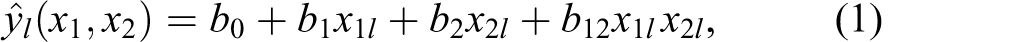Abstract
After repeated intraperitoneal injections of nickel and chromium (VI) salts to rats, we found, and confirmed by mathematical modeling, that their combined subchronic toxicity can either be of additive type or depart from it (predominantly toward subadditivity) depending on the effect assessed. Against the background of moderate systemic toxicity, the combination under study proved to possess a marked additive genotoxicity assessed by means of the random amplification of polymorphic DNA test. We also demonstrated that chromium and nickel reciprocally influenced the retention of these metals in some organs (especially in the spleen) but not their urinary excretion in this study.
Introduction
High-strength nickel (Ni)–chromium (Cr) stainless steels of austenitic type are produced on a large scale and are extensively used in electric arc welding, thus accounting for concurrent occupational exposure to Cr and Ni as one of the most widespread variants of combined toxicity. Metal plating technologies involve the presence of aerosols containing metal salt solutions in the workroom air, where both Cr and Ni plating processes are performed, and thereby arising conditions for combined exposure to these metals.
The effects and mechanisms of toxic action have been extensively studied for both of these metals, and the findings of a great number of studies have been summarized in monographs, including official documents. 1 -4 At the same time, there are very few works devoted to the combined toxicity of Cr and Ni and those few that are available were carried out a long time ago and largely involved single exposure to lethal doses. Thus, in our laboratory, we experimentally showed in mice that effects of combined exposure to lethal doses of Cr (VI) and Ni were less than additive (implicit antagonism). 5 Later on, Johansson et al 6 demonstrated antagonism of the damaging action of Ni and Cr (III) on the alveolar macrophages in rabbits. Rudolf and Cervinka, 7 who studied the effects of 24-hour in vitro exposure of primary human skin fibroblasts to Ni and Cr (VI), found that high Ni concentrations in combination with Cr (VI) induced rapid cell damage leading to necrosis, but lower (nontoxic) Ni concentrations prevented Cr-induced damage and apoptosis, mainly by overexpression of heat shock proteins.
These findings are not unexpected because subadditivity or even explicit antagonism are, in fact, common effects of combined toxicity of metals, although where exposure is chronic and a large number of effects are involved in the assessment, both additivity and superadditivity (synergy) may be observed for some of these effects. 8 -10 It was therefore difficult to predict the type of combined toxicity characteristic of Cr and Ni in the more important case of long-term exposure.
Besides its specific relevance, the issue under consideration has broader implications for expanding the experimental basis of the theory of combined toxicity, which is one of the pressing problems in general toxicology. In particular, only little has been studied regarding the toxicokinetic mechanisms of combined chronic toxicity, despite the fact that some researches point to their important role. Thus, it has been shown that lead reduces the accumulation of fluoride in bone tissue of rats 11 and inhibits oxidative biotransformation of naphthalene and, thereby, its excretion from the organism. 12 It has also been shown that combined exposure of rats to lead and cadmium resulted in reduced urine excretion of lead and increased urine excretion of cadmium, compared to groups exposed to these metals separately. 10 It is necessary to gather more data on reciprocal effects of toxic substances on their distribution in and elimination from the organism.
To characterize combined toxicity, modern toxicology usually operates the terms “additivity,” “synergism” (or synergy, or potentiation, or superadditivity), and “antagonism” (or subadditivity). However, the exact meaning of each of these terms can vary depending on the underlying paradigm of combined adverse action preferred by a researcher 8,13 -16 or, even if not explicitly, by an agency (e.g. the US Environmental Protection Agency or the American Conference of Governmental Industrial Hygienists). The most widespread concept assumes that there exist 2 fundamentally different types of combined toxicity and thus accepts 2 different meanings of the central term “additivity.” The “additivity of effects” is deemed to belong only to the realm of the so-called independent action of 2 or more substances at different biological sites evoking one and the same outcome. The “additivity of doses” or the so-called “Loewe additivity” is based on the assumption that 2 or more chemicals can act on the same biological site by the same mechanisms, differing in their potency only and thus being mutually replaceable in isoeffective doses.
