Abstract
Dimethiconol and its esters and reaction products are used in cosmetics as either skin-conditioning agents or hair-conditioning agents. The Cosmetic Ingredient Review (CIR) Expert Panel reviewed relevant data and concluded that these ingredients are safe in the present practices of use and concentration described in this safety assessment. While there is an absence of data on reproductive and developmental toxicity and limited tumorigenicity and toxicokinetics data, the Panel reasoned that these ingredients would not be absorbed through the skin, obviating concern over potential reproductive and developmental toxicity or carcinogenicity.
Keywords
Introduction
This safety assessment includes dimethiconol and its esters and reaction products. These reaction products can be categorized into 2 types: end-capped homopolymers: dimethiconol arginine, dimethiconol beeswax, dimethiconol behenate, dimethiconol borageate, dimethiconol candelillate, dimethiconol carnaubate, dimethiconol cysteine, dimethiconol dhupa butterate, dimethiconol hydroxystearate, dimethiconol illipe butterate, dimethiconol isostearate, dimethiconol kokum butterate, dimethiconol lactate, dimethiconol meadowfoamate, dimethiconol methionine, dimethiconol mohwa butterate, dimethiconol panthenol, dimethiconol sal butterate, and dimethiconol stearate and copolymers: hydrolyzed collagen propylene glycol (PG)-propyl dimethiconol, dimethiconol/methylsilanol/silicate crosspolymer, dimethiconol/silica crosspolymer, dimethiconol/silsesquioxane copolymer, dimethiconol/stearyl methicone/phenyl trimethicone copolymer, isopolyglyceryl-3 dimethiconol, trimethylsiloxysilicate/dimethiconol crosspolymer, and acrylates/dimethiconol acrylate copolymer.
The end-capped homopolymers consist of polymer chains made from dimethyl siloxyl monomers, wherein each end of the polymer chain is capped with an ester side chain (eg, dimethiconol behenate, a dimethyl siloxyl polymer which terminates on each end with the behenate ester). The copolymers consist of at least 2 monomers polymerized together. The skin-conditioning agent/hair-conditioning agent function in personal care products is associated with most of these ingredients.
Of the 28 ingredients that are being reviewed in this safety assessment, the following 10 are reported to the Food and Drug Administration (FDA) as being used in personal care products: dimethiconol, dimethiconol arginine, dimethiconol beeswax, dimethiconol cysteine, dimethiconol meadowfoamate, dimethiconol methionine, dimethi-conol panthenol, dimethiconol stearate, dimethiconol/silsesquioxane copolymer, and trimethylsiloxy-silicate/dimethiconol crosspolymer. Current use concentration data from the Personal Care Products Council (Council) also indicate that, while not reported to the Voluntary Cosmetic Registration Program (VCRP), dimethiconol behenate and acrylates/dimethiconol acrylate copolymer are also being used in cosmetic products.
The CIR Expert Panel reviewed the safety of similar chemicals, dimethicone and amodimethicone, in cosmetics and concluded that both are safe as used in cosmetic products. 1 That safety assessment noted that clinical and animal absorption studies generally reported that dimethicone was not absorbed following oral or dermal exposure. Dimethicone was not toxic following single-dose oral exposure (mice, rats, and guinea pigs), and adverse effects were not observed in rats that received up to 10% dimethicone in the diet for 90 days. Dimethicone is a minimal skin and ocular irritant and is not a sensitizer. A significant reduction in the average seminal vesicle to body weight ratio was noted in male rats dosed orally with a dimethicone sample (3.3 mL/kg/d). Absolute testis weight was significantly reduced in male rats dosed dermally with undiluted motor oil (1.5 mL/kg/d) containing an unspecified amount of dimethicone. Results for dimethicone were negative in all genotoxicity assays and in both oral (tested at 91%) and dermal (tested at unknown concentration) carcinogenicity assays using mice.
The Expert Panel also reviewed the safety of cyclomethicone, cyclotetrasiloxane (D4), cyclopentasiloxane (D5), cyclohexasiloxane (D6), and cycloheptasiloxane (D7) in personal care products and concluded that these ingredients are safe in the present practices of use and concentration. 2 Thus, there are no safety concerns relating to the potential presence of D3, D4, or D5 as an impurity in dimethiconol and dimethiconol/silsesquioxane copolymer emulsions or other materials containing dimethiconol that are being evaluated in this safety assessment.
Most of the toxicity data included in this safety assessment are related to α,ω dihydroxydimethyl-polysiloxanes associated with Chemical Abstracts Service (CAS) no. 70131-67-8 from Dow Corning. These hydroxy-terminated dimethyl siloxane (silicone) polymers are often listed in the CAS Registry and various literature references as siloxanes and silicones, dimethyl, hydroxy terminated; or dimethoxy silicone/silane, hydroxy terminated. The data herein refers specifically to Dow Corning chemicals associated with the CAS no. 70131-67-8 at concentrations of ≥95%. Siloxanes and silicones, dimethyl, hydroxy terminated and CAS no. 70131-67-8 are listed among the other chemical names/identification numbers for dimethiconol in the International Cosmetic Ingredient Dictionary and Handbook; however, the name dimethiconol is not mentioned in any of the toxicity studies. Additionally, in the CAS Registry, the name dimethiconol is associated with 2 CAS nos. 70131-67-8 and 31692-79-2; both CAS Registry files describe hydroxy-terminated dimethyl siloxane.
Chemistry
Definition and Structure
Chemical definitions, other chemical names, and cosmetic ingredient functions for the ingredients reviewed in this safety assessment are included in Table 1. 3 The ingredient moieties that have been reviewed by the CIR Expert Panel are also identified. Because the dimethiconol fatty acid (FA) moieties are of botanical origin by definition, information on the composition of oil/butter sources of these FAs is included in Table 2. Chemical structures for dimethiconol 4 and its representative siloxanes are included in Figure 1A. The chemical structures for 3 dimethiconol polymers are included in Figure 1B.
