Abstract
It is widely known that not only the gonadal estradiol (E2) but also hippocampal E2 plays an essential role in memory process. However, the role of hippocampal E2-enhanced memory mechanism during aging is largely unknown. The aim of the present study was to investigate the effect of age on E2 concentration, the expression level of its receptors, and key steroidogenic enzymes in hippocampus. We also investigated the effect of microglia activation on E2 synthesis in hippocampal neurons. The results showed that serum E2 was higher in 19-month-old (aged) rats, which exhibited spatial memory decline in the Morris water maze (MWM) test when compared to the younger rats. Hence, serum E2 may not be associated with the reduced spatial memory performance in aging. In contrast, the level of E2 and the expressions of its receptors were significantly decreased in hippocampus of aged female rat compared to younger females. Furthermore, the expressions of key hippocampal steroidogenic enzymes, steroidogenic acute regulatory protein (StAR), and cytochrome P450 (P450) also significantly decreased with age, which resulted in lower hippocampal E2 levels. In addition, we found that the microglia of aged brain highly expressed interleukin 6 (IL-6), which directly inhibited E2 synthesis in hippocampal neurons via suppression of P450 synthesis. Taken together, we summarized that the microglia-derived IL-6 inhibited hippocampal E2 synthesis in aged rats which, in turn, contributed to the deficit of spatial memory performance.
Introduction
Cognitive impairment occurs gradually and becomes progressive with age. Aged female rats, approximately 21 to 24 months old, showed impairment on a number of spatial memory tasks. 1 –3 In rats, long-term administration of estradiol (E2) enhanced spatial memory only during the initial period of 3 months but not 10 months after ovariectomization. 4 This indicated that the beneficial effect of E2 treatment is age dependent. Moreover, exogenous E2 treatment could not restore the number of spine density in the brain of 22- to 23-month-old female rats. 5 However, the mechanisms underlying age-related changes in the efficacy of E2 treatment are still unclear. Several studies reported that age-related decline in the E2 efficacy is result of a prolonged lack of systemic E2. 5 –9 The brain itself, especially hippocampus, is also a key site of E2 synthesis with a higher level (6-10 times) than serum E2. 10 Hippocampal-derived E2 has been shown to be essential for synaptic plasticity and memory formation. 11 Recent studies demonstrated that aromatase treatment abolished long-term potentiation (LTP) and decreased the number of spine synapses in ovariectomized (OVX) mice. 12–13 Our previous study demonstrated a priming role of hippocampal E2 on the induction of estrogen receptor (ER) β expression in hippocampal neurons, which is an important mechanism of exogenous E2-induced synaptic plasticity. 14 These evidences indicated that hippocampal E2 plays an important role in hippocampus-dependent memory processing. However, it is unknown whether the E2 response is influenced by endogenous E2 level, especially hippocampal E2, in aged brain.
The OVX animal is a widely used animal model for studying the mechanism of age-related memory impairment in women, as it has been shown to express similar pathophysiological features of estrogen deficiency-induced memory loss as occurring in menopausal women. 7–8,13 However, rodents exhibit regulatory mechanisms that are entirely different from humans. For example, aged rats showed a higher increase in serum E2 level than younger rats. 15–16 Therefore, extrapolation of mechanisms of ovarian steroid-related aging characteristic of rodents to humans is somewhat limited. Furthermore, the majority of studies on age-related memory impairment have used OVX rats ranging in age from 8 to 13 months old. 4,17 While many studies have been conducted to examine the effect of E2 deficiency on memory impairment, the influence of age (time) on memory impairment is currently unknown.
A number of studies have shown that brain volume declines with age at the end of adolescence, 12 months old rat. 18 Since brain shrinkage occurs before gradually decreasing levels of estrogen, there may be other factors that have influence on the aging brain besides the decreasing E2. Spontaneous brain inflammation, characterized my microglia activation, is another possible contributor to age-related memory impairment since it is amplified and prolonged in the aged brain. 19–20 Activated microglia-derived neurotoxic factors, including interleukin 6 (IL-6), suppress synaptic plasticity and hippocampal-dependent memory formation. 21 –23 Although our previous study revealed that IL-6 inhibited estrogen synthesis in an in vitro model of cultured hippocampal neurons, 24 the exact mechanisms of action are still unknown. Therefore, in the present study, we aimed to investigate the amount of E2, expression of ER, expression of steroidogenic enzymes, and activation of microglia in age-related spatial memory impairment on female rats. Furthermore, we investigated the effect of a microglia-derived cytokine, IL-6, on the biosynthesis of E2 in cultured hippocampal neurons.
Material and Methods
Animals
A total of 25 female virgin Sprague Dawley rats at different ages were purchased from National Laboratory animal Center, Mahidol University, Thailand. The rats were grouped according to ages (n = 5) as follows: 2 months (average weight 217.6 g), 5 months (average weight 288.94 g), 10 months (average weight 325.08 g), and 19 months (331.9 g). Vaginal smear was monitored daily in the morning from 2 to 19 months of age. Diestrus rats were selected. In 19-month-old group, 6 rats had regular estrus cycle. Aged rats (19 months old) received daily subcutaneous injection of 0.5 mg/mL E2 for 7 days. The animals were housed a humidity- and temperature-controlled room with a constant 12-hour light–dark cycle (lights on from 7
Spatial Memory Task
The spatial memory was assessed in the Morris water maze (MWM), a measure of hippocampally dependent spatial navigation and reference memory. The maze was a black circular pool, measuring 153 cm in diameter with a height of 60 cm. The water level was 30 cm, and its temperature was set at 22°C ± 2°C. The pool was surrounded with visual cues (such as triangle, circle, and square) and divided into 4 quadrants. A hidden circular platform (10 cm in diameter) was placed in a nonvisual cue quadrant so that its surface was 2 cm below water level. Animal behavior was recorded by a video camera.
The test was done as described in a previous study with slight modifications. 25 Training (acquisition) trials started the day after habituation days. Training took 7 consecutive days, and each day consisted of 4 trials. In each trial, the rat was placed in the pool from one of the determined points of visual cue. If the rat could find the platform within a time span of 60 seconds, it was allowed to stay on it for 10 seconds; otherwise, it was guided to the platform and remained there for 10 seconds. The time that a rat could find the platform was recorded (escape latency). The rats were then towel dried and kept in a heated cage. The intertrial time was 3 minutes for each rat.
For testing, rats were allowed to swim in the pool for 60 seconds while the platform was removed. The total distance to the target platform (center of the previous position of the platform) was recorded during this period. This total distance is called search error which is a good reflection for spatial learning in MWM test.
