Abstract
During gestation and lactation, the experimental mice dams received one of the following treatments: (a) diet free of pesticide; (b) diet enriched with endosulfan (END); 30.0 µg kg−1; (c) diet free of pesticide + oral vitamin E (α-tocopherol; 200 mg kg−1 per mouse); and (d) diet enriched with END (30.0 µg kg−1) + oral vitamin E (200 mg kg−1 per mouse). At weaning, pups and dams were killed, and selected organs as well as blood samples were collected for analyses. Compared with the control results, END induced alteration in a number of biochemical and histopathological parameters either in the dams or their offspring. The ameliorative effect of vitamin E to superoxide dismutase based on the “ameliorative index (AI)” for mothers and pups was 0.84 and 0.72, respectively. The AI for malondialdehyde reached a maximum value of nearly equal to 1.0 for dams or pups. For butyryl cholinesterase, the AI was 0.90 and 0.94 for dams and pups, respectively. In conclusion, a dietary exposure during gestation and lactation to low dose of END caused significant changes in the mother but also in the weaned animals that had not been directly exposed to this pesticide. These biological and histological alterations could be reversed to a great extent by oral supplementation of vitamin E.
Introduction
Organochlorine (OC) pesticides are among the most frequent contaminants found in the environmental compartments because they persist in the environment and bioaccumulate in organisms, partly due to their lipophilic properties. 1 Endosulfan (END) is a chlorinated cyclodiene pesticide, which was first severely restricted and then banned in 2006 in several European Union countries. This insecticide is a mixture of two stereoisomers, namely α- and β-END 2 in a ratio of 7:3. It has been used worldwide in agriculture, viticulture, and horticulture. 3 –6 END is one of the most abundant OC pesticides in the global atmosphere and is capable of undergoing long-range transport to remote locations such as the Arctic. 7 Nevertheless, the general population continues to be exposed: like other persistent OC pesticides, END residues have been detected in several foods including fruits, vegetables, and fish. 8 There is an evidence that END is acutely poisonous to humans through both accidental and intentional exposure. 9
END is classified by the World Health Organization (WHO) as a moderately hazardous (class II) pesticide; 10 however, it is genotoxic 11 and is an endocrine disrupter displaying xenoestrogenic activity. 12 END has been shown to be toxic to the liver, kidney, nervous system, and reproductive organs of laboratory animals. 3,13,14 The mode of action of END is to bind and inhibit γ-aminobutyric acid (GABA)-gated chloride channel receptor, there by inhibiting GABA-induced chloride flux across membranes. 15
Exposure to this compound can modify the activity of some enzymes involved in oxidative stress and xenobiotic metabolism, as well as testosterone metabolism and clearance. 16 Recently, Casabar et al. 17 showed that END is a strong activator of the pregnane X receptor (PXR) and an inducer of cytochrome P450 (CYP) 2B6 and CYP3A4, so it may have an impact on the metabolism of their substrates. A recent study by Ozmen et al. 18 showed that treating rabbits with END led to changes in glucose levels and histological degenerative changes in the pancreas and endocrine disturbances.
END may also be a risk factor for children whose parents have been exposed since the accumulation of OC compounds in fat tissue during the mother’s life can expose the child during pregnancy and breast feeding. 1 The transfer of OC pesticides from mother to fetus was demonstrated via the detection of the pesticides in the maternal umbilical cord, placenta, and in samples taken from the newborn. 1 Moreover, Pathak et al. 19 suggested that levels of some OC pesticides such as END in the maternal fluid or tissue are associated with preterm delivery and increased fetal oxidative stress. Pre- and postnatal exposure to END is reported to affect biogenic amines and amino acids in the prefrontal cortex. 20 Canlet et al. 21 reported in both genders an increase in plasma glucose as well as changes in levels of factors involved in the regulation of liver oxidative stress, confirming the prooxidant activities of this compound. While the toxicological effects of END have been studied in vitro and in vivo, the impact of long-term exposure to low doses through dietary intake in animal models from the fetal stage to adulthood has not yet been reported.
Pesticides, such as END, are omnipresent in environment, water, fruits, and vegetables and are considered as risk factors for human health. Consumers are mainly exposed to pesticides through diet, and the main question to be answered concerns the impact of such exposure on health. Specifically, women during pregnancy and lactation may be exposed to low doses of END from different environmental sources including food intake. From this standpoint, the present investigation is undertaking to assess effects of dietary exposure of mice dams to a low dose of END, characterized as “the acceptable daily intake” (ADI), in the absence and presence of vitamin E, as well as the effects on the offspring from the dams subjected to different treatments.
Materials and methods
Reagents
END was purchased from Fluka (Riedel de Haën, France) and was added as a component of rodent nuggets at a dose of 30 µg kg−1. This dose allowed the mice to ingest the equivalent of 0.006 mg kg−1 food per day ADI) defined for humans by Food and Agriculture Organization/WHO
22
that was extrapolated for mice on the basis of mean body weight. Information on the toxicity of END was obtained from Agritox (AFSSA, 2005. http://www.dive.afssa.fr/agritox/) or ExToxNet (ExToxNet: http://extoxnet.orst.edu/).
