Abstract
Tripeptide-1, Hexapeptide-12, their metal salts and fatty acyl derivatives, and Palmitoyl Tetrapeptide-7 function primarily as skin conditioning agents, and Palmitoyl Tripeptide-1, Palmitoyl Hexapeptide-12, Tripeptide-1, Copper Tripeptide-1, and Palmitoyl Tetrapeptide-7 are also used in cosmetic products. Typical use concentrations of these ingredients are <10 ppm. The Panel noted that the low use concentrations and negative safety test data reviewed obviate any concerns relating to the safety of these ingredients in cosmetic products. Thus, the Panel concluded that these ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
The safety of Tripeptide-1, Hexapeptide-12, their metal salts and fatty acyl derivatives, and Palmitoyl Tetrapeptide-7 as used in cosmetics (listed below) was reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel (Panel). These ingredients function primarily as skin conditioning agents in cosmetic products. 1 The ingredient name, palmitoyl oligopeptide, listed in the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary) has been retired, because it was vague and indeterminately represented 2 other ingredients. The definition for this ingredient also contained no indication of the peptide sequence(s), a point that the Expert Panel deemed critical to a declaration of safety. Palmitoyl oligopeptide now represented by the name Palmitoyl Tripeptide-1 (Gly-His-Lys [GHK] peptide sequence) or Palmitoyl Hexapeptide-12 (Val-Gly-Val-Ala-Pro-Gly [VGVAPG] peptide sequence) in the Dictionary. Unfortunately, the dictionary recites 2 possible sequences for “Hexapeptide-12,” and relevant safety data be found for only one of these. Accordingly, this safety assessment only addresses Hexapeptide-12 and derivative ingredients (eg, Palmitoyl Hexapeptide-12) which contain the peptide sequence Val-Gly-Val-Ala-Pro-Gly. Specifically, the safety of such ingredients, containing the peptide sequence Ala-Pro-Gly-Val-Gly-Val, is not addressed. Thus, the data or conclusions in this safety assessment are not applicable to other peptide sequences. In this report, “Hexapeptide-12” only represents Val-Gly-Val-Ala-Pro-Gly.
This safety assessment also includes data on a trade name material containing Palmitoyl Tripeptide-1 (Gly-His-Lys [GHK] peptide sequence) and Palmitoyl Tetrapeptide-7 (Gly-Gln-Pro-Arg [GQPR] peptide sequence) and other trade name materials in which Palmitoyl Hexapeptide-12 or Palmitoyl Tripeptide-1 is the only oligopeptide component. The Val-Gly-Val-Ala-Pro-Gly sequence is an elastin peptide and the Gly-His-Lys sequence is a liver growth factor peptide and a fragment of type I collagen. Data on the biological activity of these peptides are also included herein. Tripeptide-1 (GHK) Palmitoyl Tripeptide-1 (GHK) Myristoyl Tripeptide-1 (GHK) Copper Tripeptide-1 (GHK) Bis(Tripeptide-1) Copper Acetate (GHK) Manganese Tripeptide-1 (GHK) Hexapeptide-12 (VGVAPG) Palmitoyl Hexapeptide-12 (VGVAPG) Myristoyl Hexapeptide-12 (VGVAPG) Palmitoyl Tetrapeptide-7 (GQPR)
Chemistry
The ingredients in this report are related structurally by bearing one of 3 distinct peptide sequences, either Tripeptide-1 (GHK), Hexapeptide-12 (VGVAPG), or Tetrapeptide-7 (GQPR). The ingredients reviewed in this safety assessment include one of these 3 peptide sequences also having a fatty acyl group at the N-terminus, or as their metal salts. For example, the structures of these 3 peptides are depicted in Figure 1, each with the fatty acyl group resulting from the reaction of palmitic acid with the N terminus of the peptide (ie, Palmitoyl Tripeptide-1, Palmitoyl Hexapeptide-12 (Palmitoyl-Valine-Glycine-Valine-Alanine-Proline-Glycine only), and Palmitoyl Tetrapeptide-7).
