Abstract
Oxime reactivators are critical antidotes after organophosphate pesticide or nerve agent poisoning, directly restoring the function of inhibited acetylcholinesterase. In the continuing search for more broad-spectrum acetylcholinesterase reactivators, this study evaluated one of the leading next-generation oxime reactivators: methoxime, (1,1′-trimethylene bis[4-(hydroxyimino)methyl]pyridinium dichloride (MMB-4). The pharmacokinetics of both salts of MMB-4 (dichloride [2Cl] and dimethanesulphonate [DMS]) were characterized across a range of relevant doses (19, 58, and 116 µmol/kg, intramuscular) in a nonhuman primate model (male African green monkeys), and only subtle differences were observed between the salts. Additionally, the behavioral and cardiovascular safety of these MMB-4 salts was compared directly to other available oximes (HI-6 2Cl, HI-6 DMS, and pyridine-2-aldoxime chloride (2-PAM Cl)) at comparable projected doses. Automated operant behavioral tests were used to examine attention, motivation, visual discrimination, concept execution, and fine motor coordination after high doses of all oxime salts, and of all oximes studied, only the highest dose of 2-PAM Cl (447 µmol/kg) disrupted behavioral performance. Likewise, the effects of a range of doses of MMB-4 2Cl or DMS, HI-6 2Cl or DMS, or 2-PAM Cl on cardiovascular parameters were measured in African green monkeys implanted with telemetry devices. Only a small transient decrease in pulse pressure was observed following administration of the highest dose of MMB-4 DMS (116 µmol/kg). Thus, MMB-4 salts, up to the 9× equivalent of a projected autoinjector dose in humans, did not produce behavioral or cardiovascular toxicity in African green monkeys in the current study, and the pharmacokinetic parameters were orderly and predictable.
Keywords
Introduction
Oximes are a critical component of the standard therapy following organophosphorus (OP) nerve agent exposure and are given in conjunction with atropine and a benzodiazepine anticonvulsant (such as diazepam or midazolam). Organophosphorus nerve agents and pesticides inhibit acetylcholinesterase (AChE) by phosphorylating the enzyme at a serine residue in the active site, thereby preventing substrate binding and resulting in an excess of acetylcholine at peripheral and central sites. Atropine directly antagonizes the excess acetylcholine at muscarinic receptors, while anticonvulsants attenuate seizures initiated by the overstimulation of central cholinergic synapses. Oximes are arguably the only true antidote to OP exposure. Oximes reactivate AChE through nucleophilic attack on the phosphorus of the OP-inhibited enzyme, freeing the active site and allowing the enzyme to once again hydrolyze ACh.
Unfortunately, no single oxime can reactivate AChE across all OPs; instead, the efficacy of each oxime varies greatly from 1 OP to the next. The US Army currently fields the Antidote Treatment-Nerve Agent Auto-Injector (ATNAA) containing the monopyridinium oxime 2-PAM (pralidoxime, pyridine-2-aldoxime) and atropine. 2-PAM offers little to no reactivation against the OP nerve agents tabun, cyclosarin, or soman. 1,2 The desire to have a broader spectrum reactivator has led the US Army to investigate the bispyridinium oxime methoxime, 1,1′-trimethylene bis[4-(hydroxyimino)methyl]pyridinium dichloride (MMB-4). The MMB-4 offers some advantages in comparison to 2-PAM including greater efficacy against cyclosarin and the Rissuan V agent, VR. 3 –6
Due to obvious ethical reasons, experiments on the efficacy of oximes against OP intoxication cannot be conducted in humans. Therefore, animal models are used with the caveat of potential species differences in OP-inhibited AChE reactivation by oximes. 7 –11 The nonhuman primate is often used as a surrogate for man and typically demonstrates similar oxime reactivation of AChE to that seen in humans. 12 Thus, the present study utilized African green monkeys to investigate the pharmacokinetics and safety of MMB-4. For comparison, 2 other oximes that are currently fielded, 2-PAM and HI-6, were included in the cardiovascular and behavioral safety studies.
Materials and Methods
Subjects
Fifteen young adult male African green monkeys (Chlorocebus aethiops sabeus) of Caribbean origin were used. The average animal weight was 6.5 kg. The colony was maintained on a 12-hour light–dark cycle (lights on at 0600) with an average temperature of 21°C ± 2°C and relative humidity of 50% ± 20%. The animals were maintained at a healthy weight by allotment of daily rations (Certified Primate Diet 5048; Purina Mills Inc., St Louis, MO) and fresh fruits and vegetables. The controlled allotment of food ensured the animals would be properly motivated for food-reinforced tasks. Prior to drug administration and experimentation, the animals were trained extensively in relevant tasks. Animals participating in the pharmacokinetic study were placed individually in primate collar-restraint chairs (Primate Products Inc, Immokalee, Florida) and trained to cooperate during blood sampling and intramuscular (im) injections. Likewise, animals participating in the cardiovascular study were trained to cooperate in the home cage when pulled forward for injections. Behavioral study animals were extensively trained to receive im injections (saline, 1 mL) in the home cage at least twice weekly just minutes prior to daily behavioral testing described below. The experimental protocol was approved by the Animal Care and Use Committee at the United States Army Medical Research Institute of Chemical Defense (USAMRICD), and all procedures were conducted in accordance with the principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, National Academy Press, 1996, and the Animal Welfare Act of 1966, as amended. The USAMRICD is a research facility fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Drugs
Test compounds (≥95% purity) were synthesized at the Walter Reed Army Institute of Research and included MMB-4 2Cl (MW = 329.19), MMB-4 DMS (MW = 448.465), 2-PAM Cl (MW = 172.61), HI-6 2Cl monohydrate (1-[[[4-(aminocarbonyl) pyridinio] methoxy] methyl]-2-[(hydroxyimino)methyl]-pyridinium dichloride; MW = 377.22), and HI-6 DMS (MW = 448.47). The doses of oximes in this study were chosen based on the currently fielded treatments. The currently fielded treatment for nerve agent exposure in the US Army, the ATNAA, contains 2.1 mg atropine and 600 mg 2-PAM. The US Army treatment doctrine specifies that 3 autoinjectors of 2-PAM Cl (approximately 149 µmol/kg based on a 70 kg human) may be delivered immediately upon signs of nerve agent intoxication. By comparison, the Swedish military has fielded an autoinjector containing 500 mg of HI-6 dichloride monohydrate; 3 such autoinjectors deliver 58 µmol/kg to a 70 kg human. It is projected that a comparable dose of MMB-4 would be used (i.e., 58 µmol/kg). In the pharmacokinetic study, doses of 19, 58, and 116 µmol/kg (the equivalent of 1, 3, and 6 auto-injectors, respectively) were tested. The second experiment, with its emphasis on establishing the behavioral and cardiovascular safety or toxicity of these oximes, included higher than doctrinal doses for HI-6 and MMB-4: 116 µmol/kg (equivalent to 6 auto-injectors) and 174 µmol/kg (equivalent to 9 auto-injectors). For 2-PAM Cl, autoinjector-equivalent doses of 6 (298 µmol/kg; 6×) and 9 (447 µmol/kg; 9×) were similarly tested for safety. Applying the Food and Drug Administration’s allometric scaling formula 13 incorporating body surface area conversions from humans to animals, these doses would be approximately 46% of the human doses stated above (eg, the 9× dose would be more comparable to a 4.14× dose). Nevertheless, these doses are still higher than those typically reported in previous work with these same oximes and are, in many cases, higher than projected human doses.
The test compounds were injected intramuscularly into the lateral thigh muscle in a constant volume of 1 mL. For experiment 1 (pharmacokinetics study), a minimum of 14 days separated each injection. Both experiment 2 (behavioral study) and experiment 3 (cardiovascular study) used a within-participants design, with each participant receiving each dose of each drug across several weeks. A minimum of 10 days separated each drug injection. Vehicle control injections (isotonic saline, 1 mL) were administered on days between test compounds to serve as a basis from which to gauge the safety/toxicity of the test compounds.
Experiment 1: Pharmacokinetic Characterization of 2 MMB-4 Salts
Blood collection and sample preparation
For the computation of pharmacokinetic parameters, 8 to 9 monkeys were used for each dose of each salt. Blood samples were taken at 0 minutes (preinjection baseline), 5, 10, 20, 30, 40, 80, 160, and 320 minutes following im injection of the test compound. Whole blood (∼0.8 mL) was collected from the saphenous vein into a 1-mL syringe (coated with 1,000 units/mL heparin), the needle was removed, and the blood sample was transferred into a 2-mL microcentrifuge tube and quickly processed in a microcentrifuge at 10 k revolutions per minute for 3 minutes. The supernatant (ie, plasma) was removed via transfer pipette and placed into a 1-mL tube and flash frozen using a bath of dry ice and ethanol. The frozen plasma samples were stored −80°C until analyzed.
For analysis, frozen plasma samples were thawed on ice. When completely thawed, the samples were clarified by centrifugation in a microcentrifuge at 4°C for 5 minutes. In cases where <100 μL of plasma was collected or oxime levels were expected to be beyond the standard matrix-matched control (MMC; see below) values (3× and 6× autoinjector equivalents), samples were diluted with 1× phosphate-buffered saline. The clarified plasma was acidified with phosphoric acid to 3% and centrifuged again as described above. The supernatant was then processed by ultrafiltration through Vivaspin 500 devices with either 5 k or 10 k molecular weight cut-off, per manufacturer’s directions (Vivascience, AG, Germany). The centrifugation was performed at 4°C. The filtrate was collected for oxime analysis by high-performance liquid chromatography (HPLC).
