Abstract
4-Amino-2-nitrotoluene (4A2NT; CAS 119-32-4) is a degradation product of 2,4-dinitrotoluene. The toxicity data on 4A2NT are limited. Therefore, we collected toxicity data from rats to assess environmental and human health effects from exposures. The approximate lethal dose for both sexes was 5000 mg/kg. A 14-day toxicity study in rats was conducted with 4A2NT in the feed at concentrations of 0, 125, 250, 500, 1000, and 2000 ppm. Based on a 14-day oral dose range toxicity study with 4A2NT in the feed, 2000 ppm was selected as highest concentration for a subsequent 90-day study. An oral 90-day subchronic toxicity study in rats was conducted with concentrations of 0, 500, 1000, or 2000 ppm of 4A2NT in the feed. The calculated consumed doses of 4A2NT in the feed were 0, 27, 52, or 115 mg/kg/d for males and 0, 32, 65, or 138 mg/kg/d for females. A no-observed adverse effect level could not be determined. The lowest observed adverse effect level was 27 mg/kg/d for males and 32 mg/kg/d for female rats based upon decreased body weight gain. The decreased body weight gain in male rats was the most sensitive adverse event observed in this study and was used to derive a benchmark dose (BMD). A BMD of 23.1 mg/kg/d and BMD with 10% effect level of 15.5 mg/kg/d were calculated for male rats, which were used to derive an oral reference dose (RfD). The human RfD of 1.26 μg/kg/d was derived using current United States Environmental Protection Agency guidelines.
Introduction
2,4-Dinitrotoluene (2,4-DNT) is used in the manufacture of dyes and military explosive materials. It is degraded into a number of nitro reductive products, including 4-amino-2-nitrotoluene (4A2NT; CAS 119-32-4) through in vivo mammalian and bacterial metabolism.1–3 The 4A2NT is also an environmental degradation product of 2,4-DNT and is detected in soil and ground water at some ammunition plant manufacturing and disposal facilities. 4 There is considerable information on the toxicity of 2,4-DNT and 2,6-dinitrotoluene (2,6-DNT),2,3 but there are limited toxicity data on their metabolites. The 2A4NT (2-methyl-5-nitroaniline; CASRN 99-55-8) is one of the metabolites of 2,4-DNT, which is also an intermediate compound in the synthesis of a wide range of azo dyes and has shown to be carcinogenic in mice but not in rats. 5
The toxicity data on 4A2NT are limited. The acute oral lethal dose, 50% (LD50) of 4A2NT in wild birds was 3.2 mg/kg for red-winged blackbirds (
The aquatic toxicity study results show that the LC50 values of 4A2NT were 12 mg/L for rainbow trout (
Materials and Methods
Chemical
4-Amino-2-nitrotoluene (CAS 119-32-4, 2-nitro-4-aminotoluene, 4-methyl-3-nitroaniline, and 5-nitro-4-toluidine; Figure 1) was purchased from Aldrich Chemical Co Inc (Milwaukee, Wisconsin; lot number 07006BX, 97% pure). It was a rust-colored powder. All other chemicals used in this study were of analytical grade.
4-Amino-2-nitrotoluene.
Animals
Male and female Sprague Dawley rats were obtained from Charles River Laboratories, Wilmington, Massachusetts. The animals were examined by the attending veterinarian and found to be in acceptable health. The male rats had a weight range of 198 to 231 g, and the female rats had a weight range of 127 to 176 g. Animals were quarantined for a 1-week period before experiments. Rats were provided with water and Purina Certified Rodent Chow 5002 ad libitum. Animal use procedures were approved by the Animal Care and Use Committee at the United States Army Environmental Hygiene Agency (currently known as US Army Public Health Command [USAPHC]). Animal care and use was conducted in accordance with the principles stated in the Guide for the Care and Use of Laboratory Animals and in accordance with all applicable Federal and Department of Defense regulations. 9 The USAPHC animal care program is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International.
Methods
Diet Preparation
4-Amino-2-nitrotoluene powder was gently ground in a mortar and pestle and mixed with ground rat chow. The compound and feed were mixed weekly for 25 to 30 minutes using a Hobart mixer (Columbus, Ohio). The mixed feed lots were randomly sampled and verified for the concentration of 4A2NT via gas chromatography. After mixing, the feed was refrigerated until use. The feed was added to feeders 3 times per week with fresh feed. Consumed feed was measured to estimate the actual dose administered during the feeding study.
Acute and 14-Day Subacute Oral Toxicity
An approximate lethal dose (ALD) procedure was performed to determine the lethality in 14 days from a single oral dose of 4A2NT suspended in polyethylene glycol 200 (PEG 200). Doses of 987, 1489, 2222, 3333, 5000, or 7500 mg/kg were given to male and female rats. The 14-day feeding study was conducted in male and female rats (6 per group) at dose levels 0 (control), 125, 250, 500, 1000, and 2000 ppm. Body weight and feeder weights were recorded on days 0, 1, 3, 7, and 14. At 14 days, blood samples for hematology and clinical chemistry and selected tissues for organ weights were collected at the termination of experiments. The tissues were weighed for the calculation of organ weight-to-body weight and organ-to-brain weight ratios. The hematology parameters studied were white blood cell (WBC) count, red blood cell (RBC) count, hemoglobin (HGB), hematocrit (HCT) mean cell volume, mean cell hemoglobin (MCH), mean cell hemoglobin concentration, and platelet (PLT) count. For clinical chemistry parameters, the following parameters were measured: alkaline phosphatase (ALKP), serum glutamic pyruvic transferase (SGPT), serum glutamic oxaloacetic transferase (SGOT), blood urea nitrogen (BUN), calcium (C), cholesterol (CHOL), glucose (GLU), total bilirubin (TBIL), total protein (TP), and triglyceride (TRIG).
90-Day Feeding Study
Five groups of male and female rats (5 per group) were fed a diet containing 4A2NT at concentrations of 0, 500, 1000, or 2000 ppm based on the high dose selected from the 14-day feeding study. The calculated consumed doses from the feed were 0, 27, 52, and 115 mg/kg/d for males and 0, 32, 65, and 138 mg/kg/d for females. Body weights were recorded, and feed consumptions were calculated, and all animals were observed for any clinical signs of toxicity. After 90 days, blood samples were collected for hematology and clinical chemistry, and selected organs were taken and weighed for organ-to-body weight and organ-to-brain weight ratio calculations. The selected tissues were trimmed and preserved in formalin, and evaluations for histopathology were performed. The hematology and clinical chemistry parameters studied were similar to the one described in 14-day studies.
Statistical Analysis
Food consumption, body weights, and weight gains were statistically compared using analysis of variance, and when significance was observed, the data were further analyzed using a Duncan’s post hoc test. Hematology values, clinical chemistry values, organ-to-body weight ratios, and organ-to-brain weight ratios were statistically compared using an analysis of variance and a Newman–Kuels post hoc test. 10
Determination of BMD and Human Equivalent Dose
Benchmark dose is an alternative approach to the no-observed adverse effect level (NOAEL)/lowest observed adverse effect level (LOAEL) approach in the development of a point of departure (POD) that has been used for many years in dose–response assessment. The BMD method gained favor with the risk assessment community, because it incorporates and conveys more information than NOAEL/LOAEL method. The dose–response assessment is a 2-step process, one defining POD and other is extrapolating from POD for relevance to environmental exposure. We developed an oral RfD for 4A2NT according to the United States Environmental Protection Agency (USEPA) BMD approach. 11
The BMD and its 95% lower confidence limit for 10% effect levels (BMDL10) were determined from the dose–response relationship of the decrease in body weight gain of rats in the 90-day oral feeding study, the most sensitive dose-responsive adverse event in this study. The BMD analysis used Benchmark Dose Software (BMDS, version 2.2) to calculate the POD using the standard suite of models for continuous data.12–14 The average weights and standard deviation for each dose group were entered into the BMDS. To best fit the data into traditional BMD format, the absolute value of the difference in average weight of the dose groups from the control group was plotted against the dose administered. Of the models that yielded acceptable data fits, those with large-scaled residual were excluded. The mean of the BMDL10 of the remaining models was used as the POD for interspecies extrapolation from rats to humans. A dose metric adjustment factor (DAF) was calculated and used to derive the human equivalent dose (HED). 15
Results
Acute and Subacute Oral Toxicity
The earliest toxic effects of 4A2NT were observed at 3 hours, and deaths occurred in between 21 and 42 hours of treatment. The lethal dose occurred at 5000 to 7500 mg/kg in PEG 200 vehicle for both the sexes.
