Abstract
Tobacco use is the leading preventable cause of death. The cytotoxicity of cigarette smoke condensate (CSC), the particulate fraction of cigarette smoke without the vapor phase, has mostly been tested in short-term in vitro studies lasting from a few hours to a few days. Here, we assessed the toxicity of CSCs from 2 reference cigarettes, 3R4F and CM6, using a primary human small airway epithelial (PSAE) cell line by quantifying adenosine 5′-triphosphate (ATP), 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy-methoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS), total glutathione (reduced glutathione [GSH] + oxidized glutathione [GSSG]), and lactate dehydrogenase (LDH) release over the course of 28 days. The CSCs, 0.3 to 10 μg/mL, promoted cell proliferation at 120 hours of exposure, but demonstrated cytotoxicity at days 14 and 28. Interestingly, CSCs, 0.3 to 3 μg/mL, showed a cell death effect at day 14 but induced cell proliferation at day 28. Consistently, transformation associated with morphological changes began by day 14 and the transformed cells grew dramatically at day 28. The LDH assay appeared to be sensitive for assessing early cell damage, whereas the ATP, MTS, and GSH assays were more suitable for determining later stage CSCs-induced cytotoxicity. The ATP assay showed greater sensitivity than the MTS and GSH assays. We also assessed the toxicity of CSCs in an human Telomerase Reverse Transcriptase (hTERT)-immortalized Barrett esophagus cell line (CP-C). The CP-C cells demonstrated dose- and time-dependent cytotoxicity over the course of 28 days but displayed higher resistance to CSCs than PSAE cells. This study demonstrates that CSCs cause cytotoxicity and induce transformation related to cell resistance and cell invasion properties.
Keywords
Introduction
Cigarette smoking harms nearly every organ in the body and is the leading cause for premature death in the world. Tobacco smoke contains over 5000 chemical constituents 1 and 93 of the chemicals are harmful or potentially harmful in humans. 2 Cigarette smoke has a very strong oxidative potential, resulting in cell damage including cell integrity damage, reactive oxidative stress, inflammation, and damage to DNA, RNA, proteins, and lipids. Both the gas and particulate phases of cigarette smoke contain high levels of free radicals. 3 –6
The cytotoxic properties of cigarette smoke lead to chronic tobacco-related malignant and nonmalignant diseases. 7 In vitro cytotoxicity assays have been widely used to investigate the effect of chemicals on cells and tissues. Cell-based in vitro systems have the advantage of both a more rapid screening of samples and a relatively lower cost than studies in human and animal models. 8 Due to cells diversity and the complexity of chemical toxic properties, the predictivity of cytotoxicity assays may be different for different compounds or compound mixtures, and there may also be variable responses in different cell types. Indeed, it has been reported that the relative potency of toxicity of cigarette smoke condensate (CSC) from a variety of cigarettes is different when various types of assays and cell types are used. 9,10 These studies included several cytotoxicity assays, such as lactate dehydrogenase (LDH) assay, neutral red uptake, and MTT, in several cells lines, A549, CHO cells, human lung microvascular endothelial cells (HMVEC-L), and small airway epithelial cell (SAEC). The CSC includes the particulate fraction, but not the vapor phase, of cigarette smoke. Cytotoxicity can be examined by cell viability assays, such as adenosine 5′-triphosphate (ATP), 11,12 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetra-zolium (MTS), 12 and GSH (total glutathione [reduced glutathione, GSH + oxidized glutathione, GSSG]) assays, 13 as well as cell damage assays, such as the LDH assay. 14 Almost every cytotoxicity assay has its own strengthens and limitations. As a result, a battery of cytotoxicity assays is generally recommended. The sensitivity, reproducibility, ease of use, and compatibility with physiologically relevant cell lines are the important factors in selecting assays.
Tobacco smoke, as well as many of its individual constituents, has demonstrated extensive toxicity in in vitro (cell culture and tissues) studies. In most of these studies, when cells were treated with tobacco smoke condensates or smoke constituents at high doses, cytotoxicity (measured as cell death) was observed within a few hours to a few days. 10,15,16 These studies contribute to understanding toxic end points resulting from cigarette smoke exposures on various cells and tissues. However, an obvious limitation of these short-term studies is that they do not reflect the repeated, long-term nature of tobacco smoke exposures in most consumers of tobacco products. In addition, the very high concentrations often used in short-term studies may not be achieved in vivo. Studies on longer term exposure (14 and 28 days) to cells to CSC may provide additional information on the harm of tobacco smoke and potentially on biomarkers of harm. Other reports of tobacco smoke cytotoxicity with longer duration exposures in human and human-derived cell lines were not located.
The small airway and esophagus receive exposure to tobacco smoke. Direct and repetitive exposures to toxic chemicals, including carcinogens in tobacco smoke, result in cytotoxicity as well as genetic alterations and cellular aberrations in epithelium, which eventually lead to malignant transformation. 17 –21 In this study, the cytotoxic effects of CSCs in a primary small airway epithelial (PSAE) cell line and hTERT-immortalized esophageal epithelial cells (CP-C) were examined by the 4 cytotoxicity assays. Primary cells that are isolated directly from human may be more physiologically relevant, whereas transformed cell lines are more suitable for longer time periods. 22 Inclusion of these 2 cell lines also permitted assessing effects at various transformational stages.
Cigarette smoke condensates (CSCs) from various types of cigarettes or CSCs from the same cigarette produced with different smoking machine regimens may have different toxic effects. The toxicity of CSCs from different cigarettes and the toxicity of many components of the CSCs have been tested previously 23 –25 ; however, fewer studies have reported the toxicities of CSCs generated using different smoking regimens from the same cigarette. 10
This study was conducted to (1) compare the suitability and sensitivity of 4 commonly used in vitro cytotoxicity assays (LDH, ATP, MTS, and GSH assays) in assessing the cytotoxicity of CSCs in PSAE and CP-C cells; (2) to assess systematically cytotoxicity induced by exposure to CSC at a wide range of concentrations over the course of 28 days in human cell lines and to identify cell-specific response to CSCs in these 2 cell lines; and (3) to compare the relative potency of CSCs from 2 reference cigarettes, CM6 and 3R4F, produced by machine smoking using either International Organization for Standardization (ISO) or Health Canada Intense (Intense) regimens. The 3R4F reference cigarette was developed under the organizational structure of the University of Kentucky Tobacco and Health Research Program. The CM6 is a CORESTA monitor cigarette. The 3R4F cigarettes are manufactured to contain an exact blend containing certain amounts of burley, flue-cured, oriental, and reconstituted tobacco. In contrast, CORESTA CM6 cigarettes are made from only flue-cured lamina tobacco only, containing no tobacco stems, reconstituted tobacco sheet or expanded tobacco, and no nontobacco ingredients. Comparative smoke analysis of the 2 cigarettes has shown differences in the content of nicotine free dried particulate matter, carbon monoxide, nicotine, and carbonyl compounds. 26,27 These cigarettes can also differ in the concentration of 5 minor alkaloid compounds that have been examined. 28
Materials and Methods
Cigarette Smoking Condensate
Four CSCs, obtained from the Centers for Disease Control and Prevention, were dissolved in dimethyl sulfoxide (DMSO) and stored in −80°C until use. Detailed information about cigarette collection, smoking conditions, and preparation of CSCs was described previously. 29 The CSCs are designated 3R4F Intense, 3R4F ISO, CM6 Intense, and CM6 ISO (ISO or Health Canada Intense (Intense) regimens). Kits for CellTiter-Glo Luminescent Cell Viability assay (ATP, G7570), CellTiter 96 AQueous Non-Radioactive Cell Proliferation assay (MTS, G5421), and GSH-Glo Glutathione assay (GSH V6911) were purchased from Promega Corporation (Madison, Wisconsin). Monosodium pyruvate (P8574) and nicotinamide adenine dinucleotide (NADH; N4505) for the LDH assay were purchased from Sigma-Aldrich (St Louis, Missouri).
