Abstract
3-Nitro-1,2,4-triazol-5-one (NTO), an insensitive explosive, was evaluated to assess potential environmental and human health effects. A 14-day oral toxicity study in Sprague-Dawley rats was conducted with NTO in polyethylene glycol -200 by gavage at doses of 0, 250, 500, 1000, 1500, or 2000 mg/kg-d. Body mass and food consumption decreased in males (2000 mg/kg-d), and testes mass was reduced at doses of 500 mg/kg-d and greater. Based on the findings in the 14-day study, a 90-day study was conducted at doses of 0, 30, 100, 315, or 1000 mg/kg-d NTO. There was no effect on food consumption, body mass, or neurobehavioral parameters. Males in the 315 and 1000 mg/kg-d groups had reduced testes mass with associated tubular degeneration and atrophy. The testicular effects were the most sensitive adverse effect and were used to derive a benchmark dose (BMD) of 70 mg/kg-d with a 10% effect level (BMDL10) of 40 mg/kg-d.
Introduction
The explosive compound, 3-nitro-1,2,4-triazole-5-one (NTO), was first prepared in 1905 and its explosive properties were reported by the Los Alamos National laboratory in 1985. 1 Renewed interest in the energetic properties of NTO has been fueled by the need to develop munitions that are less prone to inadvertent detonation during transport and routine handling. The reduced sensitivity to environmental stimuli and nearly equivalent performance to traditionally fielded munitions make NTO-based formulations desirable replacements for traditional explosives such as 1,3,5-trinitro-1,3,5-triazine, and 2,4,6-trinitrotoluene (TNT). 2,3 3-Nitro-1,2,4-triazole-5-one is presently used in a number of formulations including IMX 101 and IMX 104. As a new component of munitions formulations, NTO must not only meet certain performance criteria, but must also be acceptable from the perspective of human health and the environment. For munitions workers, exposure to NTO can occur through inhalation and dermal routes as well as incidental ingestion. Development of occupational and environmental exposure standards for the protection of personnel and the environment requires toxicity information.
Published toxicity data for NTO are generally limited to in vitro and acute studies. The oral LD50 for NTO is reported to be > 5g/kg in rats and mice.
4
3-Nitro-1,2,4-triazole-5-one did not induce dermal sensitization in guinea pigs and was not an eye irritant, but did produce mild skin irritation in rabbits.
4
In an acute inhalation exposure to the highest—achievable concentrations of NTO aerosols (0.18 mg/L), there was no mortality, compound-induced clinical signs of toxicity, or gross pathological changes in rats.
5
The genotoxicity of NTO was evaluated using a battery of tests, including the Ames test, Chinese hamster ovary cell chromosome aberration test, L5178Y TK+/− mouse lymphoma mutagenesis test, and rat micronucleus test. 3-Nitro-1,2,4-triazole-5-one was not genotoxic in either in vitro or in vivo testing.
6
Toxicokinetics and metabolism data for NTO are also limited. A study of NTO in male rhesus macaques (
There are no published studies on the toxicokinetics and metabolism of NTO in other animals. However, the fate of NTO was investigated in vitro using rat liver microsomal and bacterial systems. Rat liver microsomal catalysis of NTO under a nitrogen atmosphere produced primarily an amine, 5-amino-1,2,4-triazol-3-one (ATO). In the presence of oxygen, primarily 5-hydroxy-1,2,4-trizol-3-one (urazole) was produced and ATO was produced as a minor product. 8,9 Addition of NTO to aqueous medium released protons and lowered pH. The metabolism of NTO by bacteria was pH dependent in aqueous systems. The maximum microbial reduction occurred at pH 6 in the presence of sucrose, while a ring cleavage occurred at pH 8. 9
To evaluate the subacute and subchronic mammalian toxicity of NTO, 14- and 90-day repeated-dose oral toxicity studies were conducted in Sprague-Dawley rats. The results from the 90-day study reported herein were used to establish a benchmark dose (BMD) and a 95% lower confidence limit on the 10% effect level (BMDL10) which may be used to calculate occupational exposure levels.
Materials and Methods
Test Substance
3-Nitro-1,2,4-triazol-5-one (oxynitrotriazole; CAS: 932-64-9; lot: BAE07B305001; purity: 99.6%) was procured from BAE Ordnance Systems, Inc. (Kingsport, Tennessee). Polyethylene glycol (PEG)-200 (CAS: 25322-68-3; lots: E27629 and G05625) was purchased from Mallinckrodt Baker, Inc. (Phillipsburg, New Jersey).
Test Animals
Young adult male and female Sprague-Dawley rats were obtained from Charles River Laboratories, Wilmington, Massachusetts, and were acclimatized for a minimum of 5 days upon arrival. For the 14-day study, 36 male (169 ± 7.1 g) and 36 female (130 ± 8.3 g) rats (6 weeks old) were randomly distributed into five treatment groups and a PEG-200 control group consisting of 6 rats per sex per group. For the 90-day study, 50 male (202 ± 11.2 g) and 50 female (168 ± 8.5 g) rats (7 weeks old) were randomly distributed into 4 treatment groups and a PEG-200 control group consisting of 10 rats per sex per group. Rats were distributed into groups according to body mass the day before the initiation of dosing using the LABCAT (Innovative Programming Associates, Inc., Lawrenceville, New Jersey) randomization program. Housing conditions were maintained at 64°F to 79°F, 30% to 70% relative humidity, with a 12-hour light/dark cycle. A certified pesticide-free rodent chow (Harlan Teklad, 8728C Certified Rodent Diet) and drinking quality water were available ad libitum. Rats were housed individually in suspended polycarbonate boxes with Harlan Sani-Chip bedding. Animal use procedures were approved by the Animal Care and Use Committee at the US Army Public Health Command (USAPHC). Animal care and use was conducted in accordance with the principals stated in the Guide for the Care and Use of Laboratory Animals and in accordance with all applicable regulations. 10 The USAPHC animal care program is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International (AAALAC). These studies were conducted in compliance with Good Laboratory Practice standards as described in Title 40, Code of Federal Regulations, Part 792.
Test Article Administration
3-Nitro-1,2,4-triazole-5-one is acidic when added to an aqueous medium; therefore, the NTO was suspended in PEG-200 to minimize problems with an acidic dosing medium. A separate solution/suspension in PEG 200 was prepared for each dose group in both the subacute (14-day) and subchronic (90-day) phases so that dosage volumes were consistent across groups. Each mixed dosing solution/suspension was analyzed to verify concentration prior to dosing, and stability and homogeneity were verified for selected concentrations. PEG 200 controls were administered an equivalent volume as the highest dose group for both phases (10 mL/kg and 7 mL/kg for subacute and subchronic phases, respectively). The test article was administered for either 14 or 90 consecutive days (7 days/week) using a syringe attached to a 16-gauge gavage needle. Dose levels for the subacute phase were selected based on reported LD50 values, while subchronic dose levels were based on the subacute results. In the 14-day study, 6 groups of male and female rats (6 per group/sex) were administered oral doses of 0 (PEG-200 control), 250, 500, 1000, 1500, or 2000 mg/kg-d NTO. Body mass and food consumption were measured on days 0, 1, 3, 7, and 14. All animals were observed twice daily for signs of toxicity and morbidity. In the 90-day study, 5 groups of male and female rats (10 per group/sex) were administered oral doses of 0 (PEG-200 control), 30, 100, 315, or 1000 mg/kg-d NTO. Body mass and food consumption were measured weekly, and all animals were observed twice daily for signs of toxicity and morbidity (once daily on weekends/holidays).
Urinalysis
Once within the final 2 weeks of the 90-day study, all animals were placed in metabolism cages overnight for timed free-catch urine collection. Each sample was evaluated for volume, color, and appearance. Measurements of pH, glucose, bilirubin, urobilinogen, ketone, blood, protein, nitrite, and leukocytes were determined (Siemens Healthcare Diagnostics™Multistix™ Reagent Strips, Fisher Scientific, Pittsburgh, Pennsylvania). Specific gravity was measured with a refractometer (American Optical Corp., Buffalo, New York).
Neurobehavioral Evaluation
Potential neurotoxic effects of NTO were evaluated using the functional observation battery (FOB) and motor activity assessment described in McDaniel et al. (1993) and Moser (2000). 11,12 The FOB, home cage, handheld, and open arena observations were conducted on each animal prior to initiation of dosing and weekly thereafter.