A review of current discussions around these seemingly contradicting but, in reality, quite often coexisting paradigms has been recently published by us together with presentation of our own views. 10 Specifically, we analyzed the results obtained in experimental lead–cadmium subchronic intoxication seeking to identify the type of combined toxicity with the help of 2 mathematical models based on either (a) analysis of variance (ANOVA) or (b) Mathematical Theory of Experimental Design, which correspond, respectively, to the paradigms of effect additivity and dose additivity. We applied the same approach to studying the lead–fluorine combination 11 and came to virtually the following same conclusions:
the above-mentioned mathematical models are virtually interchangeable, and thus the paradigms under consideration may be regarded as different methods of modeling the combined toxicity rather than as reflecting fundamentally differing processes; and
within both models, there exists not merely 3 types of combined toxicity (additivity, subadditivity, and superadditivity) mentioned previously, but at least 10 variants of it depending on exactly which effect is considered, on its level, on the dose levels and their ratios as well as on a particular effect of 2 toxics being unidirectional or opposite.
Materials and Methods
The experiment was carried out on outbred white female rats (from our own breeding colony) with the initial age of 4 months and body mass of around 220 to 230 g. From each exposed or control group, 10 to 12 rats were selected at random. All rats were housed in conventional conditions, breathed unfiltered air, and were fed standard balanced food. The study was planned and implemented in accordance with the “International guiding principles for biomedical research involving animals” developed by the Council for International Organizations of Medical Sciences (1985) and approved by the Ethics Committee of the Ekaterinburg Medical Research Center Medical for Prophylaxis and Health Protection in Industrial Workers.
In a preliminary experiment, we established median lethal dose (LD50) values for potassium dichromate (K2Cr2 O7) and Ni chloride (NiCl2) administered once intraperitoneally (ip) to be equal to 30 and 66 mg/kg body mass, respectively. In the main experiment, the rats were injected ip 3 times a week (up to 20 injections) with water solution of either one of these salts in a dose equivalent to 0.05 LD50 (1.5 mg/kg and 3.3 mg/kg, respectively) or both together in the same doses, the controls receiving injections of distilled water in the same volume (4 mL/rat, ie, about 20 mL/kg). The doses of the salts used by us correspond to 0.53 mg/kg of Cr and1.51 mg/kg of Ni, respectively (note 1)
We used repeated ip injections instead of inhalation exposure to toxics for more exact dosing, which is an important consideration in comparative experiments performed for quantitative estimation of effects and their interactions. After the exposure period, the following procedures were performed for all rats only once: measuring body weight. Estimating the ability of the central nervous system (CNS)’s for temporal summation of subthreshold impulses (SSTI index)—a variant of withdrawal reflex and its facilitation by repeated electrical stimulations in an intact, conscious rat.
17
(In this variant, the rat held by the experimenter is brought with its hind legs against a plate to which an electrical current is applied with a pulse length of 0.1 millisecond and a frequency of 1 pulse/second, the current strength being smoothly increased from zero to upward. The instrument records the time in seconds from the moment the current is switched on till the electric circuit is disconnected as the rat jerks away at least 1 of its legs.) Recording of the number of head dips into holes in a hole board, which is frequently used for studying the behavioral effects of toxicants and drugs.
18,19
(The rat can move freely around the board of 55 by 55 cm with 16 uniformly distributed holes of 4.5 cm in diameter, and the experimenter counts the number of head dips during the first 3 minutes after the rat is placed on the board.) Collecting urine during 24 hours to determine its output (diuresis) as well as total coproporphyrin, δ-aminolevulinic acid, and creatinine content. Collecting capillary blood from a notch on the tail for examination of the white blood cell count, hemogram, red blood cell count (as well as proportion of reticulocytes), and hemoglobin content. Rats were killed by decapitation and core blood was collected. Biochemical indices were determined from the serum, including total serum protein, albumin, globulin, alanine transaminase and aspartate transaminase, ceruloplasmin, alkaline phosphatase, γ-glutamyl transpeptidase, catalase, total SH-groups, and bilirubin. Reduced glutathione (GSH) was determined in the whole blood hemolysate.
All the above-mentioned clinical laboratory tests on blood and urine were performed using well-known techniques as described previously. 20 We used values for 24-hour renal excretion of Cr and Ni as exposure biomarkers and as indices for the ability of kidneys to eliminate these metals, the urine concentrations of which were measured using the AA-6650F atomic absorption spectrophotometer (Shimadzu Corp, Japan). 21 The same AAS technique was used to determine tissue concentrations of both metals in the liver, spleen, kidneys, and brain of rats.
The level of genomic DNA fragmentation as an index for the metals’ in vivo genotoxicity was assessed in blood leukocytes using the random amplification of polymorphic DNA test as described earlier. 22,23 The method is based on the fact that, unlike a fragmented DNA, which forms the so-called “comet tail” during agarose gel electrophoresis, a nonfragmented DNA has a very low degree of migration and virtually stays in the same place (“comet head”), the degree of migration being directly related to the degree of DNA fragmentation. To characterize the degree of damage to DNA tagged with tritium, we used “coefficient of fragmentation,” that is, the ratio of total radioactivity of all “tail” fractions to the radioactivity of the “head.” The statistical significance of differences between group arithmetic mean values was estimated using Student t test with Bonferroni correction. 24 To identify the type of combined action of Cr and Ni based on biochemical, cytological, and functional indices for the organism’s status, we used (a) 2-way ANOVA and (b) the mathematical theory of design of experiments (DOE). Both approaches are theoretically justified and described in detail by Varaksin et al. 10 As they have led to virtually the same conclusions, we describe that only in some detail based on the DOE theory.