Cell Culture
Primary human small airways epithelial cells were purchased from ATCC (PCS-301-0100). The cells were cultured in 100-mm dishes (BD Falxon Tissue Culture dish; Primia, Durham, NC) in Small Airway Epithelial Cells growth medium as recommended by the supplier. The CP-C cells purchased from ATCC (ATCC CRL-4029, Manassa, VA) were derived from a region of high-grade dysplasia in a male patient with Barrett esophagus. These cells were immortalized by transduction with a retroviral expression vector, plXSN-hERT. The CP-C cells were cultured in the complete growth medium as recommended by the supplier. Medium was changed every 3 days with new CSC for the longer term studies.
Cytotoxicity Assays
Cells were plated in 96-well plates at a density of 3000 cells per well; for each cell, agent, and treatment condition, 3 identical replicate wells were set up, treated, and analyzed in parallel. Following overnight attachment, cells were treated with 4 CSCs at concentrations of 0.3, 1, 3, 10, 30, 100, or 300 μg/mL for 6, 24, 72, 120 hours, 14, or 28 days. Concentrations were selected to permit assessments at low and higher levels of CSC. This dose range is similar to those previously reported in other in vitro studies, 30,31 and these concentrations may be reachable in smokers depending on number of cigarettes smoked and length of time smoking. 32,33 Control cells received only the solvent (DMSO). The DMSO used in this study did not show effect on cell viability. Each assay was conducted on 3 independent experiments and there were 3 replicates for each concentration for each independent assay, including the vehicle controls.
Adenosine 5′-Triphosphate Assay
The CellTiter-Glo Luminescent Cell Viability assay is a homogeneous method to determine the number of viable cells in culture based on quantitation of the ATP present. The generation of a luminescent signal is proportional to the amount of ATP present, and the amount of ATP is directly proportional to the number of cells present in culture. Cells were cultured and treated with CSC in plates with 100 μL medium in each well. The ATP levels were measured by adding 100 μL of CellTitre-Glo reagent (Promega). The plate was briefly agitated and then incubated in the dark for 10 minutes at room temperature before measuring luminescence in a Chameleon Multilabel Detection Plateform (Lablogic Systems, Inc, Brandon, Florida). Average background luminescence readings from wells containing phosphate-buffered saline alone were subtracted from the sample readings. The half maximal inhibitory concentration (IC50) values were determined from the concentration curves generated using GraphPad Prism software (GraphPad Software, Inc, La Jolla, California).
The MTS Assay
This is a colorimetric assay in which the amount of color produced is directly proportional to the number of viable cells. After CSC exposure, 20 μL of MTS labeling reagent was added to each well and incubated for 4 hours. Absorbance at 490 nm was measured 2 hours after the addition of 20 μL of MTS reagent/well in a Chameleon Multilabel Detection Plateform. The IC50 values were determined from the concentration curves generated using GraphPad Prism software.
The GSH/GSSG-Glo Assay
This is a luminescence-based system for the detection and quantification of total glutathione (GSH + GSSG), GSSG, and GSH/GSSG ratio in cultured cells. Light from luciferase is dependent on the amount of luciferin formed, which is, in turn, dependent on the amount of GSH present. This makes the luminescent signal proportional to the amount of GSH. Cells were cultured and treated with CSCs in plates with 100 μL medium in each well. After the culture medium was removed from the wells, the reagents were added to the well according to the manufacturer’s instruction. Luminescence was read in a Chameleon Multilabel Detection Plateform. The IC50 values were determined from concentration curves generated using GraphPad Prism software.
Lactate Dehydrogenase Assay
This assay measures LDH leakage from cells into the culture medium. The assay was performed according to a published method. 34 Briefly, at a designated time, 20 μL of culture medium was transferred to a clear plastic 96-well plate. Triton X-100 was added to the remaining medium to a final concentration of 1% to lyse the cells. After cell lysis, 20 μL of the culture medium was transferred to empty wells of the 96-well plate. The measurement of LDH activity was started by adding 200 μL of substrate mixture (containing 0.54 mmol/L NADH, 4.5 mmol/L sodium pyruvate, 81.3 mmol/L Tris, 203.3 mmol/L NaCl, pH 7.2) to each well of the 96-well plate. The rate of conversion of NADH to NAD+ was measured over a 5-minute period by monitoring absorbance at 340 nm using a Quant Microplate Reader (BioTek, Winooski, Vermont). Cytotoxicity was determined as the percentage of LDH activity in the cell culture medium prior to lysis versus after lysis.
Morphology
Phase-contrast micrographs were taken of controls and treated cultures for morphological observations using a Nikon eclipse TS100 microscope (Nikon Instruments, Inc, Meville, New York).
Invasion Assay
BD BioCoat Matrigel Invasion Chambers (BD Biosciences, Bedford, Massachusetts) were used to study cell invasion properties in vitro. The chambers consisted of a BD Falcon TC Companion Plate with Falcon cell culture inserts that contains an 8-µm pore size membrane with a thin layer of matrigel basement membrane matrix. Briefly, the chambers were removed from −20°C storage and allowed to come to room temperature. Medium (0.5 mL insert and 0.5 mL well) was added to rehydrate the matrigel for 2 hours at 37°C, 5% CO2. The media was removed from the wells and replaced by media with serum (chemo-attractant), followed by 0.5 mL of untreated PSAE, 0.3 or 1.0 μg/mL CSC-treated cell suspension (2.5 × 104 cells) to the matrigel inserts. The chambers were incubated in a tissue culture incubator at 37°C, 5% CO2 atmosphere for 22 hours. The noninvading cells were removed and the filter stained according to instructions provided in the assay. Cells were counted using a NIKON microscope.
Statistical Analysis
The data are expressed as mean ± standard deviation. Statistical analysis was performed with 2-way analysis of variance and included comparisons between time points for each CSC in each assay, between ISO and Intense CSCs from the same cigarette, and between cells lines at the same time point.
Results
Comparison of the Sensitivity of In Vitro Cytotoxicity Assays for Assessment of 4 CSCs in PSAE Cells
All 4 CSCs (3R4F Intense, 3R4F ISO, CM6 Intense, and CM6 ISO) demonstrated concentration-dependent cell growth or cell death effects measured by the 3 cell viability assays, ATP, MTS, and GSH assays. Figures 1 to 3 show the results of treatments with different concentrations of CSCs over the course of 28 days. The 100 and 300 μg/mL CSCs resulted in cell death in 6 and 24 hours of exposure in all 3 assays. At certain concentrations, all 4 CSCs increased cell proliferation at early time points but caused cytotoxicity at later time points. For example, 3 and 10 μg/mL of CSCs resulted in ATP content increase at 6, 24, and 72 hours exposure but decrease at 120 hours and 14 days. Cells treated with 0.3 or 1 μg/mL of CSCs caused ATP, MTS, and GSH levels to increase from 6 to 120 hours but decreased at 14 days. Interestingly, ATP, MTS, and GSH assays showed that CSCs, 0.3 to 3 μg/mL, caused cell death at 14 days, but cell growth at day 28. Over 50% cell death was reached at all time points, which enabled IC50 values to be calculated. Table 1 shows the values of IC50 for each assay and each exposure time for each CSC. There is an overall trend that IC50 values progressively decreased from 6 hours to 14 days (values that were significantly different are noted in Table 1), demonstrating time-dependent cytotoxicity of CSCs; however, IC50 values increased at 28 days compared to day14, which indicates that cells developed resistance to CSCs. For example, IC50 values of CM6 ISO measured by ATP assay were 55.5, 50.4, 37.1, 18.1, 3.6, and 9.7 μg/mL at 6, 24, 72, 120 hours, 14, and 28 days, respectively. IC50 values of ISO CSCs are lower that Intense CSCs in the first 120 hours. For example, IC50 values of 3R4F ISO versus 3R4F Intense and CM6 ISO versus CM6 Intense measured by ATP assay were 19.2 versus 23.2 and 18.1 versus 22.2, respectively, at 120-hour exposures.
