Abstract
Despite the extensive use of nanoparticles (NPs) in various fields, adequate knowledge of human health risk and potential toxicity is still lacking. The human lymphocytes play a major role in the immune system, and it can alter the antioxidant level when exposed to NPs. Identification of the hazardous NPs was done using in vitro toxicity tests and this study mainly focuses on the comparative in vitro cytotoxicity and genotoxicity of four different NPs including cobalt (II, III) oxide (Co3O4), iron (III) oxide (Fe2O3), silicon dioxide (SiO2), and aluminum oxide (Al2O3) on human lymphocytes. The Co3O4 NPs showed decrease in cellular viability and increase in cell membrane damage followed by Fe2O3, SiO2, and Al2O3 NPs in a dose-dependent manner after 24 h of exposure to human lymphocytes. The oxidative stress was evidenced in human lymphocytes by the induction of reactive oxygen species, lipid peroxidation, and depletion of catalase, reduced glutathione, and superoxide dismutase. The Al2O3 NPs showed the least DNA damage when compared with all the other NPs. Chromosomal aberration was observed at 100 µg/ml when exposed to Co3O4 NPs and Fe2O3 NPs. The alteration in the level of antioxidant caused DNA damage and chromosomal aberration in human lymphocytes.
Introduction
Metal-oxide nanoparticles (NPs) have been commercially used in biosensors, water purification devices, diagnostics, cosmetics, and therapeutic agents. The NP toxicity study was one of the fastest growing areas of research due to its applications in a wide variety of fields. 1 Cobalt (II, III) oxide (Co3O4), iron (III) oxide (Fe2O3), silicon dioxide (SiO2), and aluminum oxide (Al2O3) are commonly used NPs with a variety of applications and even toxic to human in a dose-dependent manner. 2,3
Co3O4 NPs are currently attracting enormous interest owing to their unique size and due to their various applications like pigments, sensors, electrochemistry, magnetism, energy storage, and medicine. 4,5 Fe2O3 NPs due to their magnetic properties have applications in magnetic drug, gene delivery, tissue repair, cell separation, magnetic resonance imaging, and magnetic fluid hyperthermia. The antibody-targeted Fe2O3 NPs are used for diagnosis and targeted treatment of cancer. 6 SiO2 NPs are used in the biomedical field such as DNA delivery, additives to drugs, cancer therapy, enzyme immobilization, and biosensors. 7,8 Al2O3 NPs have a broad range of applications in aircrafts, ceramic industry, catalysis, biomaterials, and drugs. 9,10
The Co3O4 NPs can easily penetrate and cause damage to the skin than the bulk material. The NPs can pass through cuts and abrasions, causing toxicity in human epidermis. 11 It was reported that hard metal industry workers who are frequently exposed to NPs have increased the risk of lung cancer. 12 The Fe2O3 NPs after given orally for 28 days to female albino rats showed toxic signs and symptoms, but no mortality was observed. The Fe2O3 NPs caused a decrease in the level of brain acetylcholinesterase, indicating that the NPs can affect the synaptic transmission and nerve conduction. 13 The release of Fe2O3 NPs into the environment causes ecological effects on aquatic organisms. It affects the early life stages of the zebrafish (Danio rerio) during embryonic development causing hatching delay, mortality, and malformation. 14 Silica is the common name for materials, which is composed of SiO2. It occurs in crystalline and amorphous forms. The 50, 100, and 200 nm sized SiO2 NPs affect the liver causing inflammation in BALB/c mice by intravenous injection. The NPs remained in spleen and liver even after 4 weeks of NPs exposure. 15 The SiO2 NPs were also found to be toxic to two human cell lines from the proximal tubule (Caki-1 and Hek293). The Caki-1 cells exhibited more toxicity than Hek293 cells. SiO2 NPs in the size range of 25 nm exhibited more cytotoxic and genotoxic effect than NPs at 100 nm for both kidney cell lines. 16 The Al2O3 NPs have been toxic to algae, bacteria, fungi, fish, plants, and mammalian cells. The NPs not only affect the human body but also cause toxic effects in aquatic system and soil microbial communities. The NPs increased the chromosomal aberrations and sister chromatid exchanges in plant cells. 17,18
Human lymphocytes are one of the major components of the immune cells, and they play a significant role in the prooxidant and antioxidant balance of immune cell activity. 19 The studies done regarding NPs toxicity have mainly concentrated on the evaluation of individual NPs toxicity. Only limited works have been done regarding the comparison of different NPs for estimating their possible hazard. This study primarily focuses on the comparative toxicity of the four different NPs, namely, Co3O4, Fe2O3, SiO2, and Al2O3 in human lymphocytes.
Materials and methods
Chemicals
The four different NPs (Co3O4, Fe2O3, SiO2, and Al2O3 NPs) were purchased from Sigma-Aldrich (St Louis, Missouri, USA). The product specification of the suppliers for Co3O4 (CAS number: 1308-06-1), Fe2O3 (CAS number: 1309-37-1), and Al2O3 NPs (CAS number: 1344-28-1) areas follows ≤50 nm and SiO2 NPs (CAS number: 7631-86-9) in the size of 10–20 nm. All the chemicals used in this study were of analytical grade.
