Abstract
Polyquaternium-22 and polyquaternium-39 are polymers that function as antistatic agents, film formers, and hair fixatives in cosmetic products. These ingredients are being used at concentrations up to 2% (polyquaternium-22, in a rinse-off product) and up to 3% (polyquaternium-39, in rinse-off and leave-on products). The unreacted monomer content of these ingredients was considered low and of no toxicological concern. Limited data showed no skin irritation/sensitization. Although these ingredients were nongenotoxic in bacterial assays, mammalian genotoxicity, carcinogenicity, and reproductive and developmental toxicity data were not available. These polymers, however, are large, highly polar molecules that would likely not be absorbed, and neither local effects in the respiratory tract nor systemic toxicity are expected following product application/exposure. The Expert Panel concluded that polyquaternium-22 and polyquaternium-39 are safe in the present practices of use and concentration in cosmetic formulations.
Introduction
This report reviews the available data relevant to the safety of polyquaternium-22 and polyquaternium-39 as used in cosmetics. These ingredients are polymers, each with a quaternary ammonium moiety. The monomers used to produce polyquaternium-22 are acrylic acid and dimethyldiallyl ammonium chloride. Polyquaternium-39 is produced from the 2 aforementioned monomers and acrylamide. Given the similarities, these ingredients were grouped together. In addition, both ingredients function as antistatic agents, film formers, and hair fixatives in cosmetic products.
The Panel, in its safety assessment of polyacrylamide, had established a limit for acrylamide monomer of 5 ppm in cosmetic formulations. 1
The Panel previously issued final reports on the safety assessment of polyquaternium-7, 2 polyquaternium-10, 3 and polyquaternium-11 4 and concluded that these cosmetic ingredients are safe in the present practices of use and concentration. This conclusion for polyquaternium-7 was reaffirmed by the Panel during a rereview of the safety of this ingredient in cosmetics in 2010. 5 The Panel also determined that the amount of residual acrylamide in polyquaternium-7 is not of toxicological concern. The Panel’s conclusions on polyquaternium-10 and polyquaternium-11 were also confirmed. 6,7
Chemistry
Definition and Structure
Polyquaternium-22 (CAS No. 53694-17-0) is defined as a copolymer of acrylic acid, diallyldimethyl ammonium chloride (DADMAC), and acrylic acid that conforms to the structural formula in Figure 1 8 : another name for this copolymer is acrylic acid-DADMAC polymer.
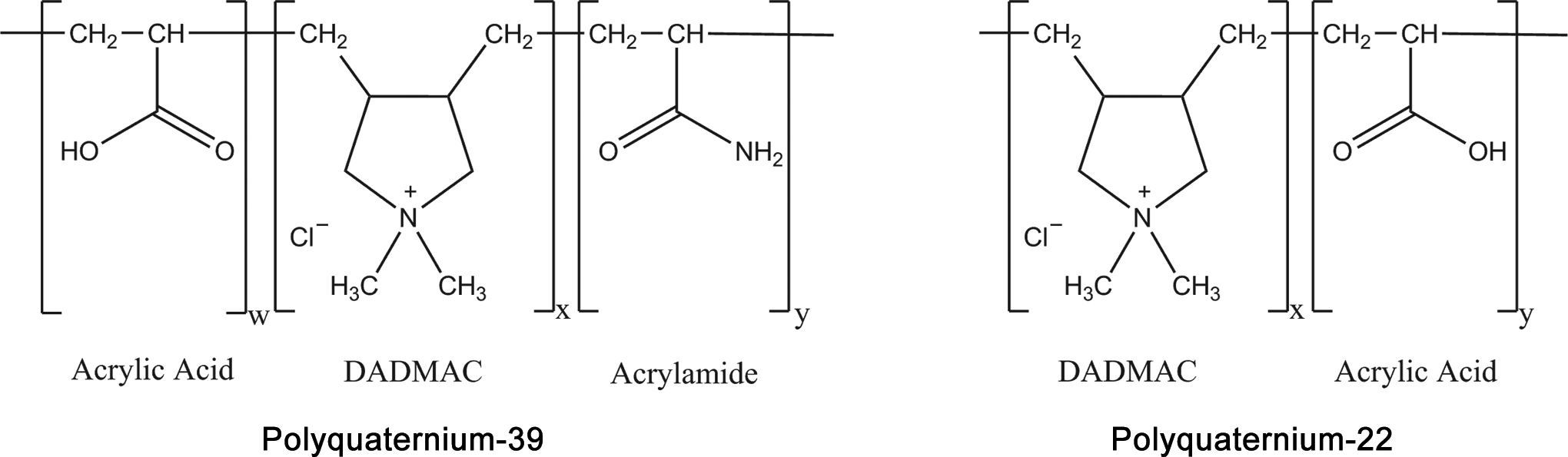
Polyquaternium-22 and polyquaternium-39. DADMAC indicates diallyldimethyl ammonium chloride. Unless specified by a manufacturer, w, x, and y have no standard definition.