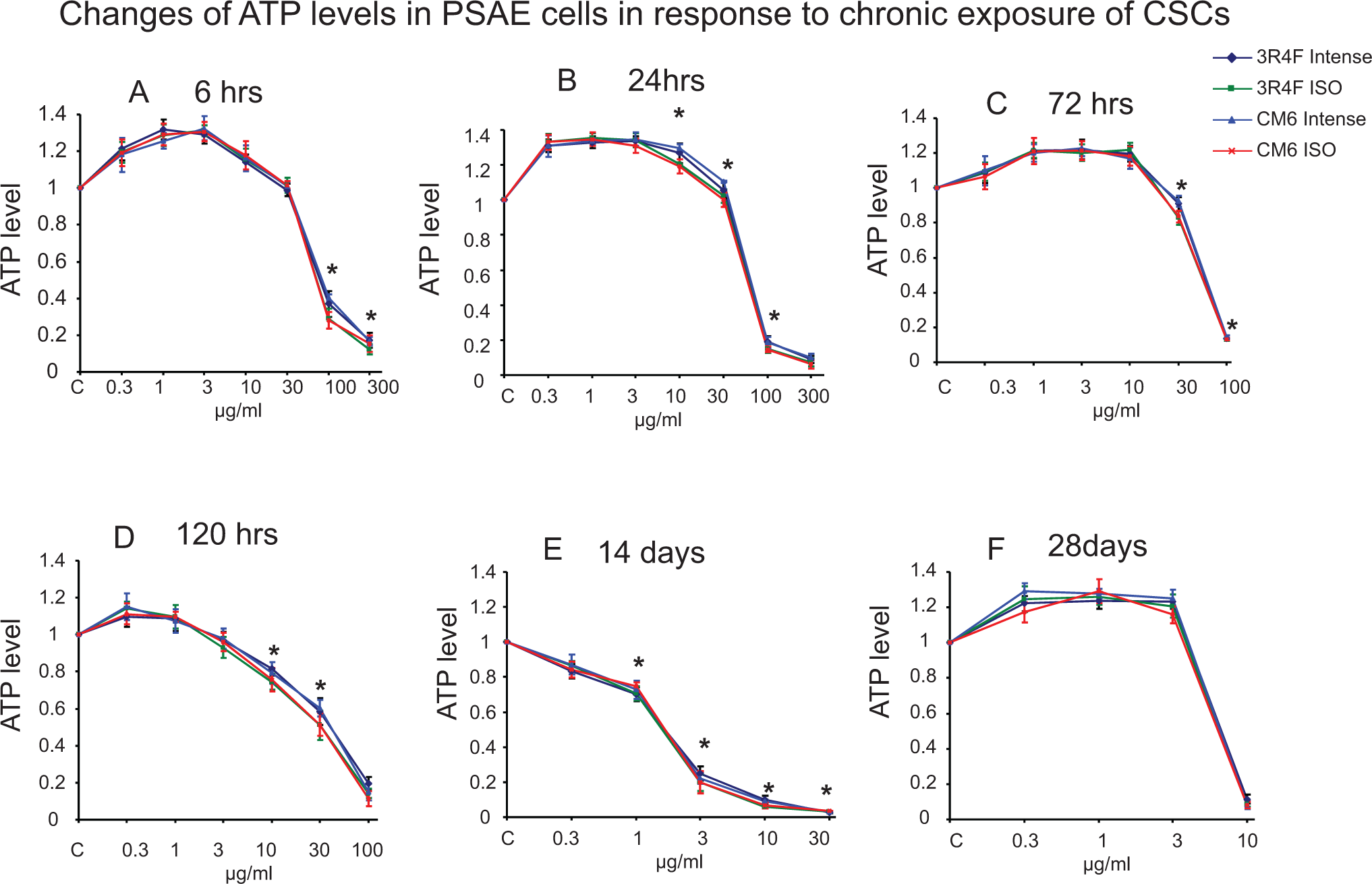
Concentration-dependent cytotoxicity of 4 CSCs on primary small airway epithelial (PSAE) cells measured by ATP assay after exposure to 4 CSCs for 6, 24, 72, 120 hours, 14, or 28 days. The ATP levels are relative to control. The data are expressed as mean ± standard deviation (SD). Each assay was conducted on 3 independent experiments, and there were 3 replicates for each concentration for each independent assay. *Indicating a statistically significant difference between International Organization for Standardization (ISO) and Intense CSCs from the same cigarette (P < 0.05). ATP indicates adenosine 5′-triphosphate; CSC, cigarette smoke condensate.
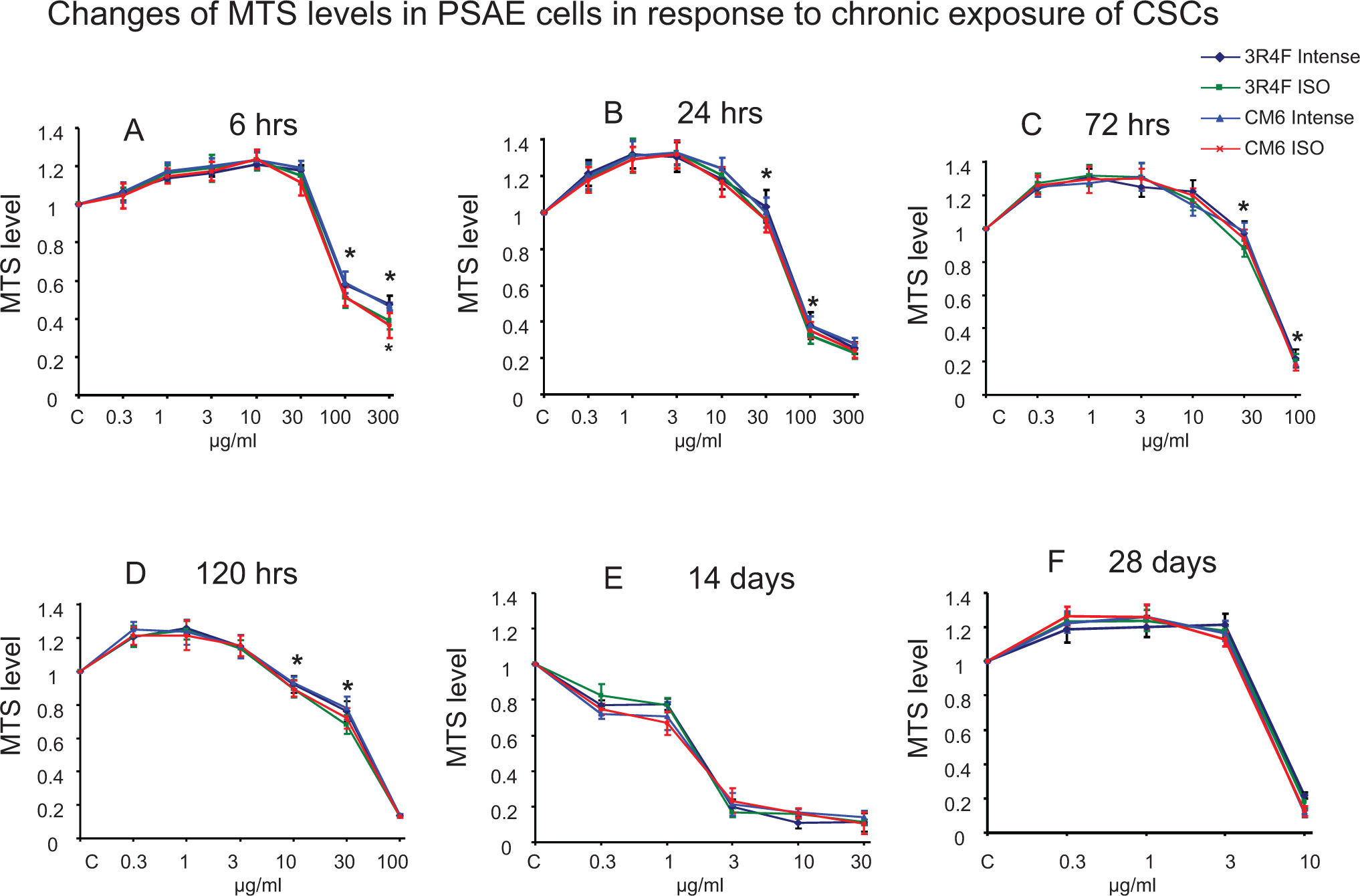
Concentration-dependent cytotoxicity of 4 CSCs on primary small airway epithelial (PSAE) cells measured by MTS assays after exposure to 4 CSCs for 6, 24, 72, 120 hours, 14 or 28 days. The MTS levels are relative to control. The data are expressed as mean ± standard deviation (SD). Each assay was conducted on 3 independent experiments and there were 3 replicates for each concentration for each independent assay. *Indicating a statistically significant difference between International Organization for Standardization (ISO) and Intense CSCs from the same cigarette (P < 0.05). CSC indicates cigarette smoke condensate; MTS, 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy-methoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium.
