Abstract
Tin(IV) oxide functions as an abrasive, bulking, and opacifying agent in cosmetic products and is used at concentrations up to 0.4% in rinse-off products and up to 1.3% in leave-on products. The Cosmetic Ingredient Review Expert Panel (Panel) noted that tin(IV) oxide is a water-insoluble inorganic metal compound and should not be percutaneously absorbed; therefore, systemic exposure is not likely. Studies of dermal application of tin(IV) oxide were considered to determine toxicity at the site of application. The Panel concluded that tin(IV) oxide is safe in the present practices of use and concentration.
Introduction
As given in the International Cosmetic Ingredient Dictionary and Handbook, tin(IV) oxide functions as an abrasive, bulking, and opacifying agent in cosmetic products. 1 The Roman numeral IV connotes that tin in this molecular structure is in the +4 oxidation state.
Chemistry
Definition and Structure
Tin(IV) oxide (CAS Nos. 1332-29-2 and 18282-10-5), dioxide of tin, is an inorganic oxide that conforms to the following structure:
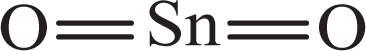
Other names for this chemical include stannic oxide, white tin oxide, tin dioxide, stannic anhydride, and flowers of tin. 1,2
Physical and Chemical Properties
Tin is a silver-white metal that is malleable and somewhat ductile. It has a highly crystalline structure and exists in 2 allotropic forms at normal pressures. Gray tin exists below 13.2°C and has a cubic structure. At 13.2°C, gray tin is converted to white tin, which has a tetragonal structure. 3,4 The white form is better known as the common, stable form at room temperature. 5 In compounds, tin can exist in the +2 or +4 oxidation state 3,4 : divalent and tetravalent oxidation states are designated as stannous and stannic, respectively. The Stock Oxidation-Number system denotes the oxidation state using Roman numerals in parentheses following the metal’s name: tin(II) and tin(IV). 6 The cosmetic ingredient, tin oxide, is tin(IV) oxide. Chemical and physical properties of tin(IV) oxide are found in Table 1. Notably, tin(IV) oxide is not soluble in water.
Chemical and Physical Properties of Tin(IV) Oxide. 32

Abbreviation: NA, not available.
Method of Manufacture
The Earth's crust contains approximately 2 to 3 ppm tin, comprising 0.0006% of the Earth's crust. 2,7 The most important tin-containing mineral is cassiterite, also known as SnO2. Other tin minerals are stannite, teallite, cylindrite, and canfieldite. After tin-containing ores are mined, they undergo further separation processing, resulting in concentrates containing 70% to 77% tin by weight, almost pure cassiterite, and are ready for smelting. 8 Elemental tin is obtained from cassiterite by reduction with coal in a reverberatory furnace. 5 Although tin(IV) oxide occurs naturally in mineral form, this is not the source of the commercial product. It is manufactured directly from tin metal by thermal oxidation, either by exposing molten tin to air in a furnace at elevated temperatures or by blowing tin powder in a stream of air through a furnace at approximately 700°C. 9
According to one source, 9 the commercial production of tin(IV) oxides yielded the following grades: average particle size of 0.3 µm (bulk density = 0.72 g/cm3), average particle size of 0.4 µm (bulk density = 1.15 g/cm3), and average particle size of 0.5 µm (bulk density = 1.35 g/cm3). Each grade is >99.0% pure and has a specific gravity of 6.9.
