Abstract
Immunohistochemical staining for the lysosome-associated membrane protein 2 (LAMP-2) has been proposed previously as an alternative to electron microscopy to identify hepatic phospholipidosis. This study used LAMP-2 immunohistochemistry (IHC) to diagnose phospholipidosis in rats exhibiting renal tubular injury. Rats were administered toreforant, a histamine H4 receptor antagonist by oral gavage at a dose of 3, 10, or 100 mg/kg/d for 6 months. Hematoxylin and eosin staining revealed renal tubular epithelial cell vacuolation, hypertrophy, degeneration, and luminal dilation in the 100 mg/kg/d group animals. Renal tubular injury was confirmed using kidney injury marker 1 (KIM-1) IHC. The involvement of phosopholipidosis in the renal injury was investigated by LAMP-2. Adipophilin IHC was included to differentiate phospholipidosis from lipidosis. Increased LAMP-2 staining was observed in the 100 mg/kg/d group animals when compared to vehicle group animals. Lysosome-associated membrane protein-2 staining was most prominent in the outer stripe of the outer medulla where KIM-1 staining was also most prominent. By contrast, adipophilin staining was not increased. Phospholipidosis was also confirmed by electron microscopy. These data support the use of LAMP-2 IHC as a diagnostic tool and suggest an association between phospholipidosis and the renal tubular injury caused by toreforant.
Introduction
Drug-induced phospholipidosis is a phospholipid storage condition characterized by excess accumulation of membranous lamellar structures in cells. 1 –3 Over 200 marketed drugs and drug candidates have been shown to induce phospholipidosis in animal studies. The functional consequence of phospholipidosis and its association with toxicity are poorly understood. There are notable examples of toxicity associated with phospholipidosis including renal toxicity by aminoglycosides. A causal relationship between phospholipidosis and toxicity, however, has not been established, and a protective role for phospholipidosis has been proposed involving clearance of drug and lipid-drug complexes. 1 –3
Phospholipidosis appears as cytoplasmic vacuoles in many cell types by light microscopy. Transmission electron microscopy (TEM) has historically been used to confirm phospholipidosis through ultrastructural demonstration of concentric lamellar bodies. Transmission electron microscopy, however, is limited by instrument accessibility and low throughput. Immunohistochemistry (IHC) has been proposed as an alternative approach to identify the cytoplasmic phospholipids that accumulate with phospholipidosis. 4 Immunohistochemical staining for the lysosome-associated membrane protein 2 (LAMP-2) and adipophilin was shown to identify hepatic phospholipidosis while differentiating from lipidosis. Application of IHC to diagnose phospholipidosis in other tissues has not been reported previously.
Toreforant is a histamine H4 receptor antagonist that has been evaluated for the treatment of various inflammatory diseases. 5 Its chemical structure exhibits cationic amphiphilic drug (CAD)-like properties that have been associated with phospholipidosis, and an in vitro fluorescence assay using a human monocyte cell line also indicated its potential for phospholipidosis. The compound has been evaluated in various toxicity studies up to 6 months duration in rats and 9 months duration in monkeys. Since renal changes including vacuolization were observed in the rat 6-month study, this investigation examined the kidney for phospholipidosis using IHC and TEM. The relationship between phospholipidosis and toxicity was investigated by comparing LAMP-2 immunostaining with labeling for kidney injury marker 1 (KIM-1).
Methods
Animals
All in-life procedures were conducted in compliance with the Animal Welfare Act and US Food and Drug Administration Good Laboratory Practices Regulations for Nonclinical Laboratory Studies. The protocol was reviewed and approved by the test facility Institutional Animal Care and Use Committee. Male and female Sprague-Dawley rats, approximately 10 weeks old at study initiation, were obtained from Charles River Laboratories. Standard procedures and conditions for animal care, feeding, and maintenance of room, caging, and environment were applied. Toreforant was formulated as suspensions in 0.5% methylcellulose and administered to rats (0, 3, 10, 100 mg/kg/d; 20/sex/group) by oral gavage for 6 months. Animals were evaluated for mortality, clinical observations, body weights, food consumption, clinical pathology, and toxicokinetics. Anatomic pathology was evaluated following necropsy.
Immunohistochemistry
Tissue sections were stained with hematoxylin and eosin (H&E) and examined using light microscopy. Because renal changes were observed only in the 100 mg/kg/d group, IHC was performed only for the vehicle and 100 mg/kg/d groups. Paraffin-embedded blocks of kidney from subsets of 10 male and 10 female rats in vehicle and 100 mg/kg/d groups were sectioned for LAMP-2, adipophilin, and KIM-1 IHC. Serial sections (4 μm) were obtained from paraffin-embedded tissue, mounted on charged slides, air-dried, heated at 60°C for 1 hour, and loaded onto an autostainer (DAKO, Carpinteria, CA). Slides underwent deparaffinization and heat-induced epitope retrieval (citrate buffer pH 6.0). For LAMP-2 IHC, sections were labeled with rabbit anti-LAMP-2 (Igp96; Cat# 51-2200, Invitrogen, Waltham, MA) at 1:800 concentration followed by donkey antirabbit biotinylated (Cat# AP182B, Chemicon International, Inc, Temecula, CA) secondary antibody at 1:2000 concentration. Normal rabbit immunoglobulin G (IgG; Cat# SC-2027, Santa Cruz Biotechnology, Dallas, TX) was used as a negative control, and normal rat liver (fasted) was used as a positive control. For adipophilin IHC, sections were labeled with mouse monoclonal to adipophilin (Cat# 610102, Progen, Heidelberg, Germany) at 1:100 concentration followed by donkey antimouse biotinylated (Cat# AP192B, Chemicon International, Temecula, CA) secondary antibody at 1:2000 concentration. Normal mouse IgG (Cat# MABC004, Chemicon International, Inc, Temecula, CA) was used as a negative control, and normal rat liver (fasted) was used as a positive control. For KIM-1 IHC (kidney injury molecule-1, sections were labeled with goat antirat TIM-1/KIM-1/HAVCA (Cat# AF3689, R&D systems, Minneapolis, MN) at 1:50 concentration followed by donkey antigoat biotinylated (Cat#AP180B, Chemicon International, Inc, Temecula, CA) secondary antibody at 1:2000 concentration. Normal goat IgG (Cat#SC-2028, Santa Cruz Biotechnology, Dallas, TX) was used as a negative control, and kidney from cisplatin-treated rat was used as a positive control. The immunoreactivity was visualized by ABC reagent (Cat# PK6100, Vector, Burlingame, CA) and diaminobenzidine (Cat# K3468, DAKO, Carpinteria, CA) followed by counterstaining with Mayer hematoxylin.
Measurement of IHC Labeling and Statistical Analysis
Qualitative image analysis was conducted for all the IHC labeling. Grading of the IHC labeling was blinded. The grading for LAMP-2 IHC labeling was as follows: 1=slight LAMP-2 labeling in the proximal convoluted tubules (PCTs); 2=mild LAMP-2 tubular cell labeling; 3=moderate LAMP-2 tubular cell labeling; 4=marked LAMP-2 tubular cell labeling including the cortex (Cx) and renal papilla (RP). The grading for adipophilin IHC labeling was as follows: 1=slight (1% to 25%) adipophilin membrane labeling in the PCT and 2=mild (25%-50%) adipophilin membrane labeling in the PCT. The grading for KIM-1 IHC labeling was as follows: 0=<5% tubular cells labeled; 1=5% to 25% tubular cells labeled; 2=25% to 50% tubular cells labeled; 3=50% to 75% tubular cells labeled; and 4=>75% tubular cells labeled and labeling in the Cx and RP included. Mann-Whitney U test was used for statistical analysis.