Dimethiconol and Its Esters and Reaction Products. 3

Composition of Oil/Butter Sources of Dimethiconol FA Moieties.a

aThe Cosmetic Ingredient Review (CIR) Expert Panel has evaluated the safety of palmitic acid, stearic acid, and oleic acid and concluded that each is safe as used in personal care products. 51
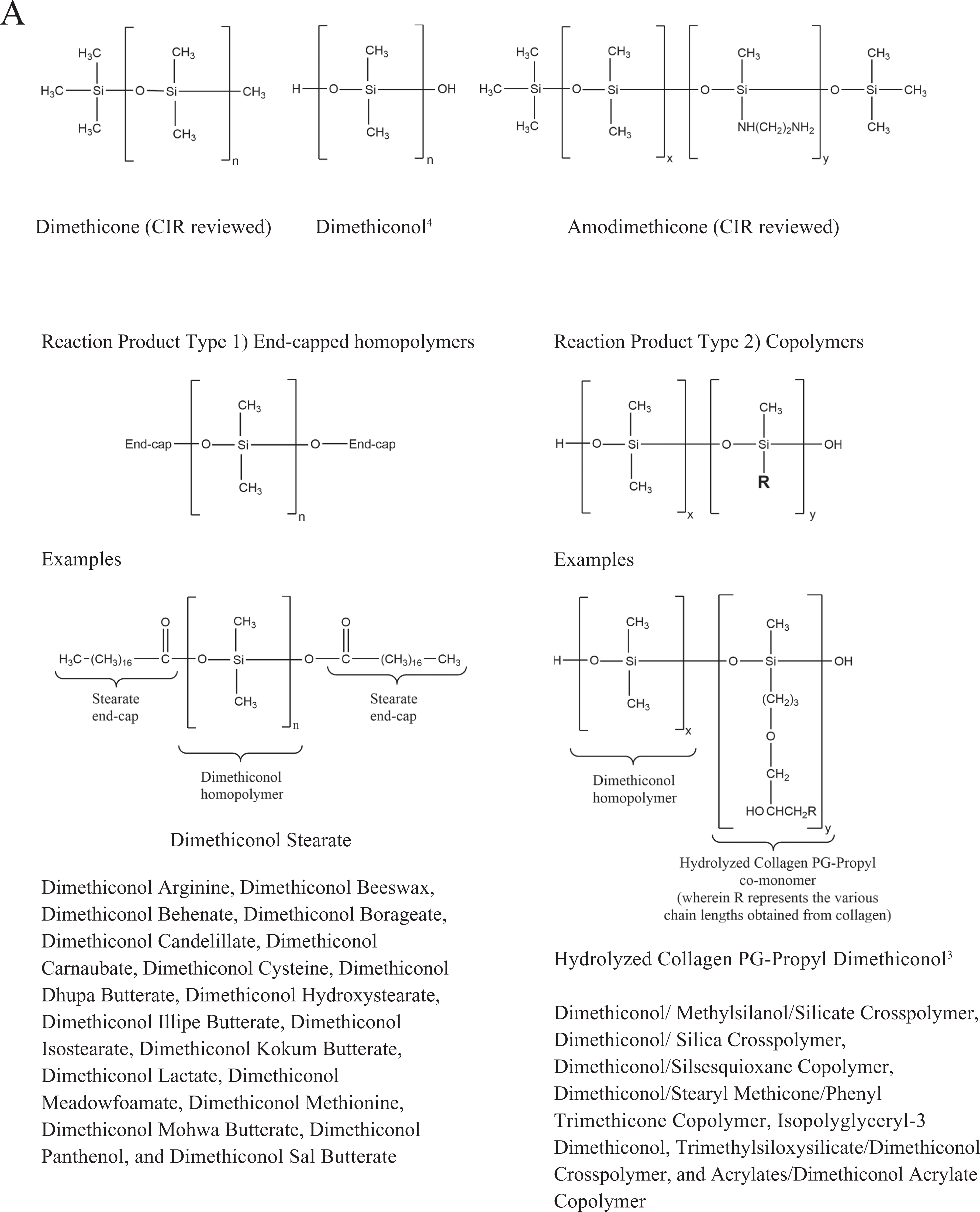
A, Structures for dimethiconol, examples of its reaction products and related, reviewed ingredients. Dimethicone (CIR reviewed) dimethiconol 4 amodimethicone (CIR reviewed) reaction product type 1 end-capped homopolymers. Examples: dimethiconol stearate dimethiconol arginine, dimethiconol beeswax, dimethiconol behenate, dimethiconol borageate, dimethiconol candelillate, dimethiconol carnaubate, dimethiconol cysteine, dimethiconol dhupa butterate, dimethiconol hydroxystearate, dimethiconol illipe butterate, dimethiconol isostearate, dimethiconol kokum butterate, dimethiconol lactate, dimethiconol meadowfoamate, dimethiconol methionine, dimethiconol mohwa butterate, dimethiconol panthenol, and dimethiconol sal butterate. Reaction product type 2 copolymers. Examples: hydrolyzed collagen PG-propyl dimethiconol, 3 dimethiconol/ methylsilanol/silicate crosspolymer, dimethiconol/silica crosspolymer, dimethiconol/silsesquioxane copolymer, dimethiconol/stearyl methicone/phenyl trimethicone copolymer, isopolyglyceryl-3 dimethiconol, trimethylsiloxysilicate/dimethiconol crosspolymer, and acrylates/dimethiconol acrylate copolymer. B, Structures for dimethiconol polymers. Dimethiconol stearate, hydrolyzed collagen PG-propyl dimethiconol, isopolyglyceryl-3 dimethiconol.
Data provided by the (Council) 5 indicate that dimethiconol stearate and dimethiconol beeswax are supplied at approximately 100% active. Similar information on the remaining ingredients included in this review was not provided.
Chemical and Physical Properties
Dimethiconol and the copolymers have a reactive hydroxyl group on the terminal portion of the molecule. The hydroxyl group is bonded directly to the silicon atom in a silicon–oxygen bond. These compounds condense, under acid or alkaline catalysis, and also undergo ethoxylation. When these compounds undergo condensation reactions in the presence of an acid or base, the molecular weight is increased (ie, an increase in the n value) and water is released. 6
In addition to the definition of dimethiconol/silsesquioxane copolymer provided in Table 1, the Silicones Environmental, Health and Safety Council of North America (SEHSC) defines dimethiconol/silsesquioxane as the product of a condensation reaction between dimethiconol and methyl trimethoxysilane and defines silsesquioxanes as siloxane polymers that contain silicon atoms bonded to 3 other silicon atoms via siloxane bonds. 7 According to the SEHSC, the SiOH groups that terminate the siloxane polymer chains in dimethiconol are reactive under certain circumstances. One common reaction is a condensation reaction with alkoxy-terminated siloxanes and alkoxy silanes. In this reaction, the SiOH groups react with the alkoxy groups to form a new siloxane (SiOSi) bond with the release of the corresponding alcohol. So, for dimethiconol/silsesquioxane, the dimethiconol polymer reacts with the methoxy groups on methyl trimethoxysilane, releasing methanol and forming new siloxane bonds. Since there are 3 methoxy groups on this silane, the reaction produces a 3-dimensional siloxane polymer network in which dimethyl siloxane polymers link together silsesquioxane units.
The limited available data on the properties of dimethiconol, dimethiconol beeswax, dimethiconol behenate, and dimethiconol/silsesquioxane copolymer (5%) and dimethiconol (20%) in anionic surfactant emulsion are included Table 3; octanol-water partition coefficients on these compounds are not included. Data on the remaining compounds reviewed in this safety assessment were not found. However, properties/composition data, including the high molecular weight, on Dow Corning materials and other materials that are considered by the silicones industry to represent dimethiconol are included in Table 4. 7
Properties of Dimethiconol and Dimethiconol Compounds.