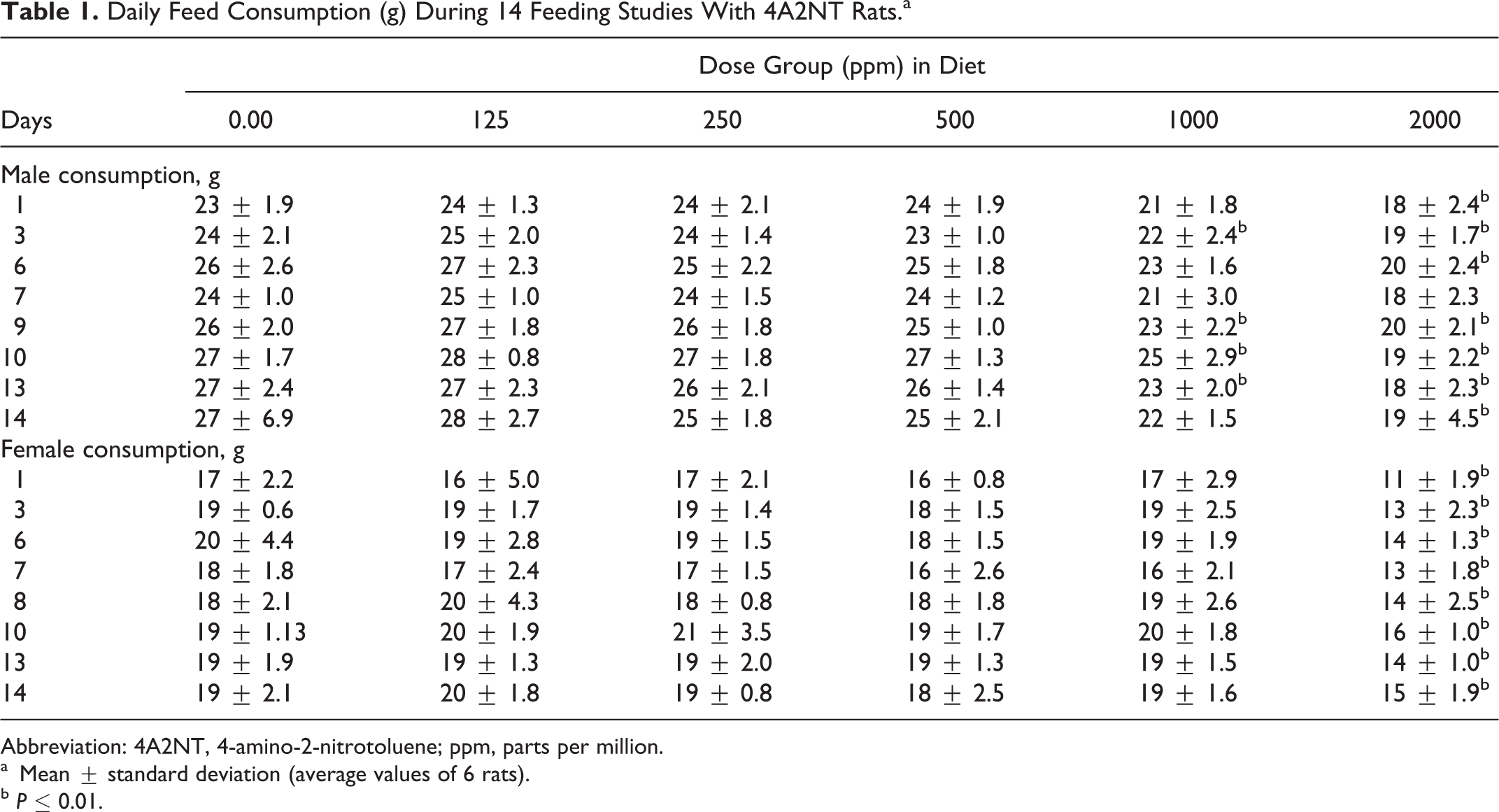
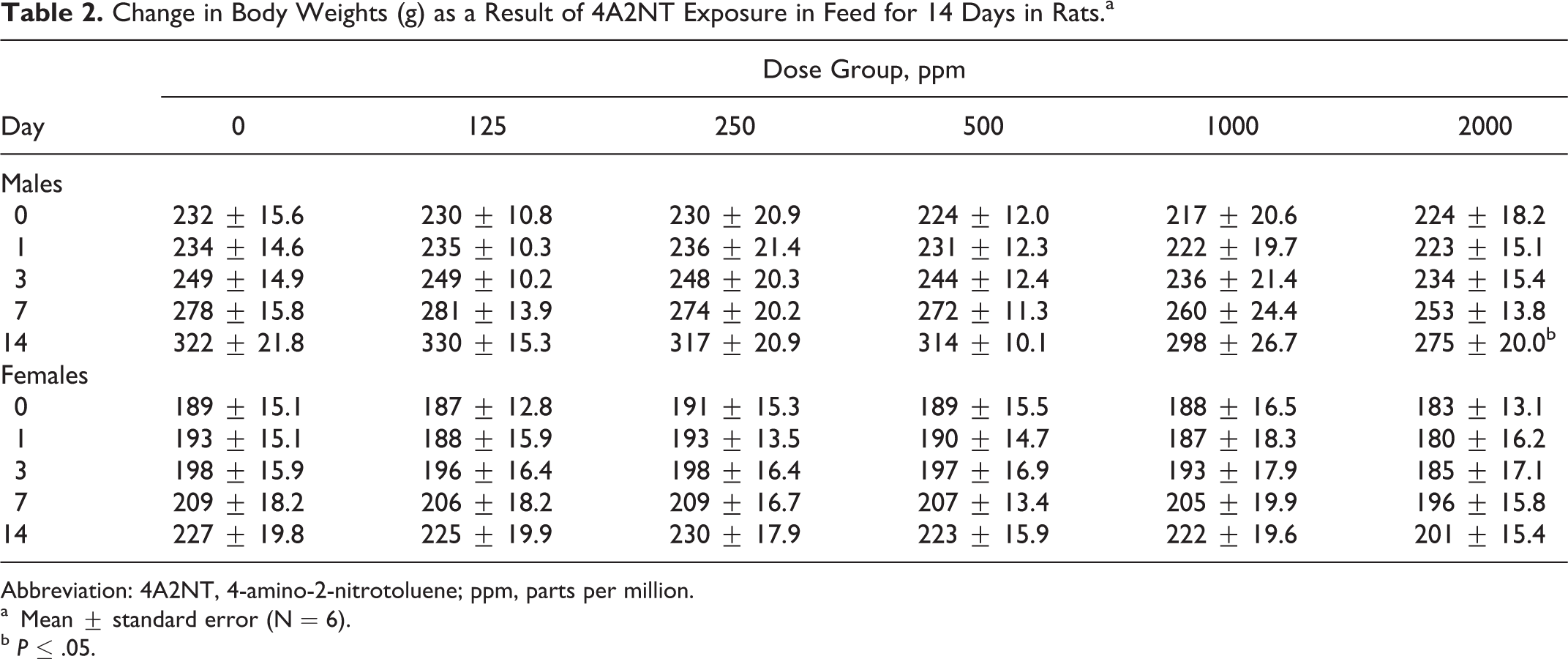
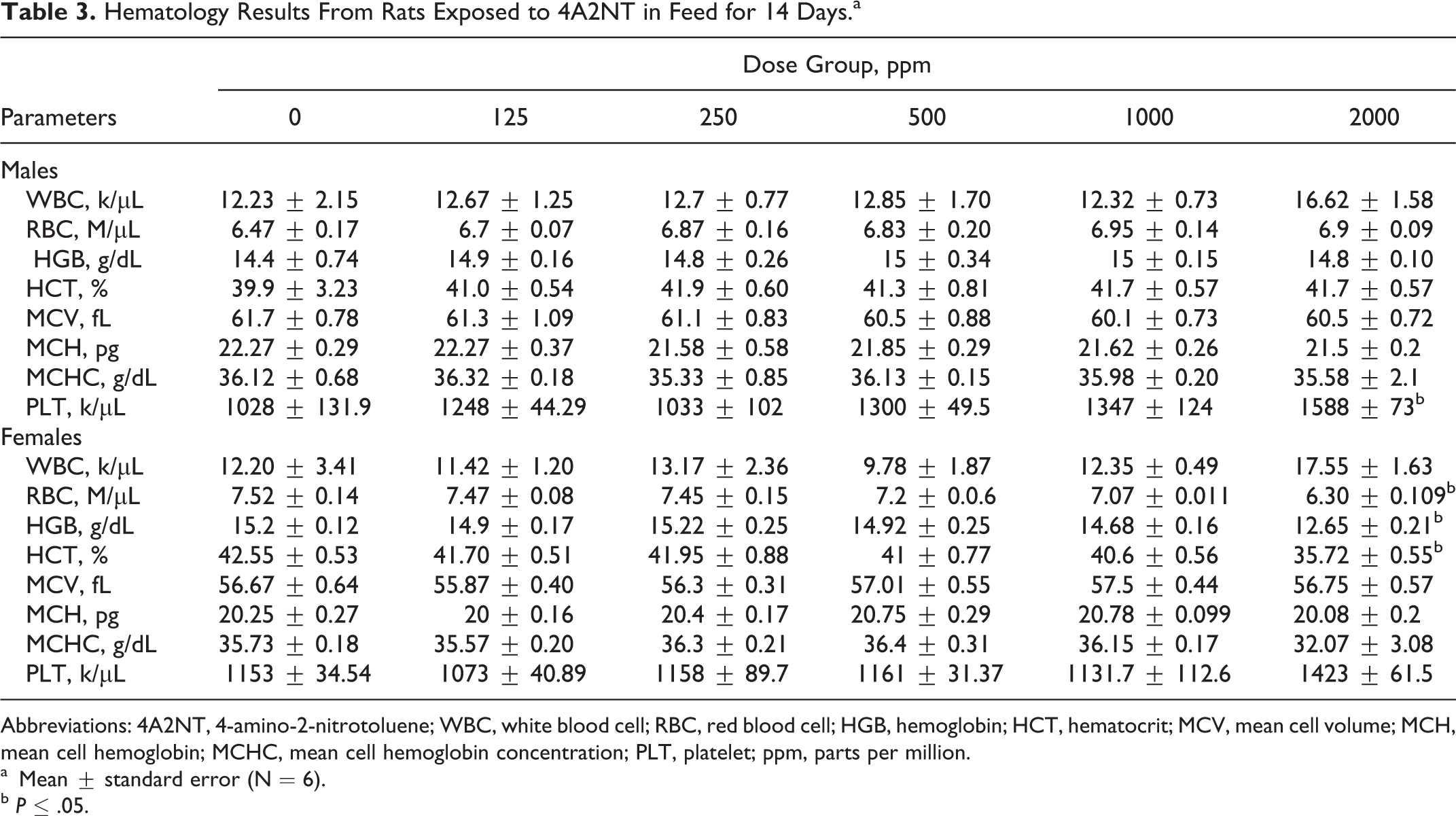
In the 14-day feeding study, all animals survived fed on diet containing 4A2NT at all dose levels (125, 250, 500, 100, and 2000 ppm) to the end of study. No clinical signs of toxicity were observed. Rats fed on the diet containing 4A2NT for 14 days showed a significantly reduced feed consumption in males (1000 and 2000 ppm) and in females (2000 ppm; Table 1). The food consumption is reduced to about 25% in the high-dose group. The body weight results of rats showed that at high dose (2000 ppm), both sexes showed decreased body weight gains, but it was significantly different in males (Table 2). In female rats at high dose, hematological parameters such as HGB, HCT, and RBC counts were decreased, indicating anemia; in males, only PLTs were increased at high dose (Table 3). The clinical parameters were increased in males (CHOL and TP) and in females (TP) at high dose (Table 4). The body weight-to-brain and testis weight ratios in males were significantly increased, while in females the body weights to liver, brain, and spleen were significantly increased at high dose when compared to controls. There is a significant increase in testis-to-brain weight ratios in males and adrenal-to-brain weight ratios in females at high dose when compared to controls 16 (data are not shown).