HPLC
MMB-4 plasma levels were determined by HPLC using a weak-cation exchange chromatography column, PolyCAT A (2.1 × 100 mm, 5 μm, 300× [PolyLC Inc, Columbia, MD]), at 25°C. The samples were placed in a temperature-controlled autosampler maintained at 6°C. After injection, MMB-4 was eluted from the column utilizing isocratic elution at a flow rate of 0.3 mL/min and a run time of 5 to 6 minutes. The HPLC buffer consisted of 0.15 mol/L ammonium acetate pH 4.5 in water and 44% (vol) acetonitrile. The sample was monitored at 300 nm.
Sample MMB-4 plasma concentrations were determined from comparison to peak area standard curves of MMB-4-spiked MMC plasma. Matrix-matched controls were prepared by spiking unadulterated control AGM plasma (Bioreclamation, Inc, Hicksville, New York) with MMB-4. The MMC concentration range was 0.3, 0.03, 0.003, and 0.0003 μmol·mL−1. The spiked MMCs were processed similar to the samples. 14
Experiment 2: Cognitive-Behavioral Safety Testing of Select Oximes
Apparatus
Each monkey had a dedicated aluminum panel that could be affixed to the front of the home cage during daily behavioral testing. Each panel consisted of a touchscreen monitor (15-inch resistive flat panel LCD; Omnivision, LLC; Roselle, IL), a pellet dispenser that delivered a 190 mg Bio-Serv (Bio-Serv, New Brunswick, New Jersey) banana-flavored precision pellet (product #F0035) as a reinforcer for each correct response, a food cup into which pellets were delivered, and a notebook computer (Dell Latitude C640 running Windows 98SE), which controlled experimental events and collected data via a custom-written Microsoft VisualBasic 6.0 (Redmond, Washington) software program. During testing, the animal’s home cage door was raised to allow unhindered access to the touch screen. All computerized events and data collection were accomplished with 0.01-second precision.
Trials
A novel vigilance and speed discrimination test was used for all behavioral safety testing. Each daily session consisted of 240 consecutive trials separated by a variable intertrial interval of 3 to 18 seconds, during which the screen was blank and responses were without effect. Each trial began with the presentation of initial-link stimulus, a large white circle in the center of the screen. The latency to press the initial-link stimulus (ie, trial-initiation latency) was recorded for each trial to index vigilance and motivation. Touching the initial-link stimulus or waiting 20 seconds initiated the choice phase of the trial. During the choice phase, 2 targets (gray squares of equal size) moved about the screen, one vertically and one horizontally. The starting positions of the targets were determined randomly. There were 3 basic trial types, each differing in the speed of the slower target: 2, 5, or 8 cm/s. The faster target always moved at a speed of 10 cm/s. Thus, the 3 trial types were designed to provide easy (10 vs 2; 80% relative speed disparity), moderate (10 vs 5; 50% relative speed disparity), and difficult (10 vs 8; 20% relative speed disparity) speed discriminations within each session. The directions of movement and the trial types were selected pseudorandomly so that an equal number of trials of each type were presented within each session. On each trial, the screen background color was either red or blue, indicating whether the faster or slower target was correct. Thus, the task required not only speed discrimination but also color discrimination to correctly master the faster/slower concept and execute the test with high levels of accuracy. The color–speed relationship was counterbalanced across animals (ie, for half of the participants, red signaled that choosing the faster target was correct; for the other half, blue signaled that the faster target was correct). Each choice phase lasted 60 seconds or until a choice response was made, whichever came first. Sessions usually lasted about an hour but were ended after a maximum of 2 hours. Only data from the first 90 minutes were included for data analysis. Monkeys completed training within 6 weeks and were stabilized on the task for over 1 month of daily sessions (Monday-Friday) at approximately the same time each day. Behavioral sessions began exactly 4 minutes after drug or vehicle injection. The 2 highest doses (6× and 9×) of all 5 oxime salts were evaluated. A within-participants design was used with each participant receiving each dose of each drug. For each dose of each drug, the 2 adjacent vehicle days (one before and one after administration of the test compound) were combined for evaluation.
Experiment 3: Cardiovascular Assessment of Oximes
Approximately 2 months following the completion of experiment 1, 6 of the 9 animals that participated in experiment 1 served as participants in this experiment. The reuse of nonhuman primates is common, and the several weeks given were more than sufficient for drug washout.
Surgery
All surgical procedures were performed using sterile technique in a dedicated surgery suite. Animals were administered ketamine (10 mg/kg, im) for ease of handling and surgical preparation. The animal was intubated and maintained on a surgical plane of anesthesia with isoflurane (3% induction; 1.5%-1.0% maintenance). A small subcutaneous pouch was dissected near the left oblique muscle to accept the transmitter (TL11M2-D70-PCT; Data Sciences International [DSI], St Paul, Minnesota). The catheter for femoral artery cannulation was tunneled subcutaneously to exit at the inner thigh incision. To emulate lead II electrocardiogram (ECG), one electrode was placed in the upper right quadrant of the chest and the other in the lower left quadrant of the chest. The upper right electrode (negative) was placed just below the pectoral muscles against the rib cage near the first interspace, and the lower left electrode (positive) was placed in an interspace on the left caudal lateral thorax beneath the abdominal muscles.
For the femoral artery catheterization, an incision was made over the femoral artery in the left, inner thigh area. The artery was first isolated from the surrounding tissues via blunt dissection, and a 60-cm length of 2-0 silk suture was passed under the freed section of artery. The suture loops were drawn to opposite ends of the section of freed artery and retracted sufficiently to lift the vessel from the surface and prevent blood flow through the freed section. A 4-cm hypodermic needle bent at a 45o angle was used to puncture the upper surface of the freed section of artery. Without withdrawing the needle, the catheter tip was placed into the bevel groove of the needle and advanced through the vessel wall while retracting lightly on the needle at an angle perpendicular to the bent end. The catheter was advanced into the vessel about 1 cm or until stopped by the downstream suture. The needle was withdrawn, leaving the catheter in place within the vessel.
All incisions were closed with 3-0 Vicryl and further treated with VetBond skin adhesive following suturing. Following suturing, the animal was administered penicillin G benzathine (40,000 units/kg, im). Once recovery from anesthesia began and the animal regained consciousness, an analgesic (buprenorphine, 0.02 mg/kg, im) was given for immediate postsurgical pain. The animal was returned promptly to the home cage and monitored periodically until determined to be adequately recovered from anesthesia.
Telemetry
The home cages were fitted with 2 PhysioTel RPC-1 receivers (DSI). The continuously recorded ECG data from experiment 3 were parsed into 2-minute segments collected every 15 minutes for 320 minutes (5 hour 20 minutes) following MMB-4 or saline administration to correspond with the time frame used in the pharmacokinetics study (experiment 1). The data were analyzed using Ponemah Physiology Platform v5.2 (DSI). The automated markings were visually confirmed and segments with artifacts removed. Occasionally, a 30 to 100 Hz low-pass filter was applied to reduce noise.
Experiment 4: Hematology and Blood Chemistry Following Repeated Oxime Administration
Just prior to the start of experiment 2 and again at its conclusion, blood samples were taken from all 6 African green monkeys during routine physical examination following sedation with ketamine (10 mg/kg, im). Each animal received multiple doses of MMB-4 2Cl, MMB-4 DMS, HI-6 2Cl, HI-6 DMS, and 2-PAM Cl. By comparing hematology and blood chemistry values before and after an extensive regimen of repeated high-dose exposure to several oximes, the safety or toxicity of this class of compounds was assessed in this nonhuman primate species.
Statistical Analysis
Experiment 1
Mean plasma concentration–time data were fit to standard pharmacokinetic models using PKSolver 2.0. 15 The data were best described by a 1-compartment model with first-order absorption and elimination. Estimated parameters were compared between salts using a 2-factor analysis of variance (ANOVA). Comparisons of values within salts were made using a Bonferroni correction for multiple comparisons.
Experiment 2
A within-participants design was used with each participant receiving each dose of each drug. For each dose of each drug, the 2 adjacent vehicle days (one before and one after administration of the test compound) were combined for evaluation. Because assumptions of normality could not be met consistently, a Wilcoxon rank-sum test (Axum 6.0; MathSoft, Inc; Cambridge, MA) was used to compare vehicle performance to performance following drug administration for each participant on 3 dependent measures: overall accuracy, choice reaction time, and trial-initiation latency.
Experiment 3
The continuously recorded data were parsed into 2-minute segments collected every 15 minutes. The derived parameters studied included diastolic, systolic, and mean blood pressures; PR, RR, QT, and QR intervals; QRS duration; and heart rate. A participant-specific correction using linear model analysis of covariance based on the methods of Spence at al 16 was applied to QT intervals. The means from the segment were analyzed using a 2-factor repeated-measures ANOVA (Prism version 5.04, GraphPad Software, Inc; La Jolla, California).
Experiment 4
Hematological and blood chemistry data were analyzed using a 2-tailed paired t test comparing baseline values obtained prior to administration of oximes and at the conclusion of the experiment when each animal had been exposed to all oximes.
Statistical significance is stated as P < 0.05 for all tests.
Results
Experiment 1: Pharmacokinetic Characterization of MMB-4 Salts
The pharmacokinetic data were best described by a 1-compartment model with first-order absorption and elimination. The mean plasma concentrations across the 5 hours and 20 minutes of sampling for all 3 doses of MMB-4 DMS and 2Cl are presented in Figure 1.
