Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) rereviewed the safety of 28 ethanolamides and found them safe in the present practices of use and concentration when they are formulated to be nonirritating, and that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed. Most of the ethanolamides are reported to function in cosmetics as hair-conditioning agents, skin-conditioning agents, and surfactant—foam boosters. The Panel reviewed available animal and clinical data, as well as information from previous CIR reports.
Keywords
Introduction
This safety assessment reviews the safety of isostearamide monoethanolamine (MEA), myristamide MEA, and stearamide MEA, as used in cosmetics. The following 25 ingredients, which are secondary carboxamides comprising the amidation products of alkyl carboxylic acids and ethanolamines, are also included in this safety assessment, for a total of 28 ingredients defined in the International Cosmetic Ingredient Dictionary and Handbook as ingredients: acetamide MEA, azelamide MEA, babassuamide MEA, behenamide MEA, C16-22 acid amide MEA, cocamide MEA, cocamide methyl MEA, cocamidopropyl betainamide MEA chloride, hydroxystearamide MEA, lactamide MEA, lauramide MEA, linoleamide MEA, oatamide MEA, oleamide MEA, oliveamide MEA, palm kernelamide MEA, palmamide MEA, palmitamide MEA, pantothenamide MEA, peanutamide MEA, ricinoleamide MEA, sunfloweramide MEA, tallowamide MEA, trideceth-2 carboxamide MEA, and undecylenamide MEA,
Most of the ethanolamides are reported to function in cosmetics as hair-conditioning agents, skin-conditioning agents, and surfactant—foam boosters.
Several ingredients on this list have previously been reviewed by the Cosmetic Ingredient Review (CIR). In 1995, CIR published a safety of isostearamide MEA, myristamide MEA, and stearamide MEA 1 and concluded that these 3 ingredients were safe for use in rinse-off products. In leave-on products, these ingredients were safe for use at concentrations that will limit the release of free ethanolamine to 5% but with a maximum use concentration of 17% for isostearamide, myristamide, and stearamide MEA. In that assessment, test data were used to establish limits for use concentrations because concentration of use data was not available. The report further stated that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
In 1993, the Panel concluded that acetamide MEA is safe as used as a cosmetic ingredient at concentrations not to exceed 7.5% in leave-on products, based on sensitization test data, and is safe in the present practices of use in rinse-off products; cosmetic formulations containing acetamide MEA should not contain nitrosating agents or significant amounts of free acetamide. 2 In that assessment, acetamide MEA was reported to be used at up to 25% in rinse-off products. This conclusion was reaffirmed. 3 Cocamide MEA was reviewed in 1999; the Panel concluded that cocamide MEA is safe as used in rinse-off cosmetic products and safe at concentrations of up to 10% in leave-on products. 4 Cocamide MEA was reported to be used at up to 25% in 1984, but the types of products, that is, rinse-off or leave-on, were not specified. The Panel also noted that cocamide MEA should not be used as an ingredient in cosmetic products containing N-nitrosating agents or in product formulations intended to be aerosolized. The leave-on concentration limit was derived from the concentration found safe at the time for cocamide diethanolamine (DEA); the inhalation caveat was based on concerns of inhalation toxicity of ethanolamine. These conclusions will be superseded by the conclusion reached in this safety assessment.
The structures and definitions of the ethanolamides are provided in Table 1. The conclusions of the previous reports, as well as those for relevant acids that have been previously reviewed by CIR, are provided in Table 2.
Definitions and Structures.

Abbreviations: aq, aqueous; CAS, Chemical Abstracts Service; cond ag, conditioning agent; MEA, monoethanolamine; surf, surfactant.
Conclusions of Previously Reviewed Ingredients and Constituents.

Abbreviation: MEA, monoethanolamine.
The data in this report apply to the safety of the 3 ingredients previously reviewed and the additional 25 ingredients that are secondary carboxamides, consisting of the amidation products of alkyl carboxylic acids and ethanolamine.
Chemistry
The ethanolamides consist of covalent, secondary amides, whereby one of the nitrogen substituents is ethanol and the second is a carbonyl attached substituent. For example, myristamide MEA is a secondary amide wherein one of the nitrogen substituents is ethanol and the second is a 14 carbon, carbonyl attached chain (Figure 1). These ingredients are not salts and do not readily dissociate in water. However, amidases, such as fatty acid amide hydrolase (FAAH) which is known to be present in human skin, could potentially convert these amides to ethanolamine and the corresponding fatty acids. 5 -7 Secondary amides do tend to react with nitrosating agents to form nitrosamides. Chemical and physical properties of ethanolamides are summarized in Table 3.