Polyquaternium-39 (CAS No. 25136-75-8) is defined as a polymeric quaternary ammonium salt of DADMAC and acrylamide. 8 The structural formula of this chemical is included in Figure 1. 9 Other names for this polymeric quaternium ammonium salt include acrylic acid; polymer with acrylamide and DADMAC; and 2-propen-1-aminium, N, N-dimethyl-N-2-propenyl-, chloride, polymer with 2-propenamide, and 2-propenoic acid. 8
Chemical and Physical Properties
Specifications for polyquaternium-22 and polyquaternium-39 are as follows 10,11 :
Polyquaternium-22
Appearance (viscose water clear liquid)
Solid (40% ± 1%)
pH (2-5, for 1% aqueous solution)
Viscosity (3,000-7,500 cps, at 25°C)
Polyquaternium-39
Appearance (clear viscose liquid)
Solid (9%-11%)
pH (5.5-7.2)
Viscosity (2,000-20,000 cps, at 25°C)
Polyquaternium-22
A trade name material for polyquaternium-22 that has a molecular weight of 4.5 × 105 is described as a clear to slightly hazy, yellow, viscous liquid with a mild, aldehydic odor. 12 The total solid content is listed as 39% to 43% and the pH range is 4.2 to 5.3. Another trade name material for polyquaternium-22 (molecular weight = 1.9 × 105) is described as a clear, yellow viscous liquid with a mild, aldehydic odor. 13 The total solid content is listed as 35% to 40% and the pH range is 4.0 to 5.5.
Polyquaternium-39
A trade name material for polyquaternium-39 (molecular weight = 1.5 × 106) is described as a clear, colorless viscous liquid with a mild, aldehydic odor. 14 The total solid content is listed as 9.4% to 10.4% for this material and the pH range is 5.5 to 7.0.
Methods of Manufacture
Polyquaternium-22
According to 1 source, the starting monomers in the manufacture of polyquaternium-22 are acrylic acid and dimethyldiallyl ammonium chloride. 15
Polyquaternium-39
Polyquaternium-39 is manufactured from acrylic acid, diallyl dimethyl ammonium chloride, and acrylamide. 15
Composition
Polyquaternium-22
Polyquaternium-22 is marketed at concentration ranges of 30% to 45% and ≥94%. The lower concentration range products are defined as aqueous solutions that may also contain preservatives. 15 The higher concentration product is described as a powder with residual moisture content. Typical residual unreacted monomers in this ingredient are acrylic acid (<1,000 ppm) and DADMAC (<1%).
Polyquaternium-39
Polyquaternium-39 is marketed at the following 3 concentration ranges—8% to 11.5%, 41% to 45%, and ≥92%. 15 The 2 lower concentration range products are preserved aqueous polymer solutions. The highest concentration product is an unpreserved powder. Typical unreacted monomers in this ingredient are acrylamide (<1 ppm), acrylic acid (<50 ppm), and DADMAC (<2%).
