Abstract
Aflatoxin B1 (AFB1), one of the most common mycotoxins found in human foods, is principally hepatotoxic; however, it also affects reproduction. The aim of the present study was to elucidate the reproductive toxic effects and possible mechanism of action of AFB1 in rats. Male Wistar rats were injected intramuscularly with doses of 10, 20, or 50 µg AFB1/kg body weight on alternate days from 45 to 100 days of age. Significant reductions in body weights, relative weights of reproductive organs, daily sperm production, epididymal sperm count, viable sperm, motile sperm, and hypoosmotic swelling-tail coiled sperm were observed. Significant decreases in testicular steroidogenic enzymes and serum testosterone levels were also observed indicating decreased steroidogenesis. In silico docking studies illustrated AFB1 binds with steroidogenic acute regulatory (StAR) protein thereby affecting the transport of cholesterol into mitochondria resulting in decreased steroidogenesis.
Keywords
Introduction
In recent years, there is an increasing concern that exposure to toxic pollutants disrupts male reproduction of wildlife and humans and plays a pivotal role in the decline of quality and quantity of human semen. 1 –3 The contributing factors for suppressed reproductive health are exposure to environmental contaminants, industrial and occupational chemicals, therapeutics, lifestyle factors, dietary toxins, and so on. 4 –6 Aflatoxins are naturally occurring toxic metabolites of Aspergillus flavus and Aspergillus parasiticus with a characteristic dihydrobisfuran moiety in their chemical structures. 7,8 Aflatoxin poisoning can produce recurrent serious health effects, which include carcinogenesis, 9 mutagenesis, 10 growth retardation, 11 and immune suppression. 12 Aflatoxins are a real threat to the health of humans by their ubiquitous and abiding occurrence in food commodities. 13 –15 Aflatoxin B1 (AFB1) is metabolized in the intestinal tissue and liver by various microsomal cytochrome P450s, resulting in AFB1-8,9-epoxide, which binds to DNA forming AFB1-guanine adducts. 16 From earlier studies, it is very clear that AFB1 also disrupts the hypothalamo–pituitary–testicular axis resulting in production of defective spermatozoa. 17 –20
Testosterone biosynthesis occurs in Leydig cells of testes in response to luteinizing hormone (LH) synthesized by the pituitary gland. The substrate for steroidogenesis is cholesterol, which is transported into mitochondria by steroidogenic acute regulatory (StAR) proteins. 21 In mitochondria, cholesterol is converted into pregnenolone by cytochrome P450 side chain cleaving enzyme. Later, pregnenolone is transported to smooth endoplasmic reticulum for conversion to testosterone. From earlier studies, it is evident that reduced steroidogenesis is a common feature in aflatoxin-mediated reproductive toxicity, although the exact mechanism is not known. 19,22 The present study is intended to elucidate the effects of aflatoxin on steroidogenesis and spermatogenesis by evaluating sperm parameters, circulatory reproductive hormone levels, and activity levels of selected steroidogenic enzymes. Furthermore, we have also studied the binding affinity of AFB1 to StAR protein using in silico tools.
Materials and Methods
Procurement and Maintenance of Experimental Animals
Male Wistar rats (40 days old; weighing between 90 and 100 g) were purchased from authorized dealer (Sri Venkateswara Traders, Bengaluru, India). Rats were housed in polypropylene cages (18 × 10 × 8 cu in) lined with sterilized paddy husk and provided filtered tap water and standard rodent feed (purchased from Sai Durga Agencies, Bengaluru, India) ad libitum. The rats were maintained in a well-controlled laboratory conditions (temperature 25°C ± 2°C under a schedule of 12-hour light and 12-hour dark cycle, humidity 50% ± 5%). The animals were examined daily for general pathological symptoms. The experiments were carried out in accordance with the guidelines of the Committee for the Purpose of Control and Supervision on Experiments on Animals, Government of India 23 and approved by the Institutional Animal Ethical Committee at Sri Venkateswara University (Tirupati, India; Resolution No. 03/2011-12/(i)/a/CPCSEA/IAEC/SVU/PSR-CHS/Dt. 26-08-2011).
Chemicals
Aflatoxin B1 (from A flavus with 98% purity by high-performance liquid chromatography) was purchased from Fermentek Ltd (Jerusalem, Israel). Dehydroepiandrosterone, androstenedione, reduced form of nicotinamide adenine dinucleotide phosphate (NADPH), and nicotinamide adenine dinucleotide (NAD) were purchased from Sigma Chemical Company (St Louis, Missouri). All other chemicals used in study were of analytical grade and obtained from local commercial sources.