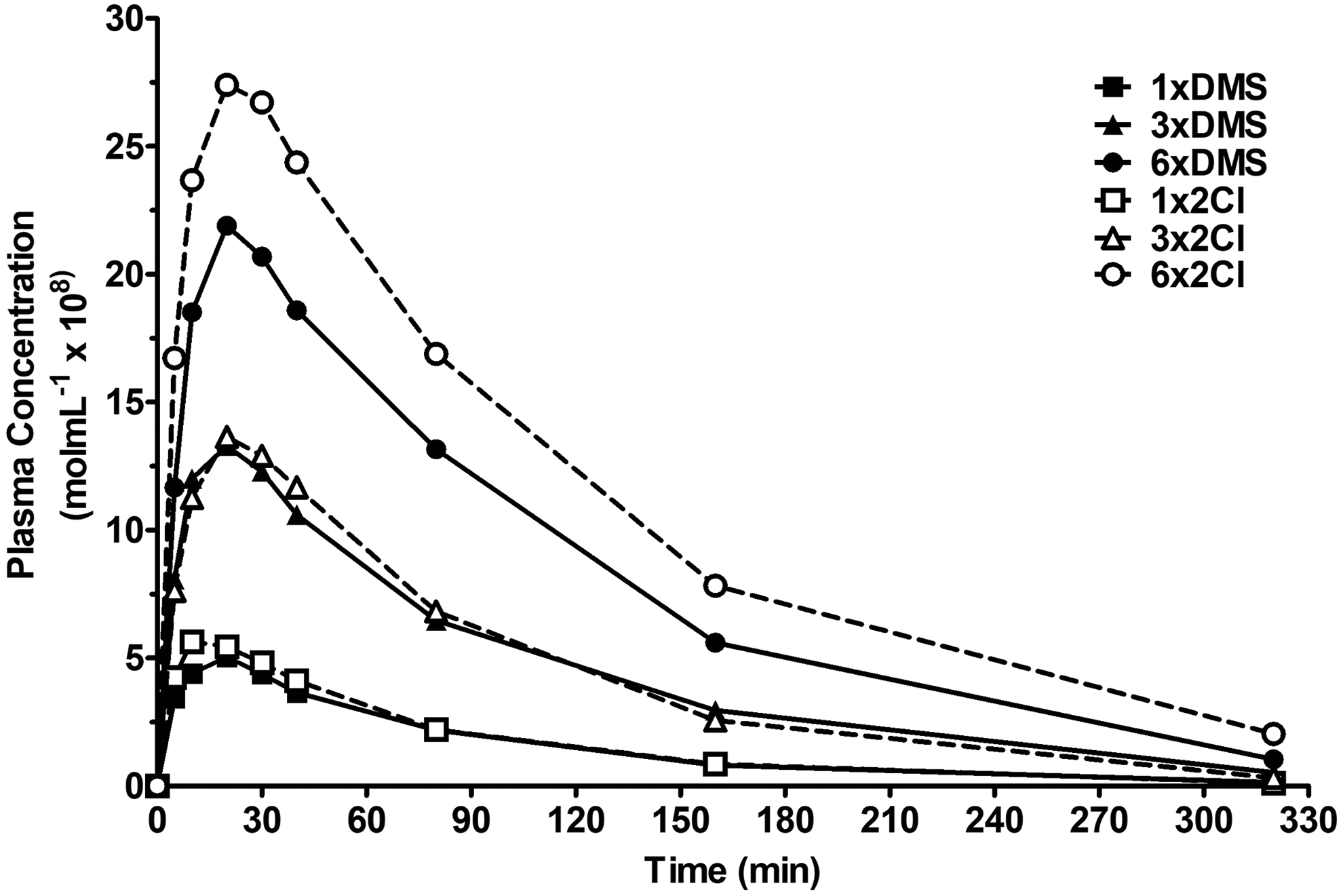
Pharmacokinetics of methoxime, (1,1′-trimethylene bis[4-(hydroxyimino)methyl]pyridinium dichloride (MMB-4) dimethanesulphonate (DMS; A) and 2Cl (B) salts (19, 58, and 116 µmol/kg) intramuscularly administered to African green monkeys are shown. Observed plasma concentration data (square, triangle, and circle) are expressed as free base (n = 7-9, ± standard error of mean [SEM]).
Pharmacokinetic estimates were determined for each animal, and the group means are shown in Table 1 for the 19, 58, and 116 µmol/kg doses for both salts. No statistically significant differences between salts were observed for any of the pharmacokinetic estimates. Rapid absorption was observed for both salts, with measurable concentrations occurring 5 minutes after im administration. Maximal plasma concentrations (Tmax) approximated 20 (13.81-23.44) minutes for all 3 doses of both salts. Variations in dose affected the apparent volume of distribution (Dose F2, 42 = 11.37, P < 0.001), which also showed a significant dose–salt interaction (F2,42 = 4.28, P < 0.05), making interpretation difficult. As expected, Cmax and AUC∞ increased dose dependently (Cmax F2,42 = 134.62, P < 0.001; AUC∞ F2,42 = 84.52, P < 0.001). Post hoc analysis demonstrated increasing values for both Cmax and AUC∞ with increasing dose (19 < 58 < 116 µmol/kg, P < 0.001 at each point for each salt). Despite the appearance of differences in Cmax for the 58 and 116 doses µmol/kg in the concentration versus time profile (Figure 1), there were no significant differences between MMB-4 salts at any of the doses. Although there was a significant main effect of dose on elimination half-life (F2,42 = 3.42, P < 0.05), there were no significant differences across doses within either of the salts.
Pharmacokinetic Parameter Estimates Determined for Both MMB-4 Dichloride (2Cl) and Dimethanesulphonate (DMS) salts (19, 58, and 116 µmol/kg) Following Intramuscular Administration To African Green Monkeys (n = 8-9).a
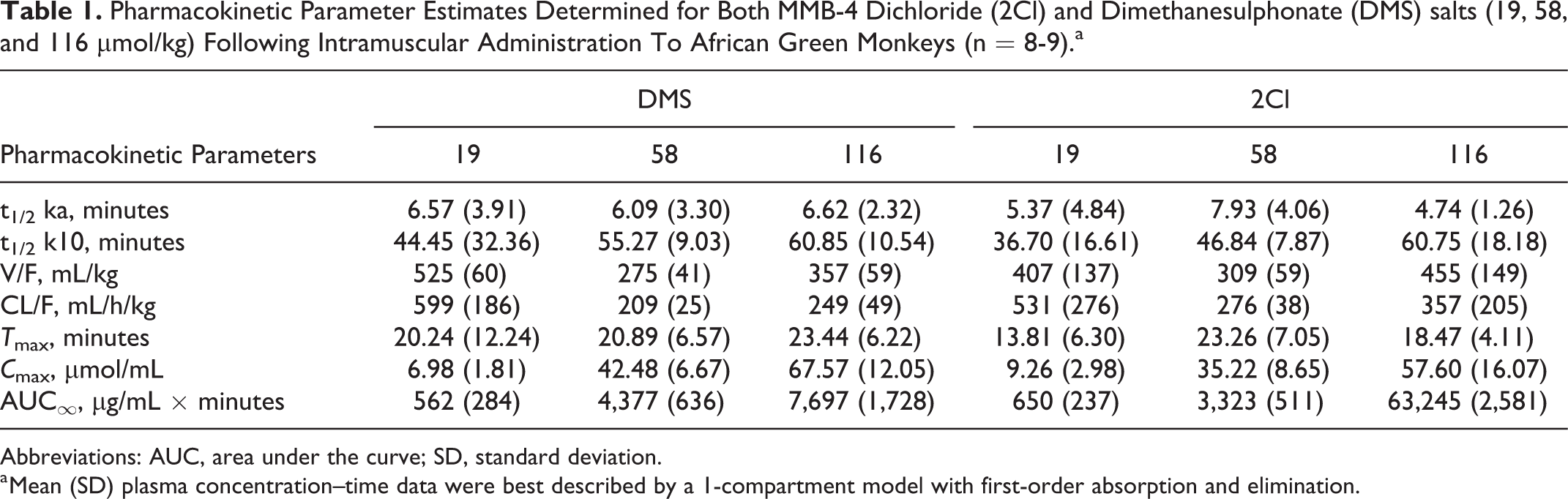
Abbreviations: AUC, area under the curve; SD, standard deviation.
a Mean (SD) plasma concentration–time data were best described by a 1-compartment model with first-order absorption and elimination.
Experiment 2: Cognitive–Behavioral Safety Testing of Select Oximes
In general, all doses of all oximes were well tolerated and produced little to no behavioral impairment. Table 2 shows the results of the statistical tests for each performance measure under the 2 highest doses of each oxime. Only the highest dose (447 µmol/kg) of 2-PAM Cl disrupted performance. Accuracy was decreased significantly, and choice reaction time and trial-initiation latency were increased significantly (Figure 2), suggesting a generalized decrease in motivation at that dose of 2-PAM Cl.
Results of Wilcoxon Rank Sum Tests (P Values) for Accuracy, Choice Reaction Time, and Trial-Initiation Latency Following the Administration of Both Salts of MMB-4 and HI-6 (116 and 174 µmol/kg), and 2-PAM (298 and 447 µmol/kg) Compared to Values Obtained After Injection of Vehicle.
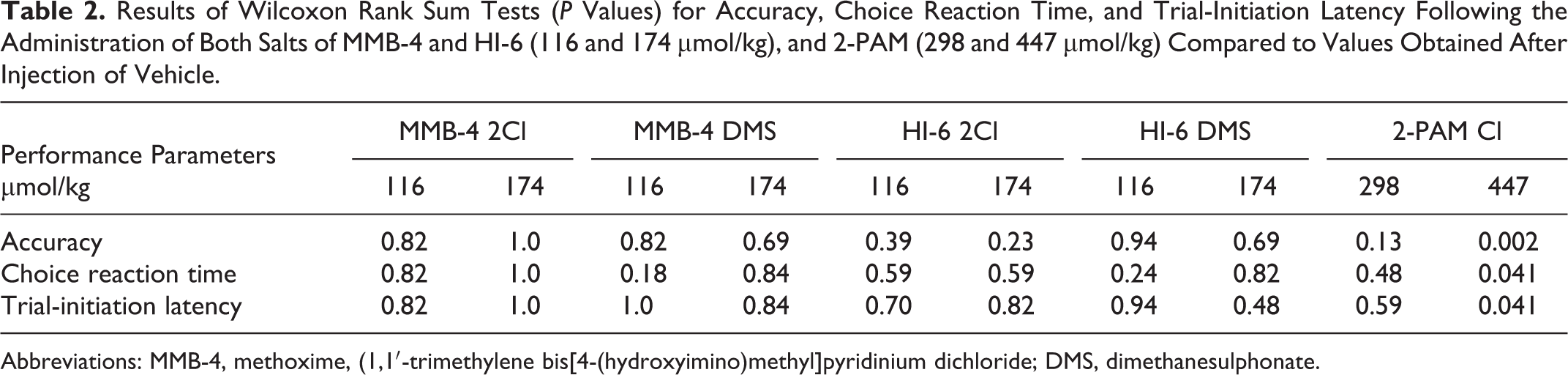
Abbreviations: MMB-4, methoxime, (1,1′-trimethylene bis[4-(hydroxyimino)methyl]pyridinium dichloride; DMS, dimethanesulphonate.
