Abstract
PEGylated oil is a terminology used to describe cosmetic ingredients that are the etherification and esterification products of glycerides and fatty acids with ethylene oxide. The Cosmetic Ingredient Review Expert Panel (Panel) considered the safety of PEGylated oils, which function primarily as surfactants in cosmetic products. The Panel reviewed relevant animal and human data provided in this safety assessment and concluded that the 130 chemically related PEGylated oils were safe as cosmetic ingredients in the present practices of use and concentration when formulated to be nonirritating.
Keywords
Introduction
In 1997, the Cosmetic Ingredient Review (CIR) published the safety assessment on polyethylene glycol (PEG)-30, -33, -35, -36, and -40 castor oil and PEG-30 and -40 hydrogenated castor oil with the conclusion “PEG-30, -33, -35, -36, and -40 castor oil are safe for use in cosmetics at concentrations up to 50% and that PEG-30 and -40 hydrogenated castor oil are safe for use at concentrations up to 100%.” 1 Because there are a large number of oils in this group and multiple PEG chain lengths, the shorthand terminology “PEGylated oils” is used to describe cosmetic ingredients that are the etherification and esterification products of glycerides and fatty acids with ethylene oxide.
Since the original review, numerous additional studies were published related to the noncosmetic use of PEG-35 castor oil and PEG-40 hydrogenated castor oil (trade names Cremophor EL and Cremophor RH, respectively) in drug delivery systems. The few of these studies that appear relevant to assessing the safety of the group of PEGylated oils in cosmetics are summarized in this safety assessment. Overall, single-dose and repeated-dose toxicity, reproductive and developmental toxicity, genotoxicity, carcinogenicity, dermal and ocular irritation, and sensitization and photosensitization data were available.
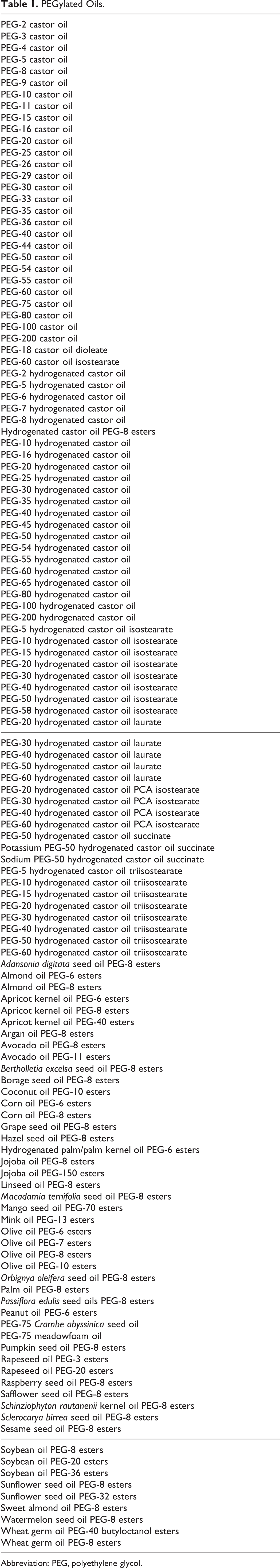
Because of similarities in chemical structure and cosmetic function, the PEGylated oils listed in Table 1 have been added to the safety assessment of PEG-30 castor oil to comprise a group of 130 cosmetic ingredients. These cosmetic ingredients include components that have been previously reviewed and concluded to be safe for use by the CIR Expert Panel, most notably the recent safety assessments on plant-derived fatty acid oils, PEGs with an average of 4 moles of ethylene oxide or greater, and alkyl PEG ethers. The ingredients, their conclusions, a summary of the findings, and published citations are found in Table 2.
PEGylated Oils.
Abbreviation: PEG, polyethylene glycol.
Summaries of Previous CIR Safety Assessments.

Abbreviations: aq, aqueous; CIR, Cosmetic Ingredient Review; LD50, median lethal dose; PEG, polyethylene glycol; UV, ultraviolet.
Chemistry
The definitions of the PEGylated oils can be found in Table 3.
Names, CAS Registry Numbers, and Definitions of the PEGylated Oil Ingredients.

Abbreviations: CIR, Cosmetic Ingredient Review; PEG, polyethylene glycol.
Just as oils and other PEGylated materials are mixtures, PEGylated oils are also mixtures. As most natural source oils are primarily triglycerides (and mono- and diglycerides) and fatty acids, PEGylated oils are primarily PEGylated glycerides, along with some PEGylated fatty acids. PEGylation of glycerides occurs not only as an etherification of the free alcohol groups of the glycerides with ethylene oxide groups but also as a transesterification, which results in net insertion of PEG groups between the glyceryl and fatty acid components of the glyceride. 2,3 For example, the primary component of castor oil, the ricinoleate triglyceride, is ethoxylated as shown in Figure 1, wherein n is equal to the number of ethylene oxide repeat units and need not be the same at all places of the molecule. It should be noted that n is not equal to X. In other words, wherein the value of X in PEG-X is equal to 2 (eg, PEG-2 castor oil), n is not equal to 2. Instead, X represents the number of stoichiometric equivalents of ethylene oxide that were added to one stoichiometric equivalent of castor oil. Therefore, the sum of all of the different n values in the mixture may be no more than X. Indeed, when 1 mole of ethylene oxide is reacted with 1 mole of fatty alcohol, adducts having no added ethylene oxide are the predominate material in the mixture. 2 Furthermore, when ethylene oxide reacts with castor oil it is approximately twice as likely that it will react at an ester site versus an alcohol site. Moreover, a percentage (13% in one specific case) of the ethylene oxide simply reacts with other molecules of ethylene oxide, resulting in some PEGs unattached to glycerides or acid groups.
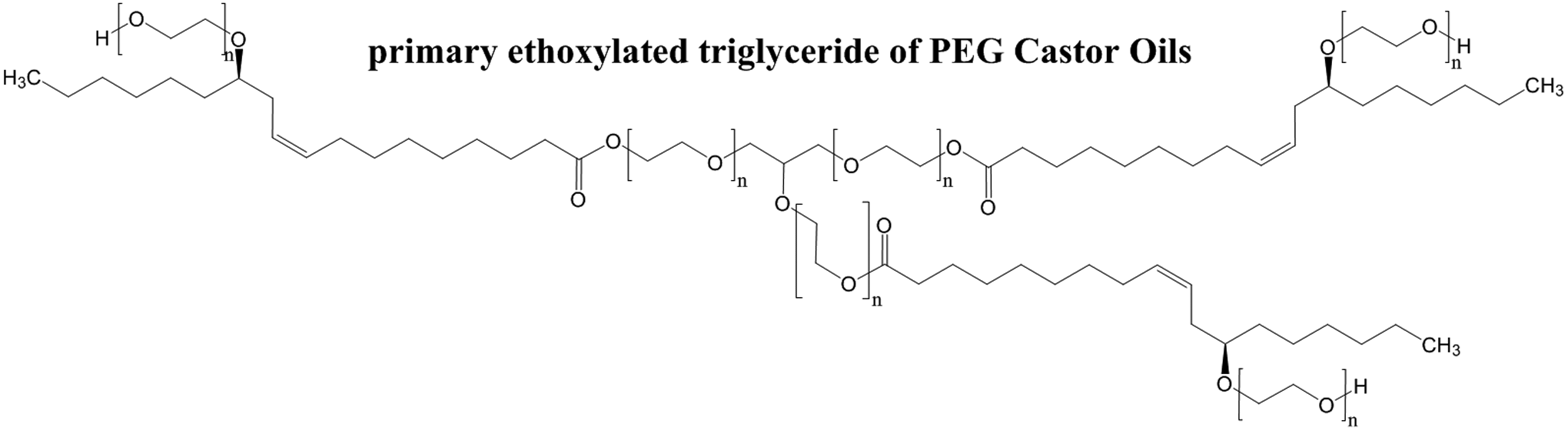
Glyceryl triricinoleyl polyethylene glycol.