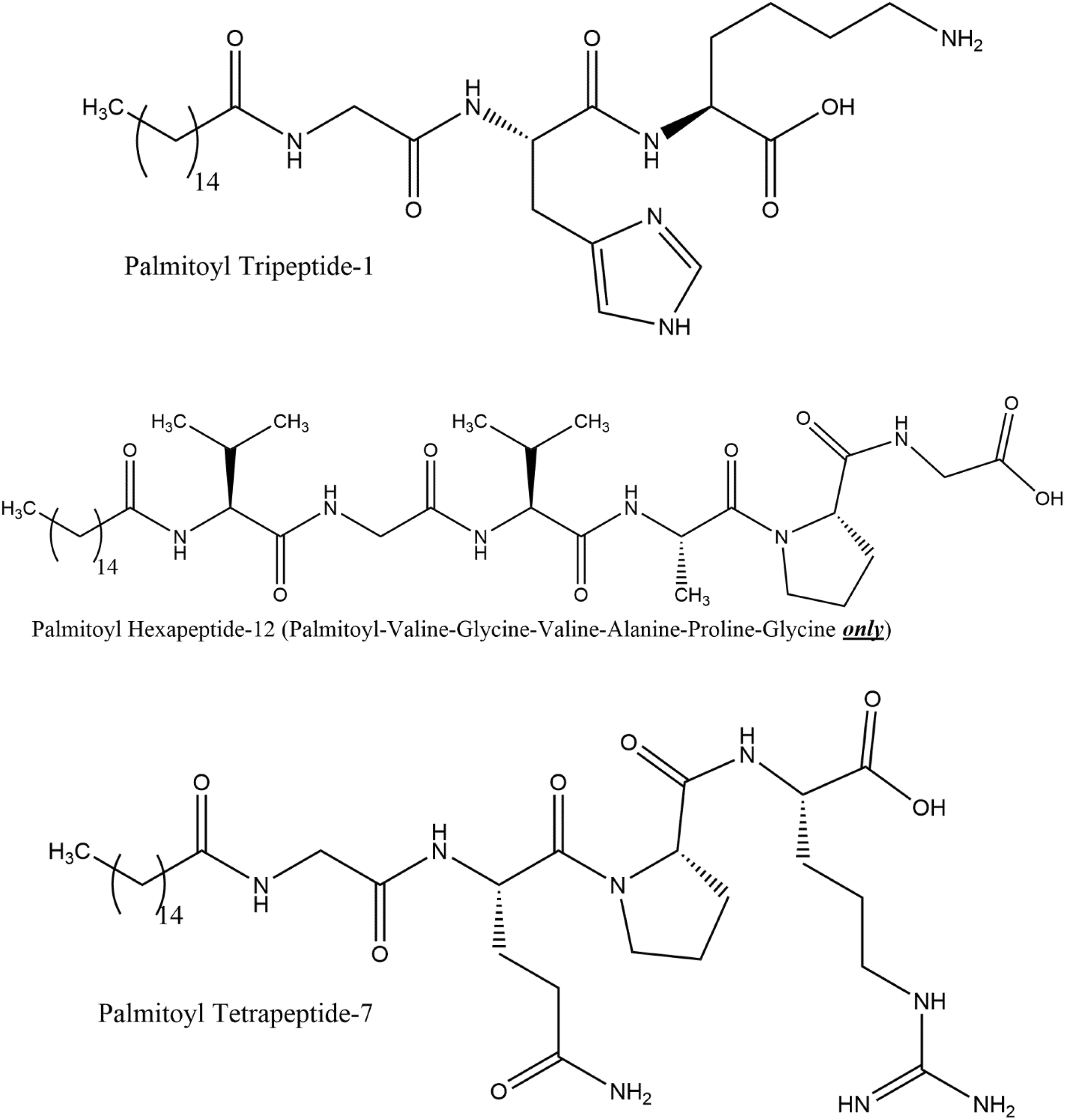
Example structures.
The definitions, structures, and functions of the ingredients in this report are included in Table 1. For example, Palmitoyl Tripeptide-1 consists of a short chain of 3 amino acids (also known as Gly-His-Lys [GHK] peptide sequence [a fragment of type I collagen]) connected via an amide bond at its N-terminus to palmitic acid. As another example, Palmitoyl Tetrapeptide-7 (Pal-GQPR) consists of a short chain of 4 amino acids (also known as GQPR peptide or glycine-glutamine-proline-arginine) similarly N-acylated with palmitic acid. The tetrapeptide portion is a fragment of the natural IgG immunoglobulins.
Definitions, Structures and Functions of the Ingredients in This Safety Assessment. 1,a
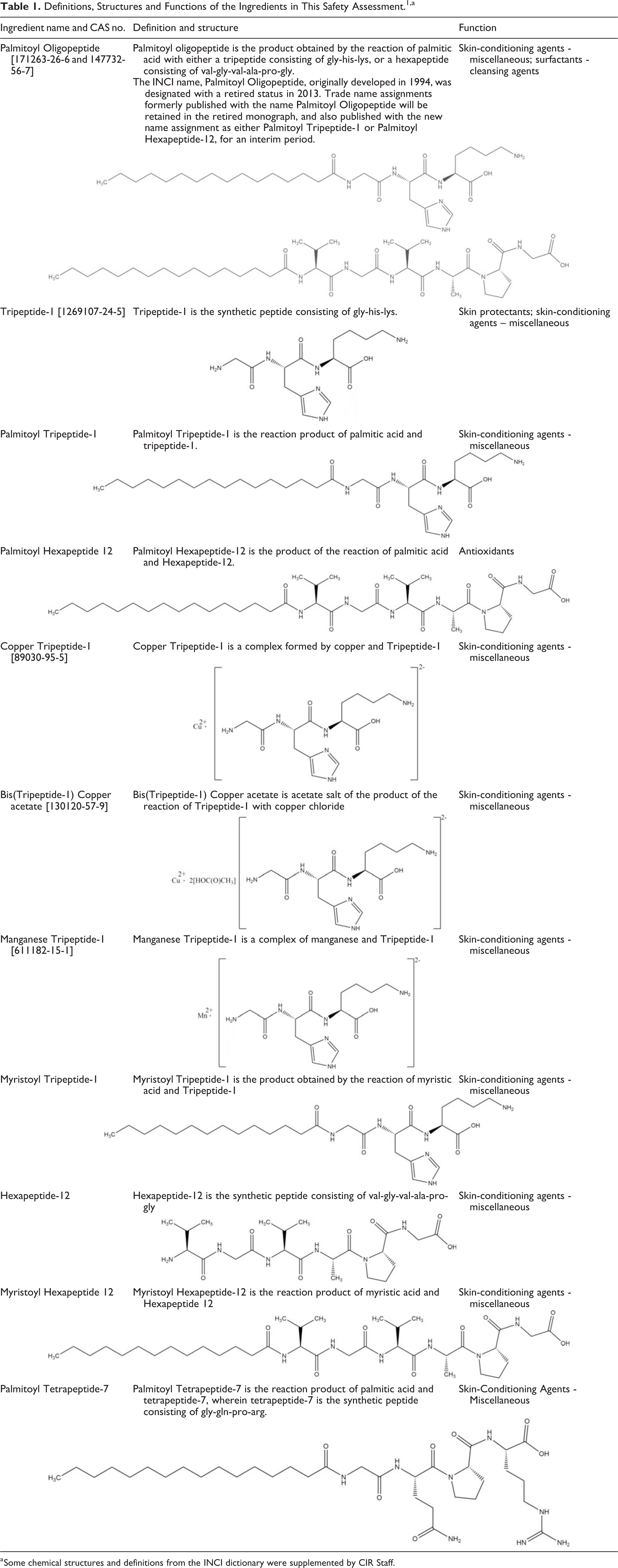
a Some chemical structures and definitions from the INCI dictionary were supplemented by CIR Staff.