H19-7 Hippocampal Neurons
To study the effect of IL-6 on steroidogenesis in neuron, rat embryonic hippocampal origin (H19-7) cells were grown on poly-L-lysine (PLL)-coated well plates (or coverslips), in Dulbecco’s modified eagle’s medium (DMEM) at 33°C as described previously. 14 H19-7 hippocampal cells were transformed with a temperature-sensitive mutant of the simian virus 40T antigen. During the differentiation process, cells were incubated at 39°C in DMEM with N2 supplements (Invitrogen, Carlsbad, California), and 10 ng/mL basic fibroblast growth factor (Sigma-Aldrich, St Louis, Missouri) was added to accelerate the formation of neuronal processes. Recombinant mouse IL-6 (R&D system Inc Minneapolis, Minnesota, USA) was added at a final concentration of 0.5 ng/mL.
Radioimmunoassay for Serum E2
The total E2 levels in serum were determined by Central Laboratory, Faculty of Medicine, Burapha University, using radioimmunoassay (RIA). This assay is based on a competitive RIA method. The basic principle of RIA is that there is competition between a radioactive and a nonradioactive antigen for a fixed number of antibody binding sites. In brief, 200 µL of serum extract was incubated with 100 µL of rat antibody against E2 for 4 hours at 4°C. [I125]-estradiol was added and incubated for 24 hours at 4°C. Proteins were precipitated and separated after centrifugation, and radiation was counted using a gamma counter. The amount of [I-125]-labeled estradiol bound to the antibody is inversely proportional to the concentration of unlabeled estradiol present. The separation of free and bound antigen is achieved using a double antibody system. The estradiol values of the samples were directly read against this standard curve. The sensitivity of the test for estradiol was 3.0 pg/mL.
Enzyme-Linked Immunosorbent Assay for Serum Anti-Mullerian Hormone and E2 in Brain Lysates
To assess the ovarian function in aged animal, anti-Mullerian hormone (AMH) level in serum and E2 in cerebral cortex or hippocampus lysates was measured by enzyme-linked immunosorbent assay (ELISA) using AMH/MIS (Immunotech-Coulter, Marseilles, France) and estradiol ELISA kit (Biochemica GmbH, Germany), respectively. The sample was pipetted into micro-assay wells, precoated with monoclonal antibody, followed by incubation at 37°C for 30 minutes. Washing buffer was used to remove excess binding before addition of the horseradish peroxidase (HRP)-conjugated reagent to each well. To develop the reaction, a chromogenic substrate was added and incubated at 37°C for 15 minutes in the dark. The reaction was stopped by the addition of 50 µL stop solution into each well. Absorbance was monitored at 540 nm using a microplate reader, and samples were quantified by interpolation based on a standard curve.
Immunohistochemical Localization of Cytochrome P450
Rat hippocampal tissues were dissected and fixed overnight at 4°C in 0.1 mol/L phosphate-buffered saline (PBS) containing 4% w/v paraformaldehyde. After being dehydrated and cleared by graded ethanol and xylene, respectively, they were embedded in paraffin. Coronal sections were cut at 5-μm thickness; these were later incubated at 37°C for 30 minutes in antigen retrieval solution (0.01 mg/mL proteinase K, 50 mmol/L Tris–HCl pH 8.0 and 5 mmol/L EDTA). Thereafter, the sections were incubated for 1 hour with 3% H2O2 to inhibit endogenous peroxidase activity. Nonspecific bindings were blocked by 2-hour incubation with 4% bovine serum albumin, 10% normal goat serum, and 0.7% Tween-20 in PBS. The sections were then incubated at 4°C overnight with 1:50 rabbit polyclonal antibody against P450 (CPY11A isoform) cholesterol side chain cleavage enzyme (p450scc; Merck Millipore, Billerica, Massachusetts). After being washed with 0.7% Tween-20 in PBS, the sections were incubated for 1 hour at room temperature with biotinylated goat antirabbit immunoglobulin G (Zymed, South San Francisco, California) at 1:500 dilution, followed by 1-hour incubation with streptavidin-conjugated HRP solution (Zymed) and 3,3′-diaminobenzidine chromogen (Pierce, Rockford, Illinois). For negative controls, the sections were incubated with 0.7% Tween-20 in PBS in the absence of primary antibody. Finally, the sections were counterstained with hematoxylin (Sigma) and examined under light microscope.
Western Blot
Hippocampus of 2-, 5-, 10-, and 19-month-old rats were removed and lysed using Pierce Ripa Buffer (Thermo Fisher Scientific Inc, Rockford, Illinois), and equal amounts of protein (30 µg) were separated on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membranes. The membranes were incubated with the following primary antibodies at a dilution of 1:1000: mouse anti-ERα, mouse anti-ERβ, mouse anti-steroidogenic acute regulatory protein (StAR), rabbit anti-P450 (CPY11A isoform), mouse anti-IL-6 mouse, mouse anti-major histocompatibility complex (MHC)-II, and mouse anti-β-actin (Merck Millipore). Membranes were probed with HRP-conjugated secondary antibodies (Zymed) and detected with Thermo Scientific SuperSignal West Pico Substrate (Thermo Fisher Scientific Inc) and captured on CL-Xposure Film (Thermo Fisher Scientific Inc).
Quantitative Real Time-Polymerase Chain Reaction
H19-7 cells were plated onto 6-well plate at a density of 1 × 106 cells/well. Total RNA was extracted using the RNeasy Mini kit (Qiagen, Hilden, Germany), and 2 µg of RNA was converted to complementary DNA (cDNA) with the high-capacity cDNA reverse transcription kit (Applied Biosystems, Foster City, California). The quantitative real time-polymerase chain reaction (qRT-PCR) was performed with the PRISM 7700 Sequence Detection System (Applied Biosystems) according to the manufacturer’s protocol. TaqMan Gene Expression Assays with FAM dye-labeled TaqMan MGB probes (Applied Biosystems), which contained customized primers, were utilized for qRT-PCR analysis. A target gene, P450 messenger RNA (mRNA; CYP11A), was analyzed in this study. Glyceraldehyde 3-phosphate dehydrogenase was used as a positive control. The relative expression of mRNA was calculated using the comparative Ct method with SDS software v.1.3.1 (Applied Biosystems).
Data Analysis
All data were expressed as mean ± standard error of the mean. Data were representative of 5 independent experiments. One-way analysis of variance with Tukey posttest was employed for comparing multiple sets of data. The level of significance of all statistical tests was P < 0.05. All data were analyzed using GraphPad Prism version 5.0 for Windows (GraphPad Software Inc, San Diego, California).
Results
Serum Hormonal Level of AMH and E2
To assess the ovarian function in aged animal, serum level of AMH, a marker for ovarian aging, was measured. The results showed that AMH level significantly increased in 5- and 10-month-old rats when compared to 2-month-old rats (P = .0496 and P = .0036) and significantly decreased in 19-month-old rats when compared to 10-month-old rats (P = .0113; Figure 1A), which indicated ovarian dysfunctions in 19-month-old rats. For serum E2, RIA study showed that E2 level was higher in the serum of female rat aged 10 to 19 months when compared to that in 2-month-old rats (P = 0.0051 and P = 0.0082; Figure 1B). Moreover, the level of E2 in estrogen-treated aged rats did not differ from untreated group (Figure 1B). These results indicated that plasma E2 was not associated with ovarian function which might be controlled by a regulatory mechanism entirely different from humans.
