Abstract
Benzyl alcohol, benzoic acid and its salts, and benzyl benzoate function mostly as fragrance ingredients/preservatives in cosmetic products. The Cosmetic Ingredient Review Expert Panel previously established concentration limits for benzyl alcohol, benzoic acid, and sodium benzoate in cosmetics and determined that the available data were insufficient to support the safety of these ingredients during inhalation exposure. After reviewing newly available data, it was concluded that benzyl alcohol, benzoic acid and its salts, and benzyl benzoate are safe in the present practices of use and concentration described in this safety assessment.
Introduction
Benzyl alcohol, benzoic acid and its salts (ie, sodium, calcium, magnesium, and potassium benzoate), and benzyl benzoate are reported to function as fragrance ingredients, pesticides, pH adjusters, preservatives, solvents, and/or viscosity decreasing agents in cosmetic products. 1
In 2001, the Cosmetic Ingredient Review (CIR) published a final report on the safety assessment of benzyl alcohol, benzoic acid, and sodium benzoate with the conclusion that benzyl alcohol, benzoic acid, and sodium benzoate are safe for use in cosmetic formulations at concentrations up to 5%. 2 Benzyl alcohol was safe for use in hair dyes at concentrations up to 10%. The available data were insufficient, however, to support the safety of these ingredients in cosmetic products in which a primary route of exposure is inhalation. The Expert Panel (Panel) stated that inhalation toxicity data were needed.
Additional new data reviewed included an Organization for Economic Cooperation and Development (OECD) Screening Information Data Sets (SIDS) for high-volume chemicals’ initial assessment report on benzoic acid, sodium benzoate, potassium benzoate, and benzyl alcohol published by the United Nations Environmental Program (UNEP) Chemicals Branch that was published in 2001, 3 the same year that the CIR Final Safety Assessment on benzyl alcohol, benzoic acid, and sodium benzoate was published. In addition to the SIDS data summaries included in this CIR safety assessment, the 2001 SIDS report may be consulted for further details. Further, the European Commissions’ International Uniform Chemical Information Database (IUCLID) on benzyl alcohol, benzoic acid, sodium benzoate, and benzyl benzoate was created in the year 2000. 4 Data from the OECD SIDS assessment and IUCLID data sets are summarized in the toxicology section. In the process of selecting new data for inclusion in the current report, efforts were made to eliminate redundancy between OECD SIDS, IUCLID, or other data found in the published literature and data included in the original final report.
Chemistry
Definition and Structure
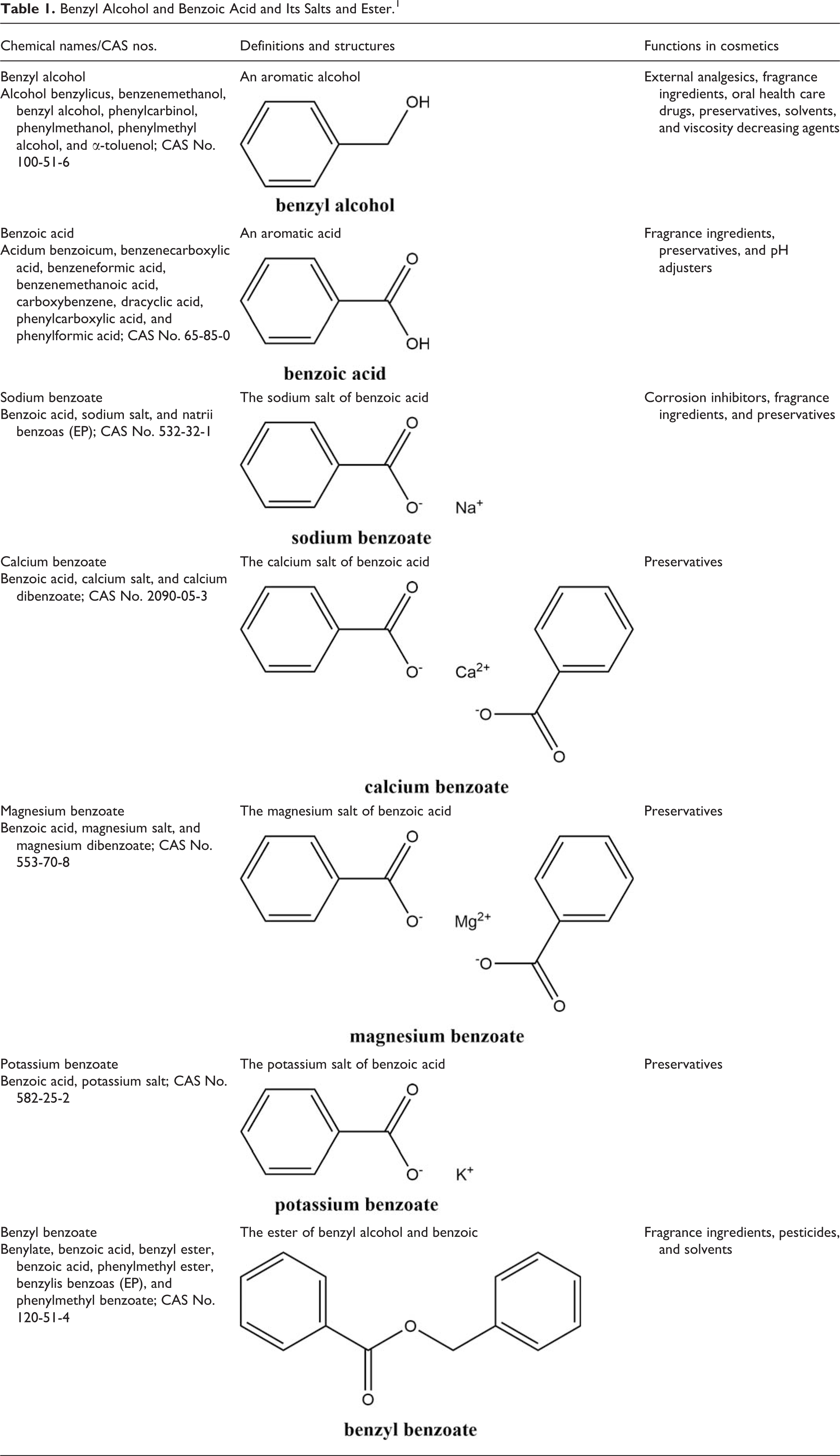
Definitions, other chemical names, cosmetic ingredient functions, and chemical structures for the ingredients reviewed in this safety assessment are listed in Table 1.
Benzyl Alcohol and Benzoic Acid and Its Salts and Ester. 1
Chemical and Physical Properties
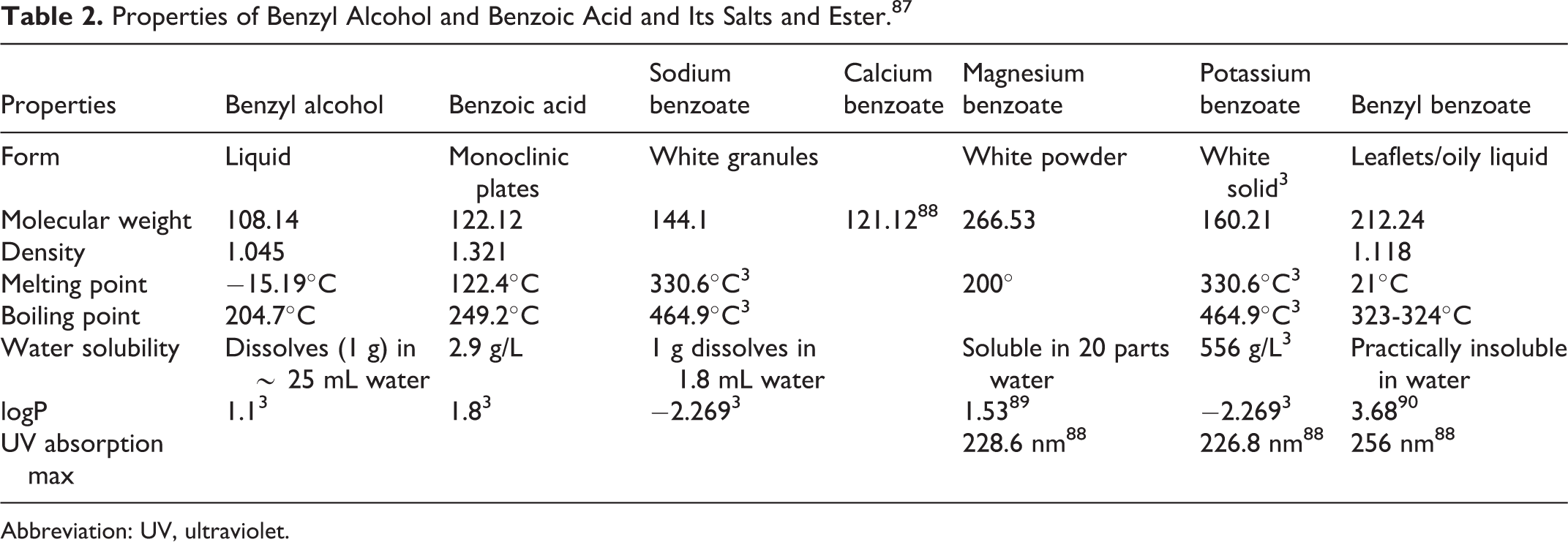
Chemical and physical properties of benzyl alcohol, benzoic acid and its salts, and the benzyl benzoate ester are listed in Table 2.
Properties of Benzyl Alcohol and Benzoic Acid and Its Salts and Ester. 87
Abbreviation: UV, ultraviolet.