Daily Feed Consumption (g) During 14 Feeding Studies With 4A2NT Rats.a
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
a Mean ± standard deviation (average values of 6 rats).
b
Change in Body Weights (g) as a Result of 4A2NT Exposure in Feed for 14 Days in Rats.a
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
a Mean ± standard error (N = 6).
b
Hematology Results From Rats Exposed to 4A2NT in Feed for 14 Days.a
Abbreviations: 4A2NT, 4-amino-2-nitrotoluene; WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean cell volume; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; PLT, platelet; ppm, parts per million.
a Mean ± standard error (N = 6).
b
Serum Chemistry Results From Rats Exposed to 4A2NT in Feed for 14 Days.a
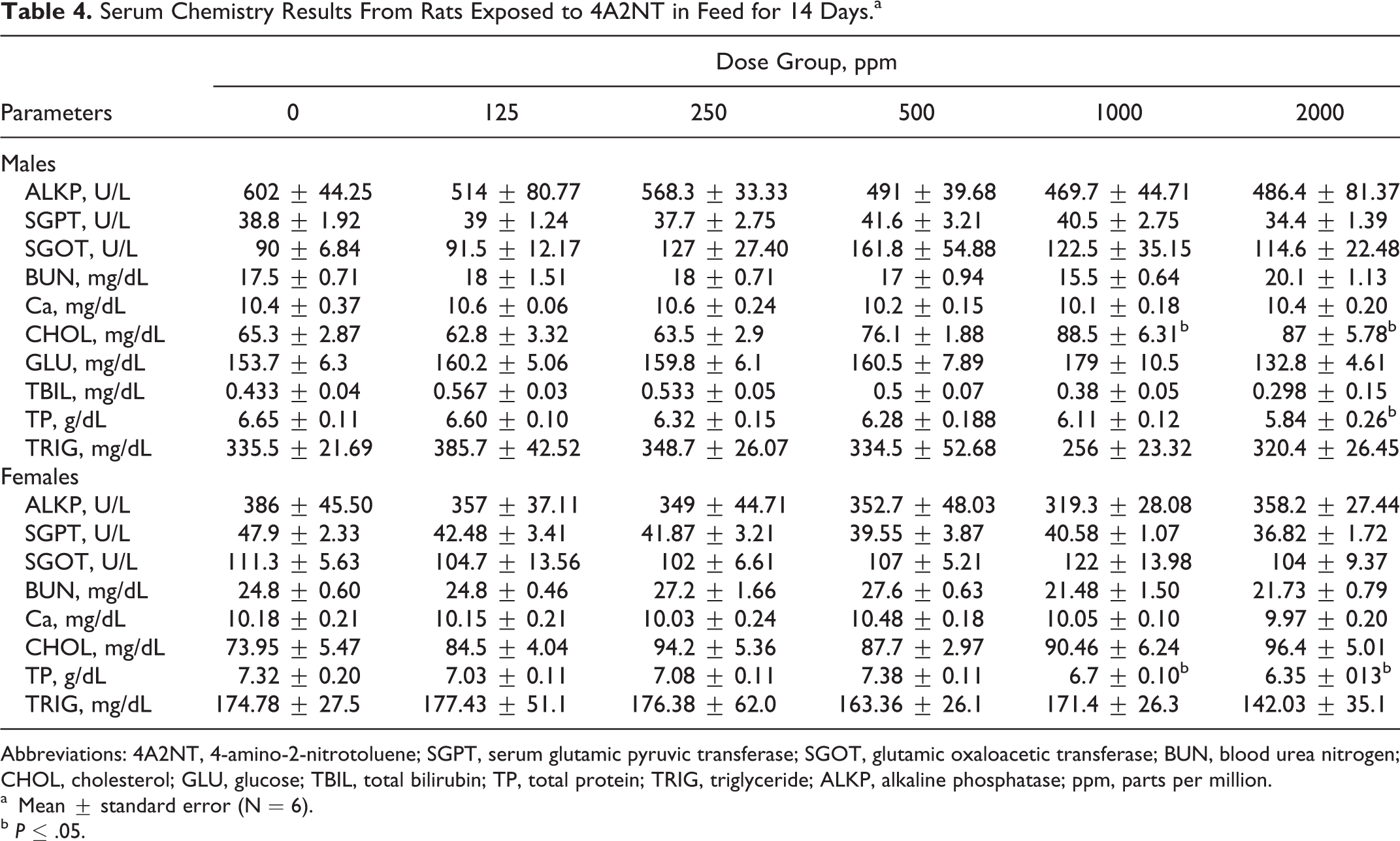
Abbreviations: 4A2NT, 4-amino-2-nitrotoluene; SGPT, serum glutamic pyruvic transferase; SGOT, glutamic oxaloacetic transferase; BUN, blood urea nitrogen; CHOL, cholesterol; GLU, glucose; TBIL, total bilirubin; TP, total protein; TRIG, triglyceride; ALKP, alkaline phosphatase; ppm, parts per million.