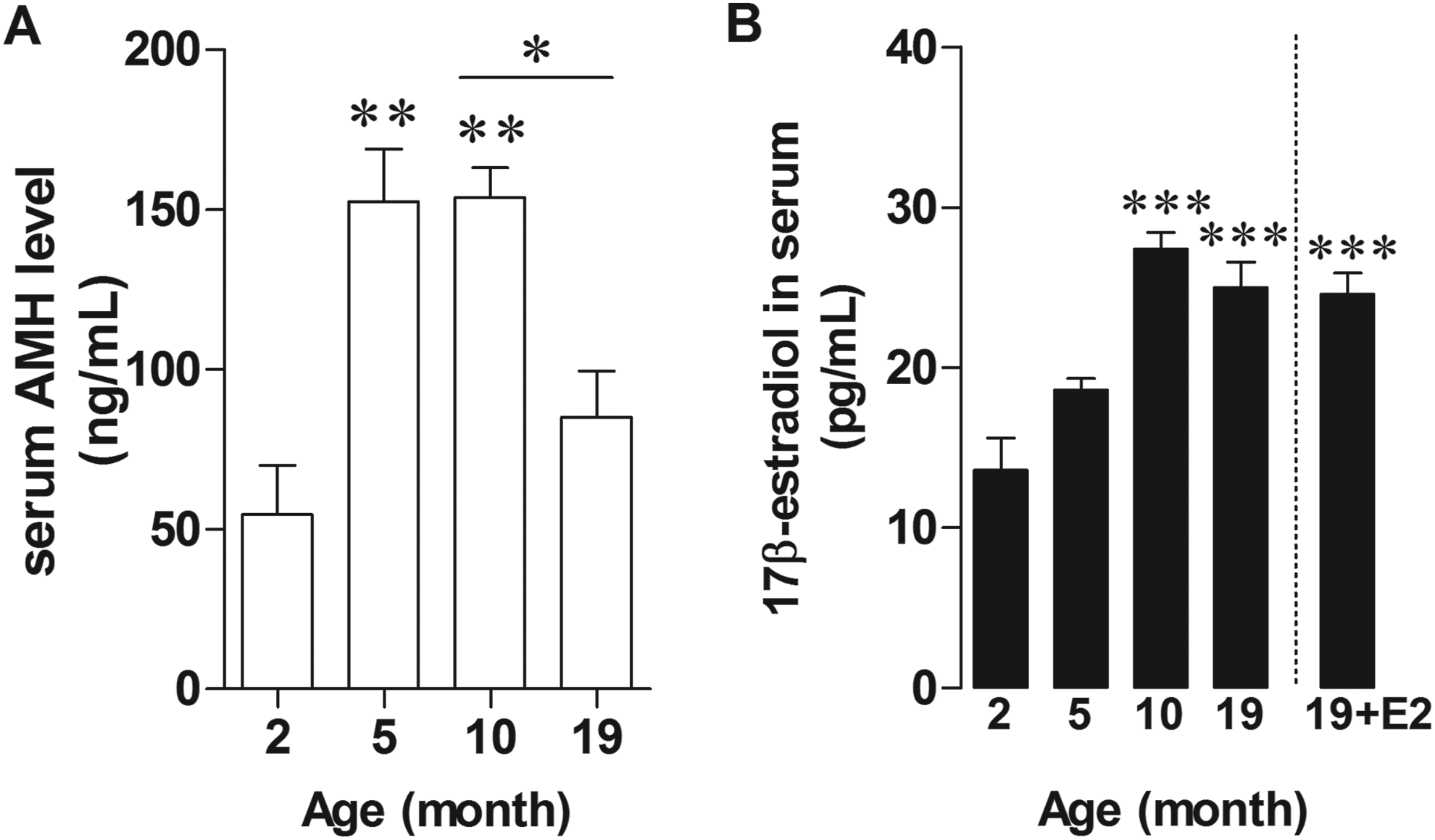
Serum concentration of AMH (ng/mL) and E2 (pg/mL) in female rats aged 2, 5, 10, 19 months old. Graphs of AMH level (A) and E2 level (B) in serum of untreated or E2-treated rats. *P < 0.005; **P < 0.0005; ***P < 0.0001 versus the 2-month-old group, n = 5. AMH indicates anti-Mullerian hormone.
Spatial Memory Impairment in Aged Female Rats Cannot be Restored by Administration of Exogenous E2
To test the effect of age on spatial memory performance, 2-, 5-, 10-, and 19-month-old female rats were assessed using MWM. The results showed that 19-month-old rats had significantly longer escape latencies in the memory trial of the 1-day (P = 0.0016; Figure 2A) and 7-day (P = 0.0038; Figure 2B) water maze test than 2-month-old rats. This strongly indicates the impairment of long-term memory in 19-month-old rats. Time latency in the estrogen-treated 19-month-old rats was slightly lower than in age-matched group, but the difference was not significant (P = .5124). Thus, exogenous E2 could not restore memory impairment in aged female rats. There was no significant difference in performance among the group on the probe test for time to find target (Figure 2C). No significant differences were observed in the swimming speed (data not shown).