Preparation of pesticide-enriched feed
END (1.5 mg) was dissolved in 1 mL methanol, and then 100 µL of this solution (containing 150 µg END) was mixed with methanol (final quantity of END 150 µg). The whole resulting solution (containing 150 µg END) was mixed with acetone (9 mL) and dispersed on 10 g of the vitamin mixture powder (vitaminic mixture 200, Scientific Animal Food Engineering (Safe), France). The vitamin powder containing 150 µg END was then homogenized in a rotavapor for 30 min at 45 C to evaporate the solvent and then mixed for 50 min at room temperature with the remaining quantity of vitamin mixture (40 g) needed to make the rodent nuggets (5 kg). The vitamin powder enriched with END was then sent to the National Institute of Agricultural Research (INRA) animal feed preparation unit (UPAE INRA, Paris, France) where vitamins (1%) and mineral complements (7%) were mixed with the other components of the rodent nuggets. The presence of END was quantified by Eurofins (Nantes, France).
The final quantity was around 30 µg kg−1 of food. Control feed was prepared as described above with a mixture of methanol/acetone (1/9, v/v) but lacking pesticide. Control feed was analyzed for the presence of the endogenous pesticide (END) and also for the presence of the most common pesticides found in the environment (including the organophosphorus, OC and pyrethroid pesticides, and polychlorobiphenyls). Results showed that nuggets in control food do not contain pesticides at the detectable level (0.01 mg kg−1).
Animals
Ten-week-old female and male C57 BL/6J mice were purchased from Charles River Laboratories (Domaine des Oncins-BP 109, 69592 L’arbresle, Cedex, France). Mean body weights were 20 ± 2 g for females and males. The animals were acclimatized for 2 weeks before starting dosing. A total of 16 virgin female mice were distributed into 8 cages. In each cage, one male was placed overnight and the presence of spermatozoa was checked in the vaginal smear in the following morning. This day was connoted as gestation day 0 (GD0). At that time, pregnant females were individually housed in clean plastic cages in the laboratory animal room (23 ± 2°C; 40% relative humidity), fed on the prepared diet and tap water was allowed ad libitum. The day of parturition was considered day 0 of lactation, postnatal day 0 (PND0). Offspring of each litter were counted, sexed, and each litter was randomly reduced to six pups of equal number of sexes to maximize the lactation performance. 24 The experimental work on animals was performed in Toxalim Unit, INRA, Toulouse (France), and in accordance to its institutional ethical committees in an accredited animal house.
Experimental design
All the 16 individually housed dams with their pups were segregated into 4 groups. During gestation and lactation, the experimental dams (M) received one of the following treatments: (a) diet free of pesticide; control (C(M)); (b) diet enriched with END (30.0 µg kg−1; END (M)); (c) diet free of pesticide + oral vitamin E (α-tocopherol; 200 mg kg −1 per mouse (V (M)); and (d) diet enriched with END (30.0 µg kg−1 + oral vitamin E (200 mg kg−1 per mouse; END/V (M)). The dose of vitamin E was divided into two equal portions and given twice a week. Pups (P) of the experimental dams were designated as C (P), END (P), V (P), and END/V (P), to sign control, END-, vitamin E-, and vitamin E + END-treated groups, respectively. At the end of weaning, blood samples were taken from the facial artery of each animal (dams and pups) and added to nonheparinized and heparinized centrifuge tubes (Multivette®, SARSTED, Germany) for separating serum and plasma, respectively. Centrifugation was performed on Sigma 1K15 Bioblock Scientific (Subra, France) for 10 min at 4°C and 4000 r min−1 (1400g). Sera and plasma were kept in eppendorf tubes and stored at (−80°C). Then, the animals were killed by cervical dislocation, and the heart, spleen, liver, kidneys, testis, or ovaries were removed and weighed. Small pieces of organs were kept in 10% formalin for histopathological studies. The remaining pieces of liver were packed in aluminum sheets and kept under nitrogen atmosphere (−80°C) for certain biochemical estimations. Fractionated blood samples and specimens of internal organs were shipped frozen, in addition to formaldehyde—reserved specimens, to the National Research Centre (NRC), Cairo, Egypt—for biochemical analyses and histopathological studies.
Biochemical analyses
These included butyryl cholinesterase (BuChE), alkaline phosphatase (ALP), urea, malondialdehyde (MDA), and superoxide dismutase (SOD). Measurements were performed spectrophotometrically using an ultraviolet–visible recording 2401 PC spectrophotometer (Shimadzu, Tokyo, Japan) and in accordance with the manufacturer’s instructions given in the pamphlets. The activity of plasma cholinesterase (BuChE; EC 3.1.1.8) was measured at 405 nm and expressed in units per liter using the method followed by Knedel and Böttger. 25 ALP (EC 3.1.1) was measured in sera at 510 nm in terms of international units per liter according to Belfield and Goldberg. 26 Concentration of urea (milligram per deciliter) was determined in plasma at 550 nm using the method of Fawcett and Scott. 27 MDA and SOD activities were determined in liver tissues, based on methods published by Satoh 28 and Nishikimi et al., 29 respectively. Spectrophotometric measurements were carried out at 534 nm for lipid peroxide, in terms of MDA, and expressed in nanomole per gram tissue. SOD measurements at 560 nm were calculated in terms of units per gram liver tissue.
Histopathological studies
Liver and kidneys samples from mice mothers and pups, in addition to ovaries from mothers, were dissected and fixed in 10% neutral formalin, dehydrated in ascending grades of alcohol, and imbedded in paraffin wax. Paraffin sections (5 mm thick) were stained for routine histological study using hematoxylin and eosin. Two slides were prepared for each mouse; each slide contained two sections for each organ. Ten field areas for each section were selected and examined for histopathological changes under light microscope (160× magnification). Histopathology was carried out in the Unit of Pathological Services, Department of Pathology, National Research Centre (NRC), Cairo, Egypt.
