Abstract
In the present study, the neuroprotective effect of 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1), a natural flavone, was investigated in comparison with another flavone, 5,7,4′-trihydroxyflavone (flavone 2) on the hippocampus of amyloid beta (Aβ)-injected rats. Rats were treated with the 2 flavones (1 mg/kg/d) for 1 week before Aβ injection. Seven days after Aβ administration, memory function of rats was assessed in a passive avoidance test (PAT). Changes in the levels of mitochondrial transcription factor A (TFAM), peroxisome proliferator-activated receptor γ coactivator 1 α (PGC-1α), phospho-adenosine monophosphate (AMP)-activated protein kinase (pAMPK), AMPK, phospho-cAMP-responsive element-binding protein (CREB), CREB, and nuclear respiratory factor 1 (NRF-1) proteins were determined by Western blot analysis. Our results showed an improvement in memory in rats pretreated with flavonoids. At the molecular level, phosphorylation of CREB, known as the master modulator of memory processes, increased. On the other hand, the level of mitochondrial biogenesis factors, PGC-1α and its downstream molecules NRF-1 and TFAM significantly increased by dietary administration of 2 flavones. In addition, flavone 1 and flavone 2 prevented mitochondrial swelling and mitochondrial membrane potential reduction. Our results provided evidence that flavone 1 is more effective than flavone 2 presumably due to its O-methylated groups. In conclusion, it seems that in addition to classical antioxidant effect, flavones exert part of their protective effects through mitochondrial biogenesis.
Keywords
Introduction
Alzheimer’s disease (AD) is the most common type of dementia that affects the middle- to old-aged individuals. 1 The rate of AD has increased to approximately 1 in 4 persons older than the age of 85. 2 Although senile plaques and neurofibrillary tangles are the main pathological hallmarks of AD, 3 oxidative stress and mitochondrial dysfunction play an important role in progression of disease. 4 The brain is particularly vulnerable to mitochondrial dysfunction, which may contribute to age-dependent pathological changes. 5 Alterations in mitochondrial efficiency are mainly linked to changes in mitochondrial mass and activities of respiratory enzymes. 6 –9 A decrease in electron transport chain (ETC) function or mitochondrial content leads to a dramatic decrease in energy production and is accompanied by detrimental side effects such as reduction in mitochondrial membrane potential (Δψm). 10 Furthermore, it has been suggested that, ETC impairment promotes reactive oxygen species (ROS) production in AD. 11 As increased ROS leads to alterations in mitochondrial morphology, dynamics, and subsequent cell death, 12 –15 the continuous renewal of mitochondria is important to maintain a healthy mitochondria population with age. It has been demonstrated that the capacity for mitochondrial biogenesis reduces with age, and this is a crucial factor involved in age-related mitochondrial dysfunction. 16,17
At the present, there is no effective therapy against AD, and all the drugs used in patients with AD provide only symptomatic treatment and do not change disease course. In the recent years, polyphenols have attracted considerable attention, especially in age-related and chronic diseases such as AD. 18,19 Among dietary polyphenols, flovonoids are the most plentiful. 20 Flavonoids may increase ability to fight oxidative stress by activation of cell survival signaling pathways. 21 Some studies indicated that flavonoids can activate the transcriptional factor cyclic adenosine monophosphate (cAMP)-responsive element-binding protein (CREB), resulting in the expression of prosurvival genes such as peroxisome proliferator-activated receptor γ (PPARγ) coactivator 1α (PGC-1α), as a potent stimulator of mitochondrial biogenesis and respiration. 21 –23 There is considerable evidence for neuroprotective effect of different subtypes of flavonoids through stimulation of CREB pathway. 24,25
Among flavonoids, 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1) and 5,7,4′-trihydroxyflavone (flavone 2) have been demonstrated as antioxidant, 26,27 anti-inflammatory, 28,29 and neuroprotective agents. 30 Previous assessments indicated that flavone 2 exerts its neuroprotective effect against Aβ neurotoxicity in an antioxidant independent manner by caspase inactivation and mitochondrial protection. 30,31 However, there is less information about the effects of flavone 1 on mitochondrial function.
In this study, we investigated the neuroprotective effects of flavone 1 in comparison with structurally different flavone, flavone 2 (Figure 1) on memory processes and possible intracellular molecules involved in the hippocampus of Aβ-injected rats, an accepted model of AD. 32 –34 The hepatotoxicity of oral administration of these 2 flavones was also evaluated by measurement of alanine aminotransferase and aspartate aminotransferase enzymes in liver and blood serum.
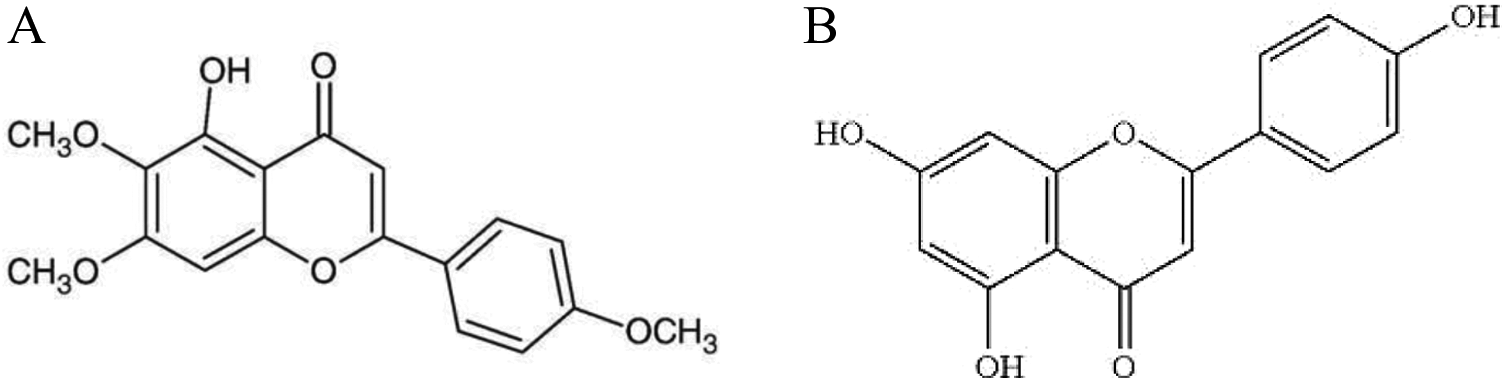
Chemical structure of (A) 5-hydroxy-6,7,4’-trimethoxyflavone (flavone 1) and (B) 5,7,4′-trihydroxyflavone (flavone 2).
