Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel assessed the safety of diethanolamine and its salts as used in cosmetics. Diethanolamine functions as a pH adjuster; the 16 salts included in this rereview reportedly function as surfactants, emulsifying agents, viscosity increasing agents, hair or skin conditioning agents, foam boosters, or antistatic agents. The Panel reviewed available animal and clinical data, as well as information from previous CIR reports. Since data were not available for each individual ingredient, and since the salts dissociate freely in water, the Panel extrapolated from previous reports to support safety. The Panel concluded that diethanolamine and its salts are safe for use when formulated to be nonirritating. These ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
Keywords
Introduction
In 1983, the Cosmetic Ingredient Review (CIR) Expert Panel issued a report on the safety of triethanolamine, diethanolamine, and monoethanolamine. 1 In 2010, the Panel decided to reopen that safety assessment as 3 separate reports and to include additional related ingredients in each of the new reviews. This assessment addresses diethanolamine and its salts. The acid salt ingredients (as recited below) would be expected to dissociate into diethanolamine and the corresponding acid, most of which have been reviewed separately. In most cases, this means that the composition of these salts is stoichiometrically 1 diethanol ammonium cation to 1 acid residue anion.
In the 1983 review, the Expert Panel concluded that diethanolamine, an ingredient that functions in cosmetics as a pH adjuster, is safe for use in cosmetic formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin. 1 Because of the potential for irritation, the concentration of diethanolamine should not exceed 5% in products intended for prolonged contact with the skin. Diethanolamine should not be used with products containing N-nitrosating agents.
In 2009, the Expert Panel reviewed the safety of DEA-dodecylbenzenesulfonate, an ingredient included in this rereview. 2 It was concluded that DEA-dodecylbenzenesulfonate is safe as used when formulated to be nonirritating.
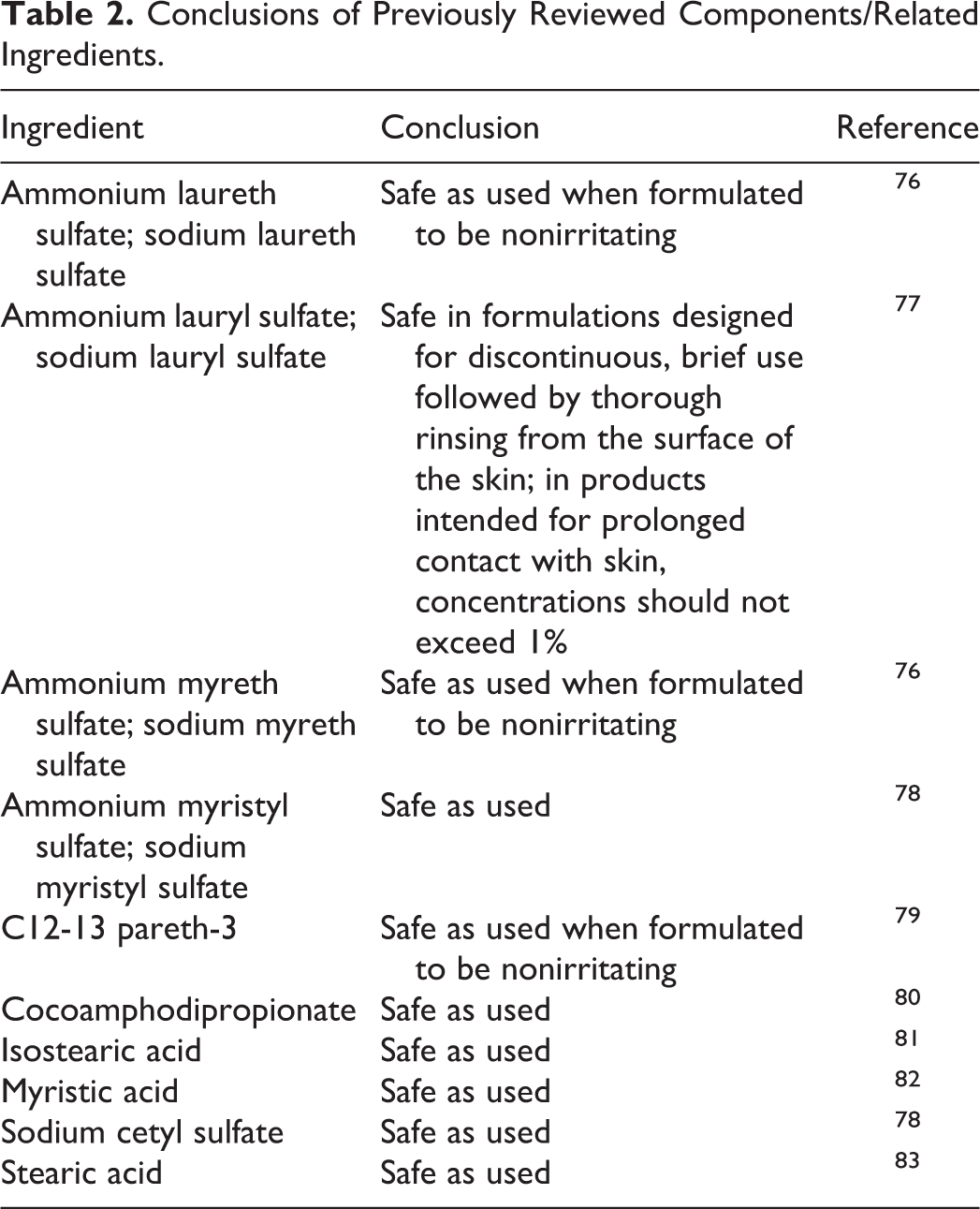
The safety of most of the components of, or ingredients related to, these salts has been reviewed by the CIR. The majority of those components or related ingredients had a conclusion of safety. The following 16 salts are included in the rereview of diethanolamine.
Inorganic Acid Salt
Diethanolamine bisulfate
Organic Acid Salts
DEA-cocoamphodipropionate
DEA-isostearate
DEA-linoleate
DEA-myristate
DEA stearate
Sulfate and Sulfonate Salts
DEA-C12-13 alkyl sulfate
DEA-C12-13 pareth-3 sulfate
DEA-C12-15 alkyl sulfate
DEA-cetyl sulfate
DEA-dodecylbenzenesulfonate
DEA-laureth sulfate
DEA-lauryl sulfate
DEA-methyl myristate sulfonate
DEA-myreth sulfate
DEA-myristyl sulfate
The definition, structure, and function(s) of each ingredient are provided in Table 1. The conclusions for the components/related ingredients that have been reviewed are provided in Table 2.
Definitions, Structures, and Function(s).
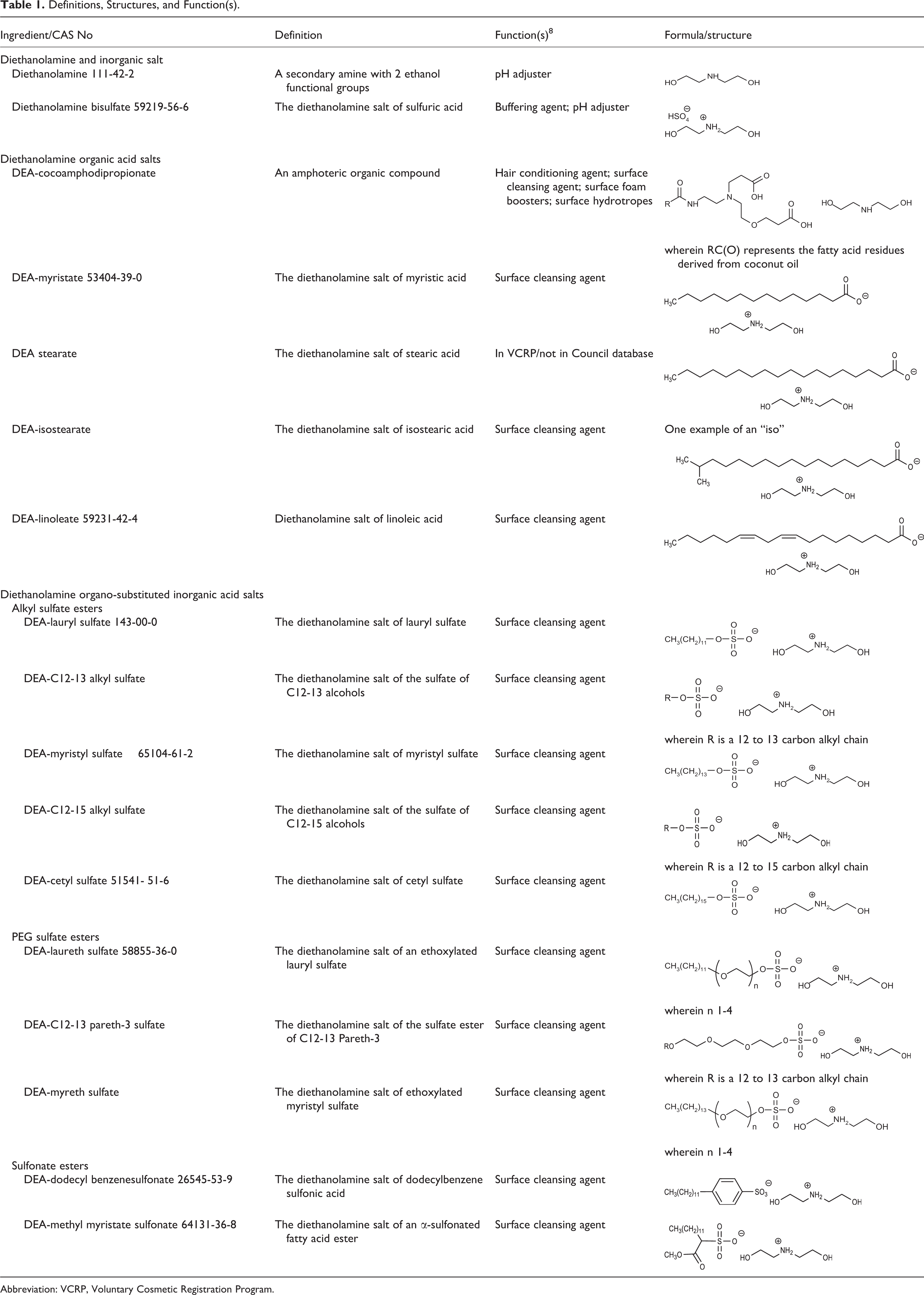
Abbreviation: VCRP, Voluntary Cosmetic Registration Program.
Conclusions of Previously Reviewed Components/Related Ingredients.
