Abstract
Copper and copper compounds have multifunctional properties (antibacterial, antiviral, and antifungal) with promising applications. Copper in its nanoparticle (Cu NPs) forms has been widely used in various industrial and commercial applications. In the current research, the cytotoxic effects of textile fabrics impregnated with copper oxide nanoparticles (CuO NPs) were studied in mammalian cell lines. CuO NPs were impregnated onto textile substrates using 2 different techniques: the sonochemical generation and impregnation of NPs from metal complexes (insitu) and a “throwing the stones” technology using commercially prepared CuO NPs. The cytotoxicity of these 2 textile fabric types was assayed on human dermal fibroblast (HDF) cells and human hepatocellular carcinoma cells (HepG2) and was evaluated by indirect contact using an MTT assay. The impregnated fabrics were not exposed to the cells, rather their leachates were used to test cytotoxicity. The fabrics were soaked into the growth media for up to 7 days, and the leachates from day 1 and day 7 were incubated with the cell lines for 24 hours prior to the testing. The discharge or leaching from antimicrobial nanomaterials into the surroundings and surface waters is posing a serious environmental threat, which needs to be addressed. Hence, with regard to product safety, it is a good approach to study the fabric leachates rather than the intact material. The results showed that CuO NPs are not toxic to HDF cells. However, cytotoxicity was seen in HepG2 cells with cell viability decreasing by 20% to 25% for all the fabrics after 24 hours.
Keywords
Introduction
Textiles are an important factor to consider in an integrated infection control program of every health-care facility. 1 In terms of pathogen transmission, textiles play a crucial role in the chain of infection caused by pathogenic microorganisms such as bacteria, fungi, and viruses. 2 Textiles of all varieties are considered to be very proficient at carrying bacteria and serving as a reservoir for the transmission of infection. 1 However, the survival of pathogens on textile substrates varies considerably. To prevent the spread of infection, hospital textiles must be processed stringently under hygienic conditions and on a professional scale.
One way of combating nosocomial infection is to use antimicrobial textiles. Antimicrobial textiles could play an important role in maintaining hygienic standards in medical settings by breaking chains of infection. 3 Antimicrobial fabrics have been shown to be very effective against a wide variety of microorganisms and fungi and have the potential to prevent the spread of pathogens. 4
The antimicrobial activity of functional textiles depends on various factors such as nature of the antimicrobial agent used, durability, and the method of application used. The selection of appropriate application method is very important. Many antimicrobial agents in the textile industry use a controlled release mechanism, whereby the agent leaches out of the textile during use. 5 These agents should not be chemically bound to the textile substrate and should only be used as a coating or a finish to allow persistent release of the agent from the textile surface into the surrounding environment. Owing to the leaching of the agent into the surrounding environment, the concentration of the antimicrobial gradually decreases under the limit of effectiveness and thus could potentially be a cause of induced resistance in microorganisms. In addition, fabrics coated with leaching antimicrobials are less durable and do not withstand repeated washings. 6
The present research is based on the use of ultrasound for in situ generation and impregnation of metal oxide nanoparticles onto nylon, polyester, and cotton fabrics as a homogeneous layer. 7 –9 In the process, antibacterial metal oxide nanoparticles are simultaneously synthesized and impregnated on a textile surface through acoustic cavitation effects caused by ultrasound. This process results in a smooth and homogeneous layer of impregnation and is capable of projecting nanoparticles toward the fabric surface at a very high speed. This induces partial melting of the fibers and adhesion of the nanoparticles to the fabric surface. 7 The research was part of an EU FP 7 project to develop antibacterial and antifungal medical textiles based on a sonochemical process.
The first phase of our study focused on the production of these textile fabrics and their antibacterial efficacy testing against a selection of gram-negative and gram-positive bacteria. 7 –11 As the fabrics under development were intended for use in hospitals, the bacterial species chosen were representative of bacteria commonly associated with health-care-associated infections.
