Abstract
Inorganic arsenic (iAs) is at the top of toxic metalloids. Inorganic arsenic-contaminated water consumption is one of the greatest environmental health threats worldwide. Human iAs exposure has been associated with cancers of several organs, neurological disorders, and reproductive problems. Nevertheless, there are no reports describing how iAs affects the anterior pituitary gland. The aim of this study was to investigate the mechanisms involved in iAs-mediated anterior pituitary toxicity both in vivo and in vitro. We showed that iAs administration (from 5 to 100 ppm) to male rats through drinking water increased messenger RNA expression of several oxidative stress-responsive genes in the anterior pituitary gland. Serum prolactin levels diminished, whereas luteinizing hormone (LH) levels were only affected at the higher dose tested. In anterior pituitary cells in culture, 25 µmol/L iAs significantly decreased prolactin release in a time-dependent fashion, whereas LH levels remained unaltered. Cell viability was significantly reduced mainly by apoptosis evidenced by morphological and phosphatidylserine externalization studies. This process is characterized by early depolarization of mitochondrial membrane potential and increased levels of reactive oxygen species. Expression of some key oxidative stress-responsive genes, such as heme oxygenase-1 and metallothionein-1, was also stimulated by iAs exposure. The antioxidant N-acetyl cysteine prevented iAs-induced effects on the expression of oxidative stress markers, prolactin release, and apoptosis. In summary, the present work demonstrates for the first time that iAs reduces prolactin release both in vivo and in vitro and induces apoptosis in anterior pituitary cells, possibly resulting from imbalanced cellular redox status.
Introduction
Arsenic is a naturally occurring metalloid ubiquitously present in the environment with potent toxic and genotoxic effects. 1 It occurs in both organic and inorganic forms; however, inorganic species are more hazardous to human health. Exposure to inorganic arsenic (iAs), mainly through contaminated water consumption, is one of the greatest environmental health threats worldwide, affecting more than 200 million people. 2,3 Inorganic arsenic contamination represents a major sanitary problem at both high and low concentrations. Human iAs exposure has been associated with cancers of several organs, 4 diabetes, 5 vascular diseases, 6 and neurological and developmental problems 7 ; however, iAs effects on the reproductive system have been poorly studied. In some reports, experimental arsenite intoxication is associated with gonad damage. In males, spermatotoxicity, testicular androgenesis inhibition, and reduced weight of the testes and accessory sex organs have been reported. 8 In female iAs-treated rats, reduced ovarian and uterine somatic indices and lower ovarian steroidogenic enzyme activity have been shown. 9
Anterior pituitary hormones play a key role in reproductive physiology. A relationship between iAs exposure and hormone levels was demonstrated through multiple linear regression models in a cohort study in which serum prolactin (PRL) levels in men were inversely associated with iAs concentrations but directly associated with thyrotropin levels. 10 In experimental animals, iAs intoxication reduced serum testosterone and gonadotropin levels in male rats, 8 whereas it inhibited steroid synthesis in female rats. 9 Thus, iAs may affect gonadotropin levels through feedback mechanisms on hypothalamus or by directly affecting anterior pituitary gland.
A previous study indicates that iAs accumulates in the pituitary, 11 however, to date, there have been no reports on iAs-mediated effects on the anterior pituitary gland.
Arsenic compound uptake, accumulation, and toxicity are dependent upon cell type and iAs species. Metalloid effects also depend strongly on valence status, methylation degree, and charge at physiological pH. 12 In many cell types, iAs undergoes oxidative methylation following uptake, these methylated metabolites being more toxic than the original compounds. 13
Reactive oxygen species (ROS) production and oxidative stress are reportedly the main mediators of iAs cytotoxicity. 14,15 It has been shown that several enzymatic elements, such as superoxide dismutase, catalase, glutathione peroxidase, thioredoxin-1 (Trx-1), and nonenzymatic elements, as glutathione, as well as oxidative stress-responsive genes, such as heme oxygenase-1 (HMOX1) and metallothionein-1 (MT-1), are altered by iAs exposure. 16 –18
Mitochondria play a crucial role in iAs-induced effects. 19 This metalloid causes morphologic changes and a rapid decline in mitochondrial membrane potential (MMP), thereby leading to apoptosis. 20
Bearing in mind the diverse evidence suggesting that iAs adversely affects endocrine glands and considering that this metalloid accumulates in the pituitary, the present study was designed to investigate whether iAs could directly affect anterior pituitary cell viability and hormone release. Here, we show that iAs exerts cytotoxic actions both in vivo and in vitro in anterior pituitary gland by decreasing PRL release and cell viability. Furthermore, these effects are mainly due to generation of oxidative stress since they can be mostly reversed by antioxidant treatment.
Materials and Methods
Ethics Statement
All experimental procedures were approved by the Institutional Animal Care and Use Committee (CICUAL) of the School of Medicine, University of Buenos Aires (Res. N 2742/2013), and were carried out in compliance with guidelines of the NIH Guide for the Care and Use of Laboratory Animals.
Drugs and Reagents
Inorganic arsenic as sodium (meta) arsenite (NaAsO2, ≥99.0%) was obtained from Sigma-Aldrich (St Louis, Missouri). Cell culture media and reagents were purchased from Gibco (Rockville, Maryland), except fetal bovine serum (FBS), obtained from GenSA (Buenos Aires, Argentina). Unless otherwise indicated, all other reagents and antibodies were purchased from Sigma-Aldrich.
Animals
Adult male Wistar rats (200-250 g) from Bioterio Central, Facultad de Farmacia y Bioquímica, Universidad de Buenos Aires were chosen to avoid hormonal fluctuation. Animals were housed with a 12-hour light–dark cycle and controlled temperature (20°C-22°C) and were fed standard laboratory chow and water ad libitum.
Dose Selection
Inorganic arsenic doses were based on bibliography. 21 –26 Taking into account that rats are less sensitive to iAs cytotoxicity than most of mammalian species due to substantial differences in iAs metabolism, 27,28 iAs doses ranged from 5 to 100 ppm excluding concentrations below 5 ppm that were shown to exert xenoestrogenic effects in anterior pituitary gland. 29
In Vivo Treatment
Rats were randomly divided into 4 groups (N = 6 per cage). Treated groups received 5, 25, or 100 ppm NaAsO2 (equivalent to 2.86, 14.32, and 57.3 ppm arsenic, respectively) in drinking water for 30 days corresponding to an average intake of 0.5, 2.5, or 10 mg NaAsO2/kg/d, in that order. In our study, the highest concentration used (100 ppm) is about 25% of oral LD50 (41 mg/kg) of NaAsO2 in rats. 30 Control group received filtered tap water. Animal weight was registered once a week and water consumption measured daily. No significant differences were observed among groups for these 2 parameters. Additionally, none of the concentrations tested caused any apparent sign of pain or distress. Following treatment, animals were killed and trunk blood was collected for PRL and luteinizing hormone (LH) determinations. Basal hypothalamus and anterior pituitary gland, without the posterior lobe, were carefully and quickly removed and stored in liquid nitrogen until further processing.
Cell Culture
Cells were obtained from pooled anterior pituitary glands by enzymatic (trypsin/DNase) and mechanical dispersion (extrusion through a Pasteur pipette) as described previously. 31 In all cases, cells were cultured for 2 days at 37°C and 5% CO2 in Dulbecco modified Eagle medium (DMEM) supplemented with 5% FBS, 10 μL/mL MEM amino acids, 2 mmol/L glutamine, 5.6 μg/mL amphotericin B, 25 μg/mL gentamicin, 50 U/mL penicillin, and 50 μg/mL streptomycin (DMEM-S-5% FBS). For spectrophotometric experiments, cells were seeded onto 96-well tissue culture plates (0.1 × 106 cells/well). For fluorescence microscopy studies, cells were seeded on glass coverslips in 24-well tissue culture plates (0.1 × 106 cells/well). For flow cytometry experiments, cells were seeded onto 24-well tissue culture plates (0.5 × 106 cells/well).
Cell Treatment
After the stabilization period (48 hours), medium was changed, and cells were incubated for different time periods in DMEM-S-5% FBS with or without iAs. Fresh sodium arsenite solutions were prepared before each experiment in distilled water.
Cell Activity Assay
The methylthiazolyldiphenyl-tetrazolium bromide (MTT) assay was used to determine cell activity as described previously. 32 In brief, cells were washed twice with Krebs-Ringer bicarbonate buffer followed by incubation with 110 μL MTT solution (0.5 mg/mL) for 4 hours at 37°C, 5% CO2. After incubation, 90 μL was removed, 100 μL 0.01 N HCl in isopropanol added to each well, and then the plate was gently shaken for 3 minutes. Optical density was determined at 600 nm in an enzyme-linked immunosorbent assay plate reader. Cell activity was considered an indicator of cell viability. 33
Nuclear Morphology Analysis
Cells were fixed in 4% formaldehyde for 30 minutes at 4°C and mounted in antifade glycerol solution containing 1 μg/mL 4,6-diamidino-2-phenylindole (DAPI), 23.3 mg/mL 1,4 diazabicyclooctane (DABCO), and 20 mmol/L Tris-HCl, pH 8. Nuclear morphology was observed and quantified with an Olympus-BX50 microscope (Japan). At least 500 nuclei from random fields were obtained from 3 independent experiments and expressed as the number of apoptotic nuclei/total number of nuclei × 100.