Throughout this report, ingredient name subheadings will include the ingredient name and its abbreviation (ie palmitoyl group [pal] and the abbreviated peptide sequence, or the abbreviated peptide sequence only) in parentheses. For example, Palmitoyl Tripeptide-1 will be written as Palmitoyl Tripeptide-1 (GHK), Hexapeptide-12 will be written as Hexapeptide-12 (VGVAPG
Physical and Chemical Properties
A supplier provided properties data on palmitoyl oligopeptide, a trade name mixture comprising Palmitoyl Tripeptide-1 and Palmitoyl Tetrapeptide-7. 2 Properties of these 2 ingredients are also included below separately.
Palmitoyl Tripeptide-1 (Pal-GHK)
Palmitoyl Tripeptide-1 is a white powder, has a molecular weight of 578.80 Da, and an estimated logP of 4.81. 2 Another trade name mixture, containing 100 ppm Palmitoyl Tripeptide-1, is reported to have a density of 1.13.
Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
Palmitoyl Hexapeptide-12 is a white powder and has a molecular weight of 737.00 Da and a logP of 5.09. 2
Method of Manufacture
General Information
Peptides have been synthesized by solid-phase fluorenylmethoxycarbonyl chemistry (FMOC protection) using an automated peptide synthesizer. 3 Palmitic acid can be coupled to the deprotected amino-terminus of the resin-bound protected peptides either manually or using the peptide synthesizer, employing the same reaction conditions used in standard amino acid coupling. Peptides and mono-palmitic acid-peptide conjugates can then be cleaved from the resin, side-chain deprotected, and purified using standard procedures.
Several strategies for the synthesis of lipidated peptides, both in solution and on solid support have been developed. 4,5 Solid support is most frequently used to synthesize peptides with longer peptide chains. Shorter peptides have been synthesized both in solution and on solid support. For example, hexa- and heptapeptides corresponding to the Ras-C-terminus and Rab-C-terminus, respectively, have been synthesized in solution. 6,7
Palmitoyl Tripeptide-1 (Pal-GHK)
Palmitoyl Tripeptide-1 can be synthesized via standard stepwise peptide synthesis. 2 The C-terminal amino acid (Lys) is protected on its acidic function, after which each N-protected amino acid (Gly, His) is sequentially coupled, adding to the amino terminus, with deprotection and amidation of the peptide at each step to elongate by one amino acid. A last coupling procedure is accomplished with palmitic acid instead of an amino acid. The protected peptide is deprotected on the side-chains of lysine and histidine and on the C-terminal acid moiety of Lys.
According to another source, Palmitoyl Tripeptide-1 can be produced via solid-phase synthesis, yielding a substituted peptide of high purity (>97%). 8
Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
Palmitoyl Hexapeptide-12 can be produced via stepwise acid-phase peptide synthesis. The C-terminal amino acid (Gly) is protected on its acid function, after which each protected amino acid (Val-Gly-Val-Ala-Pro-) is sequentially coupled, adding to the amino terminus of the peptide at each step to elongate by one amino acid. A last coupling procedure is accomplished with palmitic acid instead of an amino acid. Finally, the protected peptide is deprotected at the C-terminal function (Gly) of the peptide. 2
Hexapeptide-12 (VGVAPG)
The synthetic peptide valine-glycine-valine-alanine-proline-glycine, which contains the recognition sequence for the elastin receptor, has been produced using an automated synthesizer. 9 Reverse-phase high-performance liquid chromatography (HPLC) was used for further purification.
Copper Tripeptide-1 (GHK-Cu2+)
Copper Tripeptide-1 is prepared by combining purified glycyl-
Crystalline Copper Tripeptide-1 is prepared by dissolving glycyl-
Composition/Impurities
Palmitoyl Tripeptide-1 (Pal-GHK) and Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
According to one supplier, the impurities content of Palmitoyl Tripeptide-1 and Palmitoyl Hexapeptide-12 has been described as follows: acetate (<5%), palmitic acid (<5%), and water (<5%). 2
Tripeptide-1 (GHK)
Crude Tripeptide-1 is approximately 95% pure, likely containing some incompletely deprotected peptides. 10 However, these impurities can be removed in glass-distilled water (50 mg/mL), centrifuging at 20 000g for 1 hour at 3°C, and then lyophilizing the supernatant.
