Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of polyethylene glycol (PEG)-150 pentaerythrityl tetrastearate, which functions as a viscosity-increasing agent-aqueous in cosmetic products and is being used at concentrations up to 5%. Given the chemical structure (large molecule), skin penetration is not likely. The available toxicity data and the low ingredient use concentrations suggest that systemic toxicity would not be likely even if percutaneous absorption were to occur. Additionally, the negative human repeated insult patch test data on the undiluted ingredient were deemed sufficient for evaluating skin irritation and sensitization potential. The Panel concluded that PEG-150 pentaerythrityl tetrastearate is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Introduction
This report presents information relevant to evaluating the safety of polyethylene glycol (PEG)-150 pentaerythrityl tetrastearate as used in cosmetics. This ingredient is reported to function as a viscosity-increasing agent-aqueous in cosmetic products. 1
Chemistry
Definition and Structure
PEG-150 pentaerythrityl tetrastearate (CAS No. 130249-48-8) is the tetraester of stearic acid and a PEG ether of pentaerythritol, with an average of 150 equivalents of ethylene oxide, and it conforms to the molecular structure shown in Figure 1.
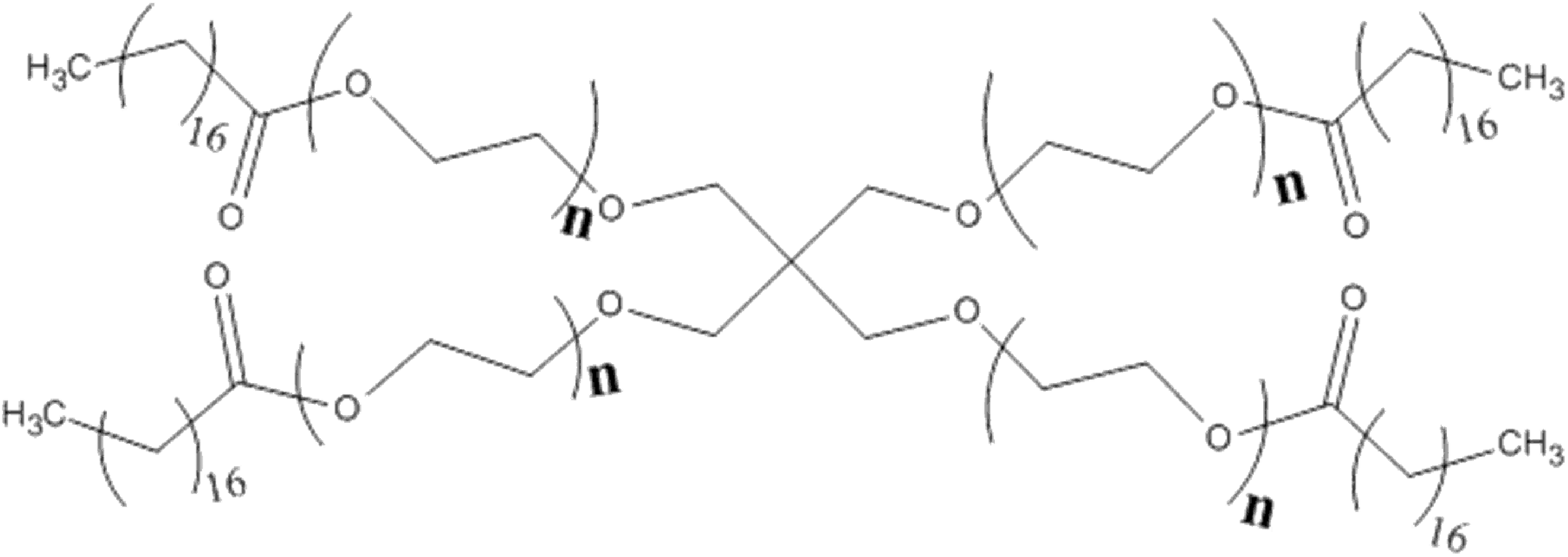
PEG-150 pentaerythrityl tetrastearate (wherein the sum of all instances of n is equal to 150).
Physical and Chemical Properties
PEG-150 pentaerythrityl tetrastearate is slightly soluble in water and has a melting point of 45°C. 2 The specifications for PEG-150 pentaerythrityl tetrastearate trade name materials are included in Table 1. 3
Specifications for PEG-150 Pentaerythrityl Tetrastearate. 3

Abbreviation: NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the total uses; totals = rinse-off + leave-on product uses.
bIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
cIt is possible that these products may be powders, but it is not specified whether or not the reported uses are powders.
