Abstract
Tavaborole, a cyclized boronic acid, has been approved by the Food and Drug Administration for the topical treatment of toenail onychomycosis. This novel, low-molecular-weight pharmaceutical compound has broad-spectrum antifungal activity against dermatophytes, yeasts, and molds responsible for the disease. Tavaborole was tested in 2-year carcinogenicity studies in mice (once daily dermal administration) and rats (once daily by oral gavage) as part of the extensive nonclinical safety program. There was no evidence of tavaborole-related neoplasms observed in either study. Based on the data gathered from these 2 carcinogenicity studies, tavaborole is considered noncarcinogenic.
Introduction
Onychomycosis is a fungal infection of the nail unit (primarily the nail bed, and often involving the nail plate and less frequently the nail matrix), most commonly occurring in the toenails. The signs of infection include a thickening of the nail plate and nail bed, discoloration, splitting of the nail plate, and lifting of the nail plate from the nail bed, known as onycholysis. Symptoms include difficulty in grooming the nails, social embarrassment, damage to socks and stockings caused by an abnormal nail plate, secondary bacterial infection, and pain or discomfort due to separation of the nail plate. Typically, the disease is caused by dermatophytes, which are ubiquitous in the environment and are the same organisms associated with athlete’s foot, jock itch, and ring worm. A large proportion of the population is susceptible to dermatophyte infections, and there does not appear to be a protective adaptive immune response, so reinfection is common in the susceptible population. 1,2
Current treatment options include both oral and topical drugs, with oral therapies resulting in relatively better efficacy outcomes. The difficulty in treating onychomycosis topically may result from the deep-seated nature of the infection within the anatomically complex nail unit (nail plate, nail bed, and nail matrix) and the inability of some drugs to effectively reach all compartments of the nail unit. Limitations of currently approved oral therapies include drug–drug interactions and systemic adverse effects, including hepatotoxicity. 3 –7 Another limitation of currently approved topical agents is their relatively low efficacy. Hence, there is a clear need for more effective topical agents to treat onychomycosis.
Tavaborole (Figure 1), a cyclized boronic acid complex (5-fluoro-1,3-dihydro-1-hydroxy-2,1-benzoxaborole), is an FDA-approved topical treatment of onychomycosis. This novel, low-molecular-weight (152 Da) compound demonstrates potent antifungal activity against dermatophytes with minimum inhibitory concentration values in the low microgram per milliliter range. 8
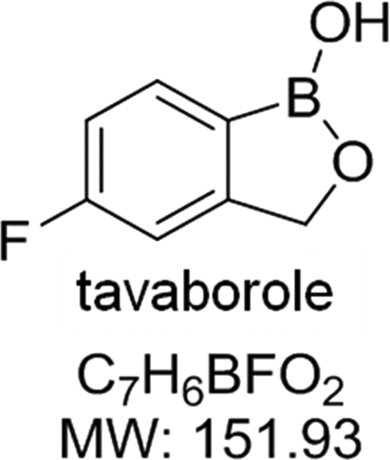
The structure, empirical formula, and molecular weight of tavaborole (CAS# 174671-46-6).
The mechanism of action of tavaborole is inhibition of fungal protein synthesis. Tavaborole represents an antifungal, which inhibits protein synthesis by inhibiting leucyl-transfer RNA (tRNA) synthetase, an aminoacyl-tRNA synthetase (AARS). 9 Although tavaborole inhibits an AARS, its mechanism of action is very different from that of known AARS inhibitors and represents a new class of inhibitors that exert their action on AARSs via the editing domain.
Tavaborole has been approved by the Food and Drug Administration (FDA) for the topical treatment of mild-to-moderate toenail onychomycosis. Preclinical safety studies have demonstrated the relative safety of this compound, including a battery of genetic toxicology studies. 10 As the treatment for onychomycosis may be expected to occur over a substantial part of a patient’s lifetime, lifelong rodent studies have been mandated by International Conference on Harmonization (ICH) Guidelines to investigate the carcinogenic potential of tavaborole using dermal (mouse) and oral (rat) treatments.
Materials and Methods
Two 2-year carcinogenicity studies were performed. One study was performed topically in mice using the same vehicle as used clinically. The other study was performed in rats by using oral gavage in a carboxymethylcellulose (CMC) vehicle. Both studies were conducted as per the published ICH Guidelines, and protocols were reviewed and approved by the appropriate Institutional Animal Care and Use Committee. The protocols were also approved by the Executive Carcinogenicity Assessment Committee (CAC) of the US FDA. The mouse study completed 1 week of dosing prior to receiving comments from the CAC. On day 8, the doses were changed to the CAC recommended doses. Additionally, toxicokinetic (TK) sampling was conducted on the first day of the new dosing schedule. Additionally, the final reports of both studies were reviewed by the Executive CAC, and the Committee agreed that the studies were adequate, noting prior Executive CAC concurrence with the protocols as well as concurrence on the results of these studies.
Dermal Carcinogenicity Study in Mice
Vehicle and test article information
The vehicle, 80% ethanol (USP, 190 proof)/20% propylene glycol (Spectrum Quality Products, New Brunswick, New Jersey), USP, was prepared approximately weekly to every other week during the study. Tavaborole used in the study had 98.3% purity and was manufactured by Anacor Pharmaceuticals (Palo Alto, California). Formulations of tavaborole were prepared weekly during the study at nominal concentrations of 50, 100, and 150 mg/mL. Prior to preparation, the bulk tavaborole was finely ground in a mortar with a pestle. All formulations were stored refrigerated when not in use and allowed to come to approximate room temperature prior to dosing. Dosing formulations were collected and evaluated for concentration at weeks 1, 2, 13, 27, 40, 53, 66, 79, 92, and 104.
Animals
Male and female, experimentally naive Crl: CD1(Icr) mice, approximately 4 weeks old at receipt, were received from Charles River Laboratories (Portage, Michigan). The mice were housed individually in suspended, stainless steel, and wire mesh-type cages in an environmentally controlled room. Fluorescent lighting was provided for approximately 12 hours/d. Temperature and humidity were continuously monitored and recorded. Meal Lab Diet (Certified Rodent Diet #5002; PMI Nutrition International, Inc St. Louis, Missouri) was available ad libitum throughout the study period. There was a total of 4 treatment groups in the study (Table 1) receiving 0%, 5%, 10%, or 15% tavaborole daily for 103 weeks (mice in groups 2, 3, and 4 were treated with 1%, 2.5%, and 10% tavaborole, respectively, on days 1-7). Separate TK groups were assigned for blood collection to measure tavaborole concentration in plasma at different time points for up to 6 months. Using a block randomization procedure, mice were assigned to the control and treatment groups. Mice assigned to the study had body weights within ±20% of the mean body weight for each sex. Mice found dead or euthanized in extremis during the first 6 days of the study were replaced.
