Abstract
Potassium cyanide (KCN) is an inhibitor of cytochrome C oxidase causing rapid death due to hypoxia. A well-characterized model of oral KCN intoxication is needed to test new therapeutics under the Food and Drug Administration Animal Rule. Clinical signs, plasma pH and lactate concentrations, biomarkers, histopathology, and cyanide and thiocyanate toxicokinetics were used to characterize the pathology of KCN intoxication in adult and juvenile mice. The acute oral LD50s were determined to be 11.8, 11.0, 10.9, and 9.9 mg/kg in water for adult male, adult female, juvenile male, and juvenile female mice, respectively. The time to death was rapid and dose dependent; juvenile mice had a shorter mean time to death. Juvenile mice displayed a more rapid onset and higher incidence of seizures. The time to observance of respiratory signs and prostration was rapid, but mice surviving beyond 2 hours generally recovered fully within 8 hours. At doses up to the LD50, there were no gross necropsy or microscopic findings clearly attributed to administration of KCN in juvenile or adult CD-1 mice from 24 hours to 28 days post-KCN challenge. Toxicokinetic analysis indicated rapid uptake, metabolism, and clearance of plasma cyanide. Potassium cyanide caused a rapid, dose-related decrease in blood pH and increase in serum lactate concentration. An increase in fatty acid-binding protein 3 was observed at 11.5 mg/kg KCN in adult but not in juvenile mice. These studies provide a characterization of KCN intoxication in adult and juvenile mice that can be used to screen or conduct preclinical efficacy studies of potential countermeasures.
Introduction
Cyanide (CN) was the first-generation chemical warfare agent used during World War I and has been placed higher on threat lists in recent years due to its increased usage in industry, increased exposure from burning plastic products in fires, 1,2 and low-level use in suicides. 3,4
Potassium cyanide (KCN) is an inhibitor of the cytochrome C oxidase (complex IV) of mitochondria. This inhibition results in an inability to utilize oxygen for the generation of high-energy phosphates (adenosine triphosphate) through oxidative phosphorylation. 5 Potassium cyanide affects organ systems with the highest demand for energy such as the central nervous, cardiovascular, and pulmonary systems. 6 Cyanide intoxication has been described as having 2 component parts: one being a systemic hypoxic state affecting the highly oxygenated biological organ systems, such as the brain, heart, and lungs; and another being a hypotensive state with bradycardia, not solely explained as sequelae of hypoxia.
The primary metabolite of CN is thiocyanate, which is excreted in the urine. 7 Minor quantities of CN may be excreted in the expired air. When CN is present in excess amounts and thiosulfate depleted, it may be metabolized by reaction with hydroxocobalamin (vitamin B12a) to cyanocobalamin (vitamin B12). Minor pathways include metabolism to formate and entry into the single carbon pool, oxidation to cyanate and subsequently CO2, and reaction with cystine to form β-thiocyanoalanine, 2-iminothiazolidine-4-carboxylic acid (in equilibrium with 2-aminothiazolidine-4-carboxylic acid). A number of other sulfur transferases can also metabolize CN.
Cyanide is highly toxic with reported median lethal dose (LD50) values of 2.7 to 8 mg CN−/kg body weight (equivalent to 6.8-20 mg KCN/kg body weight) for rats and mice gavaged with NaCN in water 8,9 and 8.5 mg/kg for KCN in mice. 10 Ferguson 11 reported lethality of 50% and 35% for rats and mice gavaged with 4 and 6 mg CN−/kg in the form of KCN, respectively. Dogs were not considered for these studies due to the relatively low amount of the detoxifying enzyme, rhodanese, which makes this species unusually susceptible to CN exposure compared to other mammals and humans. 12
A number of compounds are being investigated for efficacy in treatment of CN intoxication. 13 -17 However, little information is available on the effects of CN in young animals. Reports have indicated age differences in rhodanese activity, a key enzyme in degradation of CN. 18 The aim of this study was to characterize the natural history of oral KCN intoxication as determined by lethality, clinical signs, histopathology, biomarkers of toxicity, and both CN and thiocyanate toxicokinetics in adult and juvenile mice. The animal model will be used to test the efficacy of medical countermeasures (MCM) under the Food and Drug Administration (FDA) Animal Rule. 19
Materials and Methods
Animals
Adult CD-1 mice, aged 78 to 92 days at dosing, and juvenile CD-1 mice, aged 21 to 25 days at dosing, were obtained from Charles River Laboratories, Portage, MI and Stone Ridge, NY sites, respectively. For juvenile mice, CD-1 dams with approximately 10 nursing pups per litter were received at 10 days of age. Juvenile mice were weaned at 21 to 23 days of age and used at an age of approximately 21 to 25 days on study. All mice were quarantined for at least 10 days prior to use. General procedures for animal care, housing, and environmental conditions conformed to the current Association for Assessment and Accreditation of Laboratory Animal Care International recommendations, current requirements stated in the Eight Edition of the Guide for the Care and Use of Laboratory Animals,
20
current requirements as stated by the US Department of Agriculture through the Animal Welfare Act regulations (as amended) Public Law 99-198, and testing facility standard operating procedures. While on study, all animals were housed individually in plexiglass cages with hardwood chip bedding. The light/dark cycle was set to start at approximately 6:00
Mean Body Weights of Adult and Juvenile Mice in Each Dose Group on Day of Dosing for LD50 and Natural History Study.
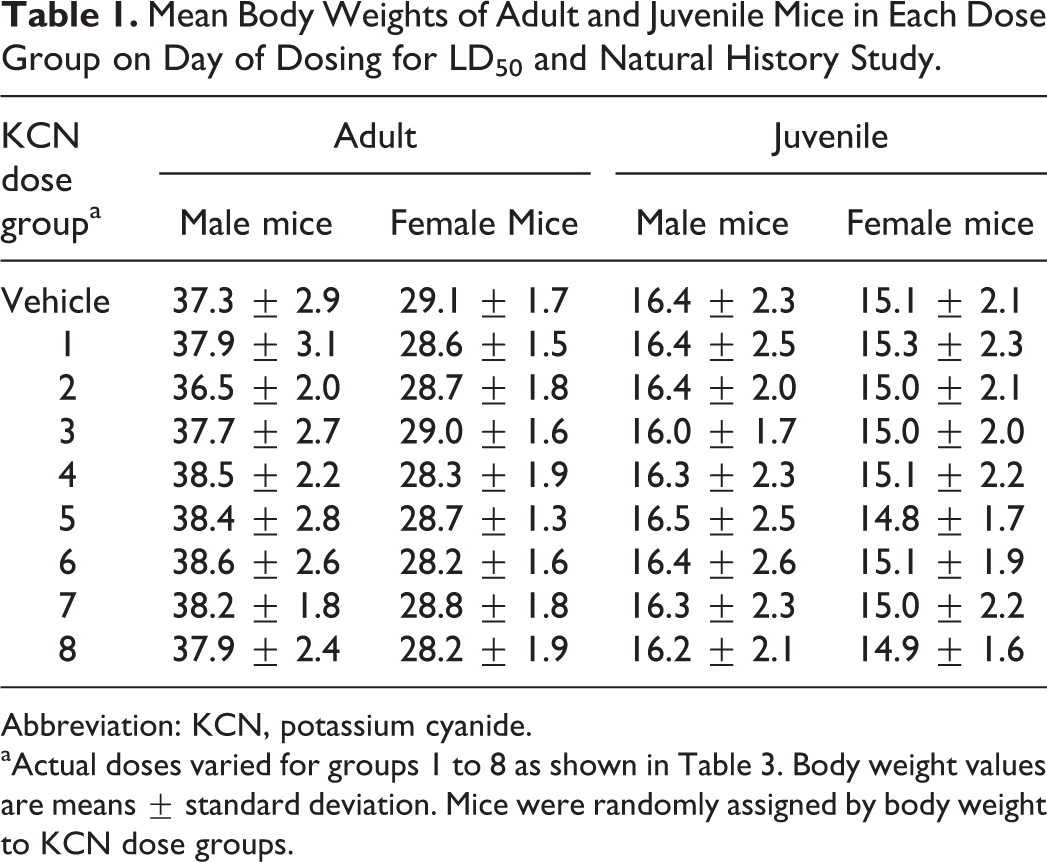
Abbreviation: KCN, potassium cyanide.
aActual doses varied for groups 1 to 8 as shown in Table 3. Body weight values are means ± standard deviation. Mice were randomly assigned by body weight to KCN dose groups.
Mean Mouse Body Weights of Adult and Juvenile Mice on Day of Dosing for Biomarkers and Histopathology Study.
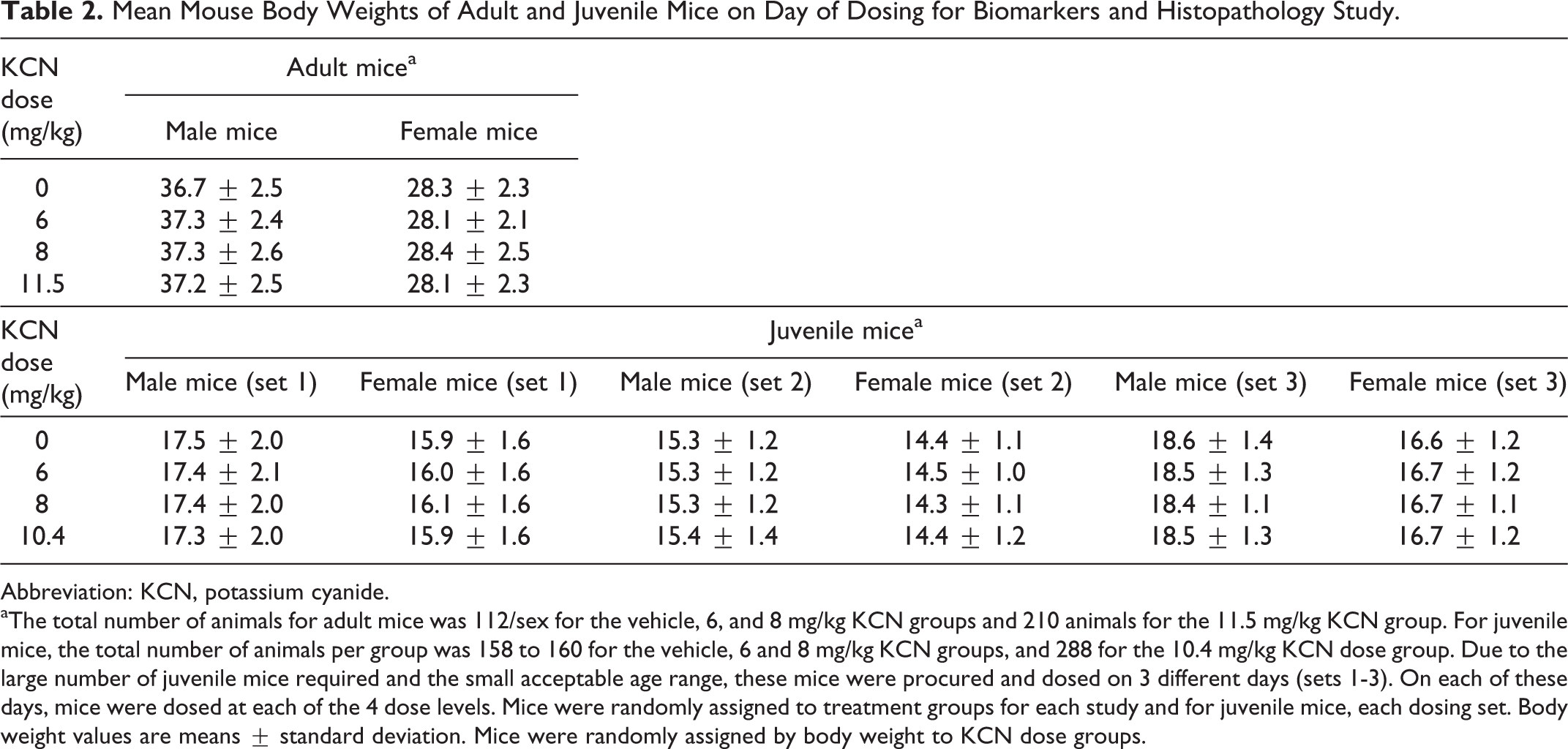
Abbreviation: KCN, potassium cyanide.
aThe total number of animals for adult mice was 112/sex for the vehicle, 6, and 8 mg/kg KCN groups and 210 animals for the 11.5 mg/kg KCN group. For juvenile mice, the total number of animals per group was 158 to 160 for the vehicle, 6 and 8 mg/kg KCN groups, and 288 for the 10.4 mg/kg KCN dose group. Due to the large number of juvenile mice required and the small acceptable age range, these mice were procured and dosed on 3 different days (sets 1-3). On each of these days, mice were dosed at each of the 4 dose levels. Mice were randomly assigned to treatment groups for each study and for juvenile mice, each dosing set. Body weight values are means ± standard deviation. Mice were randomly assigned by body weight to KCN dose groups.
Chemicals
Potassium cyanide (CAS 151-50-8, purity of 99.8%), tetrabutylammonium sulfate, 2,3,4,5,6-pentafluorobenzyl bromide, tetraborate decahydrate, thiocyanate, and potassium 13C15N-cyanide and potassium 13C15N-thiocyanate internal standards were obtained from Sigma-Aldrich (St Louis, Missouri). The I-Stat, point-of-care blood analyzer (Abbott Point of Care, Inc., Princeton, NJ), and CG4+ cartridges for measurement of blood pH and lactate were obtained from Abbott (Princeton, New Jersey). Diagnostic kits for measurement of serum cardiac injury biomarkers (troponins I and T, fatty acid-binding protein 3 [FABP3], and myosin light chain 3 [MYL3]) were obtained from Meso Scale Diagnostic (Rockville, Maryland).
Oral Dosing of Mice
Mice received a single oral dose of KCN in water. Published lethality studies were used to estimate the initial dose levels of CN to use for these studies. 10,21 Dose volumes were based on individual body weights recorded within 1 day of dosing and were approximately 10 mL/kg body weight for adult mice and 12 mL/kg for juvenile mice. Dosing solution concentrations were adjusted to deliver the target KCN doses.
Experimental Design for LD50 and Natural History Study
To verify reproducibility of the model, 3 separate toxicity/lethality studies were conducted in both sexes of adult and juvenile mice and LD50 curves generated for each. Each mouse received a single oral dose of vehicle or KCN at 1 of the 8 dose levels (Table 3). End points used to evaluate the potential toxicity were mortality, clinical signs, and body weights. Dose levels were adjusted slightly for replicates 2 and 3 based on lethality results of the first replicate. Dosing formulations were prepared separately for each replicate of the toxicity study. Six male and 6 female adult or juvenile mice were dosed at each dose level on each of the 3 days (Table 3). Two juvenile mice that died due to gavage trauma (1 from each of replicate days 1 and 2) were excluded from all analyses.
