Abstract
The function and structure of the auditory information processing system establishes a unique sensory environment for the “perfect storm.” The battle between life and death pits the cascade of an apoptotic storm, programmed cell death cascades, against simple cell death (necrosis) pathways. Live or die, the free radical biology of oxygen and hydroxylation, and the destruction of transition metal migration through the mechanical gate sensory processes of the hair cell lead to direct access to the cytoplasm, cytoplasmic reticulum, and mitochondria of the inner workings of the hair cells. These lead to subsequent interactions with nuclear DNA resulting in permanent hearing loss. The yin and yang of pharmaceutical product development is to document what kills, why it kills, and how do we mitigate it. This review highlights the processes of cell death within the cochlea.
Introduction
Ototoxicity has been broadly defined as the demonstration that certain substances, either systemic or topically administered, cause cellular damage to the tissues of the inner ear and eventually cell death and functional impairment, especially involving the end organs of the cochlear and vestibular divisions of the eighth cranial nerve. Drug-induced ototoxicity represents one of the main preventable causes of deafness. Ototoxicity is an effect shared by aminoglycoside and macrolide antibiotics, antimalarial medications, loop diuretics, platinum-based chemotherapeutic agents, some nonsteroidal anti-inflammatory drugs, and acetaminophen, when abused with narcotic medications. 1 These medications are prescribed despite their side effects, which includes inner ear toxicity, because they are lifesaving drugs or there is a lack of better treatment.
Death Is Part of Life
Active cell death implies a molecular program of cellular self-destruction. Cell death can be distinguished morphologically and biochemically into 2 types: (1) necrosis or (2) apoptosis. 2,3 As described previously by Elmore, 4 the mechanisms and morphologies of apoptosis and necrosis differ, but there is overlap between these 2 processes. Necrosis and apoptosis represent morphologic expressions of a shared biochemical network described as the “apoptosis–necrosis continuum.” 5 Whether a cell dies by necrosis or apoptosis depends in part on the nature of the cell death signal, the tissue type, the developmental stage of the tissue, and the physiologic milieu. 5,6 As highlighted by Elmore, 4 conventional histology does not always distinguish apoptosis from necrosis, and they can occur simultaneously depending on factors such as the intensity and duration of the stimulus, the extent of adenosine triphosphate (ATP) depletion, and the availability of caspases, 5 but the distinction does not play a major role in conducting preclinical ototoxicity testing under the current regulatory review process. In the nervous system, programmed cell death of sensory and motor neurons occurs in embryonic stages, but during maturation of central and peripheral nervous systems, extensive deletion of supernumerary neurons occurs through the same process. 7 –10 From an experimental standpoint, the principal problem associated with asynchronous death is that at any 1 point in time, individual cells in a population or at a position along the basilar membrane are at various stages along the cell death pathway. Knowing that the cochlea is tonotopically mapped by the position of cells along the basilar membrane, 11 –15 the presence of asynchronous cell death supports the standardized use of auditory brainstem response (ABR) assessments of threshold shifts multiple times over the study duration in order to estimate the pattern, severity, and onset of cell death attributable to a given test article. 16 –18
Humes 19 has pointed to the striking association between the inner ear and the kidney with regard to various disease processes. Three of the most prevalent classes of ototoxic agents, which include aminoglycoside antibiotics, platinum-containing chemotherapeutic agents, and loop diuretics, also have profound toxic or functional effects on the kidney. Jiang et al 20 highlight a number of physiological similarities between the cochlea and the kidney, principally the active transport of electrolytes or nutrients, and consequently, water follows to maintain iso-osmolarity. Gene expression analysis has identified at least 36 genes that are significantly expressed in both cochlea and kidney. 21 Both renal tubules and the stria vascularis are closely associated with basement membranes (of similar collagenous composition) that enclose blood vessels. Hearing loss is also associated in patients with lower estimated glomerular filtration rate and late chronic kidney disease. 22,23 Additionally, a common genetic disease, Alport syndrome, produces hearing loss and renal disease. Because of this association, insights into the mechanisms of toxicity or pharmacologic action of these compounds on the kidney may provide insights into the mechanisms of ototoxicity of these agents.
Aminoglycosides are readily taken up by renal proximal tubule cells and cochlear cells, 24 and interestingly, these antibiotics preferentially induce cytotoxicity in inner ear sensory hair cells and proximal tubule cells in vivo than for most other cell types. 19 Other ototoxic compounds, such as cisplatin (see below) and loop diuretics, are also directly toxic to both organs. 19 In addition, there is increased expression of Mpv17, a peroxisomal protein that metabolizes reactive oxygen species (ROS) in renal glomeruli and the stria vascularis of the cochlea following aminoglycoside exposure. 25
According to Akinbodewa and Okunola, 26 several factors are linked to the risk of another antibiotic, gentamicin, inducing both nephrotoxicity and ototoxicity: (1) elevated trough gentamicin levels, (2) plasma concentration time area under the curve, (3) duration of treatment on aminoglycoside, (4) concomitant vancomycin or furosemide use, (5) volume depletion, (6) elevated baseline serum creatinine, (7) increasing age, (8) presence of comorbidities, (9) liver dysfunction, (10) sepsis, (11) hypokalemia, (12) hypomagnesaemia, (13) the type of aminoglycoside, (14) the frequency of aminoglycoside dosing, and (15) the timing of aminoglycoside administration. 27,28 Studies showed that ototoxicity results mainly from apoptosis unlike dose-dependent necrosis in nephrotoxicity. 29 Almost all cells take up aminoglycosides, and most cells are able to clear these drugs from their cytoplasm relatively quickly, except for inner ear hair cells and renal proximal tubule cells that retain these drugs for extended periods of time. 24 It is thought that this retention of aminoglycosides, plus the higher metabolic rate of hair cells and the renal proximal tubules cells, contributes to their susceptibility to these drugs. 20
Cisplatin is a drug that exhibits multiorgan toxicity through oxidative stress. Cisplatin is an antineoplastic agent used in the treatment of testicular, bladder, lung, gastrointestinal, and ovarian cancers. Clinically, ototoxicity, neurotoxicity (peripheral neuropathy), and renal toxicity (nephrotoxicity) have been described. There is evidence to support a role for cisplatin-induced oxidative stress in each of these adverse effects. Both free radical generation and depletion of antioxidants have been demonstrated in kidney in response to cisplatin administration. 30 Similarly, it has been reported that attenuation of endogenous antioxidant production is a key mechanism by which cisplatin causes oxidative stress in the ear. 31 Cisplatin treatment in rats is associated with acute ototoxicity along with depletion in the cochlear antioxidant concentrations of glutathione peroxidase and glutathione reductase, elevations in superoxide dismutase concentrations, and catalase activities. 32 Gonçalves et al 33 have suggested that the nicotinamide adenine dinucleotide phosphate oxidase 3 isoform (also termed NOX3) seems to be the main source of ROS in the cochlea. These ROS react with other molecules and trigger processes such as lipid peroxidation of the plasma membrane and increase in expression of the transient vanilloid receptor potential 1 ion channel. 23 Whether this is a cause or consequence of the toxic insult to the sensory hair cells of the inner ear remains to be determined. No matter the exact precipitating cause of cellular death; histopathologically, the degenerative effects of cisplatin have been noted in outer hair cells in the organ of Corti, spiral ganglion cells, and marginal cells of the stria vascularis. 34 –36 Breglio et al 37 have recently shown that platinum is retained in the cochleae of experimental mice and treated patients with cancer months to years after cisplatin chemotherapy. Although previous work has focused upon identifying the pathways responsible for cellular hypersensitivity to cisplatin within the cochlea, the Breglio et al’s 37 data suggest that it is hyperaccumulation and not hypersensitivity that drives cisplatin ototoxicity. Cell types within the cochlea likely experience higher cumulative drug exposure than cell types in less susceptible tissues. This is in line with data from Hellberg et al, 38 which reported a positive correlation between the concentration of different platinum chemotherapeutics in cochlear endolymph and their level of ototoxicity.