Use
Cosmetic
The ingredients reviewed in this safety assessment function primarily as skin conditioning agents in cosmetic products. 1 According to information supplied to the U.S. Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), the following palmitoyl oligopeptides or oligopeptides are being used in cosmetic products: 11 palmitoyl oligopeptide (name retired, peptide sequence not stated), Palmitoyl Tripeptide-1, Palmitoyl Hexapeptide-12, Tripeptide-1, Copper Tripeptide-1, and Palmitoyl Tetrapeptide-7. The peptide sequence for palmitoyl oligopeptide is not stated in the VCRP database or in the survey of ingredient use concentrations mentioned below; however, this designation could refer to either GHK (Tripeptide-1) or VGVAPG (Hexapeptide-12).
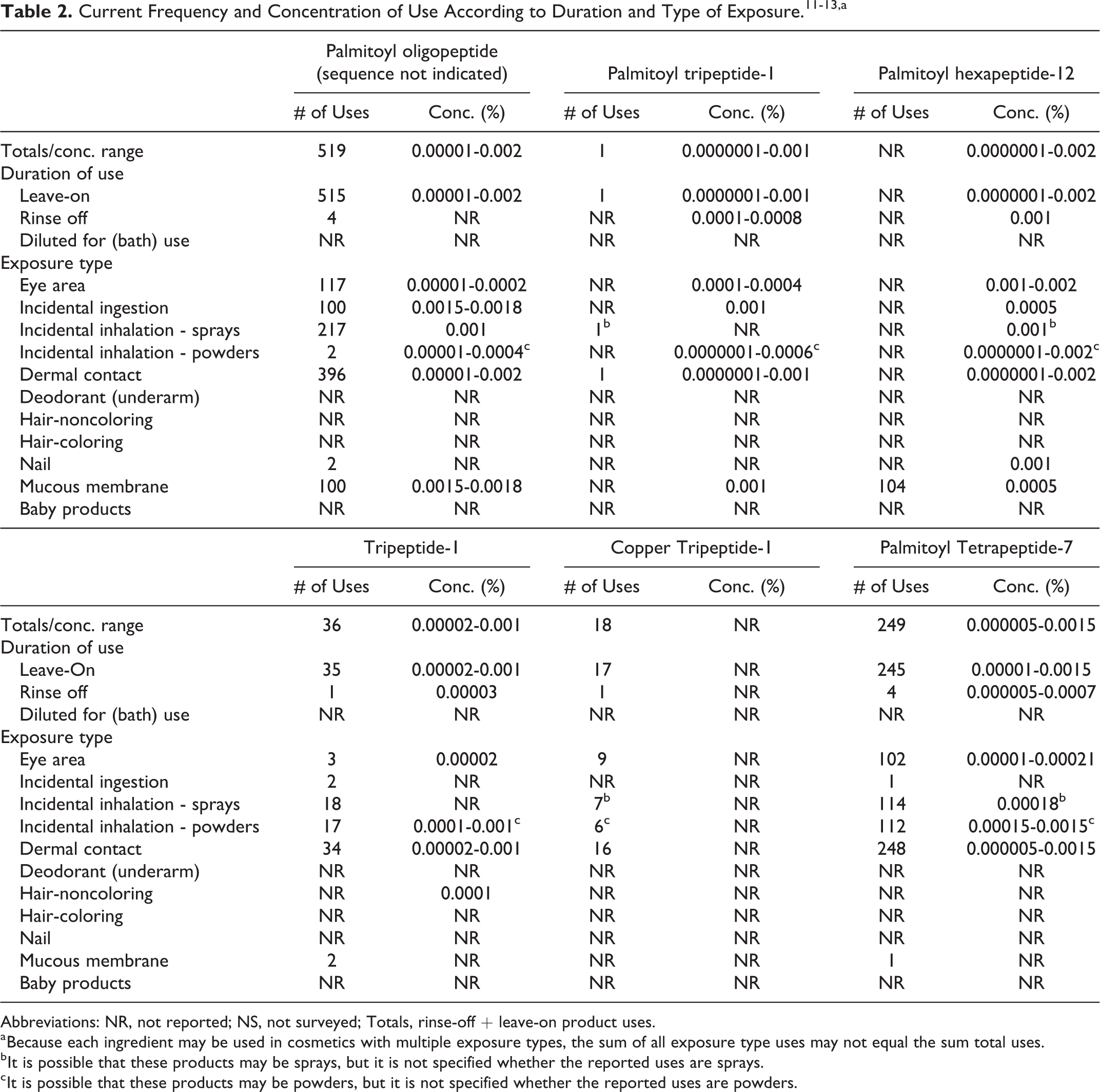
Results from a survey of ingredient use concentrations conducted by the Personal Care Products Council (Council) in 2013 and updated in 2014 indicate that, collectively, the ingredients reviewed in this safety assessment are being used at maximum concentrations ranging from 0.0000001% (Palmitoyl Tripeptide-1 and Palmitoyl Hexapeptide-12) to 0.002% (Palmitoyl Hexapeptide-12). 12,13 The highest concentration of 0.002% relates to ingredient use in leave-on products. Voluntary Cosmetic Registration Program data on ingredient use frequencies and use concentration data provided by the Council are summarized in Table 2. In addition to the data included in the survey of ingredient use concentrations, one submission indicated that peptides are being used in cosmetic products at concentrations between 1 ppm and 30 ppm, and that their use at concentrations <10 ppm is customary. 14
Abbreviations: NR, not reported; NS, not surveyed; Totals, rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
b It is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
c It is possible that these products may be powders, but it is not specified whether the reported uses are powders.