Statistical analysis
The data were analyzed using GraphPad Prism 5 (www.graphpad.com/downloads/docs/Prism5Regression.pdf) Demo and expressed as means ± SE Paired samples (t) test was used to compare the data of the control with those of treatments.
Results
Signs of toxicity and observations
All animals were carefully observed during gestation and lactation periods. Except the END-treated group END (M), the dams of the other groups showed normal behavior in contrary to the excepted group, which recorded death of one dam as well as eating of some pups. Dams of all groups showed signs of restless during gestation period. The number of pups per each female ranged between 6 and 8 of nearly 1:1 sex ratio. This led us to limit the number of mothers at 3 per group and at 6 pups per group in the course of statistical analyses performed in this study.
Body and relative organs weights
The initial starting body weight of dams recorded approximately 20 g per animal. The mean body weight of control dams
Body and organ weights of dams and their pups treated with END, with and without supplementation of vitamin E, during gestation and lactation.a

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group; n.d.: not determined.
aValues are means ± SE; n = 3 and 6 for mother and pup groups, respectively. Statistical difference from the control.
bSignificant at p ≤ 0.05.
cHighly significant at p ≤ 0.01.
dNonsignificant difference.
The mean absolute weight of some organs from control dams recorded the following: 1.58 g (liver); 0.43 g (kidneys); 0.22 g (heart); 0.13 g (spleen); and 0.03 g (ovary). Compared with the control values, there were no significant differences in weights of kidneys and ovary from the other three tested groups. END (M) group showed significant (p ≤ 0.05) increase in liver weight (2.46 g) and significant (p ≤ 0.05) decrease of spleen weight (0.09 g).
In the offspring, the mean absolute organ weights of control group C (P) recorded the following values: 0.39 g (liver); 0.13 g (kidneys); 0.06 g (heart); and 0.06 g (spleen). The vitamin E treatment V (P) induced high elevation in weights of liver, kidneys, and heart. Similar high elevation in liver weight was observed for the pesticide alone and in conjunction with the vitamin (Table 1).
Table 2 illustrates the organ weights relative to the body weights, either for the dams or the offspring at the end of lactation period. The relative liver weight of END (M) group recorded approximately 8.74%; a value which is significantly higher than that of the control value (5.45%). In the case of pups, the relative weights of liver from END (P) and V(P) groups were also significantly higher than that of the control group (4.48%), while the treatment END/V recorded high significant (p ≤ 0.01) increase. Relative weights of kidneys, spleen, and ovary from the dams did not show any significant differences than the corresponding control treatment values. High significant decreases for the spleen relative weight of pups indirectly exposed to END, END + vitamin E, and to some extent vitamin E, were observed and accounted to 0.26%, 0.34%, and 0.43% respectively, compared with 0.67% for the control value (Table 2).
Organ weights relative to body weights of dams and their pups treated with END, with and without supplementation of vitamin E, during gestation and lactation.a

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group; n.d.: not determined.
aValues are means ± SE; n = 3 and 6 for mother and pup groups, respectively. Statistical difference from the control.
b
cSignificant at p ≤ 0.05.
dHighly significant at p ≤ 0.01.
eNonsignificant difference p ≥ 0.05.
Biochemical analyses
Due to the little amount of blood sample obtainable from the tested mouse, the estimated biomarkers were carried out on ALP in serum, BuChE, and urea in plasma, as well as MDA and SOD in liver tissues.
Alkaline phosphatase
Activity of ALP in serum of treated dams and their offspring is presented in Table 3. Control mothers C (M) recorded ALP activity of 59.53 IU L−1. In comparison, END (M), V (M), and END/V (M) recorded 83.33, 58.93, and 64.00 IU L−1, respectively. Statistical analysis between the different treatments revealed no significant differences between control and both V (M) and END/V (M) groups. Highly significant (p ≤ 0.01) elevation was observed due to treatment with END alone and in conjunction with vitamin E (Table 3).
Alteration of serum ALP activity (in international units per liter) in dams and their pups following treatment of mothers during gestation and lactation periods with END alone and in conjunction with vitamin E.

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group; ALP: alkaline phosphatase.
aSignificant at p ≤ 0.05.
cHighly significant at p ≤ 0.01.
dNonsignificant difference p ≥ 0.05.
Control pups C (P) and vitamin V (P) groups recorded nearly similar ALP activities (52.67 and 52.33 IU L−1, respectively); values which were very lower than those estimated for the groups END (P) and END/V (P). The highest elevation in ALP (129.70 IU L−1) activity was induced by exposure to the tested pesticide. Administration of vitamin E in conjunction with END limited such elevation obviously (Table 3).
Butyryl cholinesterase
END (M) treatment caused declining activity of BuChE in plasma of the tested dams; recording 2832 U L−1 compared with 3581 U L−1 for control group C (M). The difference between both values was statistically high significant (p < 0.01). Statistical analysis revealed significant decrease of cholinesterase activity due to the different tested treatments. In mice pups, BuChE activity recorded 1951 U L−1 in control group C (P); a value that had no significant differences with those obtained from the other treatments (Table 4).
Alteration of plasma BuChE activity (unite per liter) in dams and their pups following treatment of mothers during gestation and lactation periods with END alone and in conjunction with vitamin E.

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group; BuChE: butyryl cholinesterase.
aSignificant at p ≤ 0.05.
cHighly significant at p ≤ 0.01.
dNonsignificant difference p ≥ 0.05.