Impurities
As stated previously, tin(IV) oxide, as manufactured from tin metal, is >99.0% pure. 9
Use
Cosmetic
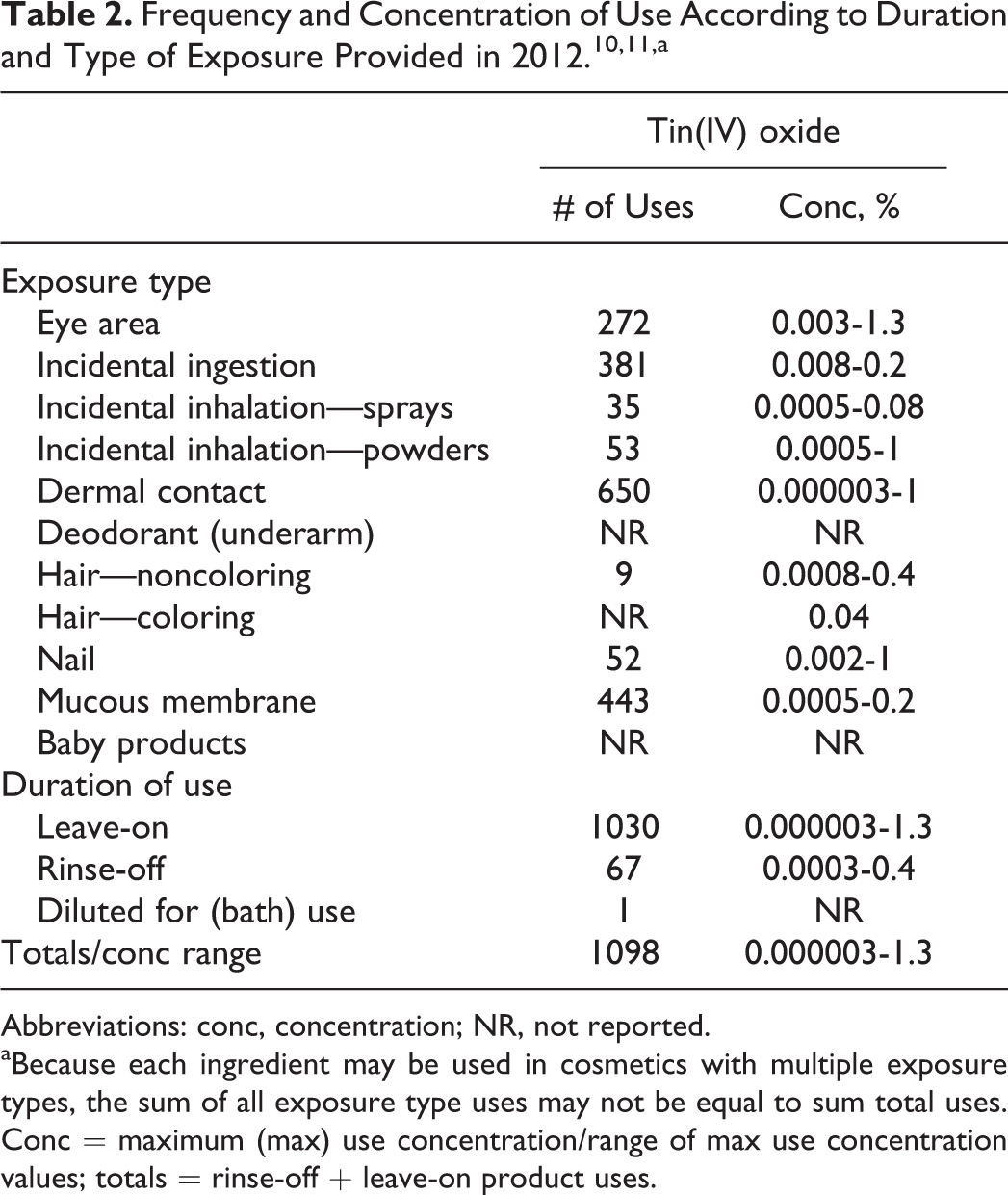
As given in the International Cosmetic Ingredient Dictionary and Handbook, tin(IV) oxide functions as an abrasive, bulking, and opacifying agent in cosmetic products. 1 According to information reported by industry to the Food and Drug Administration (FDA) as part of the FDA’s Voluntary Cosmetic Registration Program (VCRP; Table 2), tin(IV) oxide was used in 1098 cosmetic products in 2012. 10 Results from a survey of ingredient use concentrations provided by the Personal Care Products Council (also included in Table 2) in 2012 indicate that tin(IV) oxide was reported as being used at concentrations up to 0.4% in rinse-off products (maximum concentration in noncoloring shampoos) and at concentrations up to 1.3% in leave-on products (maximum concentration in eye shadow). 11
Abbreviations: conc, concentration; NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not be equal to sum total uses. Conc = maximum (max) use concentration/range of max use concentration values; totals = rinse-off + leave-on product uses.
