Abstract
The explosive 3-nitro-1,2,4-triazol-5-one (NTO) is an insensitive formulation developed to replace high energetics that are susceptible to accidental detonation from heat, shock, and impact. Although studies have shown NTO to be nontoxic at acute exposures, recent subacute and subchronic tests have demonstrated effects on testes and subsequent sperm production in rats. This study assessed endocrine disruption as a potential mechanism for these reproductive effects via the Hershberger and uterotrophic bioassays. These assays are 2 of the US Environmental Protection Agency’s tier 1 in vivo screens for the Endocrine Disruptor Screening Program that measure differences in androgen- and estrogen-sensitive tissue weights in castrated and ovariectomized rats. The gonadectomized rats were orally exposed to NTO in a corn oil vehicle at doses of 250, 500, or 1000 mg/kg body weight (bw)/d for 10 and 3 days for the Hershberger and uterotrophic assays, respectively, according to standard protocols. Male rats also received testosterone (0.2 mg/kg/d, subcutaneous) and antiandrogenic flutamide (3mg/kg/d, oral) as negative and positive controls, and females received 17 α-ethynyl estradiol (0.3 µg/d, subcutaneous) as positive controls. 3-Nitro-1,2,4-triazol-5-one caused neither a decrease in androgen-sensitive male reproductive selected tissue (seminal vesicles with fluid/without fluid, glans penis, Cowper gland, ventral prostrate, and levator ani–bulbocavernosus) weights nor a change in uterine weights. The results of this study provide no evidence to suggest that NTO acts like an estrogenic or antiandrogenic endocrine disruptor in rats at these doses.
Introduction
The energetic compound 3-nitro-1,2,4-triazol-5-one (NTO) was developed by the Los Alamos National Laboratory in 1984. 1 The danger inherent to propellants and explosives, such as 2,4,6-trinitrotoluene (TNT) and hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX), that are susceptible to accidental detonation from heat, shock, and impact drove the development of insensitive and more stable replacements such as NTO and 2,4-dinitroanisole. 3-Nitro-1,2,4-triazol-5-one showed promise as a replacement due to its detonation velocity and pressure being similar to those of RDX and its ability to be synthesized safely and inexpensively. 1 Toxicity has also been a major issue for widely used explosives like TNT and RDX, particularly at production and training sites. 2 Human and environmental health have been top priorities for all potential energetic replacements, and low toxicity is required prior to consideration for mass production.
The water solubility of NTO was determined to be 2.0 g/L3 (at 20°C). The high water solubility and potential for environmental persistence
3
suggest the potential for ground water contamination. Few environmental toxicity tests have been completed to date for NTO. An aquatic toxicity test that reported the resulting inhibitory concentration at 20% (IC20) and IC50 values for reproduction of
The Hershberger Office of Chemical Safety and Pollution Prevention (OCSPP) 890.1400) (OCSPP 890.1400) 7 and uterotrophic (OCSPP 890.1600) 8 bioassays are 2 of the 11 in vitro and in vivo tier 1 tests from the US Environmental Protection Agency’s Endocrine Disruptor Screening Program. The first tier consists of a battery of complementary and less complex tests that screen chemicals for interactions along the estrogen, androgen, and thyroid hormonal pathways through various modes of action that include receptor binding, steroidogenesis, and hypothalamic–pituitary–gonadal and hypothalamic–pituitary–thyroidal feedback. If results from the tier 1 battery demonstrate that a chemical does show potential to interact with the tested hormonal systems, additional testing would be required with tier 2 tests. The objective of this study was to test the antiandrogenic and estrogenic potential of NTO through 2 tier 1 tests, the Hershberger and uterotrophic bioassays, respectively.
Materials and Methods
Test Substances
3-Nitro-1,2,4-triazol-5-one (CAS No. 932-64-9; lot 11L375-061) was produced by BAE Systems, Ordnance Systems (Kingsport, Tennessee). Purity was determined to be 99.4%. 9 Reference agents were obtained from Sigma-Aldrich (St Louis, Missouri). All dosing suspensions/solutions were prepared by weighing the required amount of test substance and measuring the required volume of corn oil. 3-Nitro-1,2,4-triazol-5-one was ground to a fine consistency for optimal uniform suspension using a mortar and pestle after first being wetted with a small amount of the measured corn oil. After the dosing suspensions/solutions were mixed, samples were analyzed for concentration verification and homogeneity of the NTO suspension by determining the concentration of samples taken from the top, middle, and bottom of the container. Prior tests had determined NTO to be stable in corn oil for at least 6 weeks at room temperature. 9 All dosing solutions were stored at 40°F and brought to room temperature while mixed on a stir plate for 1 hour prior to dosing and throughout the dosing procedure.