Sensory motor functions and motor activity were evaluated during week 11 of dosing. Sensorimotor responses were evaluated by testing reactivity to different types of stimuli. Each rat was scored for reaction to the approach of a closed pen, auditory startle response to a loud click, tail pinch response, pinna response, and pupillary response to a pen light. Righting reflex was measured by placing the rat on its back on a padded surface. The rat was scored on how quickly it turned over onto its feet. To score aerial righting, the rat was held in the air at 20 cm with its back horizontal to a padded surface. The rat was released and scored on its ability to turn over to land on its feet. To measure hind limb landing foot splay, the back feet of each rat were moistened with water. The rat was held by the scruff of the neck and the base of the tail and dropped from 20 cm onto a cage pad to show foot impressions. Foot splay was measured as the distance between the centers of the foot prints, to the nearest 0.5 cm. This was repeated twice and the measures were averaged. Forelimb and hind limb grip strength was assessed following measurement of righting reflex and hind limb landing foot splay. Peak grip strength was measured, in kilograms, using Chatillon Digital Force Meters (Model DFM-10Ametck Inc, Largo, Florida) that were verified using standard weights. Trials were repeated twice and the average was calculated. For the forelimb test, the animal was held by the base of the tail and allowed to place forepaws on the grate; the animal was pulled away from the grate at a continuous rate until grip was released and the reading was recorded. For the hind limb test, the animal was held by the base of the tail and allowed to grasp the grate with hind paws; the animal was pulled away from the grate at a continuous rate until grip was released, and the reading was recorded.
Motor activity was assessed using a SmartFrame Open Field Activity System (Hamilton Kinder, San Diego, California). The system consisted of 4 Plexiglas motor activity chambers (41 × 41 × 38 cm) each surrounded by a frame containing 32 evenly spaced (16x and 16y, 2.5 cm apart) infrared photocells. The floor of each chamber was equipped with a board containing 9 holes equipped with infrared photocells to detect nose poke activity. Activity was measured as basic movement, immobility, x and y ambulation, and nose pokes based on the number of photo beam breaks recorded using the MotorMonitor software (Hamilton Kinder Version 4.14). After acclimation to the test room for at least 30 minutes, animals were removed from the home cage and placed individually into an open field arena for 15 minutes. Data were collected automatically by the system at 15 equally spaced times while each rat was within the enclosure.
Hematology and Clinical Chemistry
At the conclusion of the 14- and 90-day study periods, blood samples were collected from animals fasted overnight via intracardiac puncture under isoflurane anesthesia. Hematology parameters including white blood cell (WBC) count, WBC differential (% neutrophils [NEU], % lymphocytes [LYM], % monocytes [MONO], % eosinophils [EOS], % basophils [BASO]), red blood cell (RBC) count, hemoglobin (HGB), hematocrit (HCT), mean cell volume (MCV), mean cell hemoglobin (MCH), mean cell hemoglobin concentration (MCHC), red blood cell distribution width (RDW), platelet (PLT) count, and mean platelet volume (MPV) were determined using the Cell-Dyn 3700 Hematology Analyzer (Abbott Laboratories, Illinois). Clinical chemistry analyses including albumin (ALB), alkaline phosphatase (ALKP), alanine aminotransferase (ALT), blood urea nitrogen (BUN), calcium (Ca), cholesterol (CHOL), creatinine (CREA), globulin (GLOB), glucose (fasted; GLU), lactate dehydrogenase (LDH), phosphorus (PHOS), total bilirubin (TBIL), total protein (TP), sodium (Na), potassium (K), and chloride (Cl) were conducted using the VetTest 8008 Chemistry Analyzer and VetLyte Na, K, Cl Analyzer (IDEXX Laboratories, Inc., Westbrook, Maine). For the 90-day study, average prothrombin time (AVG PT) and average activated prothrombin time (AVG APTT) were also determined using the MCA 210 Microsample Coagulation Analyzer (BioData Corporation, Horsham, Pennsylvania). Serum testosterone, free triiodothyronine (T3), and free thyroxine (T4) were determined for male rats from the 90-day study using the TOSOH AIA-360 Automated Enzyme Immunoassay System (TOSOH Bioscience, Inc, South San Francisco, California). Serum samples were frozen at −35°C until the time of analysis.
Sperm Analysis
A standard size (approx. 0.1 g) midsection was cut from 1 epididymis from each male rat in the 90-day study and placed in 400 µL of room temperature Roswell Park Memorial Institute buffer. The section was then cut several times and pressed with a pestle to express the contents into the buffer. Each sample was then pipetted into a standard count analysis chamber slide (Leja, the Netherlands) and analyzed using a Hamilton Thorne Biosciences IVOS Sperm Analysis System (Beverly, Massachusetts).
Necropsy and Histopathology
All animals were euthanized using carbon dioxide (CO2) following blood collection. Necropsies were completed on all terminal animals and the following organs were removed and trimmed for determination of absolute and relative (organ/body and organ/brain) organ mass: adrenals, brain, heart, kidneys, epididymis, liver, spleen, testes, thymus, ovaries, and uterus. Tissues harvested in the 14-day study were not preserved for future microscopic evaluation because of the planned 90-day study. Selected tissues from the 90-day study were trimmed, weighed, and preserved in 10% buffered formalin. Tissues were packaged and shipped to Comparative Biosciences, Inc (Sunnyvale, California) for processing and evaluation. Tissues were gross trimmed, processed through a graded series of alcohols, embedded in paraffin, microtome sectioned at 3 to 5 µm thickness, stained with hematoxylin and eosin, and cover slipped. Slides were evaluated by an American College of Veterinary Pathology board certified pathologist and incidence and severity of lesions were scored using the accepted industry scoring system: normal, minimal, mild, moderate, and severe. A full tissue list was microscopically evaluated for the control and high-dose groups. Organs with apparent test article-related weight effects and all recognized gross lesions from all dose groups were also selected for microscopic evaluation. The testes from all dose groups were also reevaluated by an American College of Veterinary Pathology board certified military pathologist.
Statistical Analyses
Results are presented as means ± standard error of the mean (SEM) and all analyses were conducted for males and females separately. SPSS 15.0 (IBM Corp., Armonk, New York) was used to perform all analyses and statistical significance was defined as
Determination of BMD
The BMD and its BMDL10 were determined according to the United States Environmental Protection Agency’s BMD approach.
13
The dose–response relationship was based on the microscopic incidences of seminiferous tubule degeneration and atrophy. This finding represented the most sensitive dose-responsive adverse event in the 90-day study. The BMD analysis used Benchmark Dose Software (BMDS, version 2.2, U.S. EPA) to calculate the POD using the standard suite of models for dichotomous data.
14
-16
The total incidences of testicular lesions were plotted against the dose administered. Of the models that yielded acceptable
Results
14-Day Oral Toxicity
No NTO-induced mortality was observed in any of the dose groups (250, 500, 1000, 1500, and 2000 mg/kg/d) during the 14-day study period. Clinical signs of toxicity noted during the study period included rough hair coat, soft feces, diarrhea, chromodacryorrhea, urogenital staining, pale appearance, and low arousal and were observed primarily in male rats at dosages of 1000 mg/kg-d and above.
