Abstract
Information on the effects of phytoestrogens on animals has increased recently; however, there were only few studies on prenatal exposure on cellular immune response. Pregnant rats were assigned to 3 groups (12 rats per group), the first was fed control diet, the second was fed low-dose (6.5 g/100 g of diet) soy isoflavones, while the third was fed high-dose (26 g/100 g of diet) soy isoflavones. The female offspring cell-mediated immune response was determined using phytohemagglutinin (PHA) injection, and intumesce index was calculated on postnatal day 50. After 24 hours of PHA injection, blood samples were collected for tumor necrosis factor α, interferon γ (IFN-γ), and interleukin (IL)-12 determination. Spleen, thymus, and PHA-injected footpads were fixed for histopathology. Intumesce index was significantly (P < 0.05) reduced in rats’ offspring born from dams fed low- and high-dietary soy isoflavones than that in control groups. Thymic relative weights in offspring of rats fed high-dietary soy isoflavones showed a significant (P < 0.05) decrease compared to that in the control group. Female offspring where low and high-dietary soy isoflavones were fed to their dams showed a significant (P < 0.05) decrease in IFN-γ and IL-12 than that in control ones. Spleen of rats born from dams fed high dose of dietary soy isoflavones showed lymphocytic depletion in white pulp. Taking together, it is clear that dietary soy isoflavones at prenatal period had immunosuppressive effect on female offspring after PHA stimulation. This effect was mediated through reduced IFN-γ that interplayed in IL-12 production pathway thus reducing its level.
Introduction
Sex steroid hormones affect the immune system and susceptibility to disease. 1 Although estrogens can enhance both cell-mediated and humoral immune responses, there were some reports, which indicated that estrogens suppressed some cell-mediated immune responses. 2 In addition to exposure to endogenous hormones, both humans and animals are exposed to estrogenic chemicals in the environment. 3,4 These chemicals bind to sex steroid receptors but with a lower affinity than endogenous steroid hormones. 5 Besides their effects on ligand receptors, estrogenic chemicals affect several intracellular molecules, indicating that environmental estrogens may exert their effects through cell signaling pathways. 6,7
Phytoestrogens are polyphenolic nonsteroidal plant compounds with estrogen-like biological activities 8 that have the ability to trigger estrogenic activity to act as agonist 9 or antagonist, 10 depending on the tissue, estrogen receptor (ER), and concentration of circulating endogenous estrogens. 11 Soy products have become an important component of food products in adult and infant human diets in recent years. 12 Most studies have focused on the effects of estrogenic pesticides and toxic substances on immune function, and less attention has been paid to the effects of naturally occurring phytoestrogens on the immune system. 13,14 In toxicity studies, endocrine-mediated effects have been reported in rat pups of dams treated with genistein during the gestational and/or lactational periods. 15 -18
Tumor necrosis factor α (TNF-α) has a crucial role in immune regulation by modulating lymphocyte proliferation and apoptosis, which is implicated in maintaining immune homeostasis. 19 Tumor necrosis factor α activates cell inflammation, proliferation, survival, and cell death. 20 A number of cell populations from both the innate (eg, natural killer [NK] cells, NKT cells, and macrophages) and adaptive immune systems (eg, T helper 1 [Th1] cells and B cells) can secrete interferon γ (IFN-γ). 21 IL-12 mediates development and maintenance of TH1 cells by inducing production of IFN-γ by TH1 and NK cells. Interleukin (IL)-12 indirectly activates the antimicrobial and antitumor activity of macrophages and promotes cytolytic activity of NK cells. 22
With the increase in human consumption of phytoestrogens, studies that address the consequences of phytoestrogens exposure are required. Knowledge on the effects of phytoestrogens has increased recently, however, there have been only few studies concerning prenatal exposure to phytoestrogens on cellular immune response in female rats. Additionally, the immune response to intradermal phytohemagglutinin (PHA) was used for estimating T cell-mediated immunity in avian species, however, there were no available literature in rodents. Therefore, the aim of this study was to investigate the prenatal exposure of dietary soy isoflavones on immune response of female albino rats’ offspring through determination of the immune response to intradermal PHA injection via calculation of Intumesce index, determination of TNF-α, IFN-γ, and IL-12 levels and examination of histopathological changes in PHA-injected foot pad, thymus, and spleen.
Materials and Methods
Animals
Thirty-six regular cyclic adult female Wister albino rats, weighing approximately 180 to 250 g, were purchased from Laboratory Animal House at Faculty of Veterinary Medicine, Suez Canal University, Ismailia, Egypt. Rats were housed in a standard plastic cage (3 rats per cage). They were maintained under standard natural day light rhythm with an average temperature of 25°C (±1°C). They were provided casein-based diet and water ad libitum ad libitum. The experiment was approved by the Committee of Laboratory Animals at Faculty of Veterinary Medicine, Suez Canal University.
Breeding Procedures
After 2 weeks of acclimatization, the estrous cycle was monitored by daily cytological examination of vaginal smear 23 to determine the females with 3 successive regular cycles. Vaginal smears were obtained from each female once daily early in the morning. The smears were dried with a flame and stained for 1 minutes with methylene blue and examined under a microscope to determine the phase of estrous cycle. Females with regular cycles were selected for this study, while irregular ones were excluded. Mature males were introduced with proestrus virgin females in ratio of 1 male–3 females per cage at the afternoon and stay overnight. Mating was confirmed by the presence of sperms in the vaginal smears or the presence of vaginal plug, and this was designated as the zero day of pregnancy. 24
Experimental Groups and Treatment
The rats at day 0 of pregnancy were assigned to 3 independent groups (12 rats per group). The first group (n = 12) of pregnant dams were fed casein-based control diet (soy isoflavones free). The second group (n = 12) were fed low-dose (6.5 g/100 g of the diet) soy isoflavones. The third group (n = 12) were fed high-dose (26 g/100 g of the diet) soy isoflavones. All diets were formulated to fulfill all the nutritional requirements of pregnant rats. 25 Experimental diets were offered from zero day of pregnancy to the day of giving birth. After parturition, all experimental rats and their offspring were given a control diet. The number of pups per litter, number of female and male offspring per dam, and the offspring mortality rates were determined. Only female offspring were used in this experiment, while male offspring were assigned to another experiment.