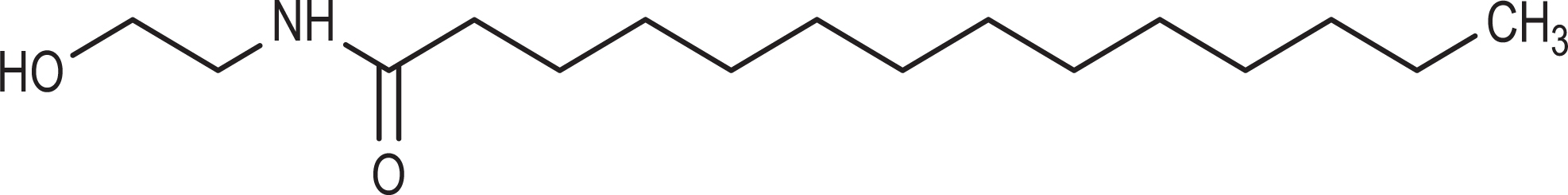
Myristamide MEA.
Physical and Chemical Properties.
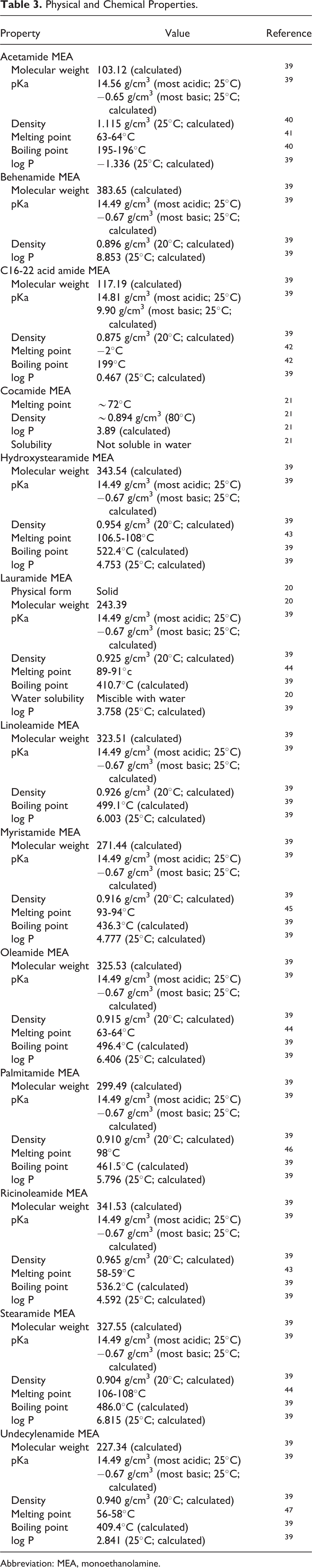
Abbreviation: MEA, monoethanolamine.
Method of Manufacture
Ethanolamine reacts with long-chain fatty acid esters in a 1:1 mole ratio to produce a 90+% pure, crystalline ethanolamide mixture. 8 Two different routes of synthesis of ethanolamides are common: direct acylation with free fatty acids and transacylation using fatty acid esters (often enzymatically). 9,10 Ethanolamides can be produced via enzymatic amidification; monoacylated ethanolamine can be isolated from the reaction mixture. The reaction is carried out using an equimolar ratio of fatty acid and ethanolamine. The enzyme is filtered upon completion of the reaction, and the product is dissolved in a mixture of methanol and chloroform. The solvent is then eliminated by evaporation, and the resulting solid is the amide.
Acetamide MEA
Acetamide MEA is prepared by the reaction of acetic acid with ethanolamine. Additional methods of production, involving acetamide and ethylene oxide, ethanolamine, and acetyl chloride, have been reported. 2
Impurities
Acetamide MEA
Analysis of 4 lots of acetamide MEA by gas chromatography-mass spectrometry indicated the presence of 0.0006% to 0.0029% ethanolamine and 0.0006% to 0.0030% acetamide. Using high-performance liquid chromatography, N-nitrosodiethanolamine was not detected (limit of detection = 0.05 ppm) in acetamide MEA. 2
Myristamide MEA
The maximum free amine content of myristamide MEA is 1.5%. 1
Stearamide MEA
Stearamide MEA has 0.8% maximum free fatty acids (as stearic acid), 0.5% to 2.0% free amine (as ethanolamine), and 54.0% to 58.0% total fatty acids (as stearic acid). 1
Use
Cosmetic
Most of the ethanolamides are reported to function in cosmetics as hair-conditioning agents, skin-conditioning agents, and surfactant—foam boosters; a few are reported to have other uses. 11 The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2011 report that stearamide MEA is used in 10 cosmetic formulations, myristamide MEA is used in 1 formulation, and isostearamide MEA does not have any uses reported. 12 Other ethanolamides included in this safety assessment have many more reported uses. For example, cocamide MEA has the highest frequency of use, with 1122 reported uses; only 33 of those uses are in leave-on products. Trideceth-2 carboxamide MEA has 189 reported uses, and acetamide MEA is reported to be used in 148 formulations.
