Abstract
Bis(2-ethylhexyl)-2,3,4,5-tetrabromophthalate (TBPH) is one of the new brominated flame retardants with adverse neurobehavioral potential. These flame retardants are often added to household furnishings where children would come into contact with them. This study explores whether oral exposure to TBPH for 28 days would impair neurobehavioral function in mice and the role of curcumin (CUR) in this process. CUR is a natural antioxidant and is thought to be of use in the treatment of neurological toxicity due to its neuroprotective effects. Learning and memory of mice exposed to TBPH was investigated using the Morris water maze. Levels of malondialdehyde (MDA), superoxide dismutase (SOD), and glutathione (GSH) were determined to assess oxidative damage. Western blot was used to detect the expression of glucose-regulated protein 78-kDa (GRP78), PKR-like ER kinase (PERK), and C/EBP homologous protein (CHOP) in the hippocampus. End-point effects were evaluated through observing post-synaptic density protein-95 (PSD-95), brain-derived neurotrophic factor (BDNF), and phosphorylated cAMP response element binding protein (p-CREB). Although TBPH exposure alone does not impair learning and memory, oxidative stress markers and endoplasmic reticulum stress–associated proteins were adversely affected in exposed mice. TBPH could significantly decrease the levels of BDNF, p-CREB, and PSD-95 in the hippocampus, and these TBPH-induced neurotoxic effects were attenuated by CUR. These findings provide further understanding of the neurotoxic effects of TBPH and the protective effect of CUR on TBPH exposure.
Keywords
Highlights
•TBPH exposure alone for 28 days does not impair learning and memory in mice. •Neurotoxic effects of TBPH may be related to OS and ERS. •TBPH reduces the levels of BDNF, p-CREB, and PSD-95 in the hippocampus. •CUR may have a protective effect against the adverse effects caused by TBPH exposure.
Introduction
Flame retardants are widely used in electronic equipment, construction materials, and furniture to prevent fires and to slow their progression. 1 Over the past few years, new brominated flame retardants (NBFRs) have largely replaced the traditional flame retardants.2,3 Bis(2-ethylhexyl)-3,4,5,6-tetrabromophthalate (TBPH), as a representative of NBFRs, is often added to household furnishings where children would come into contact with them and is either used alone or mixed with 2-ethylhexyl-2,3,4,5-tetrabromobenzoate (TBB) to replace the banned polybrominated diphenyl ethers (PBDEs). 4 TBPH’s common commercial products include FireMaster® 550, FireMaster® BZ-54, and DP-45TM.4,5 FireMaster® 550 is the most important commercial product containing TBPH, in which the proportion of TBPH is about 15%. It is mainly added to plastic polyurethane foam and used in the production of furniture, building materials, and other commodities. FireMaster ®BZ-54 consists of TBB and TBPH, of which TBPH is 30%. 6 The proportion of TBPH in DP-45TM is about 91%, a product mainly added to the insulation of wires and cables. 4 Given the current state of waste disposal in the world, there is enormous potential for TBPH to be released into the environment.
Global environmental surveys show that TBPH has been commonly detected in a variety of abiotic media such as the atmosphere, open water, soil, sediments, indoor dust, and municipal sewage.7,8 From 2008 to 2009, the detected levels of NBFRs in serum (n = 102) and breast milk (n = 105) samples from Canadian lactating women showed that the detection rates of TBPH were 16.7% and 32.4%, respectively. However, the detection level of TBPH is highest among all the five examined NBFRs, the highest level measured was 68 ng/g in serum and 24 ng/g in breast milk. 9 Subsequently, the detection results of NBFRs for 60 groups of mothers and infants from 2009 to 2010 showed that the concentration of TBPH in common food was lower than the detection limit, while indoor dust was the main way for mothers and infants to be exposed to TBPH, and the median daily intake of indoor dust (3.3 ng/d) by infants was more than twice that of mothers (1.6 ng/d). 10 Although there is limited literature on the correlation of TBPH exposure with neurobehavioral manifestations in human beings, the possible neurotoxic correlation between environmental exposure of TBPH especially in related industries with neurobehavioral changes should be paid more attention. 7
More and more evidence from studies using animal models such as fish and rodents reveal the neurotoxic effects of TBB and TBPH, with adverse effects on neurobehavioral and reproductive development. 11 Gillera et al. 12 found that FireMaster® 550 could induce memory deficits in prairie voles. Other studies demonstrated elevated anxiety and decreased social interaction in exposed male Wistar rats. 13 We undertook this study due to the relative lack of evidence of the neurotoxic effects of TBPH and the mechanisms that may cause these neurobehavioral changes.
