Abstract
Lipoxygenase (LOX)-catalyzed cooxidation of the human carcinogen benzidine (BZD) has been shown in in vitro enzyme systems. This study aimed to determine whether BZD could be activated by arachidonate 5-lipoxygenase (
Keywords
Introduction
Benzidine (BZD) is known to cause various tumors in humans and animals. 1 –3 Benzidine and many of its homologous compounds are indirect carcinogens and only become carcinogenic when they are metabolized to their active form. This does not predominantly involve cytochrome P450 (CYP450) since BZD is a very poor substrate for the reduced nicotinamide adenine dinucleotide phosphate (NADPH)-dependent oxidation by rat hepatic CYP450 or prostaglandin synthase which is present in negligible amounts in the liver. 4
Lipoxygenases (LOXs) can cooxidize many xenobiotics in the presence of polyunsaturated fatty acids, fatty acid hydroperoxides, and hydrogen peroxide. 4,5 Additionally, LOX is highly active in some CYP450-deficient extrahepatic tissues such as brain, lung, heart, skin, uterus, placenta, and fetal tissues. Therefore, LOX represents a likely alternate or additional pathway for the oxidation of xenobiotics, particularly in these tissues. 4
Liver LOX can mediate the activation of BZD, producing BZD cation radicals and BZD diimine, 5 which can bind covalently to tissue macromolecules. 6,7 Soybean lipoxygenase (SLO) 8 and purified LOX from human term placenta, 9 tissues at 10 weeks of gestation, 10 and adult human lung 11 can also activate BZD. Therefore, the LOX pathway may constitute the predominant extrahepatic mechanism of BZD activation.
In mice, it has been shown that the LOX inhibitors strongly inhibit the carcinogenesis induced by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone,
12
7,12-dimethylbenz(a)anthracene (DMBA),
13
and 12-
The 5-LOX isoenzyme is predominant in various mammalian tissues and blood cells. 4 The expression of 5-LOX increases markedly in many human malignant tumors, such as carcinomas of urinary bladder, colon, and lung, while inhibition of its expression may prevent or reverse the formation and development of malignant tumors, 17 indicating the importance of 5-LOX in the initiation and development of malignant tumors. In rats treated with DMBA, 5-LOX expression increased, while a 5-LOX-specific inhibitor could prevent DMBA-induced breast cancer, 18 indicating that 5-LOX also plays an important role in chemically induced carcinogenesis.
Earlier, we have shown the use of 5-LOX inhibitor, AA861, to investigate the effects of 5-LOX on BZD oxidation and activation. 19 However, chemical inhibitors demonstrate disadvantages, such as suboptimal selectivity, low efficiency, short-lived action, and possible complex interactions with the enzymatic substrates. 20 In contrast, RNA interference (RNAi) is an effective strategy for studying gene functions, 21 as it offers specificity and efficient inhibition for a longer duration. 22 We consider that the RNAi studies could facilitate elucidation of LOX-specific actions.
Thus, in the present study, we demonstrated the LOX-mediated cooxidation of BZD in vitro, relationship between 5-LOX expression and the activation and cytotoxicity of BZD in human tracheobronchial epithelium cells (HBECs), and the use of arachidonate 5-lipoxygenase (
Materials and Methods
Materials
Soybean lipoxygenase (activity: 150 000 Sigma units/mg of protein), BZD, nordihydroguaiaretic acid (NDGA), AA861, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), phenylmethylsulfonyl fluoride, mouse anti-β-actin monoclonal antibody, and propidium iodide (PI) were purchased from Sigma-Aldrich Co (St Louis, Missouri, USA). Human tracheobronchial epithelial cells were purchased from Shanghai Fuxiang Biotechnology Co, Ltd (Shanghai, China). Roswell Park Memorial Institute (RPMI) 1640 medium and fetal bovine serum (FBS) were supplied by Hyclone (Logan, Utah). Small hairpin RNA (ShRNA) oligonucleotides and polymerase chain reaction (PCR) primers were synthesized by Shanghai Invitrogen Biotechnology Co, Ltd. The DH5α competent cells were purchased from Takara Biotechnology Co, Ltd. (Ohtsu, Japan). Tiangen Plasmid Maxi kit was obtained from Tiangen Biotech Co., Ltd (Beijing, China). The 293T cells were supplied by American Type Culture Collection (Manassas, Virginia). The psPAX2 packaging plasmid and the pMD2.G membrane protein shell plasmid were obtained from Addgene (Cambridge, Massachusetts). Cell lysis buffer, bicinchoninic acid assay (BCA) protein assay kit, and Beyo ECL Plus Chemiluminescent kit were purchased from Beyotime Institute of Biotechnology (Haimen, China). Polyvinylidene difluoride (PVDF) membrane was purchased from Milipore (Billerica, Massachusetts). Anti-human 5-LOX polyclonal antibody was purchased from Abzoom Biolabs (Dallas, Texas). Horseradish peroxidase-conjugated goat antirabbit and goat antimouse immunoglobulin (Ig) G secondary antibodies were purchased from Jackson ImmunoResearch (Pennsylvania). Trizol reagent was purchased from Invitrogen (Grand Island, New York). Annexin V-fluorescein isothiocyanate (FITC)/PI Apoptosis Detection kit was obtained from KeyGEN Biotech (Nanjing, China).