Urea
Concentration of urea in plasma of control dams C (M) recorded 43.87 mg dL−1; a value that nearly equaled those of V (M) and END/V (M) groups. Concentration of urea in END (M)-treated group (52.67 mg dL−1) was significantly higher (p ≤ 0.01) than that of control group value. In control mice pups, concentration of urea was found to be 32.00 mg dL−1, which was very close to that of V(P) group, but significantly (p ≤ 0.01) lower than that of END (P) and END/V groups (51.33 and 42.33 mg dL−1, respectively; Table 5).
Alteration of urea concentration (in milligram per deciliter) in plasma of dams and their pups following treatment of mothers during gestation and lactation periods with END alone and in conjunction with vitamin E.

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group.
aSignificant at p ≤ 0.05.
bHighly significant at p ≤ 0.01.
cNonsignificant difference p ≥ 0.05.
MDA activity
MDA concentration was measured in liver tissues of the experimental mice subjected to different treatments. Its concentration in control mother group C (M) was found to be 81.17 nmol g−1, which was insignificantly different from that of V (M) or END/V (M) treatments. END (M) treatment induced high significant (p ≤ 0.01) elevation accounting to 104.30 nmol g−1 (Table 6).
Alteration of liver MDA concentration (in nanomole per gram tissue) in dams and their pups following treatment of mothers during gestation and lactation periods with END alone and in conjunction with vitamin E.

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group; MDA: malondialdehyde.
aSignificant at p ≤ 0.05.
bHighly significant at p ≤ 0.01.
cNonsignificant difference p ≥ 0.05.
In pups, concentrations of MDA recorded 49.90, 65.67, 43.60, and 56.33 nmol g−1, respectively, in C (P)-, V (P)-, END (P)-, and END/V (P)-treated groups. Statistical analysis showed significant difference (p ≤ 0.05) between value of C (P) and END/V (P) and high significant difference (p ≤ 0.01) between C (P) and END (P)(Table 6).
SOD activity
Alteration of SOD activity in the dams and their offspring following administration of END, with and without vitamin E, to the mothers during gestation and lactation periods is presented in Table 7.
Alteration of liver SOD (in units per gram tissue) in dams and their pups following treatment of mothers during gestation and lactation periods with END alone and in conjunction with vitamin E.

M: dams; P: pups; C: control; END: endosulfan-treated group; V: vitamin E-treated group; END/V: endosulfan + vitamin E-treated group; SOD: superoxide dismutase.
aSignificant at p ≤ 0.05.
cHighly significant at p ≤ 0.01.
dNonsignificant difference p ≥ 0.05.
In the dams, END (M) and END/V (M) treatments induced decline of SOD activity accounting to 145.0 and 175.70 U g−1 tissue, respectively, achieving high significant differences (p ≤ 0.01) compared with the control result (208.10 U g−1; Table 7).
In pups, the activity of SOD recorded 188.2, 120.00, 186.20, and 135.70 U g−1, respectively, for C (P), END (P), V (P), and END/V (P) treatment groups. Statistical analysis showed high significant (p ≤ 0.01) differences between values of C (P) and END (P) and C (P) and END/V(P) (Table 7).
Histopathological examination
Liver
Examination of the sections of liver of control dams and that treated with vitamin E showed normal architecture of a hepatic lobule. The central vein lies at the center of the lobule and surrounded by the hepatocytes with strongly eosinophilic granulated cytoplasm and distinct nuclei. Between the strands of hepatocytes, the hepatic sinusoids are shown
