Abstract
The aim of this study was to investigate the genotoxicity of aluminum oxide (Al2O3), β-tricalcium phosphate (β-TCP) (Ca3(PO4)2), and zinc oxide (ZnO) nanoparticles (NPs) that were 4.175, 9.058, and 19.8 nm sized, respectively, on human peripheral blood lymphocytes using micronucleus (MN) and chromosome aberration (CA) techniques. Aluminum oxide and β-TCP NPs did not show genotoxic effects on human peripheral blood cultures in vitro, even at the highest concentrations; therefore, these materials may be suitable for use as biocompatible materials. It was observed that, even at a very low dose (≥12.5 ppm), ZnO NPs had led to genotoxicity. In addition, at high concentrations (500 ppm and above), ZnO NPs caused mortality of lymphocytes. For these reasons, it was concluded that ZnO NPs are not appropriate for using as a biocompatible biomaterial.
Introduction
In the last decade, nanotechnology has entered every aspect of our life and attracts attention due to its rapidly wide-spreading applications. 1,2 The reason for this popularity, at the nanoscale values, the quantum physics laws take place the traditional laws of physics. Consequently, the physical, chemical, and biological features of matter such as conductivity, reactivity, elasticity, hardness, color, temperature and pressure resistance, toxicity, and biocompatibility are affected by this change. These changes have a key role for more effective, intelligent, attractive, secure, and faster solutions. 3 –5 Among the widely used applications of nanoparticles (NPs) are energy, textiles, electronics, cosmetics, food, pharmacology, and biomedical applications. 6,7 Undoubtedly, biomedical applications are vital and the most critical applications in all these areas. These applications are targeted to produce less weight, more durable, more economical, easier, and higher biocompatible materials and systems. Finally, NPs are basic building blocks to construct materials that can contribute to solutions of these problems. Determination of toxicity of NPs that are widely used due to their unique properties, such as surface area and charge, particle size, chemical and physical properties, is important for human health. 2,8,9
In this study, we aimed to investigate the potential genotoxic effects of selected NPs on human peripheral blood cells (lymphocytes in vitro). Additionally, we analyzed the genotoxic effects of these NPs using the chromosome aberration (CA) and micronucleus (MN) assays.
Materials and Methods
Chemicals and Instrumentations
Analytical grade reagents (Sigma, Germany), Fluka (Germany), Riedel-de Haent (Germany), Biochrom (Germany) and Roche (Germany) were used throughout this work without any further purification. aluminum oxide (Al2O3), β-tricalcium phosphate (β-TCP) (Ca3(PO4)2), and zinc oxide (ZnO)NPs were provided from Faculty of Science, Department of Chemistry, Ataturk University. Nanoparticles stock suspensions were prepared in distilled water, vortexed for 12 minutes, and stored at +4°C in dark. Scanning electron microscopy (SEM) analysis was performed using an FEI Quanta 250 FEG (Fei Company, Hillsboro, Oregon, USA) instrument at an acceleration voltage of 2.00 kV. In Powder X-ray experiments, Philips X’Pert Pro Diffractometer (Philips, Eindhoven, The Netherlands) was used.
Cell Cultures
This research was carried out using heparinized human blood samples from 4 healthy, nonsmoking donors (male) aged between 20 and 24 years, and they were not exposed to radiation by their occupation. Hematological and biochemical parameters of volunteers were analyzed, and no pathology was detected. Informed consent forms were signed by each donor. Cultures were set up according to a previously published protocol with a slight modification. 10 The heparinized whole blood samples (0.5 mL) were cultured in 6 mL of culture medium (Chromosome Medium B, Biochrom, United Kingdom) with phytohemagglutinin. Nanoparticles suspensions (0.5 mL) were added to blood culture tubes to achieve final concentrations of 1, 12.5, 25, 50, 100, 250, 500, 1000, and 2000 ppm. The negative and the positive control groups were distilled water and mitomycin C (MMC; at 10−7 M, Sigma), respectively. The blood cultures were incubated in complete darkness for 72 hours at 37°C in an incubator.