The photodegradation of benzoic acid aqueous solution in the presence of ultraviolet (UV) light has been reported. Benzoic acid absorbs UV light below 300 nm, and its concentration in solution was monitored using reverse-phase, high-performance liquid chromatography. The mercury lamp used emitted polychromatic radiation between 240 and 540 nm. The photon flow absorbed by the reaction system varied with the concentration of benzoic acid and with the presence of the products of degradation. When the temperature was increased from 25°C to 50°C, photodegradation was increased by 20%. Photodegradation products were not mentioned. 5
The conversion of benzyl alcohol to Benzaldehyde and benzoic acid in aqueous solution has been demonstrated using liquid chromatography/spectrophotometry. 6 When benzyl benzoate (in ethanol) was exposed to sunlight for 11 days (at 25°C), 35 products were isolated from exposed samples and identified using gas chromatography/mass spectrometry. Benzyl alcohol and benzoic acid were among the products identified. 7
Method of Manufacture
Benzyl alcohol
Large scale production of benzyl alcohol is achieved by the action of sodium or potassium carbonate on benzyl chloride. 2
Benzoic acid
Benzoic acid is produced via the decarboxylation of phthalic anhydride in the presence of catalysts. Another production method involves the chlorination of toluene to yield Benzotrichloride, which is hydrolyzed to benzoic acid. 8
Sodium benzoate
Sodium benzoate is produced by the neutralization of benzoic acid with sodium bicarbonate, sodium carbonate, or sodium hydroxide. 9
Potassium benzoate
Potassium benzoate can be prepared by reacting methyl benzoate with potassium thioacetate. 10
Benzyl benzoate
Benzyl benzoate results from the condensation of benzoic acid and benzyl alcohol. It can also be generated from Benzaldehyde via the Tishchenko reaction. 11
Ultraviolet Absorption
The following UV absorption maxima have been reported for salts and an ester of benzoic acid: 228.6 nm (magnesium benzoate), 226.8 nm (potassium benzoate), and 256 nm (benzyl benzoate). 12,13
Analytical Methods
Benzoic acid has been analyzed using mass spectrometry, 14 and potassium benzoate has been analyzed using infrared (IR) spectroscopy. 15 Benzyl benzoate has been analyzed using IR and nuclear magnetic resonance spectroscopy. 16,17
Impurities
Benzoic acid and sodium benzoate
In foods, benzoic acid and benzoate salts have a demonstrated potential for release of benzene, including in the presence of long wave ultraviolet light (UVA). Benzene formation was examined for samples contained in UV stabilized and non-UV stabilized packaging. 18 Some of the samples selected for UVA testing included model solutions prepared with 0.04% benzoate and 0.025% ascorbic acid in unbuffered water. The results of 24-hour irradiation studies indicated that, under intense UV light, benzene levels increased by as much as 53% (to 315 ng/g benzene, compared to model solutions exposed to visible light [206 ng/g benzene]) in model solutions stored in non-UV stabilized bottles. However, the use of UV stabilized polyethylene terephthalate bottles reduced benzene formation by ∼13%, relative to the non-UV stabilized bottles. Similar trends were observed following irradiation for 7 days. According to the Food and Drug Administration (FDA), the US beverage industry voluntarily reformulated beverages that were found to contain benzene levels at or above the 5 ng/g maximum contaminant level (MCL) for drinking water established by the US Environmental Protection Agency (EPA). 18 An MCL of 0.005 mg/L (5 ppb) is the EPA’s official limit for benzene in drinking water. 19 Furthermore, in a 2008 survey conducted by FDA, all of the reformulated beverages tested were shown to contain ≤1.1 ng/g benzene. Most of the beverages were reformulated by removing or reducing either benzoate or ascorbic acid or by adding EDTA. 20
Benzyl benzoate, potassium benzoate, and sodium benzoate
According to the United States Pharmacopoeia, benzyl benzoate contains less than 99% and not more than 100.5% C14H12O2, and potassium benzoate contains not less than 100.5% and not more than 99% C7H5KO2. Limits for water and heavy metals in potassium benzoate are stated as ≤1.5% and ≤0.001%, respectively. 21
The Food Chemicals Codex specification for sodium benzoate includes the following limitations: heavy metals, as Pb (≤2 mg/kg), alkalinity, as NaOH (≤0.04%), and water (≤1.5%). 22
Use
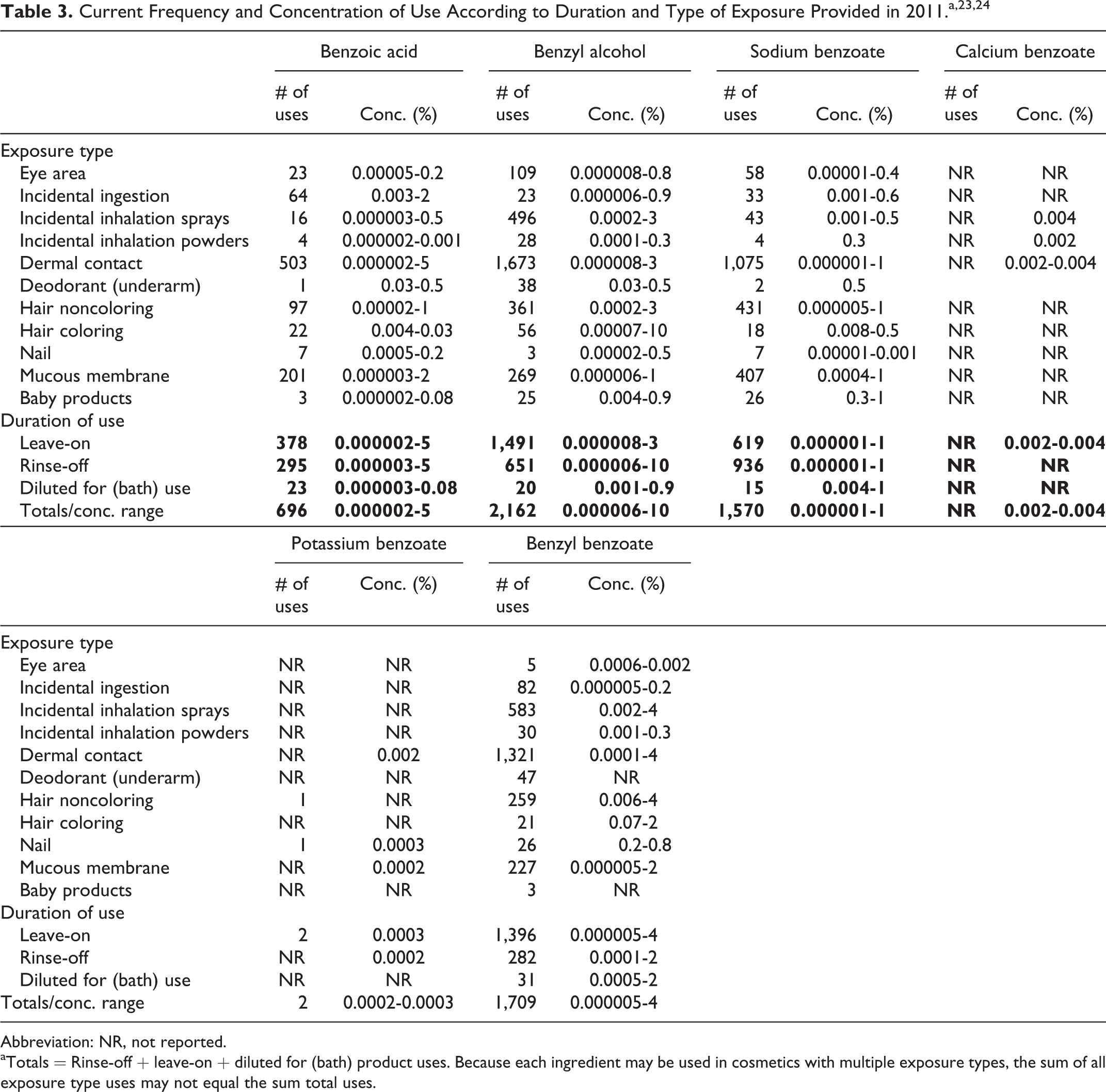
Cosmetic
The safety of the cosmetic ingredients included in this safety assessment is evaluated based on data received from the US FDA and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys conducted by the Council of maximum reported use concentrations, by product category.
According to information supplied to the VCRP in 2010, the following ingredients were being used in personal care products: benzoic acid, benzyl alcohol, sodium benzoate, potassium benzoate, and benzyl benzoate. 23 Uses of calcium benzoate and magnesium benzoate were not reported to the VCRP. These data are summarized in Table 3.
Abbreviation: NR, not reported.
aTotals = Rinse-off + leave-on + diluted for (bath) product uses. Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Independent of the VCRP data, a survey of ingredient use concentrations was conducted by the Council in 2010. The results of this survey indicated that the following ingredients were being used in cosmetic products: benzoic acid (0.000002%-5%), benzyl alcohol (0.000006%-10%), sodium benzoate (0.000001%-1%), calcium benzoate (0.002%-0.004%), potassium benzoate (0.002%-0.003%), and benzyl benzoate (0.000005%-4%). 24 No uses of magnesium benzoate were reported in the Council survey.
Personal care products containing the ingredients reported as being used may be applied to the skin, nails, or hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin, nails, or hair for variable periods following application. Daily or occasional use may extend over many years.
In the European Union, the maximum authorized concentrations for benzoic acid and sodium benzoate as preservatives in cosmetic products are rinse-off products, except oral care products (2.5%, as acid), oral care products (1.7%, as acid), and leave-on products (0.5%, as acid). For salts of benzoic acid other than sodium benzoate, the maximum authorized concentration is 0.5% (as acid). 25
The Scientific Committee on Cosmetic Products and Non-Food Products Intended for Consumers (SCCNFP) was asked to review the data submitted to support the safety of benzoic acid and its salts and esters, when used at concentrations other than those laid down in Annex VI to Directive 76/768/EEC as preservatives, for other specific nonpreservative purposes apparent from the presentation of the products and to determine whether benzoic acid and it salts and esters be used safely for nonpreservative purposes in cosmetic rinse-off products at a maximum concentration of 2.5% and in cosmetic oral care products at a maximum concentration of 1.7%. Data were provided to the SCCNFP in support of the use of benzoic acid as a nonpreservative. The SCCNFP did not find the submission appropriate for a safety evaluation of benzoic acid and sodium benzoate for the applied “other uses” in cosmetic products. 26 Subsequently, a second data set was provided, and these data were considered adequate for arriving at a conclusion on the safety of benzoic acid and sodium benzoate but not the other salts or esters. Based on these data, the Scientific Committee on Consumer Products (SCCP) issued the following opinion: The SCCP is of the opinion that benzoic acid and sodium benzoate are safe for use for preservative and nonpreservative purposes in cosmetic rinse-off products at a maximum concentration of 2.5% and in cosmetic oral care products at a maximum concentration of 1.7% and in leave-on products up to 0.5%. 27
Noncosmetic
According to FDA’s Select Committee on Generally Recognized as Safe (GRAS) substances, benzoic acid (21 CFR [Code of Federal Regulations] 184.1021) 28 and sodium benzoate (21 CFR 184.1733) 29 are classified as GRAS food substances. Sodium benzoate has also been approved by FDA for use as an antimycotic when migrating from food-packaging material (21 CFR 181.23). 30 Other CFR citations relating to FDA-approved direct/indirect food additive ingredient uses include benzyl alcohol (21 CFR 172.515, 175.105, 175.300, and 177.1210), 31 -34 benzoic acid (21 CFR 150.141, 150.161, 166.110, 175.300, and 177.1390), 35 -38 sodium benzoate (21 CFR 150.141, 150.161, and 166.110), 35 -37 calcium benzoate (21 CFR 166.110 and 178.2010), 37,39 potassium benzoate (21 CFR 166.110 and 177.1210), 37,34 and benzyl benzoate (21 CFR 172.515 and 175.105). 31,32
Benzyl alcohol is an FDA-approved diluent in color additive mixtures for external drug use. 40 In 2009, the FDA approved benzyl alcohol lotion, 5%, as a prescription medication for the treatment of head lice (Pediculosis capitis) infestation in patients aged 6 months and older. 41 The results of clinical trials supporting benzyl alcohol lotion 5% as a safe and effective topical treatment for head lice are included in the section on skin irritation and sensitization later in the report text. 42 Benzyl benzoate (10%) has been used in the treatment of scabies in developing countries. 43
Benzyl alcohol has been classified by FDA regarding its use in the following types of over-the-counter (OTC) drug products (category 1: generally recognized as safe [S] and effective [E] for the claimed therapeutic indication; category 2: not generally recognized as safe and effective or unacceptable indications; category 3: insufficient data available to permit final classification):
44
anorectal (final monograph: 1% to 4% use concentration range established; category 1) external analgesic (both category 1 and final monograph pending) external analgesic (final monograph: category 3) oral health care (category 1 pending) oral discomfort care (category 2 pending) oral discomfort care (category 3SE pending) pediculicide (final monograph: category 3) skin protectant (final monograph: category 3).
