Abstract
Topical alprostadil cream (Vitaros) is approved in Canada and Europe for the treatment of erectile dysfunction. To determine the effects on the female urogenital tract with repeated administration of the entire dose (300 μg alprostadil containing 2.5% dodecyl-2-n,n-dimethylaminopropionate hydrochloride), the vaginal pH, flora, and histology were assessed as a model for 100% transference from male to the female during unprotected sexual intercourse. Female cynomolgus monkeys were administered the entire dose of Vitaros for 14 days with a 7-day recovery. Relative to vehicle and placebo cream, the vaginal pH and microflora were determined at baseline and weekly, thereafter. Vaginal biopsies were evaluated at the end of dosing and recovery. All animals were clinically normal for the study duration, and the vaginal pH was consistent between dose groups and the dosing period. Vaginal microflora and histopathology findings of mild inflammation were generally similar across treatment groups. In conclusion, repeated vaginal exposure to Vitaros did not alter the pH, microflora, or histology after 14 daily doses, supporting the safety of Vitaros transference to the female partner.
Keywords
Introduction
Vitaros is a single-use topical cream approved for the treatment of erectile dysfunction (ED) in Canadian and European territories. 1 The active ingredient in Vitaros is alprostadil (300 μg), the pharmaceutical equivalent of the endogenous prostaglandin E1 (PGE1). Prostaglandin E1 is the product of the fatty acid cyclooxygenase pathway of arachidonic acid metabolism and naturally occurs in semen. 2 Human seminal fluid is a rich source of prostaglandins, including PGE1 and PGE2, and the total concentration of prostaglandins in ejaculate has been estimated to be approximately 100 to 200 μg/mL. 3 Because of the prostaglandin content in semen, exposure to the active ingredient of Vitaros is a low risk to female sexual partners.
Prostaglandin E1 has a wide variety of pharmacological effects including vasodilatation, inhibition of platelet aggregation, inhibition of gastric secretions, and stimulation of intestinal and uterine smooth muscle. 4,5 In vitro, alprostadil (PGE1) has been shown to cause dose-dependent smooth muscle relaxation in isolated corpus cavernosum and corpus spongiosum preparation. 6 The vasodilatory mechanism of action has resulted in a number of approved pharmaceutical products that contain PGE1. For example, approved products containing alprostadil (PGE1) as their active ingredient have been used to treat male sexual dysfunction, cardiac disorders in adults and neonates, urologic disorders, and peripheral vascular and arterial diseases. 7 -9
Vitaros is topically delivered to the tip of the penis (meatus) for the treatment of ED in men. To allow delivery of alprostadil to the corpus cavernosum, Vitaros contains the penetration enhancer, dodecyl-2-n, n-dimethylaminopropionate hydrochloride (DDAIP-HCl). The clinical development of Vitaros for the treatment of ED included two phase 3 studies exposing >1700 men and their female sexual partners to the topical alprostadil cream with significant efficacy. 10 In men, the most common treatment-related adverse events (AE) were genital pain, penile erythema, and penile burning. In women, the most commonly reported adverse event was vaginal burning. The adverse event reports were generally mild to moderate that resolved within 2 hours. 10 Longer term efficacy and safety of Vitaros were examined in an open-label phase 3 clinical trial. With continued treatment, the AEs reported for local urogenital effects decreased in patients and female partners. 11
Exposure of the vaginal epithelium to the entire dose of Vitaros that models 100% transference of the Vitaros dose from the male to the female has not been previously studied relative to the effects on vaginal pH, microflora, and epithelial proliferation. Utilizing a cynomolgus monkey model, females were vaginally administered the entire dose of Vitaros (300 μg alprostadil containing 2.5% DDAIP-HCl) for 14 days followed by a 7-day nondosing period to evaluate the impact on the vaginal pH, microflora, and histology.
