Abstract
Background and Objectives:
Diclofenac (DIC) is metabolized to reactive metabolites such as diclofenac acyl-β-
Methods:
We examined the effects of inhibiting DIC-AG and 4′OH-DIC production on covalent protein adduct formation and lactate dehydrogenase leakage using sandwich-cultured rat hepatocytes (SCRHs).
Results:
After pretreatment of SCRH with (−)-borneol (BOR, a uridine diphosphate (UDP)-glucuronosyltransferase inhibitor) or sulfaphenazole (SUL, a cytochrome P450 2C9 inhibitor) for 30 minutes, intracellular concentrations of DIC, DIC-AG, and 4′OH-DIC were determined after further treating cells with 300 μM DIC for 3 hours. The decreased levels of reactive metabolites caused by BOR or SUL pretreatment resulted in decreased lactate dehydrogenase leakage from SCRH, although the formation of covalent protein adducts was not affected.
Conclusion:
These results suggested that both DIC-AG and 4′OH-DIC may be involved in acute cytotoxicity by DIC.
Introduction
Two major processes are involved in the development of drug-induced liver injury (DILI), direct hepatotoxic effects and idiosyncratic toxicity. Direct hepatotoxic effects of drugs occur acutely when the parent drugs and/or their reactive metabolites induce formation of reactive oxygen species from mitochondria or inflammatory cells. 1 The biotransformation to reactive metabolites, that is, chemically reactive species, is occasionally the initial event in the direct hepatotoxic effects of drugs. Idiosyncratic DILI is usually occurring in fewer than 0.01% of patient treated with certain drugs but has serious consequences and is the most common reason for a drug to be withdrawn from the pharmaceutical market. 2,3 Because the mechanisms of idiosyncratic DILI are unclear, it is difficult to predict its occurrence during preclinical and clinical trials. Some reports on the mechanisms of idiosyncratic DILI demonstrated that reactive metabolites could cause tissue damage by modification of cellular proteins through formation of covalent protein adducts and, further, by induction of immune responses against such covalent protein adducts. 4 –6
Some of the nonsteroidal anti-inflammatory drugs (NSAIDs), those containing arylacetic or 2-arylpropionic acid, induce both acute and idiosyncratic DILI. For example, bromfenac was withdrawn in 1998, after less than a year on the market because of DILI.
7
Diclofenac (DIC) is used to treat patients with osteoarthritis or rheumatoid arthritis and was associated with severe hepatic toxicity.
8
–12
The DIC is mainly metabolized to diclofenac acyl-β-
Sandwich-cultured rat hepatocytes (SCRHs) are useful tools to examine hepatobiliary drug transport, drug–drug interactions, and hepatotoxicity because these cells maintain hepatocyte architecture, including tight junctions, bile pockets, and transporters. 33,34 The SCRH are also used to examine cytotoxicity by drug metabolites. 35 –37 Taking advantage of these features of SCRH, we examined whether intracellular concentrations of DIC-AG and 4′OH-DIC affected acute cytotoxicity in these cells.
The results of this study showed the involvement of reactive metabolites such as DIC-AG and 4′OH-DIC in the acute liver cytotoxicity by DIC.
Materials and Methods
Ethical Approval
The study protocol was approved by the Committee for the Care and Use of Laboratory Animals of the School of Pharmacy of Kindai University (Osaka, Japan).
Compounds and Reagents
The DIC and sulfaphenazole (SUL; a CYP2C11 inhibitor) were obtained from Sigma Aldrich (St Louis, Missouri). Diclofenac-d4 (DIC-d4), DIC-AG, and 4′OH-DIC were from Toronto Research Chemicals (Toronto, Canada). (−)-Borneol (BOR; a UGT inhibitor) was from Tokyo Kasei Kogyo (Tokyo, Japan). To assay lactate dehydrogenase (LDH) leakage, a LDH cytotoxicity detection kit was obtained from Takara (Shiga, Japan). All other chemicals and solvents were of mass spectrometry (MS) grade or the highest commercially available purity.
Hepatocyte Isolation and Primary Culture
Hepatocytes were isolated from 8-week-old male Sprague Dawley rats by a 2-step collagenase perfusion method as described previously. 38 Viability, as determined by trypan blue exclusion, was above 90%. Hepatocytes were seeded at 1 × 105 cells/cm2 on collagen-coated 24- or 96-well plates (Sumitomo Bakelite, Tokyo, Japan) in Williams’ medium E supplemented with 10% fetal bovine serum, 1 nM insulin, 1 nM dexamethasone, 100 U/mL penicillin, 100 μg/mL streptomycin, and 2 mM GlutaMAX supplement I. Plates were then incubated at 37°C in the presence of 5% CO2 and 95% air. After an initial cell attachment period of 3 hours, the medium was replaced with fresh Williams’ medium E without serum and with the addition of the whole supplements and 5 U/mL aprotinin. At 24 hours after plating, the medium was replaced with fresh serum-free medium containing BD Matrigel (BD Biosciences, San Jose, California) at 0.25 mg/mL. Medium was changed daily until experiments were conducted at 5 days after plating. We used different hepatocyte batches among Figures 1 to 4. We have confirmed that the viability and metabolic activity for DIC were not significantly different among the different hepatocyte batches.