NPs characterization
The hydrodynamic size of the NPs in Milli-Q water was measured using dynamic light scattering (DLS; 90Plus particle size analyzer, Brookhaven Instruments Corporation, Holtsville, New York, USA). Particle size of the NPs was measured using a transmission electron microscopy (TEM; JEOL-JEM-2100F TEM operating at 200 kV, Japan). The hydrodynamic size of the NPs after 0th and 24th h exposure to human lymphocyte culturing medium containing Rosewell Park Memorial Institute (RPMI-1640) medium supplemented with 1% penicillin,1% streptomycin, 10% fetal bovine serum (FBS), and 2% phytohemagglutinin (PHA) was measured using the DLS.
Toxicity studies
Culturing of human peripheral lymphocytes
Blood samples were drawn from the healthy volunteers aged 21–25 for cytotoxicity and genotoxicity studies (n = 3) according to VIT University, Institutional Ethics Committee for human studies (IECH). All donors participated in the study signed the written informed consent according to the approved procedure by VIT University, IECH (IEC/IRB no: IECH/2013/Dec 18-005). The human lymphocytes were isolated from peripheral blood using the lymphocyte separation media (HiSepTM, Hi Media, Mumbai, Maharashtra, India) and 1 × 106 cells were cultured using RPMI-1640 supplemented with 1% penicillin, 1% streptomycin, and 10% FBS. The 2% PHA was added to stimulate the cells and incubated for 24 h in 5% carbon dioxide (CO2) at 37°C. The cells were counted by tryphan blue exclusion method and the cell viability was measured using a hemocytometer. 20
Cytotoxicity by MTT assay
After 24 h of incubation, the human lymphocytes were counted and 1 × 104 cells seeded into the 96-well culture plates with different concentration of Co3O4, Fe2O3, SiO2, and Al2O3 NPs (10, 25, 50, 75, and 100 µg/ml) and incubated at 37°C for 24 h. After incubation, cells were treated with 5 mg/ml solution of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) at 37°C for 4 h in 5% CO2 incubator. Then 50 μl of dimethyl sulfoxide was added to solubilize the formed formazan. The number of viable cells was read by a Microplate Absorbance Reader (BioTek, Winooski, Vermont, USA) at 532 nm. The human lymphocytes not treated with NPs were taken as a control. 21
Lactate dehydrogenase leakage assay
The lactate dehydrogenase leakage (LDH) activity was an alternative assay to determine the cytotoxicity of NPs. The membrane integrity damage of cells was estimated using LDH by measuring the amount of enzyme released by the human lymphocytes. About 0.1 ml of Co3O4, Fe2O3, SiO2, and Al2O3 NPs (50, 75, and 100 µg/ml) were added with 0.2 mM tris(hydroxymethyl)aminomethane (Tris)–hydrochloric acid, 30 mM sodium pyruvate, and 6.6 mM nicotinamide adenine dinucleotide. The absorbance was measured at 340 nm, and the LDH release expressed in terms of percentage. 22
Oxidative stress in human lymphocytes
Oxidative stress in human lymphocytes was estimated by the reactive oxygen species (ROS) generation. In 96-well culture plates, 1 × 106 cells/ml were seeded and incubated for 24 h at 37°C. After 24 h, different concentration of NPs (50, 75, and 100 µg/ml) was added and incubated for 24 h. Then the cells were incubated at 37°C for 30 min with dichlorodihydrofluorescein-diacetate (10 mM). The intensities of fluorescence were detected with wavelength excitation at 485 nm and wavelength emission at 528 nm using a fluorescence spectrophotometer (SL174, ELICO, India). 23
LPO and antioxidant enzymes level
The lipid peroxidation (LPO) was estimated by the malondialdehyde (MDA) formation, and 0.1 ml of cell suspension was incubated at 37°C with 0.1 M of sodium phosphate buffer for 1 h. The cells were precipitated with 5% trichloroacetic acid and centrifuged at 2300g for 15 min at room temperature. Then 1% thiobarbituric acid was added and placed in boiling water for 15 min. After cooling at room temperature, the optical density was read at 532 nm using an ultraviolet–visible (UV-Vis) spectrophotometer (2201, Systronics, India) and represented as nanomoles of MDA per milligram of protein. 24
The catalase activity in human lymphocytes was estimated by adding 0.1 ml of lymphocytes with 1 ml of hydrogen peroxide (H2O2; 30 mM in phosphate buffer, pH 7.0) and 2 ml of 50 mM phosphate buffer (pH 7.0). The absorbance was measured at 240 nm using an UV–Vis spectrophotometer (2201, Systronics). 25 Glutathione (GSH) level was estimated using Ellman’s reagent. The assay mixture contained 5,5′-dithiobis-(2-nitrobenzoic acid), phosphate buffer, and human lymphocyte extract. The absorbance was measured at 412 nm for the estimation of GSH. 26 The superoxide dismutase (SOD) assay was done using the kit obtained from Sigma-Aldrich. The absorbance was read at 440 nm by the decrease in color development.