A Personal Care Products Council (Council) survey of the maximum reported use concentrations found that cocamide MEA is reported to be used at up to 18% in rinse-off formulations and at up to 5% in leave-on formulations. 13 Stearamide MEA has the highest concentration of use in leave-on formulations at up to 15%. The use information for the ethanolamides is provided in Table 4. In some cases, reports of uses were received by the VCRP, but no concentration of use data was available. For example, acetamide MEA is reported to be used in a nail formulation, but no use concentration was available. In other cases, no reported uses were received in the VCRP, but a use concentration was provided in the industry survey. For example, lauramide MEA was not reported in the VCRP to be used in baby product formulations, but the industry survey indicated that it was used in such products at 0.5%. It should be presumed that lauramide MEA is used in at least 1 baby product. Ethanolamides that are not reported to be in use, according to VCRP data and the Council survey, are listed in Table 5.
Frequency and Concentration of Use According to the Duration and Type of Exposure.
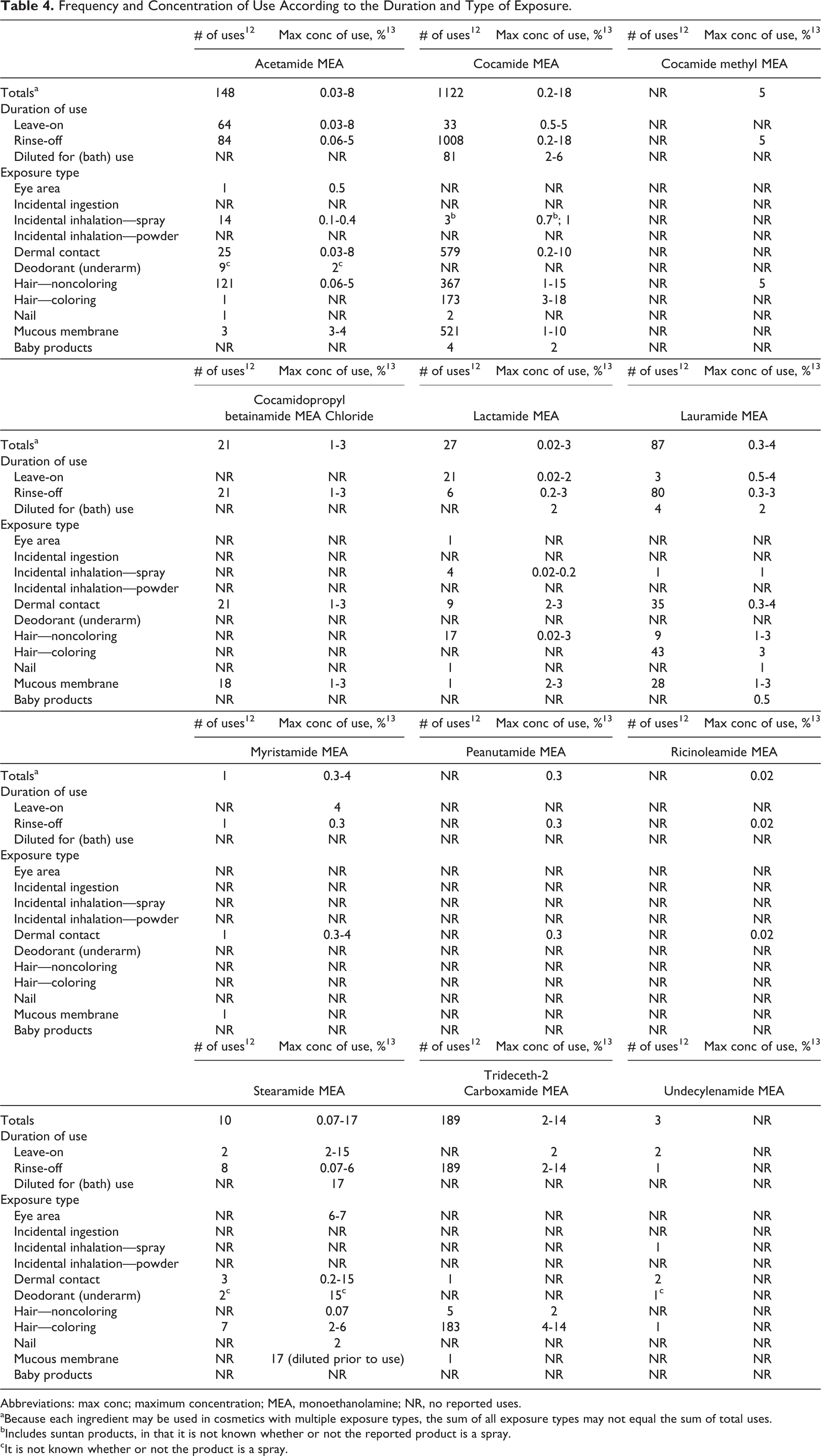
Abbreviations: max conc; maximum concentration; MEA, monoethanolamine; NR, no reported uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes suntan products, in that it is not known whether or not the reported product is a spray.
cIt is not known whether or not the product is a spray.
Not Reported to be in Use.

Abbreviation: MEA, monoethanolamine.