Method of Manufacture
PEG-150 pentaerythrityl tetrastearate is produced by reacting pentaerythritol with 150 stoichiometric equivalents of ethylene oxide. 3 The reaction is conducted under controlled conditions to ensure that moisture is very low during the ethylene oxide reaction stage to ensure minimal formation of free PEG. The resulting ethoxylated pentaerythritol is esterified with 4 equivalents of stearic acid to form the tetraester.
Composition/Impurities
Based on the acid value specification of 5.0 mg KOH/g for PEG-150 pentaerythrityl tetrastearate (for 1 tradename mixture) in Table 1, the amount of free stearic acid present would be less than 2.5%. 3 In addition to the values included in Table 1, the specifications for this trade name material include the following: moisture content (≤1%), ethylene oxide (≤1 ppm), and 1,4-dioxane (≤5 ppm).
Use
Cosmetic
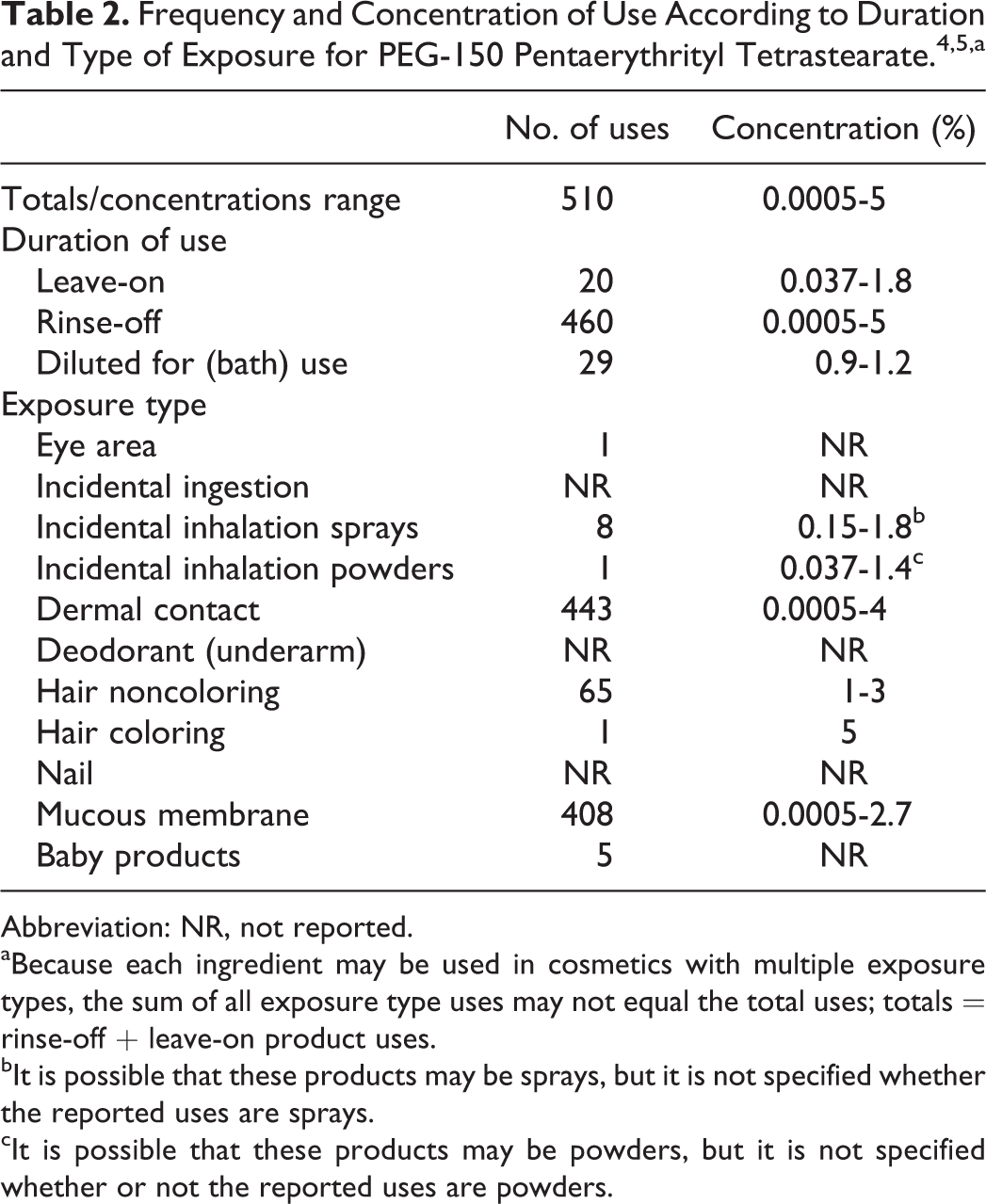
PEG-150 pentaerythrityl tetrastearate is reported to function as a viscosity-increasing agent-aqueous in cosmetic products. 1 Information on the use of this ingredient as a function of product type was supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program. 4 These data indicate that PEG-150 pentaerythrityl tetrastearate is used in rinse-off and in leave-on products, with the majority of uses in rinse-off products. The Personal Care Products Council (Council) conducted a survey of ingredient use concentrations in 2013 to 2014, and a maximum use concentration of 5% (in hair dyes and colors) was reported for PEG-150 pentaerythrityl tetrastearate. 5 The maximum reported use concentrations for rinse-off and leave-on products were 5% (hair dyes and colors) and 1.8% (tonics, dressings, and other hair grooming aids), respectively. Ingredient frequency-of-use and use concentration data are included in Table 2.
Cosmetic products containing PEG-150 pentaerythrityl tetrastearate may be applied to the skin and hair and may come in contact with mucous membranes or, incidentally, these products may come in contact with the eyes. Products containing this ingredient may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
It is possible that PEG-150 pentaerythrityl tetrastearate may be used in products that are sprayed (highest maximum use concentration = 1.8%, in tonics, dressings, and other hair grooming aids) and in face and neck powders (highest maximum use concentration = 1.4%). Because it is possible that this ingredient may be used in products that are sprayed and may be used in powders, it could be incidentally inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump spray. 6 –9 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 6,7