Electron Microscopy
Electron microscopy was conducted using kidney from 1 male and 1 female rat from the vehicle control and 100 mg/kg/d groups. Approximately, 1 mm 3 of formalin-fixed tissue were trimmed and transferred to 10% neutral-buffered formalin fixative. The samples were washed in buffer (0.1 mol/L sodium phosphate buffer, pH 7.2), postfixed in 1% osmium tetroxide in the same buffer, dehydrated in an ethanolic series culminating in acetone, and infiltrated with Spurr epoxide resin. Ultrathin (80-90 nm thick) sections were cut from each trimmed block for which an ultrathin area of interest was designated and placed on 200-mesh copper grids before being stained with uranyl acetate and lead citrate. The stained thin sections were examined by TEM at the Laboratory for Advanced Electron and Light Optical Methods at North Carolina State University, Raleigh, North Carolina.
Results
Kidney Pathology
There were no changes observed in renal parameters for the 3 and 10 mg/kg/d groups at necropsy (Table 1). In the 100 mg/kg/d group, blood urea nitrogen (BUN) was increased 1.6× and 1.3× relative to the vehicle group for males and females, respectively. Serum creatinine was increased 2.0× for males and 1.5× for females relative to the vehicle group. Kidney weights also were increased relative to those of vehicle control animals.
Renal Parameters in Rat 6-Month Study at Necropsy.
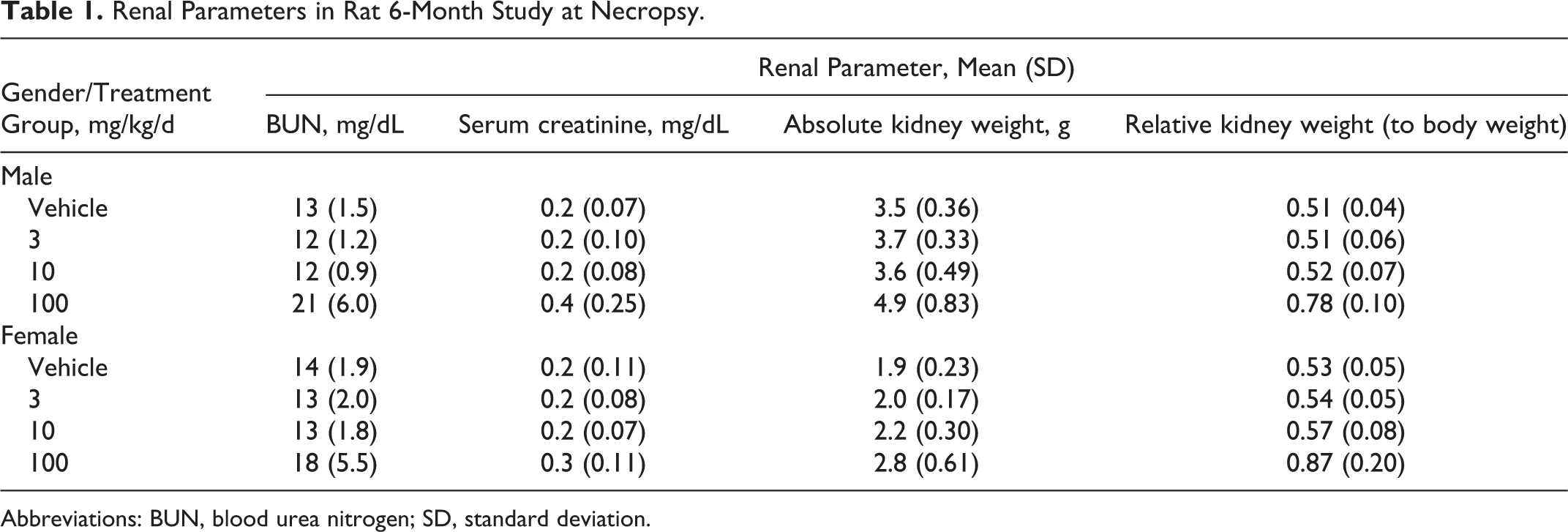
Abbreviation: BUN, blood urea nitrogen; SD, standard deviation.
There were no histomorphologic changes in H&E-stained kidney sections from the vehicle, 3, or 10 mg/kg/d group animals. Among the 100 mg/kg/d group animals, tubular epithelial cell vacuolation, hypertrophy, degeneration, and luminal dilation were observed in the outer stripe of the outer medulla (OSOM). The severity of these changes ranged from slight to marked. The affected tubules were large/dilated and lined by variably vacuolated/hypertrophic cells with abundant pale/foamy cytoplasm (Figure 1).
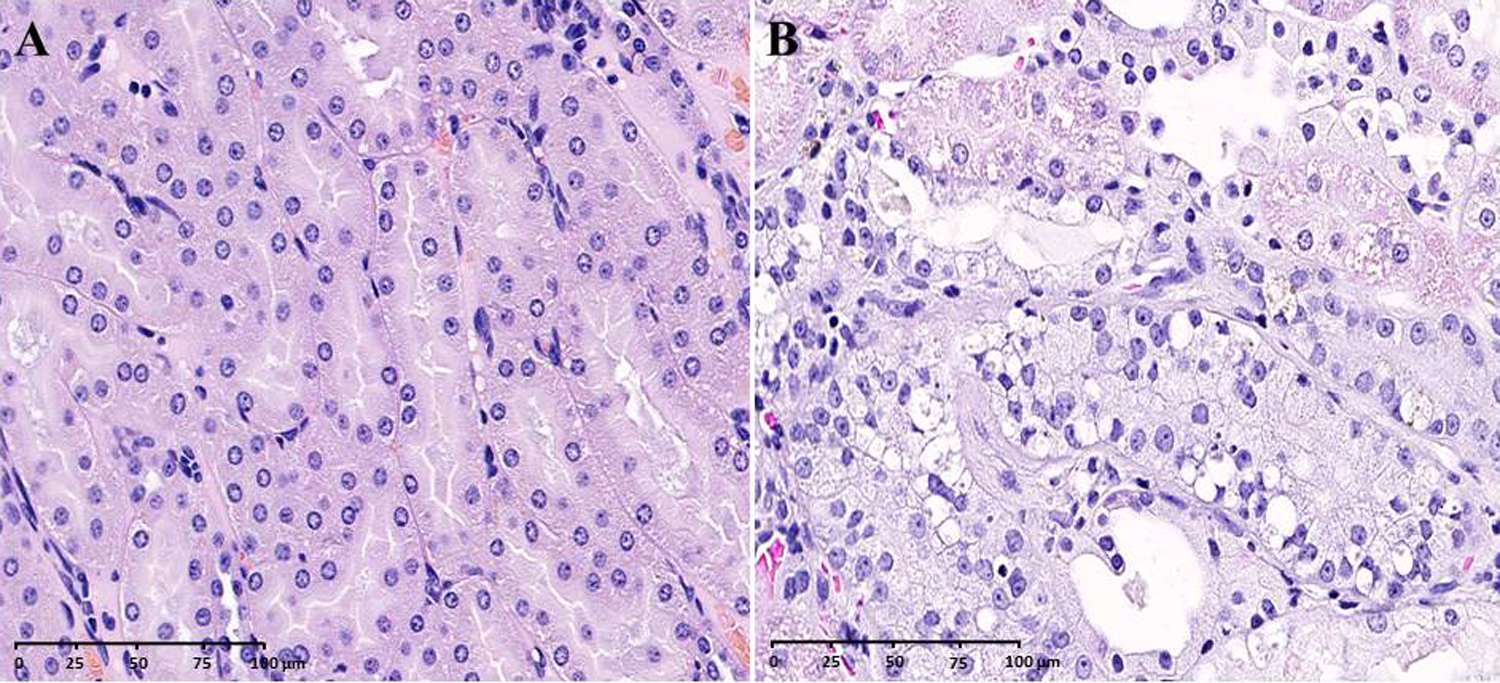
Kidney sections from (A) vehicle and (B) 100 mg/kg/d group male rats (hematoxylin and eosin [H&E] ×40). Renal tubules in the 100 mg/kg/d group were dilated and lined by vacuolated and hypertrophic cells with abundant pale, foamy cytoplasm.