However, the cytotoxicity of these fabrics was yet to be investigated. The cytotoxicity test is 1 of the biological evaluation and screening tests that use tissue cells in vitro to observe the cell viability and morphological effects by medical devices. Because of global concern of nanoparticle (NP) toxicity, it is very important to test the impregnated fabrics for their cytotoxicity. The present research investigated the cytotoxicity of these fabrics with human cell lines. The fabrics impregnated with copper oxide nanoparticles (CuO NPs) were assessed for their toxicity in human dermal fibroblast (HDF) cells and human hepatocellular carcinoma cells (HepG2) using the microculture tetrazolium (MTT) assay for cell viability. The only contact of these fabrics would be the human skin (minimal contact). Therefore, it was very important to assess HDF cell lines for their cytotoxicity. Because of their small size, there is always a risk that NPs can enter the human body through various routes and may reach the most sensitive organs. Hence, HepG2 cells were used as a second cell line for cytotoxicity testing.
Various methods have been used to estimate NP cytotoxicity in mammalian cells and although there is no agreement on whether any single method is optimal, there is now a better understanding of their limitations based on the type of end point measurement and understanding of assay type. Some of the most common cytotoxicity assays are trypan blue exclusion assay, colony-forming efficiency (CFE) assay, lactate dehydrogenase (LDH) assay, and MTT assay. Choosing the right type of assay to assess the cytotoxicity of antimicrobial nanomaterials depends upon their applications and intended use. The textile fabrics produced for this project were intended to be used only as touch surfaces (laboratory coats, scrubs, uniforms, curtains, patient apparels, and bed linen) in order to combat the spread of infection in health-care environment. Human contact with these fabrics is going to be very minimal. This is why, a basic biocompatibility assay to test cell viability was thought to be a good indicator to evaluate the cytotoxicity of these fabrics. Hence, the MTT assay was used as a standard method to assess cell viability based on the mitochondrial activity, which is a well-known target for nanoparticle cytotoxicity. 12
Materials and Methods
Preparation of fabrics
The fabrics were prepared from 2 pilot scale machines built as part of the SONO project and were used to prepare samples for testing and for the manufacture of hospital gowns and linens.
Impregnated samples were prepared using the same protocol described in the previous articles as part of the research.
7,9
–11,13
In the reaction tank, copper acetate was dissolved in a 9:1 mixture of ethanol and water. The ultrasonic transducers were turned on. When the solution reached a temperature of 55°C ± 5°C (rise in temperature due to sonication), 25% of aqueous ammonia solution was added dropwise to adjust the pH to between 8 and 9. The ammonia forms a complex with the copper ions in solution. Rolls of cotton (10 × 0.1 m, plain unbleached woven cotton 145 g/cm2) were then fed at a constant speed (22 cm/min) through the reaction tank and ultrasonic field.
7
The impregnated fabrics were washed thoroughly with water to remove traces of ammonia and ethanol and dried under vacuum at room temperature. Two sets of fabrics were compared in the work presented here:
“Throwing the stones” (TTS) technology (0.91% w/w CuO)—In this technique, the reaction tank was filled with a suspension of preformed CuO NPs in water. Ultrasound was employed to impregnate these nanoparticles onto the fabrics but not in the sonochemical synthesis of them. The CuO nanopowder was purchased from a commercial supplier (product #: 29N-0801, average particle size 80 nm as per Holland Moran Ltd, Israel). Insitu technology (0.9% w/w CuO)—This is the sonochemical method of impregnation, where the nanoparticles were formed and simultaneously impregnated onto the fabrics in a one-step process. This process was carried out using the same procedure as the laboratory scale method of impregnation.
7
High-resolution scanning electron microscopy (HR-SEM) images of the fabrics (Figure 1) showed that the TTS and insitu impregnations were not only different colors but also very different at the micro-scale. The in situ impregnation appeared as a homogeneous layer of nanoparticles on the cotton fibers. The NPs were ∼40 nm in diameter. The TTS impregnation appeared to be significantly less homogeneous with large clumps of NPs rather than an even impregnation (∼1 µm).

