Abstract
Salt forms of pharmaceutical compounds can have unique pharmacokinetic and toxicity properties. MDV1634 was evaluated for neurology indication and also demonstrated blood pressure (BP)-lowering effects in nonclinical studies. During the chemistry manufacturing campaign, 2 salt forms, dihydrochloride (2HCl) and maleate (MAL), which improved chemical stability and water solubility of the free base were identified. MDV1634.MAL showed better chemical attributes and was evaluated in toxicology studies for further development. A 28-day oral toxicity study in dogs with MDV1634.MAL demonstrated partially reversible renal toxicity. Although MAL salt is generally regarded as safe, renal toxicity is sometimes observed in rats and dogs. To evaluate contribution of each salt form to renal toxicity and BP lowering, an additional 28-day study was conducted with MDV1634.2HCL and MDV1634.MAL, which included toxicokinetics, continuous BP measurement in a subset of dogs, and sensitive urinary biomarker evaluation for temporal monitorability and reversibility of potential renal findings. In the repeat study, both salt forms showed similar exposures during the dosing period, but renal tubular toxicity was observed only with MDV1634.MAL and not with MDV1634.2HCl. The renal findings with MDV1634.MAL included early urinary biomarker changes (increase in albumin, clusterin, β2 microglobulin, and neutrophil gelatinase-associated lipocalin); elevations in serum blood urea nitrogen and creatinine; and microscopic findings of partially reversible tubular basophilia, single cell necrosis, pigmentation, and mineralization. The renal findings in contrast to the BP findings were MAL-specific and considered not related to MDV1634, thereby under scoring the importance of salt forms in pharmaceutical development.
Introduction
The contribution of the salt component during safety assessment of a new chemical entity (NCE) can be challenging. Selection of the appropriate salt form early in an NCE development can improve the chances of successful commercialization. 1 The salt form can influence the physicochemical properties of an NCE, which may in turn affect its pharmacokinetics, toxicity, and impurity profiles as well as the potential route of administration. 2,3 MDV1634 is an NCE that was evaluated for neurological indication. During the chemistry manufacturing campaign (CMC), 2 different salt forms of free base, dihydrochloride (2HCl) and maleate (MAL), were evaluated with a goal of improving the chemical stability and water solubility of the free base. MDV1634.MAL showed better chemical attributes compared to MDV1634.2HCl, and therefore the MAL salt, which is generally regarded as safe (GRAS), was synthesized for evaluation in the Good Laboratory Practices (GLPs) oral toxicity studies. A standard 28-day GLP oral toxicity study with 4-week recovery was conducted with the vehicle and MDV1634.MAL (3 doses) in beagle dogs (both sexes). Although MDV1634 was clinically well tolerated at all doses, there were MDV1634-related renal microscopic findings in both sexes (not seen in the vehicle group) at the scheduled end of dose (EOD) necropsy, including minimal/mild tubular basophilia, mild tubular degeneration/necrosis, and minimal hyaline casts at the mid and high doses in the absence of overt clinical pathology changes. The kidney findings were reversible in most dogs at the end of 4-week recovery, and the degeneration/necrosis was only partially reversible in 1 male dog in the high-dose group at 30 mg/kg twice daily. Since there were no clinical pathology correlates for the microscopic tubular findings, there were regulatory concerns for lack of monitorability of potential renal toxicity in patients during clinical development of the compound.
Although MAL salt is GRAS, MAL-specific renal tubular toxicity has been observed in nonclinical studies in dogs, where the MAL content was directly correlated with the toxicity 4 –7 and in rats at higher MAL doses. 8 –11 In addition, there are reports where maleic acid is directly correlated with the induction of renal tubular necrosis in both rats 12 and dogs. 13,14 In a study with pravadoline MAL (WIN 48098-6), the maleic acid moiety and not pravadoline itself was linked to WIN 48098-6-induced renal tubular necrosis in dogs. Pravadoline ethanesulfonate salt at comparable systemic exposures as the MAL salt did not cause the renal toxicity. 14
Alternately, the renal toxicity seen in dogs with MDV1634.MAL could be a secondary effect of the MDV1634-induced hypotension. In preliminary studies (internal data), dose-dependent blood pressure (BP) lowering was observed with MDV1634.MAL following administration of a single dose to dogs (0, 2, 20, and 60 mg/kg) in 2 cardiovascular telemetry studies, and in spontaneously hypertensive Dahl rats in a pharmacology study (0, 6, 20, and 40 mg/kg). Furthermore, evaluation of the secondary pharmacology profile in a panel of 126 targets (receptors for neurotransmitters, peptides, steroids, ion channels, enzymes, and transporters) demonstrated that the mechanism for BP lowering can be attributed to the moderate to high affinity antagonism by MDV1634 of the α2-adrenergic receptors (αAdr2C: cell-based functional IC50 281 nM; αAdr2B: cell-based functional IC50 24 nM) receptors. The α-Adr receptors, which are expressed in multiple tissues (brain, kidney, heart, and vasculature) have overall effects on BP and blood flow by affecting complex responses such as presynaptic inhibition of neurotransmitter release, diminished sympathetic efferent traffic, vasodilation, and vasoconstriction. 15 The hypotensive effects caused by αAdr2B and 2C receptor antagonism has the potential to also induce indirect adverse effects on the renal tubular cells resulting in secondary renal toxicity. Since the effect of MDV1634.MAL on BP lowering was already confirmed in the single dose telemetry and in the hypertensive rat pharmacology studies, the dose–response effect of MDV1634.2HCl on BP was further evaluated (dose response) in jacketed external telemetry (JET)-implanted male dogs in the repeat 28-day study.
The current repeat 28-day GLP oral toxicity study was therefore conducted using the 2 salt forms, MDV1634.MAL (tested at 30 mg/kg twice daily) and MDV1634.2HCl (tested at 3, 10, and 30 mg/kg twice daily), to determine whether the renal toxicity seen in dogs was MDV1634-specific, MAL-specific, or an indirect effect of BP lowering. To monitor early signals of renal toxicity (tubular or glomerular) and correlate with the observed clinical (repeat measurements) and microscopic pathology changes, sensitive urinary markers of renal toxicity (albumin [ALB], clusterin (CLU), β2 microglobulin, kidney injury molecule [KIM-1], and neutrophil gelatinase-associated lipocalin [NGAL]) were evaluated, and cystatin C level in the serum were measured. The repeat study also incorporated a more extended recovery period (6 weeks), since the 4-week recovery period in the original 28-day study demonstrated partial reversibility of the microscopic renal findings. This follow-up 28-day study in dogs has confirmed that the renal toxicity observed in the initial 28-day toxicity study was due to maleic acid content in the MDV1634.MAL salt form. The levels of maleic acid that are associated with the renal findings in dogs 14 are present in the same general range in MDV1634.MAL. Corresponding findings were not observed with the MDV1634.2HCL at similar doses and comparable exposures. Although MAL salt forms of pharmaceuticals are GRAS, the content of maleic acid in any MAL salt formulation should be carefully evaluated during pharmaceutical development.
Materials and Methods
Test Articles and Dose Formulation
MDV1634 dihydrochloride (MDV1634.2HCl) and MDV1634 maleate (MDV1634.MAL) salts were manufactured by Wilmington PharmaTech (Newark, Delaware) with Medivation Inc’s direction. Both salts were greater than 99% pure as analyzed by high performance liquid chromatography, with MDV1634 freebase quantified at 80.7% and 75.6% in the 2HCl and MAL salt forms, respectively. All doses used for the experiments represent free base concentration of the test compounds. MDV1634.2HCl and MDV1634.MAL doses were formulated weekly in ultrapure water and 0.5% (w/v) 4,000 cps aqueous methylcellulose respectively. Analytical data validated the stability of the dosing formulations for weekly preparations and for the concentration range and storage conditions used in the study. Both test compounds and formulations were stored at 4°C, protected from light, and appropriate handling precautions and personal protective equipment were used during preparation of dose formulations. All dose formulations met specifications for concentration of both salt forms and additional homogeneity specifications for the MDV1634.MAL suspension. Prior to dosing, formulations were removed from the refrigerator, allowed to warm to room temperature while being stirred for at least 30 minutes, and stirred continuously during dosing.
MDV1634.MAL: 28-Day Oral Toxicity Study With 28-Day Recovery (Original Study)
The study was conducted in accordance with United States Food and Drug Administration, GLP regulations.
Animal care and use
The original oral toxicity study was conducted at MPI Research, Mattawan, Michigan; all animals were experimentally naive, purpose-bred beagle dogs (6.5-7.5 months of age) received from Marshall BioResources, North Rose, New York. All study procedures were conducted according to a written study protocol, which was in strict compliance with the national legal regulations on the animal welfare and accepted animal welfare standards and was approved by the CRO’s Institutional Animal Care and Use Committee (IACUC).
Study design
Animals were acclimated for a minimum of 10 days prior to initiation of the study and were randomized to study groups to achieve similar (±20%) group mean body weights. Following acclimatization, 18 males (6.8-8.7 kg) and 18 females (5.2-6.7 kg) were assigned to control (vehicle) or the MDV1634.MAL treatment groups (main study and recovery) and individually housed in an environmentally controlled room. Main study dogs (n =3 per sex/group) were dosed with MDV1634.MAL suspension at 0 (0.5% methylcellulose [w/v] in deionized water), 6, 20, or 60 mg/kg/d (Groups 1-4), administered twice daily in equally divided doses (3, 10, or 30 mg/kg twice daily, 3 mL/kg, 6 hours apart) for 28 days, and sent to necropsy; additional animals (n =2 per sex) were dosed with MDV1634.MAL at 0, 20 or 60 mg/kg/d (groups 1, 3, and 4) and allowed an additional 4-week, drug-free recovery period (Supplemental Table 1). Parameters assessed during the in-life phase included clinical signs, mortality and morbidity, body weight and food consumption, ophthalmology, electrocardiology and cardiovascular evaluation, clinical pathology (chemistry, hematology, coagulation, and urinalysis), gross pathology, organ weights, histopathology, and toxicokinetic (TK) parameters. Dogs were offered Block Lab Diet (Certified Canine Diet #5007, PMI Nutrition International, Inc) and tap water ad libitum, except during designated period of the study.