In Vitro Enzyme System
Basic protocol
Benzidine and SLO were mixed in 0.1 mol L−1 acetate buffer (pH 3.5, 4.0, 4.5, 5.0, 5.5, and 6.0) and 50 mmol L−1 Tris-HCl buffer (pH 6.0, 6.5, and 7.0). After a 2-minute preincubation at 25°C, the reaction was initiated by the addition of hydrogen peroxide (Sigma Aldrich; 1 mL final volume). Temporal changes in absorbance by the oxidative product of BZD were recorded at 425 nm using an UV/VIS7501 spectrophotometer, and enzyme activity (nmol·min−1·mg−1 SLO) was calculated using the molar absorptivity of BZD dimiine (6.5 × 104 L·mol−1·cm−1). 7,8 υmax and Km were calculated from the Lineweaver-Burk plots.
Inhibitory test
Before adding BZD and hydrogen peroxide, SLO was preincubated with the specified concentration of the LOX inhibitor, NDGA, at 25°C for 3 minutes. The enzyme activity, inhibitor ratio, and the 50% inhibitory concentration (IC50) were then calculated.
Cell Culture Experiments
Cell Culture
Human tracheobronchial epithelial cells were grown in RPMI 1640 medium supplemented with 10% FBS.
Lentivirus shRNA vector construction and transfection
Small hairpin RNA against human
Positive clones were sequence verified, and the extracted plasmids were transfected into the human embryonic kidney 293T cells, together with the psPAX2 packaging plasmid and the pMD2.G membrane-protein shell plasmid for 18 hours. Cells were cultured for another 48 hours in a complete medium, and then the cellular supernatant was collected and the lentivirus was concentrated by ultracentrifugation (100 000
Based on a trial infection test, lentivirus-5-LOX-shRNA (LV-5-LOX-shRNA), lentivirus-NC-shRNA (LV-NC), and polybrene (final concentration: 5 μg/mL) were added to HBE target cells per the infection coefficient (multiplicity of infection = 20). After culturing overnight, the medium was removed and fresh medium was used to perform the transfection for 96 hours. Then, expression of the green fluorescent protein was detected under a BA400EF-UPR fluorescence microscope (Motic, Wuhan, China) with an excitation wavelength of 488 nm and an emission wavelength of 507 nm.
After transduction with lentiviral particles, cells were divided into 3 groups: HBECs (uninfected), HBE-NC cells (transfected with negative control virus), and HBE-5-LOX-shRNA (transfected with 5-LOX-shRNA lentiviral vectors). Interference efficiency was determined using the real-time fluorescence quantitative PCRs (QPCR) and Western blotting.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell viability assay
The HBECs, HBE-NC, and HBE-5-LOX-shRNA cells were seeded into 96-well plates at a density of 1 × 105 cells/mL (100 µL/well) and cultured for 24 hours. Treatment groups were exposed to BZD (0, 125, 250, 500, and 1000 μmol/L) or an equal volume of dimethyl sulfoxide (DMSO; control). In the inhibitor test, prior to their exposure to 500 μmol/L BZD, HBECs were preincubated with AA861 (0.3, 3.0, and 30.0 μmol/L). Then, 10 μL of MTT (5 g/L) was added to each well, and plates were incubated for another 4 hours. The medium was then removed, and 150 µL of DMSO was added. After shaking for 10 minutes, absorbance at 492 nm was measured (PowerWave XS2 Microplate Spectrophotometer; BioTek, Vermont).
Western blotting of 5-LOX
After treatment as described earlier, cells were lysed in cell lysis buffer containing phenylmethylsulfonyl fluoride, and the protein amount was determined by the BCA assay.
After heat denaturation for 10 minutes, proteins were separated by sodium dodecyl sulfate–polyacrylamide gel (10%) electrophoresis and then blotted onto PVDF membrane. After blocking for 2 hours, membranes were probed with rabbit antihuman 5-LOX polyclonal (1:500 dilution) or mouse anti-β-actin monoclonal antibody (1:2000 dilution), followed by horseradish peroxidase-conjugated goat antirabbit or goat antimouse IgG secondary antibodies (1:2000 or 1:4000 dilution, respectively). Membranes were developed using the Beyo ECL Plus Chemiluminescent kit. The film images were scanned and the gray scale of images was analyzed using the Image-Pro Plus 6.0 software (Media Cybernetics, Inc., Silver Spring, Maryland).
Quantitative PCR assay for ALOX5 mRNA
RNA was extracted with the Trizol reagent according to the manufacturer’s instructions. RNA was reverse transcribed into complementary DNA. Real-time PCR primers for
Comet assay for detecting DNA damage
Cells were resuspended in phosphate-buffered saline at a density of 2 to 5 × 105 cells/mL. Then, a mixture of 10 μL of cells and 65 μL of 0.5% low-melting point agarose (Promega, Madison, Wisconsin) was layered onto slides precoated with 100 μL 1% normal-melting point agarose (Biowest, Nuaillé, France) and immediately covered with a coverslip. After solidification of the gel at 4°C for 7 to 8 minutes, coverslips were removed and the slides were immersed in a cold (4°C) lysis solution at pH 10 for 1.5 to 2 hours. After washing with 0.4 mol/L Tris for 15 minutes, the slides were submerged in a fresh electrophoresis solution (1 mmol/L Na2EDTA and 300 mmol/L NaOH) for 20 minutes, and then electrophoresis was performed at 300 mA, 25 V, for 30 minutes. The slides were neutralized in 0.4 mol/L Tris and stained with 50 μL of 5 μg/mL PI. Then, the slides were photographed under a BA400EF-UPR fluorescence microscope (Motic, Wuhan, China). From each group, 50 cells were randomly selected and Olive tail moments were measured using the CASP 1.2.2 software (CaspLab, Germany).
