Abstract
Chloroform is an organic solvent used as an intermediate in the synthesis of various fluorocarbons. Despite its widespread use in industry and agriculture, exposure to chloroform can cause illnesses such as cancer, especially in the liver and kidneys. The aim of the study was to analyze the effects of chloroform on redox imbalance and pulmonary inflammatory response in adult C57BL/6 mice. Forty animals were divided into 4 groups (N = 10): female (FCG) and male (MCG) controls, and females (FEG) and males (MEG) exposed to chloroform (7.0 ppm) 3 times/d for 20 minutes for 5 days. Total and differential cell counts, oxidative damage analysis, and protein carbonyl and antioxidant enzyme catalase (CAT) activity measurements were performed. Morphometric analyses included alveolar area (Aa) and volume density of alveolar septa (Vv) measurements. Compared to FCG and MCG, inflammatory cell influx, oxidative damage to lipids and proteins, and CAT activity were higher in FEG and MEG, respectively. Oxidative damage and enzyme CAT activity were higher in FEG than in FCG. The Aa was higher in FEG and MEG than in FCG and MCG, respectively. The Vv was lower in FEG and MEG than in FCG and MCG, respectively. This study highlights the risks of occupational chloroform exposure at low concentrations and the intensity of oxidative damage related to gender. The results validate a model of acute exposure that provides cellular and biochemical data through short-term exposure to chloroform.
Introduction
Chloroform is an organic solvent used as an intermediate in the synthesis of various fluorocarbons. 1 Despite its widespread use in industry and agriculture, exposure to chloroform at moderate doses causes illnesses such as cancer, especially in the liver and kidneys. 2 –5 Chloroform is metabolized to phosgene by the cytochrome P450 oxidative complex CYP2E1 in the liver and kidneys. 6 In the bloodstream, this intermediate metabolite damages cells and binds to cytoplasmic proteins, denaturing them or interfering with the functions of mitochondria and other organelles. 2,4,7 –10 The damage caused by chloroform, therefore, is associated with its cell penetration, metabolism, and toxicity from these processes and is related to gender. 11
Contact with chloroform occurs via inhalation. Vapor suspension in baths or indoor pools corresponds to 40% to 50% of total exposure to the substance, followed by direct contact with the skin (20%-30%), and ingestion of chlorinated water (up to 20%). 12 Studies have shown that inhaled chloroform damages the nasal mucosa and has various manifestations in mice and rats, in which inflammatory response differs between males and females. 1,13 Genetic factors linked to sexual dimorphism, such as hormone production, variations in muscle and bone mass, and the predominant type of inflammatory response, can increase or decrease the production of oxidant species. 3
The innate and adaptive immune responses are the bodily defenses against toxic substances that reach the lung alveoli. The innate immune system of the lungs comprises the epithelial barrier and the acute inflammatory response that can cause tissue damage via the recruitment and activation of neutrophils, eosinophils, and macrophages. 14 Macrophages act through phagocytosis of destroyed cell debris or cells in which structure and function have been altered by chloroform. Neutrophils act through degranulation and release of substances that can destroy affected cells and other cellular debris. 15
The imbalance between oxidants and antioxidants in favor of oxidants is called redox imbalance. Increasing redox imbalance can be the result of direct exposure to chloroform but may also be generated by activated inflammatory cells such as neutrophils and macrophages. 16 The harmful potential of chloroform is believed to be associated with the intracellular production of oxidant species that cause the release of proinflammatory chemotactic factors, in turn triggering an inflammatory response in the body, mainly from macrophages and neutrophils. 1,17
The body has an antioxidant system to counteract this release and its potential negative effects. The main components of this system are the antioxidant enzymes superoxide dismutase, catalase (CAT), and glutathione peroxidase that act at the beginning of the formation of reactive species chains, preventing the accumulation of O2 and H2O2 radicals. 18 These antioxidant enzymes constitute the primary mechanism of protection against parenchymal lung injury mediated by free radicals. 18 This study evaluated the effects of ultrasonic nebulization with chloroform on the imbalance between oxidants and antioxidants in pulmonary inflammatory response and its relationship with sexual dimorphism in adult C57BL/6 mice.