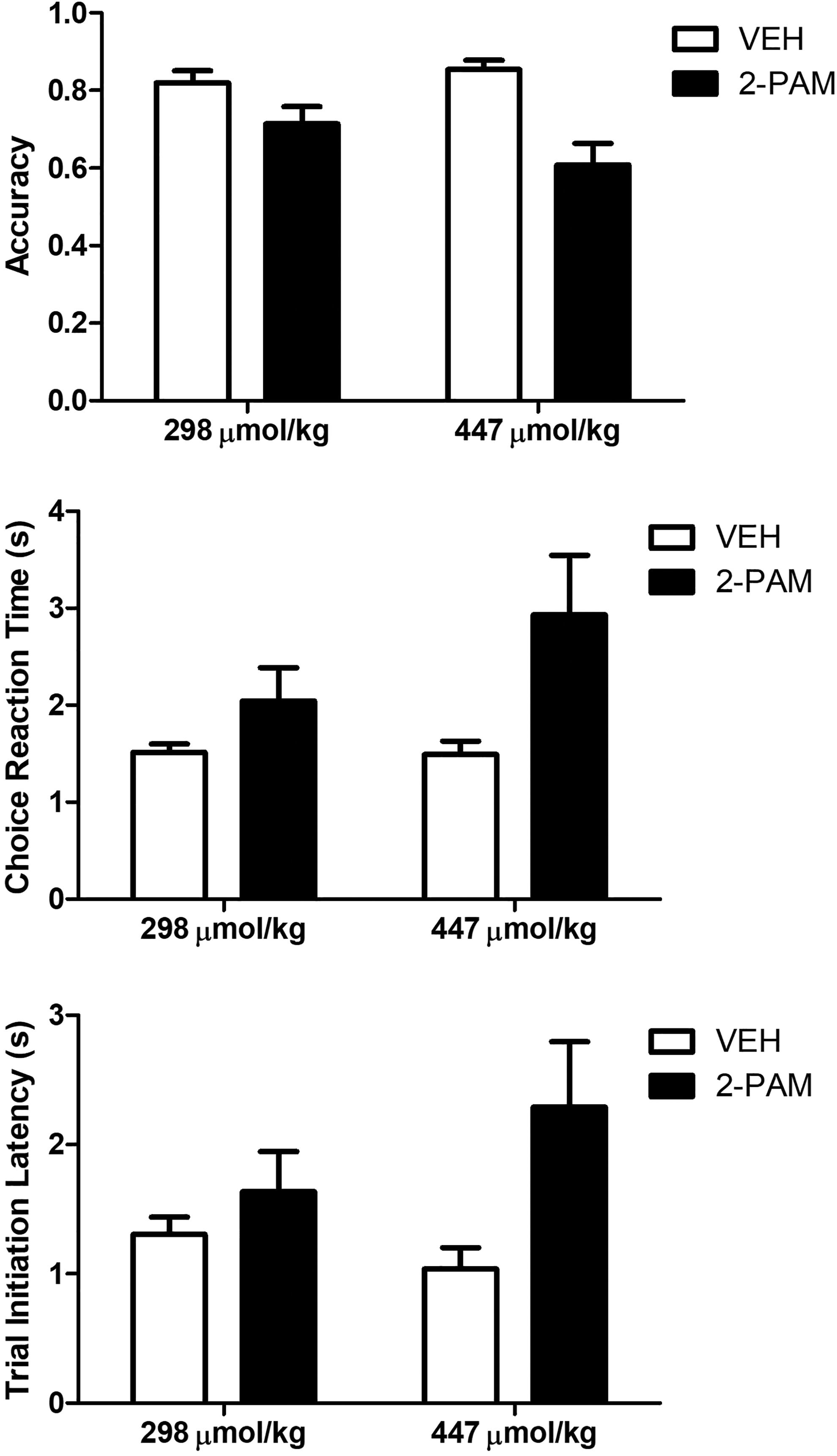
Relative to vehicle, 447 µmol/kg of 2-PAM Cl significantly reduced accuracy and increased choice reaction time and trial-initiation latency. The data are presented as the mean and standard error of mean [SEM]. **P < 0.001, *P < 0.05.
Experiment 3: Cardiovascular Assessment of Oximes
The majority of cardiovascular measures showed a significant effect of time, as the handling and injection led to elevated heart rate and blood pressures during the first 15 minutes of recording. These initial handling-induced elevations in the derived parameters decreased over time, resulting in a significant effect of time (P < 0.01-0.0001) in most measures.
Methoxime, (1,1′-trimethylene bis[4-(hydroxyimino)methyl]pyridinium dichloride
The MMB-4 DMS produced a slight decrease in pulse pressure (F1,7 = 9.41; P < 0.05) compared to values following saline injection (Figure 3). After the normal elevation in pressure produced by handling and injection, systolic and diastolic pressures appear to drop slightly more (although not statistically different) following MMB-4 DMS injection. The slight reduction in systolic pressure is reflected in the narrowing of pulse pressure. A similar, although transient and not statistically significant (P = 0.0866), decrease in pulse pressure is also seen following injection of MMB-4 2Cl.
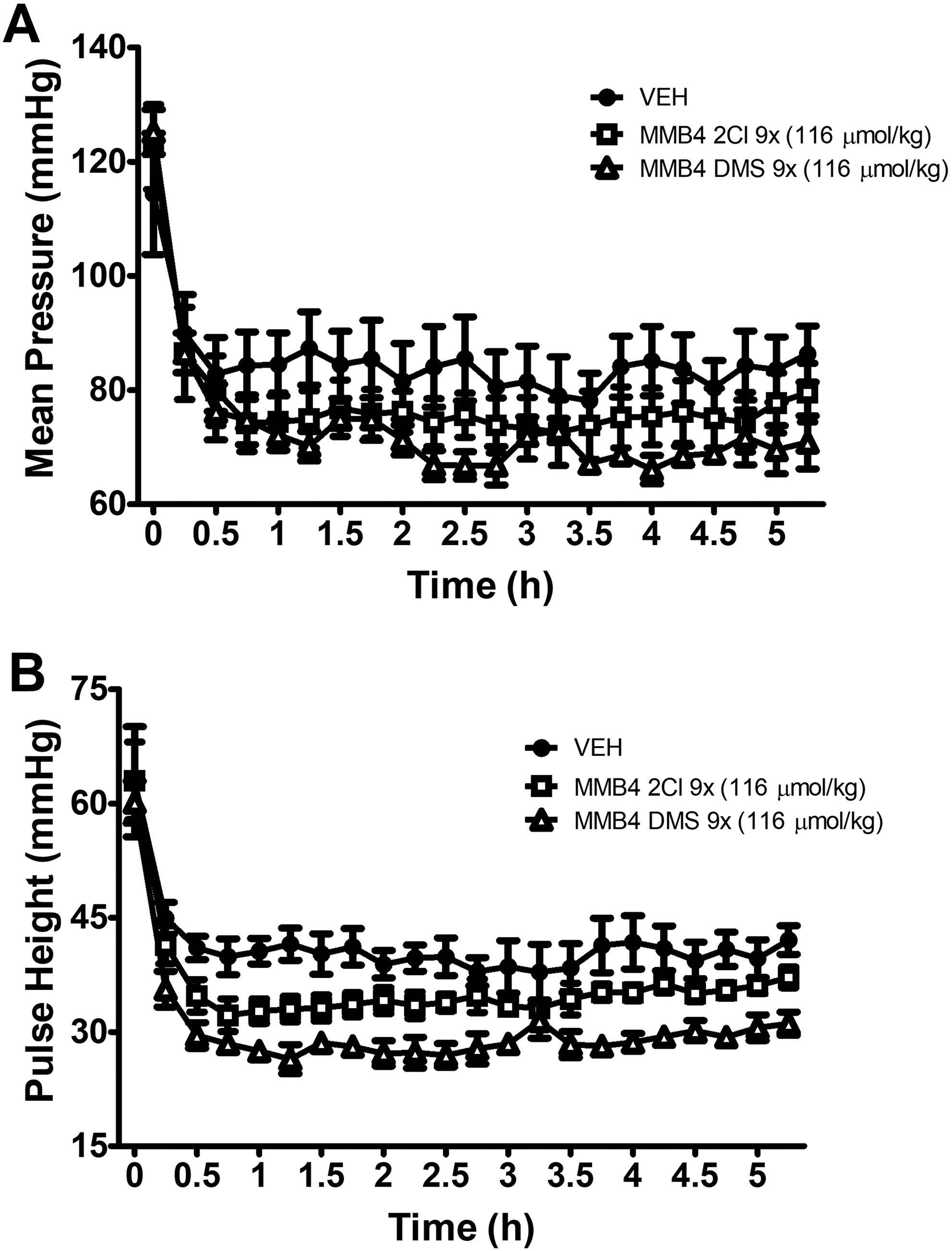
Mean arterial pressure (A) and pulse height (B) following MMB-4 dimethanesulphonate (DMS; 116 µmol/kg) and 2Cl (116 µmol/kg) administration. Minimal effects on blood pressure or pulse height were observed following administration of either salt of MMB-4. The data are presented as the mean ± standard error of mean [SEM].