A few of the ethanolamides may be used in products applied to baby skin or used near the eye area or mucous membranes. Additionally, some of the ethanolamides are used in cosmetic sprays, with reported maximum use concentrations of 0.4% acetamide MEA in aerosol hair sprays and 1% cocamide MEA and lauramide MEA in foot sprays. 13 These products could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 14 -17 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 14,16
Acetamide MEA, stearamide MEA, and undecylenamide MEA are reported to be used in deodorants, and stearamide MEA has the highest reported concentration of use in deodorants at 15%. It is not known whether or not these products are sprayed. There is some evidence indicating that, generally, deodorant spray products, compared with hair sprays, can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 14 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Monoalkylamines, monoalkanolamines, and their salts are listed by the European Commission in Annex III Part 1: the list of substances which cosmetic products must not contain, except subject to the restrictions and conditions laid down. 18 These ingredients are allowed a maximum secondary amine content of 0.5% in finished product, are not to be used with nitrosating agents, and must have a minimum purity of 99%. The maximum secondary amine content of 0.5% is allowed for raw materials, maximum nitrosamine content allowed is 50 µg/kg, and the chemicals must be kept in nitrite-free containers. Acetamide MEA and babassuamide MEA, 2 ethanolamides included in this safety assessment, are listed in this category. All the other ethanolamides are listed in the European Union inventory.
Toxicokinetics
No published toxicokinetic data were discovered, and no unpublished data were submitted.
Toxicological Studies
Acute (Single) Dose Toxicity
Dermal
Acetamide MEA
No deaths occurred when rabbits were dosed dermally with 20 mL/kg acetamide MEA. 2
Cocamide MEA
The dermal median lethal dose (LD50) of cocamide MEA was evaluated by applying 2 g/kg to the abraded and intact skin of rabbit under occlusive patches. 19,20 The dermal LD50 in rabbits was >2 g/kg.
Oral
Cocamide MEA
The acute oral toxicity of cocamide MEA (purity not specified) was evaluated in a number of studies using rats. 19 -21 In most studies, the highest dose administered was 5 g/kg or 5 mL/kg; the LD50 was greater than this dose. In a study in which doses of 1 to 32 g/kg were used, the LD50 was reported to be 7.4 g/kg in rats. 19,20 In 2 other rat studies, the oral LD50 of cocamide MEA was identified as 3.3 g/kg and >3.125 g/kg, respectively. 21 In studies in mice, the oral LD50 of cocamide MEA was >10 g/kg in most studies. However, a value of 3.125 g/kg was reported in 1 study using mice. (Details were not provided.)
Acetamide MEA
The oral LD50 of acetamide MEA has been reported as 26.95 and as 27.66 g/kg in rats. The acute LD50 of 2 hair products containing 1.3% acetamide MEA was >16.9 g/kg for 1 product and >25 mL/kg for the other; these were the highest doses tested. 2
Lauramide MEA
The oral LD50 of lauramide MEA was >2 g/kg in rats. 22
Repeated Dose Toxicity
Dermal
Acetamide MEA
In a 13-week study, a hair product containing 1.3% acetamide MEA, applied as an aqueous (aq) solution that was diluted to 50% (w/v), was not toxic to rabbits. Slight-to-moderate erythema was observed sporadically at the application site from days 44 to 84 of the study. 2
Cocamide MEA
Cocamide MEA, 25% in olive oil, was applied to mice twice a day for 1 week and the application site was not covered. 21 No dermal reactions were observed. Additional details were not provided.
Stearamide MEA
The dermal toxicity of a formulation containing 17.0% stearamide MEA was evaluated in a 4-week study in rabbits; 2 g/kg of a 10% aq solution of the formulation was applied to intact and abraded skin; no gross or microscopic lesions were observed. In a 13-week dermal study, a formulation containing 5.27% stearamide MEA was not toxic in rats. 1
Oral
Cocamide MEA
Groups of 10 male and 10 female Wistar rats were dosed by gavage with 0, 70, 250, or 750 mg/kg body weight (bw)/d cocamide MEA in olive oil; the high dose was increased to 1500 mg/kg bw after 14 days. 19 The animals were dosed once daily, 5×/wk, for 4 weeks. Recovery groups of 5 male and 5 female rats per dose level were included. None of the animals died, and no significant test article-related gross or microscopic lesions were observed. The no observed adverse effect level (NOAEL) was >750 mg/kg bw/d.
Reproductive and Developmental Toxicity
Data on the reproductive and developmental toxicity of the ethanolamides were not found. Since ethanolamine may be present as an impurity in the ethanolamides, and since amidases in the skin could possibly convert some of the ethanolamide to ethanolamine and the corresponding carboxylic acid, a summary of available data from the reports on ethanolamine and the constituents of these ethanolamides is provided subsequently.