The Whole Is More Than the Sum of Its Parts
Figure 1 shows the general simplistic view of the structure of the mammalian auditory system. Sound pressure waves from the environment are focused and directed by the pinna to enter the ear canal and oscillate or push against the eardrum (tympanum). It has been estimated that for the movement of the eardrum, the width of a single hydrogen atom is sufficient to initiate a “sound stimulus” to be processed. 39 The tympanum is the separating boundary between the outer (external) and the middle ear. To ensure test article exposure to critical transduction mechanisms of the middle and inner ear, many preclinical screening studies administer the test article by needle puncture of the tympanum (intratympanic dosing [IT]). The middle ear includes a system of small bones, or ossicles, known as the malleus (hammer), incus (anvil), and stapes (stirrup). Movement of the tympanum physically, simultaneously, and synchronously vibrates the ossicles with the frequency of air pressure waves that initiate the tympanum movements. The physical movement of the ossicles transmits the extremely small waveforms to the cochlea. The purpose of the middle ear is to bridge what would otherwise be an almost impenetrable mechanical boundary between the air-filled medium of the external and middle ear chambers and the fluid-filled spaces of the cochlea. Simple physical characteristics of fluid dynamics defines the acoustic impedance (resistance to movement) that must be overcome between the 2 physical states (air vs water). The fluid inside the cochlea must be “pushed” or moved with more intensity if the frequency of the fluid is to retain a 1:1 relationship with each oscillation or movement in the external world of the original sound pressure waves. The air-propagated sound pressure wave is simply too weak to transduce (convert) similar vibrations or oscillations onto the water media, so the ossicles provide the driving dynamics to address the impedance mismatch. The stapes “footplate” concentrates the ear drum and ossicle movements (surface area of approximately 500 mm2) to a smaller punctate spot on the cochlear membrane, about 20 times smaller, which is called the oval window. When the middle ear operates efficiently, there is a wide range of sound pressure waveforms that are effectively transmitted without loss or interference to the cochlea—the organ that will transduce the physical waveform to an electrochemical neuronal pulse that can be transmitted to the brain.
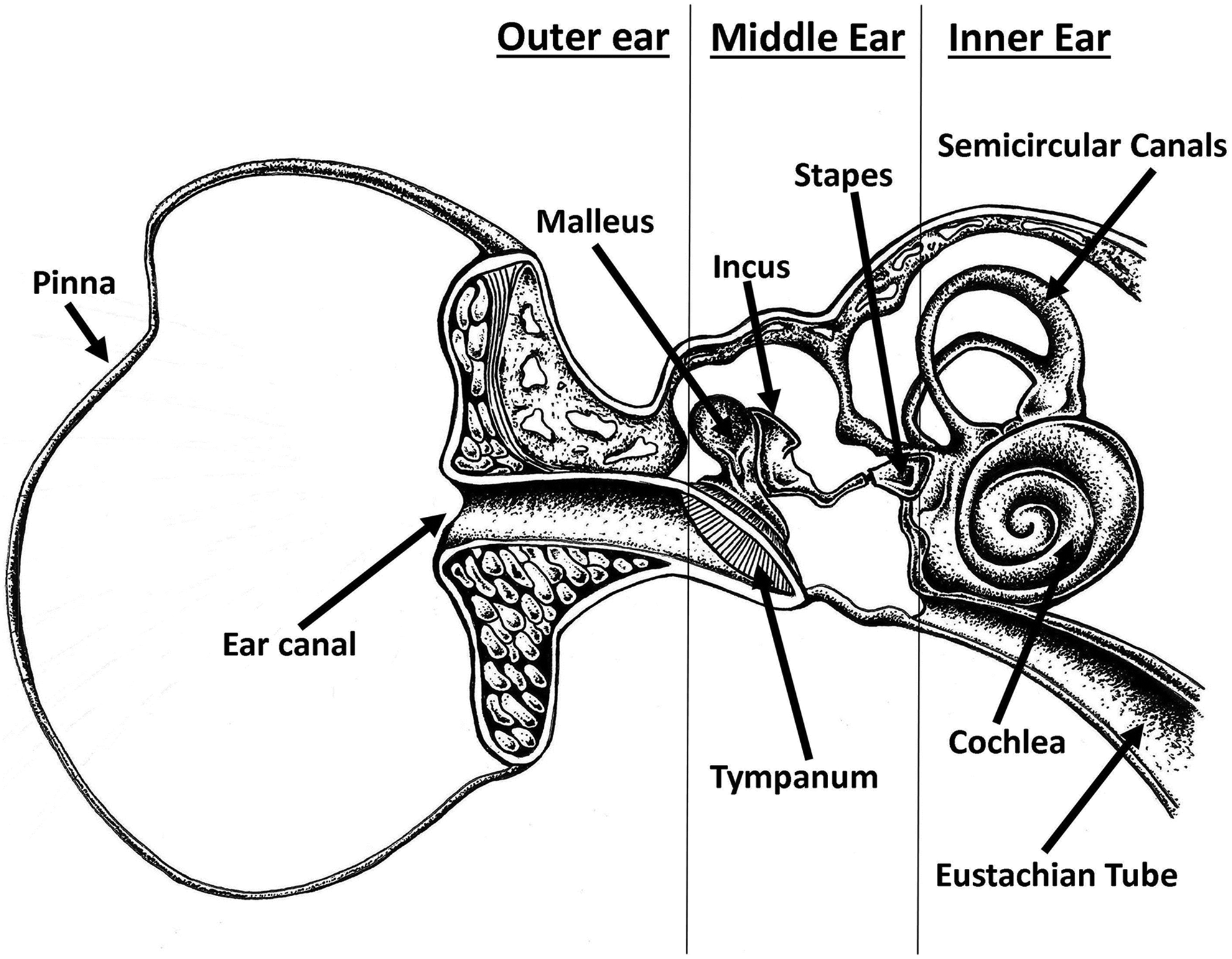
Basic anatomy of the mammalian auditory system into outer, middle, and inner ear structures. The semi-schematic drawing showing the main path of sound. Vibrations entering the external auditory canal first affects the eardrum (tympanum). Oscillations of the drum are transmitted through the middle ear chiefly by bone conduction over the chain of ossicles. The foot of the stapes carries the vibrations to the fluid-filled cochlea, in which the sensory neuron endings of the auditory nerve are immersed.