Body mass for males in the 2000 mg/kg-d NTO group was reduced by 6% (
Food Consumption in Rats Dosed Orally With NTO for 14 Days.a
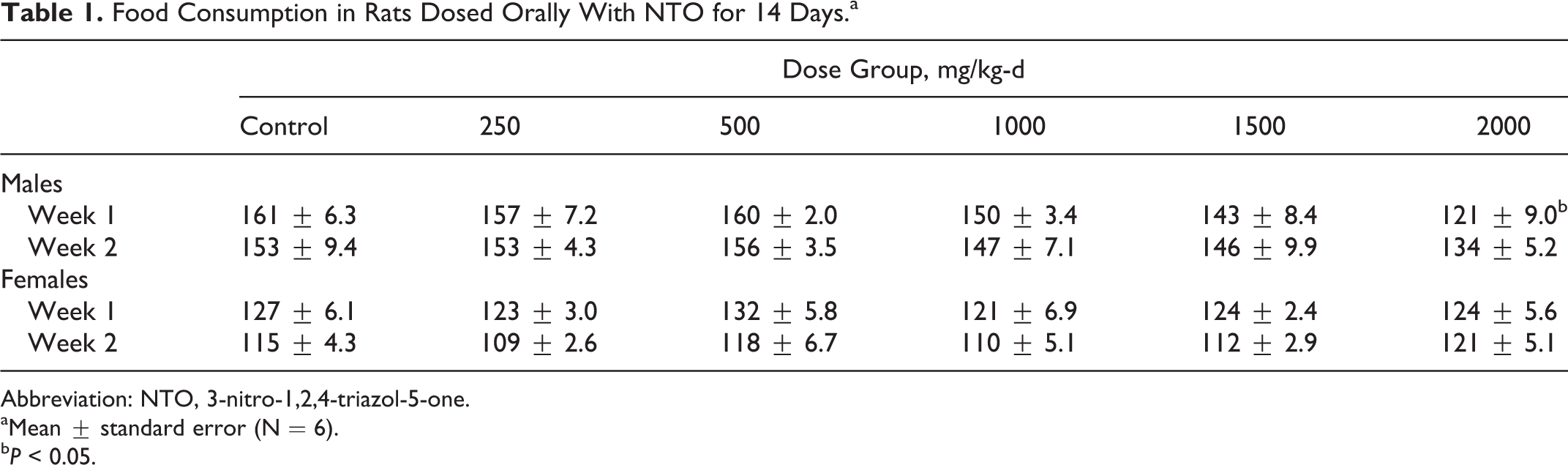
Abbreviation: NTO, 3-nitro-1,2,4-triazol-5-one.
aMean ± standard error (N = 6).
b
Body Mass (g) of Rats Orally Dosed With NTO for 14 Days.a
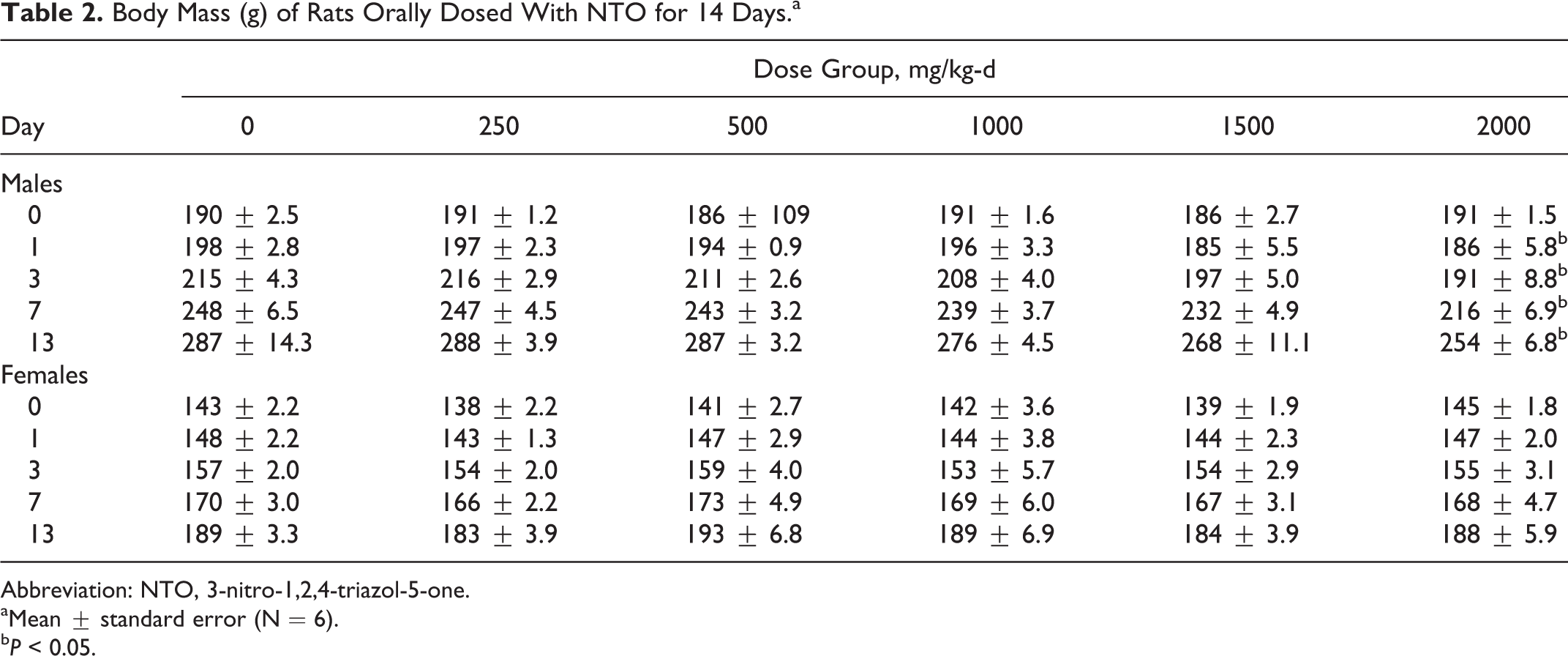
Abbreviation: NTO, 3-nitro-1,2,4-triazol-5-one.
aMean ± standard error (N = 6).
b
Hematology parameters (RBC, HGB, HCT, MCV, MCH, MCHC, RDW, PLT, MPV, WBC, NEU, LYM, MONO, EOS, and BASO) did not differ between NTO-treated and control groups. In male rats, alanine aminotransferase (ALT) and total protein (TP) levels were reduced (
Testes mass, testes-to-body mass, and testes-to-brain mass in the 500, 1000, 1500, and 2000 mg/kg-d dose groups were reduced (
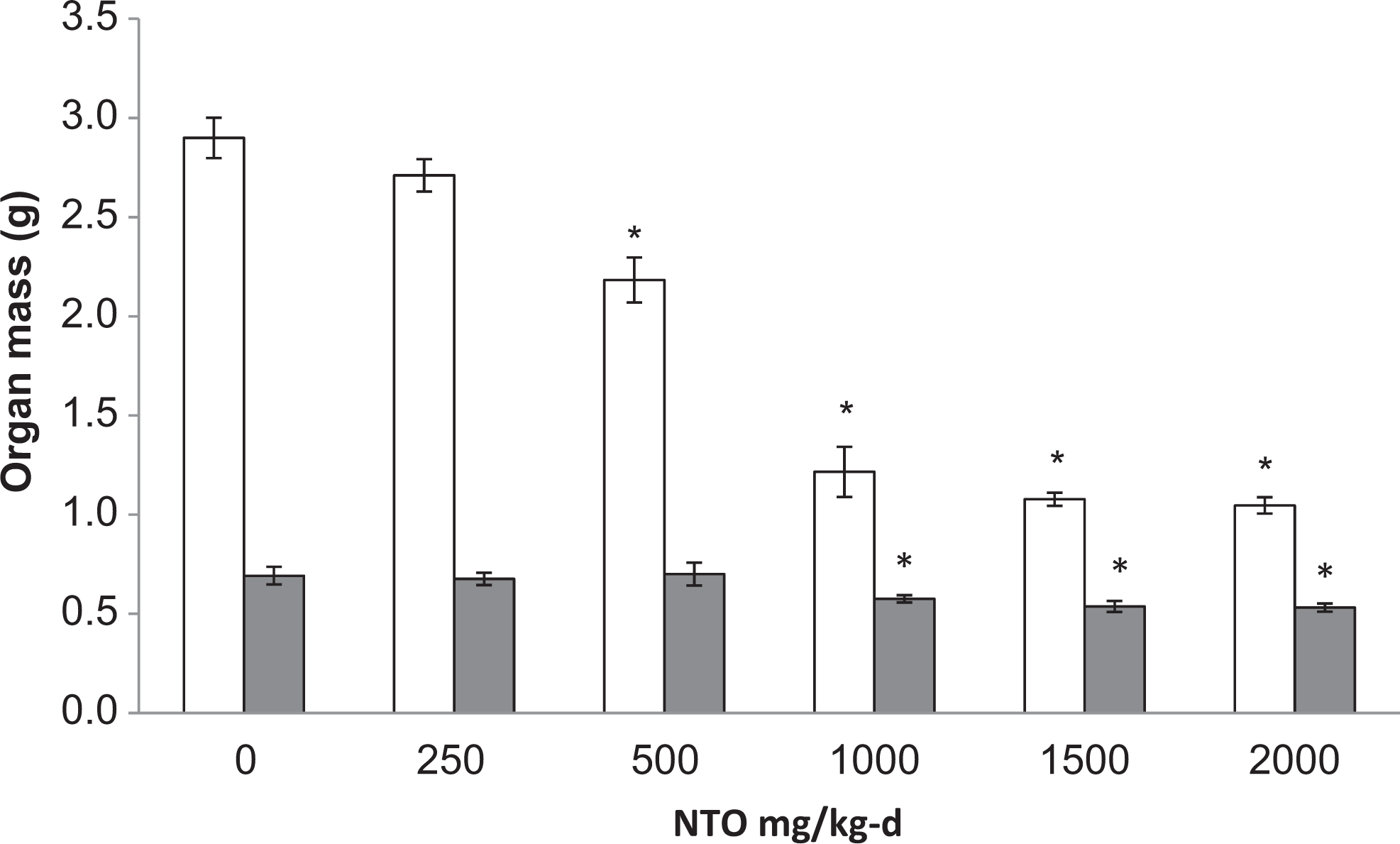
Mean mass of testes (open bars) and epididymides (solid bars) in male Sprague-Dawley rats treated orally with NTO for 14 days. Data are presented as means ± SEM for each dose group. Groups marked with an asterisk (*) are significantly different (
Gross pathology findings included 1 male rat (1500 mg/kg-d) with hydronephrosis of 1 kidney and testes that were small and soft. Findings in females included 1 rat (1500 mg/kg-d) with 3 1 mm blanched focal areas on the liver, 1 rat (250 mg/kg-d) with hemorrhagic lungs, and 1 rat (2000 mg/kg-d) with a pale liver. These findings were isolated and could not be clearly attributed to treatment.