While castor oil triglycerides are primarily (approximately 87%) composed of ricinoleic acid residues, approximately 7% are oleic acid, 3% are linoleic acid, 2% are palmitic acid, 1% are stearic acid, and a trace are dihydroxysteric acid. 1 Thus, these PEGylated castor oil ingredients, and all of the PEGylated oil ingredients, are rather complex mixtures of structurally related molecules.
The available free fatty acids found in castor oil, and the other oils, may also be esterified by the ethoxylation process, as seen in Figure 2 (and etherified with ethylene oxide groups if there are any reactive alcohol functionalities on the fatty acids).
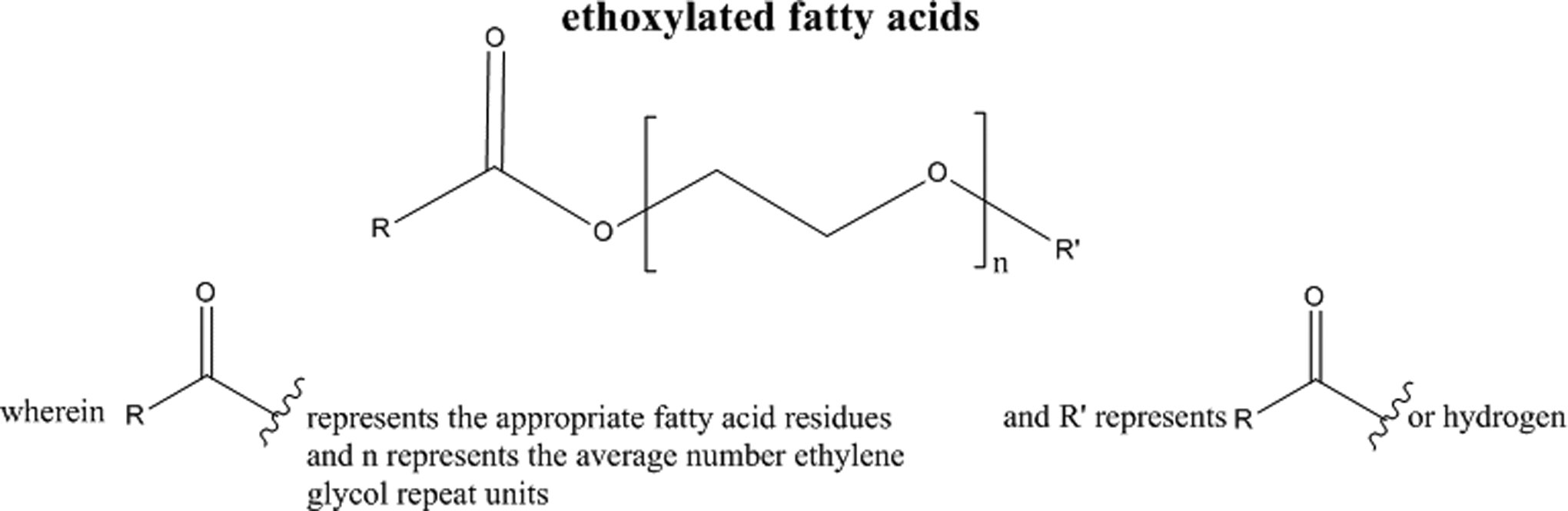
Fatty acid esterification.
Some of the ingredients in this report have been hydrogenated. For example, hydrogenation of castor oil primarily results in the reduction of the Ω-9 unsaturation of ricinoleate triglycerides (and the Ω-9 unsaturation of any free ricinoleic fatty acids). 2 Accordingly, hydrogenated castor oil is principally 12-hydroxystearic triglyceride. The resultant ethoxylated triglyceride, therefore, differs from that of PEGylated nonhydrogenated castor oil only in the loss of these double bonds, as seen in Figure 3.
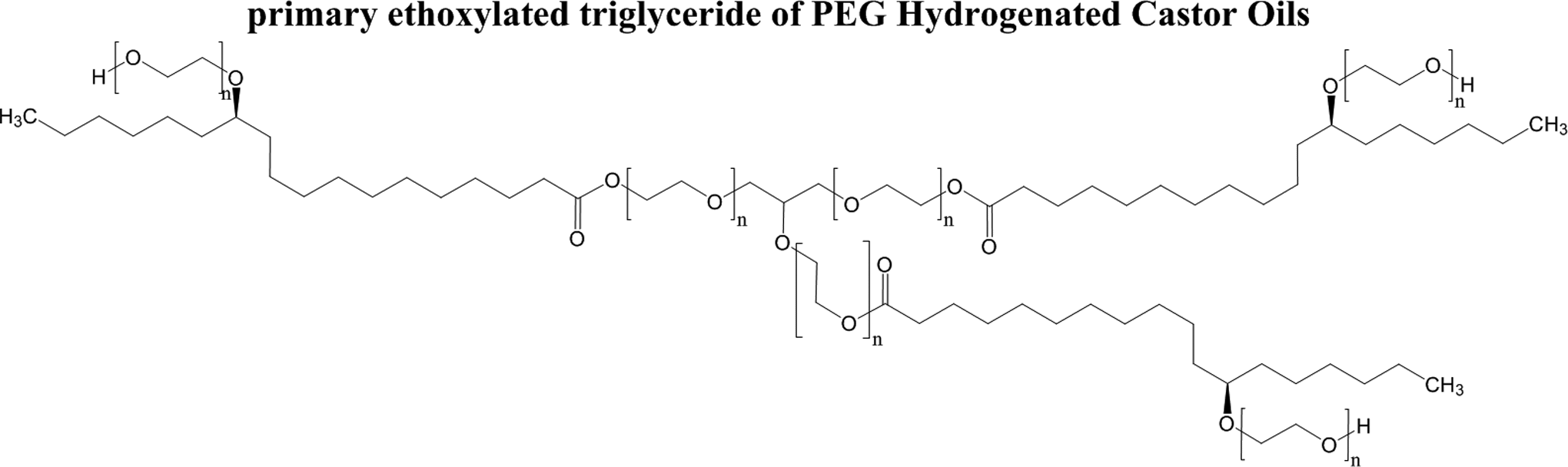
Glyceryl 12-hydroxystearyl polyethylene glycol.