Chromosome Aberration Assay
Lymphocyte cultures for CAs (chromatid or chromosome gaps and chromatid or chromosome breaks) were prepared using Evans and coworkers’ method with slight modifications. 11 After 70 hours and 15 minutes of incubation, colcemid (Sigma) at a final concentration of 10 μg/mL was added to the cultures. At the end of incubation, the cells were harvested by replacing the culture medium with a hypotonic solution (0.4% KCl) in which cells were incubated for 20 minutes at 37°C. Finally, the cells were fixed in cold Carnoy’s fixative (methanol: glacial acetic acid, 3:1vol/vol), 3 times. To prepare the slides, the cell pellets in the tubes were homogenized and dropped onto a cold slides. Finally, the slides were dried at room temperature and stained with 5% Giemsa stain solution (diluted with Sorensen buffer, pH 6.8) for 15 minutes.
Micronucleus Assay
Micronucleus assay was performed according to Fenech method. 12 Cultures were prepared as in CA assay. After 44 hours from the start of incubation, Cytochalasin B (final concentration of 3 µg/mL; Sigma) was added to the tubes. At the end of the 72-hour incubation period, the cells were treated with 0.4% KCl for 20 minutes at 37°C. Then, the cells were fixed in a cold fixative (methanol: glacial acetic acid: 0.9% NaCl, 5:1:6 vol/vol/vol). Finally, the slides were dried in air and stained with 5% Giemsa stain solution for 15 minutes. 13
Slide Analysis
Slides were observed using a Prior T-100 mA (England) light microscope at 100× magnification with immersion oil. They were also reviewed by 2 observers who were blinded to the results of CA and MN analysis. For the analysis of CAs, 30 well-spread metaphases were analyzed to define the presence of CAs. Classification of CAs (chromatid or chromosome gap and chromatid or chromosome break) was carried out in accordance with the recommendations of Environmental Health Criteria 46 for environmental monitoring of human populations. 14
Statistical Analysis
IBM SPSS statistics for Windows package program (v.18.0, IBM Corp, Armonk, New York) was used for statistical calculations. The Fisher least significant difference test was used to compare obtained data from experiments as mean values for CA/cell and MN/1000 cell data between test and control groups. Significant differences were evaluated at P < .05.
Results
Characterization of NPs