Cosmetic products containing Tripeptide-1, Hexapeptide-12, and related amides may be applied to the skin and hair, or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Palmitoyl Hexapeptide-12 is used in body and hand sprays (maximum use concentration = 0.002%). Because this ingredient is used in products that are sprayed, the ingredient could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays. 15 -18 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 15,16
Toxicokinetics
In Vivo Studies
Tripeptide-1 (GHK)
Tripeptide-1 (1% in saline; dose = 10 mg/kg) was injected into the tail vein of male rats (number not specified).
19
Blood samples were collected prior to dosing and for up to 60 minutes post-dosing. Plasma concentration–time profiles of Tripeptide-1 and its
In Vitro Studies
Tripeptide-1 (GHK)
In an enzyme assay, the liver growth factor, Tripeptide-1, was hydrolyzed by an aminotripeptidase purified from rat brain cytosol. 20
Toxicology
Acute Oral Toxicity
Palmitoyl Tripeptide-1 (Pal-GHK)
The acute oral toxicity of a trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 was evaluated using 10 Sprague-Dawley rats (5 males, 5 females). 22 The test substance was administered by gavage at a dose of 2,000 mg/kg. Dosing was followed by a 14-day observation period, after which necropsy was performed. Dosing had no effect on general behavior or body weight gain, and none of the animals died. There were no apparent abnormalities at necropsy. This mixture was classified as nontoxic (LD50 > 2,000 mg/kg).
Repeated Dose Toxicity
Palmitoyl Tripeptide-1 (Pal-GHK)
There were no clinical signs or mortalities in a cumulative skin irritation study on a trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 involving guinea pigs. Details relating to the test protocol were not provided. 23
In the guinea pig maximization test on a trade name mixture containing 100 ppm Palmitoyl Tripeptide-1, the test substance was evaluated at a concentration of 75% in a saline vehicle. 24 Clinical signs were not observed and none of the animals died during the study. Additionally, body weight gain was unaffected by test substance administration.
Ocular Irritation
In Vivo
Palmitoyl Tripeptide-1 (Pal-GHK)
The ocular irritation potential of the ingredient, a trade name mixture containing 100 ppm Palmitoyl Tripeptide-1, was evaluated using 3 male New Zealand white rabbits. 25 The test substance (0.1 mL) was instilled into the conjunctival sac of the left eye of each animal, and the eyes were not rinsed. Ocular reactions were scored at approximately 1 hour, 24 hours, 48 hours, and 72 hour-postinstillation, and then on days 5 and 8. On day 1, very slight conjunctival reactions (chemosis and redness) were observed in all 3 animals. No other ocular reactions were observed for the duration of the study. It was concluded that this mixture was a slight irritant (maximum ocular irritation index = 4.7).
Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
A trade name mixture containing 100 ppm Palmitoyl Tripeptide-12 was instilled as a single dose (0.1 mL) into the left eye of each of 3 male New Zealand white rabbits. 26 Eyes were not rinsed, and reactions were scored at 24 hours, 48 hours, and 72 hour-postinstillation. Moderate or slight conjunctival irritation (chemosis [score = 2] and redness [score = 1 or 2]) was observed in all animals for up to 4 days postinstillation. Neither iris irritation nor corneal opacity was observed. This mixture was considered a nonirritant when instilled into the eyes of rabbits. This conclusion was based on the observation that the mean scores for chemosis, redness, and degree of corneal opacity in 2 of the 3 animals did not reach the criteria for irritation under the experimental conditions of this study.
In Vitro
Palmitoyl Tripeptide-1 (Pal-GHK)
The ocular irritation potential of a trade name mixture containing 1,000 ppm Palmitoyl Tripeptide-1 was evaluated in the hen’s egg chorioallantoic membrane in vitro assay. 27 Details relating to the assay protocol were not presented. Sodium dodecyl sulfate (0.5% w/v) served as the positive control. This trade name mixture was classified as slightly irritating, but was considered “well tolerated.” The positive control was classified as an ocular irritant.
Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
The hen’s egg chorioallantoic membrane in vitro assay was also used to evaluate the ocular irritation potential of a trade name mixture containing 200 ppm Palmitoyl Hexapeptide-12. 28 The test substance was diluted to 50% (w/v) in distilled water prior to testing. The score for each egg was determined by the sum of the notations of hyperemia, hemorrhage, and coagulation (coagulation = opacity and/or thrombosis). The notation for the test substance corresponded to the arithmetic mean, rounded to the nearest tenth, of the scores obtained for 4 eggs. Sodium dodecyl sulfate (0.5% w/v) served as the positive control. The mean irritation index was 0.8 for the diluted trade name mixture and 12.0 for the positive control. The test substance was classified as practically nonirritating.