Apoptosis Determination by Annexin V–Fluorescein Isothiocyanate and Propidium Iodide Staining
The annexin V–fluorescein isothiocyanate (FITC) apoptosis detection kit (Sigma-Aldrich) was used for apoptosis determination. Briefly, after treatment, cells were rinsed with calcium- and magnesium-free Krebs buffer, trypsinized in the same buffer, and centrifuged at 1,000g for 10 minutes. Cell pellets (0.5 × 106 cells) were resuspended in 250 µL annexin binding buffer followed by addition of 5 µL FITC-conjugated annexin V (exλ: 488 nm, emλ: 535 nm, FL1) and 10 µL propidium iodide (PI; exλ: 488 nm, emλ: 585 nm, FL2) solution (100 µg/mL). Cells were incubated for 15 minutes in darkness at room temperature and were analyzed by flow cytometry with a Becton Dickinson FACScalibur flow cytometer (San Jose, California). At least 104 events were measured for each treatment. Further flow cytometric data analysis was performed with WinMDI 2.8 software (http://facs.scripps.edu).
Immunocytochemical Studies
Cells were fixed in 4% formaldehyde for 30 minutes at 4°C, permeabilized with 0.2% Triton X-100 in phosphate-buffered saline for 10 minutes at 4°C, and incubated in blocking solution (5% normal serum, 0.2% Triton X-100) for 30 minutes at room temperature. The cells were incubated with guinea pig primary antibodies (PRL, LH, growth hormone, adrenocorticotrophin (ACTH), tirotrophin (TSH), 1:1,000; S100, 1:50) for 1 hour at room temperature, and after 3 washes, secondary antibody conjugated to FITC (1:200) was added. Cells were mounted in antifade solution containing DAPI and DABCO, and nuclear morphology was observed. Data of at least 400 nuclei obtained from random fields were expressed as number of immunopositive cells with apoptotic nuclei/total number of cells with apoptotic nuclei.
Mitochondrial Membrane Potential Measurement
Mitochondrial membrane potential was measured with 3,3′-dihexyloxacarbocyanine iodide (DiOC6), a cationic, MMP-sensitive fluorescent probe that intercalates into the inner mitochondrial membrane in an electrochemical potential-dependent manner. 34 Decreased MMP is associated with reduced DiOC6 uptake. Cells were loaded with 30 nmol/L DiOC6 20 minutes before the end of treatment. Then, cells were trypsinized in calcium- and magnesium-free Krebs buffer (0.5 × 106 cells/mL) and analyzed with a Becton Dickinson FACScalibur flow cytometer (exλ: 488 nm, emλ: 535 nm, FL1). Immediately prior to measurement, 10 μg/mL PI (exλ: 488 nm, emλ: 585 nm, FL2) was added to each FACS tube to detect cells with plasma membrane disruption; 104 cells were measured per treatment. In the control group, plasma membrane integrity after trypsinization was always over 95%. Nonviable PI-positive cells were not used in the analysis. Data were analyzed using WinMDI 2.8 software.
Reactive Oxygen Species Determination
Intracellular ROS was measured with the readily cell membrane-diffusible lipophilic probe dihydrorhodamine 123 (DHR123). 35,36 Once inside the cell, DHR123 is oxidized to the mitochondrial-specific fluorescent dye rhodamine. Dihydrorhodamine 123 has been shown to be specifically oxidized by peroxynitrite and hydrogen peroxide. 37 Cells were loaded with 30 µmol/L DHR123 20 minutes before the end of treatment. Cells were then trypsinized in calcium- and magnesium-free Krebs buffer (0.5 × 106 cells/mL) and analyzed by flow cytometry (exλ: 525 nm, emλ: 535 nm, FL1). Immediately prior to measurement, 10 μg /mL PI (exλ: 488 nm, emλ: 585 nm, FL2) was added to each tube for disrupted cellular plasma membrane detection; 104 cells were measured per treatment. In control cells, plasma membrane integrity after trypsinization was always above 95%. Nonviable PI-positive cells were not used in the analysis. Data were analyzed using WinMDI 2.8 software.
RNA Isolation
Frozen tissues were homogenized with 1 mL TRIzol reagent (Invitrogen, Carlsbad, California). For in vitro experiments, 177 μL TRIzol was added to each well. After isolation, total RNA from whole tissues or 2 × 106 cells per treatment was spectrophotometrically quantified at 260 nm, and the 260/280 ratio was determined. RNA integrity was assessed by formaldehyde/formamide gel electrophoresis.
Reverse Transcription and Polymerase Chain Reactions
First-strand complementary DNA was synthesized with Moloney murine leukemia virus (M-MLV) reverse transcriptase in reverse transcription buffer containing 5.5 mmol/L MgCl2, 0.5 mmol/L dNTP, 2.5 mmol/L random hexamers, and 3.125 U/mL M-MLV reverse transcriptase. Reactions were run in a final volume of 12 µL containing 5 μg RNA. The reverse transcription reaction was done at 37°C for 50 minutes, and reverse transcriptase was inactivated by heating samples at 70°C for 15 minutes before polymerase chain reactions (PCRs). To check for genomic contamination, an assay was performed in a reaction solution without reverse transcriptase. Specific primers for HMOX1, MT-1, Trx-1, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were designed with OligoPerfect designer software (Invitrogen) from published sequences 38 and are detailed in Table 1. Glyceraldehyde-3-phosphate dehydrogenase was used as endogenous control. Samples were amplified by PCR on a thermocycler (Mastercycler; Eppendorf, Hamburg, Germany). The reaction mixture contained GoTaq PCR buffer, 1.5 mmol/L MgCl2, 200 mmol/L dNTPs, 0.625 U GoTaq polymerase, and 300 nmol/L primers. Reverse transcription-PCR methods were utilized to determine relative messenger RNA (mRNA) expression changes. Reactions were subjected to 16 to 40 PCR amplification cycles to ascertain the optimum cycle number within the linear range for PCR amplification. Each PCR reaction was hot started for 2 minutes at 94°C and finished with a 10-minute elongation at 72°C. Polymerase chain reaction amplifications of MT-1, HMOX1, and GAPDH were performed as previously described. 39 The optimum cycle number was 40 for HMOX1 and MT-1 and 30 for GAPDH. Thioredoxin-1 optimum amplification was determined after 40 cycles of 94°C melting for 45 seconds, 58°C annealing for 60 seconds, and 72°C extension for 45 seconds. Amplified products were analyzed by 2% agarose–ethidium bromide gel electrophoresis.
Primers Used for PCR Assays.

Abbreviations: bp, base pair; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; HMOX1, heme oxygenase-1; MT-1, metallothionein-1; PCR, polymerase chain reaction; Trx-1, thioredoxin-1.
Polymerase Chain Reaction Data Analysis
Polymerase chain reaction product signal intensity was determined by digital image analysis using Gel Pro Analyzer software for Windows (Media Cybernetics, LP, Silver Spring, Maryland). For statistical comparison of results from different experiments, HMOX1, MT-1, and Trx-1 intensity levels were normalized to the GAPDH band intensity in each lane.
Hormone Determination
Prolactin and LH were measured by a double antibody radioimmunoassay (RIA) 40 using reagents provided by Dr A. F. Parlow (National Hormone and Pituitary Program, Torrance, California). Recombinant PRL (NIDDK-rPRL-RP-3) and recombinant LH (LHRP-3) were used as reference preparations. NIDDK-anti-rPRL-S-9 and anti-rLH-11 were used as antisera. Assay sensitivity was 0.1 ng/mL, and intra- and interassay coefficients of variation were under 10%. All samples were measured in the same assay to prevent interassay variation. Prolactin and LH levels in control media at 24 hours were 1,430.5 ± 168.1 ng/mL and 6.31 ± 0.72 ng/mL, respectively.
Statistical Analysis
Results were expressed as mean ± standard error and evaluated by 1-way analysis of variance followed by Tukey-Kramer, Dunnett, or Student t test depending on experimental design. Differences among groups were considered significant if P < 0.05. Results were confirmed by at least 3 independent experiments.
Results
In Vivo Arsenic Exposure Increased Oxidative Stress-Responsive Genes Expression and Decreased PRL Serum Levels
Considering that iAs accumulates in nervous system and anterior pituitary gland 11,22 and exerts adverse effects on several endocrine organs, 9,41,42 we focused our investigation on iAs actions in the anterior pituitary gland. First, we addressed whether this metalloid could generate oxidative stress in basal hypothalamus and anterior pituitary gland and alter pituitary hormone release in vivo. Based on previous toxicological studies carried out in rats, 43 –45 we chose to investigate iAs effects in concentrations ranging from 5 to 100 ppm iAs, to be administrated in drinking water for 30 days. Messenger RNA expression of several well-known oxidative stress markers, such as HMOX1, MT-1, and Trx-1, was evaluated by PCR.