Cosmetic products containing tin(IV) oxide may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Tin(IV) oxide is reported as being used in dusting powders (up to 0.03%), body and hand cosmetic sprays (up to 0.06%), and other fragrance preparations (up to 0.08%) and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 12 -15 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 12,13
Noncosmetic
Tin(IV) oxide is used in a variety of manufacturing applications, including polishing glass and other metals. 2 Elemental tin is present mainly in solder alloys used in the electronics industry and is also used as a protective coating for other metals, especially those used for food containers. 16
According to a memorandum on food contact substance notification (FCN) No. 000431, the intended use of tin(IV) oxide is, in combination with silicon dioxide and titanium dioxide, as a colorant for food-contact polymers, paper and paperboard, coatings, and in printing inks applied to nonfood-contact surfaces of food-contact articles. 17 The food contact substance will be used at a level not to exceed 6% of the total colorant weight. The finished food-contact articles may contact all food types with no temperature limitation.
Tin(IV) oxide is not regulated in 21 CFR but is the subject of threshold of regulation exemption 98-004, for the use of tin(IV) oxide at a maximum level of 1.1% by weight in colorants otherwise composed of mica and titanium dioxide, provided that the maximum loading rate for the colorant in the food-contact material does not exceed 3% by weight for polymers, 5% for paper and paperboard, 15% for coatings, or 30% for ink formulations. Tin(IV) oxide is a constituent of the food contact surface that is the subject of FCN 235 (use of tin antimony oxide [also known as antimony gray cassiterite] as a pigment for all polyolefins for food contact applications). 17
Toxicokinetics
Neither data on the absorption, distribution, metabolism, and excretion of tin(IV) oxide nor data on skin penetration were found in the published literature and unpublished data were not provided.
Toxicology
Acute Oral Toxicity
An acute oral median lethal dose (LD50) of >20 g/kg has been reported for tin(IV) oxide in mice and rats (strains and ages not stated). 18
Acute Intraperitoneal Toxicity
Following intraperitoneal (ip) dosing of rats and mice (numbers and strains not stated) with tin(IV) oxide, an acute LD50 of > 6.6 g/kg was reported. 18
Repeated Dose Toxicity
Groups of 10 males and 10 females were fed diets containing 0%, 0.03%, 0.10%, 0.30%, or 1.0% tin(IV) oxide for 28 days. 19 End points monitored included mortality, growth, food consumption and utilization, hematology, urinalysis, serum biochemistries, and gross and microscopic pathology. No compound-related adverse effects were observed among rats fed tin(IV) oxide during the 4-week feeding period.
Ocular Irritation
The ocular irritation potential of an eye shadow containing 0.3% tin(IV) oxide was evaluated using 34 female patients (18-65 years old), 3 of whom withdrew for reasons unrelated to conduct of the study. 20 The participants were instructed to use the test material at least once daily for 4 weeks. A comprehensive ocular examination was performed at the end of the 4-week period. There were no adverse events, and all ophthalmologic examinations remained within normal limits. Study results did not indicate a potential for ocular irritation or hypersensitivity.
In an in vitro study, the ocular irritation potential of an eye shadow containing 1.11% tin(IV) oxide was evaluated using the chorioallantoic membrane (CAM) vascular assay (CAMVA-14 day) and the bovine corneal opacity and permeability (BCOP) test. 21 In the CAMVA-14 day assay, 2 groups of 10 White Leghorn eggs were dosed (40 µL or 40 mg) with the eye shadow at concentrations of 100% and 50% (effective concentration ≈ 0.6%), respectively. The 14-day incubation period was followed by an additional 30 ± 5 minutes of incubation. The CAM was then observed for signs of vascular hemorrhage, capillary injection, or ghost vessels (all positive responses indicating ocular irritation potential), and the RC50 was determined. The RC50 is defined as the concentration at which 50% of the treated eggs show a positive response. Positive responses were not observed in treated eggs (RC50 > 100%).