Electron Microscopy
In kidney of the 100 mg/kg/d group animals, electron microscopy revealed the presence of concentric lamellar inclusions within lysosomes, which are characteristic of phospholipidosis (Figure 2). The location of these changes correlated with the vacuolation/hypertrophy in the OSOM observed by light microscopy.
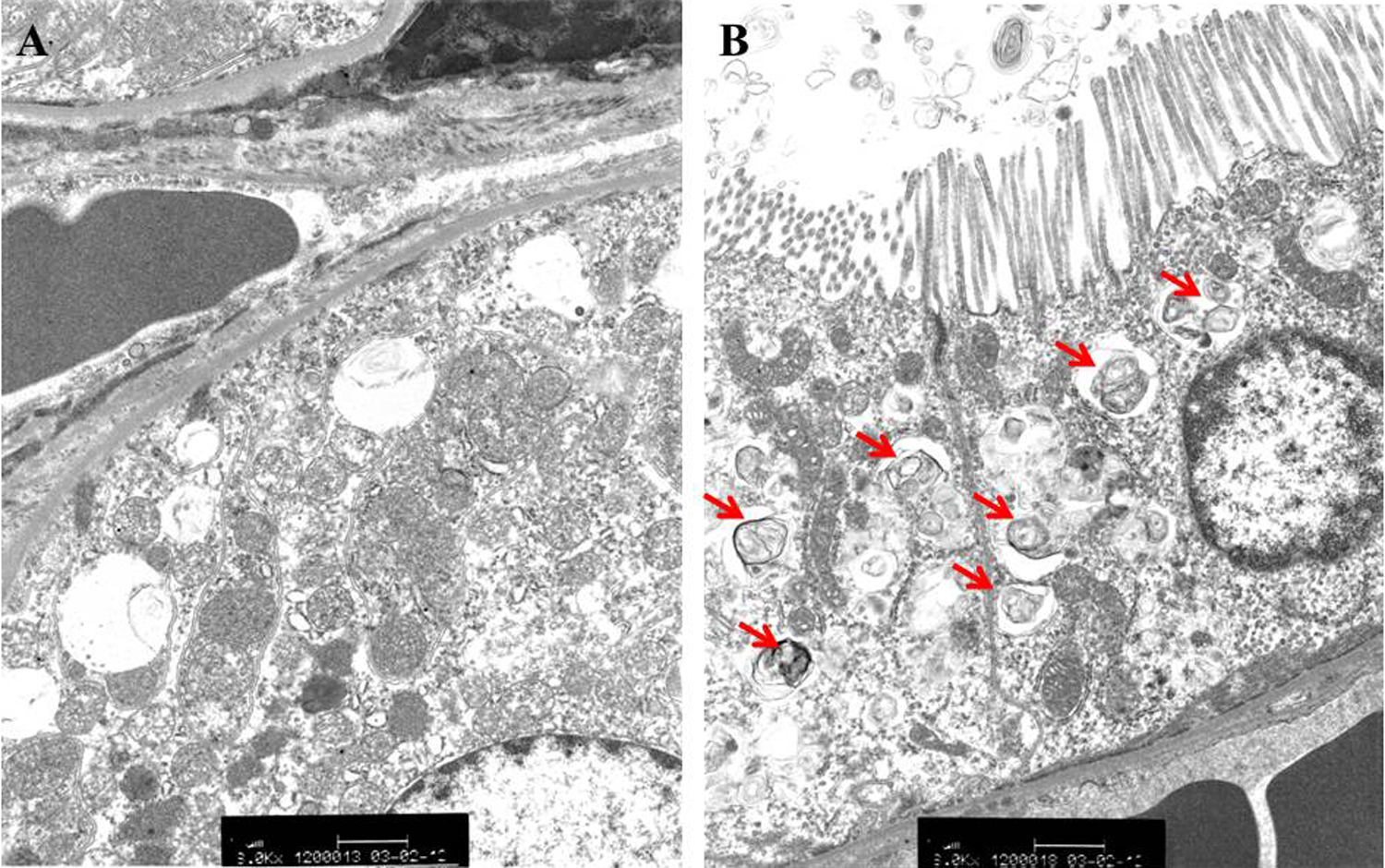
Transmission electron microscopy photomicrographs of kidney from (A) vehicle and (B) 100 mg/kg/d group male rats revealed lamellar bodies marked with red arrows in the latter.
Lysosome-Associated Membrane Protein 2 IHC
Punctate cytoplasmic LAMP-2 IHC labeling was observed in the PCTs in the male and female vehicle group rats. Tubular epithelial cells in the OSOM in rats from the 100 mg/kg/d group showed intense LAMP-2 labeling of the lysosomal membranes surrounding the phospholipid accumulations (Figure 3). LAMP-2 IHC scoring of vehicle and test article group rats is shown in Figure 4. There was a statistically significant increase in LAMP-2 labeling mainly in the OSOM tubular epithelial cells in the 100 mg/kg/d group rats when compared to the vehicle group rats (P < 0.001). Markedly increased LAMP-2 labeling in the Cx, OSOM, and RP in the 100 mg/kg/d group rats correlated with the presence of hypertrophic/vacuolated/epithelial cells with abundant pale/foamy cytoplasm from H&E stain (Figure 5).
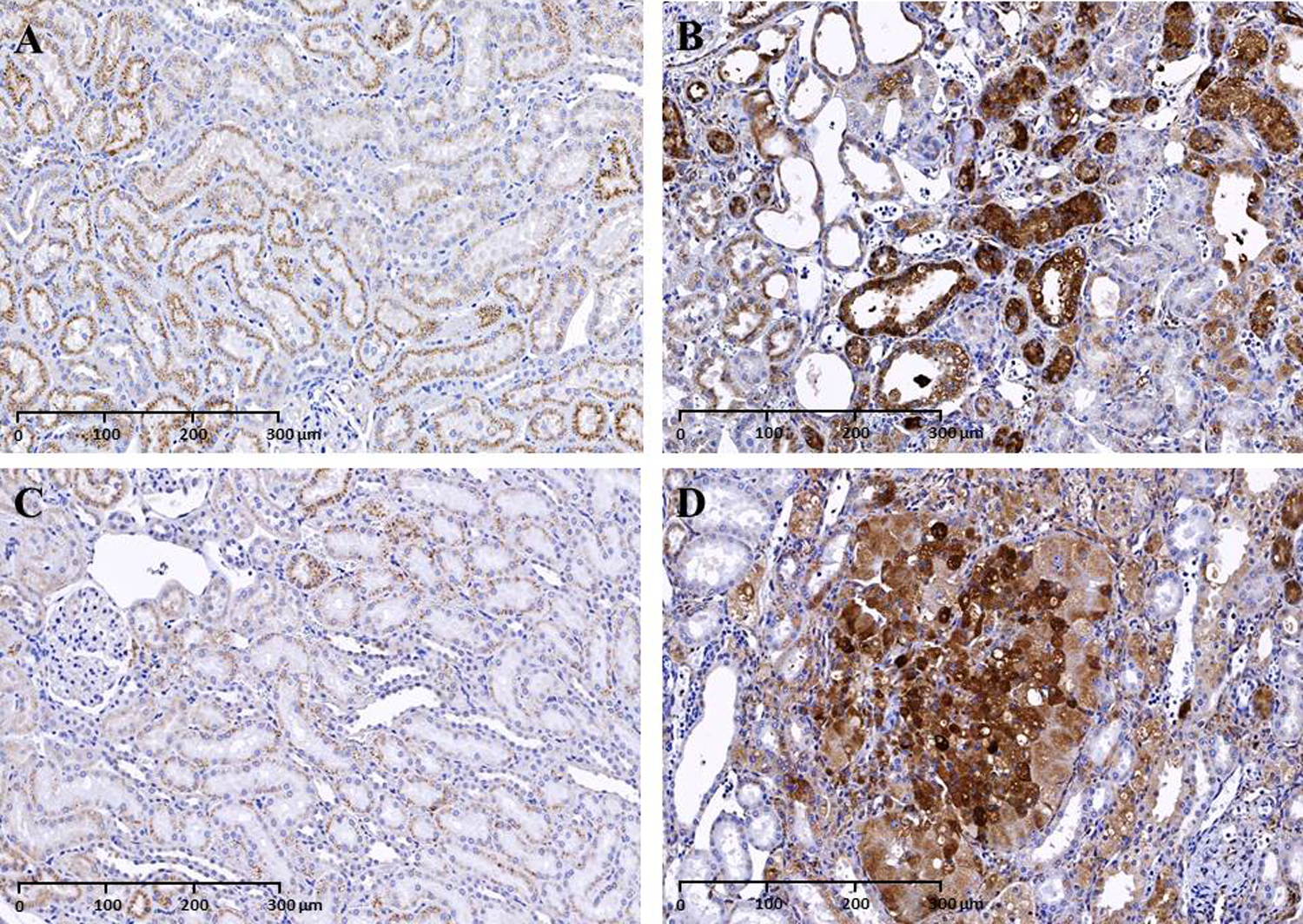
Lysosome-associated membrane protein 2 immunohistochemistry (LAMP-2 IHC), 20×: Punctate cytoplasmic LAMP-2 labeling was observed in the proximal convoluted tubules in the outer stripe of the outer medulla (OSOM) of (A) vehicle male and (C) female rat. Intense LAMP-2 labeling of the lysosomal membranes surrounding the cytoplasmic phospholipid accumulations in the OSOM of the (B) 100 mg/kg/d group male and (D) female rat.