Toxicokinetic Summary of MDV1634.MAL in the Original 28-Day Study.
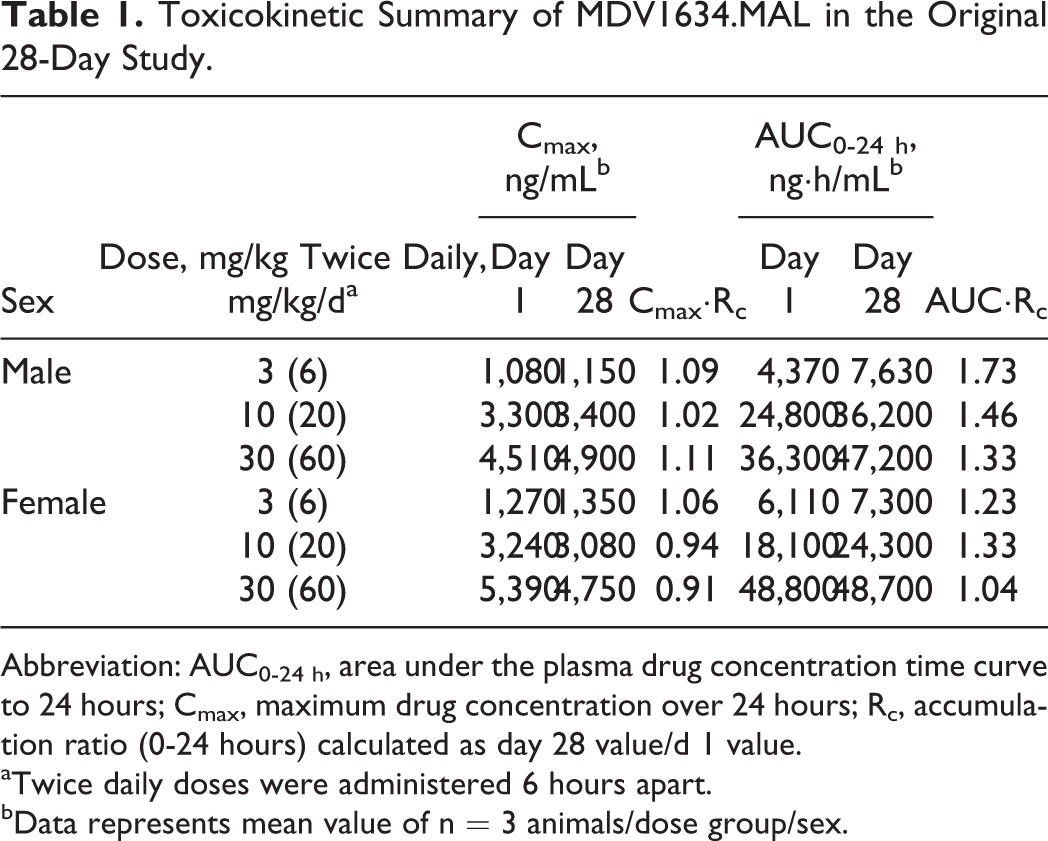
Abbreviation: AUC0-24 h, area under the plasma drug concentration time curve to 24 hours; Cmax, maximum drug concentration over 24 hours; Rc, accumulation ratio (0-24 hours) calculated as day 28 value/d 1 value.
aTwice daily doses were administered 6 hours apart.
bData represents mean value of n = 3 animals/dose group/sex.
Electrocardiographic evaluation
All animals received an electrocardiographic (ECG) examination prior to randomization, at predose, at 1 to 2 hours postdose on day 1 and during the last week of dosing, and once prior to the recovery necropsy. Standard ECGs (10 Lead) were recorded at 50 mm/s, and appropriate leads were used for the RR, PR, and QT intervals; QRS durations were measured and heart rate (HR) was determined. Corrected QT (QTc) interval was calculated using a Fridericia correction procedure, and all tracings were evaluated and reported by a consulting veterinary cardiologist.
All Main and Recovery Study animals survived until scheduled euthanasia and had a terminal body weight recorded prior to necropsy. Animals were fasted overnight before their scheduled necropsy. A sedative, ketamine HCl for injection, and dexmedetomidine, was administered by intramuscular injection before animals were transported from the animal room to the necropsy area. The animals underwent exsanguination by incision of the axillary or femoral arteries following anesthesia by intravenous injection of sodium pentobarbital.
Clinical pathology assessment
Clinical pathology evaluations were conducted on all animals prior to initiation of dosing, at the EOD and in selected animals, at the end of recovery (EOR). The animals had access to drinking water but were fasted overnight prior to scheduled sample collection. Blood samples (approximately 4.8-5.8 mL) were collected from the jugular vein into tubes containing K3EDTA for hematology; sodium citrate plasma was used for coagulation analysis, and serum was collected for the clinical chemistry evaluation. Urine samples were collected using steel pans placed under the cages for at least 16 hours, and complete urinalysis was performed.
Histopathology
Following necropsy, tissues and organs from a standard panel were collected, weighed, fixed, embedded in paraffin, sectioned, mounted on glass slides, and stained with hematoxylin and eosin (H&E). Complete list of tissues (58 tissues) was evaluated in the study. All tissues were placed in a neutral-buffered formalin for fixation except the eye (including optic nerve) and testes, which were fixed using a modified Davidson’s fixative. Eyes and testes were placed into formalin after fixation, and formalin was infused into the lung via the trachea. Histopathological analysis was conducted by a board-certified veterinary pathologist, and the findings were independently peer reviewed by a second certified pathologist.
Bioanalytical and toxicokinetic analysis
Blood samples (approximately 0.5-1 mL) were collected in K2 EDTA tubes from all animals via the jugular vein for determination of MDV1634 plasma concentrations. Samples were collected at predose and at 0.5, 1, 2, 4, 6 (prior to the second dose), 7, 10 (1 and 4 hours post second dose, respectively), and 24 hours postdose on days 1 and 28. Only the samples at 1 and 7 hours postdose (approximately t max after the first and second doses, respectively) were analyzed for animals from the control group (0 mg/kg dose). The blood samples were collected on wet ice and centrifuged under refrigeration within 1 hour of collection. The animals were not fasted prior to blood collection, except for those intervals that coincided with clinical pathology collections. All animals were bled at each time point, always starting with the control animals and progressing through the dose levels to minimize the potential for inadvertent contamination. Samples from control animals and treated groups were centrifuged/processed in a separate processing room to minimize potential for inadvertent contamination. The samples were contained in tightly capped, prelabeled, plastic vials and were stored on dry ice prior to being stored frozen at −60°C to −90°C until shipped on to Tandem Labs, Salt Lake City, Utah, for analysis of plasma concentrations of MDV1634. All analytical work was conducted by Tandem Labs using an analytical method developed and validated by that laboratory.
The toxicokinetic parameters for MDV1634 were determined by Covance Laboratories, Madison, Wisconsin, using concentration–time data from individual animals. Noncompartmental analysis in Phoenix WinNonlin (Pharsight Corporation, version 6.2.1, Sunnyvale, California) was used to calculate the TK parameters from the individual plasma MDV1634 concentration–time data for males and females. Nominal doses were used throughout the analysis. Dose-normalized TK parameters, ratios, and descriptive statistics were calculated using WinNonlin and Microsoft Excel (version 11.0). Concentration values below the lower limit of quantitation (LLOQ; 1.00 ng/mL) were treated as 0 for descriptive statistics and TK analysis. The TK parameters included Cmax (peak concentration from the 0 to 24 hours), tmax (time to peak concentration from 0-24 hours), AUC0-24 h (area under the concentration-time curve from 0-24 hours), t1/2 (elimination half-life following the first dose of the day, prior to the second daily dose), and Rc (accumulation ratio; Cmax or AUC0-24 h value on day 28 relative to the corresponding Cmax or AUC0-24 h value on day 1).
Statistical analysis
Group pairwise comparison (Levene/analysis of variance-Dunnett/Welch) tests were used to evaluate significant differences in body weight, food consumption, hematology (except leukocyte counts), coagulation, clinical chemistry, ECG intervals, and organ weights (absolute; relative to body and brain weights). Log transformation/group pairwise comparisons were used for total and differential leukocyte counts. Rank transformation followed by Dunnett test was used for urinalysis (urine volume, specific gravity, and pH changes).
MDV1634.2HCl and MDV1634.MAL: 28-Day Oral Toxicity Study With 6-Week Recovery (Current Repeat 28-Day Study)
The repeat study was performed in accordance with the Organization for Economic Co-operation and Development (OECD) principles of GLP and as accepted by regulatory authorities throughout the European Union, United States of America (Food and Drug Administration), Japan (Ministry of Health, Labour and Welfare), and other countries that are signatories to the OECD Mutual Acceptance of Data Agreement.
Animal care and use
The repeat study was conducted at Charles River Laboratories (CRL), Preclinical Services, Montreal; all animals were experimentally naive, purpose-bred beagle dogs (approximately 9 months of age) received from Marshall BioResources, North Rose, New York. All study procedures were conducted according to a written study protocol, which was in strict compliance with the national legal regulations on the animal welfare and accepted animal welfare standards and was approved by the CRO’s IACUC.