Materials and Methods
Reagents
Antibodies directed against phospho-adenosine monmo phosphate (AMP)-activated protein kinase (pAMPK), AMP-activated protein kinase (AMPK), phospho-CREB, CREB, and β-actin were obtained from Cell Signaling Technology (Beverly, Massachusetts). Mitochondrial transcription factor A (TFAM) and PGC-1α antibodies were from ABCAM (Cambridge, United Kingdom). Anti–nuclear respiratory factor 1 (NRF-1) was purchased from Santa Cruz Biotechnology (Santa Cruz, California). Flavone 1 was a gift from Plants and Drugs Research Institute, Shahid Beheshti University, and flavone 2 was from Sigma Aldrich (St Louis, Missouri). Enhanced chemiluminescence (ECL) kit was obtained from Amersham Bioscience (Piscataway, New Jersey). Polyvinylidene fluoride (PVDF) was from Millipore (Billerica, Massachusetts). All the other reagents of enzymatic assays and Aβ1-42 were obtained from Sigma Aldrich.
Animals
Adult male Albino Wistar rats weighing 230 to 270 g were used in this study. Rats were housed under standard laboratory conditions (12-hour light–dark cycle) and had free access to food and water. All experimental procedures were in agreement with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health Publication No. 80-23, revised 1996) and were also approved by the Animal Care Committee of Shahid Beheshti University of Medical Sciences.
Experimental Design
Rats were divided into 6 groups (n = 14): vehicle group, which received oral gavage of water (1 mL) daily for 1 week before the bilateral intrahippocampal injection of 3 µL phosphate-buffered saline (PBS). The Aβ group received oral gavage of water (1 mL) for 1 week before the intrahippocampal injection of Aβ (2.2 nmol/L). The other 4 groups received 1 mL solution of flavone 1 or flavone 2 every day for 7 days (1 mg/kg). Flavone treatment was stopped, and rats received Aβ or PBS in the CA1 region of hippocampus.
Preparation of Aβ1-42
To reach 200 ng/µL concentration and fibril formation, the Aβ1-42 was solubilized in PBS (0.1 mol/L) then incubated at 37°C (5 days). On the surgery day, this solution was diluted with PBS (2.2 nmol/L).
Isolation and Identification of Flavone 1
Flavone 1 was purified as yellow crystals from the aerial parts of
Surgery and Microinjection
Anesthesia was induced by ketamine hydrochloride (50 mg/kg) and xylazine (4 mg/kg) administered by intraperitoneal injection. Rats were placed on a stereotaxic frame (Stoelting, Wood Dale, IL, USA), and the skin overlying the skull was incised. Coordinates for the CA1 of hippocampus and bilateral injection of 3 µL/side PBS (vehicle) and/or Aβ were anterocaudal: AP 3.8 mm, lateral: ±3.2 mm (both respect to bregma), and vertical: 2.8 mm (from dura), according to the atlas of Paxinos and Watson. 37 All microinjections performed over a period of 60 seconds with a Hamilton syringe.
Tissue and Serum Collection
Seven days after surgery, animals were decapitated. Carotid blood was collected in centrifuge tubes. The rats’ brain were collected and washed in ice-cold PBS and then hippocampi were dissected out. Whole livers were also removed and samples were immediately stored at −80°C. Serum was separated and used freshly for the assessment of liver function test.
Western Blot Analysis
Hippocampi were lysed in lysis buffer (pH 7.4 containing Tris-HCl 1 mol/L pH 8.0, NaCl 150 mmol/L, sodium deoxycholate 0.5%, sodium dodecyl sulfate 0.1%, EDTA 100 mmol/L, NaF 5 mmol/L, Na2MoO4 1 mmol/L, NaVO3 1 mmol/L, Triton X 100 1%, and protease inhibitor cocktail). Protein concentration was measured by Bradford method 38 and then 60 μg of total protein was loaded on a 12% polyacrylamide gel and transferred to PVDF membrane. The membranes were blocked in 2% non-fat dry milk in TBST and incubated with primary antibodies (overnight at 4°C). After incubation of blots with horse radish peroxidase-conjugated secondary antibody, immunoreactive proteins were detected with the ECL reagents. To confirm equal loading of protein, β-actin antibody was reprobed on membranes. The relative density of protein bands was measured by densitometric analysis using Image J software (ver. 1.41o, NIH, USA; http://rsb.info.nih.gov/ij/).