Assay of cell apoptosis
The cells treated with BZD or BZD plus AA861 were collected by digesting with EDTA-free pancreatin and resuspended in 500 μL binding buffer from the Annexin V-FITC/PI Apoptosis Detection Kit. To this, 5 μL of Annexin V-FITC was added, followed by 5 μL of PI. Experiments were performed in the dark for 5 to 15 minutes. The cells were analyzed within 1 hour in a Moflo XDP Flow Cytometer (Beckman Coulter, Inc; Brea, California) under an excitation wavelength of 488 nm and an emission wavelength of 530 nm. The percentage of apoptotic cells was calculated by adding the fractions of early and late apoptotic cells. Samples were analyzed in triplicate.
Statistical Analysis
The results were expressed as mean ± standard deviation and analyzed for the statistical significance using the SPSS for Windows 11.5 software. The IC50 values were determined using the Probit method. Multisample means comparison was performed with the single factor analysis of variance, and the multiple comparisons were determined using the SNK-q and Dunnett T3 test. The
Results
Cooxidation of BZD by SLO
The SLO-mediated oxidation of BZD was dependent on pH and the concentrations of SLO, H2O2, and BZD in the reaction medium. The optimal oxidative conditions were pH 6.0, 90 mg/L SLO, 1.0 mmol/L BZD, and 2.0 mmol/L H2O2 (Figure 1). Under these conditions, the rate of BZD diimine production increased with the increasing SLO concentration. The maximum reaction rate (νmax) was 17.54 nmol·min−1·mg−1 and the Michaelis constant (Km) was 0.54 mmol/L. Nordihydroguaiaretic acid inhibited the SLO-mediated oxidation of BZD in a dose-dependent manner, with an IC50 value of 193.48 μmol/L.
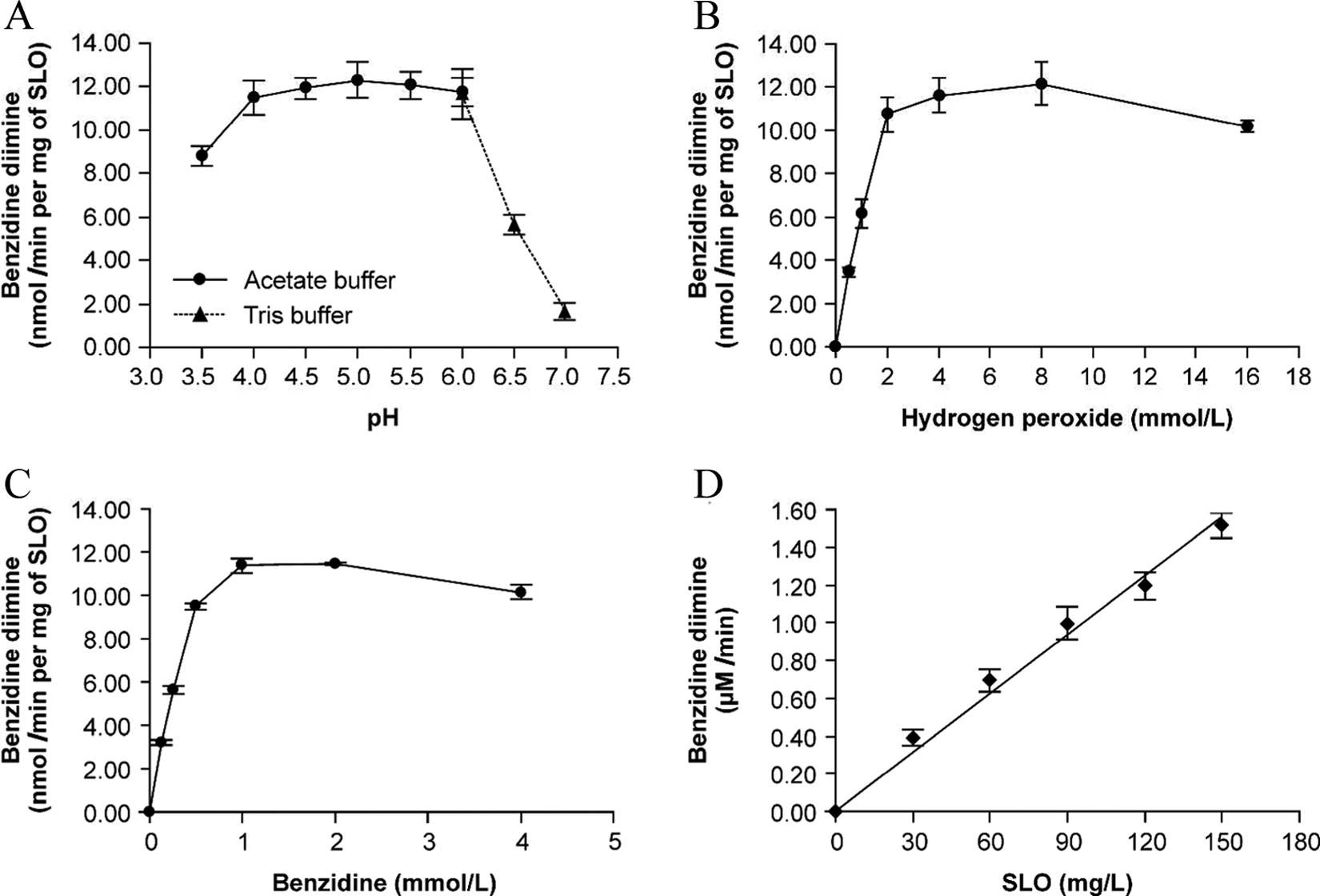
Effects of experimental conditions on oxidation of benzidine by soybean lipoxygenase (SLO). Effects of (A) pH, (B) benzidine, (C) hydrogen peroxide, and (D) SLO concentrations on SLO-mediated benzidine oxidation. Other experimental conditions were maintained at optimal oxidative conditions.