a Mean ± standard error (N = 6).
b
90-Day Oral Feeding
During the 90-day period, rats were fed on diet containing predicted concentrations of 0 (control), 500, 1000, or 2000 ppm 4A2NT. The consumed average doses calculated for males were 0, 27, 52, or 115 mg/kg/d and for females were 0, 32, 65, or 138 mg/kg/d, respectively (Table 5).
Mean Daily Consumption of 4A2NT by Rats During 90-Day Feeding.
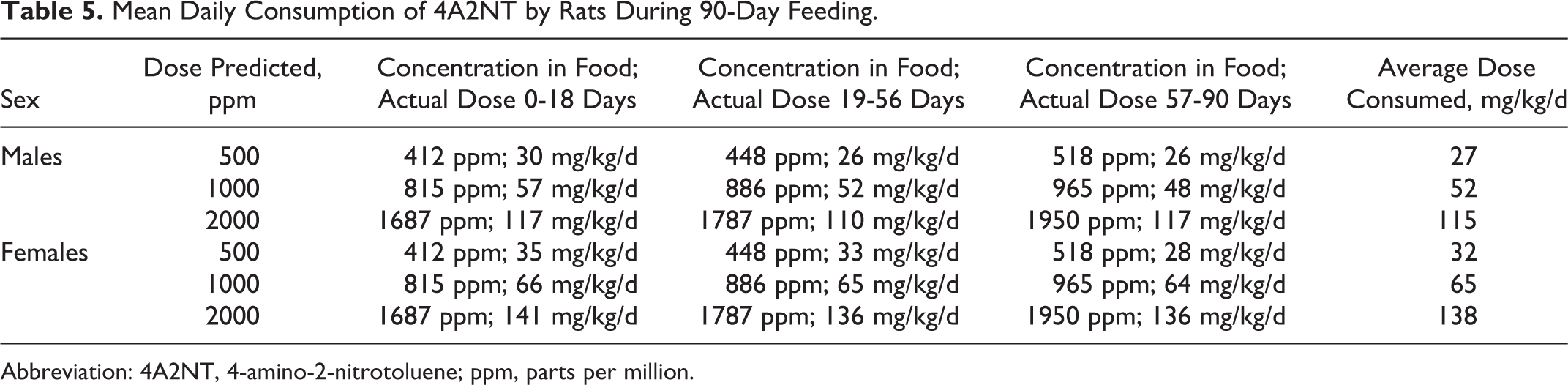
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
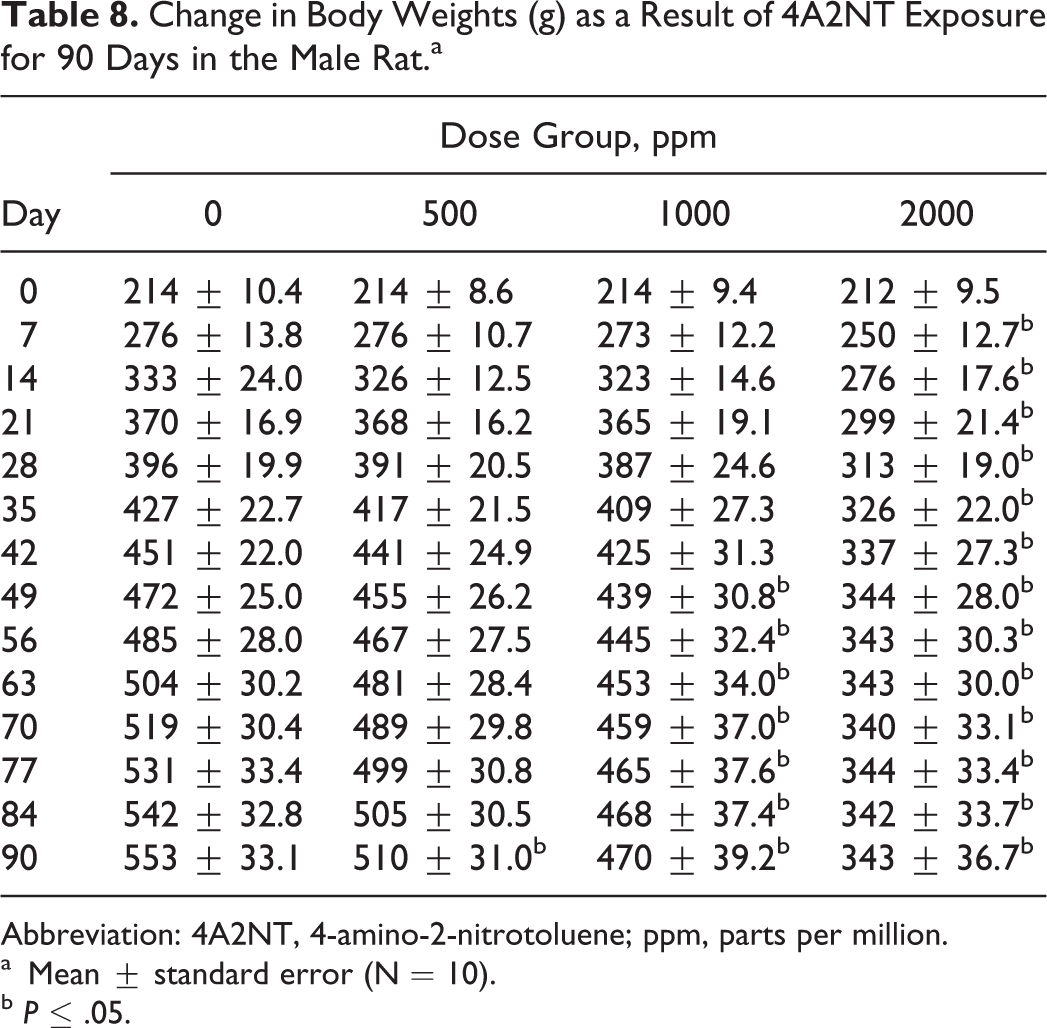
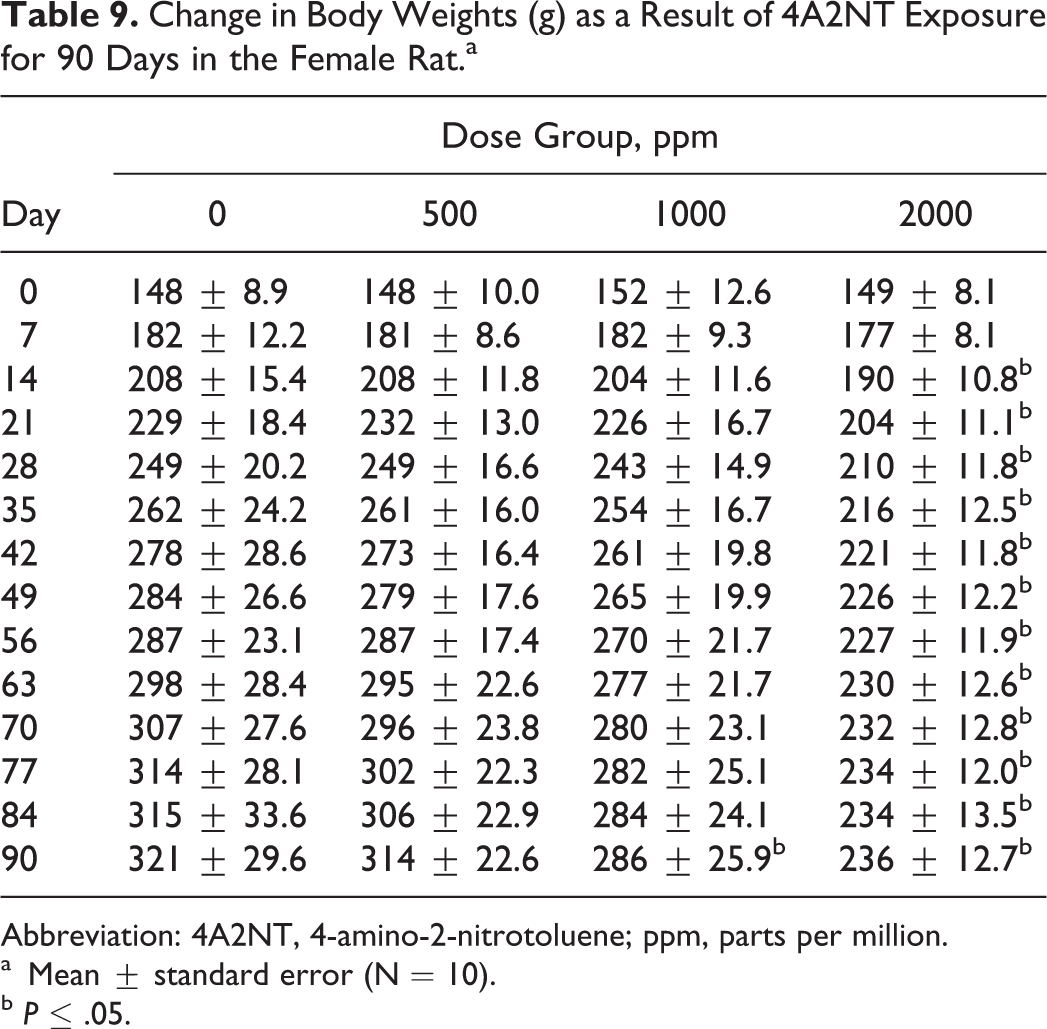