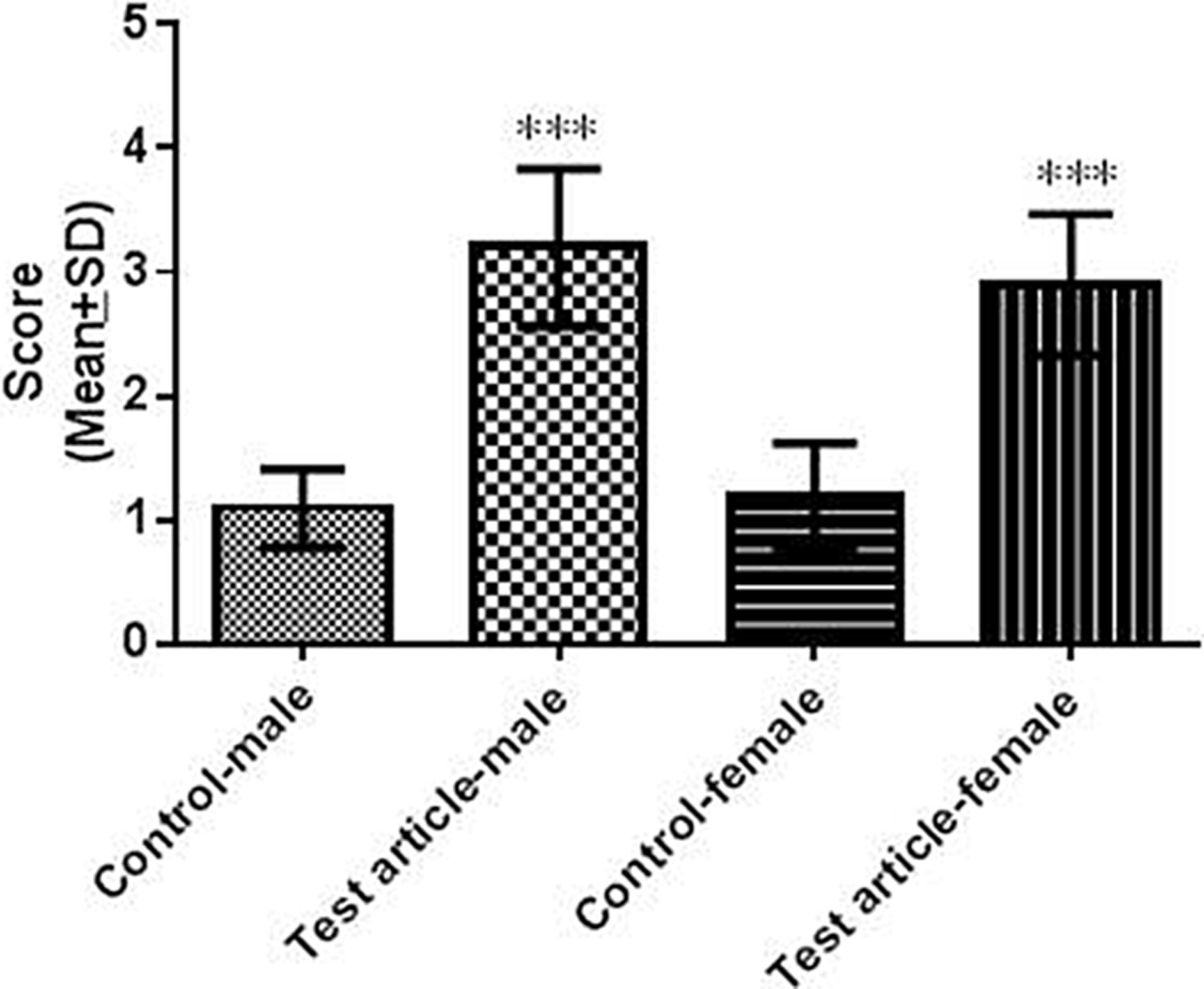
Summary of lysosome-associated membrane protein-2 immunohistochemistry (LAMP-2 IHC) scores from vehicle and 100 mg/kg/d group rats (mean ± standard deviation of 10 rats per group). Statistically significant differences are indicated by ***P < 0.001.