Experimental Design for LD50 and Natural History Study.
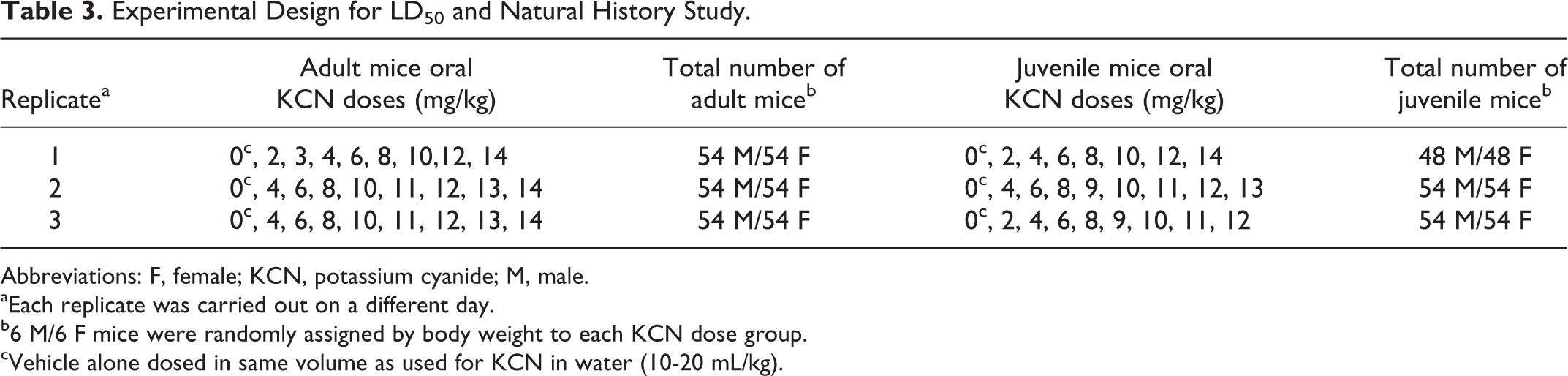
Abbreviations: F, female; KCN, potassium cyanide; M, male.
aEach replicate was carried out on a different day.
b6 M/6 F mice were randomly assigned by body weight to each KCN dose group.
cVehicle alone dosed in same volume as used for KCN in water (10-20 mL/kg).
Clinical Observations
Scheduled clinical signs were recorded at 5, 10, 15, 30, and 45 minutes and at 1, 1.5, 2, 4, 8, and 24 hours postchallenge. The percentage incidence of each adverse sign at each time point was determined based on the original number of animals dosed (ie, not corrected at each time point for survival).
Statistical Analysis
The LD50 values with 95% Fieller confidence bounds were determined by probit regressions. Probit models were fitted to the proportion of nonsurvivors as a function of the base 10 logarithms of KCN concentration (mg/kg). For each age group, it was of interest to establish parallelism and similarity of the LD50 estimates across replicates. The data from the 3 replicate experiments were fitted to 3 separate probit curves and also to a single combined curve. A χ2 likelihood ratio test was performed to compare the 2 types of model fits and to determine whether there was any significant loss of fit when the data from the 3 curves were pooled. The χ2 likelihood ratio test was conducted at the 0.05 significance level. Within each age group, data from males and females were fitted to 2 separate probit curves and also to a single combined curve to investigate parallelism and similarity of the LD50 estimates. For this analysis, data were pooled across replicates for each type of probit model fit. A χ2 likelihood ratio test was performed to compare the 2 model fits and to determine whether there was any significant loss of fit when the data from both sexes were combined. Kaplan-Meier survival curves were used to characterize the time-to-death end point. Data from both sexes were pooled for this analysis.
Experimental Design for Biomarkers and Histopathology Study
This study was conducted in adult (78 to 92 days old) and juvenile (21-25 days old) mice of both sexes to determine biomarkers of toxicity. The dose levels were selected based on the results of the previously conducted LD50 and natural history study described previously. Mice received a single oral dose of 6 mg/kg KCN, a dose that caused mild to moderate toxicity and no lethality; 8 mg/kg KCN, a dose giving moderate to severe toxicity with little lethality; and at the LD50, 11.5 mg/kg KCN for adult mice and 10.4 mg/kg KCN for juvenile mice. These doses were selected to characterize biomarker response and histopathology of KCN dose levels ranging from mildly to severely toxic. A terminal blood sample was collected, under isoflurane anesthesia, from the retro-orbital sinus prior to dosing and at 0.5, 1, 2, 4, 8, 12, and 24 hours and 7 and 28 days postchallenge for adult mice and prior to dosing and at 0.5, 2, 4, 8 and 24 hours and 7 and 28 days postchallenge for juvenile mice. Each mouse was only bled at a single time point and then euthanized. Sufficient adult or juvenile mice were dosed with KCN to give 4 male and 4 female blood samples per dose level and time point.
Analysis of plasma and urine CN and thiocyanate
Plasma CN and thiocyanate were measured in plasma and urine using a modification of the chemical ionization-gas chromatography/mass spectrometry (GC/MS) method of Bhandari et al. 22 The calibration standards, blanks, quality control samples, and experimental plasma samples were processed by derivatization using tetrabutylammonium sulfate in saturated tetraborate decahydrate in water and 2,3,4,5,6-pentafluorobenzyl bromide in ethyl acetate. Ions selected for monitoring were 208 m/z for CN, 210 m/z for 13C15N (CN internal standard), 240 m/z for thiocyanate, and 242 m/z for S13C15N (thiocyanate internal standard).
Pharmacokinetic (PK) analysis was performed using the target dose (mg/kg), target sample collection time points (hour), and the measured concentrations of CN or thiocyanate (μg/mL) in plasma. The concentration–time profiles were evaluated using the noncompartmental analysis module in the WinNonlin software program version 6.2 (Pharsight Corp, Mountain View, California). For a given concentration versus time profile, the values that were interpreted to define the terminal linear phase were identified by the WinNonlin algorithm to estimate the first-order rate constant associated with the terminal linear phase (λz) by performing a regression of the natural logarithm of the concentration over the specified time range.
The concentration–time profiles for the noncompartmental analysis were constructed using the average plasma concentrations of CN or thiocyanate from multiple animals. If a concentration–time profile did not have at least 3 time points with measurable concentrations, noncompartmental analysis was not performed. The concentration–time profiles were not considered to be adequately characterized unless the following criteria were met: (1) the coefficient of determination (r 2) for the terminal linear phase was greater than or equal to 0.85, (2) the time of the last observed concentration was greater than 3 times the half-life, and (3) area under the curve (AUC∞) had less than 20% of the area extrapolated.
Analysis of blood and serum biomarkers
Blood pH and lactate were measured immediately after sampling using an I-Stat with CG4+ cartridges. Serum creatine kinase (CK), potassium, and C-reactive protein (CRP) were measured using a Cobas C501 or C311 clinical chemistry instrument. The cardiac injury panel (serum troponins I and T, FABP3, and MYL3) were quantitated by a multiplex immunoassay method using a Meso Scale Diagnostic kit (catalog #K15161C) as per the manufacturer’s instructions. Serum mouse CK-MB isozyme and histamine were quantitated by enzyme-linked immunosorbent assay using the BioTang kits (catalog #M0583 and #M7548, respectively), as per the manufacturer’s instructions. Statistical analyses of biomarkers of damage were not performed. These analyses served to characterize the target tissues and provide a characterization of the time line of early tissue damage by CN.
Histopathology
Four mice of each age and sex were selected from each treatment group for histopathology on days 1, 7, and 28 postchallenge. The following tissues were weighed (thyroid gland postfixing), processed routinely, sectioned (between 4 and 6 μm), and stained with hematoxylin and eosin for microscopic evaluation by a board-certified veterinary pathologist: brain (olfactory bulb, cerebral cortex, midbrain, cerebellum, brain stem), cauda epididymis, heart, kidneys, liver (median and left lateral lobes), thyroid glands (with parathyroids, if present in routine section), and all gross lesions (observed at necropsy).
Results
To characterize the mouse model of oral CN intoxication, we first conducted an LD50 and natural history study to determine lethality and clinical signs of intoxication versus dose for up to 24 hours postchallenge and to establish the oral LD50 for adult and juvenile mice. A second study was conducted to measure biomarkers and conduct a histopathological evaluation of mice following CN intoxication at 3 KCN dose levels (6 mg/kg [slight toxicity], 8 mg/kg [toxic but not lethal]) and at the LD50 dose level (10.4 mg/kg for juvenile mice and 11.5 mg/kg for adult mice). The toxicokinetics of CN and its major metabolite, thiocyanate, were also determined in adult and juvenile male and female mice.
LD50 and Natural History Study
Published acute LD50 values for rats and mice ranged from 3 to 8 mg CN−/kg, equivalent to 7.5 to 20 mg/kg KCN. 21 Dose levels to define the reproducibility of LD50 determinations using CD-1 adult and juvenile mice were selected to bracket this range and to include some lower toxicity levels.
Effect of Age and Sex on Lethality of Oral Potassium CN
Adult and juvenile, male and female CD-1 mice were challenged with a single oral dose of KCN (in water) at 7 to 8 increasing dosages (2-14 mg/kg). To determine the LD50 and reproducibility of the LD50 determination, 3 KCN lethality curves were generated on each of 3 different days.
Combining all adult mice of both sexes over the 3 days of testing, the mean oral LD50 of KCN was 11.5 ± 0.4 mg/kg (mean ± 95% confidence interval; Table 4). A likelihood ratio test did not indicate a significant loss of fit (P = .058) when the data from the 3 curves for adult mice were combined, indicating reproducibility from day to day. This LD50 value is only slightly higher than a previously reported mouse LD50 value (8.5 mg/kg body weight). 10 The mean oral LD50 in adult male mice (11.8 ± 0.6 mg/kg KCN) was slightly higher than in adult female mice (11.0 ± 0.7 mg/kg), but this difference was not statistically significant due to the nonmonotonicity and greater variability in the data around the predicted curve for adult females (Figure 1).
Probit Models Fitted to Proportions Dead Versus Log10 KCN Concentration.
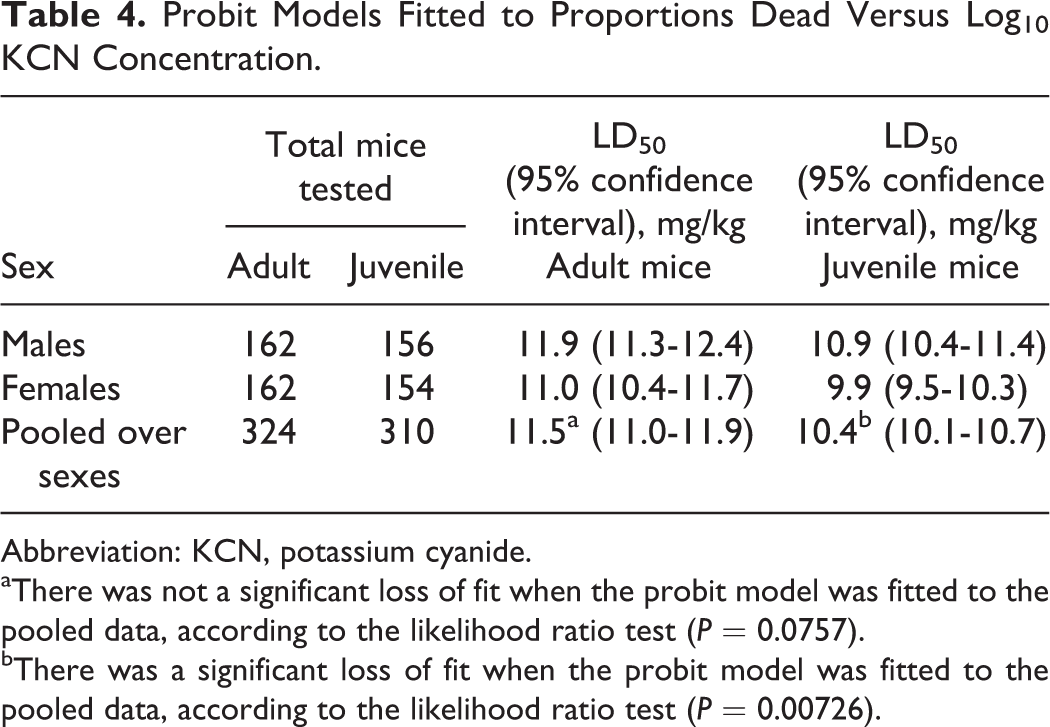
Abbreviation: KCN, potassium cyanide.
aThere was not a significant loss of fit when the probit model was fitted to the pooled data, according to the likelihood ratio test (P = 0.0757).
bThere was a significant loss of fit when the probit model was fitted to the pooled data, according to the likelihood ratio test (P = 0.00726).
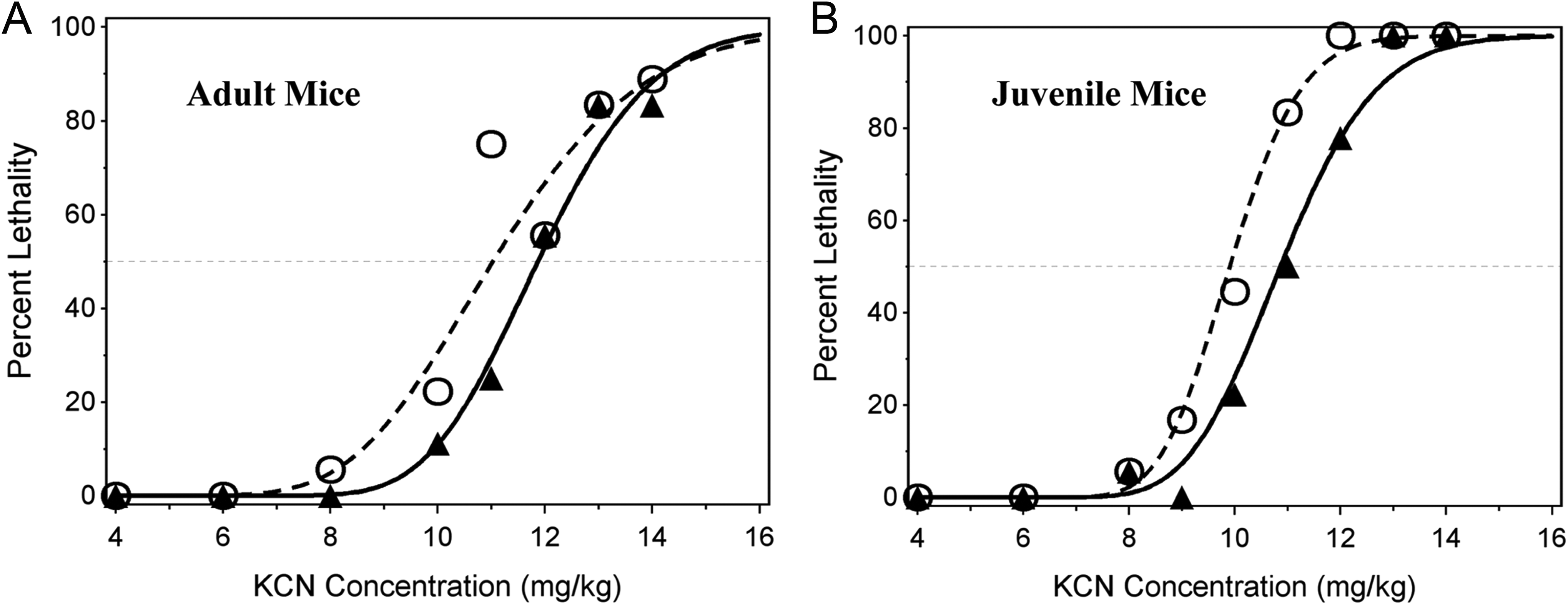
Observed and probit-predicted percent lethality versus KCN dose. Probit curves (lines) and actual data points (symbols) are shown for lethality within 24 hours postdosing in adult male (triangles) and female (open circles) mice (left figure) and for juvenile male (triangles) and female (open circles) mice (right figure). The data points are the mean of 18 mice per dose level, except at the 11 and 13 mg/kg doses for adult mice (mean of 12 mice per dose level) and at the 9 and 11 mg/kg (12 mice per dose level) and 13 and 14 mg/kg (6 mice per dose level) doses for juvenile mice. KCN indicates potassium cyanide.