Study Design for the Mouse Dermal Carcinogenicity Study.a
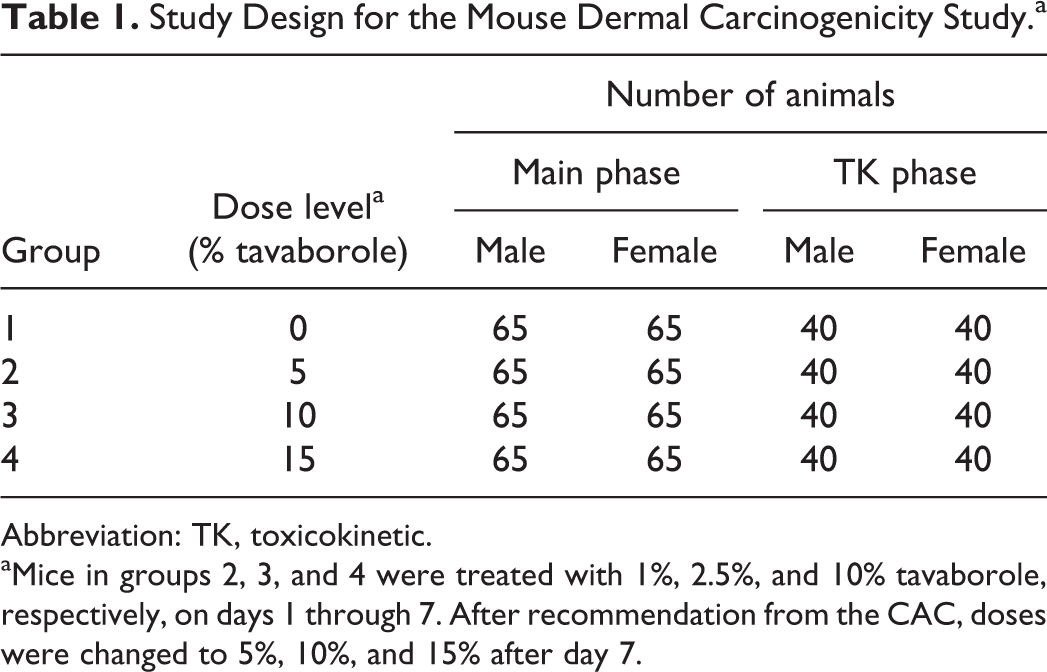
Abbreviation: TK, toxicokinetic.
aMice in groups 2, 3, and 4 were treated with 1%, 2.5%, and 10% tavaborole, respectively, on days 1 through 7. After recommendation from the CAC, doses were changed to 5%, 10%, and 15% after day 7.
Dose justification and test article administration
Doses for the definitive study were selected based on a 3-month study where mice (CD-1) were treated topically with concentrations of 1.7%, 3.3%, 5%, 10%, and 15% solutions of tavaborole. In the 3-month study, no drug-related changes were observed in body weight gain, clinical signs, hematology, or serum chemistry values. Signs of very slight erythema were present sporadically throughout the study in all tavaborole female groups but not in female control groups. Increased signs of erythema, from very slight to severe, were observed in tavaborole-treated males in the 3.3%, 5%, 10%, and 15% groups. Histopathologic evaluation of the skin found inflammatory and/or reactive changes at the application site in the 10% and 15% tavaborole groups. No other histopathologic changes attributed to tavaborole or vehicle solution were present. Based on these results from the 3-month study, doses of 0%, 5%, 10%, and 15% tavaborole solution were picked for the definitive study.
The vehicle and tavaborole were administered at approximately the same time (±2 hours) once daily for up to 104 weeks during the study by topical application to the skin. Mice assigned to TK groups received the vehicle or tavaborole for up to 180 days in the same manner as the main study groups. The dose volume for all groups was 1 µL/g. Prior to the initiation of dosing, the fur was clipped from the back of mice without abrading the skin. This clipped area comprised no less than 10% of the total body surface area. Fur was reclipped as necessary, and the 10% area of application was adjusted based upon the most recent mean body weight for each group by sex.
All mice were observed for morbidity, mortality, injury, and the availability of food and water twice daily till week 52 and thrice daily thereafter till the end of the study. Careful examination of the dosing site for signs of edema and erythema was conducted on all main study mice weekly according to the skin reaction scale based upon the Draize scale 11 for scoring skin irritation. Detailed examinations including palpation of masses were carried out weekly beginning prior to randomization till the end of the study. Body weights for all mice were measured and recorded 2 or 3 days after receipt, prior to randomization, on day 1 (main study and initial TK mice) and weekly through week 14, every other week from weeks 16 to 28, and every fourth week from weeks 32 to 104. Food consumption was measured and recorded for all main study mice weekly through week 14, every other week from weeks 16 to 28, and every fourth week from weeks 32 to 104. Ophthalmoscopic examinations were conducted on all main study mice pretest and prior to the terminal necropsy.
Plasma and TK analysis
Blood samples (approximately 0.5-1 mL) were collected from TK mice via cardiac puncture after carbon dioxide inhalation for determination of plasma concentrations of tavaborole. Samples were collected from cohorts of 3 mice/sex/group predose and at 0.5, 1, 2, 4, and 6 hours postdose on days 1 and 180. The mice were not fasted prior to sample collection. Samples were collected into tubes containing K2EDTA and stored on an ice block until centrifuging under refrigeration. Serum samples were placed in tightly capped, prelabeled plastic vials, and stored frozen at approximately 70°C until analysis using liquid chromatography-tandem mass spectrometry (LC/MS/MS). The lower limit of quantitation for the assay was 25 ng/mL. The TK parameter estimates were determined for tavaborole from mean concentration–time data. A noncompartmental module of WinNonlin software (Pharsight Corporation Carey, North Carolina) was used to calculate parameter estimates.
Postmortem study evaluations
The mice were euthanized by carbon dioxide inhalation followed by exsanguination from the vena cava and examined carefully for external abnormalities including masses. The abdominal, thoracic, and cranial cavities were examined for abnormalities and the organs removed, examined, and, where required, placed in fixative. Microscopic examination of fixed hematoxylin and eosin-stained paraffin sections was performed on protocol-designated sections of tissues by a board certified veterinary pathologist.
