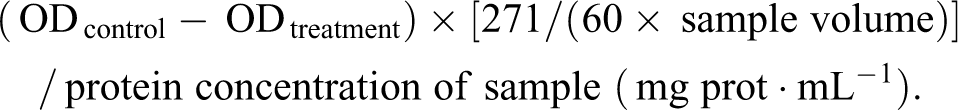Abstract
Metabolic activation of indirect-acting carcinogens in the target organ is an effective mechanism of carcinogenesis. Lipoxygenase (LOX) can co-oxidize the bladder carcinogen benzidine (BZ). However, it is not entirely clear whether BZ is activated and which enzyme is involved in its activation in bladder epithelial cells. Our results showed that BZ induced 5-LOX protein expression but had no significant influence on the expression of 15-LOX-2, CYP1B1, and CYP2E1 in SV-40 immortalized human uroepithelial SV-HUC-1 cells. BZ induced oxidative stress in SV-HUC-1 cells by increasing reactive oxygen species (ROS) and malondialdehyde levels significantly in the 100 and 200 μmol/L-BZ-treated groups and decreased the level of the antioxidant reduced glutathione significantly at 200 μmol/L BZ. Concurrently, the activity of catalase was increased, while the activity of superoxide dismutase was increased at 50 μmol/L BZ but gradually decreased with increasing concentrations of BZ (P < 0.05). However, the oxidative stress and damage in SV-HUC-1 cells caused by BZ were effectively inhibited by the 5-LOX-specific inhibitor AA861 at 10 μmol/L. Thus, 5-LOX is probably the major LOX isozyme to co-oxidize exogenous chemicals in SV-HUC-1 cells. AA861 has a protective effect on the oxidative stress and damage induced by BZ in SV-HUC-1 cells. We conclude that BZ can be activated by 5-LOX to produce ROS and oxidative stress, which may be associated with bladder cancer caused by BZ.
Introduction
Benzidine (BZ) is a known Group I human carcinogen classified by the International Agency for Research on Cancer 1 and is associated with bladder cancer and other cancers, such as skin, breast, liver, and lung cancers. 2 Although the United States, Japan, and many other countries have banned the manufacture of BZ, azo dyes based on BZ and BZ congeners are still used in the market. 3 Additionally, there is still BZ contamination in textiles and in the environment. 4
Oxidative stress is a stressed state of the body in response to environmental stimuli, reflects an imbalance between the oxidative and antioxidative systems, and mainly presents as excessive production of reactive oxygen species (ROS) and/or the depletion of antioxidant enzymes and antioxidants. As one of the toxic mechanisms of xenobiotics, oxidative stress is closely associated with chemical carcinogenesis 5 and participates in the occurrence and development of bladder cancer. 6 BZ-induced carcinogenic and mutagenic effects may be also linked with the induction of ROS, resulting in oxidative DNA damage. 7,8 Therefore, ROS production, which also causes lipid peroxidation of the cells and biomolecule damage, may be a contributing factor to the occurrence of BZ-induced tumors.
BZ is an indirect-acting carcinogen; its metabolic activation pathway may involve N-oxidation catalyzed by cytochrome P450 1A2 (CYP 1A2) 9 or prostaglandin H synthase (PHS) 10 and subsequent acetylation of the N-hydroxyl metabolite to generate the ultimate carcinogen. The active cationic radical intermediates produced in this process can co-oxidize reduced glutathione (GSH), reduced form of nicotinamide adenine dinucleotide (NADH), or reduced nicotinamide adenine dinucleotide phosphate (NADPH) and form ROS. 8,11 The cationic free radicals and ROS can oxidize DNA causing DNA damage, which may be a causative factor for BZ-induced carcinogenic and mutagenic toxicity. 8
The metabolic activation of BZ is not predominantly catalyzed by cytochrome P450 (CYP450) since BZ is a very poor substrate for NADPH-dependent oxidation by rat hepatic CYP450, and CYP450 expression is very low in some extrahepatic tissues. Additionally, BZ is not predominantly catalyzed by prostaglandin synthase, as this enzyme occurs in negligible amounts in the liver. 12 Lipoxygenases (LOXs) are a group of multifunctional non-heme iron proteins that show relatively high activity in some extrahepatic tissues with little or no CYP450, such as in the brain, lung, heart, skin, uterus, placenta, and fetal tissues. Besides dioxygenation of polyunsaturated fatty acid (PUFA), LOXs demonstrate co-oxidation activity toward many xenobiotics, including drugs, pesticides, and carcinogens, in the presence of PUFA, fatty acid hydroperoxide, or hydrogen peroxide (H2O2). 13 Therefore, LOX is regarded as a likely alternate or additional pathway for oxidation of xenobiotics, especially in CYP450-deficient extrahepatic tissues.
In in vitro acellular enzyme systems, BZ can be oxidized by purified LOX from adult human liver, 14 lung, 15 human term placenta, 16 tissues at 10 weeks of gestation, 17 and soybean to produce BZ cation radicals and BZ diimine, 14 which cause oxidative damage or bind covalently to tissue macromolecules. 18,19 Our previous study has shown that 5-LOX in human bronchial epithelial cells can also co-oxidize BZ, leading to cytotoxicity and DNA damage. 20 Metabolic activation of indirect carcinogens in the target organ is an effective mechanism of carcinogenesis. 5-Lipoxygenase is slightly expressed in normal bladder tissues and markly expressed in bladder cancer tissues. 21 It is of great interest to investigate whether 5-LOX can catalyze the activation of BZ and induce oxidative stress and cytotoxicity in the bladder, the main target organ for the carcinogenicity of BZ.