The average particle size of the sample was calculated using the Scherrer formula d = Kλ × (β CosΘ)−1, where d is the mean particle size, K is the grain shape–dependent constant 0.89, λ is the wavelength of the incident beam in nanometer, Θ is the Bragg reflection angle, and β is the line broadening at half the maximum intensity in radians. 15,16 The average particle size for Al2O3, Ca3(PO4)2, and ZnO were calculated about 4.175, 9.058, and 19.8 nm, respectively. The particles crystallize in the monoclinic system, space group C2/c (JPCDS Card No: 33-0831). The results of X-ray diffraction and SEM analysis are shown in Figure 1.
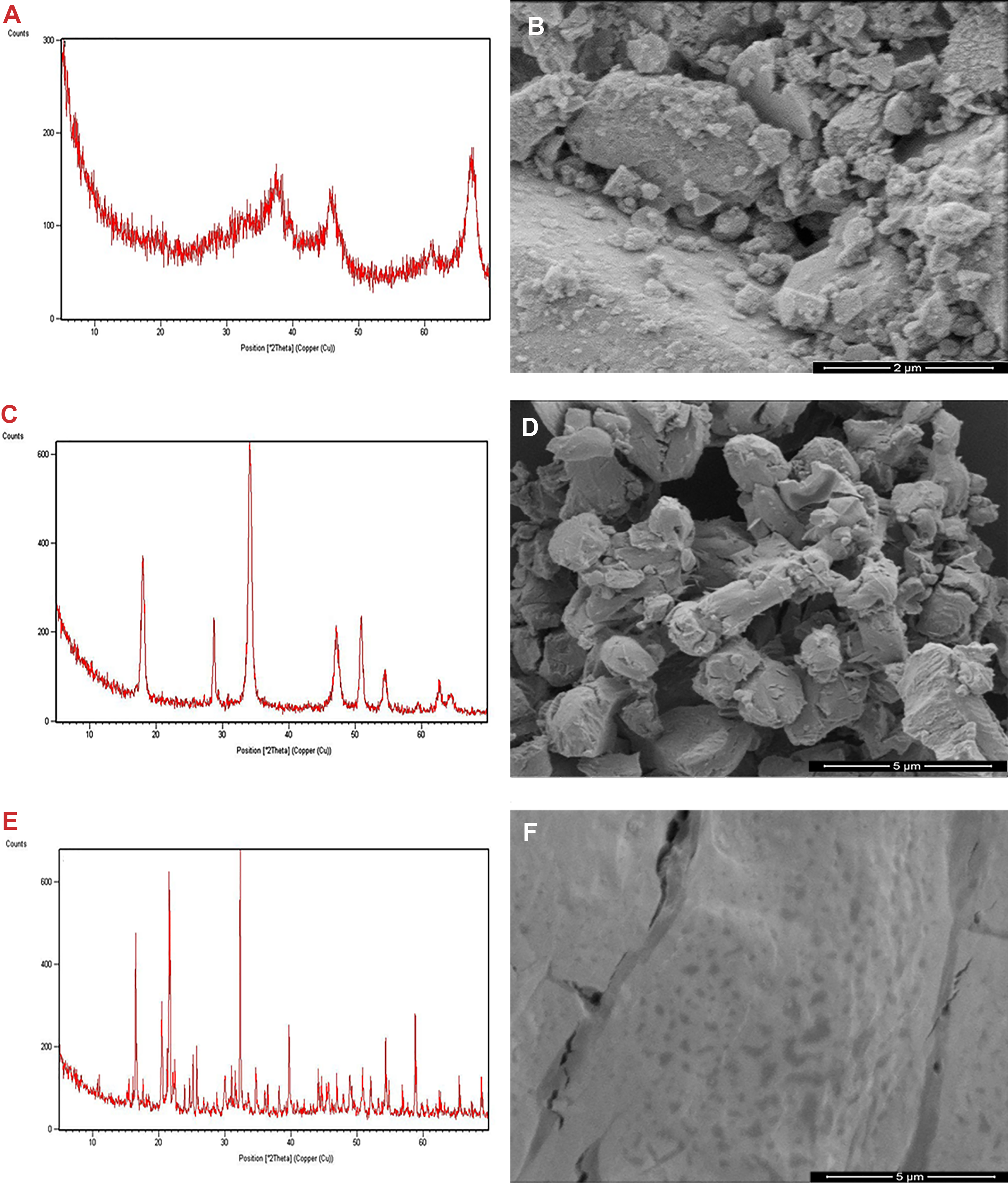
The Powder X-ray diffraction (XRD) patterns of (A) Al2O3, (C) Ca3(PO4)2, and (E) ZnO NPs. Scanning Electron Microscopy (SEM) images of (B) Al2O3, (D) Ca3(PO4)2, and (F) ZnO NPs.
Chromosome Aberration Data
Al2O3 and β-TCP NPs CA frequencies at all concentrations studied (1, 12.5, 25, 50, 100, 250, 500, 1000, and 2000 ppm) did not show any significant increases compared to the vehicle control group. The results of the CA analysis of ZnO NPs indicated a significant increase in the rate of damaged lymphocyte cells at 12.5, 25, 50, 100, and 250 ppm. High concentrations of ZnO (500, 1000, and 2000 ppm) led to inhibition of cell proliferation due to cytotoxicity. All results are shown in Figure 2.
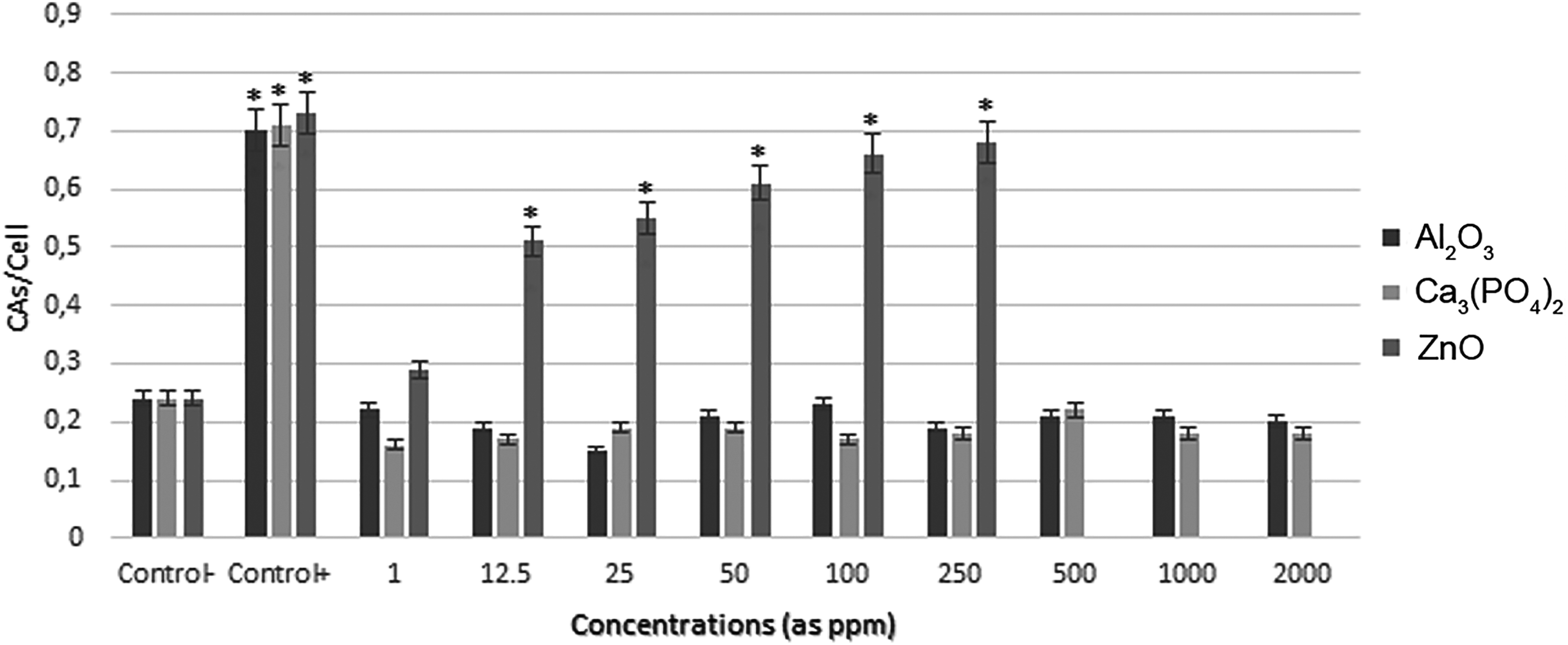
The frequencies of chromosome aberrations (Cas) in human lymphocytes treated with different concentrations of nanoparticles (NPs). Values were expressed as mean (standard deviation) for 5 cultures in each group. *Symbol indicates statistical difference from negative control at a level of 5%.
Micronucleus Frequency
The genotoxic activity of nanosize Al2O3 in the concentration range of 1 to 2000 ppm was determined after incubation of human lymphocyte for 72 hours. Micronucleus frequency also did not show any statistically significant alteration with β-TCP NPs compared to control under the same incubation conditions. The results showed statistically significant increases at 5 test concentrations of ZnO NP (from 12.5 to 250 ppm) compared to the negative control groups. At 500, 1000, and 2000 ppm, ZnO NPs caused cell death. Therefore, MN frequency was not determined for these concentrations. All results are presented in Figure 3.
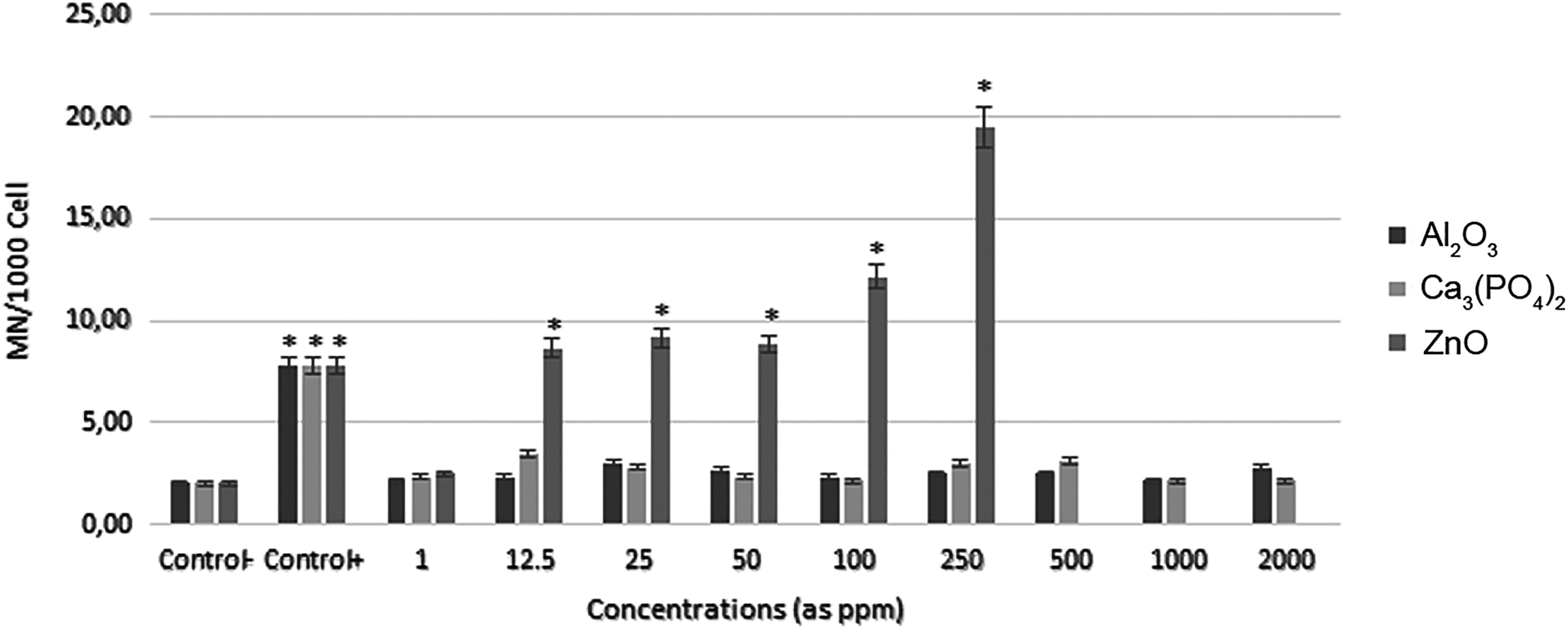