Inorganic arsenic administration did not modify hypothalamic HMOX1 or Trx-1 mRNA expression (data not shown). However, this treatment significantly increased mRNA levels of MT-1 and Trx-1 in anterior pituitary gland at all concentrations tested. Conversely, we noticed that HMOX1 expression was only augmented from 25 ppm iAs (Table 2).
In Vivo iAs Administration Increases Oxidative Stress-Responsive Genes mRNA Levels and Affects Serum PRL Release.a

Abbreviations: ANOVA, analysis of variance; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; iAs, inorganic arsenic; HMOX1, heme oxygenase-1; LH, luteinizing hormone; mRNA, messenger RNA; MT-1, metallothionein-1; PCR, polymerase chain reaction; PRL, prolactin; RIA, radioimmunoassay; SE, standard error; Trx-1, thioredoxin-1.
aRats were exposed to 5, 25, or 100 ppm iAs in drinking water during 30 days. Control group received filtered tap water. Messenger RNA expression of HMOX1, MT-1, and Trx-1 was evaluated by PCR. Densitometric values were normalized to GAPDH expression. Serum PRL and LH levels were determined by RIA. Results represent the mean ± SE and are expressed as percentage of control. ANOVA followed by Tukey-Kramer test.
b P < 0.05 versus control.
Hormone determination revealed that iAs treatment decreased PRL serum levels in a dose-dependent manner, whereas LH levels were affected only at the highest concentration used (Table 2).
In sum, these results suggest that the in vivo exposure to iAs causes oxidative stress in anterior pituitary gland but not in hypothalamus. Inorganic arsenic decreases both PRL and LH release. To investigate whether these effects are due to a direct action of the metalloid on the gland, the next experiments were performed in anterior pituitary cell cultures.
Arsenic Decreased Anterior Pituitary Cell Viability and PRL Release
To study whether iAs could affect anterior pituitary cell viability, cells were incubated with 1, 10, 25, 50, or 100 µmol/L iAs for 24 hours, and cell activity was determined by MTT assay as cell viability index. Cell viability decreased in a dose-dependent manner (Figure 1A). The estimated IC50 for the in vitro analysis after iAs incubation was 39.1 µmol/L. Since 25 μmol/L iAs significantly reduced cell activity by about 40% versus control, this concentration was henceforth chosen to address iAs cytotoxic actions in anterior pituitary cells.
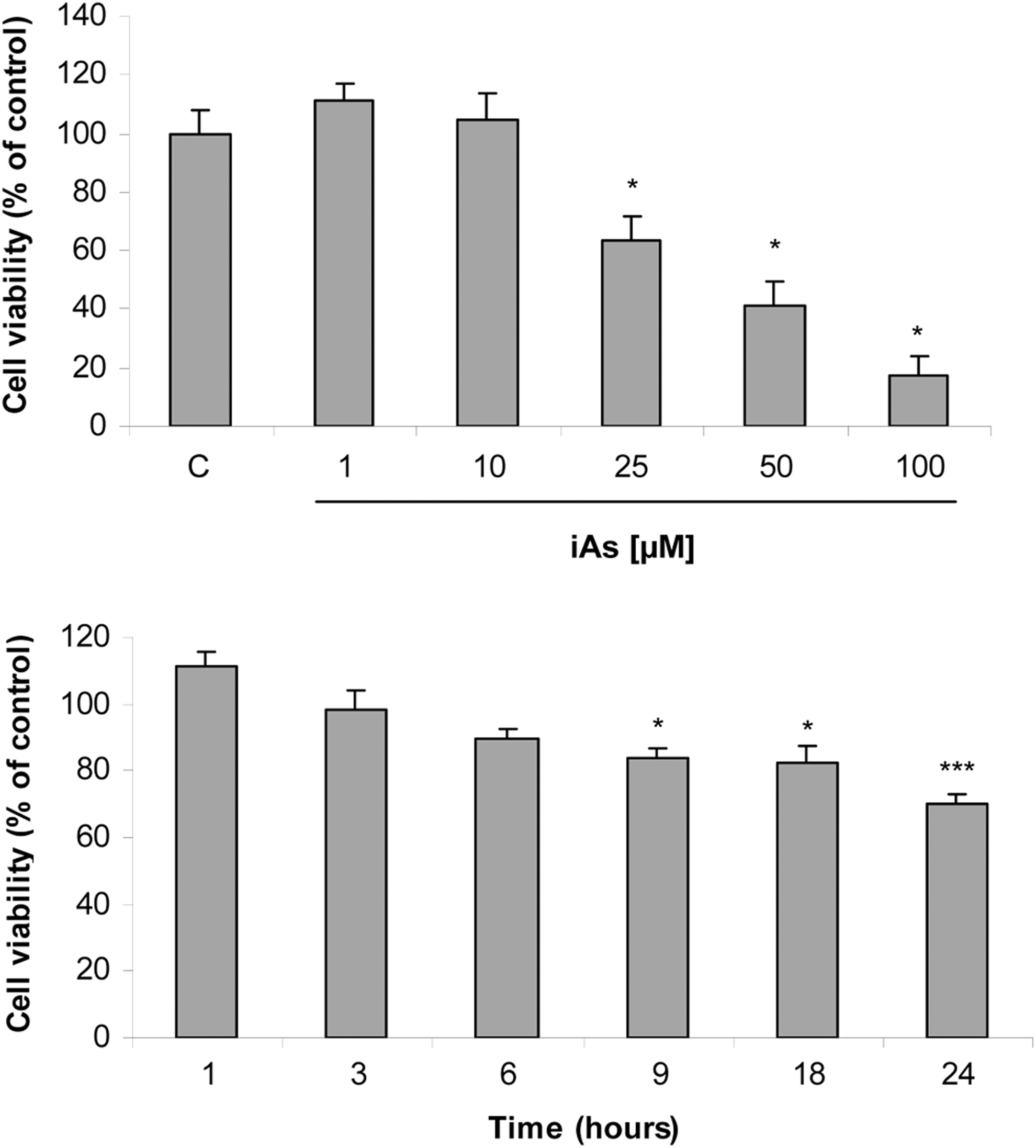
Arsenic reduces anterior pituitary cell viability. Anterior pituitary cells were incubated with (A) increasing concentrations of inorganic arsenic (iAs; NaAsO2) for 24 hours or (B) with 25 μmol/L iAs for different times. Cell activity as an index of cell viability was measured by methylthiazolyldiphenyl-tetrazolium bromide (MTT) assay. Bars represent the mean ± standard error (SE) and are expressed as a percentage of the control, n = 3. Analysis of variance (ANOVA) followed by Tukey-Kramer test, *P < 0.05; ***P < 0.001 versus respective control.
To investigate the effect of 25 μmol/L iAs over time, anterior pituitary cells were incubated with the metalloid for 1 to 24 hours. Nine hours of iAs exposure significantly decreased cell viability, which was markedly reduced after 24 hours (Figure 1B). However, 6-hour treatment was enough to significantly affect PRL release. Twenty-five μmol/L iAs did not modify LH levels at any time point tested (Figure 2). Together, these results indicate that iAs directly affects cell viability and PRL release independently of extrapituitary factors.