Single-cell gel electrophoresis assay (comet assay)
The comet assay was performed with slight alterations according to the method described by Singh et al. 27 A total of 25 µl of cell suspension was mixed with 75 µl of 0.5% low-melting temperature agarose (LMPA), and it was pre-coated with 1% normal melting temperature agarose. The slides were covered with a microscope coverslip and refrigerated for 5 min. The coverslip was removed, and a layer of 1% LMPA was added on to the slides. This was followed by immersion in ice-cold alkaline lysis solution (2.5 M sodium chloride, 10 mM Tris, 100 mM ethylenediaminetetraacetic acid (EDTA), 10% dimethylsulfoxide, and 1% Triton X-100, pH 10.0) for at least 2 h. The slides were then incubated in ice-cold electrophoresis solution (0.3 M sodium chloride and 1 mM EDTA, pH 13.0) for 20 min, followed by electrophoresis at 15 V for 25 min. After electrophoresis, the slides were neutralized and stained by spreading ethidium bromide (20 µg/ml). Analysis of comets was carried out using a fluorescence microscope (Leica DM-2500, Germany), and the percentage tail DNA was calculated based on the equation, % tail DNA = 100 − % head DNA, for the quantification of DNA damage.
Chromosomal aberrations
Chromosome aberrations were obtained from PHA-stimulated peripheral human lymphocytes. 28 Under aseptic conditions, 0.5 ml of venous blood was added and incubated at 37°C with 5 ml of RPMI-1640 medium, 1.2 ml of FBS, and 0 3 ml of PHA. At the end of 71st h of incubation, the human lymphocytes were treated with 100 µg/ml of NPs for 1 h and centrifuged at 1000 r/min for 5 min. The cells were arrested by adding 0.01% of colchicine solution and then incubated for 5 min using 6 ml of the hypotonic solution, and 6 ml of Carnoy’s fixative was added prior to slide preparations. The slides were prepared by gently placing cell suspension on a glass slide and dried on a hot plate. The chromosomes were treated with 0.25% trypsin that alters the structure of proteins followed by staining with Giemsa solution, and the slides were observed under the microscope.
Statistical analysis
The analysis of the data obtained was done using two-way analysis of variance (ANOVA) by Bonferroni posttest for cytotoxicity and oxidative stress and one-way ANOVA by Dunnett’s multiple comparison tests for DNA damage and chromosomal aberrations. All data were analyzed using GraphPad Prism 5 and found to be statistically significant at p < 0.05.
Results
NPs characterization
The NPs were characterized by measuring the hydrodynamic size using DLS, but the shape and particle size of the NPs were studied using TEM. 29 The hydrodynamic size of the Co3O4, Fe2O3, SiO2, and Al2O3 NPs in Milli-Q water was found to be 96.4 ± 0.57, 74.6 ± 0.6, 41.7 ± 2.6, and 90.2 ± 2.4 nm, respectively (Figure 1). TEM analysis confirmed that NPs are almost spherical in shape with a particle size of 35.8 ± 0.8, 43.7 ± 4.7, 17.1 ± 2.1, and 39.4 ± 3.9 nm for Co3O4, Fe2O3, SiO2, and Al2O3 NPs, respectively (Figure 2).

NPs characterization by DLS. (a) Co3O4 NPs; (b) Fe2O3 NPs; (c) SiO2 NPs; and (d) Al2O3 NPs. DLS: dynamic light scattering; NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide.

NPs characterization by TEM. (a) Co3O4 NPs; (b) Fe2O3 NPs; (c) SiO2 NPs; and (d) Al2O3 NPs. TEM: transmission electron microscope; NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide.
Hydrodynamic size of NPs in the lymphocyte culture medium
The stability of the NPs in lymphocyte culture medium was considered to be an important aspect of the cytotoxicity and genotoxicity assessment. The hydrodynamic size was used to monitor the kinetic changes in order to study the stability of the NPs in the culture medium. 22 The hydrodynamic size of Co3O4 NPs after initial exposure to the lymphocyte culture medium was found to be 206.9 ± 14.4 nm (Figure 3(a)), and an increase in the size to 675.7 ± 41.8 nm was observed after 24 h of exposure to the culture medium (Figure 4(a)). In case of Fe2O3 NPs, at 0th h the hydrodynamic size was found to be 380.6 ± 20 nm (Figure 3(b)), whereas at 24 h a drastic increase in the size to 909.07 ± 45.3 nm was observed when compared with the other metal-oxide NPs (Figure 4(b)). The size of the SiO2 NPs after 0th h of exposure was found to be 192.6 ± 6.9 nm (Figure 3(c)), and an increase in the size to 435.6 ± 29.9 nm due to agglomeration was observed after 24 h of exposure to lymphocyte cure medium (Figure 4(c)). Interestingly, the Al2O3 NPs after initial exposure to the lymphocyte culture medium had a size of 234.02 ± 11.03 nm (Figure 3(d)) when compared with the size of 560.3 ± 20.2 nm after 24 h exposure to the lymphocyte culture medium (Figure 4(d)).