Scanning electron microscopic images of the fabrics from the prototype machine (A) insitu fabric (B) TTS fabric. Images prepared at BIU (Israel). TTS indicates throwing the stones.
Leachate Preparation for Cytotoxicity Testing by Indirect Method
Fabrics impregnated with CuO NPs (both insitu and TTS from the pilot scale) were cut into round-shaped discs (∼3 cm diameter) in order to fit in the wells of a 6-well plate and sterilized by autoclaving. Control fabrics without any nanoparticles were used as a negative control, whereas a 30% v/v solution of dimethyl sulfoxide (DMSO) prepared in fresh culture medium was used as a toxicity positive control. Samples with no fabrics were also tested as a further negative control set.
The fabric samples were placed in contact with 3 mL of Dulbecco modified Eagle medium (DMEM) containing high glucose, glutamine supplemented with fetal bovine serum (FBS), and incubated at 37°C in a humidified atmosphere with 5% CO2 for 1 and 7 days. Following this, the fabric samples were removed from the growth media and the leachate medium solution was collected in universal tubes for use in the cytotoxicity studies.
Cell Culture
The HDF and HepG2 cells were obtained from the European collection of cell cultures (ECCC). The cells were maintained in DMEM containing 4.5 g/L glucose, 1% glutamine supplemented with 10% FBS, and 1% penicillin/streptomycin at 37°C in a humidified atmosphere with 5% CO2. The cells were subcultured every 2 days. The cells were washed with phosphate-buffered saline (PBS) and harvested using 0.25% trypsin. After trypsinization, counting, and appropriate dilutions, the cells were seeded at a density of 5.0 × 103 cells/well in a 96-well tissue culture–treated polystyrene plate (SARSTEDT). Cells were allowed to attach to the surface of the plate for 24 hours prior to contact with the leachate medium solution (growth media in which fabric samples impregnated with CuO NPs were soaked and incubated for 1 and 7 days) and incubated at 37°C (5% CO2).
For the exposure tests, the growth media from previously seeded cells in the 96-well plates were discarded after 24 hours of incubation. Leachate solution of 100 µL was then added to the 96-well plate and the plates were incubated at 37°C in a humidified atmosphere with 5% CO2 for 24 hours.
The MTT Assay
The MTT assay was used to measure the cytotoxicity of media exposed to CuO NPs to human cell lines. It is a colorimetric assay for measuring the cell viability. The cellular enzymes reduce the tetrazolium dye (MTT) to its insoluble formazan product giving a purple color. In brief, 5 × 103 cells/well were seeded in 96-well plates and exposed to the prepared leachate (Section 3.20.2) and incubated at 37°C in a humidified atmosphere of 5% CO2 for 24 hours. After 24 hours of exposure time, 100 µL of MTT solution was added to each well and incubated at 37°C in a humidified atmosphere of 5% CO2 for 3 hours until a purple colored formazan product developed. At the end of incubation, medium was removed from each well to avoid interference from the nanoparticles and 100 µL of DMSO was added to each well to lyse the cells. The plate was then left for 5 minutes before the absorbance was measured at 492 nm on a microplate reader (Anthos Reader 2001) using the Stingray program.
Statistics
Statistical analysis was carried out using the analysis tool pack provided with Microsoft Excel 2010. The observed cell viability following exposure to leachates from impregnated fabrics was compared with control fabrics using a single-factor analysis of variance (ANOVA) test. Statistical significance was established as P at an α level of .05. If the P value is less than .05, it rejects the null hypothesis and concludes that a significant different is present there. If the P value is greater than .05, it concludes that no significant difference exists.
Results and Discussion
Media leachates (day 1 and day 7) from the pilot scale CuO fabrics (both TTS and insitu) were exposed to HDF and HepG2 cells to determine their potential toxicity. The cytotoxicity was determined using an MTT assay. This provides a measure of cell viability as only live cells have the ability to reduce MTT solution into a purple formazan product.