Use
Cosmetic
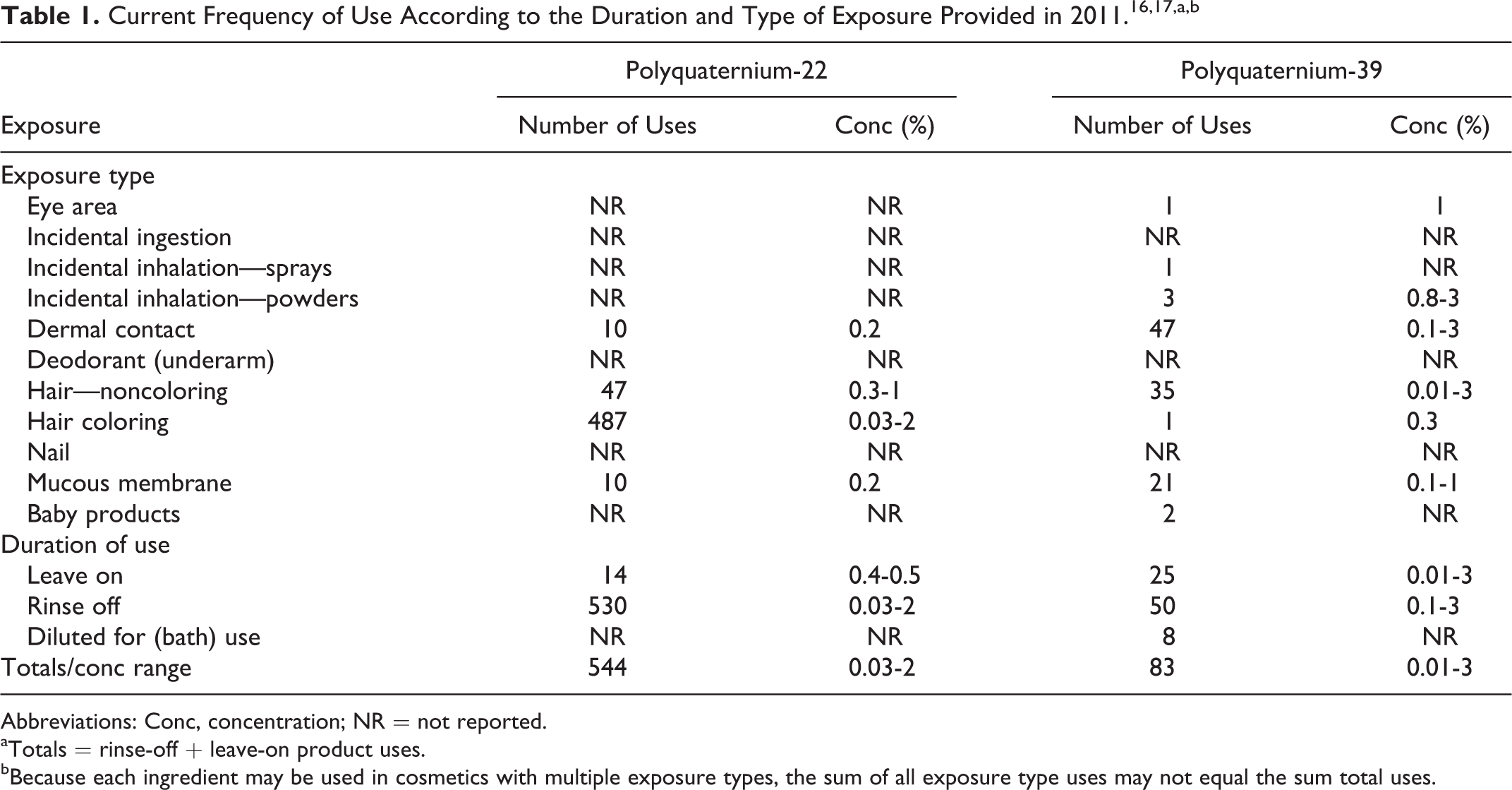
The 2 ingredients reviewed in this safety assessment function as antistatic agents, film formers, and hair fixatives in cosmetic products. 8 According to the information supplied to the US Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP) in 2013, both polyquaternium-22 (544 uses) and polyquaternium-39 (83 uses) were being used in cosmetic products. 16 These data are summarized in Table 1. Polyquaternium-22 is used primarily in hair dye products and other hair care products; polyquaternium-39 is reported to be used at lower use frequencies in these products. Results from a survey of ingredient use concentrations provided by the Personal Care Products Council (also included in Table 1) in 2011 indicate that these 2 ingredients were being used at concentrations up to 2% (polyquaternium-22, in a rinse-off product) and up to 3% (polyquaternium-39, in a rinse-off product and in a leave-on product). 17
Abbreviations: Conc, concentration; NR = not reported.