Physical and Chemical Properties
Physical and chemical properties of PEG-30, -33, -35, -36, -40 castor oil and PEG-30 and -40 hydrogenated castor oil can be found in the original safety assessment. 1 A supplier reports that PEG-30,-35, and -40 are pale yellow viscous liquids at 30°C and have a maximum water content of 0.2%. 4 PEG-40 hydrogenated castor oil is reported to be a waxy liquid at 30°C and also has a maximum water content of 0.2%.
Impurities
Since PEGs are the condensation products of ethylene oxide and water, with the chain length controlled by number of moles of ethylene oxide that are polymerized, they may contain trace amounts of 1,4-dioxane, a by-product of ethoxylation. 1,4-Dioxane is a known animal carcinogen. 5 The Food and Drug Administration (FDA) has been periodically monitoring the levels of 1,4-dioxane in cosmetic products, and the cosmetic industry reported that it is aware that 1,4-dioxane may be an impurity in PEGs and, thus, uses additional purification steps to remove it from the ingredient before blending into cosmetic formulations. 6,7
Use
Cosmetic
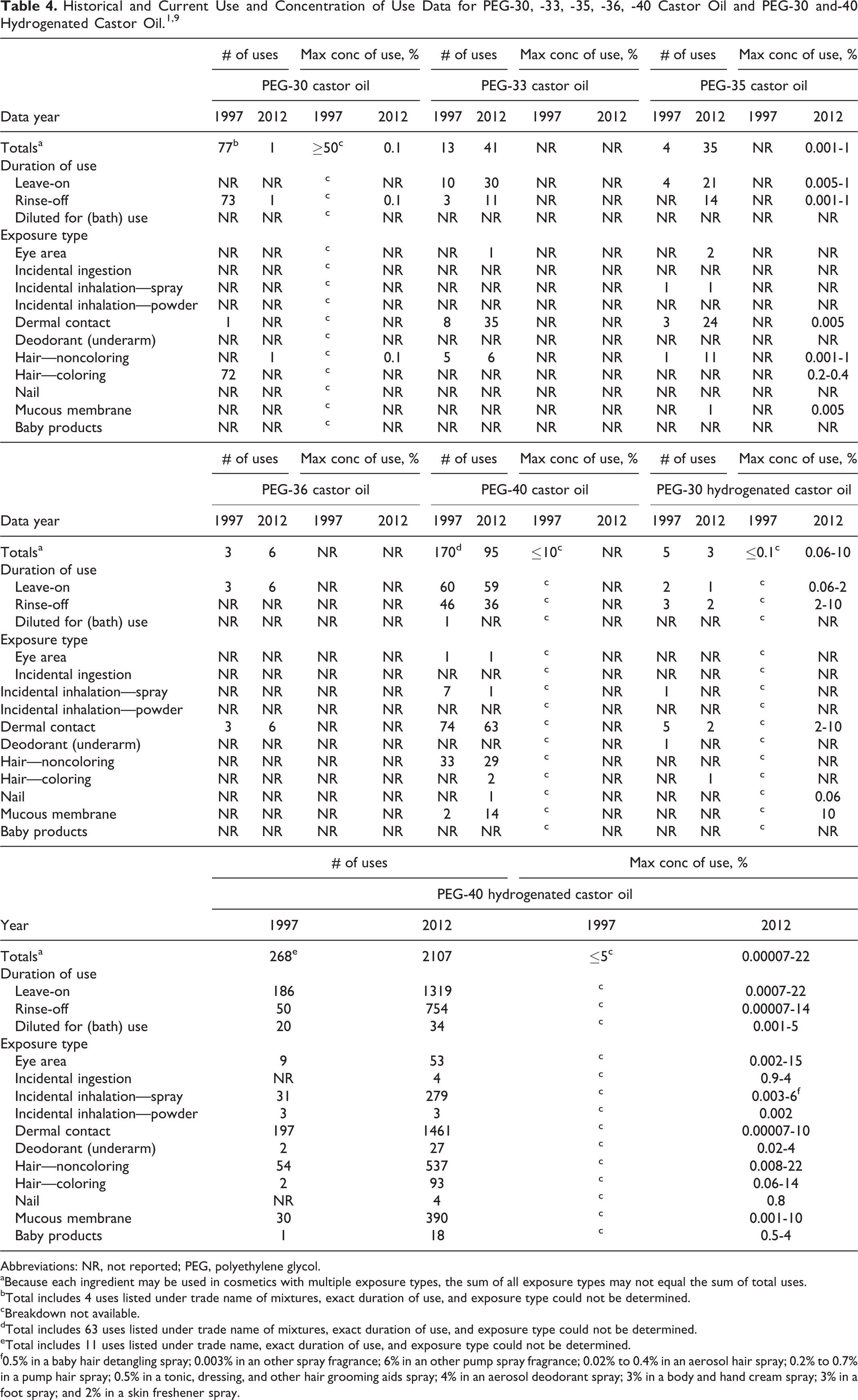
Table 4 presents the historical and current product formulation data for PEG-30, -33, -35, -36, and -40 castor oil and PEG-30 and -40 hydrogenated castor oil. These PEGylated castor oils function primarily as surfactants (emulsifying or solubilizing agents) in cosmetic formulations. 8 According to the information supplied to FDA Voluntary Cosmetic Registration Program (VCRP) database by industry in 1997, PEG-40 hydrogenated castor oil had the most uses at 268, with the majority of the uses reported in leave-on products with a dermal exposure route. 1 The ingredient with the second most uses was PEG-40 castor oil with 170 uses, most in leave-on products with a dermal exposure route. An industry survey reported use concentrations for PEG-40 hydrogenated castor oil and PEG-40 castor oil of ≤10% and ≤5%, respectively. Currently, the FDA’s VCRP database indicates that uses have decreased for PEG-30 castor oil, PEG-40 castor oil, and PEG-30 hydrogenated castor oil, with the most significant decrease occurring for PEG-40 castor oil, which now has 95 reported uses. 9 Increases in use are reported for the remaining PEGylated castor oils from the original report. The most significant increase is for PEG-40 hydrogenated castor oil, which now has 2107 reported uses (up from 268 uses). In a recent survey of use concentrations, PEG-40 hydrogenated castor oil had a maximum use concentration range of 7.0 × 10−5% to 22%, with 22% reported in leave-on noncoloring hair products. 10 PEG-30 castor oil had a maximum use concentration of 0.1% in a rinse-off noncoloring hair product.
Abbreviations: NR, not reported; PEG, polyethylene glycol.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bTotal includes 4 uses listed under trade name of mixtures, exact duration of use, and exposure type could not be determined.
cBreakdown not available.
dTotal includes 63 uses listed under trade name of mixtures, exact duration of use, and exposure type could not be determined.
eTotal includes 11 uses listed under trade name, exact duration of use, and exposure type could not be determined.
f0.5% in a baby hair detangling spray; 0.003% in an other spray fragrance; 6% in an other pump spray fragrance; 0.02% to 0.4% in an aerosol hair spray; 0.2% to 0.7% in a pump hair spray; 0.5% in a tonic, dressing, and other hair grooming aids spray; 4% in an aerosol deodorant spray; 3% in a body and hand cream spray; 3% in a foot spray; and 2% in a skin freshener spray.