Statistics
Statistical comparisons were made between the control group and tavaborole-treated groups. Data for each sex within a set were analyzed separately. The raw data were tabulated within each time interval, and the mean and standard deviation or incidence counts were calculated for each end point by sex and group. For body weights and food consumption data group-pairwise comparisons (Levene/analysis of variance [ANOVA] Dunnett/Welch) was used; for erythema, eschar, and edema formation, Cochran Mantel Haenszel test was used; for mortality data, survival analysis was used; and for tumor data, tumor analysis was used.
Carcinogenicity Study in Rats by Oral Gavage
Vehicle and test article information
The vehicle used in the study was aqueous 1% (w/v) Medium Viscosity CMC USP Grade (Sigma-Aldrich, St Louis, Missouri). Tavaborole used in the study had 98.5% purity and was manufactured by Anacor Pharmaceuticals. The test and control articles were prepared once weekly, stored at 2°C to 8°C, and dispensed daily. For each dose level, an appropriate quantity of tavaborole was weighed out and placed into an appropriate size mortar. An aliquot of 1% CMC was added and the components finely ground with a pestle to form a preliminary paste. The paste was then transferred to a beaker. A further aliquot of 1% CMC was added, and the formulation was stirred on a stir plate until homogenous. A quantum satis (QS) with 1% CMC was done, and the formulation was stirred until a homogenous suspension was achieved. After preparation and sampling, the dose suspensions were stored refrigerated in a bulk container. The bulk dose suspensions and bulk control preparation were stirred for at least 15 minutes prior to aliquoting for the day’s dose. The aliquots were allowed to equilibrate to room temperature and were also stirred for at least 15 minutes prior to dosing. The aliquots were kept on a magnetic stir plate continuously until the last rat was treated. In order to verify the concentration of the tavaborole in the formulation, samples were analyzed from all dosing formulations at weeks 1 and 13 and at 13 weekly intervals thereafter.
Animals
Male and female experimentally naive Sprague-Dawley rats, Crl: CD (SD) IGS BR, approximately 4 weeks old at receipt, were received from Charles River Canada Inc (St-Constant, Quebec, Canada). The rats were housed individually in suspended, stainless steel, and wire mesh-type cages in an environmentally controlled room. Fluorescent lighting was provided for approximately 12 hours/d. Temperature and humidity were continuously monitored and recorded. All rats were subjected to a restricted feeding regimen. Accordingly, 5 and 4 food pellets/d were offered to males and females, respectively, of a certified, commercial laboratory rodent diet (Certified Rodent Diet 4.2 g-RPR 5L35; PMI Feeds Inc, St Louis, Missouri) ad libitum, except during designated procedures for the duration of the study. There were a total of 4 treatment groups in the study (Table 2). The TK groups were maintained for blood collection to measure tavaborole concentration in plasma at different time-points for up to 6 months. Using a block randomization procedure, rats were assigned to the control and treatment groups. Rats assigned to study had body weights within ±20% of the mean body weight for each sex. Rats found dead or euthanized in extremis during the first 6 days of the study were replaced.
Study Design for the Rat Carcinogenicity Study by Oral Gavage.
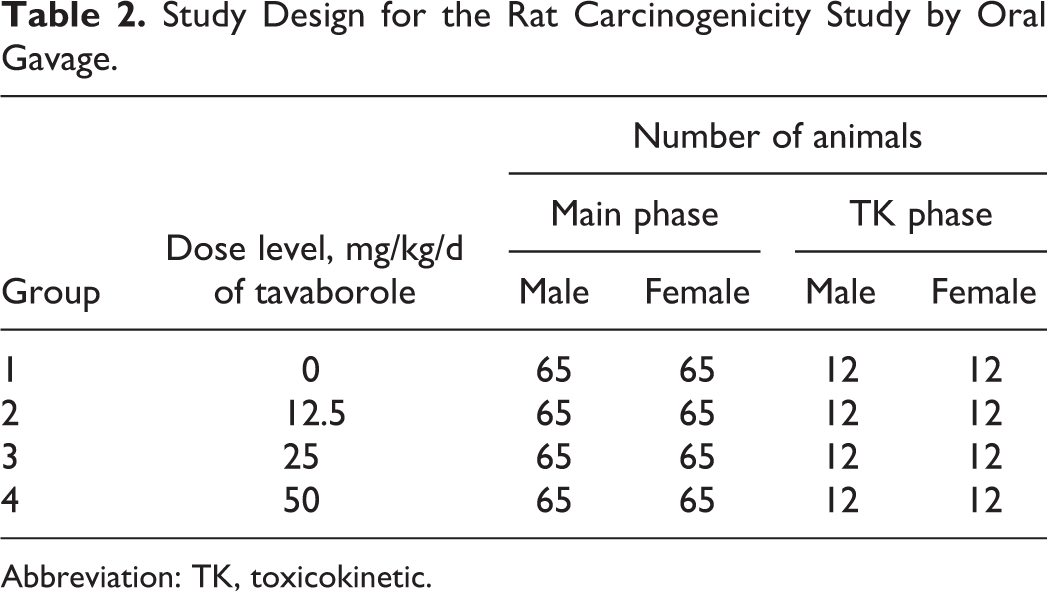
Abbreviation: TK, toxicokinetic.
Dose justification and test article administration
Doses for the definitive study were selected based on extensive rat oral toxicity studies conducted with tavaborole for periods of 28, 90, and 180 consecutive days. Specifically, in the 6-month (180 days) study, tavaborole was administered at doses of 30, 50, 100, and 200 mg/kg/d. Hyperplasia and hyperkeratosis of the nonglandular portion of the stomach were the principal adverse finding. These lesions occurred at all doses, were dose related, and showed dose-dependent increased severity and frequency of occurrence, especially apparent at ≥50 mg/kg/d. As a consequence of these findings, the lesions of the nonglandular portion of the stomach were defined as the rate limiting toxicity. Thus, the doses of 0, 12.5, 25, and 50 mg/kg/d were selected where the highest dose tested in the definitive study was above the maximum-tolerated dose.