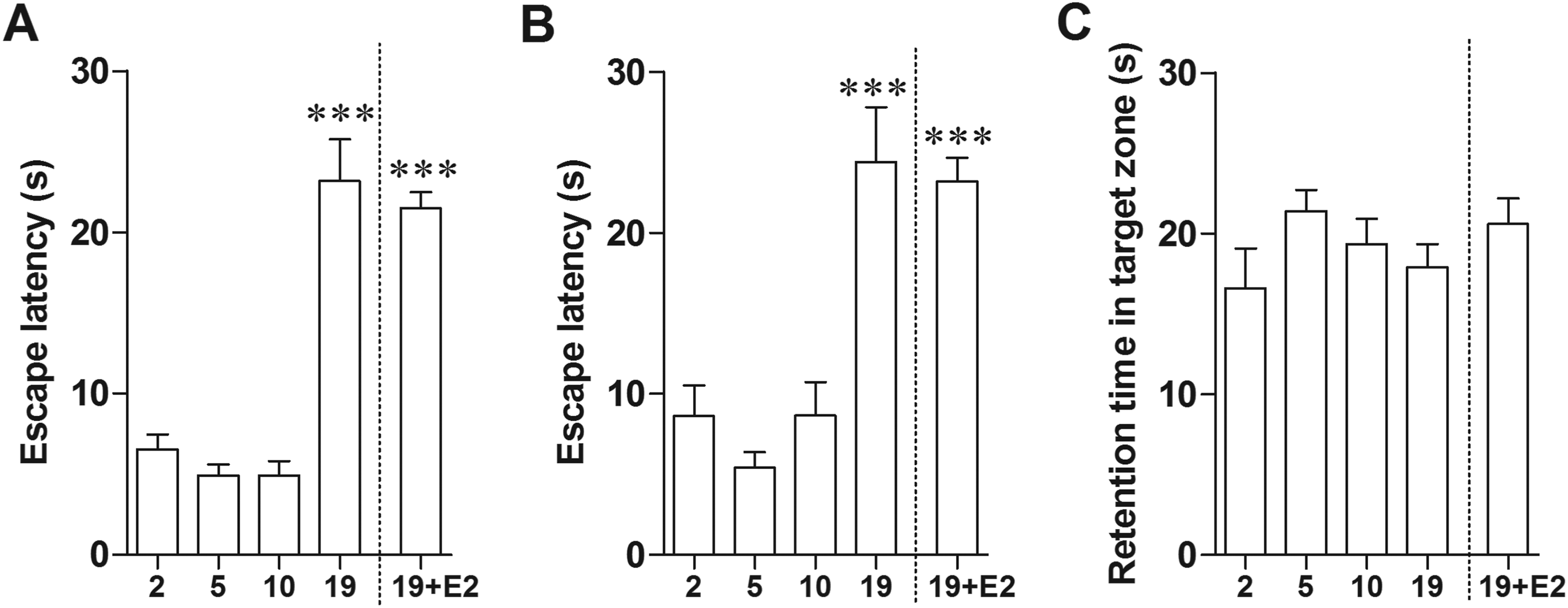
Spatial memory performance decreased in aged rats. Estrogen-treated aged rats did not perform differently from untreated aged rats. Bars represent the mean percent of escape latency of day 1 (A) and day 7 (B) as well as mean percent of retention time (C). ***P < 0.0001 versus the 2-month-old group, n = 5.
The Level of E2 in Hippocampus and Cerebral Cortex of Aged Female Rats
Since serum E2 was not associated with the reduced spatial memory performance, the concentrations of E2 in hippocampus and cerebral cortex of female rats were determined using ELISA technique. The results revealed that E2 levels were significantly increased in hippocampus of 5- and 10-month-old female rats compared to 2-month-old rats (P = 0.0234 and P = 0.0056; Figure 3A). The 19-month-old rats showed a significant decrease in the level of hippocampal E2 compared to the 10-month-old rats (P = 0.0354). In contrast, the level of E2 in cerebral cortex was also slightly decreased in the 19-month-old rats, although this decrease was not statistically significant (P = 0.2760; Figure 3B). These findings suggested the regional variations in E2 concentration in the brain during aging.
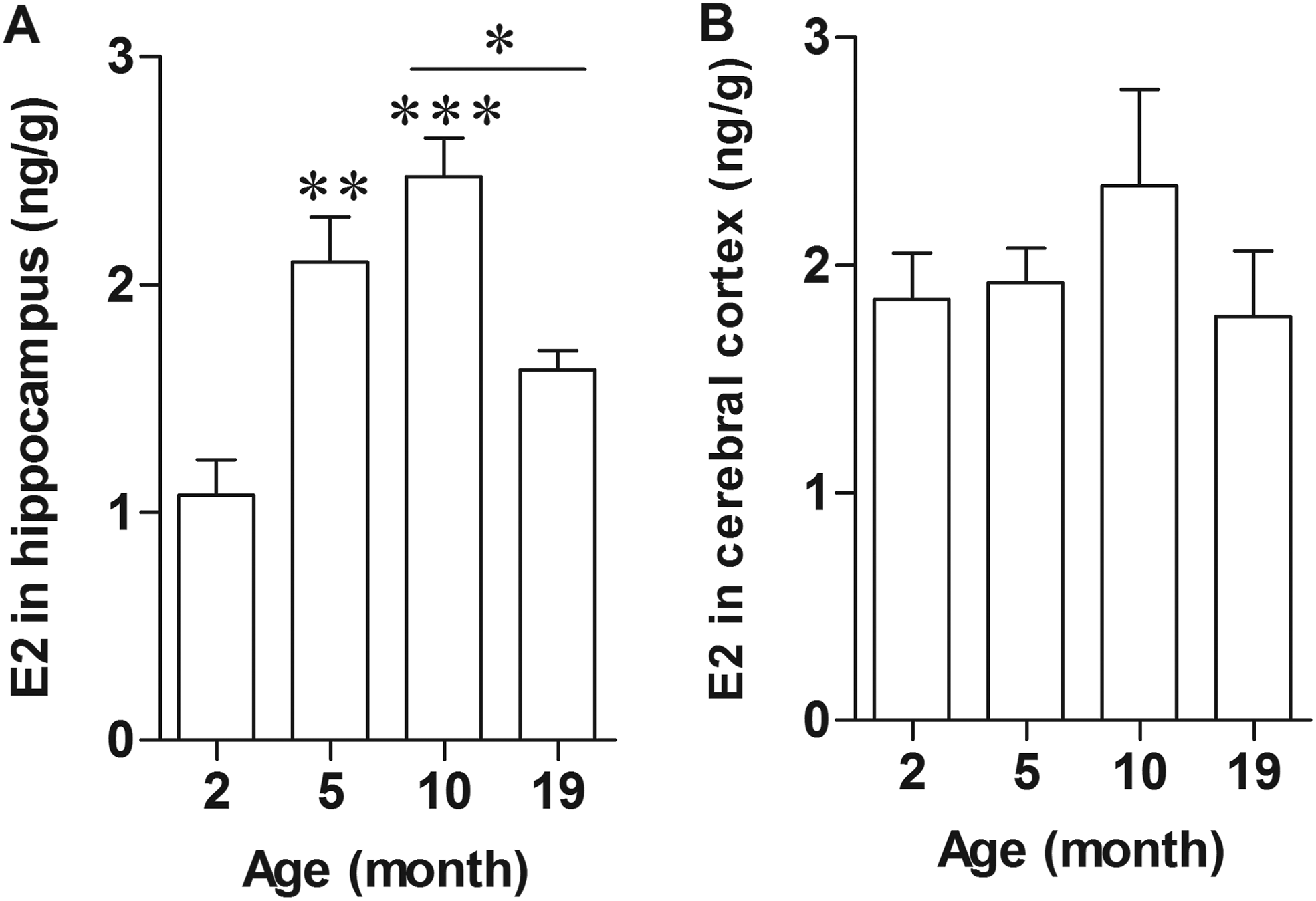
Level of E2 in hippocampus but not cerebral cortex decreased in aged rat. Graphs of E2 level in lysate of hippocampus (A) and cerebral cortex (B) of 2-, 5-, 10-, 19-month-old female rats. *P < 0.005; **P < 0.0005; ***P < 0.0001 versus the 2-month-old group, n = 5.