Significant time–drug interactions were only observed for the DMS salt and include the PR interval as well as the systolic, diastolic, and mean arterial pressures. MMB-4 2Cl had no significant effect on any of the derived parameters. A significant effect of time across parameters (with the exception of the QTc interval) was also observed (Figure 4). The effect of time on QTc was different between vehicle and MMB-4 2Cl (drug × time, P < 0.01). Mean vehicle QTc intervals increased slightly over time, whereas MMB-4 2Cl QTc intervals remained steady for the duration of the postinjection evaluation period.
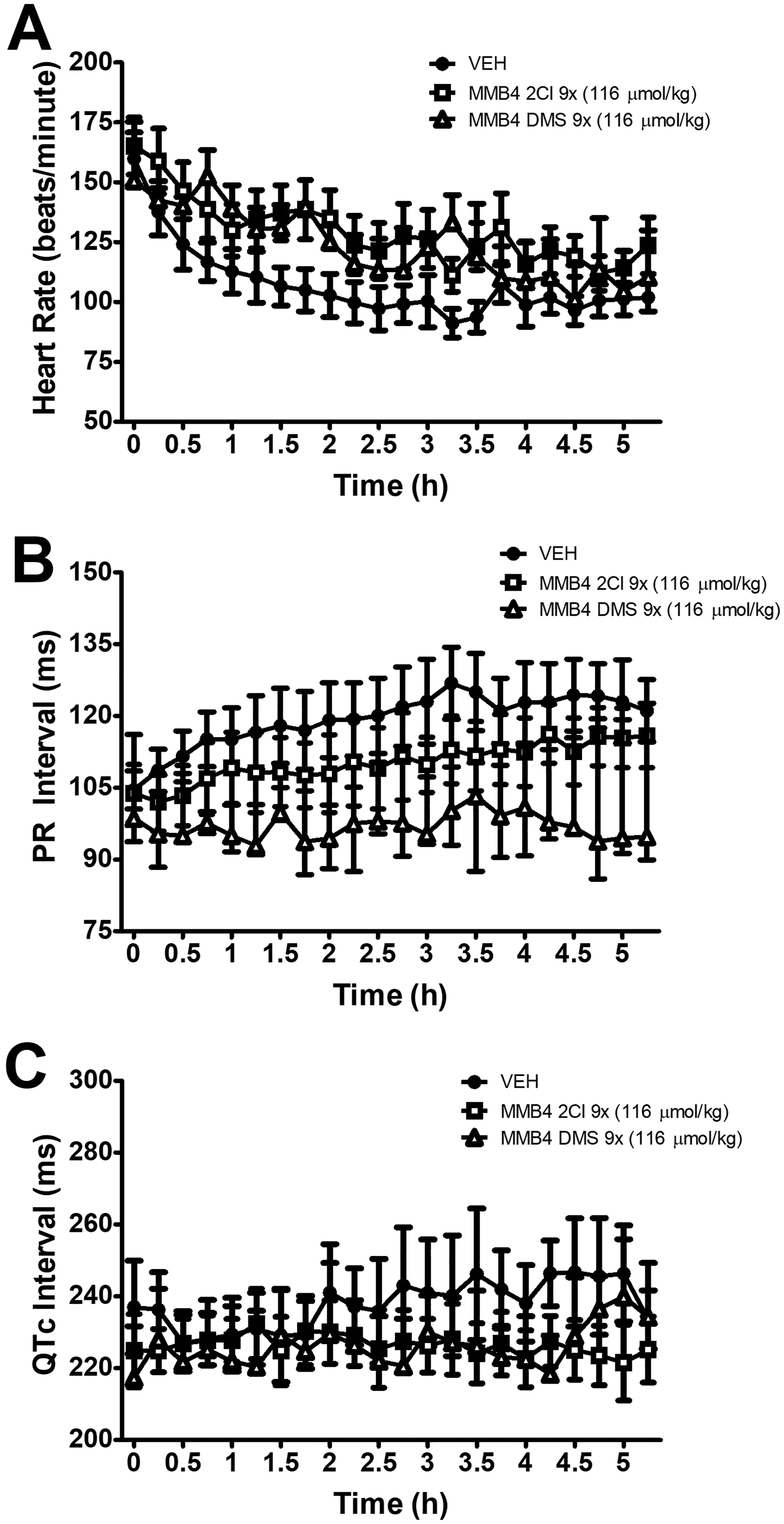
Neither salt of MMB-4 had a significant effect on heart rate (A), PR interval (B), or QTc (C). The data are presented as the mean ± standard error of mean [SEM].
HI-6
Neither salt showed a significant effect of drug on any measured parameter over the 5-hour recording (Figures 5 and 6). A significant time–drug interaction (P < 0.01) was observed with the PR interval lengthening over time following administration of HI-6 DMS but not HI-6 2Cl.
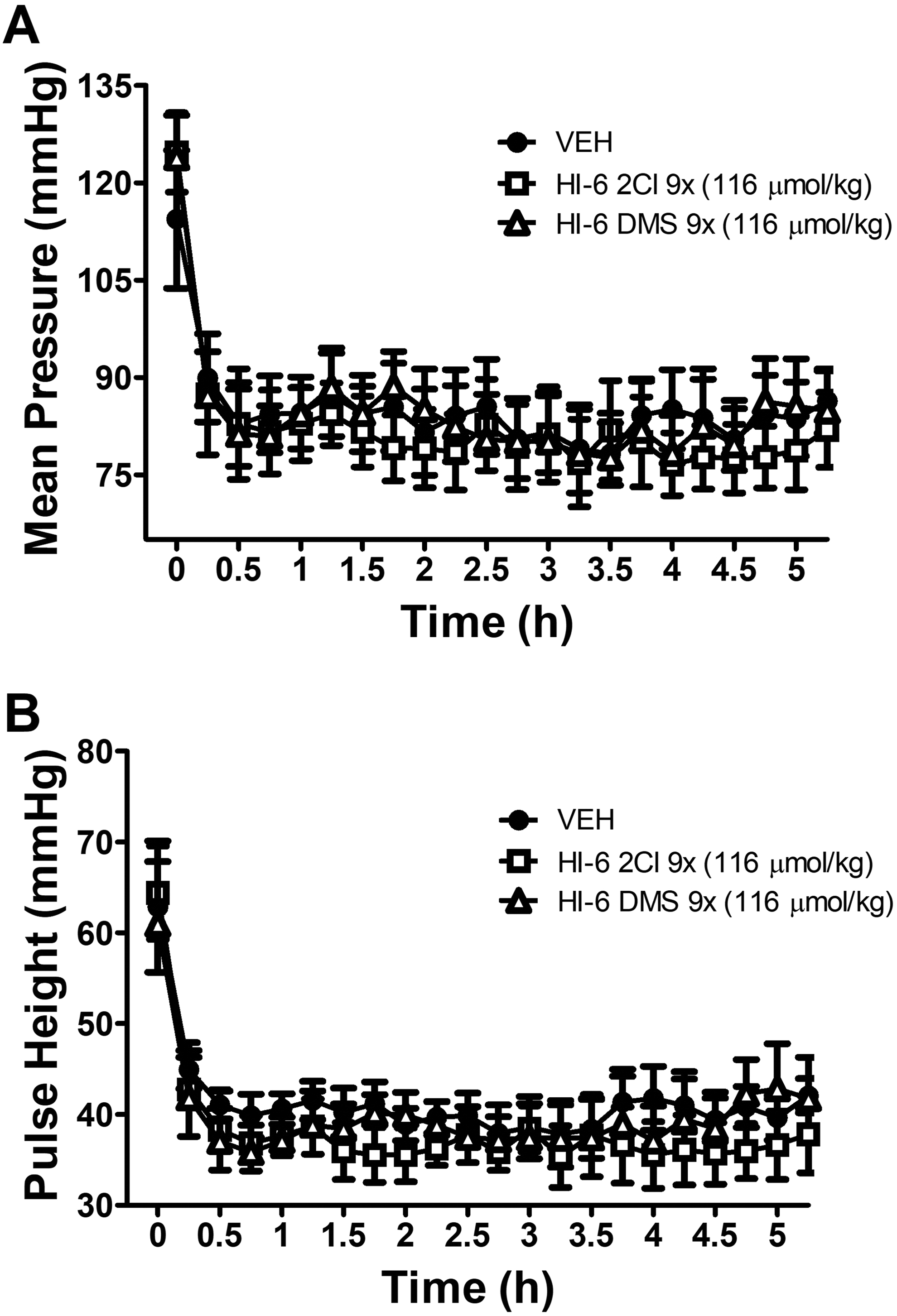
Lack of effect of either salt of HI-6 (116 µmol/kg) on mean arterial pressure (A) or pulse height (B). The data are presented as the mean ± standard error of mean [SEM].