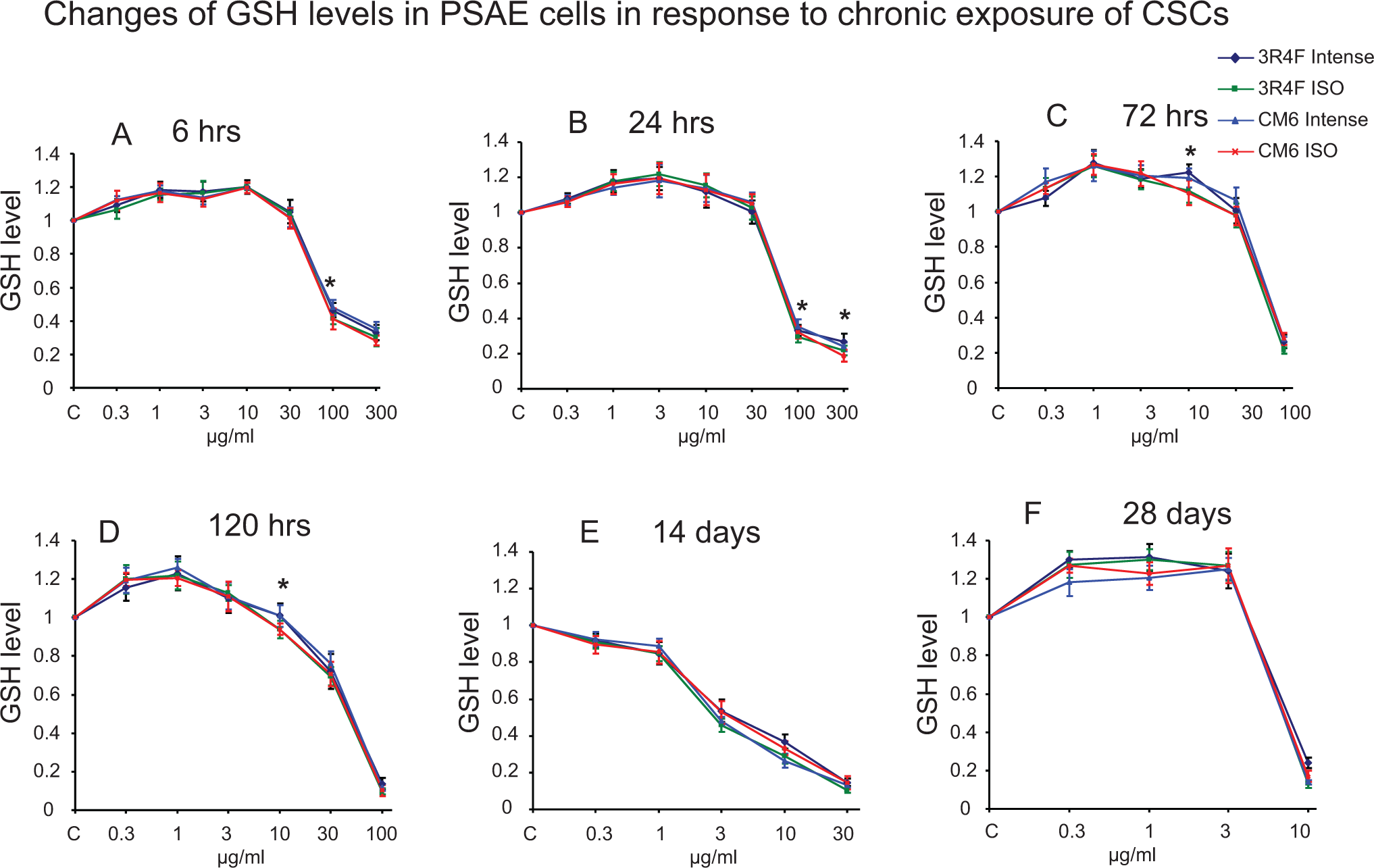
Concentration-dependent cytotoxicity of 4 CSCs on primary small airway epithelial (PSAE) cells measured by GSH assays after exposure to 4 CSCs for 6, 24, 72, 120 hours, 14 or 28 days. The GSH levels are relative to control. The data are expressed as mean ± standard deviation (SD). Each assay was conducted on 3 independent experiments and there were 3 replicates for each concentration for each independent assay. *Indicating a statistically significant difference between International Organization for Standardization (ISO) and Intense CSCs from the same cigarette (P < 0.05). CSC indicates cigarette smoke condensate.
The Values of IC50 (μg/mL) for 4 CSCs Using PSAE (Primary Small Airway Epithelial) and CP-C (hTERT-Immortalized Esophageal Epithelial) Cells.

Abbreviations: ATP, adenosine 5′-triphosphate; CSC, cigarette smoke condensate; IC50, half maximal inhibitory concentration; ISO, International Organization for Standardization; MTS, 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy-methoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium.
a P < 0.05 compared with previous time point.
b P < 0.05 compared with ATP assay value in PSAE cells at same time point.
The sensitivity of the 3 viability assays is compared in Table 1. The IC50 values of ATP assay are lower than MTS and GSH assays until 14 days but not at 28 days (values that were significantly different [P < 0.05] at 72 hours and 14 days). Accordingly, we suggest that ATP assay is more sensitive than MTS and GSH assays in assessing acute cytotoxicity of CSCs.
Figure 4 shows that cell release of LDH started to increase with treatment of 30 μg/mL CSCs at 6 hours, 10 μg/mL at 24 hours, 10 μg/mL at 72 hours, and 3 μg/mL at 120 hours. Concentration-dependent increase in LDH release was observed at 6 hours with 100 and 300 μg/mL of CSCs resulting in increases of 2.9- to 3.6-fold and 4.2- to 4.8-fold, respectively. The CSCs treatment for 24 hours at 100 μg/mL led to 6.0- to 6.9-fold increases; however, 300 μg/mL CSC treatments did not cause further increase in LDH level. Similarly, 72-hour treatments with CSCs at 1.0 to 30 μg/mL and 120-hour treatment with 1.0 to 10 μg/mL showed concentration-dependent increases in LDH release, with maximal increase of 5.4- to 6.3-fold and 4.2- to 6.2-fold, respectively. However, further increase in CSC concentrations did not caused further increase in LDH level. Incubating 0.3 to 30 μg/mL of CSCs for 14 days resulted in lower increases in LDH release, with CSCs at 10 and 30 μg/mL causing only 2.1- to 2.3-fold and 2.4- to 2.6-fold increases, respectively. Exposure to CSC at 0.3 to 10 μg/mL did not cause an increase in LDH release at 28 days.