Study design
Animals were acclimated for a minimum of 41 days prior to study start and were randomized to study groups to achieve similar group mean body weights. The extended acclimatization period in the repeat 28-day study (compared to 10 days in the original study) was based on the Standard Operating Procedures and IACUC guidance at CRL, which required a minimum of 30 days. Following acclimatization, 22 males (7.3-10.3 kg) and 22 females (5.8-8.1 kg) were assigned to control (vehicle; ultrapure water), MDV1634.2HCl (solution), or MDV1634.MAL suspension (vehicle: 0.5% aqueous methylcellulose 4,000 cps) treatment groups (main study and recovery) and socially housed (up to 3 dogs/sex, same treatment group) in an environmentally controlled room. Main study dogs (n =3 per sex) were dosed by oral gavage at 0, 6, 20, or 60 mg/kg/d (groups 1-4) with MDV1634.2HCl solution (0, 3, 10, or 30 mg/kg twice daily, 5 mL/kg, 6 hours apart) or with MDV1634.MAL suspension at 60 mg/kg/d (30 mg/kg twice daily, 5 mL/kg, 6 hours apart) for 28 days and sent to necropsy at the EOD; additional animals (n =2 per sex) were dosed at 0, 60 mg/kg/d of MDV1634.2HCl or 60 mg/kg/d of MDV1634.MAL and allowed an additional 6-week drug-free recovery period after treatment. Parameters assessed during the in-life phase included mortality and morbidity, clinical signs, body weight, food consumption, ophthalmology, electrocardiology and cardiovascular assessments (on the platform and continuous [JET monitoring on male dogs in the vehicle and MDV1634.2HCl dose groups]), clinical pathology (hematology, coagulation, and chemistry), urinalysis, biomarkers of kidney injury (urinary and serum), gross pathology, organ weights, histopathology, and TK parameters. All animals were provided a daily ration of 300 g PMI Nutrition International Certified Canine Chow No. 5007 (25% protein). The food ration was offered up to 1 hour before the first daily dosing. The feed was left in the dog’s cage for a period of up to 7 hours and then removed (prior to the second daily dosing, which was 6 hours following the first daily dosing). Due to decreases in food consumption, a can of Hill’s Prescription Diet a/d Canine food was mixed with the regular chow as of day 6 (1 male) or day 8 (remaining males and all females) of the dosing phase for the duration of the dosing and recovery periods.
Electrocardiographic evaluation
Electrocardiographic measurements were recorded in all animals on the platform once during prestudy (baseline), at 1 to 3 hours after PM dosing on day 3 and week 4 of the dosing period, and during the last week of the recovery period. Each dog (all groups) had tracings recorded at standard recording speed of 50 mm/s and standard sensitivity set at 10 mm/mV, using the limb leads I, II, III, aVR (augmented unipolar right arm lead), aVL (augmented unipolar left arm lead), and aVF (augmented unipolar left leg lead). All waveforms were qualitatively evaluated to detect rhythm or conduction disturbances or other abnormalities of the P-QRS-T waves. Also, quantitative evaluation consisted of the manual measurement of the HR and/or RR interval, PR and QT intervals, calculation of the QTc (using Van de Water formula) as well as QRS complex duration.
Quantitative and qualitative cardiovascular assessment using JET
Quantitative ECG, BP, and HR measurements were performed on the main study and recovery male dogs (group 1-4). Assigned animals were acclimated to the protective jackets for a minimum of 3 days prior to each data collection period. Monitoring frequency was at predose (baseline), on days 7, 14, and 25 during the dosing period, and once toward the end of the recovery period. Monitoring occasions were repeated as deemed necessary following ECG review. During the dosing period, duration of monitoring was for 2 hours prior to the start of the first daily dose and then for at least 18 hours following the first daily dose (recorded at a 30-second logging rate). Assessed parameters included HR (derived from BP and ECG waveforms), PR-, QRS-, QT-, RR-interval duration, TCt, and systemic arterial BP (systolic, diastolic, mean, and pulse pressures). Only BP-derived HRs were reported. The RR interval and TCt data were used for calculation purposes only and were not reported. On each occasion, ECG examinations were performed using nonsurgical (external ECG system with surface electrodes) telemetry in modified lead II configurations, and BP and HR measurements were performed using telemetry transmitters.
Qualitative ECG assessments were also conducted during each jacketed telemetry monitoring occasion on the main study control and high dose males only. ECG was evaluated twice prior to each dose (at least 30 minutes apart) and at 1, 3, 7 (1 hour after the second daily dose), and 18 hours (±30 minutes) postdose following the first daily dose. The duration of tracing was for at least 30 seconds at each time point. All waveforms were qualitatively evaluated to detect rhythm or conduction disturbances or other abnormalities of the P-QRS-T waves.
Ophthalmoscopic evaluation
Examinations were conducted on all animals prior to initiation of dosing and during the last week of the dosing period. All animals were subjected to funduscopic (indirect ophthalmoscopy) and biomicroscopic (slit lamp) examinations using 1% tropicamide as the mydriatic solution.
Clinical pathology assessment
Clinical pathology evaluations were conducted on all animals at predose, weeks 2, 3, and at the EOD and EOR. The animals had access to drinking water but were fasted overnight prior to scheduled sample collection. Blood samples were collected from the jugular vein into tubes containing EDTA for hematology, sodium citrate for plasma (coagulation chemistry measurements), and serum separators for the clinical chemistry and serum biomarker evaluation. About 2 mL blood intended for serum biomarker evaluation were kept at room temperature for at least 30 minutes prior to centrifugation for 10 minutes in a refrigerated centrifuge (set at 4°C) at 1,400 g. Serum samples were split into 2 × 250 µL aliquots and were stored in a freezer set to maintain −80°C until analysis. Cystatin C, a kidney injury biomarker, was evaluated in the serum. Serum clinical chemistry parameters including Na, K, Cl, Ca, inorganic P, glucose, blood urea nitrogen (BUN), total cholesterol, triglycerides, total bilirubin, total protein, ALB, globulin (GLOB), ALB: globulin ratio, creatinine (CREAT), creatine kinase, alkaline phosphatase, aspartate amino transferase (AST), alanine aminotransferase (ALT), and γ glutamyl transferase were assessed.
Urinalysis and urine kidney function biomarkers
Urine was collected from individual animals at predose, weeks 1, 2, and EOD, and EOR for at least 16 hours, chilled on wet ice, and stored frozen (in a freezer set to maintain −80°C) for analysis (conducted following the last sample collection). Urinalysis parameters included color, appearance/clarity, specific gravity, volume, pH, protein, glucose, bilirubin, ketones, blood, and CREAT. The urinary biomarkers included ALB, β2 microglobulin (β2 m), CLU, KIM-1, and NGAL. All kidney function biomarkers were reported normalized to urine CREAT (analyte/mg CREAT). Clusterin, KIM-1, and NGAL were assayed together using a multiplex Luminex method using Canine Kidney Toxicity Magnetic Bead Panel 1 kit (Millipore, Billerica, Massachusetts). The range of detection (lower limit to upper limit of quantitation) for KIM-1, NGAL, and CLU was 3.04 to 700 pg/mL, 208.33 pg/mL to 20,000 pg/mL, and 1,446.76 pg/mL to 1,000,000 pg/mL, respectively. Albumin and β2 m were assayed together using a multiplex Luminex method using Canine Kidney Toxicity Magnetic Bead Panel 2 kit (Millipore). Range of detection for ALB was 100.3 to 12,500 ng/mL and for β2 m was 0.2 to 25 ng/mL.
Bioanalytical and Toxicokinetic Analysis
Plasma levels of MDV1634 were measured in animals from all dose groups on days 1 and 28 of the dosing phase. For MDV1634 exposure analysis, about 0.7 mL blood was collected by jugular venipuncture into chilled tubes containing K2EDTA at predose, 0.5, 1, 3 and 6, 7, 10 and 24 hours post first dose on days 1 and 28 during the dosing period, and processed to plasma by centrifugation at 1,200 g for 10 minutes in a centrifuge set to 4°C. All blood samples through 6 hours postdose were collected within 5 minutes of the intended time, and all samples from 7 to 24 hours postdose were collected within 15 minutes of the intended time. All analysis for MDV1634 was conducted at Tandem Labs (Salt Lake City, Utah) using validated LC/MS/MS procedures (TSLM11055). Data collection was performed using Analyst from AB Sciex, statistical analyses including regression analysis and descriptive statistics including arithmetic means and standard deviations, accuracy and precision were performed using Watson Laboratory Information Management System and Microsoft Excel.
Toxicokinetic evaluation was performed using noncompartmental analysis using Phoenix WinNonlin software (version 6.2.1; Pharsight Corporation, Sunnyvale, California). Toxicokinetic parameters evaluated included maximum concentration (Cmax[0-24 h], Camax [post first daily dose], C bmax [post second daily dose]), time of maximum concentration (T max, T amax, T bmax), half-life (t 1/2), and area under the curve (AUC0-24 h, AUC0-6 h, and AUC6-24 h).
Main and Recovery Study animals surviving until scheduled euthanasia had a terminal body weight recorded prior to necropsy. Animals were fasted overnight before their scheduled necropsy. A sedative, ketamine HCl for injection, and dexmedetomidine was administered by intramuscular injection before animals were transported from the animal room to the necropsy area. The animals underwent exsanguination by incision of the axillary or femoral arteries following anesthesia by intravenous injection of sodium pentobarbital.
Microscopic Pathology
Following necropsy, tissues and organs from a standard panel were collected, weighed, fixed in formalin, embedded in paraffin, sectioned, mounted on glass slides, and stained with H&E. Complete list of tissues (52 tissues) were evaluated in the study. All tissues were fixed in neutral-buffered formalin by immersion, except the eye (including optic nerve) and testis which were fixed using modified Davidson’s fixative; eyes and testes were transferred to formalin after fixation. In addition, formalin was infused into the lung via the trachea. Histopathological analysis was conducted by a board-certified veterinary pathologist, and the findings were independently peer reviewed by a second certified pathologist.
Results
28-Day Oral Toxicity Study with MDV1634.MAL with 28-Day Recovery (Original Study)
Toxicokinetics
The TK parameters for MDV1634 are summarized in Table 1. All dogs receiving MDV1634.MAL had sufficient exposure. MDV1634 Cmax exposures were generally dose proportional in both sexes from 3 to 10 mg/kg twice daily but less than dose proportional from 10 to 30 mg/kg twice daily. There was no apparent sex difference in exposure and no accumulation after repeat dosing for 28 days.
