Abstract
Arsenic compounds can increase production of reactive oxygen species. Reactive oxygen species can induce double-strand breaks in DNA, which is a cause of chromosome aberrations (CAs). This study was conducted to determine the association between arsenic exposure and polymorphisms of genes involved in detoxification (glutathione S-transferase T1 [
Introduction
Arsenic is a toxic metalloid widely distributed in food, water, air, and soil. 1 Genotoxic effects of arsenic have been reported in vitro in mammalian cells and in vivo in laboratory animals and humans. 2 Studies have shown that the frequency of chromosomal aberration indices varies among individuals after exposure to arsenic. 3,4 Additionally, individuals exposed to arsenic exhibit different responses in either in vivo 5,6 or in vitro studies. 7 Treatment with arsenic compounds can increase production of reactive oxygen species (ROS) 8 and induce oxidative stress. 9,10 Reactive oxygen species can induce double-strand breaks of DNA, 11 which is a cause of chromosomal aberrations. 12 -14
The enzymes encoded by antioxidant genes such as glutathione S-transferase M1 (
The human
Nonhomologous end joining (NHEJ) is the prominent DNA repair system in humans. The NHEJ pathway includes several essential repair proteins, including x-ray repair complementing defective repair in Chinese hamster cells 4 (
Taken together, we hypothesize that polymorphisms in genes involved in the detoxification and the NHEJ DNA repair pathways might be involved in induction of chromosomal aberrations due to arsenic exposure.
Material and Method
The present study consisted of 123 healthy males between the ages of 19 and 47 years (mean ± standard deviation: 30.38 ± 6.24). The participants had no history of smoking or drinking alcohol, were not on medication, and had no history of diagnosed cancers, schizophrenia, bipolar disorder, and epilepsy or neuropathy. Written informed consent was obtained from each volunteer, and the study was approved by the institutional review board at our university.
Peripheral blood samples were obtained by venipuncture and collected in 2 separate tubes, one of them containing sodium heparin and the other one containing EDTA. Heparinated blood samples were used for primary culture, and the blood samples with EDTA were stored at −20°C for genotyping.
For primary cultures, triplicate 400 µL whole blood samples were inoculated into 5 mL of RPMI-1640 medium which contained 15% fetal bovin serum and 100 µL phytohamagglutinin (GIBCO, USA, Cat. No. 10576-015) in sterile (volume 10 mL) screw cap tubes. After 24 hours of incubation in a humid incubator 37°C and 5% CO2, the first tube was inoculated with sodium arsenite (NaAsO2; iAs(III); at final concentration 1 µmol/L), the second tube with mitomycin C (MMC; at final concentration 60 ηg/mL), and the third tube with no additives. After 72 hours of culture (48 hours of treatment), tubes were treated with colchicine (0.05 µg/mL; Fluka, USA) for 1 hour. Cells were then harvested by centrifugation, treated with a hypotonic solution (0.075 mol/L KCl) for 30 minutes at 37°C, and fixed 3 times in cold Carnoy fixative (3 vol of methanol:1 vol of acetic acid) for 10 minutes. Cells were resuspended in fresh fixative and dropped onto glass slides, and chromosomes were stained with Giemsa as standard cytogenetic procedures.
To avoid bias in the assessment of the slides, slides were scored by an individual who was blind to genotype and treatment conditions. To determine mitotic index (MI), number of metaphase figures were calculated in 1000 blast cells for each culture and expressed as a percentage. For each culture, 100 metaphases were randomly scored for chromatid breaks (CBs), dicentric, triradial, and quadraradial chromosomes as chromosome aberrations (CAs). Thereafter, total percentage of aberrant cells was calculated.
Genomic DNA for polymerase chain reaction was isolated from whole blood using the thawed blood samples.
35
The genotypic analysis for the
A chi-square (χ2) test was performed to determine whether distributions of the genotypes of the study polymorphisms were in Hardy-Weinberg equilibrium. Effect of iAs(III) on cytogenetic variation was analyzed using paired samples
Results
The genotypic frequencies of the polymorphisms of A424G
Cytogenetic effects of iAs(III) and MMC in comparison with each other and with negative control are indicated in Table 1. The comparisons include MI, CB, CA, and percentage of aberrant cells.
Cytogenetic Changes After Treatment With iAs(III) and MMC Compared to Negative Control.