Tavaborole and control/vehicle articles were administered at 0, 12.5, 25, and 50 mg/kg/d (at 1.25, 2.5, 5.0 mg/mL, respectively) once daily by oral gavage using a gavage needle attached to a syringe for 104 weeks. The dose volume was 10 mL/kg for all rats, including controls. The actual volume administered was calculated and adjusted based on the most recent practical body weight.
In-life examinations
All rats were observed for morbidity, mortality, injury, and the availability of food and water twice daily throughout the duration of the study. A detailed clinical examination of each rat was performed once pretreatment, weekly during the treatment period, and before necropsy. As part of this procedure, from week 26 onward, each rat was examined for the presence of palpable masses. During these examinations, particular attention was paid to the location, size, appearance, and progression (time first seen and time of disappearance, if relevant) of each palpable mass potentially representing a benign or malignant tumor. Any mass borne by a rat was given a numerical designation (ie, M1, M2, etc) according to the order of appearance. A palpable mass exceeded 2 mm to be considered for mass measurement. The physical description of the mass was limited to subcutaneous/cutaneous, fixed/movable, firm/soft, size (measured with calipers) mass not detected, and mass not measured. The description of mass appearance was limited to encrusted, abraded, and ruptured.
Body weights were recorded for all rats once prior to group assignment and approximately 1 week prior to initiation of treatment. During the treatment period, body weights were recorded for all rats on the day before the start of dosing, weekly thereafter for the first 14 weeks, and once every 4 weeks thereafter. Additionally, body weights were recorded terminally, prior to necropsy (fasted).
Plasma and TK analysis
Blood samples (approximately 1 mL) were collected (3 rats/sex/group) from TK rats by jugular venipuncture for determination of tavaborole plasma concentrations. Samples were collected at 0, 0.25, 0.5, 1, 2, and 4 hours postdose on days 1 and 180. Samples were collected into tubes containing K2EDTA and stored on an ice block until centrifuging under refrigeration. Serum samples were placed in tightly capped, prelabeled plastic vials, and stored frozen at approximately 70°C until analysis using LC/MS/MS. The TK parameter estimates were determined for tavaborole from mean concentration–time data. A noncompartmental module of WinNonlin software (Pharsight Corporation, Carey, North Carolina) was used to calculate parameter estimates.
Postmortem study evaluations
All surviving rats were euthanized with isoflurane followed by exsanguination upon completion of the treatment period. Gross pathology consisted of an external examination, including identification of all clinically recorded lesions, as well as a detailed internal examination. A full complement of tissues and organs was collected from all rats and were retained in neutral-buffered 10% formalin. Microscopic examination of fixed hematoxylin and eosin-stained paraffin sections was performed on protocol-designated sections of tissues by a board certified veterinary pathologist.
Statistics
Except for survival and tumor incidence data, numerical data obtained for all rats were subjected to calculation of group means and standard deviations. The data (males and females separately) were analyzed for homogeneity of variance using Levene Median and for normality using the Kolmogorov-Smirnov test. Homogeneous data were assessed using ANOVA and the significance of intergroup differences were determined using Dunnett test. Heterogeneous data were analyzed using the Kruskal-Wallis test, and the significance of intergroup differences was assessed using Dunn test. Differences that are significant at values of “P” that are equal to or less than 0.05 were flagged appropriately. Survival data were analyzed by the method of Peto et al. 12 Tumor incidence was analyzed separately for the 2 sexes. Statistical analysis was applied to any tumor site having a total incidence of at least 2 over all groups. The life table method 12 was used to compare tumor incidence across groups. Tumors were classified as “fatal” (“nonincidental”) or “incidental” depending on their causal association (or not) with death.
Results
Dermal Carcinogenicity Study in Mice
The results of the dosing formulation analysis indicated that the concentration of tavaborole was below the quantifiable limit in the vehicle samples. Concentrations at 5%, 10%, and 15% tavaborole were generally within the acceptable range of ±10% of target concentrations.
There were no tavaborole-related differences in mortality between control and treated males and females. Mean survival data are summarized in Table 3. There were no tavaborole-related differences in mortality between control and treated males and females. Causes of death were the type commonly seen in mice of this strain and age (heart failure/atrial thrombus, hemangiosarcoma/hemangioma, lung tumors, and lymphoma).
Survival, Body Weight Gain, and Palpable Masses in the Mouse Dermal Carcinogenicity Study; Mice Treated Daily for 103 Weeks.

Abbreviations: M, male; F, female; NA, not applicable.
Mean body weight gains (Table 3) and food consumption (data not presented) in males and females in all treatment groups were comparable to controls and unaffected by treatment. A few statistically significant increases or decreases in body weights and food consumption were observed sporadically in males and females in all treatment groups in comparison to controls; however, these differences were slight and transient in nature and were not considered toxicologically significant. No treatment-related clinical observations or an increase in the incidence of palpable masses was observed with tavaborole treatment in comparison to controls (Table 3).
A slight statistically significant increase in erythema/eschar findings was observed in males and females at 15% tavaborole (data not presented). These findings ranged from very slight to severe erythema/eschar but were only observed in very few (13) mice sporadically throughout the treatment period. Similar findings were observed, to a lesser degree and severity, in 6 mice or less at 10% tavaborole. In addition, a few statistically significant increases in edema findings were also evident in males at 15% tavaborole. These dermal findings occurred at a low incidence, were transient in nature, did not correlate with any tavaborole-related macroscopic or microscopic findings, and were not considered adverse.
No tavaborole-related ophthalmoscopic findings were observed. The observations noted were representative of pathology that would be expected for this group of animals considering age, sex, and strain.
Plasma and TK analysis
Results of the TK analysis are presented in Table 4. Tavaborole appeared to exhibit some gender-dependent TK characteristics in mice that varied with dose and study day. Following single-dose and multiple-dose administration of 5%, 10%, and 15% tavaborole topically to the skin of mice, tavaborole had mean Tmax values occurring within 0.5 to 1 hour on days 1 and 180. Tavaborole appeared to exhibit nonlinear single-dose and multiple-dose TK characteristics in male mice (generally less than proportional increases in area under the curve 0 to time t [AUC0-t] with increasing dose, where t = 6 hours) and linear single-dose TK characteristics in female mice (proportional increases in AUC0-t with increasing dose). There was little to no accumulation after 180 days of dosing.