Properties of Materials registered Under Dimethiconol INCI Name. 64
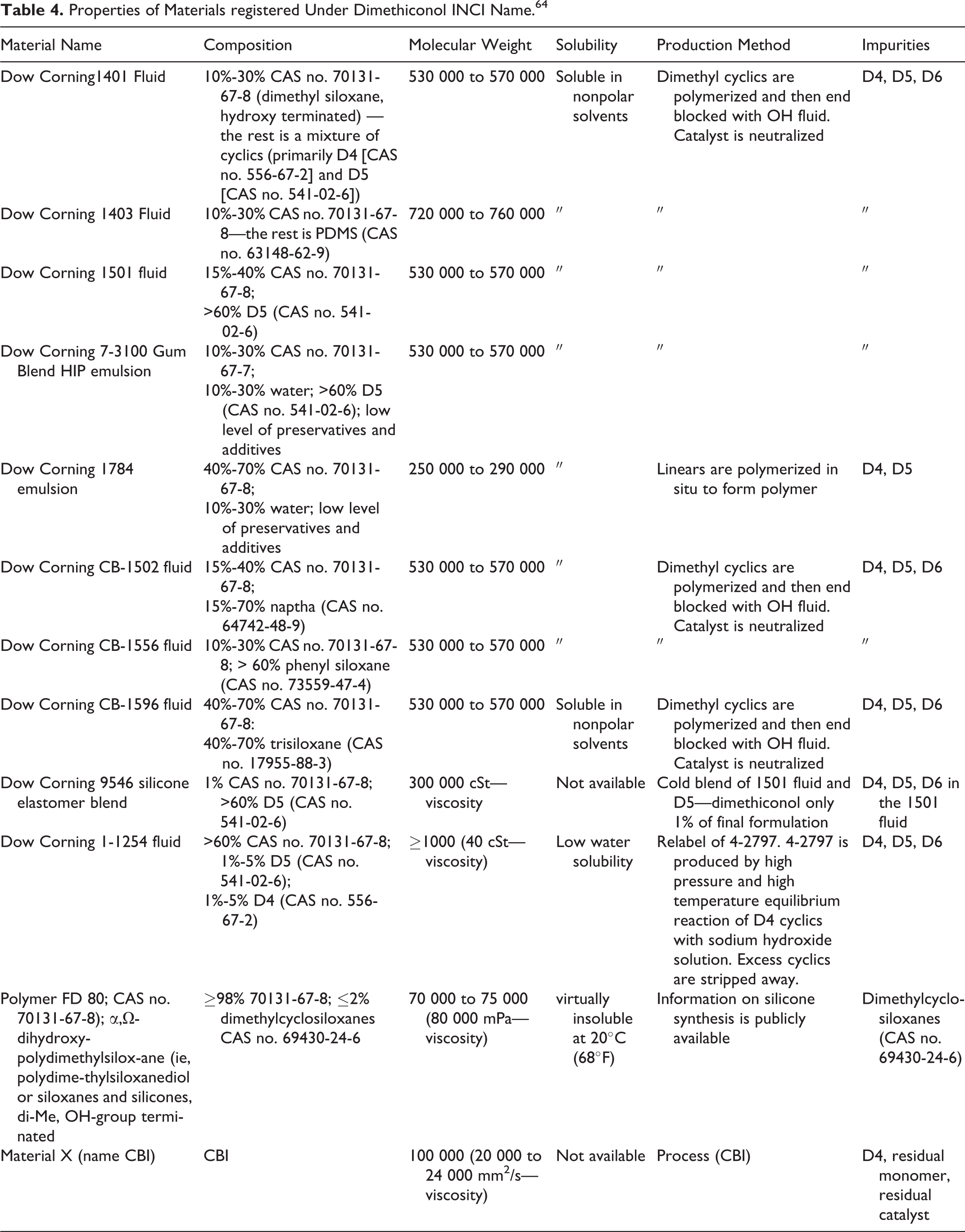
Table 5 contains data on the composition of materials (mostly trade mixtures) that contain dimethyl siloxane, hydroxy terminated (CAS no. 70131-67-8). 7 These materials are components of test materials evaluated in various toxicity tests included in this safety assessment.
Tested Materials Not Used in Cosmetics That Contain Dimethyl Siloxane, Hydroxy Terminated. 64

Analytical Methods
Dimethiconol has been analyzed via infrared spectroscopy. 8 The same method has been used to analyze dimethiconol (60%) in anionic surfactant emulsions 9 and dimethiconol/silsesquioxane copolymer (5%) and dimethiconol (20%) in anionic surfactant emulsions. 9
Ultraviolet (UV) Absorption
Ultraviolet absorption data (spectra not provided) on the following Dow silicone products were provided by the SEHSC: Dow Corning 9564 Silicone Elastomer Blend, Dow Corning® 1501 Fluid, Dow Corning 1503 Fluid, and Dow Corning Q1-3563. 7 Of these 4 materials, only Dow Corning 9564 Silicone Elastomer Blend and Dow Corning 1501 Fluid are registered under the INCI name Dimethiconol.
For Dow Corning 9564 Silicone Elastomer Blend, between 6% and 7% of UVB radiation was absorbed by this sample (2 mg/cm2). Based on the small amount of dimethiconol in this blend and the results obtained for the other samples, it was determined that there was no basis for concluding that dimethiconol was absorbing significant amounts of UVB or UVA radiation. For Dow Corning 1501 Fluid, ∼1% of the UVB radiation was absorbed by the sample. Detector noise was thought to have contributed to this finding. Absorbance in the UVA region of the spectrum was very close to zero.