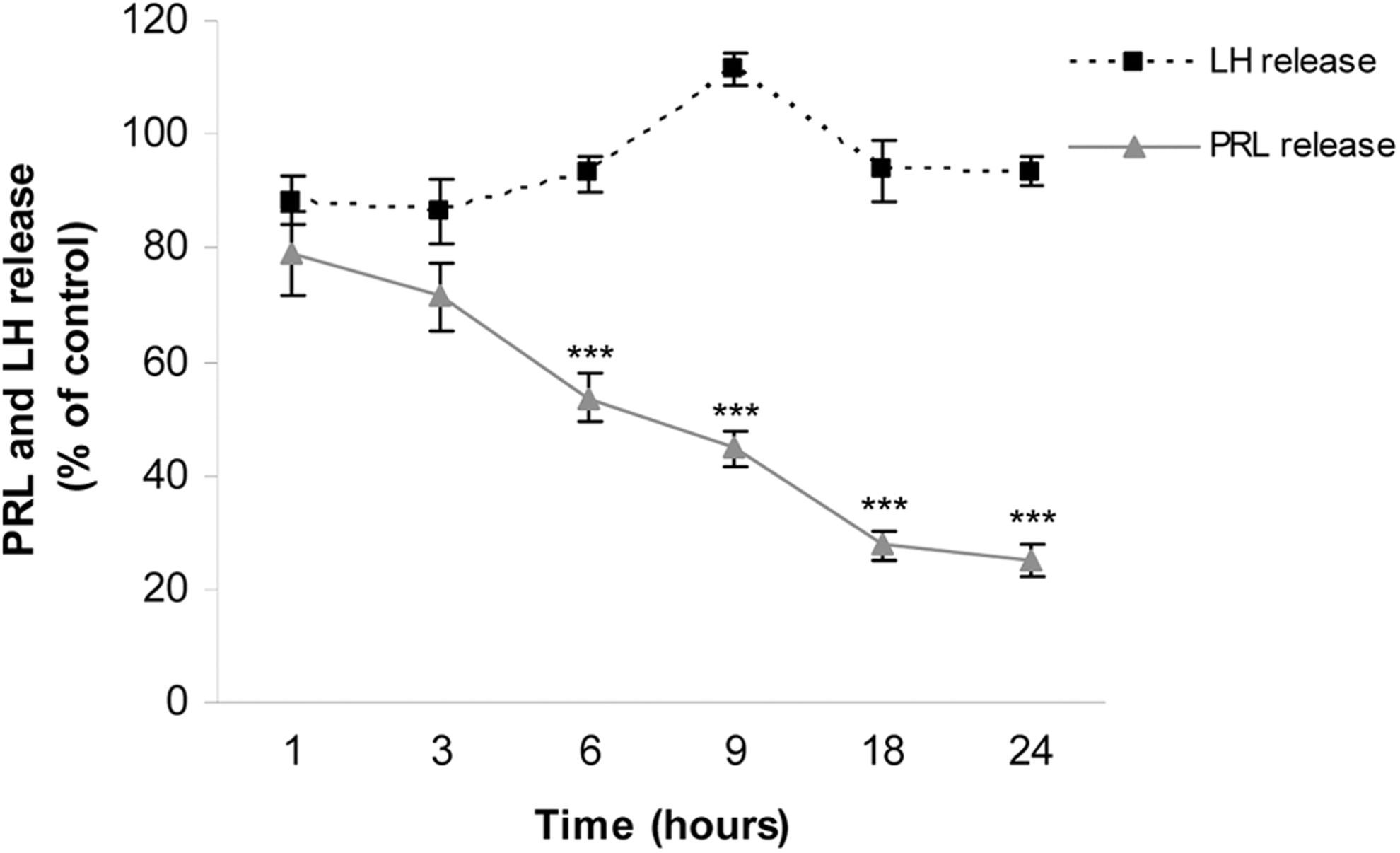
Effect of arsenic on hormone release over time. Anterior pituitary cells were incubated with 25 μmol/L inorganic arsenic (iAs; NaAsO2) for different times. Prolactin (PRL) and luteinizing hormone (LH) release into the medium was determined by radioimmunoassay (RIA). Points represent the mean ± standard error (SE) and are expressed as a percentage of the respective control, n = 3. Analysis of variance (ANOVA) followed by Tukey-Kramer test, ***P < 0.001 versus respective control.
Arsenic Induced Anterior Pituitary Cell Apoptosis
To investigate whether the decreased cell viability observed in iAs-exposed cells was due to apoptosis, we examined nuclear morphology. Anterior pituitary cells were incubated with the metalloid for different times and then fixed and stained with DAPI. Apoptotic nuclear morphology became evident after 9 hours of iAs exposure (Figure 3A). To confirm these results, apoptosis was determined by annexin V-FITC and PI staining by flow cytometry. Cells exposed to iAs displayed high annexin V-FITC fluorescence caused by externalized phosphatidylserine and low PI fluorescence reflecting intact plasma membrane, although a small percentage of necrotic cells was also observed (Figure 3B).
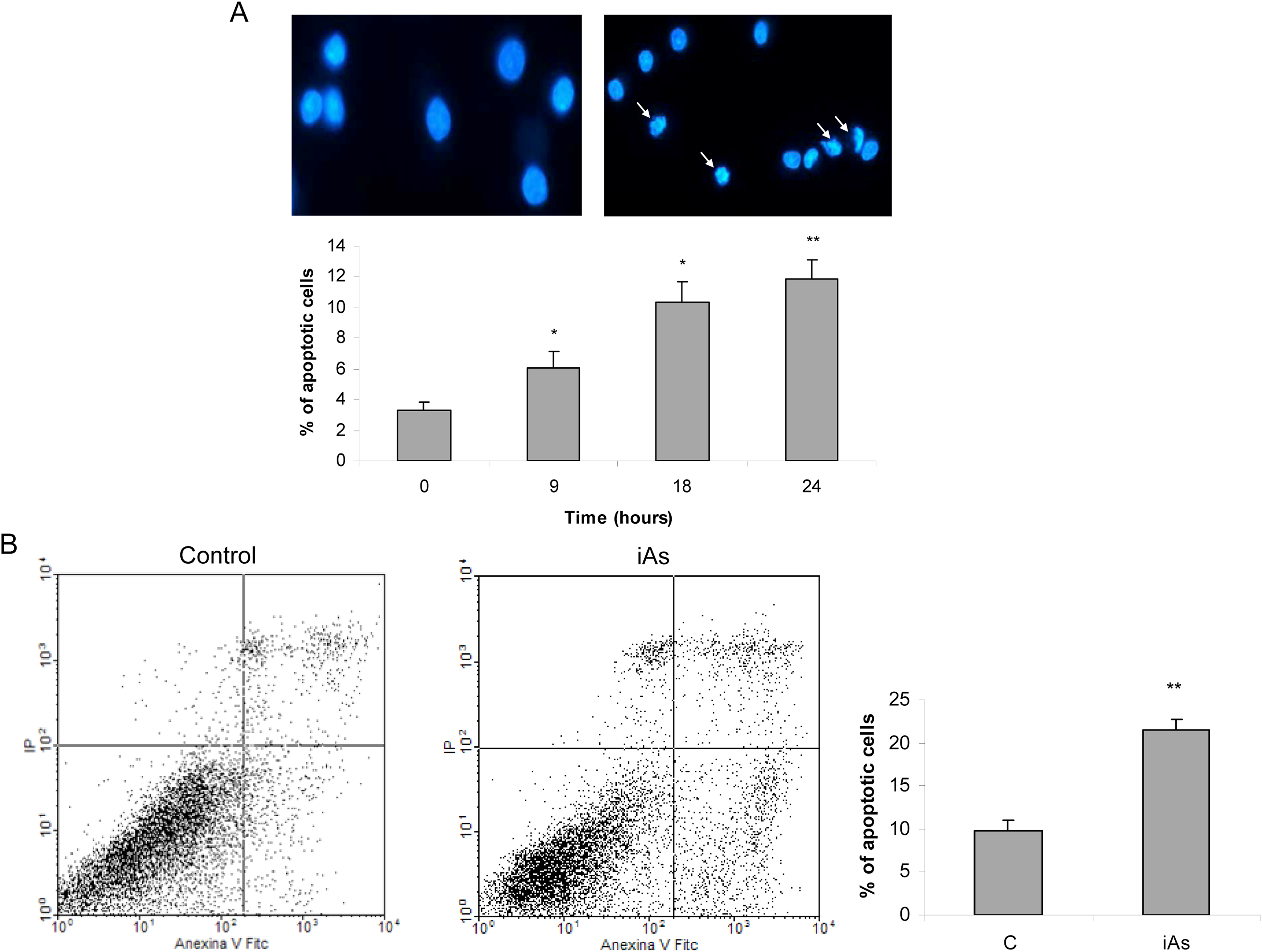
Arsenic induces anterior pituitary cell apoptosis. A, Anterior pituitary cells were incubated with 25 μmol/L inorganic arsenic (iAs; NaAsO2) for different times, and nuclear morphology was studied by 4,6-diamidino-2-phenylindole (DAPI) staining and was expressed as the number of apoptotic nuclei/total number of nuclei × 100. Pictures are representative of 3 independent experiments. Bars represent the mean ± standard error (SE), n = 3. Analysis of variance (ANOVA) followed by Tukey-Kramer test, *P < 0.05; **P < 0.01 versus control. B, Anterior pituitary cells were incubated with iAs for 18 hours and apoptosis was assessed by annexin V-FITC and propidium iodide (PI) staining and flow cytometry. Cells that were annexin V positive, PI negative were considered early apoptotic cells. Dot plots are representative of 3 independent experiments. Student t test, *P < 0.05 versus control.
Arsenic Differentially Affects Anterior Pituitary Cells Subpopulations
Anterior pituitary gland is a heterogeneous tissue consisting of several secretory and nonsecretory cell types. In order to determine whether iAs differentially affects anterior pituitary cells subpopulations, specific antibodies raised against different pituitary hormones or S100 were used to identify secretory and folliculo-stellate cells, respectively. Nuclear morphology was evaluated by DAPI staining. About 40% of total apoptotic cells resulted to be lactotrophs, and in a lesser extent somatotrophs and gonadotrophs (20%; Figure 4). Apoptosis was not detected in the rest of secretory and S100 positive cells.