Combining all juvenile mice of both sexes over the 3 days of testing, the mean oral LD50 of KCN was 10.4 ± 0.3 mg/kg. A likelihood ratio test indicated a significant loss of fit (P = .033) when the data from the 3 curves were combined, indicating significant day-to-day variability. This loss of fit was due to low (33%) mortality observed among males administered 12 mg/kg KCN on one of the experimental days. The LD50 from the combined model (10.38 mg/kg), however, was consistent with the geometric mean of the 3 daily LD50 estimates (10.34 mg/kg). The mean LD50 of oral KCN in juvenile male mice (10.9 ± 0.5 mg/kg) was slightly and statistically significantly higher than in juvenile female mice (9.9 ± 0.4 mg/kg) using the likelihood ratio test (P = 0.00726), indicating that female mice are more sensitive to KCN toxicity. For juveniles, the observed points had very smooth, almost perfectly monotone fits around the predicted lethality curves for both sexes.
The combined data from both sexes for either adult or juvenile mice fit the probit curve very well and indicate that adult mice are less sensitive to the toxicity of oral KCN compared to juvenile mice. The time to death in both adult and juvenile mice was generally dose related (Figure 2). Death occurred rapidly, usually with 30 minutes postchallenge; however, at KCN doses that were not completely lethal, some deaths occurred up to 90 minutes postchallenge in adult mice and up to 60 minutes postchallenge in juvenile mice.
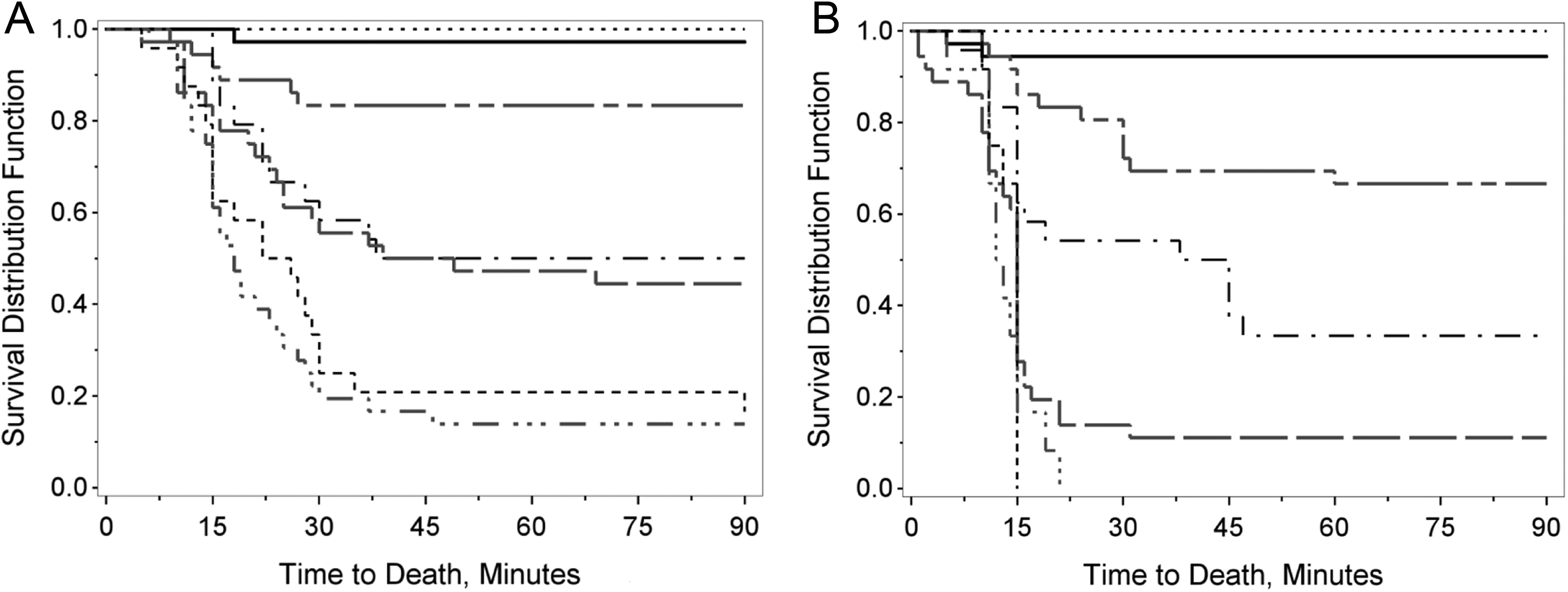
Survival distribution for adults (left) and juvenile (right) mice. Times to death of adult (graph on left) or juvenile mice (graph on right) are illustrated on these 2 graphs at oral KCN doses of 0 to 6 mg/kg (· · · · ·), 8 mg/kg (––––), 10 mg/kg (- - ______), 11 mg/kg (· – · –), 12 mg/kg (–– –– ––), 13 mg/kg (- - - - -), and 14 mg/kg (· · · – · · · –). Survival data from both sexes were pooled for these analyses. Each dose level for adult mice was generated from the survival data of 18 male and 18 female mice (vehicle, 4, 6, 8, 10, and 14 mg/kg KCN dose levels), 12 male and 12 female mice (11 and 13 mg/kg KCN dose levels), or 6 male and 6 female mice (2 and 3 mg/kg KCN dose levels). For juvenile mice, each dose level was generated from the survival data of 18 male and 18 female mice (vehicle, 4, 6, 8, 9, 10, 11, and 12 mg/kg KCN dose levels), 12 male and 12 female mice (2 mg/kg KCN dose levels), and 6 male and 6 female mice (13 and 14 mg/kg KCN dose levels). KCN indicates potassium cyanide.
Clinical Observations in Adult and Juvenile Mice
Adverse clinical signs were recorded at selected time points following oral dosing with KCN. Initial signs observed at low, nonlethal doses of KCN and at early time points following higher doses were inactivity and lethargy, most likely related to oxygen deprivation due to inhibition of electron transport. Mice showing only these 2 signs eventually recovered with no observable adverse effects.
At oral KCN doses of 2 and 3 mg/kg in adult mice (n = 6/sex) and 2 mg/kg KCN in juvenile mice (n = 12/sex), no clinical signs were observed, except for decreased activity and abnormal posture in 1 of 6 adult female mice and difficulty breathing in 1 of 12 juvenile mice at 5 minutes postchallenge with subsequent return to normal.
At 4 mg/kg KCN (n = 18/sex), decreased activity was observed in 55% to 60% of adult mice and 94% of juvenile mice of either sex at 5 to 10 minutes postchallenge, with resolution by 30 to 60 minutes postchallenge for most animals. Lethargy and abnormal posture were infrequent findings in adult mice in the first hour postchallenge, but 1 adult male mouse exhibited prostration, stupor, and pallor at 90 minutes postchallenge with recovery by 2 hours postchallenge.
At 6 mg/kg KCN (n = 18 adult mice/sex and 18 male/17 female juvenile mice), both adult and juvenile mice of both sexes showed a high incidence (>80%) of decreased activity or lethargy between 5 and 15 minutes postchallenge, with recovery starting at 15 to 30 minutes postchallenge. One or 2 animals of each sex, and each age group, displayed difficulty breathing at 5 to 30 minutes postchallenge. A single adult male mouse, 2 juvenile male mice, and 1 female juvenile mouse were prostrate at some point between 5 and 30 minutes postchallenge, having displayed lethargy prior to prostration. One female juvenile mouse was found to be gasping at 10 minutes postchallenge but survived. At 45 minutes and later postchallenge, no breathing abnormalities were detected and the only observed signs were decreased activity, rough coat, and pallor in some mice. By 2 hours postchallenge, all mice appeared normal. No deaths occurred in adult or juvenile mice at this dose level.
At 8 mg/kg KCN (n = 18/sex), 1 adult and 1 juvenile female mouse died at 18 and 10 minutes postchallenge, respectively; no adult or juvenile male mice died (Figures 3 and 4). Greater than 90% of adult and juvenile mice displayed decreased activity, lethargy, or prostration at 5 and 10 minutes post-KCN challenge. Recovery from prostration and lethargy started at 15 to 30 minutes postchallenge. Respiratory effects (difficulty breathing) were observed from 5 to 15 minutes postchallenge with incidence of 33% (adult male), 33% (adult female), 18% to 29% (juvenile male), and 22% (juvenile female). Labored breathing was observed at 15 minutes postchallenge in 17% of adult female mice (only 1 of these 3 eventually died) and at 30 minutes postchallenge in 5.6% of adult male mice (this single animal survived). Tremors and/or convulsions were observed intermittently in 6% to 17% of adult male and female mice (1-3 mice) up to 1 hour postchallenge. In juvenile mice, a higher incidence of tremors/convulsions was observed, with up to 29% of male mice and 22% of female mice demonstrating tremors and/or convulsions within 15 minutes of challenge. Recovery was faster in adult mice. At 1 hour postchallenge, 72% of adult mice (13 mice) and 50% to 59% (9-10 mice) of juvenile mice had recovered and displayed no adverse clinical signs.
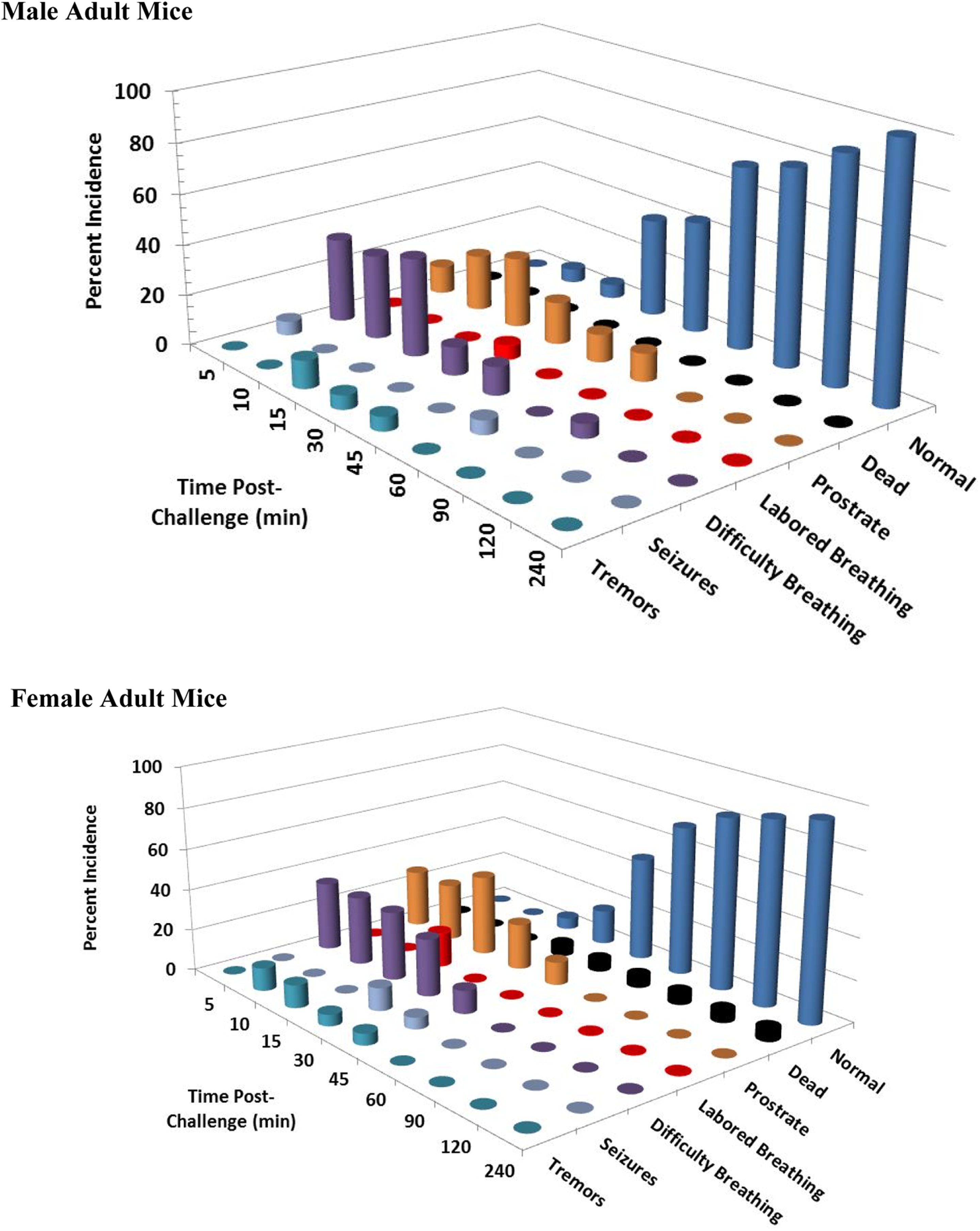
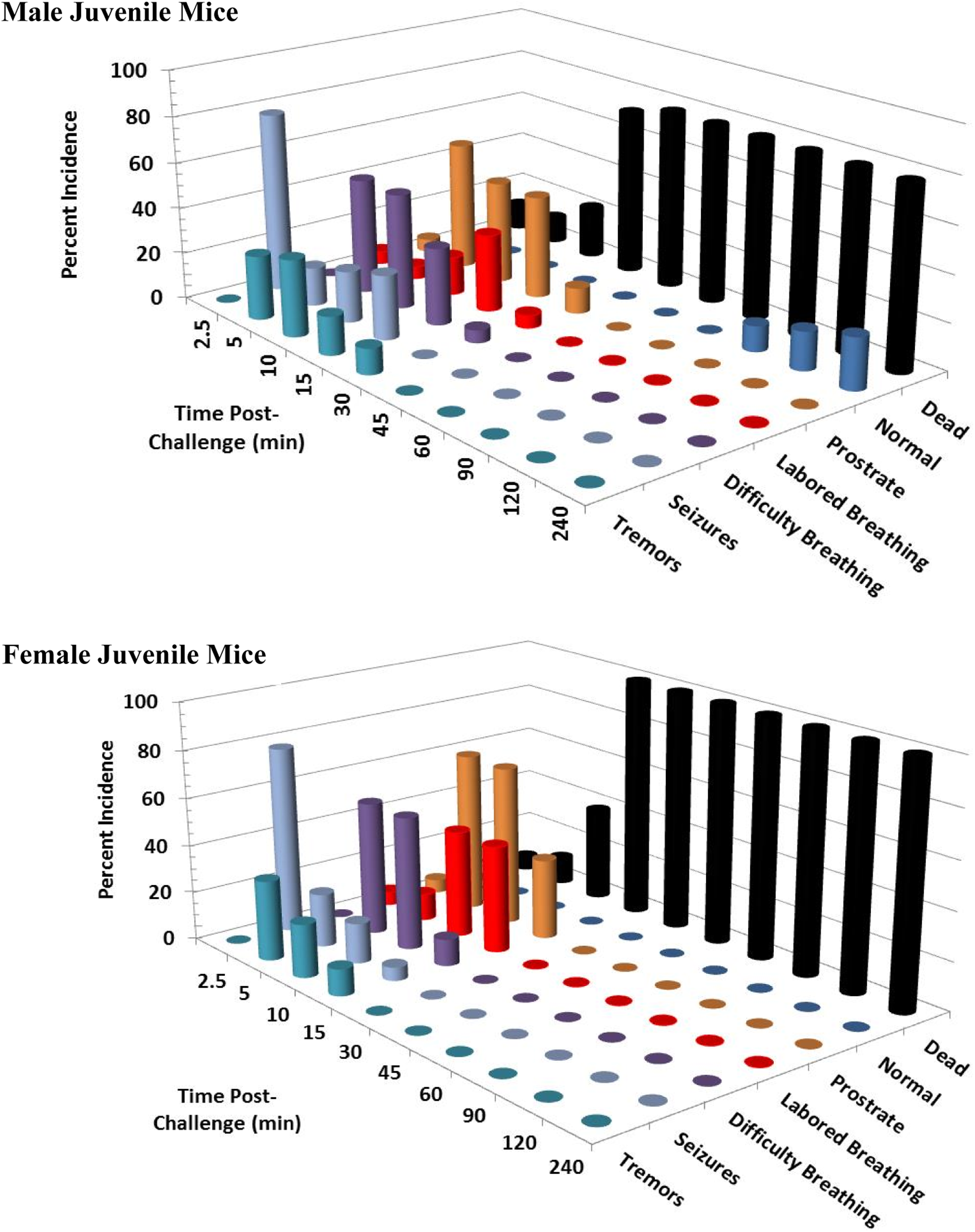
Clinical signs following a single 8 mg/kg KCN oral dose to adult mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 17 to 18 mice of each sex. KCN indicates potassium cyanide.