Experimental Design and Necropsy
Rats were acclimatized to laboratory conditions for 1 week prior to experiments. Early-pubertal healthy male rats (45 days old) were selected for the present investigation and randomly divided into 4 groups with 8 animals in each group. The animals in group 1 served as control. Rats in groups 2, 3, and 4 were injected with AFB1 at a dose of 10, 20, and 50 µg/kg body weight, respectively, dissolved in dimethyl sulfoxide, on alternative days through intramuscular route for 60 days. The dosage levels of AFB1 were based on previous studies on rats, which caused reproductive abnormalities. 18,19,24
After completion of the experimental period, the rats were fasted overnight, weighed, and killed by cervical dislocation. Testes, epididymis (caput, corpus, and cauda), prostate gland, vas deferens, and seminal vesicles were quickly removed, cleared of the adhering tissues, and weighed wet to the nearest milligram and used for the determination of tissue somatic indices (TSIs).
Testes were used for determination of daily sperm production (DSP), biochemical analysis, and enzyme assays, and epididymis was used for sperm analysis.
Analysis of Epididymal Sperm
Epididymal sperm were obtained by chopping cauda epididymis in physiological saline (0.9% NaCl in distilled water). The sperm density was determined using a Neubauer chamber (Rohem, India) as described by Belsey et al. 25 The data were expressed as millions/mL. Progressive sperm motility was evaluated by the method described earlier, 25 within 5 minutes following their isolation from cauda epididymis at 37°C, and the data were expressed as percentage of motility. The viability of the sperm was determined using 1% trypan blue reagent. 26 The sperm membrane integrity was determined by exposing the sperm to hypoosmotic solution and observed for sperm with coiled tail under the microscope (hypoosmotic swelling [HOS] tail coiling), and the percentage of sperm with coiled tail was estimated using the method described earlier. 27
Daily Sperm Production
Daily sperm production was determined in the testis by the method of Blazak et al. 28 Briefly, the testes were decapsulated and homogenized in 50 mL of ice cold 0.9% NaCl solution containing 0.01% Triton X-100 using a sterilized mortar and pestle. After thorough mixing of homogenate, the number of sperm heads was counted in Neubauer hemocytometer. The number of sperm produced per gram testicular tissue per day was calculated.
Assay of Testicular Steroidogenic Marker Enzymes
The testicular tissue was homogenized in ice-cold Tris-HCl buffer (pH 6.8). The microsomal fraction was separated and used as enzyme source. The activity levels of 3β-hydroxysteroid dehydrogenase (3β-HSD; EC 1.1.1.51) and 17β-hydroxysteroid dehydrogenase (17β-HSD; EC 1.1.1.64) were determined by the method of Bergmayer. 29 The enzyme assays were made under the conditions following 0 order kinetics after preliminary standardization regarding linearity with respect to time of incubation and enzyme concentration. Protein content in the enzyme source was estimated by the method of Lowry et al 30 using bovine serum albumin as standard. The enzyme activities were expressed as nmol of NAD converted to reduced form of nicotinamide adenine dinucleotide/mg protein/min (3β-HSD) or nmol of NADPH converted to nicotinamide adenine dinucleotide phosphate/mg protein/min (17β-HSD).
Determination of Serum Hormone levels
Blood was collected from the heart of each animal before necropsy using a heparinized syringe. The serum was separated by centrifugation at 2000g for 15 minutes after overnight storage at 4°C and stored at −20°C until all the samples were collected. Radioimmunoassay for serum testosterone was performed by the method of Rao et al. 31 The sensitivity of the assay was calculated as 0.002 ng, and intraassay variation was found to be 6.5%. Serum follicle-stimulating hormone (FSH) and LH levels were determined according to the method of Lin et al. 32 Iodination of recombinant FSH and recombinant LH with 125I was performed by the method of Greenwood et al 33 using chloramine-T as an oxidizing agent. The sensitivity of the assay was calculated as 0.008 ng for FSH and 0.006 ng for LH. The intraassay variations were 5.8% and 6.9% for FSH and LH, respectively. All the samples were run at the same time to avoid interassay variation.