Chemistry
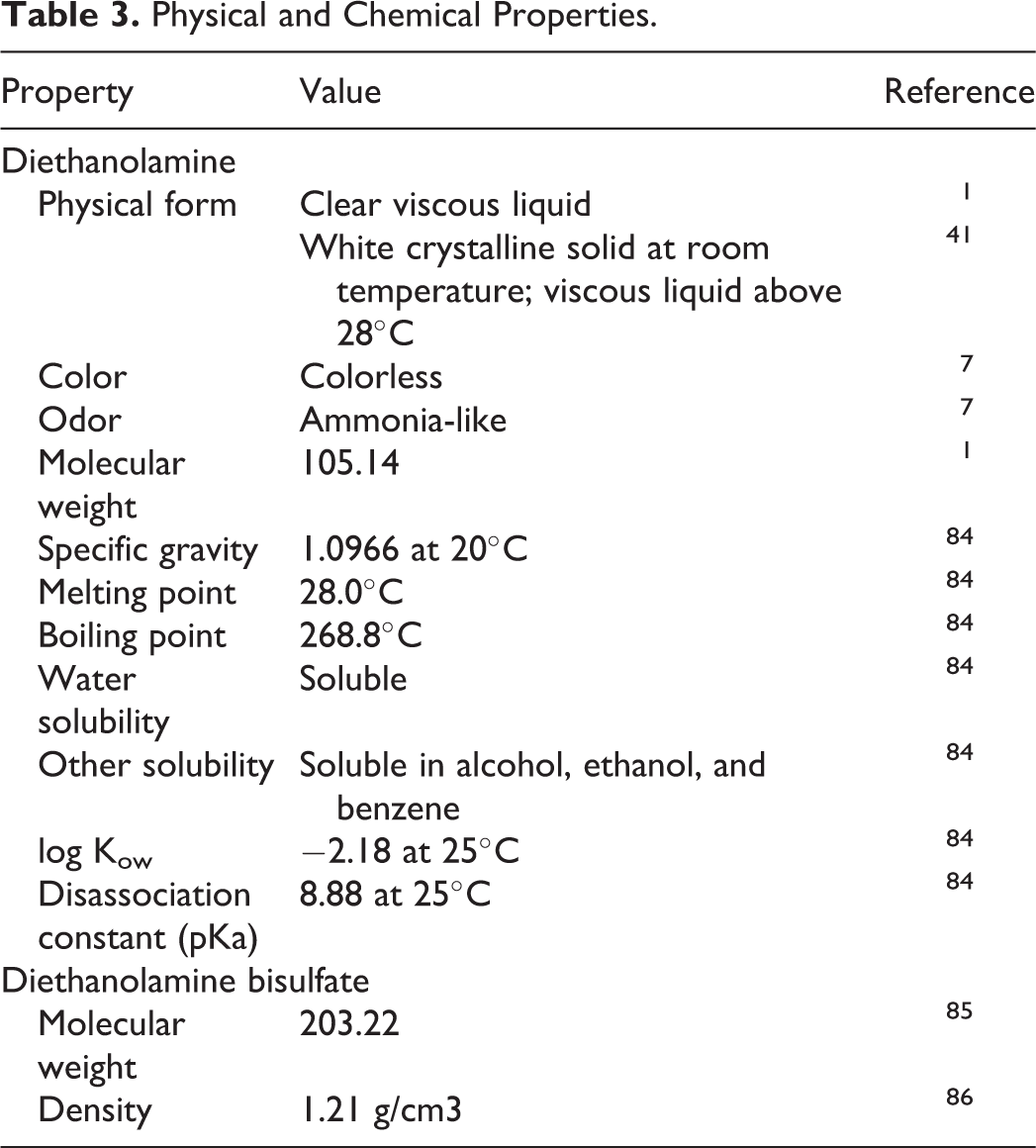
Diethanolamine (Figure 1), presented in a previous report, is an amino alcohol. Diethanolamine is produced commercially by aminating ethylene oxide with ammonia. The replacement of 2 hydrogens of ammonia with ethanol groups produces diethanolamine. Diethanolamine contains small amounts of triethanolamine and ethanolamine.

Diethanolamine.
Diethanolamine is reactive and bifunctional, combining the properties of alcohols and amines. At temperatures of 140°C to 160°C, diethanolamine will react with fatty acids to form ethanolamides. The reaction of ethanolamines and sulfuric acid produces sulfates and, under anhydrous conditions, diethanolamine may react with carbon dioxide to form carbamates. Diethanolamine can act as an antioxidant in the autoxidation of fats of both animal and vegetable origin. 1
Definition and Structure
Of concern in cosmetics is the conversion (nitrosation) of secondary amines (R1-NH-R2), such as diethanolamine (wherein R1 and R2 are each ethanol), into N-nitrosamines that may be carcinogenic. Of the approximately 209 nitrosamines tested, 85% have been shown to produce cancer in laboratory animals. 3 Nitrosation can occur under physiologic conditions. 4 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in the nitrosation of amines in aqueous (aq) solution. 5 Accordingly, diethanolamine and its salts should be formulated to avoid the formation of nitrosamines.
Acid salts
The acid salts (inorganic acid salt, organic acid salts, sulfates, and sulfonate salts), named above, are ion pairs (ie, acid residue anion and diethylammonium cation) which freely dissociate in water (eg, Figure 2). Therefore, these salts are closely related to the corresponding free acids and diethanolamine. In other words, DEA stearate is closely related to stearic acid and diethanolamine. Accordingly, the potential formation of nitrosamines should be a consideration for the ingredients in this group. Chemical and physical properties are provided in Table 3.
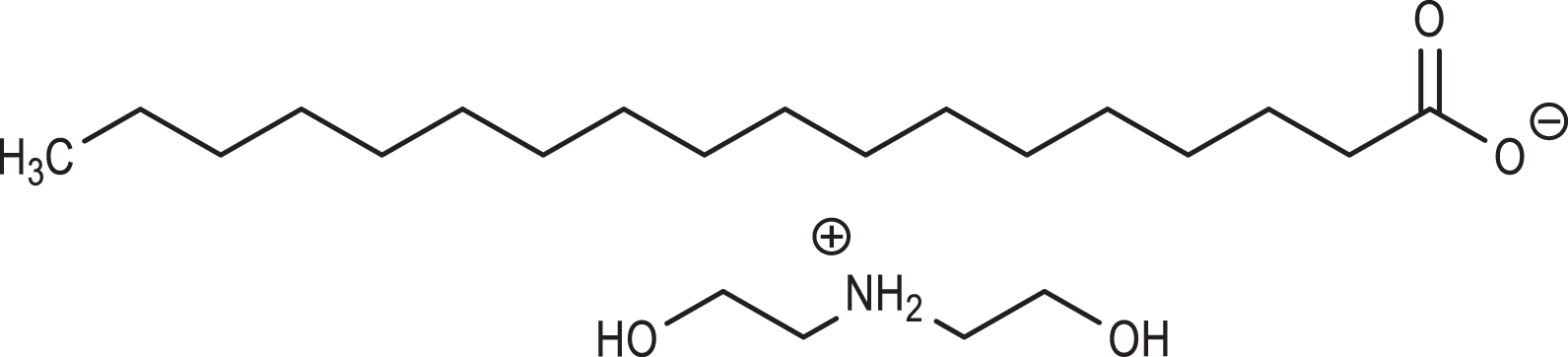
DEA stearate.
Physical and Chemical Properties.
Method of Manufacture
Diethanolamine is produced by reacting 2 moles of ethylene oxide with 1 mole of ammonia. 6 Typically, ethylene oxide is reacted with ammonia in a batch process to produce a crude mixture of approximately one-third each ethanolamine, diethanolamine, and triethanolamine, which are then separated, achieving varying degrees of single component purity.
Impurities
Dow Chemical Company reports that diethanolamine is commercially available with a minimum purity of 99.0%, containing 0.5% maximum ethanolamine and 0.5% maximum triethanolamine. 7
Use
Cosmetic
Diethanolamine functions in cosmetics as a pH adjuster. 8 Although a few of the other ingredients might also function as a pH adjuster, the majority are reported to function as a surfactant, emulsifying agent, viscosity-increasing agent, hair or skin conditioning agent, foam booster, or antistatic agent. Voluntary Cosmetic Registration Program (VCRP) data obtained from the US Food and Drug Administration in 2011 indicate that diethanolamine is used in 22 formulations, 10 are leave-on and 12 are rinse-off formulations. 9 According to data submitted in response to a survey conducted by the Personal Care Products Council (Council), diethanolamine is used at concentrations of 0.0008% to 0.3%. 10 The highest concentration used in leave-on products is 0.06% in moisturizing products. However, diethanolamine itself is usually not added to cosmetic products (Council, personal communication). Diethanolamine is often present in cosmetics because it is contained as an impurity in diethylammonium salt ingredients (eg, the ingredients assessed herein) that are used in the formulation. Based on the diethanolamine specifications for diethanolamine condensates added to cosmetic formulations and the concentration of use of those condensates, the concentration of diethanolamine present in leave-on and rinse-off formulations is reported as 0.13% and 0.64%, respectively. 10
Only 4 salts were reported to be used according to VCRP data, no more than 3 reported uses, and no concentration of use data were reported to the Council for any of the salts. 11 Frequency and concentration of use data for in-use ingredients are provided in Table 4A; ingredients not reported to be in use, according to VCRP data and the Council survey, are listed in Table 4B. 11,12
Frequency and Concentration of Use According to Duration and Type of Exposure.
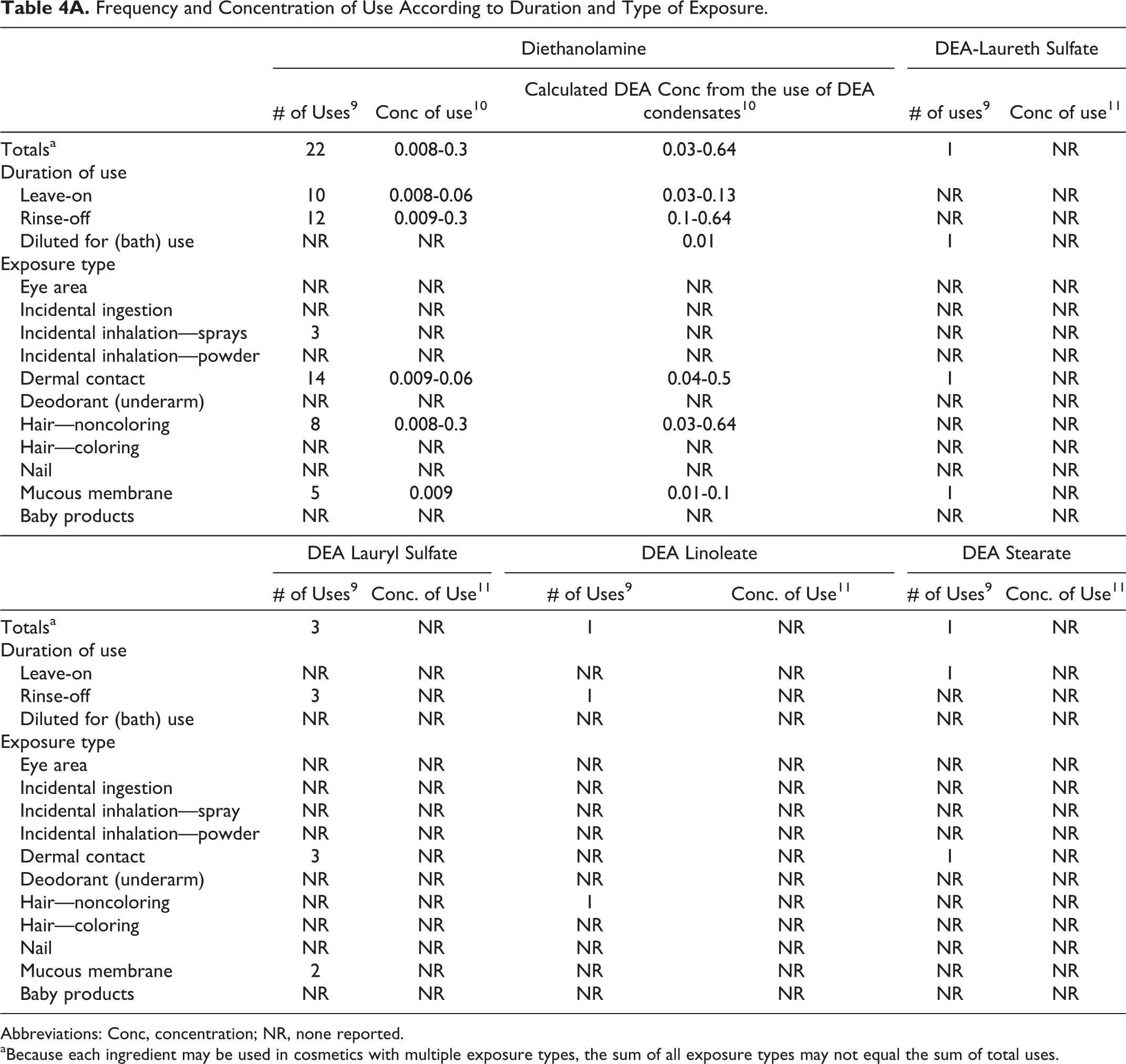
Abbreviations: Conc, concentration; NR, none reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
Ingredients Not Reported to Be in Use.

Diethanolamine is reported to be present in hair sprays. In practice, 95% to 99% of the aerosols released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 µm range. 13,14 Therefore, most aerosols incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable. 15,16 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic diameters in the range considered to be respirable. 16 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Dialkanolamines (eg diethanolamine) and their salts (ie, the ingredients included in this rereview) are on the European Union’s (EU) list of substances which must not form part of the composition of cosmetic products. 17 (Secondary amines are allowed as contaminants in raw materials at concentrations up to 0.5% in trialkylamine, trialkanolamines, and their salts and in monoalkylamines, monoalkanolamines, and their salts and at up to 5% in fatty acid dialkylamides and dialkanolamides; secondary amines are allowed at up to 0.5% in finished products containing dialkylamides and dialkanolamides.) The opinion paper issued by the Committee for Cosmetic Products and Non-Food Products stated that amines occur only in their salt form in all cosmetic products. 18 The reasoning is that all amines are alkaline compounds which are always neutralized by an acid component to produce their salts, and there is concern about the potential for nitrosamine formation; in principle, secondary amines are potential precursors of nitrosamines.