Toxicokinetics
Data on the absorption, distribution, metabolism, and excretion of PEG-150 pentaerythrityl tetrastearate were not found in the published literature, nor were unpublished data provided.
Toxicology
Acute Toxicity
Oral
The acute oral toxicity of a 25% gravimetric [sic; presumed meaning is 25% wt/wt], aqueous suspension of PEG-150 pentaerythrityl tetrastearate was evaluated using 10 Wistar albino rats (5 males, 5 females; 6 to 9 weeks old). 10 Each animal received a single oral dose of 5 g/kg body weight. Dosing was followed by a 14-day observation period, and gross necropsy was performed. None of the animals died, and it was concluded that the test substance did not induce toxicity.
Repeated Dose Toxicity
Data on the repeated dose toxicity of PEG-150 pentaerythrityl tetrastearate were not found in the published literature, nor were unpublished data provided.
Ocular Irritation
The ocular irritation potential of undiluted PEG-150 pentaerythrityl tetrastearate was evaluated using 6 New Zealand white rabbits. 10 The test substance (0.1 mL) was instilled into 1 eye of each animal, and the untreated contralateral eye served as the control. Eyes were not rinsed after test substance administration. Treated eyes were observed for corneal opacity, iritis, and conjunctivitis at 24 hours, 48 hours, and 72 hours postinstillation. The test substance did not induce ocular irritation.
Skin Irritation and Skin Sensitization
Animal
The skin irritation potential of undiluted PEG-150 pentaerythrityl tetrastearate was studied using 6 New Zealand white rabbits (3 months old). 10 The test substance (0.5 mL) was applied to 2 sites (1 abraded, 1 intact) on opposite sides of the vertebral column. Application sites were occluded for 24 hours and then evaluated for erythema, edema, and other effects at 24 and 72 hours postapplication. The test substance did not cause primary skin irritation (primary irritation index = 2.65).
Human
The skin irritation and sensitization potential of undiluted PEG-150 pentaerythrityl tetrastearate was studied using 53 subjects (18-71 years old). 11 A semiocclusive patch (1″ × 1″ gauze patch) containing 0.2 g of the test substance was applied to the upper back, between the scapulae, 3 times per week for a total of ten 24-hour induction applications. Following a 2-week nontreatment period, a 24-hour challenge patch was applied to the original site and to a new site. Reactions were scored at 24 and 48 hours postapplication. There was no evidence of a visible reaction in any of the subjects, and it was concluded that the test substance did not have skin irritation or sensitization potential in this study.