Materials and Methods
Chemicals
Vitaros containing 0.33% alprostadil and the novel excipient and penetration enhancer, 2.5% w/w DDAIP-HCl cream, was manufactured according to current Good Manufacturing Practice. Analytical results confirmed that alprostadil content was within the specifications. Control creams, one with 2.5% w/w DDAIP-HCl and the other without DDAIP-HCl, were used for comparison to Vitaros. Analytical results confirmed the presence or absence of DDAIP-HCl at the nominal level, respectively. Depo-Provera (medroxyprogesterone acetate; Pharmacia & Upjohn) and Dexdomitor (dexmedetomidine; Janssen Animal Health) were available commercially. Additional chemicals were either US Pharmacopeia or analytical grade available commercially.
Formulations
The vehicle was a potassium phosphate (0.1 mol/L)-buffered cream containing 2.5% guar gum, pH 6.6. The placebo was the vehicle cream with additional 2.5% DDAIP-HCl. Vitaros was used as supplied (0.1 mg cream contained in a unit-dose dispenser). Prepared formulations were stored refrigerated (2°C-8°C) for the study duration.
Animals
Twelve non-naive female cynomolgus monkeys (nonhuman primates [NHPs]) were used on this study (3 groups, n = 4/group), approximately 5 years old and ranging from 2.7 to 4.0 kg. Menstrual cycles were synchronized in all primates with Depo-Provera (medroxyprogesterone acetate, 30 mg, intramuscularly [IM]) administered 14 days prior to dose administration to minimize the influence of menstrual cycling. A second injection of Depo-Provera was administered 7 days after the initiation of vaginal dosing (day 7).
For dose administration, the animals were properly restrained, and the vaginal region of each animal was wiped with a dilute (1:1) saline chlorohexidine gluconate solution from vagina to anus, followed by a clean, moist (saline) gauze wiped in the same direction to avoid contamination of the vagina with fecal material. Using sterile gloves, the vagina was manually opened and the cream dispensed into the vagina. The vehicle cream and 2.5% DDAIP-HCl cream were administered using a 1.0-mL syringe inserted approximately 2 cm into the vagina. The group administered Vitaros (alprostadil cream 330 μg + 2.5% DDAIP-HCl Cream) received the entire contents of each dispenser per administration, (approximately 0.1 mL). Animals were dosed once per day for 14 days followed by a 7-day recovery. Dosing syringes and dispensers were weighed before and after dosing to confirm the amount administered.
Animals were monitored for mortality, body weight, and clinical observations throughout the dosing period. Unanesthetized animals were appropriately restrained, and vaginal pH was measured on study days −2 and −1 and prior to dose administration on days 1, 7, 14, 17, and 21. A pH electrode was carefully inserted approximately 1 cm into the vagina and the pH recorded in triplicate. At predose and on days 1, 7, 14, 17, and 21, a vaginal swab was collected from each animal to assess microbial growth. The animals were manually restrained and a sterile swab was inserted into the vagina approximately 2 cm (after cleaning per dose-administration instructions) and the vaginal walls wiped in a circular motion. The swabs were cultured in suitable conditions to detect the presence of Lactobacillus sp, Staphylococcus aureus, Yeast sp, coliforms, Streptococcus sp, and anaerobes.
After dosing phase completion on day 14 and after the 7-day recovery on day 21, vaginal biopsies (∼2 × 2 mm2) were collected. Each NHP was anesthetized with ketamine (IM, 10 mg/kg). Anesthesia was maintained with dexmedetomidine (0.02 mg/kg, IM) with heart rate, respiratory rate, and pulse oximetry continuously monitored.
Under aseptic conditions, the biopsy area was cleaned with chlorohexadine and 70% alcohol. Using a sterile biopsy punch, a ∼2 × 2 mm2 full-thickness vaginal biopsy was collected from approximately 2 cm inside of the vagina and placed into 10% formalin. Tissues were fixed in formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Microscopic examination of paraffin sections were performed on the vaginal biopsy samples from all animals. Once the biopsy was collected, animals were administered atipamezole (0.1 mL IM) to reverse the sedative effects of dexmedetomidine. All procedures were reviewed by the BioTox Sciences Institutional Animal and Use Committee.
