for 15 weeks or 1 mg/kg/wk for 52 weeks), the only observed effects were reversible decreases in neuron size and ganglia volume in sympathetic ganglia with no functional correlates.
Footnotes
Acknowledgments
The authors would like to acknowledge Satoru Oneda, SNBL, Everett, Washington, for his scientific contributions as study director of these studies and Eva Emmell and Linda Kuseryk for their contributions to data presentation and preparation of this manuscript.
Author Contributions
M. Rocca contributed to acquisition, analysis, and interpretation and drafted the manuscript. C. Han contributed to acquisition and analysis and critically revised the manuscript. M. Butt contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. T. Coogan contributed to conception and design, contributed to acquisition and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were funded by Janssen R&D and Amgen.