Cytotoxicity to HDF Cells
As shown in Figure 2, the cultured fibroblast cells (HDF) remained metabolically active followed incubation in the media exposed to the CuO fabrics leachate. The viability decreased to 97.38% for CuO TTS and to 97.92% for CuO insitu fabrics. Even in the fabrics leachate media from 7 days exposure, the cell viability did not drop significantly. There was less than a 4% decrease in cell viability for both the fabrics compared to the control sample: 96.27% for CuO TTS and 97.36% for CuO in situ. Single-factor ANOVA test from day 1 exposure and day 7 exposure revealed no significant differences between the cell viability in CuO (TTS and insitu) suspensions compared to control suspension (P > .05). The P value from day 1 exposure test was .49, and the P value from day 7 exposure was .25. P values from both day 1 and day 7 test were greater than the α value of .05, which concluded no significant difference existed. The results show that indirect contact with the CuO NPs did not induce toxicity in dermal fibroblast cells (HDF).
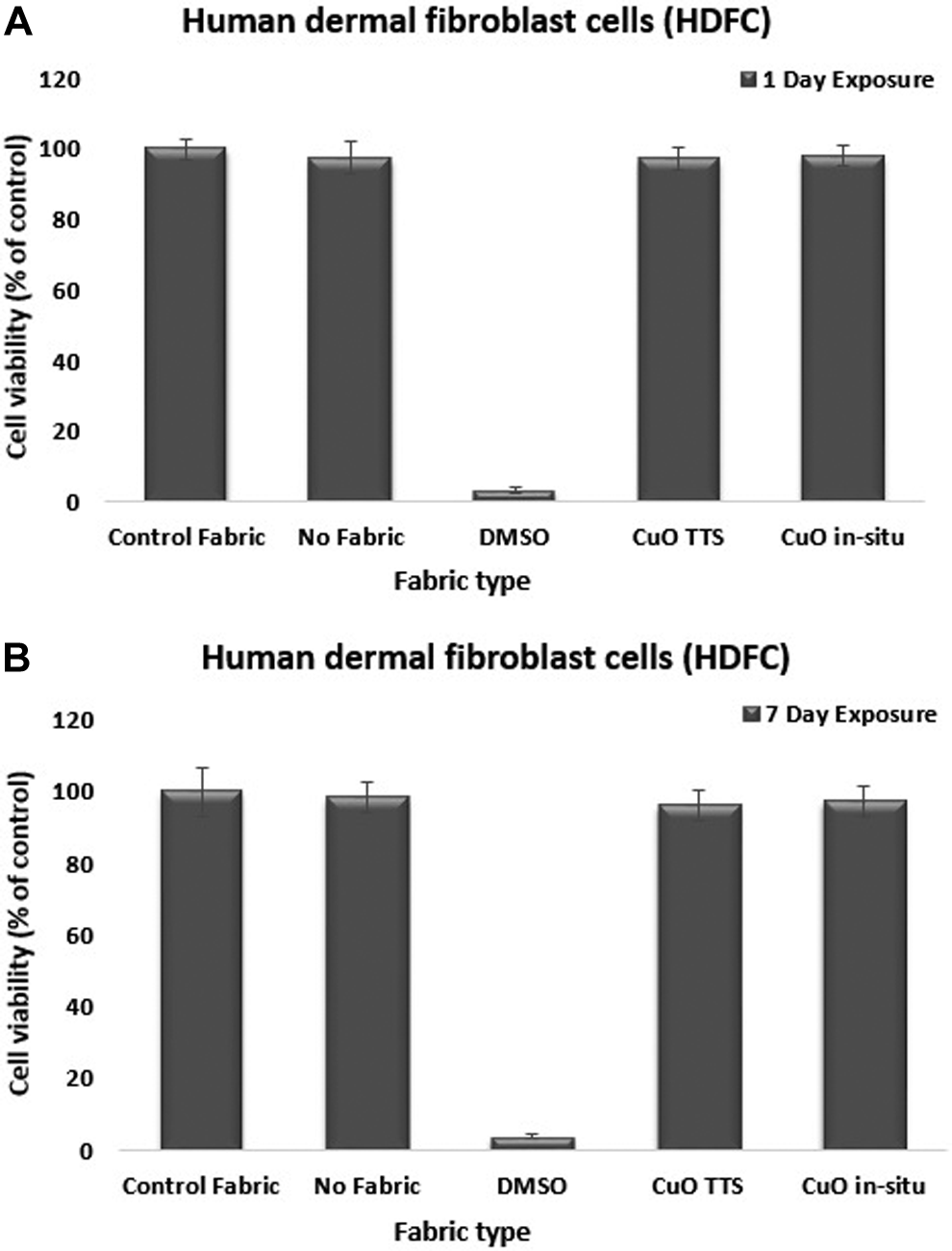
Human dermal fibroblast cells viability after (A) 1 day and (B) 7 days of exposure with the suspension from CuO impregnated fabrics. ANOVA showed that the mean counts between control and test samples are not statistically different (P > .05). Mean (SD), n = 3. ANOVA indicates analysis of variance; SD, standard deviation; CuO, copper oxide.