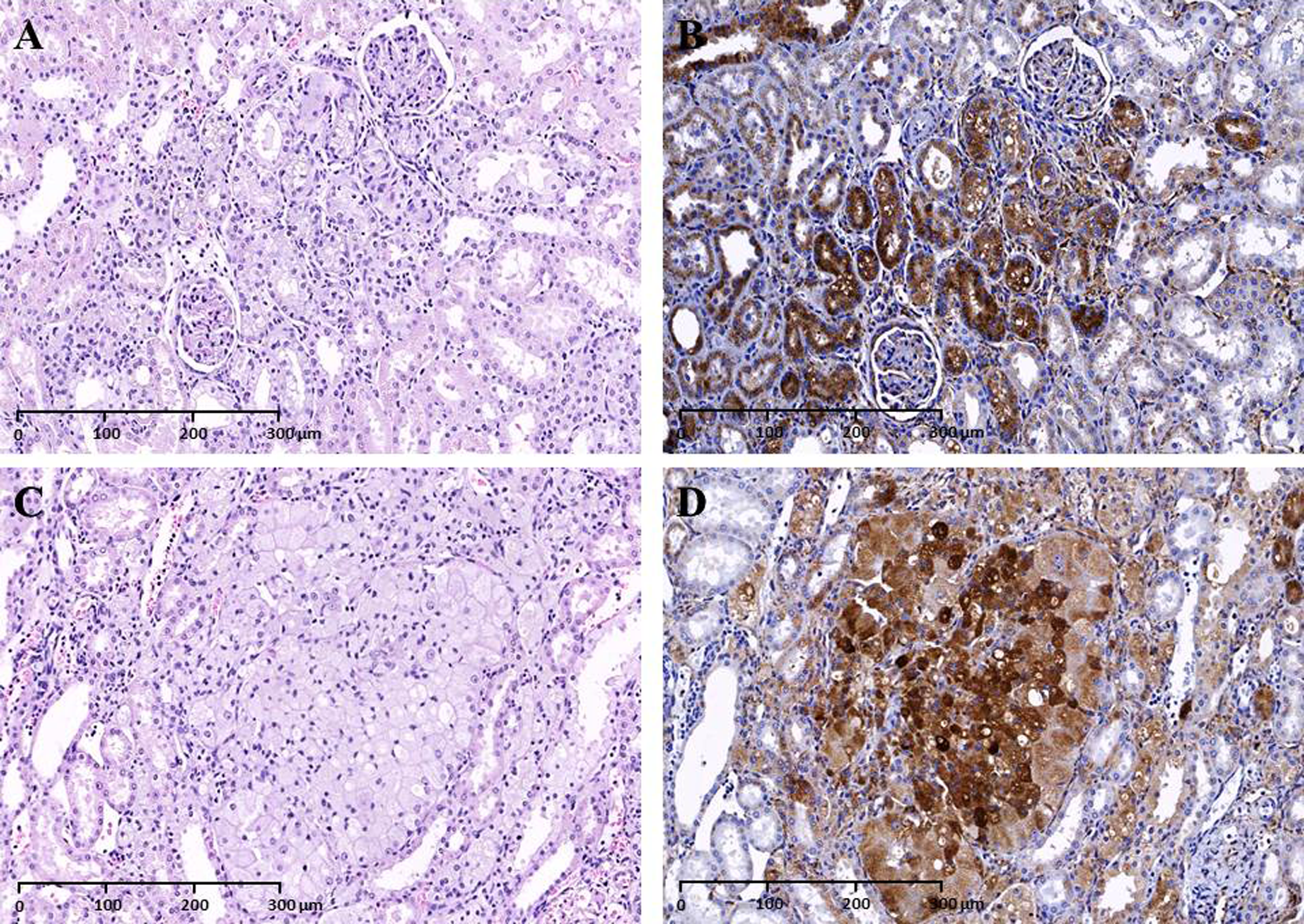
Hematoxylin and eosin (H&E) and lysosome-associated membrane protein-2 immunohistochemistry (LAMP-2 IHC), 20×: H&E staining revealed hypertrophic, vacuolated, epithelial cells with abundant pale, foamy cytoplasm in the (A) 100 mg/kg/d group male and (C) female rat; positive LAMP-2 labelling in adjacent sections of (B) test article group male and (D) female rat.
Adipophilin IHC
Prominent adipophilin IHC labeling of the tubular membrane surrounding the cytoplasmic lipid vacuoles in the PCT was observed in vehicle group rats. The lysosomal membranes surrounding the cytoplasmic phospholipid accumulations within the tubular epithelial cells from the 100 mg/kg/d group rats did not stain for adipophilin (Figure 6). Adipophilin IHC scoring from vehicle and test article group rats is summarized in Figure 7. There was a slight decrease in adipophilin labeling in the OSOM in the 100 mg/kg/d group rats when compared to the vehicle group rats. Hypertrophic, vacuolated epithelial cells with abundant foamy cytoplasm from H&E stain did not stain for adipophilin.
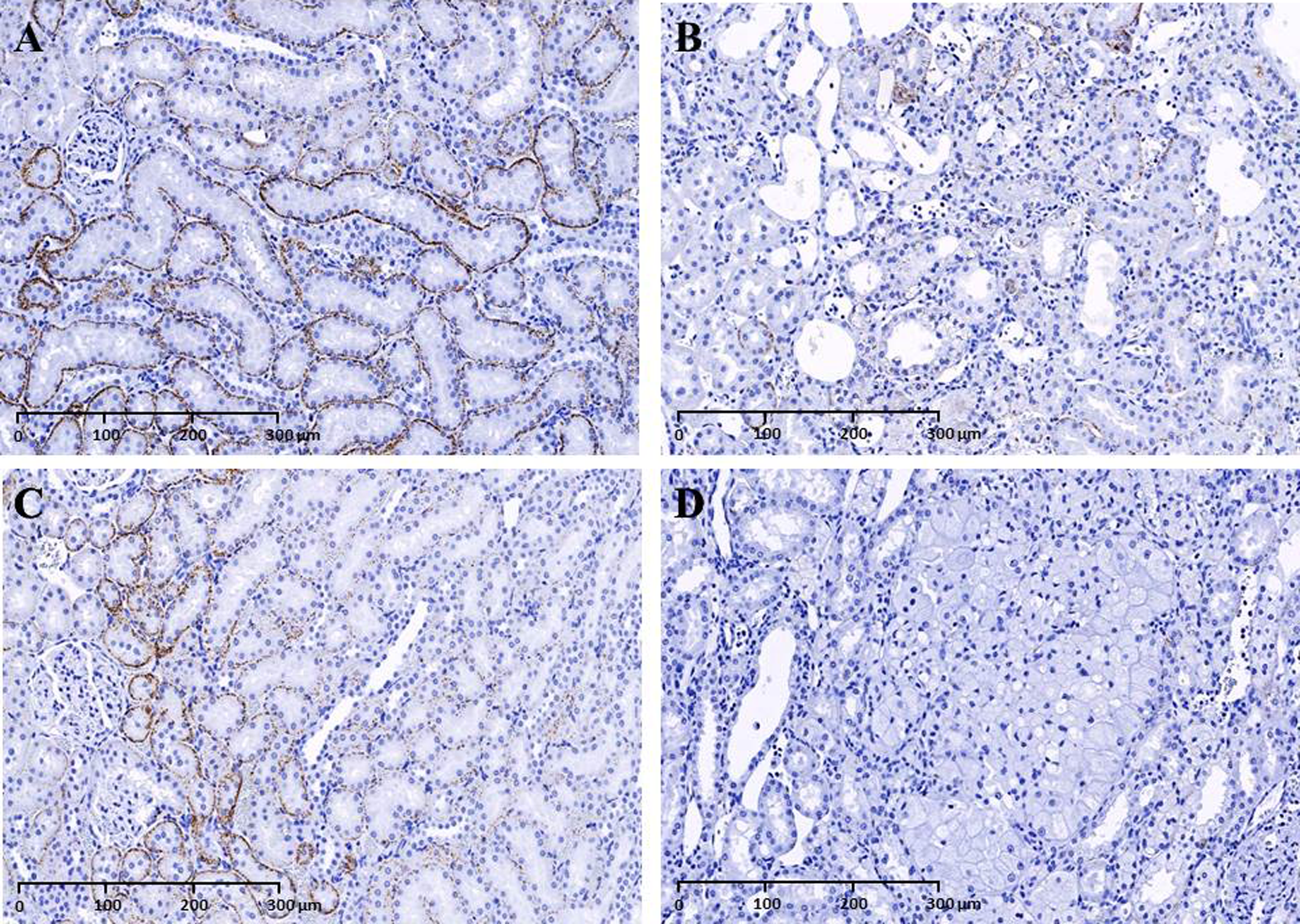
Adipophilin immunohistochemistry (IHC), 20×: decreased adipophilin labeling in the outer stripe of the outer medulla (OSOM) tubular cells in (B) test article treated male and (D) female rat compared to prominent adipophilin labeling in (A) vehicle group male and (C) female rat.