Docking Studies
Molecular docking studies were performed with AFB1 and cholesterol against StAR protein (Protein Data Bank [PDB] ID: 3POL) using PyRx virtual screening software (online free software), which uses AutoDock Vina for docking studies. 34 The interaction between protein and ligands was interpreted and visualized using PyMOL. 35
Statistical Analysis
The data were statistically analyzed using 1-way analysis of variance followed by Tukey test using SPSS 16.0. The data were expressed as mean ± standard deviation, and “P” value <0.05 was considered significant. All statistical tests were performed using SPSS 16.0 (SPSS Inc, Chertsey, United Kingdom).
Results
Body Weights and Tissue Indices
Body weights of the control and AFB1-treated rats are presented in Table 1. Significant decreases in body weights were found in AFB1-treated rats when compared to controls. Significant decreases in the indices of testis and accessory sex organs were also observed at all dosages with the exception of the testes, corpus epididymis, and prostate at 10 μg/kg compared with controls (Table 1). Food consumption (g/kg body weight/d) was almost similar in all groups (Ch. Supriya, 2014 PhD Dissertation).
Effect of Aflatoxin B1 on Body Weights (g) and Tissue Indices (g%) in Rats.a
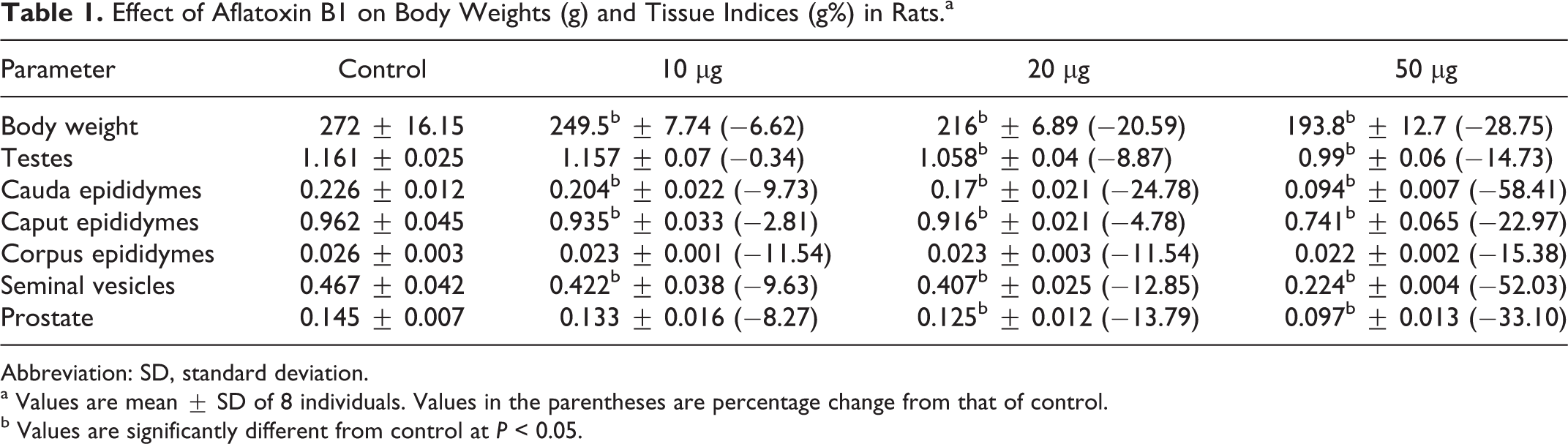
Abbreviation: SD, standard deviation.
a Values are mean ± SD of 8 individuals. Values in the parentheses are percentage change from that of control.
b Values are significantly different from control at P < 0.05.
Sperm Parameters
The effect of AFB1 on testicular DSP, cauda epididymal sperm count, sperm motility, sperm viability, and sperm HOS coiling in rats is given in Table 2. A significant decrease in DSP, sperm count, sperm motility, sperm viability, and sperm coiling percentage was observed in rats exposed to selected dosages of AFB1 when compared to controls with the exception of DSP at 10 μg.