Dietary Isoflavones Analysis
Isoflavones were extracted from the diet by mixing 1 g of sample diet with 20 mL of solution of 0.1 mol/L HCl/L and 80 mL of methanol, and then the mixture was sonicated for 20 minutes and left at room temperature for 2 hours and gravity filtered with Whatman filter paper (Clifton, New Jersey). The filtrate was centrifuged at 10,000 rpm, and the isoflavones in the supernatant were separated and quantified by high-performance liquid chromatography. Isoflavones were detected at 260 nm and quantified by comparison with external standards. 26
Body Weight
Pregnant dams were weighed weekly and their food intake (g/wk) was determined. Moreover, female offspring of each dam were weighed weekly starting from day of birth till the end of experiment (postnatal day [PND] 50).
Cell-Mediated Immune Response After PHA Injection in Foot Pad
On PND 49, offspring cell-mediated immune response was determined using PHA injection. Phytohemagglutinin challenge was performed by injection of 0.1 mL of PHA (Sigma L 9017, St Louis, Missouri) of 1 mg mL−1 dissolved in sterile phosphate-buffered saline (PBS) 27 in left foot pad of female rats in all experimental groups. Then as a control, the right foot pad of the same rat was injected with 0.1 mL of PBS. Foot pad swelling was calculated. 27 Thickness of both footpads was measured before and after PHA injection. After 24 hours, thickness of dorsoventral and lateral aspects of left and right footpads at point of injection was measured using a manual micrometer. The injections and measurements were made by the same person. The Intumesce Index was calculated as the measured ankle size—primary ankle size/primary ankle size. 28
Blood Sampling
On PND 50 of offspring age and after 24 hours of PHA injection, female rats were anaesthetized with diethyl ether, and two blood samples were collected via capillary tube from the retro-orbital venous plexus; the first one was collected in plain tubes for serum separation. Serum was stored at −20°C until determination of TNF-α, IFN-γ, and IL-12 levels. While the second blood sample was collected in EDTA for total and differential leukocytic counts.
Determination of Serum TNF-α, IFN-γ, and IL-12 Levels
Serum TNF-α was measured using rat TNF-α enzyme-linked immunosorbent assay (ELISA) kit (Cat No. 27194, IBL Co, Japan) according to manufacturer instructions. Serum IFN-γ was measured using rat IFN-γ ELISA kit (Cat No.RIf00, R&D systems, China) according to manufacturer instructions. Serum IL-12 was measured using rat IL-12/P70 ELISA kit (Cat No.CSB-E07364r, CUSABIO, China) according to manufacturer instructions.
Determination of Total Leukocytic Count and Differential Leukocytic Count
Leukocytes were counted by Neubauer’s improved haemocytometer using Turk’s solution. The number of leukocytes per cubic millimeter was calculated. 29 Percentage values for each type of white cells were calculated after spreading of blood film and staining with Giemsa stain. 30
Organs Relative Weights and Histopathology
On PND 50, female offspring were euthanized by over dose of diethyl ether. Spleen and thymus from offspring were weighed, and the relative weights were calculated. Moreover, PHA injected footpads and its associated popliteal lymph nodes, spleen, and thymus were fixed in 10% neutral buffered formalin. They were gradually dehydrated and embedded in paraffin and stained with H&E for histopathological examination. 31 The decrease in red pulp cellularity, decrease in periarteriolar lymphoid sheath size and fibrosis of spleen as well as the decreased number of lymphocytes in cortex and medulla of the thymus was estimated. 32
Statistical Analysis
All values were expressed as mean ± standard error of the mean. One-way analysis of variance was used for examining differences among groups. Intergroup comparisons were made by Tukey test. A P value of <0.05 was considered to indicate significance. All the analyses were done using GraphPad Prism (Version 5.01, GraphPad software, San Diego, California).
Results
Effect of Soy Isoflavones on Litter Size, Number of Offspring, and Mortality Rate
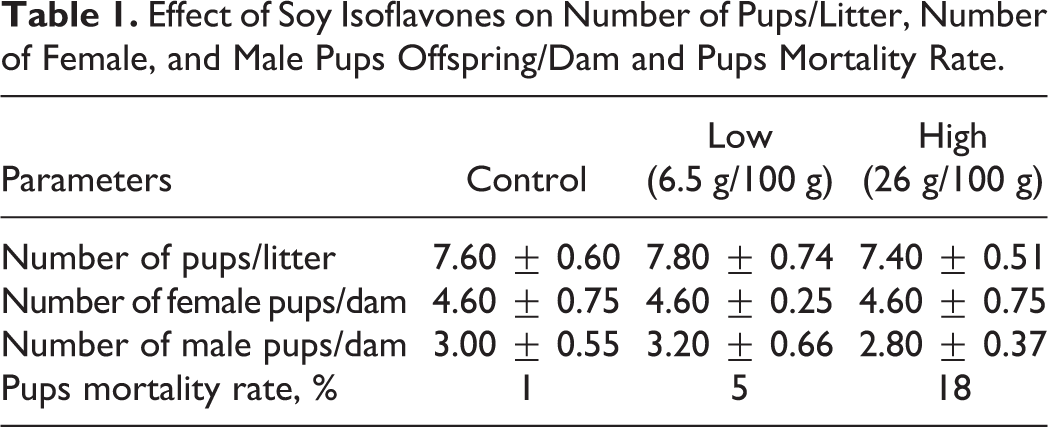
The number of pups per litter (litter size) and the number of female and male offspring per dam showed nonsignificant differences among the control and treated groups. However, offspring mortality rate was increased by 17 and 13% in offspring born to dams fed high soy isoflavones compared to offspring born to dams fed soy isoflavones free diet and offspring born to dams fed low soy isoflavones, respectively (Table 1).