The said mathematical theory was used for constructing the surface of response Y = Y(x1, x2), that is, the function describing the change in values of index Y with a change in the doses of toxicants x1 and x2. The doses of the toxicants Ni and Cr to which the animals in our experiments were being exposed (0.05 LD50) are relatively small and therefore, the response surface may be described by the simplest function:
where
Results
In this section, we only present actually measured values, while the main results of combined toxicity mathematical modeling will be dealt with in the Discussion section. To avoid data redundancy, Table 1 contains only those functional and biochemical indices of all those are listed in Materials and Methods section, which feature a statistically significant difference between at least any 2 of the compared groups. (Other indices also measured by us but not meeting this criterion are tabulated in the Supplement.) Data on the metal contents of tissues and urine are presented in Tables 2 and 3, respectively.
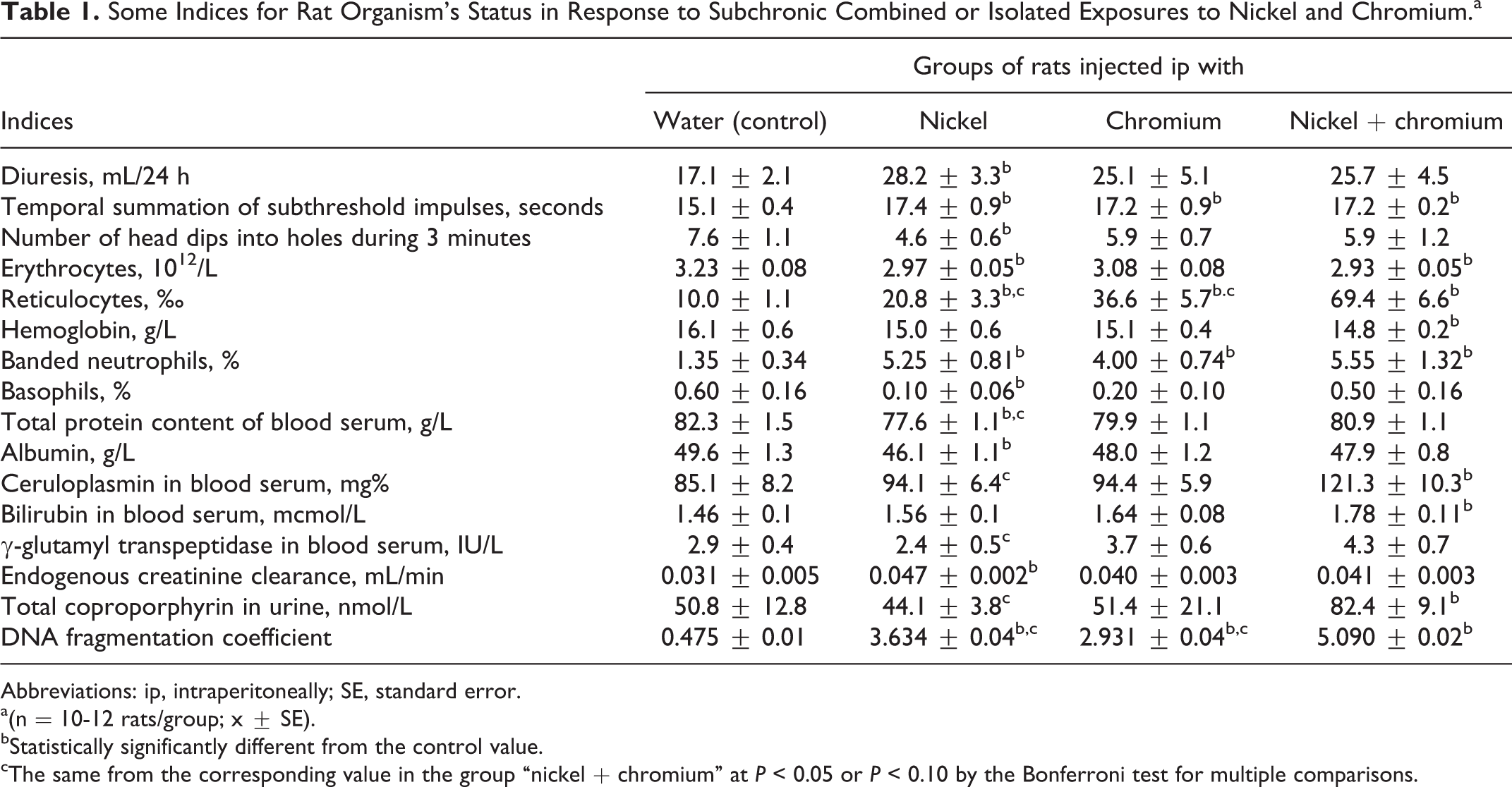
Some Indices for Rat Organism’s Status in Response to Subchronic Combined or Isolated Exposures to Nickel and Chromium.a
Abbreviations: ip, intraperitoneally; SE, standard error.