The present study investigated the effects of 5-LOX on BZ-induced cytotoxicity and oxidative stress in human uroepithelial SV-HUC-1 cells. Furthermore, isozyme expression levels of LOX in SV-HUC-1 and BZ’s regulation on 5-LOX expression were evaluated. The results provide further evidence for 5-LOX-mediated activation and the oxidative damage caused by BZ in the bladder.
Materials and Methods
Materials
Human uroepithelial cell line SV-HUC-1 was purchased from China Center for Type Culture Collection (Shanghai, China). BZ, AA861, dimethyl sulfoxide (DMSO), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma-Aldrich Co (St Louis, Missouri). Dulbecco’s Modified Eagle Medium: Nutrient Mixture F-12 (DMEM: F12) and fetal bovine serum (FBS) were supplied by Hyclone (Logan, Utah). Polyvinylidene difluoride (PVDF) membrane was purchased from Milipore (Billerica, Massachusetts). Cell lysis buffer, bicinchoninic acid assay (BCA) protein assay kit, and Beyo ECL Plus Chemiluminescent kit were purchased from Beyotime Institute of Biotechnology (Haimen, China). Anti-human 5-LOX, 12-LOX, and CYP2E1-rabbit monoclonal antibodies and anti-human CYP1B1-rabbit polyclonal antibody were obtained from Epitomics, Inc. (Burlingame, California). Anti-human 15-LOX-2 rabbit monoclonal antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, California). Anti-β-actin mouse monoclonal antibody and horseradish peroxidase-conjugated goat anti-rabbit and goat anti-mouse immunoglobulin G (IgG) secondary antibodies were obtained from Beyotime Institute of Biotechnology.
Cell Culture
SV-HUC-1 cells were cultured in DMEM:F12 supplemented with 10% FBS, 1% penicillin (100 IU/mL), and streptomycin (100 μg/mL) in a cell incubator (Sheldon Manufacturing Inc., Cornelius, Oregon) at 37°C with 5% CO2.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide Cell Proliferation Assay
SV-HUC-1 cells were seeded into 96-well plates at 3×103 cells/well (200 μL/well), cultured for 24 hours and treated with increasing concentrations of BZ (0, 25, 50, 100, 200, 400, and 800 μmol/L) for 24 hours. In the inhibitor test, prior to their exposure to 200 μmol/L BZ, SV-HUC-1 were preincubated with AA861 (0.1, 0.3, 1.0, 3.0, and 10.0 μmol/L) for 4 hours. An equal volume of DMSO was used as a control. Then, 20 μL of MTT (5 g/L) was added to each well, and plates were incubated at 37°C for another 4 hours. The medium was then discarded, and 150 μL of DMSO was added to each well. After shaking for 10 minutes, absorbance at 570 nm was measured (PowerWave XS2 Microplate Spectrophotometer; BioTek, Vermont).
Western Blot Assay
After treatment as described earlier, total proteins were extracted using RIPA lysis buffer and the concentrations were measured using the BCA kit (Beyotime Institute of Biotechnology). Protein samples (40-60 μg/lane) were separated by sodium dodecyl sulfate-polyacrylamide gel (10%) electrophoresis at 120 V and transferred to PVDF membranes at 260 mA for 90 minutes. After blocking for 2 hours, membranes were incubated overnight at 4°C with different primary antibodies: anti-human 5-LOX, 12-LOX, CYP2E1-rabbit monoclonal antibody and CYP1B1-rabbit polyclonal antibody (1:1,000 dilution; Epitomics); anti-human 15-LOX-2 rabbit monoclonal antibody (1:100 dilution; Santa Cruz Biotechnology); and anti-β-action mouse monoclonal antibody (1:2,000 dilution; Beyotime Institute of Biotechnology). The immunoblots were then incubated with horseradish peroxidase-conjugated goat anti-rabbit and goat anti-mouse IgG secondary antibodies (1:5,000; Beyotime Institute of Biotechnology) for 1 hour followed by ECL detection.
Measurement of Oxidative Stress
Measurement of intracellular ROS
The levels of intracellular ROS were determined using an ROS assay kit (Beyotime Institute of Biotechnology) following the manufacturer’s protocol. The SV-HUC-1 cells were seeded into 6-well plates and treated as described in MTT cell proliferation assay. Then, cells were harvested and washed twice with phosphate buffer saline (PBS) and incubated with DCFH-DA (10 μmol/L) at 37°C for 30 minutes in a darkroom for final analysis by RF-5301PC fluorescence spectrophotometer (Shimadzu Corporation, Kyoto, Japan) under an excitation wavelength of 488 nm and an emission wavelength of 525 nm. All determinations were performed in triplicate.