Materials and Methods
Experimental Design
Eight-week-old male and female C57BL/6 mice were housed under controlled conditions in standard laboratory cages (Laboratory of Experimental Nutrition, Department of Food, School of Nutrition, Federal University of Ouro Preto) and given free access to water and food. All in vivo experimental protocols in animals at the Federal University of Ouro Preto were approved by the ethics committee. C57BL/6 mice (N = 40) were divided evenly into 4 groups, namely, female (FCG) and male (MCG) control mice exposed to ambient air and female (FEG) and male (MEG) mice exposed to chloroform. Chloroform exposures were conducted in an 30-L volume inhalation chamber (40-cm long, 30-cm wide, and 25-cm height) located within a ventilation enclosure. To achieve the desired concentration, 0.21 mL of anhydrous chloroform (99% purity; Sigma-Aldrich, St Louis, Missouri) was diluted in 9.79 mL of water and vaporized by an ultrasonic nebulizer (Ultrasonic Inhaler Ultra Alivio Multi Power manufactured by Unique Ltda. São Paulo- Brazil.) connected to the inhalation chamber. This yields a chloroform exposure that occurred for 20 minutes, 3 times a day (morning, afternoon, and night) for a total of 60 min/d for 5 consecutive days.
The chamber chloroform concentration was 7 ppm (Adapted of Kano et al, 2002), 19 a dose chosen because previous research suggests that the concentrations of 10 ppm or below will not induce hepatotoxicity in mice (female B6C3F1) regardless of exposure duration. The rate of production of toxic metabolites and the subsequent rate of cellular damage produced by continual exposure of approximately 10 ppm chloroform are less than the maximum rates at which hepatocytes can detoxify those metabolites and repair any induced cellular damage. 4 Control and exposed animals were killed by cervical dislocation 24 hours after the last treatment.
Assessment and Analysis of Bronchoalveolar Lavage Fluid
Immediately after euthanasia, the chest of each animal was opened to collect bronchoalveolar lavage fluid (BALF). The left lung was clamped, and the right lung and the trachea were cannulated and perfused with 1.5 mL of saline solution (3 × 500 μL). The samples were kept on ice until the end of the procedure to avoid cell lysis. Total mononuclear and polymorphonuclear cell numbers were previously stained with trypan blue and determined in Neubauer chambers. Differential cell counts were performed on cytospin preparations (Shandon, Waltham, Massachusetts) stained with fast panoptic coloration kit (Laborclin, Pinhais, Paraná, Brazil). 20
Processing and Homogenizing Tissue
After BALF collection, the right ventricle of each mouse was perfused with saline to remove blood from the lungs. The right lung was clamped so that just the left lung could be perfused with 4% buffered formalin (pH 7.2) at a pressure of 25 cm H2O for 2 minutes via the trachea. The left lung was removed and immersed in a fixative solution for 48 hours. The material was then processed as follows: tap water bath for 30 minutes, 70% alcohol and 90% baths for 1 hour each, 2 baths in 100% ethanol for 1 hour each, and embedded in paraffin. Serial 5-μm sagittal sections stained with hematoxylin and eosin were obtained from the left lung for histologic analyses. After the removal of the left lung for histology, the right lung was immediately removed and stored on crushed ice in labeled tubes. The lungs were subsequently homogenized in 1 mL potassium phosphate buffer, pH 7.5, and centrifuged at 1500g for 10 minutes. The supernatant was collected, and the final volume of all samples was adjusted to 1.5 mL with phosphate buffer. The samples were stored in a freezer (−80°C) for biochemical analysis.
Antioxidant Defense and Oxidative Stress Biomarkers in Lung Homogenate
As an index of lipid peroxidation, we used the formation of thiobarbituric acid reactive substances (TBARS) during an acid-heating reaction as previously described by Draper et al. 21 Briefly, the TBARS level was estimated in accordance with the method described by Buege and Aust. 22 Carbonyl protein levels were determined according to the method described by Levine et al. 23 Lung homogenates were used to determine CAT activity. This method was based on the enzymatic decomposition of H2O2 observed spectrophotometrically at 240 nm for 5 minutes. The results were expressed as activity per milligram of protein. One unit of CAT was equivalent to the hydrolysis of 1 μmol of H2O2 per minute. 24 Total protein content was determined according to the method described by Lowry et al 25 using bovine serum albumin as a standard.