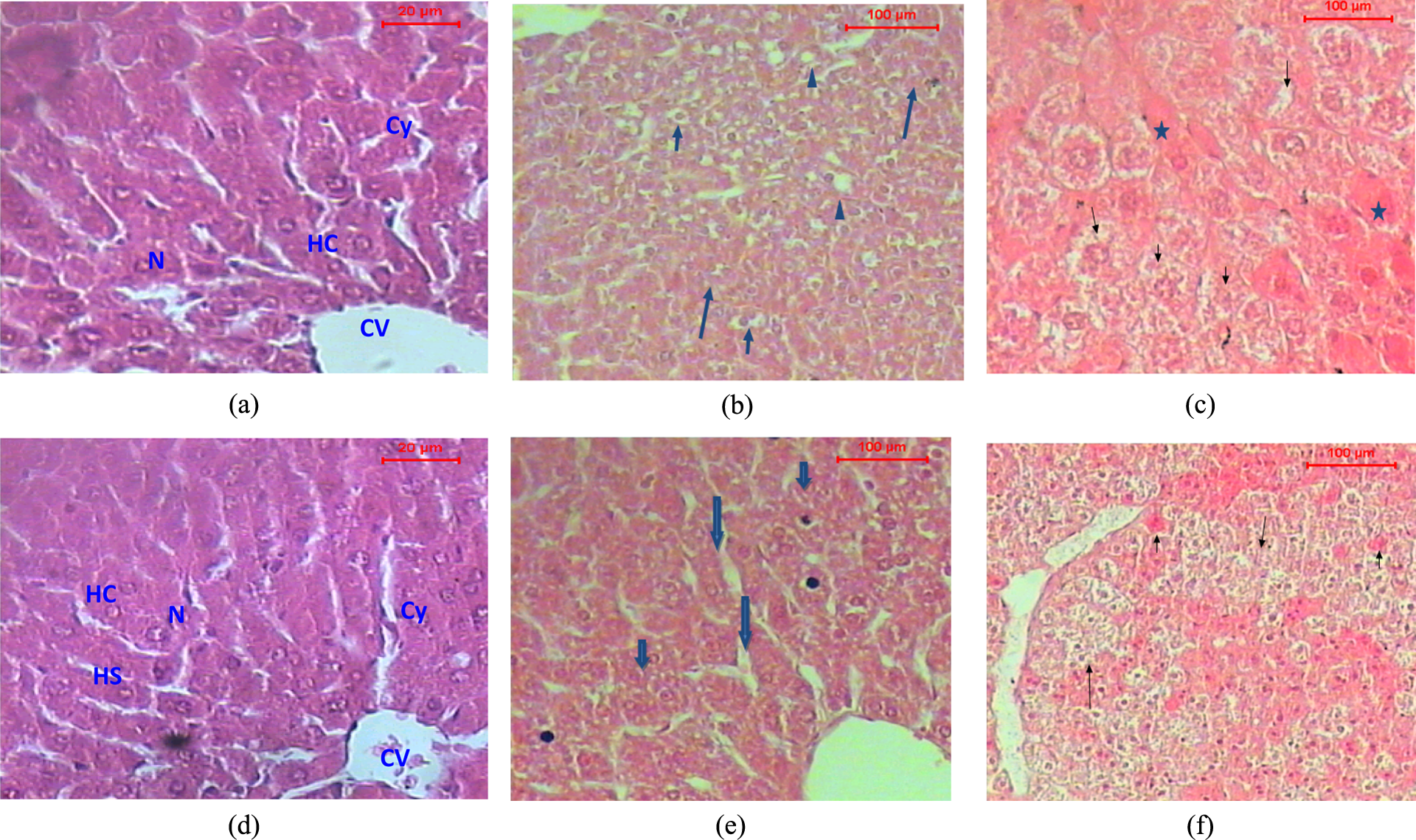
Liver sections from dams and their offspring (without sex differentiation) exposed to END, with and without vitamin E, during gestation and lactation. (a) Control C (M): A section of liver of a control dam showing the architecture of a hepatic lobule. The central vein (CV) lies at the center of the lobule surrounded by the hepatocytes (HC) with strongly eosinophilic granulated cytoplasm (Cy) and distinct nuclei (N). Between the strands of hepatocytes, the hepatic sinusoids are shown (HC). (H&E stain; ×150). (b) END (M): A section of liver of dam treated with END shows disturbance of the hepatic lobule associated with the hydropic degeneration (arrows), hemorrhagic areas (arrow heads), fatty change, and foci of necrotic cells (H&E stain; ×150). (c) END/V (M): A section of liver of dam treated with END + vitamin E shows disturbance of the hepatic lobule with hydropic degeneration (arrows). The cytoplasm appeared granulated (asterisk); H&E stain; ×150). (d)C (P): A section of liver of control offspring showing the architecture of a hepatic lobule. The central vein (CV) lies at the center of the lobule surrounded by the hepatocytes (HC) with strongly eosinophilic granulated cytoplasm (Cy) and distinct nuclei (N). Between the strands of hepatocytes, the hepatic sinusoids are shown (HS; H&E stain; ×150). N.B.: The effect of vitamin E treatment V (P) resembled that of control. (e) END (P): A section of liver of offspring treated with END shows lobules that appeared more or less like normal and hydropic degeneration (arrows), as well as congested liver sinusoids (arrowhead; H&E stain; ×150 magnification). (f) END/V (P): A section of liver of offspring treated with END + vitamin E shows disturbance of the hepatic lobules. The hydropic degeneration in the hepatocytes and hemorrhagic areas were also noticed (arrows; H&E stain; ×150 magnification). END: endosulfan; M: mice; P: pups; V: vitamin E; H&E: hematoxylin and eosin. Note: The histopathological architecture of liver sections from vitamin treatments resembled those from control treatments.
Microscopic investigation of the liver of dams treated with END ((END (M)) showed the disturbance of the hepatic lobules that were associated with the hydropic degeneration, fatty change, and foci of necrotic cells (Figure 1(b)). Administration of vitamin E in conjunction with END (END/V (M)) induced similar effects, where the cytoplasm of the hepatocytes assumed pale pink appearance such as the ground glass hepatocytes. In other hepatocytes, the cytoplasm assumed granulation (Figure 1(c)).
Sections of the liver of control offspring, and that from vitamin E treatment, showed the normal architecture of the hepatic lobules Figure 1(d)). Histopathological examination of liver of offspring from END treatment (END (P)) showed the hepatic lobules that appeared more or less normal. Congested hepatic sinusoids were found. In some cases, the foci of the necrotic hepatocytes and dilated hepatic sinusoids were also seen (Figure 1(e)).
Sections of liver of offspring belonging to the treatment of END + vitamin E (END/V (P)) showed the disturbance of the hepatic lobules. The hydropic degeneration in the hepatocytes and hemorrhagic areas were also noticed Figure 1(f)).