A repeated insult patch test on 25% aqueous (slurry) PEG-150 pentaerythrityl tetrastearate was performed using 52 subjects (19-74 years old). 12 A semiocclusive patch (1″ × 3/4″ absorbent pad) containing 0.2 mL of the test substance was applied to the upper back between the scapulae. Patches were applied 3 times per week for a total of nine 24-hour induction applications. Following a 2-week nontreatment period, a 24-hour challenge patch was applied to a new test site (adjacent to original site). Reactions were scored at 24 and 72 hours postapplication. There was no evidence of a visible reaction in any of the subjects, and it was concluded that the test substance did not have skin irritation or sensitization potential in this study.
In vitro
The skin irritation potential of PEG-150 pentaerythrityl tetrastearate (25% in distilled water) was evaluated using the MatTek Corporation EpiDerm in vitro toxicity testing system. 13 This skin model consists of normal, human-derived epidermal keratinocytes, cultured to form a multilayered, highly differentiated model of the human epidermis. This procedure involves 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl-tetrazolium bromide (MTT), a yellow, water-soluble tetrazolium salt that is reduced by succinate dehydrogenase to a purple formazan derivative in the mitochondria of viable cells. Substances that damage this mitochondrial enzyme inhibit reduction of the tetrazolium salt. Therefore, the amount of MTT reduced by a culture is proportional to the number of viable cells. Data from this assay were presented in the form of a plot (semi-log scale) of percent viabilities versus the dosing times. The estimated time for 50% viability (ET-50) for 25% PEG-150 pentaerythrityl tetrastearate was >24 hours, meaning that the test substance is expected to be nonirritating when dosed in vivo.
In another in vitro test known as the Skintex test, 10% PEG-150 pentaerythrityl tetrastearate was classified as nonirritating. Details relating to the test procedure were not provided; however, the following Skintex in vitro system test procedure is included in another publication. 14 The test substance was applied directly to the barrier membranes in 3 different volumes (30, 50, and 100 µL), and the loaded barrier was inserted into the cuvettes containing the protein reagent matrix. Three calibrators with known in vivo Draize scores and well-characterized assay performance were run in parallel, along with negative and positive controls. Membranes were exposed for 5 hours. Following the 5-hour exposure time, the membranes were removed and the optical density at 470 nm was determined using a colorimeter. The optical density measurement from the 3 calibrators had to fall within a specified range based on previously determined assay performance guidelines. If any of the calibrators fell outside of this range, or if the negative control exhibited reactivity, the assay was unqualified and the procedure repeated. Provided that the assay was qualified, the optical density obtained with the 100-µL dosage level was used in making irritant versus nonirritant determinations. The cutoff between a nonirritant and an irritant in this system corresponds to 820 opacity units (ou). Correlations between in vitro dose–response curves and in vivo dose–response curves were also based on the ou measurement obtained for the 100-µL dose of the test sample.
Reproductive and Developmental Toxicity
Data on the reproductive or developmental toxicity of PEG-150 pentaerythrityl tetrastearate were not found in the published literature, nor were unpublished data provided.
Genotoxicity
In Vitro Assays
The genotoxicity of PEG-150 pentaerythrityl tetrastearate was evaluated in the Ames test using the following bacterial strains, with and without metabolic activation, at doses up to 5,000 µg/plate: Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2uvrA. 15 The following positive controls were used: 2-aminoanthracene, 2-nitrofluorene, sodium azide, 9-aminoacridine, and methyl methanesulfonate. No appreciable toxicity was induced by the test substance, and there was no evidence of genotoxicity, with or without metabolic activation, over the range of doses tested. All positive controls were genotoxic.
In another assay (Ames test), the genotoxicity of PEG-150 pentaerythrityl tetrastearate (1 g in dimethyl sulfoxide (DMSO)) was evaluated with metabolic activation using the following Salmonella typhimurium strains: TA98, TA100, TA1535, TA1537, and TA1538. 16 The following positive controls were used: paradimethylaminobenzene disodium sulfonate, sodium azide, 2 nitrofluorene, and 2-aminofluorene. The test substance was not considered genotoxic, whereas all positive controls exhibited their expected genotoxicities.