Morphometric and Stereological Analyses
All morphometric analyses were performed at the Multiuser Laboratory of the Research Center for Biological Sciences of the Federal University of Ouro Preto. Twenty random images obtained from the histological slides of the lungs were digitized with Leica Application Suite software in association with a Leica DM5000b optical microscope and CM300 digital micro-camera. All images were scanned with a 40× objective. With the aid of Image J® (National Institute of Health, NIH, Maryland, USA), we used a representative image of a 40× magnification with a 100-µm ruler to calibrate a ruler in pixels derived from the program such that 434 pixels equaled 100 μm. Five alveolar areas (Aas) of each slide prepared from each animal were measured. 26,27
Analysis of the volume density of the alveolar septa (Vv) was performed on a test system consisting of 16 points and a known test area
28
in which the boundary line was considered forbidden to avoid an overestimation of the number of structures. The test system was connected to a monitor attached to a microscope. The number of points (PP) that touched the alveolar septa (Vv) was assessed according to the total number of test points (PTs) in the system using the equation
Statistical Analysis
The data with normal distribution were assessed through univariate analysis of variance (ANOVA; one way ANOVA) followed by the Tukey posttest. Data were expressed as mean ± standard error of mean. We used the Kruskal-Wallis test followed by Dunn’s post-test for discrete data and expressed them as median, minimum, and maximum values. In both cases, the difference was considered significant when the P value was <0.05. All analyses were performed with GraphPad Prism version 5.00 (GraphPad Software; San Diego, California) for Windows 7.
Results
Analysis of the Body Mass of the Animals
To evaluate the body mass of the animals during the 5 days of exposure to chloroform, we measured the body mass of the FEG and MEG at the end of the exposures (Figure 1). In our study, the FEG showed reduced body mass when compared to MEG. The exposure to chloroform did not affect the body mass of FCG when compared to FEG and the body mass of MCG and MEG are not significantly different.
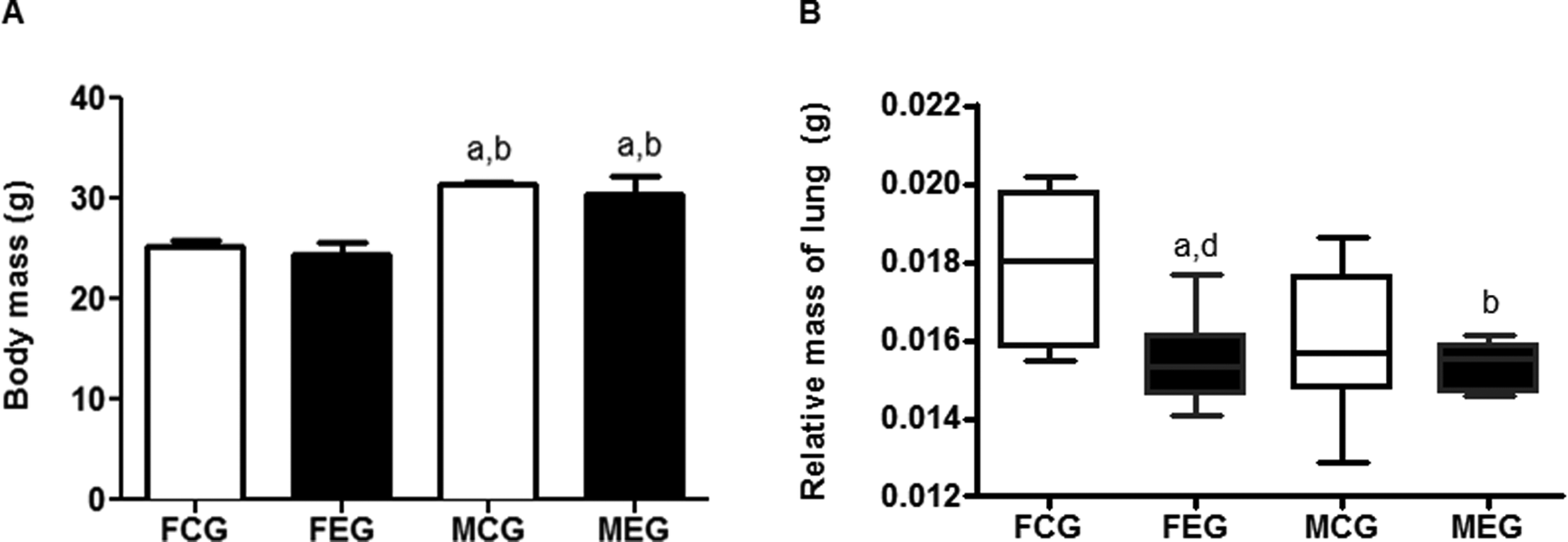
Analysis of the total mass and lung relative mass of female control (FCG), male control (MCG), female exposed (FEG), and male exposed (MEG). Data were expressed as mean ± standard error of the mean and were analyzed by analysis of variance (ANOVA) followed by Tukey posttest (P < 0.05). Data were expressed as median/minimum/maximum value and were analyzed using the Kruskal-Wallis test followed by Dunn’s post-test (P < 0.05).