Kidney
Microscopic examination of the sections of kidney of control dams, and that of vitamin E treatment, showed normal structure of the glomeruli and the renal tubules. The distal convoluted tubules could be differentiated from the proximal convoluted tubules as having larger and well-defined lumina and less affinity to stain (Figure 2(a))

Kidney sections from a dam and an offspring exposed to END, with and without vitamin E, during gestation and lactation. (a) C (M): A section of kidney of control dam showing normal structure of the glomerulus (G) and the renal tubules. DCT could be differentiated from the PCT as having larger and well-defined lumina, less affinity to stain (H&E stain; ×150). N.B.: The effect of vitamin E treatment V (M) resembled that of control. (b) END (M): A section of kidney of dam treated with END shows the degenerative and detachment of the epithelial of the renal tubules (arrows), accumulation of inflammatory cells in the interstitial spaces (asterisk), and thickness of the blood vessel (arrowheads; H&E stain; ×150). (c) END/V (M): A section of kidney of dam treated with END + vitamin E shows hypertrophy of the glomeruli (asterisks) and narrow urinary space (arrowheads). Many of the renal tubules show epithelial detachment (arrows; H&E stain; ×150). (d) C (P): A section of kidney of control offspring showing normal structure of the glomeruli (G) and the renal tubules. DCT could be differentiated from the PCT as having larger and well-defined lumina, less affinity to stain (H&E stain; ×150). (e) END (P): A section of kidney of offspring treated with END shows normal structure of the glomeruli. Some of the renal tubules show epithelial detachment (arrow) or degeneration (arrowhead) and interstitial hemorrhagic (asterisk; H&E stain; ×150). (f) END/V (P): A section of kidney of offspring treated with END + vitamin E shows disturbance of the kidney structure. Hydropic degeneration (arrows) and glomerulus degeneration (asterisk) and hemorrhagic areas (arrow heads; H&E stain; ×150 magnification). END: endosulfan; M: mice; P: pups; V: vitamin E; H&E: hematoxylin and eosin; PCT: proximal convoluted tubules; DCT: distal convoluted tubules. Note: The histopathological architecture of kidney sections from vitamin treatments resembled those from control treatments.
Microscopic examination of kidney of dams treated with END (M) showed hypotrophy of the glomeruli, wide urinary spaces, and detachment of the epithelial of the renal tubules and interstitial hemorrhage (Figure 2(c)).
Sections of kidney of control offspring and that from dams treated with vitamin E showed normal structure of the glomeruli and the renal tubules (Figure 2(d)). The pups from END treatment (END (M)) showed normal structure of the glomeruli, but some of the renal tubules showed epithelial detachment or degeneration and interstitial haemorrhagic (Figure 2(e)). Sections of kidney of offspring belonging to END/V (P) treatment showed disturbance of the kidney structure. The hydropic degeneration in the renal tubules, glomeruli degeneration, and hemorrhagic areas were seen (Figure 2(f)).
Ovary
Ovary sections from dams exposed to END, with and without vitamin E, during gestation and lactation periods showed normal ovarian follicles and cortical stroma (Figure 3(a)). The effect of vitamin E treatment V (M) resembled that of control. A section of the ovary of a dam treated with END showed degenerative of some ovarian follicles and normal cortical stroma (Figure 3(b)). The ovary of treated dams with the vitamin + END (END/V (M)) showed normal ovarian follicles and cortical stroma (Figure 3(c)).

Ovary sections from dams exposed to END, with and without vitamin E, during gestation and lactation: (a) C (M): A section of the ovary of control dam showing normal ovarian follicles (arrowhead) and cortical stroma (arrow; H&E stain; ×150). (b) END (M): A section of the ovary of dam treated with END showing degenerative effects of some ovarian follicles and normal cortical stroma (H&E stain; ×100). (c) END/V (M): A section of the ovary of dam treated with END + vitamin E showing normal ovarian follicles and cortical stroma (arrow; H&E stain; ×100). END: endosulfan; M: mice; P: pups; V: vitamin E; H&E: hematoxylin and eosin. Note: The histopathological architecture of ovary sections from vitamin treatments resembled those from control treatments.
Discussion
Pregnant women are a special risk group, given the findings showing increased risk of childhood acute lymphocytic leukemia when women use pesticides in the home and garden during pregnancy. 30 It is documented that the fetus is more vulnerable to the toxic effects of a number of environmental exposures than are children or adults. 31 Highly lipophilic compounds are expected to cross the placenta and reach the fetus during pregnancy. 32,33 Also, there are many factors involved in the transfer of chemicals into the milk and subsequently to the suckling neonate, including lipophilicity, ionization, and maternal plasma protein binding, which can all influence transport of the compound during lactation. 34 END, as a highly lipophilic compound, is expected to transfer from the dam to the pup at the plasma/milk membranes. However, the rate of transfer from the dam to the pup at the plasma/milk membranes could also be modified by plasma protein binding or active transport at the plasma/milk membrane. 32,35,36
In toxicological studies, organ and relative organ weights are important criteria for evaluation of organ toxicity. Generally, increase or decrease of organs weights than normal may be considered as a sign of toxicity. 37 Significant deviation in body or organs weights than the normal values may refer to developmental effects of END. 1,12,18
END is a contact hepatotoxin that is readily absorbed into an organism through its stomach, lung, and even through the skin. 38 It affects blood biochemistry and hematological values. 39