Similarly, benzoic acid has been classified by FDA regarding its use in the following types of OTC drug products:
44
acne (final monograph: category 3) antifungal (final monograph: category 3) oral health care—(category 3E pending) skin protectant—(final monograph: category 3).
Sodium benzoate has been classified by FDA regarding its use in menstrual/diuretic OTC drug products (final monograph: category 3), and benzyl benzoate has been approved for use in OTC pediculicides (final monograph: category 2). 44
Calcium benzoate, magnesium benzoate, and potassium benzoate are inert nonfood ingredients that, under the Federal Insecticide, Fungicide, and Rodenticide Act, are permitted by the EPA for use in nonfood use pesticide products. 45 Pesticide products containing benzoic acid as the active ingredient have been registered with EPA for use in the extermination of dust mites. 46
Benzyl alcohol has been listed as a chemical in photographic developing systems 47 and, at concentrations of 0.9% to 2%, is commonly used as an antibacterial agent in a variety of pharmaceutical formulations intended for intravenous administration. 48 Additionally, benzyl alcohol has been used in parenteral medications commonly administered to critically ill neonates. 49
Toxicokinetics
Oral Studies
Benzyl alcohol, benzoic acid, sodium benzoate, and potassium benzoate
Benzyl alcohol is metabolized to benzoic acid. Both benzoic acid and sodium benzoate are rapidly absorbed from the gastrointestinal tract of mammals and conjugated with glycine in the liver; the resulting hippuric acid is rapidly excreted in the urine. 2 Benzyl alcohol and benzoic acid and its sodium and potassium salt can be considered as a single category regarding human health because they are all rapidly metabolized and excreted via a common pathway within 24 hours. 3
Benzyl benzoate
Following oral administration (gavage, 0.5, 1, and 2 mL/kg) of benzyl benzoate to fasted female dogs, urine was collected for 5 to 6 days. All 3 doses resulted in creatinuria and urinary excretion of glycuronate conjugates. 50
Following the administration of unspecified oral doses of benzyl benzoate to cats and dogs (number and breed not stated), small amounts of hippuric acid were detected in the 24-hour urine. The metabolism of benzyl benzoate to benzoic acid in vivo has also been reported. Additional details for these 2 studies were not provided. 4
In a human patient, urinary excretion of hippuric acid was measured after oral dosing (single dose, 2.02 g) with benzyl benzoate. Urine was collected for 6 hours. Approximately 90% of the dose administered was excreted as hippuric acid in the urine. After oral dosing with 1 g, approximately 80% of the administered dose was excreted as hippuric acid. In a second patient, 71% of the administered dose was excreted as hippuric acid during the first 6 hours, and an additional 14% of the dose was excreted as such during the second 6 hours after dosing. 51
Dermal Studies
Benzoic acid
The penetration of benzoic acid through excised dorsal skin (full thickness and with stratum corneum removed) from guinea pigs was examined. Skin preparations were mounted in a 2-chamber diffusion cell. A suspension of an excess amount of benzoic acid in saline was added to donor compartments. In full-thickness skin, permeation proceeded with a short lag time. Solubility (Cd, unit = mM) and permeability (Kp, unit = ×10−2 cm/h) coefficients for benzoic acid were 32.7 ± 1.6 and 9.01 ± 1.51, respectively. Removal of the stratum corneum by tape stripping and its delipidation using an organic solvent mixture enhanced the skin penetration of benzoic acid. 52
Benzyl alcohol and benzyl benzoate
The percutaneous absorption of [7-14C]benzyl alcohol and [7-14C]benzyl benzoate in vivo was evaluated using 4 female Rhesus monkeys (10-19 years old). Each chemical was applied (4 µg/cm2, without or with occlusion [glass chamber—G or plastic wrap—P]) to a clipped 1 cm2 area of abdominal skin; the vehicle was acetone (10-20 µL/cm2). The animals remained in metabolism chairs/metabolism cages for 5 days. The amount of absorbed compound in the urine was determined by liquid scintillation counting. Mean values (±standard error of the mean, 4 animals) for urinary excretion of the administered dose were 77.4 ± 4.3% (benzyl alcohol) and 65.3 ± 13.4% (benzyl benzoate). Percutaneous absorption values (absorption, as percentage of applied dose) were as follows: benzyl alcohol (31.6% ± 4.2% [unoccluded]; 56.3 ± 14.5% [P]; 79.9% ± 7.4% [G]) and benzyl benzoate (57.0% ± 10.4% [unoccluded]; 71.2% ± 4.4% [P]; 64.7% ± 10.2% [G]). Over a 5-day period, the urinary excretion of radioactivity was 65.3% ± 13.4% following dermal application of benzyl benzoate and 56.5% ± 7.7% following dermal application of benzyl alcohol. When the acetone vehicle was replaced with a lotion (10 mg/cm2), percutaneous absorption was increased. The increase was significant for benzyl benzoate (P < .05) but not benzyl alcohol. There was no apparent correlation between the skin penetration of benzyl alcohol and benzyl benzoate and their octanol–water partition coefficients of 7.4 and 3.97, respectively. 53
The percutaneous absorption of benzyl benzoate in vitro was evaluated using human epidermis (from abdominal skin) placed in a glass diffusion chamber. Saline (5 mL) was added to the chamber and was in contact with the bottom of the epidermis. Benzyl benzoate (0.2 mL) was applied to the top of the epidermis. Six experiments were performed and the mean value for skin penetration of benzyl benzoate was 0.018% ± 0.002%. 54
The penetration of benzyl alcohol through split-thickness cadaver skin was evaluated using nonoccluded Franz diffusion cells. Benzyl alcohol (spiked with 14C radiolabel) in ethanol was applied to the skin at doses ranging from 0.9 to 10,600 µg/cm2. After 24 hours, the percentage of radioactivity that penetrated the skin increased gradually with dose, ranging from 19.8% ± 2.9% (lowest dose) to 29.2% ± 3.0% (highest dose). Also, less than 4% of the administered radioactivity was retained in the tissues at 24 hours, and evaporation accounted for the remaining percentage. Data from this study were also analyzed in relation to a finite dose diffusion/evaporation model. The results of this analysis indicated that the increase in benzyl alcohol absorption with dose was consistent with a 3-fold increase in diffusivity in the stratum corneum, as the concentration of benzyl alcohol increased from tracer levels to saturation. 55
In Vitro Study
The rate of hydrolysis of benzyl benzoate (in acetonitrile) by 80% human plasma in vitro was studied. The t½ for the in vitro hydrolysis of benzyl benzoate to benzoic acid in human plasma was 19 minutes. 56
Toxicological Studies
An OECD SIDS for high-volume chemicals’ initial assessment report on benzoic acid, sodium benzoate, potassium benzoate, and benzyl alcohol was published by UNEP in 2001, with the following recommendation relating to human safety: “Taking into account the rapid metabolism and excretion, the nonbioaccumulation, the low toxicity after acute and repeated exposures, the nonreproductive toxicity, the nongenotoxicity and the noncarcinogenicity, the low irritating and non- to very low sensitizing properties of these substances, as well as the controlled (industrial settings) and/or regulated (pharma, cosmetics and/or food) uses, these substances will pose a minimal risk to humans (workers and consumers).” 3(p35) Because the database for this safety assessment is similar to that for the 2001 CIR final report on benzyl alcohol, benzoic acid, and sodium benzoate, the available data in the OECD SIDS assessment are summarized with minimal details in sections that follow.