Age-Related Memory Impairment is Associated With Significant Alterations in Steroid-Synthesizing Enzymes in Hippocampus
In addition to the level of E2 in hippocampus, we also determined the expression of the essential estrogen biosynthetic enzymes, StAR and P450, in hippocampus of female rats, as these enzymes are critically important in steroidogenesis. StAR initiates steroidogenesis by promoting the transfer of cholesterol to the inner mitochondrial membrane, and P450 converts cholesterol to pregnenolone, which is the main precursor of other steroid hormones. The results showed that the expression of StAR significantly increased in 5- and 10-month-old rats compared to 2-month-old rats (P = 0.0458 and P = 0.0339; Figure 4A and B). Similar to hippocampal E2 level, StAR expression significantly decreased in 19-month-old rats compared to 10-month-old rats (P = 0.0099). P450 expression in hippocampus showed similar pattern with StAR (Figure 4A and C). Increase in P450 immunoreactivity was observed in hippocampus of 10-month-old rats when compared to 5- and 19-month-old rats (Figure 5A-I). In immunohistochemical detection, we did not include the picture of 2-month-old rat, as there was an obvious knife mark in the tissue section. Nevertheless, the expressions of the 2 steroid-synthesizing enzymes had already been shown more clearly in Figure 4. Moreover, we applied morphometric analysis on the hippocampus sections immunostained with P450 antibody. The results revealed the smaller hippocampus, mostly due to degeneration, in 19-month-old rats when compared to that of 5-month-old rats (Figure 5J-K.).
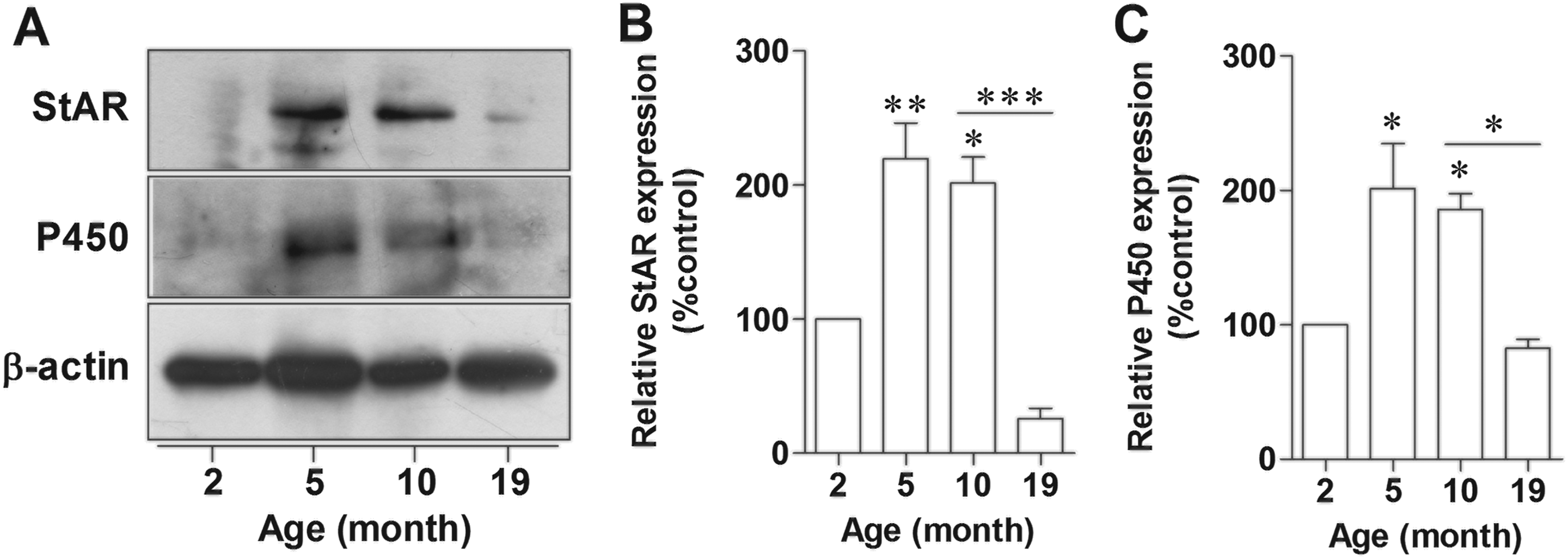
The expression of StAR and P450 in female rat hippocampus decreased in aged rat. Representative immunoblotting of StAR and P450 in hippocampus of 2-, 5-, 10-, 19-month-old female rats (A). Densitometric analyses of StAR (B) and P450 (C) normalized to β-actin at each age of female rats. *P < 0.005; **P < 0.005; ***P < 0.0001 versus the 2-month-old group, n = 5. StAR indicates steroidogenic acute regulatory protein.
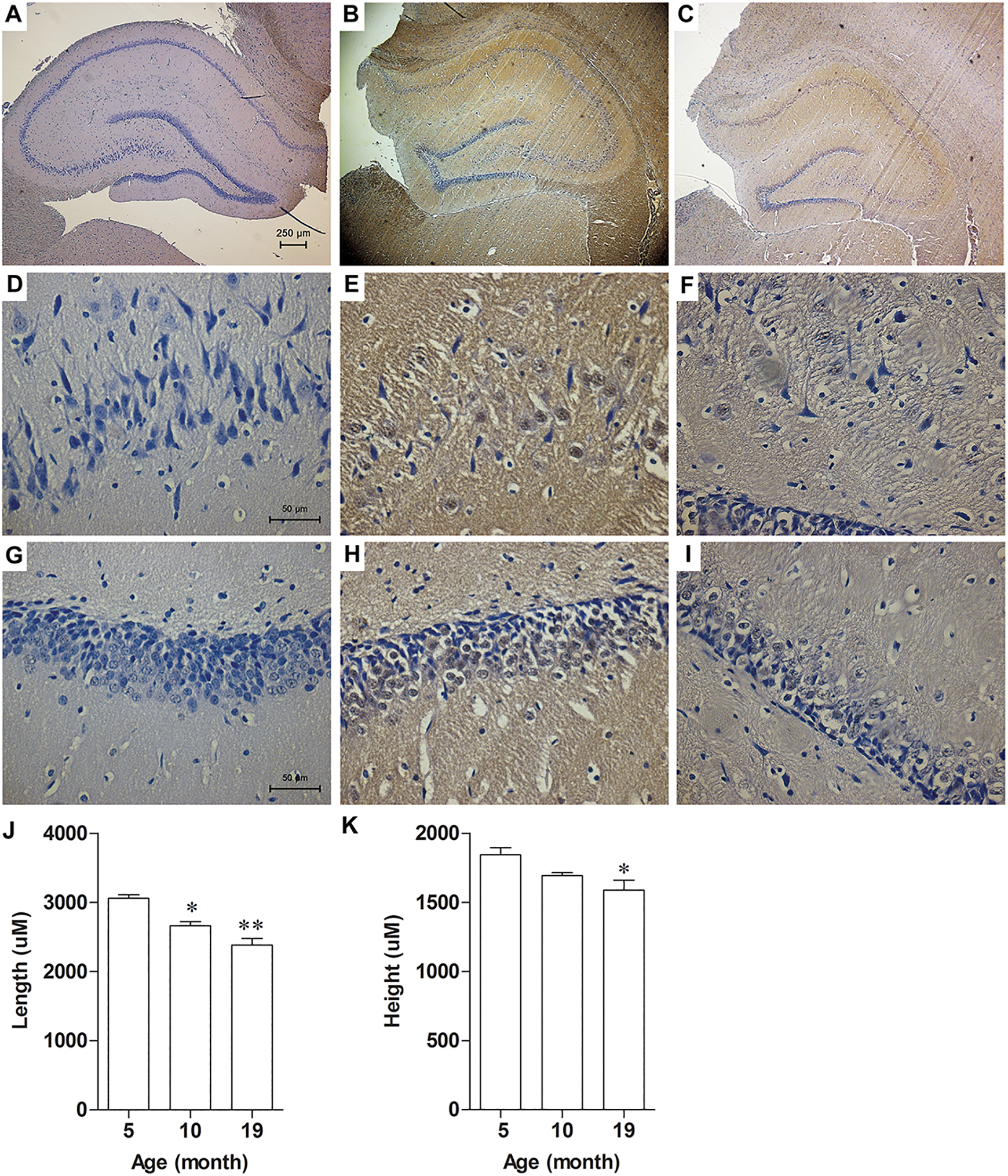
The expression of P450 in female rat hippocampus decreased in aged rat. Representative immunostainings of P450 of 5 (A, D, G)-,10 (B, E, H)-, and 19 (C, F, I)-month-old rats. Graphs showed analysis of length (J) and height (K) of hippocampus from 5-, 10-, and 19-month-old rats. *P < 0.001 vs 5-month-old, n = 5.
