Abstract
Conclusion
In the1996 safety assessment of methyldibromo glutaronitrile, the Cosmetic Ingredient Review Expert Panel stated that this ingredient is safe as used in rinse-off products and safe at
Historic and Current Uses and Concentrations of Methyldibromo Glutaronitrile.1,37,3 8
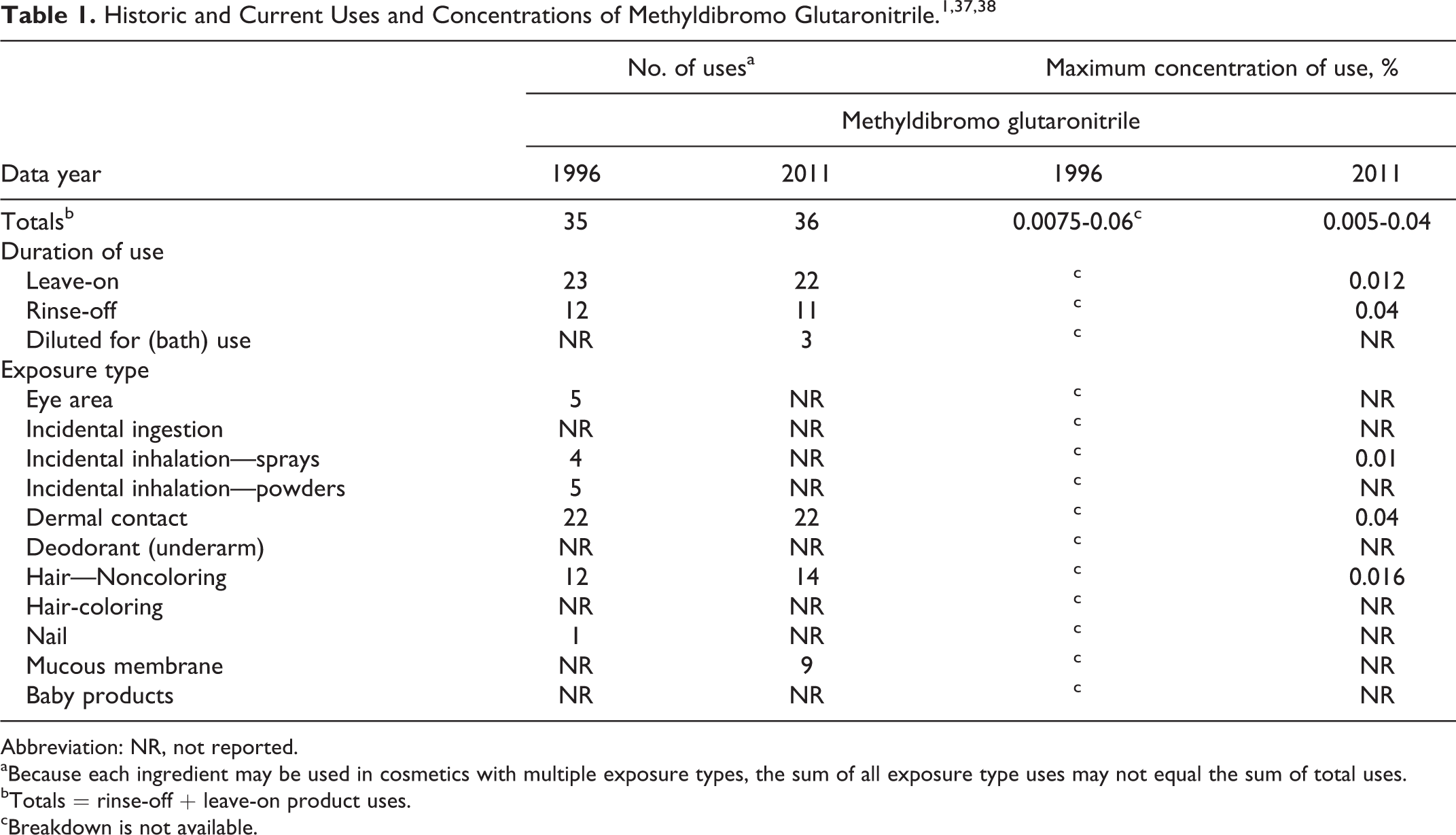
Abbreviation: NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
bTotals = rinse-off + leave-on product uses.
cBreakdown is not available.
Discussion
Information supplied to the US Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Ingredient Reporting Program indicates that in 1996, methyldibromo glutaronitrile was being used in 35 cosmetic formulations at concentrations up to and including 0.06%. Use of this ingredient in 36 product formulations in specific product categories was reported to FDA in 2011, and an industry survey produced use concentrations up to 0.04%. The European Commission had banned the ingredient from use in both leave-on and rinse-off products due to increased reports of sensitivity. However, the Expert Panel opined that many, if not most, reports of sensitization in patch test studies likely are due to testing at high concentrations such that the reactions observed are actually irritation responses.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