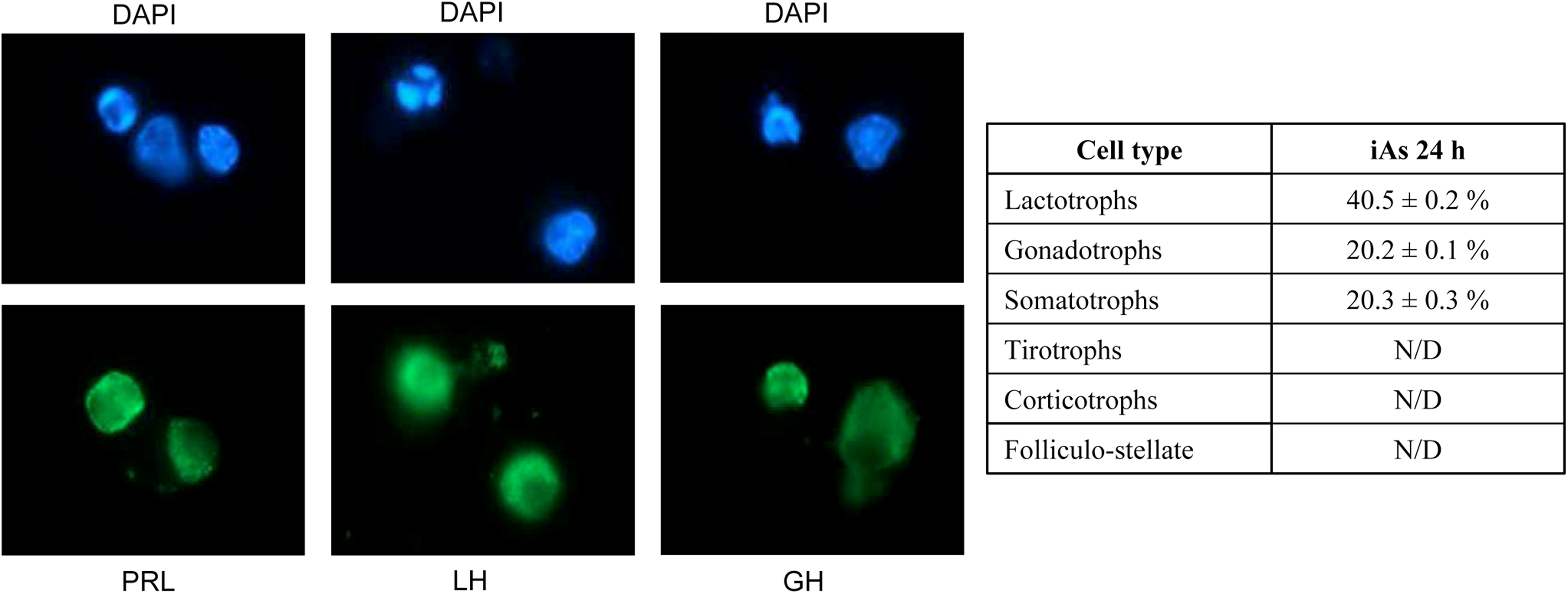
Inorganic arsenic (iAs) differentially affects anterior pituitary cells subpopulations. Anterior pituitary cells were treated with 25 μmol/L iAs (NaAsO2) for 24 hours, and then cells were immunostained with specific antibodies against different anterior pituitary hormones or S100 protein (folliculostellate cells). Nuclear morphology was studied by 4,6-diamidino-2-phenylindole (DAPI) staining. Data represent mean ± standard error (SE) and were calculated as number of immunopositive cell with apoptotic nuclei/total number of cell with apoptotic nuclei × 100, n = 3. Pictures are representative of 3 independent experiments (40×). PRL indicates prolactin; LH, luteinizing hormone; GH, growth hormone; N/D, not determined.
Arsenic Caused Early MMP Depolarization
Mitochondria are major targets for arsenic intoxication. To determine whether iAs-mediated apoptosis involves MMP changes, cells were exposed to the metalloid for different times and MMP was measured using a DiOC6 probe followed by flow cytometry. Mitochondrial membrane potential decreased after 6-hour iAs treatment and was markedly reduced after 24 hours of exposure, at which time most of the cells were already undergoing apoptosis (Figure 5).
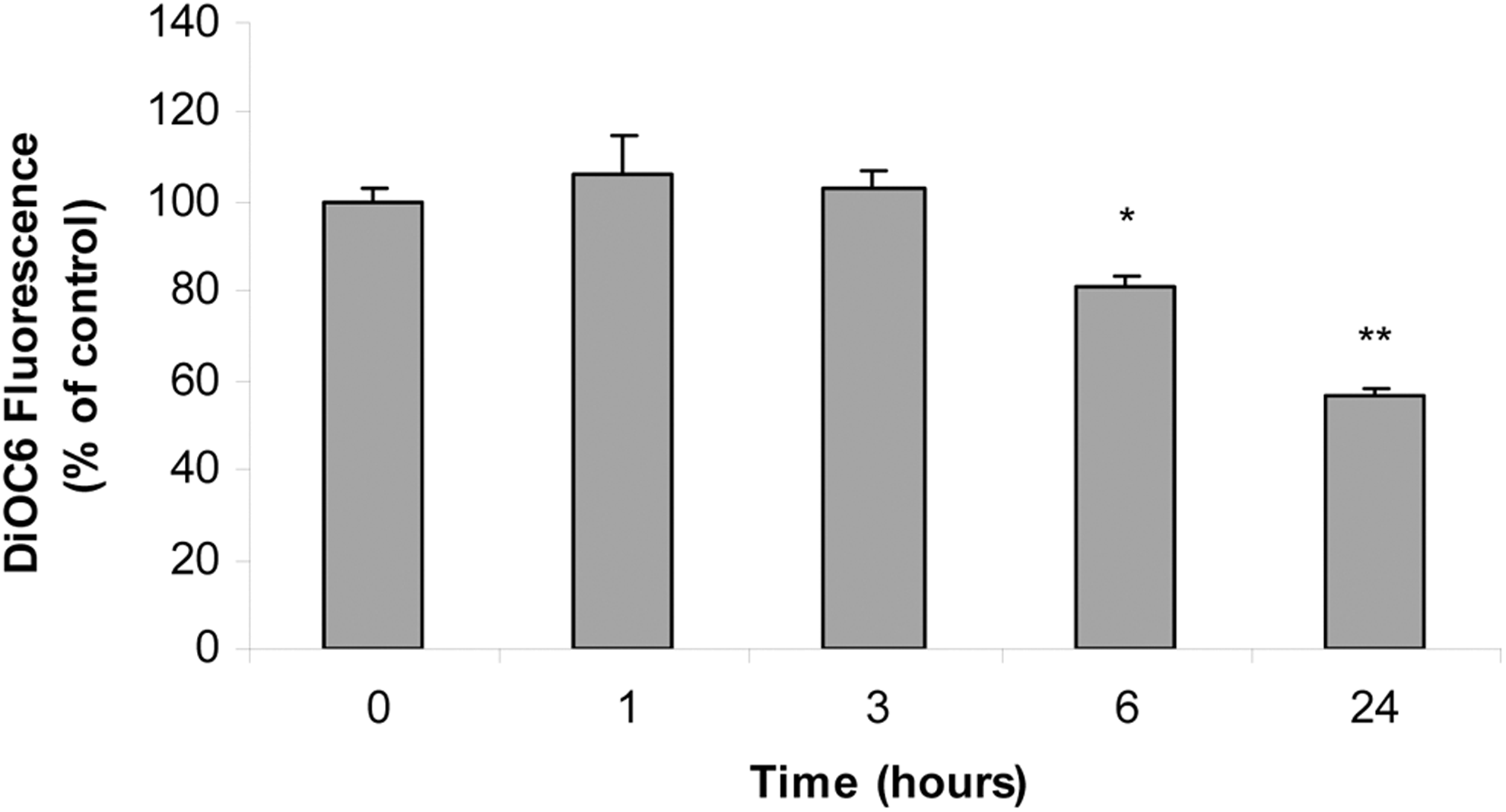
Arsenic causes depolarization of mitochondrial membrane potential after 6-hour treatment. Anterior pituitary cells were incubated with 25 μmol/L inorganic arsenic (iAs; NaAsO2) for different times. Changes in mitochondrial membrane potential (MMP) were measured by 3,3′-dihexyloxacarbocyanine iodide (DiOC6) cell staining and flow cytometry. Bars represent the mean of DiOC6 fluorescence intensity ± standard error (SE) and are expressed as a percentage of the control, n = 3. Analysis of variance (ANOVA) followed by Dunnett test, *P < 0.05; **P < 0.01 versus control.
Arsenic Treatment Increased ROS Levels
A large body of evidence gathered from different cell types indicates that iAs-induced apoptosis requires ROS production. 46 To assess whether iAs treatment changes intracellular ROS levels in our system, anterior pituitary cells were incubated with iAs for different time lapses, and ROS were measured using a DHR123 probe followed by flow cytometry. Our results show that ROS levels significantly increased during the first 3 hours of iAs incubation and decreased later in time (Figure 6).