Acute Toxicity Studies
Dermal
Benzyl alcohol, benzoic acid, sodium benzoate, and potassium benzoate
According to the OECD SIDS initial assessment report on benzoates, low acute dermal toxicity is associated with benzyl alcohol and benzoic acid and its sodium and potassium salts. 3
Benzyl alcohol and benzyl benzoate
The acute dermal toxicity of benzyl benzoate was evaluated using rats (number not stated), and a median lethal dose (LD50) of 4,000 mg/kg body weight was reported. 4 Study details were not included. In a study involving rabbits, the acute dermal toxicity of undiluted benzyl benzoate was evaluated according to the Draize procedure (no good laboratory practice [GLP]; number of animals not stated), and an LD50 of 4,448 mg/kg body weight was reported. In another study involving rabbits, acute dermal LD50s of 2,000 mg/kg and 4,000 mg/kg have been reported for benzyl alcohol and benzyl benzoate, respectively. 57
Single dermal applications of 25% benzyl benzoate (20 mL in isopropanol/water) to the backs of cats resulted in death. Additional study details were not provided. 4
Oral
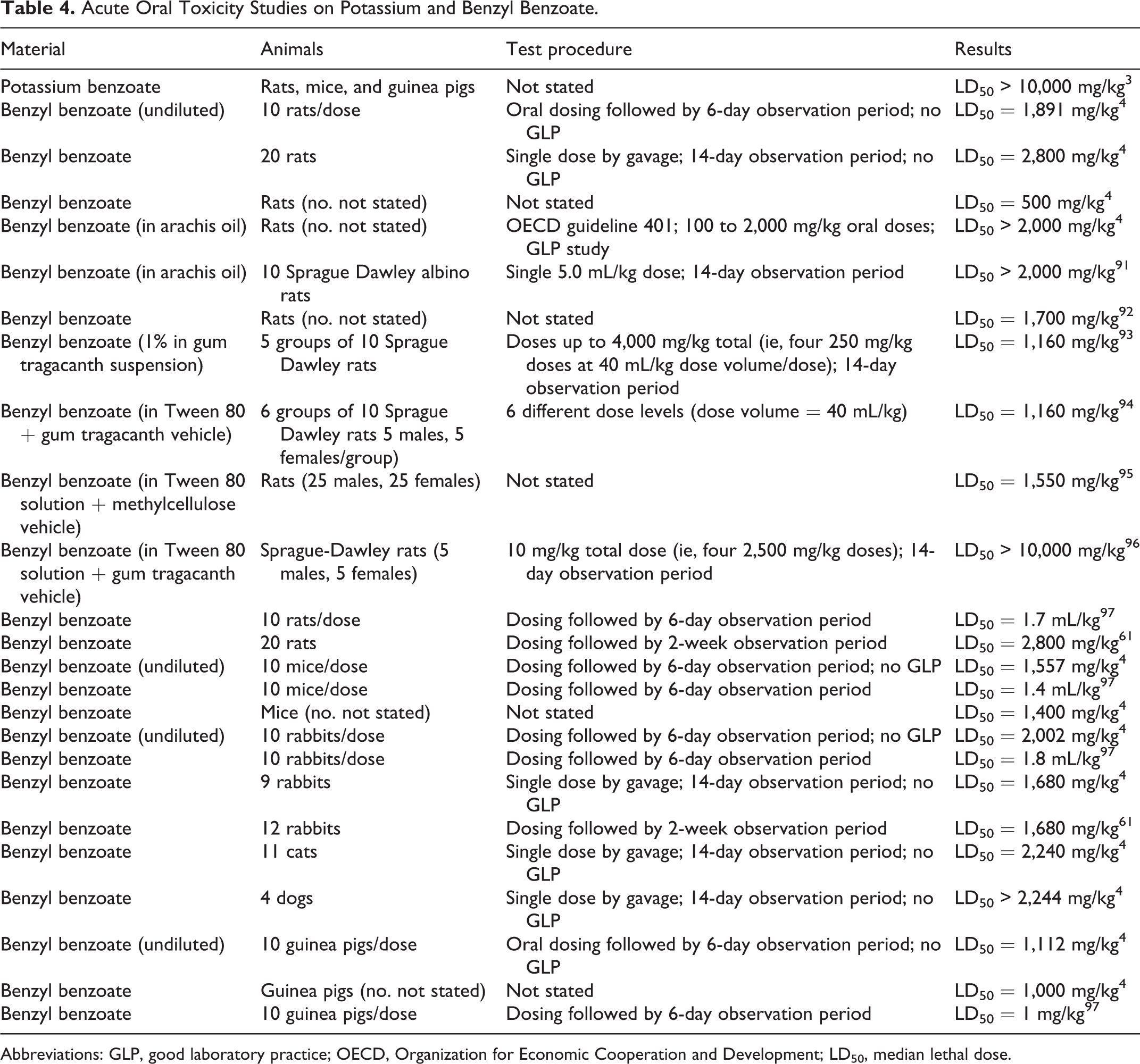
Acute oral toxicity data are summarized in Table 4.
Acute Oral Toxicity Studies on Potassium and Benzyl Benzoate.
Abbreviations: GLP, good laboratory practice; OECD, Organization for Economic Cooperation and Development; LD50, median lethal dose.
Benzyl alcohol, benzoic acid, sodium benzoate, and potassium benzoate
According to the OECD SIDS initial assessment report on benzoates, low acute oral toxicity is associated with benzoic acid and its sodium and potassium salts. The LD50 values were >2,000 mg/kg body weight, except for benzyl alcohol (LD50 = 1,610 mg/kg [rats]), which is considered harmful via the oral route. 3
Inhalation
Benzyl alcohol and benzoic acid
According to the OECD SIDS initial assessment report on benzoates, 4 hours of inhalation exposure to benzyl alcohol or benzoic acid at 4 and 12 mg/L aerosol/dust, respectively, did not cause death in rats. 3 Thus, low acute toxicity was associated with these compounds.
Intraperitoneal
Benzyl benzoate
Mice were dosed intraperitoneally with benzyl benzoate (test procedure/number of mice not stated), and an LD50 of >500 mg/kg body weight was reported. 4
Subcutaneous
Benzyl benzoate
Groups of adult C57BL/6J mice (5 per group) were injected subcutaneously (SC) with 0.1 mL benzyl benzoate (∼111 mg) daily for 4 weeks. An untreated group of sham injected animals served as the untreated concurrent control group. The adrenal glands were prepared for light and electron microscopic examination. The animals died within 3 hours of initial injection. Additional study details were not provided. 4
Guinea pigs (number not stated) received single SC injections of benzyl benzoate, and mortality rates at the administered doses were as follows: 0 (500 mg/kg), 30% (1,000 mg/kg), 80% (5,000 mg/kg), 100% (10,000 mg/kg), and 100% (20,000 mg/kg). 58
Intramuscular
Benzyl benzoate
Following benzyl benzoate intramuscular injection into cats (number not stated), doses of 2,000 and 10,000 mg/kg were described as lethal. 58
Repeated Dose Toxicity Studies
Dermal
Benzyl benzoate
The following effects were observed in groups of rats (3 males/3 females/group; strain not stated) that received dermal applications of 188, 488, 781, 1,250, or 2,000 mg/kg for 30 days: hyperplasia of squamous epithelium, degeneration of hair follicles and sebaceous glands, subcutaneous fibrosis, and hyperplasia of the thyroid gland. Seven areas of the shaved back were dosed in rotation. Dosing with 2,000 mg/kg also caused 4 deaths, decreased white blood cell numbers, and caused other blood effects. Body weight changes, decreased white blood cell numbers, and other blood effects were also observed following dosing with 1,250 mg/kg. Additional study details were not provided. 59
In a study involving rabbits (non-GLP; number and strain not stated), dermal doses of benzyl benzoate were applied daily for 90 days. 4 The range of doses tested was not stated. Deaths occurred at doses of 2,200 mg/kg body weight/day and greater. Testicular atrophy was noted at high doses, and possibly, there was also an increased incidence of focal nephritis and encephalitis. Very mild skin irritation was also observed. Additional details relating to this study were not provided. Benzyl benzoate was also applied topically to rabbits at doses of 0.5 mL/kg/day daily for 90 days. Slight dermatitis and lethargy were observed, and at higher doses, slight to moderate testicular atrophy was apparent. Increased leucocyte counts were reported and results were also suggestive of kidney damage. Benzyl benzoate also caused death, but the number of mortalities and corresponding toxic doses were not reported. Repeated dermal applications of benzyl benzoate (doses not stated) did not cause toxic symptoms in the following species: dogs, horses, cows (females), sheep, or pigs. Additional study details were not provided. 4
In another study, rabbits (number and strain not stated) received dermal applications of benzyl benzoate at doses of 0.5, 1.0, 2.0, and 4.0 mL/kg daily for 90 days. 60 Lethality and effects on the following were reported after dosing: blood effects (at 2.0 mL/kg), kidneys, skin (slight dermatitis and other effects), reproduction, leukocyte (increased 2.5× normal), testicular damage, and kidney damage. Animal deaths were not preceded by the usual pattern of systemic effects prior to death. Additional details were not included.
Five applications of benzyl benzoate (undiluted) were made to the backs of the following domestic animals: 1 sheep (250 mL dose), 1 pig (200 mL), 1 young cow (500 mL), and 1 horse (1,000 mL). The animals were observed for 2 weeks. None of the animals died. In another study, 6 applications of undiluted benzyl benzoate were made to the backs (clipped skin, 4” × 6” area) of 3 dogs as follows: 1 dog (200 mL volume) and 2 dogs (100 mL). The animals were observed for 2 weeks. No test substance-related effects were observed. 61
Benzyl benzoate was rubbed onto cutaneous lesions on the shaved upper thighs of cats (number not stated). Three applications per animal were made. Within 24 to 36 hours, the test substance was lethal at doses of 8,700 to 12,800 mg/kg. 58
Oral
Benzyl alcohol, benzoic acid, sodium benzoate, and potassium benzoate
According to the OECD SIDS initial assessment report on benzoates, repeated dose oral toxicity studies yielded a no-observed-adverse-effect level (NOAEL) of 800 mg/kg/day for benzoic acid and >1,000 mg/kg/day for the salts. 3 At higher doses, increased mortality, decreased weight gain, and liver and kidney effects were observed. Systemic toxic effects of a similar nature (eg, liver and kidney) were observed after dosing with benzyl alcohol, benzoic acid, sodium benzoate, and potassium benzoate. However, these effects were observed at higher doses of benzoic acid and its salts when compared to dosing with benzyl alcohol. Long-term studies on benzyl alcohol yielded an NOAEL of >400 mg/kg body weight per day for rats and >200 mg/kg body weight per day for mice. At higher doses, effects on body weight and lesions in the brain, thymus, skeletal muscle, and kidneys were observed. It was noted that one should take into account that oral administration was by gavage in these studies, whereby saturation of metabolic pathways is likely to occur. It was concluded that benzoic acid and its salts exhibited very low repeated dose toxicity and that benzyl alcohol exhibited low repeated dose toxicity.
Benzyl benzoate
No effects were observed in a 7-month toxicity study in which mice (number and strain not stated) were dosed orally with benzyl benzoate (800 mg/kg). 62
Inhalation
Benzyl alcohol and benzoic acid
A 4-week inhalation toxicity study of aerosolized benzyl alcohol and benzoic acid was performed using groups of Crl:CD(SD) rats. 63 Four groups of rats (10/sex/group) were exposed (nose-only) to aerosolized benzyl alcohol 5 days per week (6 h/d) for 4 weeks. Each animal received a minimum of 20 exposures of benzyl alcohol, and target exposure concentrations were 30, 100, 300, and 1,000 mg/m3 for the four groups. Two additional groups of rats were exposed to aerosolized benzoic acid (2.5 and 12.5 mg/m3) according to the same procedure. The group designated for exposure to benzyl alcohol (30 mg/m3 target) was actually exposed to a mean atmosphere concentration that was 136% of the target concentration. For the remaining groups, all mean atmosphere concentrations were within 96.7% to 107.2% of the respective target concentrations. A concurrent control group was exposed to filtered air only.