Effect of Aflatoxin B1 on Testicular Daily Sperm Production and Epididymal Sperm Parameters in Rats.a
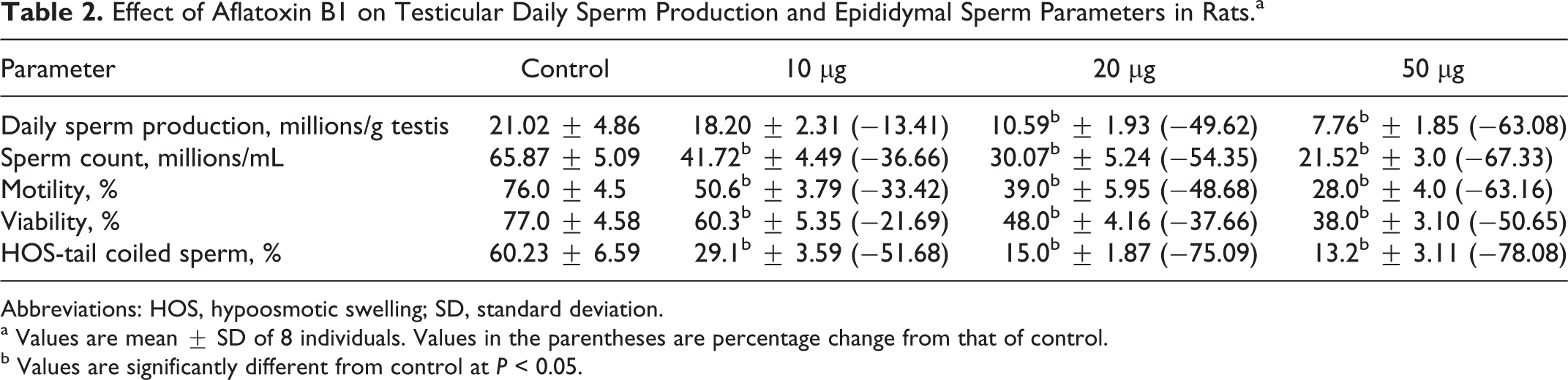
Abbreviations: HOS, hypoosmotic swelling; SD, standard deviation.
a Values are mean ± SD of 8 individuals. Values in the parentheses are percentage change from that of control.
b Values are significantly different from control at P < 0.05.
Testicular Steroidogenic Marker Enzymes and Serum Hormone Profiles
Table 3 summarizes the data on the activity levels of 3β-HSD and 17β-HSD in the testis of control and AFB1-exposed rats. The activity levels of testicular 3β-HSD and 17β-HSD were significantly decreased in rats exposed to AFB1 in a dose-dependent manner when compared to control rats (Table 3). Furthermore, a dose-dependent reduction in serum testosterone levels was also observed in rats exposed to AFB1 (Table 4). However, the levels of FSH and LH significantly increased in serum of rats treated with AFB1 when compared with the control rats (Table 4).
Effect of Aflatoxin B1 on Testicular 3β- and 17β-Hydroxysteroid Dehydrogenase Activity Levels in the Testes of Male Rats.a

Abbreviations: 3β-HSD, 3β-hydroxysteroid dehydrogenase; 17β-HSD, 17β-hydroxysteroid dehydrogenase; SD, standard deviation; NADH, reduced form of nicotinamide adenine dinucleotide; NADPH, reduced form of nicotinamide adenine dinucleotide phosphate; NADP, nicotinamide adenine dinucleotide phosphate.
a Values are mean ± SD of 8 individuals. Values in the parentheses are percentage change from that of control.
b Values are significantly different from control at P < 0.05.
Effect of Aflatoxin B1 on Serum Testosterone, FSH, and LH Levels in Rats.a

Abbreviations: FSH, follicle-stimulating hormone; LH, luteinizing hormone; SD, standard deviation.
a Values are mean ± SD of 8 individuals. Values in the parentheses are percentage change from that of control.
b Values are significantly different from control at P < 0.05.
Docking Studies
Aflatoxin B1 and cholesterol binding affinities with StAR protein (PDB ID: 3POL) were evaluated using docking studies. Aflatoxin B1 and cholesterol interact with GLN 262, LEU245, PHE184, GLU169, LEU247, ARG182, PRO181, GLU173, LEU178, LEU260, THR263, VAL259, and ALA172 at the binding site of StAR protein. Besides these interactions, hydrogen bonds with GLN262 with bond lengths of 2.8 and 2.0 were observed with AFB1 (Figure 1). Aflatoxin B1 was found to possess high binding affinity (−8.1 kcal/mol) when compared with natural ligand, cholesterol (−7.8 kcal/mol).
