Abstract
Manganese (Mn) is neurotoxic and can induce manganism, a Parkinson-like disease categorized as being a serious central nervous system irreversible neurodegenerative disease. An increased risk of developing symptoms of Parkinson disease has been linked to work-related exposure, for example, for workers in agriculture, horticulture, and people living near areas with frequent use of Mn-containing pesticides. In this study, the focus was placed on neurochemical effects of Mn. Rats were dosed intraperitoneally with 0.9% NaCl (control), 1.22 mg Mn (as MnO2)/kg bodyweight (bw)/day, or 2.5 mg Mn (as MnCl2)/kg bw/day for 7 d/wk for 8 or 12 weeks. This dosing regimen adds relevant new knowledge about Mn neurotoxicity as a consequence of low-dose subchronic Mn dosing. Manganese concentrations increased in the striatum, the rest of the brain, and in plasma, and regional brain neurotransmitter concentrations, including noradrenaline, dopamine (DA), 5-hydroxytrytamine, glutamate, taurine, and γ-amino butyric acid, and the activity of acetylcholinesterase changed. Importantly, a target parameter for Parkinson disease and manganism, the striatal DA concentration, was reduced after 12 weeks of dosing with MnCl2. Plasma prolactin concentration was not significantly affected due to a potentially reduced dopaminergic inhibition of the prolactin release from the anterior hypophysis. No effects on the striatal α-synuclein and synaptophysin protein levels were detected.
Introduction
Manganese (Mn) is a trace element widely found in nature. It is an essential nutrient for plants and animals, including humans. Being essential to plants, farmers often add Mn to different pesticides before spraying, thus allowing to reduce the number of sprayings. Furthermore, Mn is also part of the active constituents of some fungicides (maneb, mancozeb). Therefore, there exists a risk for humans for coexposure to Mn and pesticides in occupational and environmental settings. 1,2 Workers in mining, welding, and smelting industries and people living in the vicinity of such industries are at a higher risk of having Mn toxicity. 1,3 Further, it is hypothesized that humans who live in areas with Mn exposure from industry, agro-, and horticulture have an increased risk of developmental neurotoxicity. 4
Humans are most often exposed to Mn by oral ingestion via food including contaminated food and drinking water and to a minor degree via the environment. Environmental Mn exposure is often found together with lead (Pb) and arsenic (As). 5 Because Mn, Pb, and As share many underlying mechanisms of toxicity, including interference with brain neurotransmitter metabolism and induction of oxidative stress, it might be difficult to identify the causative chemical that via environmental exposure causes damage to the health. 5 Occupational inhalation exposure constitutes the primary route for high-level Mn exposure. 6 The intestinal absorption and tissue distribution of Mn are highly balanced by efficient Mn uptake and Mn efflux mechanisms, 7,8 and Mn deficiency is uncommon. 9 If not distributed to tissues, absorbed Mn is primarily excreted in the feces via the bile.
Manganese is pivotal to the function of many different metalloprotein enzymes which are important for proper protein, lipid, amino acid, and carbohydrate metabolism and for neurotransmitter functions. 10 –12 Furthermore, the efficiency of various antioxidant defense systems is Mn dependent. 13 –15 An overload of Mn can induce hepatic cirrhosis, dystonia, hypermanganesemia, and polycythemia, 7,16 as well as neurodegenerative symptoms in the central nervous system (CNS) similar to Parkinson disease (PD) and known as manganism. 17 PD is neurochemically characterized by a selective loss of nigrostriatal dopaminergic neurons and formation of ubiquitin and α-synuclein-containing cytoplasmic inclusions. 18 Manganism is characterized by specific accumulation of Mn in dopamine (DA)-rich brain regions in the basal ganglia such as globus pallidus and corpus striatum 19 and deregulation of the glutaminergic, GABAergic (γ-amino butyric acid), dopaminergic, and cholinergic system in the CNS. 15,20 In addition, α-synuclein is known to potentiate the neurotoxicity of various chemicals including metals and pesticides. 21 Manganism shares many but not all the characteristics linked to PD, 22 –25 and PD genes have been demonstrated to be involved in the regulation of Mn toxicity. 7
Astrocytes constituting about 50% of the cells in the brain seem to play an important role by binding Mn in the glutamine-synthetase system, 26 where Mn is a necessary cofactor for proper function. 27 Patients with manganism accumulate Mn in GABAergic cells in globus pallidus and have impaired GABAergic signaling, while in PD, the dopaminergic neurons in substantia nigra degenerate and possess reduced capacity to synthesize DA and to maintain proper dopaminergic signaling. Further, the dopaminergic neurons develop oxidative stress 28,29 including mitochondrial dysfunction 30 and neurodegeneration. 31 –34 Patients with manganism respond to EDTA and para-aminosalicylic acid, 35,36 whereas they do not respond to levodopa as do patients with PD. 37 Effective therapies for the treatment of manganism are not available. 10 Many studies have compared the mechanisms linked to PD and manganism 24,38 –40 and have concluded that the relationship remains unclear and therefore deserves further studies. 41 However, owing to symptom similarities between PD and manganism and the involvement of PD-related genes in Mn toxicity, 7 CNS neurotoxicity as caused by Mn may be evaluated by PD characteristic parameters.
In general, mice and rats are useful animal models to study Mn-induced neurotoxicity. 1 Specifically, a relevant Mn exposure model for manganism in rats has been proposed by applying short-term daily intraperitoneal (IP) injections of 6 or 15 mg Mn/kg bodyweight (bw) 5 days per week for 4 weeks. 23 However, to the best of our knowledge, no applicable low-dose subchronic or chronic animal model studying manganism exists but is urgently needed. Therefore, and because of handling convenience, its size which allows a sampling setup for several purposes, and because most literature data for comparison are based on studies in rats, the present study used the rat. Inhalation exposure is technically demanding and it inherently possesses pitfalls about controlling the dose of nonvolatile chemicals such as MnCl2 and MnO2 with respect to potential oral intake of the chemicals located in the fur and the exposure chamber. Intravenous injection is unsuited for daily subchronic exposure, and subcutaneous administration causes irritation and inflammation. Therefore, IP administration of MnO2 and MnCl2 was regarded as a relevant exposure route, thereby also circumventing the gastrointestinal tract and interindividual intestinal absorption rates. Furthermore, this route may build up an IP reservoir of Mn for constant absorption and distribution, and it was also argued for and applied in a comprehensive toxicokinetic study on these 2 Mn compounds. 42 The applied positive control for assuring adequate brain dissection and analytical techniques was 6-hydroxydopamine (6-OHDA), which is often used experimentally to selectively destroy dopaminergic CNS neurons. The Mn doses were based on the doses applied in the study by Roels et al. 42
The scope of the present study was to investigate the neurotoxic effects induced by Mn by applying a multidisciplinary approach. Careful cage-side inspections (animal welfare and behavior) and biological observations (drinking, eating, body weight) were performed during the dosing period. After euthanasia, the weight of corpus striatum and the rest of the brain were measured and the Mn concentration was analyzed in corpus striatum, the rest of the brain, and in plasma. In the brain, the DA concentration was selected as a target end point. In addition, the concentrations of noradrenaline (NA), 5-hydroxytryptamine (5-HT), and various relevant amino acids (glutamate [glu], taurine [tau], and GABA) and the activity of acetylcholinesterase (AChE) and butyrylcholinesterase (BuChE) were analyzed. Additionally, quantitative determination of 2 PD-relevant proteins, synaptophysin and α-synuclein, was performed by Western blotting. Furthermore, the dopaminergic system is known to inhibit prolactin release from the anterior hypophysis. 43,44 Therefore, an increased serum/plasma prolactin concentration has been proposed as an indirect marker for compromised dopaminergic activity. 45 –47
To the best of our knowledge, the present study is the first relevant low-dose subchronic daily IP injection study, thus providing new relevant data on the brain Mn concentration and effects of Mn on rat brain neurochemistry.