There were no test substance–related deaths and no effects on any of the following: body weight, food consumption, clinical pathology parameters, and organ weights. Additionally, there were no test substance–related macroscopic or microscopic findings. The no-observed-effect level (NOEL) and NOAEL were considered to be 1072 mg/m3 for benzyl alcohol and 12.6 mg/m3 for benzoic acid.
Parenteral
Benzyl benzoate
Rabbits (1 or 2 animals) were injected SC once with benzyl benzoate in olive oil (doses of 1-2.5 mL/kg) or received 4 daily 0.25 mg/kg SC injections. Effects on leukocyte counts were monitored over an 11-day period, and the animals were also examined for mortalities and clinical signs. The 2.5 mL/kg dose was lethal and clinical signs were observed. No effects were associated with the 0.25, 1, or 1.5 mL/kg doses. 64
Reproductive and Developmental Toxicity
Benzoic Acid, Sodium Benzoate, Potassium Benzoate, and Benzyl Alcohol
According to the OECD SIDS initial assessment report on benzyl alcohol, benzoic acid, and its sodium and potassium salts, benzoic acid did not induce reproductive effects in a 4-generation reproductive toxicity study (NOAEL > 750 mg/kg). 3 Groups of rats (20 rats/sex/group) received benzoic acid doses of 375 or 750 mg/kg/day in the diet continuously. Animals of the third generation were killed after 16 weeks. Also, test substance–related effects on reproductive organs (based on gross and microscopic examination) were not observed in subchronic studies (rats and mice) on benzyl alcohol and sodium benzoate.
In groups of rats fed sodium benzoate (doses up to 5,600 mg/kg/day) during each day of gestation, developmental effects were observed only in the presence of marked maternal toxicity (reduced food intake and decreased body weight; NOAEL = 1,400 mg/kg/day). All developmental effects were observed at doses ≥2,800 mg/kg/day. On days 6 through 10, 15, or 18 of gestation, dosing with sodium benzoate by gavage in hamsters (NOEL = 300 mg/kg body weight), rabbits (NOEL = 250 mg/kg), and CD-1 mice (NOEL = 175 mg/kg) did not result in maternal toxicity; doses >NOEL were not tested. In mice of an unspecified strain dosed with benzyl alcohol by gavage, an NOAEL of 550 mg/kg (only dose) body weight for developmental toxicity was reported. An LOAEL of 750 mg/kg/day (only dose) for developmental toxicity was reported for CD-1 mice dosed orally (gavage) with benzyl alcohol. In this study, maternal toxicity (increased mortality, decreased body weight, and clinical toxicity) was observed. 3
Sodium Benzoate
The effect of sodium benzoate on biochemical aspects of pregnant female albino rats and survival of their offspring was evaluated using groups of 10 Sprague Dawley rats. 65 Two groups of weanling female rats were fed (ad libitum) sodium benzoate at doses of 0.01 g and 0.0125 g in the diet, respectively, daily for 12 weeks. Standard diet was fed to the control group. After mating, the animals were fed ad libitum during pregnancy. When compared to the control group, both doses of sodium benzoate induced a decrease in serum bilirubin and an increase in serum urea.
Both doses of sodium benzoate also induced an increase in serum uric acid (P < .01 and P < .05, respectively). Serum alanine aminotransferase activity was significantly greater (P < .01) in the high-dose group but not in the low-dose group. The high dose did not induce a significant increase in serum creatinine. Statistically significant (P < .01) decreases in food intake, hemoglobin, and hematocrit were also reported. Pregnant rats that received high doses of sodium benzoate sustained a 13.6% decrease (P < .05) in mean weight of their pups, when compared to mean pup weight in the corresponding control group. 65
Benzyl Benzoate
The teratogenicity of benzyl benzoate was evaluated using 21 rats (strains not stated) per dose; doses administered in the diet were defined as 0.04% or 1.0% (≈24 or 595 mg/kg body weight/day). 4 The test substance was administered daily from day 0 of gestation to day 21 postparturition. The results of examinations for external, skeletal, or visceral anomalies indicated that benzyl benzoate did not induce harmful effects in fetuses.
In a reproductive and developmental toxicity study, pregnant rats (number and strain not stated) were fed diets supplemented with 0.04% or 1.0% benzyl benzoate from day 0 of gestation to day 21 postparturition. 66 Another group of rats was fed a control diet. There were no effects on reproductive or developmental toxicity parameters; no external, skeletal, or visceral anomalies were observed in fetuses from either treatment group. Minor variations were observed. A significantly decreased number of fetuses with incomplete sternebrae was noted in the 1% benzyl benzoate treatment group.
Testicular atrophy was observed in 2 repeated dose dermal toxicity studies (90 days) on benzyl benzoate involving rabbits. 4 The exact doses at which this finding occurred were not stated. However, testicular atrophy was observed at daily doses >0.5 g/kg/day. These studies are summarized in the Repeated Dose Toxicity and Dermal Studies section earlier in the report text.
An outbred strain of pregnant Swiss mice (Naval Medical Research Institute mice [NMRI mice]; 34 animals) were injected SC with an unspecified dose of benzyl benzoate in castor oil on days 1 and 11 of gestation. 67 The animals were killed on gestation day 17. Untreated mice served as controls. Fetal observations relating to the following were made: counts, sex, weight, and malformations. There was no evidence of test substance–related effects on fetuses. Additional details were not included.
A developmental toxicity study was performed using Drosophila melanogaster. 68 One group of males and females was raised on food medium with 4% benzyl benzoate in ethanol and the other group was raised on control feed medium. Virgin Canton-S males and females were collected daily for 4 days and then mated. The percentage of eggs hatched was 6.5 times greater in flies on feed containing benzyl benzoate, compared to those on control feed.
The safety of benzyl benzoate lotion (25% benzyl benzoate) as a topical treatment for scabies during pregnancy was assessed using a population of 444 pregnant women and their matched controls (1,776 pregnant women). 69 The study population consisted of refugee and migrant women attending antenatal clinics on the Thai-Burmese border between August of 1993 and April of 2006. Most first treatments took place during the second and third trimesters, and the overall median gestation exposure was 24.5 weeks. Treated women (444) received 559 applications of butyl benzoate lotion (79.5%, 15.5%, 4.5%, and 0.l5% receiving 1, 2, 3, and 4 treatments, respectively).
Conditional Poisson regression was used to estimate risk ratios for outcomes of pregnancy (proportion of abortions, congenital abnormalities, neonatal deaths, stillbirths, and premature babies), mean birth weight, and estimated median gestational age for scabies and scabies-free women. Regarding pregnancy outcomes, there were no statistically significant differences between women treated with the lotion and their matched controls. Thus, there was no evidence of adverse effects on pregnancy outcome due to topical application of benzyl benzoate (25%) lotion. 69
Estrogenic Effects
Benzoic acid
The estrogenic activity of benzoic acid was evaluated in the recombinant yeast human estrogen receptor (ERα) assay in vitro and in uterotrophic assays involving immature female Alpk:APfSD rats (21-22 days old) and immature female Alpk:APfCD-1 mice. 70 Immature mouse uterotrophic assays involved 3 daily SC injections of benzoic acid in corn oil (100 and 1,000 µg/kg doses; 5 mL/kg = dose volume) ending on day 4. The same protocol was used for the rat assays, with the exception that benzoic acid in arachis oil was injected (doses of 10, 100, and 1,000 µg/kg).
Benzoic acid (10−7 to 10−3 M) was negative in the recombinant yeast human ERα assay in vitro. Overall, benzoic acid produced one statistically significant and 3 statistically nonsignificant increases in average uterine weight (rat assay) and 1 significant and 6 nonsignificant decreases in average uterine weight (mouse assay). Therefore, in all uterotrophic assays, benzoic acid did not produce any reproducible evidence of estrogenic activity. Results for vehicle controls were negative and the positive control, estradiol, was uterotrophic.70
Benzyl benzoate
The estrogenic activity of benzyl benzoate (concentration range: 10−9 to 10−4 M) in vitro was evaluated in the E-screen test using MCF7 breast cancer cells. Untreated cultures served as controls. In this test, human breast adenocarcinoma cell line (MCF7 cells) proliferate in the presence of estrogen. Cell numbers were assessed by measurement of the total protein content, using the sulforhodamine B assay. Compared to control cultures, benzyl benzoate did not increase the proliferation of MCF7 cells (P > .05) over the range of concentrations tested. 71
The estrogenic activity of benzyl benzoate, benzyl salicylate, and butylphenyl methylpropional (Lilial) in the estrogen-responsive MCF7 human breast cancer cell line was evaluated using the following assays: competitive binding assay to the ER of MCF7 cytosol, competitive binding assay to recombinant ERα and ERβ, and the assay of stably transfected estrogen-responsive reporter gene (ERE-CAT) in MCF7 cells. 72 In the latter assay, the ERE-CAT vector consisted of the estrogen response element (ERE) of the vitellogenin A2 gene from −331 to −295 bp cloned into the pBLCAT2 vector upstream of the thymidine kinase promoter. Cell proliferation experiments were also performed.
The following results indicate that all 3 chemicals produced estrogenic responses in cultured human breast cancer cells in vitro. 72 At 3,000,000-fold molar excess, each chemical was able to partially displace [3H]estradiol from recombinant human estrogen receptors ERα and ERβ and from cytosolic ER of MCF7 cells.
At concentrations in the 5 × 10−5 to 5 × 10−4 M range, benzyl benzoate and the other 2 chemicals were able to increase the expression of a stably integrated ERE-CAT and of the endogenous estrogen-responsive pS2 gene in MCF7 cells; however, these effects were at a lesser extent when compared to 10−8 M 17β-estradiol (5,000-50,000 molar excess of benzyl benzoate).