Benzidine Lowers the Viability of HBECs and AA861 and LV-5-LOX-shRNA Offer Protection
The results of MTT assay indicated that BZD decreased the survival rate of HBECs in a dose-dependent manner (Table 1). Pretreatment of the cells with the 5-LOX-specific inhibitor (AA861) facilitated the survival of BZD-treated HBECs significantly in a dose-dependent fashion (Table 2). Transfection with the LV-5-LOX-shRNA also significantly increased the survival of BZD-treated HBECs; moreover, the extent of protection (42.2%) was higher than that in the AA861-treated cells (35.3%; Table 3).
Effect of Benzidine on Survival in HBECs.a
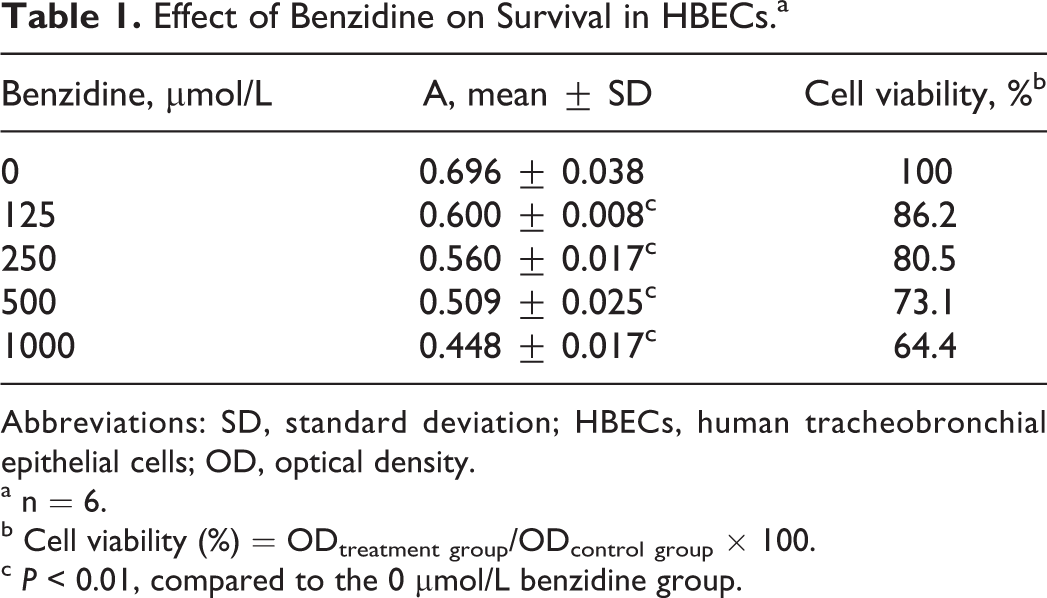
Abbreviations: SD, standard deviation; HBECs, human tracheobronchial epithelial cells; OD, optical density.
a n = 6.
b Cell viability (%) = ODtreatment group/ODcontrol group × 100.
c
Protective Effect of AA861 on Benzidine-Induced Cell Damage in HBECs.a
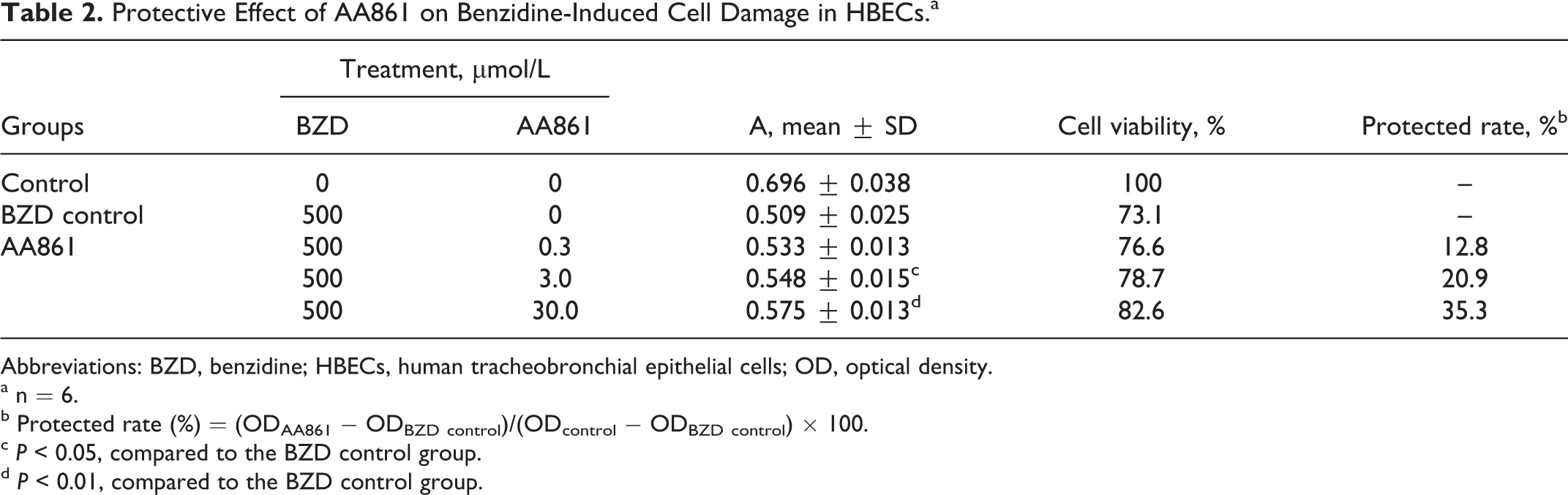
Abbreviations: BZD, benzidine; HBECs, human tracheobronchial epithelial cells; OD, optical density.