a(n = 10-12 rats/group; x ± SE).
bStatistically significantly different from the control value.
cThe same from the corresponding value in the group “nickel + chromium” at P < 0.05 or P < 0.10 by the Bonferroni test for multiple comparisons.
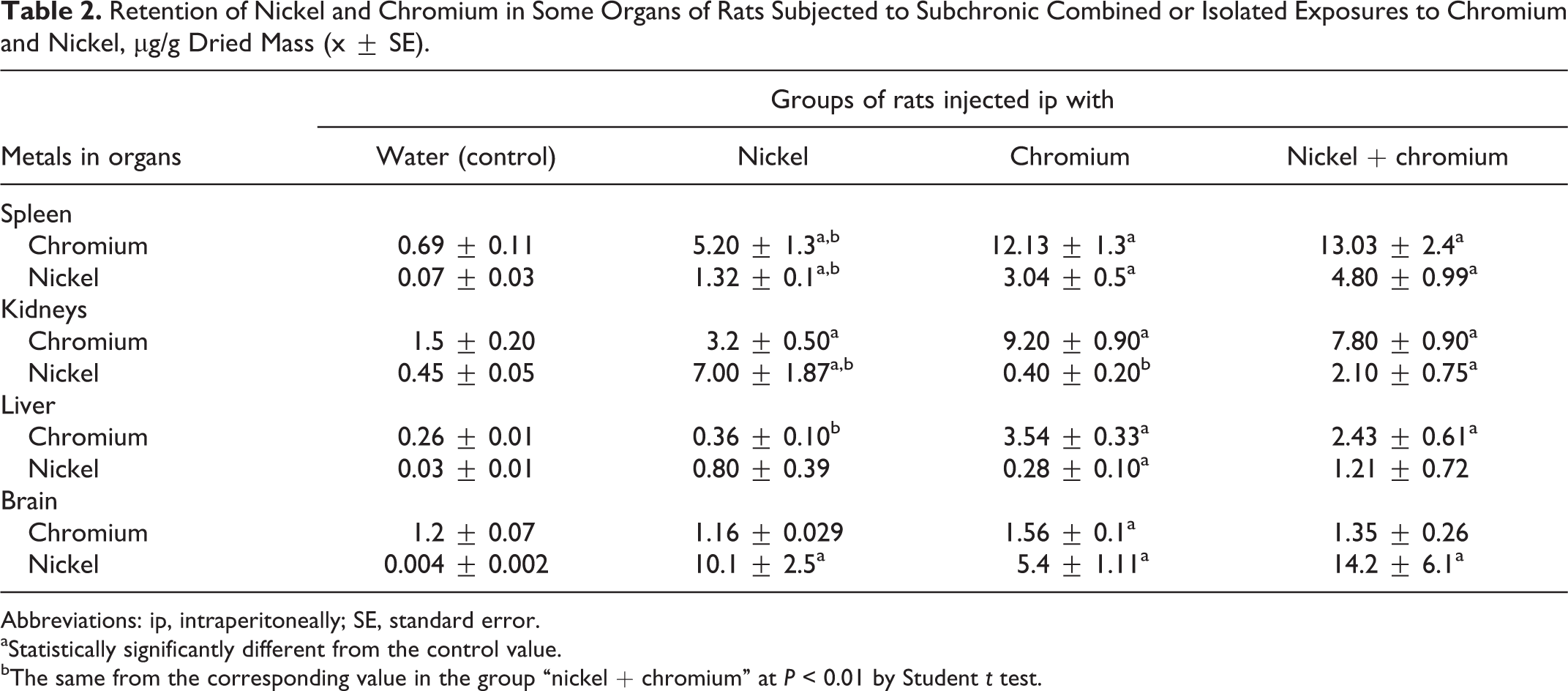
Retention of Nickel and Chromium in Some Organs of Rats Subjected to Subchronic Combined or Isolated Exposures to Chromium and Nickel, μg/g Dried Mass (x ± SE).