Effect of Soy Isoflavones on Number of Pups/Litter, Number of Female, and Male Pups Offspring/Dam and Pups Mortality Rate.
Effect of Soy Isoflavones on Body Weight and Food Intake of Dams and Their Offspring
From day zero until end of pregnancy, body weight gain showed a significant (P < 0.05) decrease in rats fed high-dietary soy isoflavones compared to that in the control group. At first and final weeks of the experiment, the offspring from dams fed low and high-dietary soy isoflavones showed a significant decrease (P < 0.05) in body weight compared to that in the control group. Interestingly, in the first day after natural birth, the offspring born from dams fed low and high-dietary soy isoflavones showed significant decrease (P < 0.05) in body weight compared to the offspring born from control dams. At week 1 of gestation, dams given low doses of soy isoflavones showed significant increase (P < 0.05) in their food intake compared to dams given high dose; however, there were no significant differences in food intake in rats at week 3 of gestation (Table 2).
Effect of Soy Isoflavones on Gestational Weight Gain, Food Intake of Pregnant Dams, and Offspring Body Weight.a
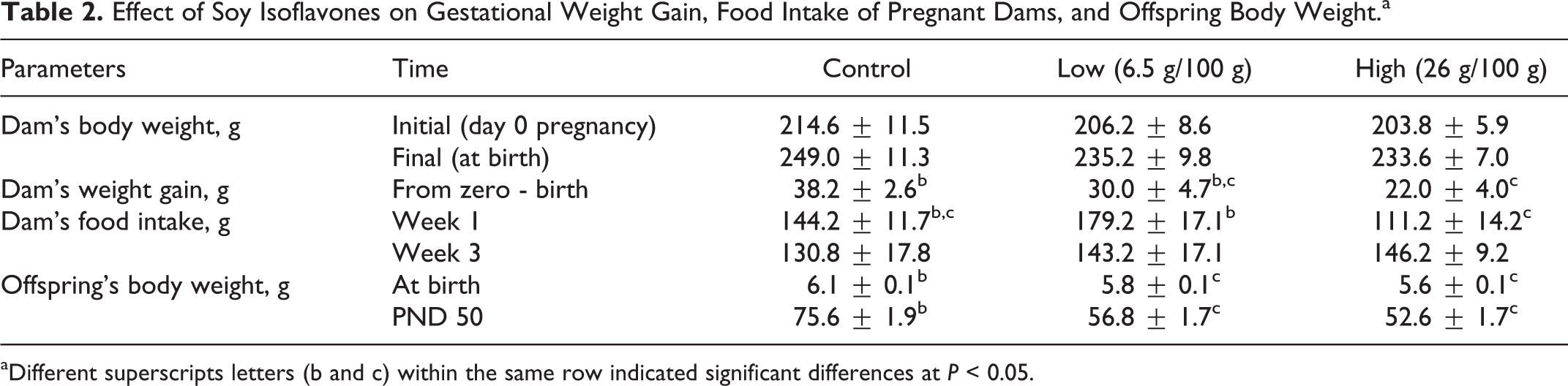
aDifferent superscripts letters (b and c) within the same row indicated significant differences at P < 0.05.
The Response of Offspring to Intradermal PHA Injection
Intumesce index of ankle was significantly (P < 0.05) reduced in offspring born from dams fed low and high-dietary soy isoflavones compared to control groups (Table 3).
Effect of Prenatal Exposure to Dietary Soy Isoflavones on Intumesce Index of Female Offspring After 24 hours of PHA Injection, TNF-α, IFN-γ, IL-12 Levels, and Relative Organ Weights.a
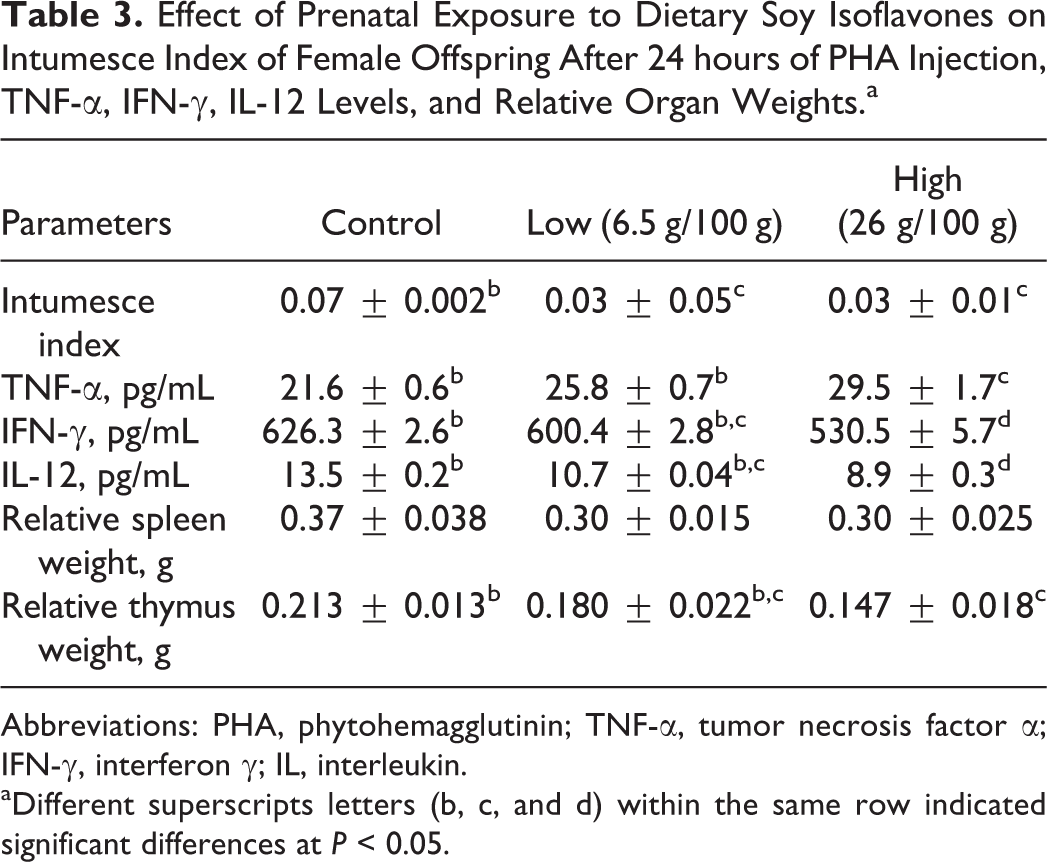
Abbreviations: PHA, phytohemagglutinin; TNF-α, tumor necrosis factor α; IFN-γ, interferon γ; IL, interleukin.