Materials and Methods
Chemicals
The fine-grained Mn (IV) oxide (CAS no. 1313-13-9) preparation used was kindly donated by Dr Harry A. Roels. According to the manufacturer, the particle size in aqueous medium is 4.791 (4.523) µm, mean (1 standard deviation [SD])) with a median diameter of 3.583 µm. The purity was >99%. This MnO2 preparation was identical to that used by Roels et al 42 and consequently allowed for comparison with his toxicokinetic study.
Immediately before use for each dosing, an appropriate amount of MnO2 powder was mixed with a corresponding volume of 0.9% NaCl in a beaker. The MnO2 was suspended by input of ultrasound energy from a Microscan Ultrasonicator (Microscan® Systems, Inc. Renton, WA) equipped with a 2 mm stainless steel probe. Meanwhile, the beaker was placed in a water bath at 0°C to 4°C to protect from heating. The energy input was 100 W at 26 kHz for 1 minute. Mn (II) chloride tetrahydrate, MnCl2–4H2O (CAS no. 13446-34-9; purity ≥98%) was obtained from Sigma-Aldrich, Brøndby, Denmark (Catalog number 221279). In contrast to MnO2, MnCl2 readily dissolved. Therefore, the physicochemical form of the 2 dosing media was a suspension of MnO2 and a solution of Mn ions from MnCl2. The 6-OHDA (CAS No. 5720-26-3; purity >99.5%) was a kind donation from NeuroSearch A/S (Valby, Denmark) . All other chemicals used were of analytical grade.
Animals—General Housing and Dosing
Six-week-old Sprague Dawley male rats (Tac: SPRD N@Mol outbred rat) with specific pathogen-free health status were purchased from Moellegaard Breeding Centre Ltd (Lille Skensved, Denmark). Upon arrival, the rats were distributed by weight into 6 groups with the same average weight. Each group was composed of 10 to 12 rats except the control group (n = 24). Rats were allowed to acclimatize to housing conditions for 1 week before the start of the study. The rats were housed conventionally, 2 per cage (Macrolon, Techniplats Gazzada S. ar. L., Buguggiate, Italy), with a 12:12-hour reversed light/dark cycle with light from fluorescent tubes from 7
The experimental groups were dosed IP 7 days per week with 0.9% NaCl (control), 1.22 mg Mn (as MnO2)/kg bw/day for 8 weeks, 1.22 mg Mn (as MnO2)/kg bw/day for 12 weeks, 2.5 mg Mn (as MnCl2)/kg bw/day for 8 weeks, and 2.5 mg Mn (as MnCl2)/kg bw/day for 12 weeks. The MnO2 and MnCl2 concentrations in the media used for IP injection were 122 and 250 mg Mn/100 mL, respectively, and the dosing volume was 1 mL/100 g bw This dosing corresponds to total cumulative Mn doses of 68.6, 102.5, 140, and 210 mg Mn/kg bw, respectively. This dosing regimen is technical and hygienic demanding and was performed carefully by trained technicians.
The sixth group, serving as a positive control group for effects on the dopaminergic system, received a single stereotactic unilateral intracranial injection of 6-OHDA in the right part of corpus striatum to induce dopaminergic cell loss. Clinical inspections, including spontaneous behavior, 2 times per day, together with biweekly bodyweight measurement were performed during the experiment by trained technicians and if deemed necessary assisted by an expert veterinarian.
Sample Preparation
The day after the last injection, the rats were decapitated in light CO2/O2 narcosis, and at autopsy, the injection site, peritoneum, and all organs were carefully inspected by expert veterinarians. Blood was collected with heparin as the anticoagulant and used for plasma preparation by centrifugation at 1,000g at 0°C to 4°C for 10 minutes. The brain was taken out and weighed. Immediately thereafter, it was cooled in 50 mL ice-cold 0.9% (wt/vol) NaCl for 2 minutes. The right and left part of corpus striatum were excised separately, weighed and pooled into 1 sample, and analyzed collectively (called the pooled corpus striatum or simply the corpus striatum). In animals dosed with 6-OHDA in the right corpus striatum, the right and left parts of corpus striatum were analyzed separately. The dissection procedures were carried out in accordance with Glowinski and Iversen 49 and by guidance from experts at NeuroSearch A/S, Greater Copenhagen area, Denmark.
Under ice-cold conditions, the corpus striatum as well as the rest of the brain were homogenized in 0.32 M sucrose by use of an Ultrasonicator (Microscan) set at 100 W at 26 kHz for typically less than 10 seconds. Samples of homogenate were immediately mixed with the following solutions and stored with the rest of the unprocessed homogenates at −80°C until analysis. Specifically, a volume of 1,000 μL homogenate was added to 2,000 μL 0.5 M perchloric acid and used for neurotransmitter analyses, 100 μL homogenate was added to 1,900 μL 0.05 M sodium hydroxide for protein analysis, and 500 μL homogenate was added to 500 μL 2% (vol/vol) Triton X-100 for cholinesterase analyses. Further details have been described previously. 48 The unprocessed homogenate was used for all other analyses and unprocessed plasma was used for the Mn and cholinesterase analyses.
Determination of Mn in Brain Tissue and Plasma by Inductively Coupled Plasma Mass Spectrometry
An aliquot of 500 µL of brain homogenate or plasma was wet ashed by 3 mL of 67% vol/vol nitric acid under pressure for 20 minutes in closed quartz bombs in a microwave oven (Multiwave; Anton Paar, Graz, Austria). The temperature reached approximately 220°C and the pressure 70 bar. Before use, the nitric acid used was purified by subboil distillation in an all-quarts apparatus (Hans Kürner, Rosenheim, Germany). The ultrapure water was obtained from an Element apparatus (Millipore, Bedford, Massachusetts). Following wet ashing, the clear residue was diluted with water to 20 mL by weighing. All sample dilutions, which were carried out in a class 100 HEPA (High Efficiency Particulate Air filter) clean air cabinet, were stored in polypropylene tubes until the time of analysis. The concentration of Mn in the diluted samples was determined by inductively coupled plasma mass spectrometry (ICP-MS) using an ELAN 6100 DRC instrument (Perkin Elmer SCIEX, Concord, Ontario, Canada) equipped with an AS-91 autosampler. During the measurement procedure, the sample tubes placed in the autosampler were flushed with class 100 HEPA-filtered air to prevent contamination. The general settings used in the standard operation mode of this instrument for the determination of Mn (55 m/z) have been detailed elsewhere. 50 The quantification was carried out using an external standard curve, and rhodium was used as internal standard throughout all analyses.
NA, DA, and 5-HT Neurotransmitter Analyses
The perchloric acid deproteinized samples were centrifuged (20 minutes, 0°C-4°C, 10,000g). To 1 aliquot of the supernatant was added internal standard (N-ω-methyl-5-HT, Sigma-Aldrich, Brøndby, Denmark, Catalog number M 1514) and used directly for the determination of 5-HT. To another aliquot was added internal standard (3,4-dihydroxybenzylamine, Sigma-Aldrich, Brøndby, Denmark, Catalog number D 7012), purified on aluminum oxide, eluted by addition of 0.2 M HClO4, and analyzed for NA and DA. Details on the sample preparation, ion-pair HPLC (High Performance/Pressure Liquid Chromatography) separation, electrochemical detection, and quantification procedures have been reported previously. 48,51
Amino Acid Neurotransmitter Analyses
Analysis was performed on the perchloric acid deproteinized samples by precolumn o-phthaldialdehyde/2-mercaptoethanol derivatization prior to HPLC separation and fluorescence detection. In brief, the perchloric acid deproteinized samples were centrifuged (20 minutes, 0°C-4°C, 10,000g). Homoserine (Sigma, H-6515) was added to the supernatant as the internal standard. Samples were separated on a RP-18 column at ambient temperature by a 0.1 M phosphate buffer with 25% methanol. The flow rate was 1 mL/min. The excitation and emission wavelengths were 230 and 455 nm, respectively. Focus was placed on glu, tau, and GABA. Details have been reported previously. 48
Determination of AChE and BuChE Activities
Triton X-100-treated samples were thawed, mixed, and allowed to incubate at ambient temperature 1 hour for tissue solubilization before they were mixed and centrifuged for 10 minutes at 0°C to 4°C and 10,000g. The resulting supernatant was used for the determination of AChE and BuChE activities at 37°C and pH 7.2 according to Ellman et al 52 on a COBAS-MIRA (Roche Diagnostics, Basel, Switzerland) robot system at 405 nm using 156 mM acetylthiocholine iodide and 218 mM butyrylthiocholine iodide as the substrate for AChE and BuChE, respectively. Further details have been published previously. 48
Protein Analysis
Protein concentration analysis was carried out according to Lowry et al 53 using bovine serum albumin (Sigma-Aldrich, Brøndby, Denmark) , Catalog number A7030) as the standard.