Sound travels rapidly in aqueous media, and the cochlea is a small coiled structure (Figure 2). Since biological fluids are incompressible and this organ is encased by a hard-bony shell, these waveforms cannot create any motion inside the chambers of the cochlea without the membrane covering the round window being allowed to move (bulge) outward every time the oval window is pushed in and vice versa. The presence of a resiliently pliable “round window membrane” at the end of the cochlear chamber allows fluid movement within the chambers of the cochlea. Two additional membranes within the cochlea, the basilar membrane and the Reissner membrane, are located along almost the entire length of the coiled organ. The Reissner membrane divides the fluid-filled spaces between the medial compartment (scala media) and the upper compartment (scala tympani). The basilar membrane divides the fluid-filled spaces between the medial compartment (scala media) and lower compartment (scala tympani). The basilar membrane has interesting mechanical properties: It is narrow and stiff at the base of the cochlea and more elastic or floppy at the far apical end of the organ. The cochlea then has 2 sources of mechanical resistance: (1) the physical stiffness of the basilar membrane itself and (2) the inertia of the cochlear fluid movements. These 2 dynamic parameters are in competition—they run in opposite directions. The stiffness gradient of the basilar membrane decreases as the waveform migrates away from the oval window (incoming signal), but the fluid inertia gradient increases down that same path. These 2 competitive forces of resistance sets the stage for the vibration traveling through the chambers of the cochlea to find a “compromise path,” which is long enough that the stiffness of the membrane has already diminished to an optimal level but not so far down the chamber length that the wave inertia resistance has intensified dramatically. Since the fluid inertia resistance is frequency dependent, the optimal compromise—the path of overall lower resistance—is also frequency dependent. The optimal compromise is long for low frequencies, which are less affected by fluid inertia, and increasingly shorter for higher frequencies. Low-frequency waves (eg, 100-200 per second) will produce vibrations at the apex (far end) of the basilar membrane—a long way from the oval window. As the frequency is increased, the place of maximal vibration on the basilar membrane shifts toward the basal end. In this manner, each interval along the basilar membrane has its own “best frequency”—a frequency that will make the cells located on the basilar membrane to vibrate more than any others (tonotopic mapping).
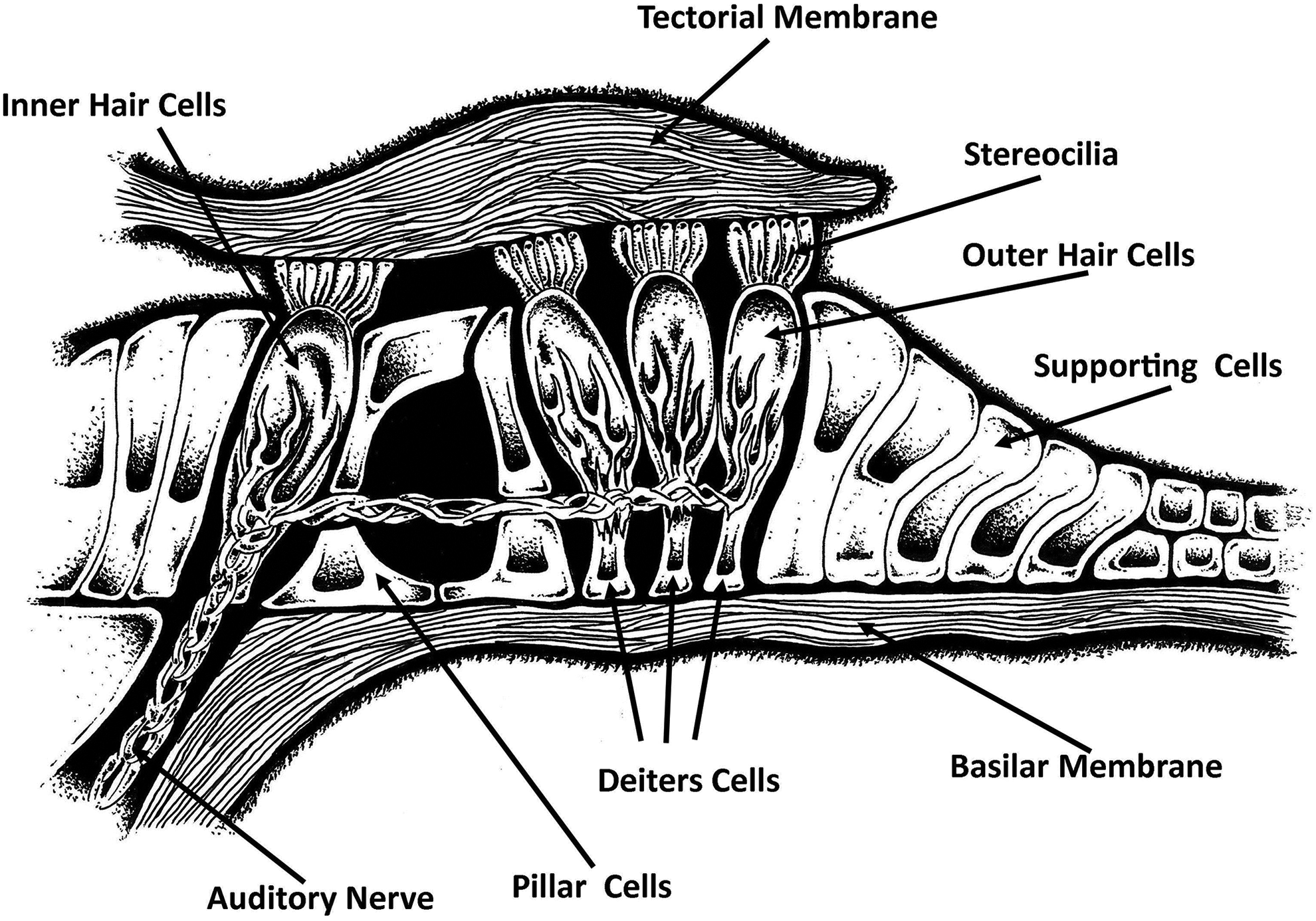
Cross section of the organ of Corti. The basilar membrane and Reissner membrane define the limits of the cochlear duct (scala media) which is located at the organ of Corti. The hair cells of the organ are the sensory receptor for transducing the traveling wave form into neural impulses relayed down the fibers of the auditory nerve.