Table 5 presents the current product formulation data for the cosmetic ingredients that were added to the PEGylated oil safety assessment, and Table 6 lists the ingredients not reported to be used. Currently, the VCRP database indicates that, of the additional ingredients, PEG-60 hydrogenated castor oil has the most uses (349) with the majority in leave-on products with a dermal exposure route. 9 The maximum use concentration range for PEG-60 hydrogenated castor oil was 4.0 × 10−5% to 18%, with the 18% reported in leave-on noncoloring hair products. 11 Olive oil PEG-7 esters had the second most reported uses (97). The maximum use concentration range for olive oil PEG-7 esters was 0.05% to 97%, with the 97% reported in a rinse-off (shaving) product.
Frequency and Concentration of Use (2012) According to Duration and Type of Exposure for Expanded PEGylated Oil Group. 9
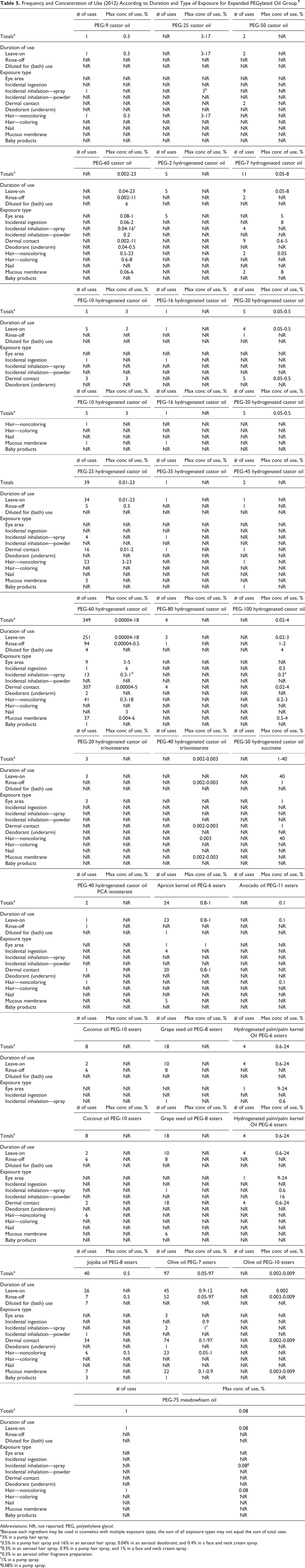











Abbreviations: NR, not reported; PEG, polyethylene glycol.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b3% in a pump hair spray.
c0.5% in a pump hair spray and 16% in an aerosol hair spray; 0.04% in an aerosol deodorant; and 0.4% in a face and neck cream spray.
d0.3% in an aerosol hair spray, 0.9% in a pump hair spray, and 1% in a face and neck cream spray.
e0.3% in an aerosol other fragrance preparation.
f1% in a pump spray.
g0.08% in a pump spray.
Not Reported to be in Use.
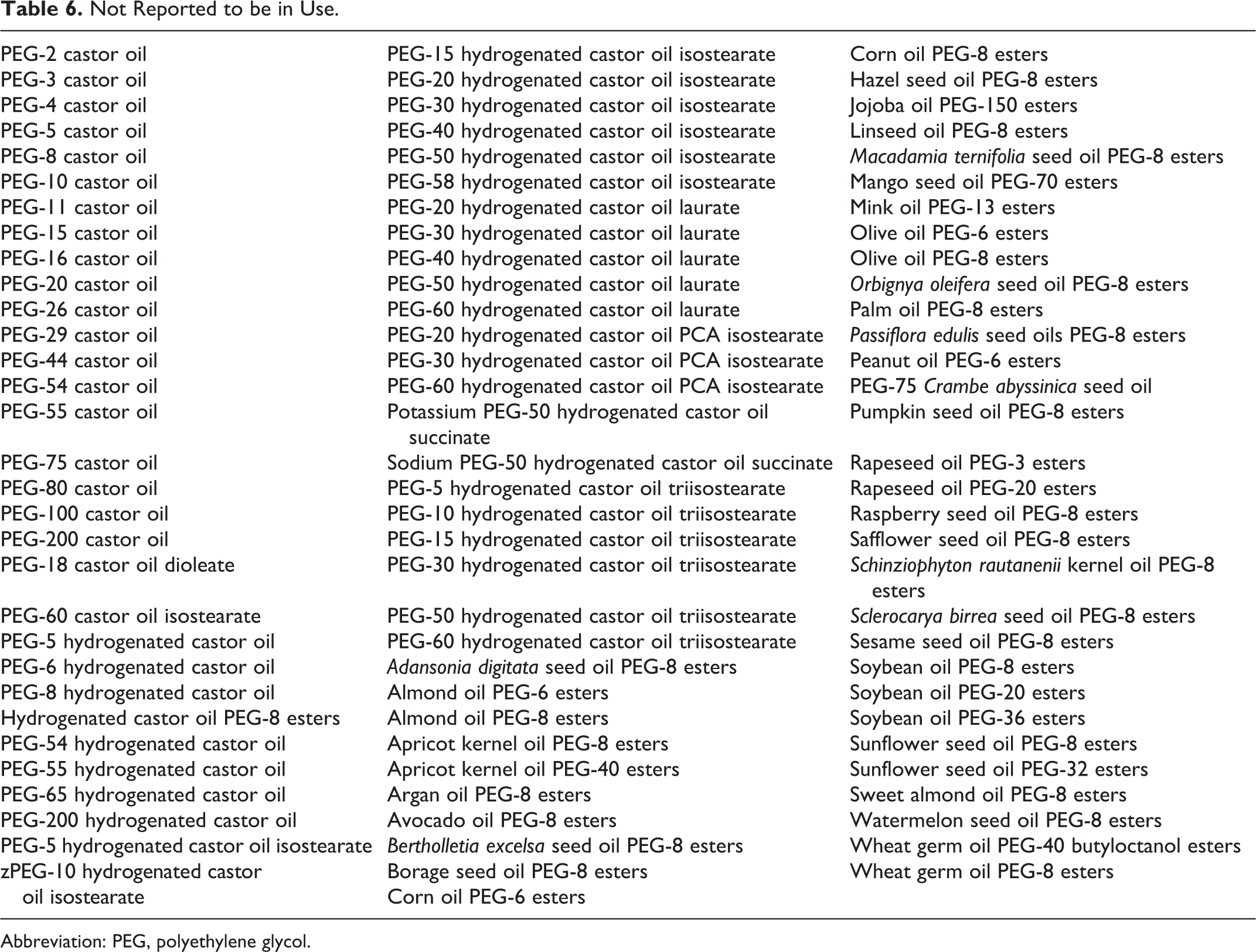
Abbreviation: PEG, polyethylene glycol.