Measurement of Mitochondrial Deoxyribonucleic acid (mtDNA) Content by Real Time-Polymerase Chain Reaction (RT-PCR)
mtDNA content of rat hippocampus was measured using real-time polymerase chain reaction amplification reactions with the SYBR Green detection method. The specific primers were designed for the rat mitochondrial cytochrome b and the rat nuclear β-actin genes, respectively, as follows: forward, 5′-CGGCTGACTAATCCGATACC-3′, reverse, 5′-TGGGAGTACATAGCCCATGA-3′ and forward, 5′-CCCAGCCATGTACGTAGCCA-3′ and reverse, 5′-CGTCTCCGGAGTCCATCAC-3′. Total intracellular DNA in brain tissues was extracted using QIAamp DNA mini kit (QIAGEN, Germantown, Maryland) following the manufacturer’s instruction. Each RT-PCR contained 50 µg of template DNA, SYBR Green Real-time PCR Master (Applied Biosystems, Waltham, Massachusetts, USA), each of the forward and reverse primers, and ultrapure water. Relative amounts of mtDNA and nuclearDNA (nDNA) copy numbers were compared (All materials in this method were gift from Dr Maghsoudi’s laboratory).
Mitochondrial Preparation
The hippocampi were isolated and homogenized in ice-cold isolation medium (0.25 mol/L sucrose, 10 mmol/L Tris, 0.5 mmol/L K+ EDTA, pH 7.4) then centrifuged at 2°C (3 minutes at 2000×
Mitochondrial Swelling Assay
Mitochondrial swelling was assessed by measuring changes in the absorbance of the suspension wavelength at 540 nm. Isolated mitochondria (0.4 mg/mL) were incubated in 2 mL of respiration buffer (150 mmol/L KCl, 5 mmol/L HEPES, 5 mmol/L K2HPO4.3H2O, 2 mmol/L
Mitochondrial Membrane Potential Assay
The Δψm was measured using fluorescence change in Rh 123 (0.1 mmol/L) at 15 minutes. For this purpose, mitochondria (0.5 mg/mL) were incubated in buffer B containing 1 µg rotenone at 25°C, then Δψm was assessed by spectrophotometer with excitation at 505 nm and recording at 534 nm after addition of Rh 123 (0.1 mmol/L). 41
Passive Avoidance Test Apparatus
The shuttle box apparatus consisted of an illuminated and a dark compartment. Two chambers were separated by a guillotine door (8 × 8 cm). The floor of both chambers consisted of stainless steel rods of 3 mm diameter . Electric shock was delivered to the floor of the dark chamber with a stimulator (50 Hz).
Passive Avoidance Test Procedure