a n = 6.
b Protected rate (%) = (ODAA861 − ODBZD control)/(ODcontrol − ODBZD control) × 100.
c
d
The Protective Effect of Lentiviral-Mediated Reduction of

Abbreviations:
a n = 6.
b Protected rate (%) = ([OD HBE-NC control group − OD HBE-NC treatment group] − [OD HBE-5-LOX-shRNA control group − OD HBE-5-LOX-shRNA treatment group])/(OD HBE-NC control group − OD HBE-NC treatment group) × 100.
c
Benzidine, AA861, and LV-5-LOX-shRNA Modulate 5-LOX Protein Expression in HBECs
As shown in Figure 2A, the extents of protein expression of 5-LOX (ca 78 kD) were standardized to those of β-actin (ca. 43 kD) for each sample. The HBECs expressed 5-LOX (lane 1). With increasing BZD concentration, relative expressions of 5-LOX (lanes 3-5) increased 24.6%, 33.5%, and 105.5% (
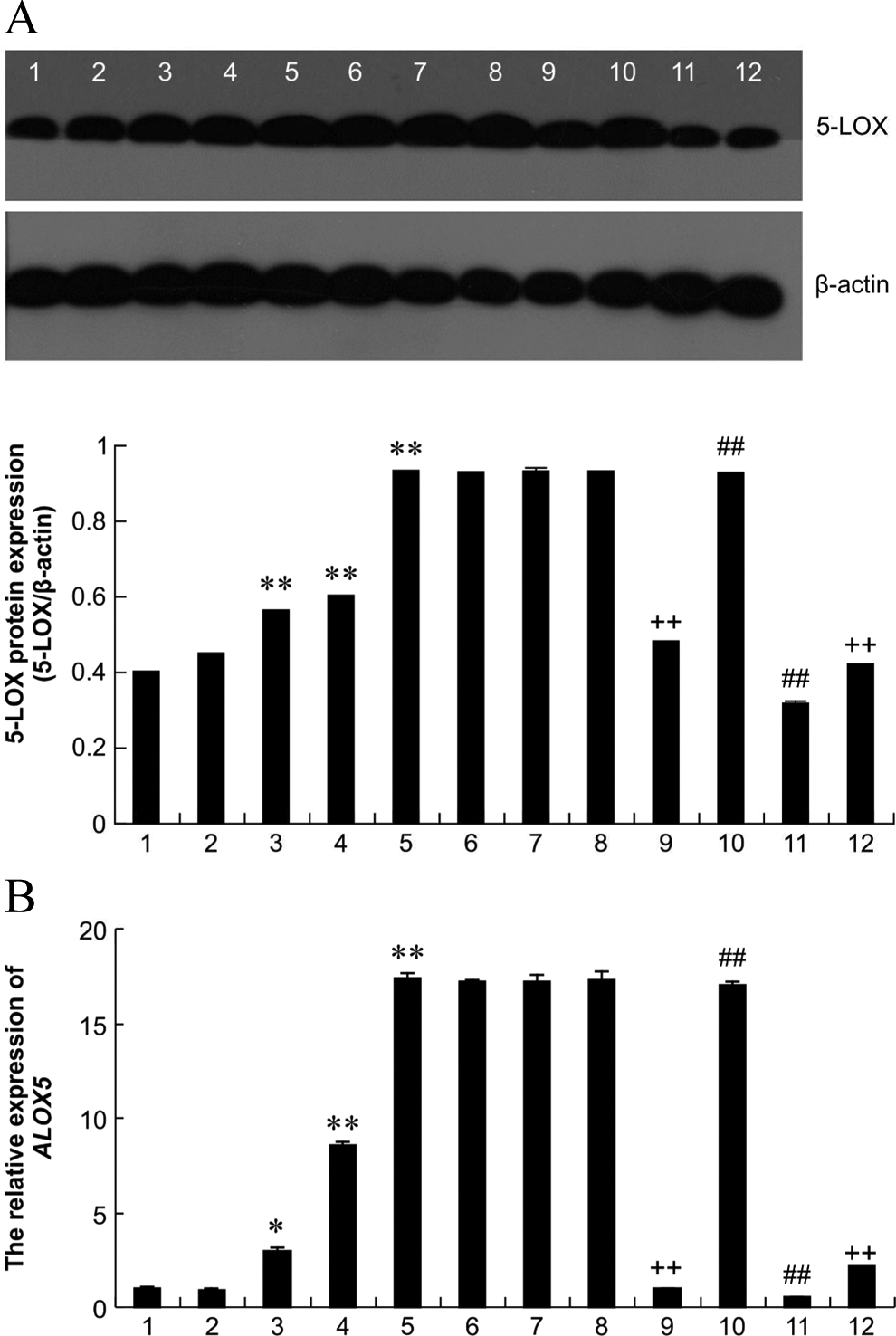
5-Lipoxygenase (5-LOX) protein and
Benzidine, AA861, and LV-5-LOX-shRNA Modulate ALOX5 mRNA Expression in HBECs
Benzidine significantly upregulated the
AA861 and LV-5-LOX-shRNA Inhibit BZD-Induced DNA Damage in HBECs
In the comet test, majority of the cells in the control group were uniformly round and lacked tailing. Clear comet tailing was observed following the BZD treatment (Figure 3). The DNA percentage in the comet tail (TDNA%) and olive tail moment values in all the BZD-treated groups were higher than those in the control group (

Comet assay images of HBECs treated with benzidine (BZD), AA861, or
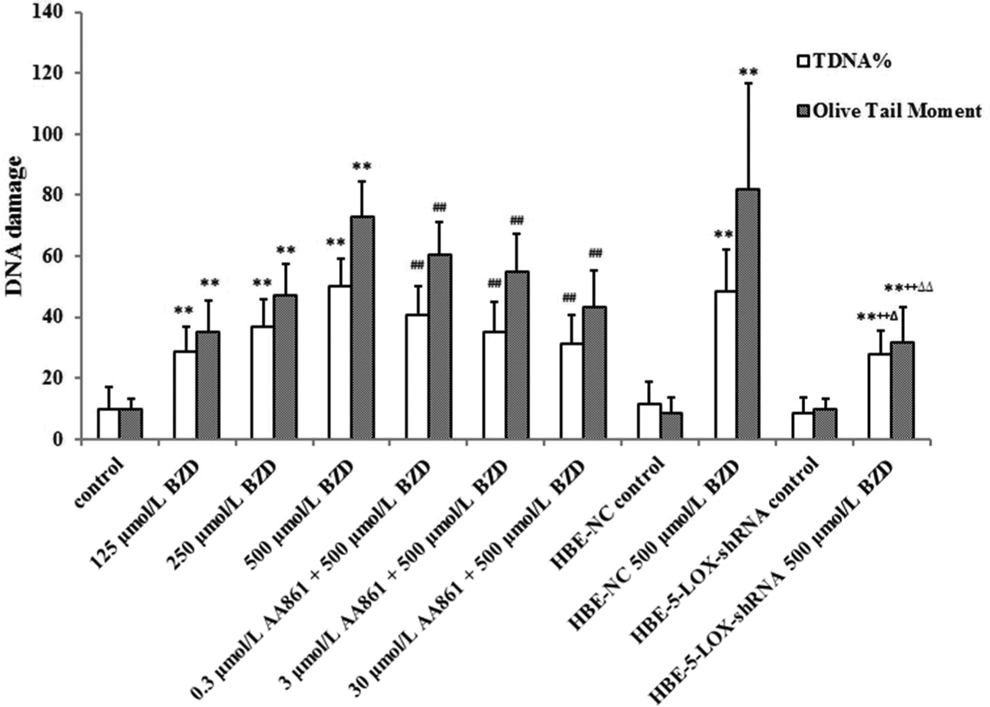
AA861 and lentiviral-mediated shRNA inhibit benzidine (BZD)-induced changes in TDNA% and olive tail moment in HBECs (n = 50). **
There was no significant difference between the TDNA% and olive tail moment values of the HBECs, HBE-NC, and HBE-5-LOX-shRNA control cells. Following treatment with 500 μmol/L BZD, the difference in values between HBECs and HBE-NC cells was not significant, but the TDNA% (27.7% ± 7.9% vs 50.0% ± 9.3% and 48.3% ± 13.7%) and olive tail moment value (32.0 ± 11.3 vs 73.2 ± 11.3 and 81.9 ± 34.7) in the HBE-5-LOX-shRNA cells were significantly lower than those in the treated HBECs and HBE-NC cells (