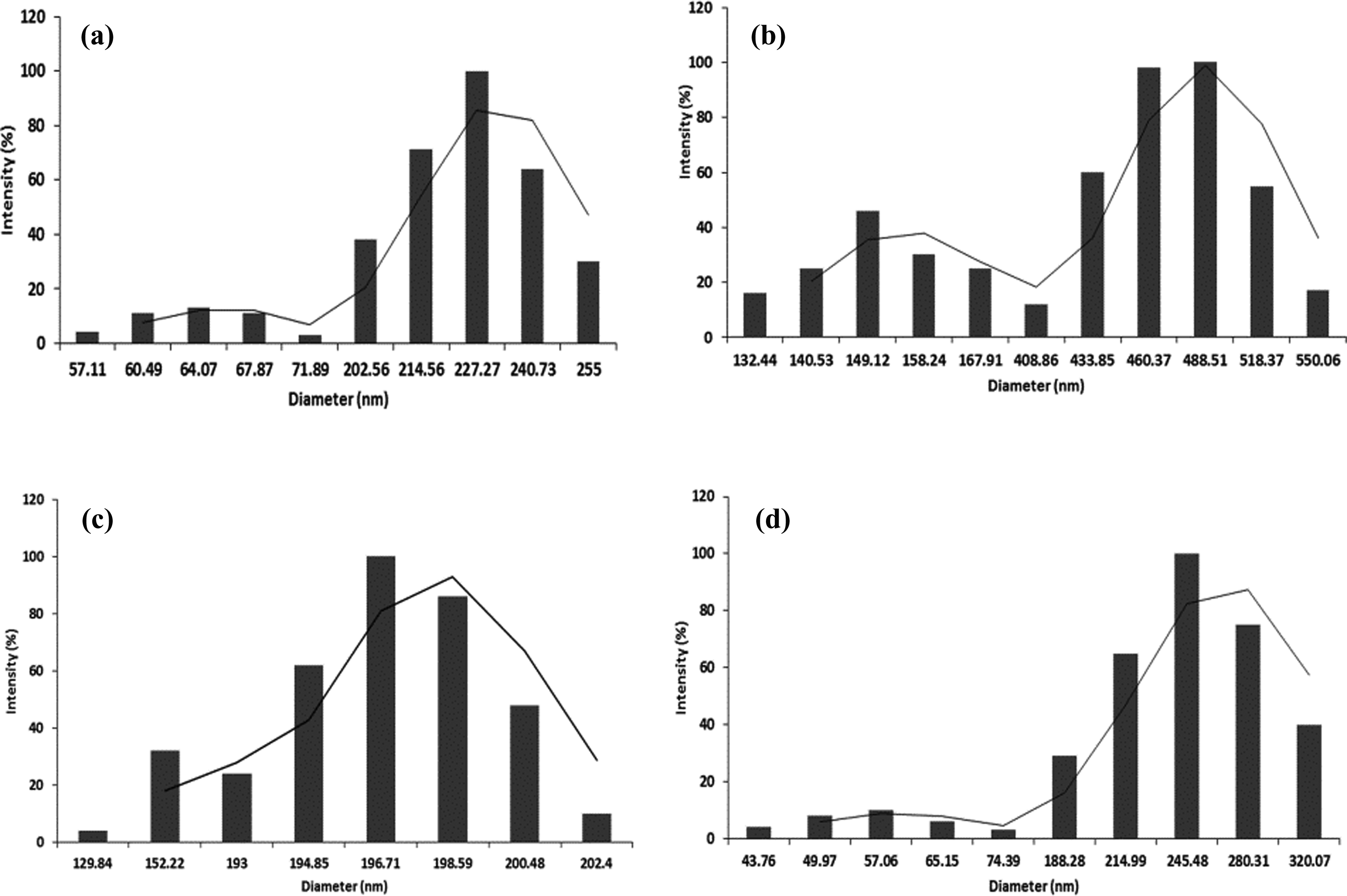
Hydrodynamic size of the metal-oxide NPs after 0th h exposure to the lymphocyte culture medium. (a) Co3O4 NPs; (b) Fe2O3 NPs; (c) SiO2 NPs; and (d) Al2O3 NPs. NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide.

Hydrodynamic size of the metal-oxide NPs after 24 h of exposure to the lymphocyte culture medium. (a) Co3O4 NPs; (b) Fe2O3 NPs; (c) SiO2 NPs; and (d) Al2O3 NPs. NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide.
Cytotoxicity of the NPs in human lymphocytes
The cytotoxicity was observed in human lymphocytes after 24 h of interaction of NPs by MTT and LDH assays. Both MTT and LDH assays have been frequently used for determining the in vitro cytotoxicity of NPs in cell culture experiments. 30 Results of MTT assay showed a significant reduction in cell viability compared with control in a dose-dependent manner. At 50, 75, and 100 µg/ml, the Co3O4 NPs showed a significant reduction in percentage cell viability (p < 0.001). Interestingly, the Fe2O3 and SiO2 NPs have a similar significant decrease in cell viability and the lowest reduction in cellular viability was observed with Al2O3 NPs. The Co3O4 NPs were found to be more toxic to human lymphocytes followed by Fe2O3, SiO2, and Al2O3 NPs in a descending order (Figure 5).

Cellular viability in human lymphocytes by MTT assay when treated with Co3O4, Fe2O3, SiO2 NPs, and Al2O3 NPs. The data obtained are expressed in terms of mean ± SD (n = 3). *p < 0.05: the significance versus control; **p < 0.01: the significance versus control; ***p < 0.001: the significance versus control. NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
The cytotoxicity was further investigated by estimating the percentage of LDH released after incubation with NPs. The Al2O3 NPs showed a significant increase in LDH level (p < 0.01) when exposed to 100 µg/ml, whereas an insignificant increase in LDH level (p > 0.05) was observed when treated at 50 and 75 µg/ml in human lymphocytes. The Co3O4 NPs showed a significant increase (p < 0.001) in LDH level when compared with Fe2O3 NPs and SiO2 NPs after 24 h of exposure to the human lymphocytes at 50, 75, and 100 µg/ml (Figure 6).