The frequencies of micronucleus (MNs) in human lymphocytes treated with different concentrations of nanoparticles (NPs). Values were expressed as mean (standard deviation) for 5 cultures in each group. *Symbol indicates statistical difference from negative control at a level of 5%.
Discussion
Nanoparticles have provided a major contribution to the development of biomedical, electronic, military, biosensors, and industrial fields. 17 –21 Aluminum oxide, β-TCP, and ZnO NPs have many potential applications in these fields. The toxic effects of these NPs have been evaluated by various research groups in different cell lines such as rat ovary, human bronchial epithelial, epidermis cells, and so on. 22 –24 In this study, dose-dependent genotoxic effects of Al2O3, β-TCP, and ZnO NPs were investigated in human lymphocytes. The CA test is an important and useful indicator to reveal the effects of biological and chemical molecules. 25 Similarly, MN assay allows the ability to reliably measure for clastogenic and anogenic abnormalities such as chromosome fracture, loss, and sticky. 26
Our results indicated that Al2O3 NPs did not show any statistically significant increase in the frequencies of CA and MN. Consequently, a size of 4,175 nm Al2O3 NPs did not cause genotoxic effect in human lymphocyte cells. Similarly, in a few studies that were performed in vitro it was not observed genotoxic and/or cytotoxic effects of Al2O3 NPs on different mammalian cell lines, such as rat fibroblasts (L929), human skin fibroblasts (BJ), mouse lymphoma, human bronchial epithelium cells, and monkey kidney cells (CHS-20) using various methods, etc. EZ4U, Comet, MLA, and MN test in the concentration range of 0 to 1250 µg/mL. 18,20,27,28 Moreover, Balasubramanyam et al’s research group reported nano-Al2O3’s toxicity on different strains of Salmonella typhimurium species with Ames test. 17 In contrast, 2 different in vitro studies on mouse cell lines reported that Al2O3 NPs were cytotoxic and genotoxic not only in high concentrations but also in very low concentrations. 22,29 When we compared these results with ours, we considered that the reason for differences is related to the NP size and using cell culture. This idea was supported by Chithrani and coworkers that the NPs with 50 nm particle size were taken to the cells faster than the NPs with 14 nm and also recommended that toxicity was influenced by various factors such as intracellular delivery rates and amounts by nanoscale dimensions. 