Assay of antioxidant enzyme (catalase and superoxide dismutase) activity
SV-HUC-1 cells were harvested and lysed with 0.3-0.5 mL PBS buffer per 106 cells by sonication for determination of the activity of catalase (CAT) and total superoxide dismutase (T-SOD) using commercial assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Activities of CAT and T-SOD were expressed as units per milligrams of protein (U/mg prot).
Catalase activity was calculated by the decrease of the yellow molybdenum acid-H2O2 complex in absorbance at 405 nm due to the hydrolysis reaction of H2O2 with CAT. One unit is defined as the amount of enzyme that catalyzes the decomposition of 1 µmol H2O2 per second at 37°C. Catalase activity (U/mg prot) was calculated according to formula in the manufacturer’s instructions as follows:
The activity of T-SOD was determined by measuring the inhibiting rate of the enzyme in response to superoxide anion (O2−) produced by xanthine and xanthine oxidase. Superoxide anion oxidized hydroxylamine to nitrite, which could be detected by absorbance at 550 nm after reacting with a reagent to form red substances. One unit of SOD activity was defined as the quantity of enzyme required to produce 50% inhibition of nitrite per 1 mL of reaction solution at 37°C. The T-SOD activity (U/mg prot) was calculated according to the manufacturer’s instructions as follows:
Measurement of nonenzymatic antioxidants (reduced GSH)
Cells were treated, harvested, and lyzed as mentioned earlier. The level of GSH was detected by absorbance at 420 nm due to the reaction of GSH with 5,5’-dithiobis-(2-nitrobenzoic acid) (DTNB) and calculated by extrapolating the data from the GSH standard as follows:
Analysis of lipid peroxidation by assay of malondialdehyde
Cells (106) were suspended in 0.5 mL of extracted solution containing 50 mM phosphate buffer (pH 7.4) and 1% polyvinylpyrrolidone and homogenized by sonication. Then, the homogenate was further processed following the instructions of the malondialdehyde (MDA) assay kit (Nanjing Jiancheng Bioengineering Institute, Nanjing). The amount of MDA was measured by the absorbance values of red TBA-MDA complex at 532 nm and calculated according to the instructions of MDA assay kit.
Statistical Analysis
The results were expressed as mean (standard deviation) and analyzed for the statistical significance using the SPSS software (version 18.0). Multisample means comparison was performed with a single factor analysis of variance, and the multiple comparisons were determined using Dunnett t-test. P < 0.05 was considered statistically significant.
Results
Effect of BZ and AA861 on the Proliferation of SV-HUC-1 Cells
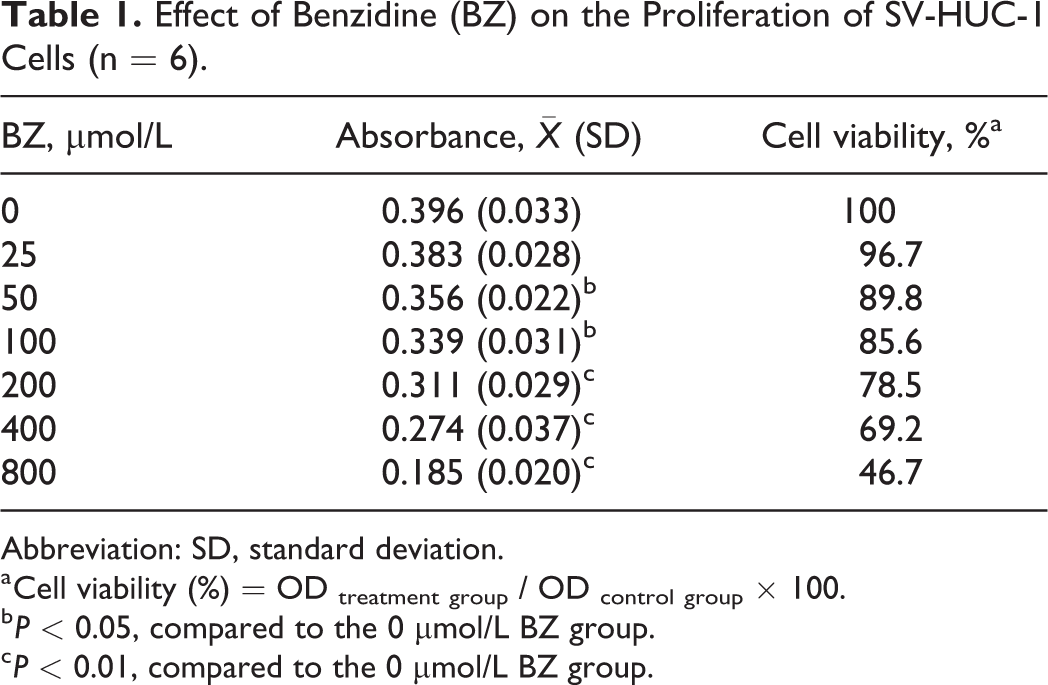
SV-HUC-1 cells were treated with different concentrations (0, 25, 50, 100, 200, 400, and 800 μmol/L) of BZ for 24 hours. Cell viability, analyzed with an MTT assay, showed a general decline with increasing concentrations of BZ (Table 1). This result suggested that BZ inhibited the proliferation of SV-HUC-1 cells in a dose-dependent manner (r = −0.949, P < 0.05). Differences in absorbance were statistically significant in samples treated with 50 μmol/L BZ or above compared to control samples.