90-Day Oral Toxicity
Observations, body mass, food consumption
There was no NTO-related mortality in any of the dose groups. Daily clinical observations were similar to those observed in the 14-day study and included changes in arousal, chromodacryorrhea, yellow stains on the ventral surface and face, dried red material on the nose and around the eyes, soft feces, and vocalization.
Food consumption did not differ between the control group and any of the NTO dose groups during the 90-day study. Body mass increased with time for all male and female dose groups and differences noted between treated and control groups were spurious and not dose or duration related.
Organ mass
Treatment-related changes in organ mass were again observed primarily for the male reproductive organs following 90 days of treatment. Mean testes mass in the 30, 315, and 1000 mg/kg-d groups was reduced (
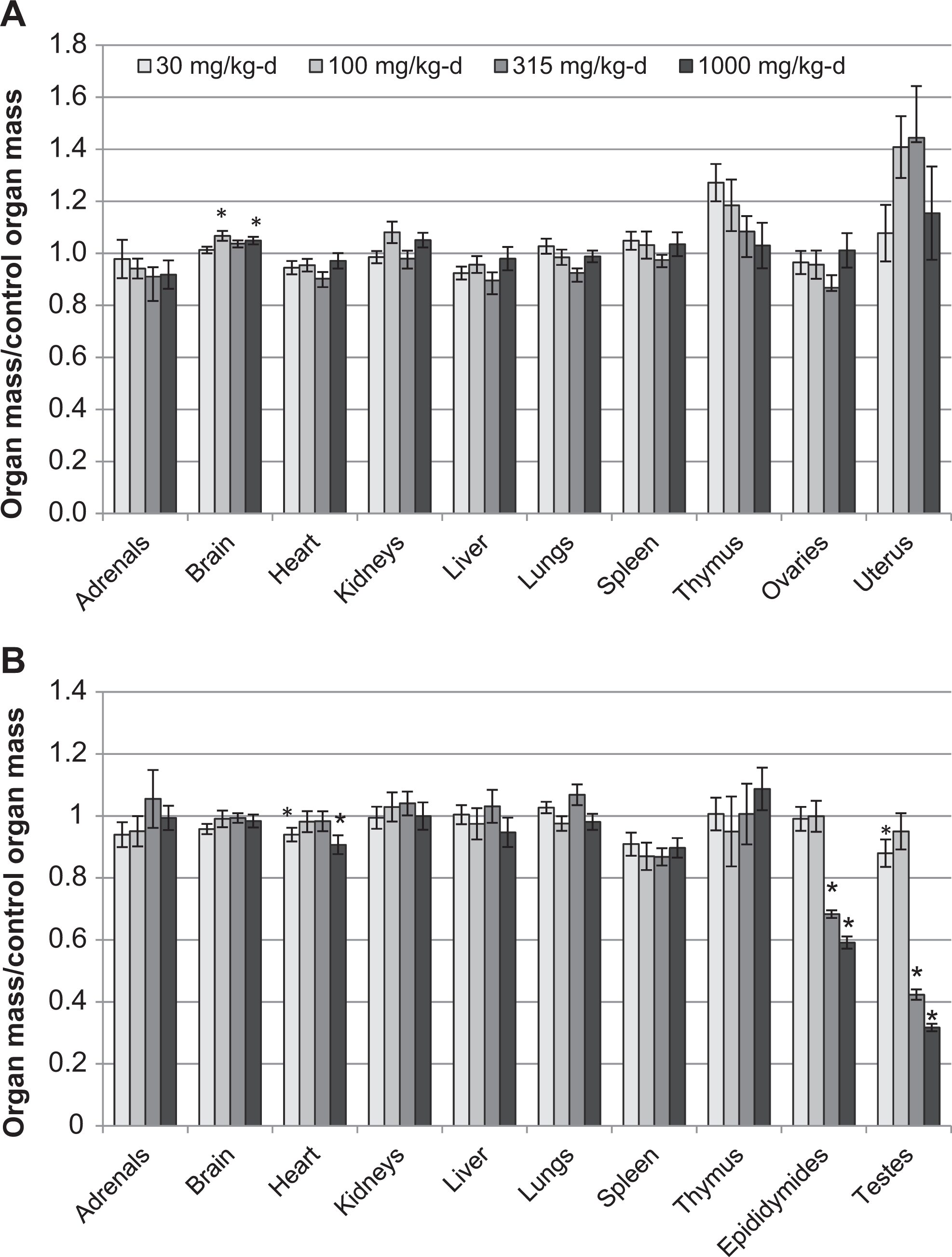
Fold change relative to controls in absolute organ mass in female (A) and male (B) Sprague-Dawley rats treated orally with NTO for 90 days. Data are presented as means ± SEM for each dose group. Groups marked with an asterisk (*) are significantly different (
Hematology and clinical chemistry