Histopathology Kidney Findings
At 10 and 30 mg/kg/d twice daily, MDV1634.MAL-related microscopic renal findings observed in both sexes included minimal to mild basophilic tubules, mild tubular degeneration/necrosis, and hyaline casts. There was a dose-dependent increase in the incidence of the tubular findings, which either affected approximately one-third of the nephrons at these doses (1/6 controls, 0/6 animals at 3 mg/kg twice daily, 3/6 animals at 10 mg/kg twice daily, and 5/6 animals at 30 mg/kg twice daily) or the findings were generally focal and affected only a few tubules when graded minimal. At 30 mg/kg twice daily, 1 male had mild basophilic tubules with multiple foci of affected tubules throughout the cortex. At the recovery necropsy (28-day [4 weeks] drug free), both males at 30 mg/kg twice daily had minimal basophilic tubules lined by thickened basement membrane with occasional, limited interstitial fibrosis surrounding the affected tubules; the findings were reversible in females at all doses. At 10 mg/kg twice daily, the findings were reversible in both sexes in recovery animals. There were no clinical pathology changes during the EOD or EOR evaluations that correlated with the microscopic findings. Based on the limited number of affected tubules and the lack of glomerular findings, the lack of changes in the clinical pathology parameters was not unexpected. The microscopic findings from representative sections are illustrated in Figure 1.
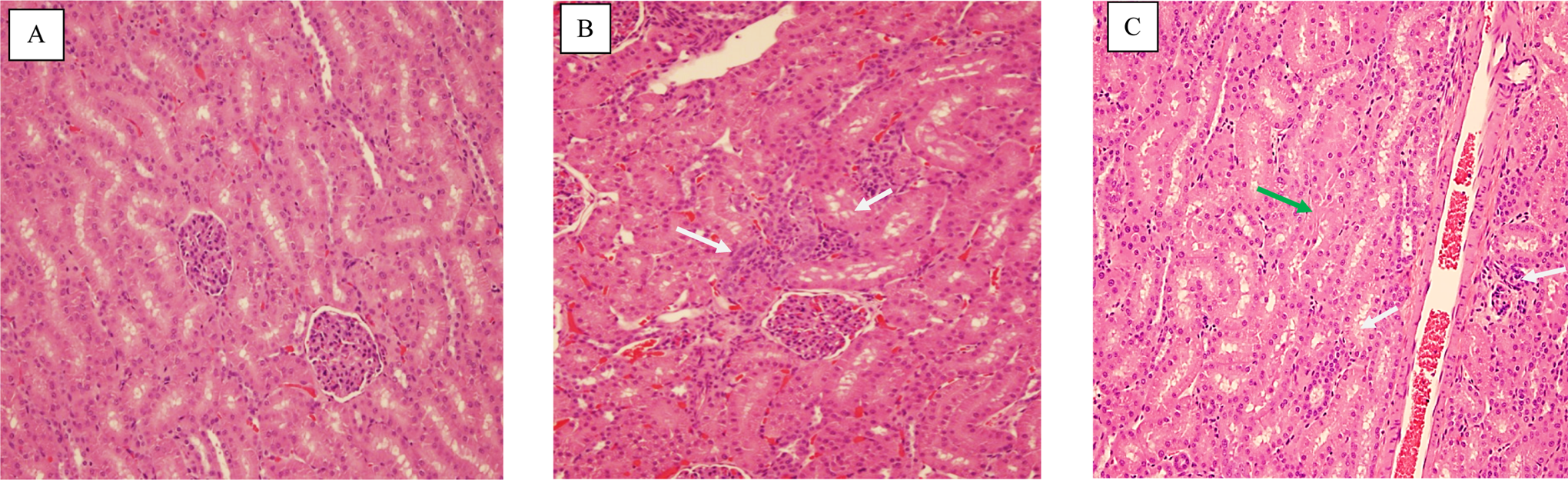
Microscopic renal changes following administration of vehicle or MDV1634.MAL for 28 days in dogs (initial study). Representative kidney micrographs from vehicle (A) or MDV1634.MAL-treated (B and C; 30 mg/kg twice daily) dogs following administration for 28 days (end of dose); the arrows demonstrate MDV1634-related tubular basophilia (B and C, white arrow) and necrosis/degeneration (C, green arrow).
28-Day Oral Toxicity Study With MDV1634.2HCl and MDV1634.MAL With 6-Week Recovery (Repeat 28-Day Study)
To evaluate whether the renal toxicity was MDV1634-specific, MAL-specific or a secondary effect of BP lowering, a repeat 28-day oral toxicity study was conducted in beagle dogs. The groups (1-5) included were a vehicle control, 3 doses of MDV1634.2HCl (3, 10, and 30 mg/kg, twice daily), and a single high dose of MDV1634.MAL (30 mg/kg, twice daily), respectively. To evaluate possible contribution of BP changes to the renal toxicity, the male dogs in the vehicle and MDV1634.2HCl dose groups were implanted with external telemetry jackets (JET implants), and the hemodynamic changes were measured over a 24-hour period on the selected days during the dosing period (Materials and Methods). Since MDV1634.MAL had already demonstrated BP lowering in a previous cardiovascular safety pharmacology study, it was expected that the MAL salt would have similar effects as the hydrochloride salt (MDV1634 exposure related) with respect to BP-lowering effects in the repeat 28-day study.
Toxicokinetics
All dogs receiving MDV1634 (2HCl and MAL) had sufficient exposure as demonstrated by TK assessments. There was no apparent sex difference in exposure and no evidence of accumulation after repeat administration of MDV1634.2HCl (3-30 mg/kg twice daily) or MDV1634.MAL (30 mg/kg twice daily) for 28 days. The sex-combined TK parameters for MDV1634 are summarized in Table 2. At 30 mg/kg twice daily, systemic exposure (Cmax and AUC0 - 24 h) was similar between the MDV1634.2HCl and MDV1634.MAL groups. Evaluation of the TK parameters in the 3 MDV1634.2HCl dose groups demonstrated that MDV1634 exposure increased with dose from 3 to 30 mg/kg twice daily, and the increases in AUC0-24 h values (but not Cmax) were generally slightly greater than dose proportional (Table 2).
Sex-Combined Toxicokinetic Summary of MDV1634.2HCl and MDV1634.MAL in the Repeat 28-Day Study.
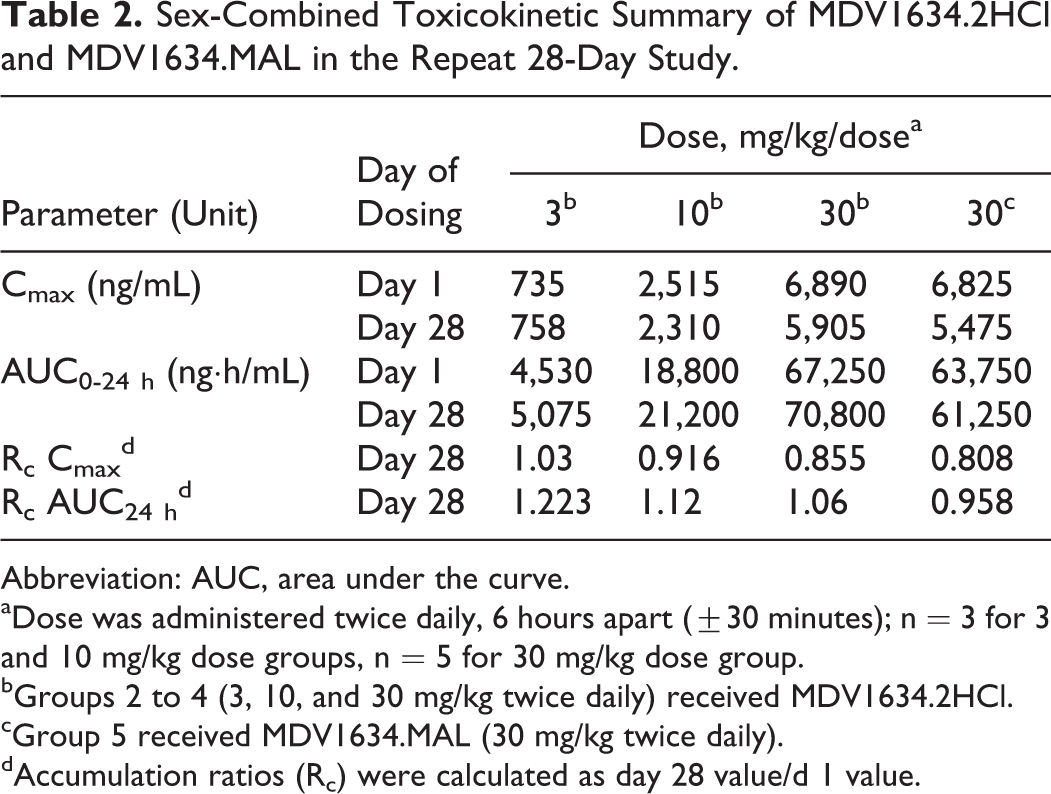
Abbreviation: AUC, area under the curve.
aDose was administered twice daily, 6 hours apart (±30 minutes); n = 3 for 3 and 10 mg/kg dose groups, n = 5 for 30 mg/kg dose group.
bGroups 2 to 4 (3, 10, and 30 mg/kg twice daily) received MDV1634.2HCl.
cGroup 5 received MDV1634.MAL (30 mg/kg twice daily).
dAccumulation ratios (Rc) were calculated as day 28 value/d 1 value.