Clinical signs following a single 8 mg/kg KCN oral dose to juvenile mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 17 to 18 mice of each sex. KCN indicates potassium cyanide.
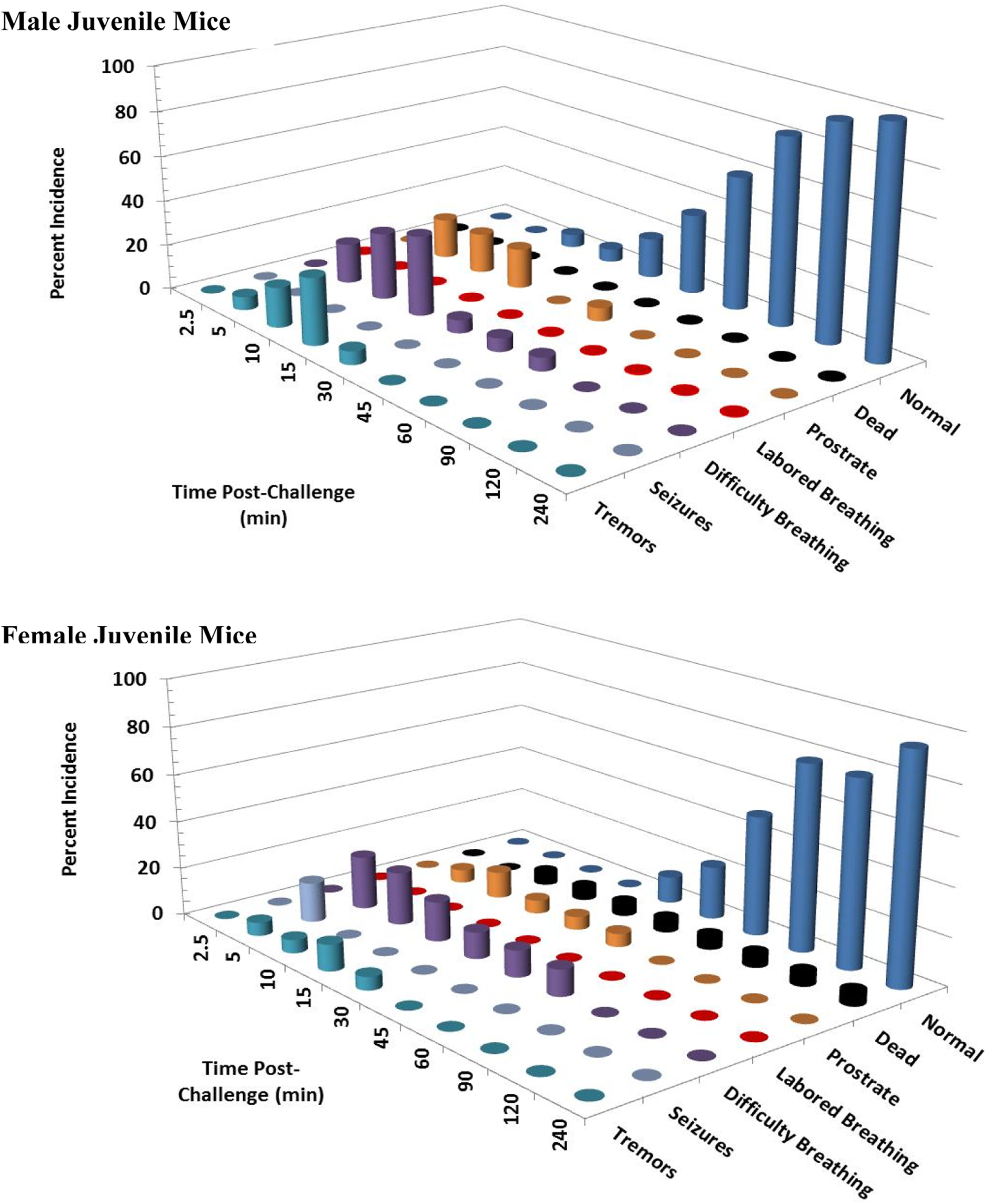
At 10 mg/kg KCN (n = 18/sex), orally administered KCN was more lethal to female versus male mice and juvenile versus adult mice (Figures 5 and 6). For adult mice, 22% of female (4 of 18) and 11% of male (2 of 18) mice died or were euthanized within 30 minutes postchallenge. While for juvenile mice, 22% of male (4 of 18) and 44% of female (8 of 18) mice died or were euthanized within 30 minutes postchallenge, except for a single male mouse that died between 30 and 60 minutes. Within 15 minutes postchallenge, 39% to 50% of adult mice and 44% to 56% of juvenile mice had difficulty breathing. Labored breathing was observed in a few adult and juvenile mice, in some cases progressing to death or euthanasia. Prostration was observed in up to 33% to 44% of adult mice and 28% to 39% of juvenile mice at 5 minutes postchallenge. By 15 minutes postchallenge, the percentages of mice that were prostrate or dead were 61.2%, 72.3%, 55.6%, and 88.9% for adult male, adult female, juvenile male, and juvenile female mice, respectively. Surviving animals showed signs of recovery with full recovery generally observed 4 hours postchallenge, although 1 adult male mouse was scored as prostrate at 240 minutes postchallenge and subsequently recovered. The incidence of tremors and/or convulsions in adult mice (either sex) fluctuated between 0% and 17% for 2 hours postchallenge. However, in juvenile mice, of either sex, the majority of animals (72%) had convulsions within 2.5 minutes of challenge. This high incidence of convulsions continued in surviving juvenile mice up to 15 minutes postchallenge; the incidence decreased at later time points due to prostration and death or recovery. A low incidence of seizures continued until 90, 120, 60, and 45 minutes postchallenge in adult male, adult female, juvenile male, and juvenile female mice, respectively.
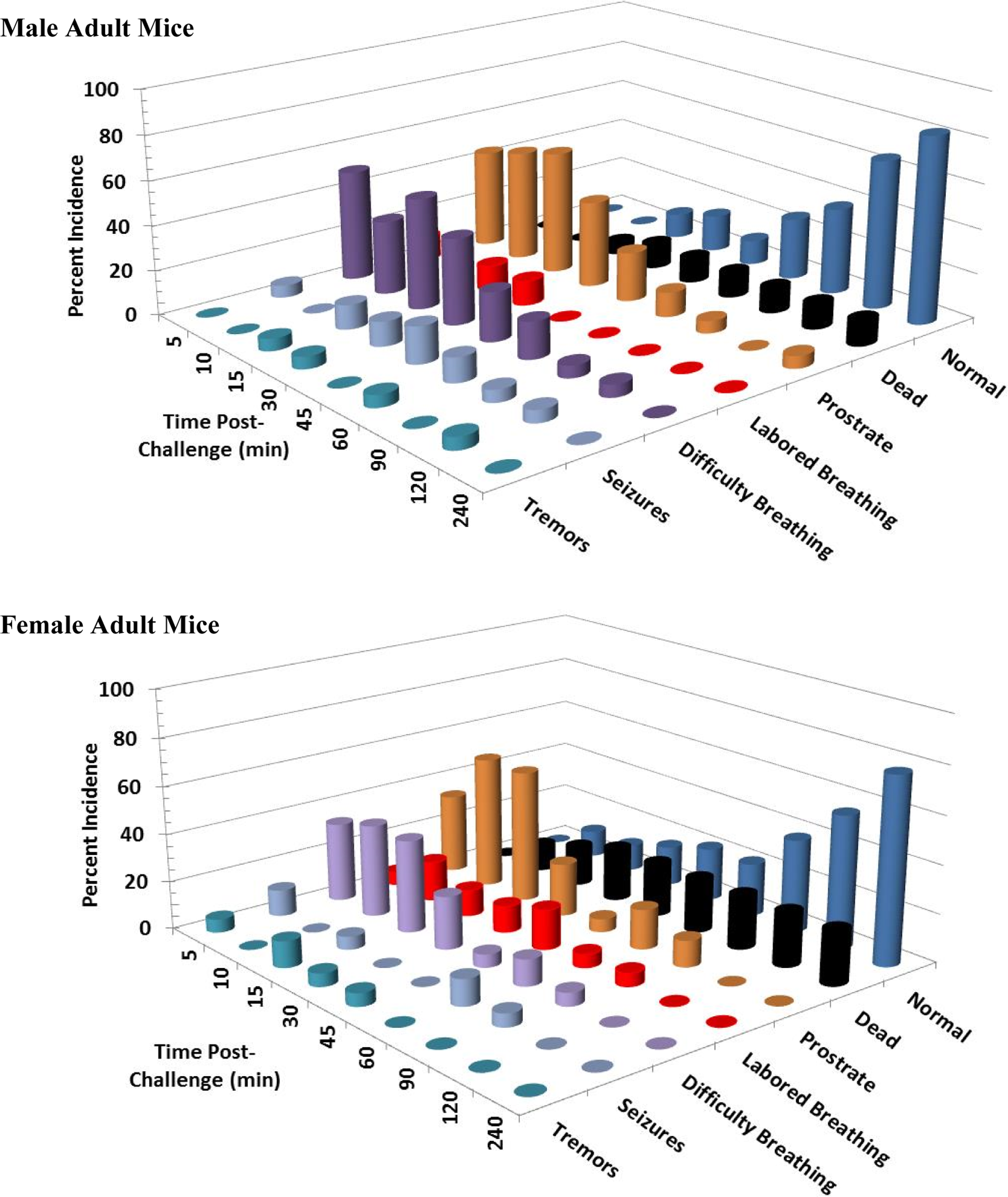
Clinical signs following a single 10 mg/kg KCN oral dose to adult mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals were appeared normal beyond this time point. This experiment was conducted with 18 mice of each sex. KCN indicates potassium cyanide.
Clinical signs following a single 10 mg/kg KCN oral dose to juvenile mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 18 mice of each sex. KCN indicates potassium cyanide.
The 11 mg/kg KCN (n = 12/sex) dose groups also showed sex and age effects on KCN lethality, with female and juvenile mice being more sensitive. Mortality percentages were 25%, 75%, 50%, and 83% for adult male, adult female, juvenile male, and juvenile female mice, respectively. All deaths occurred within 45 minutes of challenge, and 50% were within 5 minutes postchallenge. Difficult respiration and prostration occurred rapidly in both sexes and age groups. Within 15 minutes postchallenge, 50% to 75% of the adult mice (both sexes) and male juvenile mice and 100% of female juvenile mice had difficulty breathing or labored breathing. Tremors were observed in up to 25% to 33% of adult mice (both sexes) and male juvenile mice and in up to 42% of female juvenile mice generally within 30 minutes of challenge. A high incidence of seizures was observed in juvenile mice between 1 and 5 minutes postchallenge and is plotted at 2.5 minutes. The incidence of seizures in male and female juvenile mice between 1 and 5 minutes postchallenge were 58% and 83%, respectively. The subsequent rapid decrease in the incidence of seizures could be due to masking of this sign by prostration and respiratory signs as well as death. The incidence of seizures in adult mice was significantly lower than in juvenile mice and generally not observed prior to 5 minutes. One adult male mouse had seizures at 45 minutes and convulsions at 60 minutes postexposure; this animal survived to 24 hours (end of the study).