aTotals = rinse-off + leave-on product uses.
bBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Cosmetic products containing polyquaternium-22 or polyquaternium-39 may be applied to the skin and hair, or, incidentally, may come into contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come into contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
It is possible that polyquaternium-39 is being used in face and neck lotions and powders at concentrations up to 3% and is also being used in indoor tanning preparations (possibly sprays, use concentration data not available). Because this ingredient is used in aerosol/pump sprays or powders, it could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays. 18 –21 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 19,20
Noncosmetic
Polyquaternium-39 is included among the substances that may be safely used as components of paper and paperboard products that come into contact with aqueous and fatty foods. 22
Toxicokinetics
Data on the absorption, distribution, metabolism, and excretion of polyquaternium-22 or polyquaternium-39 were not found in the published literature.
Toxicology
Acute Oral Toxicity
Polyquaternium-22
The acute oral toxicity of polyquaternium-22 was evaluated using 10 healthy male albino rats. 23 The animals were dosed orally with 5 g/kg of the test material. Mortality and systemic observations were made for 3 to 4 hours postdosing and daily thereafter for 7 days. All animals survived the study and it was concluded that the median lethal dose (LD50) was >5.0 g/kg.
Polyquaternium-39
The acute oral toxicity of polyquaternium-39 was evaluated using 10 healthy male Wistar albino rats. 24 The test protocol was identical to that in the preceding study, except for the 14-day observation period after the initial 3- to 4-hour observation period. The authors concluded that the LD50 was >5.0 g/kg and that the test material was nontoxic.
Repeat Dose Toxicity
Repeat dose toxicity studies on polyquaternium-22 or polyquaternium-39 were not found in the published literature, and unpublished data were not submitted.
Ocular Irritation
Polyquaternium-22
In an ocular irritation study, 0.1 mL of polyquaternium-22 was placed in the conjunctival sac of 1 eye in each of the 6 rabbits. 25 The eyes were not rinsed after instillation. Reactions were scored at 1, 2, and 3 days postinstillation. All eyes appeared normal throughout the study, and the test material was classified as a nonirritant.
The ocular irritation potential of an oxidative dye containing 0.16% polyquaternium-22 (pH = 9.5-10) was evaluated in vitro using the hen’s egg test on the chorioallantoic membrane (HET-CAM test). 26 The dye was tested at a concentration of 10% (0.016% active polyquaternium-22), and was classified as a slight irritant (irritation scores of 2.38, 4.51, or 4.60). The positive controls, sodium hydroxide (0.1 M) and sodium dodecyl sulfate (SDS, 1%), yielded scores of 19.98 and 10.21, respectively. The negative control (0.9% sodium chloride) yielded a score of 0.
Polyquaternium-39
The ocular irritation potential of polyquaternium-39 was evaluated in a study involving 6 healthy New Zealand albino rabbits. 27 The protocol was identical to that used in the ocular irritation study discussed earlier in this section. There was no evidence of systemic effects at any time after test substance instillation. All eyes appeared normal during the study and the test material was classified as a nonirritant.
Skin Irritation and Sensitization
Animal
Polyquaternium-22
In a skin irritation study, polyquaternium-22 (0.5 mL) was applied to abraded and intact skin sites on the backs (clipped free of hair) of 6 healthy albino rabbits. 28 The total dose applied was 1.0 mL/rabbit. The test material was applied under an occlusive patch for 24 hours, and reactions were scored at 24 and 72 hours postapplication. None to slight and none to severe erythema were observed at 24 and 72 hours postapplication, respectively. The erythema observed was more severe at abraded sites than at intact sites. There was no evidence of edema. A primary irritation index (PII) of 1.1 was reported, and the test material was classified as a nonirritant.
Polyquaternium-39
The skin irritation potential of polyquaternium-39 was evaluated in a study involving 6 healthy New Zealand albino rabbits. 29 The test protocol was identical to that used in the skin irritation study discussed earlier in this section. There was no evidence of erythema, edema, or abnormal systemic signs during the study. The test material was classified as a nonirritant (PII = 0).