Western Blotting
Prior to electrophoresis, the samples (pooled corpus stratum) were sonicated under ice-cold conditions at 100 W and 26 kHz for 1 minute and diluted in sample buffer (1 M Tris–HCl, pH 8.5, 8% lithium dodecyl sulfate, 40% glycerol, 2 mM EDTA, 0.075% Coomassie blue G-250, 0.025% phenol red; Bio-Rad, Denmark) together with 20× reducing agent (TCEP (tris(2-chloroethyl)phosphine)) ; Bio-Rad, Copenhagen, Denmark) and a cocktail set of protease inhibitors (CalbiochemTM purchased from Merck, Hellerup, Denmark, Catalog number 539134). Samples for synaptophysin, α-synuclein, and β-actin analysis were processed for sodium dodecyl sulfate polyacrylamide gel electrophoresis as described in detail previously. 48 The membranes were incubated for 2 hours with rat polyclonal anti α-synuclein (1:1,000; a kind donation from Karen Bielefeldt Pedersen, Lundbeck Pharma A/S, Denmark), 54 mouse monoclonal antisynaptophysin (1:1,000; Neomarkers, Fremont, California), or mouse monoclonal anti-β-actin (1:5,000; Sigma-Aldrich, Brøndby, Denmark). After washing 3 × 15 minutes in TBS-T (Mixture of tris-buffered saline and Tween-20) buffer, the membranes were incubated for 2 hours with appropriate secondary peroxidase-labeled antibodies (1:5,000, goat antirabbit; Zymed, San Francisco, California and 1:2,000 rabbit antimouse; DAKO; Glostrup, Denmark). After that the blots were washed with TBS-T buffer for 3 × 15 minutes, bound antibodies were visualized by enhanced chemiluminescence (Bio-Rad, Copenhagen, Denmark) and measured with an imaging system (ChemiDoc; Bio-Rad, Copenhagen, Denmark). Densitometric quantification of Western blots was performed with the image analyzing program Quantity One (Bio-Rad, Copenhagen, Denmark).
Plasma Prolactin Analysis
The prolactin analysis was performed by the use of a commercially available ELISA kit (Biotrak catalog no. RPN 2563) purchased from GE Healthcare Life Sciences GmbH (DK-2605, Denmark). However, this kit is no longer available.
Statistical Analyses
The newest available SAS version was used for data processing and statistical analyses. In Tables 1 to 5, the data are expressed as mean (1 SD). A P < 0.05 was accepted as statistically significant and indicated by “*.” All data were tested for normal distribution and homogeneity of variance. When statistically significant treatment effects were detected in the 1-way ANOVA (Analysis of Variance), differences between control group and Mn-exposed groups were analyzed by post hoc test (t test). Only statistically significant effects are discussed. The striatal DA concentration is regarded as a relevant parameter for Mn neurotoxicity. Therefore, as an example, the results of the one-way ANOVA are specified for this parameter as a footnote to Table 3.
The Bodyweight and Weight of Right Corpus Striatum, Left Corpus Striatum, Pooled Corpus Striatum, and the Rest of the Brain From Rats Exposed Intraperitoneally to 1.22 mg Mn (as MnO2)/kg bw or 2.5 mg Mn (as MnCl2)/kg bw Once a Day for a Total of 8 or 12 Weeks.a,b,c
Abbreviations: bw, bodyweight; ND, not determined; 6-OHDA, 6-hydroxydopamine; SD, standard deviation.
aThe control group daily received 0.9% NaCl intraperitoneally for 12 weeks.
bThe sixth group of rats, serving as a positive control group for effects on the dopaminergic system, received a single unilateral intracranial injection of 6-hydroxydopamine (6-OHDA) in the right part of the corpus striatum.
cMean (SD); n = 24 (control group), n = 10 to 12 (dosed groups), and n = 10 (6-OHDA group).
dThis average was significantly different from the control group (P < 0.05).
The Concentration of Manganese (Mn) in Pooled Corpus Striatum (nmol/g tissue), in the Rest of the Brain (nmol/g tissue), and in Plasma (pmol/mL) From Rats Exposed Intraperitoneally to 1.22 mg Mn (as MnO2)/kg bw or 2.5 mg Mn (as MnCl2)/kg bw Once a Day for a Total of 8 or 12 Weeks.a,b,c
Abbreviations: 6-OHDA, 6-hydroxydopamine; SD, standard deviation.
aThe control group daily received 0.9% NaCl intraperitoneally for 12 weeks.
bThe sixth group of rats, serving as a positive control group for effects on the dopaminergic system, received a single unilateral intracranial injection of 6-hydroxydopamine (6-OHDA) in the right part of corpus striatum.
cMean (SD); n = 24 (control group), n = 10 to 12 (dosed groups), and n = 10 (6-OHDA group).
dThis average was significantly different from the control group (P < .05).
The Concentration of NA, DA, and 5-HT in Pooled Corpus Striatum (nmol/g Tissue) and in the Rest of the Brain (nmol/g Tissue) From Rats Exposed Intraperitoneally to 1.22 mg Mn (as MnO2)/kg bw or 2.5 mg Mn (as MnCl2)/kg bw Once a Day for a Total of 8 or 12 weeks.a,b,c,d
Abbreviations: DA, dopamine; 5-HT, 5-hydroxytryptamine; NA, noradrenaline; ND, not detectable/determined; 6-OHDA, 6-hydroxydopamine; SD, standard deviation.
aThe control group daily received 0.9% NaCl intraperitoneally for 12 weeks.
bThe sixth group of rats, serving as a positive control group for effects on the dopaminergic system, received a single unilateral intracranial injection of 6-hydroxydopamine (6-OHDA) in the right part of corpus striatum.
cMean (SD); n = 24 (control group), n = 10 to 12 (dosed groups), and n = 10 (6-OHDA group).
dDue to a technical error, the samples degraded and could not be assayed.
eExample of 1-way ANOVA results: F 5, 76 = 215.56, P = 0.000.
fThis average was significantly different from the control group (P < 0.05).
The Concentration of Glutamate (Glu), Taurine (Tau) and γ-Aminobutyric Acid (GABA) in Pooled Corpus Striatum (μmol/g tissue) and in the Rest of the Brain (μmol/g tissue) From Rats Exposed Intraperitoneally to 1.22 mg Mn (as MnO2)/kg bw or 2.5 mg Mn (as MnCl2)/kg bw Once a Day for a Total of 8 or 12 Weeks.a,b,c
Abbreviations: GABA, γ-aminobutyric acid; Glu, glutamate; 6-OHDA, 6-hydroxydopamine; SD, standard deviation; Tau, taurine.
aThe control group daily received 0.9% NaCl intraperitoneally for 12 weeks.
bThe sixth group of rats, serving as a positive control group for effects on the dopaminergic system, received a single unilateral intracranial injection of 6-OHDA in the right part of corpus striatum.
cMean (SD); n = 24 (control group), n = 10 to 12 (dosed groups), and n = 10 (6-OHDA group).
dThis average was significantly different from the control group (P < 0.05).