At 12 mg/kg (n = 18/sex), orally administered KCN caused a higher incidence of mortality and faster time to death in juvenile versus adult mice. Percentage mortalities of 50%, 56%, 78%, and 100% were recorded for adult male, adult female, juvenile male, and juvenile female mice, respectively (Figures 7 and 8). Difficulty breathing and prostration occurred rapidly in both sexes and age groups. At this dose level, 3 of the 8 surviving adult female and 5 of the 8 surviving adult male mice remained prostrate as long as 2 hours postchallenge; however, these animals eventually recovered completely by 8 hours postchallenge and survived to 24 hours (end of study). Over half of the mice, either sex or age group, had difficulty breathing or labored breathing within 5 minutes of KCN challenge. With one exception, all mice, adult and juvenile of both sexes, had respiratory signs (pale color, difficulty breathing, and/or gasping) within 10 minutes postchallenge. The incidence of tremors and seizures in adult mice increased at this dose level (5%-17% in female and 22%-28% in male mice), with the highest incidence occurring at 1 to 2 hours postchallenge. A 78% incidence of seizures was observed in male and female juvenile mice. In juvenile mice, seizures occurred at less than 5 minutes postchallenge and all but one animal had tremors and/or seizure recorded prior to 15 minutes postchallenge. By 4 hours, all surviving animals appeared normal and remained so at 24 hours postchallenge.
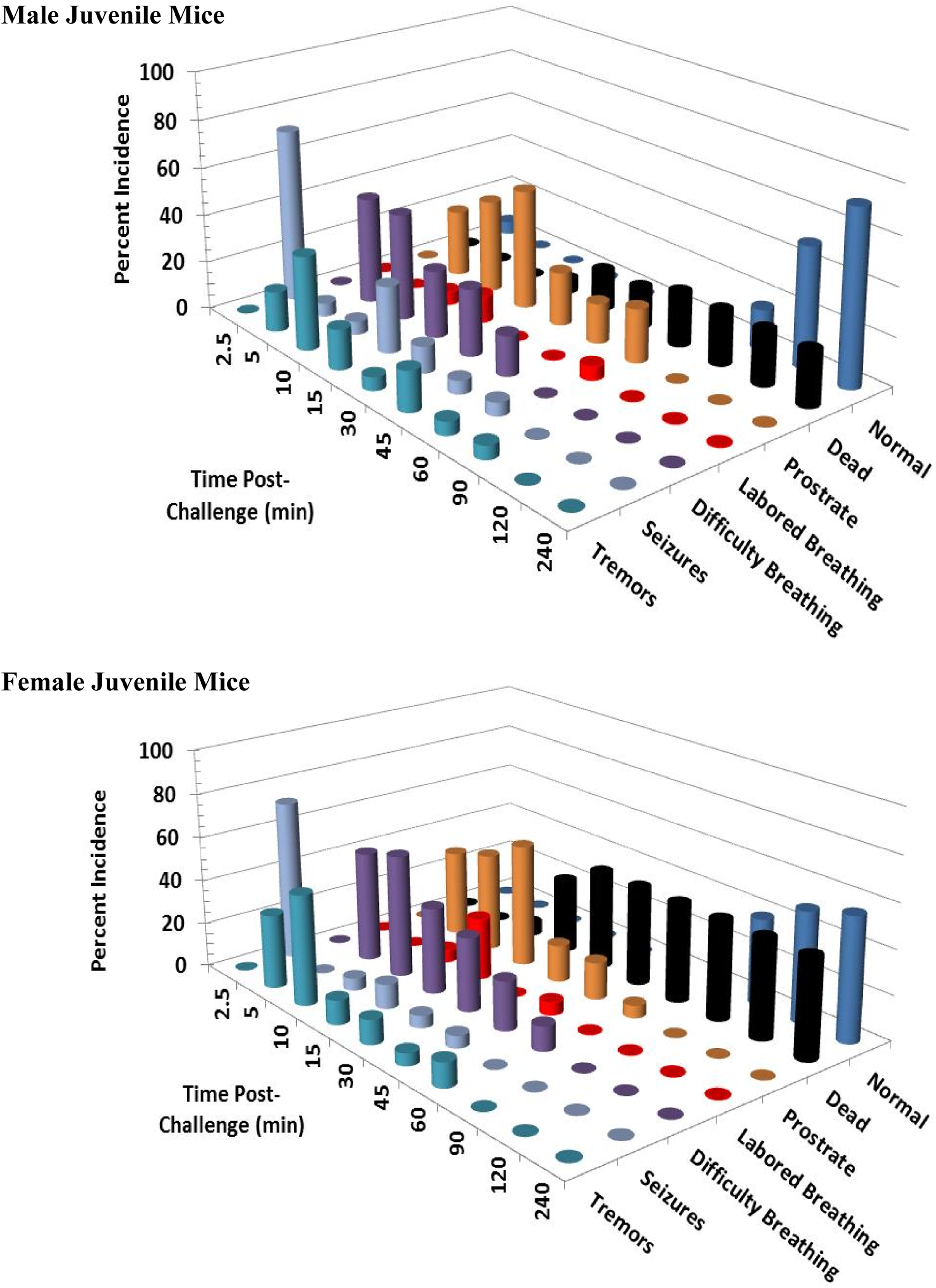
Clinical signs following a single 12 mg/kg KCN oral dose to adult mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 18 mice of each sex. KCN indicates potassium cyanide
Clinical signs following a single 12 mg/kg KCN oral dose to juvenile mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 18 mice of each sex. KCN indicates potassium cyanide.
At 13 mg/kg KCN (n = 12 adult mice/sex and 6 juvenile mice/sex), 83% of adult male and female mice died or were euthanized within 90 minutes postchallenge, all but one adult male mouse within 45 minutes. At this dose level, 100% of juvenile mice died within 15 minutes postchallenge. Difficulty breathing and prostration occurred rapidly in both sexes. At this dose level, 1 or 2 surviving mice of each sex remained prostrate as long as 1 to 1.5 hours postchallenge. Tremors were noted in a few animals from 5 to 60 minutes postchallenge. The incidence of tremors or seizures at 1 to 15 minutes postchallenge were 33%, 58%, 100%, and 100% of adult male, adult female, juvenile male, and juvenile female mice, respectively. By 4 hours postchallenge, the few surviving animals appeared normal and remained so at 24 hours postchallenge.
At 14 mg/kg KCN (n = 18 adult mice/sex and 6 juvenile mice/sex), 83% of male and 89% of female adult mice died or were euthanized within 45 minutes postchallenge (Figures 9 and 10). All juvenile mice died or were euthanized at this dose level. All animals had respiratory difficulty within 10 to 15 minutes postchallenge, most within 5 minutes. Animals rapidly became prostrate, and most deaths/euthanasia occurred between 10 and 45 minutes postchallenge in adult mice and within 15 minutes postchallenge in juvenile mice. Tremors and seizures and/or convulsions occurred in less than 5 minutes in all juvenile mice, except one that was prostrate, and in 39% and 33% of adult male and adult female mice, respectively, at 5 minutes postchallenge. Seizures were also observed in 1 surviving adult female mouse at 90 minutes postchallenge. By 4 hours, the few surviving animals appeared normal and remained so at 24 hours postchallenge.
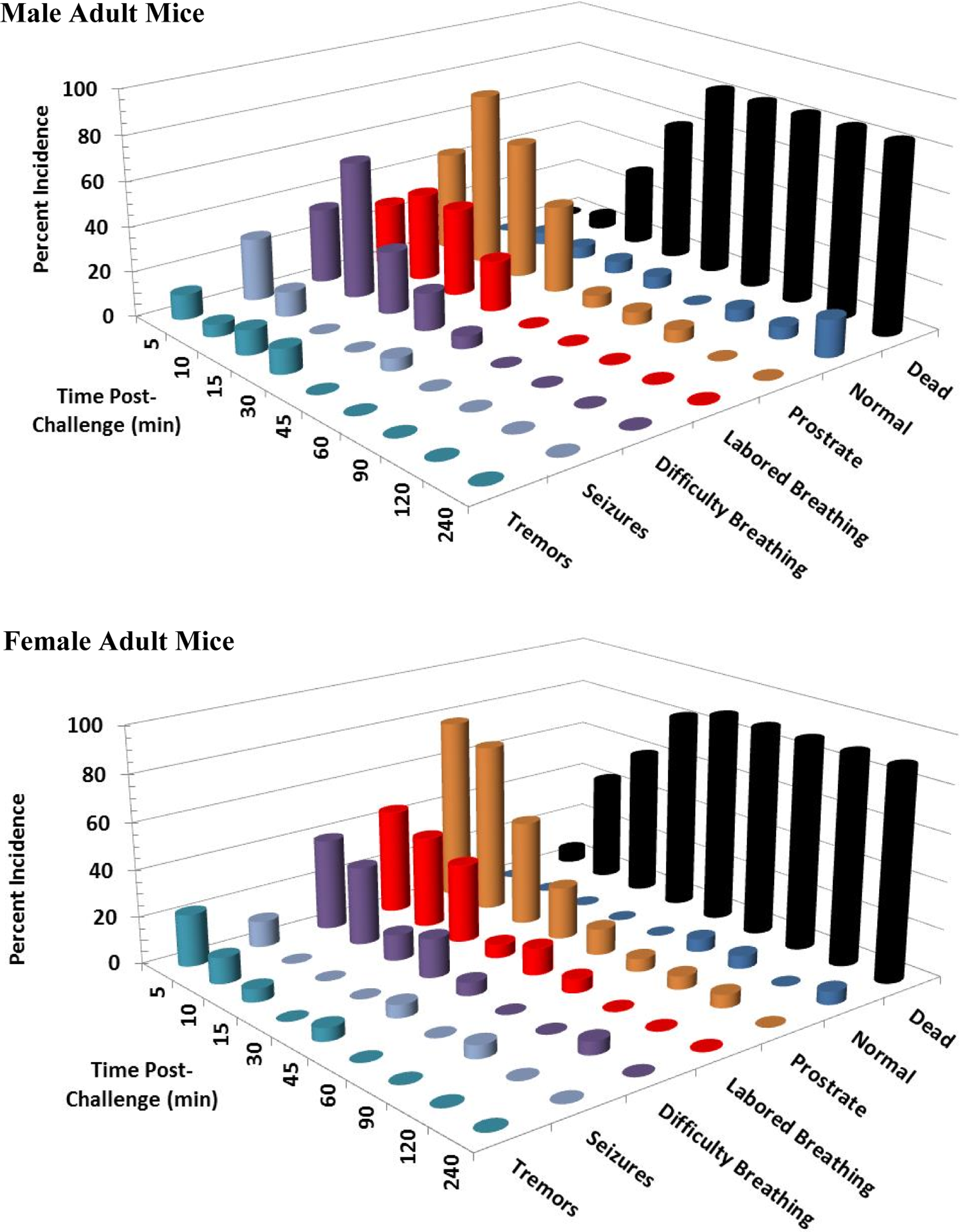
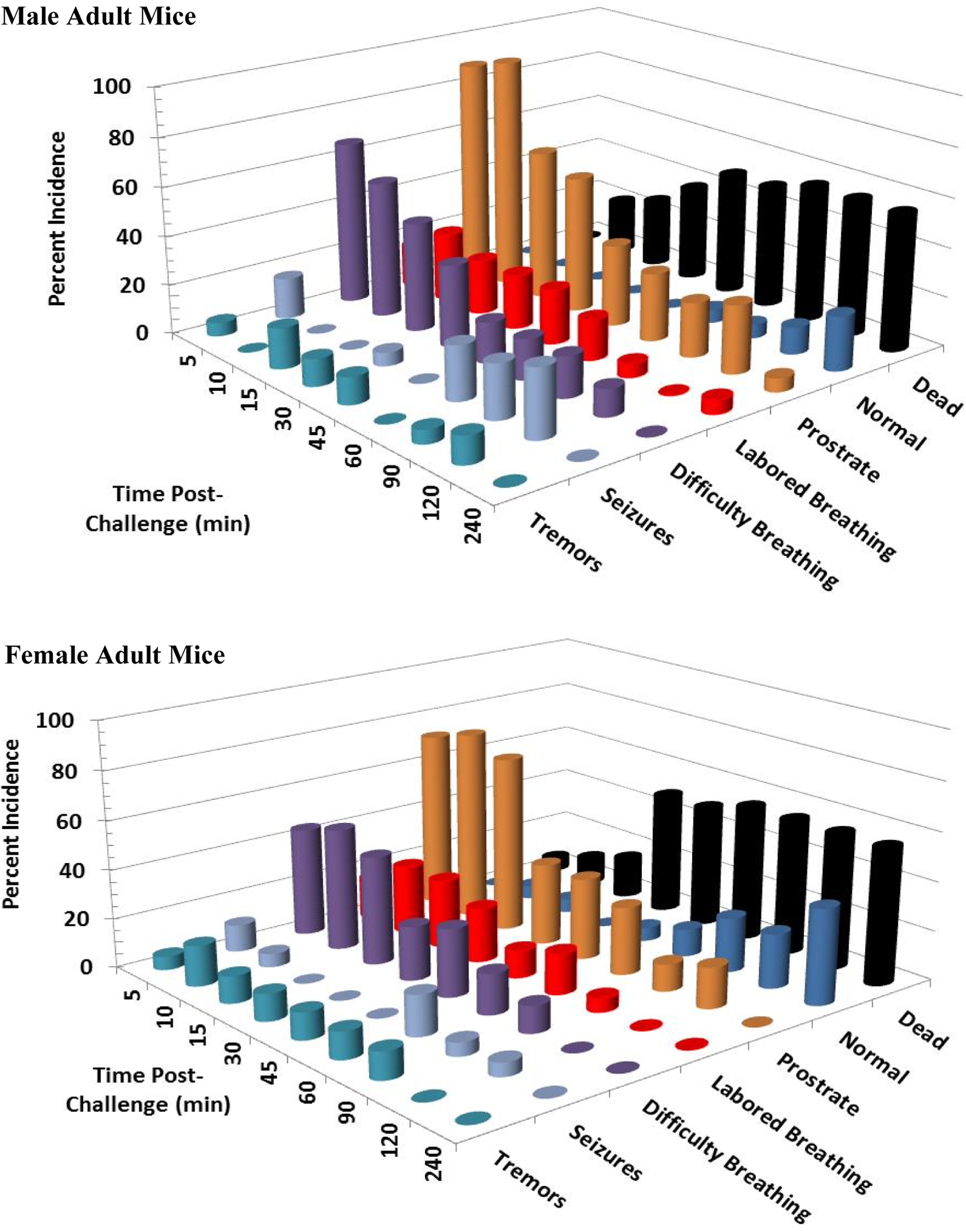
Clinical signs following a single 14 mg/kg KCN oral dose to adult mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 6 mice of each sex. KCN indicates potassium cyanide.
