Abstract
Archaeon
Introduction
For the past few decades, lipid bilayer vesicles (liposomes) have been extensively studied for drug delivery. The ability to incorporate hydrophilic, lipophilic and amphiphilic compounds into liposomes (to aqueous core or lipid bilayer) led to the development of drug, vaccine and gene delivery systems, protecting encapsulated drug from physiological media, better biodistribution of drugs, site- and time-specific unloading of cargo in therapeutic concentrations at the site of action and lowering toxic side effects of active compounds. Liposomes that are prepared from natural phospholipids are biologically inert and cause little or no antigenic, pyrogenic, allergic and toxic reactions. 5,6
A major disadvantage of liposomes as drug carriers however is their instability in a gastrointestinal tract and circulatory system; they release their cargo before reaching the target tissue or become prone to reticuloendothelial system elimination, mostly in liver and spleen. 7,8 Different approaches, such as adding poly(ethylene glycol) to liposomes to mask the surface, have been used to overcome this problem. 9 The discovery of new archaeal lipids that form liposomes that are stable in high temperature, low or high pH, resist even in the presence of phospholipases, bile salts and serum media 10 –14 and even in mixtures with conventional diester lipids 15 has led to the development of new potential drug, gene and vaccine delivery systems. Some Archaea species possess unusual bipolar tetraether lipids that bear two hydrophilic head groups located at both ends of a hydrophobic domain that stabilize and rigidify membranes of methanogens, thermophiles and psychrophyles. 16,17 Archaeosomes are also strong adjuvants for the induction of Th1, Th2 and CD8+ T cell responses to the entrapped soluble antigens and are therefore promising self-adjuvanting delivery vehicles for vaccines against intracellular infections and cancers. 18
Archaeon
In the scope of a possible use of
Endocytotic uptake of
Methods
Archaeosomes
Cells of
Cell cultures
All cells were obtained from European collection of Cell Cultures (Salisbury, UK) except EA.hy926 that were purchased from American Type Culture Collection (Manassas, Virginia, USA). They were grown as an attached monolayer culture at 37°C, 5% carbon dioxide (CO2) atmosphere in a humidified chamber.
B16-F1 mouse melanoma cells were grown in Eagle’s minimum essential medium (EMEM) with
Chinese hamster ovary (CHO) cells were grown in Ham’s nutrient mixture F-12 (PAA Laboratories, Pasching, Austria), supplemented with 2 mM
Three human cell lines were also used for studies: epithelial colorectal adenocarcinoma cells (CACO-2), human liver hepatocellular carcinoma cell line (Hep G2) and human endothelial umbilical vein cell line (EA.hy926). EA.hy926 cell line was obtained by the hybridization of primary human umbilical vein endothelial cells with the A549/8 human lung carcinoma cell line but has maintained the phenotype of endothelial cells.
29
CACO-2, Hep G2 and EA.hy926 cells were grown as described before.
30
Briefly, for CACO-2, we used EMEM (Sigma–Aldrich) supplemented with 10% FBS, 1% penicillin/streptomycin, 1%
Cytotoxicity assay
Cytotoxicity was evaluated by the colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay 31 (cell growth determination kit (CGD1), MTT based, Sigma–Aldrich), as described by the manufacturer. In brief, cells were seeded to 96-well plates (2000–10,000 cells per well) and allowed 2 h to attach to the surface. Different concentrations of archaeosomes were added to cells (5000–0.000256 µg/ml). After 4 days of incubation at 37°C in a CO2 incubator, the MTT reagent was added to cells and after 4 h purple formazan was extracted from cells by dimethyl sulfoxide and measured spectrophotometrically at 544 nm using a microplate reader (Tecan Infinite M200, Tecan Group Ltd, Männerdorf, Switzerland), and using 690 nm wavelength as a reference.