Abbreviations: ip, intraperitoneally; SE, standard error.
aStatistically significantly different from the control value.
bThe same from the corresponding value in the group “nickel + chromium” at P < 0.01 by Student t test.
Urinary Concentrations of Nickel and Chromium Normalized to Creatinine in Rats Subjected to Combined or Separate Exposures to Chromium and Nickel, μg/g (x ± SE).

Abbreviations: ip, intraperitoneally; SE, standard error.
aStatistically significantly different from the control value.
bThe same from the corresponding value in the group “nickel + chromium” at P < 0.01 by Student t test.
Table 1 shows a statistically significant difference in at least any one of the exposed group from the control group for only 16 indices of the 39 measured. This fact points to the relatively low subchronic toxicity of Ni and Cr at the doses selected for the study. Given this, of special importance becomes the fact that the DNA fragmentation coefficient was statistically significantly elevated in response to the metals, both administered separately and, particularly, in combination (P < 0.0001).
Table 2 shows that exposure to one metal of the studied combination more or less changed concentration of organs not only for this but also for another metal. (This effect is described in more detail in the Discussion section.)
Table 3 shows that urine concentrations of Cr and Ni (normalized to that of creatinine) at the end of subchronic treatment with the respective metal separately or in combination were statistically significantly increased compared to control values. However, the lowest concentration of Cr was detected in the urine of rats treated with Ni.
Discussion
As it was mentioned earlier, at the chosen doses both metals studied by us were moderately toxic even when acting in combination. The shifts they induced (see Table 1 and the Supplement) are mostly not specific for toxicodynamics of a particular poison and rather reflect general changes in the organism’s homeostasis characteristic of many other intoxications. In particular, judging by our previous experience (for instance 10 and 11 ), quite typical is a misbalance between excitation and inhibition processes in the CNS. A prevalence of inhibition manifests itself by both the increase in the index of Temporal Summation of Subthreshold Impulses and decrease in exploratory activity as measured by the number of head dips.
One important exception is a probably specific hemolytic effect of Ni discussed subsequently in connection with toxicokinetics of Cr. It is also possible that decrease in serum total protein and albumin as well as increase in ceruloplasmin are signs of hepatoxicity common for many heavy metals, Cr and Ni included 1,2 We are not sure, however, that renal hyperfiltration manifested by an increase in the endogenous creatinine clearance may be interpreted as connected with nephroptoxic action of metals.
The genotoxicity of Ni and Cr as underlying the carcinogenicity of these metals has been known for a long time 1 -4 and, in particular, their DNA-damaging action for a rat liver–derived cell line was demonstrated in parallel, 26 but, to our knowledge, the obvious additivity of this most dangerous effect of these metals has been shown here for the first time. As for the majority of the other indices (irrespective of whether or not the difference from the controls was significant), the values obtained in the group (Ni + Cr) practically do not exceed the values obtained in the groups of isolated exposure, which point to the subadditivity of action. At the same time, combined exposure accounts for some effects being more pronounced (though not always statistically significantly) compared with both groups of isolated exposure. This is demonstrated by indices such as the number of reticulocytes, blood level of reduced GSH, and some indices of damage to the liver (organ mass, ceruloplasmin, alkaline phosphatase, γ-glutamyl transpeptidase, blood serum bilirubin), and an increased concentration of coproporphyrin in urine, which can also serve as a hepatotoxicity index (note that some of these indices are not included in Table 1, considering the insufficient statistical significance of the intergroup differences). Whether the combined action in all of these effects is only additive or superadditive may only be ascertained by mathematical modeling.
In full conformity with what is stated in the Introduction section, such modeling has revealed fundamental agreement between the conclusions on the type of combined toxicity derived by 2 different modeling methods (ANOVA and DOE). However, there are distinctions between these conclusions depending on the effect (and sometimes the level of effect) by which this type is estimated. Of the theoretically possible 10 subvariants of combined toxicity that we had actually observed for the combined action of lead and cadmium,
10
we have identified only 5 for Cr and Ni: additivity—as concerns GSH, liver mass, hemoglobin, bilirubin, DNA fragmentation coefficient, and ceruloplasmin (illustrated in Figure 1A); superadditivity (synergy)—as concerns reticulocytes and coproporphyrin (illustrated in Figure 1B) subadditivity (antagonism) for unidirectional action of metals—as concerns temporal summation of subthreshold impulses, banded neutrophils, alkaline phosphatase, γ-glutamyl transpeptidase, and erythrocytes (illustrated by Figure 1C); antagonism for unidirectionally and oppositely acting Ni and Cr in different dose ranges—as concerns albumin, endogenous creatinine clearance, and number of head dips into holes (illustrated by Figure 2A); and antagonism and synergy for the effect of the same level but for different doses—as concerns basophils and total serum protein (illustrated by Figure 2b and 2c).