30 Two different in vivo studies also stated that Al2O3-NPs caused genotoxicity and neurotoxicity in rat cells. 31,32 In addition, according to the last 2 research results, it was suggested that the cause of genotoxicity is high-dose exposure in in vivo studies. The differences in results of in vivo and in vitro assays were affected by several factors such as high-dose concentrations, time course of exposure, culture conditions, and cell types. It was also reported that there were little correlation between in vivo and in vitro experiments. 33
The calcium phosphate compounds were preferred as a biomaterials due to their excellent biocompatibility and nontoxic properties. 19 The results for the present study indicated that β-TCP NPs with particle size 9.058 nm did not show genotoxic effect on human lymphocytes in the concentration range of 1 to 2000 ppm. In the literature, no information was found about the genotoxicity of β-TCP NPs. Therefore, this study was the first to investigated on toxicity of β-TCP NPs. Nevertheless, there were a few reports on the toxicity of different NP compounds that contain calcium. It was reported that CaP NPs did not cause cytotoxicity on HeLa cells and HEK 293 cells with particle size of 180 and 100 nm, respectively, with the [3-[4,5-dimethylthiazol-2-yl-)2,5-diphenyl tetrazolium bromide] (MTT) assay in vitro. 34,35 Similarly, the results of the 7-day acute oral toxicity tests and repeated-dose, 28-day subchronic oral toxicity tests indicated that CaCO3 (100-200 nm) and Ca3(C6H5O7)2 (200-400 nm) particles were not able to cause a significant and dose-dependent toxicity even at very high doses (1.3 and 2.3 g/kg) on the imprinting control region mice. 36 On the contrary, recent in vitro study on toxic effects of dolomite ([CaMg(CO3)2]) NPs claimed that these NPs (particle size 39±18 nm) had cytotoxic and genotoxic effects in concentration range of 100 to 400 µg/mL. Patil and coworkers determined that dolomite was a toxic compound, but they did not explain which element/elements cause the toxicity. 37 On the other hand, there were a few studies on toxicity of bulk size β-TCP. The combination of PDGF-BB and β-TCP that a growth factor used in molecular biology did not show systemic and local toxicity in rats. 38 Similar in vivo studies on rats and rabbits demonstrated that the bulk size β-TCP did not cause subacute systemic toxicity. 39,40