Curcumin (CUR), a well-known natural compound, has been used in a diverse array of pharmacological activities. 14 Recently, extensive research has been carried out to study the application of CUR as a health improving agent and used as a functional ingredient in foods. 15 Yadav et al. 16 reported that CUR protects arsenic-induced neurobehavioral toxicity by modulating oxidative stress and dopaminergic function in rats. It may be a significant concern whether CUR can be of use in treatment of neuronal damage caused by TBPH exposure.
In this work, we investigated the learning and memory abilities of mice exposed to TBPH using the Morris water maze (MWM). Levels of malondialdehyde (MDA), superoxide dismutase (SOD), and glutathione (GSH) were determined to assess oxidative damage. Western blot was used to detect the expression of glucose-regulated protein 78-kDa (GRP78), PKR-like ER kinase (PERK), and C/EBP homologous protein (CHOP) in the hippocampus because these proteins may be related to adverse effects caused by TBPH. 17 Indicators of end-point effects were evaluated through observing post-synaptic density protein-95 (PSD-95), brain-derived neurotrophic factor (BDNF), and phosphorylated cAMP response element binding protein (p-CREB). Additionally, we administered CUR as an antagonist to reduce the neuronal damage caused by TBPH exposure. This study aimed to explore whether oral exposure to TBPH for 28 days would impair the learning and memory of the tested mice and the role of CUR in this process.
Materials and methods
Animals and care
Five-week-old specific pathogen-free (SPF) male Balb/c mice (about 22 g) were provided by Hubei Experimental Animal Research Center. All mice were housed under SPF conditions at 20–25°C with 50–70% humidity and a 12 h light/dark cycle. A commercial diet and filtered water were provided ad libitum throughout the experiment. All experimental protocols were approved by the Office of Scientific Research Management (IAEC-2021-01-019).
Chemicals and kits
TBPH (CAS: 26040-51-7) and CUR (CAS: 458–37-7) were purchased from AccuStandard® Inc (New Haven, USA) and Sigma-Aldrich (St Louis, MO, USA), respectively. A mouse ELISA kit for PSD-95 was obtained from Enzyme-linked Biotechnology Co., Ltd (Shanghai, China). ELISA kits for BDNF and p-CREB were purchased from BlueGene (Shanghai, China). MDA and GSH colorimetric kits as well as SOD assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The rabbit anti-mouse GRP78 (ab108613) and GAPDH (ab37168) antibodies were purchased from Abcam (Cambridge, MA, USA). The rabbit anti-mouse PERK (#3192) and CHOP (#2895) antibodies were purchased from Cell Signaling Technology (Danvers, Massachusetts, USA). HRP-goat anti-rabbit (AS1107) and HRP-goat anti-mouse (AS1106) were purchased from Bioss (Beijing, China). All other chemicals used in the study were of the highest grade available commercially, unless specifically noted.
Experimental protocol
Forty Balb/c mice were randomly divided into five groups (8 mice each): control group (saline); 20 mg·kg−1·d−1 TBPH exposure group; 200 mg·kg−1·d−1 TBPH exposure group; 25 mg·kg−1·d−1 CUR group; and 200 mg·kg−1·d−1 TBPH +25 mg·kg−1·d−1 CUR group (TBPH + CUR). The daily dose of TBPH (200 mg·kg−1) was chosen based on the potential metabolism and toxicity of TBPH described by Springer et al. 4 The dosage of CUR (25 mg·kg−1) was selected as the optimal dosage according to Sunagawa et al. 18 In the TBPH + CUR group, CUR was administered intragastrically, followed by administration of TBPH 2 h later. The dosage volume for all operations was 10 mL·kg−1. Experimental mice received daily treatments for 28 days.