A comprehensive understanding of toxic effect of CuO NPs was necessary to safely expand their use. Factors such as size surface characteristics, dissolution properties, and exposure routes mediate the toxic effects of CuO NPs. In our experiments, the cytotoxicity was determined by indirect method because the reactivity of these fabrics is mainly due to the dissolution of CuO NPs at ambient conditions, which changes its properties depending on prevailing environmental conditions. Their dissolution involves both ions and particulate species that induce toxic effects by triggering reactive oxygen species (ROS) production and DNA damage. 14 Other researchers have also claimed that the dissolution of metal ions is a key factor in the toxicity of nanoparticles. Chang et al showed that the toxicity of CuO NPs was largely influenced by soluble Cu ions. 14 Brunner et al also found that the toxic effect of soluble metal oxide NPs was higher than the insoluble ones, when used at the same concentration. 15 Sabella et al in their experiments with chelating and lysosomotropic agents found that the toxicity mechanism of metallic NPs is mainly due to the release of the corresponding toxic ions. They also showed that that particles unable to release toxic ions (such as stably coated NPs or diamond and silica NPs) are not harmful to intracellular environments. 16 This is a rationale for testing NP leachates from the fabrics by indirect method rather than the intact material. There has been considerable debate that testing NP leachates should be considered while assessing nanocytotoxicity and results like these add value to such debates. Other researchers have reported that the toxic action of metal and metal oxide NPs is mainly due to the dissolution of cytotoxic metal ions and not from the particles themselves, and our results confirm those claims as well. 17 –19 Such results might indicate that metal oxide NPs dissolubility is an important factor in their toxicity. Solubilized Cu2+ ions proved to contribute substantially to the cytotoxicity of CuO NPs. Our results showed that CuO NPs from fabric surfaces into saline gets dissolved more readily and produce more copper ions. In a further experiment, saline leachates from the CuO NPs impregnated fabrics were centrifuged to separate solid CuO from dissolved copper. Inductively coupled plasma–optical emission spectrometer (ICP-OES) analysis showed that the TTS and in situ CuO fabrics behaved differently in terms of the amount of dissolved copper and the amount of solid CuO released into the saline solution. In the case of the TTS fabrics, most of the copper in the leachates was pelleted and thus most likely in the form of CuO NPs. In the case of the in situ fabrics, most of the copper in the leachates was found dissolved in the supernatant fraction. This showed that the leachates from the in situ fabrics contained a higher proportion of dissolved copper as compared to the CuO NPs.
These fabrics are intended for use as bed sheets, laboratory coats, patient gowns, and linens in health-care settings. The primary contact with fabrics would be skin only. As the NPs on the fabric surface are sonochemically impregnated (bound antibacterial), direct contact between nanoparticles and skin cells would be minimal. It would be the bodily fluids (blood, urine, sweat, and so on) from the skin which would dissolve the NPs into metal complexes or ions. Hence, indirect method of measuring cytotoxicity was preferred over direct contact method.