Age-Related Memory Impairment is Associated With Significant Alterations in ERα and ERβ Expressions in the Hippocampus of Female Rats
We next analyzed the effect of aging on the expression levels of the 2 ERs, ERα and ERβ in hippocampus of female rats. The 19-month-old rats showed significantly decreased ERα protein levels by about 40% with respect to 2-month-old rats (P = .0013; Figure 6A, B). A similar change was observed for ERβ whose protein level was decreased by 30% in the 19-month-old rats as compared to 2-month-old rats (P = .0007; Figure 6A, C). The results indicated that aging rats exhibited a decrease in ER expressions which may contribute to the decrease in responsiveness to E2 treatment in aging animals.
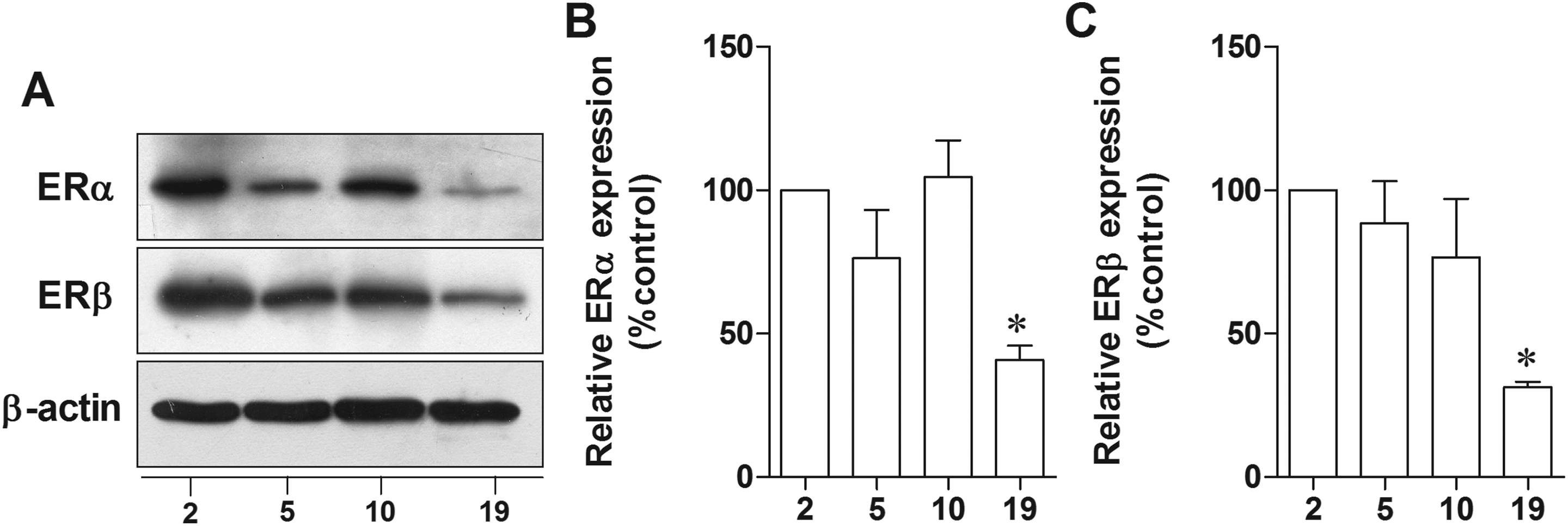
The expression of ER in female rat hippocampus decreased in aged rats. Representative immunoblotting of ERα and ERβ in hippocampus of 2-, 5-, 10-, 19-month-old female rats (A). Densitometric analyses of ERα (B) and ERβ (C) expression at each age of female rats normalized with β-actin. *P < 0.005 versus the 2-month-old group, n = 5. ER indicates estrogen receptor.
Age-Related Memory Impairment is Associated With Significant Increase in Microglial Activation Marker
Our previous study reported that activated microglia inhibited hippocampal synaptic plasticity. 24 In the present study, the phenotype of aged microglia was demonstrated using microglial activation marker, MHC-II. Results showed that MHC-II significantly increased in hippocampus of 19-month-old rats compared to 2-month-old rats (P = 0.0579; Figure 7A and B), suggesting the higher microglia activation in hippocampus of aged female rats. Among all the inflammatory cytokines, IL-6 has the most potent inflammatory properties, inducing inflammation and degeneration of the brain tissue. Similar to MHC-II, the expressed levels of hippocampal IL-6 was higher in 19-month-old rats when compared to the younger rats (P = .0649; Figure 7B and C). Our findings indicated that age-induced microglial activation and IL-6 expression damaged the hippocampal neurons. It is possible that age-related memory impairment is associated with microglial activation and chronic neuroinflammation.