In Canada, diethanolamine is completely prohibited as per the Cosmetic Hotlist; Health Canada prohibits the use of dialkanolamines (eg, diethanolamine). 19 This prohibition is based on the EU prohibition in the Cosmetics Regulation, Annex II. The use of diethanolamine in product formulations in Canada is being investigated due to the reported use at concentrations of up to 3% (Health Canada, personal communication).
Noncosmetic
Diethanolamine, in a previous report, is described as being used in the manufacture of emulsifiers and dispersing agents for textile specialties, agricultural chemicals, waxes, mineral and vegetable oils, paraffin, polishes, cutting oils, petroleum demulsifiers, and cement additives. It is an intermediate for resins, plasticizers, and rubber chemicals. Diethanolamine is used as a lubricant in the textile industry, a humectant and softening agent for hides, as an alkalizing agent and surfactant in pharmaceuticals, as an absorbent for acid gases, and in organic syntheses. 1 Diethanolamine is approved for use as an indirect food additive (21CFR175.105, 21CFR176.170, 21CFR176.180, 21CFR176.210). 20
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
In vitro
The in vitro absorption of [14C]diethanolamine in an oil-in-water emulsion was determined using fuzzy rat skin. 21 Approximately 2 mg/cm2 was applied to the skin in each diffusion cell; the solution was applied for 24 hours. A total of 1.4% of the applied dose was absorbed over 24 hours, with 4% remaining in the skin. (There was 1.9% of the applied dose remaining in the stratum corneum and 1.9% in the viable dermis/epidermis.) The researchers indicated that little of the diethanolamine that was found in the skin at 24 hours was absorbed into the receptor fluid. Similar results were reported at 72 hours.
Three full-thickness skin preparations from CD rats, CD-1 mice, and New Zealand White (NZW) rabbits and 6 from female mammoplasty patients were used to compare the dermal penetration of diethanolamine through the skin of different species. 22 [14C]diethanolamine (96.5% purity; specific activity 15.0 mCi/mmol) was applied to the skin sample undiluted, or as an aq solution, at a dose of 20 mg/cm2. Dose volumes of 35 µL undiluted [14C]diethanolamine or 95 µL of the aq solution (37% wt/wt) were applied to the exposed surface of the skin (1.77 cm2) for 6 hours. Skin sample integrity was confirmed with the use of a reference chemical, [14C]ethanol. With undiluted diethanolamine, the cumulative dose absorbed (ie, recovered in the effluent) was 0.04% in rat skin, 1.30% in mouse skin, 0.02% in rabbit skin, and 0.08% in human skin. With aq diethanolamine, an increase in the cumulative dose absorbed was seen in all species: 0.56% in rat skin, 6.68% in mouse skin, 2.81% in rabbit skin, and 0.23% in human skin. The penetration of undiluted and aq diethanolamine was greater through mouse skin than any other skin sample. With undiluted diethanolamine, penetration was similar for rat, rabbit, and human skin samples. The researchers hypothesized that the fact the penetration of diethanolamine was greater for an aq solution than for undiluted diethanolamine may be attributable to elevated skin hydration caused by the application of the aq solution to the skin.
The percutaneous absorption of diethanolamine in cosmetic formulations spiked with [14C]diethanolamine (95%-99% purity) was examined using viable human skin. 23 Two shampoo formulations, both with a [14C]diethanolamine dose of 0.49 µCi, were applied as a 1:6 aq dilution for 5 minutes; 1 shampoo contained cocamide DEA with a concentration of 0.092% free diethanolamine in the formulation and the other contained lauramide DEA with a concentration of 0.28% free diethanolamine in the formulation. The amount of the shampoo applied was 1.2 mg (diluted 1:6) and the dose was 1.9 mg/cm2. (The diethanolamine dose was 4.2 µg/cm2 for the first formulation and 7.7 µg/cm2 for the second shampoo formulation.) Two hair dye formulations, spiked with 0.49 or 0.43 µCi diethanolamine, were applied for 30 minutes. One hair dye contained lauramide DEA and the other contained cocamide and oleamide DEA. The estimated concentration of free diethanolamine in the hair dye products was 0.61%. An application of 11.2 mg was applied to the skin samples, with a dose of 17.5 mg/cm2. (The diethanolamine dose was 109.3 µg/cm2 for the first formulation and 108.9 µg/cm2 for the second hair dye formulation.) Additionally, 3 mg/cm2 of 2 body lotions (amount applied not stated) with 0.13 or 0.12 µCi diethanolamine was applied for 24 hours. (The diethanolamine dose was 1.0 µg/cm2 for the first formulation and 1.2 µg/cm2 for the second lotion formulation.) These products contained 0.0155% triethanolamine, with 0.020% free diethanolamine.
In all of these studies, very little diethanolamine was found in the receptor fluid, with only 0.1% of the applied dose recovered in the receptor fluid for the shampoo and hair dye formulations. While the amount absorbed was similar for these 2 product types, the distribution and localization were different, with most of the diethanolamine (62%-68%) penetrating from the shampoos being localized in the stratum corneum and that from the hair dyes (52%-64%) being found in deeper epidermal and dermal layers. With the lotion, 0.6% to 1.2% of the dose was recovered in the receptor fluid. Penetration from the lotions differed from each other. With the first lotion, 15.4% of the applied dose penetrated, with 0.6% found in the receptor fluid and 14.8% in the skin; approximately 65% of the penetrated diethanolamine from this formulation found in the skin was localized in the epidermis and dermis. With the second lotion formulation, 7.8% of the applied dose penetrated, with 1.2% found in the receptor fluid and 6.6% in the skin; approximately 56% of the penetrated diethanolamine from this formulation that was found in the skin was localized in the epidermis and dermis.
The researchers examined whether diethanolamine was binding to skin. Diethanolamine did not appear to be covalently bound to skin proteins, and extending the times before analysis to 48 and 72 hours did not result in any statistically significant difference when compared to the values obtained after 24 hours. Repeat application studies with viable skin did not produce a significant change in dose absorbed into receptor fluid. However, with nonviable skin, absorption into the receptor fluid from the lotion increased each day, from 0.6% on the first day to 2.6% on the third day; testing showed that the skin barrier did not remain intact for the entire 72 hours. Using a shampoo and a lotion formulation, nonviable skin gave penetration values similar to viable skin. The researchers concluded that most of the diethanolamine that penetrated was not available for systemic absorption.
Another study examining the percutaneous penetration of 7 cosmetic formulations spiked with radiolabeled and unlabeled diethanolamine was performed, mimicking simulated use conditions using fresh human skin samples. 24 Two shampoos both contained 0.98% diethanolamine and 4.02% cocamide DEA, and 2 additional shampoo formulations both contained 0.25% diethanolamine and 4.75% lauramide DEA; a 100 µL/cm2 (equivalent to 100 mg/cm2) as a 1:10 aq dilution of each formulation was applied for 10 minutes. A bubble bath containing 0.25% diethanolamine and 4.75% lauramide DEA, applied as a 1:300 dilution at a dose of 100 µL/cm2 (equivalent to 100 mg/cm2) for 30 minutes, a moisturizer containing 0.008% diethanolamine and 2% triethanolamine, applied at a dose of 5 mg/cm2 for 48 hours, and a semipermanent hair dye containing 0.075% diethanolamine and 1.42% lauramide DEA and an oxidative hair dye containing 0.25% diethanolamine and 4.74% lauramide DEA, both applied at a dose of 100 mg/cm2 for 30 minutes, were also used. Very little diethanolamine penetrated through the skin (ie, was recovered in the receptor fluid); 0.011% to 0.034% of the applied dose of the shampoo formulations penetrated, 0.024% to 0.063% of the applied dose from the hair dye formulations penetrated, and 0.508% from the bubble bath formulation penetrated through the skin. The moisturizer formulation was also applied to frozen skin samples. A total of 0.605% of the applied dose penetrated fresh skin, while 0.456% penetrated frozen skin.
The researchers also examined the penetration of a simple aq 1% solution of diethanolamine through fresh and frozen human skin samples. The cumulative 24-hour percutaneous absorption was approximately 5-fold greater in frozen skin compared to fresh skin, that is, 8.87 µg/cm2 compared to 1.73 µg/cm2. The 24-hour cumulative penetration values represented 0.433% and 0.086% of the dose for frozen and fresh skin, respectively. The amounts of diethanolamine remaining on and in the skin were 1.68% and 1.14% of the applied dose recovered from the frozen and fresh skin samples, respectively. The researchers further investigated the distribution of [14C]diethanolamine between aq and lipid fractions of viable skin strata. The radioactivity on the skin samples from 2 donors (n =12) was determined after a 24-hour exposure. At 24 hours, the cumulative permeation value was 0.405% of the applied dose for one donor (abdominal skin) and 0.067% for the other (breast skin). Tape-strip profiles for both donors appeared to indicate that the diethanolamine had become evenly distributed throughout the stratum corneum. The majority of diethanolamine was recovered in aq extract, as opposed to organic extract, of the epidermal and dermal tissue, which suggested to the researchers that the material was in the free state and not associated with the lipid fraction.
Human liver slices were incubated with 1 mM [14C]diethanolamine (>97% purity). 25 After 4 and 12 hours, 11% and 29% of the diethanolamine, respectively, absorbed into the liver slices. The radioactivity was comprised mostly of diethanolamine (85%-97%); 4 other metabolites were present at low concentrations. The liver slices were fractionated into aq, organic, and pellet fractions. The aq-extractable radioactivity was comprised primarily of diethanolamine, with up to 4 other components. Diethanolamine-derived radioactivity in the organic extracts was predominately (>90%) comprised of phospholipids containing nonmethylated headgroups. Diethanolamine was readily absorbed and incorporated into ceramides, forming mostly ceramide-phosphoethanolamine, and it was slowly methylated therein.
Dermal
Nonhuman
A single occluded dose of ∼20 mg/cm 2 (1,500 mg/kg) [ 14 C]diethanolamine was applied to a 19.5 cm2 area on the back of rats for 6 hours, and the treated site of half of the animals was rinsed after the occlusive wrapping was removed. 26 In the animals that were not rinsed, 80% of the dose was found in the wrappings and 3.6% found in the skin. In animals that were rinsed, rinsates from the wrappings contained 58% of the dose and rinsates from the skin contained 26% of the dose. Unrinsed animals absorbed 1.4% of the dose, while those animals that were rinsed absorbed 0.64%. The majority of the radioactivity was found in the carcass, liver, and kidneys.
The researchers also conducted a repeated-dose percutaneous absorption study in which as a preexposure, 1,500 mg/kg/d nonradiolabeled diethanolamine was applied occlusively under a 2 × 2 in gauze square to the backs of rats 6 h/d for 3 or 6 days. [14C]diethanolamine, 1,500 mg/kg/d, was then applied for 3 to 6 consecutive days under occlusion; each dose was left in contact with the skin for 48 hours. Totals of 21% and 41% of the dose were absorbed by the animals dosed for 3 and 6 days, respectively. The majority of the recovered radioactivity was in the wrapping. The carcass, liver, and kidneys contained most of the radioactivity in the animals. Totals of 4.3% and 13% of the radioactivity were recovered in the urine of animals dosed with nonradiolabeled diethanolamine for 3 and 6 days, respectively. Preexposure to diethanolamine resulted in a 1.5- to 3.0-fold increase in the absorption rate.
Groups of 4 to 5 male B6C3F1 mice were used in dermal studies of the absorption, distribution, metabolism, and excretion (ADME) of diethanolamine. 27 The dose, which contained 6 to 20 µCi radiolabeled, unlabeled diethanolamine and 95% ethanol, for a total volume of 15 µL per dose, was applied to a 1 cm2 area of skin under a nonocclusive covering. At 48 hours after dermal application of 8 and 23 mg/kg [14C]diethanolamine, 26.8% and 33.8% of the dose were absorbed in the mice. A statistically significant increase in the amount absorbed after dosing with 81 mg/kg, 58.1%, was observed. The amount of radioactivity found at the site of application was only 4.0%, 3.1%, and 2.2% of the dose following application of 8, 23, and 81 mg/kg [14C]diethanolamine, respectively, and the amount excreted in the urine 48 hours after application was 7.5%, 10.4%, and 16.4%, respectively. The tissue/blood ratio was greatest in the liver and kidneys.