The next stage of processing or converting the ambient air pressure waves to a pattern of electrical excitation that can be encoded by sensory neurons for further transmission to the central nervous system (CNS) requires transduction. Transduction—the physical conversion from one form of energy (fluid waveform) to an electrochemical impulse—is performed by the organ of Corti, which runs the length of the basilar membrane. The organ of Corti has a “folded” structure. At the base of the structure, the portion that sits directly on the basilar membrane, there are rows of sensory hair cells. On the modiolar side (the side closest to the center of the cochlear spiral), the organ of Corti curves upward and folds back over to form an “umbrella” or a “roof,” known as the tectorial membrane, which comes into close proximity to the stereocilia (hairs) on the sensory cells.
Unique Features of Hair Cells: The Mechanically Gated Channel
Auditory perception begins with receptor cells that are sensitive to one or more kinds of stimuli (transduced sound pressure to endolymph waveforms or mechanical bending). Specific neurons in the sensory systems, both peripheral and central, encode certain critical attributes of sensation (eg, pitch, timbre, and loudness). Some of these attributes are encoded by the pattern of activity in a population of sensory neurons. Integrated circuits of neuronal outputs act as auditory amplifiers that augment our sensitivity and are responsible for the first stages of frequency analysis. The paired cochleae are said to contain over 30, 000 receptor cells. 40 The mechanical nature of the waveform within the fluid-filled cochlea initiates transduction (conversion) of the air pressure waveform from the outer environment to the electrical impulses of the inner environment. Hair cells can measure movements of atomic dimensions and transduce stimuli ranging from static inputs to those at frequencies of tens of kilohertz. Damage to these “signal converters” accounts for most of the hearing loss induced by drug administration. 40,41
The organ of Corti is the receptor organ of the inner ear, containing well-organized placement of hair cells and a variety of supportive cells along the length of the basilar membrane. Like the basilar membrane itself, both the hair cells and the auditory nerve fibers are tonotopically organized. That is, at any position along the uncoiled basilar membrane, the structures are optimally sensitive to a particular frequency, and these frequencies are logarithmically mapped in ascending order from the apex to the base. There are 2 types of cilia-activated (hair) cells from a single row of inner hair cells and running parallel to that single row are 3 to 4 columns of v-shaped bundles of outer hair cells (Figure 1A, insert). The outer hair cells are in the vast majority within the cochlea and are supported at their base by the pharyngeal (Deiters) cells. The space between the inner and outer hair cells is controlled and maintained mechanically by pillar cells. A laminar layer of cantilevered gelatinous material lies over the top of the hair cells to form the tectorial membrane. The membrane is physically anchored at the base, and its tapered distal edge forms a fragile connection to the organ of Corti. The longest stereocilia (hairs) of the outer hair cells are tightly attached to the tectorial membrane’s lower surface. This coupling of hair cell bundles to the tectorial membrane is physically strong—so strong that the stereocilia tips are pulled free from the hair cells when the tectorial membrane is lifted from the organ of Corti (as during dissection and/or histological fixation for pathology reviews). When the basilar membrane vibrates in response to a sound, the organ of Corti and the overlying tectorial membrane are carried with it. Since the organ of Corti and the tectorial membrane pivot on different planes, the waveform oscillations displace the space between them differently, forcing a back-and-forth shearing motion between the upper surface of the organ of Corti and the lower surface of the tectorial membrane. The cilia of the hair cell bundles are then bent both up and down and laterally (Figure 3A and B). This mechanical deflection of the hair cell cilia physically opens channels allowing ions to enter into the cells. Unlike chemically or electrically gated neuron channels, the hair cells of the inner ear are simple open-/close-gated channels. If an ion is small and positively charged (eg, Ca++ or K+), it migrates through the open channel gate. 11 –15
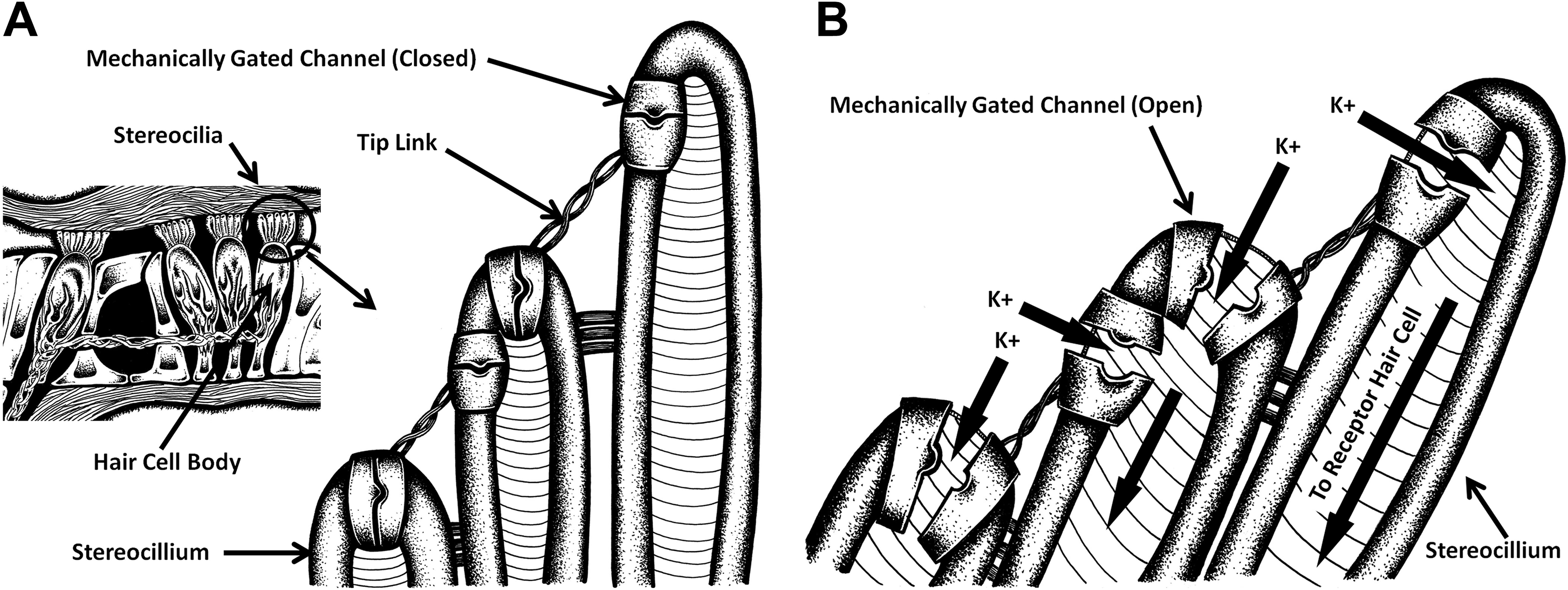