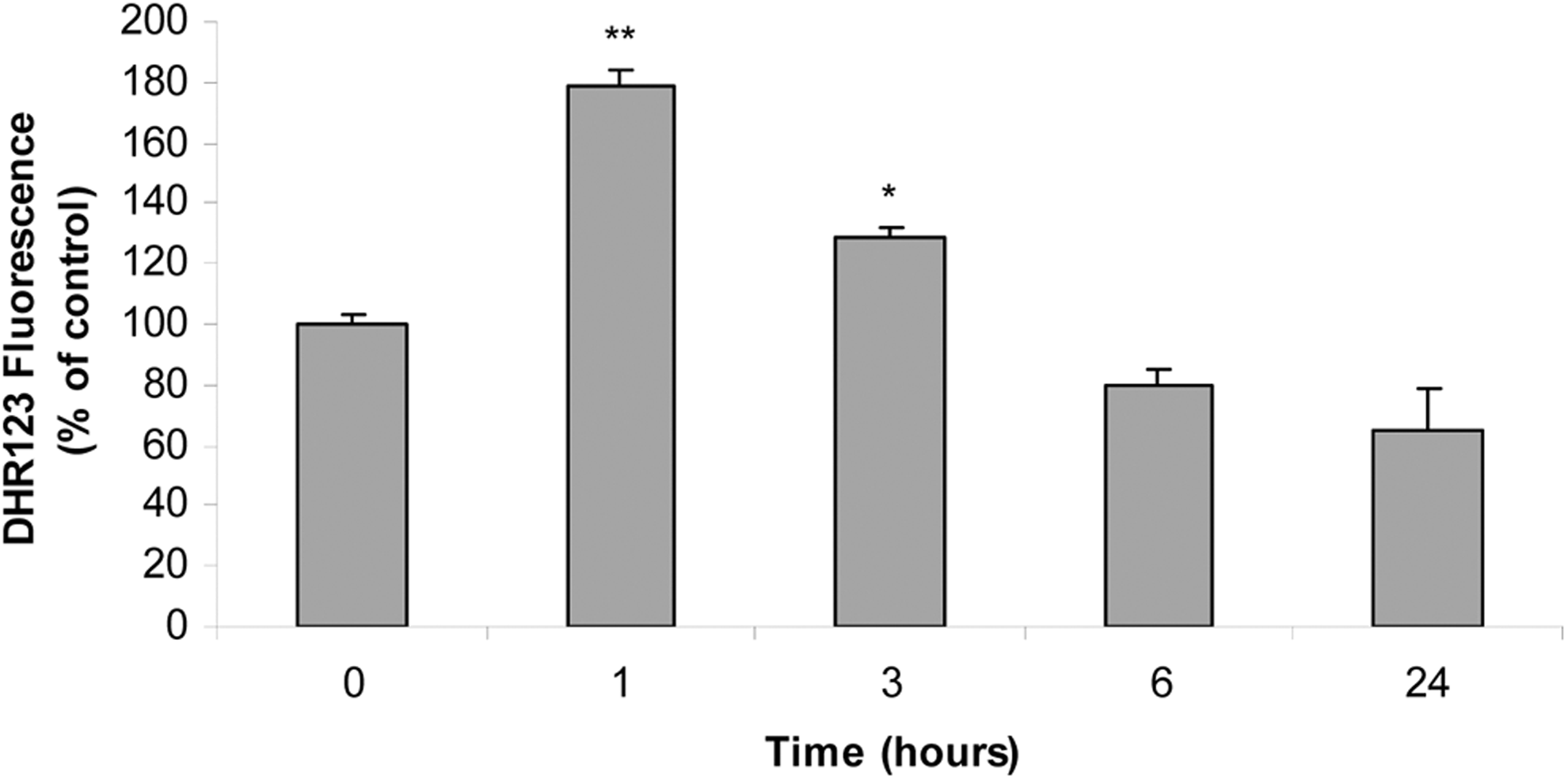
Arsenic increases reactive oxygen species (ROS) levels. Anterior pituitary cells were incubated with 25 μmol/L inorganic arsenic (iAs; NaAsO2) for different times. The ROS levels were measured by dihydrorhodamine 123 (DHR123) cell staining and flow cytometry. Bars represent the mean of DHR123 fluorescence intensity ± standard error (SE) and are expressed as a percentage of the control, n = 3. Analysis of variance (ANOVA) followed by Dunnett test, *P < 0.05; **P < 0.01 versus control.
Arsenic Induced Oxidative Stress-Responsive Gene Expression
To further examine iAs-mediated oxidative stress response, mRNA levels of HMOX1 and MT-1 were evaluated. Cells were exposed to the metalloid for 3, 6, or 9 hours, and mRNA expression was measured by PCR. Messenger RNA levels of both HMOX1 and MT-1 were significantly increased after 3 hours and were still increased after 9 hours of iAs treatment (Figure 7). Altogether, these results indicate that iAs early generates oxidative stress, which in turn triggers cell’s antioxidant response.
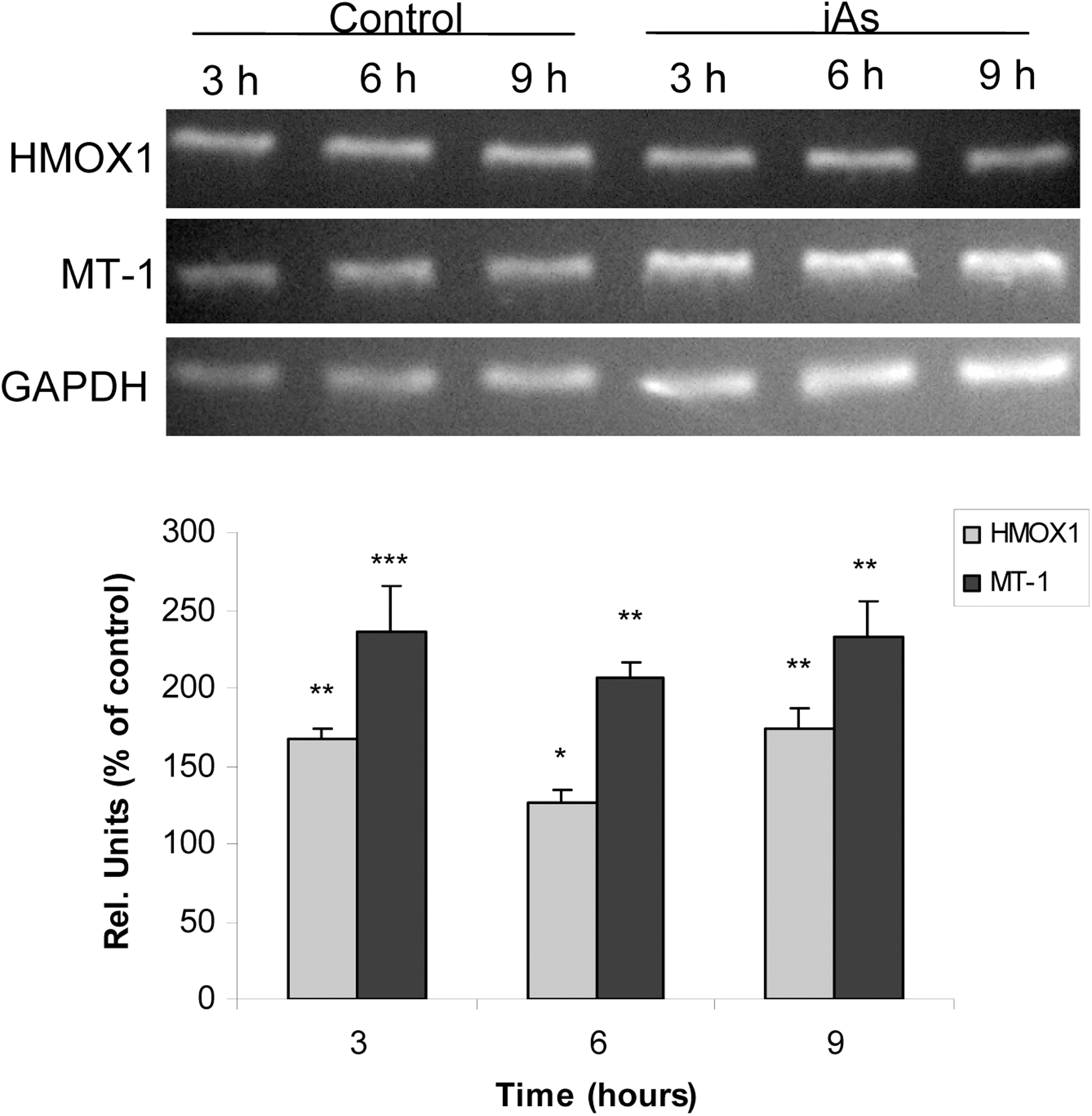
Arsenic modifies oxidative stress-responsive gene expression. Anterior pituitary cells were incubated with 25 μmol/L inorganic arsenic (iAs; NaAsO2) for different times. Messenger RNA (mRNA) expression of heme oxygenase-1 (HMOX1) and metallothionein-1 (MT-1) was evaluated by polymerase chain reaction (PCR). Representative gel images of PCR analysis are shown. Bars represent the mean ± standard error (SE) of densitometric values normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and are expressed as a percentage of the respective control, n = 3. Analysis of variance (ANOVA) followed by Tukey-Kramer test, *P < 0.05, **P < 0.01; ***P < 0.001 versus respective control.
