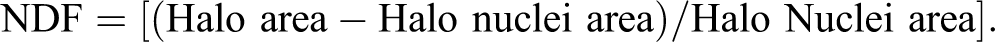Abstract
Although Halo assay is a fast and more economic technique, it is not popular compared to comet assay for the measurement of DNA damage. One of the reasons behind this was nonavailability of suitable user-friendly program. Currently, most of the researchers were analyzing halo images manually using image analysis software (Scion Image or ImageJ). To address this problem, I have developed a semiautomatic halo analysis ImageJ program, HaloJ, and applied in the assessment of DNA damage at the single-cell level. In this article, we have shown that data obtained from the HaloJ program have a very good correlation with the data obtained using comet assay analysis program such as Comet Assay Software Project. To the best of our knowledge, this will be the first program to quantify DNA damage of halo images. This program will be of great use for researchers working on the DNA damage and repair, radiation biology, toxicology, cancer biology, and so on.
Introduction
A variety of chemical compounds can damage the DNA of living cells. These damages include base damage, single-strand breaks, and double-strand breaks. If left unrepaired these damages will lead to malignant transformation. The detection of such lesions is of primary importance from a toxicological point of view. A variety of methods have been developed for detecting DNA damage. Assays such as micronucleus and sister chromatid exchange are based on the enumeration of downstream aberrations after initial DNA damage 1 –3 whose extent can be indirectly estimated. Other assays like alkaline and neutral filter elution of genomic DNA directly detect DNA strand breaks, 4 which is a sensitive but time-consuming method. Later, comet assay came into picture, which has an additional advantage of being as sensitive as the alkaline elution technique. Comet assay or single-cell gel electrophoresis is very good technique to detect DNA damage at high sensitivity. The comet assay is based on an electric field to pull damaged DNA out of cells trapped in the agarose gel. Comet assay is a very good but time-consuming technique. Comet assay has several advantages such as (1) measures DNA damage at the single-cell level, (2) only few cells are needed to carry out the assay, (3) does not require any prelabeling of DNA, and (4) a very sensitive method for detecting DNA damage. 5
Halo assay is another technique to measure DNA damage. The term “halo” refers to the shape of radially extracted DNA fragments from isolated nuclei. The symmetric shape of each halo allows precise determination of a DNA fragment without much intervention of the user. In this assay, the cells are lysed and individual nucleoids are visualized as “halos,” and thereafter, halo area can be measured by an image analysis system. Sestili and Cantoni developed a novel technique, the alkaline halo assay (AHA), which allows the measurement of DNA single-strand breakage at the single-cell level and presents similar sensitivity and some advantages with respect to the comet assay. 6 The AHA is similar to comet assay in the slide preparation but is based on entirely different principle. Instead of relying on an electric field to pull damaged DNA out of the cells, the halo assay is dependent on a short, postlysis incubation in an alkaline hypotonic buffer, which leads to self-diffusion of short DNA fragments to offer damage information. As a consequence, this method is simpler and more rapid than the comet assay. Sestili et al modified AHA to fast halo assay (FHA). 7 The behavior of single-stranded DNA fragments under the conditions of the FHA is essentially the same as in AHA. The modifications consist in the simplification of the lysis, denaturation, and staining procedures and allow, as compared with AHA, the preparation of samples within 15 minutes, with a two-third reduction in total processing time, using only 2 reagents to promote DNA extraction and staining, that is, sodium hydroxide and ethidium bromide.
Halo assay is used by limited researchers compared to comet assay because of lack of program/software for analysis of halo image. Most of the researchers analyze halo images manually using image analysis software (Scion Image or ImageJ). 7 –9 For popularizing halo assay, we have developed an ImageJ program (HaloJ) for analyzing halo images. This is a semiautomatic program that analyzed halo image in 2 steps and gave percentage of DNA in halo nuclei and nuclear diffusion factor (NDF). This program will be of great use to researchers working on the DNA damage and repair, radiation biology, toxicology, cancer biology, and so on.