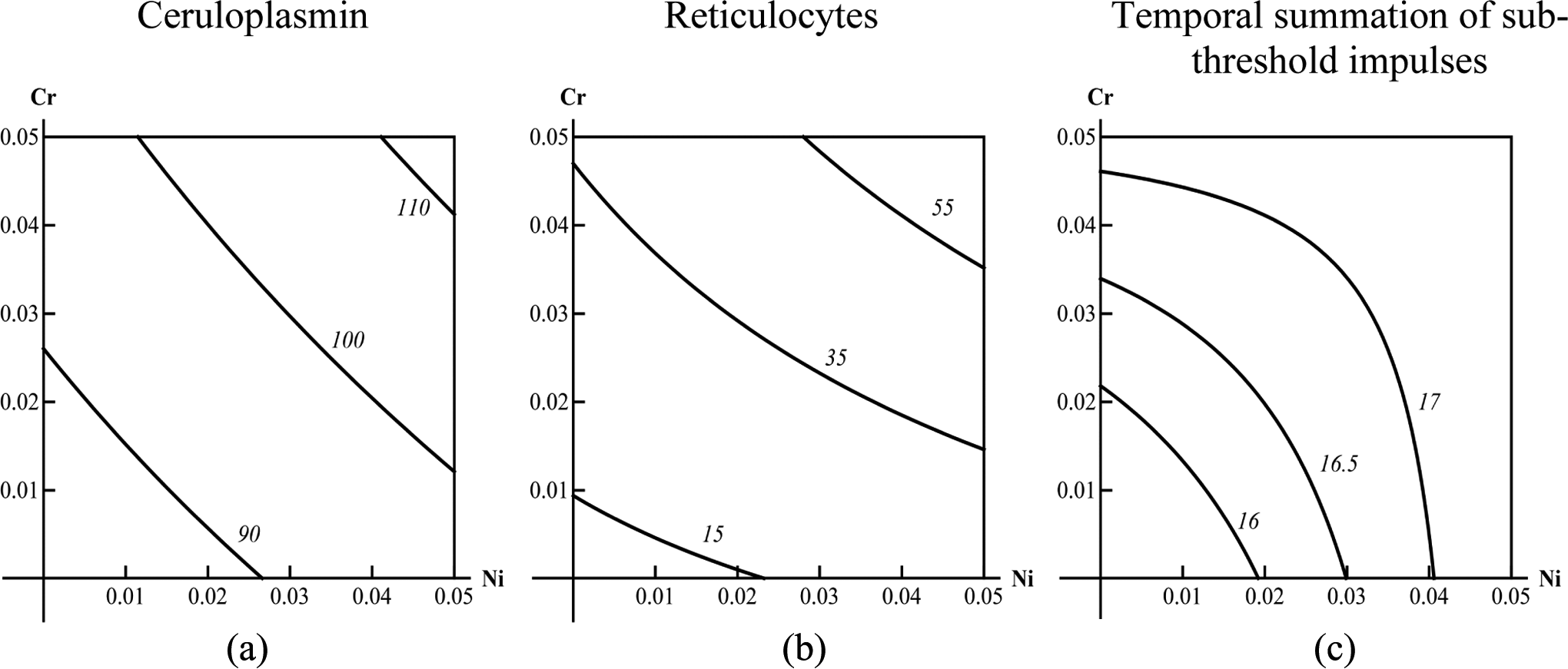
Isoboles for 3 basic types of combined exposure to nickel and chromium: (A) additivity, (B) synergy, and (C) subadditivity. The axes of coordinates show the doses of the toxicants as fractions of median lethal dose (LD50) and numbers at the isoboles show the level of effect on which the isobole is constructed.