Summary TK Parameters on Days 1 and 180 After Application of Tavaborole Solutions Once Daily Topically to the Skin of Male and Female Mice.a,b
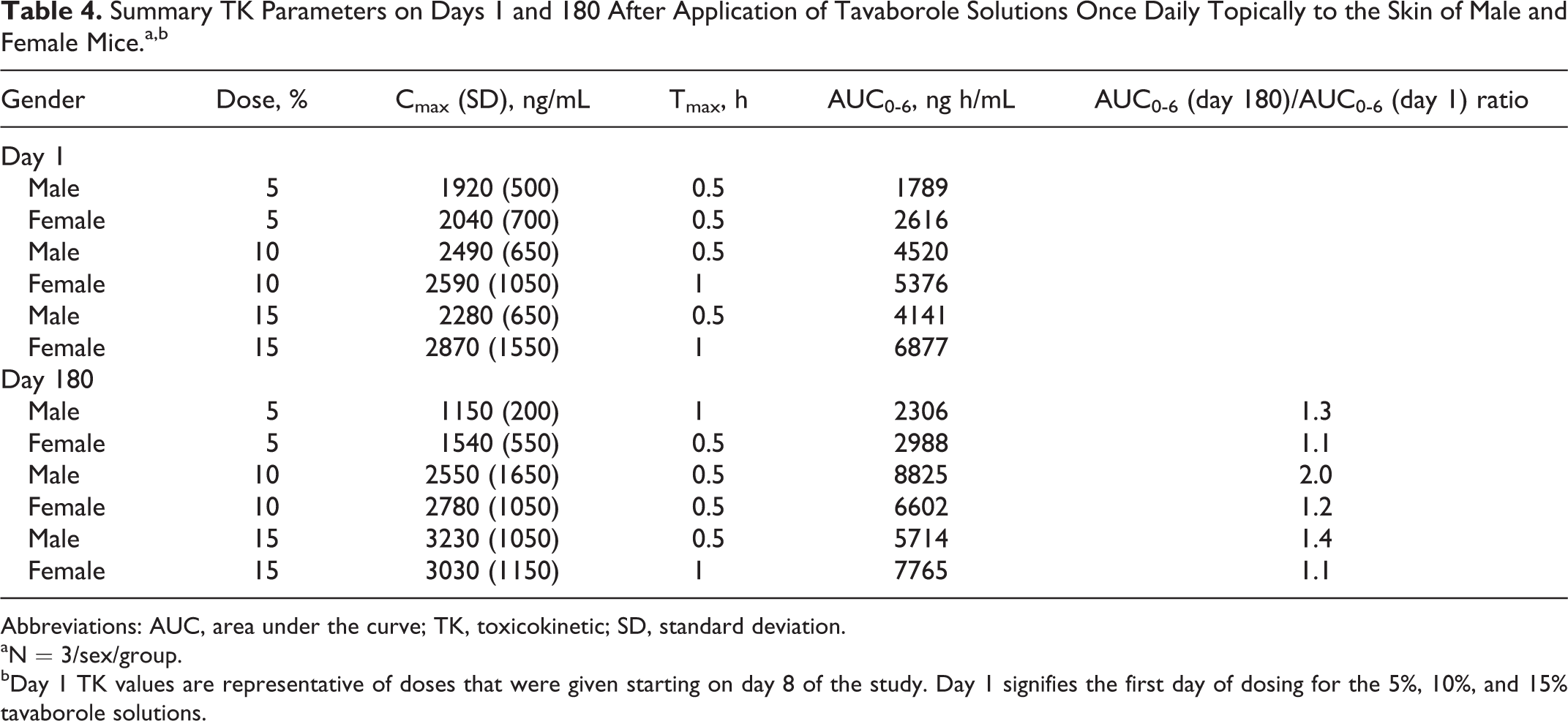
Abbreviations: AUC, area under the curve; TK, toxicokinetic; SD, standard deviation.
aN = 3/sex/group.
bDay 1 TK values are representative of doses that were given starting on day 8 of the study. Day 1 signifies the first day of dosing for the 5%, 10%, and 15% tavaborole solutions.
Postmortem study evaluations
No tavaborole-related observations were made in male or female mice at necropsy. Necropsy findings in the study were typical of observations commonly seen in mice of the same strain and age.
A summary of the incidence of major neoplastic lesions is presented in Table 5. There were no tavaborole-related neoplastic effects observed in treated males or females. Neoplasms in this study were the type typically seen in this strain and age of mouse. Any differences in tumor incidence between control and treated mice were small and not considered biologically or statistically significant.
Summary of Major Neoplastic Lesions From the Mouse Dermal Carcinogenicity Study; Mice Treated Daily for 104 Weeks.
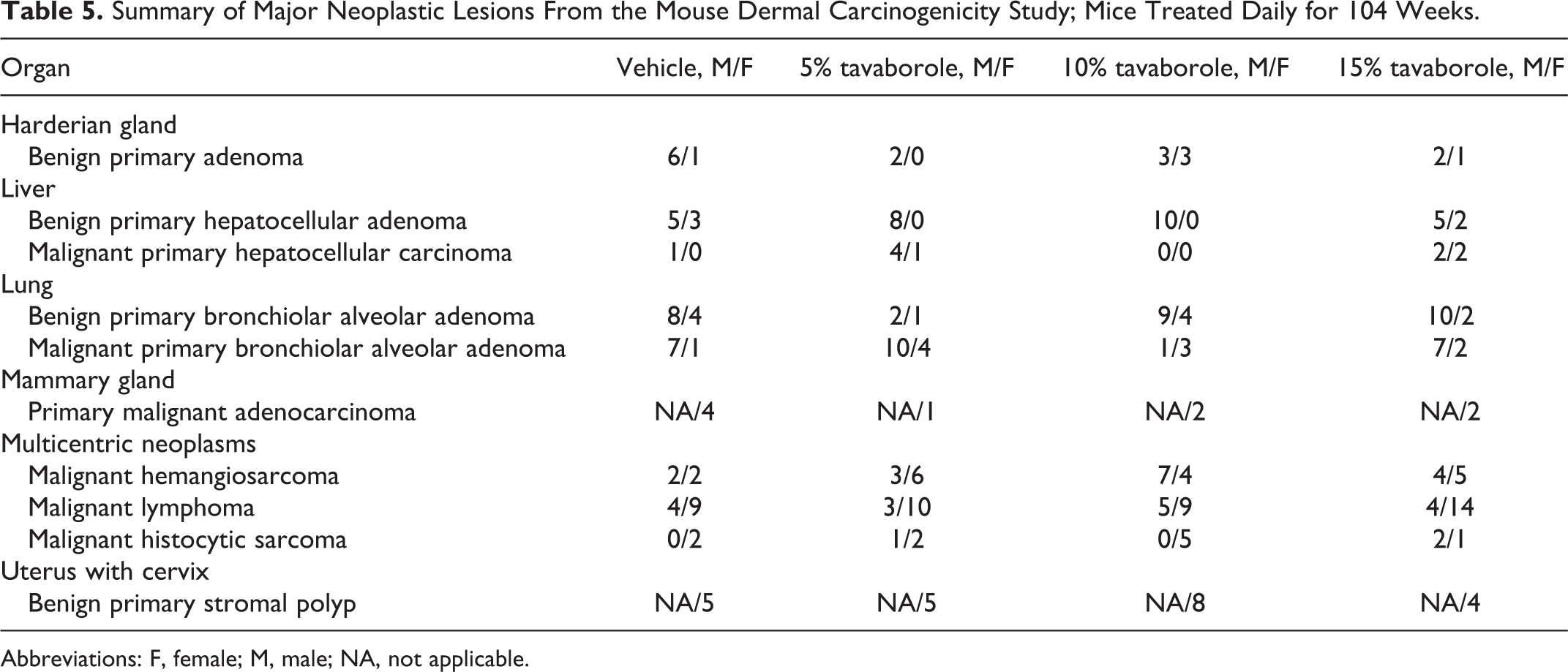
Abbreviations: F, female; M, male; NA, not applicable.