According to the studies that were performed in order to determine the genotoxic effects of ZnO NPs, this NP was a very genotoxic material even at low concentrations in general. It was claimed that the ZnO NPs caused genotoxicity leading to lipid peroxidation and oxidative stress. 41 –47 The previous studies reported that ZnO NPs induced DNA double-strand breaks frequency, olive tail moment, apoptosis, DNA breaks, and CA frequency on A549, HEK, HepG2, MEF Ogg1+/+, CHO, and human peripheral lymphocyte cells in the concentration range of 0.1 to 320 µg/mL. 23,48 –52 Similarly, our findings also demonstrated that ZnO NPs caused genotoxicity on human lymphocytes in the concentration range of 12.5 to 250 ppm. The ZnO NPs also caused cell death at 500 to 2000 ppm dose range; therefore, no cells were counted at these concentrations. On the contrary, it was reported that ZnO NPs (≤35 and 50-80 nm) did not show genotoxic effects at 0.1, 1, and 10 µg/mL concentrations on TK6 cell lines, but in the same study, genotoxic effects were only reported at 100 µg/mL using Comet assay. 53 On the other hand, Kwon’s research group investigated the genotoxic effects of ZnO NPs with 20 and 70 nm size, positively or negatively charged. According to their research results, ZnO NPs did not show any significant increase in frequency of CA in the concentration range of 3.75 to 15 µg/mL. 54
Consequently, the safety and biocompatibility of Al2O3 and β-TCP NPs were demonstrated following in vitro biocompatibility tests. According to the tests results, these NPs did not show genotoxic effects on human peripheral lymphocytes at any concentrations. Therefore, it was suggested that Al2O3 and β-TCP NPs could be used as biomaterials for biomedical applications. According to our knowledge, this article was the first study about genotoxicity of β-TCP NPs; therefore, further studies should focus on the genotoxicity and biocompatibility of β-TCP NPs. Many of the previous studies and our results showed that the ZnO NPs had genotoxic potential even at very low concentrations. Thus, we think that it is not suitable to use ZnO NPs as biomaterial. Although it was considered that the reason for ZnO NPs toxicity was Zn+2 ions, which was dissolution product of ZnO, this idea was not clear, because it was not supported by enough literature. 55 Therefore, further studies would be needed to determine the toxicokinetics of ZnO NPs.
Footnotes
Authors’ Contributions
A.G.B. and T.H. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