Morris water maze
The MWM test, described in a previous study, 19 was conducted to evaluate the acquisition and maintenance of spatial memory of the exposed mice. The maze consists of a black circular pool (diameter 2.14 m, height 80 cm) with a 9.5 cm diameter platform and is filled with water at room temperature (25°C) to a height of 50 cm. The positioning navigation MWM experiment was carried out in the third week after exposure. The mice continued to be treated with drugs during the behavioral test. Learning and memory ability was evaluated by testing how long it took the mice to locate the platform (latency). The positioning navigation experiment lasted for 5 days, following which the mice rested for 1 day, and the space exploration experiment was carried out on the 7th day to evaluate the memory retention ability of the mice by their ability to locate the position of the original platform. The same quadrant was selected as the entry point, and over a 60 s period, the latency time, the mouse trajectory, and number of times the mouse swam over the platform were recorded by the Ethevision XT Version 12.0 (Noldus Ltd, Netherlands) monitoring and analysis software.
Sampling and testing
Following 28 days of exposure, the mice animals were euthanized cervical dislocation while still under deep anesthesia and the brains excised. The collected hippocampal tissues of the brain were rinsed with cold phosphate buffer (PBS, pH=7.5) and made into a 10% homogenate by adding PBS; this was then centrifuged at 9391 g for 10 min and the supernatant was preserved at 4°C. Levels of MDA, SOD, and GSH were then measured. MDA levels in the hippocampal supernatant were determined by thiobarbituric acid (TBARS) assay and were calculated as follows: MDA (μmol·g−1 prot) = [6.45(OD532−OD600)−0.56×OD600] × sample dilution factor/homogenate protein concentration. 20 The GSH concentration in the hippocampal supernatant was measured using a colorimetric kit and calculated as follows: GSH (μmol·g−1 prot) = [(measure OD405 - blank OD405)/(standard OD405 - blank OD405)] × standard concentration × sample dilution factor/homogenate protein concentration. 20 SOD level was measured using kits according to the manufacturer’s instructions with an ELx800 absorbance microplate reader (Bio-Tek Instruments, Inc., USA).
Analysis of BDNF, p-CREB, and PSD-95
These biomarkers are used to evaluate the effects on related proteins involved in memory in the hippocampus of mice exposed to TBPH. The levels of BDNF, p-CREB, and PSD-95 in the hippocampus were measured using commercial ELISA kits, according to the manufacturer’s instructions. The sensitivities of the ELISA kits were 1.0 pg/mL for BDNF, 0.1 ng/mL for p-CREB, and 0.1 ng/mL for PSD-95. The concentrations were measured in duplicate for each sample.
Western blot
The expression of GRP78, PERK, and CHOP protein in the hippocampus from mice in each group was detected by western blot. After the frozen mouse, hippocampal tissue was lysed with RIPA lysate, the total protein was extracted, and the protein concentration in the sample was determined by BCA. The protein was separated by 10 μL 10% SDS-PAGE gel electrophoresis and then transferred to a 0.45 μm polyvinylidene fluoride (PVDF) membrane. An anti-blocking buffer was added and the solution shaken at room temperature for 1 h. Then GAPDH (5% skim milk, 1: 10,000), GRP78 (5% skim milk, 1:2000), PERK (5% BAS, 1:1000), and CHOP (5% BAS, 1:1000) antibodies were added and sealed overnight at 4°C. The membrane was washed and incubated with sheep anti-rabbit second antibody at room temperature for 1 h. The gray values and gray ratios of GAPDH, GRP78, PERK, and CHOP bands for each sample were obtained.
Statistical analysis
The obtained experimental data were expressed as mean ± standard error of the mean (SEM) and analyzed using GraphPad Prism 8.0 (San Diego, CA, USA). The differences between groups were analyzed for significance by performing a one-way analysis of variance combined with a Tukey test. A p-value of <0.05 was regarded as significant.