The Activity of AChE and BuChE in Pooled Corpus Striatum (U/g tissue) in the Rest of the Brain (U/g tissue), and in Plasma (U/L) From Rats Exposed Intraperitoneally to 1.22 mg Mn (as MnO2)/kg bw or 2.5 mg Mn (as MnCl2)/kg bw Once a Day for a Total of 8 or 12 Weeks.a,b,c
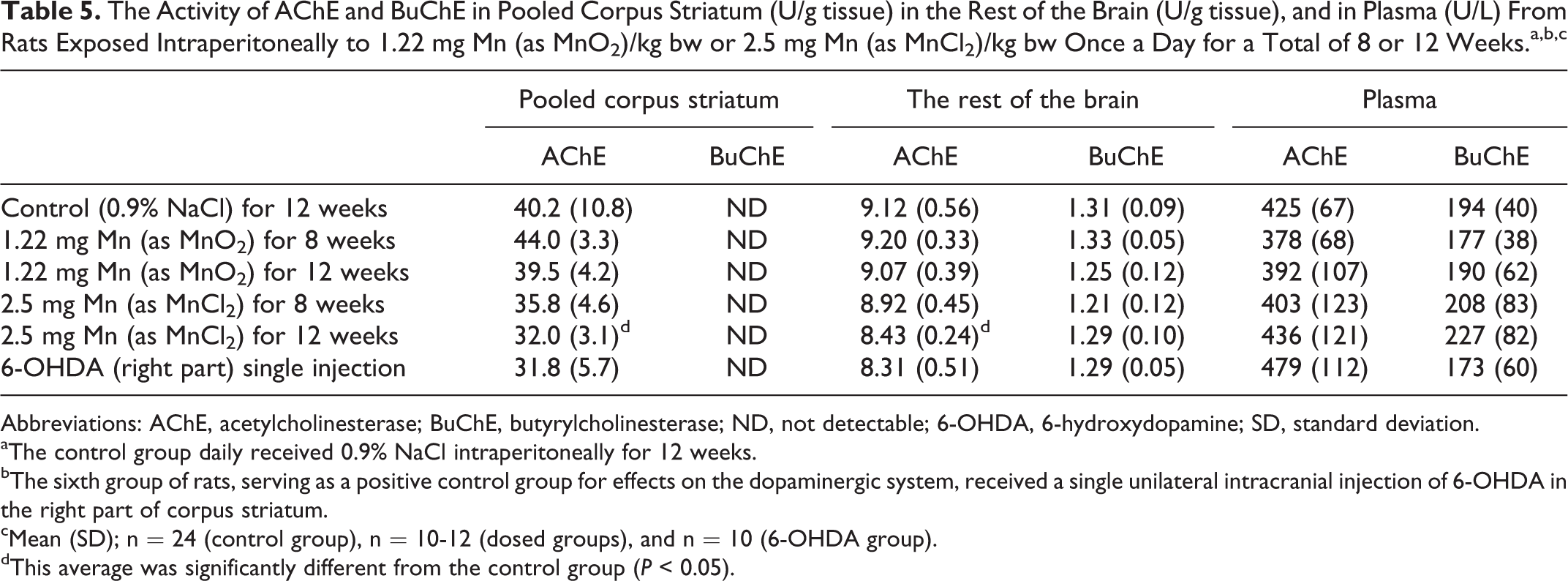
Abbreviations: AChE, acetylcholinesterase; BuChE, butyrylcholinesterase; ND, not detectable; 6-OHDA, 6-hydroxydopamine; SD, standard deviation.
aThe control group daily received 0.9% NaCl intraperitoneally for 12 weeks.
bThe sixth group of rats, serving as a positive control group for effects on the dopaminergic system, received a single unilateral intracranial injection of 6-OHDA in the right part of corpus striatum.
cMean (SD); n = 24 (control group), n = 10-12 (dosed groups), and n = 10 (6-OHDA group).
dThis average was significantly different from the control group (P < 0.05).
Results
Cage-Side Inspections
According to the daily (2 times per day) cage-side inspections, no dosing-induced serious toxic effects were observed in the rats. Both Mn dosing solutions were mildly irritating at the injection site as indicated by the lightly red colored skin, which persisted red for about 1 hour after injection. Both Mn dosing solutions affected the spontaneous behavior as reflected by a reduced spontaneous activity for about 1 hour after dosing when compared with controls.
The Mn- and 6-OHDA-dosed animals did not show any symptoms of rigidity, tremor, including resting tremor, bradykinesia, or hypokinesia, but in the period after the 6-OHDA-dosing until euthanasia 8 weeks after dosing, these animals were less active compared with controls and the IP Mn-dosed animals.
Observations at Autopsy
At autopsy, the MnO2 injections for 8 and 12 weeks were found to cause a mild irritative tissue reaction around the site of injection and in peritoneum and an accumulation of encapsulated unabsorbed material in the peritoneum and around the testicles. There was no obvious difference between 8 and 12 weeks of dosing. No organ showed any effect of Mn dosing when inspected macroscopically.
Bodyweight and Weight of Brain Regions
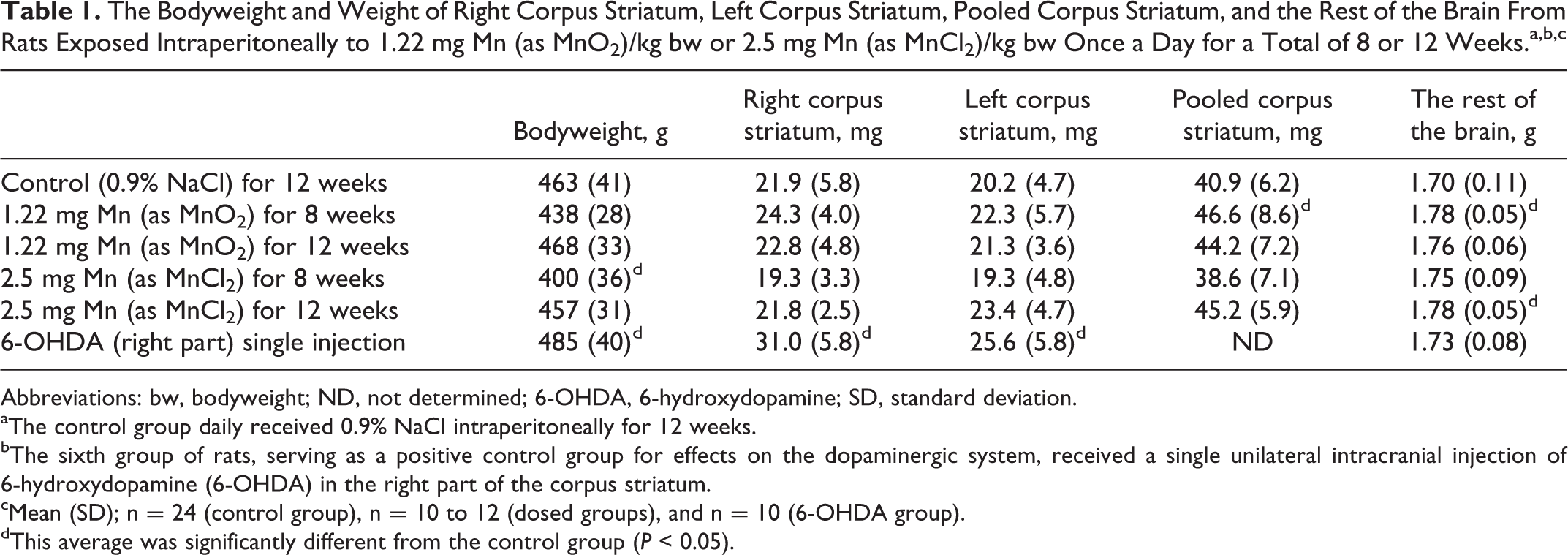
Table 1 shows the final body weight, the weight of the right and the left part of corpus striatum, the reconstructed pooled corpus striatum, and the weight of the rest of the brain.