Clinical observations
A summary of the clinical findings is presented in Table 3 and supports exposure to the compound in all dose groups administered MDV1634 (2HCl and MAL). MDV1634.2HCl was well tolerated at 3 mg/kg twice daily with no treatment-related clinical observations or ophthalmologic findings. At 10 mg/kg twice daily (MDV1634.2HCl), tremors and partly closed eyes were only sporadic (first 2 dosing days) in both sexes. At 30 mg/kg twice daily (MDV1634.2HCl and MDV1634.MAL), tremors and partly closed eyes were observed in most animals throughout the dosing period. Additional clinical signs at 30 mg/kg twice daily (both salts) included decreased activity (days 1-18), redness of eyes (days 1-49, also observed during recovery period), and protruding nictitating membrane (days 1-14). The clinical observation of protruding nictitating membrane (a manifestation of pain encountered in the eye of dogs) was not observed during the second half of the dosing period, had no histopathologic correlate in the eye at postdose necropsy, and no functional ocular changes were noted during the funduscopic (indirect ophthalmoscopy) and biomicroscopic (slit lamp) ophthalmologic examinations. There were no effects on body weight or food consumption during treatment or recovery periods in the study. The only noted exception was that 1 male in the MDV1634.MAL group had decreased food consumption (group mean change up to 38% on days 3-5); therefore, all dogs received dietary supplementation from day 8 of dosing.
Summary of Clinical Findings in Repeat 28-Day Study.
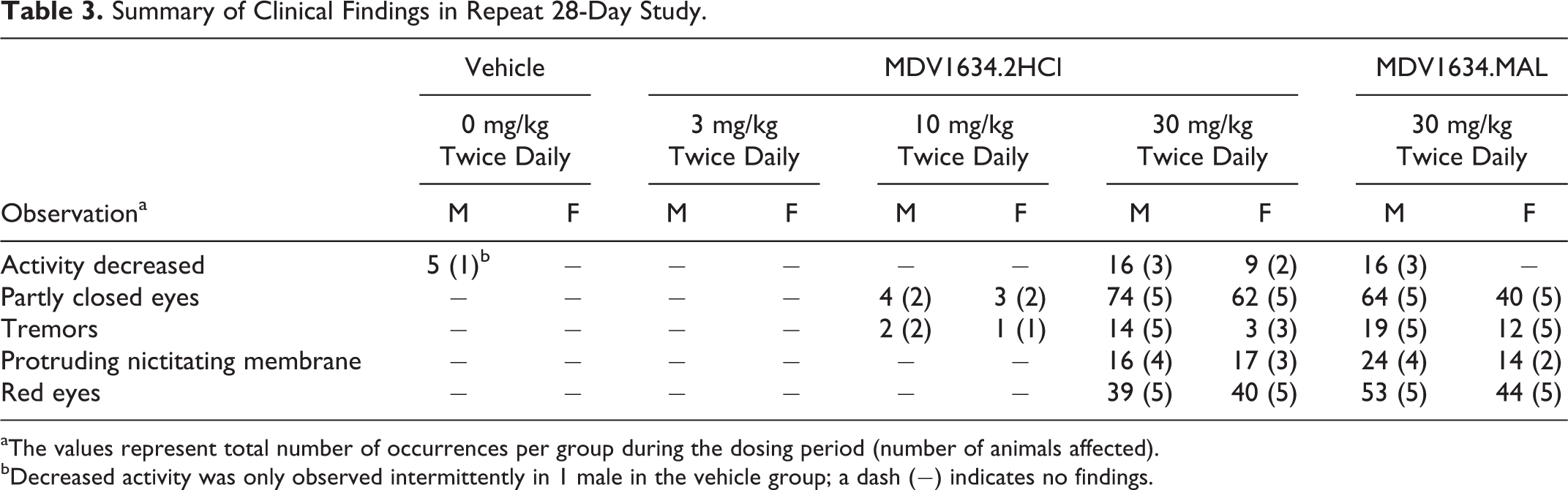
aThe values represent total number of occurrences per group during the dosing period (number of animals affected).
bDecreased activity was only observed intermittently in 1 male in the vehicle group; a dash (–) indicates no findings.
Cardiovascular Assessments in Repeat 28-Day Study
It was established in an earlier cardiovascular telemetry study that a single administration of MDV1634.MAL (6 mg/kg) decreased BP in dogs. To evaluate dose response and potential tolerability of the BP effect after repeat dosing, quantitative evaluation of BP and hemodynamic changes were performed in JET-instrumented male dogs (control and MDV1634.2HCl-treated) during the dosing period (at predose and on days 7, 14, and 25). At ≥10 mg/kg twice daily, there were MDV1634-related decreases in systolic BP with accompanying decreases in mean arterial pressure and pulse pressure throughout the dosing period. However, compensatory increases in HR were only noted during the day 7 (but not days 14 and 25) evaluation at 10 and 30 mg/kg twice daily. There were no additional ECG changes in the dogs based on the JET quantitative evaluation or additional qualitative cardiovascular evaluation that were conducted using external leads (day 3 and last week of dosing). A summary of the JET quantitative cardiovascular assessment is presented in Table 4.
Summary of JET Quantitative Cardiovascular Assessment in Male Dogs.
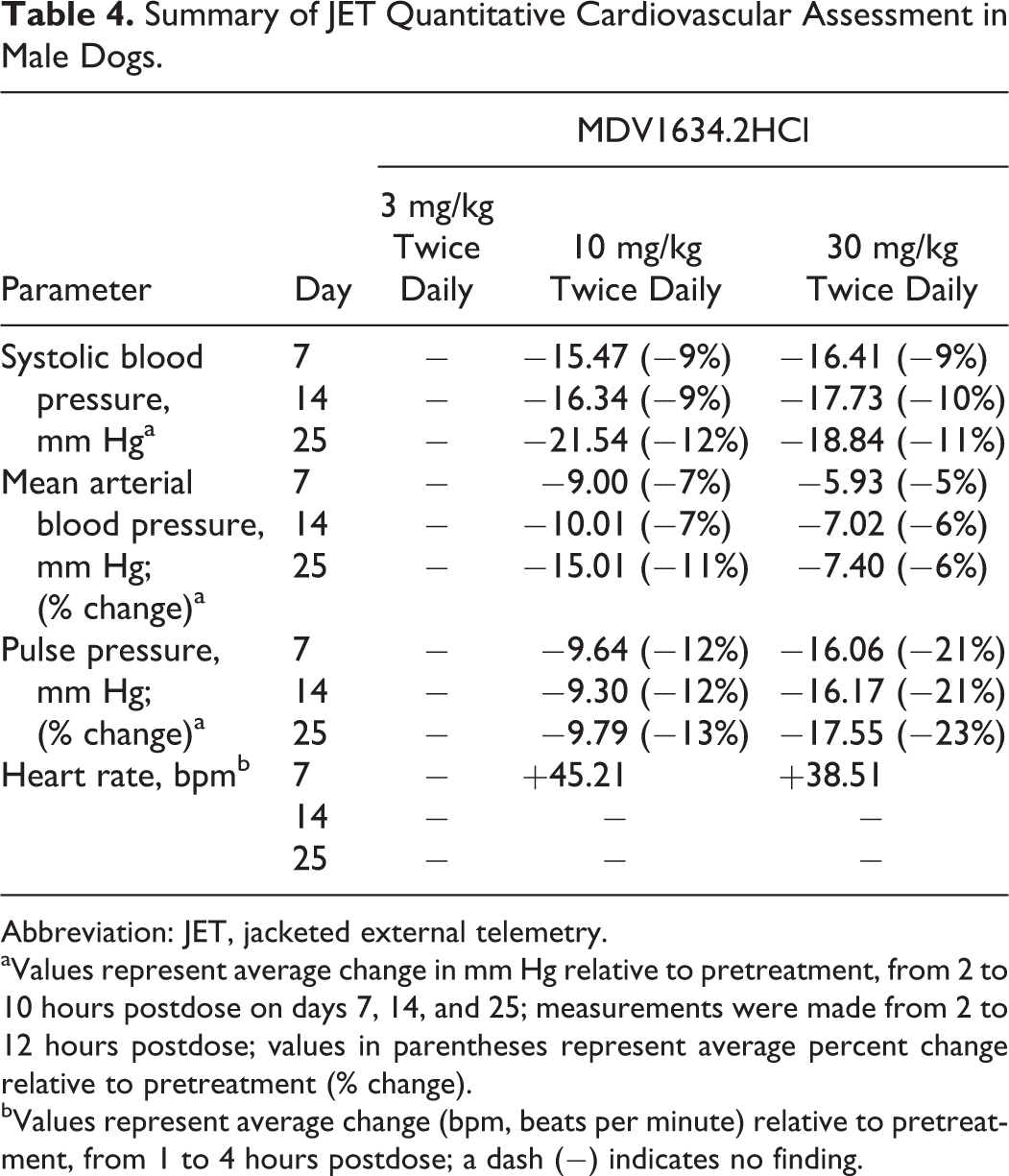
Abbreviation: JET, jacketed external telemetry.
aValues represent average change in mm Hg relative to pretreatment, from 2 to 10 hours postdose on days 7, 14, and 25; measurements were made from 2 to 12 hours postdose; values in parentheses represent average percent change relative to pretreatment (% change).
bValues represent average change (bpm, beats per minute) relative to pretreatment, from 1 to 4 hours postdose; a dash (–) indicates no finding.
Clinical Chemistry Changes in Repeat 28-Day Study
A summary of MDV1634-related serum clinical chemistry changes is presented in Table 5. At ≤10 mg/kg twice daily, there were no MDV1634.2HCl-related changes in clinical chemistry and at ≤30 mg/kg twice daily, there were no changes in hematology, coagulation, or urinalysis evaluations. At 30 mg/kg/d, MDV1634.2HCl-related changes (compared to pretreatment) included transient (week 2) increases (considered adverse) in AST (1.6-1.8× in 3/5 males and 1.6-2.8× in 2/5 females), although the changes did not have any microscopic correlate in histopathology evaluations. At 30 mg/kg/d, MDV1634.MAL-related changes (compared to pretreatment) at week 2 of dosing included minimal to mild increases in AST (1.8× in males and 1.6-2.3× in 2/5 females [persisted in week 3]), ALT (1.6× in males and 1.9× in females [persisted in week 3]), BUN (1.4-1.9× in 4/5 males [persisted in week 3] and 1.8× in 1/5 female), and CREAT (1.4-1.7× in 3/5 males and 1.5× in 1/5 females) and minimal to mild decreases in ALB (0.9× in 1/5 males and 0.9× in 1/5 females). At 30 mg/kg twice daily (MDV1634.MAL), 1 male dog presented with moderate increase in BUN (4.8×) and CREAT (4.5×) and mild decreases in ALB (0.9×), globulin (0.8×), and potassium (0.86×). The increases in BUN and CREAT were not associated with changes in urine specific gravity or urine volume and were consistent with decreased glomerular filtration, likely due to subclinical dehydration. Decreases in ALB correlated with an increased incidence of semiquantitative protein concentration that likely resulted from a loss of proteins in the urine. Decreases in potassium may have resulted from increased urinary loss. Overall the minimal to mild clinical pathology changes at 30 mg/kg twice daily in the MDV1634.2HCl or the MDV1634.MAL group did not present clear evidence of renal toxicity.