In some cases, reports of uses were received from the VCRP, but no concentration of use data were available. For example, PEG-33 castor oil is reported to be used in 41 formulations, but no use concentration data were available. In other cases, no reported uses were received from the VCRP, but a use concentration was provided in the industry survey. For example, PEG-25 castor oil was not reported in the VCRP database to be in use, but the industry survey indicated that it is used in leave-on formulations at maximum concentrations ranging from 3% to 17%. It should be presumed that PEG-25 castor oil is used in at least 1 cosmetic formulation.
PEGylated oils were reported to be used in fragrance products, hair sprays, deodorants, and indoor tanning preparations and could possibly be inhaled. For example, PEG-60 castor oil was reported to be used in aerosol hair sprays at maximum concentrations up to 16%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 12 –15 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 13,14 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 14 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
The PEGylated oils are not restricted from use in any way under the rules governing cosmetic products in the European Union. 16
Noncosmetic
The PEG-30 castor oil and PEG-40 hydrogenated castor oil may be used as nonionic surfactants in oral, topical, and parenteral drug delivery systems. 3,4,17 –23 PEGylated castor oil derivatives may also be used in animal feeds and textiles. 4
The PEG-30, -33, -35, -36, and -40 castor oil have been approved by the FDA as indirect food additives in adhesives and components of coatings (21 CFR §175.105 and §175.300) and packaging and food contact surfaces (21 CFR §176.210, §177.2800). The PEG-30 and -40 hydrogenated castor oils are approved as direct food additives (21 CFR §73.1) as well as indirect food additives in packaging and food contact surfaces (21 CFR §177.2800).
Toxicokinetics
Toxicokinetics data were available from earlier CIR safety assessments supporting the safety of alkyl PEG ethers and castor oil. These data are summarized in Table 2.
Absorption, Distribution, Metabolism, and Excretion
The disposition of PEG-35 castor oil was determined in 31 patients with cancer treated with a 1-hour infusion of paclitaxel (87.8 mg PEG-35 castor oil/mg drug). 24 Dose levels of PEG-35 castor oil ranged from 70 to 100 mg/m2. Plasma concentrations were measured. Clearance of PEG-35 castor oil appeared to be independent of infusion duration and the administered dose in the range studied (P = 0.797). Exposure measures increased in near proportion to an increase in dose. PEG-35 castor oil had a half-life and clearance of 35.7 ± 18.9 hours and 0.216 ± 0.075 L/h, respectively (P < 0.00001). The volume of distribution at steady state was 9.48 ± 2.59 L and indicated limited distribution of the excipient outside of the central compartment. These results were compared to those of the excipient Tween 80, which had a shorter terminal half-life (0.607 ± 0.245 hours) and total plasma clearance (7.70 ± 2.90 L/h). The other values were similar. The study concluded that use of PEG-35 castor oil as a formulation vehicle could result in drug interaction and excipient-related toxic side effects due to its relatively low rates of elimination.
Penetration Enhancement
A study of the development of a topical gel for treatment of acne vulgaris reports that various types of PEGs are hydrophilic penetration enhancers and are used in topical dermatological preparations. 17 The authors selected PEG-40 hydrogenated castor oil because of its properties as a very hydrophilic, nonionic solubilizer for fat-soluble vitamins A, D, E, and K, and for its stability and clarity in alcohol solution.
In a study evaluating vehicle effects on in vitro skin permeation of model drugs, caffeine and testosterone, apricot kernel oil PEG-6 esters facilitated the flux and diffusivity of caffeine across the stratum corneum, when compared to propylene glycol. 25
Toxicological Studies
Acute Toxicity
Dermal and oral acute toxicity data were available from earlier CIR safety assessments supporting the safety of alkyl PEG ethers and castor oil. These data are summarized in Table 2
Intravenous—nonhuman
PEG-X castor oil
Castor oil with an unspecified number of stoichiometric equivalents of ethylene oxide (generically listed as Cremophor) mixed with dimethyl acetamide (DMA) was evaluated as a vehicle in a diabetes drug. 26 The mixture was composed of 23% to 45% DMA/10% to 12% Cremophor in water and the dose volume was 1.67 to 3 mL/kg. Groups of 3 New Zealand White rabbits received intravenously the test material, saline, insulin, or N-methyl-2-pyrrolidone into the marginal ear vein. Blood was drawn just before injection and again at 0.25, 0.5, 1, 2, 4, and 24 hours after injection to determine glycemia values. Glycemia after injection with the DMA/Cremophor mixture remained stable and within the normal range of 3.6 to 5.0 mmol/L. These results were comparable with those for the rabbits that received saline. In addition, the test material did not elicit irritation at the site of injection.
PEG-60 hydrogenated castor oil
The toxicity of PEG-60 hydrogenated castor oil was evaluated in male and female Beagle dogs, male and female cynomolgus monkeys, male New Zealand White rabbits, male Hartley guinea pigs, and male Sprague-Dawley rats. 27 The test material was injected intravenously to groups of 3 dogs at 0.625, 1.25, 2.5, or 10 mg/kg; in groups of 3 monkeys or 5 rabbits at 50 or 100 mg/5 mL/kg; and in groups of 5 guinea pigs and 5 rats at 10 or 100 mg/5 mL/kg. Blood pressure was monitored in the dogs before the injection and 10, 30, and 60 min after injection. Blood was taken from all animals to measure plasma histamine levels. In dogs, further histopathological examinations were performed on mast cells in the liver and skin. Clinical signs of toxicity were observed until 60 min after injection in all animals.
In dogs injected with 1.25, 2.5, or 10 mg/kg of the test material, blood pressure decreased and flush, swelling, and itching were observed. Additionally, in the 10 mg/kg-dose group, a decrease in spontaneous activity was observed. An increase in plasma histamine levels was observed in the 2.5- and 10-mg/kg dose groups. Degranulation was observed after injection in the mast cells of the skin but not in the liver. No signs of toxicity were observed in monkeys, rabbits, guinea pigs, or rats, and there was no change in plasma histamine levels. The toxicity of PEG-60 hydrogenated castor oil may be species specific. 27
Repeated-Dose Toxicity
Repeat dose toxicity data were available from earlier CIR safety assessments supporting the safety of alkyl PEG ethers and castor oil. These data are summarized in Table 2.
PEG-35 castor oil
Several in situ rat studies investigated the effects of the use of Cremophor EL (PEG-35 castor oil) as a vehicle in intravenous drugs. 28 –31 Reduction in muscle mitochondria, increase in myocardial lipid peroxidation, acidosis, weight loss, and cholestatic effects have been observed at perfusion concentrations of 100 µg/L.
Reproductive and Developmental Toxicity
Reproductive and developmental toxicity data were available from earlier CIR safety assessments supporting the safety of PEGS, castor oil, and alkyl PEG ethers. These data are summarized in Table 2.
Genotoxicity
Genotoxicity data were available from earlier CIR safety assessments supporting the safety of castor oil, PEGs, and alkyl PEG ethers. These data are summarized in Table 2.