Effect of Benzidine (BZ) on the Proliferation of SV-HUC-1 Cells (n = 6).
Abbreviation: SD, standard deviation.
a Cell viability (%) = OD treatment group / OD control group × 100.
b P < 0.05, compared to the 0 μmol/L BZ group.
c P < 0.01, compared to the 0 μmol/L BZ group.
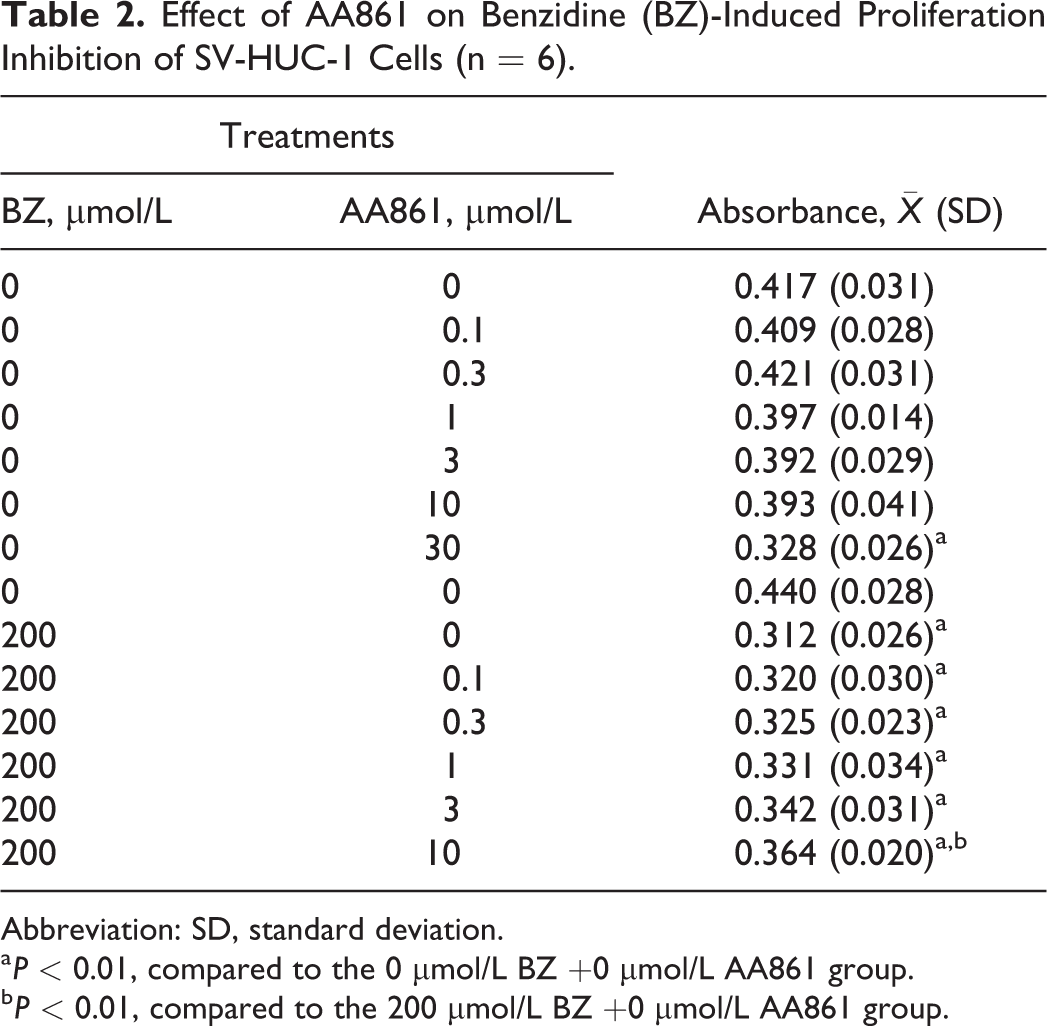
5-LOX-specific inhibitor AA861 had no significant influence on cell viability of SV-HUC-1 when the concentration was under 10 μmol/L. Pretreatment of the cells with the AA861 facilitated the survival of 200 μmol/L BZ-treated SV-HUC-1 in a dose-dependent manner and differences were statistically significant when the concentration of AA861 reached 10 μmol/L (Table 2).
Effect of AA861 on Benzidine (BZ)-Induced Proliferation Inhibition of SV-HUC-1 Cells (n = 6).
Abbreviation: SD, standard deviation.
a P < 0.01, compared to the 0 μmol/L BZ +0 μmol/L AA861 group.
b P < 0.01, compared to the 200 μmol/L BZ +0 μmol/L AA861 group.
BZ Modulates LOX, CYP1B1, and CYP2E1 Protein Expression in SV-HUC-1 Cells
The protein samples extracted from human tracheobronchial epithelial (HBE) and A431 cells were used as positive control of 5-LOX and 12-LOX protein expression, respectively. The protein expressions of 5-LOX (ca. 78 kDa) and 15-LOX-2 (ca. 75 kDa) were detected, but no 12-LOX (ca. 72 kDa) protein expression was detected in SV-HUC-1. With increasing BZ concentration (from 100 to 200 μmol/L), relative expression of 5-LOX increased 30.11% and 49.12% compared to the solvent control group (P < 0.05). There was no significant change in 15-LOX-2 expression between BZ treatment groups and control group (Figure 1).