Tavaborole-related microscopic findings were observed in the treated skin of males and females at all dose levels (Table 6). When compared to vehicle control, an increased incidence of epidermal hyperplasia, hyperkeratosis, and/or subacute/chronic inflammation was observed in the treated skin of males and females at 5%, 10%, and 15% tavaborole/d. A clear dose response was not observed. The epidermal hyperplasia consisted of an increase (greater than 1-2 cell layers thick) in the number of nucleated epithelial cells comprising the epidermis. This change was usually associated with increased collagen within the dermis, which was considered to be related to the hyperplastic process. The degree of hyperplasia depended on the number epidermal cell layers, where minimal contained 3 to 4 layers, mild contained 5 to 6 layers, moderate contained 7 to 8 layers, and severe contained greater than 9 cell layers. Incidences of moderate and severe grades of epidermal hyperplasia were usually focally adjacent to the margins of ulcers, and in these cases, the epidermal hyperplasia most likely represented reactions to the ulcers rather than tavaborole. Sometimes the epidermal hyperplasia was associated with minimal to mild hyperkeratosis and/or minimal to moderate subacute/chronic inflammation. The hyperkeratosis was characterized by excessive thickening of the keratin layer, which was primarily orthokeratotic or occasionally parakeratotic in appearance. Subacute/chronic inflammation consisted of a mixed cellular infiltrate of lymphocytes, plasma cells, and macrophages, with variable numbers of neutrophils. Incidences of moderate and severe grades of subacute/chronic inflammation were usually associated with ulcers and most likely represented reactions to the ulcers rather than the tavaborole. Microscopic observations in the treated skin of serocellular crusting, acute inflammation, edema, hemorrhage, bacterial colonies, and ulceration were minimal to severe in severity. These observations were most likely related to the various manipulations of the dosing sites (clipping, abrasion, and cleaning) rather than due to application of tavaborole. A slightly higher incidence of ulceration in the treated skin was observed in the male died on study (DOS) mice given 15% tavaborole. This observation was not noted in the female DOS mice at the same dose. This finding in the male DOS animals was considered incidental and not tavaborole related because of the low incidence, presence in only 1 sex, and limitation to the DOS mice. Remaining neoplastic microscopic findings were the type commonly seen in mice of similar strain and age and were not tavaborole related (adenoma [harderian gland, liver, and lung], lung carcinoma, hemangioma, mammary adenocarcinoma, and benign stromal polyps).
Tavaborole-Related Microscopic Observations in Treated Mouse Skin.
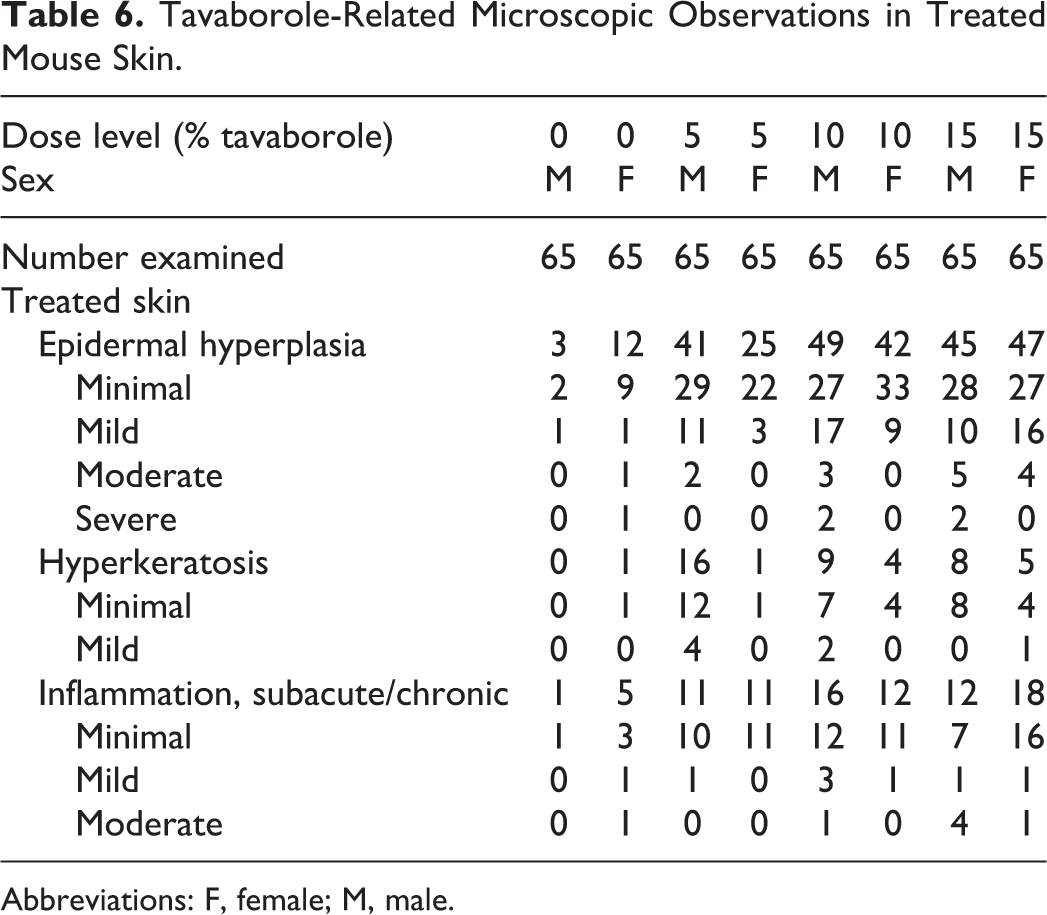
Abbreviations: F, female; M, male.