Body weight
There was a significant reduction in body weight in the group dosed with 2.5 mg Mn/kg bw/day (as MnCl2) for 8 weeks and an increased body weight when dosed with 6-OHDA compared with controls (P < 0.05). Dosing with MnO2 for 8 or 12 weeks and dosing with MnCl2 for 12 weeks did not affect the final bodyweight or the body weight gain.
Weight of corpus striatum
The weight of the pooled corpus striatum was significantly increased by dosing 1.22 mg Mn/kg bw/day as MnO2 for 8 weeks (P < 0.05). The weight of the right and left corpus striatum of animals dosed with 6-OHDA was significantly increased when compared with controls (P < 0.05).
Weight of the rest of the brain
The weight of the rest of the brain was significantly increased in animals dosed with 1.22 mg Mn/kg bw/day as MnO2 for 8 weeks (P < 0.05) and after dosing 2.5 mg Mn/kg bw/day with MnCl2 for 12 weeks compared with controls (P < 0.05).
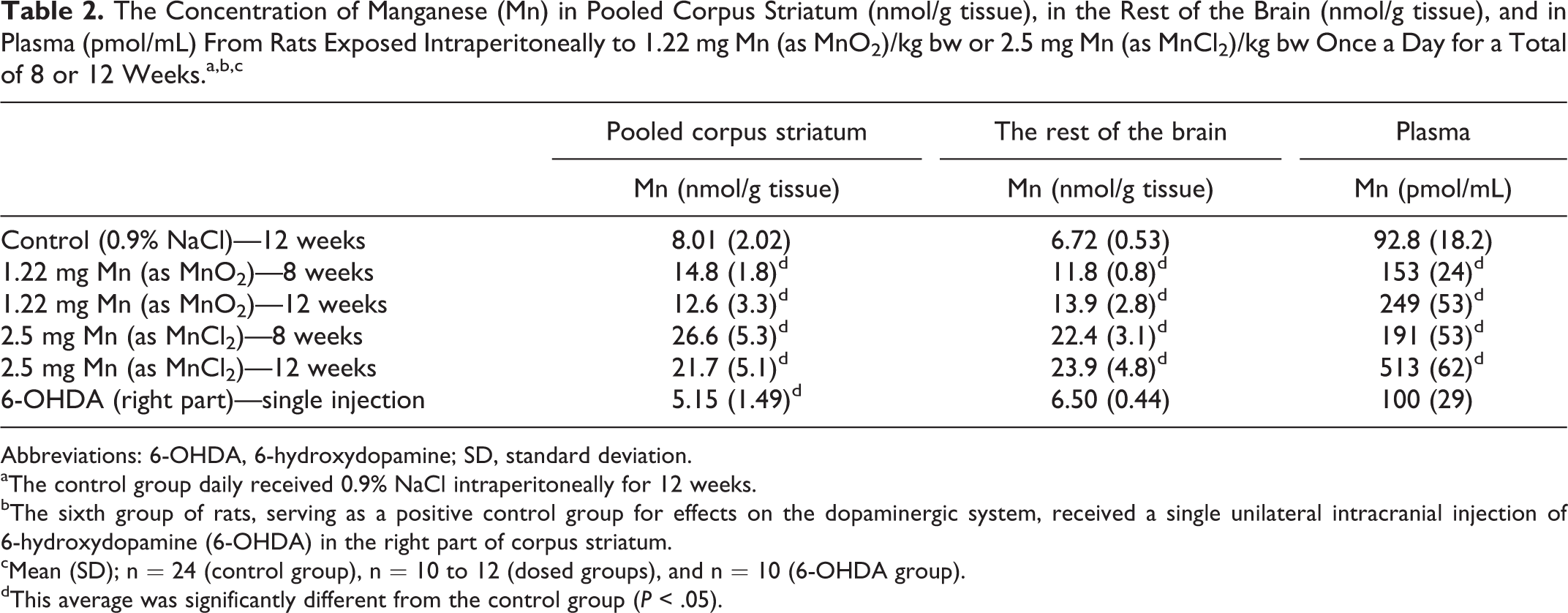
Mn Analysis and Mn Concentrations
The ICP-MS analysis was almost free from polyatomic interference (40Ar15N) and the SD for the solute (0.32 M sucrose) was negligible and did not compromise the accurate analysis of low levels of Mn (data not shown).
As seen in Table 2 in all 4 Mn dosing regimens, the Mn concentration significantly increased in corpus striatum, the rest of the brain, and in plasma (P < 0.05). Dosing with MnCl2 resulted in higher Mn concentrations than dosing with MnO2. In plasma, dosing for 12 weeks with MnO2 and MnCl2 resulted in higher Mn concentrations than following dosing for 8 weeks.
After unilateral injection with 6-OHDA, the Mn concentration in the right corpus striatum was significantly reduced compared to control pooled corpus striatum (P < 0.05). The 6-OHDA dosing did not affect the Mn concentration in the rest of the brain and in plasma (Table 2). Table 2 shows that in control animals, there was no difference in the regional Mn concentration in corpus striatum and the rest of the brain. In Mn-dosed animals, there was no regional difference in the Mn concentration in corpus striatum and the rest of the brain if dosed with MnO2 or MnCl2 for 8 or 12 weeks.
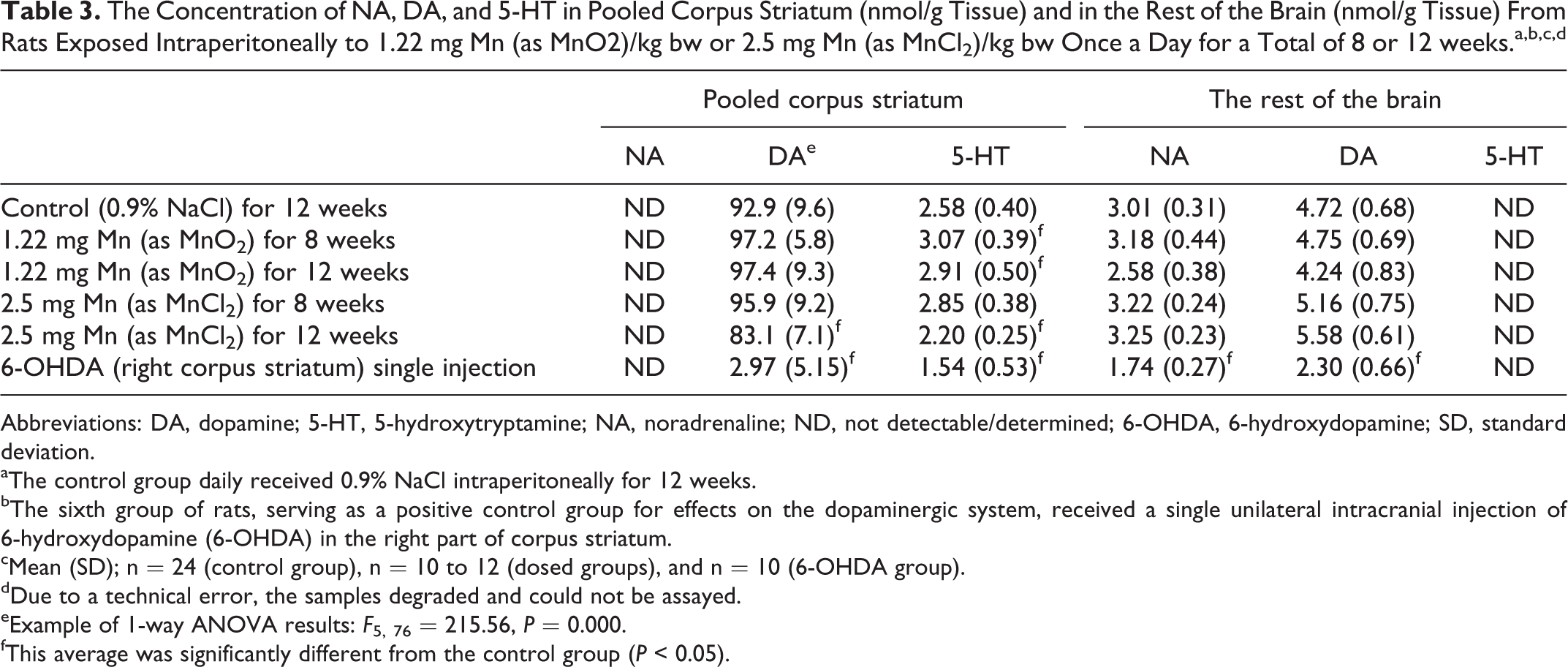
Regional NA, DA, and 5-HT Concentrations
Table 3 shows the concentration of these neurotransmitters in corpus striatum and the rest of the brain.