Passive avoidance test consists of 2 stages: training and memory test. On the training phase (7 days after Aβ injection), rats were allowed to habituate to the environment 1 hour before the training. Then, each rat was put in the illuminated chamber. Upon entry into the dark chamber, the guillotine door was closed and the rat was given 1 mA electrical shock for 1.5 seconds. After 30 seconds, rat was returned to its home cage. Two minutes later, the training was repeated. This procedure was repeated until the rat did not enter the dark chamber during 120 seconds. In memory test, 24 hours after training, rats were placed in the illuminated chamber, and after 10 seconds, the door was opened. They did not receive foot shock if they entered the dark chamber. The delay in entering the dark compartment (step-through latency [STL]) up to 300 seconds and the total time spent in the dark compartment (TDC) was recorded. 42
Statistical Analysis
All data are represented as the mean ± standard error of the mean. Comparison between the groups was made by 1-way analysis of variance followed by Tukey’s post hoc test to analyze the difference. The statistical significances were achieved when
Results
Flavonoids Improved Aβ-Induced Passive Avoidance Memory Deficits
To determine the effect of flavonoids on memory, rats were subjected to a passive avoidance task, a well-established task for investigating learning and memory processes. As shown in Figure 2A, Aβ-injected rats revealed a significant decrease in STL to enter the dark chamber and increased TDC compared to the control group. Flavonoid treatments notably increased the STL relative to Aβ-injected group. Also, in flavonoids-treated rats, TDC significantly decreased compared to the Aβ-injected rats (Figure 2B).
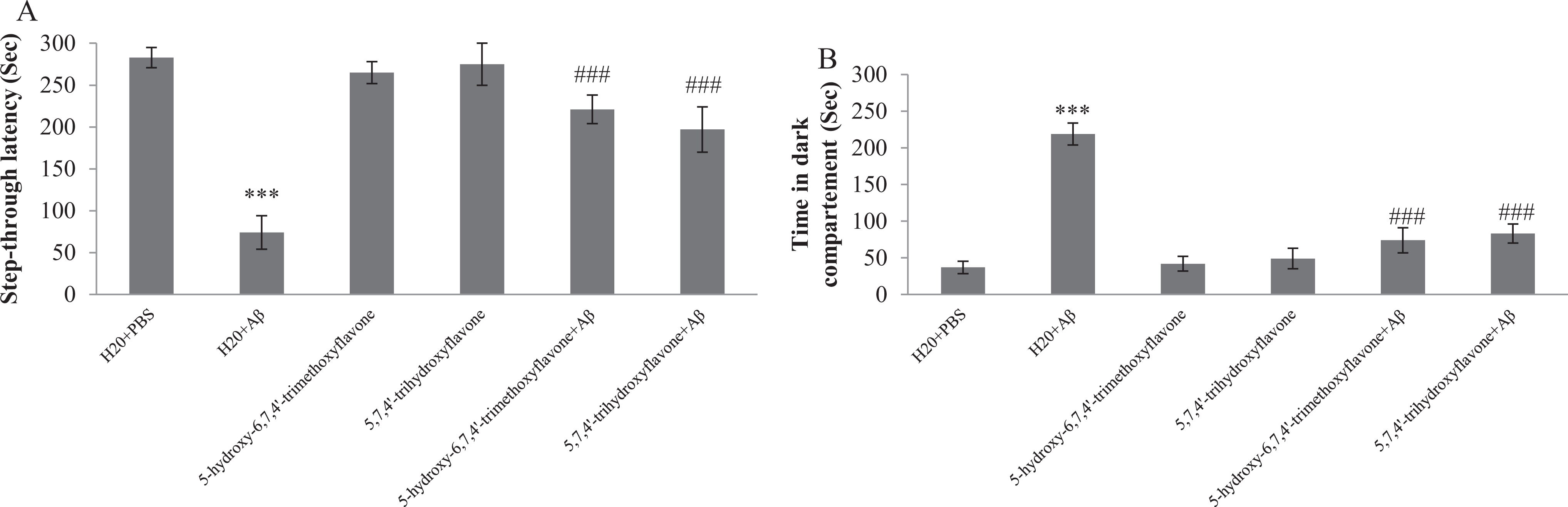
Effect of oral administration of 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1) and 5,7,4′-trihydroxyflavone (flavone 2) on memory. Step through latency (A) and time spent in dark compartment (B) during the probe test performed 24 hours after training phase. Each point shows the mean ± standard error of the mean (SEM) for 8 rats. ***
Flavonoids Increased Mitochondrial Biogenesis in Aβ-Injected Rats
Mitochondrial biogenesis has been identified as a compensatory response to cellular injury. The PGC-1α has been characterized as a master regulator of mitochondrial biogenesis. 43 Transcription factor CREB, involved in memory processes, and AMPK, cell energy sensor, are involved in regulation of PGC-1α. Activation of PGC-1α stimulates the expression of NRF-1 and TFAM as the other transcription factors involved in the mitochondrial genes expression. To review the effect of flavone 1 and flavone 2 on the induction of mitochondrial biogenesis, we determined the changes in factors involved in this pathway by Western blotting. As shown in Figure 3B and C, intrahippocampal injection of Aβ decreased phosphorylation of AMPK and CREB to about 62.9% and 68.7%, respectively, compared with control rats, while supplementation with flavone 1 prevented this reduction about 1.4- and 1.3-fold, respectively. Pretreatment with flavone 2 increased phosphorylation of AMPK and CREB by 1.2-fold when compared with Aβ-injected rats. Moreover, the level of PGC-1α, NRF-1, and TFAM in Aβ-injected rats reduced to 58.4%, 69.3%, and 54.2% compared to vehicle (Figure 3D-F).