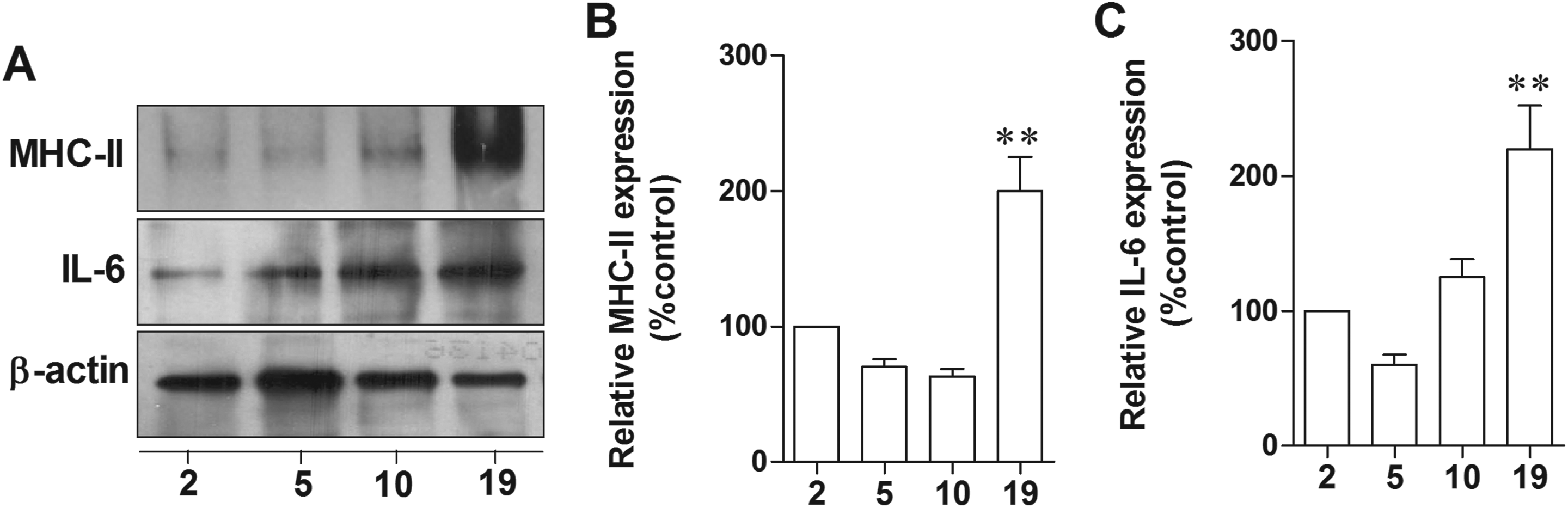
Age-induced microglia activation via increased expression of MHC II and IL-6 in female rat hippocampus. Representative immunoblotting of MHC-II and IL-6 in hippocampus of 2-, 5-, 10-, 19-month-old female rats (A). Densitometric analyses of MHC-II (B) and IL-6 (C) expression in each age of female rats normalized with β-actin. **P < 0.001 versus 2-month-old group, n = 5. MHC indicates major histocompatibility complex.
The Expression of P450 Significantly Decreased in IL-6-Treated Hippocampal Neurons
Previously, we had shown that IL-6 inhibited estrogen synthesis in an in vitro model of neuron–microglia coculture, 24 while its mechanism of action is unknown. In the present study, we demonstrated the direct effect of IL-6 on the expression of the main steroidogenic enzymes, that is, P450, in H19-7 hippocampal neurons. It was clearly shown that the expressions of P450 protein, but not mRNA, were significantly decreased in the group that was treated with IL-6 (500 pg/mL) for 10, 20, and 30 hours (P = .0568, P = .0060, and P = .0060; Figure 8). This result indicated that cytokine derived from aged microglia, including IL-6, inhibited hippocampal E2 synthesis by decreasing of P450 protein expression.
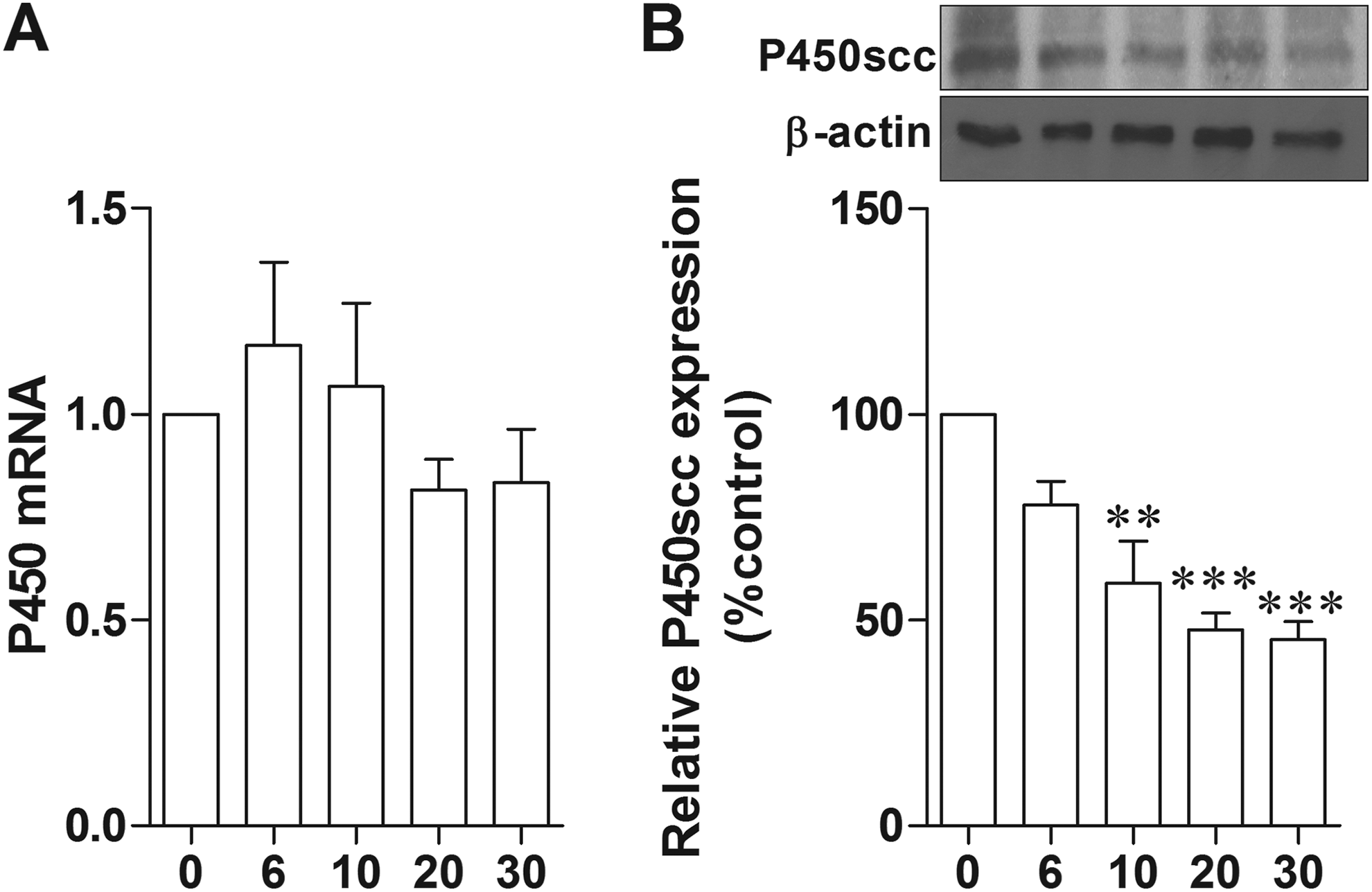
The expression of P450 was decreased in IL-6 treated H19-7 hippocampal neurons. Real-time PCR showed P450 mRNA expression after 500 pg/mL IL-6 treatments at various time points, for example, 0, 6, 10, 20, and 30 hours normalized by GAPDH (A). Representative immunoblottings of P450 in H19-7 hippocampal neurons that were incubated with 500 pg/mL IL-6 for 0, 6, 10, 20, and 30 hours (B). Densitometric analyses of P450 in each time of treatment normalized by β-actin. **P < 0.001, ***P < 0.001 vs the control (0 hour), n = 8. PCR indicates polymerase chain reaction.