MEA: In a dietary study, 0 to 7800 ppm of a composite hair dye and base containing 22% ethanolamine was fed to 60 gravid female rats on days 6 to 15 of gestation, and the rats were killed on day 19 of gestation. No developmental effects were observed. In another dietary study, 30 male rats were fed 0 to 7800 ppm of the hair dye for 8 weeks prior to mating with female rats that were being fed a basal diet, while a group of 60 female rats were fed 0 to 7800 ppm of the test substance for 8 weeks prior to mating with male rats being fed a basal diet. No effects on reproduction or fertility were observed. In rabbits, no developmental effects were observed when pregnant females were dosed by gavage on days 6 to 18 of gestation with 0 to 19.5 mL/kg/d of the hair dye containing 22% ethanolamine.
23
Arachis hypogaea (peanut) oil: Peanut oil was used as the vehicle in a fertility study in rats. It was administered orally for 28 days prior to mating and for 6 days during mating. No unusual findings were noted in the vehicle group. No unusual findings were observed in a teratogenicity study in which rats were injected with a test article, and peanut oil was used as the vehicle. Treatment-related changes were not observed in a study in which rabbits were dosed intramuscularly with 21% peanut oil on day 8 of gestation. However, in a study of the effects of oil vehicles on early embryonic lethality in mice, it was stated that plant oils proved to be unsuitable carriers of test mutagens in female dominant-lethal studies where the route of administration is via the peritoneal cavity.
24
Hydroxystearic acid: The dermal teratogenicity of 2 formulations containing 7% hydroxystearic acid was evaluated using female rats. No teratogenic effects were observed. (However, dermal irritation was reported.)
25
Palm oil: Crude palm oil was not a reproductive toxicant in a study in which male and female Wistar/NIN inbred weanling rats were fed a diet containing this ingredient (10%) prior to mating. Mean litter sizes were comparable between test and control groups. No significant changes were found in liver or kidney weight in adult animals. Neither untreated palm oil (15%) nor 15% heated palm oil in the diet induced anomalies with respect to fertility and in utero growth when fed to male and female Sprague-Dawley SPF rats prior to mating. In a study investigating the effects of palm oil on sexual maturation and endocrine function, vaginal opening was observed significantly earlier (compared to 5% corn oil control) in weanling rats fed 20% palm oil in the diet. No significant differences were observed in endocrine function as determined by measuring estradiol, prolactin, and luteinizing hormone.
26
Palm kernel oil: In the second generation resulting from the mating of adult Mongolian gerbils fed diet containing 8.75% (w/w) palm kernel oil, no statistically significant differences were found with respect to the following: frequency of litters, mean litter size, total of newborns, and suckling death. Animals receiving a basal diet served as the control group.
26
Genotoxicity
In Vitro
Acetamide MEA
Acetamide MEA, ≤5000 µg/plate, was not mutagenic in the presence or absence of metabolic activation in an Ames test, and ≤5000 µg/mL acetamide MEA did not induce unscheduled DNA synthesis in primary rat hepatocytes. 2
Cocamide MEA
In an Ames test, 50 µg/plate cocamide MEA was mutagenic in Salmonella typhimurium TA100 with metabolic activation; it was not mutagenic with S typhimurium TA98, TA1535, TA1537, or TA1538, with or without metabolic activation or in TA100 without metabolic activation. Cocamide MEA was not mutagenic in a plate incorporation assay. 4
The mutagenic potential of cocamide MEA was evaluated in an Ames test using S typhimurium TA1535, TA1537, TA1538, TA98, and TA100, with and without metabolic activation. 19,20 Cocamide MEA, evaluated at doses of 4 to 2500 μg/plate, was not mutagenic. (No data for controls were provided.)
Lauramide MEA
The mutagenic potential of lauramide MEA was evaluated in an Ames test with S typhimurium TA98, TA100, TA1535, TA1537, and TA1538 at doses of 33 to 3333 μg/plate, with and without metabolic activation. 27 Doses of 3.3 and 10 μg/plate were tested with TA1537 without metabolic activation, and a dose of 10 μg/plate was tested in strains TA100 without metabolic activation. Negative controls were used and gave expected results. Lauramide MEA was not mutagenic in this assay.
Carcinogenicity
Data on the carcinogenicity of the ethanolamides included in this report were not found in the published literature, and no unpublished data were provided. Since ethanolamine may be present as an impurity in the ethanolamides, and since amidases in the skin could possibly convert some of the ethanolamide to ethanolamine and the corresponding carboxylic acid, a summary of available data from the reports on constituents of these ethanolamides is provided.
Cocamidopropyl betaine: The carcinogenic potential of a nonoxidative hair dye formulation containing 0.09% active cocamidopropyl betaine was determined in Swiss Webster mice. A dose of 0.05 mL per mouse was applied 3 times weekly for 20 months to interscapular skin that was clipped free of hair. Dermal changes were noted, but the incidence of neoplasms in treated animals did not differ significantly from control groups.