In cell proliferation experiments, each chemical increased the proliferation of estrogen-dependent cells over a 7-day period. Cell proliferation was inhibited by fulvestrant (antiestrogen), suggesting an ER-mediated mechanism. However, over a 35-day period, the extent of proliferation in the presence of 10−4 M benzyl benzoate, benzyl salicylate, or butylphenyl methylpropional increased to the same magnitude as that observed in the presence of 10−8 M 17β-estradiol over a 14-day period (10,000 molar excess of benzyl benzoate). 72
Genotoxicity
In Vitro
Benzoic acid, sodium benzoate, potassium benzoate, and benzyl alcohol
According to the OECD SIDS initial assessment report on benzyl alcohol, benzoic acid, and its sodium and potassium salt, each chemical was not mutagenic in in vitro Ames tests. Various results (negative and positive [chromosomal/chromatid responses]) for sodium benzoate, potassium benzoate, and benzyl alcohol were obtained in other in vitro genotoxicity assays. However, while some mixed and/or equivocal in vitro chromosomal/chromatid responses have been observed, sodium benzoate and benzyl alcohol were not genotoxic in the in vivo cytogenetic assay, the micronucleus test, or in other in vivo assays. The weight of evidence of the in vitro and in vivo genotoxicity data indicates that these chemicals are not mutagenic or clastogenic. 3
In the Ames test, benzyl benzoate was not mutagenic to Salmonella typhimurium strains TA98 or TA100 at doses up to 5,000 µg/plate (plate incorporation assay) or 5,000 mg/plate (preincubation assay). 4 Benzyl benzoate also was not mutagenic to the following strains at doses up to 3 µmol/plate (preincubation assay) with or without metabolic activation: TA98, TA100, TA1525, and TA1537. In the recombination assay, benzyl benzoate was not mutagenic to Bacillus subtilis strains H17 or M45 at a concentration of 10 mg/disk.
In another Ames test, the mutagenicity of benzyl benzoate (in ethanol) was evaluated using S typhimurium strains TA98, TA100, TA1535, and TA1537. 73 Benzyl benzoate was tested at a concentration of 3 µmol/plate with and without metabolic activation and was not found to be mutagenic.
In the in vitro comet assay, the DNA-damaging potential of benzoic acid and benzyl alcohol was evaluated using human blood cells (lymphocytes). 74 This assay is able to detect single and double strand breaks in DNA. Benzoic acid was evaluated at concentrations ranging from 0.05 to 5 mM and, benzyl alcohol, at concentrations ranging from 1 to 50 mM. Benzoic acid was tested at lower concentrations (≤5 mM) because this chemical has apoptotic effects at higher concentrations. The tail moment and percentage tail DNA in chemicals evaluated were compared to results for the solvent control (distilled water). Significantly increased tail moment and percentage tail DNA were noted only with 5 mM benzoic acid and with 25 and 50 M benzyl alcohol, indicating genotoxicity. A dose response was not observed.
Benzoic acid (in dimethyl sulfoxide) was evaluated in the micronucleus test at concentrations ranging from 250 to 1,000 µg/mL (with and without metabolic activation) using L5178Y TK ± clone 3.7.2C mouse lymphoma cells. Cultures without benzoic acid served as negative controls and mitomycin C served as the positive control. Benzoic acid was nongenotoxic over the range of concentrations tested, both with and without metabolic activation. The positive control was genotoxic. 75
The genotoxicity of sodium benzoate was evaluated in the sister chromatid exchanges (SCEs) assay using human blood lymphocyte cultures incubated for 72 hours. Preparations were scored blindly for cells in their first mitosis, second mitosis, and third and subsequent divisions. Forty SCEs second division cells from each culture were scored for SCEs. Compared to negative control cultures, sodium benzoate (0.02, 0.2, 2, 4, and 8 mM) caused statistically significant delays in cell division (P < .01), indicative of weak cytostatic activity, but did not induce cytotoxicity. Also, when compared to negative control cultures, 8 mM sodium benzoate induced a statistically significant increase (P < .01) in SCEs/cell. Sodium benzoate (2 mM) induced a statistically significant decrease (P < .05) in SCEs/cell when compared to the 8 mM sodium benzoate culture. 76
The DNA-damaging activity of sodium benzoate and potassium benzoate was evaluated in the spore rec-assay using B subtilis M45 (rec−) and H17 (rec+) strains with and without metabolic activation. Sodium benzoate (in water) was tested at doses of 16 and 20 mg/disk and potassium benzoate (in mixed solution of water and ethanol [1:1]) was tested at doses of 15 and 20 mg/disk. Results for both chemicals were judged positive in this assay. 77
Photogenotoxicity
Benzoic acid and sodium benzoate
The photogenotoxicity of benzoic acid (0.5%) and sodium benzoate (0.5%) and other food additives was evaluated using Escherichia coli cell suspensions. 78 Initially, the cytotoxic and genotoxic potential of each in the absence of sunlight (200-3,000 nm; mean intensity = 5.8 × 102 Wm−2) was evaluated by allowing the cells to remain in contact with the test article for 80 minutes in the dark. Neither benzoic acid nor sodium benzoate affected cell viability (ie, not cytotoxic) or the number of spontaneous mutations in the absence of sunlight. The number of spontaneous mutations was 2.09 mutations per 108 cells. Also, these chemicals did not induce mutations in the absence of sunlight.
Plates containing the cellular suspension (5 mL) and a food additive (0.5% concentration) were exposed to sunlight for periods ranging from 0 to 60 minutes. Exposure to sunlight resulted in cell death in the presence or absence of benzoic acid or sodium benzoate. When cells were exposed to direct sunlight in the absence of benzoic acid or sodium benzoate, the number of induced mutations increased with duration of exposure (5.1 × 102 mutations per 108 cells after 60 minutes). In the presence of benzoic acid or sodium benzoate, the number of induced mutations increased with the duration of exposure to sunlight. This increase was almost additive over that observed with sunlight exposure alone. After 60 minutes, there were approximately 1 × 103 mutations per 108 cells. 78
Effect on DNA Synthesis
Sodium benzoate
Aqueous sodium benzoate, at a concentration of 7.20 mg/mL, was added to Tetrahymena pyriformis in an experimental series consisting of six 100 mL cultures. Another experimental series, without sodium benzoate, served as the control. The quantitative analysis of DNA content of the protozoan nuclei was performed using an image analysis system. Sodium benzoate caused a statistically significant increase (P < .01) in DNA content, suggestive of stimulation of mitosis. The authors speculated that this observation might represent a direct cytotoxic effect due to increased DNA content of nuclei. 79
Carcinogenicity
The following carcinogenicity study summaries are included in the CIR final report on benzyl alcohol, benzoic acid, and sodium benzoate that was published in 2001. 2
Benzyl Alcohol
Groups of 100 F344/N rats (50 each sex) were dosed orally with 200 or 400 mg/kg benzyl alcohol in corn oil, 5 days per week for 103 weeks. 2 Groups of 100 B6C3F1 mice were dosed with 100 or 200 mg/kg benzyl alcohol following the same schedule. During week 80, mice were mistakenly dosed for 4 days with 375 (low-dose group) and 750 mg/kg (high-dose group) of α-methylbenzyl alcohol. Dose-related negative trends were noted in the incidences of anterior pituitary gland neoplasms in female rats (vehicle control, 29/50; low dose, 17/47; high dose, 9/49) and of Harderian gland adenomas in male mice (8/50; 3/50; 2/50). Epithelial hyperplasia of the nonglandular stomach was noted in 4 of 50 high-dose male rats; it was not found in controls or low-dose male rats. An increased incidence of adenomas of the adrenal cortex noted in high-dose male mice (0/48; 0/44; 3/48) was within historical range and not considered compound related. The National Toxicology Program investigators considered the study negative for benzyl alcohol-induced carcinogenicity. However, reviewing the study, the EPA considered the 3 of 48 incidence of adrenal cortex adenoma to be “equivocal evidence of carcinogenic activity rather than negative.”
Benzyl Alcohol and Benzoic Acid
A skin painting study was performed using groups of 120 Eppley Swiss mice (60 per sex). 2 A nonoxidative hair dye containing 2.0% benzyl alcohol and 0.016% benzoic acid was painted onto the skin at a dose of 0.05 mL/application, 3 times weekly for 20 months. Sites were shaved of hair 24 hours before each application and a new bottle of dye was used each week. Two groups of control animals were shaved but not treated. Nine months into the study, 10 mice/sex/group were killed. Body weights and survival differed little between treatment and control groups. Varying degrees of chronic dermal inflammation were noted in all groups, including the controls. A significant (P < .01) increase in malignant lymphomas was noted in treated females (23/60). However, the researchers noted that one concurrent control group had a very low incidence (7/60% or 12%) of that tumor type. The rate was 22% for the other control group and had averaged 33% for 3 control groups in previous studies. Thus, the findings were not considered treatment related. The incidence of pulmonary adenomas and hepatic hemangiomas, which are common in this mouse strain, was similar between treated and control groups. No unusual neoplasms were observed.
Sodium Benzoate
For 18 to 24 months, groups of Fischer 344 rats (50 males and 52 females per group) received feed containing 2% or 1% sodium benzoate. 2 The doses corresponded to the maximum tolerated dose (MTD) and ½MTD as determined in 6-week toxicity studies. A control group of 25 male and 43 female rats received untreated feed. Average daily sodium benzoate intake was 280 and 202 mg, respectively, for male and female rats of the 2% group and 141 and 102 mg, respectively, for male and female rats of the 1% group. No clinical signs of toxicity or differences in average body weight or mortality rates were noted in treated rats when compared with controls. Neoplasms that were present in treated rats were similar in type and number to those in controls. No evidence of sodium benzoate-related carcinogenicity was observed.
In a life-time drinking water study, 100 Albino Swiss mice (50 of each sex) were supplied with water containing 2% sodium benzoate. 2 A control group of 200 mice was supplied with untreated water. Average daily intake of sodium benzoate was 124.0 and 119.2 mg for males and females, respectively. Sodium benzoate treatment did not affect survival. No carcinogenic effect attributable to treatment was noted at necropsy.
Other Relevant Studies
Ocular Toxicity
Benzyl alcohol
The ocular toxicity of benzyl alcohol was evaluated using 9 New Zealand rabbits. 80 The following test concentrations of benzyl alcohol (in stock solution of carboxymethylcellulose and polysorbate 80) were injected intravitreally, each into 3 eyes (15 eyes total injected): 0.0073%, 0.022%, 0.073%, 0.222%, and 0.733%. Control (2) eyes were injected intravitreally with 0.9% normal saline. Ocular toxicity was not observed at the lowest concentration; transient clinical changes (retinal hemorrhage and whitening), but no remarkable pathological changes, were noted in 1 of 3 eyes tested with 0.022% benzyl alcohol. Ocular toxicity was observed at the remaining concentrations; changes in the outer retina included loss and shortening of outer segments and receptors.
Triamcinolone acetonide (TA) has been increasingly applied intravitreally in therapy for retinal diseases, and the vehicles of TA injections usually contain benzyl alcohol as a preservative. 81 The ocular toxicity of 2 commercial vehicles (A and B) for drugs was evaluated using 3 groups of 12 New Zealand white rabbits (vehicle A, vehicle B, and balanced saline solution [BSS, negative control] groups). The 2 vehicles were similar in that each also contained benzyl alcohol (9.9 mg/mL), sodium carboxymethylcellulose (7.5 mg/mL), and polysorbate 80 (0.4 mg/mL). Each group of 12 was divided into 2 groups, each receiving a different intravitreal dose (0.1 or 0.2 mL). Ocular examination (using ophthalmoscope) results were normal for group A and the negative control group; retinal damage was obvious in group B. However, at microscopic examination, retinal toxicity was observed in groups A and B. Vehicle B induced extensive retinal necrosis and atrophy, whereas vehicle A induced localized retinal changes.