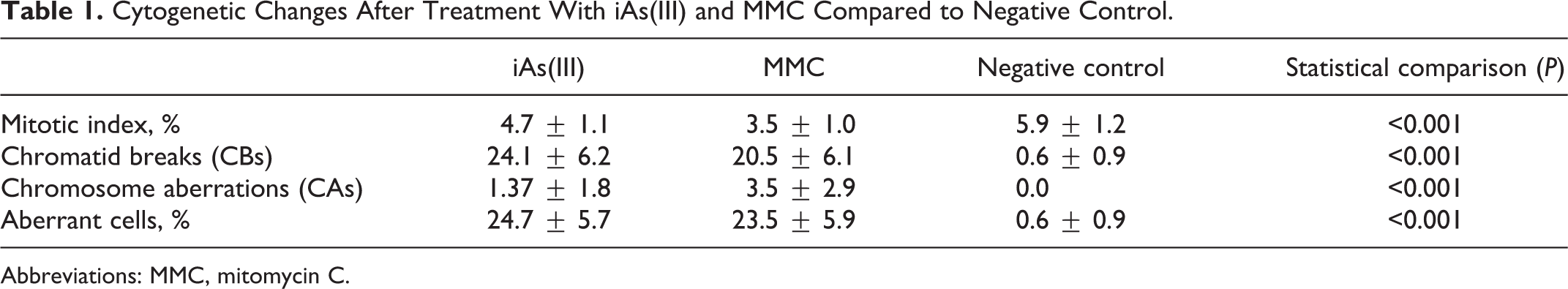
Abbreviations: MMC, mitomycin C.
In MMC treatment, significant differences for MI, CB, and CA were observed when compared with the negative control. In the case of iAs(III) treatment, MI significantly decreased in comparison with the negative control. However, the level of CB, CA, and percentage of aberrant cells significantly increased in iAs(III) treatment compared with the negative control.
Considering the
Cytogenetic Effects of iAs(III) With Respect to
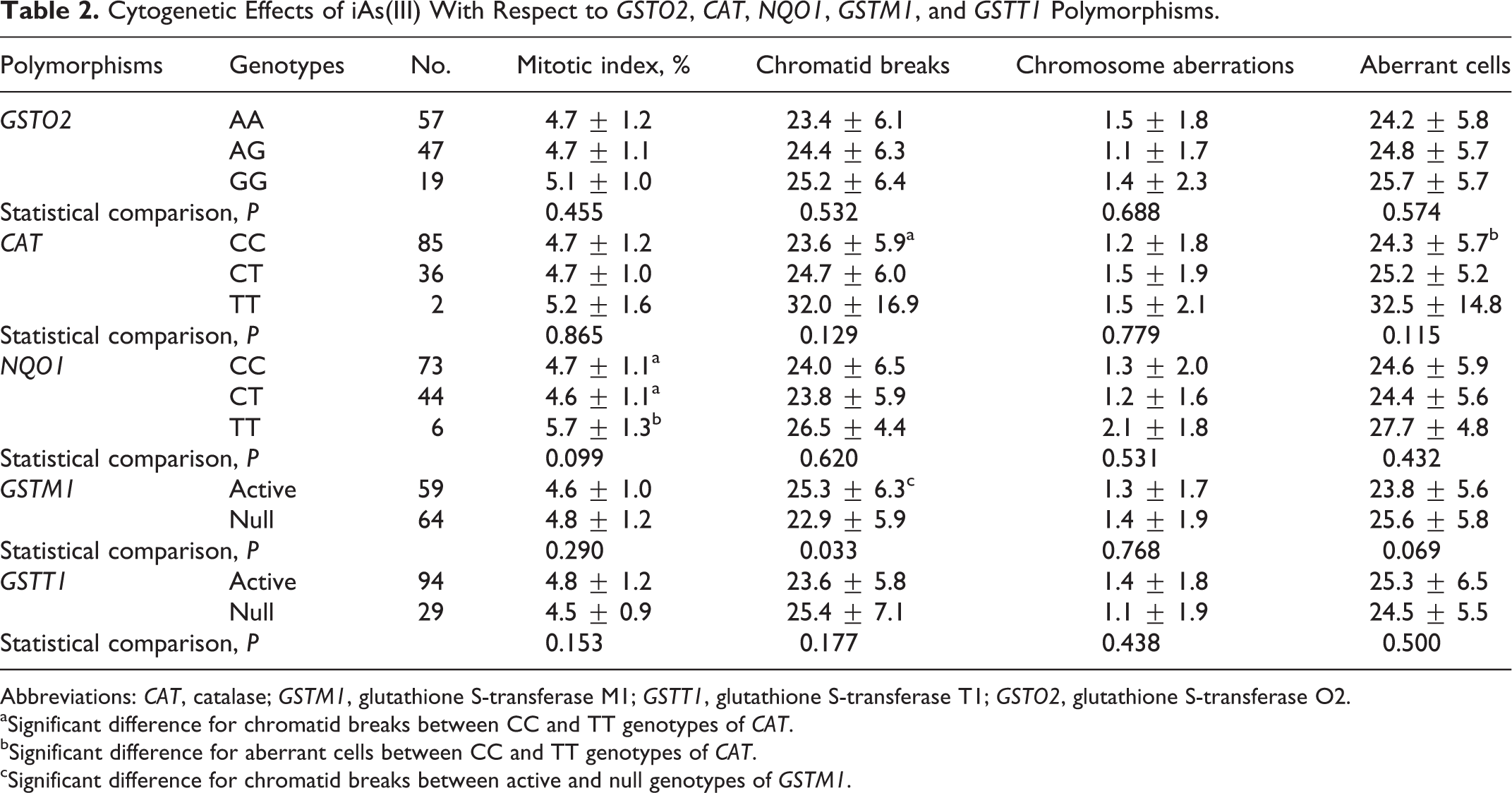
Abbreviations:
aSignificant difference for chromatid breaks between CC and TT genotypes of
bSignificant difference for aberrant cells between CC and TT genotypes of
cSignificant difference for chromatid breaks between active and null genotypes of
Considering
Cytogenetic Effects of iAs(III) With Respect to
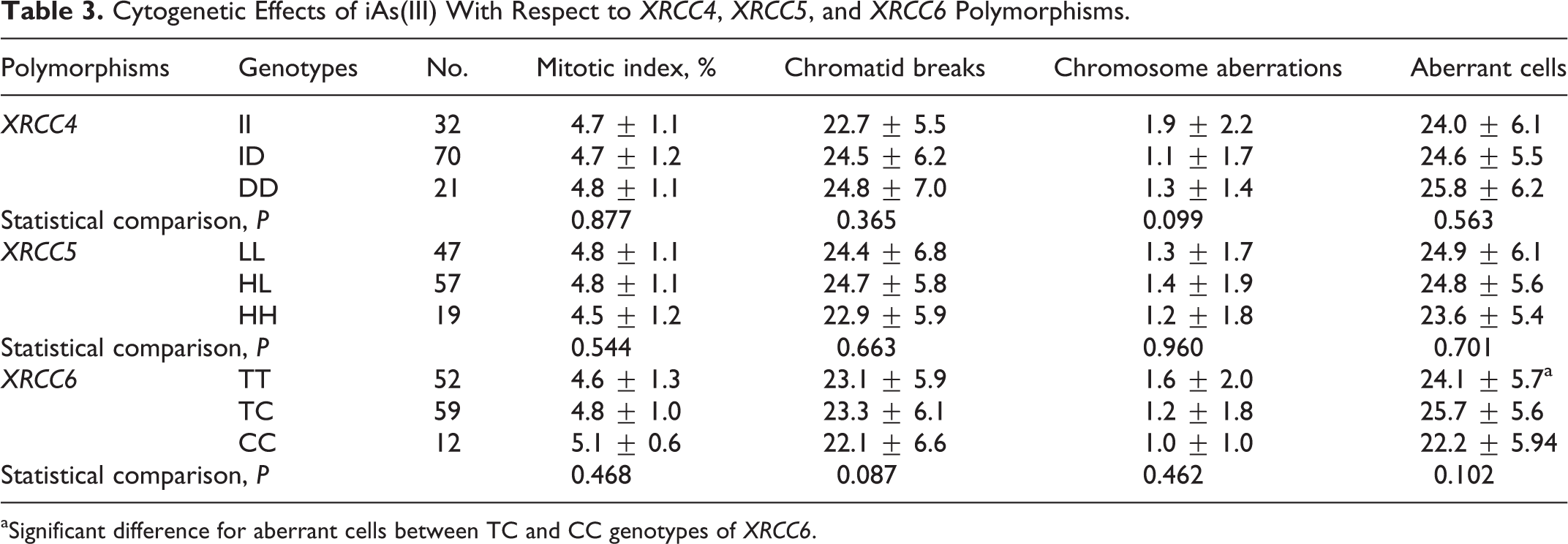
aSignificant difference for aberrant cells between TC and CC genotypes of
Discussion
Cytogenetic assessments revealed that the MI and CB values in samples treated with iAs(III) are significantly higher than those in MMC treatment. This is consistent with previous reports. 39,40
Arsenic biotransformation in persons having the A allele of the
Catalase activity in patients with the TT genotype of C-262T polymorphism is reported to be significantly reduced.
36
In our study, we found that the TT genotype of
It has been reported that CAs induced by air pollution are associated with the
It should be noted that relatively small sample size and potential false-positive findings as a result of multiple comparisons are main limitations of the present study. In summary, the present study showed that the polymorphisms of
Footnotes
Acknowledgments
The authors are indebted to the participants for their close cooperation.
Author Contributions
Fatemeh Azizian-Farsani and Gholamreza Rafiei contributed to design, acquisition, and analysis, drafted the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Mostafa Saadat contributed to conception and design, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Fatemeh Azizian-Farsani and Gholamreza Rafiei have equal contributions in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shiraz University.