Animal Husbandry
All Sprague-Dawley rats were obtained from Charles River Laboratories, Wilmington, Massachusetts. Gonadectomies were performed by the vendor. All animals were housed in rooms with allowable temperature and humidity ranges of 68°F to 79°F and 30% to 70%, respectively. An automatically controlled 14:10-hour light/dark cycle was maintained, with the dark period beginning at 1900 hours. Feed (Harlan Teklad, 2020XC Certified Rodent Diet Harlan Laboratories, Frederick, MD, USA) and water were available ad libitum. All rats were pair-housed in the same sex groups and were housed in suspended polycarbonate cages with ALPHA-dri bedding (Shepherd Specialty Papers, Elkridge, MD, USA). Individual rats were uniquely identified by number via cage card and tail markings.
Animal use procedures were approved by the US Army Public Health Command (USAPHC) Institutional Animal Care and Use Committee. Animal care and use was conducted in accordance with
Hershberger Bioassay
The Hershberger bioassay was conducted according to standard guidelines (OCSPP 890.1400). 7 Due to the results of the previous studies where NTO caused reductions in testes weights and sperm counts, 6,9 only the antagonist component was conducted; the agonist component was excluded in order to reduce unnecessary animal use. This assay is a short-term screening test that uses accessory tissues of the male reproductive tract to test chemicals for potential androgen activity. Sprague-Dawley rats were castrated on postnatal day (pnd) 42. On pnd 58, rats were assigned to 1 of the 3 dose groups by randomized weight distribution (6 rats per dose group): a corn oil vehicle control group, 3 dose groups of NTO (250, 500, and 1000 mg/kg body weight (bw)/d plus testosterone propionate (TP; CAS No. 57-82-5), TP only group (negative control), and TP plus flutamide group (positive control). This dose range was selected based on the maximum recommended dose of 1000 mg/kg/d and the results of a 90-day NTO toxicity study performed by PHC where adverse testes effects were observed at ≥500 mg/kg bw/d. 7 Rats from the control groups received the vehicle in the highest volume used with the dosed groups. Testosterone propionate, the reference androgen agonist, was given via subcutaneous injection at 0.2 mg/kg bw/d. Flutamide (CAS No. 1311-84-7), the reference antagonist, was administered at 3 mg/kg bw/d. Rats were exposed to flutamide and NTO by oral gavage. The subcutaneous injections were given to the dorsoscapular region and the maximum injection volume did not exceed 5 mL/kg bw. Rats were dosed for 10 consecutive days at approximately 24-hour intervals. Rats were euthanized on day 11, and body weight and weights of the seminal vesicles (SVs), ventral prostrate, levator ani-bulbocavernosus (LABC) muscle, Cowper glands (COWs), glans penis (GP), liver, paired kidneys, and adrenal glands were measured and recorded. One prosector completed all tissue removals as recommended by the standard guidelines.
Uterotrophic Bioassay
The uterotrophic assay was conducted according to standard guidelines (OCSPP 890.1600). 8 This assay is a short-term screening test used to identify substances with estrogen-active potential. All female Sprague-Dawley rats were ovariectomized pnd 42. The rats had a 5-day acclimation period upon arrival to PHC. They were vaginally lavaged with saline water for 5 consecutive days starting on pnd 56. Epithelial cells were analyzed for the presence of any possible remaining ovarian tissue. No ovarian cells were found, indicating that ovariectomy was complete. No indications of estrus were observed. Dosing began on pnd 61.