The results of food consumption and body weights are presented in Tables 6–9. Both male and female rats showed significant decreases in body weight gain in the 2 high-dose groups during 90-day period. The decreased body weight gain was observed in males at all dose groups 27, 52, or 115 mg/kg/d (550, 100, or 200 ppm; Table 8), while in females was observed in 2 high-dose groups 65 or 138 mg/kg/d (1000 or 2000 ppm; Table 9).
Food Consumption (g) in Male Rats on a Diet Containing 4A2NT During a 90-Day Feeding Study.a
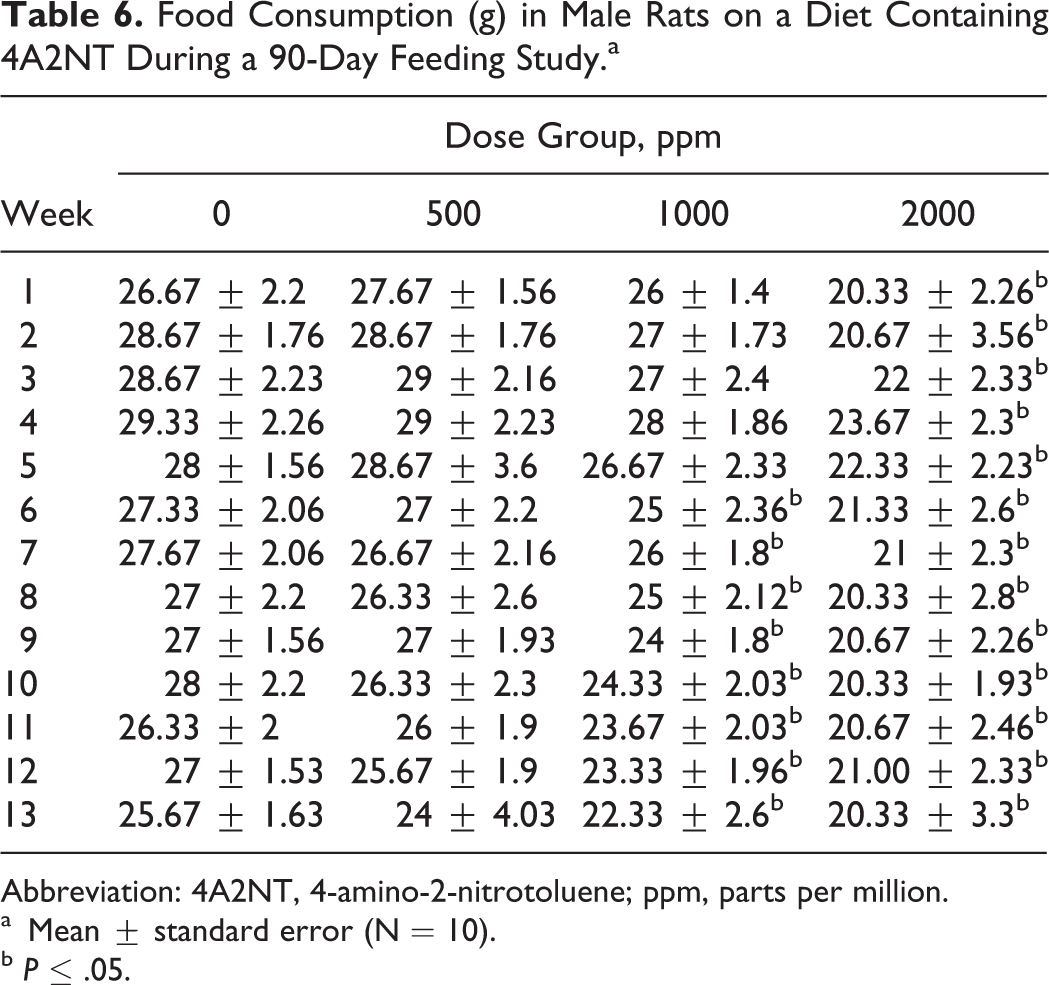
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
a Mean ± standard error (N = 10).