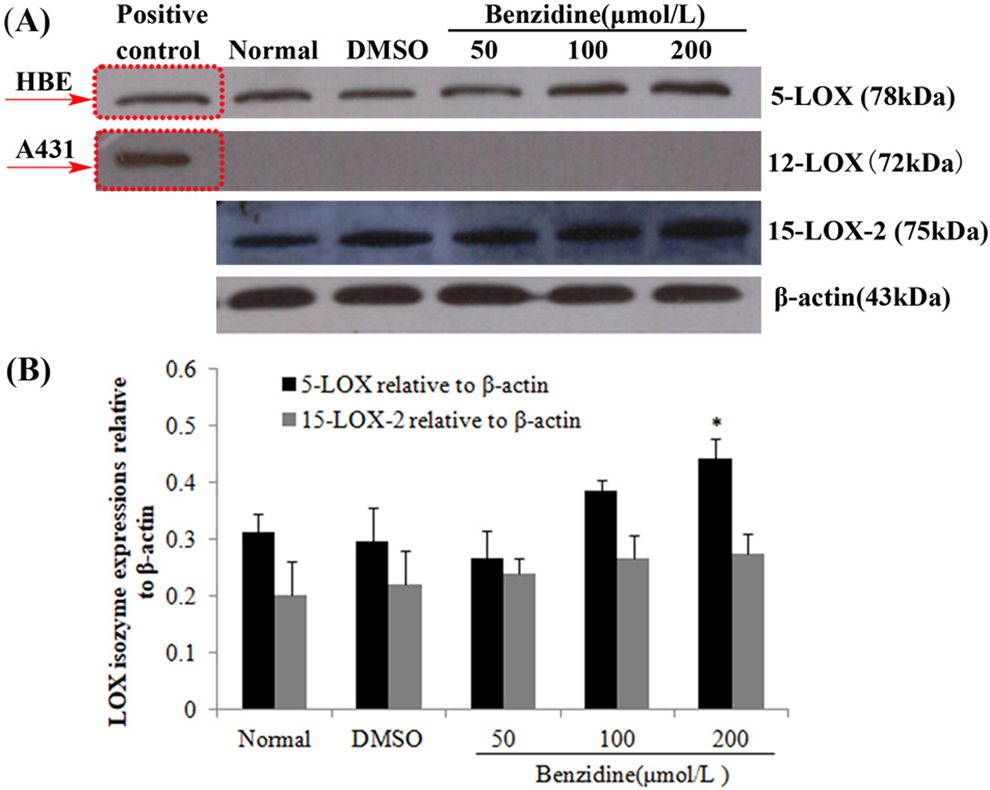
Effect of benzidine (BZ) on lipoxygenase (LOX) isoenzyme protein expression in SV-HUC-1. A, Pattern of LOX isoenzyme protein expression. B, Relative protein expression ratios of LOX isoenzyme corrected for β-actin (n = 3). Protein concentrations for 5-LOX, 12-LOX, and 15-LOX-2 protein assays were 40, 60, and 60 μg, respectively. * Significantly different compared to solvent control (P < 0.05).
Keeping experimental conditions consistent including the same primary antibody source and dilution ratios, the protein expression levels of 5-LOX, CYP2E1, and CYP1B1 were compared. Relative to the significant increases in 5-LOX protein expression, no statistically significant increases were observed in CYP2E1 and CYP1B1 protein expression mediated by BZ (Figure 2).
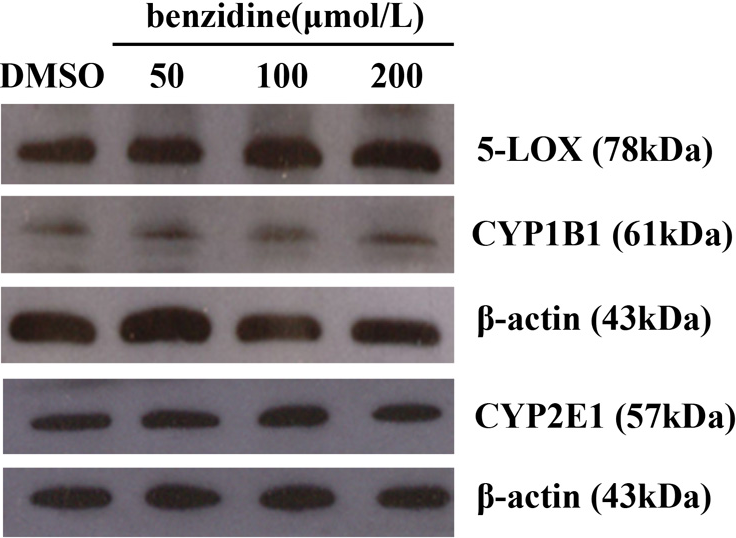
Protein expression of CYP2E1, CYP1B1, and 5-LOX in benzidine-treated SV-HUC-1 cells.