28
Hydroxystearic acid: In an 18-month subcutaneous carcinogenicity study in female Swiss-Webster mice, hydroxystearic acid was injected subcutaneously twice weekly for a total dose of 4 or 80 mg delivered in a total of 8 mL tricaprylin. Hydroxystearic acid was classified as tentatively carcinogenic in Swiss-Webster mice. Subcutaneous sarcomas were observed at the site of injection in 9 of the 28 mice (14 per dose group) that were alive at 6 months. All of the sarcomas were observed in the low-dose group. In a second study in which 9 A/He male mice received a total intraperitoneal dose of 60 mg hydroxystearic acid over a period of 4 weeks, the frequency of lung tumors was within the spontaneous occurrence.
25
Lactic acid: Female rabbits were dosed by gavage with 0.1 to 0.2 g/kg lactic acid in 100 to 150 mL water twice daily for 5 months, and 5 female rabbits were dosed orally with 0.1 to 0.7 g/kg lactic acid in 50 to 100 mL water twice daily for 16 months. No tumors were reported.
29
Lauric acid: Treatment of mice with repeated subcutaneous injections of 25 and 50 mg lauric acid was not carcinogenic.
30
Oleic acid: Intestinal and gastric tumors were found in mice receiving dietary oleic acid at daily concentrations of up to 200 mg/mouse. No malignant tumors were induced by repeated subcutaneous injections of 1 to 16.5 mg oleic acid in 2 species of mice.
30
Palmitic acid: Feeding of 50 g/kg/d palmitic acid to rats resulted in lipogranulomas observed in fat associated with the testis or ovary; this lesion was reversible upon diet substitution and attributed to dietary imbalance. Low incidences of carcinomas, sarcomas, and lymphomas were observed in mice receiving single or repeated subcutaneous injections of 25 and 50 mg palmitic acid.
30
Ricinoleic acid: None of 20 mice injected intravaginally with 2% ricinoleic acid (in gum tragacanth) had neoplasms or hyperplastic lesions of the corpus uteri, cervix uteri, vagina, or perineal skin. However, benign lung adenomas were observed in 10 of the 13 mice dosed with ricinoleic acid and in 6 of the 24 vehicle control mice that were killed after the 14th month of dosing.
31
Stearic acid: Feeding of up to 50 g/kg/d dietary stearic acid to mice was not carcinogenic. Low incidences of carcinomas, sarcomas, and lymphomas were observed in mice receiving single or repeated subcutaneous injections of ≤82 mg stearic acid.
30
Irritation and Sensitization
Irritation
Dermal
Nonhuman
Acetamide MEA
Acetamide MEA, applied neat, was a mild skin irritant in an open-patch Draize test in 12 rabbits. In another study, acetamide MEA (70% active, minimum) was not a primary irritant when applied to the intact and abraded skin of rabbits using a 24-hour occlusive patch. 2
Cocamide MEA
The irritancy potential of 50% cocamide MEA in petrolatum was evaluated in a 24-hour patch test in guinea pigs, rabbits, and hairless mice. Cocamide MEA was slightly irritating to rabbits but was not irritating to guinea pigs and hairless mice. 4
The irritation potential of cocamide MEA was evaluated in rabbits. 21 Cocamide MEA, 25%, was not irritating to rabbits when applied under an occlusive patch for 24 hours. (Additional details were not provided.)
Lauramide MEA
In a Draize study, lauramide MEA was not irritating to rabbit skin. 22 No details were provided.
Stearamide MEA
The primary irritation index of a formulation containing 17.0% stearamide MEA was 1.00/8 in a group of 3 rabbits. 1
Human
Irritation data were available from earlier CIR safety assessments indicate that acetamide MEA and cocamide MEA were not irritating; however, a 1% aq solution of a formulation containing 17% stearamide MEA produced questionable to mild reactions in 10 of the 19 subjects in the study. 1,2,4 In a 21-day, 14-subject, cumulative irritation study in which 0.2 mL of a formulation containing 5.0% stearamide MEA was applied using occlusive patches, slight irritation was observed, with a composite total score of 156/882.
Sensitization
Nonhuman
Acetamide MEA
Acetamide MEA was not a sensitizer in a maximization study using 10 guinea pigs. Induction included an intradermal injection of 5.0% acetamide MEA in propylene glycol and in Freund’s complete adjuvant and a topical application of 10% acetamide MEA. 2
Cocamide MEA. Cocamide MEA was not a sensitizer in a guinea pig maximization study. Details were not provided. 21
Human
Acetamide MEA
In earlier CIR safety assessments that support the safety of ethanolamides, aq solution of 7.5% acetamide MEA did not cause primary irritation or sensitization in a 50-subject study, however, a repeated insult patch test (RIPT) of a hair product containing 1.3% acetamide MEA showed mild reactions during induction and challenge. However, the researchers concluded that a hair product containing 1.3% acetamide MEA was not a sensitizer. 2
Stearamide MEA
In an RIPT using 100 subjects in which 0.1 mL of formulation containing 5.27% stearamide MEA was applied using occlusive patches, the formulation did not produce sensitization. 1
Ocular Irritation
Nonhuman
Irritation data from earlier CIR safety assessments show that 1.3% acetamide MEA and 5.27% stearamide MEA were not irritating to rabbit eyes however 70% minimal activity of acetamide MEA was practically nonirritating and 17% stearamide MEA was reported to be minimally to moderately irritating. 1,2
Lauramide MEA
In a Draize ocular irritation study, Lauramide MEA was highly irritating to rabbit eyes. 22 Details were not provided.