3-Nitro-1,2,4-triazole-5-one induced treatment-related changes in hematological parameters for male (RBC, MCV, MCH, and RDW) and female (MCV) rats (Table 3). Red blood cell counts of male rats demonstrated a dose-related decrease; however, this decrease (8%) was only statistically significant (
Hematology Results From Rats Dosed Orally With NTO for 90 Days.a
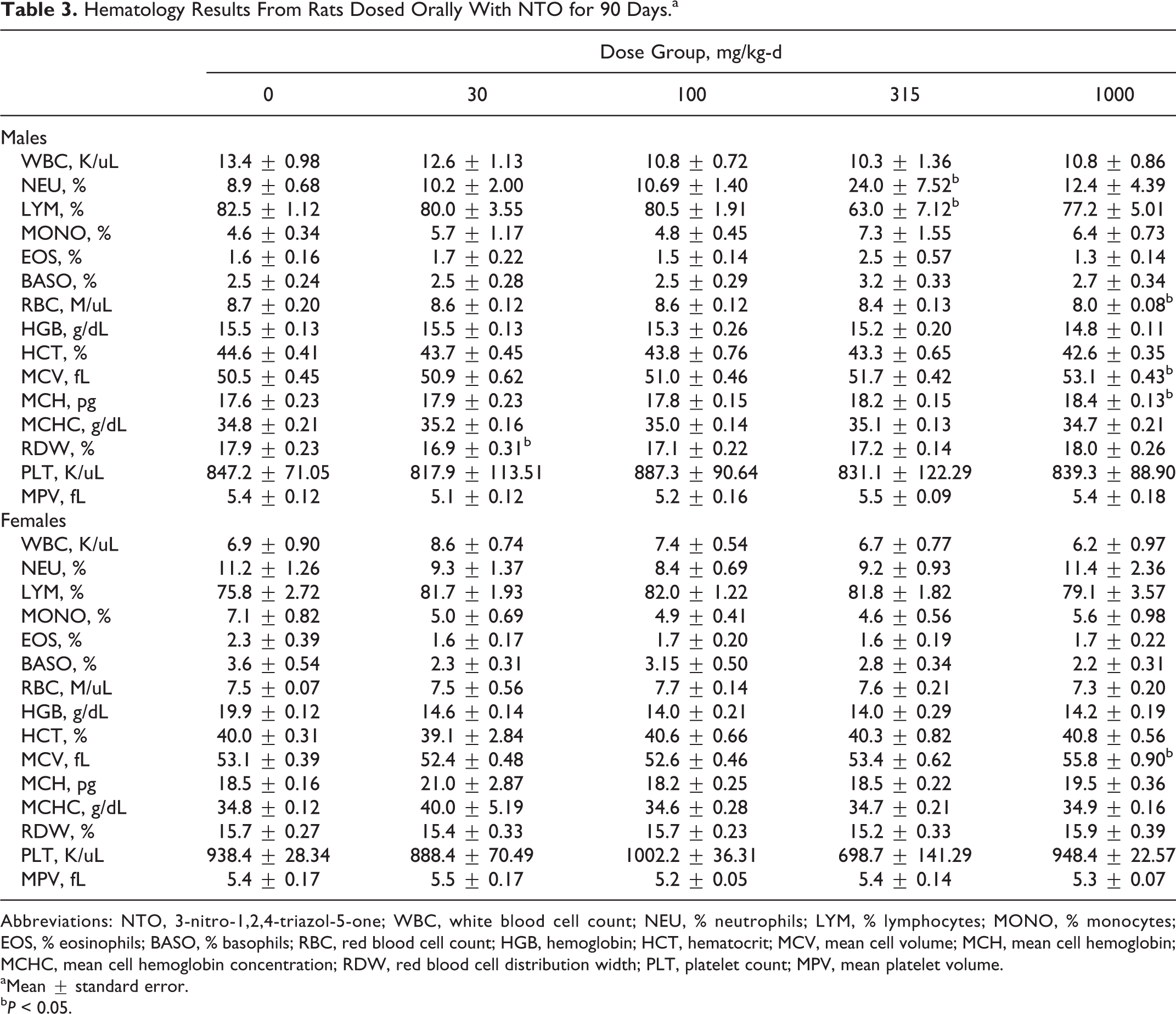
Abbreviations: NTO, 3-nitro-1,2,4-triazol-5-one; WBC, white blood cell count; NEU, % neutrophils; LYM, % lymphocytes; MONO, % monocytes; EOS, % eosinophils; BASO, % basophils; RBC, red blood cell count; HGB, hemoglobin; HCT, hematocrit; MCV, mean cell volume; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; RDW, red blood cell distribution width; PLT, platelet count; MPV, mean platelet volume.
aMean ± standard error.
b
Clinical chemistry parameters did not differ between NTO-treated groups and the control group for female rats (Table 4). In male rats, only ALB and TP demonstrated a dose–response and were reduced relative to the control (
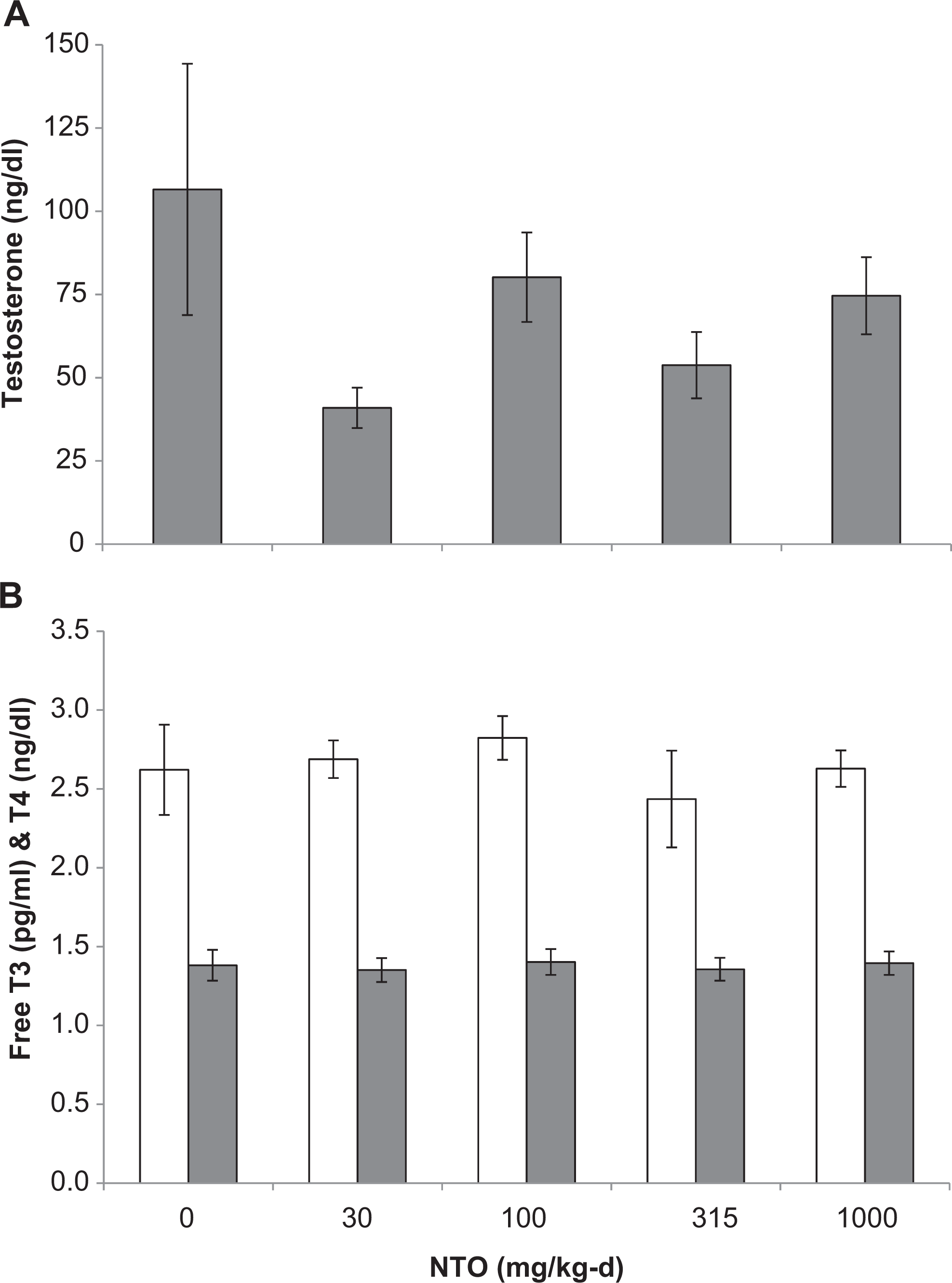
Mean testosterone (A) and free T3 (open bars) and T4 (solid bars) (B) in male Sprague-Dawley rats treated orally with NTO for 90 days. Data are presented as means ± SEM for each dose group. NTO indicates 3-nitro-1,2,4-triazol-5-one; SEM, standard error of the mean; T3, triiodothyronine; T4, thyroxine.