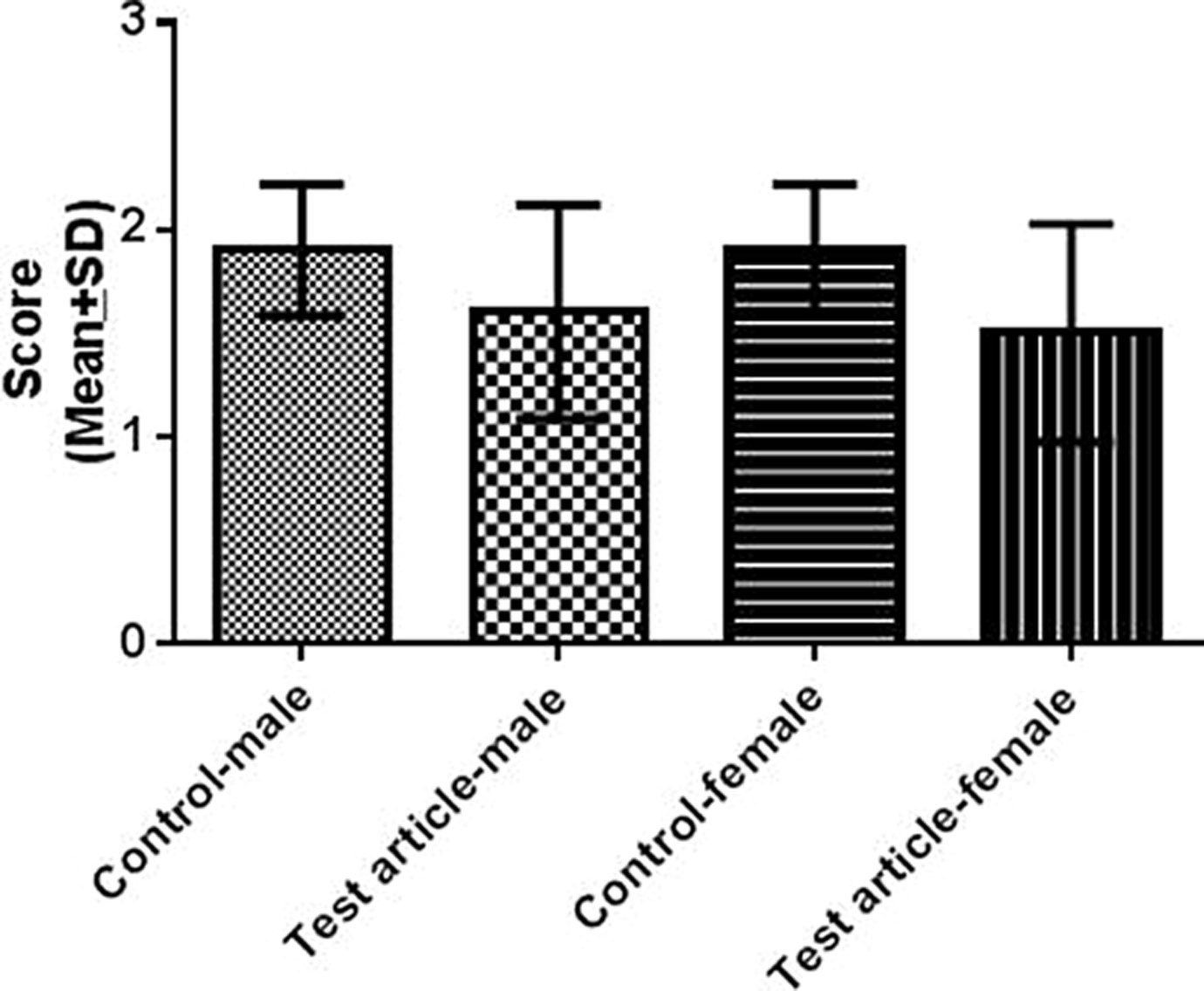
Summary of adipophilin immunohistochemistry (IHC) scores from vehicle and 100 mg/kg/d group rats (mean ± standard deviation [SD] of 10 rats per group).
Kidney injury marker 1 IHC
Kidney injury marker 1 IHC findings from vehicle and test article group rats are summarized in Figure 8. There was a statistically significant increase in KIM-1 staining in the test article group when compared to the vehicle group, P < 0.001. There was no KIM-1 labeling detected in tubular epithelial cells in vehicle group male rat and weak background labeling in vehicle group female rat. There was strong KIM-1 labeling detected in the OSOM tubular epithelial cells in the 100 mg/kg/d group rats (Figure 9). Increased KIM-1 labeling was also detected in the Cx and RP in several animals in the 100 mg/kg/d group. The distribution of KIM-1 and LAMP-2 labeling, when compared using adjacent sections, coincided (Figure 10).
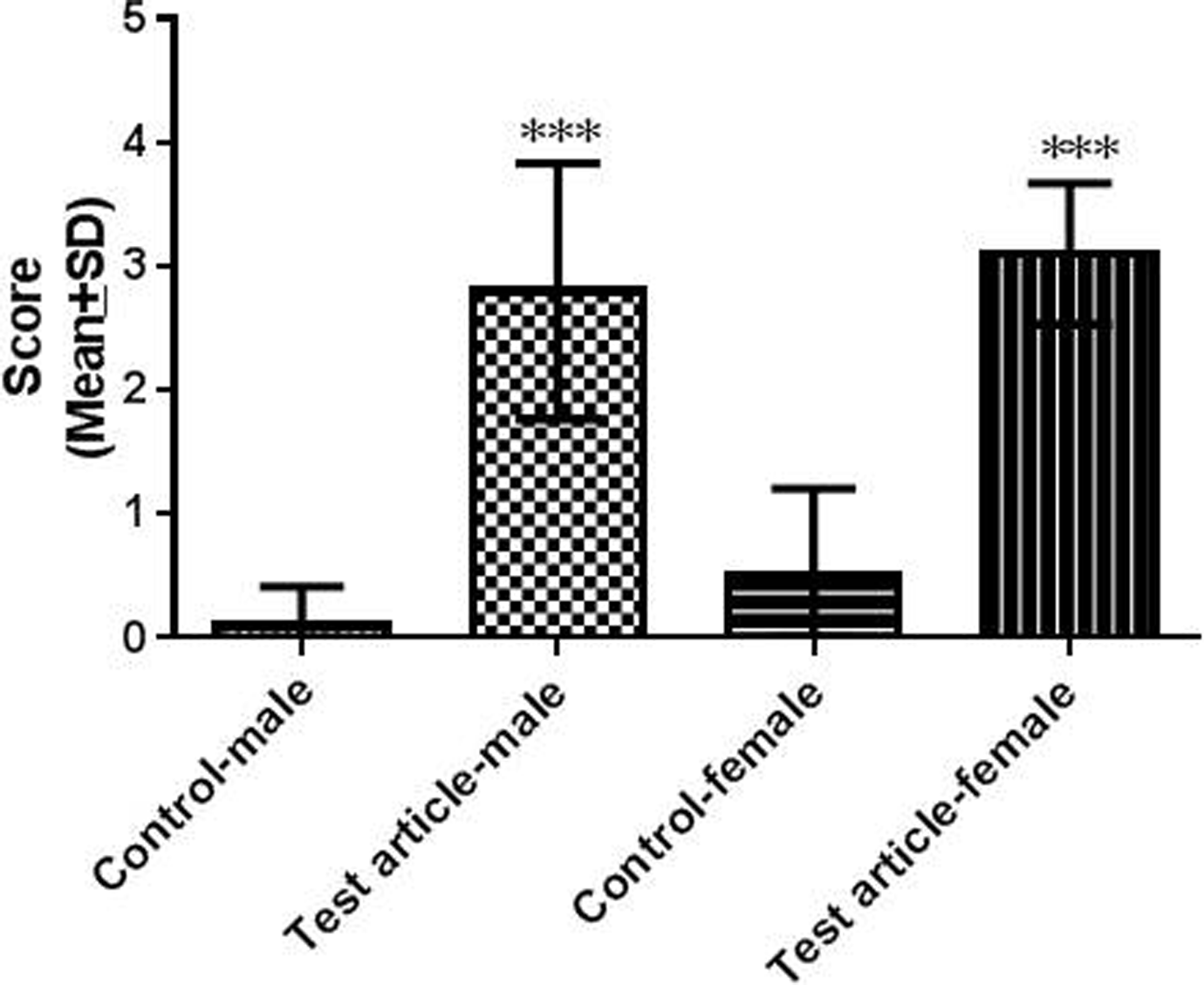
Summary of kidney injury marker 1 immunohistochemistry (KIM-1 IHC) scores from vehicle and 100 mg/kg/d group rats (mean ± standard deviation of 10 rats per group). Statistically significant differences are indicated by ***P < 0.001.