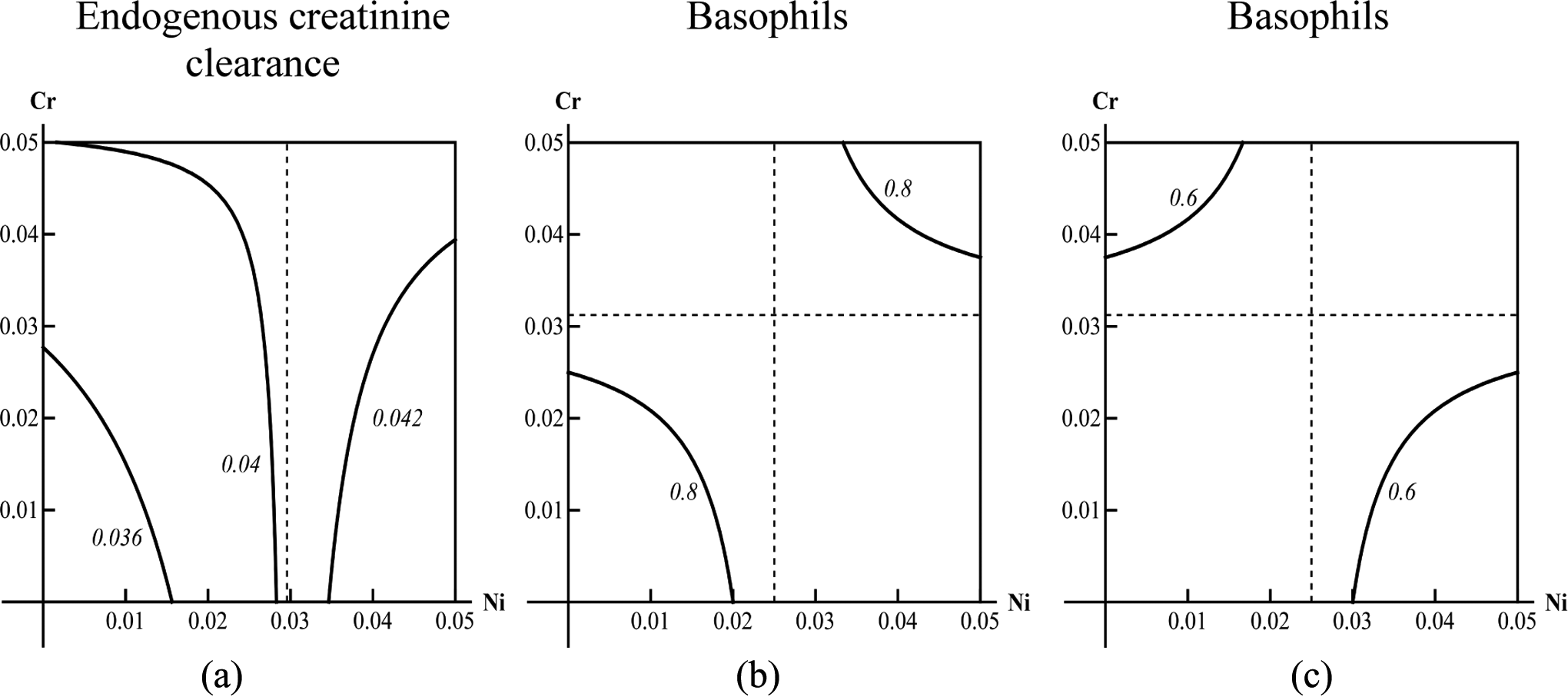
Examples of isoboles for subvariants of combined action of nickel and chromium: (A) antagonism for unidirectional (low doses of nickel) and opposing (high doses of nickel) actions of metals; (b) one of the branches of the isobole (high level of effect) which corresponds to unidirectional action of nickel and chromium (dependence of the combined action on the range of doses is the same as in A); (C) the other branch of the same isobole (low level of the same effect), which corresponds to opposite actions of nickel and chromium (dependence of the combined action on the range of doses is inverse to that in A and B).
One observation of interest is the seemingly paradoxical increase in the Cr content of the spleen above the background level in the rats that were administered Ni alone (Table 2), the increase being marked (7.5 times) and statistically highly significant. As is known, the red pulp of the spleen filters out aged erythrocytes that are recognized by the macrophages of this tissue by essentially changed surface properties and structure. Meanwhile, a circulating erythrocyte is a kind of kinetic trap for blood Cr: the erythrocyte membrane lets in only hexavalent Cr, which inside the cell is reduced to Cr (III) by various mechanisms (but basically involving GSH) and is thus sequestered within the erythrocyte until the latter is destroyed as a result of hemolysis—either intravascular, or in normal conditions, mainly intracellular (ie, carried out by macrophages, first of all, those of the spleen). 3
Meanwhile, as is apparent from Table 1, the Ni treatment caused a statistically significant decrease in the number of circulating erythrocytes along with a very substantial increase in the proportion of reticulocytes, which is characteristic of hemolytic anemias. We have no direct evidence of intracellular hemolysis being enhanced by Ni in our experiment; it is known, however, that Ni accelerates aging of erythrocytes by changing the properties of the membrane lipids and proteins. 27 Juxtaposition of these facts suggests that the elevated retention of the “background” Cr in the spleen of Ni-exposed rats may be explained by this action of Ni on the erythrocytes.