In a published comment on the preceding publication, the association of the benzyl alcohol preservative with retinal toxicity was acknowledged. 82 However, it was noted that retinal toxicity is not observed at concentrations achieved following injection of 0.1 mL preserved TA in clinical practice.
In a similar study, 4 groups of Chinchilla rabbits (pigmented) were injected intravitreally with the following 4 materials, respectively: 0.1 mL BSS, 0.1 mL TA (1.3 mg/mL), 0.1 mL vehicle (0.99% benzyl alcohol and other excipient) alone, and 0.1 mL TA + vehicle. 83 Given the materials tested, it should be noted that commercially prepared TA was described as a suspension containing 40 mg TA plus vehicle containing 0.99% benzyl alcohol, 0.75% excipient sodium carboxymethylcellulose, and 0.04% polysorbate. At microscopic examination, morphologic changes in the ciliary body, lens, and retina were observed only in 2 groups (TA + vehicle and vehicle alone). Thus, it was concluded that benzyl alcohol was toxic to these tissues when injected intravitreally.
Dermal Irritation and Sensitization Studies
Irritation and Sensitization
Skin irritation and sensitization data (human and nonhuman) are listed in Table 5.
Skin Irritation and Sensitization Studies.
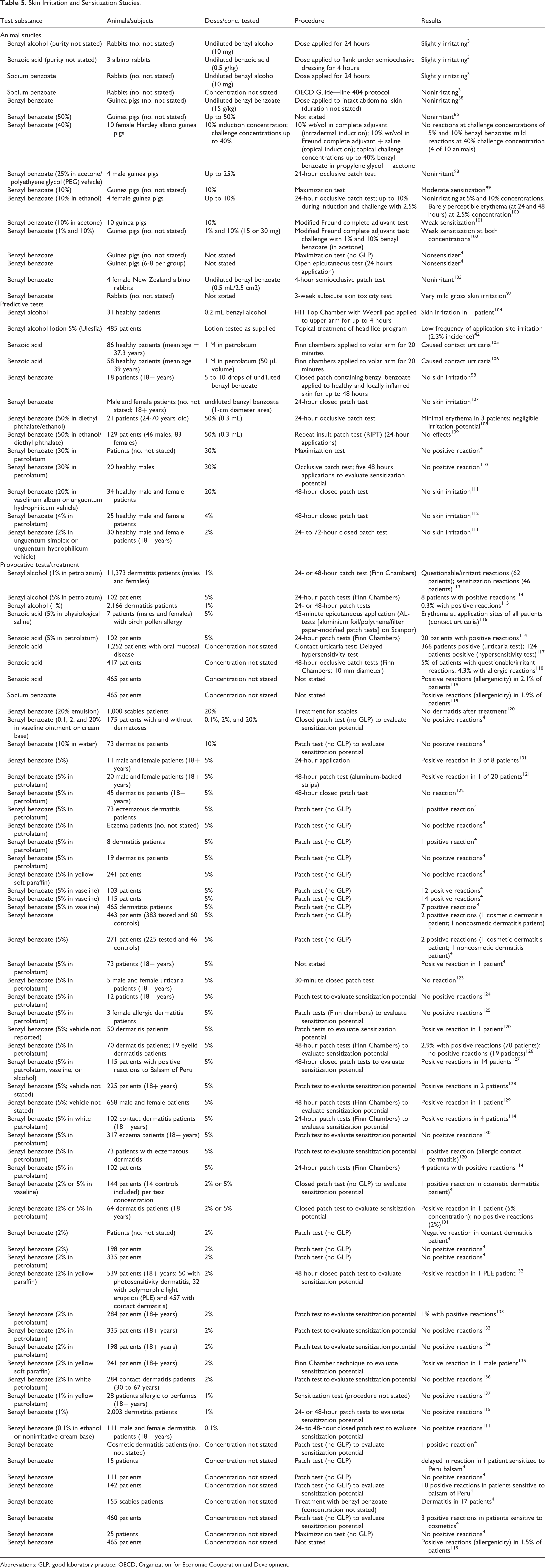
Abbreviations: GLP, good laboratory practice; OECD, Organization for Economic Cooperation and Development.
Benzyl alcohol, benzoic acid, sodium benzoate, and potassium benzoate
According to the OECD SIDS initial assessment report on benzoates, benzoic acid and benzyl alcohol were slightly irritating to the skin, whereas sodium benzoate was nonirritating. 3 Skin irritation data on potassium benzoate were not available; however, it is not expected that this compound would be irritating to the skin. 3 In the same report, benzoic acid did not induce sensitization in animal studies; however, a low incidence of positive reactions was observed in dermatologic patients patch tested. Similar results were reported for sodium benzoate, and it has been suggested that the positive reactions observed were actually nonimmunologic contact urticaria. Both positive and negative results were reported for benzyl alcohol in animal skin sensitization studies. A maximum skin sensitization incidence of 1% was reported for benzyl alcohol in human patch tests. Occupational exposure to benzyl alcohol, benzoic acid, or sodium benzoate has not resulted in skin sensitization over a period of decades.
Phototoxicity
In Vitro
Benzyl alcohol and sodium benzoate
An in vitro photohemolysis test in human erythrocyte suspensions was used to evaluate the phototoxicity of benzyl alcohol and benzyl benzoate. 84 Cells were incubated with either chemical (10− 3 mol/L) for 1 hour and exposed to UVA or short wave ultraviolet light (UVB) radiation. The UVA rich light source was a UVASUN 5000 lamp (320-460 nm; 42 mW/cm2), and the UVB rich light source was a lamp with TL 20 W/12 light bulbs (between 275 and 365 nm; 1 mW/cm2 [UVB] and 0.4 mW/cm2 [UVA]). Moderate hemolysis (∼6%) was induced by benzyl alcohol (at 10−4 mol/L) only in the presence of UVA, classifying it as a weakly phototoxic compound. Benzyl benzoate (test concentration not stated) was not phototoxic in the presence of UVA or UVB light.
Animal
Benzyl benzoate
The phototoxicity of benzyl benzoate was evaluated using hairless mice (number and strain not stated). 4 Details relating to the test protocol were not included. Following test substance application, test sites were irradiated for a total of 3 or 4, 24-hour exposures. No reactions were observed after 3 exposures; however, a slightly positive reaction was observed after 4 exposures.
The phototoxicity of benzyl benzoate was evaluated, using 5 female Hartley guinea pigs, at concentrations of 10%, 30%, and 50% in acetone. 85 At 4 hours after depilation, the test substance was applied to a circular area (diameter = 1.5 cm) on both sides of the back. Two applications (left and right side) of each concentration were made to the back of each animal. One side of each animal was covered with aluminum foil and the other was irradiated with 5 Toshiba model FL-40 BLB lamps (320-400 nm) at a distance of 10 cm for 70 minutes. Photo irritation was not observed at any of the 3 test concentrations.
Ocular Irritation Studies
Animal
Benzoic acid, benzyl alcohol, sodium benzoate, and potassium benzoate
According to the OECD SIDS initial assessment report on benzoates, benzyl alcohol (4% aqueous) and benzoic acid (undiluted) were irritating to the eyes of rabbits, and sodium benzoate (concentration not stated) was only slightly irritating. 3 Data on potassium benzoate were not available; however, it was expected that this chemical would induce slight ocular irritation.
Benzyl benzoate
Undiluted benzyl benzoate was instilled (0.1 mL) into the eyes of 6 rabbits (3 males, 3 females), and reactions were scored up to 72-hour postinstillation. 86 Ocular irritation was observed in 3 rabbits (only reactions reported) at 1-hour postinstillation.
Human
Benzyl benzoate
In an ocular irritation study involving human patients (test procedure/number of patients not stated), undiluted benzyl benzoate was classified as an ocular irritant. 4
Clinical Studies
Case Reports
Case reports involving patch, intradermal, or prick testing of benzyl alcohol, benzoic acid, sodium benzoate, or benzyl benzoate are listed in Table 6.
Case Reports.
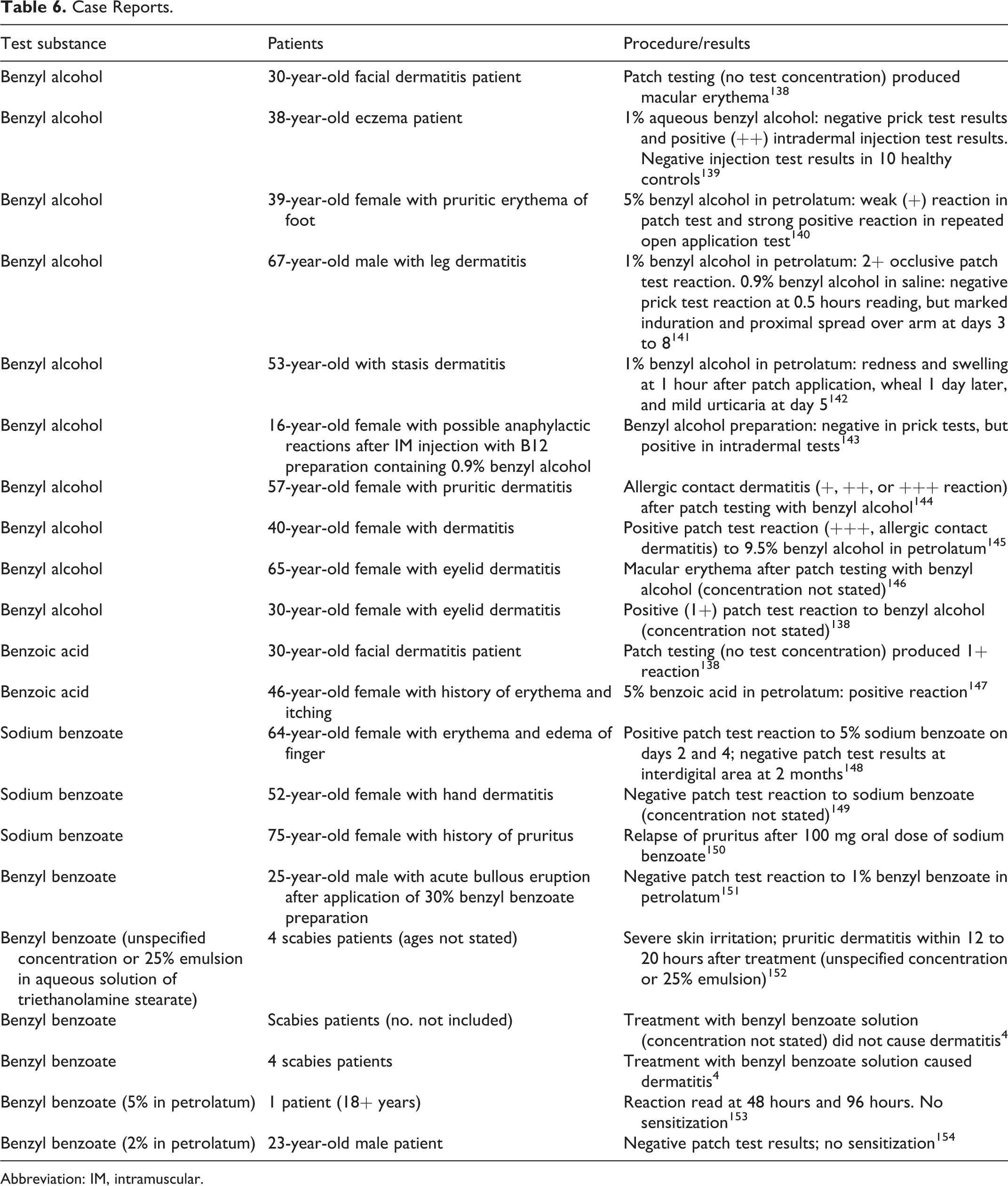
Abbreviation: IM, intramuscular.