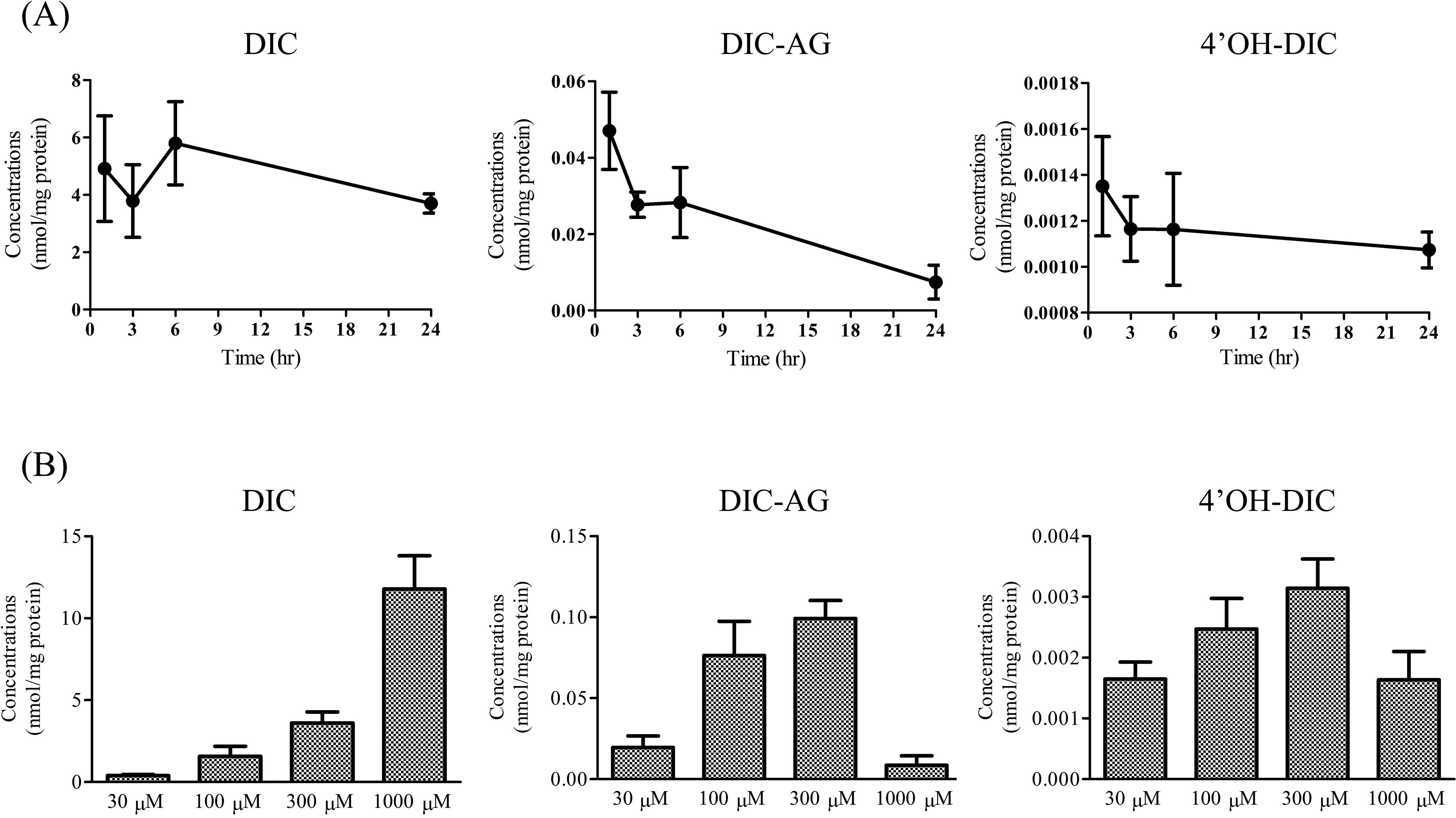
Intracellular concentrations of DIC, DIC-AG, and 4′OH-DIC in SCRH treated with (A) 300 μM DIC for 1, 3, 6, or 24 hours and (B) DIC (30, 100, 300, or 1,000 μM) for 3 hours. Values are means (SD), n = 5 to 6. DIC indicates diclofenac; DIC-AG, diclofenac acyl-β-
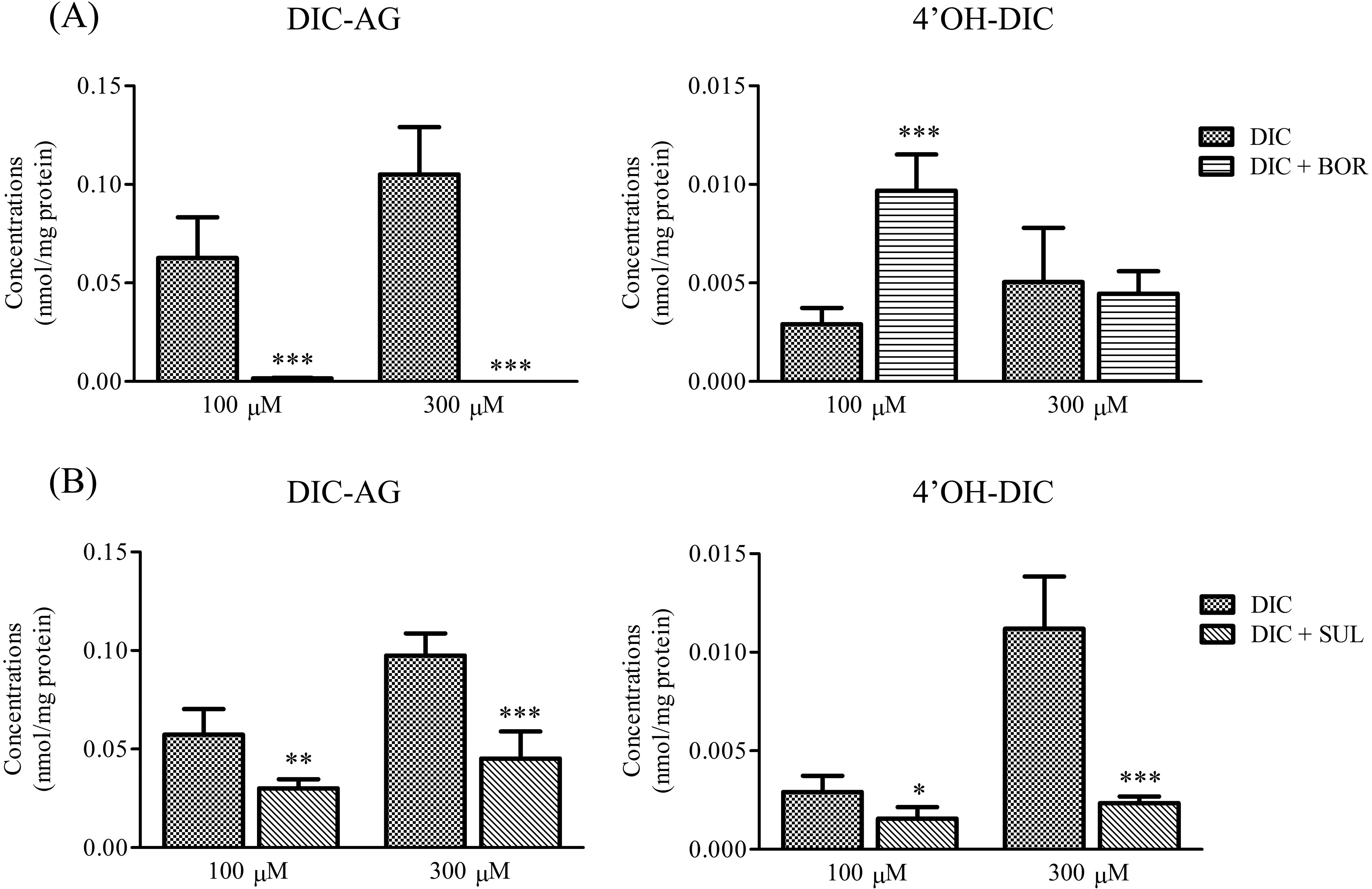
Intracellular concentrations of DIC-AG and 4′OH-DIC in SCRH treated with DIC (100, 300, or 1,000 μM) with or without (A) 1 mM BOR or (B) 1 mM SUL for 3 hours. Values are means (SD), n = 3 to 4. *P < 0.05, **P < 0.01 and ***P < 0.001 versus corresponding groups receiving only DIC. BOR indicates (−)-borneol; DIC, diclofenac; DIC-AG, diclofenac acyl-β-
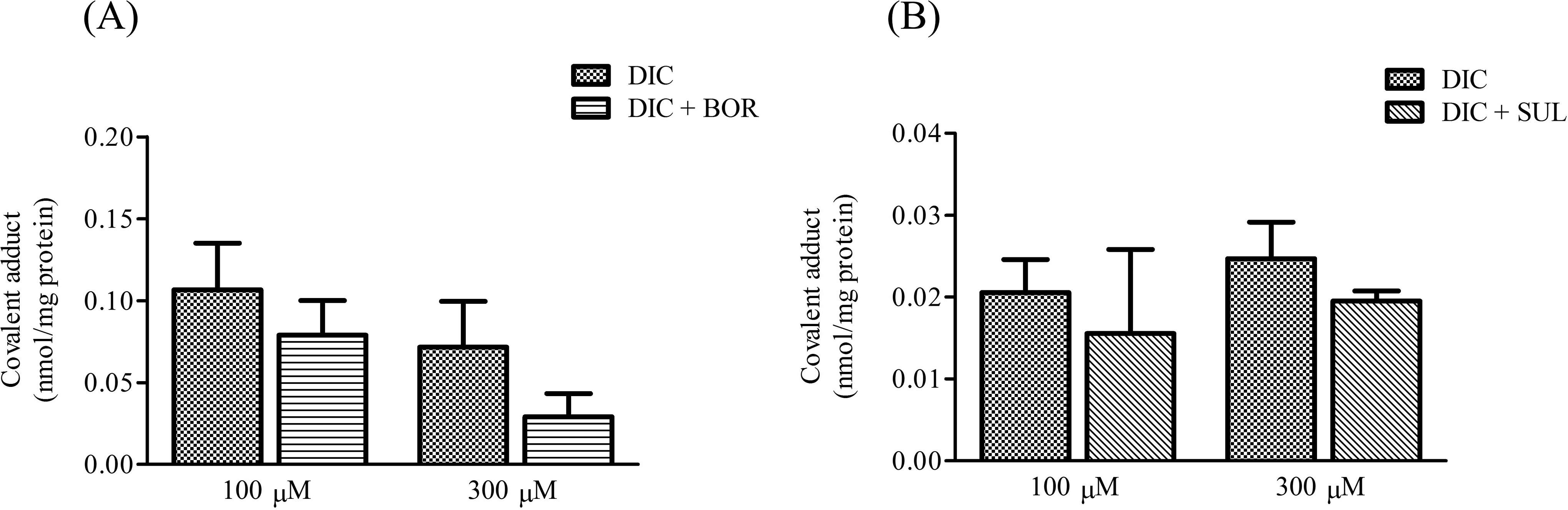
Formation of covalent protein adducts of DIC in SCRH treated with DIC (100 or 300 μM) with or without (A) 1 mM BOR or (B) 1 mM SUL for 3 hours. Values are means (SD), n = 3 to 4. BOR indicates (−)-borneol; DIC, diclofenac; SCRH, sandwich-cultured rat hepatocytes; SUL, sulfaphenazole.
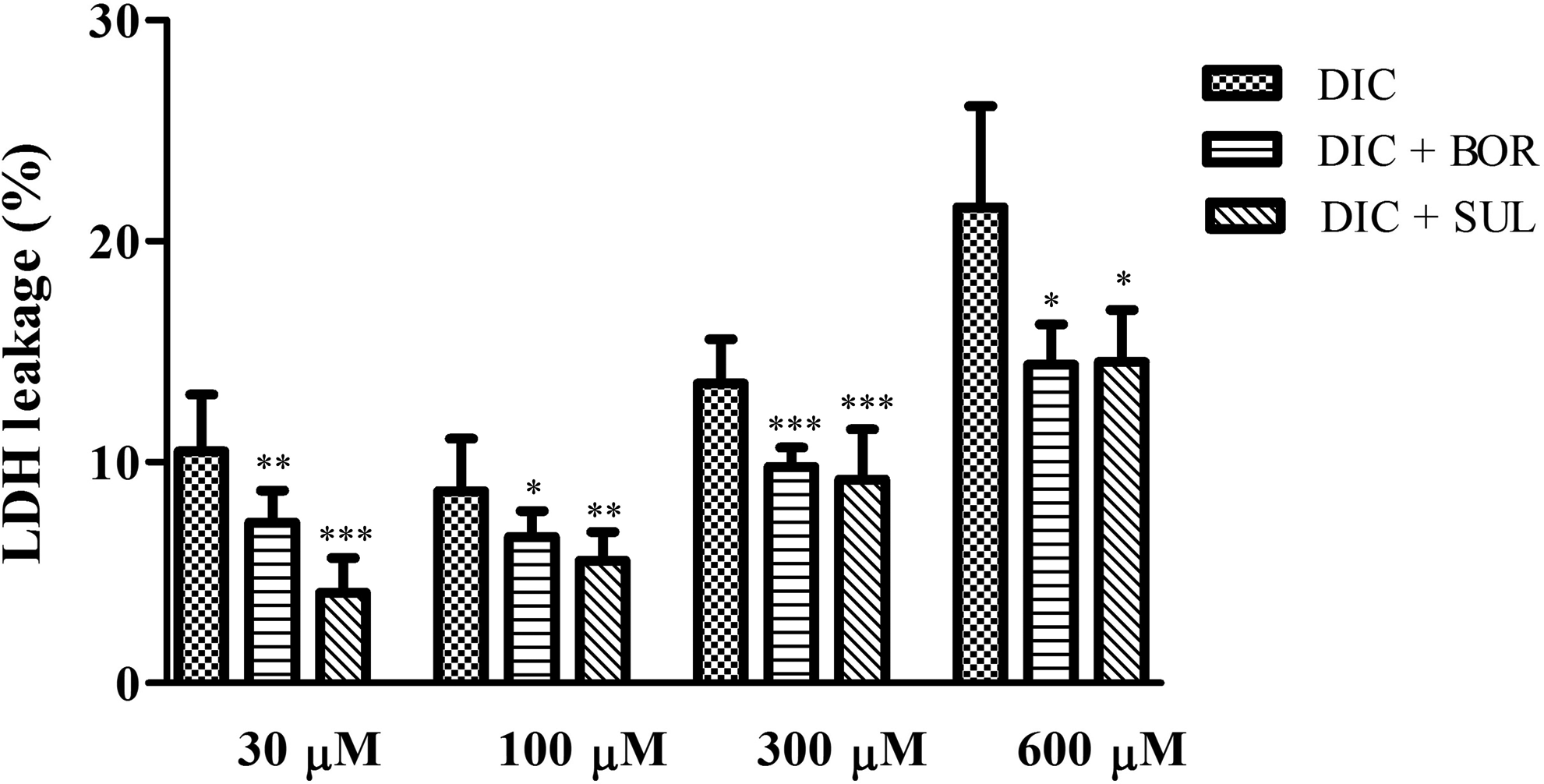
Cell toxicity in SCRH after treatment of 30, 100, 300, or 600 μM DIC for 24 hours with or without 1 mM BOR or 1 mM SUL. Values are means (SD), n = 4 to 8. *P < 0.05, **P < 0.01 and ***P < 0.001 versus corresponding groups receiving only DIC. BOR indicates (−)-borneol; DIC, diclofenac; SCRH, sandwich-cultured rat hepatocytes; SUL, sulfaphenazole.