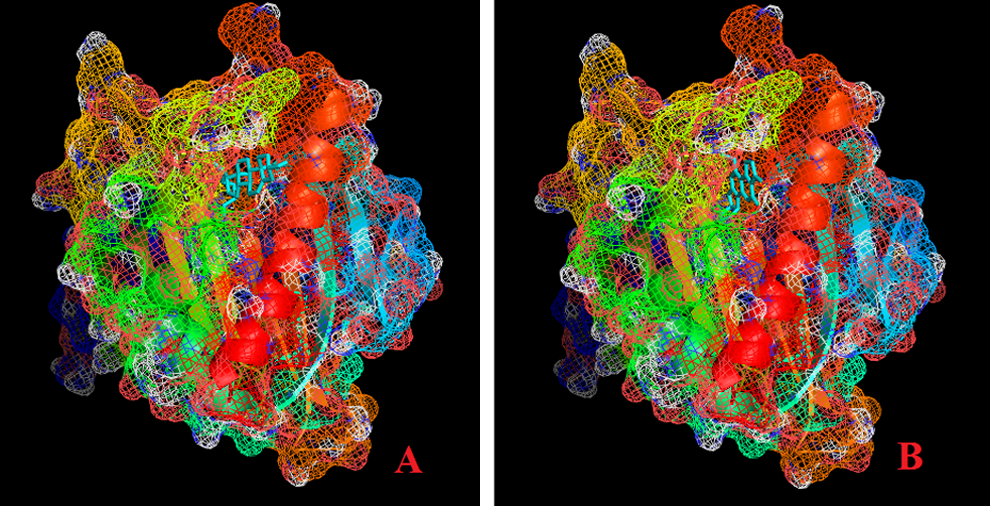
Mesh model of steroidogenic acute regulatory (StAR) protein showing hydrophobic binding pocket of cholesterol (A) and aflatoxin B1 (AFB1; B).
Discussion
In the present study, male rats were exposed to sublethal dosages (10, 20, or 50 µg/kg body weight) of AFB1 from early puberty (40 days of age) to sexual maturity (100 days of age), in order to evaluate its effect through a complete spermatogenic cycle, which takes approximately 60 days in rats. 36 The exposure of rats to 10, 20, or 50 µg/kg body weight corresponds to human exposure in areas with high AFB1 contamination leading to outbreak of aflatoxicosis. In 2 instances of aflatoxicosis, the daily AFB1 intake was estimated to have been at least 38 and 55 µg/kg body weight for an undetermined number of days. 37
The present work has shown that exposure of rats to 10, 20, or 50 µg/kg body weight of AFB1 significantly decreased the body weight gain and the relative weights of testis and other reproductive organs. The decrease in the weight of the testes may be due to degeneration of germinal epithelium. The observed loss in weight of epididymis may be due to decrease in sperm production. Similar effects were observed in male rats exposed to AFB1 by other researchers, 19,22,38 which may be due to reduced bioavailability and/or production of androgen in the AFB1-exposed rats. It is well known that the morphology and functional integrity of testes and accessory sex tissues are dependent on the bioavailability of androgens. 39 The weight of reproductive organs has also been used as a sensitive bioassay of androgens. 40 Decrease in the serum level of testosterone may be a reason behind the significant decline observed in the weight of testis and accessory sex tissues in rats exposed to AFB1. The data also reveal reduced levels of circulatory testosterone in rats exposed to AFB1, indicating a possible inhibition of androgen synthesis in experimental rats.
The effects of AFB1 on spermatogenesis were assessed by determining DSP and epididymal sperm quality and density. The epididymal sperm count and DSP were also used as important indicators to detect the reproductive toxic effects of various chemicals. 41 It is well known that production and maturation are important for the development of healthy spermatozoa. The composition of the internal epididymal milieu is responsible for sperm maturation, which is under the control of androgen. In rats, androgen-binding protein secreted by Sertoli cells into the lumen of seminiferous tubules, under FSH stimulation, is transported to the epididymis, where it accumulates at concentrations higher than those found in the testes. This leads to a high local concentration of androgens, essential for maturation of epididymal spermatozoa. 42 The effects seen in this study such as decreased quantity and deteriorated quality of sperm have been reported in rats and mice chronically treated with sublethal doses of AFB1. 43,44 It is well known that testosterone plays a pivotal role in the regulation of structural and functional integrity of reproductive organs. 45
To determine whether AFB1 affects testosterone biosynthesis, we measured serum testosterone and determined the activity levels of marker steroidogenic enzymes in testes. Steroidogenesis is a complex testicular process where cholesterol is converted to testosterone by a cascade of enzymes. In response to LH secreted by the pituitary gland, transcription of genes involved in testosterone biosynthesis in Leydig cell is induced. Among them, StAR protein is an important rate-limiting protein involved in the transport of cholesterol from cytosol to mitochondria. In mitochondria, cholesterol is converted into pregnenolone and then transported to smooth endoplasmic reticulum for further steps involved in testosterone biosynthesis.