ALPs are a group of enzymes found primarily in the liver (isoenzyme ALP-1) and bone (isoenzyme ALP-2). There are also small amounts produced by cells lining the intestines (isoenzyme ALP-3), the placenta, and the kidney (in the proximal convoluted tubules). What is measured in the blood is the total amount of ALP released from these tissues into the blood. The primary importance of measuring ALP is to check the possibility of bone disease or liver disease. Since the mucosal cells that line the bile system of the liver are the source of ALP, the free flow of bile through the liver and down into the biliary tract and gallbladder are responsible for maintaining the proper level of this enzyme in the blood. When the liver, bile ducts, or gallbladder system is not functioning properly or is blocked, this enzyme is not excreted through the bile and ALP is released into the blood stream. Thus the serum ALP is a measure of the integrity of the hepatobiliary system and the flow of bile into the small intestine. An increased serum ALP may be due to congestion or obstruction of the biliary tract, which may occur within the liver, the ducts leading from the liver to the gallbladder, or the duct leading from the gallbladder through the pancreas that empty into the duodenum (small intestine). Any of these organs (liver, gallbladder, pancreas, or duodenum) may be involved. 40 Therefore, high elevation of ALP activity in serum of END-treated dams indicate hepatotoxic effects of the tested dose, as well as extension of this adverse effect to the offspring; reflecting their high vulnerability. Coadministration of vitamin E alleviated such observed effect for the dams, while alleviation occurred to some extent for the pups (Table 3). Similar results were recently reported for the effect of vitamin E on mice exposed to atrazine herbicide. 41
BuChE, also known as pseudocholinesterase, has been recognized as an enzyme that hydrolyzes choline esters. It has already been mentioned that ChE is synthesized mainly in hepatocytes and secreted into the blood stream. 42 ChE activity is reduced in liver dysfunction due to reduced synthesis, in contrast to other serum enzymes associated with the clinical assessment of liver function whose activities increase as a result of increased release from their cellular sources following cell membrane damage. 43 In this respect, changes in ChE activity reflect the changes in hepatocellular functions and have been regarded as sensitive indicators of the diminished synthetic capacity of the hepatic parenchyma. 44 Our findings revealed that dams treated with END had exercised high significant decline of ChE (e.g. BuChE), while alteration of ChE in the suckling pups, compared with the control result, was found insignificant (Table 4); a result that agrees with Zhao et al. 45 and coincides with those previously obtained on suckling rats with the insecticide chlorpyrifos. 46 On the other hand, decreases in serum and tissue acetylcholinesterase activity are commonly reported in the END-treated animals. 47 –49
Both the dams and their pups showed high significant elevation of urea concentration following treatment with END. Coadministration of vitamin E alleviated such observed effects to some extent (Table 5). Urea is the major nitrogen-containing metabolic product of protein metabolism. Xenobiotic compounds inhibit the incorporation of amino acid into protein and cause increase of urea levels. 50
Toxicity of many xenobiotics is often associated with the production of oxygen-free radicals, more generally known as “reactive oxygen species” (ROS), which are not only toxic themselves but are also implicated in the pathophysiology of many diseases. 51 Recent studies indicate that pesticide intoxication produces oxidative stress by the generation of free radicals and by inducing tissue lipid peroxidation in mammals and other organisms. 52,53 There are several pathways by which the pesticide is thought to induce oxidative stress. It inhibits the mitochondrial electron transfer chain reaction, leading to accumulation of semi-ubiquitous, which enables it to transfer one electron (e–) to molecular oxygen to form superoxide radicals. 54 Further, it may also interfere with cellular antioxidant defense system via alteration in activities of antioxidant enzymes, namely, SOD and catalase (CAT) and status of glutathione (GSH). 55
Hincal et al. 56 reported the oxidative stress-inducing effects of END, with an increase of lipid peroxidation (LPO) and a significant alteration in GSH redox cycle in cerebral and hepatic tissues of rats. Erythrocytes exposed to 1 ppm END showed significant decrease in activities of SOD and CAT, while LPO and glutathione-S-transfarase were increased in comparison with the control values. 57 In vitro studies, the latter investigators found that vitamin E alleviated oxidative stress induced by END in rat erythrocytes. Previous studies in our laboratory on animals, using different insecticides including chlorpyrifos, methomyl, and atrazine revealed increase of LPO and decrease of SOD levels following the insecticidal treatments. 36,41,46,58 –60 Specifically, coadministration of vitamin E in conjunction with atrazine normalized the values of SOD and MDA in comparison with those of control values. 41 Such results corroborate our findings on pregnant and lactating dams and their offspring treated with END with and without vitamin E; suggesting that vitamin E acts as an effective antioxidant for END toxicity in reducing oxidative stress burden. Also, vitamin C was found to reduce the toxicity of END in liver of rabbits. 61 To the best of our knowledge, the use of vitamin E against END-induced toxicity to pregnant and/or lactating mice is investigated by the present study for the first time. Only it has been reported that prenatal and lactational exposure to END modifies expression and release of pituitary hormones and both nitrosative and oxidative stress in pubertal male rats. 62
It has been long recognized that major contributors to nonenzymatic protection against LPO are vitamin E and vitamin C, well known free radical scavenging. 63 Vitamin E is a lipid soluble, chain-breaking antioxidant playing a major protective role against oxidative stress and prevents the production of lipid peroxides by scavenging free radicals in biological membranes. Previous investigators reported that administering vitamin E may be useful in controlling the toxic effect of insecticides and chemicals. 23,64
To assess the effect of END on antioxidative stress enzymes (e.g. MDA and SOD) in a “quantitative manner”, we calculated the percentage of change in END-treated groups relative to control untreated groups, either for mothers or offspring. Based on data in Table 6, changes in MDA were found to be 28.5 and 31.6% for dams and offspring, respectively. Changes in SOD activity (refer to data in Table 7) accounted to −30.3 and −36.2% for dams and offspring, respectively. Such findings clearly demonstrate that pups have exercised degree of effects nearly equaled to that their mothers were affected.