Groups of 4 to 5 male Fischer 344 rats were used to evaluate the ADME of diethanolamine (99% purity) following dermal administration. 27 The dose, which contained 6 to 20 µCi radiolabeled, unlabeled diethanolamine, and 95% ethanol, for a total volume of 25 µL per dose, was applied to a 2 cm2 area of skin under a nonocclusive covering. Absorption increased significantly with increasing dose. After 48 hours, only 2.9% of the radioactivity was absorbed following dermal application of 2.1 mg/kg, while 10.5% and 16.2% of the radioactivity was absorbed with doses of 7.6 and 27.5 mg/kg, respectively. Of the amount absorbed, 1.2%, 4.3%, and 4.5% of the radioactivity was recovered in the skin at the dose site following application of 2.1, 7.6, and 27.5 mg/kg [14C]diethanolamine, respectively. The greatest tissue/blood ratios were found in the liver and lung.
Diethanolamine, 80 mg/kg body weight (bw) in acetone, was applied to a 2 cm2 area on the backs of 7 female C57BL/6 mice for 11 days. 28 Diethanolamine and its methylated metabolites accumulated in the liver and plasma of mice. Also, a statistically significant decrease in hepatic concentrations of choline and its metabolites was reported.
Human
Three female participants applied a lotion containing 1.8 mg diethanolamine/g to their entire body 2×/day for 1 month. 28 The participants applied 14.5 to 22.9 mL lotion/day; diethanolamine exposure following application of 20 mL/d by a 60-kg person would be 0.6 mg/kg/d diethanolamine. Blood samples were collected 1 day prior to the start of dosing, at 1 week, and at 1 month. Two of the participants completed 1 month of application, while the third completed 3 weeks. Application of the diethanolamine-containing lotion for 1 month resulted in increased concentrations of diethanolamine in plasma, when compared to control samples. (As a reference point, however, it was calculated that the concentration of diethanolamine and metabolites in the plasma of humans after a few weeks of application of the lotion was only 0.5% to 1% of the concentrations achieved in mice, where 80 mg/kg/d was applied for 11 days.)
Oral
Nonhuman
Four male Fischer 344 rats were dosed orally by gavage with 7 mg/kg aq [14C]diethanolamine to examine the ADME of diethanolamine (>97% purity). 25 The amount of radioactivity excreted in the urine at 24 and 48 hours was 9% and 22%, respectively, and the amount in the feces was 1.6% and 2.4%, respectively. At 48 hours after dosing, 27% of the radioactivity accumulated in the liver, 5% in the kidneys, and 0.32%, 0.27%, 0.19%, and 0.18% in the spleen, brain, heart, and blood, respectively. As measured in the liver and the brain, 87% to 89% of the radioactivity distributed into the aq phase, and 70% to 80% of that total radioactivity was as unchanged diethanolamine. In the liver, the remaining radioactivity in the aq phase was distributed between 3 methylated metabolite fractions, while in the brain, only 1 minor, nonmethylated metabolite was present in the aq extract.
With repeat oral dosing with [14C]diethanolamine, radioactivity continued to accumulate in tissues, reaching steady states at 4 to 8 weeks. The levels of diethanolamine equivalents in the blood, brain, and liver were much higher following 8 weeks of repeated oral doses of 7 mg/kg/d, when compared to the single-dose post-48-hour values. Again, diethanolamine was the major radioactive component. Using high performance liquid chromatography (HPLC), almost all of the organic-extractable hepatic radioactivity eluted with the phosphatidylcholine fraction, with greater than 95% of the material in the form of phospholipids containing an N,N-dimethyl-diethanolamine headgroup. In the brain, the entire organic-extractable radioactivity eluted in the phosphatidylethanolamine fraction, and it was almost entirely comprised of diethanolamine-containing headgroups.
According to the researchers, the results of this study demonstrated that diethanolamine is O-phosphorylated and N-methylated and that these metabolites are incorporated as the polar headgroups in aberrant phospholipids. The researchers felt this was evidence that diethanolamine and ethanolamine, a naturally-occurring alkanolamine, share common biochemical pathways of transformation. Retention and bioaccumulation of diethanolamine-derived radioactivity was attributed partly to aberrant phospholipids being incorporated into tissues, most probably in cell membranes.
Groups of 3 to 5 male Fischer 344 rats were used to evaluate the ADME of a single oral administration of [14C]diethanolamine (99% purity). 27 The dose administered contained 2 to 200 µCi radiolabeled, unlabeled diethanolamine and the amount of water needed to achieve a target dose of 5 mL/kg bw. Gastrointestinal absorption was nearly complete after doses up to 200 mg/kg diethanolamine. At 48 hours after dosing with 7 mg/kg, 22% of the radioactivity was excreted in the urine and 2.4% in the feces, primarily as unchanged diethanolamine. Radioactivity was not detected in carbon dioxide. Excretion was mostly unchanged diethanolamine. A total of 57% of the radioactivity was found in the body 48 hours after dosing with 7 mg/kg [14C]diethanolamine. Disposition of radioactivity was greatest in the liver (27.3%), muscle (16.3%), skin (5.1%), and kidneys (5%); only 0.2% of the radioactivity was found in the blood after 48 hours. Dose did not affect distribution in the tissues.
The researchers then evaluated the ADME of diethanolamine upon repeat oral exposure. Four rats were dosed orally with 7 mg/kg/d [14C]diethanolamine for 5 days. Approximately 40% of the total radioactivity was excreted during dosing. At 48 hours after the last dose, a total of 42% of the radioactivity was found in the tissues, with the greatest distribution in the liver (18.2%), muscle (12.4%), kidneys (5.56%), and skin (4.18%). To further investigate diethanolamine bioaccumulation, rats were dosed with 7 mg/kg/d diethanolamine, 5 d/wk, for 2, 4, or 8 weeks. During the last week of each dosing period, 69%, 79%, and 92% of the dose was excreted, respectively. After 8 weeks of dosing, most of the radioactivity was recovered as unchanged diethanolamine; however, there were also significant amounts of poorly retained metabolites, N-methyl diethanolamine, and another metabolite that was tentatively identified as a quaternized lactone. The radioactivity recovered in the liver after 2, 4, and 8 weeks of dosing was 12.3%, 7.9%, and 4.12%, respectively. It was estimated that steady state for bioaccumulation occurred after 4 weeks of repeat dosing, except for the blood, which continued to bioaccumulate diethanolamine throughout dosing. Clearance of bioaccumulated diethanolamine appears to be a first-order process, with a whole body elimination half-life of ∼6 days.
Intravenous
Nonhuman
Groups of Sprague Dawley rats (number per group not specified) received an intravenous (IV) dose of 10 or 100 mg/kg diethanolamine in physiological saline. 26 Animals were killed 96 hours after dosing. Peak concentrations appeared in the blood 5 minutes after dosing. Elimination from the blood was biphasic. Totals of 25% and 36% of the dose were excreted in the urine with 10 and 100 mg/kg diethanolamine, respectively.
Groups of 3 to 5 male Fischer 344 rats were used to evaluate the ADME of [14C]diethanolamine (99% purity) in phosphate-buffered saline after IV administration. 27 After 48 hours, 28% of the dose was excreted in the urine, 0.6% in the feces, and only 0.2% in carbon dioxide. Using HPLC, it was determined that most of the radioactivity in the urine was present as unchanged diethanolamine. A total of 54% of the dose was found in the body 48 hours after dosing with 7 mg/kg [14C]diethanolamine, and as with oral dosing, the greatest disposition was found in the liver (27%), muscle (15%), skin (4.5%), and kidneys (4%). Only 0.2% of the radioactivity was found in the blood after 48 hours.
Groups of 5 female Sprague Dawley rats were dosed IV, via the cannulated jugular vein, with 10 or 100 mg/kg [14C]diethanolamine (97.4% purity). 29 The dose volume was 2 mL/kg, and each rat received ∼4.2 µCi 14C. Blood samples were taken at various intervals up to 84 hours after dosing. The peak concentrations of radioactivity in both the plasma and the red blood cells were observed 5 minutes after dosing. Clearance of radioactivity from the plasma was calculated to be approximately 50 and 93 mL/h/kg for the low and high dose, respectively. In blood, these values were 84 and 242 mL/h/kg, respectively.
Urine and feces were collected for 96 hours after dosing. During this time, the major route of excretion was urinary; 25% and 36% of the dose was recovered in the urine. Urinary excretion was rapid at the high-dose level, with 23% of the dose recovered in the first 12 hours. Only 8.5% of the dose was recovered with the low dose during this time frame. The majority of the radioactivity was recovered in the carcass, 35% and 28% for the low and high dose, respectively. At 96 hours after dosing with 10 and 100 mg/kg diethanolamine, approximately 21% and 17% of the dose was recovered in the liver, 7% and 5% in the kidneys, and 5% and 5% in the skin. The researchers stated that because the majority of the applied dose (administered radioactivity) was recovered in the tissues, particularly in the liver and kidneys, this indicated a propensity for bioaccumulation. There was some evidence that the bioaccumulation was dose dependent.
N-Nitrosodiethanolamine Formation
The formation of N-nitrosodiethanolamine (NDELA) upon dosing with diethanolamine (99.7% purity), with and without supplemental oral sodium nitrite, was determined in male B6C3F1 mice. 30 Groups of 5 to 6 mice were dosed orally with aq diethanolamine or dermally with diethanolamine in acetone. Diethanolamine, 160 mg/kg/d, was applied for 7 d/wk for 2 weeks. One group of mice dosed dermally was allowed access to the application site, while the other was not. Studies were performed both with and without ∼40 mg/kg/d supplemental sodium nitrite in drinking water. N-Nitrosodiethanolamine was not found in the urine or blood of mice dosed with diethanolamine without nitrite or in the blood or gastric contents of those given supplemental nitrite with diethanolamine.
N-Nitrosodiethanolamine formation from diethanolamine (>99% purity) and nitrite was also examined in another study. 31 Female B6C3F1 mice were dosed dermally or orally with 4 mg/kg diethanolamine, in conjunction with oral exposure to sodium nitrite. Following 7 days of dermal dosing, no NDELA was detected in the blood, ingesta, or urine of test, vehicle control, or sodium nitrite control mice. (The limits of detection for the blood, ingesta, and urine were 0.001, 0.006, and 0.47 µg/mL, respectively.) With a single oral dose, NDELA was formed in all of the animals; the amounts of NDELA detected in the blood and ingesta of mice 2 hours postdosing were very small, 0.008 ± 0.003 µg/g and 0.424 ± 0.374 µg/g, respectively.
Toxicological Studies
Acute (Single) Dose Toxicity
Oral
In a previous safety assessment report, studies to determine the acute oral toxicity of diethanolamine were conducted in guinea pigs and rats. 1 An LD50 of undiluted diethanolamine was 0.71 to 0.80 mL/kg and 1.82 g/kg for a solution of diethanolamine in water for a group of 5 and 6 rats, respectively. Using groups of 2 to 3 guinea pigs, all guinea pigs survived dosing with 1 g/kg, but none survived dosing with 3 g/kg diethanolamine gum arabic solution. Using 90 to 120 rats, 20% aq diethanolamine had an acute LD50 of 1.41 to 2.83 g/kg, based on results of testing performed over a 10-year time period. With groups of 10 rats, a hair preparation containing 1.6% diethanolamine had an LD50 of 14.1 g/kg when diluted and 12.9 mL/kg when undiluted.
Other
The intraperitoneal LD50 of diethanolamine was 2.3 g/kg for mice in a previous safety assessment report. 1
Repeated Dose Toxicity
Dermal
In a 13-week study, 1 mg/kg of a hair dye formulation containing 2.0% diethanolamine was applied to the backs of 12 rabbits for 1 hour, twice weekly. The test site skin was abraded for half of the animals. No systemic toxicity was observed, and there was no histomorphologic evidence of toxicity in rabbit studies reported in a previous safety assessment report. 1
Diethanolamine was applied dermally to groups of 5 male and 5 female B6C3F1 mice, 5×/week for 2 weeks, at doses of 160, 320, 630, 1,250, and 2,500 mg diethanolamine/kg bw in 95% ethanol. 32 Vehicle only was applied to the controls. All of the male and 3 of the female high-dose test animals died during the study. Ulceration, irritation, and crusting were observed at the application site of male mice of the 1,250 and 2,500 mg/kg groups and females of the 2,500 mg/kg group. Microscopically, moderate to marked epidermal ulceration and inflammation were observed in these animals. Ulcerative necrosis extended into the underlying dermis. Minimally severe acanthosis, without inflammation, was seen in the 160, 320, and 630 mg/kg dose groups. Absolute and relative liver weights increased in a dose-dependent manner in males and females.