Statistical analysis was performed using Microsoft Excel (Microsoft Corp., Redmond, Washington, USA). Results from at least three independent experiments were averaged and presented as mean ± SEM. One-way analysis of variance was used to determine statistical differences between control and treated cells and
Cell labeling and confocal fluorescence microscopy
Cells were seeded to 8-well µ-slides (Ibidi GmbH, Martinsried, Germany), 10,000 cells per well (B16-F1 and CHO), or on round cover glass slides placed in 24-well plates, 20,000 cells per well (CACO-2, Hep G2 and EA.hy926). Cells were then washed and stained by membrane label Vybrant DiI (Vybrant® DiI cell-labeling solution, Molecular Probes/Invitrogen/Life Technologies Corp., Grand Island, New York, USA), following a manufacturer’s protocols (DiI diluted 1:200 in phosphate buffered saline (PBS) for 30 min at 37°C). Cells were then washed three times and used for incubation with archaeosomes. Calcein-encapsulated archaeosomes were added to stained cells at a concentration of 100 µg/1 ml of culture medium and incubated for 10 and 30 min, 1, 2 and 24 h at 37°C in a CO2 incubator. Cells were then washed three times with PBS, fixed with 4% paraformaldehyde and mounted on a glass slide.
B16-F1 and CHO cells were studied by a confocal laser scanning fluorescence microscope (CLSM) Leica TCS SP5 (Leica Microsystems GmbH, Wetzlar, Germany) using 63× water immersion objective and
Results
Cytotoxicity
Cytotoxicity of archaeosomes was tested on five cell lines (Figures 1 to 3): rodent mouse melanoma cells (B16-F1) and CHO cells (Figure 1), as well as three human cell lines—epithelial colorectal adenocarcinoma cells (CACO-2), liver hepatocellular carcinoma cell line (Hep G2; Figure 2) and endothelial umbilical vein cell line (EA.hy926; Figure 3). The archaeosomes were moderately toxic to both rodent cell lines (median lethal dose (LC50) for B16-F1 and CHO cells is around 625 µg/ml) but showed no toxicity to human CACO-2 and Hep G2 cells. However, archaeosomes exhibited strong toxic effect on Ea.hy926 cell line (LC50 around 0.8 µg/ml).
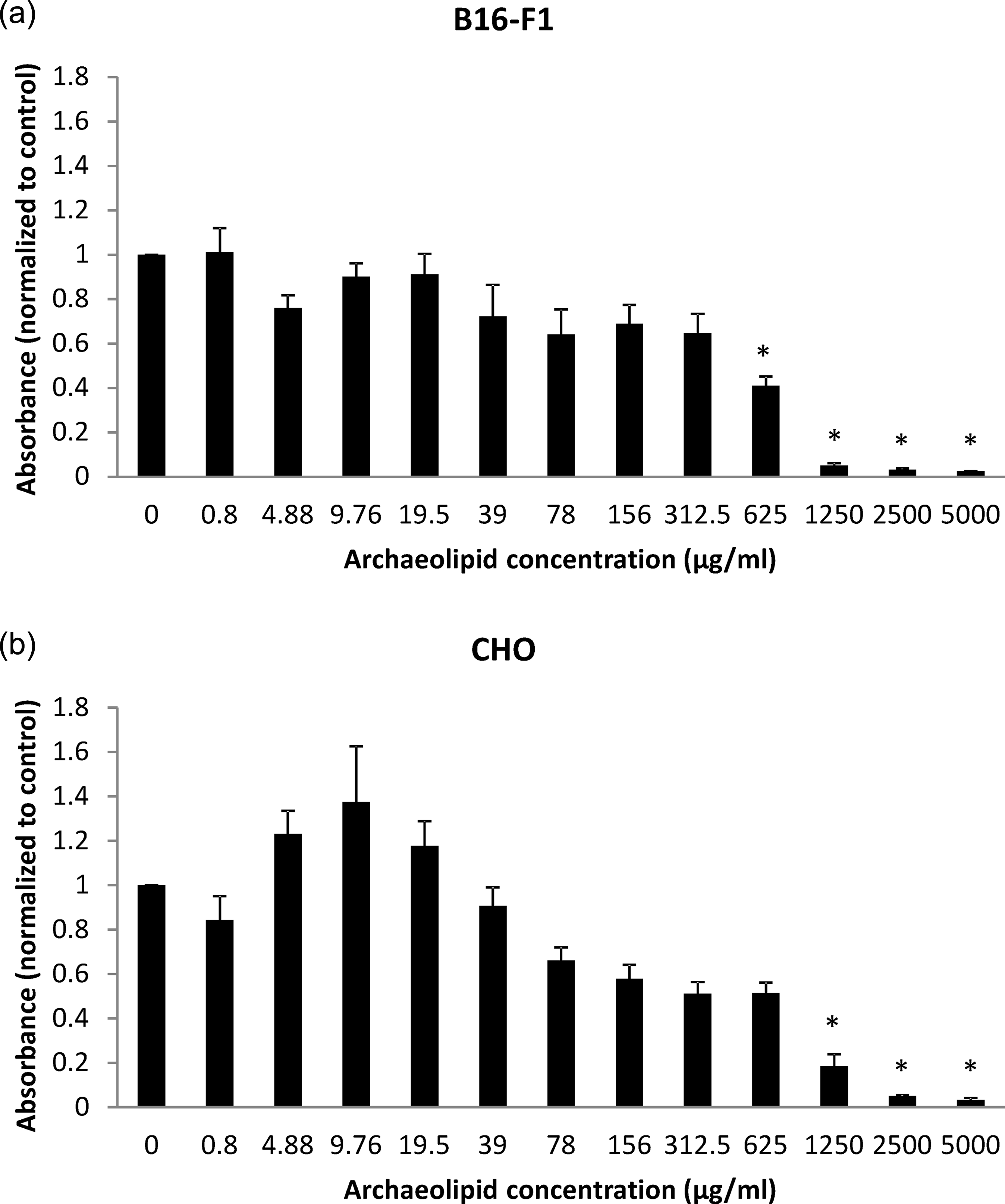
A dose-dependent cytotoxic effect of archaeosomes on rodent cell lines: B16-F1 (a) and CHO (b), measured by MTT cytotoxicity assay. Data are presented as mean ± SEM of at least three independent experiments. Significant differences from controls are designated by asterisks (*