Results
All animals were observed to be clinically normal for the duration of the study, with the exception of transient bleeding associated with pH measurements and soft stool and diarrhea that was occasionally observed. Menses was observed in 2 primates from groups 2 and 3 on day 14. There were no test article-related effects on body weight. The vaginal pH did not vary significantly between dose groups or over the dosing period (Figure 1). Lactobacillus sp was the most common organism found on the vaginal swabs for all groups, and the levels were generally similar during the dosing period with a slight increase during the recovery period (Figure 2A). Staphylococcus aureus was generally at low levels during the dosing period and increased during the recovery period (Figure 2B). In contrast, the coliforms tended to increase in all groups throughout the dosing period and decreased during recovery (Figure 2C). Vaginal swabs did not reveal the presence of Streptococcus sp, Yeast sp, or anaerobes for any animal throughout the dosing or recovery period. Histologically, all animals had mild lymphoplasmacytic infiltrates, with occasional intraepithelial lymphocytes at the end of the dosing and recovery periods (Figure 3). Acute hemorrhaging was observed at most biopsy sites and attributed to the collection procedure (Figure 3).
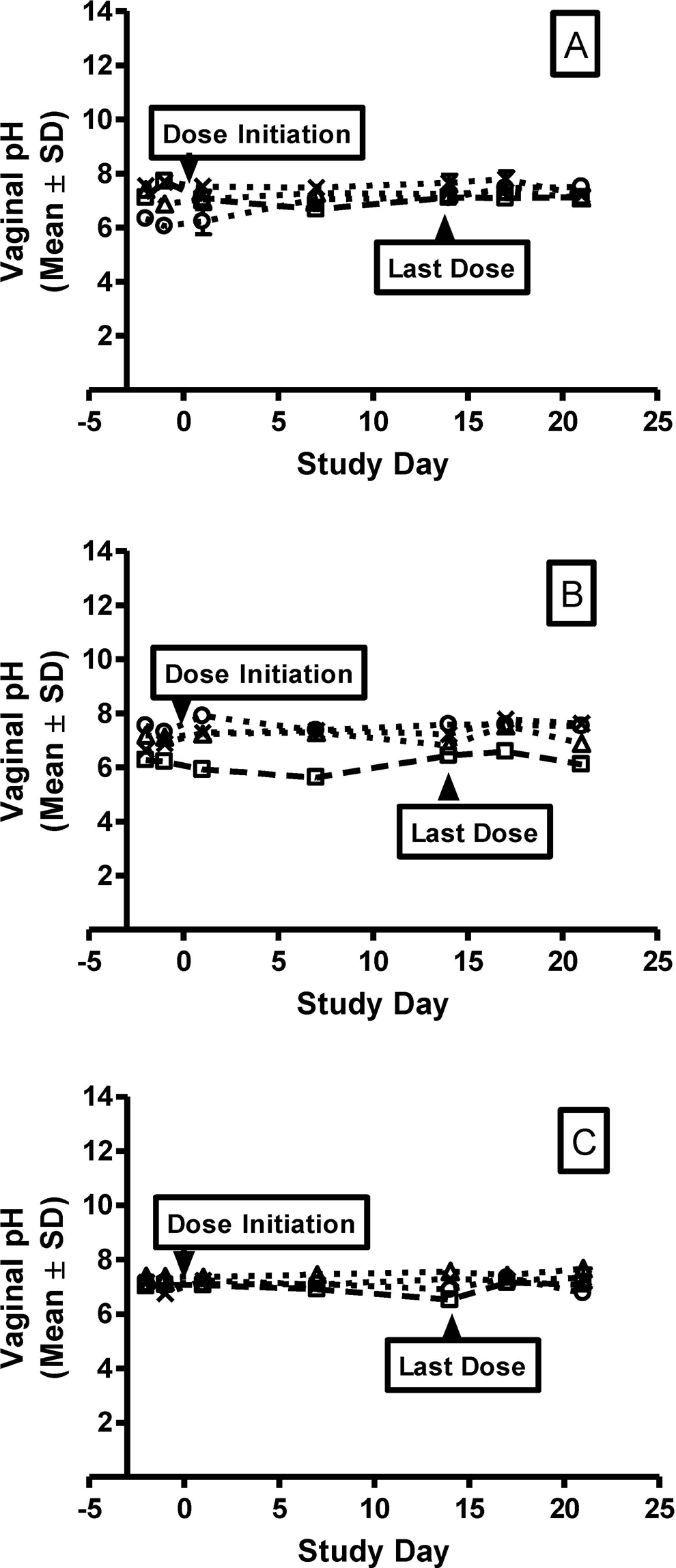
Individual vaginal pH. At each timepoint, 3 readings were collected from each animal. The individual pH measurements are plotted and values represent mean ± standard deviation for the triplicate measurements taken for (A) vehicle cream, (B) 2.5% DDAIP-HCl cream, and (C) Vitaros (alprostadil/DDAIP-HCl cream, 330 μg/2.5%). DDAIP-HCl indicates dodecyl-2-n,n-dimethylaminopropionate hydrochloride.