Diclofenac Treatment and Collection of Medium and Cell Lysate Samples
The intracellular concentrations of DIC, DIC-AG, and 4′OH-DIC, as well as covalent protein adduct formation, were evaluated in SCRH. For time-dependent studies, SCRHs were treated with 300 μM DIC. After incubation for the designated times (1, 3, 6, or 24 hours), the culture medium was removed and cells were washed twice with ice-cold phosphate-buffered saline (PBS). Next, 200 μL ice-cold PBS including 0.2 mM phenylmethylsulfonyl fluoride (PMSF), 1% Triton X-100, and 1% phosphoric acid was added to the cells. The cells were collected with a cell scraper and were homogenized by sonication. For concentration-dependent studies, SCRHs were incubated with various concentrations of DIC (30, 100, 300, or 1,000 μM). After 3-hour exposure, the cells were harvested as described above. For the BOR and SUL inhibition experiment, SCRHs were treated with DIC in the presence or absence of 1 mM BOR or 1 mM SUL, preincubating for 30 minutes. After subsequent 3-hour exposure, the cells were harvested as described above.
A 150-μL cell lysate was deproteinated by adding 150 μL acetonitrile to precipitate proteins, followed by centrifugation at 2,000g for 10 minutes. The supernatants and protein pellets were stored at −80°C until analyzed for DIC, DIC-AG, and 4′OH-DIC and for covalent protein adduct formation.
Determination of DIC and DIC Metabolites by Liquid Chromatography-Tandem Mass Spectrometry (LC–MS/MS)
The DIC-d4 (10 μL of a 0.4-μg/mL stock) was added as an internal standard to 250-µL aliquots of supernatant samples, along with 100 μL 0.2 M acetic acid and 1 mL ethyl acetate. The tubes were shaken for 10 minutes and centrifuged at 2,000g for 10 minutes. The supernatants were evaporated to dryness using a centrifugal evaporator (Tokyo Rikakikai, Tokyo, Japan) at 25°C. The residues were each dissolved in 100 μL LC mobile phase and filtered with a 0.45-μm Millex-LH filter unit (Millipore, Darmstadt, Germany). Aliquots of 10 μL were injected onto the LC–MS/MS. The LC–MS/MS equipment consisted of an LC system (UltiMate 3000 series; Thermo Scientific, Waltham, Massachusetts) and a TSQ Endura triple quadrupole MS with electrospray ionization (Thermo Scientific). For data recording and analysis, Finnigan Xcalibur software (Thermo