A, (box insert) Illustration of the organization of the basilar membrane and the relative positions of the outer and inner hair cells within the cochlea (Organ of Corti). The individual stereocilia are expanded to show how each cilia are connected to neighboring cilia and the position of the mechanical gate channels along the lateral and apical aspects of the cilia. B, Illustration to show the opening of the mechanic-transduction channels of the cilia when sheering forces of the tectorial membrane physically bend the hair cilia in concert with the frequency of the traveling wave. The channels are physically opened, and any and all ions, chemicals, molecular entities in close proximity have free access into the inner cell environment. The apical channels provide a wider gate into the cell than the laterally positioned channels.
Ca++ plays a prominent role in the performance of the outer hair cells, which possess large amounts of cytoplasmic calcium-binding proteins 42 to buffer Ca++ and pumps in the plasma membrane to extrude the ion. The main Ca++ source is likely to be influx through the mechanotransducer (MET) channels, which are about 5-fold more permeable to Ca++ than to Na+ and partially open at rest. Ca++ influx through the cilia channels during stimulation controls the extent and rate of channel adaptation. 13,43 Horváth et al 44 performed functional ratiometric Ca(2+) imaging (fura-2) in the hemicochlea preparation of hearing mice to measure purinergic receptor-mediated Ca(2+) signaling in pillar, Deiters, and Hensen cells. Their resting intracellular calcium concentration was determined and compared in the same type of preparation. The ATP induced reversible, repeatable, and dose-dependent Ca(2+) transients in all 3 cell types, showing desensitization. Inhibiting the calcium signaling of the ionotropic P2X (omission of extracellular Ca[2+]) and metabotropic P2Y purinergic receptors (depletion in intracellular Ca[2+] stores) revealed the involvement of both receptor types. Detection of P2X2,3,4,6,7 and P2Y1,2,6,12,14 receptor messenger RNAs by reverse transcription polymerase chain reaction (RT-PCR) supported this finding, and antagonism by pyridoxal-5-phosphate-6-azophenyl-2′,4′-disulphonic acid suggested different functional purinergic receptor population in pillar versus Deiters and Hensen cells. 44
Defining the mechanical attributes of the hair bundle is central to understanding transduction and amplification in the vertebrate cochlea. The traveling wave of endolymph produces a shearing force against the hair cells that are in close proximity to the rigid tectorial membrane. The hair cell cilia then pivot at their base or “ankles.” 45 The hair-bundle mechanics are dictated not only by the passive structural elements (the flexibility of the stereociliary ankles, the interciliary connections, and the gating springs) but also by the opening and closing of the MET channels. 46 It has been argued that active hair-bundle movements may also contribute to amplification in the mammalian cochlea. 47,48
Figure 3A and B shows the hair cell bundle and the individual MET channels located at the apex and side portions of the cilia. Hair cells convert deflections of their hair bundle into electrical signals by gating mechanically sensitive channels (Figure 3A and B 49 –52 ). In achieving their remarkable sensitivity, hair cells must overcome a fundamental problem; the energy at sound threshold levels is limiting. 53 Hair cells overcome this barrier by mechanically amplifying low-intensity stimuli, a mechanism termed the active process. The motion of the hair cell bundle to the sheering force of the tectorial membrane is governed by its mechanical compliance. This compliance has a passive component attributable to the flexibility of the stereociliary ankles, stiffness of the gating springs and an active component, and the gating compliance, linked to the opening and closing of the MET channels. 46 The frequency of hair bundle oscillations varied tonotopically, suggesting a role in mechanical tuning. 11 –15 Lukashkina et al 54 have suggested that outer hair cell motility delivers differential energy to apical and basal turn cochlear partitions. In order to optimize mechanical impedance matching in the stiff basal turn of the cochlea, the framework provided by Deiters and Pillar cells must be rigid so that forces delivered by the outer hair cells are almost isometric. The cochlear partition thus moves more in the apical region. Deiters and Pillar cells are more gracile and may enable the transfer of isotonic rather than isometric forces and may even be absent in low-frequency hearing. Lukashkina et al 54 suggest that differences in the ways outer hair cells are coupled to the cochlear partition at the apex and base may account for why the high-frequency regions of the cochlea are so susceptible to cochlear insults by noise and hypoxia but not by systemic administration of ototoxic agents.