Clinical Chemistry Results From Rats Dosed Orally With NTO for 90 Days.a
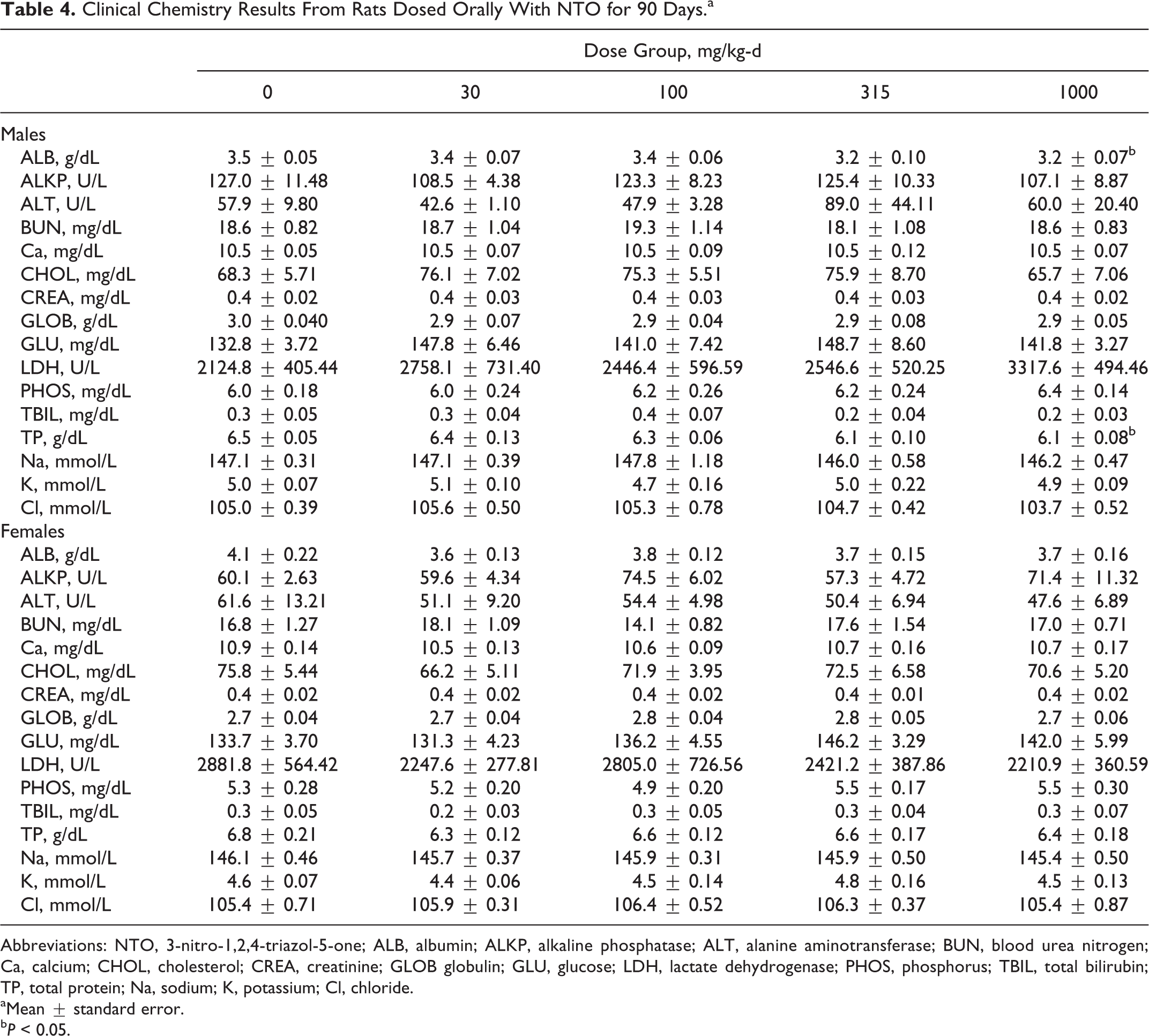
Abbreviations: NTO, 3-nitro-1,2,4-triazol-5-one; ALB, albumin; ALKP, alkaline phosphatase; ALT, alanine aminotransferase; BUN, blood urea nitrogen; Ca, calcium; CHOL, cholesterol; CREA, creatinine; GLOB globulin; GLU, glucose; LDH, lactate dehydrogenase; PHOS, phosphorus; TBIL, total bilirubin; TP, total protein; Na, sodium; K, potassium; Cl, chloride.
aMean ± standard error.
b
Urinalysis
The NTO treatment did not affect most of the urinalysis parameters including, pH, specific gravity, ketones (negative), glucose (negative), proteins, bilirubin, urobilinogen, nitrites, and leukocytes. However, urine color differed between NTO-treated groups and the control group for both males and females. For female rats, the 1000 mg/kg-d dose group had more animals with bright yellow urine compared to the control group. For male rats, the 1000 and 315 mg/kg-d dose groups had more animals with bright yellow urine compared to the control group.
Neurobehavioral evaluation
The FOB parameters (home cage, hand-held, or open arena observations) did not differ between NTO-treated and control groups. In addition, treatment with NTO did not affect any of the sensory motor or motor activity observations.
Sperm analysis
Mean sperm counts in the 315 and 1000 mg/kg-d groups were reduced by approximately 97% and 99%, respectively, when compared to controls. Sperm counts in the 30 and 100 mg/kg-d groups did not differ from controls (Figure 4).
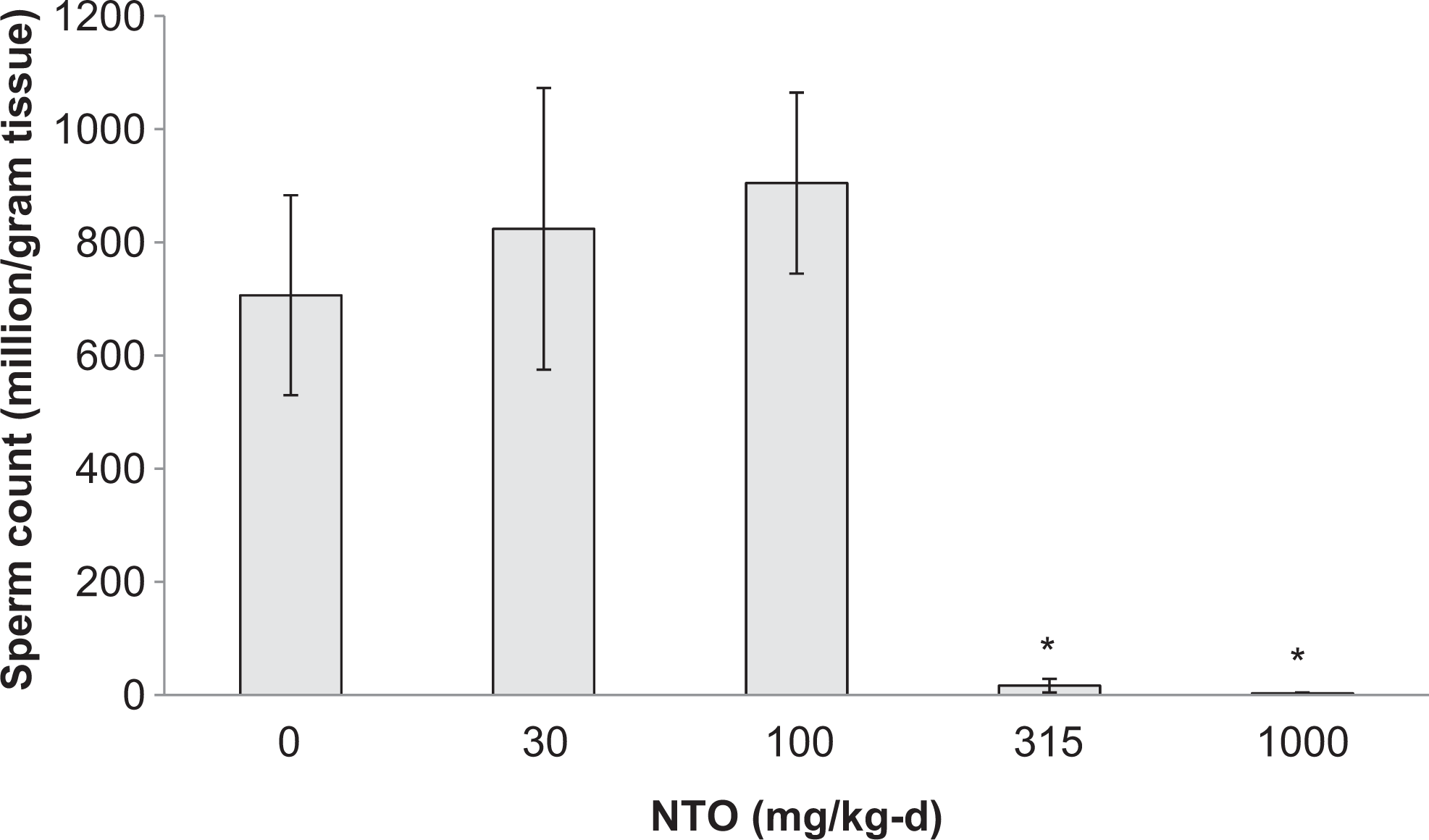
Mean epididymal sperm counts in male Sprague-Dawley rats treated orally with NTO for 90 days. Data are presented as means ± SEM for each dose group. Groups marked with an asterisk (*) are significantly different (
Histopathology
Microscopic evaluation of a full tissue list for the control and 1000 mg/kg-d animals as well as specific gross lesions for the lower dose groups indicated that the target organs were the liver and testes for NTO-treated animals. Trace to mild centrilobular hyperplasia was present in the livers of males (6 of 10) and females (8 of 9) in the 1000 mg/kg-d dose group. Microscopic findings present in the livers of all groups evaluated, including controls, were nonspecific or considered incidental changes to include inflammation, single cell necrosis, microvesiculation in the midzonal and periportal areas, and bile duct hyperplasia. Testes examined microscopically from the 315 and 1000 mg/kg-d groups consistently demonstrated moderate to severe seminiferous tubular degeneration and atrophy (6 of 7 and 9 of 9, respectively). Seminiferous tubules from the 1000 mg/kg-d group were moderately degenerate, retaining only Sertoli cells, spermatogonia, and early spermatocytes. All spermatid and late spermatocyte stages were absent in the tubules and only minimal sperm and necrotic debris was present in several epididymides. In the 315 mg/kg-d group, tubules were severely atrophic with the majority of tubules only retaining supporting Sertoli cells with no spermatic precursor cells or adult sperm. Additionally, 1 male in both the 30 and 100 mg/kg-d groups exhibited moderate to severe tubular degeneration and atrophy (Figure 5).