aDifferent superscripts letters (b, c, and d) within the same row indicated significant differences at P < 0.05.
Effect of Soy Isoflavones Administration on TNF-α, IFN-γ, and IL-12 in Offspring
Prenatal exposure to high-dietary soy isoflavones significantly (P < 0.05) increased TNF-α level in these offspring compared to offspring born to dams of low and control groups. Moreover, offspring of dams fed low and high-dietary soy isoflavones showed a significant (P < 0.05) decrease in serum IFN-γ levels compared to the control group. High-dietary soy isoflavones intake by dams resulted in a significant (P < 0.05) decrease in IFN-γ compared to that from dam fed low soy isoflavones. Offspring born from dams treated with low and high soy isoflavones showed significant (P < 0.05) decrease in levels of IL-12 compared to offspring of control dams (Table 3).
Effect of isoflavones on Total and Differential Leukocytic Counts
Low and high maternal exposure to soy isoflavones significantly (P < 0.05) reduced total leukocytic count (TLC) in offspring compared to rats born from control group. There was no significant difference in basophil, eosinophil, and neutrophil among different groups. However, offspring born from dams treated with low and high maternal exposure to soy isoflavones showed a significant (P < 0.05) decrease in monocyte and lymphocyte compared to the female albino rat offspring born from the control group (Table 4).
Effect of Soy Isoflavones Administration on Total and Differential Leukocytic Counts in Female Offspring Albino Rat.a
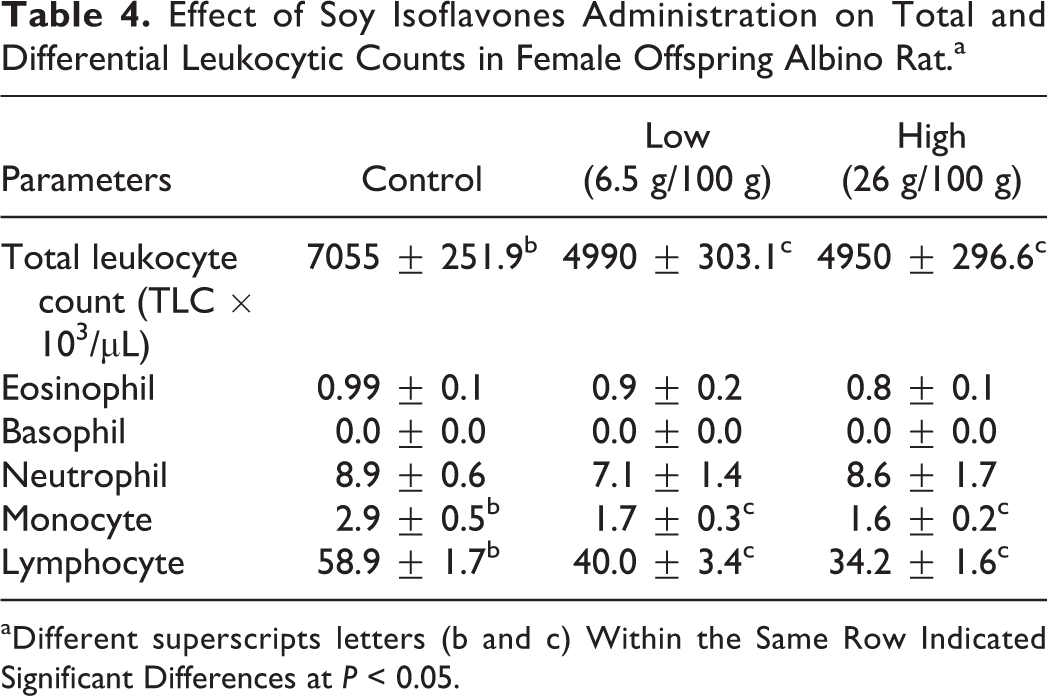
aDifferent superscripts letters (b and c) Within the Same Row Indicated Significant Differences at P < 0.05.
Thymus, Spleen Relative Weights, and Histopathology
Maternal exposure to dietary soy isoflavones didn’t change relative splenic weights in female albino rat offspring. However, thymus relative weights in offspring of dams fed high-dietary soy isoflavones showed significant (P < 0.05) decrease compared to that in the control group (Table 3).
Footpad of offspring born to control dams showed severe leukocyte infiltration, while rats born from dams fed low-dietary soy isoflavones showed mild leukocyte infiltration. Rats born from dams fed high-dietary soy isoflavones showed no leukocyte infiltration (Figure 1).