Analysis of Lung Relative Mass
The relative mass of the lungs was calculated as a simple ratio of lung mass divided by the total mass of animals, which allowed an assessment of the possible mass change due to the local action of chloroform, despite the preexisting bias of sexual dimorphism in which the body mass of MEG increased compared to that of the FEG. The relative mass of FEG lungs decreased compared with that of FCG lungs (Figure 1).
Analysis of the Total and Differential Leukocyte Counts in BALF
The ultrasonic inhalation of chloroform induced an inflammatory response in the lung parenchyma of the FEG. Exposure to chloroform caused a significant increase in BALF total leukocytes in the FEG compared with the FCG (Table 1 and Figure 2). Alveolar macrophages were more frequent in FEG BALF than in FCG BALF. No significant changes were observed in the differential counts of neutrophils and lymphocytes.
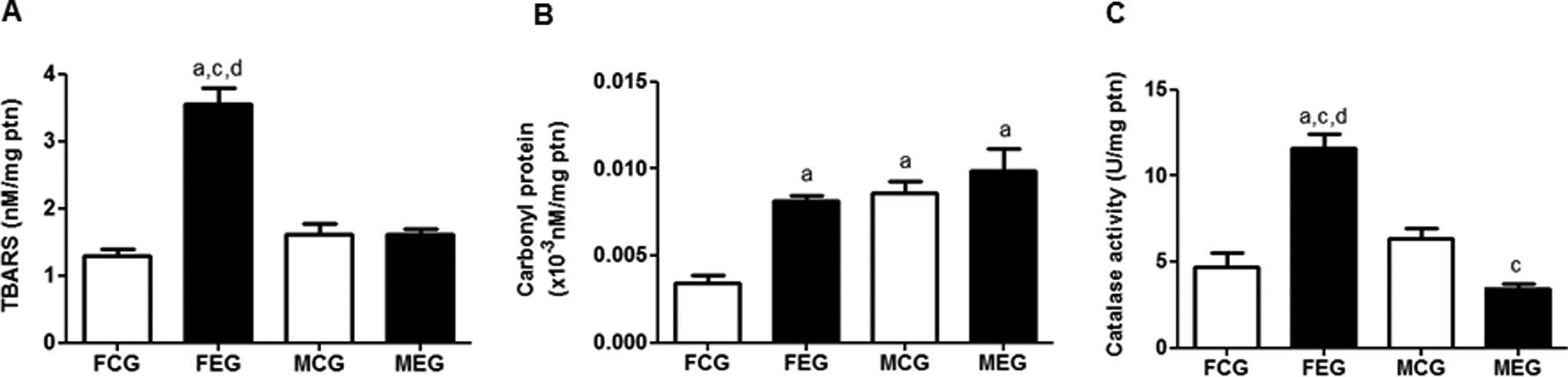
Analysis of thiobarbituric acid reactive substance (TBARS), carbonyl protein levels and catalase (CAT) activity. Data were expressed as mean ± standard error of the mean and were analyzed by analysis of variance (ANOVA) followed by Tukey post-test (P < 0.05).
Effects of Exposure to Chloroform on Cellular Influx in Bronchoalveolar Lavage Fluid From the Controls and Exposed Groups.a
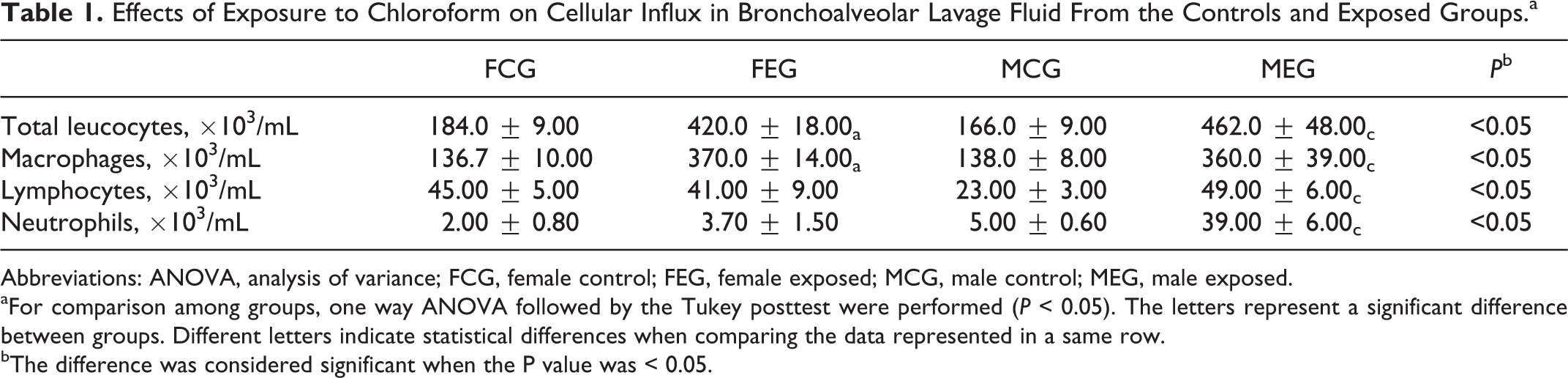
Abbreviations: ANOVA, analysis of variance; FCG, female control; FEG, female exposed; MCG, male control; MEG, male exposed.