Summary of Clinical Chemistry Changes in Repeat 28-Day Study.a
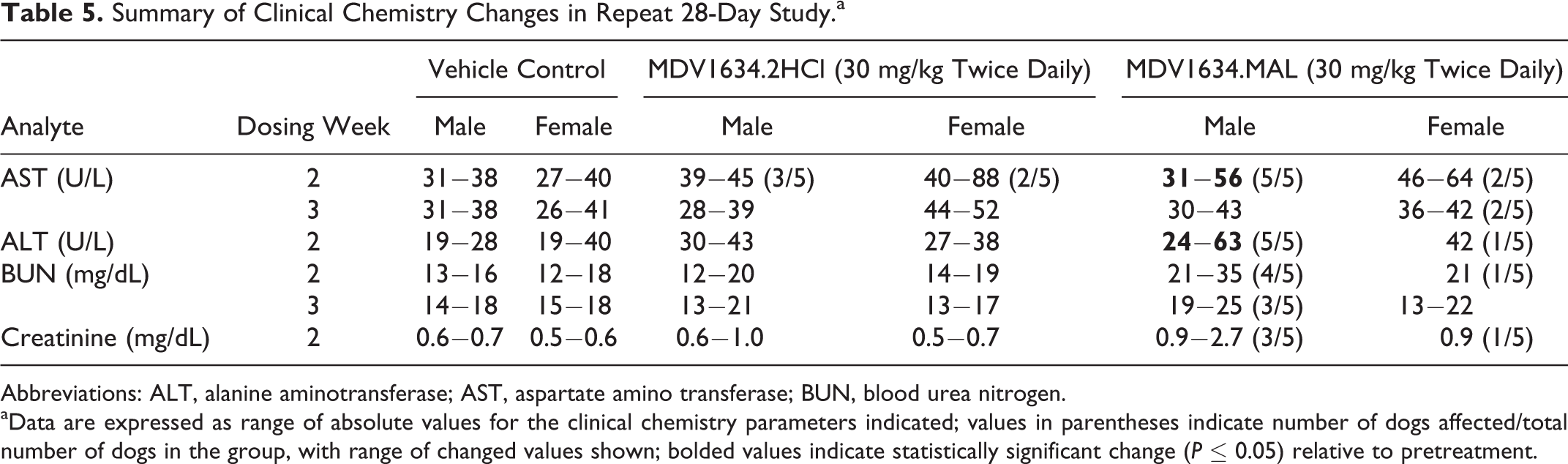
Abbreviations: ALT, alanine aminotransferase; AST, aspartate amino transferase; BUN, blood urea nitrogen.
aData are expressed as range of absolute values for the clinical chemistry parameters indicated; values in parentheses indicate number of dogs affected/total number of dogs in the group, with range of changed values shown; bolded values indicate statistically significant change (P ≤ 0.05) relative to pretreatment.
Urinary Biomarker Changes in Repeat 28-Day Study
Evaluation of early and sensitive urinary biomarkers of kidney injury during the course of the study (dosing and recovery) demonstrated moderate to severe early (dosing weeks 1-2) increases (compared to pretreatment values) in ALB, CLU, β2 microglobulin, and NGAL (Figure 2A and B; Table 6). The increases were higher in magnitude at weeks 1 and 2 compared to the end of dosing; however, the increase in NGAL persisted up to the end of the dosing period. The increase in KIM-1 was minimal and sporadic (week 1 only), and serum cystatin C changes were also minimal suggesting minimal glomerular involvement. The profile of urinary biomarker changes was supportive of tubular damage and decreased tubular reabsorption.
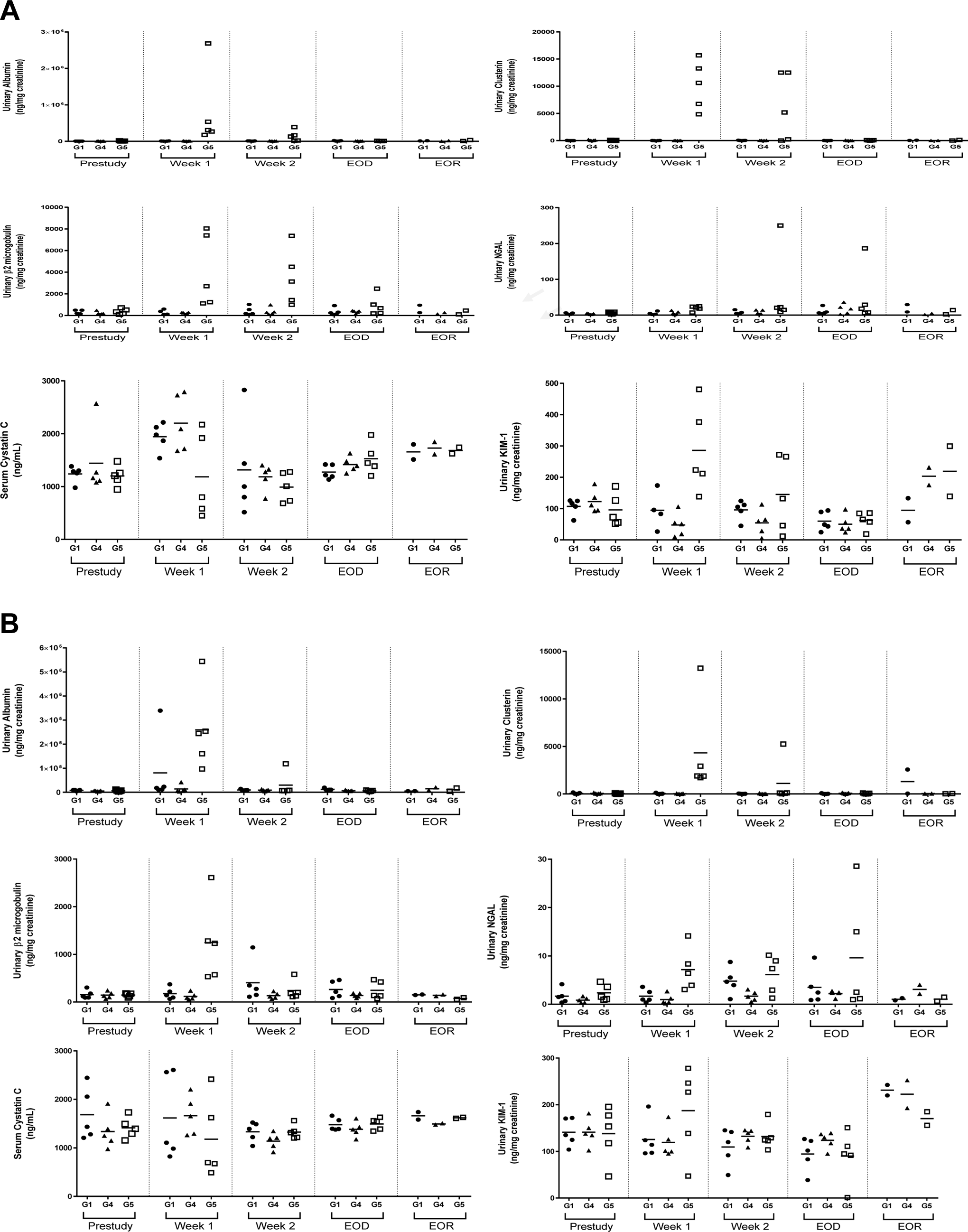
A, Summary of normalized urinary biomarkers in males in the 28-day repeat dose study with 6-week recovery (repeat study). Values represent concentration in urine (normalized to total volume and urinary creatinine). Horizontal bars indicate group mean values. B, Summary of normalized urinary biomarkers in females in the 28-day repeat dose study with 6-week recovery (repeat study). Values represent concentration in urine (normalized to total volume and urinary creatinine. Horizontal bars indicate group mean values; G1, vehicle; G4, MDV1634.2HCl 30 mg/kg twice daily; G5, MDV1634MAL 30 mg/kg twice daily; Week 1, end of 1-week dosing; Week 2, end of 2 week dosing; EOD, end of dose; EOR, end of recovery.
Fold Change in Urinary Biomarkers in Repeat 28-Day Study.