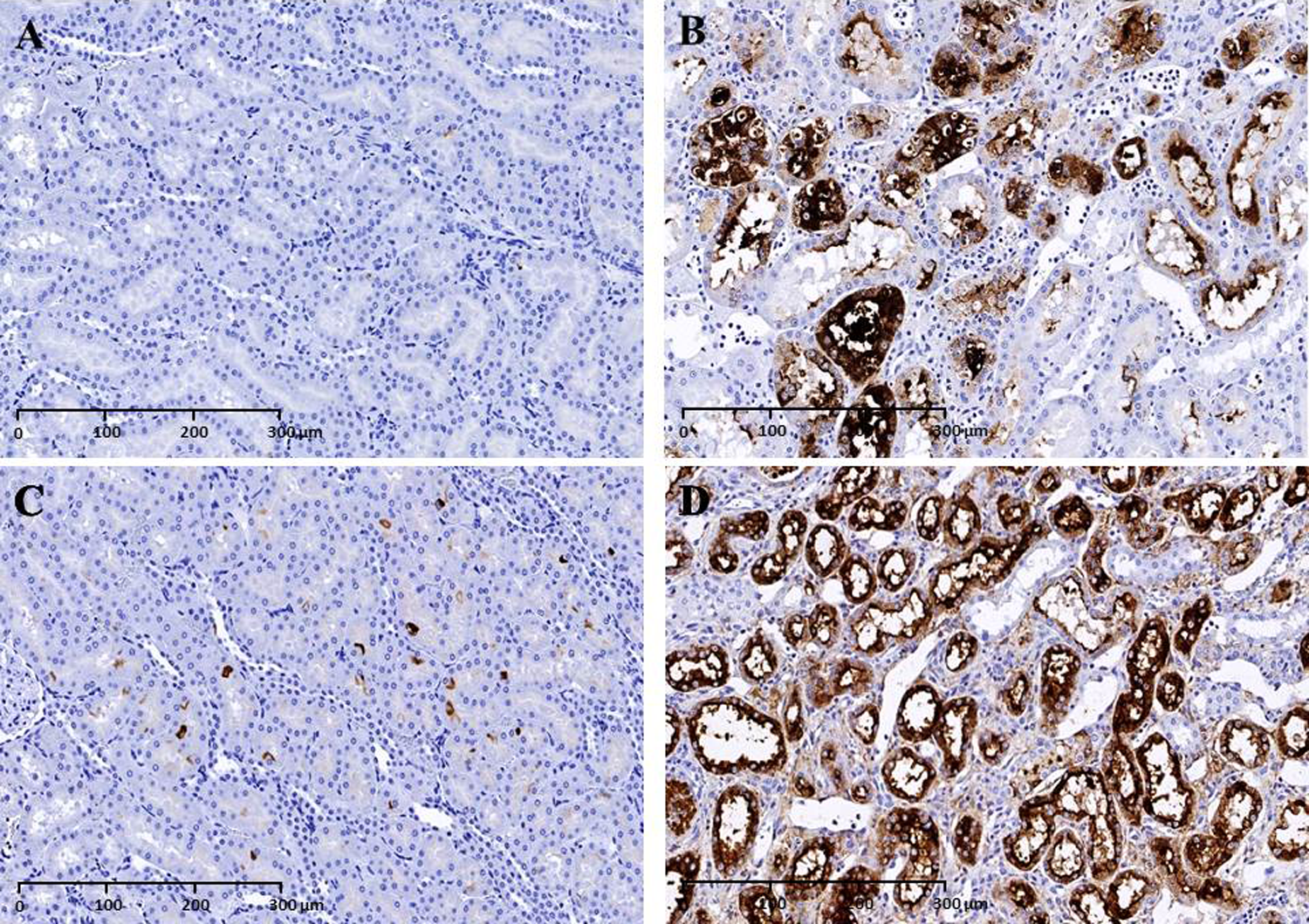
Kidney injury marker 1 immunohistochemistry (KIM-1 IHC), 20×: No KIM-1 labeling was observed in the outer stripe of the outer medulla (OSOM) tubular epithelial cells in (A) vehicle group male and slight background labeling in tubular epithelial cells in (C) vehicle group female. Intense KIM-1 labeling was detected in the OSOM tubular epithelial cells in both the (B) test article group male and (D) female rat.
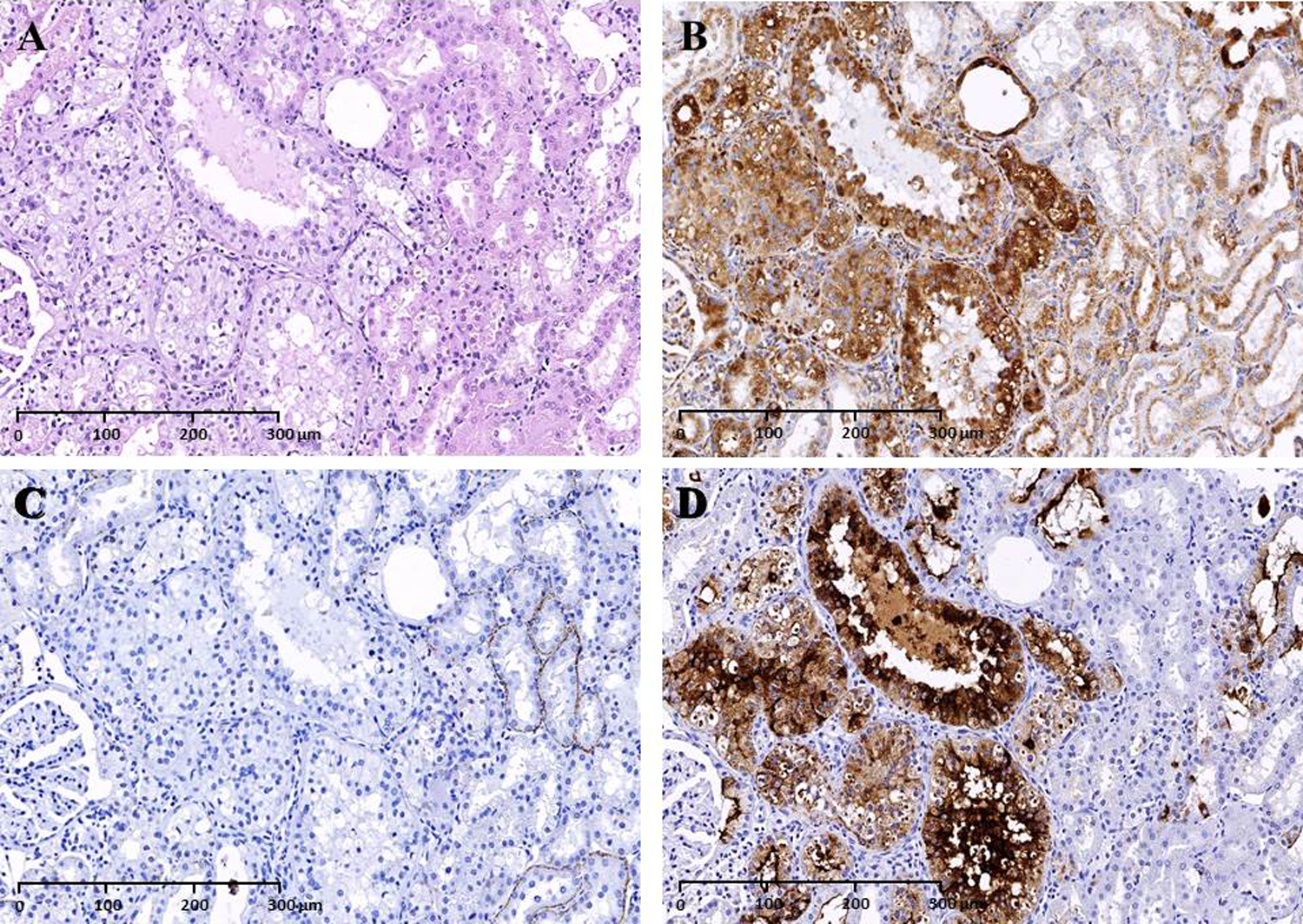
Hematoxylin and eosin (H&E), lysosome-associated membrane protein 2 (LAMP-2), adipophilin and KIM-1 IHC, 20×: Serial kidney sections of the 100 mg/kg/d group male rat, H&E revealed (A) hypertrophic/vacuolated epithelial cells with phospholipid accumulations which labelled positively with LAMP-2 (B) but not labelled with adipophilin (C). Tubular epithelial cells with phospholipid accumulations that were labelled with LAMP-2 also stained positively for KIM-1 (D).