Per OPPTS 890.1600 guidelines, a separate group of rats were administered 17α-ethynyl estradiol (EE; CAS No. 57-63-6), a reference estrogen, for determination of the EE dose to be used in the uterotrophic assay and for the verification of laboratory proficiency (dissection skills and animal model responsiveness) prior to the start of the actual assay. Of the 4 doses of EE tested to produce a uterine weight response (0.0005, 0.00075, 0.001, and 0.003 mg/kg bw/d), 0.003 mg/kg bw/d was selected as the uterotrophic assay’s reference dose because it gave the most consistent increase in uterine wet and dry weights. 17α-Ethynyl estradiol was given via subcutaneous injection in the dorsoscapular region, and NTO was administered suspended in corn oil by oral gavage for 3 consecutive days. The treatment levels of NTO included 250, 500, and 1000 mg/kg bw/d. Although the guidelines recommend that the test chemical be given by subcutaneous injection, the large volume needed to keep the highest dose of NTO in uniform suspension, exceeding the maximal subcutaneous injection volume. Oral gavage volumes did not exceed 5 mL/kg bw. The control group received the highest volume used with 1000 mg/kg/d NTO test group. The sample size for each dose group was 6 individuals, and no individual’s body weight exceeded 20% of the mean weight. Rats were assigned to groups by randomized weight distribution.
Euthanasia and necropsies were performed on pnd 64. Before dissection, the vagina was examined for opening status in immature animals. One prosector completed all tissue removals as recommended by the standard guidelines. The dissection procedure began by opening the abdominal wall starting at the pubic symphysis. Then, uterine horn and any ovarian tissue, if present, were detached from the dorsal abdominal wall. The urinary bladder and ureters were removed from the ventral and lateral side of uterus and vagina. Fibrous adhesion between the rectum and the vagina was detached until the junction of vaginal orifice and perineal skin can be identified. The uterus and vagina were detached from the body by incising the vaginal wall just above the junction between perineal skin. The uterus was detached from the body wall by gently cutting the uterine mesentery at the point of its attachment along the full length of the dorsolateral aspect of each uterine horn. The vagina was removed from the uterus just below the cervix so that the cervix remained with the uterine body. Each uterus with luminal fluid was weighed to the nearest 0.1 mg (wet uterine weight). Both uterine horns were cut longitudinally, and the uterus was placed on lightly moistened filter paper and gently pressed with a second piece of lightly moistened filter paper to completely remove the luminal fluid. The uterus without the luminal contents (blotted uterine weight) was weighed to the nearest 0.1 mg.
Statistical Analysis
Analysis of variance (ANOVA) tests were used to determine whether the average terminal body and organ weights of the treatment groups were significantly different. An F-statistic and corresponding
For the Hershberger bioassay, the TP group was the control. The 5 androgen-dependent tissues included in the test were the SV (plus fluids and coagulating glands), LABC, ventral prostate, COW, and the GP. The terminal body weight and weights of paired adrenal glands, paired kidneys, and liver were also examined. Per the guidance, because toxicity may decrease the body weight of NTO-treated groups relative to the control group, the starting body weight could be used as a statistical covariate in the analysis. This was done separately from the main analysis.
For the uterotrophic assay, the vehicle-only group was the control. Both the wet and blotted uterine weights were compared to the control. In addition, terminal body weight and the weights of the paired adrenal glands, paired kidneys, and liver were also examined. Per the guidance, the terminal body weight could be used as a statistical covariate in the analysis. This was done separately from the main analysis.
Results
The only difference in androgen-sensitive tissue weights that was observed from the Hershberger assay was the mean GP weight from the 500 mg/kg/d group being higher than that of the TP positive control group (Table 1) after removal of an outlier. Mean GP weight from the 1000 mg/kg/d treatment group was not different than that of the positive control group. As expected, androgen-sensitive tissue did increase in weight in rats exposed only to the androgen agonist TP, and these differences were not apparent in rats that were exposed to TP concurrently with the reference antagonist flutamide. No differences in terminal body weight or nonandrogen-sensitive tissue weights were observed (Table 2).
Effects of 3-Nitro-1,2,3-triazol-5-one (NTO) on Androgen-Sensitive Tissue Weights (g) From Castrated Sprague-Dawley Rats.