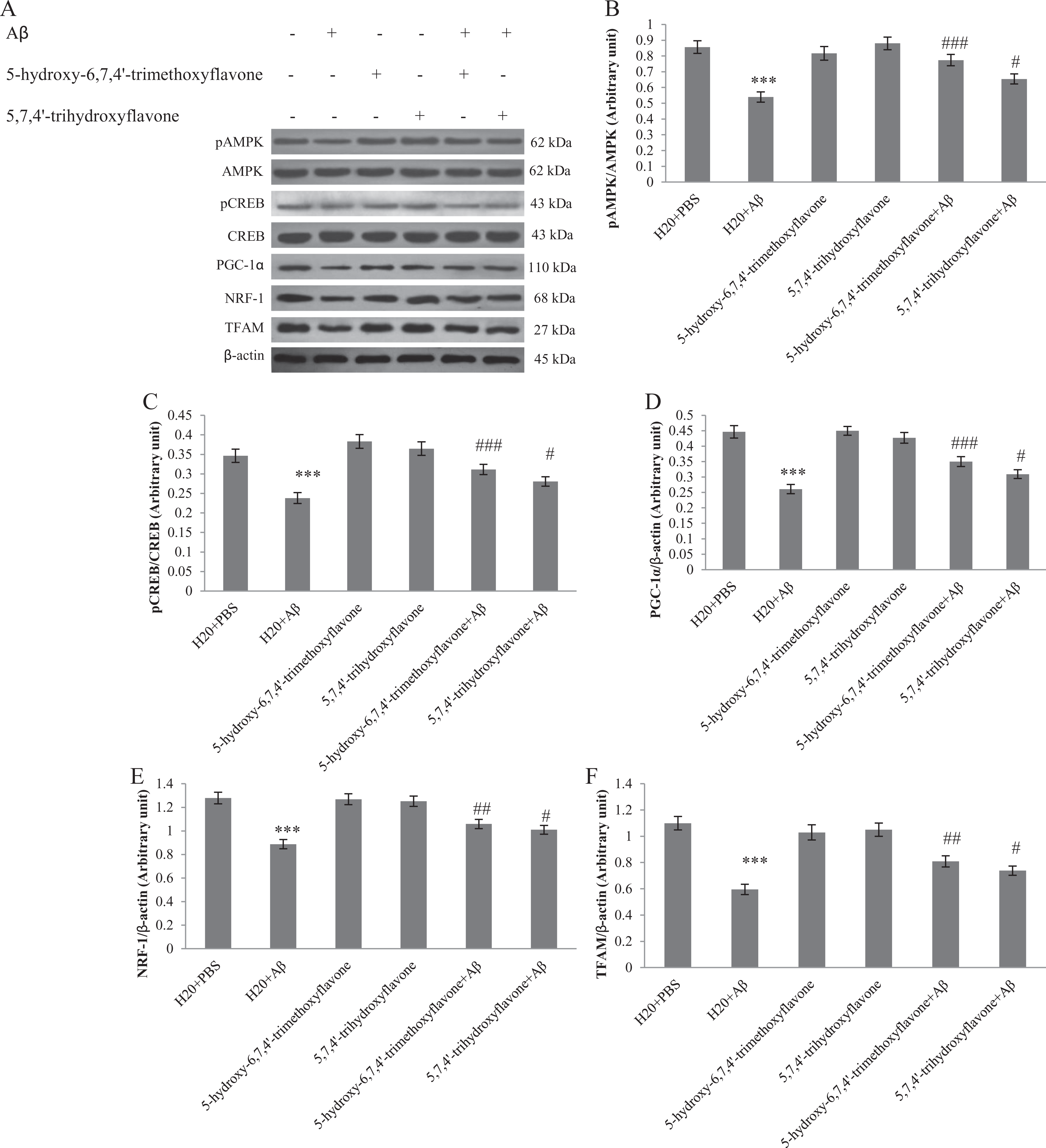
Western blot analysis to measure the effect of 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1) and 5,7,4′-trihydroxyflavone (flavone 2) pretreatment on the mitochondrial biogenesis in the hippocampus of amyloid β (Aβ)-injected rats. A, 60 µg of proteins were separated on sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), Western blotted, probed with specific primary antibodies, and reprobed with β-actin antibody (1 representative Western blot is shown; n = 6). The densities of phospho-adenosine monophosphate (AMP)-activated protein kinase (pAMPK)/AMP-activated protein kinase (AMPK) (B), phospho cAMP-responsive element-binding protein (pCREB)/cAMP-responsive element-binding protein (CREB; C), peroxisome proliferator-activated receptor γ coactivator 1α (D), anti–nuclear respiratory factor 1 (NRF-1; E), and mitochondrial transcription factor A (TFAM; F) bands were measured and their ratio was calculated. Each point shows the mean ± standard error of the mean (SEM). ***
In rats pretreated with flavone 1, the level of PGC1-α, NRF-1, and TFAM increased by 1.3-, 1.2-, and 1.3-fold, respectively, compared with Aβ group. Also, as shown in Figure 3, pretreatment with flavone 2 increased the mentioned factors to about 1.2-, 1.1-, and 1.2-fold compared to Aβ-injected rats.
In agreement with result from Western blot, measurement of mtDNA content by RT-PCR confirmed that pretreatment of Aβ-injected rats with flavones significantly increase mtDNA content in hippocampus compared to only Aβ-injected rats (Figure 4).
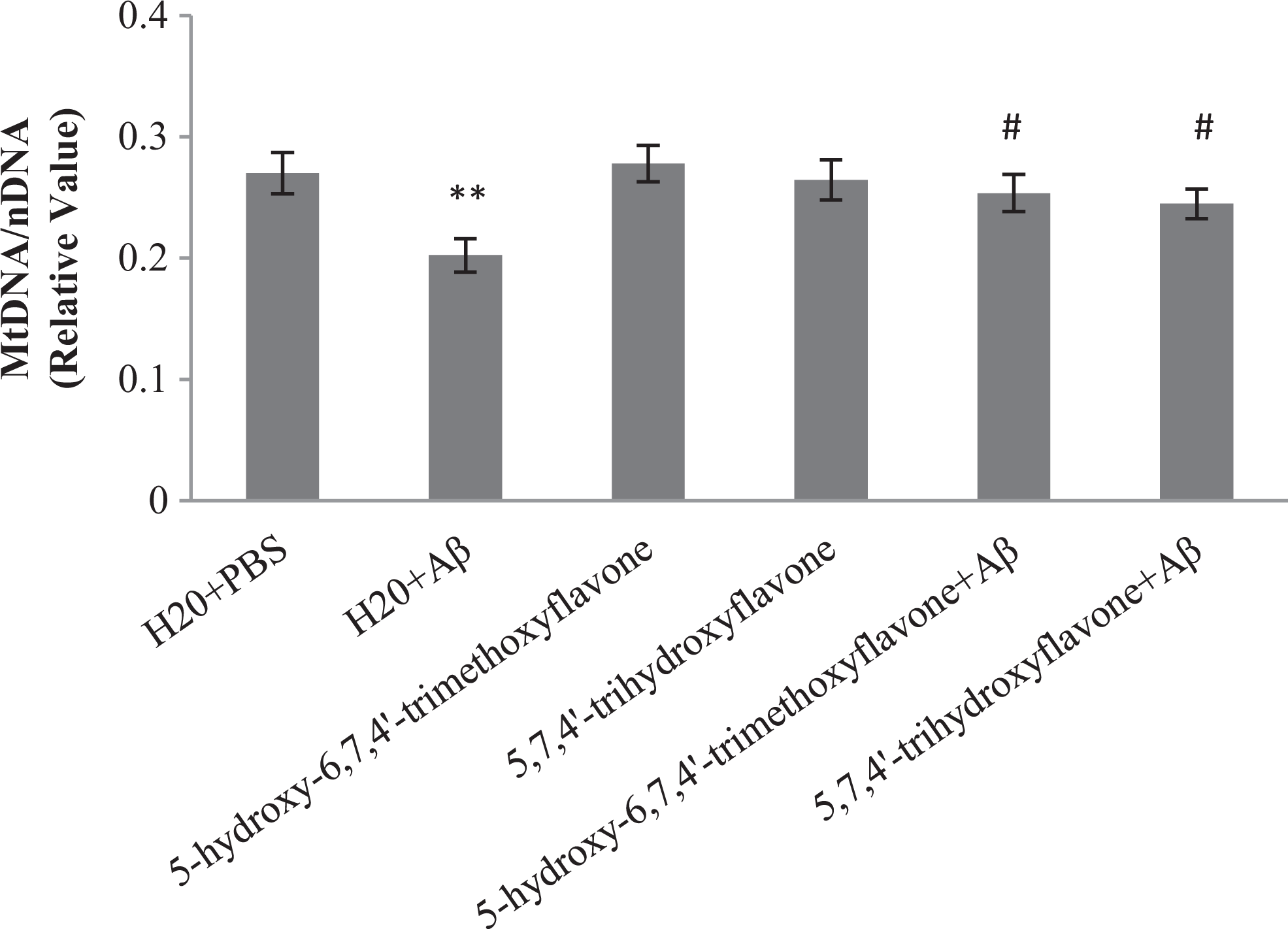
Effect of oral administration of 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1) and 5,7,4′-trihydroxyflavone (flavone 2) on relative mitochondrial deoxyribonucleic acid (mtDNA) content. Each point shows the mean ± standard error of the mean (SEM). **
Flavonoids Stabilized Δψm Reduced by Aβ in Rat Hippocampus
We investigated the inhibition of Δψm reduction with flavone 1 and flavone 2 in the presence of Aβ in rats hippocampi. An increase in discharge of Rh 123 fluorescence from mitochondria was seen, revealing the loss of Δψm. We found that Aβ reduced Δψm, whereas pretreatment by flavone 1 and flavone 2 prevented this reduction (Figure 5).