Arsenic-Induced Oxidative Stress, PRL Decrease, and Cell Death Are Prevented by N-Acetyl Cysteine Treatment
Thiol-containing molecules play a key role in cellular redox status control. To establish whether iAs-induced oxidative stress mediates cytotoxicity, cells were first incubated with N-acetyl cysteine (NAC), a thiol-protecting glutathione precursor with antioxidant capability, and then treated with the metalloid. Cells were treated for 2 hours with 2 mmol/L NAC followed by iAs incubation without NAC for 6 or 24 hours. N-acetyl cysteine treatment was able to fully prevent iAs-induced HMOX1 and MT-1-increased mRNA expression (Figure 8A). Additionally, this antioxidant completely abrogated the iAs-mediated PRL release inhibition (Figure 8B). Moreover, NAC treatment partially prevented iAs-induced apoptosis (Figure 8C). In sum, these results suggest that ROS production is involved in the iAs-mediated PRL release inhibition and cytotoxicity.
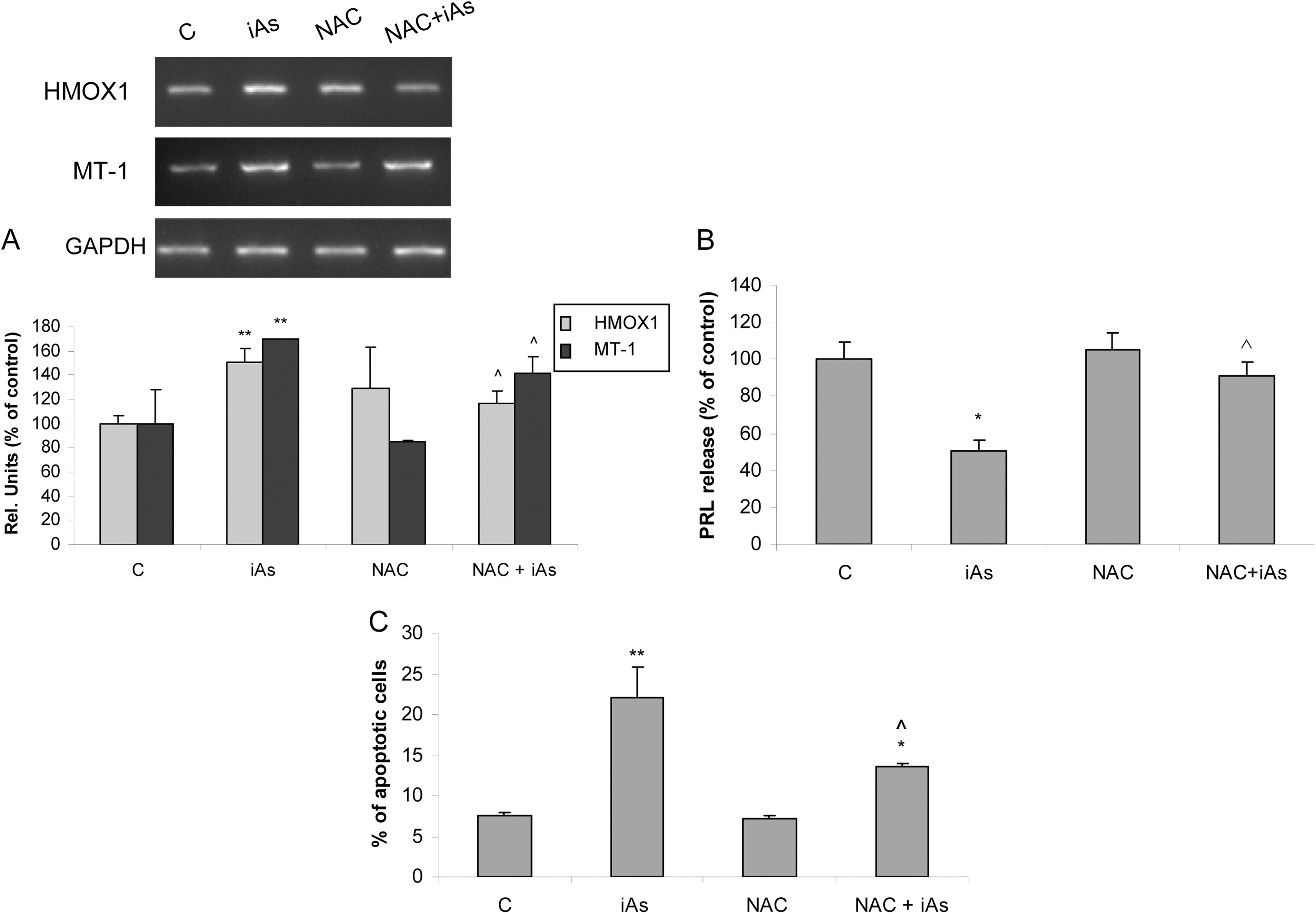
N-acetyl cysteine (NAC) prevents arsenic-induced oxidative stress, prolactin (PRL) decrease, and apoptosis. Anterior pituitary cells were preincubated with 2 mmol/L NAC followed by 25 µmol/L inorganic arsenic (iAs; NaAsO2) treatment without NAC for 6 hours (A and B) or 24 hours (C). A, Messenger RNA (mRNA) expression of heme oxygenase-1 (HMOX1) and metallothionein-1 (MT-1) was evaluated by polymerase chain reaction (PCR). Representative gel images of PCR analysis are shown. Bars represent the mean ± standard error (SE) of densitometric values normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and are expressed as a percentage of the respective control, n = 3. Analysis of variance (ANOVA) followed by Tukey-Kramer test, **P < 0.01 versus respective control, ⁁P < 0.05 versus iAs. B, PRL release into the medium was determined by radioimmunoassay (RIA). Bars represent the mean ± standard error (SE) and are expressed as a percentage of the control, n = 3. Analysis of variance followed by Tukey-Kramer test, *P < 0.05 versus control, ⁁P < 0.05 versus iAs. C, Apoptosis was determined by annexin V-fluorescein isothiocyanate (FITC) and propidium iodide (PI) staining followed by flow cytometry analysis. Bars represent the mean ± SE, n = 3. Analysis of variance followed by Tukey-Kramer test, *P < 0.05; **P < 0.01 versus control, ⁁P < 0.05 versus iAs.
Discussion
In the present study, we demonstrated for the first time that iAs exposure causes oxidative stress, thus exerting deleterious actions in anterior pituitary physiology.
We showed that 5, 25, and 100 ppm iAs administration through drinking water generates oxidative stress and affects hormone release. Although iAs maximum contaminant level in drinking water is 0.01 ppm (EPA), we used concentrations above this limit since we ran an animal-based toxicological study during a short exposure time. Importantly, this work was not directed to inform risk assessment at these iAs concentrations because of substantial differences in iAs toxicity between rat and human. In this way, iAs methylation rate is higher in rat, which in turn allows a more effective iAs urine excretion as well as a lower iAs accumulation in tissues. 27,28 For these reasons, direct comparisons between human and rat are not accurate at the same iAs dose.
Heme oxygenase-1, MT-1, and Trx-1 are involved in the first steps of the cell antioxidant defense and are considered oxidative stress markers. Heme oxygenase-1 catalyzes the first, rate-limiting step of heme degradation, which produces carbon monoxide, iron, and biliverdin, a potent antioxidant. 47,48 Heme oxygenase-1 is a well-known biomarker of iAs-induced oxidative stress and is thought to be involved in toxic metalloid adaptation. 49 Similarly, MT-1 is a metal-binding protein with high sulfhydryl content that defends against metal-induced oxidative cell injury. 50 Arsenic induces MT-1 expression both in vitro and in vivo, 51,52 possibly by binding to the MT-1 promoter. Thioredoxin-1 shields a redox-active disulfide/dithiol within the conserved active site sequence (-Cys-Gly-Pro-Cys-) and plays an important role in regulating cell redox homeostasis and cell growth, differentiation, and apoptosis. Thioredoxin-1 regulates iAs-induced apoptosis by preventing mitochondrial cytochrome c release. 53
Our results showed that iAs administration increased mRNA levels of HMOX1 and Trx-1 but did not modify their expression in hypothalamus. This could be due to a lower iAs accumulation in hypothalamus because of the blood–brain barrier 11 or to a greater sensitivity of pituitary to iAs-induced oxidative stress.
Several reports have indicated that different toxic metals such as cadmium, lead, mercury, and chromium disturb the hypothalamic–pituitary–gonadal axis and affect hormonal status. 38,54 –57 In order to expand our understanding of the effect of iAs on the pituitary, we ran experiments on anterior pituitary cells in culture. Here, we demonstrate for the first time that iAs induces apoptosis and decreases PRL release in anterior pituitary cells.