5-LOX-Specific Inhibitor AA861 Inhibits ROS Generation Induced by BZ
Average ROS fluorescence optical densities of SV-HUC-1 are shown in Figure 3A. There was no significant difference between 10 μmol/L AA861 treatment and the solvent control. Reactive oxygen species levels increased with increasing BZ concentration and statistically significant differences were observed at doses equal to or larger than 100 μmol/L BZ. Reactive oxygen species were significantly attenuated in a dose-dependent manner by 5-LOX-specific inhibitor AA861 pretreatment.
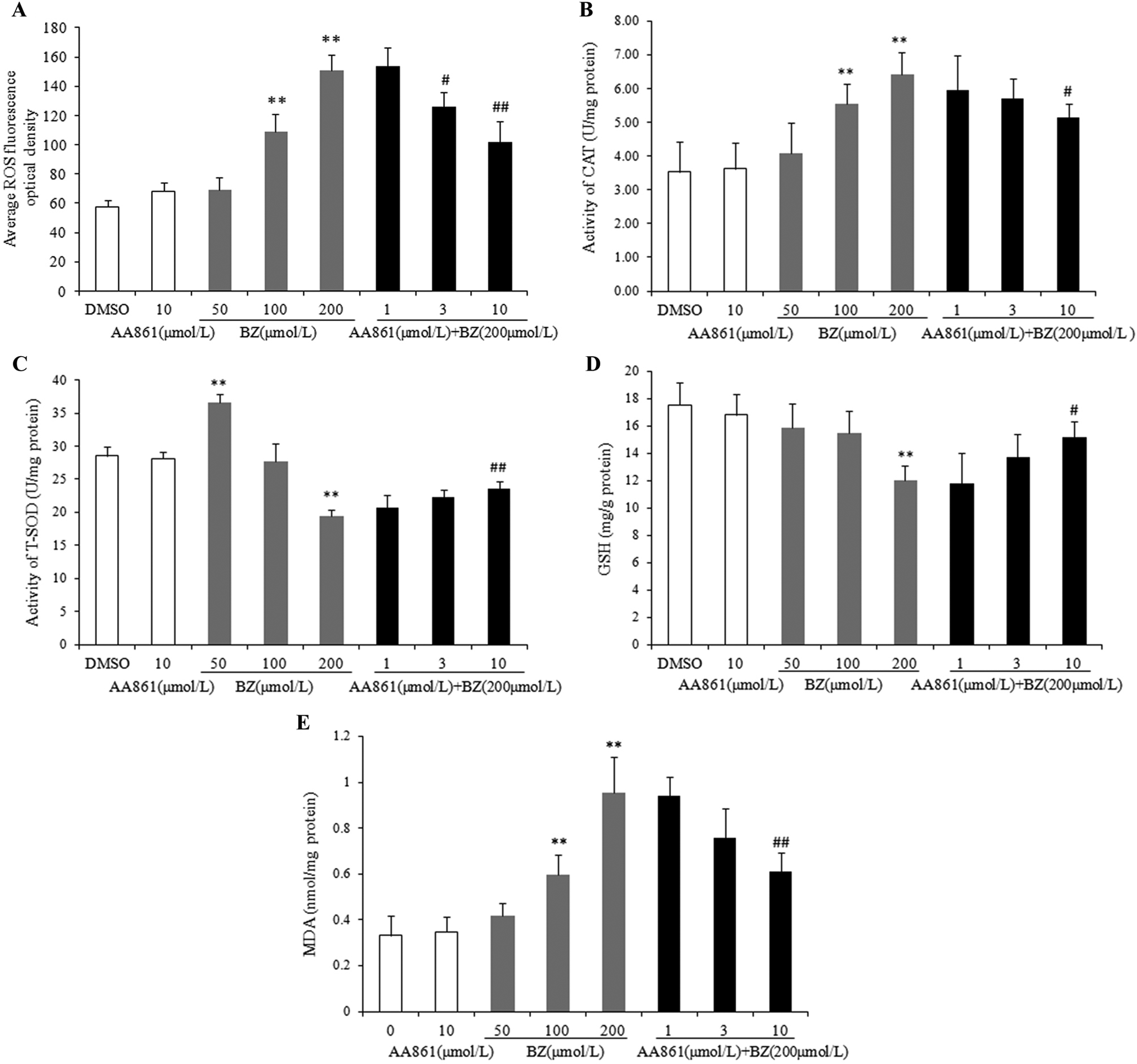
Effect of benzidine (BZ) and AA861 on reactive oxygen species (ROS) generation, antioxidative activity (CAT, T-SOD, GSH), and malondialdehyde (MDA) level in SV-HUC-1 cells. A, ROS fluorescence optical density; B, activity of CAT; C, activity of T-SOD; D, GSH level; E, MDA level. Data show the mean (SD; n = 3). *P < 0.05, **P < 0.01, compared to solvent control; #P < 0.05, ##P < 0.01, compared to 200 μmol/L BZ treatment group. CAT indicates catalase; GSH, glutathione; SD, standard deviation; T-SOD, total superoxide dismutase.