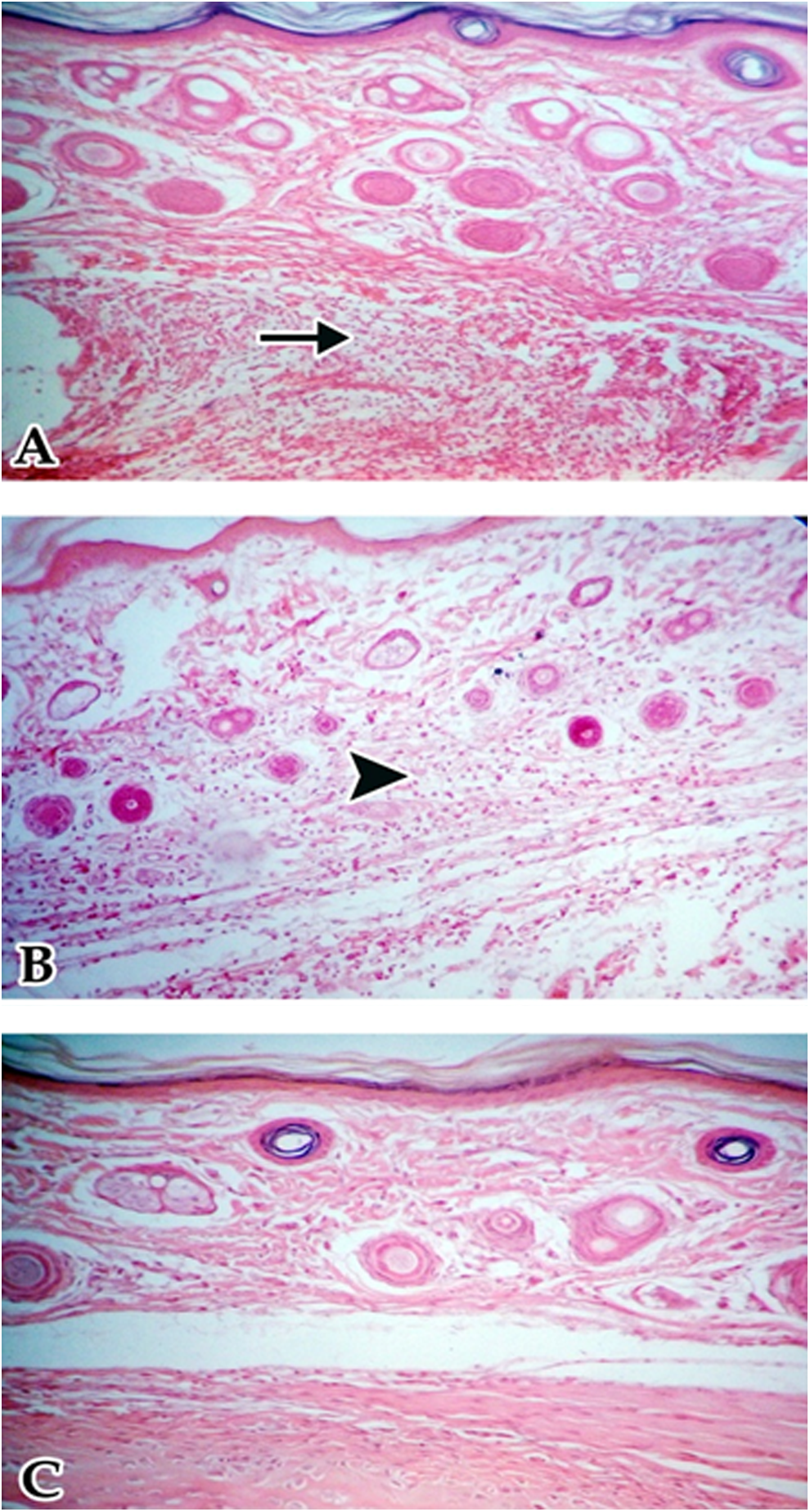
Footpad of female albino rat offspring 24 hours after phytohemagglutinin (PHA) injection. A, Control rats showed severe leukocyte infiltration (arrow). B, Rats born from dams fed low-dietary soy isoflavones showed mild leukocyte infiltration (head arrow). C, Rats born from dams fed high-dietary soy isoflavones showed no lymphatic infiltration (hematoxylin and eosin; 200×).
Spleen of offspring born to control dams showed normal white and red pulp areas, while spleen of rats born from dams fed low dose of dietary soy isoflavones showed a decrease in white and red pulps and coagulative necrosis. Spleen of rats born from dams fed high dose of dietary soy isoflavones showed lymphocyte depletion in white pulp (Figure 2).