Corpus striatum
There was a marked reduction in the DA concentration in the right part of corpus striatum in animals dosed with 6-OHDA as the positive control compared with controls (P < 0.05; pooled corpus striatum). Further, the striatal DA concentration was significantly reduced after 12 weeks of dosing with MnCl2 compared with controls (P < 0.05). Dosing with MnO2 for 8 and 12 weeks increased the 5-HT concentration in pooled corpus striatum, whereas dosing for 12 weeks with MnCl2 decreased the 5-HT concentration in pooled corpus striatum compared with controls, both types of effects were statistically significant. The unilateral dosing with 6-OHDA in the right corpus striatum reduced the 5-HT concentration in this part compared with controls (pooled corpus striatum).
The rest of the brain
Dosing with Mn did not affect the NA and DA concentrations in the rest of the brain, whereas 6-OHDA markedly reduced the concentration of NA and DA when compared with control (P < 0.05).
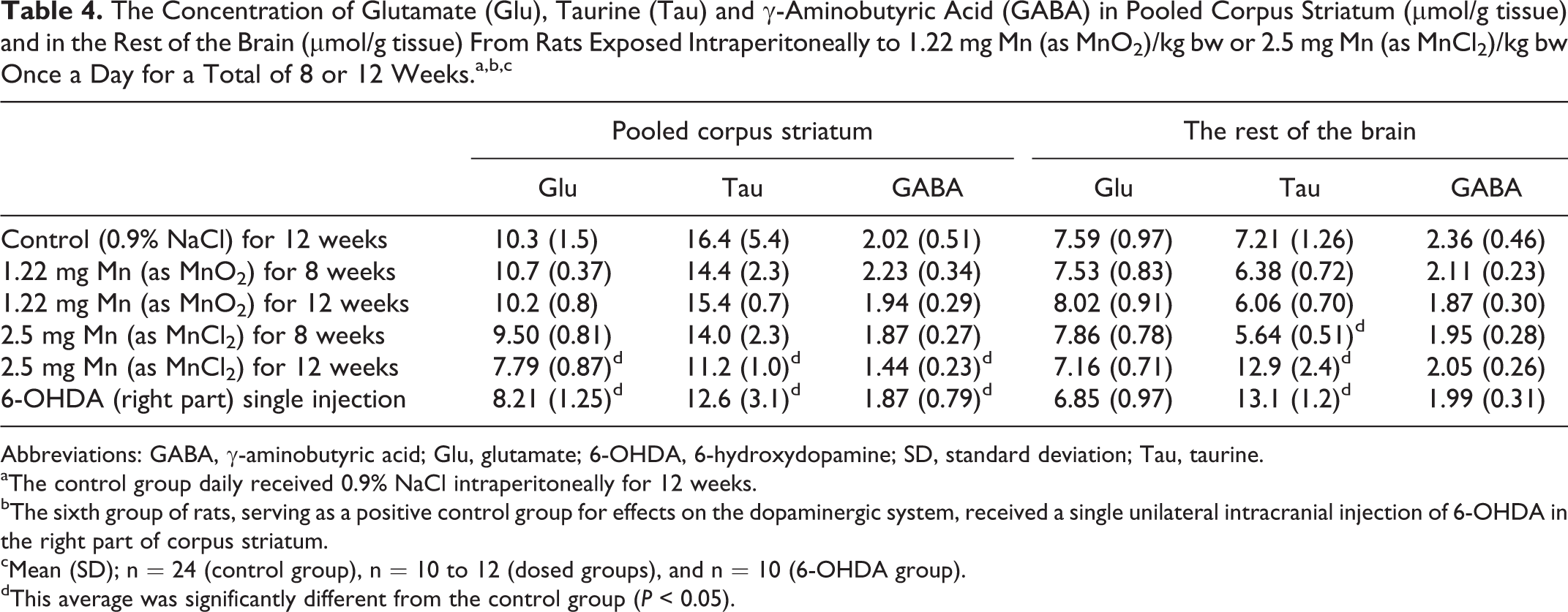
Concentrations of Amino Acids
Table 4 shows the concentration of neurotransmitter-related amino acids in corpus striatum and the rest of the brain. Dosing with MnCl2 for 12 weeks caused a significant reduction in the concentration of glu, tau, and GABA in corpus striatum when compared with controls (P < 0.05). Dosing with MnO2 was without effect on any amino acid in corpus striatum or in the rest of the brain.
The glu, tau, and GABA concentrations in the right corpus striatum was significantly reduced after dosing with 6-OHDA compared with controls (P < 0.05; pooled corpus striatum). Dosing with MnCl2 for 8 weeks reduced the tau concentration in the rest of the brain compared with controls, whereas the tau concentration increased markedly in the rest of the brain following 12 weeks dosing with MnCl2 compared with the controls, both types of effects were statistically significant.
Enzyme Activity of AChE and BuChE
Table 5 shows the activity of AChE and BuChE in corpus striatum, the rest of the brain, and in plasma. It was not possible to measure the very low activity of BuChE in corpus striatum by the present setup of sampling.
Dosing with MnCl2 for 12 weeks significantly reduced the AChE activity in corpus striatum and the rest of the brain compared with controls (P < 0.05). Acetylcholinesterase and BuChE activities in plasma were not affected by MnO2 or MnCl2 dosing. Dosing with MnO2 and 6-OHDA did not affect the AChE or BuChE activity in corpus striatum, the rest of the brain, or in plasma.
Western Blotting
Figure 1 shows a representative Western blot of the 3 proteins, β-actin, synaptophysin, and α-synuclein, in samples from pooled corpus striatum. All 3 proteins had the predicted molecular size according to the markers: β-actin (42 kDa), synaptophysin (38 kDa), and α-synuclein (17 kDa). Standard deviations of the same sample run on one gel or different gels were less than 15%. Standard samples at protein concentrations 2.5, 5, and 7.5 μg total protein showed a linear relationship for the 3 proteins (data not shown, further details have been published previously 48 ). A large interindividual variation in concentrations was observed in each group. Thus, the evaluation by densitometry failed to reveal any effect of Mn dosing.

Western blot of β-actin, α-synuclein, and synaptophysin in pooled corpus striatum from rats exposed intraperitoneally to 1.22 mg Mn (as MnO2)/kg bodyweight (bw) or 2.5 mg Mn (as MnCl2)/kg bw once a day for a total of 8 or 12 weeks. The control group daily received 0.9% NaCl intraperitoneally for 12 weeks. The sixth group of rats, serving as a positive control group for effects on the dopaminergic system, received a single unilateral intracranial injection of 6-hydroxydopamine (6-OHDA) in the right part of corpus striatum. Each lane represents samples from pooled corpus striatum at a protein concentration of 5 μg. Lane 1 shows the control group; lanes 2 and 3 show 1.22 mg Mn (as MnO2)/kg bw for 8 or 12 weeks of dosing, respectively; lanes 4 and 5 show 2.5 mg Mn (as MnCl2)/kg bw for 8 or 12 weeks of dosing, respectively; and lane 6 shows 6-hydroxydopamine (6-OHDA) dosing group.