Carcinogenicity Study in Rats by Oral Gavage
The results of the dosing formulation analysis indicated that the concentration of tavaborole was below the quantifiable limit in the vehicle samples. Dose concentrations at 1.25, 2.5, and 5.0 mg/mL tavaborole were generally within the acceptable range of ±10% of target concentrations. The results of the homogeneity analysis indicated that the suspensions were homogenous throughout as the concentration percentage from nominal ranged from 97.6% to 99.8%. The formulations were proven to be stable under conditions of storage on this study.
In-life examinations
There were no differences in the number of mortalities across the groups, and cumulative survival at the end of the study was considered to be acceptable at 66% to 77% for males and 45% to 54% for females (Table 7). Causes of death were the type commonly seen in rats of this strain and age. Mean body weight gains (Table 7) and food consumption (data not presented) in males and females in all treatment groups were comparable to controls and unaffected by treatment.
Survival, Body Weight Gain, and Palpable Masses in the Rat Oral Carcinogenicity Study; Rats Treated Daily for 104 Weeks.

Abbreviations: M, male; F, female; NA, not applicable.
The total number of masses was comparable across all groups (Table 7). The palpable masses were generally transient with 25% to 53% and 49% to 64% of masses present at necropsy for males and females, respectively. The total number of masses recorded was consistently higher in females than in males due to the presence of palpable mammary masses and was not an indication of any sex-related effects.
There were no clinical signs related to tavaborole treatment at any dose. Clinical signs such as fur thinning, ungroomed/dull/matted/stained fur, changes in activity, posture, condition, and changes in feces were observed throughout the study. However, such signs were generally noted with a similar incidence and severity across the treated and control groups and were not considered tavaborole related. There were a number of rats across all groups, which demonstrated foot lesions (ulcerative pododermatitis) over the study duration, which were consistent with chronically housed animals. These rats were treated with a topical application of iodine to prevent infection and Isopads were placed on the cage floors to allow relief from the cage floor until the wounds healed. Occasional cases of corneal opacity were noted in tavaborole-treated rats; however, this change was also observed in control animals and was therefore considered to be an incidental finding and not related to tavaborole treatment. In general, over the course of the study, rats were euthanized in extremis due to mass rupture, mass location compromising overall well-being of the animal (eating, breathing, or mobility), limb injury, or uterine prolapse.
Plasma and TK analysis
There were dose-dependent increases in exposure of tavaborole after oral administration, and exposure was slightly greater than dose proportional. Exposure to tavaborole was relatively constant from days 1 to 180. There were no clear or consistent differences in exposure and maximal plasma concentrations related to gender. Results of the TK evaluation are presented in Table 8.
Summary TK Parameters on Days 1 and 180 After Administration of 12.5, 25, and 50 mg/kg/d Tavaborole Once Daily by Oral Gavage to Male and Female Rats.
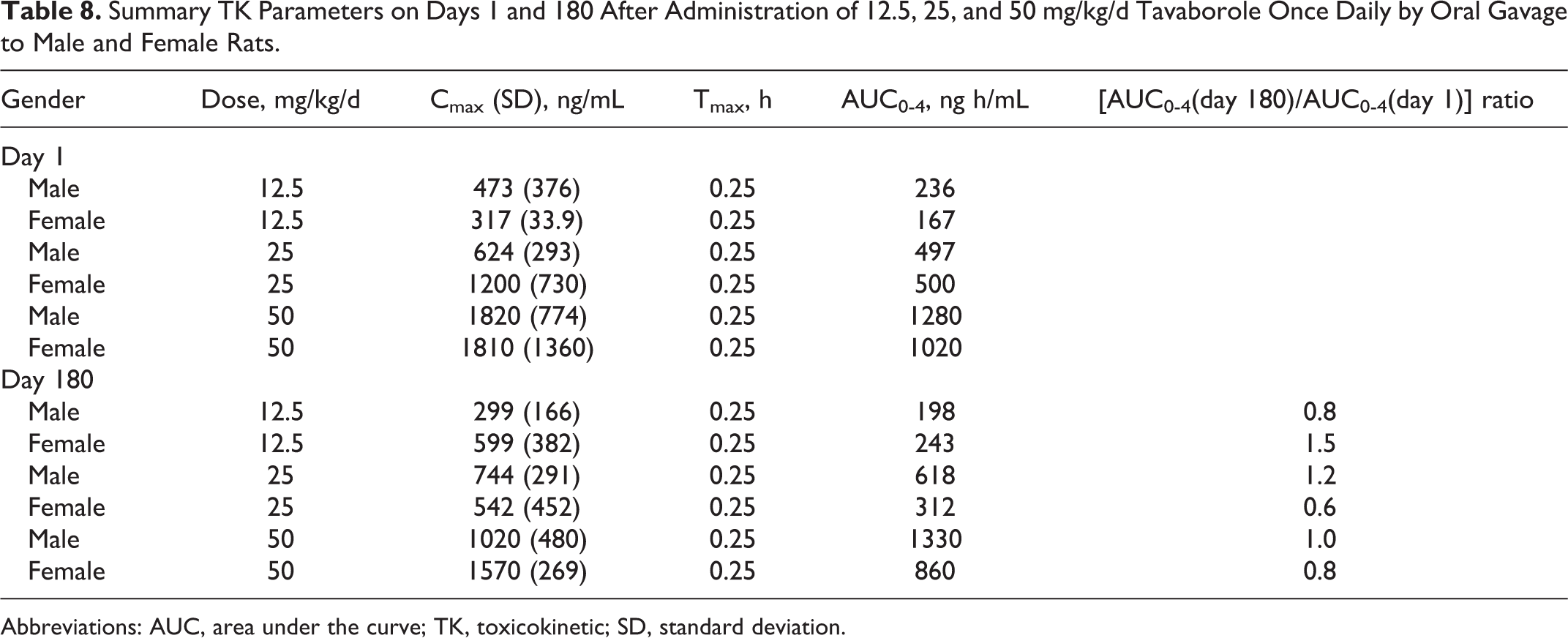
Abbreviations: AUC, area under the curve; TK, toxicokinetic; SD, standard deviation.