On the other hand, we compared the results of MDA and SOD in END/vitamin E groups with the results of control groups; to assess the ameliorative effect of vitamin E, based on calculating what the so-called “ameliorative index” (AI). As AI was approaching “1”, the amelioration reaches high degree of normalization to the normal control value. The obtained results revealed that AI of MDA accounted to a maximum value (approximately 1.0), either for mothers or pups (refer to data in Table 6). The AI of SOD equaled 0.84 and 0.72, respectively for mothers and pups (refer to data in Table 7). Here again, the results reveal that administration of vitamin E in conjunction with END has achieved high to full degree of normalization for the measured antioxidative stress enzymes, either in experimental dams or their pups at nearly similar degrees. The analysis of our data as depicted above may clearly indicate that the pups have suffered the toxic effect of END indirectly from their mothers through placental and/or breast-feeding routes. They also took benefit from administration of vitamin E to their mothers. Such findings may be considered as a new addition to END transformation via placenta and lactation, especially at a very low dose characterized as “safely acceptable” (i.e. ADI).
Similar estimations for percentage of change in cholinesterase enzyme (Table 4) due to END treatment indicate decline of activity accounting to −18.1 and −11.8% for mothers and pups, respectively. Coadministration of vitamin E with END resulted in approaching the enzyme activity to that of the normal value; where the estimated AI value equaled 0.90 and 0.94 for dams and their offspring, respectively.
Exposure of the dams to END at the equivalent dose to its ADI had resulted in noticeable histopathological effects on liver, kidney, and ovary as illustrated in Figures 1 –3. The most pronounced effects of END on liver were represented by disturbance of the hepatic lobule associated with the hydropic degeneration, hemorrhagic areas, fatty change, and foci of necrotic cells. Liver sections from the pups showed lobules that appeared more or less normal and hydropic degeneration, as well as congested liver sinusoids.
In kidney sections of END-treated mice, degenerative and detachment of the epithelial of the renal tubules, accumulation of inflammatory cells in the interstitial spaces, and thickness of the blood vessel were manifested. In kidney sections of pups, some of the renal tubules showed epithelial detachment or degeneration and interstitial haemorrhagic. The dams treated with END showed degenerative effects to some ovarian follicles.
The above-mentioned observations indicated marked changes in the overall histoarchitecture of the examined organs in response to END, which could be due to its toxic effects primarily by the generation of ROS, causing damage to the various membrane components of the cell. 56,57 Indeed, treatments received vitamin E with END showed some histopathological changes in the examined organs but were occurred at little severity compared to those of the pesticide treatment alone. This may be due to the vitamin dose that was not sufficient enough to alleviate the adverse effect of END completely. However, vitamin E was obviously able to alleviate the biochemical alterations induced by END. To the best of our knowledge, the literature offers no data about biochemical and histopathological effects of END on pups via gestation and lactation.
It has been reported that END can cause toxic effects in almost all tissues of both humans and animals, including the liver, lung, central nervous system, genital system, pancreas, and so on. 5,23,38,39 The mainly effected organ in END toxicity is the liver. At necropsy, hemorrhages can be seen in livers in acute poisoning in cattle. 5 Hemorrhages and infiltration of inflammatory cells that localized around the central vein and portal space can be seen. Interlobular mononuclear inflammatory cells among vacuolated hepatic cells and dilated congested sinusoids were reported. Apoptotic activity in liver cells increased in livers by END exposure. 49
Kidney changes in END poisoning are dose dependent. Tubular dilation, hydropic degeneration in tubular epithelium, and hemorrhage in the cortical and medulla part of the kidney were reported. 65 The effect of END is mainly on the proximal convoluted tubule cells. 66 –68 Degeneration and necrosis in kidneys may be thought that oxidative stress plays a role in changing configuration of cell membrane and seem to account for the morphologic alteration of kidney. 67
In the course of our study, some of the above documented histopathological effects were observed in the examined tissues of the dams treated with END and also in tissues of their pups. However, the dose of END used in the present investigation may be very lower than doses used in many previous studies. 23,49,61,62
Conclusion
In light of the results of the present study, it can be concluded that exposure of adult female mice to END through food at its ADI level can induce hepatic, renal oxidative stress, and histopathological effects. Since the experimental treatments were conducted on dams during gestation and lactation, the obtained results reveal possibility of placental and lactational transfer of END intoxication to the offspring. Supporting our conclusion is that highly lipophilic compounds, such as END, are expected to cross the placenta and reach the fetus during pregnancy. 32,33 Also, they can transfer to the suckling neonate through breast-feeding. 34,35 Suckling neonate seemed to be more vulnerable to toxic effects than their mothers. Coadministration of vitamin E showed noticeable ameliorating effects, either to mothers or pups. The findings may support the need to further investigate the protective effects of antioxidants against pesticide intoxication especially during pregnancy and breast-feeding as well as effects on newborn child.
Highlights Exposure of mice dams during gestation and lactation to END at ADI induced adverse effects to the dams and their pups. The pups were affected similarly as their mothers did due to their indirect exposure. Supplementation of vitamin E + END diminished oxidative stress caused by it. The results demonstrate transfer of END to the pups via placenta and lactation.
Footnotes
Acknowledgment
The authors thank Caroline Sommer, Florence Blas-y-Estrada, and Roseline Gautier from the National Institute of Agricultural Research (INRA, France). We also thank Prof. Dr Abdel-Razik H. Farrag, Department of Pathology, National Research Centre, Dokki, Cairo, Egypt, for reading the histopathological slides.
Conflict of interest
The authors declared no conflicts of interest.
Funding
The authors thank the Academy of Scientific Research and Technology, Egypt, and National Institute of Agricultural Research, France, for supporting this study within the REF BHC IMHOTEP 2011 Project No. 25382 YG.