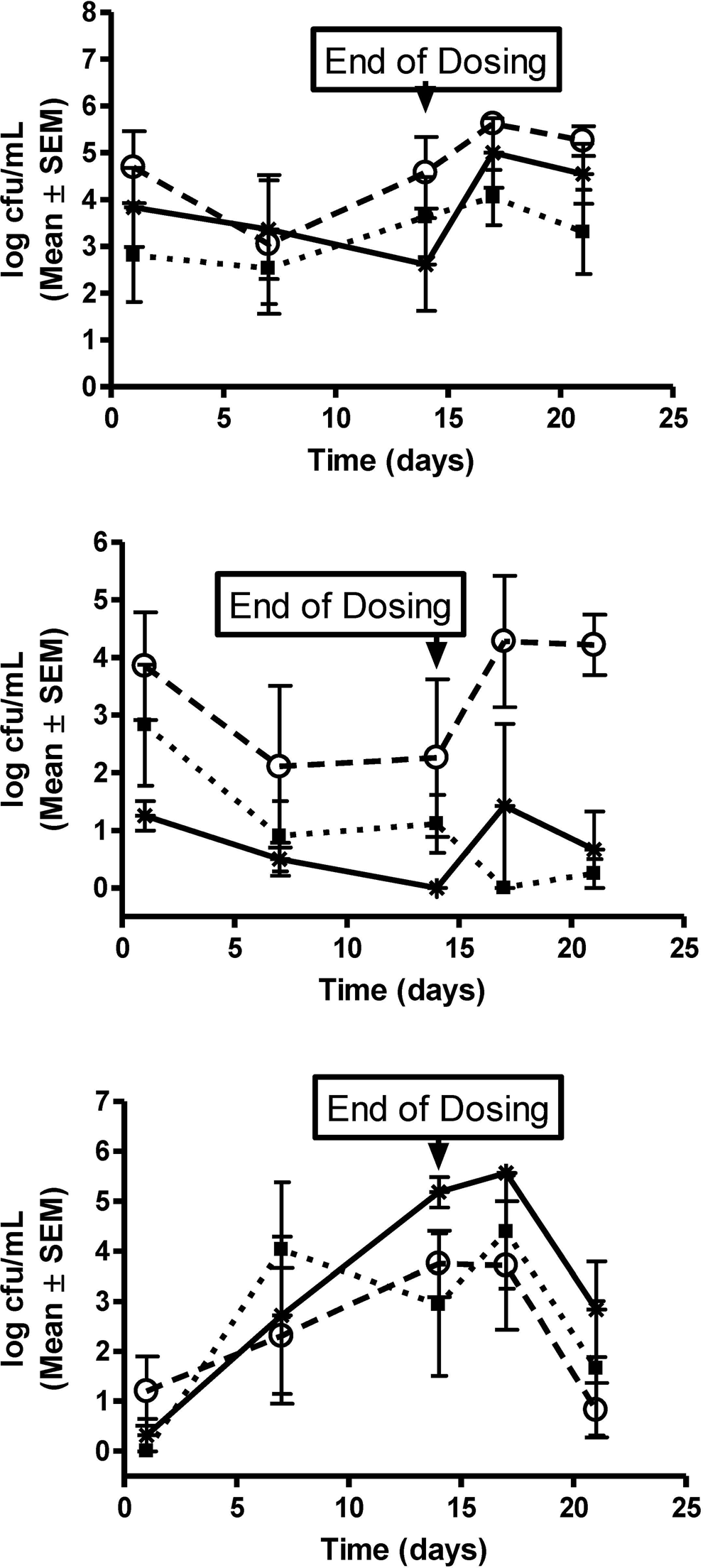
Microbial colonization. Microorganisms detected from the vaginal swabs included (A) Lactobacillus sp, (B) Staphylococcus aureus, and (C) coliforms. Values represent mean ± standard error of the mean of log10 colony forming units (log cfu)/mL for each group (n = 4); vehicle cream (▪), 2.5% DDAIP-HCl cream (^), and Vitaros (*; alprostadil/DDAIP-HCl cream, 330 μg/2.5%) over the 21-day study duration. DDAIP-HCl indicates dodecyl-2-n,n-dimethylaminopropionate hydrochloride.
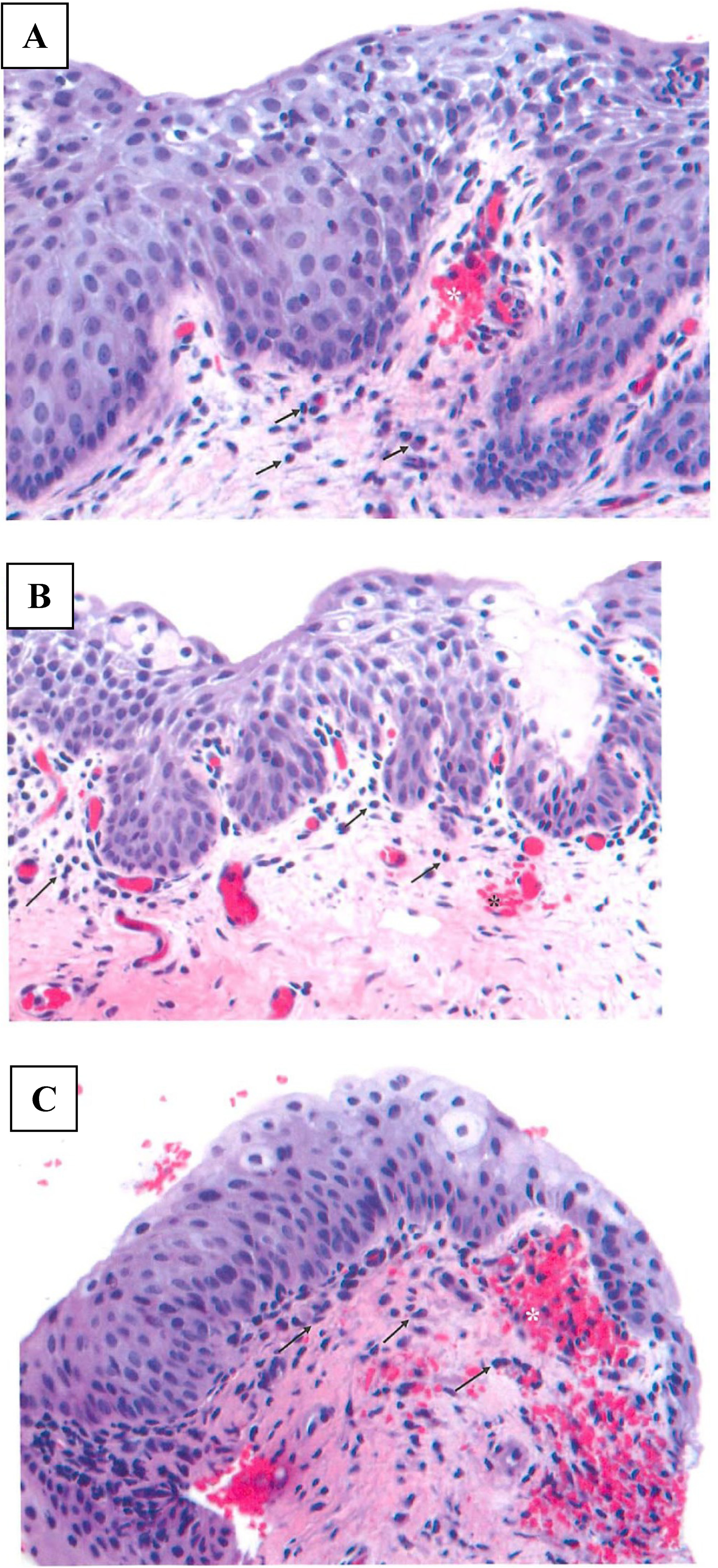
Representative photomicrographs (40× magnification). Vaginal biopsies collected after the 14th dose of (A) vehicle cream, (B) 2.5% DDAIP-HCl cream, and C) Vitaros (alprostadil/DDAIP-HCl cream, 330 μg/2.5%). Arrows delineate superficial mucosal infiltration of lymphocytes and plasma cells and the asterisk marks the superficial acute hemorrhage. DDAIP-HCl indicates dodecyl-2-n,n-dimethylaminopropionate hydrochloride.
Discussion
Normal vaginal microflora consists of Lactobacillus sp and gram-positive cocci. 12 -14 Colonization with gram-negative bacteria is associated with urinary tract infections. 