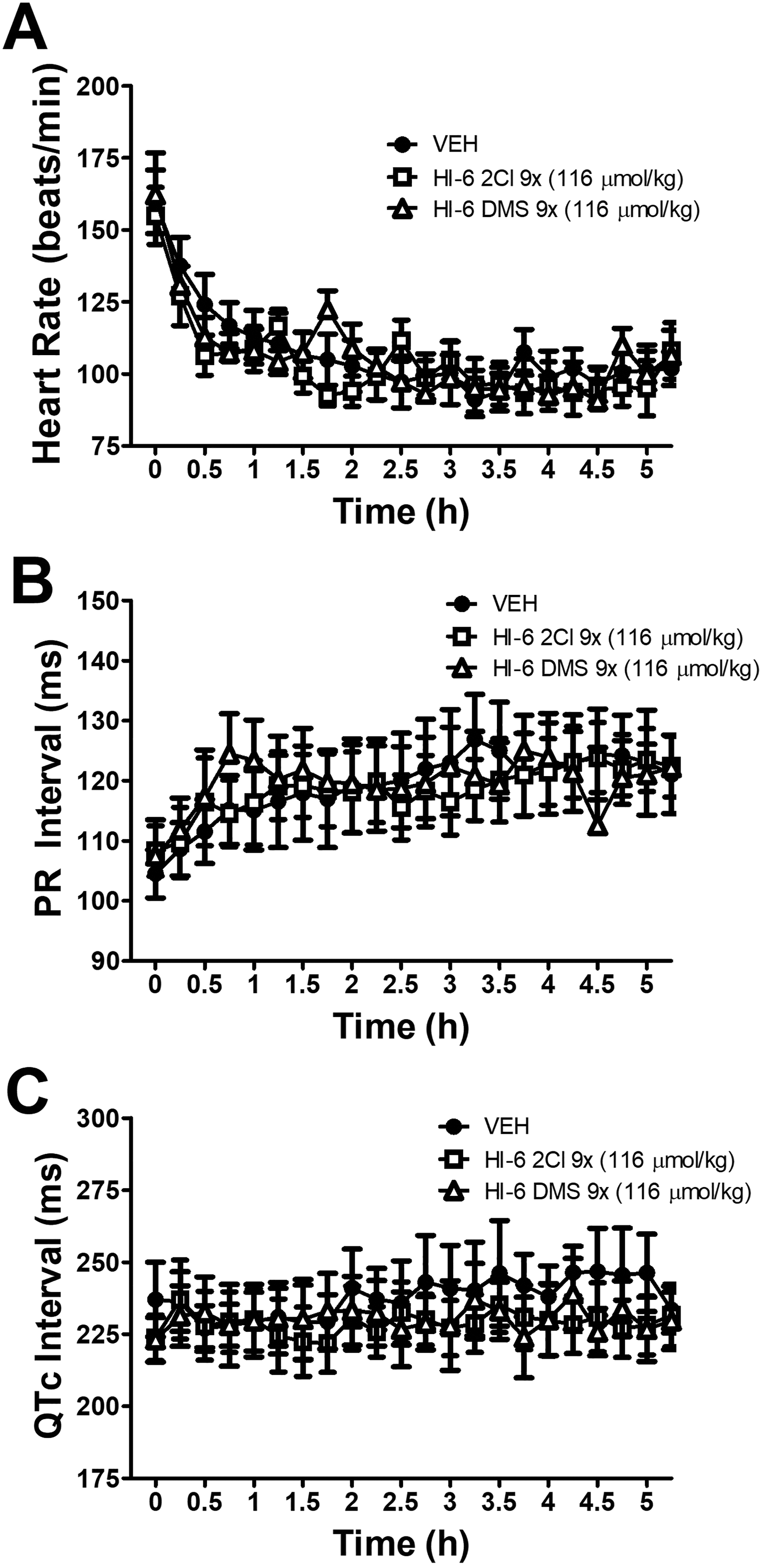
Neither salt of HI-6 had a significant effect on heart rate (A), PR interval (B), or QTc (C). The data are presented as the mean ± standard error of mean [SEM].
2-PAM
There were statistically significant time–drug interactions for all measures of blood pressure (systolic, diastolic, and mean pressures P < 0.0001; pulse pressure P < 0.01; Figure 7). There was a trend of increasing doses of 2-PAM resulting in increasing mean blood pressure, but this trend failed to reach statistical significance. By the end of the recording period, all parameters following 2-PAM administration returned to levels similar to those seen following vehicle administration. There were no significant effects on HR, PR interval, or QTc interval (Figure 8).
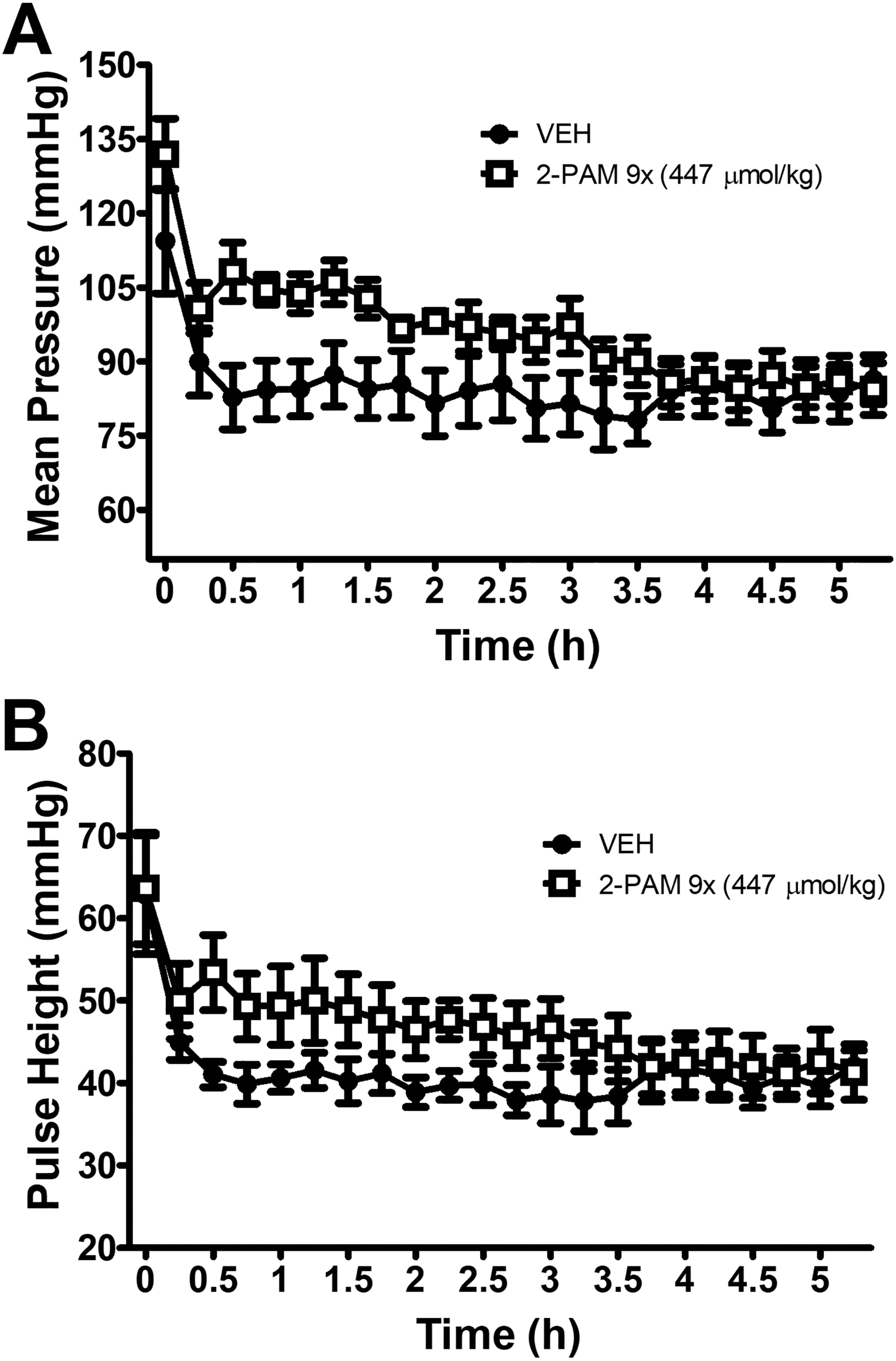
Effects of 2-PAM (447 µmol/kg) on mean arterial pressure (A) and pulse height (B). Although there was a trend for a transient increase in mean pressure and pulse height following 2-PAM administration, this trend failed to reach statistical significance. The data are presented as the mean ± standard error of mean [SEM].
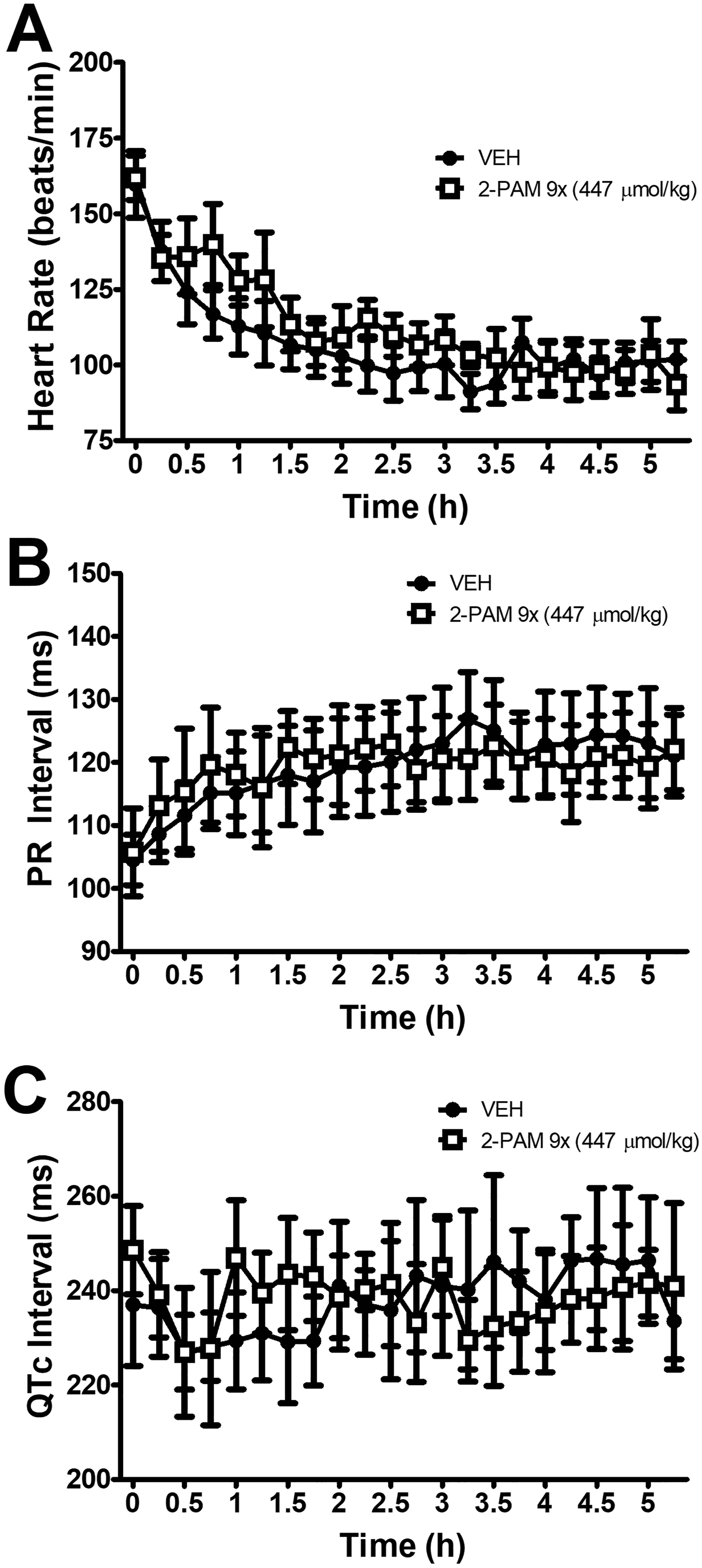
Effect of 2-PAM (447 µmol/kg) on heart rate (A), PR interval (B), or QTc (C). Similar to what was observed with the other oximes, 2-PAM had a negligible effect on cardiac parameters.