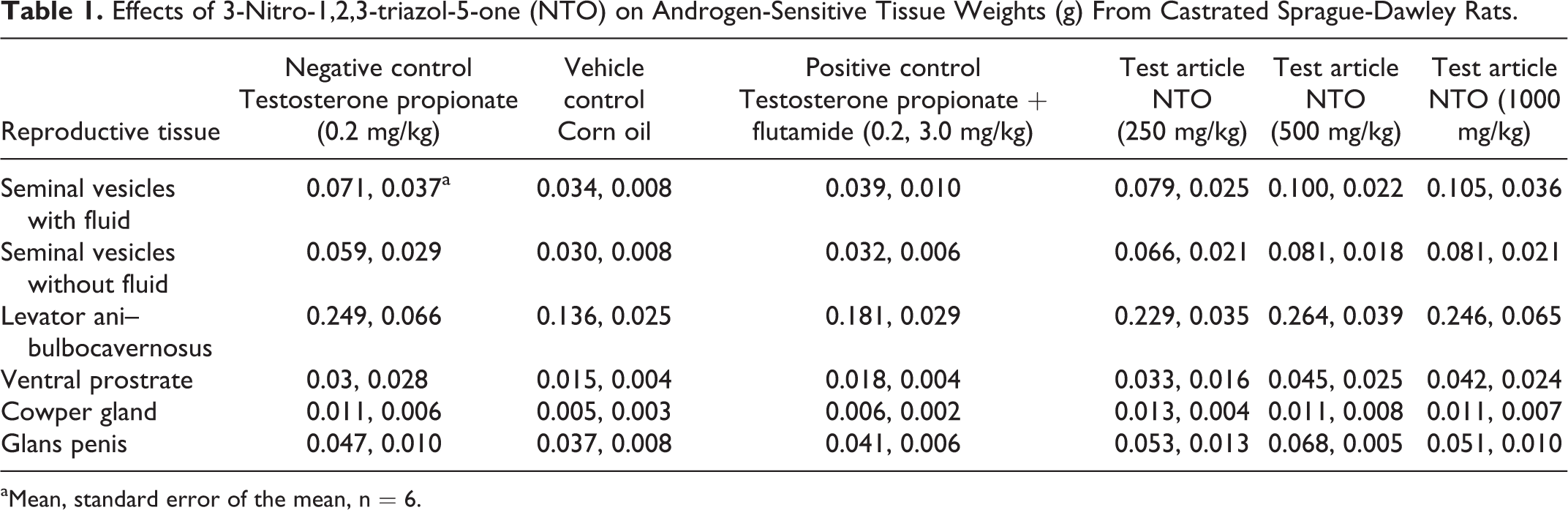
aMean, standard error of the mean, n = 6.
Effects of 3-Nitro-1,2,3-triazol-5-one (NTO) on Body and Organ Weights (g) From Castrated Sprague-Dawley Rats.
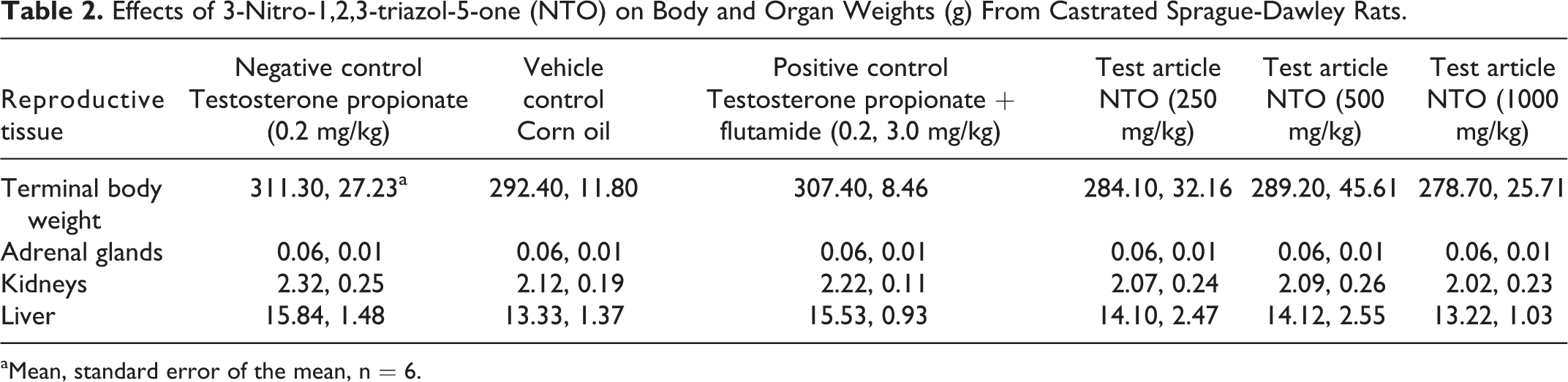
aMean, standard error of the mean, n = 6.