Arsenic-induced cell death is a dose- and time-dependent process. Our studies of morphology and phosphatidylserine externalization suggest that iAs-mediated cytotoxicity is primarily apoptosis driven. Apoptosis induction by iAs tallies with numerous reports that demonstrate the same effect in other tissues. 58 –60 Although apoptosis is the most commonly identified mechanism of iAs-mediated cell death, we observed a small percentage of annexin V- and PI-positive cells, which indicates that iAs may also induce necrosis in anterior pituitary cells, albeit to a lesser extent.
Even though arsenic toxicity is species- and cell type-specific, free radical generation and oxidative stress are common to many tissues. 16 In vivo and in vitro evidence indicates that arsenic generates oxidative stress mainly by superoxide and hydrogen peroxide production. 60 –64 Arsenic-mediated cell damage also results from the ability of trivalent arsenic to bind thiol groups of biomolecules, which inhibits their activity. 65
Our results demonstrate that iAs acutely increased ROS levels and caused early MMP depolarization, which increased over time. It has been suggested that the mitochondrial respiratory chain could be a major source of arsenite-mediated ROS production. 66 Other sources of ROS generation have also been proposed including oxidation of arsenite to arsenate 67 and glutathione depletion. 68 Because of arsenic’s thiol-oxidizing property, mitochondria are prime targets of iAs intoxication, either indirectly via ROS accumulation or directly via mitochondrial matrix condensation and enhanced membrane permeability. 69 In either case, these iAs-induced mitochondrial insults initiate apoptosis, which we observed as cells undergoing nuclear condensation and fragmentation after 9 hours of exposure. Mitochondrial membrane potential decrease and ROS production could be interrelated since both phenomena are dependent on electron transport chain (ETC) functionality. Reactive oxygen species disrupts mitochondrial membrane integrity, 70 which affects the ETC, thus reducing ETC-dependent ROS generation and MMP.
Concordant with our in vivo results, we demonstrated that in vitro iAs treatment increases mRNA expression of HMOX1 and MT-1, which further underscores that anterior pituitary gland is a direct target of iAs pro-oxidant effects.
Here, we demonstrated that ROS generation is involved in iAs-mediated cytotoxicity given that NAC treatment prevented the increase in oxidative stress markers’ expression, PRL decrease, and cell death caused by iAs. However, apoptosis prevention is only partial, which may indicate that an ROS-independent mechanism may also be involved in iAs-induced apoptosis. To this end, Morales et al demonstrated that ROS inhibition had no effect on arsenic trioxide-induced cell death in a cell line of human multiple myeloma. 71 On the other hand, we cannot rule out that intracellular NAC accumulation, previous to iAs treatment, might not be sufficient to completely protect against ROS actions.
In this study, we demonstrate that in vivo exposure to 25 and 100 ppm iAs caused a decrease in serum PRL levels, whereas only 100 ppm iAs negatively affected LH levels. In addition, in anterior pituitary cells, we found that PRL release into culture media decreased time dependently, whereas LH levels remained unchanged. This differential pattern may be due to greater sensitivity of lactotrophs to the cytotoxic effect of iAs, which was further confirmed by immunocytochemical studies. In line with these results, previous reports from our laboratory showed that cadmium and chromium VI-mediated PRL release inhibition is related to marked lactotrophs susceptibility to heavy metals. 38,57 Mechanisms responsible for iAs cytotoxicity in anterior pituitary cells are summarized in Figure 9.
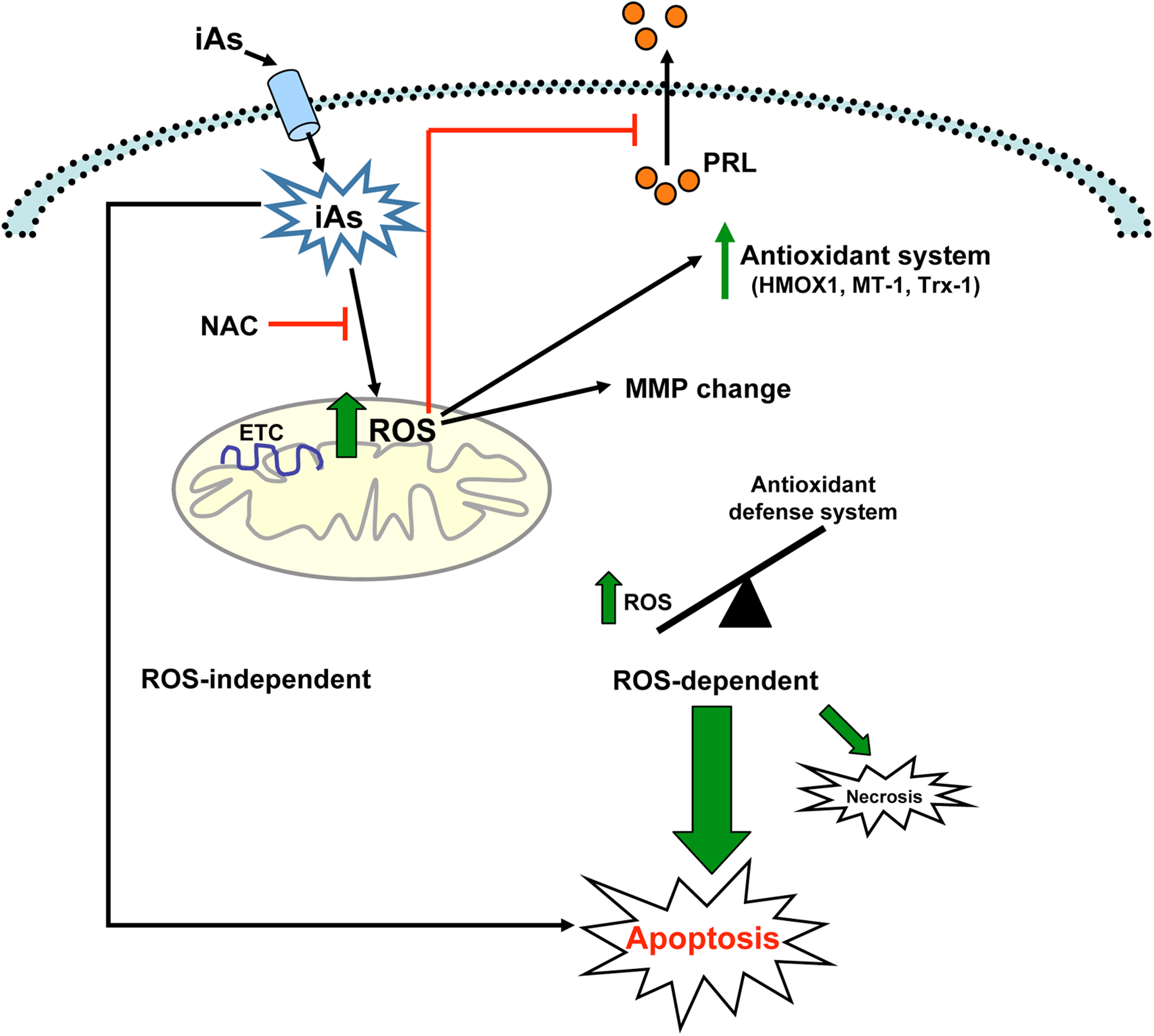
Mechanisms of inorganic arsenic (iAs)-driven toxicity in anterior pituitary cells. Once inside the cell, arsenic (iAs), as sodium arsenite, promotes reactive oxygen species (ROS) formation by affecting electron transport chain (ETC) components. This raise in ROS levels inhibits prolactin (PRL) release and stimulates the expression of several genes from cell antioxidant defense system, such as heme oxygenase-1 (HMOX1), metallothionein-1 (MT-1), and thioredoxin-1 (Trx-1). Later in time, iAs-induced ROS causes a mitochondrial membrane potential (MMP) depolarization. The imbalance of cell redox status leads to cell death mainly by apoptosis and in a lesser extent by necrosis. Inorganic arsenic toxic effects on hormone release and cell viability are partially prevented by the antioxidant N-acetyl cysteine (NAC). In addition, iAs could induce other apoptotic mechanisms independent of ROS generation.
The present work provides solid evidence in relation to the deleterious effects of arsenic on pituitary physiology. We demonstrate that arsenic directly inhibited PRL release in vivo and in vitro and induced apoptosis principally by generating oxidative stress. Understanding arsenic’s effects on pituitary physiology is of major interest because this metalloid may have adverse effects on normal endocrine function, which remains a largely unexplored field.
Footnotes
Acknowledgments
The authors thank Mercedes Imsen for her kind help with animal care and handling and Dr Victoria Lux-Lantos (Instituto de Biología y Medicina Experimental-CONICET, Buenos Aires, Argentina) for the labeled hormones we used for the RIA.
Authors’ Note
S. Ronchetti contributed to acquisition and analysis of data and drafted the manuscript. B. Duvilanski contributed to acquisition and analysis of data and critically revised the manuscript for important intellectual content. M. Bianchi contributed to acquisition of data and critically revised the manuscript for important intellectual content. J. Cabilla contributed to conception or design and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants from Agencia Nacional de Promoción Científica y Tecnológica (PICT 10-1668), Universidad de Buenos Aires (B100473), and Consejo Nacional de Investigaciones Científicas y Técnicas (PIP 5536).