For Dow Corning 1503 Fluid, absorbance in the UVA region was very close to zero—what absorbance was found was attributed to detector noise. Similar results were obtained for Dow Corning Q1-3563. Absorbance in the UVB region was very close to zero. 7
Use
Most of the ingredients reviewed in this safety assessment function either as a skin-conditioning or hair-conditioning agent in personal care products (Table 1). 3
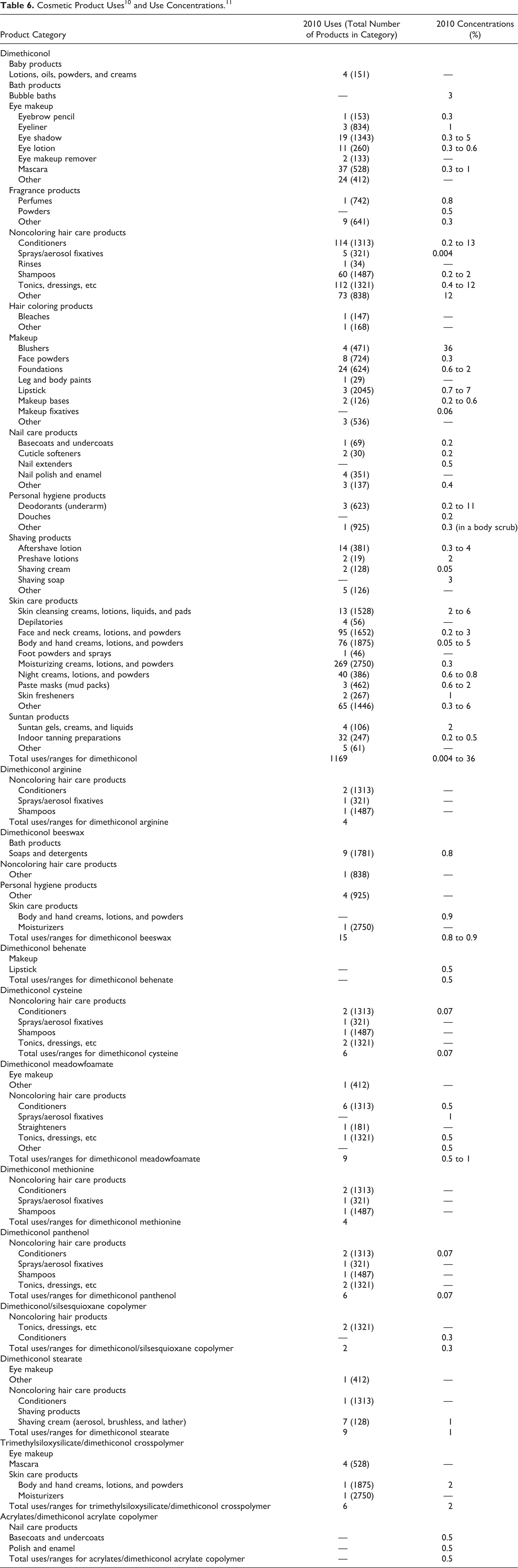
According to information supplied to the FDA by industry as part of the VCRP 10 and the results of a survey of current ingredient use concentrations that was conducted by the Council 11 obtained in 2010, the following ingredients are being used in personal care products: dimethiconol, acrylates/dimethiconol acrylate copolymer, dimethiconol arginine, dimethiconol beeswax, dimethiconol behenate, dimethiconol cysteine, dimethiconol meadowfoamate, dimethiconol methionine, dimethiconol panthenol, dimethiconol stearate, dimethiconol/silsesquioxane copolymer, and trimethylsiloxy-silicate/dimethiconol crosspolymer. Dimethiconol is the most widely used ingredient and has the highest concentrations of use; it is used in 1169 cosmetic formulations and the concentrations of use range from 0.004% to 36% in these products. These data are summarized in Table 6.
In some cases, uses are reported to the VCRP, but use concentration data are not available, eg, dimethiconol arginine. In other cases, some ingredients are not reported as in use according to the VCRP but are reported in the concentration of use survey, eg, dimethiconol behenate.
Personal care products containing these ingredients may be applied to the skin, nails, or hair, or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day, and daily or occasional use may extend over many years.
Dimethiconol and several of the esters are used in hair sprays, raising the potential for incidental inhalation exposure the aerosol properties that determine deposition in the respiratory system are particle size and density. The parameter most closely associated with deposition is the aerodynamic diameter, da, defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤10 µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract. 12,13
Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 14 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range, and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. 15 Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
Noncosmetic Use
The insecticidal activity of dimethoxy silicone/silane, hydroxy terminated has been reported. 16
Toxicokinetics
Information on absorption, distribution, metabolism and excretion of the ingredients reviewed in this safety assessment was not found. However, it is not expected that these ingredient would not be absorbed through the skin and metabolized due to their high molecular weights.
Toxicology
The published literature was not found to contain short-term toxicity, subchronic toxicity, reproductive toxicity, or phototoxicity/photosensitization data on the ingredients reviewed in this safety assessment.
Single-Dose Toxicity
Inhalation
The acute inhalation toxicity
Oral
A mixture containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl (20%) was evaluated using groups of 10 Hilltop-Wistar albino rats (5 males, 5 females/group). 17 The test substance was administered by stomach tube up to a dose of 16.0 mL/kg. None of the animals died and there were no signs of toxicity. Mottled lungs (red or pink and dark red) were noted at necropsy. The lethal dose (LD)50 was >16.0 mL/kg.
A suspension containing Dow Corning 60 000CSt, no cosolvent in corn oil (20% w/v; containing ≥95% polysiloxanes, di-Me, hydroxy terminated) was administered by gavage to rats, and a summary of the results was provided by Dow Corning Corporation. 18 A single dose of the test substance (2 g/kg body weight) was administered to 10 fasted Sprague-Dawley rats (6 weeks old) by gavage. None of the animals died and there were no overt signs of toxicity during the 14-day observation period. Lesions were not observed at gross necropsy. The LD50 was >2 g/kg body weight.
Polymer FD 80 was evaluated using Sprague-Dawley rats (5 males, 5 females). The test substance was administered by gavage at a dose of ∼2 g/kg, and necropsy was performed after day 14. None of the animals died and there was no evidence of clinical signs. The LD50 was >2 g/kg. 19
Dimethiconol stearate was evaluated using 10 fasted, Wistar-derived albino rats (5 males, 5 females).
Dermal
The acute dermal toxicity 17 of a mixture containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl) (20%) was evaluated using groups of 8 New Zealand White rabbits (4 males, 4 females/group). The test substance was applied (doses up to 16.0 mL/kg; 24-hour period) under impervious plastic sheeting to clipped, intact skin of the trunk. Skin irritation was not noted. One male rabbit and 2 female rabbits died (all at 16 mL/kg dose). Mottled lungs (males) and mottled livers/lungs (females) were noted at necropsy. There were no remarkable necropsy findings in surviving animals. LD50s were >16.0 mL/kg for males and females.
In another study, 21 the acute dermal toxicity of siloxanes and silicones, dimethyl, hydroxy terminated (22 wt. %) in Dow Corning 2-1845 microemulsion was evaluated using 12 (6 males, 6 females) New Zealand White rabbits of the Hra:(NZW)SPF strain. The undiluted test substance was applied (under an occlusive wrap) to clipped dorsal skin at a dose of 2 g/kg (dose volume = 1.9741 mL/kg) for approximately 24 hours. The following reactions (all test substance related) were observed at the application site: erythema and desquamation (6 rabbits), erythema and edema (1 rabbit), and desquamation (1 rabbit). None of the animals died during the 14-day study, and there were no test substance-related effects on body weight gain. Macroscopic findings were not observed at necropsy. It was concluded that the Dow Corning 2-1845 microemulsion was nontoxic (LD50 > 2 g/kg).