Groups of 5 male and 5 female F344/N rats were also dosed with diethanolamine 5×/week for 2 weeks, with 125, 250, 500, 1,000, and 2,000 mg diethanolamine/kg bw in 95% ethanol. 32 Vehicle only was applied to the negative controls. All of the females and 3 of the males of the 2,000 mg/kg group and 1 female of the 1,000 mg/kg group died during the study. Irritation and crusting was observed at the application site of male and female rats of the 500, 1,000, and 2,000 mg/kg groups. Microscopically, ulceration with acanthosis and inflammation was observed in these animals. Ulcerative necrosis extended into the underlying dermis. Minimal acanthosis was seen in female rats of the 250 mg/kg group, and hyperkeratosis was seen in all test groups. Absolute and relative kidney weights increased in a dose-dependent manner in males and females. Renal tubular epithelial necrosis (mild severity) was observed in the animals that died during the study. Some statistically significant changes in urinalysis parameters were observed. Mild to moderate seminiferous tubule degeneration in the testis was observed in 4 of 5 high-dose males.
To determine whether repeated dermal administration of diethanolamine induced cell proliferation and/or apoptosis in the livers of mice, 8 male and 8 female B6C3F1/Crl mice were dosed dermally with 0 and 160 mg/kg bw diethanolamine (99.6% purity) in 96% ethanol for 1 week followed by a 3-week recovery period and groups of 10 male mice were exposed dermally, 5 d/wk, to 0 or 160 mg/kg for 1, 4, or 13 weeks. 33 A dose–response relationship was examined using groups of 8 male mice; applications of 10 to 1,250 mg/kg diethanolamine were made to male mice for 1 or 13 weeks, and controls were exposed to vehicle only. (Application of 630 and 1,250 mg/kg diethanolamine was discontinued, and the animals killed, after 1 week due to severe skin lesions; the high dose was then 160 mg/kg.) No diethanolamine-related deaths were observed. Body weights were not affected by dosing. Statistically significant increases in liver weights were seen in mice dosed with ≥10 mg/kg bw/d diethanolamine for 1 or more weeks. After 1 week of dosing, increased cell proliferation in the liver was observed in males and females; this effect was reversible. Eosinophilia was found in animals dosed with ≥40 mg/kg diethanolamine for 1 or more weeks, and hepatocellular giant cells were seen in animals dosed with 160 mg/kg for 13 weeks. Repeated application of ≥10 mg/kg diethanolamine to male B6C3F1 mice caused increased liver cell proliferation. Diethanolamine had no effect on the number of apoptotic cells in the liver. Some erythema and/or focal crust formation was observed and attributed to the procedure and/or the vehicle, but not to diethanolamine.
Unoccluded dermal applications of 80, 160, 320, 630, and 1,250 mg/kg bw/d diethanolamine (purity >99%) in acetone were applied to the backs of 10 male and 10 female B6C3F1 mice, 5 d/wk for 13 weeks. 34 Vehicle only was applied to the negative control group. (The square area of the dose site was not provided.) Two male mice and 4 female mice dosed with 1,250 mg/kg diethanolamine were killed in moribund condition. Final mean bws of male mice dosed with 1,250 mg/kg were statistically significantly decreased compared to controls. Clinical signs of toxicity, observed in males and females dosed with 630 and 1,250 mg/kg diethanolamine, were irritation, crust formation, and thickening at the application site. Ulceration and inflammation were observed in the 630 and 1,250 mg/kg dose groups. Dose-dependent increases in absolute and relative liver weights, associated with hepatocellular changes, were observed, and hepatocellular necrosis was seen in males dosed with 320 to 1,250 mg/kg diethanolamine. Absolute kidney weights were statistically significantly increased in males and females of all test groups. Relative kidney weights were increased in males of all test groups and females dosed with 630 or 1,250 mg/kg diethanolamine; nephropathy was not found. Heart weights were increased in high-dose males and females, and cardiac myocyte degeneration was reported.
Unoccluded dermal applications of 32, 63, 125, 250, and 500 mg/kg bw/d diethanolamine (purity >99%) in 95% ethanol were applied to the backs of 10 male and 10 female F344/N rats, 5 d/wk for 13 weeks. 35 Vehicle only was applied to the negative control group. (The square area of the dose site was not provided.) One male and 2 females given 500 mg/kg died or were killed in moribund condition during the study. Clinical signs of toxicity, observed in males and females given 125 to 500 mg/kg diethanolamine, were irritation and crusting at the application site. The no observable adverse effect level (NOAEL) for ulceration was 125 mg/kg for male rats and 63 mg/kg for female rats. Final mean bws were statistically significantly decreased in males dosed with 250 and 500 mg/kg and females dosed with 125 to 500 mg/kg diethanolamine. Increases in absolute and relative kidney weights were observed with increased incidence of renal lesions. Nephropathy was observed in lower and mid-dose females, but a clear treatment effect was not seen in males. Increases in absolute and relative liver weights were not accompanied by an increase in hepatic lesions. Demyelination in the medulla oblongata was seen in all high-dose animals (7 females) given 250 mg/kg diethanolamine; this lesion was minimal in severity. An NOAEL was not achieved regarding hematological changes, but the researchers did note that the differences between controls and male rats exposed to 32 mg/kg diethanolamine were minimal.
Oral
A previous safety assessment of triethanolamine, diethanolamine, and monoethanolamine reported on oral studies conducted in which neonatal rats were dosed with 1 to 3 mM/kg/d diethanolamine, as a neutralized salt, on days 5 to 15 after birth, male rats were dosed with 4 mg/mL neutralized diethanolamine in drinking water for 7 weeks, and rats were fed 0 to 0.68 g/kg/d diethanolamine in feed for 90 days. 1 Repeated oral ingestion of diethanolamine produced evidence of hepatic and renal damage. Deaths occurred in the 7-week and 90-day studies. Administration of neutralized diethanolamine in the drinking water at doses of 490 mg/kg/d for 3 days or of 160 mg/kg/d for 1 week produced alterations in hepatic mitochondrial function. Oral administration of diethanolamine may affect, directly or indirectly, the serum enzyme levels, isozyme patterns, and concentrations of some amino acids and urea in the male rat liver and kidney. Repeated diethanolamine administration in the drinking water increased male rat hepatic mitochondrial ATPase and altered mitochondrial structure and function.
Female CD-1 mice, 3 per group, were dosed orally, by gavage, with 10, 100, or 1,000 mg/kg bw diethanolamine in distilled water for 7 days. 36 One animal of the 100 mg/kg group died, and the death was considered test article related. (Two deaths in the 1,000 mg/kg group were attributed to gavage error.)
Groups of 48 female B6C3F1 mice were dosed with 0, 100, 300, or 600 mg/kg bw diethanolamine 37 and groups of 48 F344 female rats were dosed with 0, 50, 100, or 200 mg/kg bw diethanolamine, 38 orally, by gavage, in distilled water for 14 days. No effect on bw gain was observed for mice in any group. Rats exposed to 10 and 200 mg/kg diethanolamine had significant decreases in bw and/or bw gains. Liver weights were increased in a dose-dependent manner in both rats and mice, and kidney weights were increased in a dose-dependent manner in rats. No effects were seen in thymus, spleen, or kidney weights of mice. In mice, diethanolamine exposure resulted in an increase in the number of B cells and a decrease in the number of CD4+CD8− (18%) T-cell subsets. A dose-dependent decrease in the antibody-forming response to sheep erythrocytes was observed with 600 mg/kg diethanolamine, and a dose-dependent decrease in the cytotoxic T-lymphocyte response was observed, which was statistically significant at the lowest dose. The natural killer cell response was not affected. In rats, exposure to diethanolamine did not alter the number of B cells, T cells, or T-cell subsets. The proliferative response to allogenic cells, as measured by the mixed leukocyte response, was increased in a dose-dependent manner; the increase in the high dose was statistically significant. Natural killer cell response and cytotoxicity of resident macrophages were decreased in diethanolamine-treated animals.
In a 13-week study, 10 male and 10 female B6C3F1 mice were dosed orally, by gavage, 5 times/week, with 50, 100, 200, 400, or 800 mg/kg diethanolamine in deionized water. 39 The negative controls were dosed with vehicle only. Two males of the high-dose group died during the study. Body weights and bw gains of high-dose males were decreased. No clinical signs of toxicity were noted. Treatment-related renal lesions were found, but details were not provided.
Groups of 10 male and 10 female B6C3F1 mice received 630, 1,250, 2,500, 5,000, or 10,000 ppm diethanolamine (>99% purity) in the drinking water for 13 weeks. 34 The pH of the solution was adjusted to 7.4. The negative controls were given untreated water. All males and females of the 5,000 and 10,000 ppm groups and 3 females of the 2,500 ppm group died during the study. Body weight gains were decreased in males of the 2,500 ppm group and females of the 1,250 and 2,500 ppm groups. No significant gross effects were noted at necropsy. Statistically significant, dose-dependent, increases in absolute and relative liver weights were observed, with hepatocellular cytological alterations and necrosis in animals given ≥2,500 ppm diethanolamine. Absolute and relative kidney weights also increased dose dependently, with statistically significant increases in mice given 1,250 or 2,500 ppm diethanolamine, and nephropathy was observed in all male test groups and female test groups given 2,500 or 5,000 ppm diethanolamine. Heart weights were increased in female mice given 2,500 ppm diethanolamine, and relative heart weights were increased in males of the 2,500 ppm group and females of the 1,250 and 2,500 ppm groups.
Groups of 10 male F344/N rats received 320, 630, 1,250, 2,500, or 5,000 ppm diethanolamine and 10 female F344/N rats received 160, 20, 630, 1250, or 2,500 ppm diethanolamine in the drinking water for 13 weeks. 35 Negative controls were given untreated water. Two males of the high-dose group died during the study. The following dose-related effects were observed: decreases in bw gains in males and females, hematological effects, increases in kidney weights accompanied by renal lesions, and increases in relative liver weights in males and females. Demyelination of the medulla of the brain and of the spinal cord was observed in all males of the 2,500 and 5,000 ppm groups and all females of the 1,250 and 2,500 ppm groups.
Groups of 10 male albino rats received feed containing 0.01%, 0.1%, or 1% (10, 100, or 1,000 mg/kg bw/day) diethanolamine (99% purity) for 32 days. 40 Nine of the 10 high-dose animals died during the study. All test animals had decreased hemoglobin and hematocrits, with an increased white blood cell count. The relative liver weights of animals fed 0.01% and 0.1% diethanolamine and the absolute liver weights of animals of the 0.1% group were increased. In a repeat 30-day study using the same procedure and dose levels, 7 of the 10 high-dose animals died and the remaining 3 high-dose animals killed, prior to study termination; bws and feed consumption were significantly decreased in this group. Again, hemoglobin and hematocrit were decreased in the 0.1% group, and the hemoglobin value was reduced in the 0.1% group.
Inhalation
Short-term inhalation of 200 ppm diethanolamine vapor or 1,400 ppm diethanolamine aerosols (duration of dosing not provided) produced respiratory difficulties and some deaths in male rats. Inhalation of 25 ppm for 216 continuous hours resulted in increased liver and kidney weights, while exposure of male rats to 6 ppm diethanolamine following a “workday” schedule for 13 weeks caused decreased growth rate, increased lung and kidney weights, and some deaths. 1
In a 2-week dose-range finding inhalation study, 10 male and 10 female Wistar rats were exposed, nose only, to target concentrations of 0, 100, 200, and 400 mg/m3 diethanolamine (>99% pure) for 6 h/d, 5 d/wk. 41 Mass median aerodynamic diameter (MMAD) was 0.6 to 1.9 µm. Test article–related effects were not seen with 100 or 200 mg/m3 diethanolamine. With 400 mg/m3 diethanolamine, decreased bws and bw gains were seen in males and increased relative and absolute liver weights were seen in females. Microscopically, no effects were seen in the respiratory tract; the larynx was not examined.