A dose-dependent cytotoxic effect of archaeosomes on human cell lines: CACO-2 (a) and Hep G2 (b), measured by MTT cytotoxicity assay. Data are presented as mean ± SEM of four independent experiments. Significant differences from controls are designated by asterisks (*

A dose-dependent cytotoxic effect of archaeosomes on human umbilical vein cell line EA.hy926 measured by MTT cytotoxicity assay. Data are presented as mean ± SEM of four independent experiments. Significant differences from controls are designated by asterisks (*
Confocal microscopy of archaeosome uptake
The archaeosomes were added to B16-F1, CHO, CACO-2, Hep G2 and EA.hy926 cells. After different incubation times, the cells were fixed and observed under a CLSM (Figure 4). We can see archaeosomes as green dots in the images. The archaeosomes interact with cells in three distinct phases (Table 1). First, they adsorb onto the cell surface ((1) e.g. as in Figure 4, CACO-2, 10 min). Then, they are transferred to the cell interior ((2) e.g. as in Figure 4, CACO-2, 2 h), and finally, they release their load (in our case calcein) into the cytoplasm ((3) e.g. as in Figure 4, EA.hy926, 24 h). Time course and intensity of the uptake of archaeosomes and release of calcein are however different in different cell types (Table 1). In B16-F1, CHO, CACO-2 and Hep G2 cells, the release of calcein into the cytoplasm is observed after 24 h of incubation with archaeosomes, while in EA.hy926 cells this phenomenon can be seen already after 30 min when intensive green coloring of the cytoplasm occurs. In the case of B16-F1, CHO, CACO-2 and Hep G2 cells, intact archaeosomes inside cells persist even after 24 h (Figure 4, CACO-2, 24 h). B16-F1 cells accumulate the least amount of archaeosomes.

Laser scanning confocal microscope images of cell lines CACO-2 and EA.hy926 incubated with archaeosomes loaded with calcein (green). Cells are labeled with Vybrant® DiI cell-labeling solution for membranes (red) prior incubation with archaeosomes. Cell types are shown on left side and incubation times of cells with archaeosomes (10 min, 2 and 24 h) are shown on top. Longitudinal sections are shown.