Auditory hair cells are probably the fastest and most sensitive of all vertebrate mechanoreceptors, and those in the mammalian cochlea may respond to vibrations (elicited by sound stimuli) with amplitudes of 1 nm, and in some species (eg, mice or bats) at frequencies as high as 100 kHz. Adaptation to these exacting stimuli is likely to involve the accessory apparatus, the external and internal attachments of the MET channel within each stereocilium, and the nature of the channel protein itself. 11,14,43,50,51
Hair Cells Have Support
Support cells in the cochlea include Hensen cells, Deiters cells, inner and outer pillar cells, Claudius cells, and inner supporting cells (mainly the inner phalangeal cells). 46,55,56 The inner and outer pillar cells are positioned to lean toward each other at the head of the organ of Corti, forming a triangular tunnel, which is filled with cortilymph. The inner hair cells and outer hair cells lie on 2 sides of the Corti tunnel, respectively. The inner phalangeal cells and Deiters cells support the inner and outer hair cells, respectively. Hensen cells form a support bar on the outer edge of the organ of Corti. 55 The outer tunnel of the inner aspect of the cochlea contains the Hensen cells, the third-row outer hair cells, and Deiters cells. There are gap junctions between supporting cells and hair cells. These gap junctions are made of connexins that are encoded by connexins genes (eg, Cx26, Cx30, etc). 56 Connexins are structurally related transmembrane proteins that assemble to form gap junctions—a channel that permits ions and small molecules to move between adjacent cells. The connexins are critically important to intercellular communications within the cochlea. Expression of Cx26 and Cx30 is not the same throughout the cochlea; its distribution shows a 3-fold longitudinal gradient of reduction from the apex to the base. The reduction is more pronounced in the Deiters cells and pillar cells than in the Hensen cells. 44,55,56 These channels of gap junctions (connexins) play an important role in recycling and regulating intracellular K+ as well as pH homeostatic mechanisms. Electrical coupling and dye coupling between supporting cells are very strong, but coupling between supporting cells and hair cells are relatively weak. As gap junctions between supporting cells conduct information rapidly while linking adjacent cells together, Hensen cells may work as a functional filter barrier that may serve to adjust inner cochlear milieu. According to Zhu et al, 56 connexin 26 (Cx26, GJB2) is a predominant gap junction isoform in the cochlea, and targeted deletion of Cx26 in Deiter cells and outer pillar cells shift the outer hair cell electromotility and reduces active cochlear mechanics with hearing loss. Cx26 expression in supporting cells appears to play a critical role in active cochlear amplification in vivo.
Parsa et al 57 reported that pillar and Deiters cells, in spite of obvious differences in size, share some common morphological features. Both types of cells are pyramidal and comprise little more than the basal cones that anchor the cells to the basilar membrane. These authors confirmed that basal cones consist mostly of an actin-rich core wrapped by microtubules and support the currently accepted concept that neither actin filaments nor microtubules penetrate into the basilar membrane. Despite differences in size between Deiters cells from different rows and different cochlear regions, the basal feet of Deiters cells are all similar in shape and size. Using confocal and scanning electron microscopy measurements of mice cochlea, Parsa et al 57 reported that the basal pole of every Deiters cells, independent of their position in the organ of Corti and along the cochlear spiral, attached to the basilar membrane within a 15.1 ± 0.3 μm-wide stripe running the length of the cochlear spiral adjacent to the row of outer pillar cells. All Deiters cells’ basal poles have similar diameter and general morphology and distributed on the stripe in a precise arrangement with a center-to-center distance of 7.1 ± 0.3 μm between neighboring cells of the same row and 5.9 ± 0.4 μm for neighboring cells in adjacent rows. Complete detachment of Deiters cells revealed an elliptical imprint on the top surface of the basilar membrane consisting of a smaller central structure with a very smooth surface surrounded by a rougher area, suggesting the presence of 2 different anchoring junctions. These previously unidentified morphological features of Deiters cells could be critical for the mechanical response of the organ of Corti.
The inner part of the cochlea including inner hair cells and pillar cells are relatively stable, which may be because supporting cells are more pliable than inner hair cells. Given the sensitivity reduction accompanied by structural contraction, the current view is that supporting cells contribute to the control of hearing sensitivity. Evidence has shown that mechanical and acoustic overstimulation can cause intracellular calcium concentration increases, accompanied by dynamic contractions of the hearing organ and the concentration changes in calcium in outer hair cells and Deiters cells, which can induce motility of the outer hair cells. The mechanism for contraction of the organ of Corti induced by intense sound or mechanical stimulation may be a change in concentration of intracellular calcium, leading to motility of outer hair cells and Deiters cells. This phenomenon has been suggested to be a form of “protective reflex” against sound trauma since a stimulation intensity exceeding the protective capacity of the hearing organ can damage outer hair cells. Such observations may be helpful in understanding why noise almost always injures outer hair cells, while inner hair cells are less frequently injured. This may be due to distortion in the support structure provided by Deiters cells, which likely involves Ca++, as the rise in Ca++ concentration can stiffen the basal platform.
Hair Cell Death
Part of pharmaceutical development and the tiered safety screening in use today to bring new drug products onto the market is not only to identify therapeutic efficacy and safety but also to identify the etiology of toxicity that may be documented on the pathway to drug approval. 58,59 Central nervous system cell death occurs throughout the normal life span and is often attributed to age-related deficits. In actuality, knowing that the specific type of cell death is not a critical or determining factor in identifying or constructing the cytocochleogram. However, it is important to understand why the hair cells, by their nature, are uniquely targeted for cell death in the hopes of being able to prevent the damage in the first place. Fetoni et al 60 suggest that it is critically important in the analysis and identification of ototoxicity in nonclinical safety studies to acknowledge the fact that sensory disruption due to damage in the organ of Corti may trigger central mechanisms of homeostatic/maladaptive plasticity. 2,22,61,62 Consistent with theories of homeostatic plasticity, many studies have already reported changes in excitatory, inhibitory, and neuromodulatory networks along with the central auditory pathway. 29,63 –67 These studies suggest that the identification of hair cell loss early in the drug development process is critical for a comprehensive safety evaluation at the time of the new drug application review process and before marketing approval.