However, we cannot rule out one more mechanism of this phenomenon associated with the above-mentioned role of GSH in the sequestration of Cr by erythrocytes. The blood level of reduced GSH equal to 0.298 ± 0.06 mmol/L in the control group was markedly, though statistically not significantly, elevated in response to Ni or Cr in combination with Ni (to 0.425 ± 0.10 and 0.505 ± 0.13 mmol/L, respectively). It is difficult to explain the mechanism of such toxic activation of the reduced GSH, but it could lead to enhanced transformation of Cr (VI) into Cr (III) and, therefore, to enhanced retention of the latter by erythrocytes and thus by the spleen as well.
It should be mentioned, however, that although the elevated accumulation of Cr in the spleen of rats exposed to combined Ni–Cr intoxication was also slightly higher than those exposed to Cr alone, this difference is neither large nor statistically significant (Table 2). It is hard to say as yet why this phenomenon is less marked in relation to Cr injected to rats than it is in relation to background Cr, especially considering the fact that the effect of Cr–Ni combination on the red blood cells is even stronger than the effect of isolated Ni exposure (Table 1). Nevertheless, in both cases, this effect is unidirectional, which counts in favor of its consistent character.
Resident macrophages are present in different organs, which means that the spleen is not the unique (although most important) location of intracellular hemolysis. One could therefore expect some increase in the background Cr under the effect of Ni also in organs other than the spleen. We can indeed see such an increase in the kidneys and an insignificant increase in the liver. However, the Cr content of these organs in response to Cr–Ni exposure, on the contrary, tended to decrease in comparison with exposure to Cr alone. In the brain, Ni did not cause any substantial changes in either the background Cr or its retention in Cr-treated rats. Thus, this toxicokinetic phenomenon should be recognized as specific to the spleen.
At the same time, our data point to the reciprocal influence of Cr on Ni distribution in the organism. Indeed, as can be seen from Table 2, for isolated Cr treatment, the Ni content of the spleen is statistically significantly increased compared to the control group by a factor of 43, and for combined Cr–Ni exposure, it is statistically significantly higher in comparison with the group of isolated Ni exposure by a factor of 3.6. The liver of the rats was found to contain almost 10 times more Ni in response to isolated Cr exposure than in the control group and 1.5 times more in response to combined exposure compared with isolated Ni exposure, although this difference is not significant. Similar intergroup differences can be seen in relation to the Ni content of the brain. In the kidneys, however, Cr intoxication did not affect the background Ni content, while after the combined Cr–Ni exposure, it was significantly lower compared with isolated Ni exposure. We have to admit that we do not have a sufficiently consistent working hypothesis for explaining any influence of Cr on the distribution of Ni.
As it is known, kidney is the main organ for both Ni and Cr excretion. 1 -4 Treatment with these metals raised their urinary excretion considerably above the background level (Table 3), but the only noticeable difference between the groups of isolated and combined exposure was decreased concentration of Ni in response to simultaneous exposure to Cr. This effect, however, was not statistically significant, and the presence of Ni in the toxic combination did not have any effect on the elimination of Cr. Paradoxically, separate treatment with Ni significantly decreased the background urine concentration of Cr, and we are not able to propose any consistent hypothesis to explain this result. Thus, the above-mentioned features of metal retention in the kidneys do not demonstrate direct correlation with the elimination of these metals with urine (cf Tables 2 and 3). We should admit, however, a limitation of this study, that is, giving us no possibility to estimate the day-to-day fluctuations of urinary concentrations (especially after each single ip injection of metals).
Conclusions
Combined subchronic toxicity of Ni and Cr can have an additive character or depart from it depending on the effect by which it is estimated.
Against the background of moderate general toxic effect, the genotoxicity of the combination studied is clearly marked and is additive, which allows us to predict an exceptional carcinogenic risk in association with industrial technologies that generate such combination.
A reciprocal influence of Ni and Cr on the distribution of the other metal in the organism is shown.
Footnotes
Author Contribution
Ilzira A. Minigaliyeva contributed to conception or design, acquisition, analysis, or interpretation, drafted the manuscript, gave final approval , and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Boris A. Katsnelson contributed to conception or design, acquisition, analysis, or interpretation, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Larisa I. Privalova, Vladimir B. Gurvich, Vladimir G. Panov, Anatoly N. Varaksin, Oleg H. Makeyev, Marina P. Sutunkova, Nadezhda V. Loginova, Ekaterina P. Kireyeva, Ekaterina V. Grigoryeva, Tatyana V. Slyshkina, Eugenia V. Ganebnykh, and Svetlana V. Grebenkina contributed to acquisition, analysis, or interpretation, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The conduct of the research and the preparation of this paper were financed by the Ekaterinburg Medical Research Centre for Prophylaxis and Health Protection in Industrial Workers with the help of the Russian Foundation for Basic Research (Grant No. 12-01-00218).
Note
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.