Based on the results of the dose-range finding study, target concentrations of 0, 15, 150, and 400 mg/m3 diethanolamine were used in a 90-day study, in which groups of 13 male and 13 female rats were exposed via inhalation to 65 exposures, 6 h/d 5 d/wk. The MMAD was 0.6 to 0.7 µm. A functional observational battery was conducted using 10 rats/gender/group. Body weight gains were reduced in males of the high-dose group. Some statistically significant effects on clinical chemistry values were seen in the mid- and high-dose group. Males and females of the high-dose group had statistically significant increases in blood in the urine, and males of the mid- and high-dose groups excreted significantly elevated amounts of renal tubular epithelial cells. Relative liver weights were statistically significantly increased in males and females of the high-dose group and females of the mid-dose group, and relative kidney weights were statistically significantly increased in males and females of the mid- and high-dose groups. Focal squamous metaplasia of the ventral laryngeal epithelium was observed in test animals of all groups, and a concentration-dependent increase in laryngeal squamous hyperplasia, and in the incidence and severity of local inflammation of the larynx and trachea, was observed. There were no indications of neurotoxicological effects.
In a third study, test groups of 10 male and 10 female Wistar rats were exposed to target concentrations of 0, 1.5, 3, and 8 mg/m3 diethanolamine using the same dosing schedule as above, and recovery groups of 10 females were exposed to 3 or 8 mg/m3, with a postexposure period of 3 months. No dose-related clinical signs were observed. Liver weights of test, but not recovery, females dosed with 8 mg/m3 were statistically significantly increased. Laryngeal effects were similar to those described above. No microscopic changes were observed in the upper respiratory tract of recovery animals. The 90-day no observed effect concentration (NOAEC) was determined to be 1.5 mg/m3 diethanolamine.
In inhalation studies, Sprague Dawley rats, Hartley guinea pigs, and beagle dogs (number per species and sex not specified) were each exposed to 0.5 ppm diethanolamine for 6 h/d, 5 d/wk, for a total of 45 exposures. 40 All animals survived until study termination. There were no clinical signs of toxicity and no evidence of irritation. No gross or microscopic lesions were observed at necropsy.
Reproductive and Developmental Toxicity
Dermal
No developmental or reproductive effects were observed in rat studies using a hair dye containing up to 2% diethanolamine, as reported in a previous safety assessment report. 1 A reproductive and developmental toxicity study was performed in which 0, 20, 80, or 320 mg/kg diethanolamine (98.5% purity) in ethanol was applied to a 2 cm2 area on the back of 15 male C57BL/6 mice, 15 per group, for 4 weeks. 42 (It was not stated whether the test area was covered.) These males were then mated with untreated females. In the parental male mice, sperm motility was significantly decreased at all dose levels and in a dose-dependent manner. In male pups, a significant decrease in epididymis weight was seen in the 80 mg/kg group at postnatal day (PND) 21 and a reduction in weights of reproductive organs of high-dose males pups was seen at PND 70. There were no significant differences in skeletal formation or in growth and development parameters.
Doses of 0, 20, 80, or 320 mg/kg diethanolamine (98.5% purity) in ethanol were applied to a 2 cm2 area on the backs of groups of 10 gravid female C57BL/6 mice on day 6 of gestation through PND 21. The bws of male and female pups of the high-dose group were statistically significantly decreased compared to controls. No specific differences in organ weights were observed in pups of the test groups as compared to controls. There were no significant differences in skeletal formation. Dose-dependent effects on growth and developmental parameters were noted. Sperm motility was decreased in male pups, but this result was not statistically significant.
CD rats and NZW rabbits were used to evaluate the potential of diethanolamine (≥99.4% purity) to produce developmental toxicity with dermal exposure. 43 Groups of 25 gravid CD rats were dosed dermally with 150, 500, or 1,500 mg/kg/d diethanolamine in deionized water, 6 h/d, on days 6 to 15 of gestation, under an occlusive covering. Dosing volume was 4 mL/kg/d. Control animals were dosed with vehicle only. Teratogenic effects were not seen at any dose. Dermal application of 500 mg/kg diethanolamine resulted in alterations in maternal hematological parameters but did not affect embryonal/fetal development. Other signs of maternal toxicity were seen at this dose and with 1,500 mg/kg/d. The NOEL for embryonal/fetal toxicity was estimated to be 380 mg/kg/d, which incorporates an adjustment for the 10% to 24% deficit in expected dose that occurred on days 12 to 15 of gestation.
Groups of 15 mated rabbits were dosed dermally with 35, 100, or 350 mg/kg/d diethanolamine in deionized water, 6 h/d, on days 6 to 18 of gestation, under an occlusive covering. Dosing volume was 2 mL/kg/d, and controls were dosed with vehicle only. Dermal administration of 350 mg/kg/d diethanolamine produced severe skin irritation in rabbits, and signs of maternal toxicity were observed at this dose. No developmental toxicity was observed, and there was no evidence of teratogenicity at any dose. The NOEL for maternal toxicity of diethanolamine in rabbits was 35 mg/kg/d, and the embryonal/fetal NOEL was 350 mg/kg/d diethanolamine.
Oral
In a Chernoff-Kavlock screening test, groups of 4 gravid CD-1 mice were dosed orally, by gavage, with 200, 380, 720, 1,370, or 2,605 mg/kg bw diethanolamine in distilled water on days 6 to 15 of gestation; a control group was dosed with vehicle only. 36 Two, 3, and 4 of 4 animals of the 720, 1,370, and 2,605 mg/kg dose groups died during the study. Rough hair coats were observed at all dose levels. Group of 50 female gravid CD-1 female mice were dosed orally, by gavage, with 0 or 450 mg/kg bw diethanolamine in distilled water on days 6 to 15 of gestation. No animals died during the study. The reproductive index and average number of live litters on day 0 were not affected by dosing, but the average number of live litters on day 3 was decreased. Mean bws and bw gains of pups were also decreased on PND 3.
Gravid Sprague Dawley rats were dosed orally, by gavage, with 50, 200, 500, 800, or 1,200 mg/kg/d diethanolamine on days 6 to 15 of gestation. 44 Maternal mortality was observed at doses of 50 to 1200 mg/kg/d. At doses of 50 and 200 mg/kg/d, no differences in gross developmental end points were found between test and control animals. The NOEL for embryonal/fetal toxicity was 200 mg/kg/d diethanolamine. However, maternal weight gains were significantly decreased at that dose. (Additional details were not provided.)
Groups of 12 gravid female Sprague Dawley rats were dosed orally, by gavage, with 50, 125, 200, 250, or 300 mg/kg bw/d diethanolamine (>98% purity) distilled water on days 6 to 19 of gestation, while controls were dosed with vehicle only. 45 Dosing volume was 5 mL/kg. Surviving dams and pups were killed on PND 21. All females dosed with 300 mg/kg diethanolamine were killed prior to study termination due to excessive toxicity. Toxicity was also observed for 1 dam dosed with 200 mg/kg, and only 5 dams of the 250 mg/kg group delivered live litters and survived until study termination. The calculated LD50 was 218 mg/kg bw/d. No significant maternal or developmental toxicity was seen with 50 mg/kg bw/d diethanolamine. Signs of maternal and developmental toxicity were seen at doses of ≥125 mg/kg bw/d and included decreased maternal weight gains, increased kidney weights in dams, increased postimplantation and postnatal mortality, and reduced live pup weights. The NOAEL for maternal toxicity and teratogenicity was 50 mg/kg/d, and the lowest-observable-adverse-effect level (LOAEL) for these parameters was 125 mg/kg/d.
Inhalation
In a range-finding study, groups of 10 gravid Wistar rats were exposed, nose only, to target concentrations of 0.1, 0.2, or 0.4 mg /L diethanolamine, 6 h/d, on days 6 to 15 of gestation. 46 (Diethanolamine purity was >98.7%.) All animals survived until study termination. Relative liver weights were increased in animals of the 0.2 mg/L group, and absolute and relative liver weights were increased in animals of the 0.4 mg/L group. No treatment-related effects were observed with 0.1 mg/L diethanolamine.
Groups of 25 gravid Wistar rats were exposed, nose only, to target concentrations of 0.01, 0.05, or 0.2 mg diethanolamine aerosol/L air, 6 h/d, on days 6 to 15 of gestation. 47,48 (Diethanolamine purity was >98.7%.) Maternal toxicity, as indicated by vaginal hemorrhage, was seen at the highest dose level. No treatment-related malformations were observed at 0.2 mg/L. The NOAEC for both maternal and developmental toxicity was 0.05 mg/L, and the NOAEC for teratogenicity was >0.2 mg/L, which was the highest dose tested in this study.
Effect on hippocampal neurogenesis and apoptosis
The effect of diethanolamine on neurogenesis was investigated using C57BL/6 mice. 49 Diethanolamine, 0, 20, 80, 160, 320, and 640 mg/kg bw in ethanol, was applied to a 2 cm2 area on the backs of gravid female mice, 6 per group, on days 7 to 17 of gestation. A dose-related decrease in litter size was observed at doses >80 mg/kg, and the decrease was statistically significant at doses of 160 to 640 mg/kg bw/d. The livers of the maternal mice were analyzed on day 17 of gestation, and hepatic concentrations of choline and its metabolites were statistically significantly decreased. In the fetal brain, treatment with 80 mg/kg bw/day diethanolamine diminished the proportion of cells that were in the mitotic phase to 50% of controls. The number of apoptotic cells of the hippocampal area was >70% higher in fetuses of diethanolamine-treated mice compared to controls. The researchers stated that the effect observed in the mouse fetal brain after administration of diethanolamine was likely to be secondary to diminished choline levels. The researchers also hypothesized that a potential mechanism for the effect of choline deficiency, and maybe diethanolamine, on progenitor cell proliferation and apoptosis involves abnormal methylation of promoter regions of genes.
The doses used in the above study were based on expected concentrations of 1% to 25% diethanolamine in cosmetic formulations. However, based on comments from the Cosmetic, Toiletry, and Fragrance Association (now known as the Personal Care Products Council), indicating the overestimation of the amount of diethanolamine contained in consumer products, 50 the researchers tested lower doses to establish a dose–response relationship. Groups of 7 mice were dosed dermally with 20 to 80 mg/kg bw diethanolamine (purity >99.5%) as described previously, with the exception that acetone was used as the vehicle. 49 The negative control group was dosed with vehicle only. Although the results reported in the earlier study with 80 mg/kg bw diethanolamine were confirmed, no differences were seen between treated and control groups with <80 mg/kg bw.
In a study to identify the potential mechanism for the alterations described above, mouse neural precursor cells were treated in vitro with diethanolamine. 51 Cells exposed to 3 mM diethanolamine had less cell proliferation at 48 hours and had increased apoptosis at 72 hours. Diethanolamine treatment decreased choline uptake into the cells, resulting in diminished choline and phosphocholine. A 3-fold increase in choline concentration prevented the effects of diethanolamine exposure on cell proliferation and apoptosis; intracellular phosphocholine levels remained low. The researcher hypothesized that diethanolamine interferes with choline transport and choline phosphorylation in neural precursor cells. Additionally, it was suggested that diethanolamine acts by altering intracellular choline availability.
Genotoxicity
In Vitro
Diethanolamine, with and without metabolic activation using liver preparations from rats induced with a polychlorinated biphenyl mixture, was not mutagenic to Salmonella typhimurium TA100 or TA1535, according to the data presented in a previous safety assessment report. 1
With or without metabolic activation, diethanolamine (0-3,333 µg/plate) was not mutagenic in Ames test using S typhimurium TA98, TA100, TA1535, or TA1537, was negative in a mouse lymphoma assay (≤400 nL/mL), and did not induce sister chromatid exchanges or chromosomal aberrations in a Chinese hamster ovary cell cytogenetic assay (≤1,500 µg/mL). 52 Diethanolamine was not clastogenic in a mouse micronucleus test (80-1,250 mg/kg, dermal application). Positive results were reported in an in vitro assay for induction of DNA single-strand breaks in isolated hepatocytes for rats, hamsters (both at ≥25 µmol/tube), and pigs (≥12.5 µmol/tube).
In studies examining species selectivity of effects caused by diethanolamine, increases in DNA synthesis were observed in mouse and rat, but not in human, hepatocytes following treatment with ≥10 µg/mL diethanolamine. 53 Additionally, when the hepatocytes were incubated in medium containing reduced choline, DNA synthesis was increased in mouse and rat hepatocytes, but not in human hepatocytes. Conversely, choline supplementation decreased diethanolamine-induced DNA synthesis in mouse and rat hepatocytes.