12 -14 The animals in this study also showed a predominance of Lactobacillus sp that was relatively consistent with treatment. With repeated manipulation associated with vaginal administration, the gram-negative colonization increased throughout the dosing period. The colonization was consistent across all groups demonstrating that coliform growth was not likely due to the treatment but manipulation associated with dosing. Although some differences were observed between groups, the initial S aureus counts also varied but a comparison between predose and postdose levels were similar. Not observed in these animals were anaerobes that may be associated with normal flora. After a 7-day recovery, the detectable microflora, that is, Lactobacillus, coliforms, and S aureus, were comparable to pretreatment levels.
Normal vaginal pH in premenopausal women is approximately 4.5 and increases to 6 to 7.5 in postmenopausal women. 15 When vaginosis is present in premenopausal women, the pH increases to a range of 5 to 6.5. 15 Reference pH values were established with multiple readings prior to the first vaginal dosing in female cynomolgus monkeys. The animals in this study had a pretreatment pH ranging from 6 to 7, a level above that seen with premenopausal women but consistent with postmenopausal vaginal pH. Although the vaginal pH from sexually mature cynomolgus monkeys was similar to that observed in premenopausal women with vaginosis, the pH remained consistent throughout the dosing and recovery periods with no evidence of associated microbial proliferation.
Vitaros application for the treatment of ED may expose the female sexual partner during sexual intercourse. As a model for 100% transference to the sexual partner, the entire dose (100 μL) was administered to female monkeys. Based on the results of this study, daily 14-day vaginal dose administration of DDAIP-HCl (2.5%) or alprostadil cream (330 μg + 2.5% DDAIP-HCl) for this dose regimen (14 days) to female cynomolgus monkeys had no meaningful effect on the clinical observations, vaginal pH, microflora, and vaginal histology of female cynomolgus NHPs in comparison to placebo cream. After a 7-day recovery period, the findings were similar to those found at the end of the dosing period.
The components of the alprostadil topical cream formulation (Vitaros) administered in 100 μL do not alter the vaginal pH, microflora, or histology after 14 daily doses. The data from the repeated application of Vitaros support safety for potential vaginal exposure of female sexual partners to the topical ED product containing alprostadil and the permeation enhancer, DDAIP-HCl.
Footnotes
Authors’ Note
Primary Laboratory of Origin: Apricus Biosciences, Inc.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research presented in this article was funded solely by Apricus Biosciences, Inc.