LDH leakage in human lymphocytes when treated with Co3O4, Fe2O3, SiO2, and Al2O3 NPs. The data obtained are expressed in terms of mean ± SD (n = 3). The LDH release for the untreated control cells is 0%. *p < 0.05: the significance versus control; **p < 0.01: the significance versus control; ***p < 0.001: the significance versus control. NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide; LDH: lactate dehydrogenase leakage.
Oxidative stress in human lymphocytes
The Co3O4 NPs were found to significantly increase (p < 0.001) ROS level in the human lymphocytes at 50, 75, and 100 µg/ml. The Al2O3 NPs showed a significant increase in ROS level at 100 µg/ml (p < 0.01). The Fe2O3 NPs and SiO2 NPs showed a significant increase in ROS level at 75 and 100 µg/ml (p < 0.001), but insignificant increase in ROS level was observed at 50 µg/ml (p > 0.05) (Figure 7).
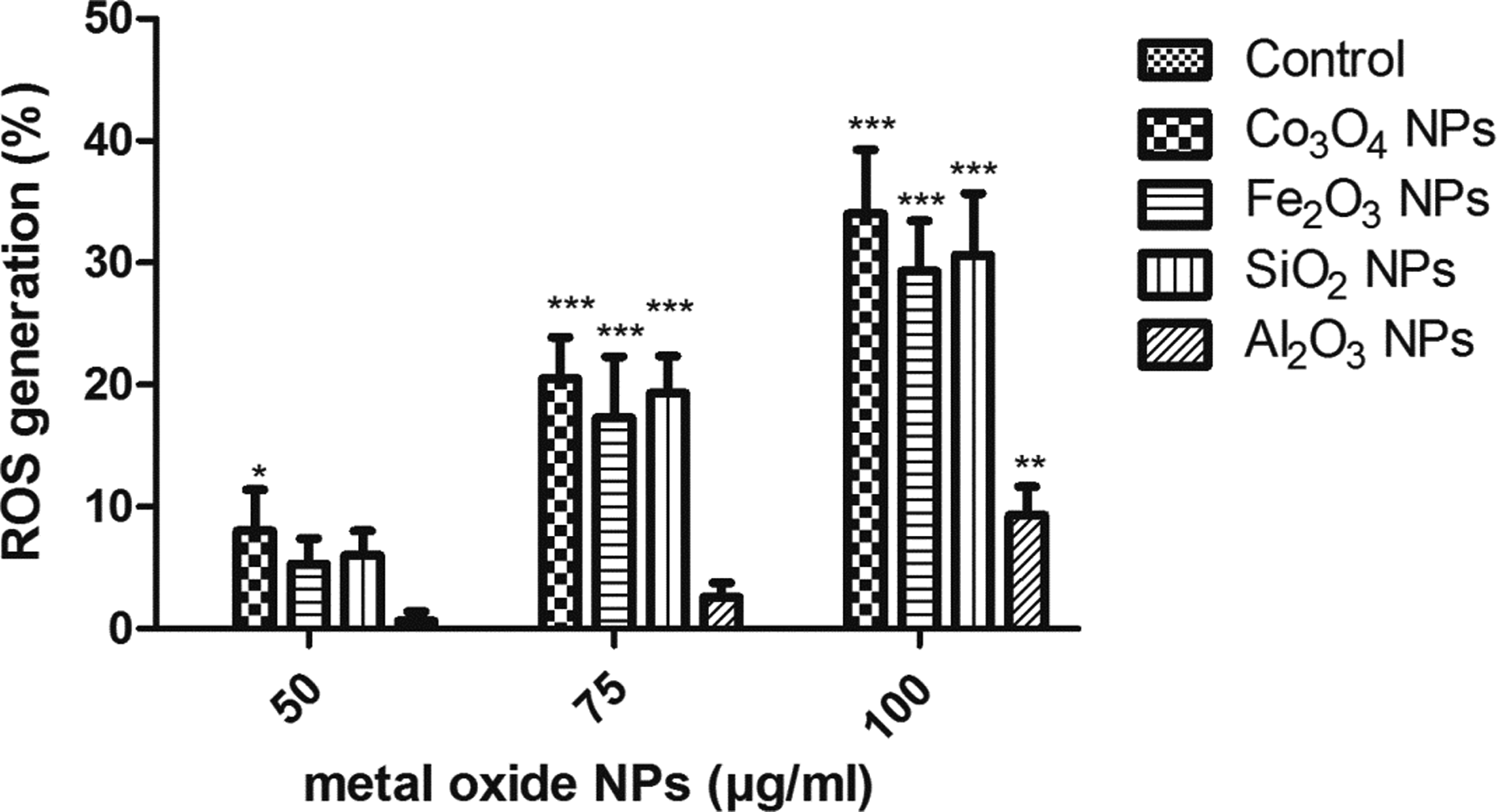
Oxidative stress estimated in human lymphocytes by measuring the ROS level. The percentage of ROS release for untreated control cells is 0%. The data obtained are expressed in terms of mean ± SD. *p < 0.05: the significance versus control; **p < 0.01: the significance versus control; ***p < 0.001: the significance versus control. ROS: reactive oxygen species.