The BCOP test is used to evaluate the potential ocular corrosivity or severe irritancy of a test substance, as measured by its ability to induce opacity and increased permeability in an isolated bovine cornea. 22 Toxic effects on the cornea are measured by decreased light transmission (opacity) and increased passage of sodium fluorescein dye (permeability). Following exposure to a test substance, the opacity and permeability measurements are combined to derive an in vitro irritancy score, used to classify the level of irritancy. In the BCOP assay, 5 corneas were dosed with 0.75 mL of a 20% (effective concentration ≈ 0.2%) solution of the eye shadow in minimal essential media. Opacity measurements and sodium fluorescein permeability were determined. The corrected mean opacity score was 0.6, and the corrected mean optical density (permeability) was −0.003. The in vitro score for the eye shadow, as calculated, was 0.56 (0.6 + 15 (−0.003) = 0.56). 21
Skin Irritation and Sensitization
The skin irritation and sensitization potential of a powder eye shadow containing 0.3% tin(IV) oxide was evaluated in a repeated insult patch test (RIPT) using 111 male and female patients (18-75 years old), 98 of whom completed the study. 23 Withdrawal from the study was not related to application of the test material. A 1 × 1 sq in semiocclusive patch containing 0.2 g of the test material was applied to the upper back (between the scapulae) of each patient 3 times per week for a total of nine 24-hour induction applications. After a 2-week nontreatment period, challenge patches were applied for 24 hours to a new test site. Reactions were scored at 24 and 72 hours postapplication. No reactions were observed, and it was concluded that the test material did not have skin irritation or allergic contact sensitization potential.
The skin irritation and sensitization potential of a lipstick containing 0.5% tin(IV) oxide was also evaluated in an RIPT (similar procedure) using 112 male and female patients (16-79 years old), 103 of whom completed the study. Withdrawal from the study was not related to application of the test material. No reactions were observed, and it was concluded that the test material did not have the potential for causing dermal irritation or allergic contact sensitization. 24 In another study, the skin irritation and sensitization potential of a lipgloss product containing 0.35% tin(IV) oxide was evaluated in an RIPT (amount per patch not stated) using 112 male and female patients (18-70 years old), 108 of whom completed the study. 25 The test protocol was identical to that used in the preceding test, with the exception that challenge sites were evaluated at 24, 48, and 72 hours postapplication. No reactions were observed, and it was concluded that the test material did not demonstrate a clinically significant potential for eliciting dermal irritation or sensitization.
Except for use of a 3/4 × 3/4 sq in semiocclusive patch and the evaluation of challenge reactions at 24 and 48 hours only, the same RIPT procedure in the preceding study was used to evaluate the primary or cumulative irritation and/or allergic contact sensitization potential of an eye shadow containing 1.3% tin(IV) oxide. 26 The initial study group consisted of 232 patients (16-79 years old), 209 of whom completed the study. Withdrawal from the study was not related to product application. It was concluded that test results for the eye shadow did not indicate a potential for dermal irritation or allergic contact sensitization.
Occupational Studies/Case Reports
Two-hundred fifteen workers (ages not stated) were exposed to tin(IV) oxide fumes at a plant, 95% of whom had at least 3 years of service. 27 Of the 215 workers who received chest x-rays, 121 had changes identified as pneumoconiosis. None of the x-ray films was suggestive of massive fibrosis or significant emphysema, and there was no evidence of massive fibrosis or nodulation. There were no differences in the following between the 121 pneumoconiotic workers and 94 nonpneumoconiotic workers: respiratory symptoms, vital capacity, chest expansion, loss time due to chest illness, and incidence of tuberculosis.