b
Food Consumption (g) in Female Rats on a Diet Containing 4A2NT During a 90-Day Feeding Study.a
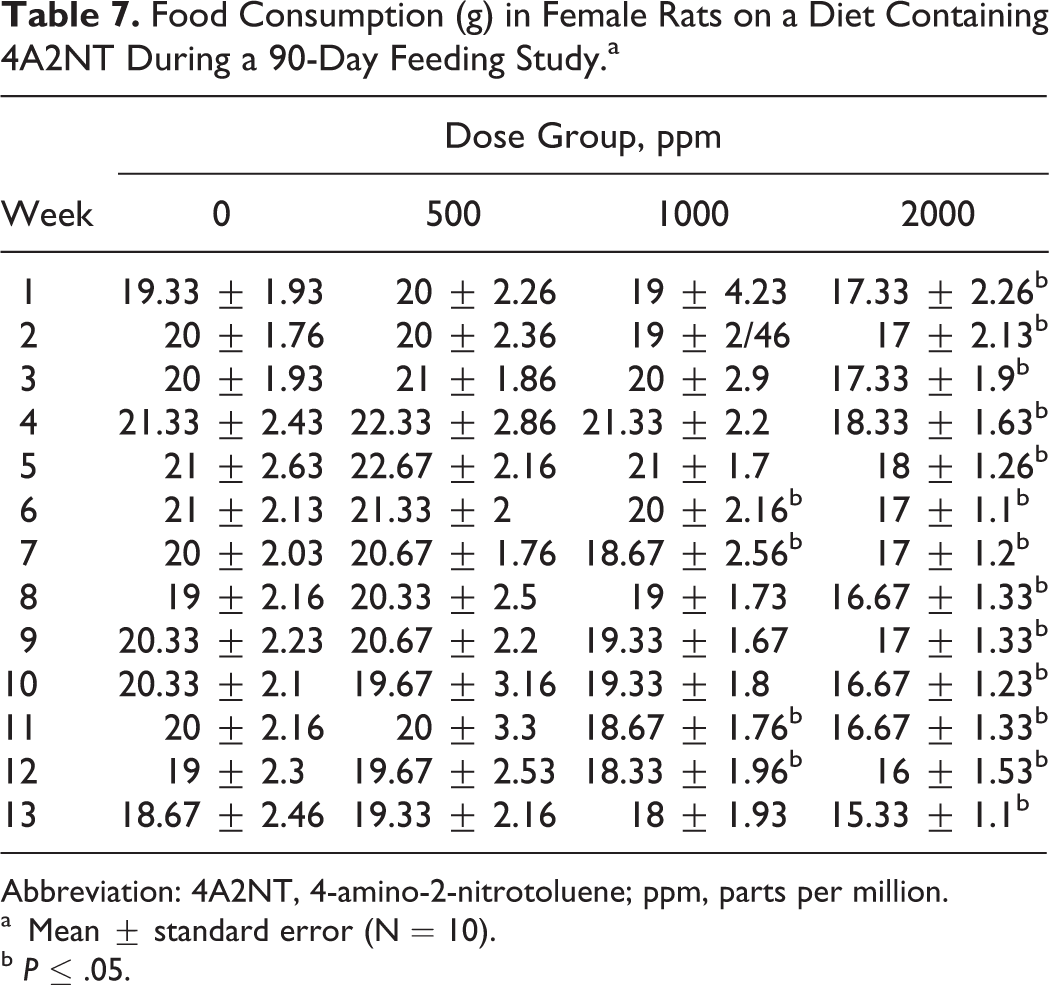
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
a Mean ± standard error (N = 10).
b
Change in Body Weights (g) as a Result of 4A2NT Exposure for 90 Days in the Male Rat.a
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
a Mean ± standard error (N = 10).
b
Change in Body Weights (g) as a Result of 4A2NT Exposure for 90 Days in the Female Rat.a
Abbreviation: 4A2NT, 4-amino-2-nitrotoluene; ppm, parts per million.
a Mean ± standard error (N = 10).
b
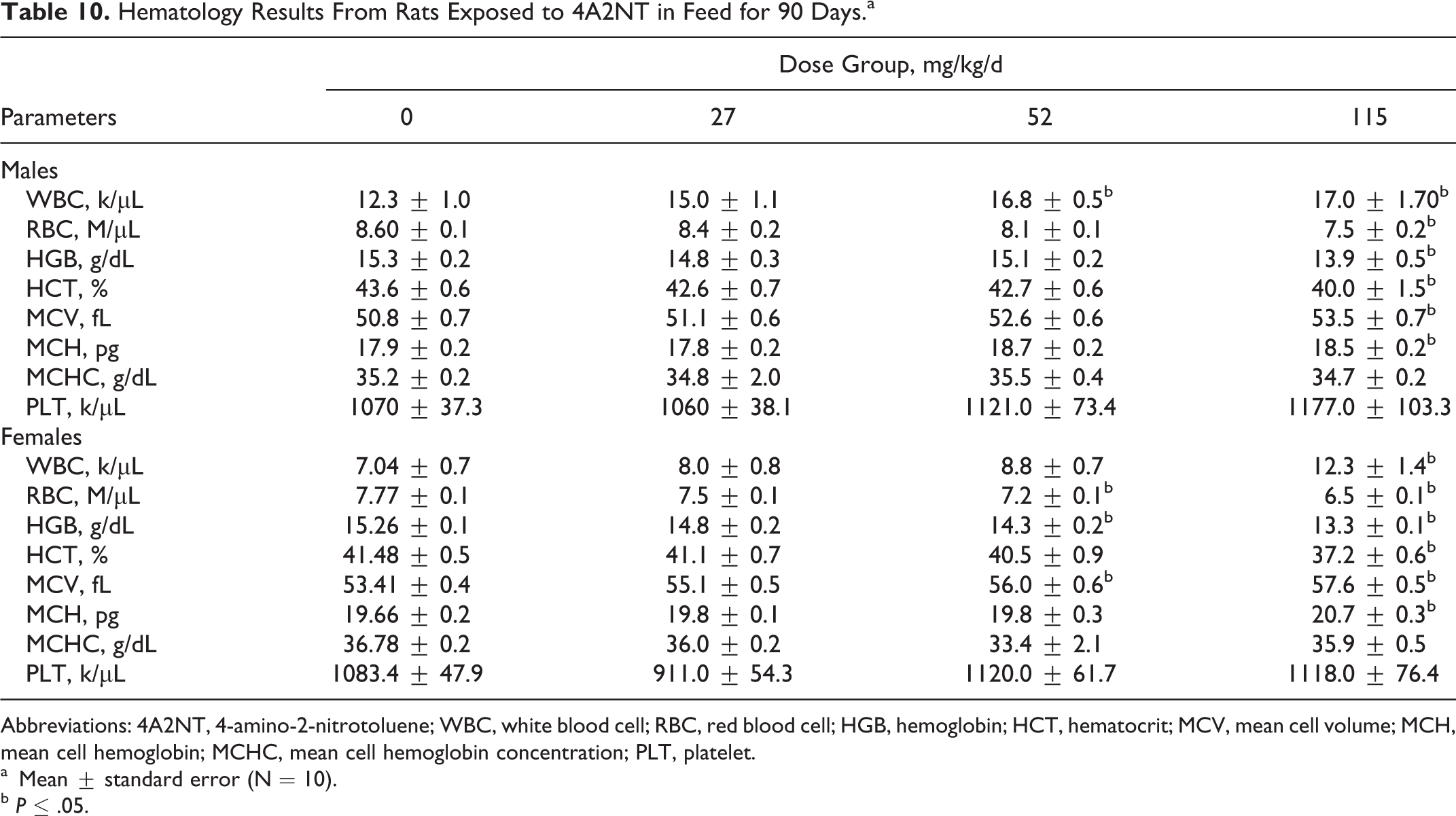
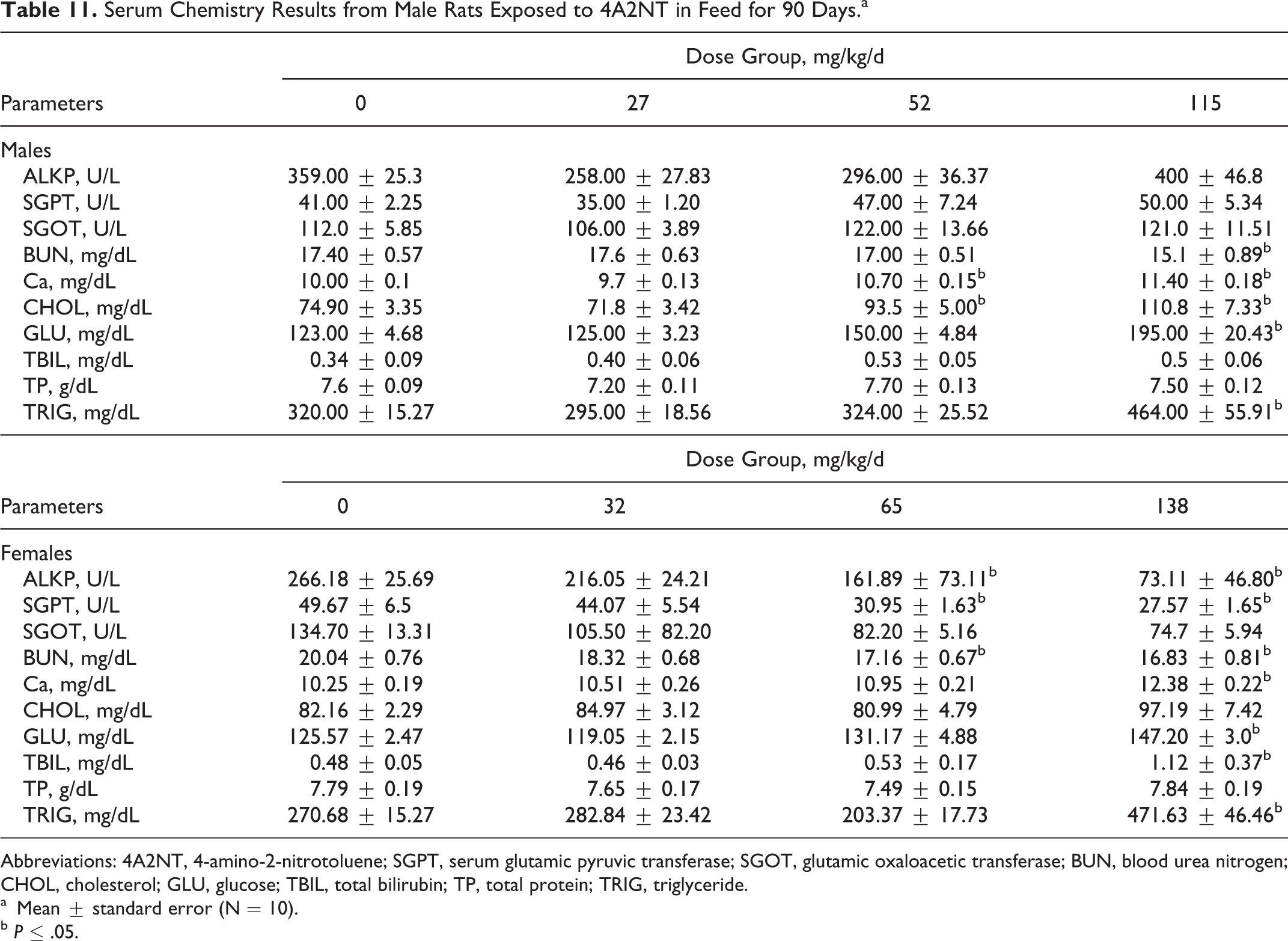
The hematological and clinical chemistry values for male and female rats are presented in Tables 10 and 11. Male (115 mg/kg/d) and female (65 and 138 mg/kg/d) rats showed decreased HGB, HCT, and RBC counts when compared to their controls, suggestive of anemia, and an increase in WBC, MVC, and MCH. The results of clinical chemistry values indicated that male rats receiving a 4A2NT dose of 52 or 115 mg/kg/d showed an increase in C, GLU, and CHOL but a decrease in BUN at the highest dose. Females receiving 65 and 138 mg/kg/d showed decreases in ALKP, SGPT, SGOT, and BUN and increases in CHOL, GLU, TBIL, and TRIG in the 138 mg/kg/d group.
Hematology Results From Rats Exposed to 4A2NT in Feed for 90 Days.a
Abbreviations: 4A2NT, 4-amino-2-nitrotoluene; WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean cell volume; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; PLT, platelet.
a Mean ± standard error (N = 10).
b
Serum Chemistry Results from Male Rats Exposed to 4A2NT in Feed for 90 Days.a
Abbreviations: 4A2NT, 4-amino-2-nitrotoluene; SGPT, serum glutamic pyruvic transferase; SGOT, glutamic oxaloacetic transferase; BUN, blood urea nitrogen; CHOL, cholesterol; GLU, glucose; TBIL, total bilirubin; TP, total protein; TRIG, triglyceride.
a Mean ± standard error (N = 10).
b