Materials and Methods
Materials
High-melting point agarose, low-melting point agarose, NaOH, Na2-EDTA, Tris-base, and propidium iodide (PI) were obtained from Sigma-Aldrich Chemicals Inc (St Louis, Missouri). All other chemicals used were of analytical grade procured locally.
Treatment of Lymphocytes for Halo and Comet Assay
To perform halo and comet assay, splenic lymphocytes were obtained from Balb/c mice. For inducing DNA damage in these lymphocytes, cells were exposed to 2-, 4-, and 8-Gy γ-radiation, and the extent of the DNA damage was monitored 15 minutes after radiation exposure.
Preparation of Slides for Alkaline Halo and Comet Assay
In brief, frosted microscope slides (Gold Coin, Mumbai, India) were covered with 200 µL of 1% normal melting agarose in phosphate-buffered saline at 45°C, immediately coverslipped, and kept at 4°C for 10 minutes to allow the agarose to solidify. Removal of coverslip from the agar layer was followed by addition of a second layer of 200 µL of 0.5% low-melting agarose (LMA) containing approximately 105 cells at 37°C. Coverslips were placed immediately, and the slides were kept at 4°C. After solidification of the LMA, the cover slips were removed, and slides were placed in chilled lysing solution containing 2.5 mol/L NaCl, 100 mmol/L Na2-EDTA, 10 mmol/L Tris-HCl, pH 10, 1% dimethyl sulfoxide (DMSO), 1% Triton X100, and 1% sodium sarcosinate, for 1 hour at 4°C. The slides were removed from the lysing solution and incubated for 20 minutes in freshly prepared alkaline buffer (300 mmol/L NaOH, 1 mmol/L Na2-EDTA, and 0.2% DMSO, pH ≥13.0). After 20 minutes of alkali incubation, 1 set of the slides was removed for halo assay and another set was subjected to electrophoresis at 25 V for 15 minutes in alkaline buffer. Alkali of halo and comet assays was neutralized with 0.4 mol/L Tris-HCl buffer, pH 7.6, for 5 minutes twice. Finally, the samples were washed once with distilled water for 5 minutes. Slides were either used for image capturing or stored at 4°C in a humidified chamber. The slides were stained with 50 µL of PI (20 µg/ml) and visualized using a Carl Zeiss Fluorescent microscope (Axioskop) with bright field phase-contrast and epi-fluorescence facility. The images (˜50 cells/slides) were captured with high-performance MRC5 camera Carl Zeiss, Germany.