The results have shown that the fabrics did not induce any toxicity to skin cells. Similar results have been reviewed by Tamaekong et al and Hostynek and Maibach as well. 20,21 Based on the predictive guinea pig maximization test (GPMT) reported by Magnusson and Kligman and the local lymph node assay (LLNA) by Kimber, the risk of dermal sensitivity to CuO is minimal. 22,23 Sweigert et al also reported that copper (II) complexes did not induce any toxicity in human dermal skin fibroblast cells and melanoma cells. 24 Cohen et al used light and electron microscopy and other biochemical testes to address the biological effects of topically applied CuO NPs and micro-sized particles (15 to 100 nm diameter) to human skin organ cultures. 25 They reported that nanoparticles were more toxic than micro-sized particles with stronger effects. However, they did not see any penetration of NPs into the skin and proposed they might have adhered to skin surface. In case of HDF cells in vitro, our results did not show any analogous effect of fabrics impregnated with CuO NPs.
Cytotoxicity to HepG2 Cells
Figure 3 shows the cytotoxicity of CuO fabrics to HepG2 cells. The cultured HepG2 cells showed some evidence of cytotoxicity from the CuO fabrics. The cell viability decreased to 80.7% and 75.7% after 24 hours, when exposed to the media leachates from the CuO TTS and CuO insitu fabrics, respectively. After 7 days of exposure, the cell viability further decreased to 78% (CuO TTS) and 72% (CuO in situ). Single-factor ANOVA test from day 1 exposure and day 7 exposure showed significant differences between the cell viability in CuO (TTS and insitu) suspensions compared to control suspension (**P < .01). All differences were concluded significant at P < .05. The P value from both day 1 and day 7 exposure test was **P < .01. The P values for both the tests were less than the α value of .05, which rejected the null hypothesis and concluded that significant differences existed between each set on day 1 and between the controls and test samples at day 7 (for Day 1 = overall P < .01; between controls P = .023 and <.01 for everything else, for Day 7 = between controls P = .152; between tests = .064; and between controls and tests P < .01). These results suggested that the HepG2 cells were sensitive to CuO.