3-Nitro-1,2,4-triazol-5-one did not cause changes in uterine weight in the uterotrophic assay (Table 3). As expected, uterine weights did increase in rats exposed to the reference agonist ethynyl estradiol. The only difference that was observed in this bioassay was mean liver weight from the 1000 mg/kg/d dose group being lower than that of the control group (Table 4).
Effects of 3-Nitro-1,2,3-triazol-5-one (NTO) on Uterine Weights (g) From Ovarectomized Sprague-Dawley Rats.
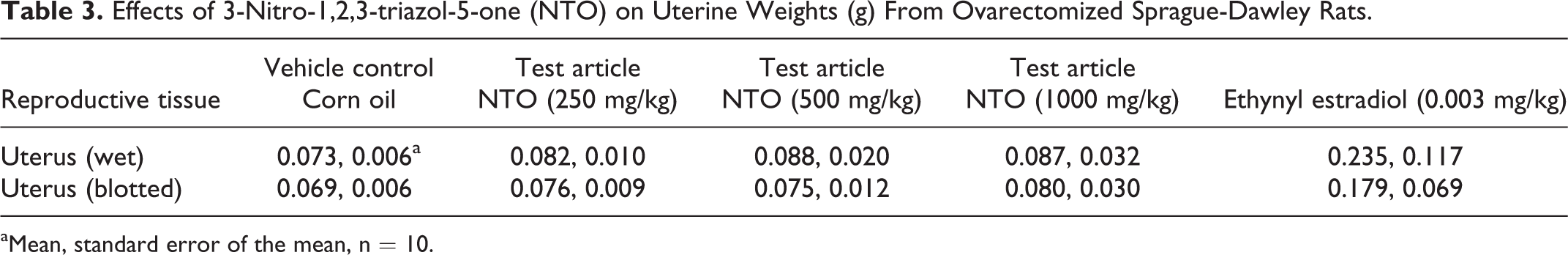
aMean, standard error of the mean, n = 10.
Effects of 3-Nitro-1,2,3-triazol-5-one (NTO) on Body and Organ Weights (g) From Ovarectomized Sprague-Dawley Rats.
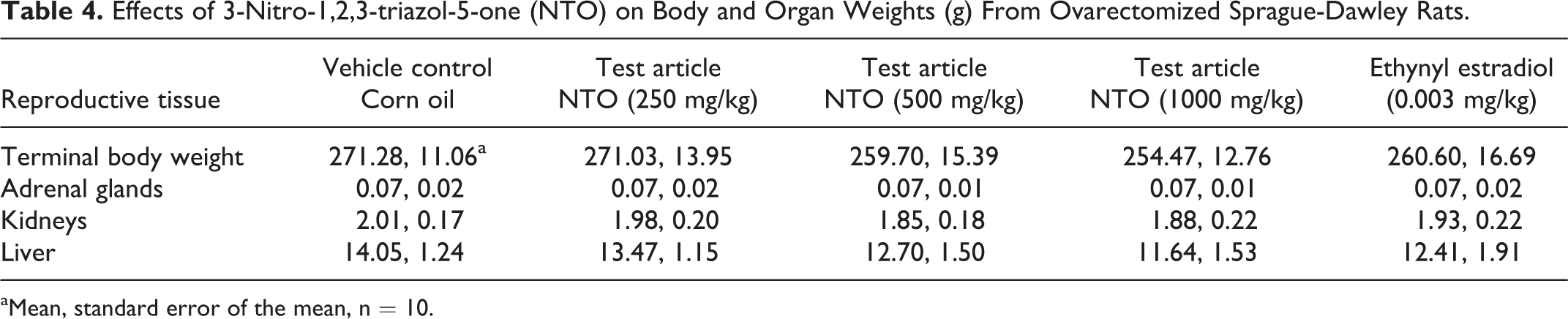
aMean, standard error of the mean, n = 10.