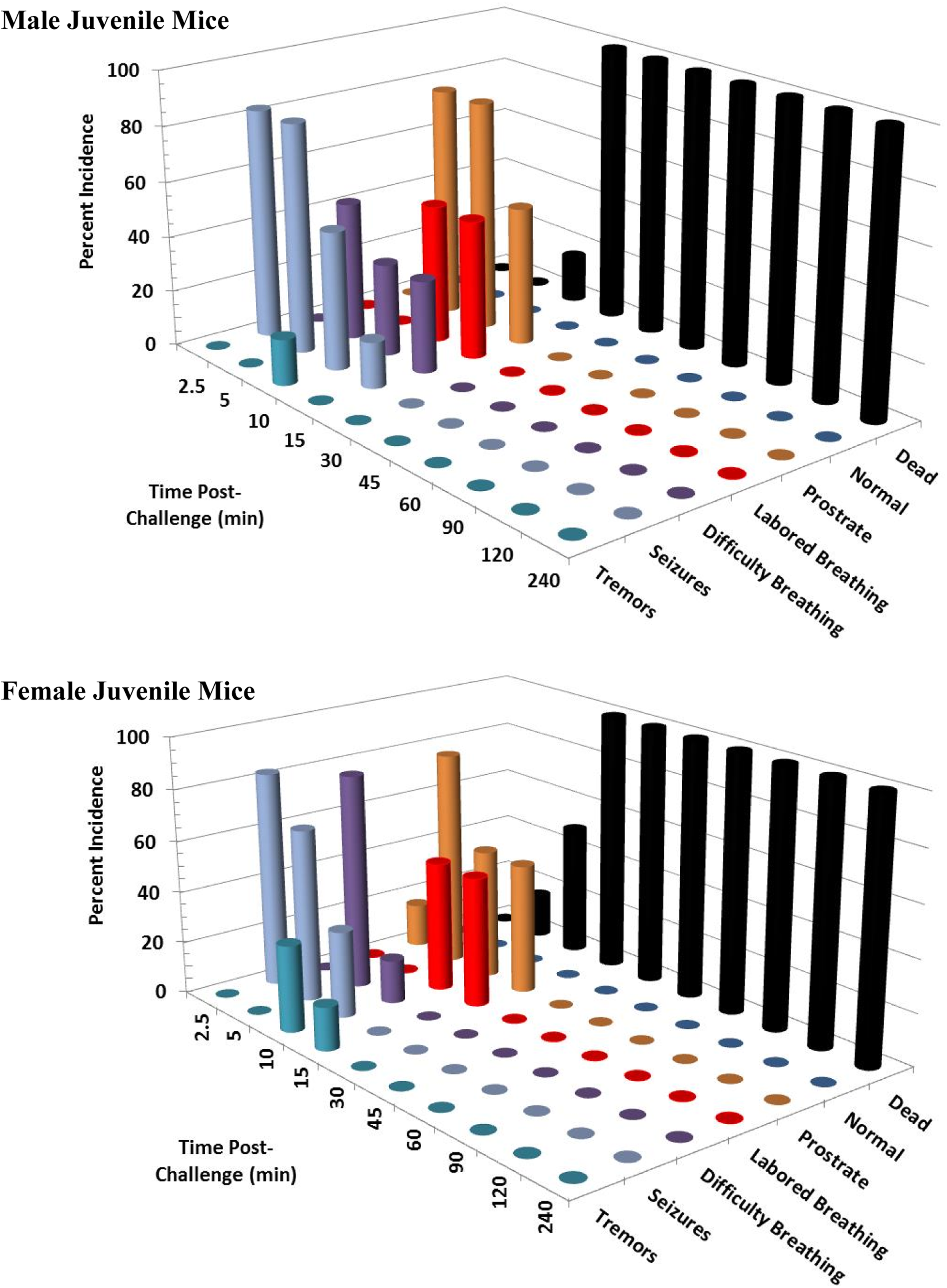
Clinical signs following a single 14 mg/kg KCN oral dose to juvenile mice. Clinical signs were recorded for 24 hours postchallenge. Only the first 240 minutes are shown, as most surviving animals appeared normal beyond this time point. This experiment was conducted with 6 mice of each sex. KCN indicates potassium cyanide.
Biomarkers and Histopathology Study
Plasma CN
Cyanide is rapidly removed from plasma primarily by metabolism to thiocyanate and subsequent urinary excretion. 22 Plasma CN PK parameters were difficult to determine accurately due to the rapid breakdown of CN in plasma. Tmax was approximately 4 hours for the low-dose adult or juvenile mice, but ≤30 minutes for the mid- and high-dose groups. Other toxicokinetic parameters could not be calculated accurately for adult or juvenile mice, due to the known instability and rapid breakdown of CN and interference by endogenous levels of CN at later time points.
Plasma thiocyanate
Due to endogenous sources of CN in the diet and environment, background levels of thiocyanate were detected in control mice. The mean background plasma thiocyanate concentrations in vehicle-treated mice throughout the 28-day study were higher in male mice (3.36 μg/mL in adult and 1.37 μg/mL in juvenile mice) than in female mice (0.50 μg/mL in adult and 0.56 μg/mL in juvenile mice). Maximum plasma thiocyanate concentrations (Tmax) were observed 4 hours postchallenge for all dose groups in both adult and juvenile mice (Tables 5 and 6 and Figure 11), except for the high-dose adult male mice (Tmax of 2 hours). This difference in Tmax values was likely not of biological relevance, as the true Tmax may fall between the 2- and 4-hour time points. The elimination half-life and apparent clearance values were similar (within 50%) in comparisons between adult and juvenile mice (Tables 5 and 6). Mean Cmax values increased with dose and were similar, within 23% between male and female mice and within 26% between adult and juvenile mice at each dose level. Mean AUC0-24 hours values increased with dose and were similar, within 12% between male and female mice and within 23% between adult and juvenile mice at each dose level. Within 24 hours, plasma thiocyanate concentrations approached background levels (Figure 11) and by 7 days postchallenge had returned to background levels.
Pharmacokinetic Parameters for Thiocyanate Following Oral Dosing of KCN in Adult Mice.
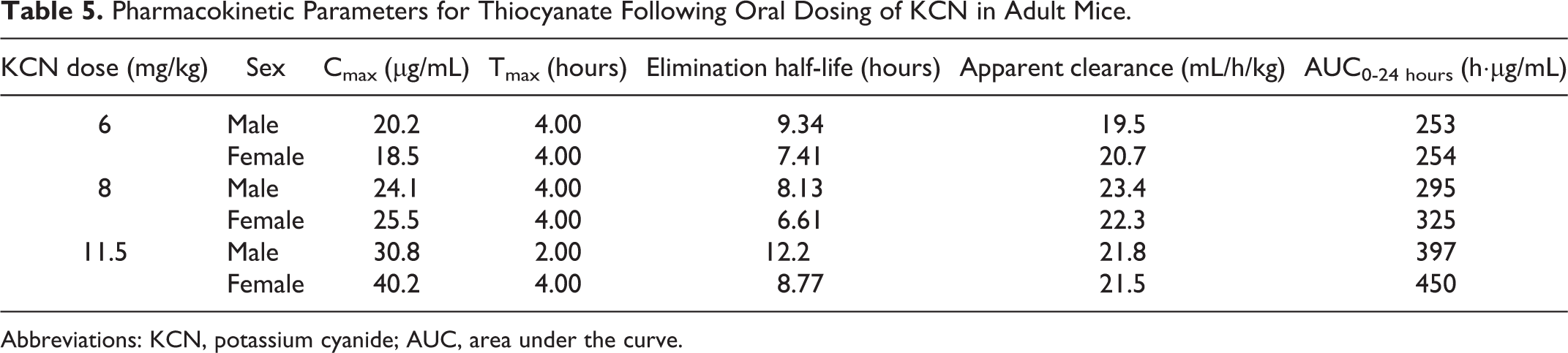
Abbreviations: KCN, potassium cyanide; AUC, area under the curve.
Pharmacokinetic Parameters for Thiocyanate Following Oral Dosing of KCN in Juvenile Mice.
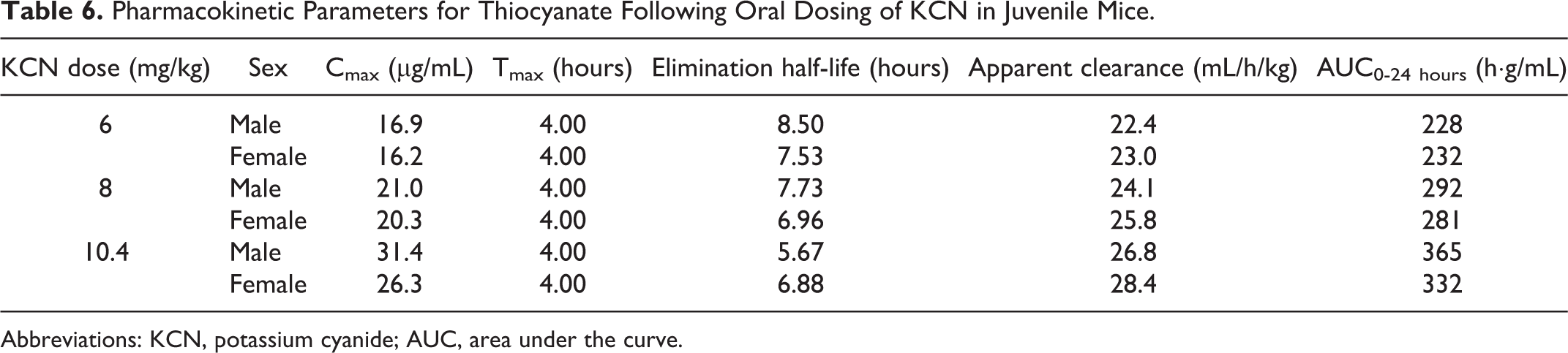
Abbreviations: KCN, potassium cyanide; AUC, area under the curve.
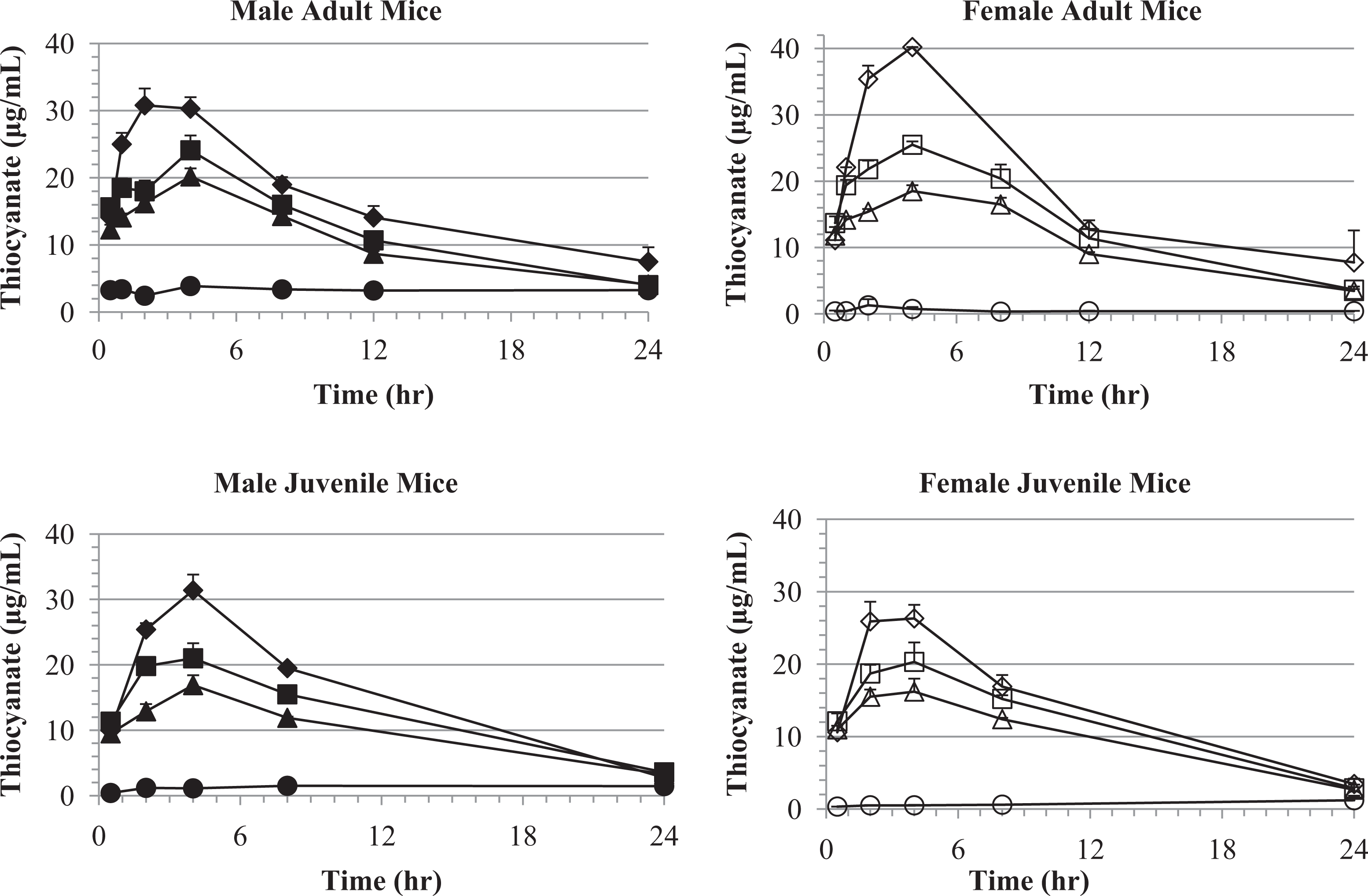
Plasma thiocyanate concentrations versus time following oral KCN challenge of male and female adult and juvenile mice. Plasma thiocyanate levels, following KCN dosing of adult mice, are shown for the vehicle (circle), 6 (triangle), 8 (square), and 11.5 (diamond) mg/kg KCN dose levels. Plasma thiocyanate levels, following KCN dosing of juvenile mice, are shown for the vehicle (circle), 6 (triangle), 8 (square), and 10.4 (diamond) mg/kg KCN dose levels. Data points indicate mean values of 4 mice plasma samples and error bars the SEM. The detection limit was 0.0565 to 0.062 μg/mL. KCN indicates potassium cyanide; SEM, standard error of the mean.
Blood pH and lactate levels
Blood pH and lactate, although nonspecific, were good markers of CN toxicity. Blood pH decreased and blood lactate concentration increased maximally at 0.5 hours post-KCN challenge in adult and juvenile mice of both sexes and then returned to normal levels by 4 hours postchallenge (Figures 12 and 13). Blood pH and lactate concentration profiles were inversely related, and both had a dose-related change in magnitude. The mean blood pH and lactate values had returned to the normal baseline values by 8 hours postchallenge in surviving animals.