Effect of AA861 on Activity Changes of CAT and SOD Induced by BZ
At 10 μmol/L AA861, there was no effect on CAT and SOD activities in SV-HUC-1 cells (Figure 3B and 3C). However, CAT activity increased with increasing BZ concentration, and the difference was statistically significant when BZ reached 100 μmol/L (Figure 3B). Intracellular SOD activity was significantly increased in a low concentration of BZ (50 μmol/L BZ) but significantly decreased in a high concentration of BZ (200 μmol/L) compared to the solvent control (Figure 3C). Pretreatment with AA861 reversed the BZ-induced change in both CAT and SOD activities (Figure 3B and 3C). The difference in activity, compared to 200 μmol/L BZ alone, was statistically significant when AA861 reaches 10 μmol/L.
AA861 Suppressed BZ-Induced Decrease of Intracellular GSH Content
BZ reduced GSH content in a dose-dependent manner compared with the solvent control group. AA861 pretreatment could attenuate the decrease of GSH content induced by BZ. When AA861 reached 10 μmol/L, there was significant difference compared to 200 μmol/L BZ treatment group (Figure 3D).
AA861 Inhibited MDA Generation Induced by BZ
Malondialdehyde is a prominent product of lipid peroxidation of PUFA that is used as a biomarker to measure the level of oxidative stress. As shown in Figure 3E, 10 μmol/L AA861 alone had no significant effect on MDA level. BZ markedly increased MDA generation in SV-HUC-1 cells. AA861 pretreatment inhibited MDA generation induced by 200 μmol/L BZ in a dose-dependent manner with a statistically significant difference at 10 μmol/L AA861 pretreatment group.
Discussion
In this study, we used the 5-LOX-specific inhibitor AA861 to investigate the effects of 5-LOX on BZ-induced oxidative stress in SV-HUC-1 cells, one of the important carcinogenic target cells for BZ. Our data showed that in SV-HUC-1, 5-LOX and 15-LOX-2 proteins were expressed. BZ exposure upregulated 5-LOX expression; inhibited the proliferation of SV-HUC-1; significantly increased ROS, CAT, and MDA levels; and reduced SOD and GSH levels. Furthermore, the inhibition of 5-LOX by AA861 pretreatment reversed BZ-induced anti-proliferation and oxidative stress.
BZ has been found in the urine of workers who handled BZ-based dyes. Dewan et al reported urinary levels of BZ in 20 workers of direct black 38 production were 0-362.5μg/L. The National Institute for Occupational Safety and Health calculated that inhalation of a BZ-derived dye of 2 mg/m3 over an 8-hour workday can result in 28-48 μg/L BZ in the urine. 22 In order to observe the effects of BZ through short-term single exposure, the concentrations of BZ used in our experiments are higher than the urinary levels of BZ reported in humans exposed to BZ-derived dye and are determined according to the results of MTT assay in this study.
The LOX pathway, which metabolizes arachidonic acid (AA), is involved in various physiological and pathological processes, such as inflammation and cancer. 23 The major isozymes of LOX in animals and man comprise 5-LOX, 12-LOX, and 15-LOX, which are designated according to the carbon number where oxygen is inserted into PUFA catalyzed by LOX. In this study, 5-LOX and 15-LOX-2 were detected in SV-HUC-1, which indicated that these 2 isozymes might be responsible for the physiological and pathophysiological roles of LOX in SV-HUC-1. These results are identical to those reported in the literature that indicates the expression of 5-LOX, 12-LOX, and 15-LOX in urothelial via the detection of AA metabolites from various LOX isozymes. 24 However, in our study, 12-LOX was not detected in experimental cells perhaps because 12-LOX is distributed predominantly in blood platelets and leukocytes and has no or lower expression levels in other normal cells, 25 or the selected experimental conditions were not optimal. Previous researchers have generally considered 5-LOX and 12-LOX to be pro-carcinogenic, but 15-LOX-2 to have a tumor-suppressive function. 26 In this study, a significant increase of 5-LOX expression and a slight nonsignificant increase of 15-LOX-2 expression in BZ-treated SV-HUC-1 cells indicated that 5-LOX may be related to the bladder carcinogenesis of BZ.
The importance of 5-LOX in the initiation and development of malignant tumors has been confirmed by the fact that 5-LOX levels increase markedly in many human malignant tumors, including bladder cancer, while inhibiting 5-LOX may prevent or reverse the formation and development of malignant tumors. 27 7,12-Dimethylbenz[a]anthracene (DMBA) upregulates 5-LOX expression, while a 5-LOX-specific inhibitor prevents DMBA-induced breast 28 and oral 29 cancer in rat and hamster, respectively, indicating that 5-LOX also plays an important role in chemically induced carcinogenesis, but its mechanism is not fully understood. Our previous studies on the effects of 5-LOX on BZ toxicity found that purified LOX catalyzes BZ to its active metabolites in in vitro noncell systems, BZ exposure upregulates 5-LOX, and 5-LOX knockdown inhibits BZ-induced DNA damage and cell apoptosis in HBE cells. 20 We hypothesized that 5-LOX mediates BZ-induced cytotoxicity and carcinogenesis through the oxidative metabolism of PUFA to produce bioactive lipid mediators such as leukotrienes and co-oxidizes BZ to its active metabolites, which, as far as we know, has not yet been reported in the literature. In our current study, we used SV-HUC-1 cells to investigate whether the above hypotheses are true at the BZ carcinogenic target site. The results showed that the 5-LOX protein levels in SV-HUC-1 cells were significantly upregulated by BZ exposure. BZ had no significant influence on the protein expression of CYP2E1 and CYP1B1, other 2 xenobiotic-metabolizing enzymes associated with bladder cancer. 30,31 We also observed the protective effect of the 5-LOX inhibitor on BZ-induced proliferation inhibition in SV-HUC-1 cells, consistent with our previous results in HBE cells. 20 These data further demonstrate that 5-LOX is involved in BZ-induced cytotoxicity in bladder cells.