Male rats in all dose groups (27, 52, and 115 mg/kg/d) showed decreased body weight gains (Table 8). Male rats at dose 27 mg/kg/d are the lowest effective dose in decreasing body weight gain when compared to controls. Higher organ-to-body weight ratios in liver, kidney, spleen, and adrenal in males were observed in high dose. In females, the organ weight-to-body weight ratios increased in kidney, liver, brain, spleen, and ovaries in the higher dose groups when compared to controls. The organ-to-brain weight ratios were increased in liver at the 2 high doses in males. Testis-to-brain weight ratios and spleen to brain weight ratios decreased at the high dose in males. In females, organ weight-to-brain weight ratios increased in liver (2 high doses) and increased in adrenals at the high dose 16 (data not shown).
All animals survived to the end of the 90-day study. At necropsy, the high-dose males and females had yellow-stained pelts, pale livers, and minimal body fat. Histopathological examination of tissues showed that the liver contained cytoplasmic vacuolization of hepatocytes at all dose levels in both sexes and in control rats. This cytoplasmic vacuolization of hepatocytes is more pronounced in males and somewhat pronounced in females in treated rats. The severity of the hepatocellular vacuolization as particularly is associated with the dose of 4A2NT. Male rats at 52 and 115 mg/kg/d dose groups showed moderate to high incidence of testicular hypospermatogenesis. The lesions consisted of reduction in spermatogenesis in large number of seminiferous tubules that were associated with the administration of test material.
Determination of BMD and BMDL10
The decrease in body weight gain by male rats was the most sensitive adverse event observed in this study. The dose–response relationships of the decreased body weight gain of male and female (adjusted to show absolute difference from control animals) are shown in Figure 2. The Power model gave the best fit of data with a smallest scaled residual of interest. The BMD and BMDL10 were, respectively, 23.1 and 15.5 mg/kg/d for males and 48.3 and 29.7 mg/kg/d for females.