Discussion
A previous study identified the testes and epididymides as potential targets of NTO in rats having been subacutely and subchronically exposed via daily oral gavages, 3 which caused NTO to be considered as a potential endocrine disruptor. In the subacute exposure (14 days), testes weights in the 500, 1000, 1500, and 2000 mg/kg/d dose groups were smaller than those from the 250 mg/kg/d and control groups. Epididymal weight was also reduced in the 1500 and 2000 mg/kg/d dose groups compared to controls. In the subchronic study (90 days), testes and epididymides weights were reduced in the 315 and 1000 mg/kg/d treated rats. Moderate to severe testicular hypoplasia, characterized by interstitial degeneration and loss of spermatogenic epithelium in the seminiferous tubules, was observed in the testes in 66% and 100% of males from the 315 and 1000 mg/kg/d dose groups, respectively. Epididymal aspermia was also observed at these dose levels.
The antagonist component of the Hershberger bioassay was conducted to test the hypothesis of NTO being a possible antiandrogenic endocrine disruptor. By castrating the male rats in the current study, the endogenous source of testosterone was removed. The animals were then supplemented with TP injections to reconstitute normal organ development. If the test compound acted as an androgen antagonist, androgen-sensitive tissue weights in these rats would have been reduced compared to the rats from the TP-only group. The only change observed in the male rats was that mean GP weight from the 500 mg/kg/d group was higher, not lower, than that of the TP positive control group. This was not a dose-dependent change as the mean GP weight from the 1000 mg/kg/d treatment group was no different than that of the positive control group. Additionally, the guidelines 8 state that a statistically significant change in any 2 or more of the 5 required androgen-dependent tissue weights is indicative of a positive result; again, this was the only change observed in the male androgen-dependent tissues.
In female rats, estradiol is needed for uterine development. The endogenous estrogen source was removed upon ovariectomy, and EE was administered to reconstitute normal uterine development in the positive control rats. If NTO acted like an estrogenic endocrine disruptor, uterine weights would have increased relative to the control rats. No changes in uterine weights were observed. In a previous pubertal development study, 9 NTO also failed to cause changes in reproductive development parameters in Sprague-Dawley; age and body weight at vaginal opening and measures of estrous cyclicity (age at first estrus, cycle length, percentage of cycling, and percentage of cycling regularly) were not affected by treatment with NTO at 500 and 1000 mg/kg/d. Age and body weight at preputial separation also remained unchanged.
Five in vitro bioassays had recently tested NTO’s potential as an endocrine disrupting chemical (estrogen receptor binding, androgen receptor binding, estrogen transactivation, aromatase, and steroidogenesis), and no effects were observed. 10 Together with the results of the current study, this lack of effects suggests that the changes in testes and sperm observed in the 90-day study were not due to NTO acting as an antiandrogenic or estrogenic endocrine disrupting chemical. Unfortunately, any thoughts as to a potential mechanism behind the gonadal regressions would be merely conjecture at this point. New mechanistic studies are currently underway to test whether the testes are a specific target of NTO toxicity. Also, while it is still possible that NTO might have targeted a more distal part of the neuroendocrine system responsible for maintaining reproductive organ function, the impairment also could have been due to a more general toxic effect. By eliminating the energy required to maintain a system not required for individual survival, that energy can be allocated to activities involved with the metabolism and elimination of a toxic substance and compensatory mechanisms involved with enhancing survivorship. Whatever the mechanism of toxicity may be, if the reproductive effects are severe enough they may affect human male fertility rates. In wildlife, reproductive effects could come at the price of an individual’s reduced environmental fitness and also potentially at the price of a population’s propagation.
This study has shown NTO to be neither antiandrogenic nor estrogenic via its lack of effects on androgen and estrogen-sensitive gonadal tissue. Additional studies are needed to elucidate a mechanism behind the gonadal regression observed in the subacute and subchronic NTO exposure studies.
Footnotes
Author Contributions
Quinn contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Bannon contributed to conception, interpretation, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Jackovitz contributed to conception and design, acquisition and analysis, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Hanna and Shifflet contributed to design, acquisition, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. Johnson contributed to conception and design, acquisition, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the US Army Research Development and Engineering Command (RDECOM) Environmental Acquisition and Logistics Sustainment Program (EASLP), Aberdeen Proving Ground, MD 21010.