Discussion
This study represents the first reported use of LAMP-2 and KIM-1 IHC to compare phospholipidosis and toxicity at the cellular level. Renal phospholipidosis and toxicity were observed in the rat 6-month toxicity study of toreforant. The 100 mg/kg/d group had various renal changes including slight to marked tubular epithelial vacuolation, hypertrophy, degeneration, and luminal dilation in the OSOM. Kidney weights were also increased. The renal changes correlated with increased BUN (30% to 60%) and serum creatinine (50% to 100%). There was no evidence of renal changes in the 3 or 10 mg/kg/d groups, and thus further evaluation by TEM and IHC were limited to the vehicle and 100 mg/kg/d groups.
Early characterization of toreforant suggested potential for phospholipidosis based on its CAD-like properties and accumulation of phospholipids in a predictive in vitro assay. 6 Foamy macrophages were observed in various organs in 1 and 3-month toxicity studies in rats and monkeys (data not shown). In the rat 6-month toxicity study, renal tubular vacuolation was observed and confirmed as phospholipdosis using TEM. Immunohistochemistry evaluation showed that vacuoles stained with LAMP-2 but not with adipophilin to differentiate phospholipidosis from lipid vacuoles. According to Obert et al, 4 the IHC method to identify phospholipidosis has advantages over other approaches such as 1 the ability to use formalin-fixed, paraffin-embedded tissues, 2 greater sensitivity than histochemical staining with Sudan Black or Oil Red-O, and 3 greater availability and ease than ultrastructural evaluation. Immunohistochemistry also enables comparison of multiple markers in the same or adjacent sections.
Renal toxicity in the 100 mg/kg/d group was confirmed using KIM-1 IHC. Kidney injury marker 1 is a transmembrane protein located in proximal tubular cells, and the ectodomain that is shed from the outside of the cells is considered to be an early marker of injury and of regeneration/repair. 7 –9 Kidney injury marker 1 is generally not detected in normal renal tissue but is highly expressed in dedifferentiated proximal tubular epithelial cells in human and rodent kidneys after ischemic or toxic injury. The hypertrophic and vacuolated tubular epithelial cells in the 100 mg/kg/d group were positively labeled by KIM-1 IHC indicating cellular injury. In adjacent sections, the hypertrophic and vacuolated tubular epithelial cells also were labeled by LAMP-2 IHC suggesting an association between phospholipidosis and renal tubular injury. Colocalized KIM-1 and LAMP-2 immunostaining, however, does not necessarily indicate a causal relationship between phospholipidosis and toxicity. Cells can sequester phospholipids and lamellar bodies in lysosomes without injury. 1 –3
Although the majority of compounds that induce phospholipidosis have not been associated with toxicity, there are some notable exceptions including amiodarone and aminoglycoside antibiotics. Amiodarone, an antiarrhythmic drug, has been shown to cause interstitial pneumonitis and pulmonary interstitial fibrosis in patients. 10,11 Some investigations suggested a link between the induction of amiodarone-induced phospholipidosis in alveolar macrophages and pulmonary toxicity, but a causal relationship was not established. Other data suggest that phospholipidosis is not involved in amiodarone-induced pulmonary toxicity. Phospholipidosis has been reported in patients without pulmonary toxicity, and amiodarone-induced phospholipidosis in rodents is not associated with fibrosis or significant functional alterations. 12 Sun et al 13 reported that the accumulation of phospholipids precedes a decrease in cell function that precedes cell death. These results could be interpreted that phospholipidosis is not directly associated with toxicity but rather toxicity results when lamellar bodies reach high levels. Other mechanisms such as oxygen-free radicals appear to have a more plausible causative role in amiodarone-induced pulmonary toxicity.
The aminoglycoside, gentamicin, induces phospholipidosis in proximal tubule epithelium and has been associated with renal tubular injury in patients and experimental animals. 14,15 Gentamicin accumulates in lysosomes where it produces membrane destabilization, lysosomal aggregation, alteration in lipid metabolism, and phospholipidosis. The extent of phospholipidosis correlates with toxicity, and inhibition of phospholipidosis using polyaspartic acid inhibits toxicity suggesting a causal relationship between phospholipidosis and toxicity. However, other research indicates that cytosolic concentration of gentamicin rather than accumulation in lysosomes is critical for toxicity. When a critical threshold of lamellar bodies in lysosomes is reached, gentamicin is released from lysosomes into the cytosol. Several different mechanisms may be involved to cause apoptosis or necrosis. Cytosolic gentamicin could act on mitochondria to inhibit respiration and ATP production and induce oxidative stress to activate apoptosis. Gentamicin could also form an oxidant complex with mitochondrial iron capable of inducing cell death. Lysosomes could also rupture to release proteases and cathepsins into the cytosol to cause cell death.
To date, a causal link between phospholipidosis and toxicity has not been demonstrated. 2 The prevailing theory is that phospholipidosis is an adaptive response to the presence of the drug and is not a toxic condition. Its role may be protective by clearing drug and lipid-drug complexes. Observations from the rat 6-month study with toreforant of colocalized KIM-1 and LAMP-2 immunostaining in the renal tubular epithelium are not conclusive on the role of phospholipidosis in renal toxicity.
Toreforant has been evaluated in multiple clinical studies up to 6 months in duration without any reported occurrences of phospholipidosis or renal toxicity. 5 Human exposures in these studies were much less than exposures associated with phospholipidosis and renal injury in rats (data not shown). For example, a Phase 2b rheumatoid arthritis study evaluated doses up to 30 mg/d for 6 months. The area under the curve margin was 4 when the 30 mg/d cohort was compared to the no observed adverse effect level of 10 mg/kg/d in the rat 6-month study. The margin was 50 relative to the 100 mg/kg/d group where renal phospholipidosis and toxicity were observed. Additional safety measures can be incorporated into clinical studies by monitoring for renal injury using noninvasive methods such as serum creatinine, BUN, and urinary KIM-1. Monitoring for phospholipidosis is not common, but bis(monoacylglycerol)phosphate (BMP) has been proposed as a biomarker. 16,17 Urinary BMP and KIM-1 in the rat 6-month study would have provided potential correlative data for renal phospholipidosis and toxicity; however, these assessments were not included because phospholipidosis was diagnosed retrospectively and blood and urine samples were not available for analysis.
In summary, this study demonstrated that a clinical drug development compound, toreforant, induced phospholipidosis and renal tubular injury in rats. Colocalized KIM-1 and LAMP-2 immunostaining indicated an association between phospholipidosis and renal toxicity, but a causal relationship could not be established. Toreforant has a large safety margin, and the ability to monitor for phospholipidosis and renal toxicity will enable its evaluation in human clinical trials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