Carcinogenicity
Dermal
The National Toxicology Program (NTP) evaluated the carcinogenic potential of diethanolamine (>99% purity) in ethanol using B6C3F1 mice and F344/N rats. 52 Groups of 50 male and 50 female mice were dosed dermally with 0, 40, 80, or 160 mg/kg/d, 5 d/wk, for 103 weeks. Survival of dosed females, but not males, was significantly decreased in a dose-dependent manner. Mean bws of test animals were decreased at various intervals throughout the study. In male mice, the incidences of hepatocellular adenoma and hepatocellular adenoma and carcinoma (combined) in all dose groups, and the incidence of hepatocellular carcinoma and hepatoblastoma in the 80 and 160 mg/kg groups, were statistically significantly increased compared to controls. In female mice, the incidence of hepatocellular neoplasms was significantly increased. Male mice also had a dose-related increase in the incidences of renal tubule hyperplasia and renal tubule adenoma or carcinoma (combined) and an increase in the incidence of renal tubule adenoma. In male and female mice, incidences of thyroid gland follicular cell hyperplasia were increased. Hyperkeratosis, acanthosis, and exudate were treatment-related changes observed at the application site. It was concluded that there was clear evidence of carcinogenic activity of diethanolamine in male and female B6C3F1 mice based on increased incidences of liver neoplasms in males and females and increased incidences of renal tubule neoplasms in males.
In the rats, groups of 50 males were dosed dermally with 0, 16, 32, or 64 mg/kg bw diethanolamine and groups of 50 females with 0, 8, 16, or 32 mg/kg bw, 5 d/wk, for 103 weeks. The only treatment-related clinical finding was irritation at the application site. Minimal to mild nonneoplastic lesions were found at the site of application of dosed males and females. The incidence and severity of nephropathy in dosed females, but not males, was significantly greater in the treated groups compared to controls. There was no evidence of carcinogenic activity of diethanolamine in male or female F344/rats.
The carcinogenic potential of diethanolamine was evaluated using a Tg·AC (zetaglobin v-Ha-ras) transgenic mouse model. 54,55 Groups of 10 to 15 female homozygous mice were dosed dermally with 0, 5, 10, or 20 mg diethanolamine/mouse in 95% ethanol, 5×/week, for 20 weeks. Diethanolamine was inactive in Tg·AC mice.
Previously, the International Agency for Research on Cancer (IARC) Working Group concluded that diethanolamine was not classifiable as to its carcinogenicity to humans (group 3), based on inadequate evidence in humans and limited evidence in experimental animals for the carcinogenicity of diethanolamine. 56 Recently, however, taking into consideration that no or very few epidemiological studies assessing agent-specific exposure are available, the IARC Working Group concluded that, in the absence of adequate epidemiological information, there was sufficient evidence of carcinogenicity in experimental animals for a classification of diethanolamine as possibly carcinogenic to humans (group 2B). 57 (This change was based on a change in the criteria for classification, not new animal data.).
Possible mode of action for carcinogenic effects
Choline deficiency has been shown to increase spontaneous hepatocarcinogenesis in rats. 58,59 Male F344 rats fed diets deficient in labile methyl donors, specifically choline and methionine, absent any known carcinogen, had increased incidence of liver cancer. Addition of choline to the diet prevented this effect. Hepatic effects were also seen in mice. 60 Male B6C3F1 mice fed a choline-deficient diet resulted in all animals developing either liver nodules or tumors.
In the NTP studies, diethanolamine also increased renal tubule neoplasms in male mice. 52 In a study in which male Sprague Dawley rats were fed a choline-deficient diet for 6 days, followed by a normal diet for up to 119 days, acute renal lesions consisting of tubular epithelial cell necrosis were observed immediately after being fed a choline-deficient diet. 61 Chronic renal lesions consisting of interstitial nephritis characterized by fibrosis and scarring were observed 28 to 119 days after being fed with the choline-deficient diet. (Hepatic lesions were also observed.) The proximal convoluted tubule was most severely affected.
Disposition data indicate that diethanolamine is less readily absorbed across rat skin than mouse skin, 22 resulting in lower blood and tissue levels of diethanolamine in rats than in mice, suggesting that, in rats, the levels of diethanolamine that occur are not high enough to markedly alter choline homeostasis. If true, species differences observed in tumor susceptibility could be a function of the internal dose of diethanolamine. Alternatively, species differences in tumor susceptibility may explain the increased incidence of hepatocarcinogenesis in B6C3F1 mice compared to rats exposed to diethanolamine. It is important to note that both rats and mice are reported to be much more susceptible to choline deficiency than humans. 62
A species-selective inhibition of gap junctional intercellular communication by diethanolamine in mouse and rat, but not in human, hepatocytes with medium containing reduced choline concentrations provided additional support that the mechanism for diethanolamine-induced carcinogenicity in rodents involves cellular choline deficiency. 63 Also, Bachman et al have hypothesized the diethanolamine-induced choline deficiency leads to altered DNA methylation patterns, which facilitates tumorigenesis. 64
Two other hypotheses as to the mode of action of diethanolamine carcinogenicity as possible alternatives to the intracellular choline deficiency hypothesis have been proposed. 65 One involves the nitrosation of diethanolamine to NDELA and the other the formation of diethanolamine-containing phospholipids. The researcher did not find these likely for the following reasons. Regarding the first alternate hypothesis, nitrosation to NDELA, NDELA was not detected in mouse plasma or urine after cotreatment of mice with diethanolamine and nitrite. In regard to the second hypothesis, altered phospholipids, while diethanolamine has been shown to be incorporated into phospholipids, without qualitative or quantitative differences between rats and mice, carcinogenic effects are seen only in mice, making it unlikely that incorporation of diethanolamine into the phospholipids is a major determinant of carcinogenic response. Leung et al reviewed the information available and also felt that choline deficiency is the mechanism responsible for liver tumor promotion in mice. 66
The localization of β-catenin protein in hepatocellular neoplasms and hepatoblastomas in B6C3F1 mice exposed dermally to 0 to 160 mg/kg bw diethanolamine for 2 years were characterized, and genetic alterations in the Catnb and H-ras genes were evaluated. 67 A lack of H-ras mutations in hepatocellular neoplasms and hepatoblastomas led the researchers to suggest that the signal transduction pathway is not involved in the development of liver tumors following diethanolamine administration.
Irritation and Sensitization
Irritation
Skin
Nonhuman
The primary skin irritation potential of diethanolamine using rabbits was reported in a previous safety assessment report. Undiluted diethanolamine was reported to be moderately irritating and no irritation was observed with 10% aq diethanolamine following the same protocol. Using groups of 6 rabbits, application of 30% and 50% diethanolamine using semiocclusive patches to intact and abraded skin of rabbits produced essentially no irritation. 1
Human
Various concentrations of diethanolamine were found to be nonirritating in studies described in a previous safety assessment report. 1
Mucosal
Nonhuman
The ocular irritation potential of 30% to 100% diethanolamine was evaluated using rabbits. 1 A 30% aq solution, diethanolamine was essentially nonirritating, while a 50% aq solution was a severe irritant. Instillation of 0.02 mL undiluted diethanolamine produced severe injury to rabbit eyes. A hair preparation containing 1.6% diethanolamine had a maximum average irritation score of 0.7 of 110 for rinsed and unrinsed eyes.
Sensitization
Nonhuman
Diethanolamine had an EC3 (estimated concentration of a substance expected to produce a stimulation index of 3) value of 40% in a mouse local lymph node assay (OECD guideline 429), resulting in a categorization of weak potency of for skin sensitization. 68 In a maximization study using 15 Dunkin-Hartley guinea pigs, intradermal and epicutaneous induction used 1% and 17.6% aq diethanolamine, respectively, after 10% sodium lauryl sulfate pretreatment. 69 At challenge with 0.7%, 3.5%, or 7% diethanolamine, 1 of 15 animals reacted to the lowest and highest challenge concentrations after 2 but not 3 days. In a second maximization test using 20 Himalayan spotted guinea pigs, the intradermal induction, epicutaneous induction, and epicutaneous challenge concentrations were 5%, 75%, and 25% diethanolamine in physiological saline, respectively. 69,70 Freund’s complete adjuvant was used at intradermal induction. Two animals had mild erythema at day 1, and 1 animal had mild erythema at day 2. Diethanolamine was not a sensitizer.
Human
Formulations containing 1.6% and 2.7% diethanolamine, tested undiluted in 12 and 100 participants, respectively, and a formulation containing 2% diethanolamine, tested as a 10% solution in distilled water in 165 participants, were not sensitizing in clinical studies, as reported in a previous safety assessment report. 1
Provocative testing
Over a 15-year period, provocative patch testing using diethanolamine was performed on 8,791 patients. 69 There were 157 (1.8%) positive reactions to diethanolamine, and most of the reactions (129; 1.4%) were weak positives. There were 17 (0.2%) irritant reactions reported. Cosensitization was reported; 77% of the patients who reacted to diethanolamine also tested positive to ethanolamine. Occupational sensitization was reported; of 7,112 male patients, 1.0% who did not work in the metal industry had positive reactions to diethanolamine, as opposed to 3.1% and 7.5% of those working in the metal industry and those exposed to water-based metalworking fluids, respectively.
Metal workers who were patients with dermatitis were patch tested with 2% diethanolamine in petrolatum (pet). 71 The patches were applied for 1 to 2 days. On day 3, one of 174 patients (0.6%) had a positive reaction.
Patch testing was performed with 2.5% diethanolamine in pet in 200 patients with suspected metalworking fluid contact dermatitis. 72 (All patients were metalworkers.) Patches were applied for 1 or 2 days. On day 3, 12 patients had positive reactions, 6 (doubtful), 5 (+), and 1 (++). The % positive reactions was 3.0%.
Miscellaneous Studies
In male albino rats, following repeated oral administration of 320 mg/kg/d in drinking water of radiolabeled diethanolamine, a decrease was seen in the amount of choline incorporated into the liver and the kidneys after 1, 2, and 3 weeks as compared to 0 weeks. Ethanolamine and choline phospholipid derivatives were synthesized faster and in greater amounts and were catabolized faster than diethanolamine phospholipid derivatives. This was not seen with a single 250 mg/kg injection of diethanolamine. 1
Inhibition of Choline Uptake
The ability of diethanolamine to alter cellular choline levels was examined in a number of studies. In the study described previously in which B6C3F1 mice were dosed orally and dermally with 160 mg/kg/d diethanolamine in conjunction with oral sodium nitrite, a pronounced decrease in choline and its metabolites was observed in the livers. 30 The smallest decreases were seen in the mice that were dosed dermally and not allowed access to the test site, and the greatest increase was seen in the mice dosed orally.
In Syrian hamster embryo (SHE) cells, diethanolamine inhibited choline uptake at concentrations ≥50 µg/mL, reaching a maximum 80% inhibition at 250 to 500 µg/mL. 73 Diethanolamine also decreased phosphatidylcholine concentrations in the phospholipids, was incorporated into SHE lipids, and transformed SHE cells in a concentration-dependent manner. Excess choline blocked these biochemical effects and inhibited cell transformation, and the researchers hypothesized there is a relationship between the effect of diethanolamine on intracellular choline availability and utilization and its ability to transform cells. In a study performed to test the hypothesis that diethanolamine treatment could produce biochemical changes consistent with choline deficiency in mice, it was found that diethanolamine treatment caused a number of biochemical changes consistent with choline deficiency in mice. 74 Hepatic concentrations of S-adenosymethionine decreased. Biochemical changes were seen without fatty livers, an observation often associated with choline deficiency.
The dose–response, reversibility, and strain dependence of diethanolamine effects on hepatic choline deficiency were studied. 74 B6C3F1 mice were dosed dermally with diethanolamine in ethanol for 4 weeks. Control animals were either not dosed or dosed with ethanol only. The pattern of changes observed in choline metabolites after diethanolamine treatment was very similar to that observed in choline-deficient mice, and the NOEL for diethanolamine-induced changes in choline homeostasis was 10 mg/kg/d. Fatty livers were not observed. The reactions were dose dependent and reversible. Strain dependency was evaluated by dosing male C57BL/6 mice with 0 or 160 mg diethanolamine/kg bw for a similar 4-week period. Hepatic choline deficiency was found to be strain dependent. Dermal application of 95% ethanol decreased hepatic betaine levels, suggesting that the use of ethanol as a vehicle for dermal application of diethanolamine could exacerbate the biochemical effects of diethanolamine.