AA861 and LV-5-LOX-shRNA Protect Against BZD-Induced Apoptosis in HBECs
Benzidine increased the early apoptosis rate (0.60% ± 0.17%, 0.80% ± 0.14%, 23.89% ± 2.89%, and 33.09% ± 4.10% in 0, 125, 250, and 500 μmol/L BZD-treated cells, respectively) and apoptotic index (1.08 ± 0.29%, 1.28 ± 0.29%, 25.34 ± 2.79%, and 42.56 ± 3.13%) of HBECs; these effects were statistically significant when BZD concentration exceeded 250 μmol/L (
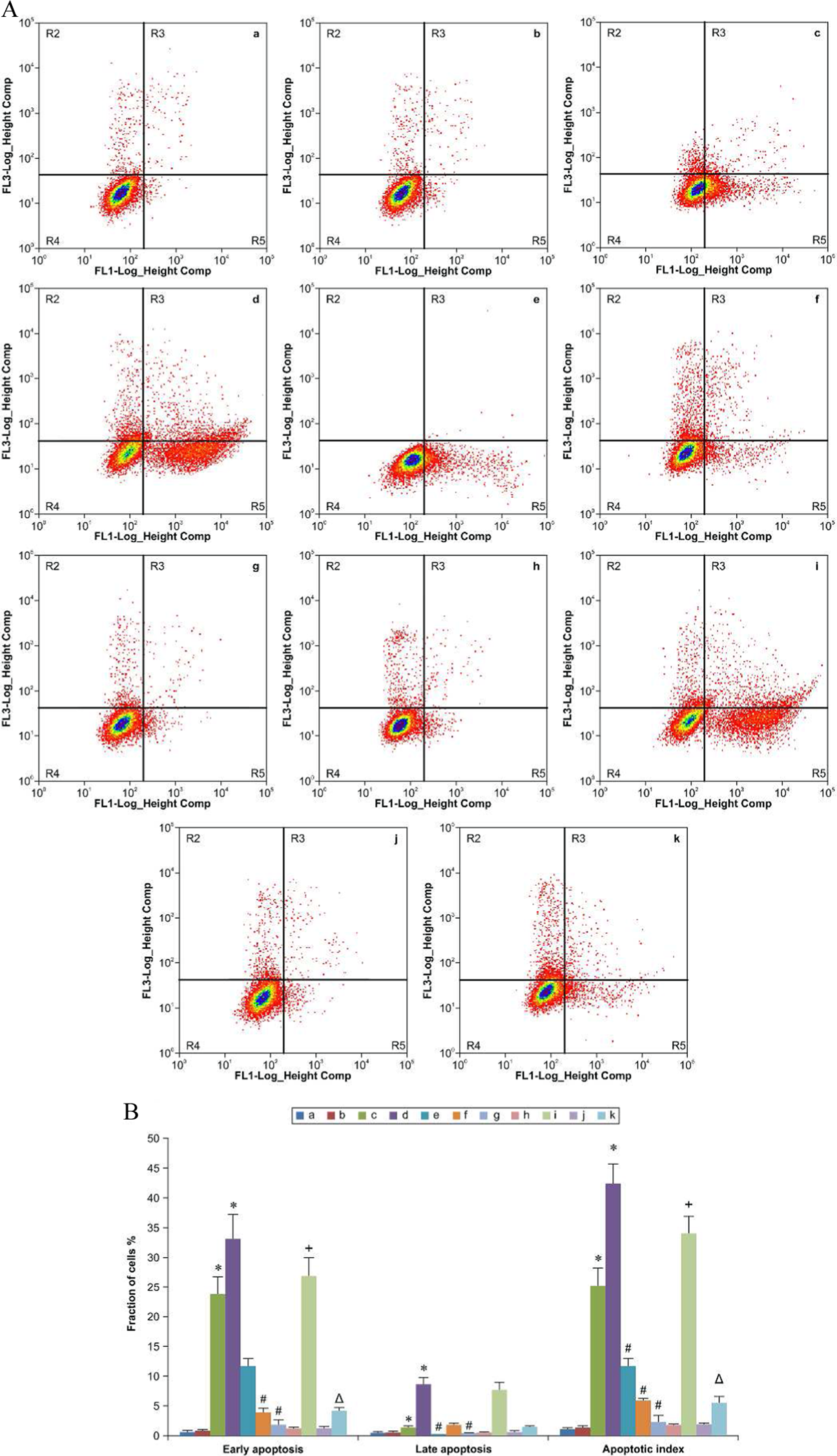
The protection of AA861 and lentiviral-5-LOX-shRNA against apoptosis induced by BZD in HBECs. (A) Graphs obtained by flow cytometry after Annexin V-FITC/PI dual staining. (B) Early apoptosis rate, late apoptosis rate, and apoptotic index. Data are expressed as the mean ± SD (n = 3); (a) solvent control, (b) 125 μmol/L BZD, (c) 250 μmol/L BZD, (d) 500 μmol/L BZD, (e) 0.3 μmol/L AA861 + 500 μmol/L BZD, (f) 3 μmol/L AA861 + 500 μmol/LBZD, (g) 30 μmol/L AA861 + 500 μmol/L BZD, (h) HBE-NC control group, (i) HBE-NC 500-μmol/L BZD-treatment group, (j) HBE-5-LOX-shRNA control group, (k) HBE-5-LOX-shRNA 500-μmol/L BZD-treatment group. *
Discussion
The CYP450 family is a critical player in the metabolism of endogenous and exogenous compounds. Studies have suggested that the levels and activity of CYP450 are low to undetectable in some extrahepatic tissues. Thus, it is difficult to explain oxidative metabolism of xenobiotics in these tissues. Lipoxygenases, a group of multifunctional single-strand peptide enzymes with relatively high activity in many mammalian extrahepatic tissues, are considered to represent an alternative pathway for the oxidation of endogenous and exogenous compounds in the extrahepatic tissues. 4