Ocular irritation potential of this trade name mixture was also evaluated in the Statens Seruminstiut Rabbit Corna (SIRC) fibroblastic cell line using the neutral red releasing method. 28 Sodium dodecyl sulfate and sodium chloride served as positive and negative controls, respectively. The IC50, defined as the test substance concentration that inhibited 50% of cell survival and growth, was >50%, and the mortality at 50% dilution was 37.9%. It was concluded that the test substance caused negligible cytotoxicity.
Palmitoyl Tetrapeptide-7 (Pal-GQPR)
The hen’s egg chorioallantoic membrane in vitro assay was used to evaluate the ocular irritation potential of a trade name mixture containing 500 ppm Palmitoyl Tetrapeptide-7. The assay procedure stated in the preceding section was used. The test material was classified as slightly irritating (mean irritation index = 3.75). 29
Skin Irritation and Sensitization
The following skin irritation and sensitization data are also summarized in Table 3.
Skin Irritation and Sensitization Studies.

Abbreviation: HRIPT, human repeated insult patch test.
Animal
Palmitoyl Tripeptide-1 (Pal-GHK)
A trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 was evaluated for its skin irritation potential using 3 male New Zealand white rabbits. 30 This mixture was applied to scarified or nonscarified skin of the flank (0.5 mL on 6 cm2 area, clipped free of hair), using an occlusive hypoallergenic dressing, for 24 hours. Reactions were scored at 24 hours and 72 hours postapplication. At 24 hour-postapplication, slight erythema was observed on both flanks of 2 rabbits. These were the only reactions observed during the study. This trade name mixture was classified as a nonirritant (PII = 0.3).
A cumulative skin irritation study on this mixture was performed using 10 guinea pigs (5 males, 5 females). 23 The test substance was applied to the left flank (0.05 mL on a 2 cm × 2 cm area, clipped free of hair) once daily for 14 consecutive days. The right flank was treated with purified water (control). The test site was not covered with a dressing during the application period. Reactions were evaluated immediately prior to each application and approximately 24 hours after the last application by comparing the reactions on both flanks. The animals were killed and cutaneous samples were removed from treated sites. Cutaneous reactions were not observed during the study. However, a very slight beige coloration of the skin was observed in each animal. It was concluded that the test substance was a nonirritant in guinea pigs (maximum weekly mean irritation index = 0).
The skin sensitization potential of this mixture was studied using 30 guinea pigs (strain not stated) in the maximization test. 24 The test group consisted of 20 animals (10 males, 10 females) and the control group consisted of 10 animals (5 males, 5 females). During induction day 1, test animals were injected intradermally with the test substance (1% in 0.9% isotonic saline vehicle [injection volume = 0.1 mL]) in the presence of Freund’s complete adjuvant. The test substance (0.5 mL) was cutaneously applied to test animals on induction day 8. The control group was treated only with vehicle during the induction period. The challenge phase was initiated after a 12-day nontreatment period. A dry compress containing the test substance (75% in saline vehicle [0.5 mL]) was applied, under an occlusive dressing to the right flank, and vehicle only (0.5 mL) was applied to the left flank of all animals. The compress and occlusive dressing were removed at the end of the 24-hour application period. Challenge reactions were evaluated at 24 hours and 48 hours after removal. The animals were then killed and cutaneous samples were obtained from challenge sites. Microscopic examination was not performed on cutaneous samples. Cutaneous reactions were not observed during the challenge phase. It was concluded that test substance did not induce sensitization in guinea pigs.
Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
A trade name material containing 100 ppm Palmitoyl Hexapeptide-12 was evaluated in a skin irritation study involving 3 male New Zealand white rabbits. 31 A dry compress containing the test substance was applied (0.5 mL on 6 cm2 area, clipped free of hair) for 4 hours under a semiocclusive dressing. Reactions were scored at 24 hour, 48 hour, and 72 hour postremoval. Moderate cutaneous reactions (erythema, but no edema) were observed, and these reactions were reversible within 24 hours or 48 hours. Cutaneous reactions were not observed on days 3 and 4. This trade name mixture was considered a nonirritant (mean erythema score <1.0).
Human
Palmitoyl Tripeptide-1 (Pal-GHK)
The skin irritation potential of a trade name mixture containing 1,000 ppm Palmitoyl Tripeptide-1 was evaluated using 10 healthy adult volunteers. 27 The mixture was applied to dorsal skin (∼0.02 mL on 50 mm2 area), using an occlusive patch (Finn chamber on Scanpor), for 48 hours. Untreated sites (covered with occlusive patch) served as negative controls. Reactions were scored 30 minutes after patch removal. Neither irritation nor significant cutaneous intolerance was observed (primary irritation index [PII] = 0). There was also no evidence of a secondary effect. The test substance was classified as “very well tolerated”.
The skin sensitization potential of this trade name mixture was evaluated in a human repeated insult patch test (HRIPT) using 52 subjects. 32 The study was initiated with 57 subjects (16 to 79 years old), 5 of whom withdrew for reasons unrelated to ingredient application. During induction, patches (type not stated) were applied 3 times per week for a total of nine 24-hour induction applications. Non-treatment periods during the induction phase were described as 24 hours following each Tuesday and Thursday patch removal and 48 hours following each Saturday removal. The challenge phase was initiated following a 2-week nontreatment period. Challenge patches were applied for 24 hours to a new test site that was adjacent to the induction patch site. Reactions were scored 24 hours and 72 hours after patch application. Barely perceptible (+) to moderate (2+) reactions were observed during induction and/or challenge phases. However, it was noted that these transient, low-level responses were considered clinically insignificant. It was concluded that the test substance did not indicate a clinically significant potential for dermal irritation or allergic contact sensitization.