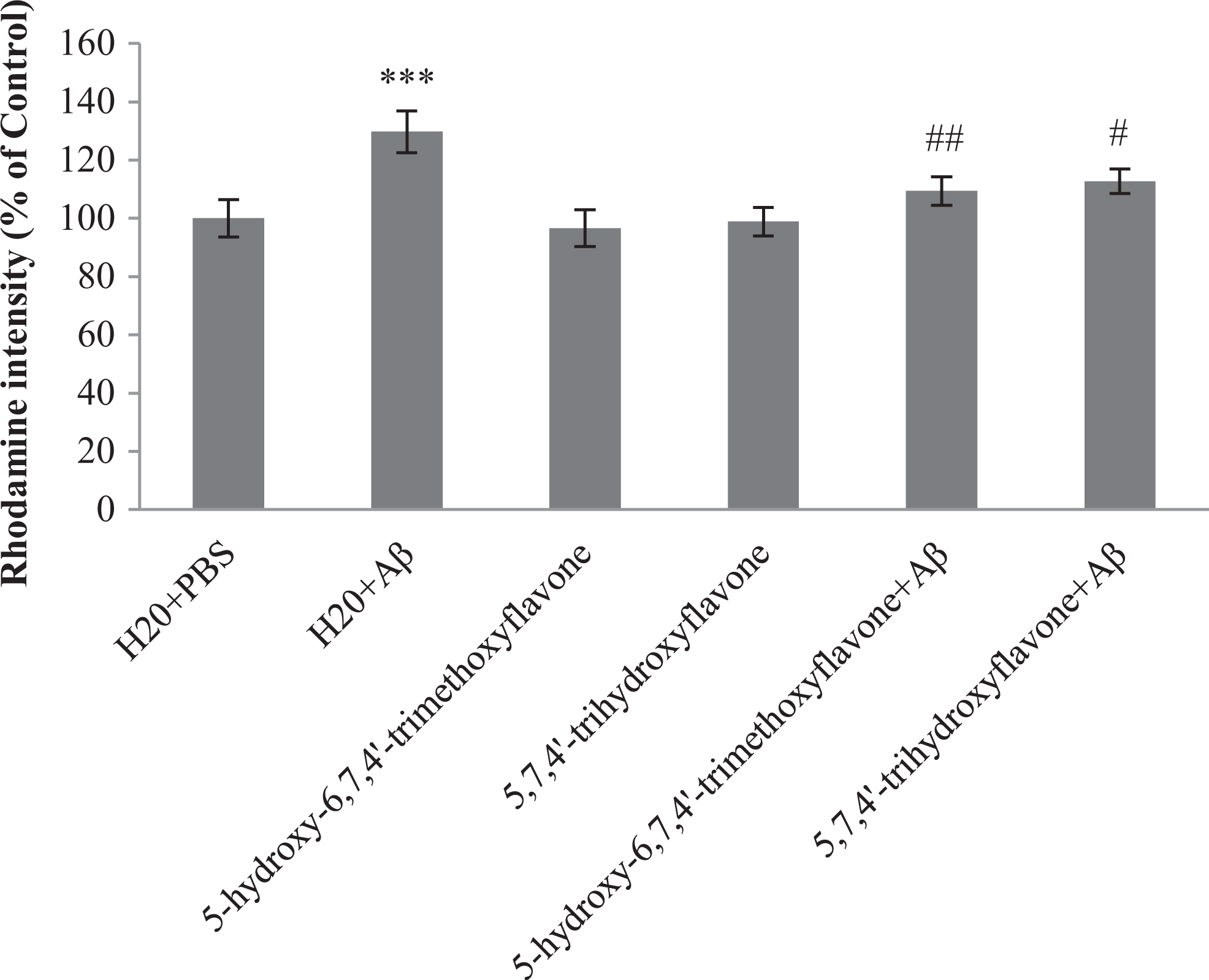
Effect of 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1) and 5,7,4’-trihydroxyflavone (flavone 2) pretreatment on the loss of Δψm induced by amyloid β (Aβ). Δψm was measured using Rh 123 fluorescence. Each point shows the mean ± standard error of the mean (SEM). ***
Flavonoids Decreased Mitochondrial Swelling in Aβ-Injected Rats
Opening of permeability transition pore (PTP) was indicated by mitochondria swelling, which resulted in apoptosis. 44 To study the brain mitochondrial swelling, we measured the absorbance of hippocampi mitochondria in all groups. As shown in Figure 6, Aβ caused a decrease in the absorbance of mitochondria, indicating that mitochondria in this group were swollen. Supplementation with flavone 1 and flavone 2 prevented mitochondrial swelling compared to vehicle/Aβ-injected rats.