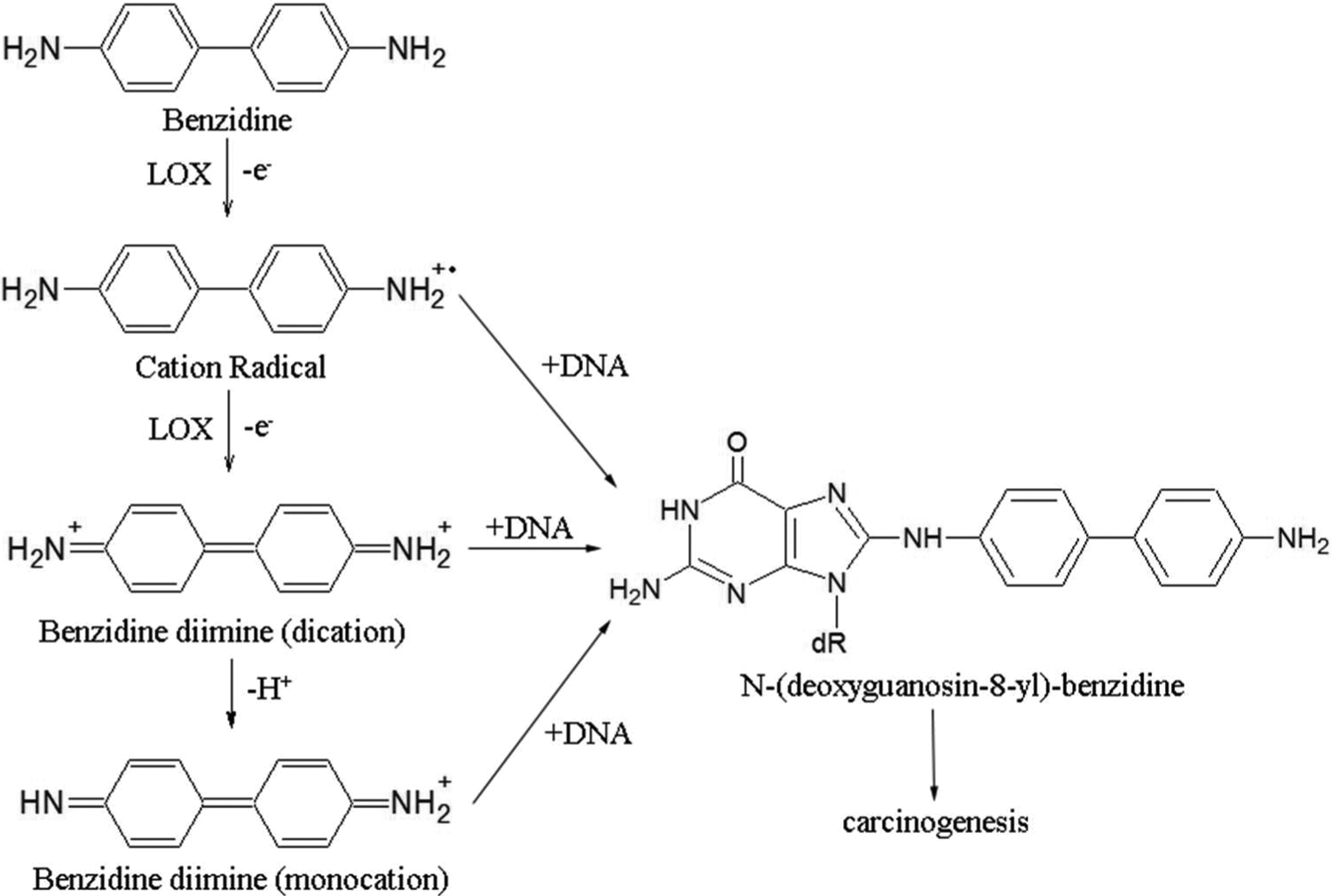
The SLO has been widely studied as a model LOX to understand the structure and function of LOX in general in cell-free enzyme systems. 25 In the current study, we confirmed that LOX (SLO) can catalyze oxidation of BZD to BZD diimine in vitro, which is reactive toward C-8 of guanine to yield N-(deoxyguanosin-8-yl)-benzidine as the major carcinogen-DNA adducts (Figure 6). 6,26 This suggested that the oxidative activation of BZD might be mediated by 5-LOX in HBECs, which could be one of the mechanisms underlying the BZD-related carcinogenesis.
Many epidemiological and animal experimental studies have indicated that BZD is carcinogenic and teratogenic. 27 In vivo, BZD is metabolized and activated to an electrophilic agent that can covalently bind to DNA or induce other forms of DNA damage, causing gene mutation, resulting in tumorigenesis. 1,28 Because the respiratory tract is both the route of exposure and the target organ of BZD and there is LOX activity in lung tissue, we select HBECs in our study. Our results suggested that activation products of BZD were cytotoxic to HBECs by causing DNA damage and inducing apoptosis. Determination of DNA damage, and the apoptosis and cell proliferation inhibition rates, can therefore indirectly reflect the levels of metabolic activation of BZD.
The 5-LOX is one of the most important LOX isoenzymes in vivo.
29
Given its relation to oncogenesis and tumor progress, we hypothesized that 5-LOX participated in BZD-induced carcinogenesis by catalyzing BZD oxidation and activation. To confirm the actions of the 5-LOX, we used both a pharmacological 5-LOX-specific inhibitor, AA861, and the reliable, highly specific, and efficient RNAi molecular biological strategy. The infection efficiency of the HBE-5-LOX-shRNA lentivirus in HBECs was above 80%. Western blotting and QPCR results demonstrated that LV-5-LOX-shRNA infection could successfully lower the