Palmitoyl Hexapeptide-12 (Pal-VGVAPG)
A trade name mixture containing 200 ppm Palmitoyl Hexapeptide-12 was evaluated for skin irritation potential using 10 adult volunteers. 28 A single 48-hour application of the test substance (diluted to 50%) was made, under an occlusive patch, on dorsal arm skin. Neither irritation nor significant cutaneous intolerance was observed (PII = 0). There was also no evidence of a secondary effect. Diluted test material was considered very well tolerated.
An HRIPT on this trade name mixture was performed using 53 healthy adult volunteers. 33 The test substance was diluted to a concentration of 50% prior to application. The test procedure involved 48-hour occlusive patch applications of the diluted test substance (area of application not specified). Eight induction applications were made, followed by challenge patch application. Neither skin irritation (mean irritation index [induction] = 0.04) nor sensitization was observed.
Palmitoyl Tetrapeptide-7 (Pal-GQPR)
The skin irritation and sensitization potential of a trade name mixture containing 500 ppm Palmitoyl Tetrapeptide-7, was evaluated in an HRIPT involving 52 healthy male and female subjects (age range: 18 to 79 years). 34 The test material (0.2 mL) was applied to a 3/4″ × 3/4″ occlusive patch that was placed on the upper back between the scapulae. During the induction phase, patches were applied (24 hours) 3 times per week for a total of 9 induction applications. After a 2-week nontreatment period, a 24-hour challenge patch was applied to a new site that was adjacent to the original application site. Reactions were scored at the time of patch removal and at 24 hour and 72 hour-postapplication. It was concluded that results for the test material did not indicate a potential for dermal irritation or allergic contact sensitization.
Reproductive and Developmental Toxicity
Data on the reproductive and developmental toxicity of Tripeptide-1, Hexapeptide-12, their metal salts and fatty acyl derivatives, and Palmitoyl Tetrapeptide-7 were neither found in the published literature nor submitted.
Genotoxicity
Palmitoyl Tripeptide-1 (Pal-GHK)
The genotoxicity of a trade name mixture containing 1,000 ppm Palmitoyl Tripeptide-1 was evaluated in the Ames test, with and without metabolic activation, using the following Salmonella typhimurium strains: TA98, TA100, TA1535, and TA1538. 35 The test material (0.1 mL in ethanol solution) was nongenotoxic. In another assay, the genotoxicity of a trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 was evaluated, with and without metabolic activation, using the following S typhimurium strains: TA98, TA102, TA1535, and TA1537. 36 At doses up to 5,000 µg/plate, the test material was classified as nongenotoxic.
Palmitoyl Tetrapeptide-7 (Pal-GQPR)
The Ames test was used to evaluate the genotoxicity of a trade name mixture containing 500 ppm Palmitoyl Tetrapeptide-7, in the following S typhimurium strains: TA98, TA100, TA1535, TA1537, and TA1538. 37 The test material (1 mL, in 9 mL of DMSO) was evaluated with and without metabolic activation. The test material was considered nonmutagenic in all bacterial strains.
Carcinogenicity
Data on the carcinogenicity of Tripeptide-1, Hexapeptide-12, their metal salts and fatty acyl derivatives, and Palmitoyl Tetrapeptide-7 were neither found in the published literature nor submitted.
Summary
The safety of the following ingredients in cosmetics is reviewed in this safety assessment: Tripeptide-1, Palmitoyl Tripeptide-1, Myristoyl Tripeptide-1, Hexapeptide-12, Palmitoyl Hexapeptide-12, Myristoyl Hexapeptide-12, Copper Tripeptide-1, Bis(Tripeptide-1) Copper Acetate, Manganese Tripeptide-1, and Palmitoyl Tetrapeptide-7.
The ingredients reviewed in this safety assessment function primarily as skin conditioning agents in cosmetic products. According to information supplied to the USFDA by industry as part of the VCRP, the following palmitoyl oligopeptides are being used in cosmetic products: palmitoyl oligopeptide (name retired, peptide sequence not stated), Palmitoyl Tripeptide-1, Tripeptide-1, Copper Tripeptide-1, and Palmitoyl Tetrapeptide-7. The peptide sequence for palmiotoyl oligopeptide is not stated in the VCRP database or in the Council’s survey of ingredient use concentrations; however, the sequence could be either GHK (Tripeptide-1) or VGVAPG (Hexapeptide-12).
Results from a survey of ingredient use concentrations conducted by the Council in 2013 and updated in 2014 indicate that, collectively, the ingredients reviewed in this safety assessment are being used at maximum concentrations ranging from 0.0000001% (Palmitoyl Tripeptide-1 and Palmitoyl Hexapeptide-12) to 0.002% (Palmitoyl Hexapeptide-12). The highest concentration of 0.002% relates to ingredient use in leave-on products. In addition to the data included in the survey of ingredient use concentrations, one submission indicated that peptides are being used in cosmetic products at concentrations between 1 ppm and 30 ppm, and that their use at concentrations of <10 ppm is customary.