Image Processing Using HaloJ, an ImageJ Macro
For assessment of the areas and contents of DNA in halo nuclei, we developed an HaloJ macro for public domain image analysis software, ImageJ 1.47v (http://rsbweb.nih.gov/ij/download.html). This macro is a semiautomatic program that analyzed halo and gave percentage of DNA in halo nuclei and NDF. The macro convert selected image automatically into 32-bit black and white images followed by the Lookup Tables (LUT) to a given LUT image (eg, ICA, ICE, etc). The above-mentioned 2 steps take place automatically. After these steps, 2 action commands pop-up sequentially: (step 1) draw a circle around the Halo Nucleus; Click OK direct to (step 2) draw a circle around the Halo. This completes analysis of a single halo image. Pressing Click OK diverts to next pop-up, Next Halo? If there are several images then let YES checked and press OK. Continue the above-mentioned steps until all halos of the opened windows were analyzed. For analyzing new image file, uncheck YES of NEXT Halo? Pop-up and press OK. This will direct to NEXT IMAGE? Pop-up. Press OK to open new image for analysis. Follow the instructions until all images are analyzed. To understand macro steps, a flowchart is provided in Scheme 1. Results of this analysis will pop-up in the Result window. The result window shows halo parameters such as percentage of halo nuclei DNA and NDF along with other information about the images. The intensities of fluorescence signals are proportional to the amount of PI binding to DNA and also proportional to the amount of DNA (because DNA is stained randomly with this dye). The level of DNA damage can be quantified with NDF, which can be derived from surface areas of halo and nucleus as follows:
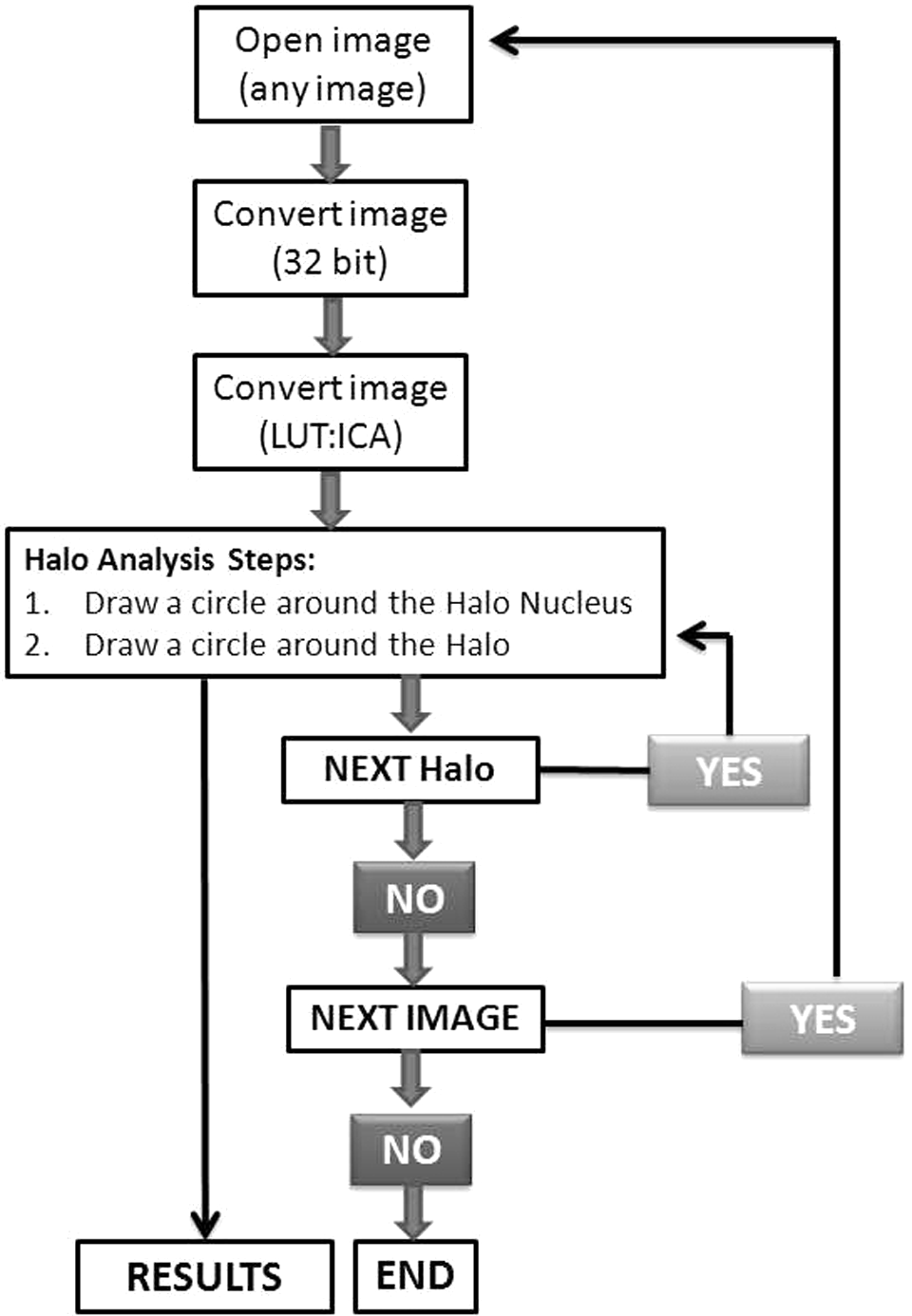
A flow chart of sequential operation in HaloJ program.