A clinical study of 19 male employees (most <30 years old) exposed to tin(IV) oxide dust and fumes at a plant was performed. 28 Impairment of pulmonary function was not observed in any of the patients, and there were no reports of work disability from any clinical cause. Physical examinations did not reveal any abnormal lung findings or significant findings in general. All of the values for vital capacity, maximal breathing capacity, resting minute volume, and respiratory reserve were within normal limits. The absence of alteration in these ventilatory tests indicate that there was no significant degree of obstructive emphysema or of diffuse pulmonary fibrosis. Based on the methods used, it was noted that the only type of pulmonary function alteration that could have escaped detection would have been impaired diffusion of the alveolo-capillary block type, which is found in cases of asbestosis and berylliosis.
Based on chest roentgenograms, 1 patient was classified as completely normal, 8 were classified as stannosis suspects, and 10 were classified as having tin(IV) oxide pneumoconiosis. It was noted that patients with less than 3 years of exposure may be classified as either normal or suspects but did not present with pulmonary nodulation. After 3 years of exposure to tin(IV) oxide, nodular stenosis was found in all cases, and advanced stages occurred with increasing frequency as the years of exposure increased. Of the 10 employees with roentgenographic changes classified as stannosis, 6 had been exposed to tin(IV) oxide fume. The most advanced changes were observed in 4 of these 6 employees. Of the 4 employees, 1 had likely been exposed exclusively to tin(IV) oxide dust and had first-stage stannosis, and the remaining 3 employees (exposure to dust and fumes) had varying degrees of change. Of the 10 employees with lung changes, 6 were asymptomatic and the following signs were reported for the remaining 4: moderate anorexia (2 employees), cough with serious expectoration (1 employee), and scapular pain (1 employee). For the 10 cases of stannosis, the hemograms and sedimentation rates were within normal limits. Traces of albumin in the urine were reported for 3 of the cases, and the blood Kahn reaction was normal in all cases. The authors noted that the results of this study corroborate the conclusion that tin(IV) oxide fume, and not tin(IV) oxide dust, is more likely to be the cause of stannosis. 28
Stannosis is the form of pneumoconiosis (nonfibrotic form) that results from the inhalation of tin in the form of tin(IV) oxide fumes or dust. Tin(IV) oxide accumulates in the pulmonary parenchyma. Lung radiography results for a man (age not stated) who had worked in the smelter of a tin mine for 26 years revealed moderately profuse small nodules, some of which were metallic in density. 29 The patient was asymptomatic and clinically normal. Lung function tests were not performed. Results, 8 years later, revealed an increase in the profusion of small opacities, particularly in the left mid-zone of the lung. The patient remained asymptomatic. Another case report involved a 55-year-old male employee of a detinning plant for 15 years. He was exposed to tin(IV) oxide fumes as well as clouds of coal dust on the job, and lung function test results yielded a forced vital capacity of 90% and a forced expiratory volume that was 96% of predicted values. Lung radiography results revealed very profuse bilateral nodules (∼3 mm in diameter). At lung biopsy, focal aggregations of macrophages containing dust particles (black particles) were observed in some of the air spaces and in the perivascular and peribronchiolar connective tissue. Electron probe analysis results indicated that tin was present in the dust. 29
A 50-year-old female (nonsmoker) with stannosis was exposed to tin(IV) oxide fumes for 33 years. 30 There were also exposures to biomass fuels and asbestos. A chest x-ray revealed common nolular lesions, and thorax high-resolution computed tomography revealed widespread interlobular thickening and peribronchial thickening. Subpleural nodules with metallic density were observed in the upper and middle lobe of the right lung. Bronchial lavage cytology was defined as class II, and histiocytic cells and focal fibrosis were detected on transbronchial lung biopsy. The patient died 6 months later due to respiratory failure.
Reproductive and Developmental Toxicity
Data on the reproductive and developmental toxicity of tin(IV) oxide were not found in the published literature and unpublished data were not provided.
Genotoxicity
Tin(IV) oxide was administered (doses not stated in Chinese publication summary) to rats through the trachea, and cytological preparations were made at various intervals in order to determine effects on micronucleus frequency and karyorrhexis of rat bone marrow cells and lung macrophages. 31 Results indicated that tin(IV) oxide can induce micronuclei and karyorrhexis in bone marrow cells. On the 1st and 10th day, the frequency of karyorrhexis was higher than that of micronuclei and differed significantly from that of the control, and vice versa, on the 20th and 30th days. Tin(IV) oxide can also induce micronuclei and karyorrhexis in lung macrophages. On the 10th and 20th days, the frequency of karyorrhexis was the same as that in bone marrow cells.