An acute dermal toxicity study summary on Dow Corning 60 000CSt, no cosolvent was provided by Dow Corning Corporation. 18 The test substance was applied to the skin of each of 10 (5 males, 5 females) New Zealand white rabbits for 24 hours. Erythema was observed at application sites, having cleared by day 7. None of the animals died and there were no signs of systemic toxicity during the 14-day observation period. An acute dermal LD50 of >2 g/kg body weight was reported.
The acute dermal toxicity of polymer FD 80 was evaluated using Sprague-Dawley rats (5 males, 5 females). The test substance was applied to the skin at a dose of ∼2 g/kg, and necropsy was performed after day 14. None of the animals died and there was no evidence of pathological clinical signs. The LD50 was > 2 g/kg. 22
Repeated-Dose Toxicity
In an 8-month feeding study, 23 6 of 18 rabbits were fed 0.05% Dow Corning special polymer 5-26-64, a polymerized siloxane containing siloxanes and silicones, dimethyl, hydroxy terminated, in a basal diet. The remaining 12 rabbits comprised the control group (basal diet only). Both groups had equal numbers of males and females. None of the animals died during the feeding period, and all animals were killed after 8 months. While incomplete testicular development was reported in 2 treated males, the finding is consistent with historical results in untreated animals. Other endpoints such as liver and kidney toxicity were not different between controls and treatment animals. In a subsequent analysis of the methodology, however, it was noted that there was no way to determine the actual dose level that the rabbits received and no discussion on test article concentration, stability, homogeneity, suggesting that the findings should not be used in health and safety assessments. 7
A chronic feeding study on Dow Corning special polymer, identified as siloxanes and silicones, dimethyl, hydroxy terminated, was conducted using 30 albino weanling rats. 24 The test group consisted of 10 rats (5 males, 5 females), and these animals were fed a basal diet consisting of 0.05% Dow Corning special polymer for 1 year. The control group (10 males, 10 females) was fed basal diet only. The only reported deaths were 2 rats in the control group. There were no test substance-related effects on hematological or clinical chemistry values. Gross evidence of severe pulmonary disease was noted at necropsy. Inflammatory changes in the lungs or tubular degenerative changes in the kidneys were fairly common in test and control groups and were not considered test substance related. It was concluded that administration of the test substance did not induce adverse effects in rats. Subsequently, Dow Corning determined that the preceding study is scientifically invalid based on the following rationale: “No ability to determine the actual dose level that the rabbits [rats] received. No discussion on test article concentration, stability, homogeneity. We do not feel that this study should be used in health and safety assessments and do not want the general public using it because of limitations.” 7
Ocular Irritation
The ocular irritation potential of an emulsion (Dow Corning 35 emulsion) containing siloxanes and silicones, dimethyl, hydroxy terminated at a concentration of 13% and another emulsion (Dow Corning 22 emulsion) containing siloxanes and silicones, dimethyl, hydroxy terminated at a concentration of 38% was evaluated using 2 groups of 10 albino rabbits (1 per test substance). 25 Two drops of each test material were instilled into 1 eye. While Dow Corning 35 emulsion did not induce a significant ocular response in rinsed or unrinsed eyes and Dow Corning 22 emulsion elicited slight, transient conjunctivitis only in the unrinsed eye, the method was considered not robust enough to satisfy current health safety testing standards.
The ocular irritation potential of a mixture 17 containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl) (20%) was evaluated using groups of 6 New Zealand White rabbits (3 males, 3 females). The test substance was instilled into the lower conjunctival sac of 1 eye per animal per group at volumes up to 0.1 mL. Six eyes were dosed per test volume. Dose volumes of 0.005, 0.01, and 0.1 mL induced moderate, persistent corneal and conjunctival injury (in all rabbits per group). Moderate iritis was also observed at a dose volume of 0.1 mL. All reactions had cleared by day 21 postinstillation. Study results indicated that the test material caused moderate, persistent injury when instilled into the eye.
An ocular irritation study summary on Dow Corning 60 000CSt, no cosolvent was provided by Dow Corning Corporation. 18 The undiluted test substance was instilled (0.1 mL) into the right eye of each of 3 female New Zealand white rabbits (3-4 months old). Conjunctival erythema, chemosis, and discharge were observed in all rabbits, having cleared by 72-hour postinstillation. Lesions of the cornea or iris were not observed. The test substance was classified as a nonirritant.
The SEHSC 26 also provided a summary of an ocular irritation study using rabbits dosed with Dow Corning PA Fluid. Direct contact with the test substance resulted in very slight redness in the unrinsed rabbit eye through 48 hours. The rinsed eye was clear at 24-hour postexposure.
The ocular irritation potential of dimethiconol stearate was evaluated using 6 healthy, young adult New Zealand albino rabbits. 27 The test substance (0.1 mL) was instilled into the 1 eye of each animal; contralateral eyes served as controls. Ocular lesions were evaluated according to the Draize scale (0-110). An ocular irritation score of 0 was reported for each rabbit, and dimethiconol stearate was classified as nonirritating to the eyes of rabbits.
Mucous Membrane Irritation
The mucous membrane irritation potential of 3 Dow Corning materials (TX-102A, TX-102B, and TX-102C) 28 containing 82.1% siloxanes and silicones, dimethyl, hydroxy terminated was evaluated using 6 dogs (2 dogs per test material). Each material (amounts ranging from 8 to 18 g) was maintained in contact with the hard palate for 7 hours, using an aluminum mold previously shaped to the contour of the roof of the mouth. At the end of the contact period, the oral cavity was examined for evidence of irritation or lesions. The animals were killed on day 8, and punch biopsy specimens of the hard palate were obtained and examined microscopically. Test materials TX-102A and TX-102B did not induce irritation of the hard palate. Test material TX-102-C induced slight edema of the hard palate in both dogs; the edema had cleared by the end of the 8-day observation period. Results of microscopic examinations were not reported. However, according to the SEHSC, microscopic examinations were considered normal for all samples in this study. 29
A mucous membrane irritation study on Dow Corning 4-2797 (X7-9192), dimethylsiloxane hydroxy-terminated fluid was provided by Dow Corning Corporation. 30 Following application of the test substance (0.5 g) to the vaginal mucosa of each of 6 New Zealand white rabbits (10-12 weeks old), there were no signs of irritation, weight loss, or clinical signs of toxicity during the 72-hour observation period.
Skin Irritation and Sensitization
The skin irritation potential of a mixture 17 containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl) (20%) was evaluated using 6 New Zealand White rabbits (3 males, 3 females). The test substance was applied (0.5 mL, 4-hour application) under a gauze patch to clipped, intact skin. The patch was covered with impervious sheeting. Reactions were scored according to the following Draize scale: 0 (no erythema) to 4 (severe erythema). Skin irritation was not observed in any of the rabbits, and the test material was classified as a nonirritant.