Scientific) was used. Analysis was performed using a reversed-phase column (COSMOSIL 5C18-AR-II, 2.0 mm × 150 mm, 5 μm; Nacalai Tesque, Kyoto, Japan). The column temperature was set at 40°C and the autosampler was maintained at 4°C. The mobile phase (8.5 mM ammonium acetate and 0.0075% [vol/vol] formic acid in water/methanol [50:50, vol/vol]) was pumped at a flow rate of 0.2 mL/min. Retention times were 5.2 minutes for 4′OH-DIC, 7.6 minutes for DIC-AG, 13.9 minutes for DIC-d4, and 13.9 minutes for DIC. The MS/MS conditions were performed as previously described. 39 The eluate was transferred into the electrospray probe, starting at 1.5 minutes after injection, by switching the MS inlet valve. The conditions of electrospray ionization were set at 3500 V spray voltage, 400°C capillary temperature with the nitrogen sheath, and 10 arbitrary units auxiliary gases. The MS scan was operated in the positive ionization mode. The selected reaction monitoring mode was used with argon as the collision gas at 1.5 mTorr. Compound-dependent parameters are shown in Table 1. The mass resolutions were set at 0.7 full width at half height (unit resolution). The obtained area of DIC, DIC-AG, and 4′OH-DIC were normalized by the area of DIC-d4.