PEG-60 Hydrogenated Castor Oil
The genotoxic potential of PEG-60 hydrogenated castor oil was studied in a reverse mutation test in Salmonella typhimurium strains TA100, TA98, TA1535, and TA1537 and in Escherichia coli strain WP2uvrA, with and without metabolic activation. 32 The test concentrations ranged from 313 to 5000 μg/plate. The positive controls were 9-aminoacridine, sodium azide, 2-(2-furyl)-3-(5-nitro-2-furyl)-acrylamide, and 2-aminoanthracene. No biologically relevant increases in revertant colony numbers were observed in any test strain at any concentration, with or without metabolic activation. Controls yielded expected results. The study concluded that PEG-60 hydrogenated castor oil was not genotoxic.
The above-mentioned research study also investigated the genotoxic potential of PEG-60 hydrogenated castor oil in a chromosome aberration study with Chinese hamster V79 cells, with and without metabolic activation. 32 The test concentrations ranged from 313 to 5000 μg/mL. The positive controls were mitomycin C and dimethylnitrosamine. The test material without metabolic activation had dose-related inhibition of cell proliferation after 24- and 48-hour treatments. With metabolic activation, the test material induced only slight inhibition of cell proliferation even at the highest concentration after 6 hours treatment. PEG-60 hydrogenated castor oil did not induce chromosome aberrations at any dose, with or without metabolic activation. The controls yielded expected results. The study concluded that PEG-60 hydrogenated castor oil was not genotoxic.
The same study also researched the genotoxic potential of PEG-60 hydrogenated castor oil in a mouse micronucleus test using BDF1 male and female mice. 32 A dose range finding experiment preceded the main study. In the main study, groups of 5 mice of each sex received single intraperitoneal injections of 2000 mg/kg body weight PEG-60 hydrogenated castor oil. Control groups received physiological saline or mitomycin C. Bone marrow cells were collected at 24, 48, or 72 hours. The number of micronucleated erythrocytes in 1000 polychromatic erythrocytes and the number of polychromatic erythrocytes in 1000 erythrocytes were recorded for each mouse. In the dose range-finding study, no deaths occurred. In the main study, no treatment-related mortalities or clinical signs of toxicity were observed. No significant increase in micronucleated polychromatic erythrocytes or significant decrease in the ratio of polychromatic to normochromatic erythrocytes were observed in male or female mice that received the test material. The controls yielded expected results. The study concluded that PEG-60 hydrogenated castor oil was not genotoxic.
Carcinogenicity
Carcinogenicity data were available from earlier CIR safety assessments supporting the safety of PEGs and alkyl PEG ethers. These data are summarized in Table 2.
Irritation and Sensitization
Irritation
Irritation data were available from earlier CIR safety assessments supporting the safety of PEGs, mink oil, and plant-derived fatty acid oils. These data are summarized in Table 2.
Dermal—nonhuman
PEG-35 castor oil
A skin irritation study of a pharmaceutical microemulsion that contained 20% (w/w) PEG-35 castor oil was performed in male guinea pigs (strain not specified). 33 The hair on the backs of the guinea pigs was removed 24 hours before treatment, and the animals were divided into a group with intact skin and a group with skin injury from scarification. These groups were again subdivided into single and multiple applications. There were a total of 5 guinea pigs in each subgroup. All guinea pigs received the test material and a control cream. Single application animals were treated for 24 hours, and the test sites were inspected for erythema and edema 1, 24, 48, and 72 hours after material removal. Multiple-application animals were treated for 24 hours, followed by assessment for skin irritation 1 hour after material removal, in a total of 7 applications. The test sites were observed for an additional 3 days after the last application. While very slight irritation was observed on average at the 1-hour observation point in guinea pigs treated with multiple applications with damaged skin, the average scores were still in the range that was considered to indicate “no irritation.” No irritation was observed in any of the single-application animals or in the intact skin of the multiple-application animals. It was concluded that single and multiple applications of the microemulsion that contained 20% (w/w) PEG-35 castor oil did not cause irritation effects in guinea pigs.
PEG-40 hydrogentated castor oil
A dermal irritation test was performed in mice (species and number not described) to investigate the potential irritancy of a microemulsion that contains 20% PEG-40 hydrogenated castor oil. 23 A single dose of 10 μL of the test microemulsion was applied to the left ear of the mouse. The right ear served as a control. The mice were observed for development of erythema for 6 days. No signs of irritation were observed in the mice. The authors concluded that the formulation containing 20% PEG-40 hydrogenated castor oil would probably not irritate human skin.
The dermal irritancy potential of a microemulsion gel system that contained 20.66% PEG-40 hydrogenated castor oil as a surfactant was studied in male albino rats using the Draize method. 34 Animals were divided into 3 groups of 6: a negative control (no treatment), a positive control (0.8% aqueous formalin), and the test formulation. The rats received a dose of 0.5 g of the formulation on a 5-cm2 area on the shaved dorsal side daily for 3 consecutive days. Signs of erythema and edema were monitored daily for 3 days. After 3 days, the rats were killed, and skin samples were taken for histopathological examination. No signs of irritation were observed in the test formulation. The controls yielded expected results. Histopathological examination found no apparent signs of skin irritation. The study concluded that the test formulation that contained 20.66% PEG-40 hydrogenated castor oil was not a skin irritant.
Ocular
PEG-35 castor oil
Several different formulations of a potential glaucoma drug in a nanoemulsion were tested for ocular irritation potential. 35 A few of these formulations contained PEG-35 castor oil as a surfactant. Groups of 6 New Zealand albino rabbits received test formulations that contained 0% to 13% PEG-35 castor oil. In each rabbit, the right eye received 50 μL of the tested formulation, while the left eye was used as a control. The rabbits received the test formulation every 2.5 hours through a period of 7.5 hours/d for 3 successive days and once on the fourth day. Eyes were examined according to the Draize method 1 and 24 hours after the last instillation. The eyelids, cornea, iris, conjunctiva, and anterior chamber were inspected for inflammation or other toxic reactions. The eyes were then stained with fluorescein and examined under ultraviolet light to verify possible corneal lesion. A few nanoemulsion formulations that contained up to 13.5% PEG-35 castor oil were found to be nonirritating and tolerated well by the rabbit eye. Cross-sections from the corneas of rabbits’ eye after application of the tested formulations together with a control section showed that both corneal structure and integrity were unaffected by treatment.
Sensitization
Sensitization data were available from earlier CIR safety assessments that support the safety of PEGs, mink oil, plant-derived fatty acid oils, and alkyl PEG ethers. These data are summarized in Table 2.
Clinical Use
Case Studies
PEG-35 castor oil
A 40-year-old female undergoing chemotherapy treatment for breast cancer had a cutaneous lupus erythematous-like reaction within 24 hours of intravenous administration of the drug paclitaxel that contained the diluent, PEG-35 castor oil. 36
When treatment was switched to a formulation of paclitaxel bound to albumin, without PEG-35 castor oil, no lupus-like reactions were observed. The case study concluded that PEG-35 castor oil induced the lupus-like reaction and suggested that previously reported incidences of lupus-like reaction in chemotherapy patients were from this diluent and not from the chemotherapeutic agent.