Alteration in the level of LPO and antioxidant enzymes
The oxidative stress was evaluated in human lymphocytes by measuring the level of LPO and antioxidant enzymes like catalase, GSH, and SOD (Table 1). The higher production of intercellular ROS and reduced antioxidant level is the primary reason for toxicity due to metal-oxide NPs. 31 The Co3O4 NPs when treated with human lymphocytes showed a significant increase in LPO level and reduction in catalase and SOD level at 50, 75, and 100 µg/ml. An insignificant reduction in GSH level (p > 0.05) was observed at 50 µg/ml while a significant reduction was observed in GSH level at 75 and 100 µg/ml (p < 0.001), indicating that the oxidative stress created by Co3O4 NPs was higher when compared with all the three NPs used for this study.
Alterations in the level of LPO and antioxidant enzymes in human lymphocytes.a

LPO: lipid peroxidation; H2O2: hydrogen peroxide; MDA: malondialdehyde; GSH: reduced glutathione; SOD: superoxide dismutase; NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide.
aThe data obtained are expressed in terms of mean ± SD.
b p < 0.01: the significance versus control.
c p < 0.001: the significance versus control.
d p < 0.05: the significance versus control.
Interestingly, both Fe2O3 and SiO2 NPs at 75 and 100 µg/ml showed a significant reduction in catalase and GSH level after 24 h of exposure to the human lymphocytes in a dose-dependent manner. The Fe2O3 NPs at 75 and 100 µg/ml showed a significant increase in LPO level and a significant reduction in SOD level (p < 0.001). The SiO2 NPs showed an insignificant increase in LPO and a decline in SOD level at 50 µg/ml (p > 0.05). The least alteration in the level of antioxidant enzymes was observed with Al2O3 NPs. The Al2O3 NPs showed an insignificant reduction in GSH and SOD level at 50 and 75 µg/ml (p > 0.05) but at 100 µg/ml the GSH level was found to be significant (p < 0.05). The Al2O3 NPs had an insignificant increase in LPO level at 75 and 100 µg/ml (p > 0.05). The oxidative stress was evidenced by the induction of ROS, LPO, and depletion of catalase, GSH, and SOD in human lymphocytes in time and dose-dependent manner.
Tail DNA migration
Single-cell gel electrophoresis or comet assay was a reliable assay to measure the genotoxicity of any compound. The comet assay was a simple, most reliable, and inexpensive method to detect DNA damage in individual cells when treated with various chemicals, radiation, and drugs. This technique requires only few cells for obtaining the results within few hours. 32 The obtained results from the comet assay indicated that Co3O4 NPs and Fe2O3 NPs were able to cause significant and dose-dependent increase in percentage (%) tail DNA damage compared with control (p < 0.001). Tail DNA damage of 10.1% and 9.3% was observed when exposed for 24 h to Co3O4 NPs and Fe2O3 NPs at 100 µg/ml, respectively (Table 2). The SiO2 NPs showed an insignificant increase of 3.4% tail DNA at 100 µg/ml (p > 0.05). The highest % tail DNA was observed in Co3O4 NPs and the least % tail DNA was observed in Al2O3 NPs (Figure 8). The Al2O3 NPs showed an insignificant increase of 2.8% tail DNA at 100 µg/ml (p > 0.05).
Tail DNA migration in human lymphocytes treated with Co3O4, Fe2O3, SiO2 c, and Al2O3 d NPs.a

NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide; LDH: lactate dehydrogenase leakage.
aThe % tail DNA data obtained are expressed in terms of mean ± SD.
b p < 0.001: the significance versus control.
c p < 0.05: the significance versus control.
d p < 0.01: the significance versus control.
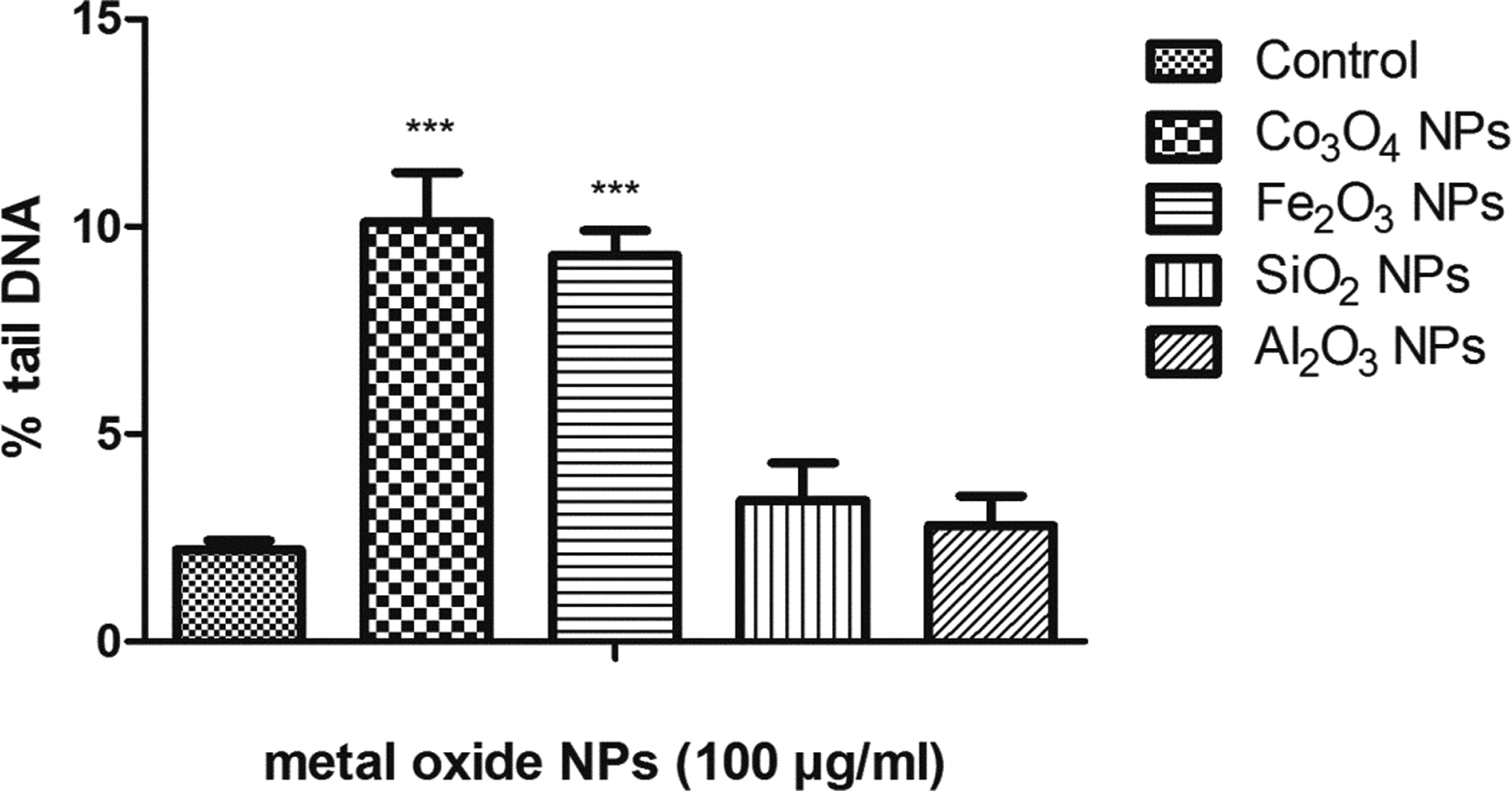
DNA damage (% tail DNA migration) in human lymphocytes. (a) Control; (b) Co3O4 NPs; (c) Fe2O3 NPs; (d) SiO2 NPs; (e) Al2O3 NPs; and (f) percentage tail DNA. The data obtained are expressed in terms of mean ± SD. *p < 0.05: the significance versus control; **p < 0.01: the significance versus control; ***p < 0.00: the significance versus control. NP: nanoparticle; Al2O3: aluminum oxide; Co3O4: cobalt (II, III) oxide; Fe2O3: iron (III) oxide; SiO2: silicon dioxide; LDH: lactate dehydrogenase leakage.
Chromosomal aberrations