The impurity content of both Palmitoyl Tripeptide-1 and Palmitoyl Hexapeptide-12 has been described as follows: acetate (<5%), palmitic acid (<5%), and water (<5%). Crude Copper Tripeptide-1 is reported to be approximately 95% pure, but can be further washed to remove residual, nondeprotected peptides, resulting from incomplete synthesis.
After IV injection, Tripeptide-1 was rapidly degraded to L-histidyl-L-lysine, which was rapidly eliminated (in minutes) from circulating blood. It has been reported that Tripeptide-1 is unstable in human plasma and is rapidly degraded by aminopeptidases. In an enzyme assay, the liver growth factor, Tripeptide-1, was hydrolyzed by an aminotripeptidase purified from rat brain cytosol.
A trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 was nontoxic (LD50 > 2,000 mg/kg) in an acute oral toxicity study involving rats. Studies designed to evaluate the repeated dose toxicity of the ingredients reviewed in this safety assessment were not found in the published literature. However, neither treatment-related clinical signs/mortalities were reported in cumulative skin irritation/sensitization studies on this mixture in a guinea pig maximization test.
A trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 was slightly irritating to the eyes of rabbits. A trade name mixture containing 100 ppm Palmitoyl Tripeptide-12 was nonirritating to the eyes of rabbits. In the hen’s egg chorioallantoic membrane in vitro assay for evaluating ocular irritation potential, a trade name mixture containing 1,000 ppm Palmitoyl Tripeptide-1 was classified as an irritant, a trade name mixture containing 200 ppm Palmitoyl Hexapeptide-12 was practically nonirritating, and a trade name mixture containing 500 ppm Palmitoyl Tetrapeptide-7 was slightly irritating. In the in vitro neutral red release assay (SIRC fibroblastic cell line) for evaluating ocular irritation potential, a trade name mixture containing 200 ppm Palmitoyl Hexapeptide-12 caused “unimportant cytotoxicity.”
In skin irritation studies (single application) involving rabbits, a trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 and a trade name mixture containing 100 ppm Palmitoyl Hexapeptide-12 were classified as non-irritants. A trade name mixture containing 100 ppm Palmitoyl Tripeptide-1 was classified as a nonirritant in a cumulative skin irritation study involving guinea pigs. This mixture also did not induce skin sensitization at a challenge concentration of 75% in the guinea pig maximization test.
In human skin irritation studies (single application), a trade name mixture containing 1,000 ppm Palmitoyl Tripeptide-1 and a different mixture containing 200 ppm Palmitoyl Hexapeptide-12 (50%) were classified as non-irritants. Human repeated insult patch test results for these 2 trade name mixtures, and another trade name mixture containing 500 ppm Palmitoyl Tetrapeptide-7, were negative for skin irritation and sensitization.
Ames test results for Palmitoyl Tripeptide-1 and Palmitoyl Tetrapeptide-7 were negative with and without metabolic activation in S typhimurium bacterial strains.
Discussion
Use concentration data indicate that the ingredients reviewed in this safety assessment are being used at concentrations up to 0.002%, a value reported for Palmitoyl Hexapeptide-12 in leave-on products (in night products [not spray] and in body and hand sprays). Information substantiating the use of peptides at concentrations between 1 ppm and 30 ppm in cosmetic products, and customary use at concentrations of <10 ppm, was also evaluated. The Panel agreed that the data on peptide use should be relied upon as typical use concentrations for all of the ingredients reviewed in this safety assessment, which include Tripeptide-1, Hexapeptide-12, their metal salts and fatty acyl derivatives, and Palmitoyl Tetrapeptide-7. Thus, given the low use concentrations of these ingredients, together with the negative repeated dose toxicity, skin irritation and sensitization, and genotoxicity data, it was determined that the available data support the safe use of these ingredients in cosmetic products. The Panel noted that this safe conclusion is applicable only to ingredient names associated with the following known peptide sequences: GHK, VGVAPG, and GQPR.
Palmitoyl Hexapeptide-12 is used in body and hand sprays (maximum use concentration = 0.002%). Because this ingredient is used in products that are sprayed, the ingredient could possibly be inhaled. The Panel discussed the issue of incidental inhalation exposure from propellant and pump sprays and powders, and considered pertinent data indicating that incidental inhalation exposures to this ingredient in such cosmetic products would not cause adverse health effects. These data considered include data characterizing the potential for this ingredient to cause repeated dose toxicity, dermal irritation or sensitization, and genotoxicity. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Panel concluded that the following cosmetic ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment. Tripeptide-1 (GHK) Palmitoyl Tripeptide-1 (GHK) Myristoyl Tripeptide-1 (GHK)a Copper Tripeptide-1 (GHK) Bis(Tripeptide-1) Copper Acetate (GHK)a Manganese Tripeptide-1 (GHK)a Hexapeptide-12 (VGVAPG)a Palmitoyl Hexapeptide-12 (VGVAPG) Myristoyl Hexapeptide-12 (VGVAPG)a Palmitoyl Tetrapeptide-7 (GQPR)
aNot reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report is available from the Executive Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Authors’ Contributions
Wilbur Johnson contributed to conception and design, acquisition, analysis, and interpretation, and drafted the manuscript; Bart Heldreth contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript; Lillian Gill, Wilma Bergfeld, Donald Belsito, Ronald Hill, Curtis Klaassen, Daniel Liebler, James Marks, Ronald Shank, Thomas Slaga, and Paul Snyder contributed to conception and design, analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.