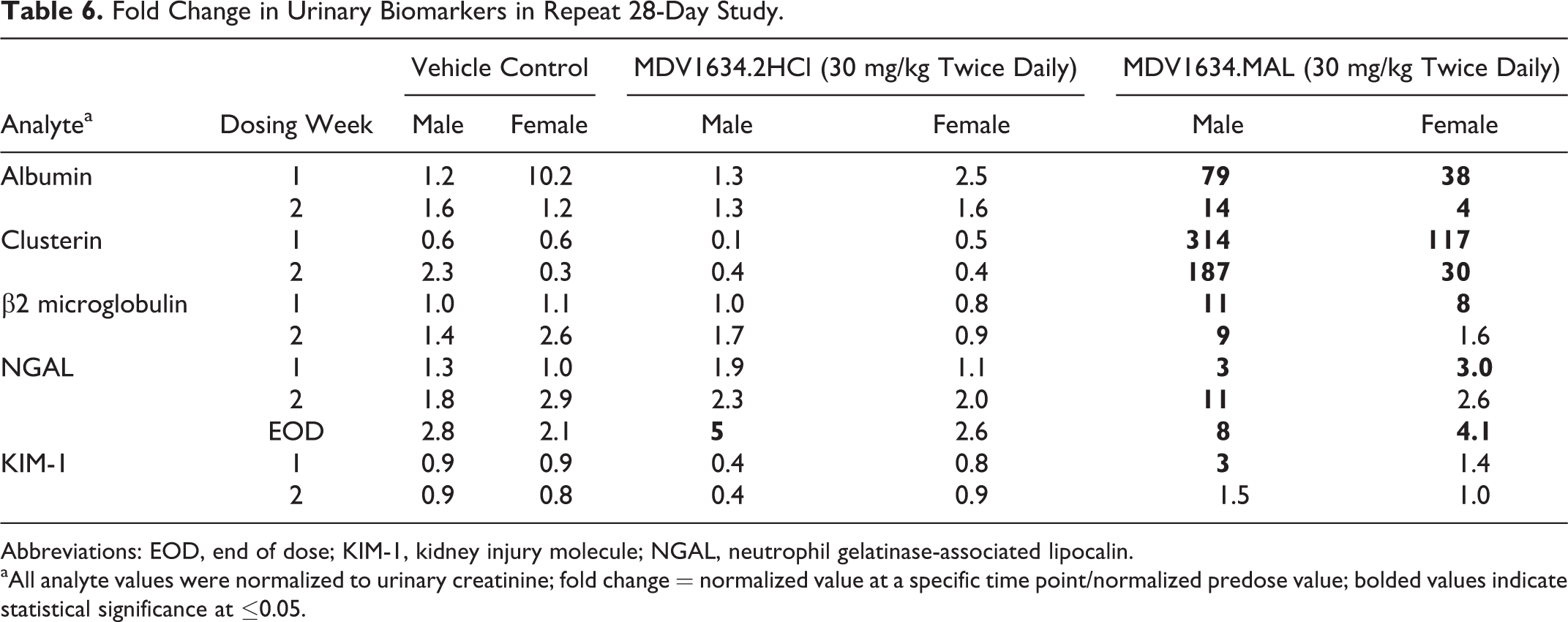
Abbreviations: EOD, end of dose; KIM-1, kidney injury molecule; NGAL, neutrophil gelatinase-associated lipocalin.
aAll analyte values were normalized to urinary creatinine; fold change = normalized value at a specific time point/normalized predose value; bolded values indicate statistical significance at ≤0.05.
Microscopic Changes in Repeat 28-Day Study
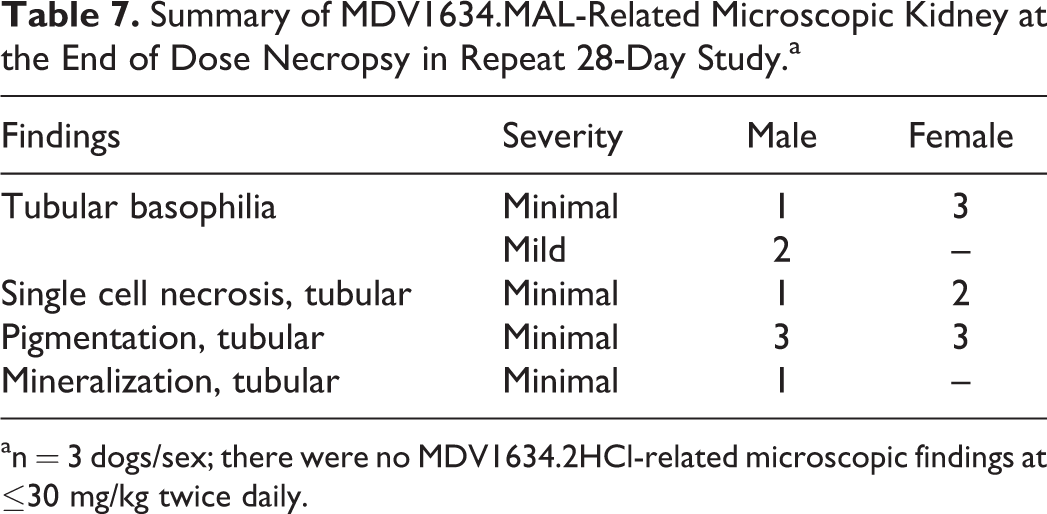
There were no MDV1634.2HCl-related microscopic changes in any dose group at ≤30 mg/kg twice daily. At 30 mg/kg twice daily, MDV1634.MAL-related microscopic changes (Table 7, Figure 3A and B) included tubular single cell necrosis (Figure 3A and B), pigmentation (Figure 3B), and tubular basophilia (Figure 3B), which were observed at the end of dosing in a few dogs and correlated with early changes (weeks 1 and 2) in ALB, CLU, and β2-microglobulin as well as early (week 1) and EOD changes in NGAL (Figure 2A and B). Renal tubular basophilia suggestive of potential drug-related tubular toxicity was more severe in the male dog, and 1 male dog also had mineralization. In most dogs, the findings were bilateral and predominately noted at the corticomedullary junction but with increasing severity, tubular basophilia was also noted in the cortex. All microscopic pathology changes were reversed at the end of the 6-week recovery period in females; however, tubular basophilia, single cell necrosis, and pigmentation were still noted in 1 male. There were no MDV1634-related glomerular microscopic findings at the EOD or following 6-week recovery.
Summary of MDV1634.MAL-Related Microscopic Kidney at the End of Dose Necropsy in Repeat 28-Day Study.a
an = 3 dogs/sex; there were no MDV1634.2HCl-related microscopic findings at ≤30 mg/kg twice daily.
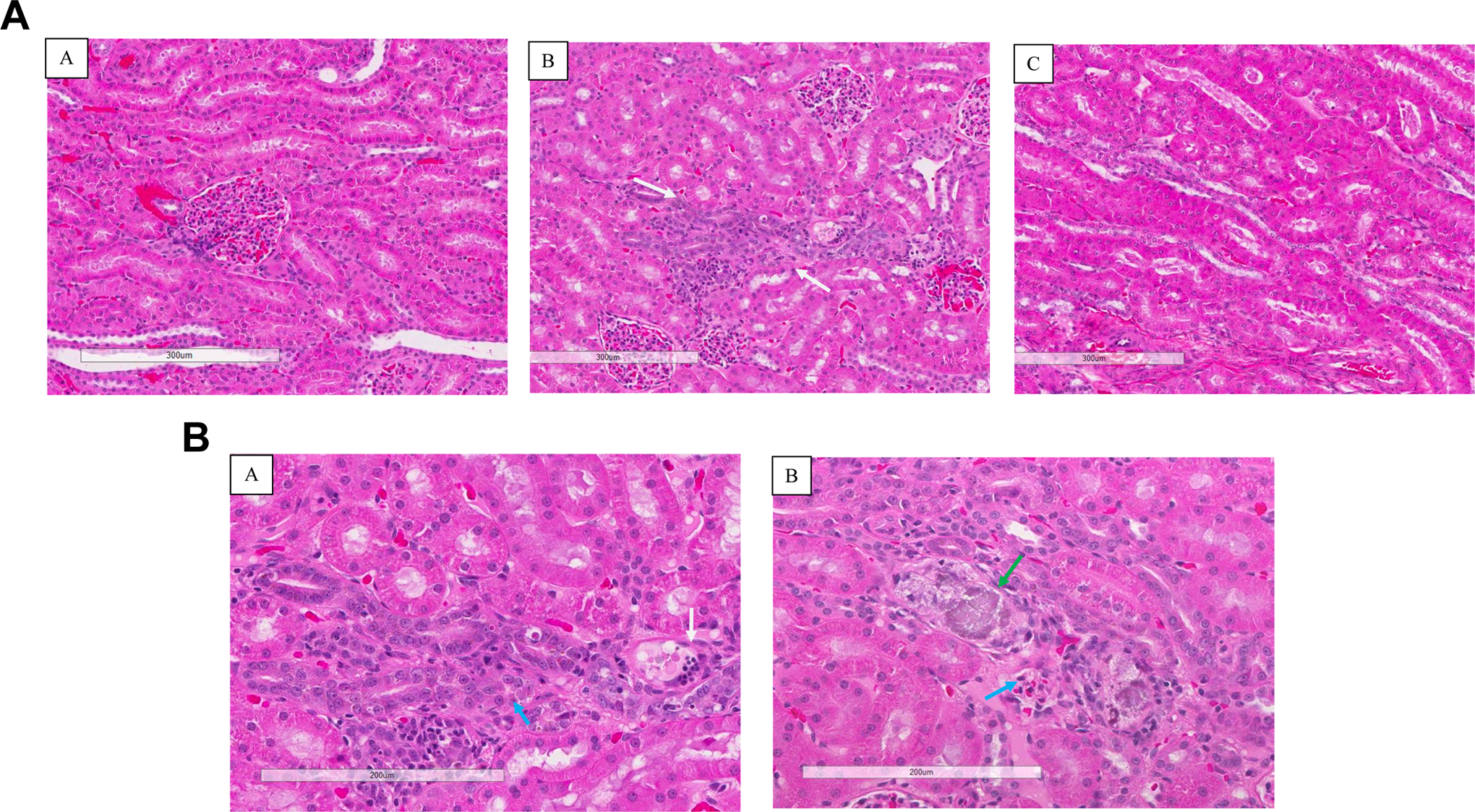
A, Microscopic renal changes following administration of vehicle, MDV1634.2HCl or MDV1634.MAL for 28 days in dogs (repeat 28-day study). Representative kidney micrographs from vehicle (A), MDV1634.MAL-treated (B; 30 mg/kg twice daily) and MDV1634.2HCl (C; 30 mg/kg twice daily) dogs following administration for 28 days (end of dose); the arrows demonstrate MDV1634-related tubular basophilia (B, white arrow). No basophilia is visible in MDV1634.2HCL-treated dog (C). Microscopic renal changes following administration of MDV1634.MAL (30 mg/kg twice daily) for 28 days in dogs (repeat 28-day study). Representative kidney micrographs (higher magnification) from MDV1634.MAL-treated dogs, following administration for 28 days (end of dose). The arrows demonstrate MDV1634-related single cell necrosis (A, white arrow), mineralization (B, green arrow), and pigment (A and B, blue arrow).