Tandem Mass Spectrometry (MS/MS) Parameters of Individual Compounds.
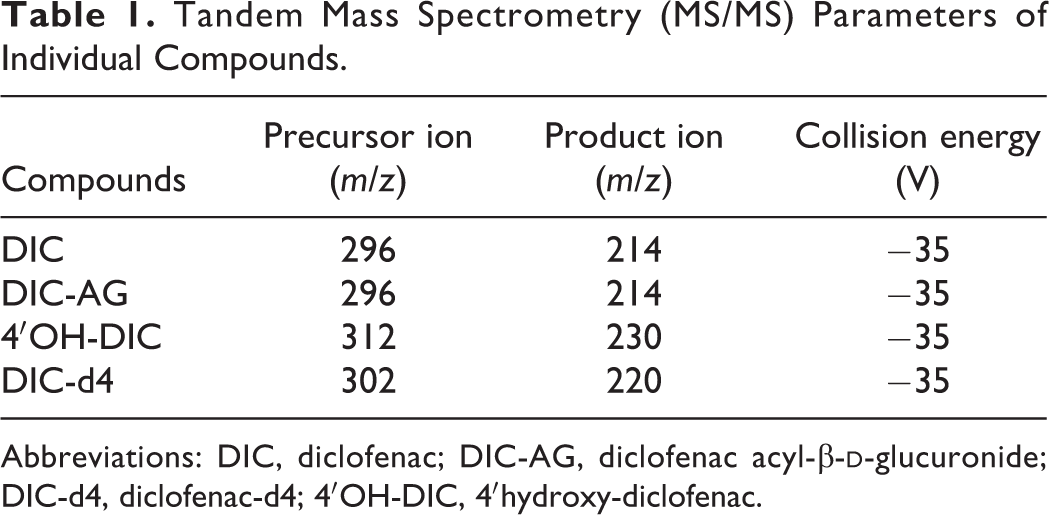
Abbreviations: DIC, diclofenac; DIC-AG, diclofenac acyl-β-
Determination of Covalent Protein Adduct Formation by LC–MS/MS
Protein adduct formation of DIC-AG to hepatocellular proteins was determined as described previously. 26 Briefly, protein pellets were extensively washed with methanol:ether (3:1, vol/vol). After centrifugation, pellets were dried and incubated with 2.5 mL 1 M sodium hydroxide at 80°C for 1 hour. The DIC–protein adducts were extracted with sodium hydroxide and ethyl acetate. The extracts were dried and these residues were reconstituted in 200 μL mobile phase and 40-μL aliquots were injected onto the LC–MS/MS for analysis.
Cytotoxicity Assays
The cytotoxicity of DIC was estimated by determining LDH levels in the extracellular medium of SCRH, using an LDH cytotoxicity detection kit. The LDH release was measured spectrophotometrically at 492 nm (620 nm reference wavelength) 24 hours after DIC (30, 100, 300, or 600 μM), with or without BOR (1 mM) or SUL (1 mM). The percentage of LDH released in each sample minus the spontaneous LDH release control was calculated by comparing its absorbance to that indicating maximum LDH release (1% Triton X-100-treated control cells) minus the spontaneous LDH release control.
Statistical Analyses
Statistical analysis was performed using Dunnett test after an analysis of variance. Values of P < 0.05 were considered to indicate statistical significance.