Results
Effects of TBPH and CUR treatment on spatial learning and memory in mice
Figure 1 presents the heatmaps of the five different treatment groups showing the 60 s mouse trajectories while searching for the platform. There was insignificant reduction in density of target area trajectories of mice in 200 mg·kg−1 TBPH group as compared to normal. There was also no significant increase in purposeful searching for the platform in the co-exposure group (TBPH + CUR) group compared with the 200 mg·kg−1 TBPH group. Heatmaps of mouse trajectories in the MWM for the different treatment groups (n=8). The yellow, green, and blue tracks represent the exercise residence times of the mice over the whole area. Yellow tracks indicate the longest time spent in motion, followed by green then blue, and no trace means that the mice did not move into that region.
Figure 2(a) shows that during the 5-day training period in the positioning navigation experiment, the average time taken to find the hidden platform gradually decreased in all groups, but no significant difference was found (p > 0.05), all groups showing a purposeful search for the hidden platform. In addition, there was no significant difference in swimming speed between the five groups of mice (p > 0.05) (Supplementary Table S1). Figure 2(b) shows that compared with the control, less time is spent in the target quadrant by the TBPH group, but the difference is not significant. Similarly, Figure 2(c) shows that even though there was a reduction in the number of times the TBPH mice crossed the platform area, the result is also not statistically significant compared with the control. The results of the 20 mg·kg−1 TBPH group compared with the 200 mg·kg−1 TBPH group showed that the search time in the target quadrant and the number of crossings of the platform area decreased with the increase in TBPH concentration. In contrast, the search time in the target quadrant and the number of crossings of the platform area in the TBPH + CUR group were higher than in the 200 mg·kg−1 TBPH group. Results of MWM for the different treatment groups (n=8). (a) The escape latency (s); (b) the time spent in the target quadrant; and (c) the number of crossings of the platform area.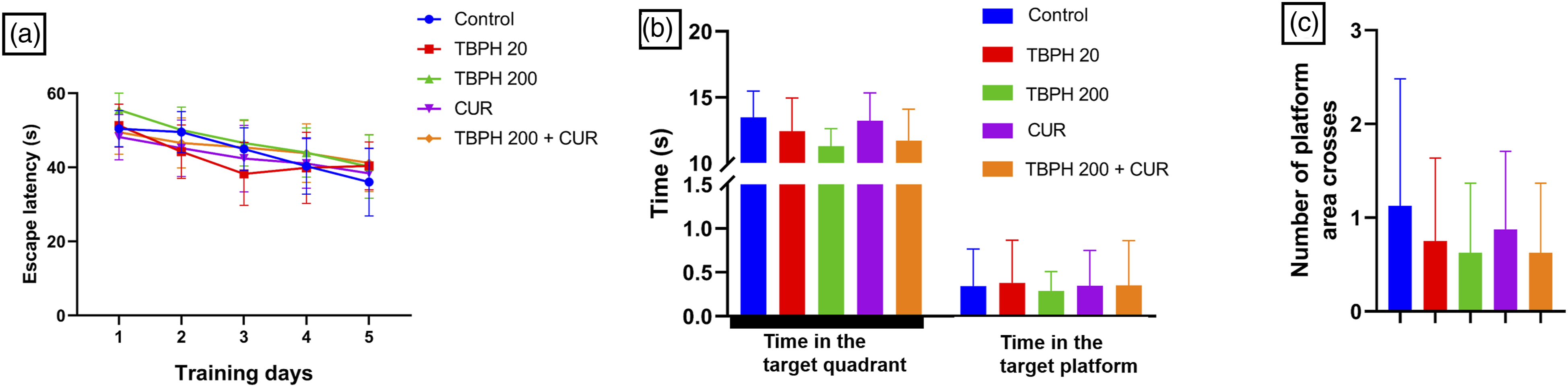
Effects of TBPH and CUR treatments on MDA, SOD, and GSH levels in the hippocampus of mice
As shown in Figure 3, there was significant (p < 0.05) increase in tissue MDA and SOD level and reduction in tissue GSH level as compared to control though the higher dose (200 mg·kg−1) being highly significant (p < 0.01). However, CUR significantly (p < 0.01) reversed the TBPH-induced increase in MDA, and SOD, and decrease in GSH level as compared to the TBPH (200 mg·kg−1) group. Oxidative damage levels in the hippocampus of mice from the different treatment groups (n=7). (a) The concentration of MDA; (b) the concentration of SOD; and (c) the concentration of GSH. * p < 0.05, ** p < 0.01, 200 mg·kg−1 TBPH group compared to the control; # p < 0.05, 200 mg·kg−1 TBPH group compared to the TBPH + CUR group.