Carcinogenicity
Data on the carcinogenicity of PEG-150 pentaerythrityl tetrastearate were not found in the published literature, nor were unpublished data provided.
Summary
PEG-150 pentaerythrityl tetrastearate is reported to function as a viscosity-increasing agent-aqueous in cosmetic products. Frequency-of-use data provided by the FDA indicate that this ingredient is being used in rinse-off and leave-on products, with the majority of uses in rinse-off products. The Council conducted a survey of ingredient use concentrations in 2013 to 2014, and a maximum use concentration of 5% (in hair dyes and colors, rinse-off products) was reported for PEG-150 pentaerythrityl tetrastearate. The maximum reported use concentration for leave-on products was 1.8% (tonics, dressings, and other hair grooming aids).
PEG-150 pentaerythrityl tetrastearate is produced by reacting pentaerythritol with 150 equivalents of ethylene oxide. The reaction is conducted under controlled conditions of very low moisture to ensure minimal formation of free PEGs. The resulting ethoxylated pentaerythritol is esterified with 4 equivalents of stearic acid to form the tetraester. Additionally, the specifications for impurities limit ethylene oxide and 1,4-dioxane to 1 and 5 ppm, respectively.
A 25% wt/wt aqueous suspension of PEG-150 pentaerythrityl tetrastearate (dose = 5 g/kg) was nontoxic in an acute oral toxicity study involving albino rats.
In an ocular irritation study involving rabbits, undiluted PEG-150 pentaerythrityl tetrastearate was classified as a nonirritant. Undiluted PEG-150 pentaerythrityl tetrastearate also did not cause primary skin irritation (abraded or intact skin) in rabbits. Via in vitro tests evaluating the skin irritation potential of PEG-150 pentaerythrityl tetrastearate at concentrations of 25% aqueous and 10%, this ingredient was determined not to be an irritant. Neither skin irritation nor sensitization was observed in human repeated insult patch tests in which subjects were patch-tested with undiluted PEG-150 pentaerythrityl tetrastearate (53 subjects) or a 25% aqueous solution (52 subjects) of this ingredient.
PEG-150 pentaerythrityl tetrastearate was not genotoxic in the Ames test using the following Salmonella typhimurium strains, with or without metabolic activation: TA98, TA100, TA1535, TA1537, and TA1538.
Data on the toxicokinetics, repeated dose toxicity, carcinogenicity, or reproductive and developmental toxicity of PEG-150 pentaerythrityl tetrastearate were not found in the published literature.
Discussion
Use concentration data indicated that the maximum reported use concentrations for PEG-150 pentaerythrityl tetrastearate in rinse-off and leave-on products were 5% and 1.8%, respectively. Because the method of manufacture ensures minimal formation of free PEG and the specifications for impurities limit ethylene oxide and 1,4-dioxane to 1 and 5 ppm, respectively, the Panel agreed that concerns about these impurities in the finished cosmetic product are not warranted. Furthermore, after considering the large size of this molecule, the Panel agreed that percutaneous absorption is not expected. The absence of the potential for percutaneous absorption, the negative genotoxicity data, and data results for skin irritation and sensitization provided the Panel with a sufficient basis for assessing the safety of PEG-150 pentaerythrityl tetrastearate when used as a viscosity-increasing agent in cosmetic products.
It is possible that PEG-150 pentaerythrityl tetrastearate may be used in products that are sprayed (highest maximum use concentration = 1.8%, in tonics, dressings, and other hair grooming aids) and in face and neck powders (highest maximum use concentration = 1.4%). Although use in these types of products has not been confirmed, the Panel discussed the issue of incidental inhalation exposure from propellant, pump sprays, and powders and considered pertinent data indicating that incidental inhalation exposures to this ingredient in such cosmetic products would not cause adverse health effects. The data considered include data characterizing the potential for this ingredient to cause acute oral toxicity and ocular or dermal irritation or sensitization. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that PEG-150 pentaerythrityl tetrastearate is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
W. Johnson contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. B. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. L. J. Gill, W. F. Bergfeld, D. V. Belsito, R. A. Hill, C. D. Klaassen, D. C. Liebler, J. G. Marks, R. C. Shank, T. J. Slaga, and P. W. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.