Plasma Prolactin Concentration
The concentration of prolactin was measured in all plasma samples from control animals and the group dosed with MnCl2 for 12 weeks. The concentration was 100% ± 45% (indexed control) and 155% ± 78% (Mn dosed), respectively. These results did not show any statistically significant effect of Mn dosing.
Discussion
In the present study, we investigated the neurotoxic effects following subchronic exposure to MnCl2 and MnO2. The observed accumulation of encapsulated unabsorbed material in the peritoneum and around the testicles specifically in animals dosed with MnO2 might represent a reservoir for further continuous systemic absorption and distribution.
Increased weight was observed in the corpus striatum after dosing with MnO2 (1.22 mg/kg bw/day) for 8 weeks and 6-OHDA and in the rest of the brain of animals dosed with MnO2 (1.22 mg/kg bw/day) for 8 weeks. Dosing with MnCl2 (2.5 mg/kg bw/day) for 12 weeks increased nonsignificantly the weight of the pooled corpus striatum by 10.5% and the rest of the brain significantly by 4.7%. However, gross autopsy did not reveal any signs of inflammation in the brain after Mn exposure as previously reported, 55 but staining of tissue for pathology was not assessed. The increased weight in the 6-OHDA-dosed animals and reduced physical activity compared to control and Mn-dosed animals might be related to decreased 5-HT levels. 56
All Mn dosing regimens increased the Mn concentration in the corpus striatum, the rest of the brain, and in plasma. In general, animals dosed with MnCl2 had approximately 2-fold higher Mn concentration in the plasma and the rest of the brain compared with animals dosed with MnO2. This difference in concentration reflects a higher accumulated Mn dose in animals dosed with MnCl2. There was no difference in the Mn concentration in corpus striatum and the rest of the brain when dosed with MnO2 or MnCl2 for 8 or 12 weeks. In the plasma, dosing for 12 weeks with MnO2 and MnCl2 resulted in higher Mn concentrations than dosing for 8 weeks. Although plasma Mn levels are higher after 12 weeks of dosing, there was no increase in the accumulation of Mn in corpus striatum or the rest of the brain. This is in agreement with previously reported results that 4 weeks of dosing with 2.4- to 6-fold higher Mn doses did not increase the Mn concentrations in corpus striatum. 15,23 These results indicate that the animals may have reached an upper obtainable Mn concentration in the brain, which could be caused by balance between the divalent metal transporter 1, the primary divalent Mn-ion transporter into cells, and the Mn-outflux mechanism 8 and by saturation of Mn-binding sites.
In a comprehensive toxicokinetic study by Roels et al, 42 male rats were IP administered with MnCl2 or MnO2 (1.22 mg Mn/kg bw) once a week for 4 weeks and euthanized 4 days after last dosing. Using the identical MnO2 preparation and dose (1.22 mg/kg bw) and a comparable ICP-MS analytical method, Roels et al 42 found that repeated administration of MnO2 increased the blood Mn concentration by 82% (about 14,370 pmol/mL). This percentage increase is comparable to what was observed in the present study where blood Mn concentration increased by 65% and 168% after 8 and 12 weeks of repeated IP administrations. Additionally, the Mn blood concentration significantly increased by 106% and 453% after 8 or 12 weeks of administration with MnCl2 (2.5 mg/kg/bw/day), respectively. This is a higher increase than reported by Roels et al, 42 (61% increase) compared with controls. However, in the present study, the observed absolute plasma Mn concentrations after MnCl2 or MnO2 dosing were significantly lower than what observed by Roels et al, about 58- to 94-fold lower for MnO2-dosed animals, even though the analytical methods used for measurements were comparable. The reason for this rather large discrepancy is unknown.
The striatal Mn concentration in the present study increased by 85% or 57% after 8 or 12 weeks administration of MnO2 (1.22 mg/kg bw/day), where Roels et al 42 reported a 125% increase in the striatal Mn concentration (about 6.47 nmol/g). These are lower percentage increases taking into consideration the daily administration for 8 and 12 weeks, hence a higher total cumulative dose. The discrepancy cannot be explained by different analytical methods, but more likely by a different dissection procedure of the corpus striatum.
In addition to the higher increase in plasma Mn concentrations, the striatal Mn concentration increased by 232% and 171% after 8 or 12 weeks of administration of a dose of MnCl2 (2.5 mg/kg bw/day) compared to a 33% increase in the striatal Mn concentration (about 9.60 nmol/g) previously reported after dosing with MnCl2. 42 The higher percentage increases in plasma Mn concentration in striatum and plasma reflects the higher dose given in the present study compared to the study of Roels et al. 42
Overall, the present study showed Mn concentrations in corpus striatum, which were comparable to those of Roels et al 42 and O’Neal et al 23 using IP injection. However, the Mn-increased concentrations were more than 2-fold higher in the present study than those found by Roels et al 42 but 2-fold lower when compared to the study by O’Neal et al using 6 or 15 mg Mn/day (as MnCl2), 5 d/wk for 4 weeks. 23 These differences in Mn concentrations might be explained by different dosing regimens and/or dissection procedures.
The concentrations of the 3 CNS neurotransmitters NA, DA, and 5-HT are important biomarkers of brain integrity and therefore often applied in studies on CNS neurotoxicity of chemicals. 48 The low striatal NA concentration was not detectable by the applied sampling setup. The Mn dosing did not affect the NA concentration in the rest of the brain, whereas 6-OHDA markedly reduced the concentration of NA in the rest of the brain. No adequate study was identified in the literature using IP Mn administration for comparison. Manganism and Mn dosing of laboratory animals are associated with dopaminergic dysfunction 6 and are known to induce dopaminergic neurodegeneration. 34,57 Importantly, a 10.5% reduction in striatal DA concentration after 12 weeks of dosing with MnCl2 was found, whereas there was no effect on the DA concentration in the rest of the brain. The cause of the reduced striatal DA concentration might be compromised DA synthesis, reuptake, or regional degradation. The literature documents different effects on the DA concentration in CNS after IP Mn dosing. Most of the studies describe a reduction especially in corpus striatum as shown in mice, rats, and monkeys. 58 –63 Others have shown unchanged DA concentration 64,65 and 2 investigations have shown an increased concentration. 23,66 However, from the literature, it can be concluded that when optimal absorption and distribution in the brain are obtained, the effect is a Mn-induced reduction in the striatal DA concentration, 67 as also found in the present study. The positive control, 6-OHDA, strongly decreased the DA concentration in the right part of corpus striatum and in the rest of the brain. Dosing with MnO2 for 8 and 12 weeks increased the 5-HT concentration in corpus striatum, whereas dosing for 12 weeks with MnCl2 and 6-OHDA decreased the striatal 5-HT concentration (14.7% for MnCl2), when compared with controls. The mechanisms, if any, underlying this Mn species-dependent different effect on the striatal 5-HT concentration are not known. However, one study showed that IP injection of 6 or 15 mg Mn/day, 5 d/wk as MnCl2 to rats for 4 weeks did not affect the striatal 5-HT concentration. 23
The amino acids, glu, tau, and GABA, play an important role for proper CNS neurotransmission, 68,69 particularly the excitatory neurotransmitter glu 70,71 and the inhibitory neurotransmitter GABA are regarded to play central roles. 72,73 Dosing with MnCl2 for 12 weeks reduced the striatal concentration of glu (24.4%), tau (31.7%), and GABA (28.7%), whereas MnO2 dosing had no effect on the concentration of glu and GABA in corpus striatum and in the rest of the brain. Interestingly, tau concentration was reduced in the corpus striatum after 8 weeks of dosing with MnCl2, whereas it was increased in the rest of the brain after 12 weeks of dosing. Taurine is an antioxidant, 5 therefore, the increased tau concentrations in the rest of the brain could be a result of a compensatory mechanism increasing the antioxidant milieu to protect labile compounds such as DA from oxidation. 74 Remarkably, these increases in the tau concentration were to the same level as those caused by 6-OHDA. Dosing with 6-OHDA also reduced the striatal glu, tau, and GABA concentrations, whereas 6-OHDA increased the tau concentration in the rest of the brain.