Summary
This report assesses the safety of 28 ethanolamides. This safety assessment originated as a rereview of isostearamide MEA, myristamide MEA, and stearamide MEA and was expanded to include 25 additional related ingredients. Some of these ingredients have been previously reviewed by the CIR and are included because they fit scientifically and conceptually within this family of ingredients.
Amidases, such as FAAH, which is known to be present in human skin, could potentially convert the ethanolamides to ethanolamine and the corresponding fatty acids. The yield of ethanolamine from metabolism of ethanolamides in human skin is unknown. Also, secondary amides do tend to react with nitrosating agents to form nitrosamides. Impurity data were available for acetamide, myristamide, and stearamide MEA: acetamide MEA contained up to 0.0029% ethanolamine, 0.0030% acetamide, and no N-nitrosodiethanolamine; myristamide contained a maximum of 1.5% ethanolamine; and stearamide MEA contained up to 2.0% free amine (as ethanolamine).
Most of the ethanolamides are reported to function in cosmetics as hair-conditioning agents, skin-conditioning agents, and surfactant—foam boosters; a few are reported to have other uses. In 2011, stearamide MEA was reported to have only 10 uses, myristamide MEA had 1, and isostearamide had none. Cocamide MEA has the highest frequency of use, with 1122 reported uses; only 33 of those are in leave-on products. Cocamide MEA is reported to be used at up to 18% in rinse-off formulations and at up to 5% in leave-on formulations. Stearamide MEA has the highest concentration of use in leave-on formulations at up to 15%. In Europe, monoalkylamines, monoalkanolamines, and their salts are on the list of substances that must not form part of the composition of cosmetic products, except subject to restrictions and conditions laid down. Acetamide MEA and babassuamide MEA are included in this list. These restrictions include a maximum secondary amines contaminant content of 0.5% in finished products, a maximum secondary amines content of 0.5% in raw materials, and a maximum nitrosamine content of 50 µg/kg.
In an acute dermal study in rabbits, the LD50 for cocamide MEA was >2 g/kg and for acetamide MEA, it was >20 mL/kg. In oral studies, the LD50 of cocamide MEA was >5 g/kg in rats and >10 g/kg in mice. The oral LD50 values in rats for acetamide and lauramide MEA were 27 and >2 g/kg, respectively. In repeated dose dermal studies, acetamide MEA (50%; 13 weeks in rabbits), cocamide MEA (25%; 1 week in mice), and stearamide MEA (10% solution of a formulation containing 17% in rabbits, applied for 4 weeks; 5.27% in rats, applied for 13 weeks) were not toxic. In a 14-day oral study, the NOAEL of cocamide MEA in rats was >750 mg/kg/d, the highest dose tested.
No data on the reproductive and developmental toxicity or carcinogenicity of the ethanolamides included in this report were found. Available data from previous CIR reports on ethanolamine and some of the constituents were summarized, and no relevant toxic effects were noted.
Acetamide MEA (≤5000 µg/plate), cocamide MEA (≤2500 μg/plate), and lauramide MEA (≤3333 μg/plate) were not mutagenic in Ames tests with or without metabolic activation. Acetamide MEA did not induce unscheduled DNA synthesis in primary rat hepatocytes.
Acetamide MEA was a mild skin irritant in rabbits and in humans, and a formulation containing 0.5% acetamide MEA was not an irritant. Cocamide MEA was slightly irritating to rabbit skin and was not irritating to guinea pigs and hairless mice. In clinical testing, 50% cocamide MEA in petrolatum was not irritating. A formulation containing 17% stearamide MEA had a primary irritation score of 1/8 in rabbits, and in a clinical single-insult occlusive patch test, a 1% aq solution of this formulation produced questionable reactions in 7 and mild reactions in 3 of 19 subjects; in a clinical cumulative irritation study, 5% stearamide MEA produced slight irritation. Acetamide MEA and cocamide MEA were not sensitizers in guinea pigs. In clinical testing, a solution of 7.5% acetamide MEA, a formulation containing 1.3% acetamide MEA, and a formulation containing 5.27% stearamide MEA were not sensitizers.
Acetamide MEA (70% minimum activity) was practically nonirritating to rabbit eyes, and formulations containing 1.3% acetamide MEA and 5.27% stearamide MEA were not irritating to rabbit eyes. A formulation containing 17% stearamide MEA was a moderate ocular irritant, and lauramide MEA was highly irritating to rabbit eyes.