aFor comparison among groups, one way ANOVA followed by the Tukey posttest were performed (P < 0.05). The letters represent a significant difference between groups. Different letters indicate statistical differences when comparing the data represented in a same row.
bThe difference was considered significant when the P value was < 0.05.
A significant increase in total leukocytes was observed in MEG BALF compared to those of MCG BALF (Table 1 and Figure 2). Alveolar macrophages were more frequent in MEG BALF than in MCG BALF. The number of lymphocytes in MEG BALF was higher than that in MCG BALF. Neutrophils were present in higher numbers in MEG BALF compared with MCG BALF (Table 1).
Determination of TBARS and Carbonyl Protein Levels
Imbalance between oxidants and antioxidants in the lung parenchyma was evaluated by measuring redox imbalance biomarkers such as TBARS and carbonyl protein, which indicate damage to cell membranes and oxidative modification of proteins. TBARS content of the lung homogenate of the FEG increased compared with those in the homogenates of the FCG and MEG (Figure 2). Exposure to chloroform increased carbonyl protein levels in the FEG compared with that in the FCG.
Analysis of CAT Activity
Catalase is an enzyme present in most aerobic organisms and is responsible for the intracellular conversion of H2O2 into water and oxygen. Activity of CAT increased in the FEG compared to the FCG and MEG. Activity of CAT activity decreased in the MEG compared to the MCG (Figure 2).
Morphometric and Stereological Analyses
Histological analysis of our experimental model showed increased Aa in the FEG compared with the FCG and in the MEG compared with the MCG (Figure 3). In addition, the Vv in the FEG decreased compared to that in the FCG. It also decreased in the MEG compared to de MCG (Figures 3 and 4).