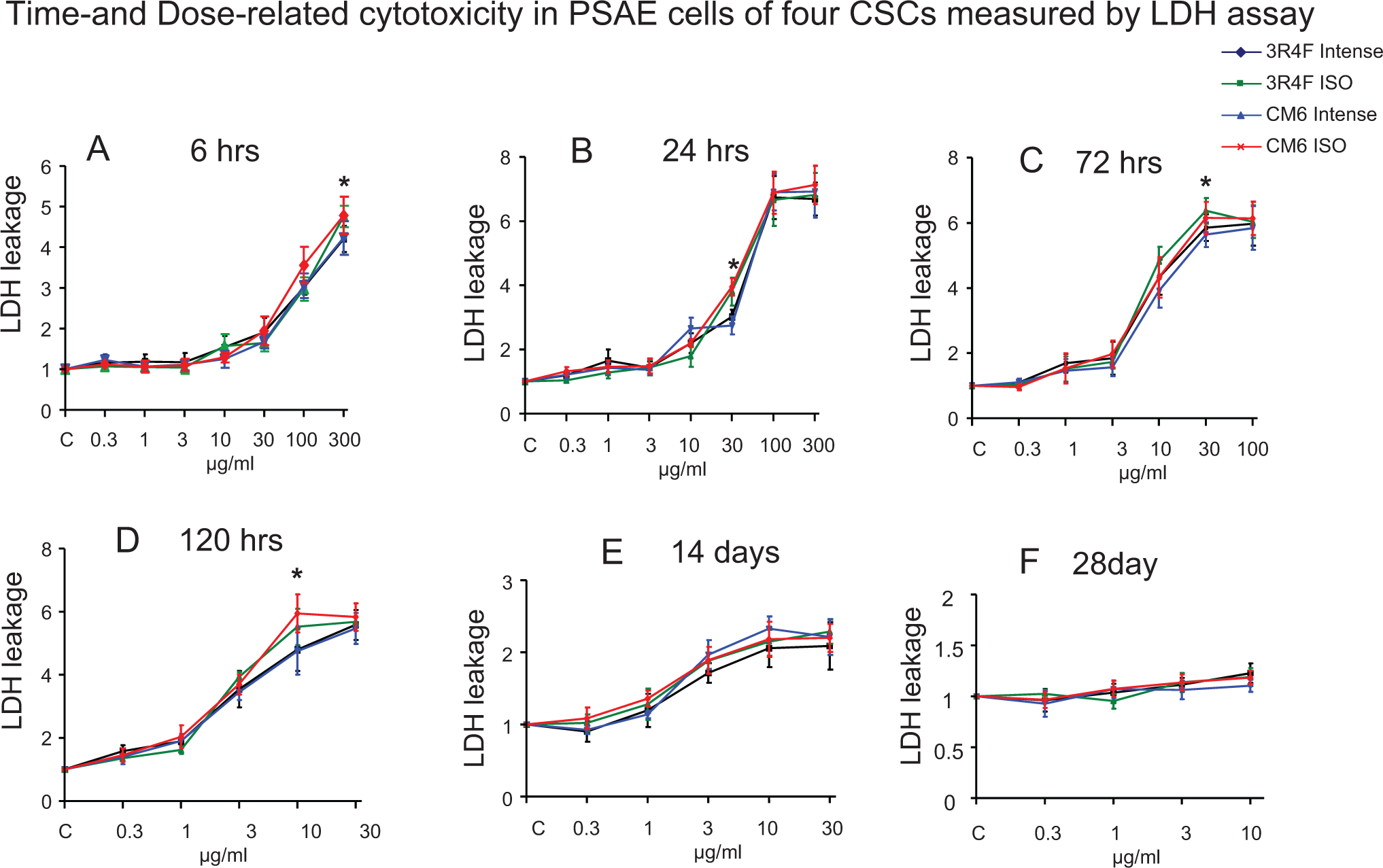
Lactate dehydrogenase (LDH) release from primary small airway epithelial (PSAE) cells treated with 4 CSCs for 6, 24, 72, 120 hours, 14 or 28 days. The LDH levels are relative (fold change) to control. The data are expressed as mean ± standard deviation (SD). Each assay was conducted on 3 independent experiments and there were 3 replicates for each concentration for each independent assay. *Indicating a statistically significant difference between International Organization for Standardization (ISO) and Intense CSCs from the same cigarette (P < 0.05). CSC indicates cigarette smoke condensate.
The relative toxicity of the 4 CSCs to PSAE cells was established. The ISO CSCs caused more cell death than Intense CSCs as determined by all 3 cell viability assays, and statistically significant differences were reached at several concentration–time points (Figures 1 –3). For example, ISO CSCs caused significantly lower ATP levels at 100 μg/mL at 6 hours, 10 and 30 μg/mL at 24 hours, and 10 and 30 μg/mL at 120 hours compared to Intense CSCs (P < 0.05). However, significant differences in MTS and GSH levels were not found among treatment with ISO and Intense CSCs at days 14 and 28 and in ATP levels at day 28. Consistently, compared with Intense CSCs, ISO CSCs caused significantly higher LDH release treated at several time–concentration points (P < 0.05). For example, 30 μg/mL 3R4F ISO, CM6 ISO, 3R4F Intense, and CM6 Intense caused 5.5, 6.1, 4.3, and 4.2-fold increases, respectively, of LDH levels at 72 hours. However, significant difference in LDH release was not found among treatments with ISO and Intense CSCs at days 14 and 28 (Figure 4).
Cytotoxicity of CSCs on CP-C Cells Measured by ATP and LDH Assays
To evaluate the toxicity of CSCs on CP-C cells, ATP and LDH assays were employed. The 300 μg/mL CSCs did not result in more than 50% cell death at 6 and 24 hours; therefore, IC50 value was not achieved. Similar to the data obtained in PSAE cells, the ATP assay showed that same concentrations of CSCs caused an increase or a decrease in ATP content at different time points (Figure 5). For example, 0.3 to 30 μg/mL CSCs resulted in ATP content increases until 72 hours, but 30 μg/mL CSCs caused reduced ATP content in 120 hours and did not cause further decreases in ATP content on days 14 and 28. On days 14 and 28, 100 μg/mL of CSC achieved more than 95% cell death measured by the ATP assay. A significant difference in the potency of toxicity among the 4 CSCs in this cell line was not found by the ATP assay (Figure 5). The results from the ATP assays demonstrated that IC50 values were higher in CP-C cells compared to PSAE cells (values that were significantly different [P < 0.5] are noted in Table 1). For example, IC50 values of 3R4F Intense at 72 hours were 110.2 and 39.9 μg/mL in CP-C and PSAE cells, respectively. The IC50 values of ISO-CSCs are lower than correspondent Intense CSCs. Unlike PSAE cells, in the LDH assay CP-C cells continued to show increases in concentration-dependent release on day 28, although the degree of increases was lower than earlier time points, demonstrating continued cell damage (Figures 4 and 6). Similar to the findings in PSAE cells, exposure to ISO CSCs resulted in statistically significantly higher LDH release compared to Intense CSCs in several time–concentration points.