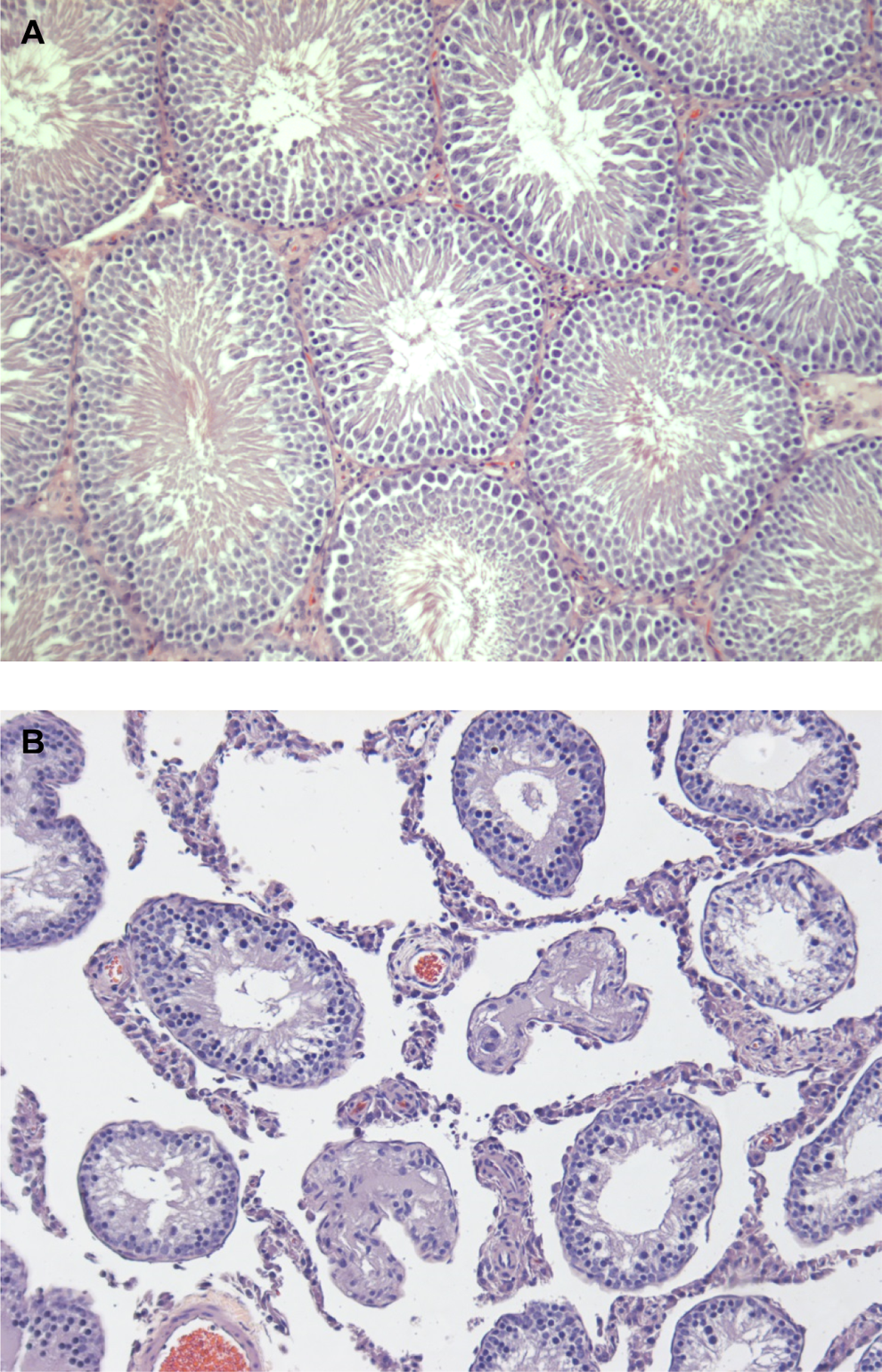
Testis from male Sprague-Dawley rats treated PEG-200 (A, ×10) or 1000 mg/kg-d NTO (B, ×10) orally for 90 days. In treated rats, seminiferous tubules were shrunken and had lost multiple germ cell layers. Often tubules were atrophic and lined only by a remaining layer of Sertoli cells. NTO indicates 3-nitro-1,2,4-triazol-5-one; PEG, polyethylene glycol.
BMD and BMDL10
The incidence of seminiferous tubular degeneration and atrophy observed in male rats was the most sensitive adverse event observed in this study. The incidence of adverse findings was used in the BMD model and the lower 95% confidence interval of a 10% response rate was calculated. The output from 9 models (Table 5) was evaluated for acceptable
Results of Benchmark Dose Modeling of Testes Histopathology in Rats Orally Dosed With NTO for 90 Days.
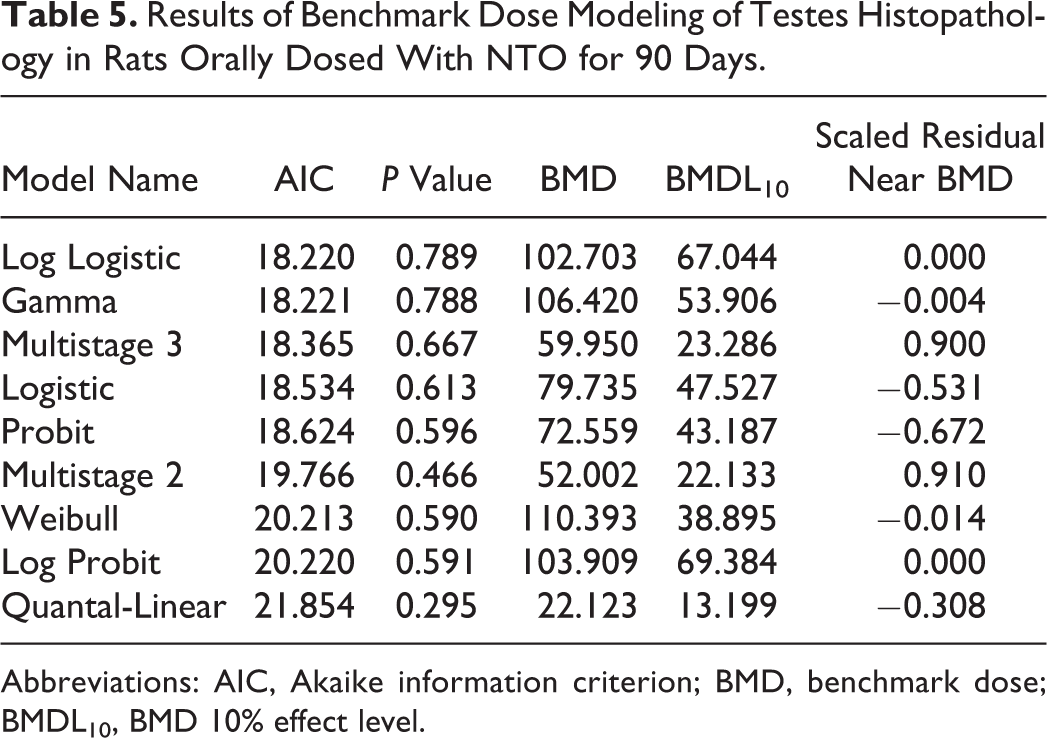
Abbreviations: AIC, Akaike information criterion; BMD, benchmark dose; BMDL10, BMD 10% effect level.