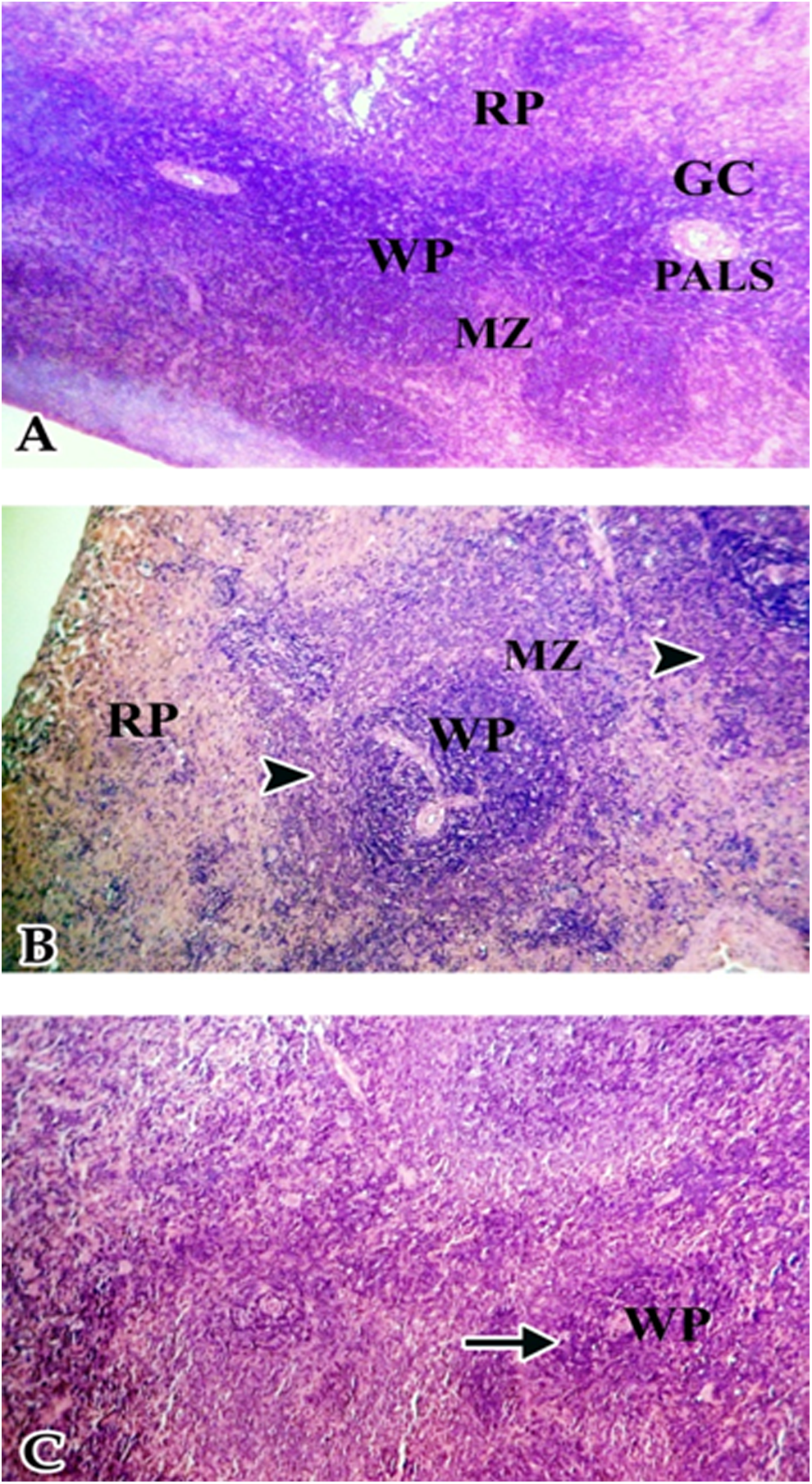
Spleen of female albino rat offspring 24 hours after phytohemagglutinin (PHA) injection. A, Spleen from control rats showed white pulp (WP) area, red pulp (RP) area, marginal zone (MZ), germinal center (GC), and periarteriolar lymphoid sheath (PALS). B, Spleen of rats born from dams fed low dose of dietary soy isoflavones showed decrease in WP, RP cellularity, and coagulative necrosis. C, Spleen of rats born from dams fed high dose of dietary soy isoflavones showed lymphocyte depletion in white pulp (arrow; hematoxylin and eosin; 200×).
Thymus of offspring born to control dams showed distinct cortex with a uniformly dense population of lymphocyte and normal medulla. Rats born from dams exposed to low-dietary soy isoflavones showed tangible body macrophages with intracytoplasmic apoptotic bodies. Rats born from dams exposed to high dose of soy isoflavones showed severe lymphocyte depletion, vacuolation, and reduction in lymphatic tissue in cortex and medulla (Figure 3).
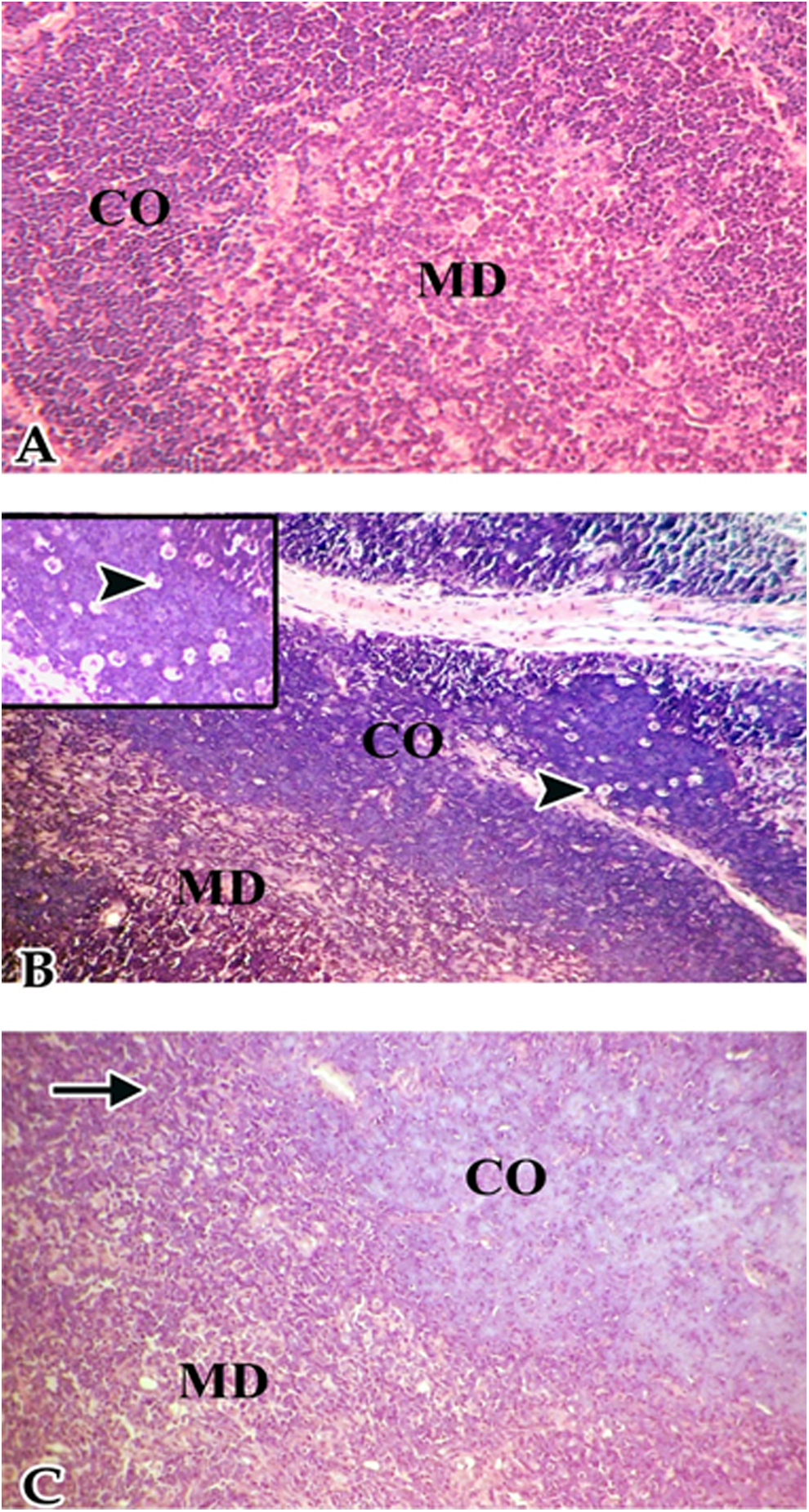
Thymus of female albino rat offspring 24 hours after phytohemagglutinin (PHA) injection. A, Thymus of control rats showed distinct cortex (CO) with a uniformly dense population of lymphocyte and normal medulla (MD). B, Rats born from dams exposed to low-dietary soy isoflavones showed tangible body macrophages with intracytoplasmic apoptotic bodies (head arrows). C, Rats born from dams exposed to high dose of soy isoflavones showed severe lymphocyte depletion, vacuolation (arrows), and reduction in lymphatic tissue in cortex and medulla, (hematoxylin and eosin; 200×).