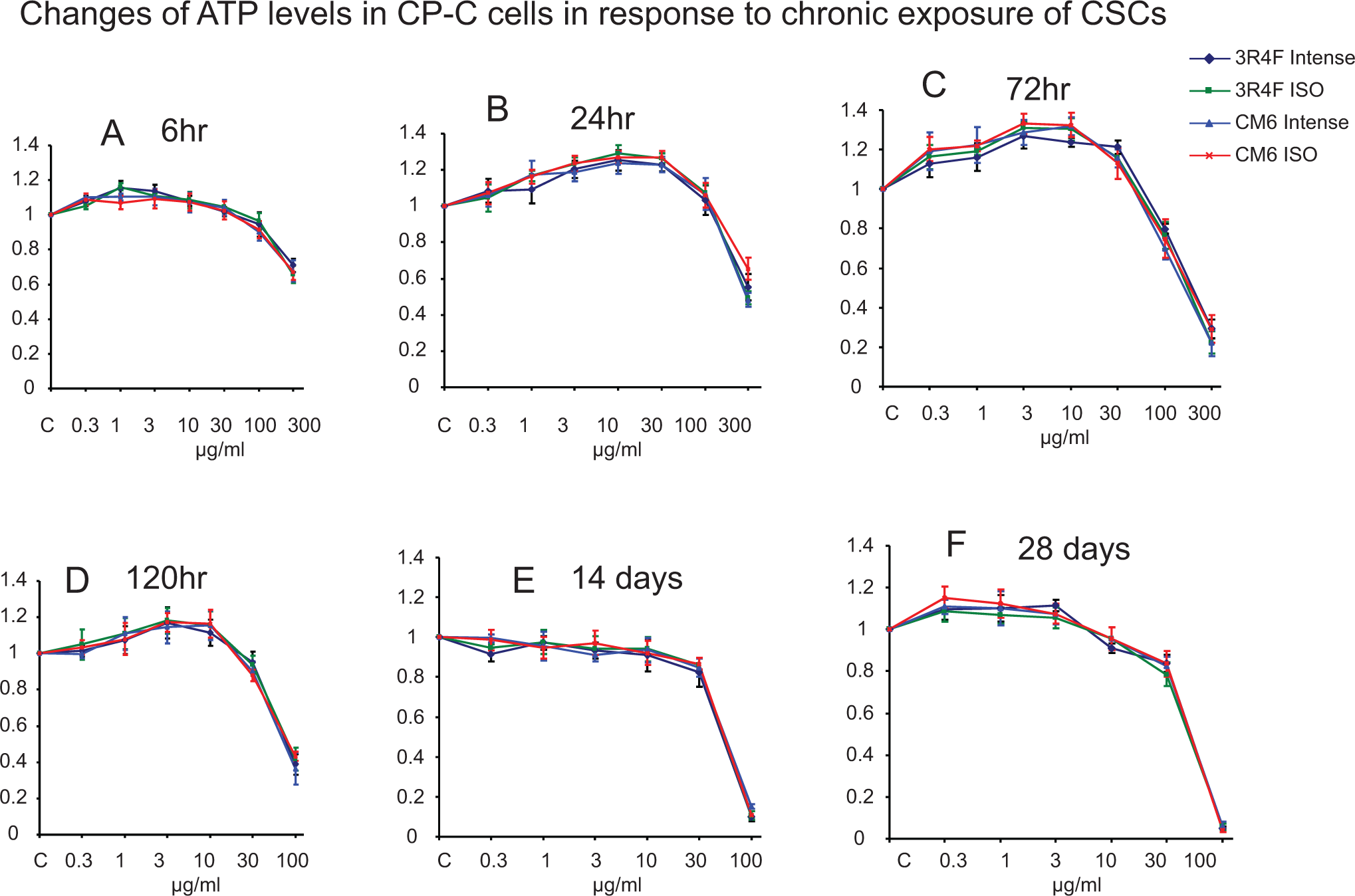
Concentration-dependent cytotoxicity of 4 CSCs on CP-C cells measured by ATP assays after exposure to 4 CSCs for 6, 24, 72, 120 hours, 14, or 28 days. The ATP levels are relative to control. The data are expressed as mean ± standard deviation (SD). Each assay was conducted on 3 independent experiments and there were 3 replicates for each concentration for each independent assay. ATP indicates adenosine 5′-triphosphate; CSC, cigarette smoke condensate.
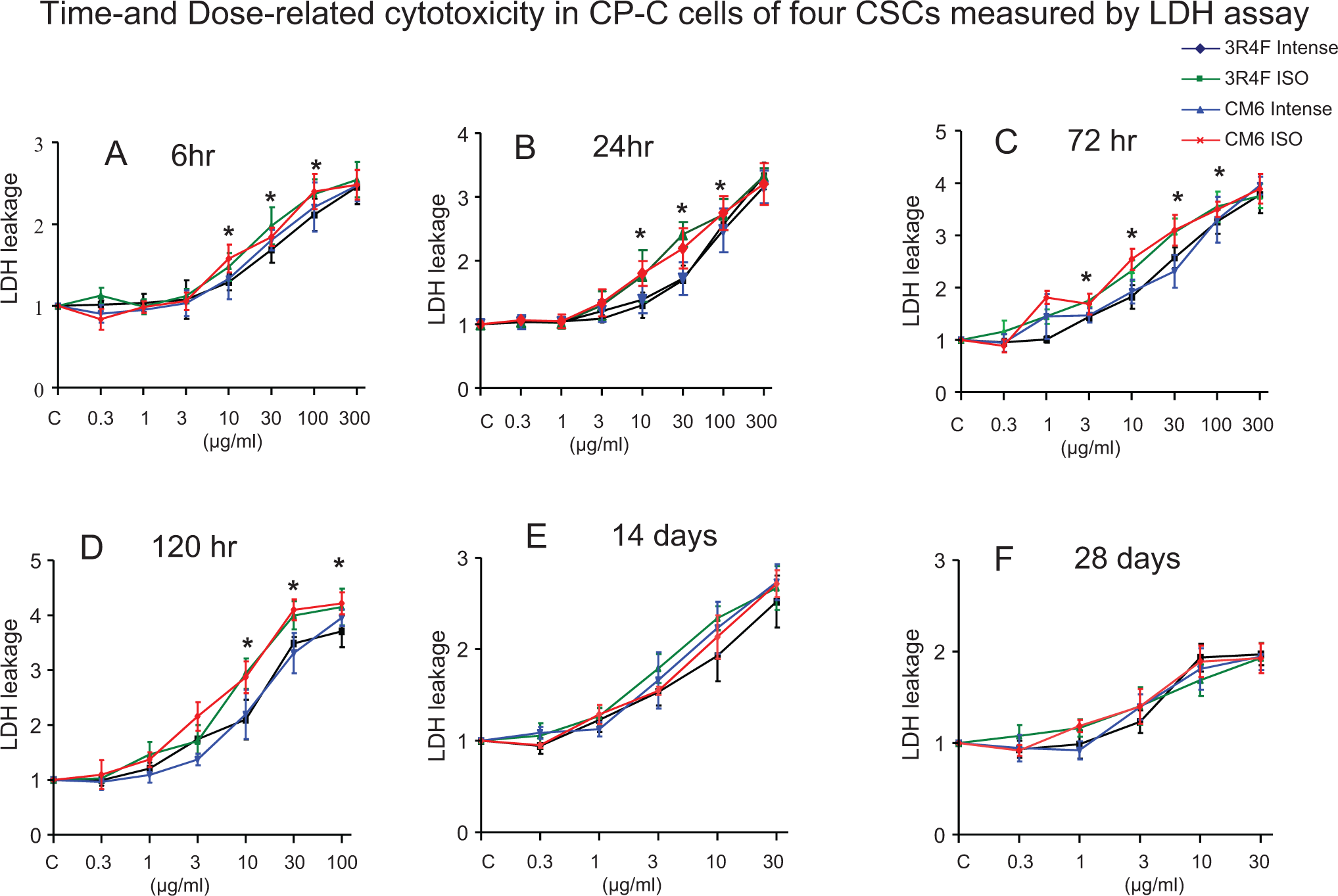
Lactate dehydrogenase (LDH) release from CP-C cells treated with 4 CSCs for 6, 24, 72, 120 hours, 14 or 28 days. The LDH levels are relative (fold change) to control. The data are expressed as mean ± standard deviation (SD). Each assay was conducted on 3 independent experiments and there were 3 replicates for each concentration for each independent assay. *Indicating a statistically significant difference between International Organization for Standardization (ISO) and Intense CSCs from the same cigarette (P < 0.05). CSC indicates cigarette smoke condensate.
Morphological Changes Induced by CSCs
Microscopic observations revealed that after 14 days of exposure to 0.3 μg/mL of CSC, PSAE cells acquired changes associated with the cell transformation, such as elongated, fibroblast-like form, and focal areas, as well as loss of contact inhibition, shown in the representative photographs (Figure 7). The transformational changes became more dramatic on day 28.