A skin irritation study on Dow Corning 60 000CSt, no cosolvent was provided by Dow Corning Corporation. 18 The undiluted test substance (0.5 g/240 cm2) was applied to clipped/shaved skin of the back of each of 3 female New Zealand white rabbits (3-4 months old). The application site was covered with a cotton gauze patch (2.5 × 2.5 cm) secured with porous tape for 4 hours. Reactions were scored for up to 72-hour postremoval. None of the rabbits had signs of dermal irritation or corrosivity, and the test substance was classified as a nonirritant.
The skin irritation potential of dimethiconol stearate was evaluated using 6 healthy, New Zealand albino rabbits. 31 The test substance (0.5 g under a 2.5-cm2 patch) was applied to intact and abraded skin sites on the trunk, clipped free of hair. The entire trunk was wrapped with a rubberized elastic cloth during the 24-hour application period. Reactions were scored at 24 and 72 hours according to the following scales: 0 (no erythema) to 4 (severe erythema to slight eschar formation) and 0 (no edema) to 4 (extreme edema). Skin irritation was not observed in any of the animals tested (primary irritation index = 0).
A skin irritation and sensitization study on Dow Corning X7-9192, dimethyl siloxane, hydroxy terminated was provided by Dow Corning Corporation. 32 In the primary irritation test, the test substance (0.1 mL in H2O, under Finn chamber [2 × 4 cm patch]) was applied to the skin of each of 4 young adult guinea pigs. Concentrations ranging from 25% to 100% were applied and reactions were scored for up to 72-hour postapplication. Skin irritation was not observed over the range of test concentrations.
The sensitization potential of Dow Corning X7-9192 (5% in water emulsion) was evaluated in the maximization test using groups of 10 (5 males, 5 females) guinea pigs. Intradermal injections (0.1 mL) of the test substance were administered on day 0. On day 7 of induction, the test substance was applied under an occlusive patch for 48 hours. During the challenge phase, initiated on day 21, the test substance was applied under an occlusive patch for 24 hours. Reactions were scored on days 23 and 24. Sodium chloride (0.9%) and dinitrochlorobenzene (DNCB) (0.1%) served as vehicle and positive controls, respectively. The test substance did not induce sensitization. 32
Skin sensitization data on Dow Corning 60 000CSt, no cosolvent in Dow Corning 360 Medical Fluid (5% w/v) were provided by Dow Corning Corporation. 18 The maximization test involved the following groups of male Hartley guinea pigs (4 weeks old): 20 test, 10 vehicle controls (Dow Corning 360 Medical Fluid), and 10 positive controls (DNCB in propylene glycol, 1% w/v). The first induction involved intradermal injections (0.1 mL per injection) of the undiluted test substance, vehicle control, and positive control in the respective groups. The second induction involved the 48-hour application of a 2 × 4 cm pad saturated with each substance per group. At 2 weeks after the last induction, test animals were challenged with the undiluted test substance (0.3 mL), and both control groups were also challenged with respective materials. Positive responses were not observed in test or vehicle control animals, and the test substance was not considered a skin sensitizer.
Prior to the following maximization test, 3 preliminary studies (4 guinea pigs per study) were conducted to evaluate the skin irritation potential of polymer FD 80. 33 In study #1 (for induction), moderate irritation was observed in 4 guinea pigs at 24 and 48 hours after intradermal injection with 50% polymer FD 80 in liquid paraffin, and weak to moderate irritation was observed in these animals after injection at a concentration of 10%. In another study (study #2, for induction), undiluted polymer FD 80 (0.5 mL) and the same polymer at a concentration of 50% in liquid paraffin were each applied to an 8-cm2 area of skin for 48 hours using occlusive patches. A weak irritant response was observed in 1 guinea pig patch tested with 50% FD 80 in study #2. In the final preliminary study (study #3, for challenge), skin irritation was not observed following a 24 or 48 hours occlusive patch application of undiluted or 50% FD 80 in liquid paraffin to a 4-cm2 area of skin. It was concluded that polymer FD 80, as supplied, was a nonirritant.
The skin sensitization potential of polymer FD 80 was evaluated in the maximization test using 2 groups of 20 Dunkin-Hartley albino guinea pigs, one of which was the control group. The induction phase consisted of 0.1 mL intradermal injections of 10% or 20% polymer FD 80 in liquid paraffin and 48 hours occlusive patch applications of undiluted polymer FD 80 (0.5 mL) to an 8-cm2 area of skin. The challenge phase involved a 24-hour occlusive patch application of undiluted polymer FD 80 (0.5 mL) to a 4-cm2 area of skin. It was concluded that polymer FD 80 did not induce sensitization. Sensitization reactions also were not observed in control guinea pigs treated with liquid paraffin. 33
Genotoxicity
In the Ames spot plate test and overlay plate test, 34 the mutagenicity of uncured and cured Dow Corning X3-5040 sealant containing approximately 75% siloxanes and silicones, dimethyl, hydroxy terminated was evaluated using the following Salmonella typhimurium strains with and without metabolic activation: TA98, TA100, TA1535, TA1537, and TA1538. The test substance was extracted with dimethylsulfoxide (DMSO) by continuous shaking at 150 rpm/37°C for 24-hour prior to the test, and doses up to 500 μL/plate were tested. The positive control for activation assays was 2-anthramine, and the nonactivation assay positive controls were sodium azide, 9-amino acridine, and 2-nitrofluorene. Dimethylsulfoxide was used as the solvent control. In both the spot and overlay plate tests, results for the test substances and solvent control were negative in all strains, both with and without metabolic activation, and the positive controls were mutagenic. The test materials were considered nonmutagenic.
In another Ames plate test, 35 a mixture containing siloxanes and silicones, dimethyl, hydroxy terminated (concentration not stated; solvent, acetone) was evaluated using the following S typhimurium strains with and without metabolic activation: TA98, TA100, TA1535, TA1537, and TA1538. Concentrations up to 150 µL/plate were tested. The positive control for activation assays was 2-anthramine in DMSO, and the nonactivation assay positive controls were sodium azide, 2-nitrofluorene, and quinacrine mustard. Results for the test substance were negative in all strains, both with and without metabolic activation, and the positive controls were mutagenic. The test substance was considered nonmutagenic.
A study on Dow Corning Q4-2797, dimethylsiloxane, hydroxy-terminated fluid was provided by the Dow Corning Corporation. 36 This fluid (in DMSO, doses up to 5000 µg/plate) was evaluated in the Ames test using the following bacterial strains with and without metabolic activation: S typhimurium strains TA97, TA98, TA100, and TA 1535, and Escherichia coli strain WP2. The following positive controls were used: sodium azide, 4-nitroquinoline-N-oxide, daunomycin, and N-methyl-N-nitro-N-nitrosoguanidine (with metabolic activation) and 2-anthramine and 2-aminofluorene (without metabolic activation). The test substance was not mutagenic to any of the strains tested. All positive controls were mutagenic.