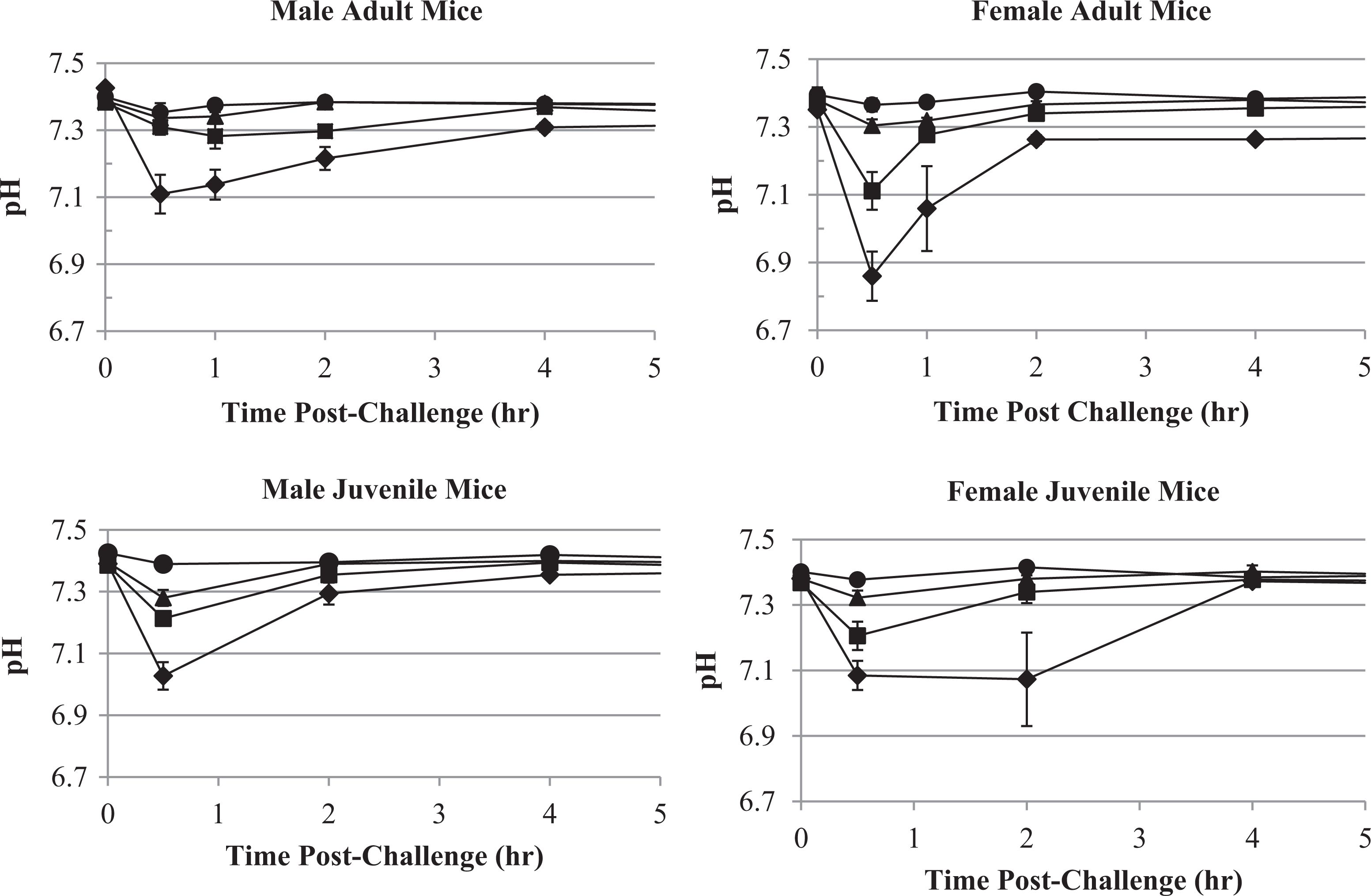
Blood pH versus KCN dose and time following oral KCN challenge of adult and juvenile mice. Blood pH following KCN dosing of adult mice are shown for the vehicle (circle), 6 (triangle), 8 (square) and 11.5 (diamond) mg/kg KCN dose levels. Blood pH following KCN dosing of juvenile mice are shown for the vehicle (circle), 6 (triangle), 8 (square) and 10.4 (diamond) mg/kg KCN dose levels. Data points indicate mean values and error bars the SEM. KCN indicates potassium cyanide; SEM, standard error of the mean.
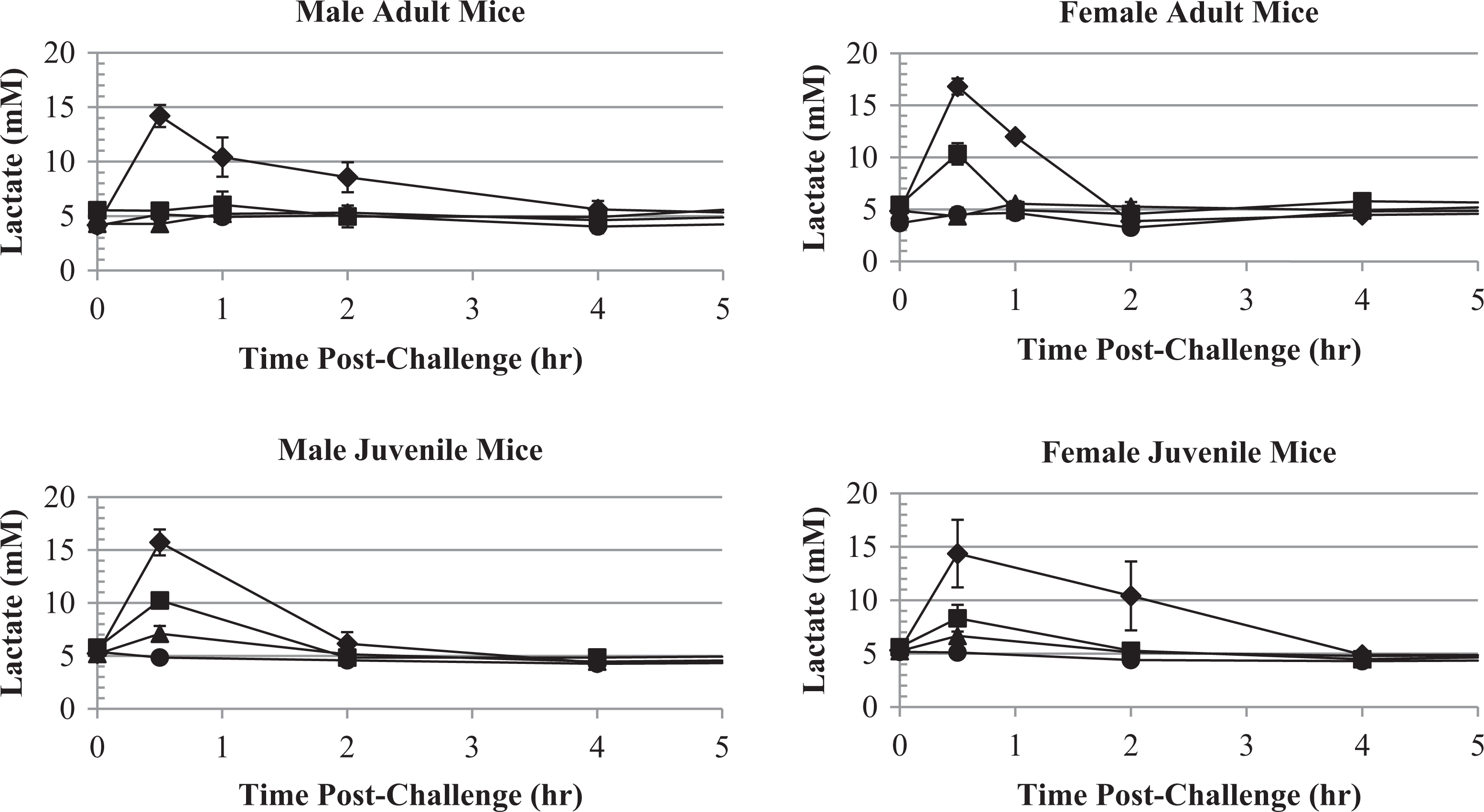
Blood lactate concentrations versus KCN dose and time following oral KCN challenge of adult and juvenile mice. Blood lactate concentration after oral KCN dosing of adult or juvenile mice are shown for the vehicle (circle), 6 (triangle), 8 (square) and 11.5 (diamond; adult mice), or 10.4 (diamond; juvenile mice) mg/kg KCN dose levels. Data points indicate mean values and error bars the SEM. KCN indicates potassium cyanide; SEM, standard error of the mean.
At the 6 and 8 mg/kg KCN dose levels, adult male mice, which were the least sensitive to KCN, had little change in blood pH and lactate compared to vehicle-treated animals. Juvenile mice, which were more sensitive to KCN, displayed a significant decrease in pH and increase in lactate, even at the 6 mg/kg KCN dose level. Therefore, although pH and lactate responses were by no means exclusive to KCN exposure, they were a good marker of toxicity and may even be used to determine the expected outcome in mice. In this study, since blood samples were terminal, a correlation between pH or lactate and mortality could not be directly determined.
Biomarker Analysis
Several markers of heart or muscle damage were measured following KCN challenge. Troponins I and T, FABP3, MYL3, and CK-MB were analyzed as specific biomarkers of cardiac damage. 23 No significant changes in troponins I and T, MYL3, or CK-MB were detected following KCN challenge in adult or juvenile mice. A significant increase in FABP3 was observed in the first 0.5 to 2 hours post-KCN challenge in adult mice (Figure 14). This increase was statistically significant only at the highest KCN dose level (11.5 mg/kg). No increase was observed in juvenile mice; however, this could be due to the higher baseline concentrations (∼100-200 ng/mL) observed in the vehicle control group for juvenile mice.
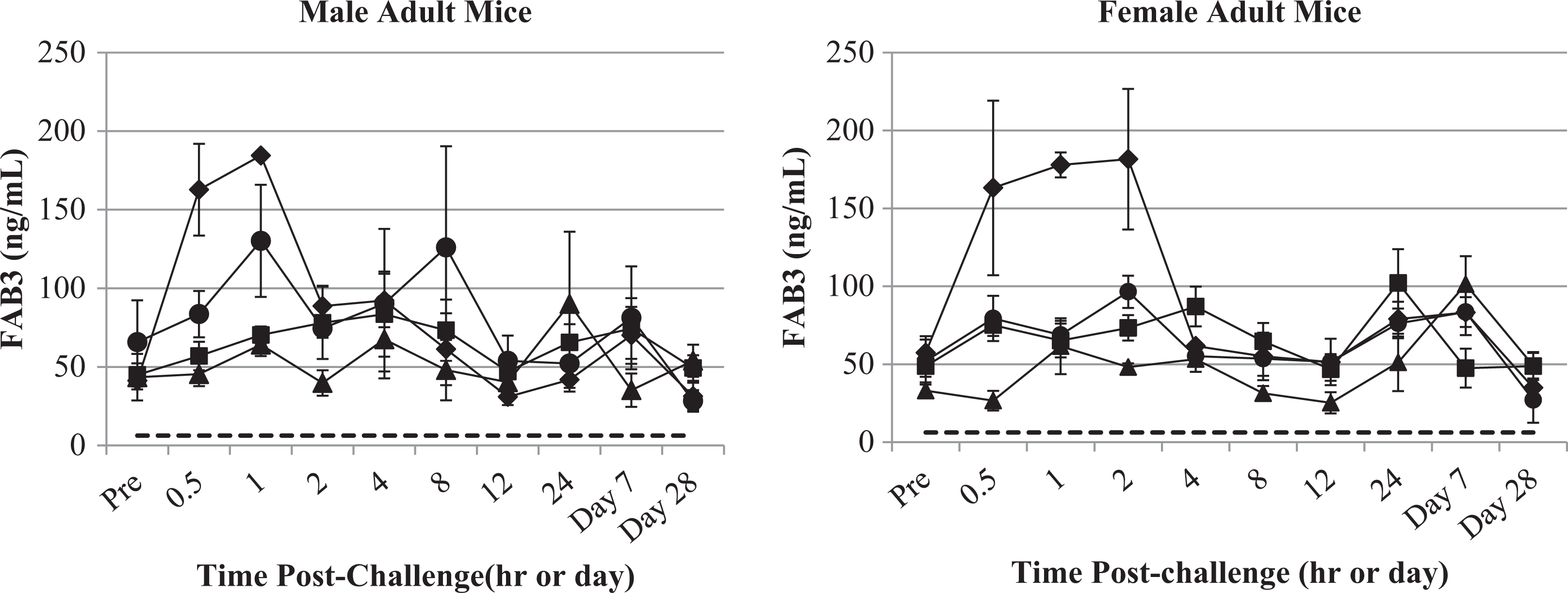
Serum FABP3 concentrations versus time following oral KCN challenge of adult mice. Serum FABP3 concentration after oral KCN dosing of male and female adult mice are shown for the vehicle (circle), 6 (triangle), 8 (square), and 11.5 (diamond) mg/kg KCN dose levels. Dotted line indicates lower limit of quantitation of the assay. Data points indicate mean values and error bars the SEM. FABP3 indicates fatty acid-binding protein 3; KCN, potassium cyanide; SEM, standard error of a mean.
Serum CK, potassium, histamine, and CRP measurements in a pilot study demonstrated no significant difference between KCN-challenged and control mice in the first 24 hours postchallenge and therefore were not analyzed in this study.
Gross Necropsy and Organ Weights
There were no gross necropsy or microscopic findings clearly attributed to oral administration of KCN in juvenile or adult CD-1 mice at time points ranging from 24 hours to 28 days post-KCN challenge with a few exceptions. The mean adult female heart weight and heart to body weight ratio, at 8 mg/kg KCN on day 8, were significantly lower (by 14%-15%) than the respective controls; however, no significant differences were observed at the higher dose (11.5 mg/kg) or on other days. The mean adult male heart organ to body weight ratio was significantly lower (∼22%) than controls at the 6 mg/kg KCN dose level on day 2 but not at higher dose levels. The mean adult male brain to body weight ratio was significantly lower on day 2 postchallenge at the 6 and 8 mg/kg KCN dose (∼6.5% and 15%, respectively) but not at the 11.5 mg/kg dose level or on other study days. All of these significantly lower organ weights and organ to body weight ratios in adult mice were sporadic and did not present a clear dose–response relationship to KCN. No statistically significant differences in organ to brain weight ratios were found in adult mice.