PEG-7 hydrogenated castor oil
A 73-year-old male was reported to have an eczematous rash on the face and neck after use of a sunscreen lotion. 37 The patient was patch tested with an extended British Contact Dermatitis Society standard series, medicament series, facial series, photoallergic series, and the sunscreen. A positive reaction was elicited only with the patient’s sunscreen, both on nonirradiated and irradiated skin. When tested with the individual ingredients of the sunscreen, a positive reaction occurred to 10% PEG-7 hydrogenated castor oil in petrolatum (+ on days 2 and 4). Negative reactions were observed to the remaining sunscreen ingredients.
PEG-60 hydrogenated castor oil
A 27-year-old male undergoing maintenance chemotherapy for acute myeloblastic leukemia developed a high-grade fever and erythroblastopenia within 6 hours of after intravenous administration of the chemotherapy drugs that included enocitabine. 38 The enocitabine product contained PEG-60 hydrogenated castor oil. When enocitabine was administered alone, the same symptoms reoccurred. Coculturing the patient’s bone marrow with enocitabine or PEG-60 hydrogenated castor oil found significant growth inhibition of late erythroid progenitors in the presence of the patient’s immunoglobulin G (IgG). The researchers of the study believe that PEG-60 hydrogenated castor oil acted as a hapten and caused the immunological suppression of the growth of erythroid progenitors through a hypersensitive reaction mediated by IgG.
Summary
The review of the safety of PEG-30, -33, -35, -36, and -40 castor oil and PEG-30 and -40 hydrogenated castor oil as used in cosmetics has been expanded to include additional PEGylated castor oils and other PEGylated plant-derived oils. Data from the original report on the PEGylated castor oils are summarized in Table 2, so this section only summarizes newly identified information on the original ingredients and the expanded ingredients.
Just as oils and other PEGylated materials are mixtures, PEGylated oils are mixtures. As most natural source oils are primarily triglycerides (and mono- and diglycerides) and fatty acids, PEGylated oils are primarily PEGylated glycerides, along with some PEGylated fatty acids.
The PEGs are the condensation products of ethylene oxide and water, with the chain length controlled by number of moles of ethylene oxide that are polymerized. PEGs may contain trace amounts of 1,4-dioxane, a by-product of ethoxylation.
The PEGylated castor oils function primarily as surfactants that function as emulsifying or solubilizing agents in cosmetic formulations. Of the 130 PEGylated oil ingredients described as cosmetic ingredients, only 35 have uses reported by either the FDA’s VCRP database or the Personal Care Products Council. Current FDA data indicate that PEG-40 hydrogenated castor oil has the largest number of reported uses, 2107, with a use concentration range of 7.0 × 10− 5% to 22%, with the 22% reported in leave-on noncoloring hair products.
PEG-30 castor oil and PEG-40 hydrogenated castor oil may be used as nonionic surfactants in oral, topical, and parenteral drug delivery systems. PEGylated castor oil derivatives may also be used in animal feeds and textiles. PEG-30, -33, -35, -36, and -40 castor oil have been approved by the FDA as indirect food additives in adhesives and PEG-30 and -40 hydrogenated castor oil are approved as direct and indirect food additives.
Various types of PEGs are hydrophilic penetration enhancers and are used in topical dermatological preparations.
Unspecified PEG castor oil did not elicit irritation at the site of injection in rabbits when evaluated as a vehicle in an intravenous drug.
The acute toxicity of PEG-60 hydrogenated castor oil was evaluated in male and female Beagle dogs, male and female cynomolgus monkeys, male New Zealand White rabbits, male Hartley guinea pigs, and male Sprague-Dawley rats. Toxicity, including decreased blood pressure, flush, swelling, itching, and increase histamine levels, was observed in dogs injected with 1.25, 2.5, or 10 mg/kg of the test material. No signs of toxicity were observed in monkeys, rabbits, guinea pigs, or rats.
Several rat studies investigated the effects of the use of Cremophor EL (PEG-35 castor oil) as a vehicle in intravenous drugs. Effects on cardiac and skeletal function have been observed at a concentration of 100 µg/L.
The genotoxic potential of PEG-60 hydrogenated castor oil was studied in a reverse mutation test, in a chromosome aberration study, and in a mouse micronucleus test: the studies concluded that PEG-60 hydrogenated castor oil was not genotoxic.
A study of single and multiple applications of a microemulsion that contained 20% (w/w) PEG-35 castor oil did not cause irritation effects in guinea pigs. A dermal irritation test performed in mice concluded that a formulation containing 20% PEG-40 hydrogenated castor oil would probably not irritate human skin. A study of the dermal irritancy potential of a microemulsion gel system in rats concluded that the test formulation containing 20.66% PEG-40 hydrogenated castor oil was not a skin irritant. Nanoemulsions containing up to 13.5% PEG-35 castor oil were nonirritating in rabbit eyes.
Case studies of adverse events occurring in patients with cancer were reported following use of chemotherapy drugs that contained PEG-35 castor oil and PEG-60 hydrogenated castor oil. Another case study reported an adverse dermatological event following use of a sunscreen containing PEG-7 hydrogenated castor oil.
Discussion
Overall, toxicokinetics, single-dose and repeated-dose toxicity, reproductive and developmental toxicity, genotoxicity, carcinogenicity, dermal and ocular irritation, and sensitization data were available for PEG castor oils. Similarly, extensive supportive data exist for the components of the PEGylated oils, which are complex mixtures of the etherification and transesterifcation products of fatty acid glycerides and fatty acids derived from the identified plant source with the equivalents of ethylene oxide to reach the desired PEG length. PEG compounds will be present unattached to glycerides or fatty acid groups. Because of this unique chemistry, the Panel determined that the available data in previous safety assessments of PEGs and of plant-derived fatty acids supported the safety of PEGylated oils in a “read-across” fashion.
The Panel expressed concern regarding the possible presence of ethylene oxide and trace amounts of 1,4-dioxane as impurities in any cosmetic ingredient containing a PEG moiety. They stressed that the cosmetic industry should continue to use the necessary purification procedures to remove these impurities from the ingredient before blending it into cosmetic formulations.
The Panel also expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
A safety assessment of diethylene glycol (aka PEG-2) has not been completed. A safety assessment of the PEGs group of ingredients, however, includes PEG-4, which was found to be safe for use in cosmetics. Since PEG-4 is a mixture that includes PEG-2, PEG-2 also would be safe for use in cosmetics.
The Panel recognized that some of these ingredients can enhance the penetration of other ingredients through the skin. They cautioned that care should be taken in formulating cosmetic products that may combine these ingredients with any ingredients whose safety was based on their lack of dermal absorption or when dermal absorption was a concern.
The Panel looked at changes in the pattern of use and concentration of use since the original safety assessment of PEG castor oils and noted that the earlier safety assessment had specified safe up to 50% use concentration. As PEG castor oils and the rest of the PEGylated oils are now used at concentrations below 50% in leave-on products, the Panel determined that concentration limits need no longer be specified. Products using these ingredients should be formulated to be nonirritating.