This result can be saved as .txt file and can be imported to excel for further analysis.
Analysis of Comet Assay Images
The quantification of the DNA strand breaks of the stored comet images was done using the Comet Assay Software Project (CASP) software by which percentage of DNA in tail, tail length, tail moment, and Olive tail moment could be obtained directly. 10
Statistical Analysis
The graphs were plotted between mean ± standard error of mean.
Results and Discussion
There are several methods available for measuring DNA damage. Each technique has its inherent drawbacks and also specific advantages. Comet assay is a well-known assay to measure toxicity induced by various stressors. Halo assay is an alternative assay for measuring the DNA damage. There are different types of comet and halo assays. Alkaline comet assay is the most common comet assay, which is used to measure total DNA damage (DNA double- and single-strand breaks). The AHA has some analogies with alkaline comet assay but is based on a different principle. The most important difference is that AHA does not use electrophoresis to separate damaged DNA from undamaged DNA, but a short postlysis incubation in an alkaline buffer is needed. Sestili et al improved the AHA to FHA with the same reliability and sensitivity but with additional advantages such as inexpensive, more rapid, and noticeably simple. 7 Despite all these advantages, halo assay is less known than comet assay because of lack of suitable program that researchers can use to analyze their images. To give a momentum to halo assay, we developed an ImageJ-based program (HaloJ) for the analysis of halo images. This is a semiautomatic program that analyzes halo image in 2 steps and gives percentage of DNA in halo nuclei and NDF. To validate this program, we compared the results obtained after performing halo assay (analyzed by HaloJ software) with the results of comet assay (analyzed using CASP software). 10
Our study shows that exposure of lymphocytes to γ-radiation leads to a dose-dependent decrease in the percentage of DNA in halo nuclei and increase in NDF of the halo images (Figure 1). Similar results were obtained from alkaline comet assay (Figure 2). Further halo parameters (percentage of DNA in halo nuclei and NDF) were correlated with the comet parameters (percentage of DNA in tail and Olive tail moment). Our observation shows that halo parameters significantly correlated with the comet parameters (Figure 3). This program is developed for research purposes only and not intended for regulatory use. To the best of our knowledge, this is the first program for the semiautomatic analysis of halo images. This program is free (a copy of the program can be obtained by sending mail to
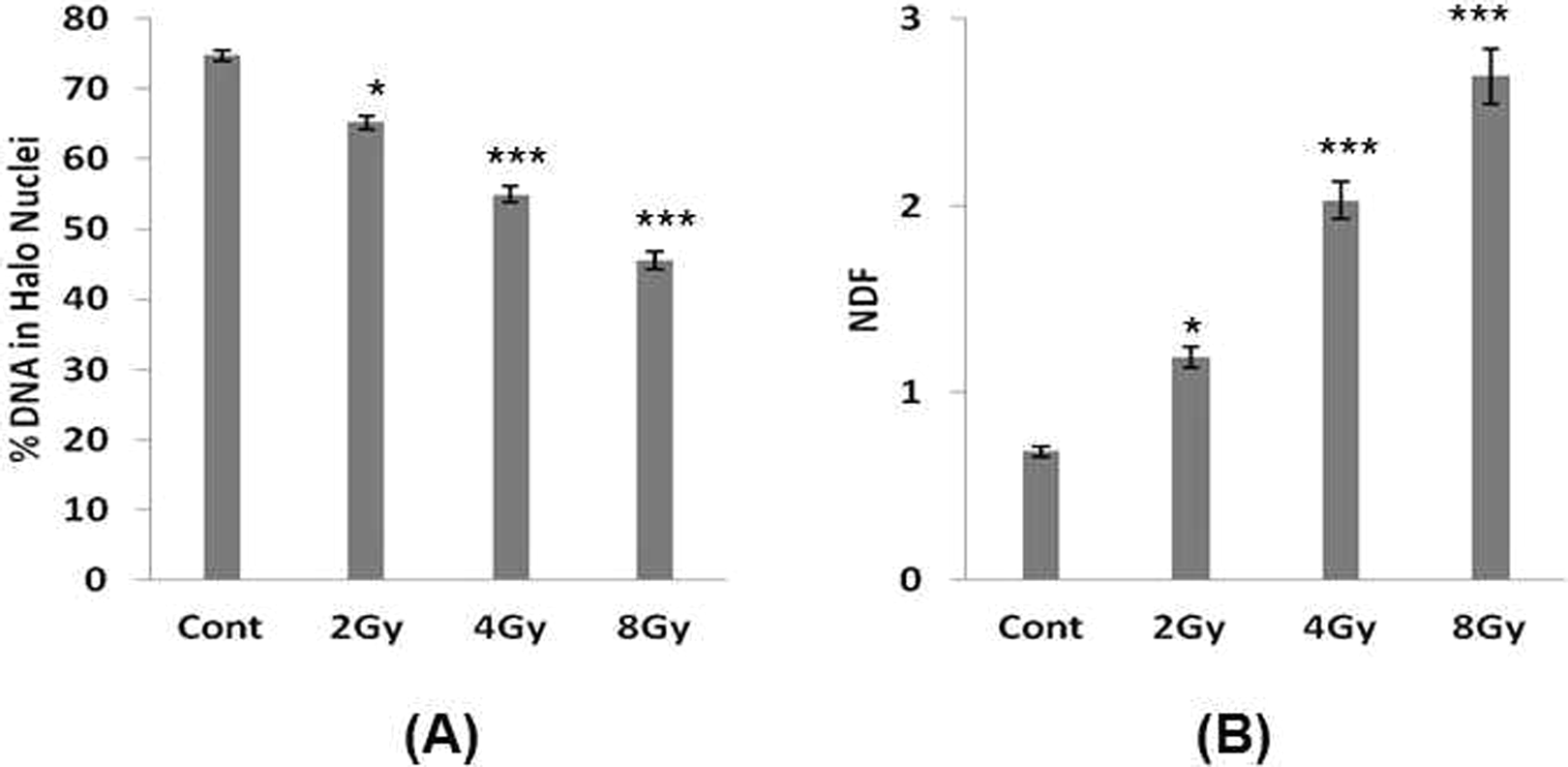
Measurement of radiation-induced DNA damage in lymphocytes using halo assay, and halo images were analyzed using HaloJ program. A, Percentage of DNA in halo nuclei and (B) nuclear diffusion factor (NDF). Each bar represents mean ± standard error of mean (SEM) obtained by quantitating ˜50 images from halo assay. Three independent experiments were performed. Data are significant at *P < 0.05, ***P < 0.001 compared to control.
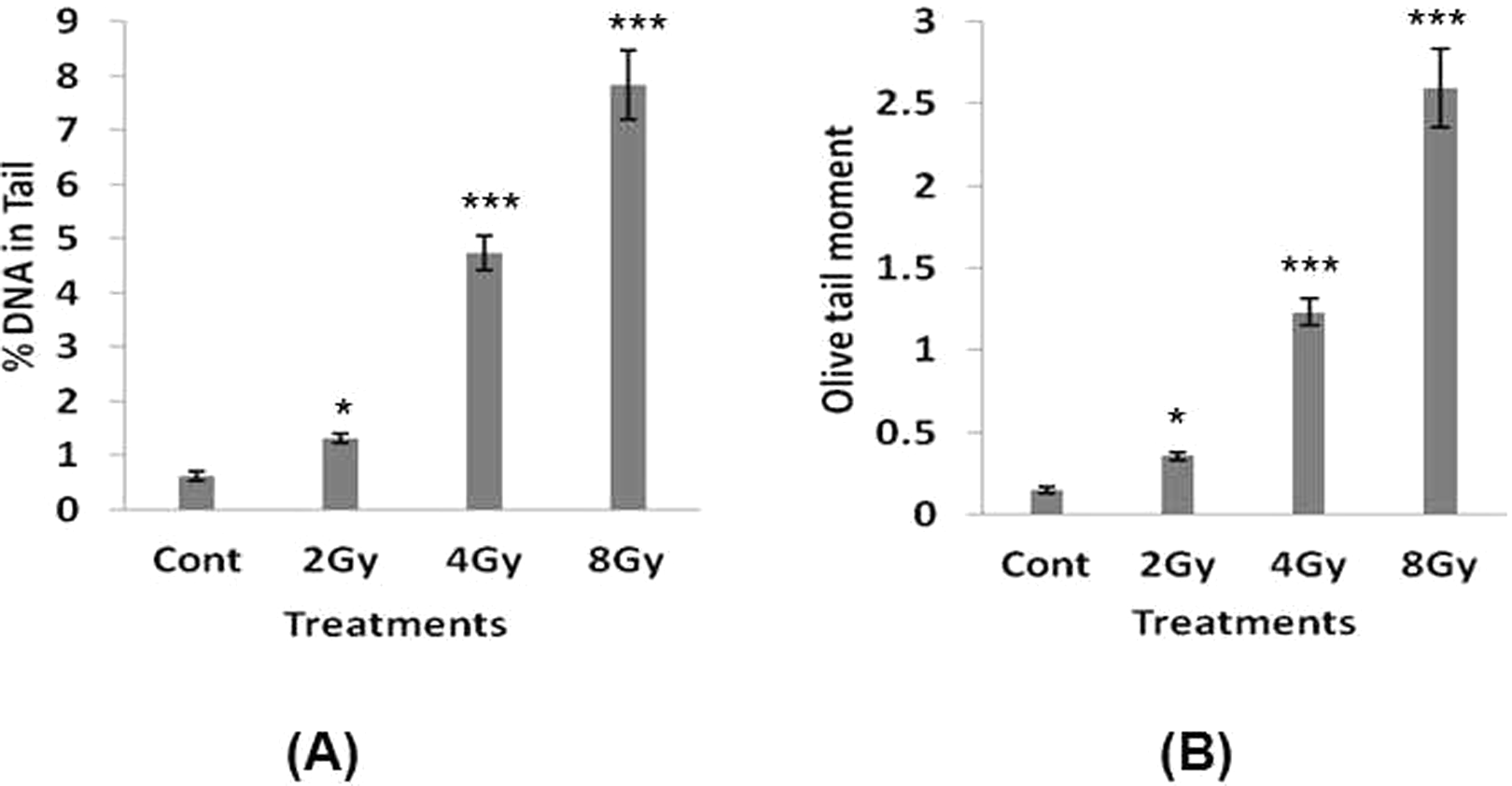
Measurement of radiation-induced DNA damage in lymphocytes using comet assay, and the images were analyzed using Comet Assay Software Project (CASP) program. A, Percentage of DNA in tail and (B) Olive tail moment (OTM). Each bar represents mean ± standard error of mean (SEM) obtained by quantitating ˜50 images from comet assay. Three independent experiments were performed. Data are significant at *P < 0.05, ***P < 0.001 compared to control.