Discussion
Insensitive munitions are less sensitive to shock and high temperatures than traditional munitions and are therefore safer for military personnel to store, handle, and use. 3-Nitro-1,2,4-triazole-5-one is a key component in this group of new munition compounds. Although NTO was developed more than 100 years ago and its explosive properties were studied in the 1980s, there was very little toxicity information available. Since NTO was identified for development as a key component in the new insensitive munitions, toxicity information to support exposure criteria for manufacturing workers as well as soldiers was needed. The study reported here provides toxicity information from subacute and subchronic testing in rats.
In the 14-day study, male rats receiving 1000 mg/kg-d and greater exhibited decreased body mass and decreased food consumption, relative to control rats. Additionally, there were limited effects on clinical chemistry parameters in male rats. The most marked effects were seen in the male reproductive system. At doses as low as 500 mg/kg-d, there were decreases in testes mass. Reductions in epididymal mass also occurred at doses as low as 1000 mg/kg-d. Based on these findings, 1000 mg/kg-d was selected as the highest dose for the subchronic study.
In the 90-day study, there were no dose-related changes in mortality, clinical signs, body mass, neurobehavior, or food consumption. Treatment with NTO affected urine color, with treated animals having bright yellow urine compared to the pale yellow or straw-colored urine of control animals. This color was likely due to the presence of NTO in the urine as the NTO solutions/suspensions were bright yellow in color, especially at higher concentrations. Although Hoyt et al
7
did not find detectable levels of NTO in urine collected from rats given a single oral dose of 24 mg/kg NTO, it is still likely that the bright yellow urine color was due to NTO elimination. The urine samples in the Hoyt et al
7
study were collected at 8 hours after administration of a single oral dose, the time at which urine NTO concentrations had returned to baseline in rhesus macaques (
The most striking effects were on the male reproductive system with a marked, dose-dependent decrease in testes mass which was associated with moderate to severe seminiferous tubule degeneration and atrophy in animals receiving NTO at doses of 315 mg/kg-d and greater. Sperm counts in the 315 and 1000 mg/kg-d dose groups were also reduced by 97.7% and 99.5% of sperm counts in the control group.
The effects on the male reproductive system were confirmed in a reproductive/developmental screening test in which rats were dosed for 2 weeks prior to mating and 2 weeks during mating. 18 The male rats exhibited reduced testes mass and histological changes in the high-dose group similar to those seen in this study. Male rats in the high-dose group in the reproductive screening study also had no motile sperm. Despite the testicular toxicity and oligospermia, the NTO did not affect reproductive performance. The absence of effects on reproductive performance likely resulted from the short duration of the premating dosing period inherent in the study design.
There are no data to indicate the mode of action for the toxic effects of NTO on the testes. Virtually all of the traditional munition compounds as well as some of their precursors and breakdown products including TNT, 1,3,5-trinitrobenzene, 2,4-dinitrotoluene, and 1,3,5-dinitrobenzene (DNB) can cause testicular degeneration and reduced sperm counts. 19-23 Testicular toxicity from DNB exposure has been studied in detail, both in vivo and in vitro. 24,25 Using light and electron microscopy and biochemical end points, 1,3-DNB has been shown to target Sertoli cells. The absence of an effect of NTO on serum testosterone levels suggests that NTO testicular toxicity may be via direct cell toxicity rather than via indirect hormone-mediated effects. 26,27 Sertoli cells, responsible for the maintenance and support of germ cells, are targets of direct testicular toxicants. 26 Although Sertoli cells are sensitive to chemical damage and are a possible target of NTO, they are resistant to cell death and respond to chemical insult with biochemical disturbances. 28 Testes from rats exposed to NTO demonstrated tubular atrophy and degeneration and loss of germ cells, with only Sertoli cells being retained in the most severely affected tubules, indicating either germ cells or Sertoli cells as targets of NTO toxicity. Regardless of the target of NTO toxicity, the prolonged dosing used in this study, combined with maturation depletion and progressive germ cell loss, resulted in tubules containing only Sertoli cells. 29 The degree of damage evident in the testes provides little information on the mode of action of NTO. More in-depth studies focused on the time course of events are required to better elucidate the target cells and mechanism of toxicity. 30
The US Environmental Protection Agency initially recommended that NTO exposure criteria be based on toxicity information from amitrole (3-amino-1,2,4-triazole, CAS 61-82-5), a compound structurally similar to NTO. Amitrole is a systemic triazole herbicide. Like NTO, amitrole exhibits low acute systemic toxicity, is a mild skin and eye irritant, and is nonmutagenic in in vitro tests. Amitrole is a developmental and reproductive toxicant in both rats and rabbits, causing reduced fertility and a number of fetal malformations. In chronic cancer bioassays, amitrole produced liver and thyroid cancer in mice, and pituitary and thyroid cancer in rats. 31 In the 90-day subchronic toxicity study, NTO did not induce pathological changes in the thyroid and no effects on thyroid hormone levels were observed. Although toxic effects on the testes were seen in both the 14-day and 90-day studies, the reproductive/developmental screen did not demonstrate functional reproductive changes or gross developmental effects of NTO. 18
Triazoles have also been reported to have impacts on the nervous system, including clinical signs such as hyperactivity, tremors, convulsions and postural changes, effects on neurobehavioral evaluations including decreased rearing, decreased motor activity, and cognitive developmental dysfunction, and pathological effects including lesions in the brain and peripheral nervous system, and decreases in brain mass. 32-36 Rats exposed to NTO did not demonstrate clinical signs of neurotoxicity and no brain lesions were found. Additionally, the neurobehavioral evaluation including FOB and motor activity did not demonstrate effects of NTO on the nervous system during the 90-day study. The only changes in brain mass observed in this study occurred in brain-to-body mass ratios in males in the 2000 mg/kg-d group in the 14-day study and in absolute mass in females in the 100 and 1000 mg/kg-d groups in the 90-day study. These changes were increases rather than the decreases reported for other triazoles and were more likely either a spurious finding or, in the case of the mass ratios, attributable to the decreases in body mass. There is evidence suggesting that pesticides, such as amitrole and other triazoles, which interfere with thyroid function may also indirectly cause neurotoxicity by altering thyroid hormone-mediated events during development. 37 Developmental neurotoxicity has been reported for tebuconazole 35 as have effects on thyroid hormones 38 ; however, the link remains speculative. 39 NTO did not, however, affect thyroid hormone levels, indicating that thyroid-mediated developmental neurotoxicity is unlikely for NTO.
The most sensitive toxic end point from the current subchronic oral exposure in rats was testicular degeneration and atrophy with associated oligospermia. The incidence of adverse findings was used as a point of departure to perform a BMD extrapolation of the data. In the BMD model, the lower 95% confidence interval of a 10% response rate was calculated. 14,16 This BMDL10 of 40 mg/kg-d can be used by regulators and other public health professionals to develop guidance values to protect the health of military and civilian populations who may be exposed to NTO.
Footnotes
Acknowledgments
The authors gratefully acknowledge the following people for their assistance with this project: Theresa L. Hanna, Matthew A. Bazar, Patricia A. Beall, Michael J. Quinn, Arthur J. O’Neill, Mark R. Way, Wilfred C. McCain, Martha L. Thompson, and Karl P. Kroeck.
Authors’ Note
The views expressed in this article are the views of the authors and do not reflect the official policy of the Department of the Army, the Department of Defense, or the US government.
Author Contribution
Crouse, L contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; gave final approval; and agree to be accountable for all aspects of work ensuring integrity and accuracy. Lent, E contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; agree to be accountable for all aspects of work ensuring integrity and accuracy. Leach, G contributed to conception and design; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: funded by the U.S. Army Armament Research, Development, and Engineering Center (MIPR number MIPR8FGM009485, 2008).