Summary
The following cosmetic ingredients are reviewed in this safety assessment: benzyl alcohol, benzoic acid, sodium benzoate, calcium benzoate, magnesium benzoate, potassium benzoate, and benzyl benzoate. Most of these ingredients function as fragrance ingredients/preservatives in cosmetic products. Together, data reported to the FDA VCRP in 2010 and the results of a 2010 industry survey performed by the council indicated use of the following ingredients in cosmetics: benzyl alcohol, benzoic acid, sodium benzoate, calcium benzoate, potassium benzoate, and benzyl benzoate. According to this industry survey, ingredient use concentrations have ranged from 0.000001% (sodium benzoate) to 5% in leave-on products (benzoic acid) and 10% in rinse-off products (benzyl alcohol).
Among the methods of manufacture identified are the production of benzyl alcohol via the action of sodium or potassium carbonate on benzyl chloride and the production of benzoic acid via the decarboxylation of phthalic anhydride. Hydroxyl radical generated by the metal-catalyzed reduction of O2 and H2O2 by ascorbic acid can attack benzoic acid to produce benzene under conditions that are prevalent in beverages. Additionally, exposure to UV light and elevated temperature over the shelf life of beverages may result in benzene formation in products containing benzoic and ascorbic acids. The US beverage industry voluntarily reformulated beverages that were found to contain benzene levels at or above the MCL for drinking water established by the US EPA. Limits for heavy metal impurities in food-grade sodium benzoate (as Pb, ≤2 mg/kg) and pharmaceutical-grade potassium benzoate (heavy metals, ≤0.001%) have been established.
Benzyl alcohol is metabolized to benzoic acid via simple oxidation. Benzoic acid and sodium benzoate are rapidly absorbed from the gastrointestinal tract of mammals, conjugated with glycine in the liver, and then excreted as hippuric acid. In in vitro studies, the percutaneous absorption of benzoic acid through guinea pig skin and the percutaneous absorption of benzyl alcohol and benzyl benzoate through human skin was reported. The percutaneous absorption and urinary excretion of benzyl alcohol and benzyl benzoate was observed following dermal application to Rhesus monkeys.
Benzoic acid (up to 1,000 µg/kg, SC injection) did not induce any reproducible evidence of estrogenic activity in uterotrophic assays involving immature female Alpk:APfSD rats and female Alpk:APfCD-1 mice. However, benzyl benzoate induced estrogenic responses in cultured human breast cancer cells in vitro. At 3,000,000-fold molar excess, benzyl benzoate was able to partially displace [3H]estradiol from recombinant human estrogen receptors ERα and ERβ and from cytosolic ER of MCF7 cells. The antiparasitic activity of benzyl benzoate has been demonstrated in vitro.
Neither acute (up to 12 mg/L aerosol/dust) nor repeated inhalation (up to 1,000 mg/m3) exposures to benzyl alcohol or benzoic acid caused death in rats. Results of the repeated dose inhalation toxicity study also indicated no test substance–related macroscopic or microscopic findings for either test substance. Overall, the results of acute oral and dermal toxicity studies indicated a low level of toxicity for benzyl alcohol, benzoic acid, and benzyl benzoate. The same was true for repeated dose oral toxicity studies on benzoic acid and its salts and benzyl alcohol. Systemic/dermal effects induced by benzyl benzoate were observed in rats receiving repeated dermal doses up to 2,000 mg/kg and in rabbits receiving repeated dermal doses up to 4.0 mL/kg. However, test substance–related toxic signs were not observed in other species that received repeated doses of benzyl benzoate.
Developmental effects only were observed in the presence of marked maternal toxicity in rats fed sodium benzoate in the diet (≥ 2,800 mg/kg/day; NOAEL = 1,400 mg/kg/day). Testicular atrophy was observed in rabbits that received repeated dermal doses of benzyl benzoate (>0.5 g/kg/day). However, overall, benzyl alcohol, benzoic acid, and sodium benzoate were not classified as reproductive/developmental toxicants in oral and dermal animal studies. There was no evidence of adverse effects on pregnancy outcome due to topical application of benzyl benzoate (25%) lotion in pregnant women.
Mixed/equivocal results were apparent in in vitro assays, but negative results were reported in in vivo genotoxicity assays on benzyl alcohol, benzyl benzoate, benzoic acid, and its sodium and potassium salt. The photogenotoxicity of benzoic acid (0.5%) and sodium benzoate (0.5%) has been demonstrated in vitro using bacterial cell suspensions. Benzyl alcohol was negative for carcinogenicity when dermally tested on mice at 2.0% in a nonoxidative hair dye. National Toxicology Program considered it negative for carcinogenicity following 2 years of oral dosing in rats (up to 400 mg/kg) and mice (up to 200 mg/kg), but the EPA considered the 3 of 48 incidence of adrenal cortex adenoma (male mice) to be equivocal evidence of carcinogenic activity rather than negative. Following dermal application to mice, a nonoxidative hair dye containing 2.0% benzyl alcohol and 0.016% benzoic acid was negative for carcinogenicity. Sodium benzoate was negative for carcinogenicity when administered orally at up to 2% to rats (in feed for up to 2 years) or mice (in a lifetime drinking water study).
Undiluted benzyl alcohol and benzoic acid were slightly irritating, and benzyl benzoate was nonirritating to the skin at concentrations up to 50% in animal studies. Undiluted benzyl benzoate was a nonirritant in human patients. In animal studies, benzyl benzoate induced sensitization reactions ranging from none to moderate. Moderate sensitization was observed at a concentration of 10% benzyl benzoate, but not at lower concentrations, and there was 1 report of mild sensitization at a concentration of 40%. Benzyl benzoate was classified as nonsensitizer when tested at a concentration of 30% in healthy human patients. Most of the skin sensitization studies on benzyl benzoate involved groups of patients, and mixed results regarding sensitization potential were reported.
A slightly positive reaction was observed in a phototoxicity study involving hairless mice tested with an unspecified concentration of benzyl benzoate (four 24-hour exposures). Photo irritation was not observed in guinea pigs tested with benzyl benzoate at concentrations up to 30%. Moderate photohemolytic activity in human erythrocytes was observed in the presence of benzyl benzoate, but significant photohemolytic activity was not associated with sodium benzoate or benzyl alcohol in these cells.
Benzyl alcohol (4% aqueous), benzoic acid (undiluted), and its sodium salt (concentration not stated) were irritating to the eyes of rabbits, and it was expected that potassium benzoate would be irritating as well. Benzyl benzoate (undiluted) was irritating to the eyes of rabbits and humans.
Discussion
The CIR Panel noted gaps in the available safety data for some of the ingredients in this safety assessment. The available data on many of the ingredients are sufficient; however, structure activity relationships and toxicokinetics suggest that the available data can be extrapolated to support the safety of the entire group.
Previously, the absence of inhalation toxicity data prompted a finding that the available data were insufficient to support the safety of these ingredients in cosmetic products that may have resulted in inhalation, for example, spray products. However, the council provided a 4-week inhalation toxicity study (rats) of aerosolized benzyl alcohol and benzoic acid. After reviewing this study, the Panel determined that these data, in conjunction with data available in the original safety assessment, are sufficient to support the safety of the ingredient named in this safety assessment.
The Panel did note the formation of adenomas in the adrenal cortex of male B6C3F1 mice receiving a high dose of benzyl alcohol in an oral carcinogenicity study. The low rate of occurrence was within historical control values for this benign tumor. The Panel acknowledged that EPA reviewed the results of this study and concluded that the results suggest equivocal evidence of carcinogenicity, but it was the Panel’s view that there was no evidence of carcinogenicity in the animals.
Data from model studies, pertaining to the beverage industry, reviewed by the Panel raised the possibility that benzene could be formed from benzoic acid and benzoates in cosmetics. The yields of benzene, however, that could be generated by photodegradation are sufficiently low that they do not constitute a relevant hazard.
In its previous safety assessment of benzyl alcohol, benzoic acid, and sodium benzoate, the Panel established a 5% safe concentration limit for benzyl alcohol, benzoic acid, and sodium benzoate and a 10% safe concentration limit for benzyl alcohol in hair dyes. On review of the current practices of use and concentration, it was clear that industry has adhered to those limits. Accordingly, reference to present practices of use and concentration is sufficient to assure safety.
Conclusion
The CIR Panel concluded that the following cosmetic ingredients are safe in the present practices of use and concentration described in this safety assessment: benzyl alcohol benzoic acid sodium benzoate calcium benzoate magnesium benzoate potassium benzoate benzyl benzoate
Magnesium benzoate is not in current use. Where this ingredient to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in the group. This conclusion supersedes the conclusion published in 2001.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L St, NW, Suite 1200, Washington, DC 20036, USA.
Authors’ Contribution
Johnson contributed to conception and design; contributed to acquisition, analysis, and interpretation; and drafted manuscript. Andersen, Bergfeld, Belsito, Hill, Klaassen, Liebler, Marks, Shank, Slaga, and Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.