The LV-5-LOX-shRNA infection demonstrated a stronger inhibition of the BZD-induced decrease in cell survival rate and DNA damage than caused by AA861. However, the inhibitory action on apoptosis was weaker when compared to that of AA861, possibly because AA861 is a reducing inhibitor, which could have affected apoptosis by alternate mechanisms. AA861 had little influence on the
In summary, in the present study, we demonstrated that LOX catalyzed the BZD oxidation which in turn upregulated the expression of 5-LOX in HBECs. Moreover, the 5-LOX inhibitor AA861 and LV-5-LOX-shRNA transfection inhibited the BZD-induced DNA damage and apoptosis in HBECs. Therefore, the 5-LOX-mediated oxidative activation of BZD may be one of the mechanisms underlying BZD-related carcinogenesis. Additionally, this study also indicated that RNAi and pharmacological 5-LOX-specific inhibitor AA861 might be used as an approach for the prevention and treatment of 5-LOX-mediated activation and toxicity of exogenous chemicals, such as BZD.
Footnotes
Author Contribution
Yun Huang contributed to conception, design, acquisition, analysis, interpretation, drafting and critically revising manuscript. Jianan Hu contributed to conception, design, interpretation, and critically revising manuscript. Qingping Tan contributed to conception, design, acquisition, analysis and drafting manuscript. Yue Wu and Hongxiang Zhu contributed to acquisition, analysis. Minru Xiong contributed to conception and design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Natural Science Foundation of China 81072331 and Specialized Research Foundation for the Doctoral Program of Higher Education of Ministry of Education of the People’s Republic of China 200605330210.