Results
Intracellular Concentrations of DIC, DIC-AG, and 4′OH-DIC After DIC Treatments to SCRH
The time courses of intracellular concentrations of DIC, DIC-AG, and 4′OH-DIC after 300 μM DIC treatments are shown in Figure 1A. Uptake of DIC into SCRH was relatively rapid and intracellular DIC levels were maintained for up to 24 hours after DIC treatments. Detectable levels of DIC-AG were observed at 1 hour after DIC treatments and were markedly reduced at 24 hours. Similar intracellular concentrations of 4′OH-DIC were observed at up to 24 hours after DIC treatment. Intracellular levels of DIC-AG and 4′OH-DIC were less than 1% and 0.1%, respectively, of DIC levels. The intracellular concentrations of DIC, DIC-AG, and 4′OH-DIC were examined in cells treated with 30, 100, 300, or 1,000 μM DIC (Figure 1B). The intracellular concentrations of DIC increased in a dose-dependent manner. Levels of DIC-AG and 4′OH-DIC were elevated as DIC doses were increased up to 300 μM. However, the intracellular concentrations of DIC-AG and 4′OH-DIC became lower at 1,000 μM DIC. At 3 hours after DIC treatments, the intracellular concentrations of DIC, DIC-AG, and 4′OH-DIC showed relative small variations and relative high levels. Therefore, we examined the effects of BOR and SUL on the intracellular concentrations of DIC, DIC-AG, 4′OH-DIC, and covalent protein adduct.
Effects of BOR and SUL on the Intracellular DIC-AG and 4′OH-DIC Concentrations
We examined the effects of BOR or SUL on intracellular concentrations of DIC-AG and 4′OH-DIC (Figure 2). The intracellular concentrations of DIC-AG in cells pretreated with BOR, then incubated with DIC at either 100 or 300 µM, were significantly lower than those not receiving BOR. Levels of 4′OH-DIC increased significantly, with BOR pretreatment, at 100 μM DIC but were only slightly changed at 300 μM DIC. With SUL pretreatment, both 4′OH-DIC and DIC-AG levels were significantly lower than in cells not receiving SUL.
Effects of BOR or SUL on the Formation of Covalent Protein Adducts
The formation of covalent protein adducts in SCRH was determined after DIC treatment with or without BOR or SUL (Figure 3). The BOR and SUL pretreatments had little effect on formation of covalent protein adducts in DIC-treated cells, although there was a decreasing trend of the formation of covalent protein adduct by BOR and SUL.
Effects of BOR or SUL on the DIC-Induced Cytotoxicity in SCRH
To clarify involvement of reactive metabolites such as DIC-AG and 4′OH-DIC and of covalent protein adducts in DIC-induced cytotoxicity, we examined LDH leakage from SCRH after DIC treatment with or without BOR or SUL (Figure 4). As the dose of DIC was increased, the LDH leakage was elevated. Both BOR and SUL pretreatments significantly reduced cytotoxicity by DIC at 30 to 600 μM. In SCRH treated only with 1 mM BOR or 1 mM SUL, there was little LDH released into the medium (data not shown).
Discussion
The detailed mechanisms linking acute toxicity to formation of reactive metabolites or covalent protein adducts remain unclear, although covalent protein adducts could be involved in idiosyncratic DILI. To clarify involvement of reactive metabolites and covalent protein adducts of DIC in acute liver toxicity, we examined effects of CYP or UGT inhibitors on intracellular reactive metabolites levels, covalent protein adduct formation, and LDH leakage in DIC-treated SCRH.
At 24 hours after DIC treatment, intracellular levels of reactive metabolites such as DIC-AG and 4′OH-DIC were low (Figure 1A). Preliminary study showed little changes in UGT and CYP activities in hepatocytes by DIC treatments. During elimination of intracellular DIC-AG, it is further metabolized to DIC-S-acyl-glutathione thioester by transacylation reactions with GSH 15 and its efflux from cells is mediated by multidrug resistance-associated proteins. 40 Recently, it was reported that the α/β hydrolase domain-containing 10 was involved in deglucuronidation of the acyl glucuronides of mycophenolic acid, tolmetin, probenecid, and DIC. 41,42 The intracellular concentrations of DIC-AG at 1, 3, and 6 hours after DIC treatment were approximately 1% those of DIC. We choose the DIC dose of 300 μM for subsequent experiments because the intracellular concentrations of DIC-AG and 4′OH-DIC were decreased at 1,000 μM DIC due to cytotoxicity by DIC itself. At 1,000 μM DIC treatment, the detachments of a large part of hepatocytes from plate were observed for ∼20 minutes after addition of DIC. The concentrations of DIC, DIC-AG, and 4′OH-DIC in the medium were not determined in our study because intracellular concentrations of drugs and drug metabolites are more likely to affect cytotoxicity. 35