Interactions between archaeosomes from
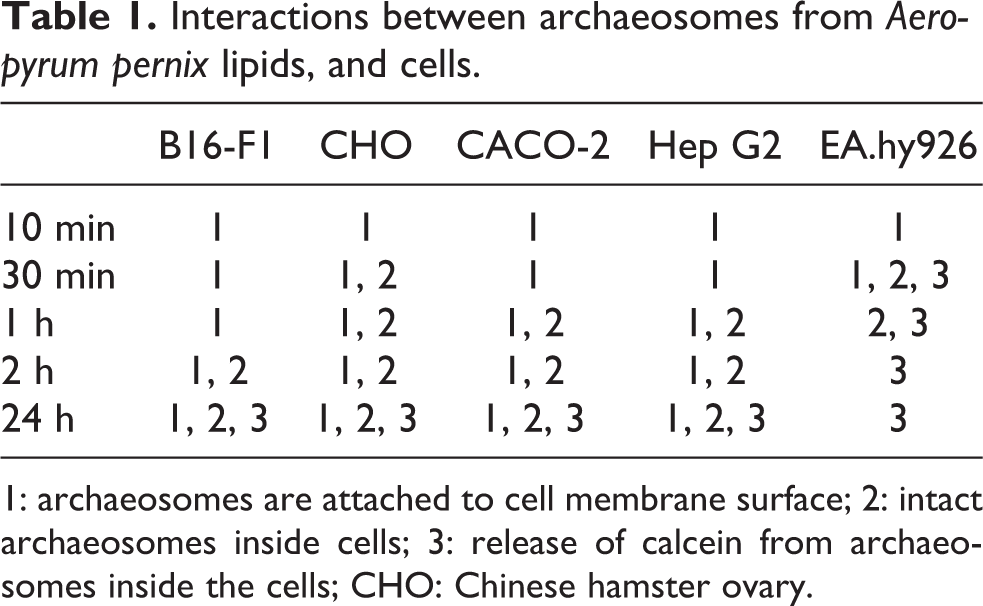
1: archaeosomes are attached to cell membrane surface; 2: intact archaeosomes inside cells; 3: release of calcein from archaeosomes inside the cells; CHO: Chinese hamster ovary.
Discussion
For a possible use of
Although most of the liposomes are inert and lack toxicity toward cells, toxicity studies of liposomes on various cell lines have been reported in the literature. Filion and Phillips 32 found that liposomes composed of cationic lipids and fusogenic dioleoylphosphatidylethanolmine are highly toxic in vitro toward macrophages. This was in line with the observation that higher amounts of lipids were associated with the cells of macrophage origin than the hepatocytes. 33 On the other hand, fibroblasts were sensitive toward two different phosphatidylcholine liposomes. 34
Our results show that archaeosomes composed of lipids from archaeon
Numerous in vitro studies have already revealed that liposomes can be internalized by cells with endocytosis. 35 –37 After binding to the cell surface, the liposomes are internalized by phagocytosis in phagocytic cells, and in nonphagocytic cells the main endocytotic pathway is clathrin-mediated or caveolae-mediated endocytosis, although liposomes can enter the cells also by macropinocytosis or other pathways (reviewed in the literature 38 ). Charged liposomes (either positively or negatively charged) are endocytosed faster and to a greater extent than neutral liposomes by endocytotic cells, although different cells have different liposome-binding sites on their surface. 39,40
Phagocytic uptake of liposomes prepared from ether lipids extracted from various archaeal species was several times greater than the uptake of liposomes composed of conventional lipids (dipalmitoylphosphatidylcholine, dimyristoylphosphatidylcholine: dimyristoylphosphatidylglycerol:cholesterol). It has also been shown that interactions of archaeosomes with the nonphagocytic cells were significantly lower than in macrophages. 24 Results of Sprott and his colleagues suggest endocytosis of archaeosomes, rich in surface-exposed phosphoserine head groups via a phosphatidylserine receptor. Archaeosomes composed of certain other archaeal lipids (lacking phosphoserine head groups) are suggested to enter the cells after energy-independent surface adsorption. 41,42
In EA.hy926 cells, we can see calcein released in the cytoplasm shortly after adding archaeosomes loaded with calcein to the medium. A fast release of large amounts of calcein from archaeosomes can be related to higher rate of archaeosome uptake and release in these cells due to fast endocytosis. This is in line with high cytotoxicity of archaeosomes of
Archaeosomes from
To conclude, our in vitro study shows that archaeosomes composed of lipids from archaeon
Footnotes
Acknowledgments
The authors would like to thank Dr Mojca Benčina for her invaluable help with confocal microscopy.
Funding
The study was supported by the Slovenian Research Agency (ARRS; Grant numbers J2-3639, P2-0249, P4-0121 and MR-1000-06-310166). Research was conducted in the scope of the EBAM European Associated Laboratory (LEA).