In comparison, daily IP injection of mice (5.0 mg Mn/kg bw as MnCl2 for 9 weeks) did not affect the content of striatal glu and GABA. 75 In rats, no effects on glu, GABA, and tau concentrations in the cerebral hemisphere were seen using 4 or 8 IP injections (25 mg MnCl2/kg bw/day every 48 hours). 15 In a recent study, it was concluded that the GABAergic nervous system is more vulnerable to Mn toxicity than is the glutamatergic. 23 However, this greater vulnerability of the GABAergic system does not seem satisfactory to be supported by the present findings. Overall, although they do not contradict the present findings, these 3 studies report no effect of IP injection of Mn on the GABAergic system, which is consistent with present findings.
Pharmacological manipulation of the cholinergic and dopaminergic systems are useful for the treatment of symptoms of PD. 76 However, understanding the function and involvement of the cholinergic and dopaminergic systems is also important in manganism where imbalances in the system may result in changes in the AChE activity and induction of oxidative stress. 20,77,78 In the CNS, Mn is known to bind to AChE causing inhibited activity and thereby to activate the cholinergic system, 79 and it is hypothesized that this increased activity could act as neuroprotection. 38
It was not possible to measure the very low activity of BuChE in corpus striatum in the present sampling setup. Twelve weeks of subchronic dosing with MnCl2 significantly reduced the AChE activity in striatum and the rest of the brain. This is in accordance with studies showing that the AChE activity in the brain seems to relay on the duration of the Mn administration by IP injection where subacute exposure is reported to increase the activity, 55,77,80 while long-term IP administration decreases the activity. 77 This emphasized the importance of more knowledge about the integrated effect of Mn on the striatal cholinergic and DAergic systems in the understanding of the mechanisms underlying Mn neurotoxicity. 20
These differently reduced striatal neurotransmitter concentrations and enzyme activity seem not to be a general consequence of increased weight of corpus striatum (10.5%). A potential explanation could be that striatal astrogliosis overcompensated for the weight loss caused by a dying of neurons and the fact that astrocytes do not possess the same capacity to synthesize the neurotransmitters and AChE as neurons except for glu and GABA.
Synaptophysin and α-synuclein are synaptic proteins known to play an essential role in the pathogenesis of PD and suggested to facilitate Mn-induced neurotoxicity and loss of dopaminergic neurons. 81 –83 Unfortunately, the present study was not able to detect any Mn-induced changes in the levels of synaptophysin and α-synuclein in corpus striatum due to the large interindividual variations within each group.
Manganese has a relatively short half-life in the blood but a fairly long half-life in different tissues that acts as a reservoir for accumulation especially in the bones and for redistribution from the bones. 6 Therefore, the Mn concentration in the blood or the plasma does not always reflect the Mn concentration in the target tissues, particularly not in the brain. 84,85 It is well known that the dopaminergic system acts as an inhibitor of prolactin release from the anterior hypophysis. 43,44 This is why increased serum prolactin concentration has been proposed as an indirect marker for compromised dopaminergic neurotransmission in humans as reported in occupational and environmental epidemiological studies. 45 –47 In the present study, the dopaminergic system was found compromised in striatal tissue, and a trend of increased plasma prolactin concentrations (55%) was observed after 12 weeks of dosing with MnCl2, although the effect was not statistically significant. There have been reported linearity between repeated IP Mn dosing and plasma prolactin concentrations in rats, 86 whereas others have reported no effect of Mn dosing. 87 An explanation of not reaching a statistically significant effect in the present study could be the large individual variability between animals and not to be related to the analysis as such, as evidenced by reanalysis of the same sample (data not shown).
Conclusion
This study shows that 8 or 12 weeks of Mn dosing with MnCl2 or MnO2 in rats significantly increased Mn in circulation, but more important the accumulation in corpus striatum and the rest of the brain having reached a possible maximum steady-state level. Dosing with MnO2 modestly increased the 5-HT levels in the corpus striatum, independent of exposure period. Interestingly, 12 weeks of MnCl2 dosing caused a significant decrease in the DA (10.5%) and 5-HT (14.7%) levels in corpus striatum, but not in the rest of the brain. In addition, 12 weeks of MnCl2 dosing significantly decreased the concentration of the CNS neurotransmitters, glu (24.4%), tau (31.7%), and GABA (28.7%), in corpus striatum but increased the tau concentration in the rest of the brain, suggesting a compensatory mechanism for tau synthesis in the rest of the brain. Furthermore, the striatal AChE activity was significantly reduced (20.4%) after 12 weeks of MnCl2 dosing. The same pattern of changes in corpus striatum was observed after a single dose of 6-OHDA.
In daily life, humans are exposed to multiple chemicals simultaneously or sequentially, and there is strong evidence that the toxicity of an individual chemical can be altered by other chemicals. 88 In the CNS, the neurotoxicity of a single chemical may also be influenced by the brain’s capability for compensation for the effect on a particular target by adjusting another system. However, when exposed to combination, multiple targets could be involved, and the homeostatic and compensatory capabilities could be restricted and thereby induce a cumulative damage. 89 Consequently, the further evaluation of Mn neurotoxicity and the applicability of the low-level subchronic rat model obtaining maximum Mn concentrations in corpus striatum and the rest of the brain ideally could be studied after coexposure to differently acting neurotoxic chemicals. The present study is an integrated part of such studies that are in progress.
Footnotes
Authors’ Note
The study was performed under conditions approved by the Danish Animal Experiments Inspectorate (Council for Animal Experimentation) and by the in-house Animal Welfare Committee for Animal Care and use at the National Food Institute, Division of Toxicology and Risk Assessment, Søborg, Denmark.
Acknowledgments
The authors thank the Danish Environmental Protection Agency Programme for Pesticide Research for funding the project and Annette Landin, Amer Mujezinovic, Trine Gejsing, Karen Roswall, Karen Bielefeldt Pedersen at Lundbeck Pharma A/S, Denmark, who provided the α-synuclein antibody, and the animal caretaker group for their excellent technical work. Special thanks are given to Dr Harry A. Roels at Louvain Centre for Toxicology and Applied Pharmacology, Université Catholique de Louvain, Brussels, Belgium, for his donation of the fine-grained MnO2 preparation and NeuroSearch A/S, Denmark, for providing 6-OHDA and for technical assistance to stereotactic intracranial injection of 6-OHDA and during brain dissection procedure.
Author Contributions
Brian S. Nielsen contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Erik H. Larsen contributed to design, contributed to acquisition, analysis, and interpretation, and critically the revised manuscript. Ole Ladefoged contributed to conception and design and contributed to acquisition, analysis, and interpretation. Henrik R. Lam contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. Brian S. Nielsen performed the majority of practical work and wrote the majority of the manuscript with contributions from all the coauthors. Erik H. Larsen developed the method of analysis and supervised the Mn analyses. Ole Ladefoged was responsible for the daily animal housing, dosing, and animal welfare during the study and he assisted as an expert veterinarian and performed the pathological evaluations. Henrik R. Lam provided the original idea, designed the study, and developed and supervised the neurochemical analyses. The authors have read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research from the Danish Environmental Protection Agency Programme for Pesticide Research. The author(s) received no financial support for the authorship, and/or publication of this article