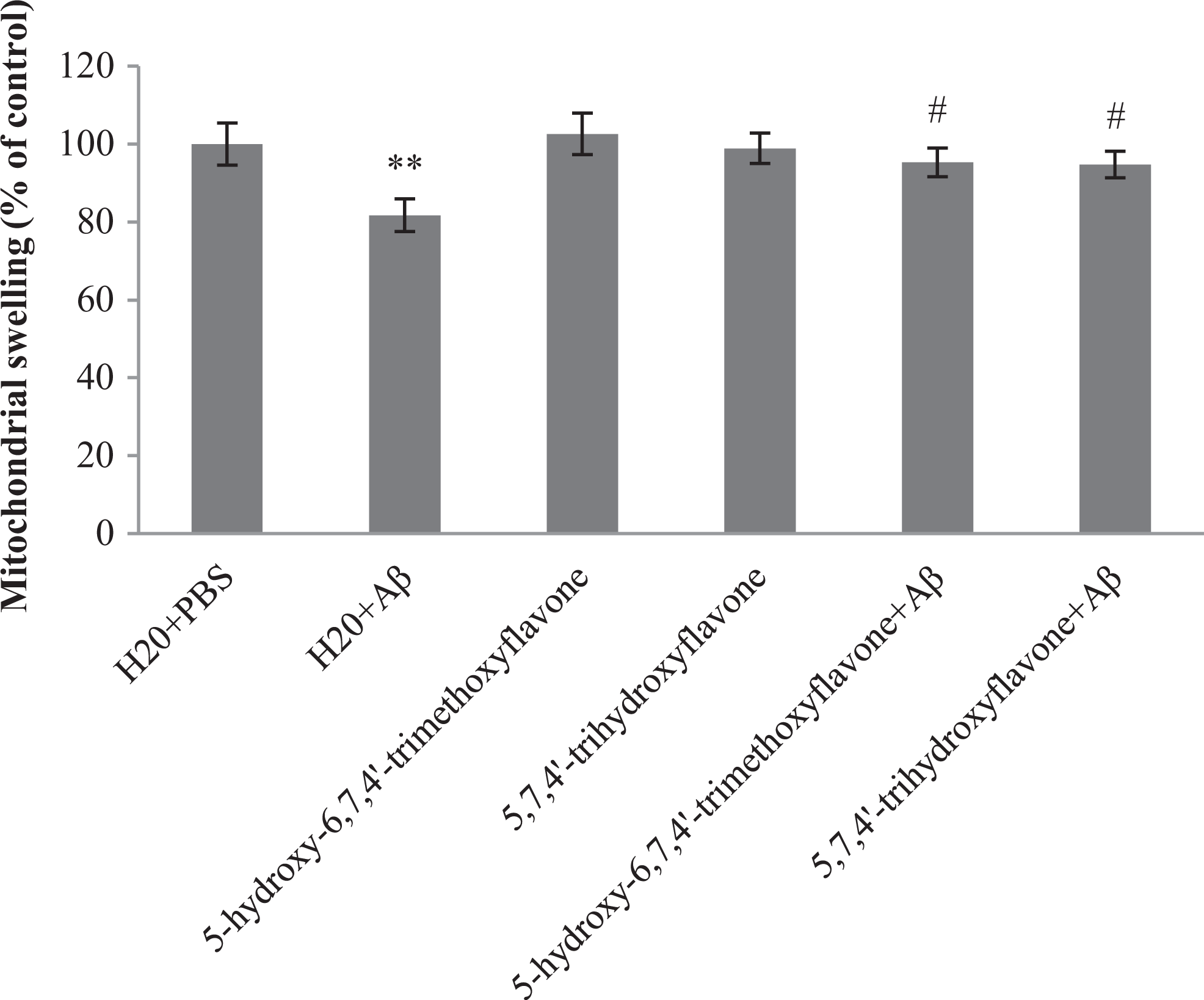
Effect of 5-hydroxy-6,7,4′-trimethoxyflavone (flavone 1) and 5,7,4′-trihydroxyflavone (flavone 2) pretreatment on the mitochondrial swelling induced by amyloid β (Aβ). Each point shows the mean ± standard error of the mean (SEM). **
Also, the level of alanine aminotransferase and aspartate aminotransferase enzymes in liver and serum did not show any changes in rats consuming flavones compared to control group (data not shown).
Discussion
In the present investigation, we showed that 2 flavones 5-hydroxy-6,7,4′-trimethoxyflavone and 5,7,4′-trihydroxyflavone could improve memory deficits induced by Aβ via modulation of mitochondrial function. Our data strongly suggest that these compounds protect hippocampal neurons against Aβ toxicity via mitochondrial biogenesis induction and inhibition of mitochondrial swelling and Δψm reduction.
Classically, protective effects of flavonoids, particularly in brain, have been attributed to their antioxidant property, which enhances ROS scavenging and modulates intracellular redox status. 45 However, mounting evidence suggests that the diverse bioactivity of flavonoids cannot be limited to their antioxidant activities. This suggestion was strongly confirmed by the series of experiments. These experiments showed that the protective effect of flavonoids in the central nervous system (CNS) is exerted via interaction with master molecules of intracellular signaling pathways involved in neuronal survival and differentiation, synaptic plasticity, and memory and cognition. Our data from PAT clearly indicated improvement of memory in Aβ-injected rats fed with flavonoids. This finding was perfectly in agreement with previous reports which showed the potential of some other groups of flavonoids to betterment of memory and cognition functions. 46
To explore the molecular basis of the effect of flavone 1 and flavone 2 on memory, we determined the protein levels of some intracellular molecules. As the master molecule of memory processes in neuronal cells, phosphorylated form of CREB (p-CREB) increased in the presence of 2 flavones, as was expected. The p-CREB translocates to the nucleus and activates cAMP response element on promoter of several downstream genes including PGC-1α, the best-known intracellular mediator of mitochondrial biogenesis. 47 PGC-1α is the activator of NRF-1, a nuclear transcription factor that contributes to the expression of genes encoding mitochondrial proteins 48 such as TFAM, which plays a crucial role in both maintaining mtDNA 49 and increasing its transcripts. 50,51 Moreover, PGC-1α is involved in ROS metabolism by inducing ROS detoxifying enzymes including catalase, superoxide dismutase, and glutathione peroxidase. Thus, it appears that known traditional function of flavonoids could be partially due to modulation of PGC-1α.