Experiment 4: Hematology and Blood Chemistry Following Repeated Oxime Administration
Hematology results indicated an increase (72%) in neutrophil count following the chronic treatment with oximes (Table 3). The baseline mean neutrophil count was 1.39 (0.38; mean [SD]) 103 cells/mL and increased to 2.39 (1.00) 103 cells/mL following the experiment (P < 0.05). The blood chemistry panels (Table 4) revealed mostly modest, although statistically significant, differences (P < 0.05) between pre- and postadministration samples with respect to inorganic phosphorus (−19%), lactate dehydrogenase (+80%), blood urea nitrogen (+17%), and chloride levels (−4%). The differences in pre- and postexperiment lactate dehydrogenase levels reached statistical significance, despite the large standard deviation in the postexperiment values (132) compared to the baseline values (22).
Hematology Results for Blood Drawn Prior to Any Oxime Administration (Pre) and Following Repeated Dosing With Oximes (Post).a
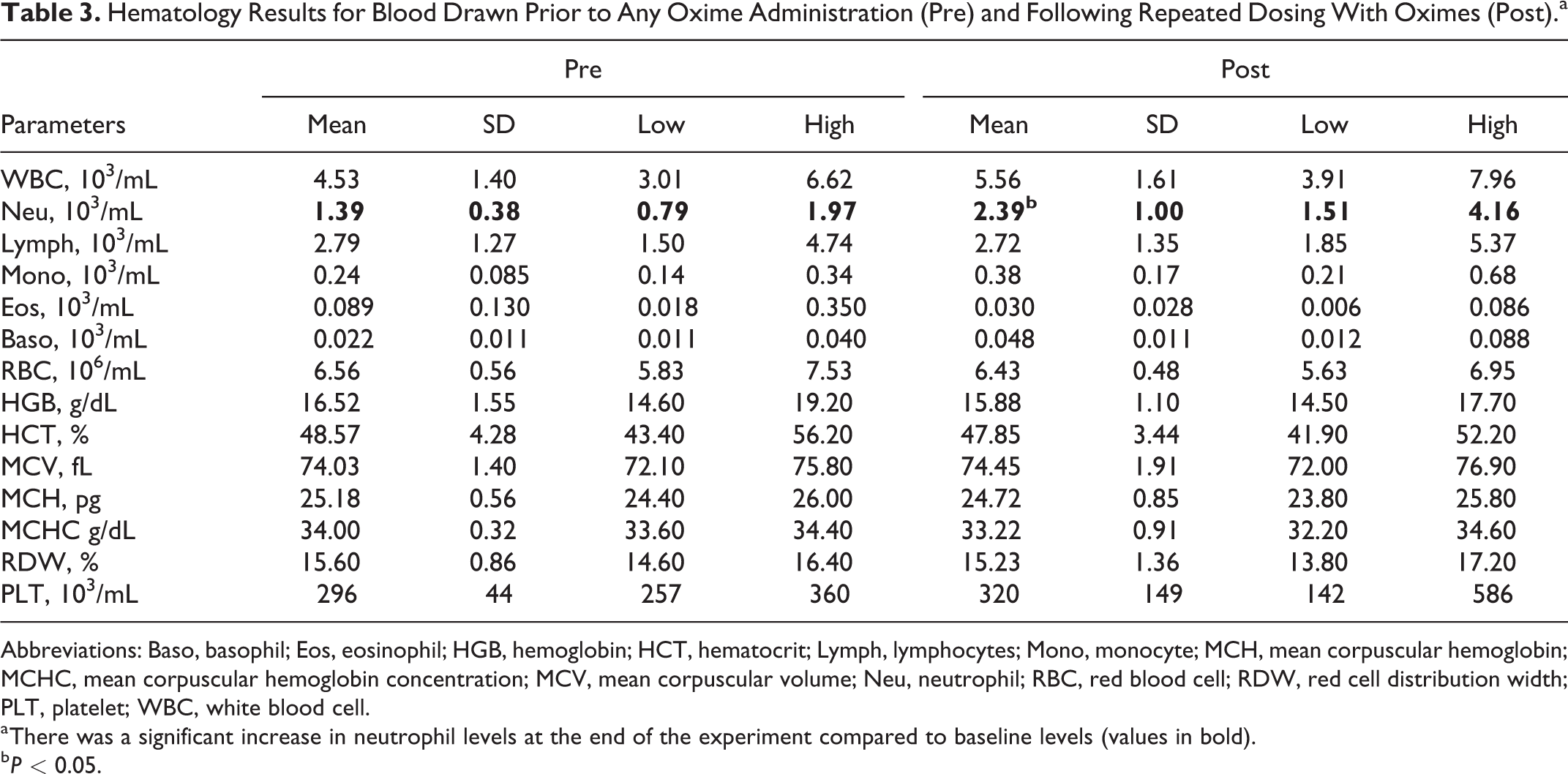
Abbreviations: Baso, basophil; Eos, eosinophil; HGB, hemoglobin; HCT, hematocrit; Lymph, lymphocytes; Mono, monocyte; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; Neu, neutrophil; RBC, red blood cell; RDW, red cell distribution width; PLT, platelet; WBC, white blood cell.
a There was a significant increase in neutrophil levels at the end of the experiment compared to baseline levels (values in bold).
b P < 0.05.
Blood Chemistry Results for Blood Drawn Prior to Any Oxime Administration (Pre) and Following Repeated Dosing With Oximes (Post).
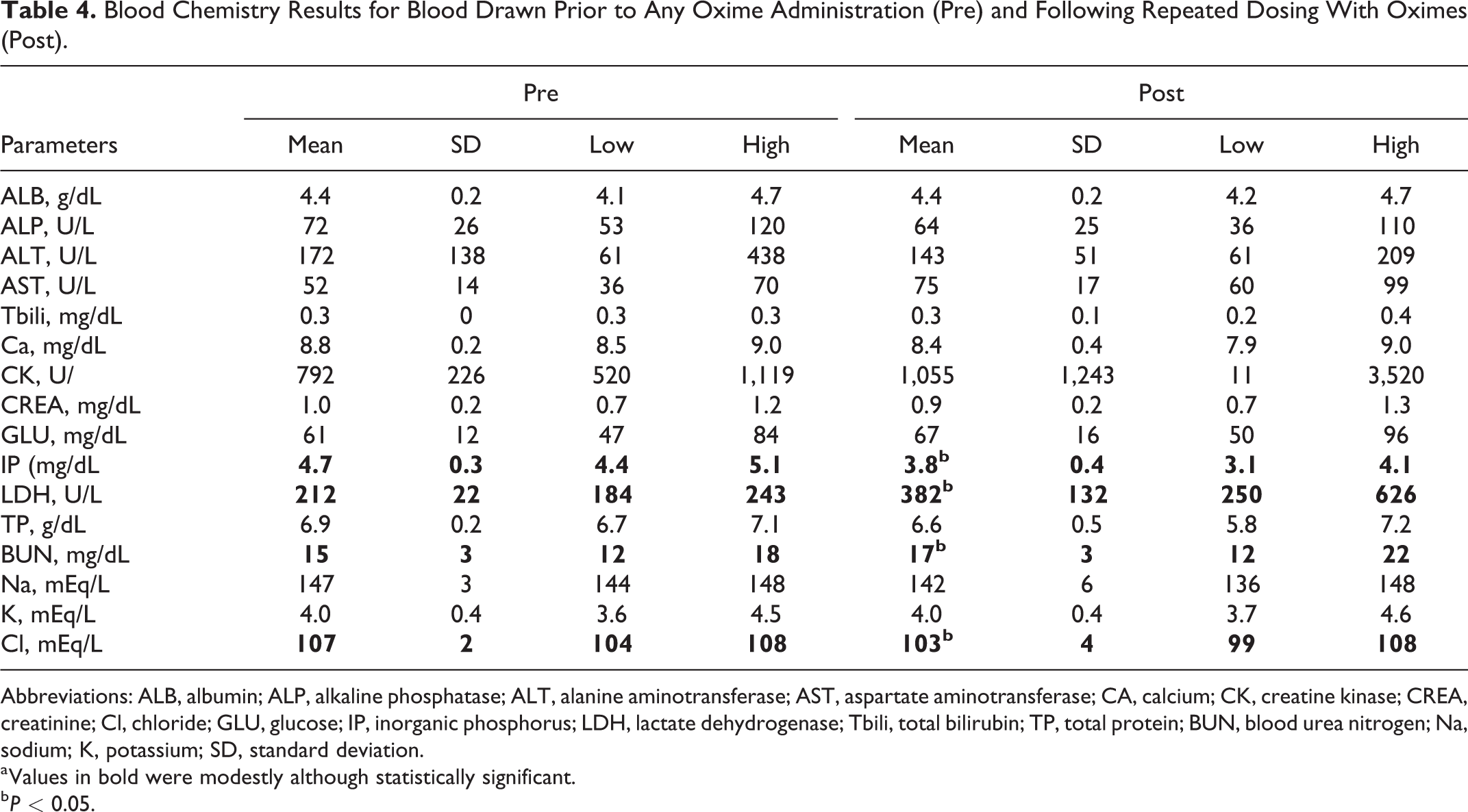
Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CA, calcium; CK, creatine kinase; CREA, creatinine; Cl, chloride; GLU, glucose; IP, inorganic phosphorus; LDH, lactate dehydrogenase; Tbili, total bilirubin; TP, total protein; BUN, blood urea nitrogen; Na, sodium; K, potassium; SD, standard deviation.
a Values in bold were modestly although statistically significant.
b P < 0.05.