Discussion
A comparative evaluation of the hydrochloride and MAL salt forms of MDV1634 revealed that the acute hypotensive effect seen with MDV1634.MAL following single administration in a cardiovascular telemetry study at ≥6 mg/kg/d (internal data) and the sustained effects seen with MDV1634.2HCl at ≥10 mg/kg twice daily (repeat 28-day study) is considered MDV1634-related, can be correlated to the high affinity antagonism of the αAdr2B and moderate antagonism of αAdr2C receptors by MDV1634, and is independent of the salt form. However, the observed renal toxicity in dogs was limited to the MAL but not the hydrochloride salt form at similar MDV1634 dose (30 mg/kg twice daily) and exposures and therefore not considered MDV1634-related. In the original 28-day oral toxicity study with MDV1634.MAL, histopathological findings in both sexes at scheduled necropsy included dose-dependent increase in the incidence of basophilic tubules (increased severity in male dogs), mild tubular degeneration and/or necrosis, and hyaline casts. Similar renal toxicity findings were also observed in the current repeat 28-day study in the MDV1634.MAL group but not in the MDV1634.2HCl group (30 mg/kg twice daily) at similar MDV1634 exposures (Cmax and AUC0-24 h). Also, similar to the EOR findings in the original study (4-week recovery), all changes were reversible in the repeat study at the end of a 6-week recovery with the exception of tubular basophilia, single cell necrosis, and pigmentation, which was observed in 1 male dog at 30 mg/kg twice daily in the MDV1634.MAL group (both studies).
Although there are approved pharmaceuticals which are MAL salts, and MAL salt has a GRAS designation, there are 2 reports in dogs where MAL (maleic acid) salt of active compounds were implicated in the induction of acute tubular necrosis. In the first report, 14 an initial study with pravadoline MAL (WIN48098 6) demonstrated evidence of maleic acid-induced acute tubular necrosis in beagle dogs following a single oral administration of 40 mg/kg pravadoline MAL (WIN48098 freebase 31 mg/kg; maleic acid content 9 mg/kg). A follow-up study with equimolar dosages of the ethane sulfonate salt of WIN48098 (WIN48098 7, 40 mg/kg/d) or maleic acid (≥9 mg/kg/d) alone in dogs confirmed that the acute tubular necrosis observed with WIN48098 6 (40 mg/kg/d) was attributable to maleic acid and not to WIN48098. In the second report, 13 administration of MAL salt of TC 5619 to dogs in a 14-day toxicology study resulted in renal toxicity at the MAL counterion exposures ≥9.6 mg/kg/d (TC 5619 ≥30 mg/kg/d). Additional studies demonstrated that administration of MAL or maleic acid to dogs also resulted in increased excretion of low-molecular-weight analytes such as Na, K, phosphate, 4 amino acids, 6,7 and bicarbonate, 5 which suggested that MAL-induced toxicity is potentially related to the dysfunction in the proximal tubule. In the original 28-day study conducted with MDV1634.MAL, the histopathologic tubular changes were observed at 10 and 30 mg/kg twice daily (20 and 60 mg/kg/d), where the corresponding maleic acid doses were 6 and 19 mg/kg/d, respectively (Supplemental Table 2). This study suggested that repeated exposure (28 days) to maleic acid content of even 6 mg/kg/d was sufficient to induce microscopic tubular changes, and the findings were only partially reversible following 28-day recovery when the maleic acid content was higher (19 mg/kg/d). In the repeat 28-day study, the MDV1634 exposures were similar between MDV1634.2HCl (AUC0-24 h, 70,800 ng·h/mL) and MDV1634.MAL (AUC0-24 h, 61250 ng·h/mL) at 30 mg/kg/d, but the microscopic tubular changes, similar to the original 28-day study, were observed only with MDV1634.MAL (maleic acid, 19.2 mg/kg/d) but not with MDV1634.2HCl (HCl, 14.4 mg/kg/d). These results confirm that the partially reversible tubular toxicity observed in the 28-day dog study was attributable to the maleic acid counterion and not to MDV1634.
Dogs may be more sensitive than other species to the nephrotoxic effects of maleic acid, since no clinical or histopathologic evidence of nephrotoxicity was observed in mice, rats, and monkeys that received WIN48098 6 MAL at daily oral dosages of 650, 400, and 100 mg/kg, respectively. Nevertheless, when renal toxicity is observed following high-dose administration of maleic acid or MAL salt to mice or rats, the nature of the renal injury is always tubular in nature. Proximal tubular necrosis following single intraperitoneal or intravenous administration of maleic acid or sodium MAL (200-600 mg/kg) was observed in mice and rats. Maleate-induced, dose-dependent cytotoxicity of isolated CD-1 mouse renal cortical tubules 16 and in vivo administration of high-dose sodium MAL (600 mg/kg) resulted in proximal tubular necrosis within 24 hours in CD-1 mice. Although maleic acid can induce proximal tubular dysfunction, 8 ultramicroscopic lesions 9 and metabolic effects including impairment of renal gluconeogenesis and aminoaciduria 10 (observed in rats), the exact mechanism for the MDV1634.MAL-induced tubular damage is still unclear.
The renal findings were considered MAL specific at ≥10 mg/kg/d twice daily and not attributable to MDV1634. The clinical findings of decreased activity and partly closed eyes (attributed to histamine H1 receptor antagonism), and tremors, protruding nictitating membrane and red eyes were observed at generally similar incidence and severity in both salt forms and therefore considered related to MDV1634 exposure. Similarly, the BP changes were attributable to MDV1634 and considered independent of salt form. However, the sustained hypotension observed with MDV1634.2HCl (≥10 mg/kg twice daily) during the repeat 28-day administration period did not contribute to renal toxicity even at 30 mg/kg twice daily, despite similar MDV1634 exposure as the MAL group. Based on the BP decreases observed in the single-dose cardiovascular study with MDV1634.MAL (internal data, not shown), it is likely that the dogs administered MDV1634.MAL also had a sustained hypotensive effect during the duration of dosing. Therefore, we concluded that MDV1634-induced hypotension was not the cause of the renal toxicity observed with MDV1634.MAL, because it was present with both salt forms and renal toxicity was only observed with the MAL salt form.
MDV1634 was being evaluated for a neurology indication that required chronic long-term exposure to the drug. Therefore, the ability to monitor any early signs of potential renal toxicity is critical to the progression of a compound undergoing clinical development. In the original 28-day study, there was lack of clinical pathology (measured at the EOD and EOR) correlates for the observed histopathologic renal changes raising a concern for clinical monitorability of potential renal toxicity. The concern was addressed in the repeat 28-day study by including clinical pathology measurements at multiple time points, and sensitive urinary biomarker evaluation was included at the same time points during the dosing period and at the EOR. Conventional biomarkers of kidney injury such as serum CREAT and serum urea occur coincident with or following kidney injury and lack sensitivity and prodromal utility, thereby providing suboptimal value as renal safety biomarkers in drug discovery and development. 17 –19 Seven urinary renal safety biomarkers, including KIM-1, CLU, ALB, total protein, β2-microglobulin, cystatin C, and trefoil factor 3 in urine has been qualified by the Predictive Safety Testing Consortium and was accepted by the regulatory agencies to monitor drug-induced kidney injury in 2010. In the repeat 28-day study, traditional clinical chemistry markers such as BUN and serum CREAT showed only minimal to mild increases throughout the dosing period in the MDV1634.MAL group but not in the MDV1634.2HCl groups, although there were no changes in urine volume or specific gravity. In the MDV1634.MAL group, there were also moderate to severe increases (compared to pretreatment values) during the dosing period in the urinary levels of ALB, clusterin, β2 microglobulin, and NGAL. Increases in the urinary biomarker in the MDV1634.MAL group were seen early (by weeks 1-2) during the dosing period, and the presentation of the biomarker changes were consistent with drug-induced tubular dysfunction and renal injury. 20 Histopathological evaluation of the kidney also confirmed that the renal injury was tubular in nature in the MDV1634.MAL group induced by the MAL counterion, and most maleic acid-induced renal injury in nonclinical studies has also been shown to be tubular in nature. 4 –11 Lack of glomerular injury was confirmed both by histopathology and by the absence of consistent changes in serum cystatin C (a marker of decreased glomerular filtration rate 21 ). Therefore, the increases in urinary ALB were considered to be due to decreased tubular reabsorption, rather than glomerular injury. The biomarkers used in this study (KIM-1, ALB, β2-microglobulin, clusterin, and serum cystatin C) in concert with traditional clinical pathology assessments and histopathology have been qualified by the regulatory agencies as acceptable kidney toxicity biomarkers in rodents, 22 –24 and there are some reports of urinary biomarker correlation in the monkeys. 25 However, validation of these sensitive urinary biomarkers in dogs is not complete for preclinical toxicology studies, but their utility has been demonstrated in the clinic. 21,26 By comparing to the gold standard histopathological evaluation, our study confirms that MAL counterion-induced kidney injury is tubular in nature and for the first time correlates the tubular urinary biomarkers with the histopathological findings in dogs.
In conclusion, MDV1634.2HCl is considered a suitable salt for clinical evaluation. Although MAL salts are GRAS, the content of MAL counterion in a compound undergoing clinical development should be carefully evaluated in toxicity studies in dogs (more sensitive species) to ensure its safety as a pharmaceutical agent.
Footnotes
Acknowledgments
The authors would like to thank Dr Joe Guiles for his intellectual input and CMC support for the MDV1634.MAL and MDV1634.HCl salt forms used in the repeat 28-day study. We also thank Ms Ria Falvo at the Charles River Laboratory, Montreal for serving as the study director in the repeat 28-day study and MPI for conducting the original 28-day study.
Author Contributions
M. Guha contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. L. Nguyen contributed to analysis and interpretation and critically revised manuscript. F. Poitout-Belissent contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. A. Bedard contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript. K. Raman contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medivation, Inc provided support for the studies reported in this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