The Panel noted that adverse reactions have been reported for PEG-35 castor oil used as a vehicle in intravenous drugs. Because this route of exposure does not occur from cosmetic use, the Panel considered that such data were not relevant to assessing the safety of the ingredient in cosmetics.
The Panel discussed the issue of incidental inhalation exposure from fragrance products, hair sprays, deodorants, and indoor tanning preparations. There were no inhalation toxicity data available. These ingredients are reportedly used at concentrations up to 16% in cosmetic products that may be aerosolized (ie, cosmetic sprays) and in other products that may become airborne (ie, cosmetic powders). The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. In addition, the Panel considered other data available to characterize the potential for PEGylated oils and related ingredients to cause reproductive, developmental, and carcinogenic effects in animals (as described in the original safety assessment of PEG castor oils) and dermal irritation and sensitization. They noted the low systemic toxicity at high doses in several acute and repeat exposure studies, little or no irritation or sensitization in multiple tests of dermal exposure and in clinical studies, little or no irritation in multiple ocular tests, the absence of genotoxicity in bacterial and mammalian test systems, and no reproductive or developmental toxicity or carcinogenicity in animal feeding studies. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that the 130 PEGylated oil ingredients listed subsequently are safe in the present practices of use and concentration in cosmetics when formulated to be nonirritating. This conclusion supersedes the earlier conclusion issued by the Expert Panel in 1997. PEG-2 castor oil* PEG-3 castor oil* PEG-4 castor oil* PEG-5 castor oil* PEG-8 castor oil* PEG-9 castor oil PEG-10 castor oil* PEG-11 castor oil* PEG-15 castor oil* PEG-16 castor oil* PEG-20 castor oil* PEG-25 castor oil PEG-26 castor oil* PEG-29 castor oil* PEG-30 castor oil PEG-33 castor oil PEG-35 castor oil PEG-36 castor oil PEG-40 castor oil PEG-44 castor oil* PEG-50 castor oil PEG-54 castor oil* PEG-55 castor oil* PEG-60 castor oil PEG-75 castor oil* PEG-80 castor oil* PEG-100 castor oil* PEG-200 castor oil* PEG-18 castor oil dioleate* PEG-60 castor oil isostearate* PEG-2 hydrogenated castor oil PEG-5 hydrogenated castor oil* PEG-6 hydrogenated castor oil* PEG-7 hydrogenated castor oil PEG-8 hydrogenated castor oil* Hydrogenated castor oil PEG-8 esters* PEG-10 hydrogenated castor oil PEG-16 hydrogenated castor oil PEG-20 hydrogenated castor oil PEG-25 hydrogenated castor oil PEG-30 hydrogenated castor oil PEG-35 hydrogenated castor oil PEG-40 hydrogenated castor oil PEG-45 hydrogenated castor oil PEG-50 hydrogenated castor oil PEG-54 hydrogenated castor oil* PEG-55 hydrogenated castor oil* PEG-60 hydrogenated castor oil PEG-65 hydrogenated castor oil* PEG-80 hydrogenated castor oil PEG-100 hydrogenated castor oil PEG-200 hydrogenated castor oil* PEG-5 hydrogenated castor oil isostearate* PEG-10 hydrogenated castor oil isostearate* PEG-15 hydrogenated castor oil isostearate* PEG-20 hydrogenated castor oil isostearate* PEG-30 hydrogenated castor oil isostearate* PEG-40 hydrogenated castor oil isostearate* PEG-50 hydrogenated castor oil isostearate* PEG-58 hydrogenated castor oil isostearate* PEG-20 hydrogenated castor oil laurate* PEG-30 hydrogenated castor oil laurate* PEG-40 hydrogenated castor oil laurate* PEG-50 hydrogenated castor oil laurate* PEG-60 hydrogenated castor oil laurate* PEG-20 hydrogenated castor oil PCA isostearate* PEG-30 hydrogenated castor oil PCA isostearate* PEG-40 hydrogenated castor oil PCA isostearate PEG-60 hydrogenated castor oil PCA isostearate* PEG-50 hydrogenated castor oil succinate Potassium PEG-50 hydrogenated castor oil succinate* Sodium PEG-50 hydrogenated castor oil succinate* PEG-5 hydrogenated castor oil triisostearate* PEG-10 hydrogenated castor oil triisostearate* PEG-15 hydrogenated castor oil triisostearate* PEG-20 hydrogenated castor oil triisostearate PEG-30 hydrogenated castor oil triisostearate* PEG-40 hydrogenated castor oil triisostearate PEG-50 hydrogenated castor oil triisostearate* PEG-60 hydrogenated castor oil triisostearate*
Adansonia digitata seed oil PEG-8 esters* Almond oil PEG-6 esters* Almond oil PEG-8 esters* Apricot kernel oil PEG-6 esters Apricot kernel oil PEG-8 esters* Apricot kernel oil PEG-40 esters* Argan oil PEG-8 esters* Avocado oil PEG-8 esters* Avocado oil PEG-11 esters
Bertholletia excelsa seed oil PEG-8 esters* Borage seed oil PEG-8 esters* Coconut oil PEG-10 esters Corn oil PEG-6 esters* Corn oil PEG-8 esters* Grape seed oil PEG-8 esters Hazel seed oil PEG-8 esters* Hydrogenated palm/palm kernel oil PEG-6 esters Jojoba oil PEG-8 esters Jojoba oil PEG-150 esters* Linseed oil PEG-8 esters*
Macadamia ternifolia seed oil PEG-8 esters* Mango seed oil PEG-70 esters* Mink oil PEG-13 esters* Olive oil PEG-6 esters* Olive oil PEG-7 esters Olive oil PEG-8 esters* Olive oil PEG-10 esters
Orbignya oleifera seed oil PEG-8 esters* Palm oil PEG-8 esters*
Passiflora edulis seed oils PEG-8 esters* Peanut oil PEG-6 esters* PEG-75 Crambe abyssinica seed oil* PEG-75 meadowfoam oil Pumpkin seed oil PEG-8 esters* Rapeseed oil PEG-3 esters* Rapeseed oil PEG-20 esters* Raspberry seed oil PEG-8 esters* Safflower seed oil PEG-8 esters*
Schinziophyton rautanenii kernel oil PEG-8 esters*
Sclerocarya birrea seed oil PEG-8 esters* Sesame seed oil PEG-8 esters* Soybean oil PEG-8 esters* Soybean oil PEG-20 esters* Soybean oil PEG-36 esters* Sunflower seed oil PEG-8 esters* Sunflower seed oil PEG-32 esters* Sweet almond oil PEG-8 esters* Watermelon seed oil PEG-8 esters* Wheat germ oil PEG-40 butyloctanol esters* Wheat germ oil PEG-8 esters*
*Not in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable with others in this group.
Footnotes
Author Contribution
Burnett contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the article, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Gill, Bergfeld, Belsito, Hill, Klaassen, Liebler, Marks, Shank, Slaga, and Snyder contributed to conception and design, contributed to analysis and interpretation, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from Lillian J. Gill, Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.