A study on Dow Corning 60 000CST no cosolvent was also provided by Dow Corning Corporation. 18 Test substance doses up to 5000 µg/plate were evaluated in an Ames test using the following bacterial strains with and without metabolic activation: S typhimurium strains TA98, TA100, TA1535, TA1537, and E coli strains WP2uvrA and WP2uvrA (pKM101). The following positive controls were used: 2-aminoanthracene (with metabolic activation), and sodium azide, 2-nitrofluorene, 9-aminoacridine, and methyl methanesulfonate (without metabolic activation). The test substance was not mutagenic to any of the strains tested. All positive controls were mutagenic.
Implant Tumorigenicity
Chronic implantation studies of polysiloxanes were conducted using 38 pure-bred Beagle dogs (∼5-7 months old). 37 The 4 implanted materials (implant mass not stated) were defined as follows: siloxanes and silicones, dimethyl, hydroxy terminated (68%) in uncured DC 386; siloxanes and silicones, dimethyl, hydroxy terminated (72%) in uncured DC 382; siloxanes and silicones, dimethyl, hydroxy terminated (96%) in uncured DC 5392; and siloxanes and silicones, dimethyl, hydroxy terminated (80%) in uncured medical adhesive type A. Except for uncured medical adhesive type A, the remaining compositions of materials tested (ie, uncured DC 382 and DC 386) are unknown. The implants (intramuscular, intraperitoneal, and subcutaneous) were removed at intervals of 3, 9, 24, and 36 months. Neither gross nor microscopic findings revealed a pattern of polymer-induced systemic toxicity. The materials tested also did not induce any untoward chronic tissue reactions, and there was no evidence of tumorigenesis over a 3-year testing period.
Clinical Assessment of Safety
Skin Irritation and Sensitization
Dimethiconol
In an RIPT,
38
the skin irritation and sensitization potential of a body lotion containing 1.125% dimethiconol (0.2 g per 1
Dimethiconol Behenate
In another RIPT (occlusive patches, similar procedure), 39 the skin irritation and sensitization potential of lip product containing 0.5% dimethiconol behenate was evaluated using 50 subjects ranging in age from 18 to 70 years. The dose per cm2 was not stated. There were no visible skin reactions in any of the subjects, and it was concluded that the lip product did not demonstrate a potential for eliciting dermal irritation or sensitization.
Dimethiconol Beeswax
The skin sensitization potential of a test product identified as undiluted dimethiconol beeswax was evaluated in an RIPT using 102 subjects (29 males, 73 females; >18 years old) with no significant active skin pathology. 40 During induction, the test material was applied to the back (0.025 g/cm2 skin, 8 mm Finn chambers) of each subject, for a total of 10 occlusive patch applications. Each chamber remained in place for 48 hours. Following a 12-day nontreatment period, an occlusive challenge patch was applied for 48 hours to a new site on the back of each subject. Reactions were scored at 48- and 96-hour postapplication according to the following scale: 0 (no reaction) to 4 (erythema, edema, and bullae). Dimethiconol beeswax did not induce skin irritation or sensitization in this study.
Summary
The following ingredients are reviewed in this safety assessment: dimethiconol, dimethiconol arginine, dimethiconol beeswax, dimethiconol behenate, dimethiconol borageate, dimethiconol candelillate, dimethiconol carnaubate, dimethiconol cysteine, dimethiconol dhupa butterate, dimethiconol hydroxystearate, dimethiconol illipe butterate, dimethiconol isostearate, dimethiconol kokum butterate, dimethiconol lactate, dimethiconol meadowfoamate, dimethiconol methionine, dimethiconol/methylsilanol/silicate crosspolymer, dimethiconol mohwa butterate, dimethiconol panthenol, dimethiconol sal butterate, dimethiconol/silica crosspolymer, dimethiconol/silsesquioxane copolymer, dimethiconol stearate, dimethiconol/stearyl methicone/phenyl trimethicone copolymer, hydrolyzed collagen PG-propyl dimethiconol, isopolyglyceryl-3 dimethiconol, trimethylsiloxysilicate/dimethiconol crosspolymer, and acrylates/dimethiconol acrylate copolymer. The skin-conditioning agent/hair-conditioning agent function in personal care products is associated with most of these ingredients.
Of the 28 ingredients that are being reviewed in this safety assessment, the following 12 are reported to the FDA as being used in personal care products: dimethiconol, acrylates/dimethiconol acrylate copolymer, dimethiconol arginine, dimethiconol beeswax, dimethiconol behenate, dimethiconol cysteine, dimethiconol meadowfoamate, dimethiconol methionine, dimethiconol panthenol, dimethiconol stearate, dimethiconol/silsesquioxane copolymer, and trimethylsiloxysilicate/dimethiconol crosspolymer. Dimethiconol is the most widely used ingredient, and hs the highest concentrations of use; it is used in 1169 cosmetic formulations, and the concentrations of use range from 0.004% to 36% in these products. Most of the toxicity data included in this safety assessment are on siloxanes and silicones, dimethyl, hydroxy terminated; dimethoxy silicone/silane, hydroxy terminated; and Dow Corning materials containing 95% or greater CAS no. 70131-67-8 (polysiloxanes, di-Me, hydroxy terminated). The CAS number for these chemical names is identified as 70131-67-8 in these studies. Siloxanes and silicones, dimethyl, hydroxy terminated, and CAS no. 70131-67-8 are listed among the other chemical names/identification numbers for dimethiconol in the International Cosmetic Ingredient Dictionary and Handbook.
In an acute inhalation toxicity study, neither deaths nor toxic signs were reported for rats exposed to vapor substantially saturated with a mixture containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl (20%) for 6 hours. Similar results were reported for the same mixture in an acute oral toxicity study involving rats (LD50 > 16 mL/kg), for polymer FD 80 (LD50 > 2 g/kg, rats), and for a suspension containing Dow Corning 60 000CSt, no cosolvent in corn oil (20% w/v; LD50 > 2 g/kg, rats). The latter test substance contains 95% or greater CAS no. 70131-67-8 (polysiloxanes, di-Me, hydroxy terminated). Dimethiconol Stearate was also classified as nontoxic in an acute oral toxicity study involving rats (LD50 > 5 g/kg).
Following a single dermal application of a mixture containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl (20%), irritation was not observed at application sites and 3of 8 rabbits died (LD50 > 16 mL/kg). Both siloxanes and silicones, dimethyl, hydroxy terminated (22 wt. %) in Dow Corning 2-1845 microemulsion and Dow Corning 60 000CSt, NO CO-SOLVENT (contains ≥95% polysiloxanes, di-Me, hydroxy terminated) were nontoxic (LD50 > 2 g/kg) in acute dermal toxicity studies involving rabbits; skin irritation was observed at application sites. Polymer FD 80 was also classified as nontoxic (LD50 > 2 g/kg) in a dermal toxicity study.