Free Radical Biology and Oxidative Stress: Charged Particles in the Cytoplasm
“Spin trapping” is one available technique used to detect and identify short-lived free radicals through the use of electron paramagnetic resonance (EPR) spectroscopy. Electron paramagnetic resonance spectroscopy detects short-lived chemical species, such as the unpaired electrons of free radicals. In the cases when the half-life of radicals is too short to detect with EPR, compounds known as “spin traps” are used to react covalently with the radical products and form more stable adducts that will also have paramagnetic resonance spectra detectable by EPR spectroscopy. Hester et al 68 investigated the ability of a spin trap, α-phenyl-tert-butyl-nitrone (PBN), to prevent acute antibiotic-induced cochlear dysfunction. The PBN is a nitrone with free radical trapping properties. Guinea pigs’ auditory thresholds were monitored prior to and following antibiotic treatments in combination with PBN or vehicle. The thresholds progressively increased in the antibiotic-treated control group, beginning with high frequencies and later including ever lower frequencies. These threshold shifts were significantly greater (P < 0.05) than those seen in the PBN-pretreated groups. Hester et al 68 suggested that while ROS are expressed in apoptotic cascades initiated by aminoglycoside therapies, their findings indicate that PBN provided protection against acute antibiotic-induced threshold sensitivity losses and identified nitrogen reactive species as the precipitating neuroactive target for this type of ototoxicity. The PBN treatments also reduced the cell death associated with carbon monoxide–induced ototoxicity 69 as well as CNS damage induced by experimentally induced ischemia. 70
Curing the Effects of the “Cure” May Be a Radical Idea
Indeed, a cost–benefit analysis supports the continued use and availability of platinum-based cancer treatments and aminoglycoside interventions in infectious diseases. As described earlier, one main side effect of these treatments is the a priori knowledge of permanent sensorineural hearing loss induced by selective inner ear sensory hair cell death. A voluminous set of published data have accumulated on aminoglycoside-initiated cell death processes. However, fewer studies exist defining the mechanisms of antibiotic uptake by hair cells. Alharazneh et al 71 investigated 2 proposed mechanisms of aminoglycoside transport into mammalian hair cells: MET channels and endocytosis. Rat cochlear explants were cultured as whole organs in gentamicin-containing media. Two-photon imaging of Texas Red-conjugated gentamicin (GTTR) uptake into live hair cells was found to be rapid and selective. Hypocalcemia, which increases the open probability of MET channels, increased aminoglycoside entry into hair cells. Three blockers of MET channels (curare, quinine, and amiloride) significantly reduced GTTR uptake, whereas the endocytosis inhibitor concanavalin A did not. Alharazneh et al 71 reported pharmacologic blockade of MET channels by both curare and quinine but not concanavalin A or dynosore. The channel blockade prevented hair cell loss when challenged with gentamicin for up to 96 hours. Taken together, these data strongly suggest that the patency of MET channels (see Figure 3A and B) mediate the entry of the antibiotic into hair cells. Kirkwood and O’Reilly 72 have more recently reported that application of d-tubocurarine (dTC) and a structurally similar alkaloid, berbamine, blocked MET channel entry of aminoglycosides into mouse outer hair cells. Kirkwood and O’Reilly 72 noted that while berbamine permeates the MET channel more easily than dTC, berbamine blocks the hair cell’s basolateral K+ current but dTC does not. These data suggest that while the MET channel may serve as the port of entry of aminoglycoside into the hair cell cytoplasm, cell death may not require changes in membrane potassium currents to initiate the death cascade.
Once admitted into the hair cells, many intracellular targets/mediators are reported to be involved in aminoglycoside ototoxicity. This complexity has presented challenges in defining the actual molecular mechanisms, their temporal sequence, and also candidate therapeutic targets. 73 It is also unclear whether redundancy may exist and whether aminoglycosides have multiple intracellular targets, making the efficacy of singular target approaches uncertain. Nonetheless, one of the attractive targets for aminoglycosides is the ribosome, as biochemical and molecular evidence strongly suggest their interaction. These data suggest that limiting permeation of the aminoglycosides through the MET channel or preventing their entry into the hair cell endolymph should be the initial therapeutic target for the prevention of hair cell death and hearing loss. This methodical strategy to identify possible curative or restorative interventions may become part of ototoxicity screening of all new chemical entities in the future.
Metals and Oxidative Damage: “Heavy Metal” Ions or Rock Bands Do the Trick
Connor 74 has clearly identified that excess exposure to both essential and nonessential heavy metals can lead to a variety of neurological disorders that selectively affect a variety of organs and cells in the body. The complicated network of neural cells includes interactions between neurons and various types of support cells (eg, glial cells). Additionally, CNS cells interact by a variety of other factors (growth factors, cytokines, caspases, etc) for mutual support, regulation, and protection. 74 The diverse but highly specific nature of the symptoms produced by each metal indicates that they can interact with a restricted population of cellular targets, ultimately resulting in unique clinical manifestations. 75 The symptoms, which can be reversible or irreversible, often present with different patterns and outcomes depending on the net accumulated dose of any given metal. There are some common pathological conditions that result from excess exposure to heavy metals which unfortunately have not received widespread recognition and thus have not been extensively investigated. For example, chronic exposure to several heavy metals such as Co, Mn, Cd, Pb, and Hg has the potential to affect hearing in humans and experimental animals based on previous studies including case reports and ex vivo studies. 75 Understanding exactly how these metals induce hearing deficits is complicated by the fact that the inner ear is an extremely complex system that is composed of a diverse collection of sensory, neural, and supporting cells that must act in synchrony to produce a neurophysiological signal terminating in the central auditory system.
Castellanos and Fuente 76 recently reviewed the literature on the adverse effects of heavy metal exposures on the human auditory system. The auditory effects of arsenic, cadmium, lead, manganese (MN), mercury, and mixtures of heavy metals were found. However, it was discovered that there is a paucity of studies confirming the effects of chromium, cobalt, copper, nickel, or zinc. Lead-exposed humans were found to present with higher (ie, worse) hearing thresholds than nonexposed controls. Lead-exposed humans had longer ABR interpeak latencies than nonexposed controls, suggesting that this chemical may adversely affect the brain stem auditory pathways. Similar results have been found in pediatric populations exposed to lead. Interactions between coexposure to lead and noise at moderate and high levels have also been found. Castellanos and Fuente 76 reported that arsenic may be associated with higher hearing thresholds in adults and children. For cadmium and mercury, studies have found an association between exposure to these agents and higher hearing thresholds as well as adverse central auditory effects.