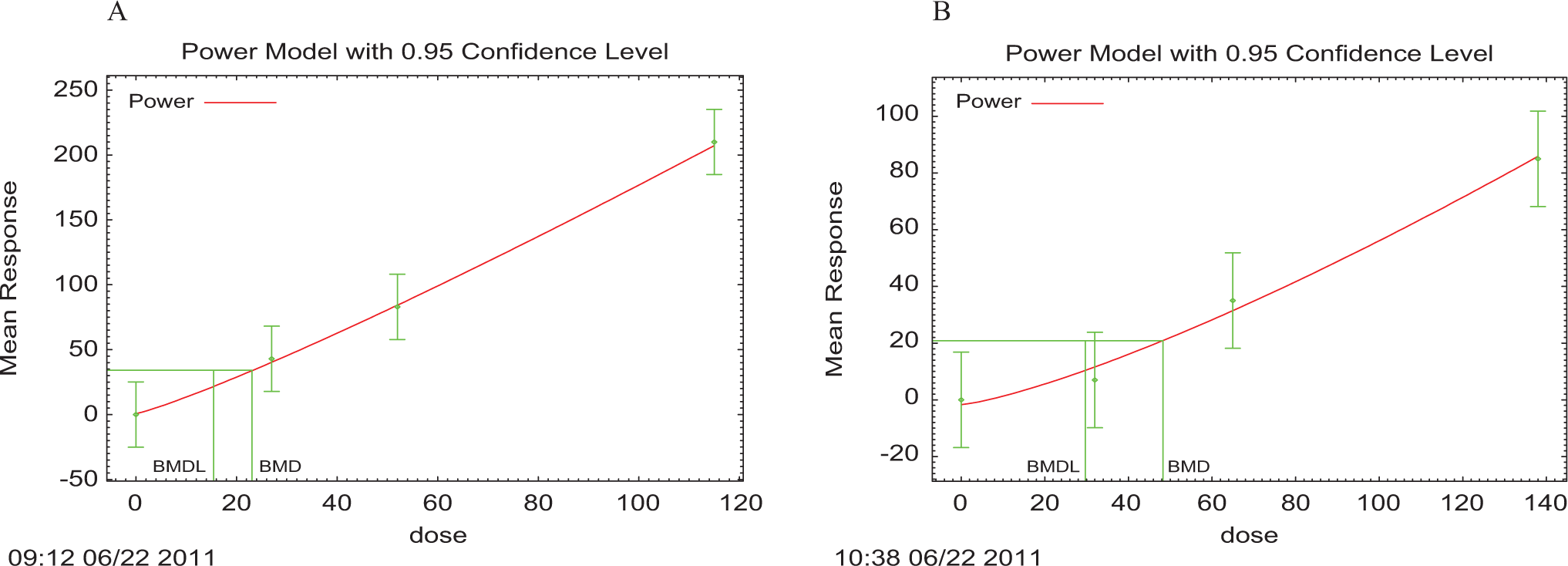
The experimental data and the BMDS output with 95% confidence interval for the power model of continuous data for the relationship between ingested dose (mg/kg/d) and the absolute difference in decreased body weight gain for each dose group compared to control (weight loss of 0) for males (A) and females (B). BMDS, Benchmark Dose Software.
Derivation of the HED
A BMDL10 of 15.5 mg/kg/d was calculated for the observed decrease in body weight gain of male rats. For the calculation of the HED,14,15 the recommended DAF for oral dosimetry is used, that is, the rat/human body weight ratio raised to the 1/4 power (BW(a)/BW(h))0.25, which in the case of rats is 0.24 (DAF = (0.25 kg/70 kg)0.25 = 0.24). The HED was computed as HED = BMDL10 × DAF; HED = 15.5 mg/kg/d × 0.24 = 3.8 mg/kg/d.
Selection of Uncertainty Factors and Derivation of the RfD
In order to calculate the RfD, composite uncertainty factors were selected. Because the HED was calculated using a DAF, UFA was set equal to 3.14,15 For human uncertainty factor, the default value of 10 was used for the unknown variation in sensitivity among the members of the human population. A factor of 10 was assigned for the lack of chronic data and an additional UF of 10 for data gaps. Thus, the RfD was calculated as the HED divided by a composite uncertainty factor of 30 (3 × 10 × 10 × 10 = 3000); RfD = 3.8 mg/kg/d/3000 = 1.26 μg/kg/d.
Discussion
There is considerable toxicity information on 2,4-DNT and 2,6-DNT, as these compounds are used as intermediates in the manufacture of polyurethanes, dyes, munitions, and military applications. It has been shown that these compounds degrade through oxidative or reductive pathways. The reductive pathway results in the formation of metabolites such as 4A2NT, 2A4NT, and 2,4-diaminotoluenes(2,4-DAT), azoxytoluene isomers, and 4-acetamido-2-nitrotoluene.1,3,17 These degradation products were detected at the production waste disposal sites and in ground water. 4 There are no toxicity data on 4A2NT to develop safety standards to assess risk related to the exposure to human and ecosystems. The acute ALD of 4A2NT was determined in PEG 200 vehicle to be about 5000 mg/kg for both sexes of rats which is similar to the reported oral LD50 of parent compound 2,4-DNT in rats. 18
The results of the 14-day feeding study with 4A2NT showed decreased body weight gain at the highest dose (2000 ppm); this high dose was selected as the highest dose group for the subsequent 90-day feeding study. The daily food consumption of rats on diet containing 4A2NT at dose levels of 0, 125, 250, 500, 100, and 200 ppm showed a significant reduction in food consumption at 2 high-dose groups of males and at 2000 ppm in females. This reduced food consumption, and toxic effect of 4A2NT resulted in reduced body weight gain in both sexes at high dose. At this high dose, certain hematological parameters such as HGB, HCT, and RBC counts decreased, suggestive of anemia in female rats. Body weight-to-brain and body weight-to-testis weights showed an increase when compared to controls. This may be due to a decrease in the body weights at high dose. The subacute toxicity studies of parent compound, 2,4-DNT, in rats results also showed decreased body weight gain as low as 34 mg/kg/d in male and 38 mg/kg/d in female rats. 19 The degradation product, 4A2NT, produced toxic effects in body weight gains and in certain hematological parameters in our present study.
The results of the subchronic study (90 days) in rats, when exposed to 4A2NT in the diet at the consumed dose levels 0, 27, 52, or 115 mg/kg/d for male and 0, 32, 65, or 138 mg/kg/d for female rats showed significant decrease in body weight at lowest group 27 mg/kg/d group in males and in two high-dose groups in females. This may be due to rats’ reduced food consumption of feed containing 4A2NT and toxic effect of test chemical (Tables 6 and 7). The other reductive metabolite, 2A4NT, in subchronic toxicity study also showed decreased body weight gain in diet up to 169 mg/kg/d for male and female rats and 128.7 for male and 136.6 mg/kg/d for female mice 5
The subchronic study hematological results showed decreases in HGB, HCT, and RBC levels in male and female rats, suggestive of anemia that were also reported with parent compounds 2,4-DNT in rats and mice of subchronic toxicity19,20 and other nitroaromatic compounds such as 2,4,6-trinitrotoluene (TNT)21,22 and 1,3,5-trinitrobenzene (TNB).23,24 There were no biologically meaningful differences in the hematological parameters studied. The changes in clinical chemistry values may be associated with alteration in hepatocellular changes in rats exposed to at high dose. The 4A2NT produced a positive response in Ames
4-Amino-2-nitrotoluene showed hepatic effects in both sexes and testicular effects in male rats, similar to other nitroaromatic compounds such as TNT and TNB21–24 and meta-dinitrobenzene. 27 The testicular effects observed in male rats may be a direct action of the chemical on the testis as seen with other nitroaromatic compounds and may be the effect of food restriction.28,29 It showed hepatic effects (cytoplasmic vacuolization) in male and female rats. The changes consisted of indistinctly bound cytoplasmic vacuoles, morphologically similar to postprandial glycogen and fat accumulation. In some rats, the vacuolization was more prominent in the periportal region, while in others the change was distributed throughout the liver. Presence of the change in control group suggests the vacuolization was due to in part postprandial accumulation of glycogen and fat, but increased severity in rats that received test chemical suggests compound-related accentuation of the change (Dr George A Parker Pathologist, Hillsborough, North Carolina). 16 Male rats dosed at two high doses (52 and 115 mg/kg/d) showed testicular effects (atrophy, hypospermia, and mild dilation of seminiferous tubules) in males. These effects are similar to parent compound 2,4-DNT.19,20 Recent subacute studies of 2,4-, 2,6-, and 3,5-DNT isomers also showed these hematological, hepatic, and testicular effects in rats. 30
Based upon decease in body weight gain in male rats, the determined BMDL10 was 15.5 mg/kg, and the oral human RfD was determined to be 1.26 μg/kg/d.
Footnotes
Acknowledgements
We thank Mr James Daniel for support, Dr William Eck for critical review of manuscript, and Ms Allison Jackovitz for the help in the preparation of tables.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Installation Management Command/Army Environmental Command, Fort Sam Houston, Texas .