A minimum of 50 metaphases per sample were scored for the chromosome aberration assay. When compared with control, an insignificant increase (p > 0.05) in chromosomal aberrations was observed when exposed to SiO2 and Al2O3 NPs (p > 0.05). The Co3O4 NPs (p < 0.001) and Fe2O3 NPs (p < 0.05) showed a significant increase in chromosomal aberration when compared with control at 100 µg/ml (Figure 9).

Chromosomal aberration in metaphase chromosomes of human lymphocytes: (a) control; (b) chromosome break; and (c) chromosome deletion. (d) The graphs show that data obtained are represented in terms of mean ± SD. Chromosomal aberrations, *p < 0.05: the significance versus control; **p < 0.01: the significance versus control; ***p < 0.001: the significance versus control.
Discussion
The use of NPs has been increased in various fields, and it was very important to investigate the toxic effects of the NPs in human health. The toxicity of NPs was mainly due to the structure, solubility, shape, and surface/mass ratio. 33 The hydrodynamic size of the NPs was found to be larger due to the agglomeration of the NPs in the prepared culture medium, and the agglomerates formed are greater when compared with the primary particle size. 34 The release of Co2+ ions was the main reason for the aggregation and toxicity of the Co3O4 NPs. The Co3O4 NPs form agglomerates of different size due to its magnetic properties in the cell culture medium. 31 The amine-modified Fe2O3 NPs were able to agglomerate in the cell culture medium but failed to induce cytotoxicity, whereas the carboxylated Fe2O3 NPs induce greater cytotoxicity and redox-related gene expression in the human lymphocyte cells. 35 The surface charge and ionic strength are the main reasons for agglomeration of the SiO2 NPs in the lymphocyte culture medium. 36 The addition of FBS has effects on the stability of Al2O3 NPs. 37 The lymphocyte culture medium contains amino acids, vitamins, and salts that cause decrease in electrostatic repulsive forces and increase in ionic strength forming NPs aggregation. The serum contains proteins that prevent the aggregation of the NPs by creating a protein corona. The agglomeration of NPs was lower at 10% FBS than the serum-free media. 38 The agglomeration of NPs was less in 10% FBS than the serum-free media. 31
The in vitro toxicity testing is an alternative testing method instead of animal experiments to study the nanotoxicity of the NPs. 30 Direct contact of the NPs to the human lymphocytes has been implicated to produce damage to mitochondria by influencing the electron flow and altering the mitochondrial functionality. The NPs damage the cells, and stimulate the release of cytoplasmic LDH, which further catalyzes the conversion of the tetrazolium salt to formazan. 39 The internalization and easy penetration of NPs into the cells were found to be the main reasons regarding the size-dependent toxic effects of NPs. 40 The toxicity in human lymphocytes was mainly due to the pathway that involves the endocytosis of NPs agglomerate. 41,42
The previous toxicity studies done have reported that cytotoxic effects and alteration in the level of oxidative stress biomarkers were observed after 24 h of exposure to the NPs. Similar results were obtained in this study in a time and dose-dependent manner. The human lymphocytes showed alteration in the level of oxidative stress biomarkers and antioxidant level after 24 h of exposure to the metal-oxide NPs. 43 Oxidative stress occurs when ROS disturbs the balance between oxidative and antioxidant defense levels. The ROS was due to the superoxide radical, H2O2, and the hydroxyl radical, which causes DNA damage and apoptotic cell death. The cytotoxicity observed with NPs might be due to the pro-inflammatory effects through ROS-mediated mechanism. 44,45 The Co3O4 NPs enter the cell, and they remain in the cytoplasm causing the induction of ROS. 46 The LPO was due to the oxidative degradation of polyunsaturated fatty acids, and it causes membrane damage and structural integrity to the human lymphocytes. The LPO was assessed by measuring the level of MDA, the final product of lipid breakdown. 47 The antioxidants like catalase, GSH, and SOD protect the human lymphocytes from oxidative damage because of their ability to scavenge ROS such as superoxide anion and hydroxyl radical. The free radicals are controlled by the antioxidant enzymes by maintaining a balance between the prooxidant and antioxidant process. 