Oxidative stress, an imbalance between levels of ROS and the ability of the body to remove it, is thought to play a vital role in the pathophysiological mechanisms of human diseases, including cancer, cardiovascular disease, diabetes, rheumatoid arthritis, Alzheimer disease, and Parkinson disease. 32 Reactive oxygen species, including •O2 −, H2O2, hydroxyl radical, singlet oxygen, and ozone, is the byproduct of normal cellular aerobic metabolism. A low concentration of ROS is necessary for certain subcellular events such as cell signaling, enzyme activation, gene expression, and regulation of apoptosis. 32 In the body, there are also antioxidant enzymes, such as SOD, glutathione peroxidase (GPx), and CAT, and antioxidants, such as vitamin C, alpha tocopherol, GSH, and flavonoids, that remove excessive ROS and maintain the balance of the oxidation and anti-oxidation systems. The SOD catalyze the dismutation of •O2 − to H2O2. Subsequently, H2O2 is quickly reduced to H2O by 2 other enzymes, CAT and GPx. Glutathione is an important antioxidant. As a cofactor for the GPx, it can reduce ROS and itself become oxidized states. 33 Some environmental stressors, such as metal toxicity, ultraviolet radiation, and ionizing radiation, can increase ROS production dramatically. When the level of oxidation exceeds antioxidant defenses, oxidative stress occurs, which may result in significant damage to cells, including oxidative damage to DNA, oxidative modification of proteins, and lipid peroxidation, 34 of which MDA is a prominent product. Thus, ROS, SOD, CAT, GSH, and MDA were used in our study as markers for oxidative stress to investigate whether 5-LOX mediates BZ-induced cytotoxicity in bladder cells via oxidative stress.
Our results demonstrated that BZ treatment significantly increased ROS level, leading to the consumption of GSH and ultimately resulting in lipid peroxidation, indicated by the overproduction of MDA in SV-HUC-1 cells, which is thought to contribute to the genotoxicity of BZ. 8 Accompanied by an increase in ROS, the activity of antioxidant enzymes SOD and CAT increased to scavenge ROS and may be an early protective response of cells to oxidative stress damage. 35 However, when the concentration of BZ was above 100 μmol/L, the activity of SOD was reduced. The enhancement of SOD activity at low concentrations of BZ and inhibition at high concentrations is consistent with previous reports. 36 Superoxide dismutase activity inhibition at high concentrations of BZ may be due to the fact that high BZ-induced ROS production overwhelms the cell’s antioxidant ability, potentially triggering ROS-induced ROS release, 37 followed by ROS accumulation-induced structural and functional damage of membrane and mitochondria, ultimately resulting in SOD activity inhibition. 38 These data suggest that BZ induces oxidative stress through the overproduction of ROS and the inhibition of SOD activity.
The present study also demonstrated that 5-LOX contributes to BZ-induced oxidative stress and cytotoxicity since the 5-LOX-specific inhibitor AA861 inhibited BZ-induced overproduction of ROS and MDA and decreased GSH levels, SOD activity, and cell viability. It has been reported that reactive free radical intermediates and ROS formation in the bioactivation of BZ is catalyzed by various endogenous enzyme systems such as CYP450 and PHS. 8 Our previous studies have showed that LOX can also mediate the co-oxidation of BZ to produce reactive free radical intermediates. 18 Therefore, 5-LOX promotes BZ-induced ROS generation possibly through catalyzing the oxidation and activation of BZ and then triggering oxidative stress and lipid peroxidation, which may be one of the mechanisms for 5-LOX-mediated cytotoxicity of BZ in SV-HUC-1, and associated with bladder cancer caused by BZ.
Conclusions
This study has further demonstrated that in human uroepithelial cells (SV-HUC-1), one of the important carcinogenic target cells for BZ, 5-LOX was involved in BZ-induced oxidative stress and cytotoxicity, which was one of the mechanisms of BZ-induced mutation and carcinogenicity. Our results provide further evidence that 5-LOX can catalyze the bioactivation of BZ to form reactive free radical intermediates and ROS, and this may be one of the pro-carcinogenic mechanisms for 5-LOX to the bladder carcinogenesis of BZ.
Footnotes
Authors’ Note
Yun Huang and Shaoling Huang equally contributed to this work.
Author Contributions
Shaoling, H contributed to acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Yun, H contributed to conception and design, analysis and interpretation, drafted manuscript, and critically revised manuscript. Yue, W contributed to analysis and interpretation and critically revised manuscript. Minlan, P, Xinge, Z, and Jing, W contributed to acquisition and critically revised manuscript. Jianan, H. contributed to conception and design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China under grant numbers 81072331.