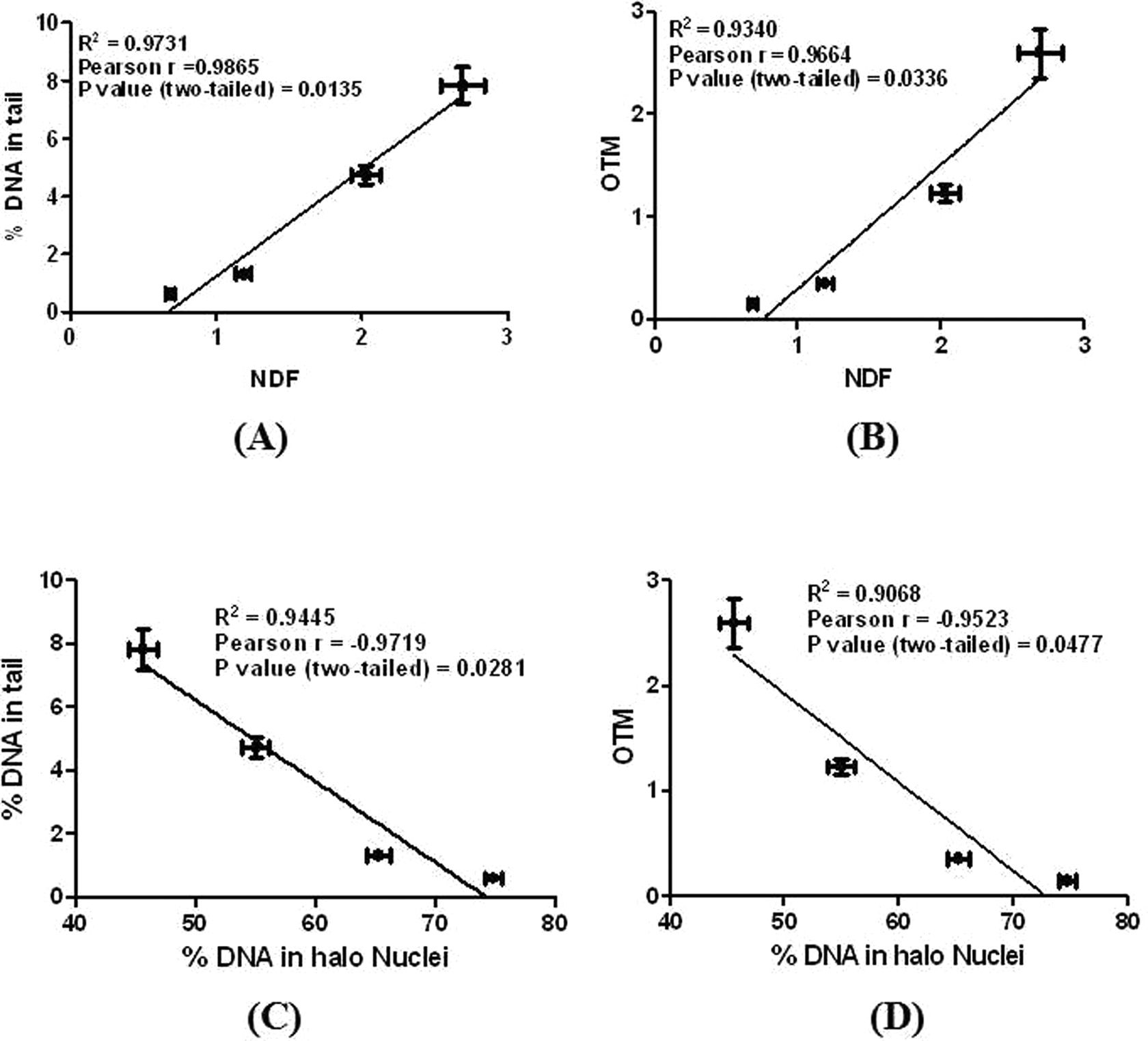
Correlation between results obtained from HaloJ analysis and Comet Assay Software Project (CASP) analysis. A, Correlation between percentage of DNA in tail and NDF, (B) correlation between OTM and NDF, (C) correlation between percentage of DNA in tail and Percentage of DNA in halo nuclei, and (D) correlation between OTM and percentage of DNA in halo nuclei. NDF indicates nuclear diffusion factor; OTM, Olive tail moment.
Footnotes
Acknowledgments
Authors would like to thank Dr S. Santosh Kumar, Radiation Biology & Health Sciences Division, Bhabha Atomic Research Centre for critical review of the article. I also thank Mr P. P. K. Venkata, Computer Division, Bhabha Atomic Research Centre, for his suggestions during writing the program.
Author Contribution
Dharmendra Kumar Maurya was involved in entire experiments planing, writing program, performing experiments and writing the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.