There are contradictory reports about the mitochondrial biogenesis in AD brains. While some researchers have reported increase in ETC gene expression and mtDNA, 52,53 others have demonstrated that aging and age-related disease such as AD reduces capacity for mitochondrial biogenesis in neuronal cells. 16,17,54 –57 In a previous study, we showed that intrahippocampal injection of Aβ decreased PGC-1α level, and the downstream of PGC-1α, NRF-1, and TFAM protein levels diminished after Aβ injection. 58 In the present study, our data showed that pretreatment of Aβ-injected rats with flavone 1 and flavone 2 prevented the reduction in PGC-1α, NRF-1, and TFAM levels. This suggests that flavonoids could induce cell resistance against Aβ neurotoxicity by activating signaling pathway associated with mitochondrial biogenesis. Moreover, improvement in memory in flavones-treated rats, at least in part, could be attributed to increased level of mitochondrial biogenesis factors. Hayashi and coworkers have demonstrated that overexpression of TFAM protects mtDNA against detrimental effect of ROS and subsequently reverses memory deficit in mice. 59
AMPK, PGC-1α regulator factor at the post-translational level, is a cellular energy sensor that is allosterically activated by intracellular increase in AMP–adenosine triphosphate ratio. 60 Impairment of AMPK activity during aging seems to be one of the key factors linked to deficiency in mitochondrial biogenesis and function in aged animal 60 and age-related disease. 57 In agreement with other literature, our data revealed that Aβ injection reduced phosphorylation of AMPK level in the rats’ hippocampus, while oral supplementation with flavonoids increased phosphorylated form of AMPK. Lu and colleagues reported that quercetin, a flavonoid, exerts its neroprotective effect through activation of AMPK phosphorylation against oxidative stress and Aβ accumulation in mouse brain. 61 It is thought that the activation of p-AMPK may decrease Aβ generation by regulation of Amyloid precursor protein (APP) processing. 62,63 Also, it has been shown that AMPK improved memory and cognitive function via induction of neurogenesis and neuronal expansion. 62
Not only increase in mitochondrial biogenesis but also prevention of mitochondrial dysfunction is important to fight against oxidative injury. To support our data about the effectiveness of flavone 1 and flavone 2, we measured mitochondrial swelling and Δψm. Opening of PTP was indirectly demonstrated by mitochondrial swelling. At the physiological concentration of calcium ions, opening and closing of PTP is reversible and is regulated by the Δψm. This function of PTP provides calcium homeostasis in the cell. 64 The collapse of Δψm and swelling are pivotal events that have been described in several cellular apoptotic pathways. 44,65,66 Aβ is a well-known inducer of Δψm reduction and subsequent apoptosis. 67 It has been demonstrated that Aβ decreased Δψm and caused an uncoupling of respiration and mitochondrial swelling as changes in ultrastructural morphology. 68,69 Our data indicated that presence of Aβ caused synchronized reduction in Δψm and induction of mitochondrial swelling. Supporting our investigation, Mungarro-Manchaca found that the Aβ peptide increased mitochondrial dysfunction and induced morphological changes, including mitochondrial swelling. 70 Sirk et al also reported that Aβ decreased Δψm, increased ROS production, and altered mitochondrial morphology in an in vitro model. 71 The neuroprotective effect of flavonoids may be due to their ability to prevent the opening of PTP and thereby inhibit mitochondrial swelling. 72 Oral administration of flavone 1 and flavone 2 influenced both the swelling and Δψm with different potency. Flavone 1 increased Δψm and decreased mitochondrial swelling in the presence of Aβ.
Based on our finding, flavone 1 is more effective than flavone 2 for inhibition of mitochondrial dysfunction and induction of mitochondrial biogenesis. The ability of flavonoids to affect CNS depends on their penetration through blood–brain barrier (BBB). Flavonoids with less polarity, for instance O-methylated derivatives, are more permeable to cross the BBB. 73 So methylation at positions C-6, C-7, and C-4′ in flavone 1 enhances the ability of this flavones to penetrate the BBB, and consequently, it could exert more neuroprotective effects.
Taken together, flavonoids are able to alleviate memory and cognition dysfunction in Aβ-injected rats at least partially through induction of mitochondria biogenesis and inhibition of some mitochondrial alterations including Δψm reduction and swelling. There is a reverse relation between polarity of flavonoid molecule and its penetration through BBB. Therefore, further studies to design less polar flavones derivatives could be a therapeutic strategy for treatment of AD.
Footnotes
Author Contributions
Arsalandeh, Farshad: contributed to acquisition; drafted manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Ahmadian, Shahin: contributed to analysis and interpretation; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Foolad, Forough: contributed to analysis and interpretation drafted manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Fariba Khodagholi: contributed to interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Farimani, Mahdi M: contributed to analysis; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Shaerzadeh, Fatemeh: contributed to conception and design; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by Iran National Science Foundation (grant number 91001416).