Carcinogenicity
Data on the carcinogenicity of tin(IV) oxide were not found in the published literature and unpublished data were not provided.
Summary
The safety of tin(IV) oxide (dioxide of tin) in cosmetics is reviewed in this report. Elemental tin is obtained from cassiterite by reduction with coal in a reverberatory furnace. Tin(IV) oxide is manufactured directly from tin metal by thermal oxidation. According to information supplied to the FDA by industry as part of the VCRP in 2012, tin(IV) oxide was being used in 1098 cosmetic products. Furthermore, results from a survey of ingredient use concentrations provided by the Personal Care Products Council in 2012 indicate that tin(IV) oxide was being used at concentrations up to 0.4% in rinse-off products (max concentration in noncoloring shampoos) and up to 1.3% in leave-on products (max concentration in eye shadow).
An acute oral LD50 of >20 g/kg has been reported for tin(IV) oxide in mice and rats. Following ip dosing of rats and mice with tin(IV) oxide, an acute LD50 of >0.6600E + 4 mg/kg was reported. No test substance-related adverse effects were observed in rats fed tin(IV) oxide at concentrations up to 1.0% in the diet for 28 days.
An eye shadow containing 0.3% tin(IV) oxide did not cause ocular irritation in 31 patients who participated in a 4-week product use study (daily applications). In vitro assay results relating to the ocular irritation potential of an eye shadow containing 1.11% tin(IV) oxide were negative when the product was diluted to concentrations of 0.6% and 0.2% prior to testing. In RIPTs, neither a lipstick (0.5% tin(IV) oxide, 103 patients), lipgloss product (0.35% tin(IV) oxide, 108 patients), powder eye shadow (0.3% tin(IV) oxide, 98 patients), nor another eye shadow (1.3% tin(IV) oxide, 209 patients) induced skin irritation or allergic contact sensitization.
In occupational settings, stannosis, a form of pneumoconiosis, has been observed in workers exposed to tin(IV) oxide fumes. Tin(IV) oxide administered intratracheally (dose not stated) induced micronuclei and karyorrhexis in rat bone marrow cells in vivo. Data on the toxicokinetics, carcinogenicity, or reproductive and developmental toxicity of tin(IV) oxide were not found in the published literature and unpublished data were not provided.
Discussion
The Panel noted that tin(IV) oxide is a water-insoluble inorganic metal compound and should not be percutaneously absorbed; therefore, systemic exposure was not a concern. This would explain the absence of both published and unpublished studies regarding toxicokinetics, reproductive and developmental toxicity, and carcinogenicity. Based on the results of dermal application studies on product formulations containing tin(IV) oxide, there is no indication that this ingredient would have any irritation or sensitization potential at use concentrations up to 1.3% in cosmetic products.
Because tin(IV) oxide was reported to be used in products that may be sprayed, the Panel discussed the issue of incidental inhalation exposure. In the absence of typical inhalation toxicity studies, the Panel considered other pertinent data that were available (ie, occupational exposure data). Stannosis, a form of pneumoconiosis, has resulted from occupational exposure to tin(IV) oxide dust/fumes, but the Panel agreed that this level of exposure to tin(IV) oxide would not result from cosmetic use. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, however, along with the highly insoluble nature of tin(IV) oxide, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings. Acute or repeated dose toxicity following exposure to tin(IV) oxide via other routes was not a concern, based on relatively high oral and ip LD50 values reported for rats and mice and negative results (rats) in a 28-day oral feeding study.
Conclusion
The CIR Expert Panel concluded that tin(IV) oxide is safe in the present practices of use and concentration described in this safety assessment.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
W. Johnson contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. B. Heldreth contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. L. Gill, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, analysis and interpretation, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy,
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.