Discussion
Isostearamide MEA, myristamide MEA, and stearamide MEA, along with acetamide MEA and cocamide MEA, have been reviewed previously by the CIR Expert Panel. This amended safety assessment includes those 5 ingredients and 23 additional ethanolamides that are also secondary carboxamides comprising the amidation products of alkyl carboxylic acids and ethanolamine. This amended safety assessment was originated as a rereview of isostearamide MEA, myristamide MEA, and stearamide MEA and was expanded to include the additional ethanolamides, the safety of which was supported by the data available in the original safety assessment and by other published and unpublished studies.
The Panel noted gaps in the available safety data for other end points for many of the ethanolamides included in this group. Because these ingredients are secondary amides, whereby one of the nitrogen substituents is ethanol and the second is a carbonyl attached substituent, their chemical structures are similar, their structure–activity relationships are expected to be similar, and their functions in cosmetics are similar, supporting the use of the available data to support the safety of all the ethanolamides included in this safety assessment.
Published toxicokinetics data are lacking, as are reproductive and developmental toxicity and carcinogenicity data. However, the Panel was of the opinion that dermal penetration would be limited based on the size and the lipophilicity of these ethanolamides. Based on the totality of the data set, the Panel did not think these ingredients would have reproductive or carcinogenic effects.
Also, while it is a metabolic possibility that amidases present in human skin could potentially convert the ethanolamides to ethanolamine and the corresponding acid, such potential amidase activity would at most cleave a small fraction of the applied ethanolamides. If these ethanolamides were cleaved into ethanolamine and the respective acids, the substantial data available on ethanolamine and other constituents indicate that these components do not have reproductive or carcinogenic effects.
Because it could be possible that DEA may exist as an impurity, the Panel reiterated its discussion regarding the positive findings reported in a dermal carcinogenicity study of DEA. The hepatocarcinogenicity that was reported in mice was considered to have little relevance to the safety of DEA in personal care products. Additionally, renal lesions reported in mice could have been the result of DEA-induced choline deficiency, a mechanism that has little relevance in humans. If DEA-induced choline deficiency was not the cause of the renal lesions, it was thought there was still no carcinogenic risk to humans because DEA does not appear to penetrate human skin to any significant extent at concentrations relevant to human exposures from the use of personal care products.
The ethanolamides consist of covalent, secondary amides. The Panel was concerned that secondary amides tend to react with nitrosating agents to form nitrosamides. Because of the potential for this process to occur, ethanolamides should not be used in cosmetic products in which N-nitroso compounds may be formed.
The potential exists for dermal irritation with the use of products formulated using ethanolamides. The Panel specified that products containing ethanolamides must be formulated to be nonirritating. Test data indicate that these ingredients are not sensitizers.
Because the ethanolamides can be used in products that may be aerosolized, including aerosol hair sprays and foot sprays, the Panel discussed the issue of incidental inhalation exposure. In the absence of inhalation data, the Panel noted that there was lack of systemic toxicity in dermal and oral single and repeated dose toxicity studies, negative results for genotoxicity in Ames tests and an unscheduled DNA synthesis assay, and little to no irritation potential and no sensitization potential. Further, this ingredient is reportedly used at concentrations of ≤1% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. However, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. Inhaled droplets/particles deposited in the nasopharyngeal and bronchial regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. Nevertheless, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
Conclusion
The CIR Expert Panel concluded that the ethanolamides listed subsequently are safe in the present practices of use and concentration described in this safety assessment when formulated to be nonirritating. The Expert Panel cautioned that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed. acetamide MEA, azelamide MEA*, babassuamide MEA*, behenamide MEA*, C16-22 acid amide MEA*, cocamide MEA, cocamide methyl MEA, cocamidopropyl betainamide MEA chloride, hydroxystearamide MEA*, isostearamide MEA*, lactamide MEA, lauramide MEA, linoleamide MEA*, myristamide MEA, oatamide MEA*, oleamide MEA*, oliveamide MEA*, palm kernelamide MEA*, palmamide MEA*, palmitamide MEA*, pantothenamide MEA*, peanutamide MEA, ricinoleamide MEA, stearamide MEA, sunfloweramide MEA*, tallowamide MEA*, trideceth-2 carboxamide MEA, and undecylenamide MEA,
Were the ingredients not in current use (as indicated by *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
M. M. Fiume contributed to conception and design; acquisition, analysis, and interpretation; and drafted the article. B. A. Heldreth contributed to conception and design; acquisition, analysis, and interpretation; drafted the article; and critically revised the article. Gill, L., W. F. Bergfeld, D. V. Belsito, R. A. Hill, C. D. Klaassen, D. C. Liebler, J. G. Marks, T. J. Slaga, and P. W. Snyder contributed to conception and design, analysis and interpretation, and critically revised the article. R. C. Shank contributed to conception and design and analysis and interpretation. Former CIR Director F. Alan Anderson contributed to conception and design, analysis, and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Conflict of interest: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