Cells of the organ of Corti, like all other cell types, require iron for DNA synthesis, mitochondrial respiration, and other critical cell functions. As described earlier, the complicated network of cells of the auditory pathways includes interactions between neurons of various types that also involve iron.
Iron
Iron is a transition element. Ferroptosis (iron apoptosis) is a unique form of programmed death, characterized by cytosolic accumulation of iron, lipid hydroperoxides, and their metabolites and affected by the fatal peroxidation of polyunsaturated fatty acids in the plasma membrane. It is a major driver of cell death in neurodegenerative neurological diseases. One important feature from a radical point of view is variable valence of iron, which allows it to undergo changes in oxidation state involving 1 electron. Iron has 2 common valences: iron II (ferrous ion) and iron III (ferric ion). Hydrogen peroxide is formed in many aerobic cells. Iron plays an important role in the production of free radicals and the generation of ROS. The reaction of iron ions and hydrogen peroxide is a reliable source of hydroxyl radicals (OH−) as well. Neuroinflammation leads to upregulation in divalent metal transporter 1 on the surface of astrocytes, microglia, and neurons, making them highly sensitive to iron overload in the presence of high levels of non-transferrin-bound iron, thereby affording such levels a dominant role in respect of the induction of iron-mediated cell death.
Conlon and Smith 77 reported changes in auditory function induced by coadministration of gentamicin and iron. Results showed that animals receiving iron in combination with gentamicin demonstrated a more rapid and profound elevation in auditory thresholds compared to animals receiving gentamicin alone. This effect was dose dependent, but iron alone maintained normal thresholds throughout the treatment period. These results provide further evidence for the intrinsic role of iron in aminoglycoside ototoxicity and highlight a potential risk of aminoglycoside administration in patients with elevated serum iron. There is now substantial evidence to suggest that this form of neurotoxicity is due, at least in part, to the production of free radicals, either in response to excessive stimulation of the N-methyl-D-aspartate (NMDA) receptor by aminoglycosides or from the formation of oxidative compounds following the binding of aminoglycosides to iron. 78 If activation of NMDA receptors by aminoglycosides contributes to hair cell death, it should be possible to modify the chemical structures of the aminoglycosides in ways that minimize the unwanted action. Researchers are also currently evaluating drugs that could be coadministered with aminoglycosides in order to interfere with the apoptotic cascade. For example, NMDA receptor antagonists, nitric oxide synthase inhibitors, caspase inhibitors, neurotrophins, and free radical scavengers have been demonstrated to protect against aminoglycoside-induced cochlear toxicity in experimental studies. The next step will be to pursue these effects using in vivo models and drugs that are likely to have favorable pharmacokinetic and side effect profiles in humans. Administration of an iron chelator has already been reported to prevent ototoxicity induced by gentamicin 79 or cisplatin. 6,80
Copper
The biological reactivity of copper is the basis for both its pivotal role in enzyme activity and its toxicity. 68,81 Recent studies on multiple cell systems, including an immortalized cochlear cell line, 82 have identified copper transporter 1 (Ctr1) as the major influx transporter for platinum-based drugs, such as cisplatin. 83,84 It has been well established that the uptake of cisplatin through Ctr1 can be competitively inhibited by increased extracellular copper. 82,85 The cellular influx and efflux of cisplatin is not solely controlled by actions at Ctr1; several different transporters such as the copper transporter 2 (Ctr2), 86 P-type copper transporting ATPases (ATP7A and ATP7B 29 ), organic cation transporter 2, 87 and the multidrug and toxin extrusion transporter 1. 88
Consistent with an earlier report on 3- to 4-week-old mouse cochleae, 82 Ctr1 was localized in the organ of Corti, epithelium of the stria vascularis, and spiral ganglion neurons (SGN) of postnatal day 3 rat cochlear organotypic cultures. 89,90 These structures contain Ctr1 in greater abundance than supporting cells and may help to explain why these structures represent the 3 major targets of cisplatin-induced cochlear damage. 89,91 Collectively, these data suggest that high levels of extracellular cisplatin are not sufficient to induce hair cell loss or stereocilia damage; rather, intracellular accumulation of cisplatin is necessary for ototoxicity and stereocilia damage to occur and identifies a significant role of copper in this process. 92
Manganese
Manganese is critically important to enzyme and membrane transport systems. It is an abundant and essential metal. 93 The degenerative effects associated with persistent Mn exposure may have deleterious consequences to other organs including the auditory system. 89,93 –101 Ding et al 90 clearly demonstrated that millimolar levels of Mn can damage hair cells and SGN. In vitro, Mn induced an atypical pattern of damage; SGN were more vulnerable to Mn than hair cells, and among the 2 hair cell populations, inner hair cell damage was greater than outer hair cells. Mirzaee et al 102 measured the effect of welding fumes and noise on the function of outer hair cells in rabbits and reported significant reductions in auditory-evoked amplitudes which were further reduced when combined with inhalation of welding fumes that contained high level of Mn plus other metals and other gases.
Conclusion
Experimental and clinical studies have elucidated some of the pathogenic mechanisms, courses, and risk factors for ototoxicity, which have led to the establishment of relevant safety assessment protocols for studies to be conducted prior to marketing approval. Further studies are warranted to better understand cellular pathways leading to degeneration, helping to build more efficient preventive interventions and contribute to our understanding of other degenerative processes such as genetic disorders. The general inclusion of ototoxicity assessments in nonclinical safety evaluation programs remains elusive. We know enough about hearing loss and its relationship with both internal and external environmental insults. To move forward, we need to know more about the “how’s and why’s” of auditory sensory organ failure and intervention strategies that can prevent cell loss before market approval. The restricted access to the sensory end organ and the elusive skills required for isolation, dissection, and processing of tissues for pathological review is the first hurdle in the process. Identifying cell death and preventing it before new drug application approval seems unlikely, but we at least attempt to combat this issue head on.
Footnotes
Authors’ Note
All contributors assisted in literature review, consolidation, and preparation of the manuscript. This manuscript was considered “work product” as a function of employment at MPI Research.
Author Contribution
Gauvin and Yoder substantially contributed to conception or design, drafted the manuscript, and gave final approval. Zimmermann and Tapp contributed to acquisition, analysis, or interpretation of data, critically revised the manuscript for important intellectual content, and gave final approval. All authors agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