Effects of TBPH and CUR treatments on levels of BDNF, p-CREB, and PSD-95 in the hippocampus of mice
Figure 4 presents the levels in BDNF, p-CREB, and PSD-95 in the hippocampus of mice from each group. There was significant decrease in BDNF and PSD-95 (p<0.05 and p<0.01, respectively) though the decrease in p-CREB levels was statistically insignificant following exposure to higher dose (200 mg/kg); however, lower dose did not produce significant changes as compared to control. CUR treatment restored the TBPH-induced depletion of BDNF, PSD-95, and p-CREB levels though insignificantly (p>0.05). The expression of memory-related proteins in the different treatment groups (n=7). (a) The level of BDNF; (b) the level of p-CREB; and (c) the level of PSD-95. * p < 0.05, ** p < 0.01, 200 mg·kg−1 TBPH group compared to the control.
Effects of TBPH and CUR treatments on expressions of GRP78, PERK, and CHOP in the hippocampus of mice
Figure 5 shows the western blotting analysis of GRP78, PERK, and CHOP protein expression in the hippocampal tissue of mice, demonstrating a significant up-regulation of GRP78, PERK, and CHOP in the 200 mg·kg−1 TBPH group (p < 0.01), compared to the control. Also, there was significant reduction in expression of GRP78 and CHOP in the TBPH + CUR group as compared to the TBPH (200 mg/kg) group (p < 0.05). Relative expression of GRP78, PERK, and CHOP protein in hippocampal tissue of mice from each group (n=3). (a) Western blot protein band, a: control, b: 20 mg·kg−1 TBPH group, c: 200 mg·kg−1 TBPH group; d: 25 mg·kg−1 CUR group, and e: 200 mg·kg−1 TBPH +25 mg·kg−1 CUR group; (b) the ratio of the gray value of GRP78 to the gray value of GAPDH; (c) the ratio of the gray value of PERK to the gray value of GAPDH; and (d) the ratio of the gray value of CHOP to the gray value of GAPDH. ** p < 0.01, compared to the control; # p < 0.05, 200 mg·kg−1 TBPH group compared to the TBPH + CUR group.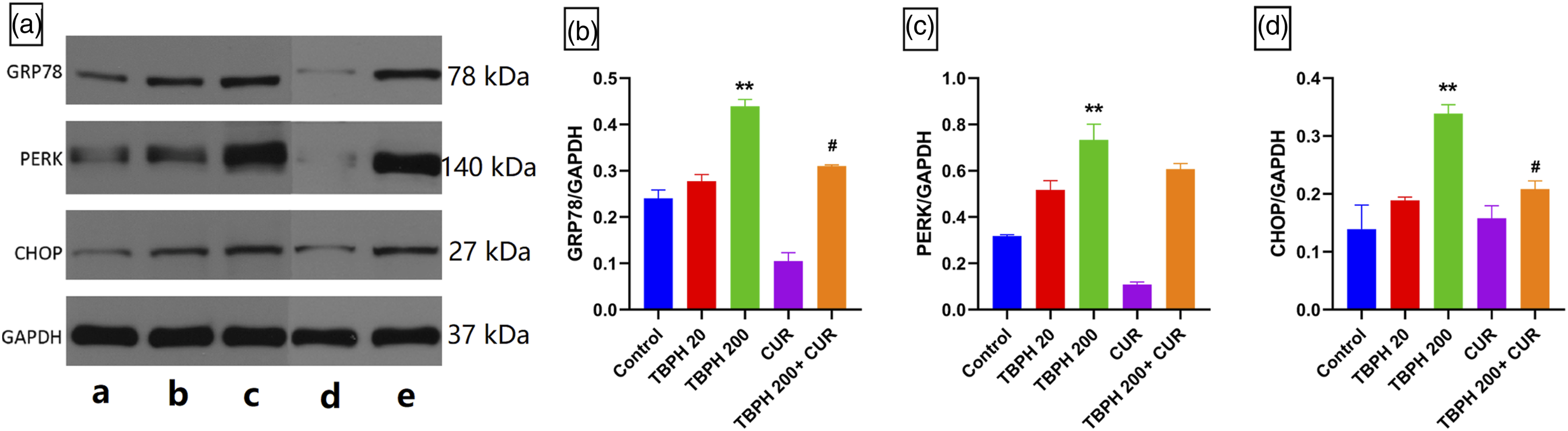
Discussion
Research into the neurotoxicity of TBPH is limited, and the few existing studies are mainly focused on FireMaster® 550.11,21 In a rat experiment, 10 days after perinatal female rats were orally exposed to FireMaster ®550 (0, 300, or 1000 μg/day), it was found that the level of 5-hydroxytryptamine (5-HT) in the placenta and the fetal forebrain decreased significantly. A decrease in 5-HT levels in the fetus may directly affect early nervous system development of the fetus, further showing that maternal exposure to FireMaster ®550 affected the nervous system development of the fetus by crossing the blood–fetal barrier. 21 In this study, although TBPH exposure for 28 days did not reduce the learning and memory of mice, higher dose (200 mg/kg) TBPH could increase oxidative stress level in hippocampus, up-regulate the expression of ER stress proteins in hippocampal neurons, and have negative effects on learning and memory-related proteins. In addition, the results of our study suggest that CUR could be considered as candidate to prevent TBPH exposure–induced neuronal damage.