Occupational Exposure
The National Institute for Occupational Safety and Health recommended exposure limits time-weighted average for diethanolamine is 3 ppm (15 mg/m3). 75 The Occupational Safety and Health Administration does not have a permissible exposure limit for diethanolamine.
Summary
This report assesses the safety of diethanolamine and 17 diethanolamine salts as used in cosmetics. The salts are expected to dissociate into diethanolamine and the corresponding acid. Diethanolamine typically contains some amount of ethanolamine or triethanolamine; according to 1 supplier, diethanolamine has a minimum purity of 99.0%, with 0.5% maximum ethanolamine and 0.5% maximum triethanolamine. N-Nitrosodiethanolamine may also be present in diethanolamine or diethanolamine-containing ingredients.
Diethanolamine functions in cosmetic formulations as a pH adjuster. While a few of the other ingredients function as a pH adjuster, the majority reportedly function as surfactants, emulsifying agents, viscosity increasing agents, hair or skin conditioning agents, foam boosters, or antistatic agents. In 2011, diethanolamine was reported to be used in 22 formulations at concentrations of 0.0008% to 0.3%. The highest leave-on concentration reported was 0.06%. In Europe, dialkanolamines and their salts (ie, diethanolamine and the acid salts) are on the list of substances which must not form part of the composition of cosmetic products. (Secondary amines are allowed as contaminants in raw materials at concentrations up to 0.5% in trialkylamine, trialkanolamines, and their salts and in monoalkylamines, monoalkanolamines, and their salts and at up to 5% in fatty acid dialkylamides and dialkanolamides; secondary amines are allowed at up to 0.5% in finished products containing dialkylamides and dialkanolamides.) In Canada, the use of dialkanolamines is prohibited, based on the European ruling.
In vitro absorption studies were performed using mouse, rat, and human skin. In in vitro studies using mouse and rat skin, 1.3% and 0.04%, respectively, of the applied dose of undiluted [14C]diethanolamine was absorbed. In studies using human skin samples, the absorption of undiluted diethanolamine, as well as concentrations of <1% diethanolamine in combination with fatty acid dialkanolamides, was less than 1% of the applied dose. Penetration of diethanolamine in aq solutions was greater than when diethanolamine was undiluted. In studies using human liver slices, diethanolamine was absorbed; the aq-extractable radioactivity was primarily unchanged diethanolamine, while analysis of the organic extracts suggested that diethanolamine was incorporated into ceramides and slowly methylated.
In dermal studies with diethanolamine, absorption through mouse skin was greater than through rat skin and absorption increased with duration of exposure. In the tissues, the liver generally had the greatest disposition of radioactivity. Urine was the principal route of elimination. In oral studies, diethanolamine accumulated in the tissues, with the greatest disposition being in the liver; radioactivity was primarily as unchanged diethanolamine. Urinary excretion was also primarily as unchanged diethanolamine. In a repeated-dose study, stead state for bioaccumulation occurred after 4 weeks; however, diethanolamine continued to bioaccumulate in blood throughout dosing.
Mice exposed orally to sodium nitrite were dosed orally and dermally with 4 mg/kg diethanolamine. A small amount of NDELA was formed following a single oral dose of diethanolamine. No NDELA was detected following dermal dosing with diethanolamine.
In acute oral studies on diethanolamine using groups of 2 to 3 guinea pigs, all survived dosing with 1 g/kg and none survived dosing with 3 g/kg diethanolamine gum arabic solution. In rats, LD50 values have been reported as 0.71 to 0.80 mL/kg for undiluted diethanolamine, 1.82 g/kg for aq diethanolamine, and 1.41 to 2.83 g/kg for 20% aq diethanolamine. In repeat dermal testing with diethanolamine in mice and/or rats, irritation was observed at the site of application site following unoccluded dosing with ≥125 mg/kg to rats and 630 mg/kg to mice. Increases in liver and kidney weights were observed in most studies, while decreases in bw were observed sporadically. In repeat oral testing with diethanolamine, increases in liver and kidney weights and decreases in bws were seen in mice and rats. Deaths, believed to be test article related, occurred in most of the studies and included a mouse given 100 mg/kg diethanolamine by gavage. In inhalation studies with diethanolamine in rats, liver and kidney weights were increased. In 13-week studies with ≤400 mg/m3 diethanolamine, irritation effects were observed in the larynx. The 90-day NOAEC was 1.5 mg/m3 diethanolamine.
In a developmental toxicity study in which gravid mice were dosed dermally with 20 to 320 mg/kg diethanolamine from day 6 of gestation through PND 21, no effects on skeletal formation were observed, but dose-dependent effects on some growth and developmental parameters were observed. In a study in which parental mice were treated dermally with diethanolamine for 4 weeks prior to dosing, sperm motility was decreased at all doses (20-320 mg/kg) in a dose-dependent manner. In rats and rabbits, dermal dosing with up to 1,500 mg/kg and 350 mg/kg diethanolamine, respectively, during gestation did not have any fetotoxic or teratogenic effects. The NOEL for embryonal/fetal toxicity was 380 mg/kg/d for rats and 350 mg/kg/d for rabbits.
In an oral developmental toxicity study in which rats were dosed with up to 1,200 mg/kg/d diethanolamine on days 6 to 15 of gestation, maternal mortality was observed at doses of ≥50 mg/kg; the NOEL for embryonal/fetal toxicity was 200 mg/kg/d. In a study in which gravid rats were dosed orally with up to 300 mg/kg/d diethanolamine, the dams of the 300 mg/kg group were killed due to excessive toxicity; the LD50 was calculated to be 218 mg/kg. The LOAEL for both maternal toxicity and teratogenicity was 125 mg/kg/d.
In a developmental toxicity study in which rats were exposed by inhalation to diethanolamine on days 6 to 15 of gestation, the NOAEC for both maternal and developmental toxicity was 0.05 mg/L and the NOAEC for teratogenicity was >0.2 mg/L.
Diethanolamine was nongenotoxic in a number of assays. Positive results were reported in an in vitro study assaying the induction of DNA strand breakage in isolated rat, hamster, and pig hepatocytes. Increases in DNA synthesis, in vitro, were reported in mouse and rat hepatocytes treated with DEA but not in human hepatocytes. Choline decreased the DEA-induced increase in DNA synthesis in the mouse and rat hepatocytes.
The carcinogenic potential of dermally applied diethanolamine was evaluated by the NTP in B6C3F1 mice and F344/N rats. Diethanolamine produced clear evidence of carcinogenic activity in male and female mice (40-160 mg/kg) based on increased incidences of liver neoplasms in males and females and increased incidences of renal tubule neoplasms in males; there was no evidence of carcinogenicity in male (16-64 mg/kg) and female rats (8-32 mg/kg). The IARC Working Group concluded that, in the absence of adequate epidemiological information, there was sufficient evidence of carcinogenicity in experimental animals for a classification of diethanolamine as possibly carcinogenic to humans. Because diethanolamine is not mutagenic or clastogenic, a nongenotoxic mode of tumorigenic action is indicated. A plausible mode of action for diethanolamine carcinogenicity in rodents involves cellular choline deficiency.
Undiluted diethanolamine was moderately irritating to rabbit skin. In humans, a hair formulation containing 1.6% diethanolamine was a cumulative irritant. However, no irritation was reported during the induction phase of sensitization studies with formulations containing 1.6% to 2.7% diethanolamine. Diethanolamine was not a sensitizer in guinea pig maximization studies with intradermal and epicutaneous induction applications of 1% and 17.6%, respectively, or in humans tested with a formulation containing 2.7% diethanolamine that was tested at 100%. In an ocular irritation study using rabbits, a 30% aq solution of diethanolamine was essentially nonirritating and a 50% aq solution was a severe irritant.
Discussion
The CIR Expert Panel noted that few safety data were available for each of the 17 diethanolamine salts included in this group, and therefore utilized the data available for diethanolamine, in conjunction with previous safety assessments of the components of the diethanolamine salts, to assess the safety of these ingredients. The available data could be extrapolated to support the safety of most of these acid salts because they are expected to dissociate into diethanolamine and the corresponding acid and due to the structural similarities of the acid component with previously assessed components.
Dermal carcinogenicity studies performed on diethanolamine by the NTP reported “clear evidence of carcinogenic activity” in male and female B6C3F1 mice, based on increased incidences of liver neoplasms in males and females and increased incidences of renal tubule neoplasms in male mice. However, there was “no evidence of carcinogenic activity” following dermal application of diethanolamine to male and female F344/N rats. Diethanolamine-induced choline deficiency has been postulated to explain the hepatocarcinomas found in the mice, based on reports that diethanolamine exposure leads to choline deficiency in rodents and choline deficiency causes liver tumors in rodents. Humans are much less susceptible to the effects of choline deficiency than rodents. Although this does not explain the differences between rats and mice treated with diethanolamine, an additional factor, dermal penetration, may. The dermal penetration of diethanolamine was higher through mouse skin than through rat or human skin. Thus, the amount of diethanolamine absorbed across rat skin may have been insufficient to substantially perturb choline homeostasis in the rats, which could be another important reason why liver tumors were observed in the mice but not in the rats in the NTP study. Given that nongenotoxic mechanisms of action have been postulated for diethanolamine-induced carcinogenicity in animal studies, the dermal penetration of diethanolamine is sufficiently well-characterized, the concentration of use of diethanolamine is quite low (≤0.06% in leave-on products), and the hepatocarcinogenicity in mice reported in the NTP study are considered to have little relevance to the safety of use of diethanolamine in personal care products.
Further, renal lesions have been reported in rats fed choline-deficient diets. Although the NTP study reported renal neoplasms in male mice (adenoma or adenoma and carcinoma combined) and not in female mice or male or female rats, the cytotoxic effects (and the regenerative mechanisms stimulated as a result) of diethanolamine-induced choline deficiency, in conjunction with the greater penetration through mouse skin, may explain the development of renal tumors seen in male mice, an end point, again, which has little relevance to the safety of diethanolamine in humans. If renal neoplasms in male mice are not caused by diethanolamine-induced choline deficiency, the Panel still believes that the use of diethanolamine in cosmetics does not pose a carcinogenic risk to humans. Not only is diethanolamine most likely carcinogenic in rodent studies by an epigenetic mechanism, but diethanolamine does not appear to penetrate human skin to any significant extent at concentrations relevant to human exposures from the use of personal care products.
The conversion (nitrosation) of secondary amines, such as diethanolamine, into N-nitrosamines that may be carcinogenic also is a concern. It is because of this concern that dialkanolamines and their salts are prohibited for use by the EC and Canada. The Panel has chosen not to prohibit the use of these ingredients but instead state that products containing diethanolamine and its salts should be formulated to avoid the formation of nitrosamines.
The Panel was concerned that the potential exists for dermal irritation with the use of products formulated using diethanolamine or its salts. The Panel specified that products containing diethanolamine or its salts must be formulated to be nonirritating.
Diethanolamine is used in cosmetic products that may be inhaled during use. In practice, however, the particle sizes produced by cosmetic aerosols are not respirable. Additionally, the safety of use of diethanolamine in products that may be inhaled is supported by the inhalation studies in animals included in this report. Although limits exist for occupational exposure to diethanolamine, exposure via a cosmetic product is not expected to be of the same level as occupational exposure of the individual chemical.
Conclusion
The CIR Expert Panel concluded that diethanolamine and the 16 related salts listed below are safe in the present practices of use and concentration when formulated to be nonirritating. The Expert Panel cautions that ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed. Diethanolamine Diethanolamine bisulfate* DEA-C12-13 alkyl sulfate* DEA-C12-15 alkyl sulfate* DEA-C12-13 pareth-3 sulfate* DEA-cetyl sulfate* DEA-cocoamphodipropionate* DEA-dodecylbenzenesulfonate* DEA-iostearate* DEA-laureth sulfate DEA-lauryl sulfate DEA-linoleate DEA-methyl myristate sulfonate* DEA-myreth sulfate* DEA-myristate* DEA-myristyl sulfate* DEA stearate
*Were ingredients in this group not in current use to be used in the future; the expectation is that they would be used at concentrations that would be nonirritating in formulation.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Monice M. Fiume contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. Bart Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Lillian J. Gill, F. Alan Andersen, Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill, Curtis D. Klaassen, Daniel C. Liebler, James G. Marks, Ronald C. Shank, and Paul W. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