Human
Polyquaternium-22
The skin irritation potential of an oxidative dye containing 0.16% polyquaternium-22 (pH = 9.5-10) was evaluated using 30 individuals (ages not stated). 26 In a single, occlusive patch test, a 10% concentration of the dye (0.016% active polyquaternium-22) was applied for 24 hours. Reactions were scored at 24, 48, and 72 hours postapplication, and the dye was not predicted to be a skin irritant (Organ Injury Scale [OIS] grade = 0.07). The negative control (water) yielded a score of 0, and the positive control (1% SDS) yielded a score of 0.92. The same test concentration of the dye was also applied to 30 individuals (ages not stated) in a multiple patch test. 26 Three 24-hour applications of the dye were made to each individual and reactions were scored according to the same schedule. The dye was not predicted to be a skin irritant (OIS = 0.07). Water (negative control) yielded a score of 0% and 0.3% SDS (positive control) yielded a score of 0.50.
Polyquaternium-39
The skin irritation and sensitization potential of polyquaternium-39 was evaluated in a human repeat insult patch test (HRIPT) using 154 adult individuals (131 women and 23 men; 18-79 years old). 30 The numbers of individuals who completed the induction and challenge phases were 153 and 150, respectively. An occlusive patch (2 cm × 2 cm) moistened with the test material (∼0.2 mL) was applied to the back for 24 hours. Patch applications were made on either side of the midline. Patches were applied on Mondays, Tuesdays, Wednesdays, and Thursdays during induction. There were no patch applications on Fridays, Saturdays, or Sundays. Week number 4 was the nontreatment period, and the challenge phase began at week number 5. Patches were applied to a new test site (24-hour application) on Monday through Thursday. No adverse skin changes were observed during induction. Erythema of negligible significance was observed on either 1 or 2 occasions on each of 2 individuals. Adverse skin effects were not observed during the challenge phase. It was concluded that the test material did not induce skin irritation or sensitization.
Reproductive and Developmental Toxicity
Reproductive/developmental toxicity data on polyquaternium-22 or polyquaternium-39 were not found in the published literature, and unpublished data were not submitted.
Genotoxicity
Polyquaternium-22
The genotoxicity of polyquaternium-22 was evaluated, with and without metabolic activation, using the Ames test on the following bacterial strains: Salmonella typhimurium strains, TA98, TA100, TA1535, and TA1537, and Escherichia coli, strain WP2 uvr A. 31 The test material was evaluated at doses ranging from 100 to 5,000 µg/plate. Positive responses were not observed during the study, and it was concluded that the test material was nongenotoxic in this assay. Results were also negative in another study (same protocol and doses) of a similar polyquaternium-22 trade material. 32
Polyquaternium-39
In another study, the genotoxicity of polyquaternium-39 was evaluated using the Ames test according to the protocol in the preceding study, with the same S typhimurium strains. 33 The test material was nongenotoxic, with and without metabolic activation, over the range of doses tested.
Carcinogenicity
Carcinogenicity data on polyquaternium-22 or polyquaternium-39 were not found in the published literature, and unpublished data were not submitted.
Summary
The monomer precursors for polyquaternium-22 are acrylic acid and dimethyldiallyl ammonium chloride. Polyquaternium-39 is manufactured from acrylic acid, DADMAC, and acrylamide. Acrylic acid (<50 ppm or <1,000 ppm) and dimethyldiallyl ammonium chloride (<1% or <2%) are present in both polymers, but acrylamide monomer (<1 ppm) may be present only in polyquaternium-39. Both ingredients function as antistatic agents, film formers, and hair fixatives in cosmetic products.
According to the information supplied to the US FDA by industry as part of the VCRP in 2011, both ingredients were being used in cosmetic products. Polyquaternium-22 is used primarily in hair dye products and other hair care products; lower use frequencies for polyquaternium-39 in these products were reported. Results from a survey of ingredient use concentrations provided by the Personal Care Products Council in 2011 indicate that these 2 ingredients were being used at concentrations up to 2% (polyquaternium-22, in a rinse-off product) and up to 3% (polyquaternium-39, in a rinse-off product).