Intracellular DIC-AG and 4′OH-DIC levels were significantly decreased by BOR and SUL pretreatment (Figure 2). The BOR inhibits the glucuronidation of drugs by decreasing levels of the cofactor uridine 5′-diphospho-glucuronic acid and, thus, UGT activity. 43 The BOR pretreatment markedly reduced the production of DIC-AG in cells treated with 100 or 300 μM DIC. However, intracellular 4′OH-DIC concentrations were significantly increased in cells treated with 100 μM DIC. The CYP2C-mediated oxidative reaction could be considered as complementary to reduction in glucuronidation by UGT, although the mechanisms responsible for the increased 4′OH-DIC levels in our experiments remain unclear. The SUL pretreatment decreased intracellular concentrations of not only 4′OH-DIC but also DIC-AG. The SUL was reported to have only a weak inhibitory effect on UGT, a half maximal inhibitory concentration (IC50) value >300 μM. 44 It was also reported that DIC inhibited UGT activity in a dose-dependent manner. 45
In previous reports, relatively high concentrations of DIC (>200 μM) induced cytotoxicity in vitro. 27,46 In our study, at 30 and 100 μM DIC, mild cytotoxicity was observed (Figure 4). Cytotoxicity by DIC was decreased by pretreatment with BOR or SUL. These results suggested that DIC-AG and/or 4′OH-DIC were involved in mediating the cytotoxic effects of DIC on SCRH. A relatively high accumulation of DIC-AG might be important for inducing cytotoxicity because we observed similar levels of LDH leakage in DIC-treated cells receiving BOR and SUL. In preliminary study, there were little changes in cytotoxicity between BOR- or SUL-only treatment and both BOR and SUL treatment at 30 to 600 μM of DIC. Further studies are needed to clarify the effects of both BOR and SUL treatments on the levels of metabolites and covalent protein adducts in hepatocytes. Relatively high UGT2B activities and poor detoxification activity by GSH could result in hepatotoxicity. Miyashita et al demonstrated that DIC-AG enhanced inflammatory responses and cytotoxicity for CD14+ cells via the p38 mitogen-activated protein kinase pathway in human peripheral blood mononuclear cells. 47 The bioactivation of NSAIDs also produced a reactive acyl-CoA intermediate. 11,48 However, an acyl-CoA intermediate was not involved in formation of covalent protein adducts. 25 Further studies are needed to clarify whether an acyl-CoA intermediate is involved in acute cytotoxicity.
Immunochemical detection of hepatic covalent protein adducts formed by reactive metabolites of DIC revealed selective adduct formation to 50, 70, 110, and 140 kDa proteins in the livers of mice treated with DIC. 49 In our experiments, formation of covalent protein adducts was not correlated with LDH leakage (Figures 3 and 4), suggesting that covalent protein adducts of DIC-AG were not involved in the induction of acute cytotoxicity in SCRH. The involvement of covalent protein adducts in acute cytotoxicity might, therefore, be negligible. The contribution ratios of DIC-AG and 4′OH-DIC to the induction of acute cytotoxicity are unclear. It was reported that formation of covalent protein adducts was not involved in acute toxicity or morphologic changes following DIC administration. 50 We found that formation of covalent protein adducts did not change, although intracellular concentrations of reactive metabolites were significantly decreased, in SCRH pretreated with BOR and SUL. Shen et al demonstrated that the production of 4′OH-DIC did not affect the formation of the protein adducts. 21 The DIC-induced liver injury through immunological mechanisms was caused by the formation of covalent protein adducts. 51 In preliminary study, the intracellular accumulation of covalent protein adducts increased in a time-dependent manner up to 24 hours after DIC treatments. Further studies are needed to clarify the formation of covalent protein adducts at additional time points to gain insight into the formation and elimination pathways and kinetics of reactive metabolites.
Several NSAIDs, such as bromfenac, ibufenac, and benoxaprofen, were withdrawn from the market because of idiosyncratic DILI. The NSAID-induced liver injury usually occurs within 6 to 12 weeks. 52 Idiosyncratic DILI could arise from immune-mediated haptenization of reactive metabolites and from danger signals. 53,54 The presence of immune cells is necessary to investigate the immunological mechanisms of idiosyncratic DILI. Our experiments using cultures of only SCRH were, therefore, limited to examining the acute cytotoxicity by DIC.
Our findings indicated that reactive metabolites such as DIC-AG and 4′OH-DIC are involved in acute liver cytotoxicity by DIC. The metabolic activities of enzymes involved in formation of reactive metabolites could be key factors in the induction of acute hepatotoxicity.
Footnotes
Author Contributions
A. Kawase contributed to conception and design and contributed to analysis and interpretation. R. Hashimoto contributed to conception and contributed to acquisition and analysis. M. Shibata contributed to conception and contributed to acquisition. H. Shimada contributed to conception and contributed to interpretation. M. Iwaki contributed to conception and design and contributed to interpretation. All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the “Antiaging” Project for Private Universities, with a matching fund subsidy from the Japanese Ministry of Education, Culture, Sports, Science and Technology (MEXT). This research was also supported in part by the MEXT-Supported Program for the Strategic Research Foundation at Private Universities, 2014 to 2018 (S1411037).