Discussion
The current study investigated the prenatal exposure of dietary soy isoflavones on immune response of female albino rats’ offspring through calculation of Intumesce index, determination of TNF-α, IFN-γ and IL-12 levels as well as histopathological examination of PHA injected foot pad, thymus and spleen. Studies conducted on animals have shown that effects of phytoestrogens vary depending on species, sex, routes of administration, dose and exposure time. 33 There is general agreement that soy-rich diets can be beneficial to adults, but the effect of prenatal and neonatal feeding of such compounds has been debated. 34 The basis of this controversy is that soy products generally contain isoflavones such as genistein, which can suppress cell-mediated immune function. 35,36
In the present study, the low and high doses of isoflavones ingested by the dams during pregnancy were within the same range of the doses taken by Asian population. Asian populations consumed 20 to 50 g of soy daily, which is their major source of phytoestrogens, comparable to a daily intake of 20 to 80 mg of phytoestrogens. 37 Moreover, dietary genistein (1000 or 1500 ppm) decreased cell-mediated immunity in female C57BL/6 mice while producing serum genistein concentrations in the physiological range for humans under certain nutritional conditions. 36
At week 3 of the experiment, exposure of pregnant dams to low and high-dietary soy isoflavones increased the food intake without reaching a significant level compared to control ones. However, ingestion of high-dietary soy isoflavones caused significant (P < 0.05) reduction in body weight gain of these pregnant dams. Significant reduction in body weight gain after administration of high concentration of genistein has been previously observed. 34,38,39 However, previous work stated that repeated administration of genistein and ingestion of phytoestrogens in diet by pregnant female rats had no effect on maternal body weight. 40,41 The decrease in body weight gain in spite of absence of significant changes in feed consumption may be due to loss of large amount of ingested food energy due to increased locomotor activity not tested in this study. 42 In another explanation, the decrease in body weight gain implies that the estrogenic hormone action of phytoestrogens is beneficial to body fat regulation and decrease level of leptin that is produced in adipose tissue. 43 Moreover, genistein induced lipolysis and inhibited lipid synthesis. 34
On PND 50, the significant decrease in body weight of offspring born to dams given low and high doses of soy isoflavones was interesting. This may indicate that the immune system in these pups was severely affected that reflected negatively on their body weights. In a previous study, the smaller gain in body mass of offspring may be a consequence of a systemic toxicity after isoflavones administration during the gestational period that had compromised the quantity and/or quality of the milk of the first breast feedings as well as may have influenced the metabolic system of the offspring. 38 The prenatal exposure to dietary phytoestrogens produced decrease in fetal weight at birth 39 due to crossing of dietary phytoestrogen through placenta to cause its effect on fetuses. 41
In the present study, the serum, tissue, or milk phytoestrogen were not measured in dams or offspring as the plasma half-life for daidzein and genistein has been estimated to be 7.9 hours in adults with peak plasma concentrations occurring 6 to 8 hours after ingestion of soy powder. 44
Phytohemagglutinin is a plant-derived mitogen that stimulates the recruitment of leucocytes involved in both adaptive and innate immune responses at site of injection producing a measurable tissue swelling 27,45 that is commonly used to estimate T-cell mediated immunity. Phytohemagglutinin-induced swelling is related to sensitive immune cell activity in House Sparrows. 27 There was a significant (P < 0.05) reduction in the intumesce index of ankle of females born to dams fed low and high-dietary soy isoflavones. Individual offspring maternally exposed to these estrogenic chemicals produced weaker inflammatory responses to PHA than did control individuals, suggesting that prenatal exposure to dietary soy isoflavones is immunosuppressive. This could be explained by the ability of isoflavones, especially genistein, for binding the mammalian ER-α and induce a myriad of physiological changes in mammalian models, including steroidogenesis and cell proliferation. 46 Among these changes, the reduced T-cell abundance and reduced cell-mediated immune function 14,36 were manifested here by reduced thymus weight as well as its lymphoid depletion. In vivo immunosuppressive function of high soy diet or genistein was also reported to delay the rejection of rat cardiac allografts. 47 Additionally, the results of the present work suggested a prolonged adverse effects of genistein in adult animals after cessation of exposure as the perinatal estrogen treatment can have permanent immune adverse effects in mice and humans. 48
Surprisingly female offspring, prenatally exposed to high-dietary soy isoflavones demonstrated significantly (P < 0.05) higher TNF-α level than control offspring. Dietary isoflavones incorporation by dose 20 mg/kg could increase serum TNF-α, while higher dose (250 mg/kg) reversed this effect. 49 The elevated levels of TNF-α may contribute to the elevated basal lipolysis 50 that may be inferred here by the reduced weight gain of offspring in response to maternal treatment with dietary soy isoflavones.