Discussion
The pharmacokinetic parameters were estimated using a 1-compartment model with first-order absorption and elimination. Intramuscular administration of either salt of MMB-4 resulted in a rapid rise in plasma concentration with a Tmax of approximately 20 minutes. There were few differences in parameters between salts. Cmax and AUC∞ increased in a dose-dependent manner.
There is a paucity of studies in the literature characterizing the pharmacokinetics of MMB-4. The pharmacokinetic estimates of the highest dose tested (116 µmol/kg, im) in the current study using African green monkeys are similar to those obtained for MMB-4 (100 µmol/kg, im) in swine, except that a much longer elimination half-life (158 minutes) was reported in swine. 17 The elimination half-life of 46.84 minutes for 58 µmol/kg MMB-4 2 Cl in this study is similar to the 50 minutes half-life reported in rabbits at 50 µmol/kg. 18 The highest dose (116 µmol/kg or 55.5 mg/kg) of MMB-4 DMS tested in this study had an elimination half-life of 60.85 minutes, similar to values reported in rhesus monkeys. 19 Interestingly, apparent volumes of distribution (L/kg) and apparent clearances (ml/h/kg) appear to be conserved across species, with the exception of rats, as noted by Hong et al. 19
The current study evaluated the cardiovascular effects of MMB-4 and 2 other fielded oximes, HI-6 and 2-PAM. There were minimal to no significant effects of lower doses of the oximes, so only the highest doses (116 µmol/kg for HI-6 and MMB-4 salts and 447 µmol/kg for 2-PAM Cl) are presented in the results. An elevation in heart rate and blood pressure was observed at the start of all trials regardless of treatment (oxime or vehicle) due to handling and injection. A slight apparent increase in heart rate over vehicle following MMB-4 administration of either salt was noted but failed to reach statistical significance. Slight to modest increases in heart rate have been reported in swine 17 and beagles 20 following MMB-4 2Cl or MMB-4 DMS, respectively.
Blood pressure appears to be one of the more sensitive measures of the cardiovascular effects of oximes. Other studies have reported elevation in blood pressure following administration of MMB-4 and 2-PAM which was not seen in the current study. Stemler et al 17 reported a transient increase in blood pressure with 2-PAM followed by prolonged elevation 3.5 to 4 hours after oxime administration. In the current study, mean pressure following 2-PAM administration was elevated compared to vehicle, although not statistically significant, over the first 3 hours and indistinguishable from vehicle by 4 hours after administration. Mean, systolic, and diastolic pressures were not significantly affected following MMB-4 administration and, if anything, showed a trend for a decrease in blood pressure. The significant effect of MMB-4 DMS on pulse height reflected the small decrease in systolic pressure with no effect on diastolic pressure. Roche et al 20 reported an increase in diastolic and systolic pressures in dogs 2 to 5 hours after MMB-4 DMS administration that returned to normal by 16 hours. The long duration of the increase in blood pressure may have been exasperated by returning the dogs to their home cages from a sling 5 hours after administration of MMB-4. Unfortunately, that study did not include vehicle animals or conditions to control for the effects associated with restraint in the sling and subsequent return to the home cage. Although the current study recorded blood pressure for only 5.5 hours after MMB-4 administration, by that time the majority of MMB-4 at any of the doses tested was cleared from the plasma. Also, it is highly unlikely that MMB-4 would directly affect blood pressure at later time points considering the obvious lack of effect at earlier time points.
The behavioral test employed in this study required several aspects of cognition and behavior of military relevance: attention, situational awareness, speed discrimination, decision-making, and fine motor coordination. Treatment with either salt of MMB-4 in high doses did not impair accuracy, choice reaction time, or trial-initiation latency in the current study. These results indicate that im injection of 116 and 174 µmol/kg of the oximes MMB-4 2Cl, MMB-4 DMS, HI-6 2Cl, and HI-6 DMS are devoid of significant cognitive–behavioral effects as tested in African green monkeys.
The only oxime-induced impairments came at the highest dose (447 µmol/kg) of 2-PAM with significant reductions in accuracy, choice reaction time, or trial-initiation latency. Given that all 3 measures of performance were affected, a general decrease in motivation (eg, as a result of gastrointestinal upset, headache, or nausea) may be responsible. Only a limited number of studies have addressed the behavioral toxicity of oximes. Oral conditioned taste aversion in rats has been demonstrated following administration of high doses of pralidoxime mesylate (P2S). 21,22 However, the same dose did not disrupt responding under a fixed ratio 20 schedule of water reinforcement in rats. 22 Extremely high doses of HI-6 can affect open field performance, disrupt operant responding, and produce a conditioned taste aversion in rats. 23,24 Pralidoxime mesylate-induced impairments in open field performance and motor coordination have been observed following intraperitoneal administration at 644 µmol/kg but not at 322 µmol/kg. 24 The current study and those from the literature suggest that oximes are relatively safe and only cause behavioral disturbances (untoward side effects) at doses well above the fielded or projected therapeutic dose in man. Safety concerns are further reduced because the projected administration of oximes would only occur after a suspected OP poisoning.
Blood chemistry and hematology assays were conducted prior to oxime administration and again at the end of an extensive regimen of repeated oxime injections. Each monkey received multiple doses of multiple oximes across several weeks. The blood tests were used as a general measure of safety. The elevation in neutrophils at the conclusion of the experiment was statistically significant but is not likely of physiological significance. An elevation in neutrophils can indicate an infection but also may be transient, resulting from vigorous exercise. The neutrophil values reported at the beginning of the experiment are lower than typically reported values for African green monkeys, whereas the postoxime values were within the normal range, 25,26 further minimizing the likely significance of the observed neutrophil changes. The slight drop in inorganic phosphorus levels can indicate malnutrition or hypothyroidism but is likely not a significant finding in isolation (Ca and K were normal), and the levels were within the range of reported values. 25 The lactate dehydrogenase (LDH), an indicator of tissue and cellular damage, was highly variable in the postoxime values with a mean of 382 and a range of 250 to 636 U/L compared to baseline values (mean: 212, range: 184-243). Hambleton et al 27 also reported a large range of LDH values (125-2,537) with a mean of 629 U/L. The lack of a significant increase in other hematological measures of tissue damage (alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase) in addition to the values obtained in this study falling at the lower end of the reported range suggests that tissue damage is unlikely. The slight increase in blood urea nitrogen and decrease in Cl levels are mostly likely physiologically insignificant as both fall within normal ranges reported in the literature. 25,27 Overall, the differences reported are most likely due to type I error and represent normal variations in blood chemistry and hematology measures in African green monkeys.
In summary, this study determined pharmacokinetic estimates for both the 2Cl and DMS salts of MMB-4 at doses equivalent to 1, 3, and 6 autoinjectors in a 70-kg human adult. There were differences between salts with both showing a Tmax of about 20 minutes and dose-dependent Cmax values. Higher than doctrinal doses (up to 9× autoinjector) of MMB-4 and HI-6 did not produce behavioral toxicity; however, the highest dose of 2-PAM (447 µmol/kg) did disrupt operant performance. The oximes in this study produced few physiological effects with minor transient changes in blood pressure seen only at the highest doses. Despite the large doses of multiple oximes, neither blood chemistry nor hematological values were outside the normal range seen in African green monkeys. These data suggest the relative safety of these compounds even at doses above those predicted to be therapeutic in man.
Footnotes
Authors’ Note
The views expressed in this article are those of the author(s) and do not reflect the official policy of the Department of Army, Department of Defense, or the U.S. Government.
Acknowledgment
We dedicate this manuscript to the memory of our dear colleague Dr Gregory E. Garcia. The authors would also like to acknowledge Harry Singh and Patrick Mattern from the Walter Reed Army Institute of Research for their contributions to plasma assay methods.
Author Contributions
Moffett, M. contributed to analysis and interpretation and drafted the manuscript. McDonough, J. contributed to conception and design and critically revised the manuscript. McMonagle, J. contributed to acquisition and critically revised the manuscript. Myers, T. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. T.M.M and J.H.M. designed the experiments, J.D.M. and T.M.M. performed the experiments, G.E.G. performed pharmacokinetic analysis of blood samples, and M.C.M. and T.M.M. analyzed data and wrote the manuscript. The following people contributed to this research from the USAMRICD: Jacob Skovira, Megan Lyman, Maura Pannell, and M.A.J. Ken Despain.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Defense Threat Reduction Agency, Medical S&T Division.