Previous studies have confirmed that the commercial FireMaster ®550 flame retardant–containing TBPH has sex-specific effects on socioemotional behavior in prairie voles, 12 while this study focused on the neurological changes caused by TBPH alone. The results of the Morris water maze test performed after 28 days of continuous exposure to TBPH showed that although the time spent searching for the target quadrant and the number of platform crossings of the treated groups were lower than those in the control, there was no significant decrease in learning and memory, indicating that TBPH exposure alone does not have a strong neurological effect. However, since the exposure period for this experiment was only 28 days, the above observation is still worthy of further study.
Other studies have shown that TBPH is a brominated homologue of bis(2-ethylhexyl) phthalate (DEHP) and has a structure similar to DEHP. The similar structure and metabolic pathway of TBPH to DEHP suggest that TBPH may have similar biological toxic effects as DEHP. 22 Increasing evidence from experimental and clinical studies have shown that oxidative stress (OS) plays an important role in the pathogenesis of learning and memory impairment caused by DEHP.23,24 We therefore explored possible pathways such as OS and endoplasmic reticulum stress (ERS) to investigate whether TBPH has similar adverse effects. Abnormally high levels of free radicals and reduced antioxidant levels in cells can increase lipid peroxidation and eventually lead to histopathological changes or organ dysfunction. 25 The results from our study show that there were significant changes in MDA, SOD, and GSH levels in the hippocampus of the mice in the higher dose (200 mg/kg) TBPH group compared with those in the control, suggesting that TBPH could cause peroxidation damage, and that oxidative stress induced by TBPH may be responsible for hippocampal damage in mice.
Abnormal lipid accumulation is often associated with perturbed endoplasmic reticulum (ER) proteostasis in cells. 26 The ER is a critical organelle for protein synthesis, folding, and quality control of these proteins. 26 Numerous genetic and environmental insults impede the ability of cells to properly fold and posttranslationally modify secretory and transmembrane proteins in the ER, leading to a buildup of misfolded proteins in this organelle, a condition known as ER stress. 27 Chronic ER stress and defects are emerging as key contributors to a growing list of human diseases, including diabetes, neurodegeneration, and cancer. 28 GRP78, PERK, and CHOP are markers of ER stress. 29 As expected, our results demonstrated that a significant up-regulation of GRP78, PERK, and CHOP in the 200 mg·kg−1 TBPH group occurred, suggesting that ER stress–related proteins may also be involved in the TBPH-induced adverse effects.