Interferon-γ is produced from number of cell populations of innate (NK cells, NKT cells, macrophages, and myelomonocytic cells) and adaptive immune systems (Th1 cells, cytotoxic T lymphocyte, and B cells). 21 In this study, female offspring of dams subjected to high- and low-dietary soy isoflavones revealed lower IFN-γ compared to control offspring. Interestingly, NF-κB regulates IFN-γ transcription and has been shown to interact with ERs. 51 In addition, the direct role of ER-α in regulation of IFN-γ production during the course of chronic bacterial infection and inflammation was previously investigated. 35 Another explanation for low IFN-γ concentration was that genistein-induced inhibition of prostaglandin E2 production. 52 The low serum IFN-γ concentrations in soy isoflavone maternally fed animals were consistent with reduced lymphocytes count, Intumesce index, and histopathological picture that were observed in the current study.
Interleukin -12 mediates development and maintenance of Th1 cells by inducing production of IFN-γ by Th1 and NK cells. Moreover, IL-12 indirectly activates the antimicrobial and antitumor activity of macrophages and promotes cytolytic activity of NK cells and lymphokine-activated killer cells. 22 The present study demonstrated that prenatal exposure to dietary soy isoflavones resulted in a significant (P < 0.05) reduction in IL-12 in female offspring compared to offspring of dams fed control diet. This reduction in IL-12 demonstrated the immunosuppressive effect of dietary soy isoflavones when given during the prenatal period. Current results could be attributed to inhibitory effect of dietary soy genistein on NF-κB production or prevention of NF-κB binding to DNA. 53 NF-κB plays a pivotal role in IL-12 gene expression 54,55 ; it is possible that the blockade of NF-κB signaling pathway is a molecular mechanism in the inhibition of IL-12 gene expression by dietary soy isoflavones. Moreover, soy phytoestrogens may inhibit IL-12 signaling through JAK-STAT pathway in T-cells and subsequent Th1 differentiation. 56 The phytoestrogens exposure in utero would produce lasting effect on cytokine responses in the absence of further exposure because perinatal estrogen treatment can have permanent immune adverse effects in mice and humans. 48 The critical period of exposure to genistein appears to occur during prenatal and neonatal development. Early exposure to genistein to pregnant female rats that exposed to low (5 mg/kg diet), or high (300 mg/kg diet) genistein diets throughout gestation and lactation may have organizational effects on immune systems later in life. Synthesis of IFN-γ was marginally higher among genistein-exposed than genistein free males. 57
This effect reduced the inflammatory response to PHA injection in the foot pad at the histopathological level and as a reduced intumesce index, TLC, lymphocyte, and monocyte counts. In contrary, no effect of dietary phytoestrogens on offspring IL-12 level was found. 35
In this study, high and low-dietary soy isoflavones fed to pregnant dams significantly (P < 0.05) decreased TLC, lymphocyte, and monocyte in both low and high offspring compared to control offspring. This reduction was in accordance with the immunosuppressive picture demonstrated by reduced serum IFN-γ concentrations that impeded IL-12 production. These cytokines are responsible for chemotaxis of the leukocytes to the site of inflammation induced by PHA injection. 58,59
In the present study, splenic relative weight of female offspring was not affected by maternal dietary soy isoflavones intake. The absence of differences in splenic relative weight in female offspring may be attributed to the variation in effects of dietary soy phytoestrogens depending on sex. 33 On other hand, it was reported that splenic hematopoiesis is augmented by estrogen treatment 60 which may relatively increase splenic size.
Female offspring born from dams that were fed high-dietary soy isoflavones had a significantly (P < 0.05) lowered thymus relative weight compared to control and low-dietary isoflavones fed groups. This reduction in thymus weight could be attributed to the effect of soy isoflavones on the thymus. Previous in vitro 61 and in vivo 62 reports suggested that daidzein may be immunostimulatory, and its metabolite may not be capable of producing genistein’s thymolytic effects. Additionally, genistein could interact with the thymus through ERs and non-ER-mediated mechanisms. It is not surprising that exposure to dietary isoflavones mimics estrogen’s hormone action as it acts as estradiol, and the estrogen treatment was reported to induce thymic atrophy and immune suppression in developing rodents. 63,64
Histopathologically, spleens of offspring born to dams fed low and high doses of dietary soy isoflavones showed lymphocyte depletion in white pulp. Dams fed high dose of soy isoflavones had severe lymphocyte depletion, vacuolation, and reduction in lymphatic tissue in cortex and medulla. These results could be explained by the effect of soy isoflavones, especially genistein, that could interact with thymic ERs; ERα and ER β 14,64 during the prenatal period where normal thymic development is dependent on E2/ ERα signaling pathway. 65 Moreover, soy isoflavones could affect ERs that are present in splenic CD8+ cells, 66,67 monocytes B-cells, and T-cells. 68 The decrease in these cell populations would explain the lower concentrations of IFN-γ and IL-12 in offspring of dams fed soy isoflavones. 51,69
In conclusion, taking together all data, it was clear that dietary intake of soy isoflavones during the prenatal period had immunosuppressive effect on female offspring after PHA stimulation. This effect was mediated by reduced IFN-γ that affected the IL-12 production pathway, and the depletion in thymus that manifested by reduction in their weights. All these changes were reflected in the reduced inflammatory and immune response to PHA stimulation in foot pad (reduced Intumesce index and microscopic inflammatory reaction in foot pad).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