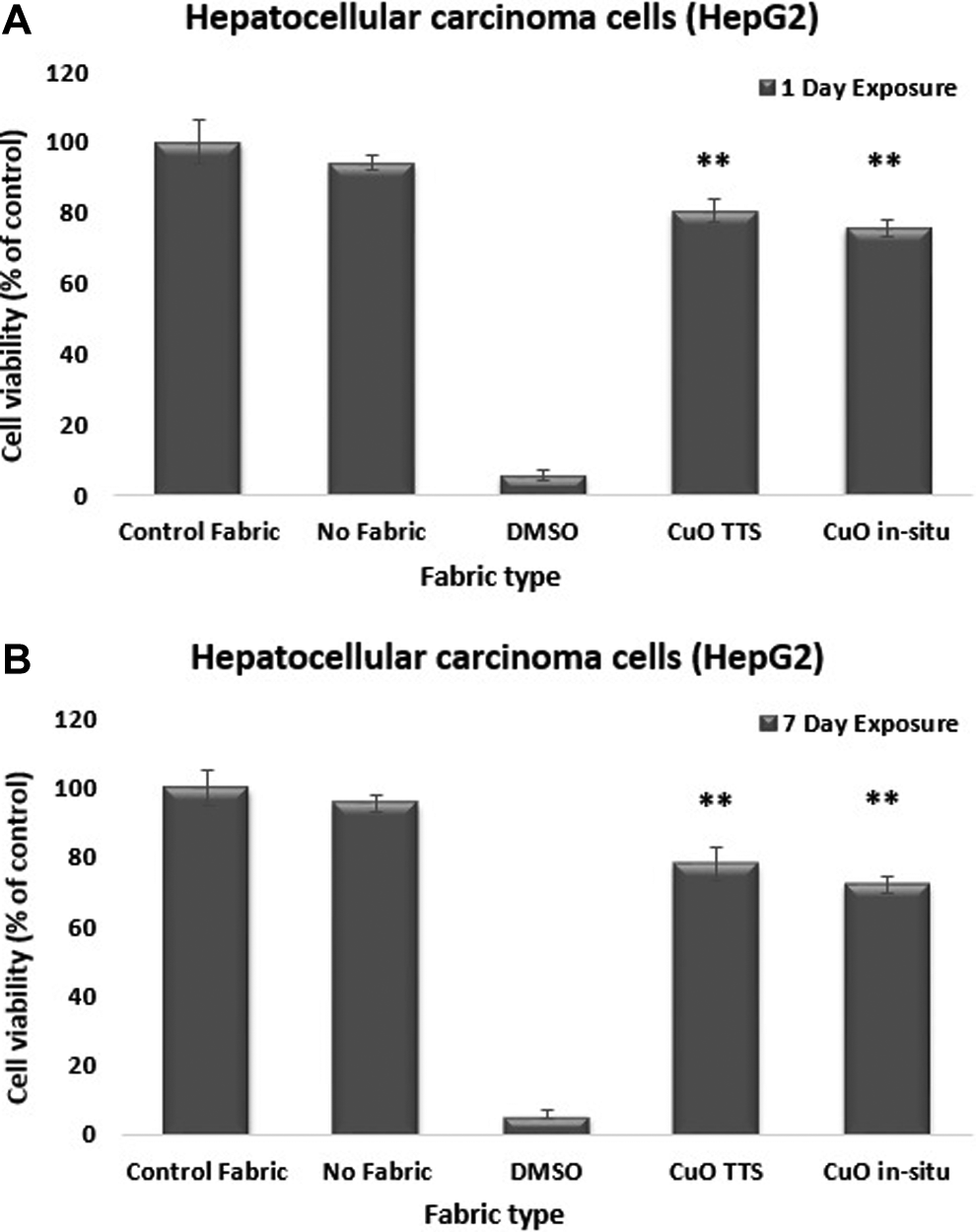
Human hepatocellular carcinoma cells (HepG2) viability after (A) 1 day and (B) 7 days of exposure with the suspension from CuO impregnated fabrics. Statistically significant differences existed in the mean counts between control and test samples postincubation (**P < .01). Mean (SD), n = 3. SD indicates standard deviation.
Wang et al in their study examined the cytotoxicity of zinc oxide (ZnO), copper oxide (CuO), titanium dioxide (TiO2), and cobalt (II, III) oxide (Co3O4) NPs to channel catfish hepatocytes and human HepG2 cells. 26 They reported that the HepG2 cells were very sensitive. The results showed that 25 mg/L of CuO and ZnO NPs induced toxicity in HepG2 cells. The cell viability with both ZnO and CuO NPs was reduced to 85% after 48 hours of exposure. Qiao et al in their studies treated human cervical carcinoma cells (HeLa) with increasing concentrations of the Cu(II) complex for 48 hours and measured the viable cells using MTT assay. 27 Fluorescence microscopic images showed that the Cu(II) complex induced apoptosis of HeLa cells mediated by intrinsic mitochondrial apoptotic pathway. In an another study, Piret et al investigated the toxic effects of copper(II) oxide NPs and CuO NPs with different specific surface areas, different shapes, and different sizes but with similar hydrodynamic diameter in suspension on HepG2 cells. 28 They found that both the NPs type induced cellular toxicity with the generation of ROS. They also showed that the Cu2+ ions released from the CuO NPs into the cell culture medium played a very small part in the toxicity by these NPs on HepG2 cells. This can be related to our results where NPs are dissolving into metal complexes or ions due to which only a little toxicity was observed from our tests.