Verma and Nair 22 observed a significant increase in cholesterol concentrations in testes of AFB1-treated mice, which may be due to incomplete utilization of cholesterol or impaired steroidogenesis. Similar decrease in serum testosterone level has been reported in rats following exposure to AFB1. 46,47 Follicle-stimulating hormone and LH act on Sertoli and Leydig cells and thus regulate spermatogenesis and steroidogenesis, respectively. 48 The decrease in serum testosterone levels could be due to diminished responsiveness of Leydig cells to LH and/or direct inhibition of testosterone synthesis in rats exposed to AFB1. The increase in serum FSH levels indicates an impairment of spermatogenesis in AFB1-treated rats and reflects germ cell loss or damage to Sertoli cells, thereby affecting the feedback regulation of FSH secretion. 49 Hasanzadeh et al 50 observed a significant increase in serum levels of FSH with a decrease in testosterone in AFB1-treated rats when compared to controls. The lowered levels of circulatory testosterone with elevated levels of FSH and LH indicate an intact pituitary–testicular axis in AFB1-treated rats.
To determine the mechanism of action of AFB1, we performed docking studies between StAR protein and AFB1. Earlier studies have shown that exposure to AFB1 induces swelling of mitochondria, and dysfunction due to accumulation of calcium during aflatoxicosis has also been reported. 51,52 Moreover, aflatoxin metabolism occurs in mitochondria resulting in mitochondrial swelling, which suggests AFB1 interaction with StAR protein. It is evident from the present study that AFB1 possesses high binding affinity when compared with natural ligand, cholesterol. Similar binding sites and affinities were observed with other StAR domain-containing proteins (Mus musculus [1JSS] with binding affinity −9.0 kcal/mol and −10.5 kcal/mol and Homo sapiens [2PSO] with binding affinity −8.6 kcal/mol and −8.9 kcal/mol for AFB1 and cholesterol respectively). From the data, it is evident that AFB1 competes with cholesterol thereby affecting the cholesterol transport into the mitochondria by StAR protein. The reduced cholesterol levels in the mitochondria might be responsible for the decreased steroidogenesis and spermatogenesis.
In conclusion, the results of the study suggest that AFB1 binds competitively to StAR protein thereby affecting cholesterol transport into mitochondria resulting in reduced biosynthesis of testosterone. Since testosterone is very much essential for spermatogenesis, the decreased circulatory testosterone levels affect DSP and quality of the sperm. Whether the decrease in sperm count and quality affect fecundity of AFB1-treated rats cannot be determined from the present data. Hence, additional in-depth fertility studies are needed involving artificial insemination using a fixed number of sperm from the cauda epididymis and also using the male’s ability to sire offspring in a fixed-time period. These studies will help us to determine the effect of AFB1 not only on the status of male libido but also on the fecundity of male rat. Our findings are significant because humans and animals are continuously exposed to aflatoxins and thus may be more prone to higher incidence of reproductive disorders. Finally, the data presented in this study provide strong evidence that AFB1 is a potent endocrine disruptor in vivo.
Ethical Standards
Animal handling and experiments were in accordance with the guidelines prepared by the Committee for the Purpose of Control and Supervision on Experiments on Animals, Government of India (CPCSEA, 2003) and approved by the Institutional Animal Ethical Committee at Sri Venkateswara University, Tirupati, India (Resolution No.03/2011-12/(i)/a/CPCSEA/IAEC/SVU/ PSR-CHS/Dt. 26-08-2011).
Footnotes
Authors’ Note
PSR conceived the idea, participated in its design, supervised the work, evaluated the data, and coordinated the study. ChS and BPG treated the animals and maintained the rat colony. ChS carried out sperm analyses and enzyme assays. BPG performed in silico studies. PSR determined the hormone levels. PSR, ChS, and BPG drafted the manuscript for publication. All authors read and approved the final manuscript. ChS and BPG should be regarded as joint first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported financially by the University Grants Commission through MRP grant to PSR. CHS and BPG were supported by a UGC-RGNF and CSIR-JRF, respectively.