As a result of TBPH exposure, a series of changes in abnormal molecular protein levels or expression (end effects) caught our attention. PSD-95 is a cytoskeleton protein that is one of the most abundant proteins in the post-synaptic dense region and is abundantly expressed in the brain, especially in the hippocampus. 30 It is a necessary protein molecule in the early stage of memory formation. 30 Pharmacological studies have found that BDNF plays an important role and is necessary in the later stage of memory formation. 31 p-CREB is also a molecular marker of hippocampal memory processing. 32 In this study, we found that TBPH decreases the levels of BDNF, p-CREB, and PSD-95 in the hippocampus, suggesting that TBPH may negatively affect hippocampal synaptic plasticity.
Reportedly, CUR has a variety of pharmacological effects, which can alleviate memory impairment in rodents, and can play a role in the prevention and treatment of memory impairment. 33 As an antioxidant, CUR can increase the activity of SOD, reduce the depletion of GSH, and inhibit the apoptosis pathway of mitochondria. 34 The results of this study also show that CUR could significantly attenuate the increase in MDA levels and the decrease in GSH levels induced by TBPH. Additionally, Wei et al. 35 reported that CUR attenuated dichloroacetic acid (DCAA)–induced oxidative stress and attenuated the inflammation response and impaired synaptic plasticity, while elevating p-CREB, BDNF, and PSD-95 levels, thereby conferring neuroprotection against DCAA-induced neurotoxicity. Our study showed that CUR could increase the levels of p-CREB, BDNF, and PSD-95 that had been reduced by TBPH exposure. Although there are some difficulties with using CUR in practical application, such as low solubility, instability under physiological conditions, and low absorption rate, the clinical pharmacological effects of CUR still have research value. 36 The results of a study of 40 subjects (aged 51–84 years) with mild memory impairment showed that those who received CUR (Theracurmin® containing 90 mg of CUR) twice a day improved their memory and attention in non-dementia adults up to 28% over 18 months compared with the control. 37 Therefore, we can postulate that CUR may contribute to the reduction of TBPH-mediated neurotoxicity, at least to some extent. Moreover, our results further show that the neuroprotective effect of CUR may be related to its ability to reduce the level of oxidative stress and the expression of proteins related to the ER stress pathway.
Most studies evaluating the toxic effects of TBB or TBPH used commercial mixtures such as FireMaster ®550.11,12 Considering the extensive use of industrial products containing TBPH in daily life, exposure to TBPH is inevitable. However, as this study shows, exposure to TBPH alone is not enough to significantly damage neurobehavior. The limitations of our study are that whether prolonged exposure has a similar effect remains unknown and whether animals of other species are suitable for this study requires further research. Overall, the results of this study are still meaningful since the demonstrated protective effect of CUR on TBPH exposure provides a potential approach to reduce the neurotoxic effects of TBPH.
Conclusion
Firstly, our in vivo experiment provides evidence that continuous 28-day TBPH exposure does not cause significant neurobehavioral damage. Secondly, we observed that TBPH increases the level of peroxidation and the expression of ERS-related proteins in the hippocampus, while reducing the level of memory-related proteins. More importantly, we found that CUR has a neuroprotective effect on TBPH exposure, which may be related to its own antioxidant capacity.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211058876 – Supplemental Material for Brominated flame retardant TBPH induced oxidative damage and reduced the expression of memory-related proteins in mice, with no discernable impairment of learning and memory
Supplemental Material, sj-pdf-1-het-10.1177_09603271211058876 for Brominated flame retardant TBPH induced oxidative damage and reduced the expression of memory-related proteins in mice, with no discernable impairment of learning and memory by Zhang Bao and Yin Jing in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