Polyquaternium-22 and polyquaternium-39 were classified as nontoxic (LD50 > 5 g/kg) in acute oral toxicity studies. These 2 ingredients were also classified as nonirritants when instilled into the eyes of rabbits. An oxidative dye containing 0.16% polyquaternium-22, tested at a concentration of 10% (0.016% active polyquaternium-22) in an in vitro HET-CAM test, was classified as a slight ocular irritant.
Polyquaternium-22 and polyquaternium-39 were classified as nonirritants when applied, under an occlusive patch, to the skin of albino rabbits. In single and multiple application occlusive patch tests, an oxidative dye containing 0.16% polyquaternium-22, tested at a concentration of 10% (0.016% active polyquaternium-22), was not predicted to be a skin irritant in human individuals. In an HRIPT (occlusive patches), polyquaternium-39 (0.2 mL applied) was classified as a nonirritant and nonsensitizer.
According to Ames test, polyquaternium-22 and polyquaternium-39 were nongenotoxic when tested at doses up to 5,000 µg/plate in the following S typhimurium strains: TA98, TA100, TA1535, and TA1537; in tests of 2 trade materials, polyquaternium-22 was also nongenotoxic in E coli strain WP2 uvr A when tested according to the same procedure. Polyquaternium-39 was not evaluated for genotoxiciy in E coli strain WP2 uvr A, in this study.
The following types of data on polyquaternium-22 or polyquaternium-39 were not found in the published literature: toxicokinetics, repeat dose toxicity, reproductive and developmental toxicity, and carcinogenicity.
Discussion
Relevant data on the composition of polyquaternium-22 and polyquaternium-39 were available. Because these polymers are very large, polar compounds, the Cosmetic Ingredient Review Expert Panel focused its attention on the unreacted monomer levels, which were reported to be <1,000 ppm acrylic acid and <1% dimethyldiallyl ammonium chloride for polyquaternium-22, and <1 ppm acrylamide, <50 ppm acrylic acid, and <2% dimethyldiallyl ammonium chloride for polyquaternium-39. The potential for residual acrylamide monomers was a concern, and the Panel previously established a limit of 5 ppm acrylamide in cosmetic formulations. In this case, the level of acrylamide is <1 ppm in the ingredient, and the ingredient is used at a maximum concentration of 3%, so acrylamide levels are well below the previously established limit. Therefore, the Expert Panel considered these unreacted monomers to be present at levels that would not cause toxicity, given the low use concentrations of these ingredients.
The available bacterial genotoxicity data on both ingredients are negative. The Panel considered the absence of mammalian genotoxicity, carcinogenicity, and reproductive and developmental toxicity data as well as the absence of skin sensitization data on polyquaternium-22. The Panel expressed no toxicity concerns based on their understanding that these polymers are large, highly polar molecules that would likely not be absorbed, and negative acute oral exposure studies indicating that systemic toxicity from cosmetic use is not a concern. The Panel also noted that there is little skin irritation/sensitization potential, based on the negative skin irritation data on polyquaternium-22 and polyquaternium-39 and the negative skin sensitization data on polyquaternium-39.
The Panel discussed the issue of incidental inhalation exposure from propellant and pump sprays, and face and neck powders, relating to the use of polyquaternium-39 in these products. Inhalation toxicity data were not available. However, the Panel considered pertinent data indicating that incidental inhalation exposure to polyquaternium-39 in such cosmetic products would not cause adverse health effects, including chemistry data and data characterizing the potential of these ingredients to cause acute oral toxicity, dermal irritation, or sensitization. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that, in cosmetic formulations, polyquaternium-22 and polyquaternium-39 are safe in the present practices of use and concentration, described in this safety assessment, in cosmetics.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC.
Author Contributions
W. Johnson contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript; B. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript; L. Gill contributed to conception and design, contributed to analysis and interpretation, and critically the revised manuscript; F. Alan Andersen contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; W. Bergfeld contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; D. Belsito contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; R. Hill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; C. Klaassen contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; D. Liebler contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; J. Marks contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; R. Shank contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; T. Slaga contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.