Morphology of primary small airway epithelial (PSAE) cells exposed to CSC. Photographs of morphological changes noted with 0.3 μg/mL of CSC (3R4F Intense) for 14 (B) and 28 days (D) of treatment are shown. Also included are photograph of controls for days 14 (A) and 28 (C). CSC indicates cigarette smoke condensate.
Invasion Activity Induced by CSCs
To determine the effect of CSC on cell invasion, the invasive capacities of PSAE cells without and with CSC (3R4F Intense and 3R4F ISO) exposures of 0.3 or 1.0 μg/mL dose for 14 or 28 days were examined. The CSC was found to stimulate lung cell invasion. The invasion was greatest at the higher CSC dose and longer duration (Figure 8).
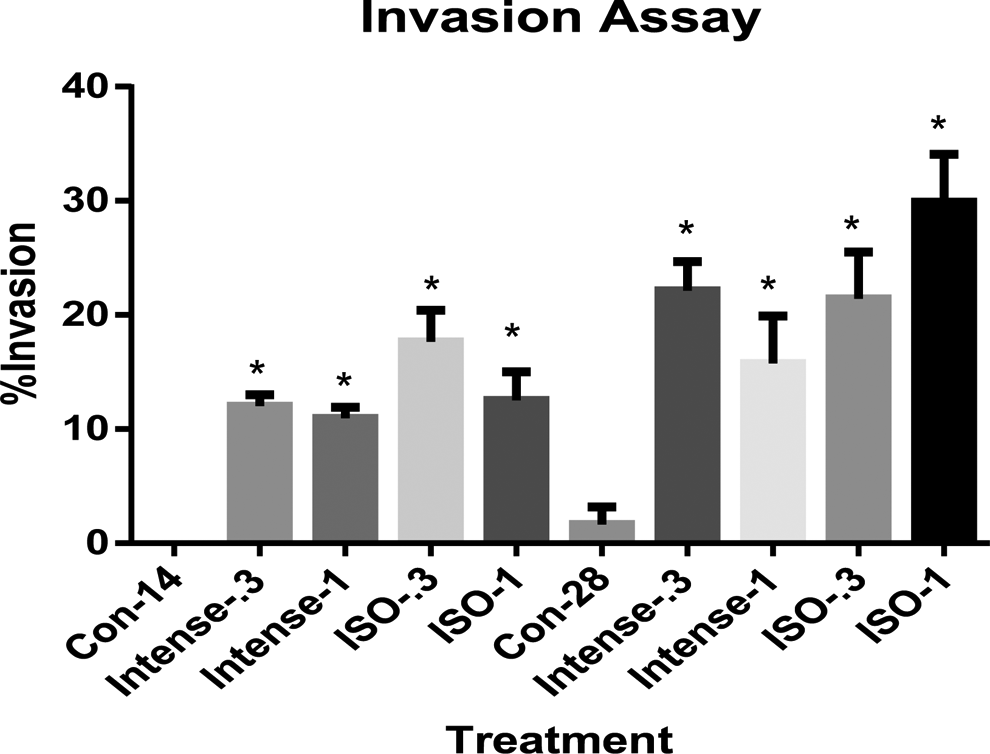
Effect of CSC exposure on primary small airway epithelial (PSAE) cells invasion. Cells were treated with 0.3 or 1.0 μg/mL CSC (3R4F Intense or 3R4F International Organization for Standardization [ISO]) for 14 or 28 days. The data are expressed as mean ± standard deviation (SD) of at least 3 determinations. Controls at each time point are included. *Indicating a statistically significant difference compared to control (P < 0.05). CSC indicates cigarette smoke condensate.
Discussion
Cigarette smoke is a mixture of more than 5000 constituents. Many constituents are cytotoxic with more than 93 chemicals being harmful or potentially harmful in humans. Cytotoxicity assays can be an informative part of the battery of in vitro toxicology tests in evaluating the harm of tobacco products and can also be used to quickly explore the mechanisms involved in chemical toxicity. The end points tested in in vitro toxicity assays include cell viability, cell proliferation rate, cytosolic enzyme leakage due to damage of cell membrane, and oxidative stress. 8 Assays measuring these end points need to be adequately compared for sensitivity and predictivity. Moreover, special consideration should be addressed on the variation of results from different doses and exposure times. A thorough assessment of in vitro model systems can increase confidence in using these cytotoxicity assays to assess potential harm of tobacco smoke and its constituents. In this study, we systematically described the effects of CSCs produced from 2 reference cigarettes machine smoked under ISO or Health Canada Intense regimens with a wide dose range over a course of 28 days on a PSAE cell line and an hTERT-immortalized Barrett esophagus cell line.
All 4 assays (LDH, ATP, MTS, and GSH assays) demonstrated the effect of concentration and the lengths of exposure on cytotoxicity. The ATP, MTS, and GSH assays showed that the same concentrations of CSC may result in PSAE cell proliferative or cell death effects depending on the duration of exposure time, confirming the complexity of the effects of CSCs. Doses of 100 and 300 μg/mL CSCs demonstrated a cell death effect in as short as 6 hours, which illustrates the high cytotoxicity of tobacco smoke in cultured cells. Exposure to CSCs, 0.3 to 3 μg/mL, caused ATP, MTS, and GSH level increases until 72 hours of exposure, but decreases at 14 days. The CSCs at 0.3 to 30 μg/mL induced cell death at day 14. At day 28, 10, and 30 μg/mL of CSCs led to further cell death, but 0.3 to 3 μg/mL CSCs resulted in cell growth. Our data demonstrate that both dose and duration are important considerations when assessing the cytotoxicity of CSCs.
Among the 3 tested cell viability assays, the ATP assay appeared to be more sensitive than MTS and GSH assays. For example, exposure to 30 μg/mL CSCs for 120 hours resulted in 40% to 49% of ATP reduction, but only 23% to 32% and 15% to 27% reduction of MTS and GSH, respectively. Moreover, IC50 values obtained by ATP assay are lower than those of MTS and GSH assays until 14 days of exposure. Table 1 also shows that IC50 values of the 3 assays are comparable at day 28. This may be explained by the development of resistance to transformed cells to CSCs.
Our microscopic observations found transformational changes at 14 days, such as foci formation and disrupted cell morphology induced by CSCs, and the transformed cells became predominant at 28 days of exposure (Figure 7). The growing colonies observed at 28 days are likely the cells that have undergone genetic and epigenetic alterations causing compromised ability to execute effective programmed cell death. Invasion assay results demonstrated that PSAE cells had obtained the capacity to invade basement membrane revealing changes associated with progressive events in the carcinogenesis process. These observations confirmed that longer term exposure of primary and immortalized cells to tobacco smoke leads to acquisition of precancerous properties. The relative insensitivity of the cytotoxicity assays to CSCs at later time points in this study suggests that these cell systems gain resistance to CSC toxicity. This interpretation is consistent with previous reports. 21,35 It has been reported that the complex process of cellular transformation involves genetic alteration and modulation of multiple signaling pathways, including mitogen-activated protein kinase-extracellular signal regulated kinases (MAPK-ERK) signaling pathway in bronchial epithelial cells. 36,37 Recent studies have reported that chronic exposure to low concentrations of CSC or low concentrations of many tobacco carcinogens, including tobacco-specific N-nitrosamines, nicotine, polycyclic aromatic hydrocarbons, and so on, resulted in disruption of growth control, acquisition of migratory capacity, and epithelial-to-mesenchymal transition (EMT). 21,35 These studies may explain the finding in this study that CSC exerts cytotoxic effects in the short term but produces cell proliferative effects with continued exposure. Our observations indicated that transformational changes induced by CSCs confer resistance to CSC toxicities, suggesting the importance of choosing the combinations of time and concentration to collect and analyze data in investigating CSC in a model system. Therefore, accessing a broad range of concentrations of CSCs over a longer exposure time, although laborious, is a useful strategy to assess the effects of CSCs in in vitro studies.