Even though CuO fabrics induced toxicity in HepG2 cells, they may still be a safer alternative to silver. A study by Faedmaleki et al examined the toxicity of silver NPs (AgNPs) in mice primary liver cells and in HepG2 cells. 29 The HepG2 cells were exposed to 1, 2, 3, 4, 5, 7.5, and 10 ppm of AgNPs, and mice primary liver cells were exposed to 1, 10, 50, 100, 150, 200, and 400 ppm of AgNPs for 24 hours. The results showed a higher toxicity of silver NPs to both mice liver cells and HepG2 cells. Viability percentage measured by MTT assay showed a dose-dependent decrease in viability of both HepG2 cells and primary liver cells. The cell viability dropped to less than 50% with 3 ppm exposure and by 15% with 5 ppm Ag NPs exposure.
CuO NPs induced cytotoxicity in numerous cell types, which is often related to apoptosis, genotoxicity, and oxidative stress. 30 –32 The toxicity lies within the delivery and the subsequent intracellular dissolution. 33 With the method used for this research, it was not clear whether the toxicity to HepG2 cells was due to the CuO NPs being taken up by the cells or due to the dissolution of NPs as Cu ions. However, further testing could determine whether the NPs are locating into the cellular target areas or getting dissolved as free metal ions. There are a lot of analytical methods to characterize CuO NPs. 34–35 Boyles et al used dynamic light scattering (DLS) and zeta potential measurements (Malvern Zetasizer Nano ZSP) to determine hydrodynamic diameter and an estimation of surface charge of CuO NPs, respectively. They observed a substantial agglomeration in DLS measurements. The dissolution of CuO NPs was assessed by coupled plasma–atomic emission spectroscopy using an Activa instrument (Horiba Jobin Yvan). 34 Chusuei et al investigated the physiochemical properties that govern the cytotoxicity of CuO, ZnO, NiO, TiO2 nanoparticles in human bronchial epithelial cells (BEAS-2B) and human broncho alveolar carcinoma–derived cells (A549). 35 They determined the dissolution kinetics of metals from metal oxide NPs in the cell culture medium by ICP mass spectrometry and graphite furnace atomic absorption (GFAA) analysis over a period of 24 hours. The results showed that the addition of NPs to the cell culture medium did not significantly alter the pH. The results also indicated that cytotoxicity is a function of metal ion dissolution from the NPs. The researchers have claimed that the release of Cu2+ from their respective oxides is most likely to contribute to their toxicity. 35
However, as the SONO fabrics are only intended for use as wearable fabrics or for touch surfaces, they would only come in contact with the skin. Vandebriel and De Jong reported that NPs do not efficiently cross the skin. 36 The results presented here with the skin fibroblast cells show that the CuO fabrics are not toxic to skin cells.
Conclusion
Cytotoxicity studies have demonstrated that the CuO fabrics are not toxic to (>95% cell viability for all fabrics) HDF cells. However, some cytotoxicity was observed in hepatocellular carcinoma cells (HepG2). Compared to other antibacterial agents, CuO has shown very little cytotoxicity to HepG2 cells. 37 Studies have shown that the NPs do not translocate though the skin barrier. 36 These fabrics are not intended for use as wound dressings but for use as bed sheets, curtains, and laboratory coats to reduce the spread of microbial contamination. Hence, the risk of NP-mediated damage to human cells should be minimal and the fabrics could be used safely in health-care settings to reduce cross contamination.
Developments in antimicrobial fabrics clearly hold promise in medical applications in the near future. However, their impact on the human health and environment needs to be well researched and established before they can gain commercial acceptance and wide spread use.
Project website: http://www.fp7-sono.eu
Footnotes
Acknowledgments
The authors are grateful for the financial support through the EU FP7 programme for providing the funding for this project (SONO, FP7 project number: 228730) and the Faculty of Health and Life Sciences at Coventry University for the additional support.
Author Contributions
G. Singh contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. J. Beddow contributed to conception and critically revised manuscript; Mee, C. contributed to acquisition and critically revised manuscript. L. Maryniak contributed to design and critically revised manuscript. M. Joyce contributed to design and critically revised manuscript. T. Mason contributed to design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The EU FP7 programme (SONO, FP7 project number: 228730).