Postmortem study evaluations
No tavaborole-related macroscopic observations were made in male or female rats. All necropsy findings observed in this study represented common findings in laboratory rats in studies of this type and duration and were considered unrelated to administration of tavaborole.
There were no tavaborole-related neoplastic findings. The occurrence of all tumors was considered consistent with studies of this type and duration in this strain of rat and therefore was considered unrelated to tavaborole exposure.
The only tavaborole-related microscopic findings were in the stomach (Table 9) and consisted of hyperplasia of the nonglandular stomach. The finding was considered higher in males and females receiving tavaborole at the high-dose only (50 mg/kg/d). Hyperplasia of the nonglandular gastric mucosa was not associated with any additional proliferative or neoplastic findings in the stomach. Remaining nonneoplastic microscopic findings were the type commonly seen in rats of similar strain and age and were not tavaborole related.
Principal Microscopic Findings in the Rat Carcinogenicity Study; Rats Treated Daily for 104 Weeks.
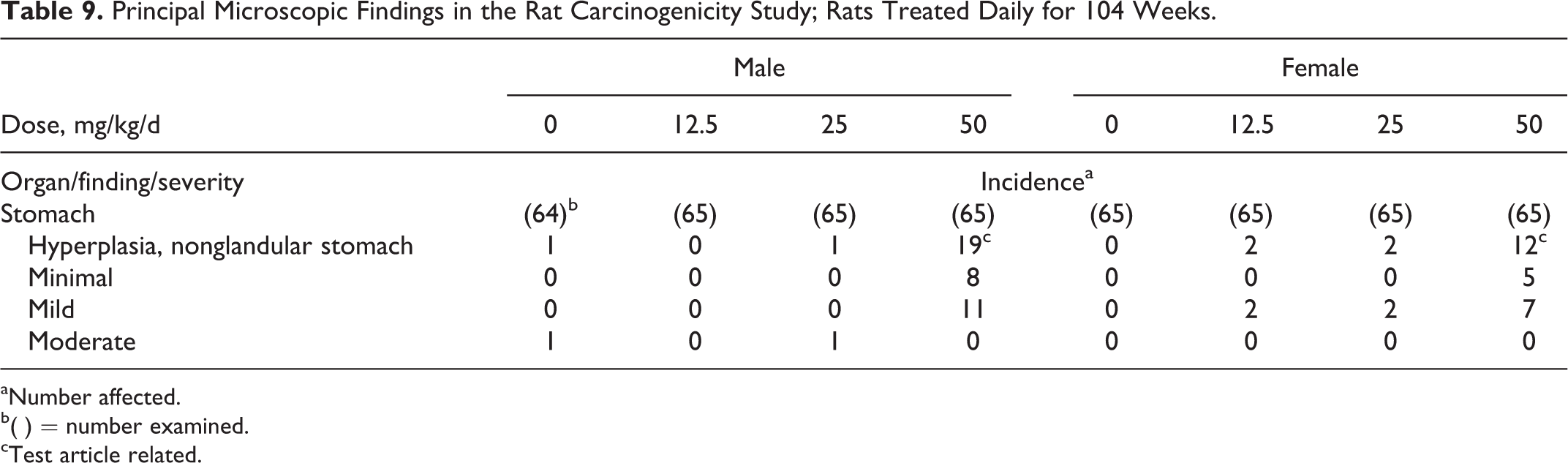
aNumber affected.
b( ) = number examined.
cTest article related.
Discussion
The creation of boron-containing drugs goes back to the early 1970s when aryl boronic acids were shown to be inhibitors of serine proteases such as subtilisin and chymotrypsin. 13 Due to the unique electronic properties of boron, with its empty p orbital, these compounds were potent enzyme inhibitors due to their ability to react with a serine hydroxyl in the enzyme active site. Over the intervening years, drug researchers have exploited this inherent reactivity of boronic acids and designed inhibitors against a number of therapeutically important enzymes, including thrombin, b (β) lactamases, hepatitis C virus protease, and dipeptidyl peptidase IV. 14 –16 The compound discussed in this article, 5-fluorobenzoxaborole (tavaborole), is an example of a boron compound where the boron was incorporated in a ring system fused to an aromatic ring. This is different from traditional boronic acid chemistry and represents a new class of antifungal drug. 8
Tavaborole Topical Solution, 5% has been FDA-approved for the treatment of onychomycosis. The carcinogenic potential of tavaborole, belonging to a new class of antifungal compounds (oxaborole), was investigated in traditional 2-year carcinogenicity studies in mice and rats. In the mouse, tavaborole was applied topically at 0%, 5%, 10%, and 15%. The lowest tavaborole dose (5%) used in the study represented the clinically relevant concentration. However, higher concentrations were tested to provide additional safety information. Although tavaborole is administered topically in humans, tavaborole was administered by oral gavage in the rat at 0, 12.5, 25, and 50 mg/kg/d. There was no tavaborole-related neoplastic finding in either species. In the mouse study, no drug related neoplastic findings were noted at topical doses up to 15% tavaborole solution (89 times the maximum recommended human dose MRHD) based on AUC comparisons).
10
In the rat study, no drug related neoplastic findings were noted at oral doses up to 50 mg/kg/d tavaborole (14 times the MRHD based on AUC comparisons).
10
The current investigations provide evidence that tavaborole, a representative compound of the benzoxaborole class, is not carcinogenic in rodents and is suitable for long-term use in humans.
Footnotes
Authors’ Contribution
V. Ciaravino and Chanda contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, and gave final approval. D. Coronado contributed to conception, analysis and interpretation, select item, critically revised the article, and gave final approval. C. Lanphear,I. Shaikh, and W. Ruddock contributed to design, acquisition, analysis, and interpretation, critically revised the article, and gave final approval.
Authors’ Note
KERYDIN™ (tavaborole) topical solution, 5% has been developed by Anacor Pharmaceuticals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Carcinogenicity studies described in this article were funded by Anacor Pharmaceuticals and conducted at MPI Research (Mouse Study) and ITR Laboratories Canada Inc. (Rat Study).