The LDH assay, which measures cytosolic enzyme released to culture medium, is probably the simplest type of cytotoxicity assay. The sensitivity and confounding variables of LDH assay in testing cytotoxicity of tobacco smoke remains to be clarified. 8 Figure 4 shows that increase in LDH release from PSAE cells occurred at exposures to 10 μg/mL of CSCs for 24 and 72 hours and 3 μg/mL of CSCs at 120 hours, whereas ATP, MTS, and GSH levels did not show cytotoxicity under these conditions. These data, being in line with a previous report, 16 indicate that cell membrane compromise may be an early pathobiological response to CSC exposure. In our experiment, cell culture media were changed at 72 hours; therefore, the LDH measured at 120 hours reflects LDH released from 72 to 120 hours. The high LDH levels at 120 hours at all tested concentrations of CSC indicated continued cell damage in this period. It came to our attention that CSCs caused increases in ATP, MTS, and GSH levels but also increased LDH release in both PSAE and CP-C cells at the same time–concentration points. The reasons underlie that apparent discrepancy is not known. However, a few explanations may be proposed. First, although cell membrane damage occurred and LDH release increased, the cells may continue to be viable, and the ATP content, MTS, and GSH may not start to decline. Conversely, these cell viability markers may even increase as a protective mechanism. The coexistence of viable, damaged, and dead cells after treatment with CSCs, as described previously, 10 may weaken the association of cell viability biomarkers with the cell number, particularly at early time points. Second, antiapopotic effect of some constituents may promote cell growth and generation of ATP, MTS, and GSH. Third, some constituents in CSC may affect the reactions of the assays. Therefore, the complexity of CSC and altered cell metabolic status induced by CSC may weaken the association of ATP, MTS, and GSH values with cell numbers. We also noticed, with CSC exposures, that the degrees of cell death measured by ATP, MTS, and GSH levels were not consistent with LDH levels. For example, significantly more reductions in ATP, MTS, and GSH were observed in cells treated with 100 μg/mL compared with 30 μg/mL of CSCs at 72 hours; however, 30 and 100 μg/mL of CSCs caused similar fold increase in LDH levels. These observations can be explained by many factors: first, more protease released from dying cells may inactivate, degrade, or metabolize released LDH; second, a higher dose of CSCs caused more cell death in shorter time, and the released LDH was degraded at the time of measurements; third, high levels of constituents in CSCs may affect the rate of conversion of NADH to NAD+. Therefore, although an increase in LDH release is an early marker of cell damage, the LDH values are affected by many confounding variables and may not always reflect the true degree of cell damage or cell death. The CSC-induced LDH release from PSAE on day 14 was much lower compared with earlier time points, and very little increase in LDH level were observed on day 28, indicating the developed resistance of cells to CSCs. The increased resistance to CSCs is consistent with our microscopic observation showing clonal expansion of transformed cells on day 14, and the transformed cells continued to grow over the course of 28 days.
The toxic potency of the 4 CSCs was also compared in this study. The 3R4F and CM6 CSCs generated by the same smoking machine method showed similar cytotoxic potencies and CM6- and 3R4F-ISO demonstrated statistically higher cytotoxicity than Intense CSCs in several time–concentration points in all 4 tested assays (Figures 1 –6). Further analysis of the harmful and potential harmful constituents from CSCs generated by ISO and Intense regimens may assist in understanding the reasons underlying the difference in cytotoxicity identified in our study among the tested CSCs. 25
In CP-C cells, exposure of CSCs also caused decreases in ATP content and increases in LDH release, but the degree of changes was lower compared to PSAE cells (Figures 1 and 5). Our data did not show statistically significant difference in the potency of toxicity among the 4 CSCs at the tested concentrations in CP-C cells, suggesting that more sensitive cell-viability assays need to be identified for assessing toxicity of CSCs on CP-C cells. The CSCs did cause increases of LDH release in a concentration-dependent manner for all the tested concentrations in all time points. Consistently, CSC caused higher fold increases in LDH release in PSAE cells compared to CP-C cells. Similar to PSAE cells, CP-C cells also developed resistance to CSCs at days 14 and 28, demonstrated by less increase in LDH release compared with earlier time points. The CSCs did, however, continue to cause increased LDH release from CP-C cells at day 28. These observations further demonstrate the need for the kinetics in assessing CSC toxicity in a model system, particularly in comparing the cytotoxic properties of CSCs in different cell model systems.
Due to the complexity of the effects caused by the chemical mixture present in CSC, evaluation of the relative harm of the tested CSCs remains a challenge. Indeed, numerous tobacco constituents have shown cytotoxic properties. Accumulating evidence suggests that many constituents in CSCs could contribute to tobacco-related lung carcinogenesis by regulating 2 processes critical for tumorigenesis, cell growth, and apoptosis. 38 –40 West et al reported that nicotine or NNK attenuated apoptosis and partially induced a transformed phenotype in 2 sets of primary human airway epithelial cell cultures: human bronchial epithelial cells derived from large airways, which are the precursor cells for squamous cell carcinomas, and SAECs, which are the precursor cells for adenocarcinomas. Nicotine and NNK exert antiapopotic effects in as early as a few hours. 40 Nicotine contributes to tumor promotion and progression with antiapoptotic and mitogenic properties. 39 Human bronchial epithelial cells underwent EMT, which contributes to various malignant features of cancer cells and a prognostic marker for early-stage NSCLC, after only 72 hours of CSC exposure. 41 Therefore, CSCs have both cytotoxic and antiapoptic effects. The synergistic, additive, and antagonist effects among the constituents lead to very complex overall effects. Accordingly, although cytotoxicity assays contribute to understanding the harm of the complex chemical mixture of CSC, these assays actually reflect the combination of cytotoxic and antiapoptotic properties of CSC. The cytotoxicity of a CSC may not completely reflect the cytotoxicity and biological harm of cigarette smoke, particularly the longer term tumorigenic effects from exposure to chemicals in the particulate and gas phases of aerosolized cigarette smoke. Therefore, studies of biological changes in other aspects, such as genotoxicity, quantitative transformation studies, and cellular signal pathway studies should be considered in combination with cytotoxicity data in evaluating the true harm of tobacco products.
In conclusion, we compared 4 cytotoxicity assays in assessing CSCs-induced cytotoxicity in 2 human cell lines. Our results suggest that the LDH assay is suitable in assessing early CSC-induced cell damage in these cell lines. The ATP measurement seems to be more sensitive than MTS and GSH assays. The CSCs generated under the ISO smoking machine conditions demonstrated higher cytotoxicity compared with CSCs generated under Health Canada Intense conditions when tested at the same concentration (µg/mL). This observation is not unexpected as cigarette smoke generated under Intense conditions has a higher water content than smoke generated under the less intense conditions. It is also worth noting that while these 2 smoking machine conditions allow condensate to be produced under more or less intense machine smoking conditions in an attempt to maximize differences in their smoke chemistry, neither regimen replicates how people smoke cigarettes. Cytotoxicity assays reflect the combined effects of cell damage and possibly the onset and progression of tumorigenic transformational changes. Studies that include extended time points may increase the value of cytotoxicity assays in assessing the harm of tobacco smoke. The results of in vitro cytotoxicity assays alone do not represent the full toxic potential of tobacco products. Therefore, the value of cytotoxicity assays in assessing the harm of tobacco smoking should be interpreted in combination with other assays.
Footnotes
Acknowledgments
We thank the Center for Disease Control and Prevention for providing CSCs. We acknowledge Dr Kenneth Wiley for his assistance in literature search and setting up database for this study. We thank Dr Patricia Richter for her valuable inputs in study design and implementation.
Authors’ Note
The views and opinions expressed in this article are those of the authors only and do not necessarily represent the views, official policy, or position of the US Food and Drug Administration or any of its affiliated institutions or agencies.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employees of the US Food and Drug Administration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