In juvenile female mice, there was a statistically significant increase in the mean liver weight (and liver to brain weight ratio) on day 28 postchallenge at the low dose (6 mg/kg). This was due to a marked increased liver weight in a single animal. This value was a single occurrence and likely not related to KCN exposure. The heart to brain weight ratios, at 28 days postchallenge, in juvenile male, but not female mice, were significantly higher in the 8 and 10.4 mg/kg KCN dose groups (∼11% and 14% increase, respectively) relative to controls. There was no histological correlation, and the increase was likely not related to KCN exposure. The mean kidney weight was significantly higher (∼7%) in female, but not male, mice receiving the 6 mg/kg KCN dose compared to control mice 28 days post-dosing. This value was a single occurrence and likely not related to KCN exposure. The mean kidney weights in all other KCN dose groups at 28 days postchallenge were similar to control kidney weights.
Histopathology
Due to the lack of microscopic findings at the high dose, only the high dose and vehicle control groups were evaluated microscopically. There were no gross lesions present in brain (olfactory bulb, cerebral cortex, midbrain, cerebellum, and brain stem), cauda epididymis, heart, kidneys, liver (median and left lateral lobes), or thyroid glands (with parathyroids, if present) of any adult or juvenile mice that could be attributed to KCN exposure.
Six adult male mice receiving the 11.5 mg/kg dose of KCN had microscopic lesions with uncertain relationship to KCN exposure. Three adult mice, 1 euthanized 24 hours postchallenge and 2 mice at 7 days postchallenge, had minimal to mild acute tubular necrosis in the kidneys, most notably near the corticomedullary junction. This lesion can be caused by prolonged hypoxia. One of these mice also had degeneration (vacuolization and Wallerian degeneration of the neuropil) in the brain stem. Although many CD-1 mice had spongiosis in the hippocampus as a background lesion unrelated to KCN exposure, this mouse was the only one found to have degenerative lesions in the brain stem.
Two male mice, euthanized at 24 hours or 7 days postchallenge (11.5 mg/kg KCN), had minimal necrosis of hepatocytes within the liver sections, and 1 male mouse, euthanized 7 days following dosing, had minimal necrosis of cardiac myofibers. Although all of these lesions can be attributed to prolonged hypoxia, the lack of concomitant lesions within female mice does not fully support a CN effect. All other lesions (liver bile duct hyperplasia, renal tubular cysts, minimal renal inflammation, and hepatic extramedullary hematopoiesis) are common incidental findings in mice and unrelated to KCN administration.
In juvenile mice, there were no gross lesions in any mice at any time point. Histopathologically, renal tubular cysts and hepatic extramedullary hematopoiesis were regarded as common incidental findings in mice and unrelated to KCN administration. Hypospermia in the epididymis was seen in many control and treated animals, an age-related immaturity finding not attributable to KCN.
In summary, there were no gross necropsy or microscopic findings clearly attributed to oral administration of 10.4 or 11.5 mg/kg KCN to juvenile or adult CD-1 mice, respectively, at time points ranging from 24 hours to 28 days after administration of oral KCN.
Discussion
A well-characterized model of oral KCN intoxication is required for preclinical Good Laboratory Practice (GLP) testing of new therapeutics for KCN intoxication under the FDA Animal Rule. 19 The objective of this study was to demonstrate a small animal model for use in testing MCM to CN intoxication. Selection of the appropriate animal model(s) is critical for licensure of MCM.
The Animal Rule requires that the natural history of the chemical agent intoxication in the animal model selected should be analogous to intoxication in humans. The pathophysiological mechanism of intoxication should also be similar. In addition, the physiological and biochemical responses to the chemical agent should be similar to that observed in humans. These end points, in addition to death, may be of utmost importance in establishing efficacy of a potential MCM. This study also addressed the question of whether the pathophysiology was dependent on the sex or age of the animal model. The dose–response relationship and toxicokinetic properties of the chemical agent and its metabolites may be useful in formulating the appropriate therapeutic dosage.
Comparison of the Natural History of CN in Mice to That of Humans
Selection of mice as a model of CN toxicity is acceptable based on the mechanism of CN toxicity, since inhibition of cytochrome C oxidase occurs in all mammalian species. As in humans, the shift from aerobic to anaerobic metabolism in mice following KCN intoxication led to blood lactate accumulation.
The mean oral LD50 of KCN was determined to be 11.5 ± 0.4 mg/kg (mean ± 95% confidence interval) in adult mice and 10.4 ± 0.3 mg/kg in juvenile mice (data from both sexes combined). A slight difference between sexes was observed in both age groups, with the LD50 in female mice being approximately 1 mg/kg less than in male mice of the same age group. The time to death in both adult and juvenile mice was generally dose related. Death occurred rapidly, usually with 30 minutes postchallenge; however at KCN doses that were not completely lethal, some deaths occurred up to 90 minutes postchallenge in adult mice and up to 60 minutes postchallenge in juvenile mice.
Reported lethal doses in human oral intoxication are similar to those observed in mice. Lethal oral doses of CN compounds have been reported to range from 50 to 200 mg CN (approximately 0.7-2.9 mg/kg body weight for CN; 1.8-7.3 mg/kg of the KCN salt), with death usually occurring within 1 hour. 24 Another report indicated that a dose of 600 mg KCN (8.6 mg/kg for 70 kg person) resulted in a deep coma 2 hours later, bradypnea, and severe lactic acidosis as seen in mice in the present study. 25 In a suicide attempt, a chemist was reported to have survived ingestion of an estimated 30 mg/kg dose of CN with no specific treatment. 26 Although there appears to be individual variability in the lethal dose of CN to humans, possibly due to estimations of dosage as well as differences in supportive and/or pharmaceutical treatment, these values are within an order of magnitude of the doses and time line that we found for KCN intoxication in mice. Therefore, the dose–response of CN in mice and humans is similar.
Clinical signs following challenge of mice with CN were similar to those observed in humans and other animals. Human ingestion of greater than 200 mg CN (approximately 3 mg/kg for a 70-kg person) is associated with altered consciousness, coma, convulsions, hypotension, cardiac arrhythmias, cardiovascular venous collapse, noncardiogenic pulmonary edema, and ultimately death. 27 Similar findings were observed in mice in this study.
At low doses of CN, humans show signs of weakness, headache, confusion, nausea, and vomiting. Mild to moderate clinical signs were observed in adult and juvenile mice dosed at 4 to 6 mg/kg KCN including decreased activity, lethargy, and restlessness up to 1 hour postchallenge with a few occurrences of respiratory difficulty and prostration. At doses at or above 8 mg/kg KCN, the incidence and severity of clinical signs in both adult and juvenile mice increased with dose and included lethargy, altered respiration, tremors, seizures, prostration, labored respiration, and death. These clinical signs are similar to those observed in intoxicated humans. 27 At ≥10 mg/kg KCN, there was a much higher incidence of seizures, generally occurring within 5 minutes of KCN challenge in juvenile mice. Clinical signs generally occur rapidly, within 5 to 15 minutes postchallenge, in both mice and humans at high doses. Most deaths in mice occurred within 45 minutes of challenge. In humans, survival times of several minutes to several hours have been reported following ingestion of lethal doses of CN. 27 Ansell and Lewis 28 reported the death of a woman at 30 minutes following ingestion of 2.5 g of NaCN. In mice, seizures were sometimes observed up to 90 minutes postchallenge and did not correlate with death. Mice surviving beyond 2 hours generally recovered and exhibited normal clinical signs by 8 hours postchallenge. Most human survivors of acute CN poisoning are free of sequelae, although some anoxic encephalopathy may occur. 29
McMahon and Birnbaum 18 reported that older C57BL/6N mice (10-12 months and 25-30 months of age) were more susceptible to oral KCN toxicity, at sublethal doses, than younger mice of 2 to 3 months of age. In the present study, the adult mice were approximately 2.5 months of age and thus similar to the younger mice in the publication by McMahon and Birnbaum. 18 These young adult mice were less sensitive to KCN when compared to juvenile mice (21-25 days of age) in the present study. Based on these 2 studies alone, it appears that very young juvenile mice, less than a month old, or aging mice, greater than 10 to 12 months old, are both more sensitive to KCN toxicity than young adults.
The results suggest that rapid intervention is required to prevent lethality in mice at KCN doses ≥8 mg/kg in both juvenile and adult mice and that female mice are slightly more sensitive than male mice. Rapid treatment is also indicated as observed for human CN poisoning. 30 The results also indicate a very rapid seizure response in juvenile mice possibly due to the less developed blood–brain barrier in juvenile animals. This may place the young at particular risk to CNS effects. Although the incidence of seizures in this study was low in adult mice, this could be partially due to masking by the lethargy, prostration, and respiratory difficulties in these animals. At doses up to the LD50, there were no gross necropsy or microscopic findings clearly attributable to oral administration of KCN in surviving juvenile or adult CD-1 mice at time points ranging from 24 hours to 28 days post-KCN exposure.
Markers of CN Poisoning and Triggers for Treatment
Pharmacokinetic analysis indicated a rapid absorption of CN with Tmax at less than or equal to 0.5 hours for the mid and high doses and within 4 hours for the low doses. A rapid decrease in plasma CN levels occurred in both adult and juvenile mice reaching endogenous levels within 24 hours postchallenge. Rapid clearance necessitates early analysis of blood CN in cases of human poisoning. 27 The clearance of the major metabolite of CN, thiocyanate, from plasma was slower than that of CN with peak concentrations of thiocyanate at 2 to 4 hours postchallenge and a terminal elimination half-life of approximately 6 to 9 hours for adult and juvenile mice at all KCN dose levels, except for male mice at the high dose having a half-life of approximately 12 hours. Mean thiocyanate Cmax and AUC0-24 hours values increased with dose and were not significantly different between sexes or between adult and juvenile mice at each dose level.
Decreased blood pH and increased blood lactate, consistent with anaerobic metabolism due to inhibition of oxidative phosphorylation, were nonspecific but rapid markers of CN intoxication. These biomarkers correlated well quantitatively and temporally with adverse clinical signs. The magnitude of the pH decrease (∼0.5 units) and lactate increase (∼3-fold) were similar in adult and juvenile mice. A slightly greater decrease in blood pH, or prolonged decrease in blood pH, observed in female mice is consistent with the slightly higher lethality of KCN observed in female mice. Human acute CN poisoning is also characterized by metabolic acidosis with markedly elevated plasma lactate and excessive venous oxygenation due to lack of oxygen uptake by tissues. 27 Although not specific for CN poisoning, blood pH and lactate levels, which can be measured almost real time using an I-Stat-type instrument, can be used in the experimental setting as a trigger for treatment. However, due to the rapidity of CN poisoning, testing therapeutics at multiple predetermined time points quickly after poisoning may be more feasible; using a separate cohort of mice to conduct a parallel blood pH/lactate study.
Of the cardiac biomarkers tested, only FABP3 gave a dose-related response (increase) to KCN, which was only significantly different from the vehicle control at the LD50 (11.5 mg/kg) in adult mice. However, no lesions were observed in heart tissue examined microscopically. Serum FABP3 in juvenile mice did not increase with KCN dosing at any dose level possibly due to the significantly higher background level of FABP3 in juvenile compared to adult mice. This marker has not been tested in clinical cases of CN poisoning.
Conclusion
In conclusion, this study presents a reproducible model of oral CN intoxication in the adult and juvenile (21-25 days old) male and female CD-1 mouse. The FDA Animal Rule 19 requires that there is a reasonably well-understood pathophysiological mechanism for the toxicity of the chemical substance, in this case CN, and its amelioration or prevention by the product. The mechanism of CN poisoning has been clearly characterized in multiple species, including mice, with a similar mechanism, that is, inhibition of cytochrome C oxidase, resulting in hypoxia leading to death at lethal doses. 31 -34 The mouse model demonstrates that the effects of KCN are rapid and, at doses up to a least the LD50, generally completely reversible in surviving mice. The toxicokinetics of CN in mice is consistent with the rapid clinical signs and recovery. Based on the rapid toxicity observed, treatment would have to be initiated quickly in mice, as is the case for humans, following exposures for optimal results.
The adult and juvenile mouse models can be used to screen or conduct efficacy studies of potential countermeasures of KCN intoxication. Although a 10% difference in LD50 was observed between male and female mice, in general, the clinical picture was similar. Therefore, for testing of countermeasures, unless there is a suspected sex difference in the mechanism of the therapeutic, the use of the more sensitive sex, female mice, is probably sufficient for efficacy testing. For countermeasures with expected pediatric use, testing of efficacy in juvenile mice is warranted due to the increased CN toxicity in young mice. A specific “trigger” for treatment commonly used to correlate the timing of treatment between animals models and actual human intoxications is difficult to determine with CN due to rapid intoxication and death. In actual intoxication, patients would also most likely receive supportive treatment prior to determining the cause of intoxication and treatment with MCM. Rapid analysis of blood pH and/or lactate using a point-of-care device, such as the I-Stat, could be used as a trigger for treatment in animal efficacy studies since these are early clinical changes. However, it should be kept in mind that these measurements are not specific to CN, and additional information, such as patient history, excessive venous oxygen, and blood CN or thiocyanate levels, would be required to confirm human poisoning. Seizures, lethargy, and/or breathing difficulties could also be used as triggers to determine whether a countermeasure is effective in later stages of intoxication. Clinical signs, described in this article, are consistent with previous studies in animals and humans 27,35 and, combined with mortality, would serve as critical end points for evaluating countermeasure efficacy.
Due to rapid toxicity at high doses of CN, it may be more practical to initially evaluate the efficacy of potential countermeasures in this mouse model at selected time points early postchallenge (ie, 5, 10 and 15 minutes). The use of a small animal model in an acute toxicity screen provides a relatively inexpensive screen that can be used to test the potential of new countermeasures. Promising therapeutic candidates can then be tested more fully in the mouse or other animal models to address the requirements of the FDA Animal Rule. 19 It is expected that the FDA Animal Rule will require 2 animal models for testing the efficacy of a CN MCM, including a large animal model, such as swine or nonhuman primate; however, the mouse model characterized in this study will fulfill the requirement for one of these 2 animal models.
Footnotes
Author Contributions
Patrick J. Sabourin contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. Christina L. Kobs contributed to design, contributed to acquisition and analysis, and drafted the manuscript. Seth T. Gibbs contributed to design, contributed to analysis and interpretation, and drafted the manuscript. Peter Hong contributed to design, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. Claire M. Matthews contributed to design, contributed to analysis and interpretation, and drafted the manuscript. Kristen M. Patton contributed to design, contributed to acquisition and interpretation, drafted the manuscript, and critically revised the manuscript. Carol L. Sabourin contributed to conception and design, contributed to interpretation, and critically revised the manuscript. Edgar J. Wakayama contributed to conception and design, contributed to interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded in whole or in part with Federal funds from Biomedical Advanced Research and Development Authority, Department of Health and Human Services, under HHSO1002011000005I/Task Order HHSO10033006T.