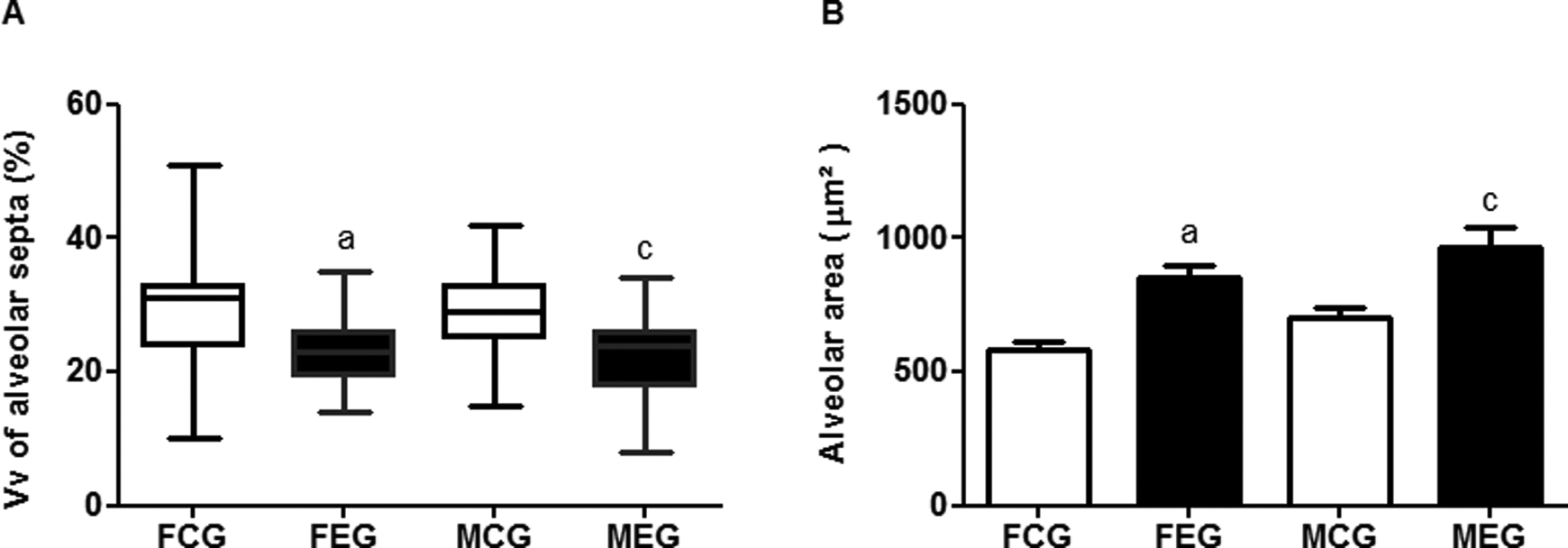
Comparison of density of alveolar septa and alveolar area. Data were expressed as median/minimum/maximum value and were analyzed using the Kruskal-Wallis test followed by Dunn’s post-test (P < 0.05). Data were expressed as mean ± standard error of the mean and were analyzed by analysis of variance (ANOVA) followed by Tukey post-test (P < 0.05).
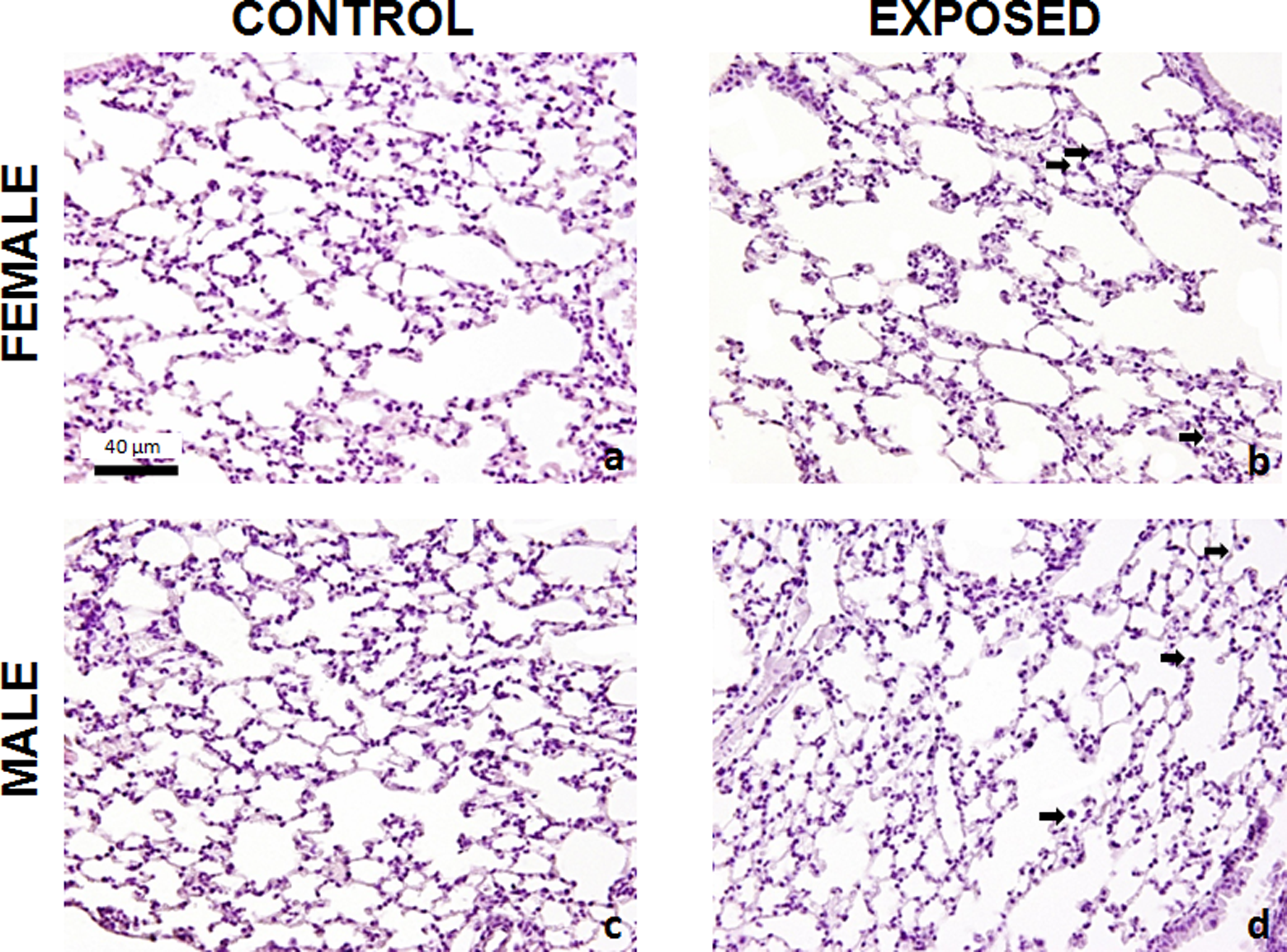
Photomicrographs of lung sections stained with hematoxylin and eosin of FCG, MCG, FEG, and MEG. A, Normal lung tissue FCG. B, Alveolar expansion and lower density of alveolar septa of FEG compared with FCG. C, Normal lung tissue from MCG. D, Alveolar expansion and lower density of alveolar septa of MEG compared to the MCG. Arrows indicate influx of inflammatory cells into the lung parenchyma of groups exposed to chloroform. Bar = 40 µm. FCG indicates female control; FEG, female exposed; MCG, male control; MEG, and male exposed.
Discussion
In this study, we analyzed total and relative mass of the lungs and total and differential cell counts in the BALF of animals acutely exposed to chloroform to evaluate the oxidative damage resulting from such exposure as indicated by lipid peroxidation, oxidative modification of proteins, and antioxidant enzyme CAT activity. Both Aa and Vv were assessed through histologic and morphometric analyses.
The inhalation of chloroform induces inflammation in various organs from the respiratory tree to the liver and kidneys. 9 Acute lung inflammation derives mainly from macrophages and neutrophils, which release inflammatory mediators and reactive oxygen species (ROS) in response to irritants, as well as from a variety of elastolytic enzymes, including neutrophil elastase, metalloproteinases 2, 8, 9, 12, cathepsin G, K, L, and S, and proteinase 3, which contribute to the destruction of the lung parenchyma. 30
Hackstein et al reported that different strains of mice (BALB/c, C57BL/6, DBA/2, C3H, and 129SV) exhibited distinct inflammatory responses. 31 However, Lanzetti et al, 32 Bezerra et al, 33 and Broeckaert et al 34 demonstrated that C57BL/6 mice exposed to different toxic substances such as cigarette smoke and ozone showed moderate to severe immune and/or inflammatory responses. In this context, C57BL/6 mice show a more pronounced inflammatory response when compared to the other strains of mice.
Sexual dimorphism is a common occurrence among mammalian species. Beginning at a specific stage of embryonic development, genetic factors derived from male and female hormones become pronounced and consequently lead to the proliferation of various cell families in differing amounts. Environmental factors such as level of physical activity and social tasks and functions may also influence the unlocking of genes for various characteristics. This gene inheritance and natural attributions derived from gene activation are largely responsible for the differences between male and female animals. 35
The difference between the total mass of the FEG and MEG may not have been influenced by acute exposure to chloroform because the acute exposure was insufficient to induce a systemic inflammatory reaction that could interfere with the total mass of these animals. We believe that the difference in mass between the exposed groups was derived from an existing sexual dimorphism evident in the controls; compared with the FCG, the MCG had a greater total mass.