Transient ocular irritation, not classified as moderate or severe, was observed following the instillation of Dow Corning 60 000CSt, no cosolvent or Dow Corning PA Fluid (≥95% polysiloxanes, di-Me, hydroxy terminated in both) into the eyes of rabbits. Dimethiconol stearate was classified as nonirritating to the eyes of rabbits. However, a mixture containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl) (20%) induced moderate, persistent conjunctival and corneal injury and iritis in rabbits. This mixture did not induce skin irritation in rabbits.
Both a mixture containing dimethoxy silicone/silane, hydroxy terminated (80%) and 1-propamine, 3-(trimethoxysilyl)-N-(3-trimethoxysiloyl propyl) (20%) and undiluted Dow Corning, no Cosolvent (contains ≥95% polysiloxanes, di-Me, hydroxy terminated) were not irritating to the skin of rabbits. The same was true for dimethiconol stearate (0.5 g applied to skin) in rabbits.
Three Dow Corning materials (TX-102A, TX-102B, and TX-102C) containing 82.1% siloxanes and silicones, dimethyl, hydroxy terminated were maintained in contact with the hard palate of dogs; only 1 (TX-102)-induced irritation (slight edema). Neither signs of vaginal mucosal irritation, weight loss, nor clinical signs of toxicity were observed in rats receiving an application of Dow Corning 4-2797 (X7-9192), dimethylsiloxane hydroxy-terminated fluid (contains ≥95% polysiloxanes, di-Me, hydroxy terminated) to the vaginal mucosa.
Dow Corning X7-9192, dimethyl siloxane, hydroxy terminated (contains ≥95% polysiloxanes, di-Me, hydroxy terminated) was not a skin irritant in guinea pigs patch tested with concentrations up to 100% and, at a concentration of 5% in a water emulsion, also did not induce sensitization in the maximization test. Maximization test results for undiluted polymer FD 80 and Dow Corning 60 000CSt, no cosolvent in Dow Corning 360 Medical Fluid (5% w/v) were also negative in guinea pigs. This Dow Corning material contains ≥95% polysiloxanes, di-Me, hydroxy terminated.
Negative Ames test results were reported for the following chemicals: uncured and cured Dow Corning X3-5040 sealant containing ∼75% siloxanes and silicones, dimethyl, hydroxy terminated (doses up to 500 µL/plate); a mixture containing siloxanes and silicones, dimethyl, hydroxy terminated (up to 150 µL/plate); Dow Corning 4-2797, dimethylsiloxane, hydroxy-terminated fluid (contains ≥95% polysiloxanes, di-Me, hydroxy terminated; up to 5000 µg/plate); and Dow Corning 60 000CST no cosolvent (contains ≥95% polysiloxanes, di-Me, hydroxy terminated; up to 5000 µg/plate).
In chronic implantation studies (38 pure-bred Beagle dogs), 4 materials containing siloxanes and silicones, dimethyl, hydroxy terminated at concentrations of 68%, 72%, 80%, and 96%, respectively, were tested. The materials were removed at various intervals up to 36 months postimplantation, and neither gross nor microscopic findings were indicative of polymer-induced toxicity or tumorigenesis.
Negative results were reported in the following RIPTs evaluating skin irritation and sensitization potential: body lotion containing 1.125% dimethiconol (0.2 g on 1″ × 1″ patch; 104 subjects), lip product containing 0.5% dimethiconol behenate (50 subjects), and undiluted dimethiconol beeswax (0.025 g/cm2; 102 subjects).
Discussion
The Panel noted gaps in the available safety data for some of the dimethiconol compounds in this safety assessment. The available data on dimethiconol are sufficient, however, and similar structural activity relationships, biologic functions, and cosmetic product usage suggest that the available data may be extrapolated to support the safety of the entire group. For example, while there is an absence of data on reproductive and developmental toxicity and limited tumorigenicity and metabolism data, the Panel reached consensus that these ingredients would not be absorbed through the skin and metabolized due to their high molecular weights, thereby obviating further concern over potential reproductive and developmental toxicity or carcinogenicity. The Panel also noted the lack of safety concerns regarding the potential presence of small molecule impurities or degradation products.
The potential adverse effects of inhaled aerosols depend on the specific chemical species, the concentration and the duration of the exposure and their site of deposition within the respiratory system. In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range, and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. Particles with an aerodynamic diameter of ≤10 µm are respirable. In addition to the negative acute inhalation toxicity data considered in this safety assessment, the Expert Panel determined that dimethiconol cysteine, dimethiconol methionine, and dimethiconol panthenol can be used safely in hair sprays, because the product particle size is not respirable.
Because some of the dimethiconol reaction products reviewed in this safety assessment contains a plant-derived moiety, the Expert Panel expressed concern regarding pesticide residues and heavy metals that may be present in these cosmetic ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
The Expert Panel noted that the hydrolyzed collagen moiety is a component of one of the dimethiconol polymers included in this safety assessment, hydrolyzed collagen PG-propyl dimethiconol. The Panel has expressed concern over the inherent danger of transmission of infectious agents with the use of human or animal derived cosmetic ingredients, eg, collagen. However, hydrolyzed collagen is derivatized collagen, and, therefore, does not pose a risk relative to the transmission of infectious diseases.
Conclusion
The CIR Expert Panel concluded that the ingredients listed below are safe as cosmetic ingredients in the present practices of use and concentration described in this safety assessment. Were ingredients in this group not in current use (as indicated by an *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group. End-Capped Homopolymers Dimethiconol Dimethiconol arginine Dimethiconol beeswax Dimethiconol behenate Dimethiconol borageate* Dimethiconol candelillate* Dimethiconol carnaubate* Dimethiconol cysteine Dimethiconol dhupa butterate* Dimethiconol hydroxystearate* Dimethiconol illipe butterate* Dimethiconol isostearate* Dimethiconol kokum butterate* Dimethiconol lactate* Dimethiconol meadowfoamate Dimethiconol methionine Dimethiconol mohwa butterate* Dimethiconol panthenol Dimethiconol sal butterate* Dimethiconol stearate Copolymers Hydrolyzed collagen PG-propyl dimethiconol* Dimethiconol/methylsilanol/silicate crosspolymer* Dimethiconol/silica crosspolymer* Dimethiconol/silsesquioxane copolymer Dimethiconol/stearyl methicone/phenyl trimethicone copolymer* Isopolyglyceryl-3 dimethiconol* Trimethylsiloxysilicate/dimethiconol crosspolymer Acrylates/dimethiconol acrylate copolymer
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L St, Suite 1200, Washington, DC 20036, USA.
Authors’ Contribution
Johnson contributed to conception and design; contributed to acquisition, analysis, and interpretation; and drafted manuscript. Andersen, Bergfeld, Belsito, Heldreth, Hill, Klaassen, Liebler, Marks, Shank, Slaga, and Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.