19 The Fe2O3 NPs generate redox cycling resulting in intracellular iron overload, and large surface area of the NPs promotes the generation of ROS. The Fe2O3 NPs showed an enhanced MDA level and reduced GSH level in rats similar when exposed to human lymphocytes. 48 When exposed to SiO2 NPs, the excessive ROS can cause injuries and impair the function of lymphocytes by aggravating the effect of LPO and damaging the intracellular components of a cell completely including its proteins, lipids, and DNA. 49 Oxidative stress and alteration in the level of antioxidant enzymes observed with alumina NPs are mainly due to the pro-inflammatory effects through ROS-mediated mechanism. 50
The oxidative stress disturbs the cellular macromolecules such as lipids, proteins, and DNA when exposed to NPs. The DNA damage occurs mainly due to mutations, deletions, cross-linking, and formation of adducts leading to apoptosis. 51,52 The caspases play a significant role in the initiation and execution of apoptosis causing cellular DNA damage by Co3O4 NPs in human lymphocytes. 31 The Fe2O3 NPs cause chromosomal DNA fragmentation and nuclear condensation in human lymphocytes. The Fe2O3 NPs arrest the G0/G1 phase in a dose-dependent manner. 53 The genotoxicity of SiO2 NPs was due to chromatin modification, ROS induction, and DNase liberation from the lysozyme. The cellular production of ROS can cause damage to cellular macromolecules such as DNA. 54 Studies done in female rats with Al2O3 NPs −30 nm, and 40 nm did not increase the % tail DNA in rat peripheral human lymphocytes after oral exposure. 55 The Al2O3 NPs do not cause DNA damage in human peripheral lymphocytes when compared with the other three NPs.
The chromosomal aberration is a genetic marker to study the genotoxicity risk of human leukocytes. Alteration in the chromosome could be any changes in either the number of chromosomes or the structure of chromosome. The chromosomal aberrations like break and deletion were observed after exposed to different NPs. Chromosomal aberrations were believed to be an important aspect resulting in cell cycle arrest, and it was an important marker for cancer risk. 56 The Co3O4 and Fe2O3 NPs were able to cause chromosome break and deletion leading to deleterious effect on humans. The penetration of metal ions into the cell causes cross-link in the DNA, sister chromatid exchange, and mutations leading to cancer risk when exposed to metal-oxide NPs. 57,58
The possible mechanism that induces cytotoxicity and genotoxicity when exposed to the metal-oxide NPs is shown in Figure 10. The metal-oxide NPs were able to generate ROS and oxidative stress by the mitochondrial dysfunction in human lymphocytes. The ROS generation, a by-product of oxidative phosphorylation, causes damage in mitochondrial macromolecules including the mitochondrial DNA leading to mutation and cancer in humans. The NPs cause chromosomal aberrations and DNA damage, causing cell cycle arrest and finally to cell death. 59 The imbalance in the ROS production when exposed to the metal-oxide NPs caused DNA damage by the oxidation of purine molecules. The metal-oxide NP-treated cells showed mitotic arrest, chromosomes break, and deletion in human lymphocytes. 57,60 The results have proven that the metal-oxide NPs were able to cause DNA damage and chromosomal aberration due to the generation of ROS, which leads to cell death in human lymphocytes.

Possible mechanism of metal-oxide NPs when exposed to human lymphocytes. NP: nanoparticle.
Conclusion
The cytotoxicity and genotoxicity studies revealed that the Co3O4 NPs were found to be more toxic to human lymphocytes when compared with other metal-oxide NPs. The least toxicity was observed in the case of Al2O3 NPs. When exposed to Co3O4 NPs and Fe2O3 NPs, the human lymphocytes exhibited increase in genotoxicity when compared with SiO2 and Al2O3 NPs. The cytotoxicity and oxidative stress lead to DNA damage and chromosomal aberrations in human lymphocytes. The results highlight the toxicity associated with the exposure of the NPs and suggest that proper attention needs to be paid regarding the use of the NPs since it can cause harmful effects to humans.
Footnotes
Acknowledgement
The authors are thankful to the VIT University management for funding the work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