Barnes et al have reported parenchymal damage due to an imbalance between proteolytic and antiproteolytic agents and redox imbalance, which leads to protein and lipid depletion. 36 Furthermore, the production of proinflammatory mediators such as interleukins (ILs), especially IL-1β and IL-6, in the acute phase of inflammation in addition to the recruitment of leukocytes can alter local metabolism, increasing glucose consumption due to the high cellular activity of the inflammatory process, thereby leading to increased consumption of lipids and proteins as alternative energy sources. 37
To assess whether exposure to chloroform promotes a cellular influx from the peripheral blood to the lung parenchyma, we performed BALF cell counts. Our results with chloroform corroborate the findings of previous studies in which the exposure of target organs to toxic substances such as cigarette smoke and formaldehyde caused an influx of macrophages and polymorphonuclear cells by inducing oxidative stress. 18,38,39 The total cell counts in the BALF fluid of male mice corroborated results published by Templin et al, 9 Golden et al, 10 and Wolf and Butterworth. 40 The presence of chloroform in the lung parenchyma in mice in both the FEG and the MEG induced an inflammatory response. The differential counts, however, showed a greater differentiation of cell types acting in response to the damage in the parenchyma of the MEG as well as the FEG, although the response was primarily from macrophages. When stimulated by damaged cells, macrophages produce proinflammatory cytokines that act in an autocrine manner to enhance phagocytosis and production of ROS. The presence of neutrophils and especially lymphocytes can reduce this autocrine action of proinflammatory ILs because these other cell types can also produce anti-inflammatory cytokines, such as IL-4, IL-10, IL-13, and transforming growth factor-β. 41 Most of these cytokines are produced by T CD4 + lymphocytes, and they reduce the affinity of macrophage and neutrophil receptors for IL-1, IL-6, and tumor necrosis factor α. It also suppresses the production of these substances as a paracrine action in macrophages and neutrophils. 41 This multicellular mechanism of inflammation in the MEG, along with the presence of lymphocytes, may explain the improved response of this group to redox imbalance and the consequent reduced cellular damage induced by the ultrasonic inhalation of chloroform.
In our study, the activity of the enzyme CAT was analyzed to understand more thoroughly its contribution to redox imbalance during chloroform exposure. Excess ROS produced as a result of the intense inflammatory process triggered a compensatory mechanism of the immune system in an attempt to reverse the stress and prevent oxidative damage. This mechanism led to an increase in the production of antioxidant enzymes such as CAT. The depletion of basal stocks of CAT in the absence of oxidative damage in the TBARS analysis indicates that stimulation for the production of ROS by inflammatory cells in the MEG was lower than that in the FEG and insufficient to damage the lung parenchyma. These data are contrary to those of other studies in which males showed increased oxidative damage in the liver, kidneys, and nasal cavity associated with the inflammatory response due to chloroform exposure. 3,13 The findings of Larson et al, 3 however, are contrary to those of Fang et al 11 who suggested that the tissue damage caused by chloroform differs between males and females in the enzyme pattern found in various organs.
Lipid peroxidation can be defined as a cascade of biochemical events that occurs as a result of the action of free radicals on unsaturated lipids in cell membranes and the destruction of those lipids. Pulmonary inflammatory response to acute infections is primarily related to macrophages, as shown in this study. Inflammatory cells are responsible for the increased production of ROS. If an inflammatory process is not controlled by anti-inflammatory mechanisms, signaling by the autocrine action of macrophages through ILs stimulates the production of ROS, and consequently, antioxidant enzymes are unable to contain the damage resulting from the elevation in free radicals. 42
The presence of carbonyl protein complex is therefore another marker of oxidative damage. Our results of protein carbonyl are in line with those of Campos et al 18 who showed that exposure to cigarette smoke, such as chloroform in our study, elevated production of ROS. Antioxidant enzymes are unable to control the inflammatory process completely, allowing oxidative damage and the formation of carbonyl protein complexes.
Our results about histological and stereological analyses of the lung parenchyma corroborate those of Bezerra et al, 17,20 Nagato et al, 17 and Keatings et al 30 who showed that a variety of elastolytic enzymes are released by the cells of the immune system in severe or poorly controlled inflammatory responses in addition to the elevation in ROS, and these enzymes contribute to the destruction of the lung parenchyma. Elastin fragments released during the degradation of elastic fibers and excessive production of ROS recruit more inflammatory cells into the lung parenchyma, increasing the inflammation and destruction of lung tissue and thereby reducing the elasticity of the parenchyma and distending the alveoli. 36 These factors are also associated with Vv in stereological analysis. 43
In summary, our overall results showed that short-term exposure to chloroform causes pulmonary inflammation. However, the intensity of the oxidative damage is related to gender. These results validate an acute model of exposure in which cellular and biochemical data can be obtained from short-term exposure to inhaled chloroform at low concentrations.
Footnotes
Acknowledgments
We thank the Laboratory of Experimental Nutrition (LABNEX-UFOP) and the Laboratory of Metabolic Biochemistry (LBM-UFOP).
Authors’ Note
Túlio Henrique Versiani de Oliveira and Keila Karine Duarte Campos contributed equally to this study. The work was done at The Federal University of Ouro Preto (UFOP).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Support Program of the Federal University of Ouro Preto (UFOP), under Contract No. 003/2014.