Discussion
In the present study, we demonstrated the decrease in endogenous E2, ER, StAR, and P450 expressions in hippocampus in aging rats, concomitant with a significant impairment of spatial memory, although serum E2 was increased. These findings suggest that age-related memory impairment in female rats is not dependent on systemic E2 circulating. This supports findings in a previous study by Barker and Galea that serum E2 concentrations are not reflective of brain tissue E2 concentrations. 17
Several studies reported the different regulatory mechanisms controlling the synthesis of steroid hormones between human and rat. E2 level in rats, unlike those in women and mice, 26 falls to a steadily moderate level and then increases in old rat as reported previously, 15–16 and here, we also found that serum E2 level in 19-month-old rats was higher than those in younger rats. There are approximately 40% of the 19-month-old rats that had irregular estrous cycles. Consequently, it is important to use an additional indicator to estimate ovarian aging, and AMH levels have been suggested to be a suitable marker. 27 In human, serum AMH serum level was almost undetectable at birth. It subtly increased after puberty and became stable until adulthood and then decreased as a sign of follicular failure. 28 Here, we also found that serum E2 level in 19-month-old female rats was higher than those in the younger rats. In addition, the levels of serum AMH slightly increased in 5- and 10-month-old rats when compared to 2-month-old rats. It then significantly decreased in 19-month-old rats when compared to 10-month-old rats. The levels of serum AMH in rat detected by ELISA in this study were in the range of 60 to 120 ng/mL similar to those previously reported. 27 Since E2 is a part of pituitary–ovarian feedback system, the serum E2 levels reflect both pituitary and ovarian functions. Therefore, AMH would be a better index for ovarian dysfunction in old rats. Recently, there has been growing interest to compare aging between rat and human at different phases of their life. The overall findings indicated that every day of the animal is approximately equivalent to 34.8 human days or 1 rat month is comparable to 3 human years. 29 Therefore, the animals used in this experiment are equivalent to human, with the ages of 6, 15, 30, and 57 years, respectively.
Moreover, aging rat and human exhibit a decrease in AMH level, which is a marker for ovarian aging. 27 Our result is thus similar to the reported finding that AMH level is lower in women aged 48 years old than those at 44 and 39 years old. 30
It is well established that aging animals and humans show decline in the ability to acquire and remember information. In rodent, spatial memory performance that required hippocampus shows decrement along the life span. Although most studies have investigated male rats, few studies in females also showed decline in age-related spatial memory performance. 2 Aged female rats, approximately 21 to 24 months old, showed impaired spatial task including MWM when compared to 2- to 4-month-old rats. 3 We also clearly showed memory impairment in 19-month-old rats whose serum E2 level was still high. This indicated that circulating E2 has no effect on improving memory impairment during aging. However, endogenous E2 level could be the key factor that helps to ameliorate memory impairment. Here, we showed that endogenous E2 and its receptors in hippocampus significantly decreased in aged rats in correlation with spatial memory impairment. Previously, it was reported that aged OVX rats exhibited a lower level of ERα expression than young OVX, and they suggested that this contributed to a decrease in responsiveness to E2 in aging animals. 5 Moreover, another previous study showed that prolonged OVX alters alternative splicing of ERβ in female rat brain, which resulted in the decrease in E2 efficacy during aging. 6,31 The decrease in E2 efficacy during aging, and the role of its receptor-mediated actions are the still object of controversy. However, specific mechanism responsible for age (but not systemic E2 deficiency)-related decline in spatial memory remains unclear.
It is plausible that endogenous hippocampal E2 is a major factor that controls memory during aging, since the level of serum E2 is not related to memory impairment in aging rats. In this study, we showed for the first time that StAR and P450 hippocampal steroidogenic enzymes significantly decreased with age, which is related to the decreased level of E2 in hippocampus. Taken together, our data suggest that the inhibitions of steroidogenesis in hippocampus could be a major factor contributing to the memory impairment in aging rats, whether the same phenomenon happens in human remains to be proven.
Our previous study demonstrated that activated microglia-inhibited synaptogenesis is IL-6 dependent, and IL-6 also suppressed secretion of E2 in the culture medium of hippocampal neurons. 24 Consequently, we suggested that IL-6 inhibits E2 synthesis in hippocampal neurons. However, the mechanism of how IL-6 inhibits E2 biosynthesis in hippocampal neuron is unknown. The present study clearly showed that IL-6 was highly expressed in aged hippocampus and blocked the expression at the protein level but not the mRNA level of P450 gene. This gene plays a role in the conversion of cholesterol to pregnenolone, which is the precursor to all other steroid hormones including E2. In conclusion, we proposed that IL-6 derived from aged microglia directly inhibited biosynthesis of E2 in the hippocampal neuron which leads to the age-related memory impairment.
Footnotes
Authors’ Note
Siriporn Chamniansawat contributed to conception and design, analysis and interpretation, drafted the manuscript, and critically revised the manuscript; Chattraporn Sawatdiyaphanon contributed to conception and design analysis, and interpretation and drafted the manuscript. Both authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgment
We express our gratitude to Emeritus Prof. Prasert Sobhon and Assoc. Prof. Narongrit Thongon of the Faculty of Allied Health Sciences, Burapha University, and Assoc. Prof. Sukumal Chongthammakun of the Faculty of Science, Mahidol University, for helpful suggestions and proofreading. We also thank our student advisory board members, Pilaiporn Ngamsamai, Suchawadee Kongnoi, and Nittayakorn Manoo, for their work on immunohistochemical staining process.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was supported by the research grants from the Thailand Research Fund (RSA6080056) and Burapha University through National Research Council of Thailand (137/2560) to S. Chamniansawat.
